UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
|
|
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934. |
For the fiscal year ended
|
|
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission File No.
(Exact name of registrant as specified in its charter)
|
(State or other jurisdiction of incorporation or organization) |
(IRS Employer Identification Number) |
|
|
|
|
(Address of principal executive offices) |
(Zip Code) |
(
Registrant’s telephone number, including area code:
Securities registered pursuant to Section 12(b) of the Act:
|
Title of each class |
Trading Symbol |
Name of each exchange on which registered |
|
|
|
The |
|
|
|
The |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐
Indicate by check mark whether the Registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See definitions of “large accelerated filer”, “accelerated filer”, “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☐ Accelerated filer ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared its audit report.
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to § 240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes
The aggregate market value of Common Stock held by non-affiliates of the registrant on June 30, 2023, based on the closing price on that date was $
Number of shares of Common Stock outstanding as of February 29, 2024:
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive Proxy Statement to be filed with respect to its 2024 Annual Meeting of Stockholders are incorporated by reference in Part III of this document.
Table of Contents
| PART I | 1 | |
| ITEM 1. | BUSINESS | 1 |
| ITEM 1B. | UNRESOLVED STAFF COMMENTS | 28 |
| ITEM 1C. | CYBERSECURITY | 28 |
| ITEM 2. | PROPERTIES | 28 |
| ITEM 3. | LEGAL PROCEEDINGS | 29 |
| ITEM 4. | MINE SAFETY DISCLOSURES | 29 |
| PART II | 29 | |
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES | 29 |
| ITEM 6. | RESERVED | 29 |
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS | 30 |
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK | 36 |
| ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA | 36 |
| ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE | 36 |
| ITEM 9A. | CONTROLS AND PROCEDURES | 36 |
| ITEM 9B. | OTHER INFORMATION | 37 |
| ITEM 9C. | DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS | 37 |
| PART III | 37 | |
| ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE | 37 |
| ITEM 11. | EXECUTIVE COMPENSATION | 38 |
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS | 38 |
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS, AND DIRECTOR INDEPENDENCE | 38 |
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES | 38 |
| PART IV | 39 | |
| ITEM 15. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES | 39 |
| ITEM 16. | FORM 10-K SUMMARY | 41 |
| SIGNATURES | 42 | |
This Annual Report (including the following section regarding Management’s Discussion and Analysis of Financial Condition and Results of Operations) contains forward-looking statements regarding our business, financial condition, results of operations and prospects. Words such as “expects,” “anticipates,” “intends,” “plans,” “believes,” “seeks,” “estimates” and similar expressions or variations of such words are intended to identify forward-looking statements but are not the exclusive means of identifying forward-looking statements in this Annual Report. Additionally, statements concerning future matters, including statements regarding our business, our financial position, the research and development of our products and other statements regarding matters that are not historical are forward-looking statements.
Although forward-looking statements in this Annual Report reflect the good faith judgment of our management, such statements can only be based on facts and factors currently known by us. Consequently, forward-looking statements are inherently subject to risks and uncertainties and actual results and outcomes may differ materially from the results and outcomes discussed in or anticipated by the forward-looking statements. Factors that could cause or contribute to such differences in results and outcomes include without limitation those discussed under the heading “Risk Factors” below, as well as those discussed elsewhere in this Annual Report. Readers are urged not to place undue reliance on these forward-looking statements, which speak only as of the date of this Annual Report. We undertake no obligation to revise or update any forward-looking statements in order to reflect any event or circumstance that may arise after the date of this Annual Report. Readers are urged to carefully review and consider the various disclosures made in this Annual Report, which attempt to advise interested parties of the risks and factors that may affect our business, financial condition, results of operations and prospects.
PART I
|
ITEM 1. |
BUSINESS |
Overview
GeoVax Labs, Inc. (“GeoVax” or “the Company”) is a clinical-stage biotechnology company developing human vaccines and immunotherapies against infectious diseases and solid tumor cancers using novel proprietary platforms. GeoVax’s product pipeline includes ongoing human clinical trials for a next-generation Covid-19 vaccine and a gene-directed therapy against advanced head and neck cancers. Additional research and development programs include preventive vaccines against Mpox (formerly known as monkeypox) and smallpox, hemorrhagic fever viruses (Ebola Zaire, Ebola Sudan and Marburg), Zika virus and malaria, as well as immunotherapies for multiple solid tumors. The Company’s portfolio of wholly owned, co-owned, and in-licensed intellectual property, stands at over 155 granted or pending patent applications spread over 24 patent families, which are discussed in greater detail in the “Our Intellectual Property” section.
Our Product Development Pipeline
The table below summarizes the status of our product development programs, which are discussed in greater detail in the following pages.
|
Indication |
Current Status |
|
Coronavirus/Covid-19 Vaccines |
|
|
Booster to mRNA vaccines |
Clinical – Phase 2 (enrollment completed) |
|
Primary vaccine for immunocompromised/cell transplant patients |
Clinical – Phase 2 (actively enrolling) |
|
Booster for immunocompromised/CLL patients |
Clinical – Phase 2 (actively enrolling) |
|
Cancer Therapy |
|
|
Solid Tumors (Advanced Head and Neck Cancer)* |
Clinical – Phase 1/2 (enrollment completed) |
|
Solid Tumors (MUC1) |
Preclinical/IND-Enabling |
|
Other Infectious Disease Vaccines |
|
|
Mpox & smallpox |
Preclinical/IND-Enabling |
|
Hemorrhagic Fever Viruses Ebola Zaire** |
Preclinical/IND-Enabling |
|
Ebola Sudan** |
|
|
Marburg** |
|
|
Zika** |
Preclinical/IND-Enabling |
|
Malaria** |
Exploratory |
* Orphan Drug status granted
** Indication within FDA Priority Review Voucher program
Our Coronavirus Vaccine Programs
Severe respiratory illnesses caused by the SARS-CoV-2 virus, remain a serious public health issue of international concern. SARS-CoV-2 is an enveloped, single-stranded, positive-sense RNA virus belonging to the family Coronavidae within the genus beta-coronavirus. The genome of SARS-CoV-2 encodes one large Spike (“S”) protein that plays a pivotal role during viral attachment to the host receptor and entry into host cells. The S protein is the basis for most approved vaccines used to protect against SARS-CoV-2. Neutralizing antibodies targeting the receptor binding domain (“RBD”) subunit of the S protein block the virus from binding to host cells. Over 90% of all neutralizing antibodies produced in response to infection are directed to the RBD subunit.
Based on our research, there are currently forty vaccines authorized for use in one or more countries around the world, including four in the United States. These vaccines are primarily designed to induce antibodies specific for the S protein of SARS-CoV-2 but rely on different mechanisms for presentation or expression of the S antigen, including recombinant proteins, whole inactivated virus, defective adenovirus vectors (three different types) or mRNA. Unfortunately, the continued adaptation and mutation of SARS-CoV-2 has resulted in the emergence of variants that are not optimally neutralized by antibodies induced by currently available vaccines, reducing clinical efficacy. This has required the continued adjustment of vaccine composition and the repeated administration of booster doses. Moreover, these current vaccines tend to stimulate only modest T-cell responses, which have been shown to be critical to critical for induction of long-term immune memory and for protection against severe Covid-19 disease. Recently, the FDA indicated the likely need for continued vaccine adjustments and boosters at least annually, similar to the approach used for influenza virus vaccines.
Modified Vaccinia Ankara (MVA) is the viral vaccine vector platform utilized in a number of our vaccine candidates, including our next generation SARS-CoV-2 product, GEO-CM04S1. There are several potential advantages to SARS-CoV-2 vaccines based on MVA vectors:
|
● |
MVA has a large genetic coding capacity which provides the foundation for vaccines based on multiple SARS-CoV-2 proteins, instead of the singular focus on the S protein. This approach induces immune responses with greater breadth of specificity. |
|
● |
MVA is known to effectively induce T-cell responses in addition to antibodies and the responses are durable. |
|
● |
MVA does not replicate in human cells which contributes to it being an extremely safe vaccine platform for human vaccines. |
As a result of the combination of these properties, MVA is an ideal vaccine vector platform for the design of the next generation Covid-19 vaccines. This is especially true when the need to correct for suboptimal vaccine-induced immune responses of certain patient populations with compromised immune systems is considered, including patients suffering from and/or being treated for a variety of cancers, organ transplant patients, and renal dialysis patients.
GEO-CM04S1 for Immunocompromised/Cell Transplant Patients – The CDC and other global public health agencies identify immunocompromised patients, including patients who have received treatment for hematologic malignancy, as highest risk for SARS-CoV-2 disease. SARS-CoV-2 infection can be very serious in this vulnerable population of hematology patients, including autologous (auto) and allogeneic (allo) hematopoietic cell transplant (HCT), and recipients of chimeric antigen receptor (CAR)-T cell therapies.
Given the serious impact of other respiratory viruses in this vulnerable patient population, it is anticipated that hematology recipients of cell therapy may develop severe clinical disease, profoundly impacting morbidity and survival. Increasingly, there is evidence of the high risk of severe morbidity, hospitalization and death resulting from SARS-CoV-2 in hematology patients. The efficacy and safety of a SARS-CoV-2 vaccine remains to be established in the different immunocompromised patient populations and it is likely that candidate SARS-CoV-2 vaccines may differ in their efficacy and safety for these patients.
Our vaccine candidate, GEO-CM04S1, is based on a synthetic, attenuated Modified Vaccinia Ankara (sMVA) vector expressing both spike (S) and nucleocapsid (N) antigens of the SARS-CoV-2 virus and was initially developed at City of Hope (COH) Medical Center for immunocompromised patients. In a placebo-controlled Phase 1 clinical trial of healthy adults conducted by COH, GEO-CM04S1 was shown to be safe and immunogenic. In November 2021, GeoVax entered into a license agreement with COH, granting GeoVax exclusive worldwide rights to further develop and commercialize the vaccine.
GEO-CM04S1 is being studied in an ongoing Phase 2 clinical trial (ClinicalTrials.gov Identifier: NCT04977024) to evaluate its safety and immunogenicity as a primary vaccine, compared to either the Pfizer/BioNTech or Moderna mRNA-based vaccine, in patients who have previously received either an allogeneic hematopoietic cell transplant, an autologous hematopoietic cell transplant or chimeric antigen receptor (CAR) T cell therapy. MVA-vector based vaccines tend to produce an immune response quickly – in less than 14 days – with only mild side effects. The trial is also the first to compare an investigational multi-antigenic SARS-CoV-2 vaccine to the current Food and Drug Administration (FDA)-approved mRNA vaccines from Pfizer/BioNTech and Moderna in people who are immunocompromised. Such patients have often shown a suboptimal immune response after receiving currently available Covid-19 vaccines.
GEO-CM04S1 is also being studied as a booster vaccine in an investigator-initiated clinical trial (ClinicalTrials.gov Identifier: NCT05672355), in immunocompromised patients with chronic lymphocytic leukemia (CLL). Despite a high vaccination rate, CLL patients may be at high risk for lethal Covid-19 infection due to their compromised ability to generate antibody responses against Covid infections or vaccinations. GEO-CM04S1 may be more effective at inducing protective immunity in CLL patients since MVA strongly induces T cell expansion even in the background of immunosuppression. Targeting both the S and N protein antigens broaden the specificity of the immune responses and can protect against the loss of efficacy associated with the reduced antibody responses. The study is examining the use of two injections of GEO-CM04S1 three months apart to assess immune responses in these vulnerable patients, with the Pfizer-BioNTech Bivalent vaccine as the control arm.
GEO-CM04S1 as a Booster Vaccine – GEO-CM04S1 is also being studied in a Phase 2 trial (ClinicalTrials.gov Identifier: NCT04639466), evaluating its use as a heterologous booster vaccine to current FDA-approved mRNA vaccines from Pfizer/BioNTech and Moderna.
Because GEO-CM04S1 is designed to stimulate potent humoral and cellular immune responses against both the S and N proteins of SARS-CoV-2, GeoVax believes its administration as a booster will induce a broader and more sustained immune response than that seen after mRNA vaccine boosting. In addition, GEO-CM04S1 may offer better protection against the significant sequence variation observed with the S antigen because the N antigen tends not to change significantly amongst variants.
The Phase 1 trial of GEO-CM04S1 (known at the time as COH-CM04S1) was designed as a dose-escalation safety study in healthy individuals between the ages of 18 to 55, who had not been previously infected or vaccinated with SARS-CoV-2. The primary objectives were to evaluate the safety, tolerability and immunogenicity of the GEO-CM04S1 administered at three different dose levels by intramuscular (IM) injection. Follow-up studies of the volunteers are continuing to better characterize their immune responses.
The Phase 2 booster study, for which patient enrollment was completed in September 2023, includes 63 healthy adults who were previously vaccinated with one of the FDA-approved SARS-CoV-2 mRNA vaccines, manufactured by either Pfizer/BioNTech or Moderna. The study is designed as a comparison trial to evaluate the safety profile and immunogenicity of 2 dose levels of GEO-CM04S1 when administered as a heterologous booster. The immunological responses measured throughout the study will include SARS-CoV-2 neutralizing antibody and T-cell responses against SARS-CoV-2 variants of concern (VOC), including the Delta and contemporaneous Omicron variants.
In February 2024, we announced positive initial safety and immune response findings at one month following vaccine administration. While the study is designed to evaluate the safety and immunogenicity of two GEO-CM04S1 dose levels. The trial remains blinded to dose of vaccine received, with study subjects being followed for a total of one year. To date, there have been no serious adverse events, and adverse events were in line with other routine vaccinations. The immunological responses measured throughout the study period include both neutralizing antibodies against multiple SARS-CoV-2 variants (including contemporaneous Omicron variants) and specific T-cell responses. Consolidated data from all subjects tested one-month post-vaccination, documented statistically significant increases in neutralizing antibody responses against multiple SARS-CoV-2 variants, ranging from the original Wuhan strain through Delta and Omicron XBB 1.5; additional testing against the JN.1 variant is underway.
GEO-CM02 as a Pan-Coronavirus Vaccine – As noted, most of the first-generation SARS-CoV-2 vaccines were designed to encode the S protein of the SARS-CoV-2 virus with the goal of inducing high levels of neutralizing antibodies. However, the limitations of this approach in the face of continually emerging variants is now obvious and alternative vaccine designs are being developed.
GeoVax has developed a design strategy for vaccines to induce broader immunity through inclusion of multiple, genetically conserved structural and nonstructural proteins from SARS-CoV-2 and other beta-coronaviruses with the potential to infect humans. This is possible, through the use of our MVA platform to incorporate other sequence-conserved structural and nonstructural proteins as targets for T-cell responses to increase the breadth and function of vaccine-induced immune responses. This strategy provides the basis for generating a universal vaccine with augmented potential to alleviate the burden of disease caused by circulating coronaviruses. Unique when compared to other vaccines approved or under development, the GeoVax pan-coronavirus vaccine candidates are specifically designed to provide protective immunity against SARS-CoV-2 and other similar coronaviruses which could pose a risk of human infection.
In January 2021, the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH), awarded the GeoVax a Small Business Innovative Research (SBIR) grant in support of the Company’s vaccine development efforts. The Phase 1 grant, titled, “Preclinical Development of GV-MVA-VLP Vaccines Against COVID-19,” supported the ongoing design, construction and preclinical testing of our MVA-vectored vaccine candidates. This program currently is serving as the foundation for further internal experimental design and small animal model testing focusing on the use of highly conserved viral nonstructural proteins as additional T-cell immunogens to further increase the functional and specificity of induced immune responses.
In September 2023, data for GEO-CM02, which contains S, membrane (M) and envelope (E) proteins, were presented during the Keystone Symposia on Molecular and Cellular Biology held in Atlanta, Georgia. The presentation highlighted the following data: (i) the GEO-CM02 vaccine induced immune responses that were efficacious against the original Wuhan strain and BA.1 Omicron variant with a single dose, (ii) animals were protected prior to the detection of neutralizing antibodies, likely indicating a critical T-cell contribution, and (iii) GEO-CM02 significantly reduced or eliminated inflammation and immunopathology in the lungs of vaccinated animals. Together, these data indicate that immunization with the multi-antigen MVA-vectored vaccine can protect against severe disease and death induced by SARS-CoV-2 infection, regardless of the variant.
Our Cancer Therapy Programs
Gedeptin® – Gedeptin is a novel patented product/technology for the treatment of solid tumors through a gene therapy strategy known as Gene-Directed Enzyme Prodrug Therapy (GDEPT). In September 2021, GeoVax entered into an assignment and license agreement with PNP Therapeutics, Inc. (“PNP”), granting GeoVax exclusive worldwide rights to develop and commercialize Gedeptin. The Gedeptin technology was developed with funding support from the National Cancer Institute (NCI), part of the NIH. GeoVax’s license to Gedeptin includes the rights to expand the use of Gedeptin to all human diseases and/or conditions including, but not limited to, other solid tumors.
In GDEPT, a replication-deficient adenovirus vector is used to infect and transduce tumor cells with a nonhuman gene, which expresses an enzyme that can convert a inactive prodrug into an active antitumor compound, in situ. A cycle of Gedeptin therapy consists of intra-tumoral injections of Gedeptin followed by administration of a prodrug, fludarabine phosphate, over a pre-defined time period. A Phase 1 dose ranging study, evaluating the safety of a single cycle of Gedeptin therapy, found the therapy to be well tolerated, with evidence of a reduction in tumor size in patients with solid tumors.
A multi-site Phase 1/2a trial (ClinicalTrials.gov Identifier: NCT03754933), evaluating the safety and efficacy of repeat cycles of Gedeptin therapy in patients with recurrent head and neck squamous cell carcinoma (HNSCC), with tumor(s) accessible for injection and no curable treatment options is ongoing, with patient enrollment closed in December 2023.
The trial design involves repeat administration using Gedeptin followed by systemic fludarabine phosphate, in order to gain preliminary information on the utility of multiple cycles of Gedeptin therapy, prior to expansion towards a larger patient trial. The initial stage of the study is being funded by the FDA pursuant to its Orphan Products Clinical Trials Grants Program. The FDA has also granted Gedeptin orphan drug status for the intra-tumoral treatment of anatomically accessible oral and pharyngeal cancers, including cancers of the lip, tongue, gum, floor of mouth, salivary gland and other oral cavities. This trial will guide the design of larger studies involving patients at earlier stages in the disease process. These studies may lead to labeling discussions with the FDA and initiation of further Gedeptin investigations including in combination with immune checkpoint inhibitors for additional cancerous and non-cancerous tumor indications.
MUC1-based Immunotherapy – Tumors are often able to inhibit the body’s natural immune system by producing inhibitory factors as a mechanism of immune resistance, especially against the T cells that are specific for tumor antigens and can kill cancer cells. The field of immuno-oncology has received new momentum with the discovery and commercial launch of immune checkpoint inhibitors (ICIs), a type of monoclonal antibodies (Mabs). ICIs block the naturally occurring and tumor-induced immune checkpoints, thus allowing T cells to more fully function and respond against tumor cells.
Unlike conventional therapies (e.g. radiation, chemotherapy, antibody, etc.), therapeutic cancer vaccines have the potential to induce responses that not only contribute to the control of tumors but also establish immunological memory that can suppress and prevent tumor recurrence. Convenience, safety, and low toxicity of cancer vaccines could make them invaluable tools to be included in future immunotherapy approaches in combination with ICIs for treating tumors. Currently, there are only a few vectored cancer vaccines being tested in combination with ICIs, all of which are in early clinical stages.
We are developing our GV-MVA-VLP™ vaccine platform, which is based on the aberrantly glycosylated forms of the cell surface-associated MUC1 protein, and is expressed on a wide range of cancers, including breast, colon, ovarian, prostate, pancreatic, and lung, with the goal of raising therapeutic anti-tumor antibodies and T cell responses in cancer patients.
|
● |
We have produced a MVA-VLP-MUC1 vaccine candidate, demonstrated VLP production by electron microscopy using MUC1 immunogold staining, and showed that the VLPs express a hypo-glycosylated form of MUC1 in human cell lines. |
|
● |
We collaborated with Dr. Olivera Finn, a leading expert in cancer immunotherapy at the University of Pittsburgh, who was one of the first medical researchers to show that many tumors express an abnormal form of MUC1 that is recognized by the immune system as foreign. Our collaboration with Dr. Finn has shown that a combination of our MVA-VLP-MUC1 vaccine candidate with a MUC1 synthetic peptide was capable of breaking tolerance to human MUC1 in transgenic mice and inducing immune responses with efficacy against challenge in a lymphoma tumor model. |
|
● |
In 2022, we initiated potential IND enabling small animal studies with Dr. Pinku Mukherjee at the University of North Carolina at Charlotte to define the optimal course and schedule of vaccination to define a protocol that can be evaluated in a Phase 1 clinical trial. These studies, which are ongoing, include assessment of the therapeutic benefit the vaccine in combination with a synthetic peptide, adjuvant and immune checkpoint inhibitor using a human MUC1 transgenic mouse model to optimally match existing and previously tested cancer treatment regimens. |
MVA as Smallpox and Mpox Vaccine - MVA was originally developed for use as a smallpox vaccine more than 30 years ago. Its preferred use is for individuals with compromised immune systems; individuals that would be put at risk if administered the normal smallpox vaccine (“vaccinia”) which can replicate in human cells. It is also approved as the vaccine for other orthopox vaccines, including Mpox. As such, an added potential benefit of our vaccines is that in those regions where Mpox or smallpox are of concern, vaccines built on an MVA vaccine platform will likely also provide protection against Mpox and smallpox.
MVA is the vaccine currently used and stockpiled in the US Strategic National Stockpile for immunization against the MPox and smallpox viruses. GeoVax previously demonstrated that an experimental HIV vaccine, utilizing NIH-MVA as the vaccine vector, protected non-human primates challenged with a lethal dose of the Mpox virus. Further, in August 2022, the City of Hope team, which originally developed GEO-CM04S1, published results demonstrating that both their proprietary sMVA (synthetic MVA) and GEO-CM04S1 (referred to as “COH04S1” in the publication) elicited robust orthopoxvirus-specific binding and neutralizing antibody responses. The authors concluded that GEO-CM04S1 and sMVA represent unique vaccine candidates to control the unforeseen global Mpox outbreak.
In response to the global need to address the continued emerging threat from Mpox and the unique opportunity offered by MVA-based vaccines, in November 2022, GeoVax secured rights from the NIH covering preclinical, clinical and commercial uses of the NIH-MVA against Mpox or smallpox viruses. The Company is now evaluating development and regulatory pathways towards expanding the public health options available to reduce and manage the risk of Mpox worldwide. The Company intends to successfully develop and commercialize GEO-MVA, becoming the first U.S.-based supplier of MVA as a vaccine against Mpox and smallpox.
Our Hemorrhagic Fever Virus Vaccines (Ebola Zaire, Ebola Sudan, Marburg)
Ebola (EBOV, formerly designated as Zaire ebolavirus), Sudan (SUDV), and Marburg viruses (MARV) are the most virulent species of the Filoviridae family, causing hemorrhagic fever illnesses with up to a 90% fatality rate in humans. In December 2019, FDA approved the first live recombinant Ebola vaccine for prevention of Ebola disease by Zaire virus. This rVSV-ZEBOV showed safety concerns in Phase 1 trials and by virtue of being replication competent could pose threats to immunocompromised individuals, such as those infected with HIV living in West Africa where recent Ebola epidemics started.
To address the unmet need for a product that can respond to future hemorrhagic fever outbreaks, we are developing vaccines utilizing our GV-MVA-VLP™ platform. As previously noted, the MVA vector itself is considered safe, having originally been developed for use in immunocompromised individuals as a smallpox vaccine. We expect our vaccines may not only protect at-risk individuals against EBOV, SUDV and MARV, but also potentially reduce or modify the severity of other re-emerging pathogens such as Bundibugyo, Ivory Coast, and Reston viruses, based on antigenic cross reactivity and the elicitation of T cells to the more conserved matrix proteins (e.g. VP40 or Z) in addition to standard GP proteins used by us and other manufacturers. Thus, the GeoVax GV-MVA-VLP™ approach could offer a unique combination of advantages to achieve breadth and safety of a pan-filo vaccine. In addition to protecting historically higher-risk populations in Africa, it is also intended to prevent the spread of disease to the US and globally, and for preparedness against terrorist release of any of bio-threat pathogens in the US and globally.
Our initial preclinical studies in rodents and nonhuman primates for our MVA-VLP-EBOV vaccine candidate have shown significant levels of protection against lethal doses of EBOV. Recent studies in lethal challenge guinea pig models demonstrated that GeoVax vaccines MVA-VLP-SUDV and MVA-VLP-MARV conferred 100% protection from death. These vaccines were subsequently evaluated in a rigorous cynomolgus macaque infectious challenge model. Vaccination protected nonhuman primates from viremia, weight loss and death following challenge with a dose of Sudan or Marburg virus that is lethal in nonvaccinated animals. Evaluation of immune responses following vaccination demonstrated presence of both neutralizing antibodies and functional T cells, indicating a breadth of responses that combine for optimal protection. The nonhuman primate studies conducted in collaboration with NIAID and the U.S. Department of Defense (DoD) have been completed and potential clinical development programs are being considered.
Other Infectious Disease Programs
GEO-ZM02 for Zika – Zika disease is an emerging infectious disease caused by the Zika virus (ZIKV) and has been linked to an increase in microcephaly in infants and Guillain-Barre syndrome (a neurodegenerative disease) in adults. ZIKV is a member of the Flaviviridae family, which includes medically important pathogens such as dengue fever, yellow fever, Japanese encephalitis, tick-borne encephalitis, and West Nile viruses. Public health officials recommend avoiding exposure to ZIKV, delaying pregnancy, and following basic supportive care (fluids, rest, and acetaminophen) after infection.
To address the unmet need for a ZIKV vaccine, we are developing novel vaccine candidates constructed using our GV-MVA-VLP platform. MVA has an outstanding safety record, which is particularly important given the need to include women of child-bearing age and newborns among those being vaccinated. Our Zika vaccine is designed based on the NS1 gene product to eliminate the risk of Antibody Dependent Enhancement (ADE), which is a serious side effect observed when a vaccinated individual doesn’t have a fully protective immune response which actually causes a more virulent reaction if infected.
Our initial preclinical studies in rodents using our GEO-ZM02 vaccine candidate demonstrated 100% single-dose protection against a lethal dose of ZIKV delivered directly into the brain. In rhesus macaques, vaccination with GEO-ZM02 induced immune responses that effectively controlled the virus replication despite the fact the vaccine is not designed to induce ZIKV neutralizing antibodies. Further development of GEO-ZM02 will be dependent upon partnering support.
In January 2023, GeoVax announced that the U.S. Patent and Trademark Office issued a Notice of Allowance for Patent Application No. 17/000,768 titled, “Method for Generating a ZIKV Immune Response Utilizing a Recombinant Modified Vaccinia Ankara Vector Encoding the NS1 Protein.” Preclinical studies demonstrated a single dose of GEO-ZM02 provided 100% protection against a lethal dose of Zika virus.
GEO-MM02 for Malaria – According to recent data from the World Health Organization, globally, malaria causes 227 million infections and 619,000 deaths annually. Despite decades of vaccine research, vaccine candidates have failed to induce substantial protection. Most of these vaccines are based on individual proteins that induce immune responses targeting only one stage of the malaria parasite’s life cycle. GeoVax’s MVA-VLP malaria vaccine candidates incorporate antigens derived from multiple stages of the parasite’s life cycle and are designed to induce an immune response with durable functional antibodies and CD4+ and CD8+ T cell responses, all hallmarks of an ideal vaccine-induced immune response.
We have collaborated with the Burnet Institute, a leading infectious diseases research institute in Australia, for the development of a vaccine to prevent malaria infection. The project included the design, construction, and characterization of multiple malaria vaccine candidates using GeoVax’s GV-MVA-VLP™ vaccine platform combined with malaria Plasmodium falciparum and Plasmodium vivax sequences identified by the Burnet Institute. The vaccine design, construction, and characterization were performed at GeoVax with immunogenicity and challenge studies in animal models conducted at Burnet Institute using their unique functional assays.
HIV – Due to our corporate refocusing of development efforts prioritizing our SARS-CoV-2 and cancer immunotherapy programs, and to a lack of continuing government support for our HIV vaccine development efforts, in early 2022 we decided to suspend active development of these programs. Our technology and intellectual property will remain available for out-license or partnering, but we are no longer devoting any significant corporate resources to this program.
Of interest, recent results from a clinical study of a combinational HIV therapy that included GeoVax’s HIV booster vaccine candidate, MVA62B were presented at the Conference on Retroviruses and Opportunistic Infections (CROI) held February 19-22, 2023 in Seattle, Washington. The data presented were part of an effort led by researchers at the University of California, San Francisco (UCSF), to develop a combinational therapy aimed at inducing remission in HIV-positive individuals (a “functional cure”). The primary objectives of the proof-of-concept trial were to assess the safety and tolerability of the combinational therapy and to determine the viral load “set-point” during antiviral treatment interruption. Secondary objectives were to assess immune responses and changes in viral reservoir status. The clinical trial was led by Steven Deeks, M.D. of UCSF, a world leader in therapeutic approaches to HIV infections, and was one of the most comprehensive tests to date for the ability of synergistic approaches to control HIV infection. The studies were conducted with funding from amfAR, The Foundation for AIDS Research. The overall goal of this trial was to induce immune responses that could potentially control HIV replication in patients in the absence of antiviral drugs. The data presented by the UCSF researchers indicated very high levels of immunogenicity (particularly T cell immunity), despite the fact these occurred in technically immunocompromised patients. These results further validate the ability of the MVA-VLP platform to stimulate a robust T-cell response to various diseases.
Further development of our Zika, Malaria and HIV programs will be dependent upon additional funding support via federal grants, corporate collaborations, or other sources.
Our GV-MVA-VLP™ Platform
GeoVax’s GV-MVA-VLP™ vaccine platform utilizes Modified Vaccinia Ankara (MVA), a large virus capable of carrying several vaccine antigens, that expresses proteins that assemble into virus-like particles (VLP) immunogens in the person receiving the vaccine. The production of VLPs in the person being vaccinated can mimic the virus production that occurs in a natural infection, stimulating both the humoral and cellular arms of the immune system to recognize, prevent, and control the target infection. The MVA-VLP derived vaccines can elicit durable immune responses in the host similar to a live-attenuated virus, while providing the safety characteristics of a replication-defective vector.
Vaccines typically contain agents (antigens) that resemble disease-causing microorganisms. Traditional vaccines are often made from weakened or killed forms of the virus or from its surface proteins. Some newer vaccines use recombinant DNA (deoxyribonucleic acid) technology to generate vaccine antigens in bacteria or cultured cells from specific portions of the DNA sequence of the target pathogen. The generated antigens are then purified and formulated for use in a vaccine. We believe the most successful of these purified antigens have been non-infectious virus-like particles (VLPs) as exemplified by vaccines for hepatitis B (Merck’s Recombivax® and GSK’s Engerix®) and Papilloma viruses (GSK’s Cervarix®, and Merck’s Gardasil®). Our approach uses recombinant DNA and/or recombinant MVA to produce VLPs in the person being vaccinated (in vivo) reducing complexity and costs of manufacturing. In human clinical trials of our HIV vaccines, we believe we have demonstrated that our VLPs, expressed from within the cells of the person being vaccinated, can be safe, yet elicit both strong and durable humoral and cellular immune response.
VLPs mimic authentic viruses in form but are not infectious or capable of replicating and can cause the body’s immune system to recognize and kill targeted viruses to prevent an infection. VLPs can also train the immune system to recognize and kill virus-infected cells to control infection and reduce the length and severity of disease. One of the biggest challenges with VLP-based vaccines is to design the vaccines in such a way that the VLPs will be recognized by the immune system in the same way as the authentic virus would be. We design our vaccines such that, when VLPs for enveloped viruses like HIV, Ebola or Marburg are produced in vivo (in the cells of the recipient), they include not only the protein antigens, but also an envelope consisting of membranes from the vaccinated individual’s cells. In this way, they are highly similar to the virus generated in a person’s body during a natural infection. VLPs produced in vitro (in a pharmaceutical plant), by contrast, have no envelope or, envelopes from the cultured cells (typically hamster or insect cells) used to produce them. We believe our technology therefore provides distinct advantages by producing VLPs that more closely resemble the authentic viruses. We believe this feature of our immunogens allows the body’s immune system to more readily recognize the virus. By producing VLPs in vivo, we believe we also avoid potential purification issues associated with in vitro production of VLPs.
Figure 1 below shows examples of thin section electron micrographs of actual viruses and VLPs for these viruses expressed by GeoVax MVA-VLP vaccines.
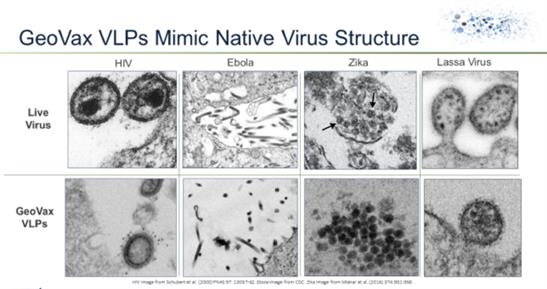
Figure 1. Comparison of MVA-VLPs and native virus structures
In the MVA-VLP platform, we take advantage of MVA’s large “coding capacity” to insert genes that encode multiple proteins, the combination of which is adequate to support the generation of VLPs by the MVA infected cells. Utility has been demonstrated for multiple vaccine candidates wherein the MVA-encoded viral matrix proteins and glycoproteins assemble into VLPs. MVA was originally developed as a safer smallpox vaccine for use in immune-compromised individuals. It was developed by attenuating the standard smallpox vaccine by passaging it (over 500 passages) in chicken embryos or chicken embryo fibroblasts, resulting in a virus with limited ability to replicate in human cells (thus safe) but with high replication capability in avian cells (thus cost effective for manufacturing). The modifications also resulted in the loss of immune evasion genes which assist the spread of wild type smallpox infections, even in the presence of human immune responses.
We collaborated with the laboratory of Dr. Bernard Moss at NIH/NIAID on four different generations of MVA vectors, spanning over 15 years of collaboration, to effectively express vaccine proteins that assemble into VLPs. These efforts led to the development of different shuttle vectors and the identification of multiple insertion sites for introducing foreign genes encoding the vaccine target proteins into MVA in a manner that optimizes each product for manufacturing stability. Each MVA-VLP vaccine has up to two expression cassettes, each encoding one or more antigens selected from pathogens of interest. At a minimum, each vaccine expresses two antigens required for VLP formation. In the case of HIV and hemorrhagic fever vaccines for example, a viral matrix protein and an envelope glycoprotein. We use a synthetic early late promoter that provides high, yet not lethal, levels of insert expression, which is initiated immediately after infection in cells of the vaccinated individual.
Our GV-MVA-VLP™ vaccine platform affords other advantages:
|
● |
Safety: Safety for MVA, generally, has been shown in more than 120,000 subjects in Europe, including immunocompromised individuals during the initial development of MVA and more recently with the development of MVA as a safer vaccine against smallpox. Our HIV vaccines demonstrated outstanding safety in multiple human clinical trials. |
|
● |
Durability: Our technology raises highly durable (long-lasting) vaccine responses. We hypothesize that elicitation of durable vaccine responses is conferred on responding B cells by the vaccinia parent of MVA, which raises highly durable responses for smallpox. |
|
● |
Limited pre-existing immunity to vector: Following the eradication of smallpox in 1980, smallpox vaccinations subsequently ended, leaving all but those born before 1980 and selected populations (such as vaccinated laboratory workers and first responders) unvaccinated and without pre-existing immunity to MVA-derived vaccines. A potential interference of pre-existing immunity to a vector may be more problematic with those vectors related to parent viruses used in routine vaccinations (e.g. measles) or constitute common viruses that infect people of all ages (e.g. cytomegalovirus). |
|
● |
Repeated use of the platform for different vaccines used in sequence. In mouse experiments, we have shown that two of our vaccines (e.g. GV-MVA-VLP-Zika followed by GV-MVA-VLP-Ebola) can be given at <4 week intervals without any negative impact on their immunogenicity (lack of vector immunity). |
|
● |
No need for adjuvants: MVA generally stimulates strong innate immune responses and does not require the use of adjuvants. |
|
● |
Protection against Mpox and Smallpox: MVA vectored vaccines have been previously shown to provide potential protection against Mpox and Smallpox. |
|
● |
Thermal stability: MVA is stable in both liquid and lyophilized formats (> 6 years of storage). |
|
● |
Genetic stability and manufacturability: If appropriately engineered, MVA is genetically stable and can reliably be manufactured in either the established Chick Embryo Fibroblast cell substrate, or novel continuous cell lines that support scalability as well as greater process consistency and efficiency. |
Government Regulation
Regulation by governmental authorities in the United States and other countries is a significant factor in our ongoing research and development activities and in the manufacture of our products. Complying with these regulations involves considerable expertise, time and expense.
In the United States, drugs and biologics are subject to rigorous federal and state regulation. Our products are regulated under the Federal Food, Drug and Cosmetic Act (FD&C Act), the Public Health Service Act, and the regulations promulgated under these statutes, and other federal and state statutes and regulations. These laws govern, among other things, the testing, manufacture, safety, efficacy, labeling, storage, record keeping, approval, advertising and promotion of medications and medical devices. Product development and approval within this regulatory framework is difficult to predict, takes several years and involves great expense. The steps required before a human vaccine may be marketed in the United States include:
|
● |
Preclinical laboratory tests, in vivo preclinical studies and formulation studies; |
|
● |
Manufacturing and testing of the product under strict compliance with current Good Manufacturing Practice (cGMP) regulations; |
|
● |
Submission to the FDA of an Investigational New Drug application for human clinical testing which must become effective before human clinical trials can commence; |
|
● |
Adequate and well-controlled human clinical trials to establish the safety and efficacy of the product; |
|
● |
The submission of a Biologics License Application to the FDA, along with the required user fees; and |
|
● |
FDA approval of the BLA prior to any commercial sale or shipment of the product |
Before marketing any drug or biologic for human use in the United States, the product sponsor must obtain FDA approval. In addition, each manufacturing establishment must be registered with the FDA and must pass a pre-approval inspection before introducing any new drug or biologic into commercial distribution.
The Emergency Use Authorization (EUA) authority granted to the FDA allows the FDA to help strengthen the nation’s public health protections against certain threats by facilitating the availability and use of medical countermeasures needed during public health emergencies. Under section 564 of the FD&C Act, the FDA Commissioner may allow unapproved medical products or unapproved uses of approved medical products to be used in an emergency to diagnose, treat, or prevent serious or life-threatening diseases or conditions caused by threat agents when there are no adequate, approved, and available alternatives. This potentially may provide a faster pathway to market for our Covid-19 or other infectious disease vaccine candidates. This was the approval pathway followed by Pfizer-BioNTech and Moderna for their respective Covid-19 vaccines.
Because GeoVax does not manufacture vaccines for human use within our own facilities, we must ensure compliance both in our own operations and in the outsourced manufacturing operations. All FDA-regulated manufacturing establishments (both domestic establishments and foreign establishments that export products to the United States) are subject to inspections by the FDA and must comply with the FDA’s cGMP regulations for products, drugs and devices.
The FDA determines compliance with applicable statutes and regulations through documentation review, investigations, and inspections. Several enforcement mechanisms are available to the FDA, ranging from a simple demand to correct a minor deficiency to mandatory recalls, closure of facilities, and even criminal charges for the most serious violations.
Even if FDA regulatory clearances are obtained, a marketed product is subject to continual review, and later discovery of previously unknown problems or failure to comply with the applicable regulatory requirements may result in restrictions on the marketing of a product or withdrawal of the product from the market as well as possible civil or criminal sanctions.
Whether or not the FDA has approved the drug, approval of a product by regulatory authorities in foreign countries must be obtained prior to the commencement of commercial sales of the drug in such countries. The requirements governing the conduct of clinical trials and drug approvals vary widely from country to country, and the time required for approval may be longer or shorter than that required for FDA approval.
We also are subject to various federal, state and local laws, regulations, and recommendations relating to safe working conditions, laboratory and manufacturing practices, the experimental use of animals, and the use and disposal of hazardous or potentially hazardous substances used in connection with our research. The extent of government regulation that might result from any future legislation or administrative action cannot be accurately predicted.
Recent Government Initiatives
US Regulators and Senior White House and Congressional Leaders have recently announced multiple objectives and initiatives that may impact GeoVax. These include:
|
- |
Project NextGen and the Rapid Response Partnership Vehicle (RRPV) -- Supporting the Biomedical Advanced Research and Development Authority (BARDA) in its objective to develop innovative vaccine platforms facilitating the production of a next generation broader performing Covid vaccines – GeoVax’s MVA-vectored Covid-19 vaccine candidates (e.g., GEO-CM04S1) is a logical candidate given the features and benefits noted throughout this report. |
|
- |
The reshoring and protection of the domestic biotech ecosystem – GeoVax represents the first domestic source for Smallpox and Mpox production which is currently controlled by a single foreign entity. |
|
- |
Replenishing the US stockpile with additional vaccines addressing Mpox and Hemorrhagic Fevers – GeoVax has multiple products in advanced stages of development. |
|
- |
Assisting African countries in their quest to prevent an array of debilitating illnesses including those caused by hemorrhagic fever viruses Fevers – GeoVax has multiple products in advanced stages of development. |
FDA Tropical Disease Priority Review Voucher Program
Section 524 of the FD&C Act authorizes the FDA to award priority review vouchers (PRVs) to sponsors of approved tropical disease product applications that meet certain criteria. To qualify for a PRV, a sponsor’s application must be for a drug or biological product for the prevention or treatment of a “tropical disease,” must otherwise qualify for priority review, and must contain no active ingredient (including any salt or ester of an active ingredient) that has been approved in any other application under Section 505(b)(1) of the FD&C Act or section 351 of the Public Health Services Act. Priority review means that the FDA aims to render a decision in 6 months.
The PRV may be sold. For example, a small company might win a voucher for developing a drug for a neglected disease and sell the voucher to a large company for use on a commercial disease. The price of the voucher depends on supply and demand. The voucher’s value derives from three factors: shifting sales earlier, longer effective patent life due to earlier entry, and competitive benefits from earlier entry relative to competitors. Top-selling treatments can yield billions in sales each year, so being approved months earlier can be worth hundreds of millions of dollars to the voucher. Since the first voucher sale in 2014, the price of the vouchers has ranged from $68 million to $350 million.
GeoVax believes that its vaccine programs in Ebola, Sudan, Marburg, Lassa Fever, Malaria and Zika may each be eligible for a PRV and we intend to apply for a PRV at the appropriate time. There can be no assurance, however, that we will qualify or be approved for a PRV.
Manufacturing
To be successful, our products must be manufactured in commercial quantities in compliance with regulatory requirements and at an acceptable cost. To date, we have not commercialized any products, nor have we demonstrated that we can manufacture commercial quantities of our product candidates in accordance with regulatory requirements. If we cannot manufacture products in suitable quantities and in accordance with regulatory standards, either on our own or through contracts with third parties, it may delay clinical trials, regulatory approvals and marketing efforts for such products. Such delays could adversely affect our competitive position and our chances of achieving profitability. We cannot be sure that we can manufacture, either on our own or through contracts with third parties, such products at a cost or in quantities that are commercially viable.
Rather than establishing the necessary facilities to manufacture any of the clinical or commercial supplies of our products, our strategy is to rely on established, recognized third-party contract manufacturers to produce materials needed for research and clinical trials. We have arrangements with third party manufacturers for the supply of products for use in our planned clinical trials. These suppliers operate under the FDA’s Good Manufacturing Practices and (in the case of European manufacturers) similar regulations of the European Medicines Agency. We anticipate that these suppliers will be able to provide sufficient supplies to complete our currently planned clinical trials. Various contractors are generally available in the United States and Europe for manufacture of materials for clinical trial evaluation, however, it may be difficult to replace existing contractors for certain manufacturing and testing activities and costs for contracted services may increase substantially if we switch to other contractors.
The raw materials and other supplies that are used in the production process for our vaccines and that we use in our research activities are generally available from a number of commercial suppliers and we believe we will be able to obtain sufficient quantities of such materials and supplies for all foreseeable clinical investigations.
Transition to high-yield, scalable MVA manufacturing process – Currently, MVA vaccines are manufactured in cells cultured from chicken embryonic fibroblasts (CEF), a suboptimal and time-consuming process useful primarily for niche markets and stockpile reserves. After exploring various approaches to growing MVA, utilizing continuous avian cell lines in bioreactors more suitable for high-yield, commercial-scale manufacturing, we have accelerated activities towards fully implementing a proprietary, continuous avian cell line manufacturing system that will provide lower-cost, scalable versatility for broad MVA vaccine and immunotherapy applications. To this end, in September 2023, we announced the signing of a commercial multi-product license agreement for ProBioGen's AGE1.CR.pIX® suspension cell line, an innovative and proven platform that enables high-yield and scalable production, ensuring efficient industrial manufacturing processes. The AGE1.CR.pIX cell line's versatility allows it to support a wide range of viruses and vaccine types, enhancing its suitability for various vaccines in development and as a replacement for traditional production systems. MVA grows particularly well on this cell line, making it even more advantageous for vaccine development.
Developing a high-yield, high-capacity process to produce MVA-based vaccines and immunotherapies constitutes a transformational development – for GeoVax, biomedicine, and the public’s health. By advancing our MVA manufacturing to a modern, interchangeable process, we are on course to expand MVA applications from stockpile-based solutions for niche medical markets to respond to world needs on a timely basis, whenever and wherever they arise. We believe that this capability puts us in the position to be the first supplier of MVA-based vaccines to implement such a transformative manufacturing process and becoming the first U.S.-based supplier of the MVA vaccine to prevent Mpox, Smallpox and other pox-related viruses.
Competition
Our product candidates face, and will continue to face, intense competition from large pharmaceutical companies, specialty pharmaceutical and biotechnology companies as well as academic and research institutions. We compete in an industry that is characterized by rapid technological change; evolving industry standards; emerging competition; and new product introductions. Competitors have existing products and technologies that will compete with our pipeline candidates and technologies and may develop and commercialize additional products and technologies that will compete with our pipeline candidates and technologies. Because competing companies and institutions may have greater financial resources than us, they may be able to provide broader services and product lines; and make greater investments in research and development. Competitors may also have greater development capabilities than we do and have substantially greater experience in undertaking nonclinical and clinical testing of products, obtaining regulatory approvals and manufacturing and marketing pharmaceutical products. They may also have greater name recognition and better access to customers.
We face general market competition from several subsectors of the vaccine development field, including large, multinational pharmaceutical companies including Sanofi, GSK, Merck, Janssen, Mitsubishi Tanabe, Takeda, and Pfizer; mid-size pharmaceutical companies and emerging biotechnology companies including Dynavax, Novavax Inc., Moderna, BioNTech and Bavarian Nordic; and academic and not-for-profit vaccine researchers and developers including the NIH. The industry is typified by extensive collaboration, licensing, and merger and acquisition activity despite the intense competition.
More than forty Covid-19 vaccines are currently authorized for use in one or more countries around the world, including three in the United States (from Pfizer/BioNTech, Moderna, and Janssen). All these vaccines are based on the S protein of the SARS-CoV-2 virus but rely on different mechanisms for presentation or expression of the S antigen, including whole, inactivated virus, defective adenovirus vectors (three different types) or mRNA. The World Health Organization reports that there are 180 Covid products in clinical development.
A number of companies are developing various types of therapeutic vaccines or other immunotherapy approaches to treat cancer including Advaxis, Immune Design, Oncothyreon, Bavarian Nordic, Roche Pharmaceuticals, Merck & Co, Bristol Myers Squibb, and AstraZeneca plc.
There are currently no FDA licensed and commercialized Zika vaccines, or hemorrhagic fever virus vaccines (other than for Ebola Zaire) available in the world market. We are aware of several development-stage and established enterprises, including major pharmaceutical and biotechnology firms, which are actively engaged in vaccine research and development in these areas. For hemorrhagic fever viruses, these include NewLink Genetics and Merck, Johnson & Johnson, Novavax, Inovio and GlaxoSmithKline. For Zika, these include NewLink Genetics, Inovio, Merck, Butantan Institute and NIH (NIAID). In December 2019, the FDA approved the first vaccine (ERVEBO®) for prevention of Ebola Zaire, developed by Merck.
In October 2021, the WHO approved the first malaria vaccine, RTS, S. It requires 4 doses and is based on a single antigen and has modest efficacy (approximately 50%, depending on the age of subjects), the WHO has defined a Road Map for developing and licensing of next generation malaria vaccines. More recently, a vaccine, R21/Matrix-M™️, jointly developed by Oxford University and the Serum Institute of India has met the WHO targeted efficacy to block both infection and transmission of malaria with at least a 75% efficacy rate.
Our Intellectual Property
Our commercial success depends in part on our ability, and our licensors’ ability, to obtain and maintain proprietary protection for our clinical product candidates, including our Modified Vaccinia Ankara-Virus-Like Particle (MVA-VLP) based vaccines, our in-licensed synthetic MVA Covid-19 vaccine candidate, and our in-licensed Gedeptin gene-directed enzyme prodrug therapy, and methods of treatment using the same.
We, and in collaboration with our licensors for our in-licensed assets, seek patent protection on each of our product and developmental candidates and, where applicable, on combinations with other therapeutic and/or antigenic agents and dosing schedules. Our success also depends on our ability to operate without infringing on the proprietary rights of others and to prevent others from infringing our proprietary rights. Our policy is to seek to protect our proprietary position by, among other methods, filing U.S. patent applications and, where appropriate, foreign patent applications covering our proprietary technology, inventions, and improvements that are important to the development and implementation of our business. We collaborate with our licensors to ensure the filing of U.S. patent applications and, where appropriate, foreign patent applications covering our in-licensed technology, inventions, and improvements that are important to the development and implementation of our business. We also rely on trade secrets, know-how, continuing technological innovation and potential in-licensing opportunities to develop and maintain our proprietary position. Additionally, we expect to benefit, where appropriate, from statutory frameworks in the United States, Europe, and other countries that provide a period of clinical data exclusivity to compensate for the time required for regulatory approval of our clinical product candidates.
We continually assess and refine our intellectual property strategies as we develop new technologies and product candidates. We plan to file additional patent applications based on our intellectual property strategies where appropriate, including where we seek to improve our basic technology, adapt to competition, or to improve business opportunities. Further, we plan to file patent applications, as we consider appropriate under the circumstances, to protect new technologies that we develop. Our patent filing strategy typically includes seeking patent protection in the United States and, wherein appropriate, in additional countries where we believe such protection is likely to be useful.
As of December 31, 2023, our owned, co-owned, and in-licensed patent estate, on a worldwide basis, includes 17 granted or allowed U.S. patent applications, 17 pending U.S. patent applications; 63 granted foreign patents, 62 pending foreign patent applications, 3 Patent Cooperation Treaty (PCT) application, and 2 U.S. provisional applications spread over 24 patent families. The term of individual patents depends upon the laws of the countries in which they are obtained. In the countries in which we currently file, the patent term is 20 years from the earliest date of filing of a non-provisional patent application which serves as a priority application. In addition, we plan to seek patent term adjustments, restorations, and/or patent term extensions where applicable in the United States and other jurisdictions. For example, depending upon the timing, duration, and specifics of FDA approval of our vaccine products, some of our U.S. patents may be eligible for a patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, commonly referred to as the “Hatch-Waxman Amendments,” and codified as 35 U.S.C. § 156. 35 U.S.C. § 156 permits restoration of the patent term of up to five years as compensation for patent term lost during product development and FDA regulatory review process. Patent term restoration, however, cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date. The patent term restoration period is generally one half the time between the effective date of an IND and the submission date of a Biologics License Application (BLA), plus the time between the submission date of a BLA and the approval of that application, except that the review period is reduced by any time during which the applicant failed to exercise due diligence. Only one patent applicable to an approved vaccine product is eligible for such an extension and the application for the extension must be submitted prior to the expiration of the patent. The USPTO, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. A similar kind of patent extension, referred to as a Supplementary Protection Certificate, is available in Europe. Legal frameworks are also available in certain other jurisdictions to extend the term of a patent. We currently intend to seek patent term extensions on any of our, or our exclusively licensed, issued patents in any jurisdiction where we have a qualifying patent and the extension is available; however, there is no guarantee that the applicable regulatory authorities, including the FDA in the United States, will agree with our assessment of whether such extensions should be granted, and even if granted, the length of such extensions. Further, even if our patent is extended, the patent, including the extended portion of the patent, may be held invalid or unenforceable by a court of final jurisdiction in the United States or a foreign country.
Our current patent portfolio includes 2 patent families directed to various aspects of our DNA and MVA-based HIV vaccines, their genetic inserts expressing multiple HIV protein components, composition, structure, claim of immunization against multiple subtypes of HIV, routes of administration, safety and other related factors and methods of therapeutic and prophylactic use thereof including administration regimes. We have in-licensed one patent from Emory University and the U.S. National Institutes of Health (NIH) relevant to our HIV-vaccine program. This patent expires in 2028, exclusive of any patent term adjustments or extensions. We wholly own one patent family, including two granted U.S. patents (US 11,098,086 and US 11,897,919), directed to specific vaccine administration methods which, where issued, valid, and enforceable, will expire in 2037, exclusive of any patent term adjustments or extensions.
We wholly own one granted U.S. patent (US 11,701,418) directed to preventive vaccines against Ebola virus, and one granted U.S. patent (US 11,896,657) directed to Marburg virus and uses thereof. These patents, where issued, valid, and enforceable, will expire in 2036, exclusive of any patent term extensions.
We wholly own one granted U.S. patent (US 11,638,750) directed to preventive vaccines against Zika virus and uses thereof. This patent where issued, valid, and enforceable, will expire in 2037, exclusive of any patent term adjustments.
We wholly own two granted U.S. patents (U.S. 11,311,612 and US 11,857,611) directed to preventive vaccines against malaria and use thereof. These applications, where issued, valid, and enforceable, will expire in 2038, exclusive of any patent term adjustments or extensions.
We wholly own 3 patent families, which includes two granted U.S. patents (U.S. 11,278,607 and U.S. 11,413,341), and granted foreign applications in Australia, Europe (validated in Germany, Spain, France, Great Britian, Italy, Poland, Turkey, and Switzerland), China, Japan, India directed to our immuno-oncology vaccine compositions and methods of use thereof. Applications are pending in the United States, Australia, Canada, China, and Hong Kong,. The patent applications of these families, where issued, valid, and enforceable, will expire between 2037-2040, exclusive of any patent term adjustments or extensions.
We wholly own one pending patent family directed to various MVA-based vaccines for the treatment of SARS CoV-2. Applications have been filed in the United States, Argentina, Australia, Brazil, Canada, China, Hong Kong, the European Patent Office, Israel, Japan, South Korea, Mexico, South Africa, and Taiwan. The patent applications in these families, if issued, valid, and enforceable, will expire in 2041, exclusive of any patent term adjustments or extensions. We have non-exclusively in-licensed from the U.S. National Institutes of Health (NIH) 2 patent families directed to certain aspects of our MVA-viral backbone used in our SARS-CoV2 vaccine, which will expire between 2027 and 2032. We have non-exclusively in-licensed from the NIH 2 patent families relating to coronavirus spike protein compositions relevant to our MVA SARS-CoV2 vaccine candidates. The patent applications for these families, where issued, valid, and enforceable, will expire between 2037 and 2041, exclusive of any patent term adjustments or extensions.
We co-own with Leidos, Inc. one patent family directed to viral constructs for use in enhancing T-cell priming during vaccination. Applications have been filed in United States, Australia, Brazil, Canada, China, Europe, Israel, India, Japan, Korea, Mexico, and Taiwan. The patent applications in this patent family, if issued, valid, and enforceable, will expire in 2042, exclusive of any patent term adjustments or extensions.
The MVA backbone that we have been using in our vaccines was provided to us by the laboratory of Dr. Bernard Moss of the NIAID, Laboratory of Viral Diseases (LVD). We have a non-exclusive commercial license to the NIH MVA backbone for our SARS CoV-2 vaccine with the NIAID of the National Institutes of Health NIH on behalf of the United States, which includes the use of certain patents and patent applications arising from the Moss laboratory and the provided materials. This non-exclusive commercial license was further amended in December 2023 to expand the Field of Use to include the use of our SARS-CoV-2 vaccine against smallpox and/or monkeypox. We also have a non-exclusive research and development license to use the MVA backbone for our other vaccine candidates. If we later decide to commercialize vaccine candidates that are under the research and development license, we will need to negotiate appropriate commercialization licenses. These in-licensed NIH patents and patent applications, if and where issued, valid, and enforceable, will expire between 2027 and 2032.
In November 2022, we executed a Material Transfer Agreement (MTA) with the National Institutes of Health (NIH) for the clinical and commercial use of an unmodified (parental) MVA 1974/NIH Clone I as a vaccine against monkeypox virus. The MTA is royalty-free, non-exclusive, and worldwide.
We have exclusively in-licensed five patent families from the City of Hope in the field of vaccine products targeted for prevention, reduction, amelioration or treatment of coronaviruses, including Covid-19, pursuant to an Exclusive License Agreement entered into on November 9, 2021, and as further amended on April 11, 2023. The in-licensed patent families are directed to synthetic MVA vectors, including synthetic MVA vaccines encoding one or more SARS-CoV-2 antigens, and their methods of production and use including for the prevention of a coronavirus and monkeypox infection, and encompass COH04S1, a multi-antigenic pan-SARS vaccine currently undergoing Phase 2 human clinical trials. These in-licensed City of Hope patent families, if issued, valid, and enforceable, will expire between 2041 and 2043, exclusive of any patent term adjustments or extensions.
We have also exclusively in-licensed two additional patent families from the City of Hope in the field of vaccine products targeted for prevention, reduction, amelioration or treatment of Covid-19 variants. The in-licensed patent families are directed to synthetic MVA vectors, including synthetic MVA vaccines encoding one or more SARS-CoV-2 variant antigens, and their methods of production and use. Applications have been filed in the United States. These in-licensed City of Hope patent families, if issued, valid, and enforceable, will expire in 2042, exclusive of any patent term adjustments or extensions.
We have exclusively in-licensed two patent families from the University of Alabama at Birmingham (“UAB”) and the Southern Research Institute pursuant to an Assignment and License Agreement with PNP Therapeutics, Inc. entered into on September 28, 2021. The two patent families are directed to the use of tail-mutant purine nucleoside phosphorylase enzymes and fludarabine for the treatment of cancer, and cover aspects of the use of our Gedeptin clinical product candidate. These in-licensed patent families, where issued, valid, and enforceable, will expire between 2029 and 2032, exclusive of any patent term adjustments or extensions.
We cannot be certain that any of the current pending patent applications we have or have licensed, or any new patent applications we may file or license, will ever be issued in the United States or any other country. Even if issued, there can be no assurance that those patents will be sufficiently broad to prevent others from using our products or processes. Furthermore, our patents, as well as those we have licensed or may license in the future, may be held invalid or unenforceable by a court, or third parties could obtain patents that we would need to either license or to design around, which we may be unable to do. Current and future competitors may have licensed or filed patent applications or received patents and may acquire additional patents or proprietary rights relating to products or processes competitive to ours. In addition, any claims relating to the infringement of third-party proprietary rights, or earlier date of invention, even if not meritorious, could result in costly litigation, lengthy governmental proceedings, divert management’s attention and resources and require us to enter royalty or license agreements which are not advantageous to us, if available at all.
We also expect to benefit, where appropriate, from statutory frameworks in the United States, Europe, and other countries that provide a period of regulatory exclusivity to compensate for the time and cost required in securing regulatory approval of our clinical products. For example, in 2010, the United States enacted the Biologics Price Competition and Innovation Act (BPCIA). Under the BPCIA, innovator manufacturers of biological products may be granted 12 years of exclusive use before biosimilar versions of such products can be licensed for marketing in the U.S. This means that the FDA may not approve an application for a biosimilar version of our products until 12 years after the date the product is approved for sale (with a potential six-month extension of exclusivity if certain pediatric studies are conducted and the results accepted by the FDA), although a biosimilar application may be submitted four years after the date we receive approval from the FDA to sell our product. Additionally, the BPCIA establishes procedures by which potentially relevant patents may be shared and litigation over patents may proceed in advance of approval. The BPCIA also provides incentives to biosimilar applicants by providing a period of exclusivity to the first biosimilar of a product approved by the FDA. The 12-year data exclusivity provision of the BPCIA does not prevent a competitor from seeking marketing approval of one of our products, or a product similar thereto, by submitting its own, original Biologics License Application (BLA).
We intend to benefit, where applicable, from additional market exclusivity provisions in various jurisdictions that reward the treatments of rare diseases. For example, in the United States under the Orphan Drug Act of 1983, the FDA may grant orphan designation to a vaccine product intended to prevent or treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the United States, or more than 200,000 individuals in the United States and for which there is no reasonable expectation that the cost of developing and making the product available in the United States for this type of disease or condition will be recovered from sales of the product. Orphan designation must be requested before submitting a BLA. After the FDA grants orphan designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. If a product that has orphan designation subsequently receives the first FDA approval for the disease or condition for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications to market the same drug for the same indication for seven years from the date of such approval, except in limited circumstances, such as a showing of clinical superiority to the product with orphan exclusivity by means of greater effectiveness, greater safety, or providing a major contribution to patient care, or in instances of drug supply issues. Competitors, however, may receive approval of either a different product for the same indication or the same product for a different indication; in the latter case, because health care professionals are free to prescribe products for off-label uses, the competitor’s product could be used for the orphan indication despite our orphan exclusivity.
We are not a party to any litigation, opposition, interference, or other potentially adverse proceeding with regard to our patent positions. However, if we become involved in litigation, interference proceedings, oppositions or other intellectual property proceedings, for example as a result of an alleged infringement or a third-party alleging an earlier date of invention, we may have to spend significant amounts of money and time and, in the event of an adverse ruling, we could be subject to liability for damages, invalidation of our intellectual property and injunctive relief that could prevent us from using technologies or developing products, any of which could have a significant adverse effect on our business, financial conditions or results of operations. In addition, any claims relating to the infringement of third-party proprietary rights, or earlier date of invention, even if not meritorious, could result in costly litigation, lengthy governmental proceedings, divert management’s attention and resources and require us to enter royalty or license agreements which are not advantageous if available at all.
In addition to patents, we rely upon unpatented, proprietary trade secrets and know-how and continuing technological innovation to develop and maintain our competitive position. We seek to protect our proprietary information, in part, using confidentiality agreements with our commercial partners, collaborators, employees, and consultants, and invention assignment agreements with our employees. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with a third party. These agreements may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our commercial partners, collaborators, employees, and consultants use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
Primary License Agreements
City of Hope License – On November 9, 2021, we entered into an Exclusive License Agreement (COH License) with City of Hope (COH), a California nonprofit public benefit corporation, under which the Company obtained exclusive worldwide rights to further develop and commercialize COH04S1, a multi-antigenic SARS-CoV-2 vaccine currently undergoing Phase 2 human clinical trials. The COH License grants GeoVax exclusive rights to key patents, know-how, regulatory filings and clinical materials for use against Covid-19. The terms of the COH License, include an upfront fee consisting of an initial payment to COH of $5,000,000 within 30 days of the effective date of the COH License, and additional payments of $3,000,000 and $2,000,000 on the first and second anniversaries, respectively, of the effective date of the COH License. The terms also include milestone payments due upon the achievement of selected development, regulatory and sales events. The Company will also pay COH an annual royalty on net sales of products covered by the patents licensed from COH on a country-by-country and licensed product-by-licensed product basis, subject to specified reductions.
Gedeptin License – On September 28, 2021, we entered into an Assignment and License Agreement (the “Gedeptin License”) with PNP Therapeutics, Inc. (“PNP”) under which the Company obtained exclusive worldwide rights to key intellectual property, including Gedeptin patents, know-how, regulatory filings, clinical materials, and trademarks. The Gedeptin patent portfolio was originally licensed from the University of Alabama at Birmingham (“UAB”) and Southern Research Institute (“SRI”) by PNP. Under the terms of the Gedeptin License, the Company is the successor to PNP under the Exclusive License Agreement between UAB, SRI and PNP, and has acquired the exclusive rights to develop and commercialize Gedeptin, a novel patented product for the treatment of solid tumors.
The terms of the Gedeptin License, include (i) an upfront payment at closing, (ii) milestone payments due upon the achievement of selected development and regulatory events, and (iii) quarterly support payments for the lesser period of three years or the Company’s filing for FDA approval of its Biologics License Application on the use of Gedeptin for the treatment of head and neck cancer in humans. The Company will also pay tiered percentage annual royalties in the low-to-mid teens on Net Sales (as defined in the Gedeptin License) of products covered under the Gedeptin License on a country-by-country and product-by-product basis, subject to specified reductions. The Company also issued a warrant to PNP, exercisable at any time following March 28, 2022, and prior to September 28, 2026, for up to 6,667 shares of the Company’s common stock at an exercise price of $195.00 per share. The Gedeptin License will remain in effect during the original term, which concludes upon FDA approval of a generic or biosimilar product, and then will automatically renew for 5-year additional terms, subject to customary termination rights.
NIH Licenses – On December 16, 2022, the Company entered into a Clinical Materials Transfer Agreement (MVA Vaccine Agreement) under which the Company has the right to develop and commercialize the unmodified (parental) MVA 1974/NTH Clone l strain as a vaccine against Mpox and smallpox.
On November 25, 2020, the Company entered into a Patent and Biological Materials License Agreement for Internal Research Use (the “Research License”) with the U.S. Department of Health and Human Services (HHS), as represented by NIAID, in support of the Company’s non-clinical development of vaccines against numerous pathogens. The Research License allows GeoVax to use these materials and patent rights owned by agencies of the HHS in combination with the Company’s proprietary technology for the creation of preventive and/or therapeutic Modified Vaccinia Ankara Virus-Virus Like Particle (MVA-VLP) vaccines against Ebola-Zaire virus, Ebola-Sudan virus, Lassa virus, Marburg virus, Zika virus and malaria. The agreement also extends to the Company’s research and development efforts in certain oncology areas. The agreement provides GeoVax with nonexclusive rights for the nonclinical development and manufacturing of its vaccine and immunotherapy candidates using HHS patents and materials.
On October 22, 2020, the Company entered into a Patent and Biological Materials License Agreement (the “Covid License”) with HHS, as represented by NIAID, in support of the Company’s development of a vaccine against SARS-CoV-2, the virus that causes Covid-19. The Covid License allows GeoVax to use these materials and patent rights owned by agencies of the HHS in combination with the Company’s proprietary technology for the creation of a preventive Modified Vaccinia Ankara Virus-Virus Like Particle (MVA-VLP) vaccine that primes and/or boosts the immune system against Covid-19. The Covid License provides GeoVax with nonexclusive rights to develop, manufacture and commercialize its Covid-19 vaccine and includes access to NIAID’s patent rights in the stabilized SPIKE protein, which is the protein that SARS-CoV-2 uses to gain entry into human tissue. In December 2023, the Covid License was amended to expand GeoVax’s commercial license to include Mpox and smallpox as additional indications.
Research and Development
Our expenditures for research and development activities were $20.7 million and $9.1 million during the years ended December 31, 2023 and 2022, respectively. As our vaccines continue to go through the process to obtain regulatory approval, we expect our research and development costs to increase. We have not yet formulated any plans for marketing and sales of any vaccine candidate we may successfully develop. Compliance with environmental protection laws and regulations has not had a material effect on our capital expenditures, earnings or competitive position to date.
Scientific Advisors
We seek advice from our Scientific Advisory Board, which consists of a number of leading scientists, on scientific and medical matters. The current members of our Scientific Advisory Board are:
|
Name |
Position/Institutional Affiliation |
|
|
Harriet L. Robinson, PhD. |
Chief Scientific Officer Emeritus, GeoVax |
|
|
Stanley A. Plotkin, MD |
Professor Emeritus, University of Pennsylvania, Adjunct Professor, Johns Hopkins University |
|
|
Barney S. Graham, MD, PhD |
Senior Investigator (retired), Vaccine Research Center, NIAID |
|
|
Scott C. Weaver, PhD |
Director, University of Texas Medical Branch Institute for Human Infections and Immunity |
|
|
Olivera J. Finn, PhD |
Distinguished Professor of Immunology and Surgery, University of Pittsburgh |
Properties
Our principal executive offices are located in Smyrna, Georgia, where we lease approximately 8,400 square feet of office and laboratory space. Our lease for the premises is currently scheduled to terminate on December 31, 2025. We do not currently own any real property. We believe that our current facilities are adequate to meet our immediate needs, but that if we require additional space, we will be able to obtain additional facilities on commercially reasonable terms.
Human Capital Resources
We currently have seventeen full-time employees. None of our employees are covered by collective bargaining agreements and we believe that our employee relations are good. We also engage consultants and independent contractors to fulfill key roles and/or provide expert services on both an ongoing and short-term basis.
We believe that our future success largely depends upon our continued ability to attract and retain highly skilled employees. We provide our employees with competitive compensation, opportunity for equity ownership, and a robust employment package that promotes wellness across all aspects of their lives, including healthcare, retirement planning, and paid time off.
Corporate Background
Our primary business is conducted by our wholly owned subsidiary, GeoVax, Inc., which was incorporated under the laws of Georgia in June 2001. Our address is 1900 Lake Park Drive, Smyrna, Georgia 30080, and our telephone number at that address is 678-384-7220. The predecessor of our parent company, GeoVax Labs, Inc. (the reporting entity) was originally incorporated in June 1988 under the laws of Illinois as Dauphin Technology, Inc. (“Dauphin”). In September 2006, Dauphin completed a merger with GeoVax, Inc. As a result of the merger, GeoVax, Inc. became a wholly owned subsidiary of Dauphin, and Dauphin changed its name to GeoVax Labs, Inc. In June 2008, the Company was reincorporated under the laws of Delaware. We currently do not conduct any business other than GeoVax, Inc.’s business of developing new products for the treatment or prevention of human diseases. Our principal offices are in Smyrna, Georgia (metropolitan Atlanta).
Available Information
Our website address is www.geovax.com. We make our SEC filings, such as proxy statements, Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to those reports available on this website under “Investors – SEC Reports,” free of charge, as soon as reasonably practicable after we electronically file or furnish such materials to the SEC. We also make available our Code of Business Conduct on this website under the heading “Investors – Corporate Governance”. Information contained on our website is not incorporated into this Annual Report.
|
ITEM 1A. |
RISK FACTORS |
Ownership of our securities involves a high degree of risk. You should carefully review and consider the risks, uncertainties and other factors described below before you decide whether to own our securities. Any of these factors could materially and adversely affect our business, financial condition, operating results and prospects and could negatively impact the market price of our common stock, and you may lose some or all of your investment. The risks and uncertainties described below are not the only ones facing our Company. Additional risks and uncertainties that we are unaware of, or that we currently deem immaterial, may also impair our business operations. You should also refer to the other information contained in this Form 10-K, including our financial statements and the related notes.
Summary of Risk Factors
Our business is subject to numerous risks and uncertainties, discussed in more detail in the following section. These risks include, among others, the following key risks:
Risks Related to Our Business and Capital Requirements
|
● |
We have a history of operating losses, and we expect losses to continue for the foreseeable future. |
|
● |
We have received a going concern opinion from our auditors. |
|
● |
Our business will require continued funding. If we do not receive adequate funding, we may not be able to continue our operations. |
|
● |
Significant disruptions of information technology systems or breaches of information security systems could adversely affect our business. |
Risks Related to Development and Commercialization of Product Candidates and Dependence on Third Parties
|
● |
Our products are still being developed and are unproven. These products may not be successful. |
|
● |
We depend upon key personnel who may terminate their employment with us at any time. If we were to lose the services of any of these individuals, our business and operations may be adversely affected. |
|
● |
Regulatory and legal uncertainties could result in significant costs or otherwise harm our business. |
|
● |
We face intense competition and rapid technological change that could result in products that are superior to, or earlier to the market than, the products we will be commercializing or developing. |
|
● |
Our product candidates are based on new medical technology and, consequently, are inherently risky. Concerns about the safety and efficacy of our products could limit our future success. |
|
● |
We may experience delays in our clinical trials that could adversely affect our financial results and our commercial prospects. |
|
● |
Failure to obtain timely regulatory approvals required to exploit the commercial potential of our products could increase our future development costs or impair our future sales. |
|
● |
State pharmaceutical marketing compliance and reporting requirements may expose us to regulatory and legal action by state governments or other government authorities. |
|
● |
Changes in healthcare law and implementing regulations, as well as changes in healthcare policy, may impact our business in ways that we cannot currently predict, and may have a significant adverse effect on our business and results of operations. |
|
● |
We may not be successful in establishing collaborations for product candidates we seek to commercialize, which could adversely affect our ability to discover, develop, and commercialize products. |
|
● |
We do not have manufacturing, sales or marketing experience. |
|
● |
Our products under development may not gain market acceptance. |
|
● |
We may be required to defend lawsuits or pay damages for product liability claims. |
|
● |
Reimbursement decisions by third-party payors may have an adverse effect on pricing and market acceptance. If there is not sufficient reimbursement for our products, it is less likely that they will be widely used. |
Risks Related to Our Intellectual Property
|
● |
Our success depends on our ability to obtain, maintain, protect and enforce our intellectual property and our proprietary technologies. |
|
● |
We could lose our license rights to our important intellectual property if we do not fulfill our contractual obligations to our licensors. |
|
● |
Other parties may claim that we infringe their intellectual property or proprietary rights, which could cause us to incur significant expenses or prevent us from selling products. |
|
● |
Any inability to protect our or our licensors’ intellectual property rights in the United States and foreign countries could limit our ability to prevent others from manufacturing or selling our products. |
|
● |
Changes in United States patent law could diminish the value of patents in general, thereby impairing our ability to protect our product candidates. |
|
● |
The patent protection and patent prosecution for our product candidates is dependent in part on third parties. |
Risks Related to Our Common Stock
|
● |
The market price of our common stock is highly volatile. |
|
● |
The sale or issuance of additional shares of our common stock or other equity securities could result in additional dilution to our stockholders. |
|
● |
Certain provisions of our certificate of incorporation which authorize the issuance of shares of preferred stock may make it more difficult for a third party to effect a change in control. |
|
● |
We have never paid dividends and have no plans to do so. |
|
● |
Public company compliance may make it more difficult for us to attract and retain officers and directors. |
|
● |
Our Certificate of Incorporation and Bylaws may be amended by the affirmative vote of a majority of our stockholders. |
|
● |
Broker-dealers may be discouraged from effecting transactions in shares of our common stock if we are considered to be a penny stock and thus subject to the penny stock rules. |
Risks Related to Our Business and Capital Requirements
We have a history of operating losses, and we expect losses to continue for the foreseeable future.
As a research and development-focused company, we have had no product revenue to date and revenues from our government grants and other collaborations have not generated sufficient cash flows to cover operating expenses. Since our inception, we have incurred operating losses each year due to costs incurred in connection with research and development activities and general and administrative expenses associated with our operations. We incurred a net loss of approximately $26 million for the year ended December 31, 2023. We expect to incur additional operating losses and expect cumulative losses to increase as our research and development, preclinical, clinical, and manufacturing efforts expand. Our ability to generate revenue and achieve profitability depends on our ability to successfully complete the development of our product candidates, conduct preclinical tests and clinical trials, obtain the necessary regulatory approvals, and manufacture and market or otherwise commercialize our products. Unless we are able to successfully meet these challenges, we will not be profitable and may not remain in business.
We have received a going concern opinion from our auditors.
We have received a "going concern" opinion from our independent registered public accounting firm, reflecting substantial doubt about our ability to continue as a going concern. Our consolidated financial statements contemplate that we will continue as a going concern and do not contain any adjustments that might result if we were unable to continue as a going concern. Our ability to continue as a going concern is dependent upon our ability to raise additional capital and implement our business plan. If we are unable to achieve or sustain profitability or to secure additional financing on acceptable terms, we may not be able to meet our obligations as they come due, raising substantial doubts as to our ability to continue as a going concern. Any such inability to continue as a going concern may result in our stockholders losing their entire investment. There is no guarantee that we will become profitable or secure additional financing on acceptable terms.
Our business will require continued funding. If we do not receive adequate funding, we may not be able to continue our operations.
To date, we have financed our operations principally through the sale of our equity securities and through government grants and clinical trial support. We will require substantial additional financing at various intervals for our operations, including clinical trials, operating expenses, intellectual property protection and enforcement, for pursuit of regulatory approvals, and for establishing or contracting out manufacturing, marketing and sales functions. There is no assurance that such additional funding will be available on terms acceptable to us or at all. If we are not able to secure the significant funding that is required to maintain and continue our operations at current levels, or at levels that may be required in the future, we may be required to delay clinical studies or clinical trials, curtail operations, or obtain funds through collaborative arrangements that may require us to relinquish rights to some of our products or potential markets.
We may pursue additional support from the federal government for our vaccine and immunotherapy development programs; however, as we progress to the later stages of our development activities, government financial support may be more difficult to obtain, or may not be available at all. Therefore, it will be necessary for us to look to other sources of funding to finance our development activities.
We expect that our current working capital will be sufficient to support our planned level of operations into the second quarter of 2024. We will need to raise additional funds to significantly advance our vaccine development programs and to continue our operations. In order to meet our operating cash flow needs we plan to seek sources of non-dilutive capital through government grant programs and clinical trial support. We may also plan additional offerings of our equity securities, debt, or convertible debt instruments. Should the financing we require to sustain our working capital needs be unavailable or prohibitively expensive when we require it, the consequences could have a material adverse effect on our business, operating results, financial condition and prospects.
Significant disruptions of information technology systems or breaches of information security systems could adversely affect our business.
We rely upon a combination of information technology systems and traditional recordkeeping to operate our business. In the ordinary course of business, we collect, store, and transmit confidential information (including, but not limited to, personal information and intellectual property). We have also outsourced elements of our operations to third parties, including elements of our information technology systems and, as a result, we manage a number of independent vendor relationships with third parties who may or could have access to our confidential information. Our information technology and information security systems and records are potentially vulnerable to security breaches, service interruptions, or data loss from inadvertent or intentional actions by our employees or vendors. Our information technology and information security systems and records are also potentially vulnerable to malicious attacks by third parties. Such attacks are of ever-increasing levels of sophistication and are made by groups and individuals with a wide range of expertise and motives (including, but not limited to, financial crime, industrial espionage, and market manipulation).
While we have invested, and continue to invest, a portion of our limited funds in our information technology and information security systems, there can be no assurance that our efforts will prevent security breaches, service interruptions, or data losses. Any security breaches, service interruptions, or data losses could adversely affect our business operations and/or result in the loss of critical or sensitive confidential information or intellectual property, and could result in financial, legal, business, and reputational harm to us or allow third parties to gain material, inside information that they may use to trade in our securities.
Risks Related to Development and Commercialization of Product Candidates and Dependence on Third Parties
Our products are still being developed and are unproven. These products may not be successful.
To become profitable, we must generate revenue through sales of our products. However, our products are in varying stages of development and testing. Our products have not been proven in human clinical trials and have not been approved by any government agency for sale. If we cannot successfully develop and prove our products and processes, or if we do not develop other sources of revenue, we will not become profitable and at some point, we would discontinue operations.
We depend upon key personnel who may terminate their employment with us at any time. If we were to lose the services of any of these individuals, our business and operations may be adversely affected.
The success of our business strategy will depend to a significant degree upon the continued services of key management, technical and scientific personnel and our ability to attract and retain additional qualified personnel and managers. Competition for qualified personnel is intense among companies, academic institutions and other organizations. The ability to attract and retain personnel is adversely affected by our financial challenges. If we are unable to attract and retain key personnel and advisors, it may negatively affect our ability to successfully develop, test, commercialize and market our products and product candidates.
Regulatory and legal uncertainties could result in significant costs or otherwise harm our business.
To manufacture and sell our products, we must comply with extensive domestic and international regulation. In order to sell our products in the United States, approval from the U.S. Food and Drug Administration (the “FDA”) is required. Satisfaction of regulatory requirements, including FDA requirements, typically takes many years, and if approval is obtained at all, it is dependent upon the type, complexity and novelty of the product, and requires the expenditure of substantial resources. We cannot predict whether our products will be approved by the FDA. Even if they are approved, we cannot predict the time frame for approval. Foreign regulatory requirements differ from jurisdiction to jurisdiction and may, in some cases, be more stringent or difficult to meet than FDA requirements. As with the FDA, we cannot predict if or when we may obtain these regulatory approvals. If we cannot demonstrate that our products can be used safely and successfully in a broad segment of the patient population on a long-term basis, our products would likely be denied approval by the FDA and the regulatory agencies of foreign governments.
We face intense competition and rapid technological change that could result in products that are superior to, or earlier to the market than, the products we will be commercializing or developing.
The market for vaccines that protect against or treat human infectious diseases is intensely competitive and is subject to rapid and significant technological change. We have numerous competitors in the United States and abroad, including, among others, large companies with substantially greater resources than us. If any of our competitors develop products with efficacy or safety profiles significantly better than our products, we may not be able to commercialize our products, and sales of any of our commercialized products could be harmed. Some of our competitors and potential competitors have substantially greater product development capabilities and financial, scientific, marketing and human resources than we do. Competitors may develop products earlier, obtain FDA approvals for products more rapidly, or develop products that are more effective than those under development by us. We will seek to expand our technological capabilities to remain competitive; however, research and development by others may render our technologies or products obsolete or noncompetitive or result in treatments or cures superior to ours.
Our product candidates are based on new medical technology and, consequently, are inherently risky. Concerns about the safety and efficacy of our products could limit our future success.
We are subject to the risks of failure inherent in the development of product candidates based on new medical technologies. These risks include the possibility that the products we create will not be effective, that our product candidates will be unsafe or otherwise fail to receive the necessary regulatory approvals, and that our product candidates will be difficult to manufacture on a large scale or will be uneconomical to market.
Many pharmaceutical products cause multiple potential complications and side effects, not all of which can be predicted with accuracy and many of which may vary from patient to patient. Long term follow-up data may reveal previously unidentified complications associated with our products. The responses of potential physicians and others to information about complications could materially adversely affect the market acceptance of our products, which in turn would materially harm our business.
We may experience delays in our clinical trials that could adversely affect our financial results and our commercial prospects.
We do not know whether planned pre-clinical and clinical trials will begin on time or whether we will complete any of our trials on schedule, if at all. Product development costs will increase if we have delays in testing or approvals, or if we need to perform more or larger clinical trials than planned. Significant delays may adversely affect our financial results and the commercial prospects for our products and delay our ability to become profitable.
We rely heavily on independent clinical investigators, vaccine manufacturers, and other third-party service providers for successful execution of our clinical trials, but do not control many aspects of their activities. We are responsible for ensuring that each of our clinical trials is conducted in accordance with the general investigational plan and protocols for the trial. Moreover, the FDA requires us to comply with standards, commonly referred to as Good Clinical Practices, for conducting, recording, and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of trial participants are protected. Our reliance on third parties that we do not control does not relieve us of these responsibilities and requirements. Third parties may not complete activities on schedule or may not conduct our clinical trials in accordance with regulatory requirements or our stated protocols. The failure of these third parties to carry out their obligations could delay or prevent the development, approval and commercialization of our product candidates.
Failure to obtain timely regulatory approvals required to exploit the commercial potential of our products could increase our future development costs or impair our future sales.
None of our vaccines are approved by the FDA for sale in the United States or by other regulatory authorities for sale in foreign countries. To exploit the commercial potential of our technologies, we are conducting and planning to conduct additional pre-clinical studies and clinical trials. This process is expensive and can require a significant amount of time. Failure can occur at any stage of testing, even if the results are favorable. Failure to adequately demonstrate safety and efficacy in clinical trials could delay or preclude regulatory approval and restrict our ability to commercialize our technology or products. Any such failure may severely harm our business. In addition, any approvals we obtain may not cover all of the clinical indications for which approval is sought or may contain significant limitations in the form of narrow indications, warnings, precautions or contraindications with respect to conditions of use, or in the form of onerous risk management plans, restrictions on distribution, or post-approval study requirements.
State pharmaceutical marketing compliance and reporting requirements may expose us to regulatory and legal action by state governments or other government authorities.
Several states have enacted legislation requiring pharmaceutical companies to establish marketing compliance programs and file periodic reports on sales, marketing, pricing and other activities. Similar legislation is being considered in other states. Many of these requirements are new and uncertain, and available guidance is limited. Unless we are in full compliance with these laws, we could face enforcement action, fines, and other penalties and could receive adverse publicity, all of which could harm our business.
Changes in healthcare law and implementing regulations, as well as changes in healthcare policy, may impact our business in ways that we cannot currently predict, and may have a significant adverse effect on our business and results of operations.
In the United States and foreign jurisdictions, there have been, and continue to be, several legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of product candidates, restrict or regulate post-approval activities, and affect our ability to profitably sell any product candidates for which we obtain marketing approval. Among policy makers and payors in the United States and elsewhere, including in the European Union, there is significant interest in promoting changes in healthcare systems with the stated goals of containing healthcare costs, improving quality and/or expanding access. In the United States, the pharmaceutical industry has been a particular focus of these efforts and has been significantly affected by major legislative initiatives.
There has also been heightened governmental scrutiny recently over the manner in which drug manufacturers set prices for their marketed products. There have been several Congressional inquiries and proposed bills, as well as state efforts, designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drug products. In June 2017, the FDA issued a Drug Competition Action plan intended to lower prescription drug prices by encouraging competition from generic versions of existing products. In July 2018, the FDA issued a Biosimilar Action Plan, intended to similarly promote competition to prescription biologics from biosimilars. In August 2020, the Inflation Reduction Act included provisions to increase Medicare’s ability to negotiate prescription drug prices.
Individual states in the United States have also become increasingly aggressive in passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures. For example, in September 2017, the California State Assembly approved SB17, which requires pharmaceutical companies to notify health insurers and government health plans at least 60 days before any scheduled increases in the prices of their products if they exceed 16% over a two-year period, and further requiring pharmaceutical companies to explain the reasons for such increase and, effective in 2016, Vermont passed a law requiring certain manufacturers identified by the state to justify their price increases.
We expect that these, and other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and lower reimbursement, and in downward pressure on the price that we receive for any approved product. Any reduction in reimbursement from Medicare or other government-funded programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our drugs, once marketing approval is obtained.
We may not be successful in establishing collaborations for product candidates we seek to commercialize, which could adversely affect our ability to discover, develop, and commercialize products.
We expect to seek collaborations for the development and commercialization of product candidates in the future. The timing and terms of any collaboration will depend on the evaluation by prospective collaborators of the clinical trial results and other aspects of a product’s safety and efficacy profile. If we are unable to reach agreements with suitable collaborators for any product candidate, we will be forced to fund the entire development and commercialization of such product candidates, ourselves, and we may not have the resources to do so. If resource constraints require us to enter into a collaboration agreement early in the development of a product candidate, we may be forced to accept a more limited share of any revenues the product may eventually generate. We face significant competition in seeking appropriate collaborators. Moreover, these collaboration arrangements are complex and time-consuming to negotiate and document. We may not be successful in our efforts to establish collaborations or other alternative arrangements for any product candidate. Even if we are successful in establishing collaborations, we may not be able to ensure fulfillment by collaborators of their obligations or our expectations.
We do not have manufacturing, sales or marketing experience.
We do not have experience in manufacturing, selling, or marketing. To obtain the expertise necessary to successfully manufacture, market, and sell our products, we must develop our own commercial infrastructure and/or collaborative commercial arrangements and partnerships. Our ability to execute our current operating plan is dependent on numerous factors, including, the performance of third-party collaborators with whom we may contract.
Our products under development may not gain market acceptance.
Our products may not gain market acceptance among physicians, patients, healthcare payers and the medical community. Significant factors in determining whether we will be able to compete successfully include:
|
● |
the efficacy and safety of our products; |
|
● |
the time and scope of regulatory approval; |
|
● |
reimbursement coverage from Medicare, Medicaid, insurance companies and others; |
|
● |
the price and cost-effectiveness of our products, especially as compared to any competitive products; and |
|
● |
the ability to maintain patent protection; and |
|
● |
the market demand is not readily known. |
We may be required to defend lawsuits or pay damages for product liability claims.
Product liability is a major risk in testing and marketing biotechnology and pharmaceutical products. We may face substantial product liability exposure in human clinical trials and for products that we sell after regulatory approval. We carry product liability insurance and we expect to continue such policies. However, product liability claims, regardless of their merits, could exceed policy limits, divert management’s attention, and adversely affect our reputation and demand for our products.
Reimbursement decisions by third-party payors may have an adverse effect on pricing and market acceptance. If there is not sufficient reimbursement for our products, it is less likely that they will be widely used.
Market acceptance of products we develop, if approved, will depend on reimbursement policies and may be affected by, among other things, future healthcare reform measures. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which drugs they will cover and establish payment levels. We cannot be certain that reimbursement will be available for any products that we may develop. Also, we cannot be certain that reimbursement policies will not reduce the demand for, or the price paid for our products. If reimbursement is not available or is available on a limited basis, we may not be able to successfully commercialize products that we develop.
Risks Related to Our Intellectual Property
Our success depends on our ability to obtain, maintain, protect, and enforce our intellectual property and our proprietary technologies.
In general, our commercial success will depend in part on our and our licensors’ ability to obtain, maintain, protect, and enforce our intellectual property and proprietary technologies, including patent protection and trade secret protection for our product candidates, proprietary technologies and their uses as well as our ability to operate without infringing, misappropriating, or otherwise violating the intellectual property rights of others. If we or our licensors are unable to obtain, maintain, protect, or enforce our intellectual property rights or if our intellectual property rights are inadequate for our technology or our product candidates, our competitive position could be harmed, which could have a material adverse impact on our business, results of operations, financial conditions, and prospects. There can be no assurance that our patent applications will result in patents being issued or that issued patents will afford sufficient protection against competitors with similar technologies, nor can there be any assurance that the patents if issued will not be infringed, misappropriated, violated, designed around or invalidated by third parties. Even issued patents may later be found invalid or unenforceable or may be modified or revoked in proceedings instituted by third parties before various patent offices or in courts. The degree of future protection for our intellectual property is uncertain. Only limited protection may be available and may not adequately obtain, maintain, protect, and enforce our rights or permit us to gain or keep any competitive advantage. These uncertainties and/or limitations in our ability to properly obtain, maintain, protect, and enforce the intellectual property rights relating to our product candidates could have a material adverse effect on our financial condition and results of operations.
We cannot be certain that the claims in our in-licensed pending patent applications will be considered patentable by the U.S. Patent and Trademark Office, or USPTO, courts in the United States or by the patent offices and courts in foreign countries, nor can we be certain that claims that may ultimately issue from our patent applications will not be found invalid or unenforceable if challenged. If we or our licensors are unable to obtain or maintain patent protection with respect to our product candidates, our business, financial condition, results of operations, and prospects could be materially harmed.
We could lose our license rights to our important intellectual property if we do not fulfill our contractual obligations to our licensors.
Our rights to significant parts of the technology we use in our products are licensed from third parties and are subject to termination if we do not fulfill our contractual obligations to our licensors. Termination of intellectual property rights under any of our license agreements could adversely impact our ability to produce or protect our products. Our obligations under our license agreements include requirements that we make milestone payments to our licensors upon the achievement of clinical development and regulatory approval milestones, royalties as we sell commercial products, and reimbursement of patent filing and maintenance expenses. Should we become bankrupt or otherwise unable to fulfill our contractual obligations, our licensors could terminate our rights to critical technology that we rely upon.
Other parties may claim that we infringe their intellectual property or proprietary rights, which could cause us to incur significant expenses or prevent us from selling products.
Our success will depend in part on our ability to operate without infringing the patents and proprietary rights of third parties. The manufacture, use and sale of biologic products have been subject to substantial patent litigation in the biopharmaceutical industry. Such lawsuits often relate to the validity or infringement of patents or other proprietary rights of third parties. Pharmaceutical companies, biotechnology companies, universities, research institutions or other third parties may have filed patent applications or may have been granted patents that cover aspects of our products or our licensors’ products, product candidates or other technologies.
Future or existing patents issued to third parties may contain patent claims that cover our products or their use or manufacture. In particular, the patent landscape in the Covid-19 vaccine space is crowded, and a large number of patent applications have been filed by numerous entities since January 2020, including for the use of certain SARS-CoV-2 antigens and antigenic combinations, including from Moderna, Janssen Pharmaceuticals, Inc., Sementis LTD., VaxBio, Inc., Oxford University, BioNTech, Ichan School of Medicine at Mount Sinai, Diosynvax LTD., The University of Alberta, University of Texas, and Tonix Pharmaceuticals. If a third party were to assert an infringement claim against us in the future with respect to our current products or with respect to products that we may develop or license, such litigation or interference proceedings could force us to:
|
● |
stop or delay selling, manufacturing or using products that incorporate, or are made using the challenged intellectual property; |
|
● |
pay damages; or |
|
● |
enter into licensing or royalty agreements that may not be available on acceptable terms, if at all. |
Any litigation or interference proceedings, regardless of their outcome, may delay the regulatory approval process, be costly and require significant time and attention of our key management and technical personnel.
Any inability to protect our or our licensors’ intellectual property rights in the United States and foreign countries could limit our ability to prevent others from manufacturing or selling our products.
We will rely on trade secrets, unpatented proprietary know-how, continuing technological innovation and, in some cases, patent protection to preserve our competitive position. Our patents and licensed patent rights may be challenged, invalidated, infringed or circumvented, and the rights granted in those patents may not provide proprietary protection or competitive advantages to us. We and our licensors may not be able to develop patentable products with acceptable patent protection. Even if patent claims are allowed, the claims may not issue, or in the event of issuance, may not be sufficient to protect the technology owned by or licensed to us. If patents containing competitive or conflicting claims are issued to third parties, we may be prevented from commercializing the products covered by such patents or may be required to obtain or develop alternate technology. In addition, other parties may duplicate, design around or independently develop similar or alternative technologies.
Some of our patent families and our in-licensed patent families are in an early stage of prosecution and cannot be enforced against third parties practicing the technology claimed in such applications unless, and until, patents are issued from such applications, and then only to the extent the issued claims cover the third-party technology. There can be no assurance that our patent applications will result in patents being issued or that issued patents will afford sufficient protection against competitors with similar technologies. There can be no assurance that the patents if issued will not be infringed, misappropriated, violated, designed around or invalidated by third parties. Even issued patents may later be found invalid or unenforceable or may be modified or revoked in proceedings instituted by third parties before various patent offices or in courts. The degree of future protection for our intellectual property is uncertain. Only limited protection may be available and may not adequately obtain, maintain, protect, and enforce our rights or permit us to gain or keep any competitive advantage. These uncertainties and/or limitations in our ability to properly obtain, maintain, protect, and enforce the intellectual property rights relating to our product candidates could have a material adverse effect on our financial condition and results of operations.
We may not be able to prevent third parties from infringing or using our intellectual property, and the parties from whom we may license intellectual property may not be able to prevent third parties from infringing or using the licensed intellectual property. We generally attempt to control and limit access to, and the distribution of, our product documentation and other proprietary information. Despite efforts to protect this proprietary information, unauthorized parties may obtain and use information that we may regard as proprietary. Other parties may independently develop similar know-how or may even obtain access to these technologies.
The laws of some foreign countries do not protect proprietary information to the same extent as the laws of the United States, and many companies have encountered significant problems and costs in protecting their proprietary information in these foreign countries.
Furthermore, even if our or our licensors’ patent applications are granted, the patent term may be inadequate to protect our competitive position on our product candidates for an adequate amount of time. Patents have a limited lifespan. In the United States, if all maintenance fees are timely paid, the natural expiration of a patent is generally 20 years from its earliest United States non-provisional filing date. Various extensions may be available, but the life of a patent, and the protection it affords, is limited. Even if patents covering our product candidates have been or are obtained, once the patent life has expired, we may be open to competition from competitive products. Given the amount of time required for the development, testing, and regulatory review of product candidates, patents protecting our product candidates might expire before or shortly after such candidates are commercialized. As a result, our patent portfolio may not provide us with sufficient rights to exclude others from commercializing product candidates similar or identical to ours.
Changes in United States patent law could diminish the value of patents in general, thereby impairing our ability to protect our product candidates.
As is the case with other biotechnology companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing biotechnology patents is costly, time-consuming, and inherently uncertain. Changes in either the patent laws or in the interpretations of patent laws in the United States and other countries may diminish the value of our intellectual property and may increase the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of issued patents. We cannot predict the breadth of claims that may be allowed or enforced in our patents or in third-party patents. In addition, Congress or other foreign legislative bodies may pass patent reform legislation that is unfavorable to us.
For example, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, signed into law on September 16, 2011, includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are prosecuted, redefine prior art and provide more efficient and cost-effective avenues for competitors to challenge the validity of patents. These include allowing third-party submission of prior art to the USPTO during patent prosecution and additional procedures to attack the validity of a patent by USPTO administered post-grant proceedings, including post-grant review, inter partes review, and derivation proceedings. Further, because of a lower evidentiary standard in these USPTO post-grant proceedings compared to the evidentiary standard in United States federal courts necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a district court action. Accordingly, a third party may attempt to use the USPTO procedures to invalidate our patent claims that would not have been invalidated if first challenged by the third party as a defendant in a district court action. Thus, the Leahy-Smith Act and its implementation can increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business, financial condition, results of operations and prospects. After March 2013, under the Leahy-Smith Act, the United States transitioned to a first inventor to file system in which, assuming that the other statutory requirements are met, the first inventor to file a patent application is entitled to the patent on an invention regardless of whether a third-party was the first to invent the claimed invention. A third party that files a patent application in the USPTO after March 2013, but before we file an application covering the same invention, could therefore be awarded a patent covering an invention of ours even if we had made the invention before it was made by such third party. This will require us to be cognizant going forward of the time from invention to filing of a patent application. Because patent applications in the United States and most other countries are confidential for a period of time after filing or until issuance, we cannot be certain that we or our licensors were the first to either (i) file any patent application related to our product candidates and other proprietary technologies we may develop or (ii) invent any of the inventions claimed in our or our licensor’s patents or patent applications. Even where we have a valid and enforceable patent, we may not be able to exclude others from practicing the claimed invention where the other party can show that they used the invention in commerce before our filing date or the other party benefits from a compulsory license. However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business, financial condition, results of operations and prospects.
In addition, the patent positions of companies in the development and commercialization of biologics and pharmaceuticals are particularly uncertain. Recent United States Supreme Court and Federal Circuit rulings have narrowed the scope of patent protection available in certain circumstances and weakened the rights of patent owners in certain situations. For example, recent Federal Circuit rulings such as Ariad Pharms., Inc. v. Eli Lilly & Co., 598 F.3d 1336, 1340 (Fed. Cir. 2010) (en banc), Wyeth & Cordis Corp. v. Abbott Labs, 720 F.3d 1380 (Fed. Cir. 2013), Enzo Life Scis., Inc. v. Roche Molecular Sys., 928 F.3d 1340 (Fed. Cir. 2019), and Idenix Pharms. LLC v. Gilead Scis. Inc., 941 F.3d 1149 (Fed. Cir. 2019), and Amgen Inc. v. Sanofi, 598 U.S. 594 (2023) have significantly heightened the standard for securing broad claims to pharmaceutical and biological products. In addition, recent Federal Circuit rulings such as In re Cellect, 81 F.4th 1216 (Fed. Cir. 2023) have expanded the bases for invalidating a patent under the judicially created doctrine of obviousness-type double patenting.
In addition to heightened patentability requirements, recent Supreme Court and Federal Circuit cases relating to biosimilar product approval under the BPCIA, have held that the “patent dance” provisions of the statute, which are intended to resolve any patent infringement issues before the approval of a biosimilar, are discretionary, and a biosimilar applicant can opt out by refusing to provide a copy of its application and manufacturing information to the biologic sponsor (see Sandoz Inc. v. Amgen Inc.,137 S. Ct. 1664 (2017). It may be that we do not learn of a biosimilar application until after FDA publishes its approval (see Immunex v. Samsung Bioepsis, 2:19-cv-117555-CCC-MF (D.N.J. Apr. 30, 2019). In addition to increasing uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by Congress, the United States federal courts, the USPTO, or similar authorities in foreign jurisdictions, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents and patents we might obtain in the future.
The patent protection and patent prosecution for our product candidates is dependent in part on third parties.
We or our licensors may fail to identify patentable aspects of inventions made in the course of development and commercialization activities before it is too late to obtain patent protection on them. Therefore, we may miss potential opportunities to strengthen our patent position. It is possible that defects of form in the preparation or filing of our patents or patent applications may exist, or may arise in the future, for example, with respect to proper priority claims, inventorship, claim scope, or requests for patent term adjustments. If we or our licensors, fail to establish, maintain, or protect such patents and other intellectual property rights, such rights may be reduced or eliminated. If our licensors are not fully cooperative or disagree with us as to the prosecution, maintenance, or enforcement of any patent rights, such patent rights could be compromised. If there are material defects in the form, preparation, prosecution, or enforcement of our patents or patent applications, such patents may be invalid and/or unenforceable, and such applications may never result in valid, enforceable patents. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business.
We rely on third parties to file and prosecute patent applications and maintain patents and otherwise protect the licensed intellectual property under some of our license agreements. We have not had and do not have primary control over these activities for some of our in-licensed patents or patent applications and other intellectual property rights. We cannot be certain that such activities by third parties have been or will be conducted in compliance with applicable laws and regulations or will result in valid and enforceable patents or other intellectual property rights. Pursuant to the terms of the license agreements with some of our licensors, the licensors may have the right to control enforcement of our licensed patents or defense of any claims asserting the invalidity of these patents and even if we are permitted to pursue such enforcement or defense, we will require the cooperation of our licensors. We cannot be certain that our licensors will allocate sufficient resources or prioritize their or our enforcement of such patents or defense of such claims to protect our interests in the licensed patents. Even if we are not a party to these legal actions, an adverse outcome could harm our business because it might prevent us from continuing to license intellectual property that we may need to operate our business. If any of our licensors fail to appropriately prosecute and maintain patent protection for patents covering our product candidates, our ability to develop and commercialize those product candidates may be adversely affected and we may not be able to prevent competitors from making, using, and selling competing products.
In addition, even where we have the right to control patent prosecution of patents and patent applications we have acquired or licensed from third parties, we may still be adversely affected or prejudiced by actions or inactions of our predecessors or licensors and their counsel that took place prior to us assuming control over patent prosecution.
Our technology acquired or licensed from various third parties may be subject to retained rights. Our predecessors or licensors often retain certain rights under their agreements with us, including the right to use the underlying technology for non-commercial academic and research use, to publish general scientific findings from research related to the technology, and to make customary scientific and scholarly disclosures of information relating to the technology. It is difficult to monitor whether our predecessors or licensors limit their use of the technology to these uses, and we could incur substantial expenses to enforce our rights to our licensed technology in the event of misuse.
In addition, the research resulting in certain of our in-licensed patent rights and technology was funded in part by the United States government. As a result, the government may have certain rights, or march-in rights, to such patent rights and technology. When new technologies are developed with government funding, the government generally obtains certain rights in any resulting patents, including a nonexclusive license authorizing the government to use the invention for noncommercial purposes. These rights may permit the government to disclose our confidential information to third parties and to exercise march-in rights to use or allow third parties to use our licensed technology. The United States government also has the right to take title to these inventions if the applicable licensor fails to disclose the invention to the government or fails to file an application to register the intellectual property within specified time limits. The government can exercise its march-in rights if it determines that action is necessary because we fail to achieve practical application of the government-funded technology, because action is necessary to alleviate health or safety needs, to meet requirements of federal regulations, or to give preference to United States industry. In addition, our rights in such inventions may be subject to certain requirements to manufacture products embodying such inventions in the United States. Any exercise by the government of such rights could harm our competitive position, business, financial condition, results of operations, and prospects.
Risks Related to Our Common Stock
The market price of our common stock is highly volatile.
The market price of our common stock has been, and is expected to continue to be, highly volatile. Certain factors, including announcements of new developments by us or other companies, regulatory matters, new or existing medicines or procedures, concerns about our financial position, operating results, litigation, government regulation, developments or disputes relating to agreements, patents or proprietary rights, may have a significant impact on the market price of our stock. In addition, potential dilutive effects of future sales of shares of common stock by us, and subsequent sales of common stock by the holders of our options and warrants could have an adverse effect on the market price of our shares.
In addition, the securities markets from time-to-time experience significant price and volume fluctuations that are unrelated to the operating performance of particular companies. These market fluctuations may also materially and adversely affect the market price of our common stock.
The sale or issuance of additional shares of our common stock or other equity securities could result in additional dilution to our stockholders.
In order to meet our operating cash flow needs, we may plan additional offerings of our equity securities, debt, or convertible debt instruments. The sale of additional equity securities could result in significant additional dilution to our stockholders. The incurrence of indebtedness could result in debt service obligations and operating and financing covenants that would restrict our operations. We cannot assure investors that financing will be available in amounts or on terms acceptable to us, if at all.
We are obligated to issue additional shares of our common stock in connection with our outstanding warrants if the warrant holders choose to exercise them. There are warrants exercisable for approximately 2.0 million shares, 374,000 of which are prefunded ($-0- exercise price) with the remainder having a weighted-average exercise price of $14.58. The exercise of these warrants will cause us to issue additional shares of our common stock and will dilute the percentage ownership of our shareholders.
Certain provisions of our certificate of incorporation which authorize the issuance of shares of preferred stock may make it more difficult for a third party to effect a change in control.
Our certificate of incorporation authorizes our Board of Directors to issue up to 10,000,000 shares of preferred stock. The shares of preferred stock may be issued in one or more series, the terms of which may be determined at the time of issuance by our Board of Directors without further action by the stockholders. These terms may include voting rights, including the right to vote as a series on particular matters, preferences as to dividends and liquidation, conversion rights, redemption rights and sinking fund provisions. The issuance of any newly issued preferred stock could diminish the rights of holders of our common stock, and therefore could reduce the value of our common stock. In addition, specific rights granted to future holders of preferred stock could be used to restrict our ability to merge with, or sell assets to, a third party. The ability of our Board of Directors to issue preferred stock could make it more difficult, delay, discourage, prevent or make it costlier to acquire or effect a change-in-control, which in turn could prevent the stockholders from recognizing a gain in the event that a favorable offer is extended and could materially and negatively affect the market price of our common stock.
We have never paid dividends and have no plans to do so.
Holders of shares of our common stock are entitled to receive such dividends as may be declared by our Board of Directors. To date, we have paid no cash dividends on our shares of common stock and we do not expect to pay cash dividends on our common stock in the foreseeable future. We intend to retain future earnings, if any, to provide funds for operations of our business. Therefore, any potential return investors may have in our common stock will be in the form of appreciation, if any, in the market value of their shares of common stock.
Public company compliance may make it more difficult for us to attract and retain officers and directors.
The Sarbanes-Oxley Act, the Dodd-Frank Act, the JOBS Act, the FAST Act, and rules subsequently implemented by the SEC have required changes in corporate governance practices of public companies. As a public company, we expect these rules and regulations, and amendments to them, to contribute to our compliance costs and to make certain activities more time-consuming and costly. As a public company, we also expect that these rules and regulations may make it difficult and expensive for us to obtain director and officer liability insurance and we may be required to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. As a result, it may be difficult for us to attract and retain qualified persons to serve on our board of directors or as executive officers.
Our Certificate of Incorporation and Bylaws may be amended by the affirmative vote of a majority of our stockholders.
Under the Delaware General Corporation Law, a corporation’s certificate of incorporation may be amended by the affirmative vote of the holders of a majority of the outstanding shares entitled to vote, and a majority of the outstanding shares of each class entitled to vote as a class, unless the articles require the vote of a larger percentage of shares. Our Certificate of Incorporation, as amended, does not require the vote of a larger percentage of shares. As permitted under the Delaware General Corporation Law, our Bylaws give our board of directors the power to adopt, amend, or repeal our Bylaws. Our stockholders entitled to vote have concurrent power to adopt, amend, or repeal our Bylaws.
Broker-dealers may be discouraged from effecting transactions in shares of our common stock if we are considered to be a penny stock and thus subject to the penny stock rules.
The SEC has adopted a number of rules to regulate “penny stocks” that restrict transactions involving stock which is deemed to be penny stock. Such rules include Rules 3a51-1, 15g-1, 15g-2, 15g-3, 15g-4, 15g-5, 15g-6, 15g-7, and 15g-9 under the Exchange Act. These rules may have the effect of reducing the liquidity of penny stocks. “Penny stocks” generally are equity securities with a price of less than $5.00 per share (other than securities registered on certain national securities exchanges or quoted on Nasdaq if current price and volume information with respect to transactions in such securities is provided by the exchange or system). Our securities have in the past constituted, and may again in the future, if we are delisted from Nasdaq, constitute, “penny stock” within the meaning of the rules. The additional sales practice and disclosure requirements imposed upon U.S. broker-dealers may discourage broker-dealers from effecting transactions in shares of our common stock, which could severely limit the market liquidity of such shares and impede their sale in the secondary market.
A U.S. broker-dealer selling penny stock to anyone other than an established customer or “accredited investor” (generally, an individual with net worth in excess of $1,000,000 (exclusive of personal residence) or an annual income exceeding $200,000, or $300,000 together with his or her spouse) must make a special suitability determination for the purchaser and must receive the purchaser’s written consent to the transaction prior to sale, unless the broker-dealer or the transaction is otherwise exempt. In addition, the “penny stock” regulations require the U.S. broker-dealer to deliver, prior to any transaction involving a “penny stock”, a disclosure schedule prepared in accordance with SEC standards relating to the “penny stock” market, unless the broker-dealer or the transaction is otherwise exempt. A U.S. broker-dealer is also required to disclose commissions payable to the U.S. broker-dealer and the registered representative and current quotations for the securities. Finally, a U.S. broker-dealer is required to submit monthly statements disclosing recent price information with respect to the “penny stock” held in a customer’s account and information with respect to the limited market in “penny stocks”.
Stockholders should be aware that, according to the SEC, the market for “penny stocks” has suffered in recent years from patterns of fraud and abuse. Such patterns include (i) control of the market for the security by one or a few broker-dealers that are often related to the promoter or issuer; (ii) manipulation of prices through prearranged matching of purchases and sales and false and misleading press releases; (iii) “boiler room” practices involving high-pressure sales tactics and unrealistic price projections by inexperienced sales persons; (iv) excessive and undisclosed bid-ask differentials and markups by selling broker-dealers; and (v) the wholesale dumping of the same securities by promoters and broker-dealers after prices have been manipulated to a desired level, resulting in investor losses. Our management is aware of the abuses that have occurred historically in the penny stock market. Although we do not expect to be in a position to dictate the behavior of the market or of broker-dealers who participate in the market, management will strive within the confines of practical limitations to prevent the described patterns from being established with respect to our securities.
|
ITEM 1B. |
UNRESOLVED STAFF COMMENTS |
None
|
ITEM 1C. |
CYBERSECURITY |
Risk oversight and management is a key role for the Board and its committees. The Board is responsible for identifying and understanding the Company’s principal risks and ensuring that appropriate systems are implemented to monitor, manage and mitigate those risks. The committees of the Board have oversight over risks within their respective mandates.
Oversight of cybersecurity is integrated into the responsibilities of the Board. The Nominating and Governance Committee (the “NGC”) has been assigned oversight of cybersecurity matters, particularly as they relate to financial risk and controls, integrity of financial data and public disclosures, and security of overall digital data.
Management is responsible for the implementation of risk management strategies and for the operational oversight of company-wide cybersecurity strategy, policy, and standards to assess and prepare us to address cybersecurity risks. We have evolving processes for assessing, identifying and managing cybersecurity risks, which are built into our information technology function and are designed to help protect our information assets and operations from cyber threats, protect employee and corporate information from unauthorized access or attack, as well as secure our networks and systems. Such processes include physical, procedural and technical safeguards, response plans, and routine review of our policies and procedures to identify risks and refine our practices. We engage independent third parties to assess and implement our cybersecurity procedures and enhance our oversight.
The NGC receives periodic updates from management regarding cybersecurity matters and is notified between such updates regarding any significant new cybersecurity threats or incidents. We do not believe that there are currently any known risks from cybersecurity threats that are reasonably likely to materially affect us or our business strategy, results of operations or financial condition.
|
ITEM 2. |
PROPERTIES |
Our principal executive offices are located in Smyrna, Georgia, where we lease approximately 8,400 square feet of office and laboratory space. Our lease for the premises is currently scheduled to terminate on December 31, 2025. We do not currently own any real property. We believe that our current facilities are adequate to meet our immediate needs and that if we require additional space, we will be able to obtain additional facilities on commercially reasonable terms.
|
ITEM 3. |
LEGAL PROCEEDINGS |
We are not currently a party to any material legal proceedings. We may from time to time become involved in various legal proceedings such as those arising in the ordinary course of business.
|
ITEM 4. |
MINE SAFETY DISCLOSURES |
Not applicable.
PART II
|
ITEM 5. |
MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market Information
Our common stock is currently traded on The Nasdaq Capital Market under the symbol “GOVX”.
Holders
On February 29, 2024, there were 14 holders of record of our common stock. The majority of our shares of common stock are held by brokers and other institutions on behalf of stockholders, and we are unable to estimate the total number of stockholders represented by these record holders.
Dividends
We have never declared or paid cash dividends on our common stock and do not anticipate paying any dividends on our common stock in the foreseeable future. We expect to retain future earnings, if any, for reinvestment in our business. We will not be permitted to pay dividends on our common stock unless all dividends on any preferred stock that may be issued have been paid in full. We currently do not have any plans to issue additional preferred stock. Any credit agreements which we may enter into may also restrict our ability to pay dividends. The payment of dividends in the future will be subject to the discretion of our board of directors and will depend, among other things, on our financial condition, results of operations, cash requirements, future prospects and any other factors our board of directors deems relevant.
Recent Sales of Unregistered Securities
There were no sales of unregistered securities during the period covered by this report that have not previously been reported on a Current Report on Form 8-K or a Quarterly Report Form 10-Q.
Issuer Purchases of Equity Securities
We did not repurchase any of our equity securities during the fourth quarter of 2023.
|
ITEM 6. |
RESERVED |
Not Applicable.
|
ITEM 7. |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
The following discussion and analysis of our financial condition and results of operations should be read together with “Selected Financial Data” and our consolidated financial statements and the related notes beginning on page F-1. This discussion contains forward-looking statements that involve risks and uncertainties because they are based on current expectations and relate to future events and our future financial performance. Our actual results may differ materially from those anticipated in these forward-looking statements because of many important factors, including those set forth under “Risk Factors” and elsewhere in this Annual Report.
Overview and Recent Developments
GeoVax is a clinical-stage biotechnology company developing immunotherapies and vaccines against infectious diseases and solid tumor cancers using novel vector vaccine platforms. GeoVax’s product pipeline includes ongoing human clinical trials for a next-generation Covid-19 vaccine and a gene-directed therapy against advanced head and neck cancer. Additional research and development programs include preventive vaccines against Mpox and smallpox, hemorrhagic fever viruses (Ebola Zaire, Ebola Sudan and Marburg), Zika virus and malaria, as well as immunotherapies for multiple solid tumors.
Our programs are in various stages of development, the most significant of which are summarized below along with recent developments:
GEO-CM04S1 – Immunocompromised/Cell Transplant Trial
|
● |
GEO-CM04S1 is currently undergoing a Phase 2 clinical trial (ClinicalTrials.gov Identifier: NCT04977024), evaluating its safety and efficacy, compared to either the Pfizer/BioNTech or Moderna mRNA-based vaccine, as a preventive Covid-19 vaccine in high-risk immunocompromised patients (e.g. patients who have previously received either an allogeneic hematopoietic cell transplant, an autologous hematopoietic cell transplant or chimeric antigen receptor (CAR) T cell therapy). |
|
● |
In September 2023, the journal, Vaccines, published data from the open-label safety portion of the trial indicating that GEO-CM04S1 is highly immunogenic, inducing both antibody responses, including neutralizing antibodies, and T cell responses. |
|
● |
In September 2023, preclinical vaccine efficacy data for GEO-CM02 were presented during the Keystone Symposia on Molecular and Cellular Biology, Vaccinology During and After Covid-19, demonstrating that our multi-antigen SARS-CoV-2 vaccine, GEO-CM02, induced efficacious immune responses against the original Wuhan strain and BA.1 Omicron variant with a single dose. The data generated in the GEO-CM02 studies validate our hypothesis that vaccines such as GEO-CM04S1, which are designed to induce both antibodies and T-cells to multiple viral structural proteins, can address the issue of viral variation and escape from the immune system. |
|
● |
In October 2023, we announced commencement of the planned site expansion for this trial to accelerate patient enrollment. In addition to study enrollments completed at the City of Hope Medical Center (Duarte, California), the trial is now open to eligible patients at Wake Forest Baptist Medical Center (Winston Salem, North Carolina), the University of Massachusetts Medical Center (Worcester, Massachusetts), and the Fred Hutchinson Cancer Center (Seattle, Washington). |
GEO-CM04S1 – Healthy Booster Trial
|
● |
GEO-CM04S1 is undergoing the Phase 2 portion of a Phase 1/2 trial (ClinicalTrials.gov Identifier: NCT04639466), evaluating its use as a universal Covid-19 booster vaccine to current FDA-approved two-shot mRNA vaccines from Pfizer/BioNTech and Moderna. |
|
● |
In September 2023, we announced the completion of patient enrollment for this trial. |
|
● |
In February 2024, we announced positive initial safety and immune responses findings at one-month following vaccine administration. |
GEO-CM04S1 – Immunocompromised/CLL Trial
|
● |
In July 2023, an investigator-initiated Phase 2 clinical trial (ClinicalTrials.gov Identifier: NCT05672355) of GEO-CM04S1 began, evaluating its use as a Covid-19 booster vaccine in patients with chronic lymphocytic leukemia (CLL), compared to the Pfizer/BioNTech mRNA-based vaccine. |
Gedeptin® – Advanced Head and Neck Cancer Trial
|
● |
Gedeptin® is currently undergoing a Phase 1/2 clinical trial (ClinicalTrials.gov Identifier: NCT03754933) for treatment of patients with advanced head and neck squamous cell carcinoma (HNSCC). This trial is being funded in part by the U.S. Food & Drug Administration (FDA) pursuant to its Orphan Products Clinical Trials Grants Program. The trial is designed to inform the design of a larger patient trial that also may involve patients with other anatomically accessible oral and pharyngeal cancers, including cancers of the lip, tongue, gum, floor of mouth, salivary gland and other oral cavities. |
|
● |
In January 2024, we announced closure of patient enrollment for this trial. |
|
● |
In July 2023, interim data were presented at the American Association for Cancer Research (AACR) and the American Head and Neck Society (AHNS) joint Head and Neck Cancer Conference, indicating that administration of Gedeptin® is safe and feasible, with observation of tumor growth impairment in a majority of the patients. |
Advanced Vaccine Manufacturing Process Development
|
● |
In September 2023, GeoVax and ProBioGen AG announced the signing of a commercial license agreement for ProBioGen’s AGE1.CR.pIX® suspension cell line. The agreement enhances our manufacturing capabilities for our entire Modified Vaccinia Ankara (MVA)-based vaccine portfolio. This follows the May 2023 execution of a Master Services Agreement with Advanced Bioscience Laboratories, Inc. (ABL) to support current Good Manufacturing Practices (cGMP) production of our vaccine candidates through late-stage development toward eventual commercialization. These agreements move the Company toward fully implementing a continuous cell line manufacturing system that will provide lower-cost, scalable versatility for our entire MVA-based vaccine portfolio. |
Intellectual Property Development
|
● |
In July 2023, the U.S. Patent and Trademark Office issued Patent No. 11,701,418 B2 to GeoVax, pursuant to the Company’s patent application No. 15/543,139 titled “Replication-Deficient Modified Vaccinia Ankara (MVA) and Matrix Protein (VP40), covering GeoVax’s vector platform for expressing ebolavirus antigens in virus-like particles (VLPs) utilizing an MVA viral vector. The claims encompass multiple ebolavirus strains, including Sudan ebolavirus, Zaire ebolavirus, Taï Forest ebolavirus, and Reston ebolavirus. |
|
● |
In August 2023, the U.S. Patent and Trademark Office issued a Notice of Allowance to GeoVax for Patent Application No. 17/726,254 titled “Compositions and Methods for Generating an Immune Response to Treat or Prevent Malaria”. The allowed claims cover compositions comprising GeoVax’s modified vaccinia Ankara (MVA) vector expressing Plasmodium antigens and methods of inducing an immune response to malaria utilizing the compositions. The compositions and methods covered in the allowed claims are useful both prophylactically and therapeutically and may be used to prevent and/or treat malaria. The patent (No. 11,857,611) was issued in January 2024. |
|
● |
In October 2023, the U.S. Patent and Trademark Office issued a Notice of Allowance to GeoVax for Patent Application No. 17/584,231 titled “Replication Deficient Modified Vaccina Ankara (MVA) Expressing Marburg Virus Glycoprotein (GP) and Matrix Protein (VP40).” The allowed claims generally cover GeoVax’s vector platform for expressing Marburg virus antigens in virus-like particles (VLPs) utilizing an MVA viral vector. The patent (No. 11,896,657) was issued in February 2024. |
|
● |
In October 2023, the U.S Patent and Trademark Office issued a Notice of Allowance to GeoVax for Patent Application No. 17/409,574 titled “Multivalent HIV Vaccine Boost Compositions and Methods of Use.” The allowed claims generally cover a priming vaccination with a DNA vector encoding multiple HIV antigens in virus-like particles (VLPs), followed by a boost vaccination with GeoVax’s vector platform for expressing HIV-1 antigens in VLPs utilizing an MVA viral vector. The patent (No. 11,897,919) was issued in February 2024. |
|
● |
In February 2024, the Japanese Patent Office issued a Decision of Grant notifying GeoVax of the allowance of the Company’s Patent Application No. 2022-153352 titled “Compositions and Methods for Generating an Immune Response to a Tumor Associated Antigen.” The allowed claims are directed to recombinant MVA viral vectors comprising specific MUC-1 nucleic sequences used in GeoVax’s MUC-1 tumor-associated antigen immunotherapy program. Pharmaceutical compositions for inducing immune responses, preventing or reducing neoplasm growth, or treating cancer are also covered by the granted claims. This represents an extension of the GeoVax MVA-VLP platform that was originally developed for vaccines targeting infectious diseases. |
General Corporate
Effective January 31, 2024, following approval by our stockholders at a special meeting held on January 16, 2024, we effected a reverse stock split of our common stock at a ratio of 1-for-15. The purpose of the reverse split was to regain compliance with the $1.00 minimum bid price required for continued listing on The Nasdaq Capital Market under Nasdaq Listing Rule 5550(a)(2). On the effective date, every fifteen issued and outstanding shares of the Company's Common Stock was converted automatically into one share of the Company's Common Stock without any change in the par value per share. The total number of issued and outstanding shares of Common Stock was reduced proportionately from 29,757,823 shares to approximately 2,039,240 shares. On the effective date, the Company’s publicly-traded warrants were adjusted to require fifteen warrants to be exercised to receive one share of common stock at a price of $75 per share.
Our corporate strategy is to advance, protect and exploit our differentiated vaccine/immunotherapy technologies leading to the successful development of preventive and therapeutic vaccines and immunotherapies against infectious diseases and various cancers. Our goal is to advance products through to human clinical testing, and to seek partnership or licensing arrangements for achieving regulatory approval and commercialization. We also leverage third party resources through collaborations and partnerships for preclinical and clinical testing with multiple government, academic and corporate entities.
We have not generated any revenues from the sale of the products we are developing, and we do not expect to generate any such revenues for at least the next several years. Our product candidates will require significant additional research and development efforts, including extensive preclinical and clinical testing. All product candidates that we advance to clinical testing will require regulatory approval prior to commercial use and will require significant costs for commercialization. We may not be successful in our research and development efforts, and we may never generate sufficient product revenue to be profitable.
Financial Overview
Revenues
We have not generated any revenues from product sales to date. Our product candidates will require significant additional research and development efforts, including extensive preclinical and clinical testing. All product candidates that we advance to clinical testing will require regulatory approval prior to commercial use and will require significant costs for commercialization. Our grant revenues relate to grants and contracts from agencies of the U.S. government in support of our vaccine development activities. We record revenue associated with these grants as the related costs and expenses are incurred.
Research and development expenses
Since our inception, we have focused and we continue to focus significant resources on our research and development activities, including developing our vector platform and analytical testing methods, conducting preclinical studies, developing manufacturing processes, and conducting clinical trials. Research and development costs are expensed as incurred and consist primarily of the following:
|
• |
personnel costs in our research, development and regulatory functions, which include salaries, benefits and stock-based compensation; |
|
• |
expenses incurred under agreements with contract research organizations (“CROs”), that conduct clinical trials on our behalf; |
|
• |
expenses incurred under agreements with contract manufacturing organizations (“CMOs”), that manufacture product used in the clinical trials; |
|
• |
expenses incurred in procuring materials and for analytical and release testing services required to produce vaccine candidates used in clinical trials; |
|
• |
process development expenses incurred internally and externally to improve the efficiency and yield of the bulk vaccine; |
|
• |
laboratory supplies, vendor expenses and other third-party contract expenses related to preclinical research activities; |
|
• |
technology license fees; |
|
• |
consultant expenses for services supporting our clinical, regulatory and manufacturing activities; and |
|
• |
facilities, depreciation and other general overhead expenses. |
We expect our research and development expenditures to increase during 2024 and beyond as we advance our existing and future product candidates into and through clinical trials and pursue regulatory approval, especially with regard to the Gedeptin and GEO-CM04S1 clinical programs. We do not provide forward-looking estimates of costs and time to complete our research programs due to the many uncertainties associated with biotechnology research and development. Due to these uncertainties, our future expenditures are likely to be highly volatile in future periods depending on the outcomes of the trials and studies. As we obtain data from preclinical studies and clinical trials, we may elect to discontinue or delay certain development programs to focus our resources on more promising product candidates. Completion of preclinical studies and human clinical trials may take several years or more, but the length of time can vary substantially depending upon several factors. The duration and the cost of future clinical trials may vary significantly over the life of the project because of differences arising during development of the human clinical trial protocols, including the length of time required to enroll suitable patient subjects, the number of patients that ultimately participate in the clinical trial, the duration of patient follow-up, and the number of clinical sites included in the clinical trials.
General and administrative expenses
Our general and administrative expenses consist primarily of personnel costs in our executive, finance and investor relations, business development and administrative functions, including stock-based compensation. Other general and administrative expenses include consulting fees, professional service fees for accounting and legal services, lease expenses related to our offices, insurance premiums, intellectual property costs incurred in connection with filing and prosecuting patent applications, depreciation and other costs. We expect our general and administrative expenses to continue to increase in the future as we support expanded research and development activities, prepare for potential commercialization of our current and future product candidates, maintain compliance with requirements of Nasdaq and the Securities and Exchange Commission, and other general corporate activities.
Critical Accounting Policies and Estimates
This discussion and analysis of our financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States. The preparation of these financial statements requires management to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses and related disclosure of contingent assets and liabilities. On an ongoing basis, management evaluates its estimates and adjusts them as necessary. We base our estimates on historical experience and on various other assumptions that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ materially from these estimates under different assumptions or conditions.
Our significant accounting policies are summarized in Note 2 to our consolidated financial statements for the year ended December 31, 2023, which are included in this Form 10-K. We believe the following critical accounting policies affect our more significant judgments and estimates used in the preparation of our consolidated financial statements:
Revenue Recognition
We recognize revenue in accordance with FASB Accounting Standards Codification Topic 606, Revenue from Contracts with Customers (ASC 606). The standard is principle-based and provides a five-step model to determine when and how revenue is recognized. The core principle is that an entity should recognize revenue when it transfers promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services.
We receive payments from government entities under non-refundable grants in support of our vaccine development programs. We record revenue associated with these grants when the reimbursable costs are incurred and we have complied with all conditions necessary to receive the grant funds. From time to time, we may enter into collaborative research and development agreements for specific vaccine development approaches and/or disease indications whereby we receive third-party funding for preclinical research under certain of these arrangements. Each agreement is evaluated in accordance with the process defined by ASC 606 and revenue is recognized accordingly.
Stock-Based Compensation
We account for stock-based transactions in which the Company receives services from employees, directors or others in exchange for equity instruments based on the fair value of the award at the grant date. Stock-based compensation cost for awards of common stock is estimated based on the price of the underlying common stock on the date of issuance. Stock-based compensation cost for stock options or warrants is estimated at the grant date based on each instrument’s fair value as calculated by using the Black-Scholes option pricing model. We recognize stock-based compensation cost as expense ratably on a straight-line basis over the requisite service period for the award. See Note 6 to our financial statements for additional stock-based compensation information.
Research and Development Expense
Research and development costs are charged to expense as incurred and consist of costs incurred in the discovery, development, testing and manufacturing of our product candidates. These expenses consist primarily of (i) salaries, benefits, and stock-based compensation for personnel, (ii) laboratory supplies and facility-related expenses to conduct development, (iii) fees paid to third-party service providers to perform, monitor and accumulate data related to our preclinical studies and clinical trials, (iv) costs related to sponsored research agreements, (v) costs to procure and manufacture materials used in clinical trials, and (vi) license fees and other expenses associated with technology license agreements.
The Company accrues for estimated costs of research and development activities conducted by third-party service providers, which may include the conduct of preclinical studies and clinical trials, and contract manufacturing activities. When evaluating the adequacy of the accrued liabilities, the Company analyzes progress of the studies or trials, including clinical trial participant enrollment, completion of events, invoices received and other events. Advance payments for research and development activities are deferred and included in prepaid expenses and other assets. The deferred amounts are expensed as the related goods are delivered or the services are performed.
Off-Balance Sheet Arrangements
We have no off-balance sheet arrangements that are likely or reasonably likely to have a material effect on our financial condition or results of operations, other than the operating lease for our office and laboratory space.
Results of Operations
The following table summarizes our results of operations for the years ended December 31, 2023 and 2022:
|
2023 |
2022 |
Change |
||||||||||
|
Grant and collaboration revenue |
$ | - | $ | 81,526 | $ | (81,526 | ) | |||||
|
Operating expenses: |
||||||||||||
|
Research and development |
20,720,766 | 9,123,479 | 11,597,287 | |||||||||
|
General and administrative |
6,022,173 | 4,986,611 | 1,035,562 | |||||||||
|
Total operating expenses |
26,742,939 | 14,110,090 | 12,632,849 | |||||||||
|
Loss from operations |
(26,742,939 | ) | (14,028,564 | ) | (12,714,375 | ) | ||||||
|
Total other income (expense) |
776,177 | 7,439 | 768,738 | |||||||||
|
Net loss |
$ | (25,966,762 | ) | $ | (14,021,125 | ) | $ | (11,945,637 | ) | |||
Grant Revenues
There were no grant revenues during 2023. As of December 31, 2022, all grant funds available for use directly by GeoVax were expended.
Research and Development Expenses
Our research and development expenses were $20,720,766 for the year ended December 31, 2023, as compared to $9,123,479 for 2022, representing an increase of $11,597,287 (127%). The increase during 2023 relates primarily to costs of conducting clinical trials for GEO-CM04S1 and Gedeptin, costs of manufacturing materials for use in our clinical trials, technology license fees, personnel costs, costs of preclinical research activities and higher travel costs. Research and development expense for 2023 and 2022 includes stock-based compensation expense of $291,094 and $225,031, respectively, associated with employee stock options.
General and Administrative Expenses
Our general and administrative expenses were $6,022,173 for the year ended December 31, 2023, as compared to $4,983,611 for 2022, representing an increase of $1,035,562 (21%). The increase during 2023 relates primarily to higher personnel costs, investor relations consulting costs, legal fees, patent costs and travel expenses. General and administrative expense for 2023 and 2022 includes stock-based compensation expense of $783,863 and $677,043, respectively, associated with employee and consultant stock options and stock awards.
Other Income
Interest income was $776,177 and $7,439 for the years ended December 31, 2023 and 2022, respectively. The variances between years are primarily attributable to the cash available for investment and to interest rate fluctuations.
Liquidity and Capital Resources
The following tables summarize our liquidity and capital resources as of December 31, 2023 and 2022, and our cash flows for the years then ended:
|
As of December 31, |
||||||||
|
Liquidity and Capital Resources |
2023 |
2022 |
||||||
|
Cash and cash equivalents |
$ | 6,452,589 | $ | 27,612,732 | ||||
|
Working capital |
4,365,861 | 24,190,836 | ||||||
|
Year Ended December 31, |
||||||||
|
Cash Flow Data |
2023 |
2022 |
||||||
|
Net cash provided by (used in): |
||||||||
|
Operating activities |
$ | (25,173,639 | ) | $ | (19,030,208 | ) | ||
|
Investing activities |
(48,946 | ) | (134,258 | ) | ||||
|
Financing activities |
4,062,442 | 35,353,328 | ||||||
|
Net increase (decrease) in cash and cash equivalents |
$ | (21,160,143 | ) | $ | 16,188,862 | |||
Operating Activities – Net cash used in operating activities of $25,173,639 for 2023 was primarily due to our net loss of $25,966,762, offset by non-cash items such as depreciation expense and stock-based compensation expense, and by changes in our working capital accounts. Net cash used in operating activities of $19,030,208 for 2022 was primarily due to our net loss of $14,021,125, offset by non-cash items such as depreciation expense and stock-based compensation expense, and by changes in our working capital accounts.
Investing Activities – Net cash used in investing activities was $48,946 and $134,258 for 2023 and 2022, respectively, and relates to purchases of property and equipment.
Financing Activities – Net cash provided by financing activities was $4,062,442 for 2023, consisting of net proceeds from the exercise of warrants. Net cash provided by financing activities was $35,353,328 for 2022, consisting of (i) aggregate net proceeds of $27,727,194 from offerings of our common stock and (ii) $7,626,134 of net proceeds from the exercise of warrants.
Funding Requirements and Sources of Capital
To date, we have not generated any product revenue. We do not know when, or if, we will generate any product revenue and we do not expect to generate significant product revenue unless and until we obtain regulatory approval and commercialize one of our current or future product candidates. We anticipate that we will continue to generate losses for the foreseeable future, and we expect the losses to increase as we continue the development of, and seek regulatory approvals for, our product candidates, and begin to commercialize any approved products. We are subject to all of the risks incident to the development of new products, and may encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may harm our business. We anticipate that we will need substantial additional funding in connection with our continuing operations. We have funded our operations to date primarily from sales of our equity securities and from government grants and clinical trial assistance.
As of the date of this Annual Report, we expect our existing cash and cash equivalents will be sufficient to fund our operations through the second quarter of 2024. This projection takes into consideration contractual commitments we have made, and expect to make, in the normal course of operating our business, which include (i) obligations to our employees, (ii) our lease obligations, (iii) payments due under license agreements for various technologies and patent rights associated with our product development activities, (iv) arrangements with contract research organizations (“CROs”), contract manufacturing organizations (“CMOs”), and other third-party vendors for clinical trials services and production of materials for use in our clinical trials, and (v) other various firm purchase commitments and contractual obligations related to production and testing of our product candidates and the general operation of our business.
Our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement that involves risks and uncertainties and is based on assumptions that may prove to be wrong; actual results could vary materially.
We have based our projections of operating capital requirements on assumptions that may prove to be incorrect, and we may use our available capital resources sooner than we expect. Our future capital requirements will depend on many factors, which include but are not limited to:
|
● |
the timing and costs of our ongoing and planned clinical trials; |
|
● |
the timing and costs of manufacturing material for use in clinical trials; |
|
● |
the number and scope of our research programs and the speed at which they are advanced; |
|
● |
the progress and success of our preclinical and clinical development activities; |
|
● |
the costs involved in prosecuting and enforcing patent claims and other intellectual property rights; |
|
● |
the costs to attract and retain skilled personnel; |
|
● |
the costs to maintain and expand our infrastructure to support our operations, our product development, and planned future commercialization efforts; |
|
● |
the terms and timing of establishing and maintaining collaborations, licenses and other similar arrangements; |
|
● |
the costs associated with any products or technologies that we may in-license or acquire; and |
|
● |
the costs and timing of regulatory approvals. |
We will need to continue to raise additional capital to support our future operating activities, including progression of our development programs, preparation for commercialization, and other operating costs. Financing strategies we may pursue include, but are not limited to, the public or private sale of equity, debt financings or funds from other capital sources, such as government funding, collaborations, strategic alliances or licensing arrangements with third parties. There can be no assurances additional capital will be available to secure additional financing, or if available, that it will be sufficient to meet our needs on favorable terms. If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we may have to significantly delay, scale back or discontinue the development of one or more of our product candidates.
|
ITEM 7A. |
QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
Our exposure to market risk is limited primarily to interest income sensitivity, which is affected by changes in the general level of United States interest rates, particularly because a significant portion of our investments are in institutional money market funds. The primary objective of our investment activities is to preserve principal while at the same time maximizing the income received without significantly increasing risk. Due to the nature of our short-term investments, we believe that we are not subject to any material market risk exposure. We do not have any derivative financial instruments or foreign currency instruments.
|
ITEM 8. |
FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
Our consolidated financial statements and supplemental schedule and notes thereto as of December 31, 2023 and 2022 and for the two-year period ended December 31, 2023 together with the independent registered public accounting firm’s report thereon, are set forth on pages F-1 to F-16 of this Annual Report.
|
ITEM 9. |
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
There were no disagreements with our accountants on matters of accounting or financial disclosure, or other reportable events requiring disclosure under this Item 9.
|
ITEM 9A. |
CONTROLS AND PROCEDURES |
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures designed to ensure that financial information required to be disclosed in our reports filed or submitted under the Securities Exchange Act of 1934, as amended (the Exchange Act), is recorded, processed, summarized, and reported within the required time periods, and that such information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow for timely decisions regarding disclosure.
We carried out an evaluation, under the supervision and with the participation of our management, of the effectiveness of the design and operation of our disclosure controls and procedures as of the end of the period covered by this Form 10-K. Based on that evaluation, our Chief Executive Officer and Chief Financial Officer concluded that, as of December 31, 2023, our disclosure controls and procedures were effective to provide reasonable assurance that information required to be disclosed by us in reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC rules and forms.
Management’s Report on Internal Control Over Financial Reporting
Management is responsible for establishing and maintaining adequate internal control over financial reporting, as defined in Rule 13a-15(f) of the Exchange Act. Management has assessed the effectiveness of our internal control over financial reporting as of December 31, 2022, based on criteria established in Internal Control - Integrated Framework (2013), issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). As a result of this assessment, management concluded that, as of December 31, 2023, our internal control over financial reporting was effective in providing reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting that occurred during our most recent fiscal quarter that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Limitations on Controls
Management does not expect that our disclosure controls and procedures or our internal control over financial reporting will prevent or detect all error and fraud. Any control system, no matter how well designed and operated, is based upon certain assumptions and can provide only reasonable, not absolute, assurance that its objectives will be met. Further, no evaluation of controls can provide absolute assurance that misstatements due to error or fraud will not occur or that all control issues and instances of fraud, if any, within the Company have been detected.
|
ITEM 9B. |
OTHER INFORMATION |
|
ITEM 9C. |
DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS |
Not Applicable.
PART III
|
ITEM 10. |
DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
Information required by this Item is included in our definitive proxy statement for our 2024 annual meeting of stockholders to be filed with the SEC under the captions “Directors and Executive Officers” and “Corporate Governance” and is incorporated herein by this reference.
Code of Business Conduct and Ethics
Our Board of Directors has adopted a written Code of Business Conduct and Ethics, a copy of which is available on our website at www.geovax.com. The Company will provide a copy of the Code of Ethics upon request to any person without charge. Such requests may be transmitted by regular mail in the care of the Corporate Secretary. We require all officers, directors and employees to adhere to this code in addressing the legal and ethical issues encountered in conducting their work. The code requires that employees avoid conflicts of interest, comply with all laws and other legal requirements, conduct business in an honest and ethical manner, and otherwise act with integrity and in our best interest. Employees are required to report any conduct that they believe in good faith to be an actual or apparent violation of the code. The Sarbanes-Oxley Act of 2002 requires certain companies to have procedures to receive, retain and treat complaints received regarding accounting, internal accounting controls or auditing matters and to allow for the confidential and anonymous submission by employees of concerns regarding questionable accounting or auditing matters. We have such procedures in place.
The Company will post on its website, www.geovax.com, or will disclose on a Form 8-K filed with the SEC, any amendments to, or waivers from, a provision of the Code of Ethics that applies to the Chief Executive Officer or the Chief Financial Officer, or persons performing similar functions, and that relate to (i) honest and ethical conduct, including the ethical handling of actual or apparent conflicts of interest between personal and professional relationships; (ii) full, fair, accurate, timely, and understandable disclosure in reports and documents that the Company files with, or submits to, the SEC and in other public communications made by the Company; (iii) compliance with applicable governmental laws, rules and regulations; (iv) the prompt internal reporting of violations of the Code of Ethics to an appropriate person or persons identified in the code; or (v) accountability for adherence to the Code of Ethics. Any waiver granted to an executive officer or a director may only be granted by the Board and will be disclosed, along with the reasons therefor, on a Form 8-K filed with the SEC. No such waivers were granted in 2023.
|
ITEM 11. |
EXECUTIVE COMPENSATION |
The information required by this Item is included in our definitive proxy statement for our 2024 annual meeting of stockholders to be filed with the SEC under the captions “Corporate Governance” and “Executive Compensation” and is incorporated herein by this reference.
|
ITEM 12. |
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
The information required by this Item is included in our definitive proxy statement for our 2024 annual meeting of stockholders to be filed with the SEC under the captions “Security Ownership of Principal Stockholders, Directors and Executive Officers” and is incorporated herein by this reference.
Securities Authorized for Issuance Under Equity Compensation Plans
The following table sets forth certain information as of December 31, 2023 with respect to compensation plans under which our equity securities are authorized for issuance.
|
Plan Category |
Number of securities to be issued upon exercise of outstanding options, warrants and rights |
Weighted-average exercise price of outstanding options, warrants and rights |
Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) |
|
Equity compensation plans approved by stockholders |
134,609 |
$28.41 |
200,000 |
|
Equity compensation plans not approved by stockholders (1) |
18,203 |
$75.00 |
-0- |
(1) Represents stock purchase warrants issued in 2020 as part of deferred salary conversions by members of management and the board of directors.
A description of our equity compensation plans can be found in footnote 6 to our 2023 consolidated financial statements, which are filed as exhibits this document.
|
ITEM 13. |
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
The information required by this Item is included in our definitive proxy statement for our 2024 annual meeting of stockholders to be filed with the SEC under the captions “Corporate Governance” and “Certain Relationships and Related Party Transactions” and is incorporated herein by this reference.
|
ITEM 14. |
PRINCIPAL ACCOUNTING FEES AND SERVICES |
The information required by this Item is included in our definitive proxy statement for our 2024 annual meeting of stockholders to be filed with the SEC under the caption “Ratification of Appointment of the Independent Registered Public Accounting Firm” and is incorporated herein by this reference.
PART IV
|
ITEM 15. |
EXHIBITS AND FINANCIAL STATEMENT SCHEDULES |
|
(a) |
Documents filed as part of this report: |
| (1) | Financial Statements | Page |
| Report of Independent Registered Public Accounting Firm | F-2 | |
| Consolidated Balance Sheets as of December 31, 2023 and 2022 | F-3 | |
| Consolidated Statements of Operations for the years ended December 31, 2023 and 2022 | F-4 | |
| Consolidated Statements of Stockholders’ Equity for the years ended December 31, 2023 and 2022 | F-5 | |
| Consolidated Statements of Cash Flows for the years ended December 31, 2023 and 2022 | F-6 | |
| Notes to Consolidated Financial Statements | F-8 |
|
2) |
Financial Statement Schedules |
The following financial statement schedule is set forth on page F-16 of this Annual Report on Form 10-K:
Schedule II—Valuation and Qualifying Accounts for the years ended December 31, 2023 and 2022
All other financial statement schedules have been omitted because they are not applicable or not required or because the information is included elsewhere in the Consolidated Financial Statements or the Notes thereto.
|
(3) |
Exhibits Required by Item 601 of Regulation S-K |
___________________
|
* |
Filed herewith. |
|
** |
Indicates a management contract or compensatory plan or arrangement. |
|
(1) |
These interactive data files shall not be deemed filed or a part of a registration statement or prospectus for purposes of Sections 11 or 12 of the Securities Act of 1933, as amended, or Section 18 of the Securities Exchange Act of 1934, as amended, or otherwise subject to liability under these sections. |
|
(2) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed October 4, 2006. |
|
(3) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed June 23, 2008. |
|
(4) |
Incorporated by reference from the registrant’s Annual Report on Form 10-K filed March 8, 2010. |
|
(5) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed April 14, 2010. |
|
(6) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed April 28, 2010. |
|
(7) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed August 2, 2013. |
|
(8) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed October 23, 2013. |
|
(9) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed May 14, 2015. |
|
(10) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed June 16, 2016. |
|
(11) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed August 4, 2017. |
|
(12) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed September 7, 2018. |
|
(13) |
Incorporated by reference from the registrant’s Annual Report on Form 10-K filed March 26, 2019. |
|
(14) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed April 30, 2019 |
|
(15) |
Incorporated by reference from the registrant’s Quarterly Report on Form 10-Q filed November 7, 2019. |
|
(16) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed January 21, 2020. |
|
(17) |
Incorporated by reference from the registrant’s Annual Report on Form 10-K filed March 24, 2020. |
|
(18) |
Incorporated by reference from the registrant’s Quarterly Report on Form 10-Q filed November 12, 2021. |
|
(19) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed June 26, 2020. |
|
(20) |
[Reserved] |
|
(21) |
Incorporated by reference from Amendment No. 3 to registrant’s Registration Statement on Form S-1 (File No. 333-239958) filed September 8, 2020. |
|
(22) |
Incorporated by reference from Amendment No. 4 to registrant’s Registration Statement on Form S-1 (File No. 333-239958) filed September 23, 2020. |
|
(23) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed September 25, 2020. |
|
(24) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed October 26, 2020. Pursuant to Item 601(b)(10) of Regulation S-K, certain confidential portions of this exhibit have been omitted as the Company has determined (i) the omitted information is not material and (ii) the omitted information would likely cause competitive harm to the Company if publicly disclosed. |
|
(25) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed November 30, 2020. Pursuant to Item 601(b)(10) of Regulation S-K, certain confidential portions of this exhibit have been omitted as the Company has determined (i) the omitted information is not material and (ii) the omitted information would likely cause competitive harm to the Company if publicly disclosed. |
|
(26) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed February 11, 2021. |
|
(27) |
Incorporated by reference from the registrant’s Annual Report on Form 10-K filed March 23, 2021. |
|
(28) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed September 29, 2021. Pursuant to Item 601(b)(10) of Regulation S-K, certain confidential portions of this exhibit have been omitted as (i) the Company has determined the omitted information is not material and (ii) the Company customarily and actually treats the omitted information as private or confidential. |
|
(29) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed November 10, 2021. Pursuant to 399 601(b)(10) of Regulation S-K, certain confidential portions of this exhibit have been omitted as (i) the Company has determined the omitted information is not material and (ii) the Company customarily and actually treats the omitted information as private or confidential. |
|
(30) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed January 20, 2022. |
|
(31) |
Incorporated by reference from the registrant’s Annual Report on Form 10-K filed March 9, 2022. |
|
(32) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed May 27, 2022. |
|
(33) |
Incorporated by reference from the registrant’s Quarterly Report on Form 10-Q filed August 3, 2022. |
|
(34) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed December 8, 2022. |
|
(35) |
Incorporated by reference from the registrant’s Annual Report on Form 10-K filed March 23, 2023. |
|
(36) |
Incorporated by reference from the registrant’s Quarterly Report on Form 10-Q filed May 4, 2023. Pursuant to Item 601(b)(10) of Regulation S-K, certain confidential portions of this exhibit have been omitted as (i) the Company has determined the omitted information is not material and (ii) the Company customarily and actually treats the omitted information as private or confidential. |
|
(37) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed July 18, 2023. |
|
(38) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed December 4, 2023. |
|
(39) |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed January 31, 2024. |
|
ITEM 16. |
FORM 10-K SUMMARY |
None.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
GEOVAX LABS, INC. |
||
|
By: |
/s/ David A. Dodd |
|
|
Name: |
David A. Dodd |
|
|
Title: |
President and Chief Executive Officer |
|
|
Date: |
February 29, 2024 |
|
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been duly signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
|
Name |
Position |
Date |
||
|
/s/ David A. Dodd |
Director President and Chief Executive Officer |
February 29, 2024 |
||
|
David A. Dodd |
(Principal Executive Officer) |
|||
|
/s/ Mark W. Reynolds |
Chief Financial Officer |
February 29, 2024 |
||
|
Mark W. Reynolds |
(Principal Financial and Accounting Officer) |
|||
|
/s/ Randal D. Chase |
Director |
February 29, 2024 |
||
|
Randal D. Chase |
||||
|
/s/ Dean G. Kollintzas |
Director |
February 29, 2024 |
||
|
Dean G. Kollintzas |
||||
|
/s/ Nicole Lemerond |
Director |
February 29, 2024 |
||
|
Nicole Lemerond |
||||
|
/s/ Robert T. McNally |
Director |
February 29, 2024 |
||
|
Robert T. McNally |
||||
|
/s/ Jayne Morgan |
Director |
February 29, 2024 |
||
|
Jayne Morgan |
||||
|
/s/ John N. Spencer, Jr. |
Director |
February 29, 2024 |
||
|
John N. Spencer, Jr. |
GEOVAX LABS, INC.
INDEX TO 2023 CONSOLIDATED FINANCIAL STATEMENTS
| Report of Independent Registered Public Accounting Firm | F-2 |
| Consolidated Balance Sheets as of December 31, 2023 and 2022 | F-4 |
| Consolidated Statements of Operations for the years ended December 31, 2023 and 2022 | F-5 |
| Consolidated Statements of Stockholders’ Equity for the years ended December 31, 2023 and 2022 | F-6 |
| Consolidated Statements of Cash Flows for the years ended December 31, 2023 and 2022 | F-7 |
| Notes to Consolidated Financial Statements | F-8 |
| Financial Statement Schedule: | |
| Schedule II – Valuation and Qualifying Accounts for the years ended December 31, 2023 and 2022 | F-16 |
 |
235 Peachtree Street NE Suite 1800 Atlanta, GA 30303 |
404 588 4200 wipfli.com |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Stockholders and the Board of Directors of GeoVax Labs, Inc.
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of GeoVax Labs, Inc. and subsidiary (the “Company”) as of December 31, 2023 and 2022, and the related consolidated statements of operations, stockholders’ equity and cash flows for the years then ended and the related notes to the consolidated financial statements and schedule (collectively, the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2023 and 2022, and the results of its operations and its cash flows for the years then ended, in conformity with accounting principles generally accepted in the United States of America.
Emphasis of a Matter
Substantial Doubt about the Company’s Ability to Continue as a Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the consolidated financial statements, the Company expects to continue to generate operating losses in the foreseeable future and will require additional funding to continue its research and development activities. This raises substantial doubt about the Company’s ability to continue as a going concern. Management’s plans in regard to these matters are also described in Note 2 to the consolidated financial statements. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
Critical audit matters are matters arising from the current period audit of the consolidated financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. We determined that there were no critical audit matters.
/s/
We have served as the Company’s auditor since 2005.
February 29, 2024
|
GEOVAX LABS, INC. |
||||||
|
CONSOLIDATED BALANCE SHEETS |
|
December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
ASSETS |
||||||||
|
Current assets: |
||||||||
|
Cash and cash equivalents |
$ | $ | ||||||
|
Prepaid expenses |
||||||||
|
Total current assets |
||||||||
|
Property and equipment, net |
||||||||
|
Other assets |
||||||||
|
Total assets |
$ | $ | ||||||
|
LIABILITIES AND STOCKHOLDERS’ EQUITY |
||||||||
|
Current liabilities: |
||||||||
|
Accounts payable |
$ | $ | ||||||
|
Accrued expenses |
||||||||
|
Total current liabilities |
||||||||
|
Commitments (Note 4) |
||||||||
|
Stockholders’ equity: |
||||||||
| Common stock, $ par value: | ||||||||
|
Authorized shares – |
||||||||
|
Issued and outstanding shares – |
||||||||
|
Additional paid-in capital |
||||||||
|
Accumulated deficit |
( |
) | ( |
) | ||||
|
Total stockholders’ equity |
||||||||
|
Total liabilities and stockholders’ equity |
$ | $ | ||||||
See accompanying notes to consolidated financial statements.
|
GEOVAX LABS. INC. |
||||||
|
CONSOLIDATED STATEMENTS OF OPERATIONS |
|
Years Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Grant revenue |
$ | $ | ||||||
|
Operating expenses: |
||||||||
|
Research and development |
||||||||
|
General and administrative |
||||||||
|
Total operating expenses |
||||||||
|
Loss from operations |
( |
) | ( |
) | ||||
| Other income: | ||||||||
|
Interest income |
||||||||
|
Net loss |
$ | ( |
) | $ | ( |
) | ||
|
Basic and diluted: |
||||||||
|
Net loss per common share |
$ | ( |
) | $ | ( |
) | ||
|
Weighted average shares outstanding |
||||||||
See accompanying notes to consolidated financial statements.
GEOVAX LABS, INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
|
Total |
||||||||||||||||||||
|
Common Stock |
Additional |
Accumulated |
Stockholders’ |
|||||||||||||||||
|
Shares |
Amount |
Paid-in Capital |
Deficit |
Equity |
||||||||||||||||
|
Balance at December 31, 2021 |
$ | $ | $ | ( |
) | $ | ||||||||||||||
|
Sale of common stock and warrants for cash |
||||||||||||||||||||
|
Issuance of common stock upon warrant exercise |
||||||||||||||||||||
|
Issuance of common stock for services |
||||||||||||||||||||
|
Stock option expense |
- | - | ||||||||||||||||||
|
Net loss for the year ended December 31, 2022 |
- | - | ( |
) | ( |
) | ||||||||||||||
|
Balance at December 31, 2022 |
( |
) | $ | |||||||||||||||||
|
Issuance of common stock upon warrant exercise |
- | |||||||||||||||||||
|
Issuance of common stock for services |
||||||||||||||||||||
|
Stock option expense |
- | - | ||||||||||||||||||
|
Net loss for the year ended December 31, 2023 |
- | - | ( |
) | ( |
) | ||||||||||||||
|
Balance at December 31, 2023 |
$ | $ | $ | ( |
) | $ | ||||||||||||||
See accompanying notes to consolidated financial statements.
|
GEOVAX LABS. INC. |
|||||||||||||||||
|
CONSOLIDATED STATEMENTS OF CASH FLOWS |
|
Years Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Cash flows from operating activities: |
||||||||
|
Net loss |
$ | ( |
) | $ | ( |
) | ||
|
Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||
|
Depreciation and amortization |
||||||||
|
Stock-based compensation expense |
||||||||
|
Changes in assets and liabilities: |
||||||||
|
Grant funds receivables |
||||||||
|
Prepaid expenses and other current assets |
( |
) | ( |
) | ||||
| Other assets | ( |
) | ||||||
|
Accounts payable and accrued expenses |
( |
) | ( |
) | ||||
|
Total adjustments |
( |
) | ||||||
|
Net cash used in operating activities |
( |
) | ( |
) | ||||
|
Cash flows from investing activities: |
||||||||
|
Purchase of equipment |
( |
) | ( |
) | ||||
|
Net cash used in investing activities |
( |
) | ( |
) | ||||
| Cash flows from financing activities: | ||||||||
|
Net proceeds from sale of common stock and warrants |
||||||||
|
Net proceeds from warrant exercises |
||||||||
| Net cash provided by financing activities | ||||||||
|
Net increase (decrease) in cash and cash equivalents |
( |
) | ||||||
|
Cash and cash equivalents at beginning of period |
||||||||
|
Cash and cash equivalents at end of period |
$ | $ | ||||||
See accompanying notes to consolidated financial statements.
GEOVAX LABS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Years Ended December 31, 2023 and 2022
|
1. |
Nature of Business |
GeoVax Labs, Inc., headquartered in the Atlanta, Georgia metropolitan area, is a clinical-stage biotechnology company incorporated under the laws of the State of Delaware. GeoVax Labs, Inc. and its wholly owned subsidiary, GeoVax, Inc., a Georgia corporation, are collectively referred to herein as “GeoVax” or the “Company”.
The Company is focused on developing immunotherapies and vaccines against cancers and infectious diseases using novel vector vaccine platforms. GeoVax’s product pipeline includes ongoing human clinical trials for a next-generation Covid-19 vaccine and a gene-directed therapy for advanced head and neck cancer. Additional preclinical research and development programs include preventive vaccines against Mpox (monkeypox), hemorrhagic fever viruses (Ebola Zaire, Ebola Sudan, Marburg, and Lassa Fever) Zika virus, and malaria, as well as immunotherapies for solid tumors.
|
2. |
Summary of Significant Accounting Policies |
Principles of Consolidation
Basis of Presentation and Going Concern
We believe that our existing cash resources will be sufficient to continue our planned operations into the second quarter of 2024. We are devoting substantially all of our present efforts to research and development of our vaccine and immunotherapy candidates and will require additional funding to continue these activities. We plan to pursue additional cash resources through public or private equity or debt financings, government grants/contracts, arrangements with strategic partners, or from other sources. There can be no assurance that additional funding will be available on favorable terms or at all. These factors collectively raise substantial doubt about the Company’s ability to continue as a going concern within one year from the date these financial statements are issued. Management believes that we will be successful in securing the additional capital required to continue the Company’s planned operations, but that our plans do not fully alleviate the substantial doubt about the Company’s ability to operate as a going concern.
The accompanying consolidated financial statements have been prepared assuming that we will continue as a going concern, which contemplates realization of assets and the satisfaction of liabilities in the normal course of business for the twelve-month period following the issue date of these financial statements. The financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might result from the outcome of the uncertainties described above.
The accompanying consolidated financial statements, and all share and per share information contained herein, have been retroactively restated to reflect the reverse stock split described in Note 10.
Use of Estimates
Cash and Cash Equivalents
We consider all highly liquid investments with a maturity of three months or less when purchased to be cash equivalents. Our cash and cash equivalents consist primarily of bank deposits and money market accounts. The recorded values approximate fair market values due to the short maturities.
Fair Value of Financial Instruments and Concentration of Credit Risk
Financial instruments that subject us to concentration of credit risk consist primarily of cash and cash equivalents, which are maintained by high credit quality financial institutions. The carrying values reported in the balance sheets for cash and cash equivalents approximate fair values.
Property and Equipment
Property and equipment are stated at cost, less accumulated depreciation and amortization. Expenditures for maintenance and repairs are charged to operations as incurred, while additions and improvements are capitalized. We calculate depreciation using the straight-line method over the estimated useful lives of the assets (generally years). We amortize leasehold improvements using the straight-line method over the term of the related lease.
We recognize leases in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Update (ASU) No. 2016-02, Leases (ASU 2016-02), which requires lessees to classify leases as either financing or operating leases based on the principle of whether or not the lease is effectively a financed purchase by the lessee. This classification determines whether lease expense is recognized based on an effective interest method or on a straight-line basis over the term of the lease, respectively. A lessee is also required to record a right-of-use asset and a lease liability for all leases with a term of greater than 12 months regardless of their classification. In the case of our facility lease agreement which has an effective term of less than 12 months, we made an accounting policy election to not recognize lease assets and liabilities and record lease expense on a straight-line basis over the lease term.
Impairment of Long-Lived Assets
We review long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability of assets to be held and used is measured by a comparison of the carrying amount of the assets to the future net cash flows expected to be generated by such assets. If we consider such assets to be impaired, the impairment to be recognized is measured by the amount by which the carrying amount of the assets exceeds the expected future net cash flows from the assets.
Accrued Expenses
As part of the process of preparing our financial statements, we estimate expenses that we believe we have incurred, but have not yet been billed by our third-party vendors. This process involves identifying services and activities that have been performed by such vendors on our behalf and estimating the level to which they have been performed and the associated cost incurred for such service as of each balance sheet date.
Net Loss Per Share
Basic and diluted loss per common share are computed based on the weighted average number of common shares outstanding, including prefunded warrants outstanding as of December 31, 2023. The Company’s additional potentially dilutive securities, which include stock options and stock purchase warrants, have been excluded from the computation of diluted net loss per share as the effect would be antidilutive. The securities that could potentially dilute basic earnings per share in the future and that have been excluded from the computation of diluted net loss per share totaled
Revenue Recognition
We recognize revenue in accordance with FASB Accounting Standards Update 2014-09, Revenue from Contracts with Customers (ASU 2014-09), which created a new Topic, Accounting Standards Codification Topic 606. The standard is principle-based and provides a five-step model to determine when and how revenue is recognized. The core principle is that an entity should recognize revenue when it transfers promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services.
We have received payments from government entities under non-refundable grants in support of our vaccine development programs. We record revenue associated with these grants when the reimbursable costs are incurred and we have complied with all conditions necessary to receive the grant funds. From time to time, we may enter into collaborative research and development agreements for specific vaccine development approaches and/or disease indications whereby we receive third-party funding for preclinical research under certain of these arrangements. Each agreement is evaluated in accordance with the process defined by ASU 2014-09 and revenue is recognized accordingly.
Research and Development Expense
Research and development costs are charged to expense as incurred and consist of costs incurred in the discovery, development, testing and manufacturing of our product candidates. These expenses consist primarily of (i) salaries, benefits, and stock-based compensation for personnel, (ii) laboratory supplies and facility-related expenses to conduct development, (iii) fees paid to third-party service providers to perform, monitor and accumulate data related to our preclinical studies and clinical trials, (iv) costs related to sponsored research agreements, (v) costs to procure and manufacture materials used in clinical trials, and (vi) license fees and other expenses associated with technology license agreements.
We accrue for estimated costs of research and development activities conducted by third-party service providers, which may include the conduct of preclinical studies and clinical trials, and contract manufacturing activities. When evaluating the adequacy of the accrued liabilities, we analyze progress of the studies or trials, including clinical trial participant enrollment, completion of events, invoices received and other events. Advance payments for research and development activities are deferred and included in prepaid expenses and other assets. The deferred amounts are expensed as the related goods are delivered or the services are performed.
Patent Costs
Our expenditures relating to obtaining and protecting patents are charged to expense when incurred and are included in general and administrative expense.
Period-to-Period Comparisons
Our operating results are expected to fluctuate for the foreseeable future. Therefore, period-to-period comparisons should not be relied upon as predictive of the results for future periods.
Income Taxes
We account for income taxes using the liability method. Under this method, deferred tax assets and liabilities are recognized for the estimated future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted rates in effect for the year in which temporary differences are expected to be recovered or settled. Deferred tax assets are reduced by a valuation allowance unless, in the opinion of management, it is more likely than not that some portion or all of the deferred tax assets will be realized.
Stock-Based Compensation
We account for stock-based transactions in which the Company receives services from employees, directors or others in exchange for equity instruments based on the fair value of the award at the grant date. Stock-based compensation cost for awards of common stock is estimated based on the price of the underlying common stock on the date of issuance. Stock-based compensation cost for stock options or warrants is estimated at the grant date based on each instrument’s fair value as calculated by the Black-Scholes option pricing model. We recognize stock-based compensation cost as expense ratably on a straight-line basis over the requisite service period for the award. See Note 6 for additional stock-based compensation information.
Other Recent Accounting Pronouncements
There have been no recent accounting pronouncements or changes in accounting pronouncements which we expect to have a material impact on our financial statements, nor do we believe that any recently issued, but not yet effective, accounting standards if currently adopted would have a material effect on our financial statements.
|
3. |
Balance Sheet Components |
Prepaid Expenses – Prepaid expenses consist of the following as of December 31, 2023 and 2022:
|
2023 |
2022 |
|||||||
|
Prepaid clinical trial costs (current portion) |
$ | $ | ||||||
|
Prepaid insurance premiums |
||||||||
|
Prepaid rent |
||||||||
|
Other prepaid expenses |
||||||||
|
Total prepaid expenses |
$ | $ | ||||||
Property and Equipment – Property and equipment consist of the following as of December 31, 2023 and 2022:
|
2023 |
2022 |
|||||||
|
Equipment and furnishings |
$ | $ | ||||||
|
Leasehold improvements |
||||||||
|
Total property and equipment |
||||||||
|
Accumulated depreciation and amortization |
( |
) | ( |
) | ||||
|
Total property and equipment, net |
$ | $ | ||||||
Depreciation expense was $
Other Assets – Other assets consist of the following as of December 31, 2023 and 2022:
|
2023 |
2022 |
|||||||
|
Prepaid clinical trial costs (noncurrent portion) |
$ | $ | ||||||
|
Prepaid technology license fees |
||||||||
|
Deposits |
||||||||
|
Total other assets |
$ | $ | ||||||
Accrued Expenses – Accrued expenses consist of the following as of December 31, 2023 and 2022:
|
2023 |
2022 |
|||||||
|
Accrued license fees |
$ | $ | ||||||
|
Payroll-related liabilities |
||||||||
|
Other accrued expenses |
||||||||
|
Total accrued expenses |
$ | $ | ||||||
|
4. |
Commitments |
Operating Lease. We lease approximately
License Agreements. We have entered into license agreements for various technologies and patent rights associated with our product development activities. These agreements may contain provisions for upfront payments, milestone fees due upon the achievement of selected development and regulatory events, minimum annual royalties or other fees, and royalties based on future net sales. Due to the uncertainty of the achievement and timing of the contingent events requiring payment under these agreements, the amounts to be paid by us in the future are not determinable.
Other Commitments. In the normal course of business we enter into various contracts and purchase commitments including those with contract research organizations (“CROs”) and contract manufacturing organizations (“CMOs”) for clinical trials services and production of materials for use in our clinical trials. Most contracts are generally cancellable, with notice, at the Company’s option. Payments due upon cancellation may consist of payments for services provided or expenses incurred to date, or cancellation penalties depending on the time of cancellation.
5. Stockholders’ Equity
January 2022 Private Placement – On January 19, 2022, we closed a private placement of
May 2022 Private Placement – On May 27, 2022, we closed a private placement of
December 2023 Warrant Exercise Inducement – On December 2, 2023, we entered into a warrant exercise inducement letter with the holder of the January 2022 Warrant and the May 2022 Warrant, pursuant to which the holder agreed to fully exercise each warrant (aggregate of
Other Common Stock Transactions – During 2023 and 2022 we issued
Common Stock Reserved for Future Issuance – Common stock reserved for future issuance consists of the following at December 31, 2023:
|
Shares |
||||
|
Stock warrants outstanding |
||||
|
Stock options outstanding |
||||
|
Stock options authorized for future grants |
||||
|
Total |
||||
Stock Options
We have two stock-based incentive plans (the “Stock Incentive Plans”) pursuant to which our Board of Directors may grant stock options or other stock awards to our employees, directors and consultants. A total of
A summary of the Company’s stock option activity during 2023 is presented below.
|
Number of Shares |
Weighted- Average Exercise Price |
Weighted- Average Remaining Contractual Term (yrs) |
Aggregate Intrinsic Value |
|||||||||||||
|
Outstanding at December 31, 2022 |
$ | $ | - |
|||||||||||||
|
Granted |
||||||||||||||||
|
Exercised |
||||||||||||||||
|
Forfeited or expired |
( |
) | ||||||||||||||
|
Outstanding at December 31, 2023 |
$ | $ | ||||||||||||||
|
Exercisable at December 31, 2023 |
$ | $ | ||||||||||||||
Stock Warrants
The table below summarizes information concerning warrants outstanding as of December 31, 2023.
|
Issue Date |
Number of Shares |
Exercise Price |
Expiration |
|||||||||
|
June 2020 |
$ |
June 2025 |
||||||||||
|
September 2020 |
September 2025 |
|||||||||||
|
September 2020 |
March 2024 |
|||||||||||
|
February 2021 |
August 2024 |
|||||||||||
|
September 2021 |
September 2026 |
|||||||||||
|
December 2023 |
- |
- | ||||||||||
|
December 2023 |
June 2029 |
|||||||||||
|
Outstanding at December 31, 2022 |
||||||||||||
As a result of anti-dilution price adjustments related to our equity transactions in December 2023, the exercise price of the June 2020 Warrants was reduced from $
|
6. |
Stock-Based Compensation Expense |
Stock-based compensation expense related to stock options is recognized on a straight-line basis over the requisite service period for the award and is allocated to research and development expense or general and administrative expense based upon the classification of the individual to whom the award is granted.
We use the Black-Scholes model for determining the grant date fair value of our stock option grants. This model utilizes certain information, such as the interest rate on a risk-free security with a term generally equivalent to the expected life of the option being valued and requires certain other assumptions, such as the expected amount of time an option will be outstanding until it is exercised or expired, to calculate the fair value of stock options granted. We granted no stock options during 2023. The significant assumptions we used in our fair value calculations for stock options granted during 2022 were as follows:
|
Weighted average risk-free interest rates |
% | |||
|
Expected dividend yield |
% | |||
|
Expected life of option (in years) |
||||
|
Expected volatility |
% |
The weighted-average grant date fair values of stock options granted during 2023 and 2022 were $-
We also have issued shares of restricted common stock to consultants and recognize the related expense over the terms of the related agreements. As of December 31, 2023, there is $
The following table summarizes our total stock-based compensation expense for employees, directors and consultants for the years ended December 31, 2023 and 2022:
|
2023 |
2022 |
|||||||
|
Stock options: |
||||||||
|
Research and development |
$ | $ | ||||||
|
General and administrative |
||||||||
|
Total stock option expense |
||||||||
|
Stock awards (consultants): |
||||||||
|
General and administrative |
||||||||
|
Total stock-based compensation expense |
$ | $ | ||||||
|
7. |
Retirement Plan |
We participate in a multi-employer defined contribution retirement plan (the “401k Plan”) administered by a third-party service provider, and the Company contributes to the 401k Plan on behalf of its employees based upon a matching formula. During the years ended December 31, 2023 and 2022 our contributions to the 401k Plan were $
|
8. |
Income Taxes |
At December 31, 2023, we have a consolidated federal net operating loss (“NOL”) carryforward of approximately $
Deferred income taxes reflect the net effect of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. We have established a full valuation allowance equal to the amount of our net deferred tax assets due to uncertainties with respect to our ability to generate sufficient taxable income to realize these assets in the future. The table below presents significant components of our deferred tax assets and liabilities at December 31, 2023 and 2022.
|
2023 |
2022 |
|||||||
|
Deferred tax assets: |
||||||||
|
Net operating loss carryforward |
$ | $ | ||||||
|
Research and development tax credit carryforward |
||||||||
|
Stock-based compensation expense |
||||||||
|
Accrued expenses |
||||||||
|
Total deferred tax assets |
||||||||
|
Deferred tax liabilities |
||||||||
|
Depreciation |
||||||||
|
Net deferred tax assets |
||||||||
|
Valuation allowance |
( |
) | ( |
) | ||||
|
Net deferred tax asset after reduction for valuation allowance |
$ | - |
$ | - |
||||
A reconciliation of the U.S. federal income tax rate to the Company’s effective tax rate is as follows:
|
2023 |
2022 |
|||||||
|
U.S. federal statutory rate applied to pretax loss |
% | % | ||||||
|
State income tax (benefit) |
||||||||
|
Permanent differences |
( |
) | ( |
) | ||||
|
NOL carryforward expiration |
( |
) | ( |
) | ||||
|
R&D tax credits, net of expiration |
||||||||
|
Change in valuation allowance and other adjustments |
( |
) | ( |
) | ||||
|
Effective tax rate |
% | % | ||||||
|
9. |
Grant Revenue |
During 2022 we received payments from government entities under our grants from the National Institute of Allergy and Infectious Diseases (NIAID) and from the U.S. Department of Defense in support of our vaccine research and development efforts. We record revenue associated with government grants as the reimbursable costs are incurred. Total revenues recorded for these grants during 2022 was $
|
10. |
Subsequent Events |
Reverse Stock Split and Reduction of Authorized Shares of Common Stock
At a special meeting of our stockholders held on January 16, 2024, our stockholders approved an amendment to our certificate of incorporation to (i) reduce our authorized shares of common stock from
Common Stock Transactions
In January 2024, we issued
GEOVAX LABS, INC.
SCHEDULE II – VALUATION AND QUALIFYING ACCOUNTS
For the Years Ended December 31, 2023 and 2022
|
Additions (Reductions) |
||||||||||||||||||||
|
Description |
Balance at Beginning Of Period |
Charged to Costs and Expenses |
Charged to Other Accounts |
Deductions |
Balance at End Of Period |
|||||||||||||||
|
Reserve Deducted in the Balance Sheet From the Asset to Which it Applies: |
||||||||||||||||||||
|
Allowance for Deferred Tax Assets |
||||||||||||||||||||
|
Year ended December 31, 2023 |
$ | $ | $ | - |
$ | - |
$ | |||||||||||||
|
Year ended December 31, 2022 |
$ | $ | $ | - |
$ | - |
$ | |||||||||||||