UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
(Mark One)
For the fiscal year ended
OR
For the transition period from to
Commission file number:
(Exact name of registrant as specified in its charter)
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
(Address of principal executive offices) (Zip Code)
Registrant’s telephone number, including area code: (
Securities registered pursuant to Section 12(b) of the Exchange Act:
Title of each class
|
Trading Symbol(s)
|
Name of each exchange on which registered
|
The |
Securities registered pursuant to Section 12(g) of the Exchange Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes ☐
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer |
☐ |
Accelerated filer |
☐ |
☒ |
Smaller reporting company |
||
|
|
Emerging growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. |
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐ |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No
The aggregate market value of the registrant’s voting and non-voting common equity held by non-affiliates of the registrant (without admitting that any person whose shares are not included in such calculation is an affiliate) computed by reference to the price at which the common equity was last sold as of the last business day of the registrant’s most recently completed second fiscal quarter was approximately $
As of March 1, 2024, the registrant had
DOCUMENTS INCORPORATED BY REFERENCE
The following documents (or parts thereof) are incorporated by reference into the following parts of this Form 10-K:
Certain information required in Part III of this Annual Report on Form 10-K is incorporated by reference from the Registrant’s Proxy Statement for the 2024 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission.
TABLE OF CONTENTS
|
|
|
|
7 |
|
Item 1. |
7 |
|
Item 1A. |
33 |
|
Item 1B. |
67 |
|
Item 1C. |
67 |
|
Item 2. |
70 |
|
Item 3. |
70 |
|
Item 4. |
70 |
|
|
71 |
|
Item 5. |
71 |
|
Item 6. |
71 |
|
Item 7. |
Management’s Discussion and Analysis of Financial Condition and Results of Operations |
72 |
Item 7A. |
81 |
|
Item 8. |
81 |
|
Item 9. |
Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
81 |
Item 9A. |
81 |
|
Item 9B. |
82 |
|
Item 9C. |
Disclosure Regarding Foreign Jurisdictions that Prevent Inspections |
82 |
|
83 |
|
Item 10. |
83 |
|
Item 11. |
83 |
|
Item 12. |
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
83 |
Item 13. |
Certain Relationships and Related Transactions, and Director Independence |
83 |
Item 14. |
83 |
|
|
84 |
|
Item 15 |
84 |
|
Item 16. |
87 |
|
|
88 |
2
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), that relate to future events or our future financial performance regarding, among other things, the plans, strategies and prospects, both business and financial, of the Company. These statements are based on the beliefs and assumptions of our management team. Although we believe that our plans, intentions and expectations reflected in or suggested by these forward-looking statements are reasonable, we cannot assure you that we will achieve or realize these plans, intentions or expectations. Forward-looking statements are inherently subject to risks, uncertainties and assumptions. Generally, statements that are not historical facts, including statements concerning possible or assumed future actions, business strategies, events or results of operations, are forward-looking statements. These statements may be preceded by, followed by or include the words “believes,” “estimates,” “expects,” “projects,” “forecasts,” “may,” “will,” “should,” “seeks,” “plans,” “scheduled,” “anticipates” or “intends” or similar expressions. Forward-looking statements contained in this Annual Report on Form 10-K include, but are not limited to, statements about:
3
These and other factors that could cause actual results to differ from those implied by the forward-looking statements in this Annual Report on Form 10-K are more fully described in Item 1A under the heading “Risk Factors.” The risks described under the heading “Risk Factors” are not exhaustive. Other sections of this Annual Report on Form 10-K, such as the description of our Business set forth in Item 1 and our Management’s Discussion and Analysis of Financial Condition and Results of Operations set forth in Item 7 describe additional factors that could adversely affect our business, financial condition or results of operations. New risk factors emerge from time to time, and it is not possible to predict all such risk factors, nor can we assess the impact of all such risk factors on our business or the extent to which any factor or combination of factors may cause actual results to differ materially from those contained in any forward-looking statements. Forward-looking statements are not guarantees of performance. You should not put undue reliance on these statements, which speak only as of the date hereof. All forward-looking statements attributable to the Company or persons acting on the Company’s behalf are expressly qualified in their entirety by the foregoing cautionary statements. We undertake no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
4
SUMMARY OF RISK FACTORS
Our business is subject to numerous risks and uncertainties that you should consider before investing in our securities. Some of the principal risk factors are summarized below:
5
These and other material risks we face are described more fully in Item 1A, Risk Factors, which investors should carefully review prior to making an investment decision with respect to the Company or its securities.
6
PART I
All brand names or trademarks appearing in this report are the property of their respective holders. Use or display by us of other parties’ trademarks, trade dress, or products in this report is not intended to, and does not, imply a relationship with, or endorsements or sponsorship of, us by the trademark or trade dress owners. Unless the context requires otherwise, references in this report to the “Company,” “we,” “us,” and “our” refer to Hyperfine, Inc. and its wholly-owned subsidiaries, including Hyperfine Operations, Inc., or Legacy Hyperfine, and Liminal Sciences, Inc., or Liminal, as the case may be.
Item 1. BUSINESS
Overview
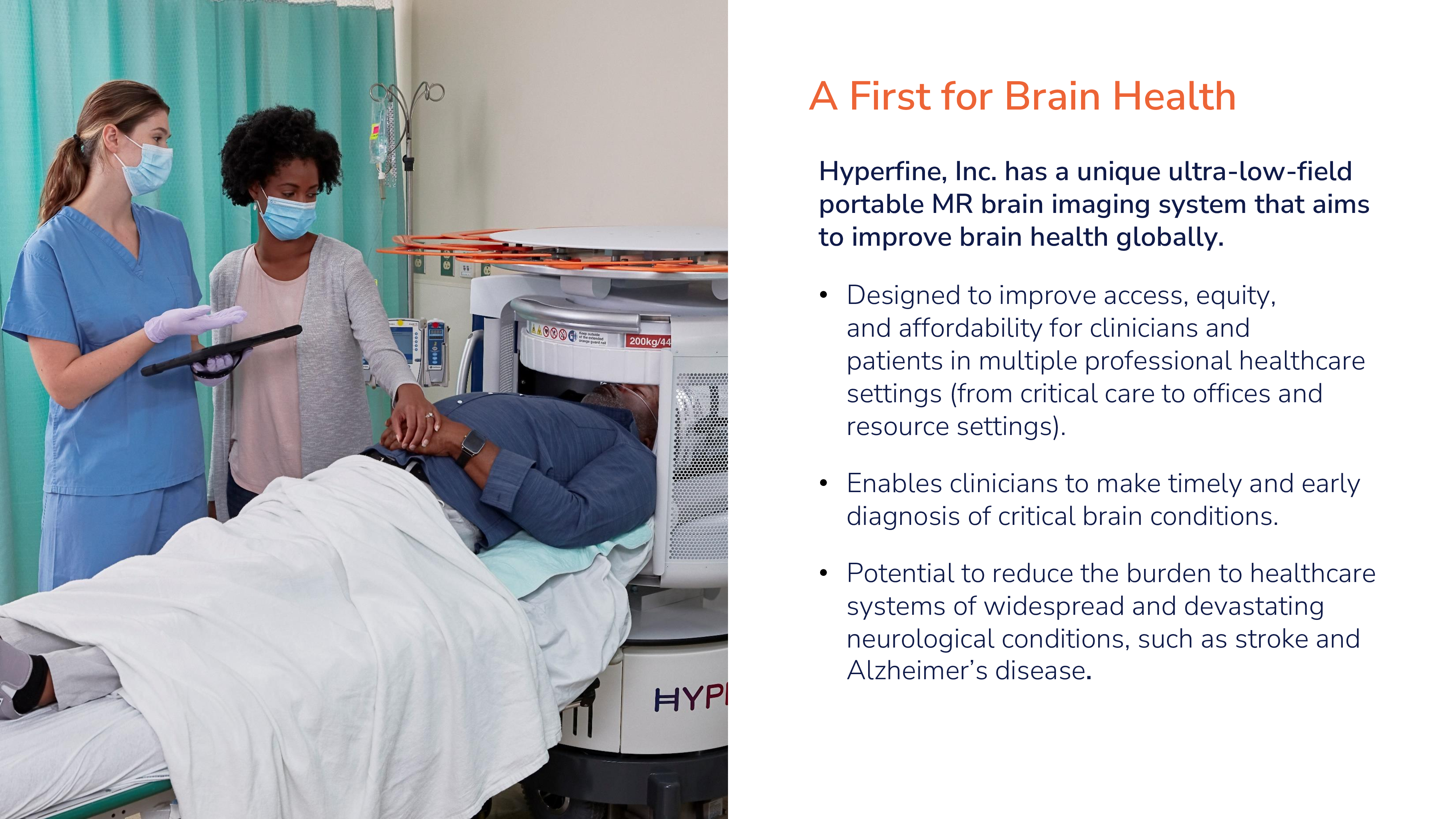
We are an innovative health technology business with a mission to revolutionize patient care globally through accessible, affordable, clinically relevant ultra-low-field (“ULF”) magnetic resonance (“MR”) brain imaging. Our Swoop® Portable MR Imaging® System (“Swoop® system”) produces high-quality images at a lower magnetic field strength than conventional magnetic resonance imaging (“MRI”) scanners. Our Swoop® system is designed to transform brain MR for the patient, the clinician and the provider, and to provide a highly differentiated experience for patients, timely imaging to clinicians, and favorable economics for hospital administrators. The Swoop® system is a portable, ULF MRI device for producing images that display the internal structures of the head where full diagnostic examination is not clinically practical. When interpreted by a trained physician, these images provide information that can be useful in determining a diagnosis. Healthcare professionals can use the Swoop® system to make effective clinical diagnoses and decisions in various care settings where conventional MRI devices are inaccessible. The easy-to-use interface and portable design of our Swoop® system make it easily and readily accessible anywhere in a hospital, clinic, or patient care site and it does not require any special facilities accommodations nor specialized personnel to operate safely. ULF MR does not expose patients to harmful ionizing radiation and compares favorably in this regard to X-ray computed tomography (“CT”) or positron emission tomography (“PET”).
The demand for MR imaging has been increasing due to the aging population and the rising prevalence of neurological, neurodegenerative, and cardiovascular conditions, as well as the trends towards decentralized healthcare in mature as well as low- and middle-income countries. Healthcare professionals and insurers recognize imaging as an effective, non-invasive diagnostic tool for evaluation and ongoing monitoring. The Swoop® system is the next generation brain imaging device designed to increase access to MRI in a cost-effective manner and expand the current $35 billion imaging market.
Despite its advantages, many healthcare institutions worldwide lack the facilities, specialized operators, and capital necessary to acquire and maintain expensive conventional MRI devices. The Swoop® system is the first U.S. Food and Drug Administration (“FDA”)-cleared, portable, ULF, MR brain imaging system and is capable of providing imaging at multiple sites of care such as intensive care units, clinics, emergency departments or physicians’ offices, and can inform the timely detection, diagnosis, monitoring and treatment of acute and chronic conditions inside and outside the hospital. We designed the Swoop® system to address the limitations of conventional imaging technologies and make brain MR imaging accessible nearly anytime and anywhere across professional healthcare settings. We believe the adoption of the Swoop® system by healthcare professionals has clinical and economic benefits throughout healthcare communities in both high and low resource settings.
The Swoop® system is AI-powered and integrates deep learning, a form of AI, for the reconstruction and denoising of T1, T2, and fluid-attenuated inversion recovery (“FLAIR”) sequences. The Swoop® system also incorporates deep learning denoising in the diffusion-weighted imaging (“DWI”) sequences for image post-processing. The integration of deep learning does not require any additional steps from the user. As a result, deep learning can enhance the image quality and, consequently, the diagnostic value of images generated at ULF. The algorithms are designed to improve ULF image quality, while reducing the impact of scan artifacts. The images created with these algorithms were validated by expert radiologists. Furthermore, the Swoop® system is used clinically every day as the first mover in the field of portable ULF MRI, and with an install base that continues to expand. The learnings from this field experience have served to improve our software, AI, and denoising algorithms resulting in the image quality and performance boosts of our product over the eight software releases since our initial clearance. As we move forward, we are continuously investing in improving our AI-powered image quality and leveraging each imaging-focused software release to further improve the Swoop® system performance. Furthermore, we possess a portfolio of 165 issued patents worldwide and 127 patents pending as of February 15, 2024.
Legacy Hyperfine received initial 510(k) clearance for brain imaging of patients of all ages from the FDA in 2020 and has now received eight subsequent clearances from the FDA after the initial clearance. We received our most recent clearance from the FDA in October 2023 for the latest update of our Swoop® system AI-powered software. This updated software expanded the Swoop® system’s AI denoising capabilities by incorporating deep learning denoising in the DWI sequence for image post-processing. The Swoop® system has also received marketing authorization for brain imaging in several countries, including the European Union (CE), the United Kingdom (UKCA), Canada, Australia and New Zealand.
7
Our initial commercial focus has been in the U.S. market and with customers that use the Swoop® system inside the hospital setting, including clinical use in pediatric and adult applications. To support these use cases, we have partnered with leading institutions to collect clinical evidence. There have been a large number of presentations at medical meetings and publications on the use of the Swoop® system in these hospital settings. We have run two studies in these areas, SAFE-MRI ECMO (Assessing the Safety and Feasibility of bedside portable low-field brain Magnetic Resonance Imaging in patients with ECMO) presented at the Asia-Pacific Chapter of the Extracorporeal Life Support Organization (APELSO) conference in November 2023 and HOPE PMR (Portable MRI for Children with Neurological Injury—A Pilot Study using Hydrocephalus as an Index Condition) which completed enrollment in mid-2023. We are actively involved in additional market adoption studies to validate the use of the Swoop® system for patients with acute ischemic stroke where the Swoop® system could be used in the emergency department. There are four study sites actively enrolling in the ACTION PMR (ACuTe Ischemic strOke detectioN) clinical study. As a prospective, international, multi-site observational study, ACTION PMR aims to examine the integration of brain imaging with the Swoop® system into the stroke diagnosis and treatment workflow. Furthermore, we have committed to conducting a utility study for the use of the Swoop® system for the monitoring of Alzheimer’s patients for Amyloid related Imaging Abnormalities (ARIA), associated with the novel Amyloid targeted therapies being developed by biopharmaceutical companies, the first of which was approved by the FDA in 2023. The FDA-approval of these new drugs requires multiple and frequent patient MRIs to monitor for ARIA throughout the course of treatment. We believe the portable Swoop® system could offer a highly differentiated, cost effective, and accessible solution for this new use case. The ability to place and use the Swoop® system in any professional care setting can significantly simplify the care navigation for these patients. Both stroke and Alzheimer’s disease represent potential worldwide incremental opportunities for our company in new care settings.
We are building our direct commercial infrastructure in the United States and have third-party distributors responsible for commercialization efforts in Canada, Australia and New Zealand. We have also added Europe to our geographic commercial focus and plan to develop our business in the European continent through a hybrid approach of direct and third-party resources.
In December 2022, we suspended our Liminal program to develop a device to non-invasively measure key vital signs in the brain. Also, in December 2022, we announced an organizational restructuring designed to decrease our operating costs and create a more streamlined organization to support our business. As a result, we terminated approximately 13% of our global workforce including, among others, the employees of our Liminal subsidiary. In connection with the restructuring, we incurred $1.0 million of costs consisting primarily of cash severance costs, other severance benefits, fixed asset impairment costs and other related restructuring costs. We completed the restructuring as of the first quarter of 2023.
Our wholly-owned subsidiaries, Legacy Hyperfine and Liminal were founded in 2014 and 2018, respectively, by Dr. Jonathan Rothberg, a serial entrepreneur who received the Presidential Medal of Technology and Innovation in 2016 for inventing a novel next-generation DNA sequencing method and has founded more than ten healthcare and technology companies, including 454 Life Sciences, Ion Torrent, CuraGen, Butterfly Network, and Quantum-Si. Legacy Hyperfine has raised over $160 million in equity investments and partnership milestones from leading institutional investors, including GV (formerly Google Ventures), and grants, including the Bill & Melinda Gates Foundation (the "BMGF").
Our Core Competitive Strengths
We believe that our competitive strengths include the following:
There is a large and growing MRI market, and we have the potential to augment conventional MRI capacity to bring benefit to patients, clinicians and providers around the world. We believe our solution addresses a vast unmet need across the global market by adding a portable, accessible and affordable brain MR system to increase the existing capacity of conventional high-field MRI scanners as imaging rates continue to grow across the aging population and patients with neurological and neurodegenerative conditions and as efficient and readily available MRI scanners deployed in different sites of care become increasingly valuable in high, middle and low resources countries globally. Our solution is designed to complement conventional MRI and seamlessly integrates into the hospital workflow, by processing orders from the Hospital Information Systems (HIS) and allowing users to upload images into their picture archiving and communication system (“PACS”), or directly onto our cloud PACS, which then makes images easily available for diagnostic purposes. To support information security reviews with customers we have obtained several certifications including HITRUST CSF® v9.4 Risk-based, 2-year, SOC2 Type 1 and SOC2 Type 2.
We believe the Swoop® system can expand the existing $35 billion global imaging market (expected to grow at a 5.7% CAGR from 2022 to 2030) by making MRI available to a broader set of patients in both developed and emerging markets, as well as improve the utilization of conventional MRI scanners through decreased wait times and more efficient patient flow. Our initial focus is on patients in intensive care units and emergency departments, where timeliness of MRI is critical, as well as pediatric patients where minimizing exposure to ionizing radiation from CT is important. An MRI scan can be essential for diagnosis and urgent intervention. The Swoop® system can be wheeled directly to a patient’s bedside and offers a prompt solution for those
8
patients who require an MRI scan but are too critically ill to be transported to a conventional MRI scanner and who may otherwise be forced to forego a scan, wait until a scanner is available, or wait until their condition stabilizes.
Hyperfine has a unique ultra-low-field portable MR brain imaging system that aims to improve brain health globally
The Swoop® system does not emit ionizing radiation and therefore does not have the increased risk of cancer that comes with CT imaging. The absence of ionizing radiation can benefit patients with conditions that require frequent follow-up with multiple scans per year, such as pediatric hydrocephalus. In certain circumstances, such as in the management of patients with delirium or altered mental status, familiarity or keeping the surrounding environment as similar as possible can be critical, which we believe makes portable devices like our Swoop® system particularly valuable since patients do not need to move to distant radiology suites for conventional MRI scans. Studies show that 37% of patients report anxiety-related reactions in an isolated room for imaging. With our Swoop® system, we can offer a quieter, calmer and more comfortable experience with the option of a family member or other caregiver being present by the patient’s side during the scanning process.
Our portable Swoop® system also helps avoid the risk of patient injury during transport through the ability to bring the system to the patient. By performing scans for urgent and critically ill patients at the bedside, we can help prevent the adverse incidents that occur with approximately 26–79% of critically ill patients during transport. The Swoop® system can eliminate the labor-intensive and high-risk process of transporting patients on ventilators or connected to other life-sustaining devices, which can be especially valuable in the staff shortage environment that many healthcare institutions continue to experience.
9
The Swoop® system is designed to increase global MRI capacity and to enable brain imaging anywhere
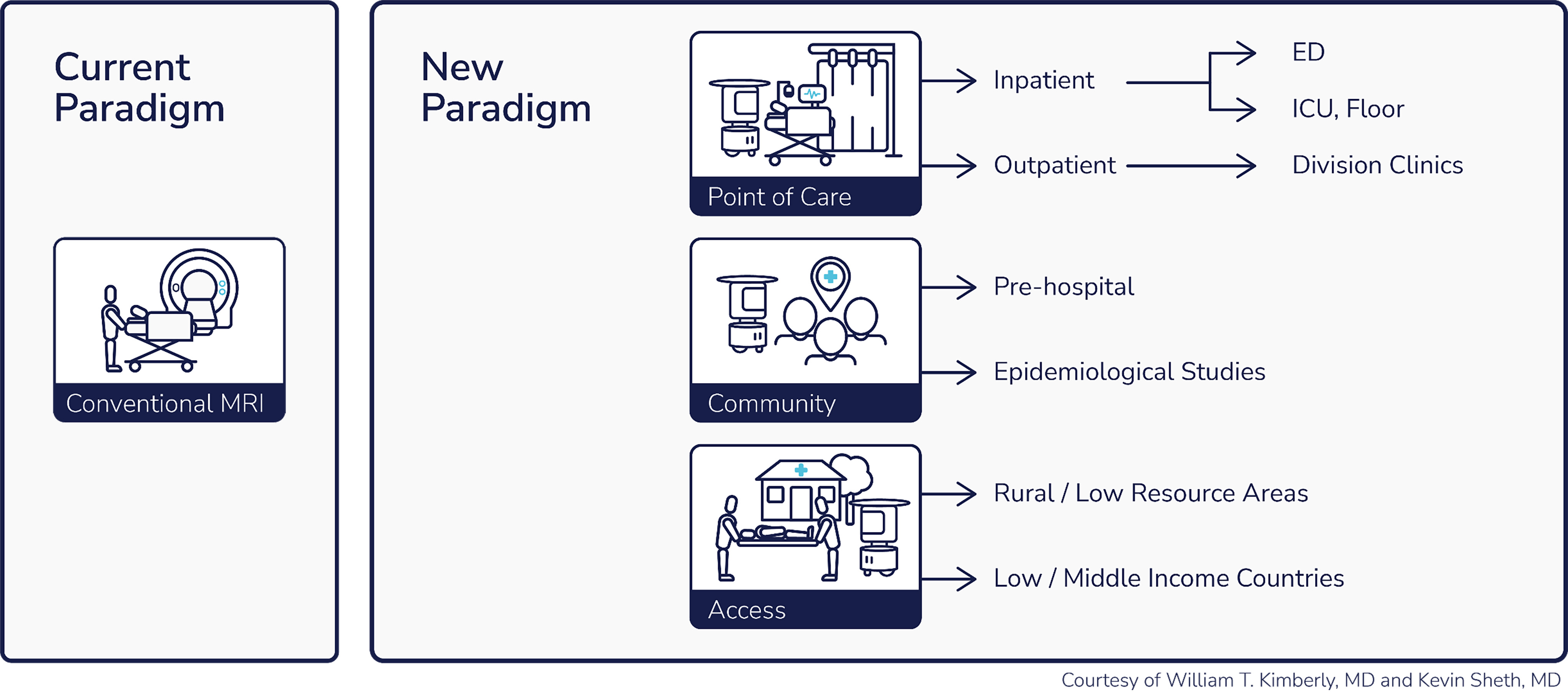
Conventional MRI scanners require specially trained technicians fully dedicated to operating those systems and increase the time and cost related to nurses and porters transporting patients to the MRI unit. Our Swoop® system is designed to simplify the image acquisition process. We have designed our system to be user-friendly and require minimal training to be operated, including a simple user interface accessed through a Hyperfine provided tablet. The Swoop® system’s portability and accessibility at the bedside can further allow more time for healthcare professionals to focus on other important activities related to patient care, diagnosis, and treatment.
As healthcare costs continue to rise, we believe our Swoop® system has the potential to allow for significant potential cost reductions that can benefit the entire imaging ecosystem. Our Swoop® system has reduced hardware costs, using modern computational power and deep learning advances. The cost benefits of our Swoop® system are not limited to a customer’s initial purchase of the system. Our customers can benefit by not having to spend on additional cooling, power, and high maintenance expenses typically required throughout the lifetime of conventional MRI scanners. Unlike conventional MRI scanners, the use of the Swoop® system also does not require a specialized radio frequency (RF) room to safely house the MRI scanner, which can result in a lower total cost of installing an MRI and allow flexibility in the use of space for other essential patient care activities.
10
Potential Opportunities
Our portable Swoop® system has the potential to streamline the care navigation for Alzheimer’s patients on the novel Amyloid Targeted Therapies. We believe the Swoop® system has the potential to represent a valuable tool in the screening and monitoring of Alzheimer’s patients considered for and or treated with the newly approved Amyloid Targeted Therapies (“ATT”), the first of which, Leqembi (by Biogen/Esai), was approved by the FDA in late 2023. The labeling approved by the FDA for Leqembi requires that patients on the drug undergo multiple MRI’s to monitor risk of ARIA. We believe conventional MRI capacity is not readily nor easily available to patients at the levels that may be needed for such monitoring in the event ATTs become widely prescribed. Undergoing a conventional MRI represents an additional burden to patients, given long wait times for MRI across the United States and the fact that conventional MRI systems are not always in the same location as the infusion centers or treating neurologists. The portable Swoop® system can be safely and conveniently located near the treating clinician or the infusion center, which can streamline the complex care navigation for these patients.
Our portable Swoop® system has the potential to to accelerate characterization and diagnosis of acute ischemic stroke. In 2023, we launched an international observational study, ACTION PMR, which has enrolled over 100 patients as of March 2024, with enrollment from all four participating sites. The goal of the study is to use portable brain imaging to identify strokes and salvageable brain tissue.
We have the opportunity to make brain MR accessible and affordable to patients in low resource settings globally when conventional MRI is not available. We have also initiated a global research program in close collaboration with Kings College London, supported by grant funding from the BMGF to assess the clinical feasibility of our Swoop® system in providing readily available brain imaging to infants between the ages of 0 to 24 months in low to medium income countries and now have deployed Swoop® systems in African and Asian countries.
Our Strategies
Our strategies include the following:
11
We have also partnered with several institutions to initiate utility studies to evaluate the use of the Swoop® system in screening and monitoring Alzheimer’s patients on the novel Amyloid Targeted Therapies. Alzheimer’s disease represents a potential large incremental and growing global market opportunity for our technology. We believe the Swoop® system can significantly streamline the care navigation and lower the burden for these patients and their clinicians and meaningfully increase the reach of Alzheimer’s diagnosis and treatment across patients of diverse demographics and income levels.
Our vision of providing affordable and accessible imaging that enables earlier detection and remote management of health conditions worldwide was initially made possible by grant funding from the BMGF. Through our engagement with BMGF, we deployed the Swoop® system to low-middle resource settings without readily-accessible MRI technology. The multiple grants provided by the BMGF, which commenced funding in the spring of 2020, support the deployment of 45 Swoop® systems across African and Asian countries. The ongoing investigation is designed to provide data to validate the potential use of the Swoop® system in measuring the impact of maternal anemia, malnutrition, infection, and birth-related injury.
Industry and Market
MRI is a non-ionizing radiation risk imaging modality widely used by healthcare professionals across various clinical settings for the medical diagnosis of patients, staging of disease, and continued assessment following treatment. MRI is noninvasive, sometimes eliminates the need for surgical intervention or invasive procedures when used correctly, and offers superior soft tissue contrast resolution compared to other imaging modalities like CT. It is a more sensitive and potentially objective measure of brain tissue and injury. MRI is used to examine CNS, musculoskeletal, and other diseases. The prevalence and incidence rates of these diseases continue to increase across the globe. According to a United Nations report, up to one billion people, nearly one in six of the world’s population, suffer from neurological disorders, including Alzheimer’s and Parkinson’s disease, stroke, multiple sclerosis, epilepsy, migraine, brain injuries, and neuro infections, with some 6.8 million dying of these disorders each year.
The aging population and rising prevalence of cancer, cardiovascular, neurological, and orthopedic conditions have augmented the demand for MRI. Healthcare professionals and insurers recognize imaging as a cost-effective and non-invasive diagnostic for prevention, early detection and ongoing monitoring. The portable Swoop® system represents a new paradigm in brain imaging that we believe will drive improved access and efficiency and expand the current $35 billion imaging market. Given the significant patient populations needing diagnostic imaging, we have positioned ourselves in an underpenetrated market with substantial room for growth. While the current imaging market is mainly limited to high-resource countries, we believe our system can help make MRI imaging more accessible in mature and low resource settings globally, leading to an increase in both MRI penetration rates and the size of the overall market opportunity.
Market needs
Despite MRI’s advantages to diagnose and monitor patients through treatment, access to MRI scanners can be challenging. Numerous challenges are associated with the use of conventional MRI devices:
12
Due to these challenges, the adoption of conventional MRI scanners has been limited across medical settings in the United States and globally, especially in rural locations where many individuals only have access to small clinics. Conventional MRI scanners also include additional costs of establishing an MRI suite, patient support areas, machine installation and servicing, including costly or inaccessible cryogens for cooling, software upgrading, and maintenance that burden hospitals and clinics with limited ongoing funding.
There are significant benefits to diagnosing a disease at multiple sites of care in its early stages, which can reduce the time to treatment and improve the quality of life for those patients. We have taken advantage of technological advances in electronics, computing and AI to develop an MRI device that is not only portable but also uses an ultra-low-field 64mT (0.064T) permanent magnet, which is much lower than the 1.5T or higher field strength of conventional MRI scanners, and does not require cryogenic cooling. Our advanced technology allows healthcare professionals to conduct an MRI scan at the patient’s bedside in the hospital or any clinical setting to begin early diagnosis, intervention, and ongoing treatment.
According to a 2008 report from the World Health Organization, 90% of the world does not have access to MRI, primarily due to socioeconomic factors. Many low-resource countries recognize the benefit of investing in their healthcare infrastructure, and it is expected to cause a spur in growth for the global MRI market. For example, India and China are two of the fastest-growing markets building its healthcare infrastructure in rural areas. The ability of these countries to build the facilities needed to house these large systems and train highly specialized personnel to operate conventional MRI scanners presents a challenge.
Products and Services
Our Swoop® Portable MR Imaging® System
We designed our Swoop® system to address an unmet need in accessible, affordable, portable medical imaging through a unique combination of hardware and AI-powered software services. Our hardware is powered using modern computational power and deep learning advances. Our software is designed to address the traditional ease-of-use and integration challenges often presented by specialized medical technologies. Our system operates from a Wi-Fi-capable tablet and integrates with picture archiving and communication systems (“PACS”) to enable fast and confident clinical decision-making.
13

Features
Portable Brain Neuroimaging—FDA-Cleared for MRI of the Brain and Head in Patients of All Ages
Portable brain imaging has only been possible using CT. CT can visualize bones, blood vessels with contrast agents, and hemorrhage well, but is not as sensitive as MRI at imaging the brain’s anatomy. Additionally, CT delivers a significant amount of ionizing radiation. Exposing patients to radiation increases the risk of developing cancer, which limits CT’s use for critically ill patients and makes it particularly hazardous for pediatric patients.
The gold standard for neuroimaging is MRI, which can provide excellent high-resolution images of the soft tissues of the brain without being obscured by the skull. MRI can provide critical insight into brain trauma and disease but historically has not been portable and not available at multiple sites of care. Because of their size, weight, and safety issues, conventional MRI scanners were only available in hospitals, major medical centers, and outpatient imaging providers, meaning that patients typically must be transported to the MRI scanner.
We have developed a new category of medical imaging, portable MRI, that is smaller, lighter weight, and lower cost than conventional MRI scanners yet maintains the soft tissue visualization capabilities critical for brain imaging. Since launching our FDA-cleared portable Swoop® system in 2020, brain imaging is now available for patients of all ages at practically any site of patient care in the United States.
Ultra-Low-Field System
To engineer this new category of portable MRI, we made several significant design changes with respect to conventional MRI, particularly the magnetic field strength. Over the past 40 years, the goal for improving conventional MRI scanners has been to attain higher magnetic field strength. In 2017, the FDA cleared the first 7T MRI, after 20 years of development. It was noted that the added field strength allows for better visualization of smaller structures and subtle pathologies that may improve disease diagnosis. We have taken a different approach by developing our Swoop® system to have an ultra-low-field magnet of 0.064T, which enables MRI to become portable because, unlike conventional MRI scanners, the field strength of the magnet in our system does not require a specialized room to house or use the MRI scanner safely. This field strength and form factor comes from a unique optimization of the magnet size, weight, field uniformity, and patented design of the permanent magnet structure that provides sufficient image clarity for diagnostic purposes.
There are additional benefits to operating an MRI system with an ultra-low-field magnet. It reduces the risk of iron-containing objects becoming projectile and injuring patients or operators, a typical concern of conventional MRI scanners. Furthermore, the radiofrequency pulses used in conventional MRI are responsible for 55% of the FDA-reported adverse events from MRI, causing skin and internal burns in some patients. Operating at 0.064T means using lower energy radiofrequency pulses and significantly reducing associated safety risks.
14
Motion Correction
Portable MRI at the point of care can provide MRI insights to more patients than previously possible. All MRI scans regularly suffer from quality problems due to patient motion, with approximately 30% of all scans from inpatient or emergency department exams having moderate or severe image quality issues. We have developed software that compensates for subtle motion to improve image quality in the most challenging, and often most in need, patients. We believe that with continued development, our technology can produce diagnostic scans without requiring the operator to make expert adjustments to the scanning procedure due to typical patient movements.
Noise-Cancellation Technology
Designing an ultra-low-field magnet is not sufficient to enable portable MRI. Portable MRI must also address the electromagnetic interference surrounding us. Electromagnetic interference makes conventional MRI outside of a shielded room unsafe and impossible. Conventional MRI scanners are permanently installed in a special room where the walls, floor, and ceiling are encased in copper or aluminum to provide an environment for conventional MRI machines to operate, in which all man-made and natural electromagnetic interference is prevented from entering. Installation of these shielded rooms typically costs more than $100,000.
We have developed proprietary, patented AI-powered noise-cancellation technology to enable portable MRI. Our technology measures the external electromagnetic interference and subtracts that from the interference that swamps the MRI signals.
Delivery of Multiple Sequences with Tissue Contrasts
MRI can provide images with different soft tissue contrasts through various sequences that can highlight a range of pathologies. These contrasts are standard in conventional MRI and allow for the differentiation of various tissue types aiding in establishing the diagnosis. Our Swoop® system generates images with contrast weightings with which physicians are most familiar and which are most clinically useful for the target use cases: T1, T2, fluid-attenuated inversion recovery (“FLAIR”), and diffusion-weighted imaging (“DWI”) with apparent diffusion coefficient (“ADC”) maps.
Image Quality
We deliver diagnostic-quality images to healthcare professionals. The images from our Swoop® system are higher in contrast resolution than other portable medical brain imaging systems, such as portable CT scanners. Our portable Swoop® system also delivers images that are diagnostic and clinically relevant in many use cases when high field MRI is not available. Our MRI signal is produced at 0.064T compared to 1.5T or higher produced by conventional, fixed MRI scanners. We believe that the Swoop® system provides the potential to improve the quality of care for patients who have limited or no access to conventional MRI, which includes 90% of the world’s population.
Controlled by an Easy-to-Use Wireless Tablet
As we seek to reach new markets and users with our Swoop® system, we have sought to make the operation of the device as intuitive and easy to use as possible. We believe it is important to consider usability when significantly changing how a medical device is used, specifically in MRI, where conventionally, the operator is required to have several years of training. We believe this is particularly important when used in hospital environments, including with unstable patients in the critical care and emergency situations such as stroke, where speed is essential.
The interfaces to the Swoop® system are simple, intuitive buttons, joystick controls to drive the system, and a familiar tablet controller for image acquisition and viewing. The user interface provided on the tablet offers a playlist of protocols based on the use case that can be started, stopped, and rearranged as needed. In addition to being easy to use and the consequential acceleration of hospital workflows that can result, our system provides standardized images across all placement sites due to our uniform manufacturing specifications and a consistent set of sequences that individual operators do not customize. Conventional MRI scanners are operated by highly-trained technologists and can have variations in image resolution and contrast weighting across sites due to institutional policies and radiologist preferences. We believe the standardization of images across scanners and sites will significantly benefit the ability of radiologists and other healthcare professionals to read our images efficiently and ultimately build a data repository of images that can serve as training sets to further improve our image quality and the Swoop® system user experience.
Integration with Picture Archiving and Communication System (PACS) and Secure Image Upload to the Cloud
Similar to other medical devices in hospitals, we designed our Swoop® system to seamlessly integrate with the hospital informational technology (“IT”) infrastructure, such that scans can be ordered easily and sent to PACS to be read by a radiologist. For applications
15
where access to such infrastructure is unavailable, we also offer a secure cloud based PACS where healthcare professionals, including teleradiology service providers, can view images from anywhere in the world. We believe the combination of portable MRI, where scans can be obtained outside the conventional MRI suite, and teleradiology can significantly improve patient care and increase access.
Design
Location Flexibility
Despite the weight of our Swoop® system being 1,400 pounds, its powered drive system means operators can move the system around the hospital with minimal effort. The Swoop® system can be moved from bed-to-bed and easily positioned in tight spaces because it turns on the spot with a zero-turn radius.
Open Layout Designed to Improve the Patient Experience and Potentially Reduce Patient Anxiety
For an MRI scan in a traditional setting, a patient arrives at the radiology department of a hospital and typically enters through a door covered with radiation warnings and other hazard symbols. The patient then proceeds to a waiting room where they undergo a lengthy safety questionnaire and are asked to remove all jewelry and clothes (to their underwear) and put on a hospital gown. Wait times vary from a half hour to several hours before the patient enters the console room and is led through a large metal door into the RF screen room by themselves. Typical conventional MRI scanners are long tubes where the patient is positioned on a motorized bed, and RF coils are attached around the patient who has been instructed to lay still. The MRI technician uses the motorized bed to push the patient into the long tube of the large superconducting magnet, leaves the room, closes the metal door to the scan room, and tells the patient over an intercom that the scan is about to start. The patient experiences an extremely loud environment throughout the scanning experience. The conventional MRI procedure is often a daunting experience for the patient that can cause significant anxiety, especially for pediatric patients who are separated from their families during this time.
Unlike conventional MRI, our Swoop® system is entirely contained in a system that is just 55 inches tall and 34 inches wide and is designed to scan patients in their beds. Parents, family, or caregivers can be close to the patient as they are scanned, with just their head in the transparent head RF coil. The system is quiet enough to allow constant verbal contact with the patient, which can create a considerably less distressing experience for the patient compared to that of a conventional MRI scanner.
Powered Using a Standard Wall Outlet
Transportable throughout any hospital environment, our Swoop® system plugs into a standard wall outlet (100-230 VAC, 50/60 Hz, 15A) and uses less than 900W of electricity. This ability is achieved with low-power electronics, including efficient power supplies and power amplifiers, coupled with a zero-power consumption permanent magnet. Our Swoop® system does not require many of the components of conventional MRI, including the liquid helium used in conventional MRI superconducting magnets or the associated safety and supporting infrastructure, the chilled water-cooling systems for the power electronics and gradient coils, and the room air conditioning needed to extract the heat generated in the separate electronics machine room, or the special 480 V, 3-phase, 200A power supply.
Services
Our Swoop® system service and support program includes support and technical assistance with hardware and software issues. A combination of remote and onsite support in collaboration with onsite technical staff solves software issues. In addition, it also includes our Hyperfine Image Viewer, a cloud PACS that users can use to upload images for storage purposes. The service and support program also grants access to our future software upgrades. Recent upgrades include our FDA-cleared image quality improvements that enhances the quality of images in the presence of subtle motion and other potential future upgrades designed to improve the patient workflow and diagnosis.
Our People
Legacy Hyperfine was founded in 2014 by Dr. Jonathan Rothberg. Our mission is to provide accessible and affordable imaging and monitoring through MRI to revolutionize healthcare for people around the world.
As of February 15, 2024, we had 131 employees, all of whom were full-time employees and of whom 25 work in sales, clinical and marketing, 84 work in research, development, manufacturing and operations, and 22 work in general and administrative capacities. As of February 15, 2024, 127 of our employees were located in the United States and, internationally, four were located in the United Kingdom and Europe. None of our employees are represented by a labor union or are subject to a collective bargaining agreement.
16
Dr. Rothberg and our business have been recognized for leadership. Legacy Hyperfine was founded in 2014 by Dr. Jonathan Rothberg, a serial entrepreneur that received the Presidential Medal of Technology and Innovation in 2016 for inventing a novel next generation DNA sequencing method and has founded more than 10 healthcare and technology companies, including 454 Life Sciences, Ion Torrent, CuraGen, Butterfly Network and Quantum-Si.
Information About Our Executive Officers and Directors
The following persons were our executive officers and directors as of February 15, 2024:
Name |
Position |
Executive Officers |
|
Maria Sainz |
President, Chief Executive Officer and Director |
Brett Hale |
Chief Administrative Officer, Chief Financial Officer, Treasurer and Corporate Secretary |
Thomas Teisseyre, Ph.D. |
Chief Operating Officer |
Directors |
|
R. Scott Huennekens |
Chairperson of the Board of Directors |
Jonathan M. Rothberg, Ph.D. |
Founder of Hyperfine and member of the Board of Directors |
John Dahldorf |
Chief Financial Officer, Adagio Medical |
Ruth Fattori |
Managing Partner, Pecksland Partners Senior Advisor, Boston Consulting Group |
Daniel J. Wolterman |
Chief Executive Officer, Wolterman Consulting LLC |
Environmental, Social and Governance Practices
We are committed to delivering products and solutions that build a healthier and more sustainable world for this and future generations. We have an Environmental, Social, and Governance (“ESG”) program and internal governance structure that we will adapt and expand as determined through our business operating reviews. We seek to align our ESG program and governance structure with our business strategy, the priorities of our stakeholders, our commitments and ambitions, and our need to adapt to changes in societal, environmental, and regulatory expectations. Our management team works to facilitate alignment with ongoing ESG efforts, which will include gathering input from internal and external stakeholders to help inform our ESG strategy and focus areas. We seek to support the transition to a more circular economy. We seek to reduce medical imaging equipment waste by promoting and enabling the reuse of equipment and parts. Every scraped system is disassembled by our service team and magnets are demagnetized and removed from the steel. Machines are refurbished or dismantled, harvested, and recycled, reducing waste and contributing to a circular economy.
Environmental Stewardship: We recognize the importance of taking measures to reduce our environmental footprint. The Swoop® system is a low-footprint MR-based device that can be deployed in a variety of settings including low- and middle-income countries. The Swoop® system is powered from a standard electrical outlet and does not require any special construction at site of care like conventional MRI requires. As we grow our business, we have initiated certain projects to begin tracking our environmental impact, and where feasible, have taken measures to increase our sustainability efforts. We support the employee-led Hyperfine Sustainability Resource Group and our environmental efforts include our commitment to reduce, reuse or recycle where possible or appropriate and energy efficient projects to lower energy use within our office areas and laboratories.
Human Capital Management: We believe that our people are the reason for our success and we have organized ourselves to maximize productivity and performance. We maintain a high bar for talent and actively work to build diversity within our workforce. Critical to achieving our strategic goals is our ability to build and retain an exceptional team in which each member plays a unique and important role.
We recognize that maintaining an engaged and top-notch workforce and a connection with the communities we serve are critical to our success. Comradery and community are at the core of who we are as a company and are integral facets of our human capital management strategy. We are inspired by each other and the possibilities of what we can achieve together. We understand that in order to drive innovation, we must continuously improve our human capital management strategies and find ways to foster engagement and growth within our organization. To this end, below are some of our initiatives:
Professional Development Programs and Opportunities: Our greatest asset is our employees and we aspire to provide them with opportunities so they can continue to grow and excel in their functions and our company. Professional growth of our employees leads to engagement, development and allows us to leverage opportunities so we can hire and promote key talent from within. Through
17
development planning, we strive for employees at all levels to focus on strengthening the skills required in their current role and potentially their next role. We are focused on building a culture of continuous coaching, feedback and open communication between managers and their direct reports throughout the entire year. We provide managers and employees with training on how to conduct effective forward-looking performance conversations and to set effective goals that are realistic, measurable, attainable, relevant and timebound.
Diversity, Equity and Inclusion: Our commitment to maintaining a top-performing company means investing in and creating ongoing opportunities for employee development in a diverse and inclusive workplace. We believe that a diverse workforce not only positively impacts our performance, fosters innovation, inspires us to achieve greater results, increases our collective capabilities and strengthens our culture, but it also cultivates an essential pipeline of experienced leaders for management. Hiring for diversity of thought, background and experience, and diversity of personal characteristics such as gender, race and ethnicity is intentional and continues to be an area of focus as we build and grow our workforce. We support the employee-led Hyperfine Women’s Resource Group and are an active member of external diversity groups that support diversity mentorship within the health technology field.
Compensation, Equity and Benefits: We have designed a broad-based compensation program that is designed to attract, retain and motivate our employees to deliver sustainable long-term value. We seek to deliver performance-driven, market competitive reward opportunities commensurate with company and individual performance. Many of our employees receive equity grants and cash bonuses in addition to base salaries and our benefits package. We believe that providing employees with an ownership interest in our company further strengthens the level of employee engagement. Furthermore, equity awards help align the interests of our employees with the long term interests of our stockholders. We also offer employees a health insurance package.
Governance, Ethics, and Compliance: Our board of directors is committed to robust corporate governance practices, risk oversight, stockholder rights, diversity, equity and inclusion, corporate sustainability, ethics and compliance in order to protect the long term interests of our company, stockholders and the patients we serve. Our board of directors adopted corporate governance principles applicable to us, including responsible oversight and management of the Company, effective controls and processes, compliance with SEC and Nasdaq Stock Market rules and regulations, maintaining an engaged board of directors and a board structure that recognizes the importance of compliance, diversity, appropriate compensation practices, and succession planning, among other matters.
We will continue to evolve and strengthen our human capital management strategies, increase our environmental efforts, maintain and continue to improve our corporate governance practices, and anticipate reporting on other corporate sustainability measures over time.
Marketing, Sales and Pricing
Marketing
Our marketing efforts are focused on accelerating awareness of our products and capabilities in order to create a strong reputation with clinicians and healthcare administrators. Our go-to-market approach features a targeted sales organization complemented by an array of promotional activities including media coverage, tradeshow exhibition, advertising, and live product demonstration. We principally target ICU, comprehensive and primary stroke accredited facilities. In the future, we plan to leverage this approach for both our Swoop® system and our future products that have similar end markets.
We recognize the role of education in accelerating clinical adoption of our products across the patient care pathway, including healthcare professionals who currently may not themselves be primary users of MRI technology. To support adoption of our product and in addition to our simplified product interface, we have built a team of clinically trained clinical support specialists to guide and coach clinicians on the unique features of our device and on the specific clinical application of our technologies.
Sales and Pricing
The Swoop® system is commercially available in the United States, Canada, Australia, and New Zealand. The system also has received CE certification for the European Union and UKCA certification in the United Kingdom. We are building our direct commercial infrastructure in the United States and also sell our products in other countries either through direct sales or through distributors.
We are primarily commercializing our device through one business model, which is ownership accompanied by an annual service and support agreement. In this model, the Swoop® system is typically sold with a service and support agreement that begins after a one year warranty and is sold initially in either 36- or 60-month terms. In certain cases, the Swoop® system is sold without an accompanying service and support agreement. We offer our customers payment term options that include, among others, either an upfront payment for the Swoop® system and annual payment for service and support or an annual payment option for the term of their agreement. The annual payment option contains a portion of interest for the Swoop® system over the term.
18
This model provides for the sale of the Swoop scanner, along with an off-the-shelf tablet for use with the scanner, to the customer based on agreed payment terms, which is more affordable than the average cost of $1.2 million for conventional MRI scanners.
To a lesser extent, we have also commercialized our device through a subscription bundle model in prior years, in which we sell the use of our device and an off-the-shelf tablet for use with the scanner, plus the same service and support benefits provided in the ownership with service and support model. The subscription bundle model is no longer available for commercial and research use of the device in 2023 and 2022.
To help ensure our customers receive the highest level of customer service, we plan to continue to sell directly to customers and provide ongoing customer support. However, as we expand internationally subject to regulatory authorization in those countries, we may continue to leverage distributors to sell our product depending on the commercial strategy for each country assessed on a country-by-country basis. Through our business model, we aim to provide MRI systems that are more affordable than conventional MRI systems and achieve our vision of increasing accessibility to MRI worldwide.
Suppliers and Manufacturing
Our Swoop® system is built using both custom-made and off-the-shelf components supplied by outside manufacturers and vendors located in the United States, Europe and Asia. One key custom-made component in our Swoop® system is the magnet, which is manufactured by a single source supplier in Europe. The majority of the other components for the Swoop® system are off-the-shelf or made using standard processes.
We purchase some of our components and materials used in manufacturing, including magnets, field programmable gate arrays (“FPGAs”), central processing units (“CPUs”) and molded plastics, from single sources. Although we believe that alternative sources of these components and materials would be available, it would take time to identify and validate replacement components, which could negatively affect our ability to supply the Swoop® system on a timely basis. We cannot give assurances that any alternative supplier would be able to recreate the manufacturing processes currently in use. To mitigate this risk, we typically carry a significant inventory of critical components. We are also working with our Swoop® system device manufacturer, Benchmark Electronics, Inc. (“Benchmark”), to add an additional magnet supplier to the manufacturing process to mitigate the risk to supply of our magnets by the current use of a single supplier.
All of our Swoop® system devices are manufactured, tested, shipped and supported by Benchmark from its facilities in Nashua, NH. We believe that this manufacturing strategy is efficient and conserves capital. However, in the event it becomes necessary to utilize a different contract manufacturer for our Swoop® system products, we would experience additional costs, delays and difficulties in doing so, and our business could be harmed.
Key Agreements
Manufacture and Supply Agreement with Benchmark Electronics, Inc.
In October 2018, Legacy Hyperfine entered into a Manufacture and Supply Agreement with Benchmark (the “MSA”). Under the MSA, Benchmark agreed to manufacture our products pursuant to binding purchase orders. Each month, we have agreed to provide Benchmark with a binding purchase order for a period specified by the MSA, as well as a non-binding forecast for each month within such period. If we do not provide the monthly purchase order and forecast update, then the first forecast month of the then-current forecast becomes binding so that a rolling binding commitment to purchase product for the specified period is maintained. The parties have agreed to meet periodically regarding any minimum order quantities of components under the MSA. We also have certain inventory related obligations, including the obligation to purchase excess and obsolete components from Benchmark. Excess components are determined based upon the amount of component inventory that exceeds the build plan for the specified period discussed above. We would be required to purchase such excess inventory and be credited back against future purchases of finished products as the inventory of components is reduced to the amount needed to meet the rolling build plan. Obsolete materials are immediately invoiced once identified.
Under the terms of the MSA, we granted Benchmark a non-exclusive, non-transferable, revocable, fully-paid, royalty-free license, without the right to sublicense, to use our technology solely to manufacture our products. The MSA provides that we will own any right, title and interest in any improvements or modifications to our technology made in the course of performance of Benchmark’s obligations under the MSA. We and Benchmark also agreed to indemnify each other against certain third-party claims.
Following the MSA’s initial three-year term, the MSA renews automatically for additional two-year terms unless either party gives 180 days’ prior written notice before the end of the then-current term to the other party electing not to renew the agreement. The MSA or any purchase order under the MSA may be terminated by either party for convenience upon 90 days’ prior written notice to the other party. The MSA may also be terminated by either party by written notice upon the occurrence of (i) a breach by the other party under the agreement which is not cured within 30 days after written notice by the terminating party, (ii) the other party becomes
19
insolvent, dissolves, liquidates or ceases to conduct business or (iii) the occurrence of payment-related breaches. Benchmark may also terminate the agreement upon the filing of any petition against us under bankruptcy or similar laws, where such petition is not vacated within 10 days via court order.
Competition
Several large companies, such as General Electric, Siemens, Philips, Hologic, Fuji, Toshiba, Canon and Hitachi currently dominate the medical imaging market. We expect that the existing market participants will remain key players in the future.
As a general matter, we view competition on two levels:
We view high-field MRI more as a complementary than a competitive technology. Particularly in the U.S., we are seeing substantial interest among our customers for the implementation of Swoop® system to augment traditional, high-field MRI workflows and increase throughput of high-field scanners.
Intellectual Property
Protection of our intellectual property is a strategic priority for our business. We rely on a combination of patents, trademarks, trade secrets and other intellectual property rights protections and contractual restrictions to protect our proprietary technologies.
The patents owned and in-licensed by us are generally directed to the architecture of MRI systems and related technology. We have developed a portfolio of issued patents and pending patent applications directed to commercial products and technologies for potential development. We believe that our intellectual property is a core strength of our business, and our strategy includes the continued development of our patent portfolio.
Our Swoop® System and Related Technology
As of February 15, 2024, we held an exclusive field of use licenses or ownership of 165 U.S. and foreign patents, and 127 U.S. and foreign patent applications. These patents and applications cover various components and techniques incorporated into and generally directed to our Swoop® system, including magnet design and manufacturing, electronics and circuitry, mechanical aspects, safety features, noise compensation technology, image formation and analysis software, and various other aspects of MRI systems. These issued patents and pending patent applications (if they were to issue as patents) have expected expiration dates ranging between 2035 and 2043. We cannot be certain that any patents will be issued from any of our pending patent applications, nor can we be certain that any of our existing patents or any patents that may be granted to us in the future will provide us with protection.
License Agreements
We have entered into licenses in the ordinary course of business relating to our technologies or other intellectual property rights or assets.
Exclusive License Agreements with The General Hospital Corporation (d/b/a Massachusetts General Hospital)
Legacy Hyperfine entered into an exclusive license agreement with The General Hospital Corporation (d/b/a Massachusetts General Hospital) (“MGH”) effective in May 2014 (the “May Agreement”) and an exclusive license agreement with MGH effective in June 2014 (the “June Agreement”), respectively, under each of which Legacy Hyperfine acquired an exclusive and worldwide license to specified patent rights owned by MGH relating to MRI technology. The licenses were granted to us subject to the right of MGH and not-for-profit academic, government and other not-for-profit institutions to make and to use the subject matter described or claimed in the rights granted under the licensed patents for research and educational purposes and, for any licensed patents that are supported by federal funding, subject to certain rights, conditions and limitations imposed by U.S. law, including a royalty-free, non-exclusive license granted to the U.S. government and a requirement that any products used or sold in the United States must be manufactured substantially in the United States.
20
Under the terms of each of the license agreements, we agreed to pay MGH an annual maintenance fee and agreed to reimburse MGH for certain patent related fees and costs incurred by MGH, including past patent fees and costs. If we were to enter into a sublicense under either license agreement, we would be obligated to pay MGH a percentage in the mid-teens of certain consideration paid to us by the sublicensee. The aggregate amount we paid under these agreements was $159,000.
At the end of the third quarter of 2022, we provided MGH with written notice that we were terminating our license agreements effective in the fourth quarter of 2022. The licensed patents are not used in our current products and we do not expect to use them in future products.
Government Regulation
Diagnostic and therapeutic medical devices like those we develop and distribute are subject to regulation by numerous regulatory bodies, including the U.S. Food and Drug Administration (“FDA”) and comparable international regulatory agencies. These agencies require developers of medical devices to comply with applicable laws and regulations governing the development, testing, manufacturing, packaging, labeling, marketing and distribution of medical devices. Devices are generally subject to varying levels of regulatory control, the most comprehensive of which requires that a clinical evaluation program be conducted before a device can be approved for marketing and commercial distribution. In addition, healthcare regulatory bodies in the United States and around the world impose a range of requirements related to paying for medical devices and the procedures in which they are used, including laws intended to prevent fraud, waste, and abuse of healthcare dollars.
U.S. Laws and Regulations
In the United States, medical devices are subject to extensive regulation at the federal level by the FDA under the Federal Food, Drug, and Cosmetic Act (“FDCA”) and its implementing regulations. The laws and regulations govern, among other things, medical device design and development, nonclinical and clinical testing, pre-market clearance, authorization or approval, establishment registration and product listing, product manufacturing, product packaging and labeling, product storage, advertising and promotion, product distribution, recalls and field actions, servicing and post-market clinical surveillance. A number of U.S. states also impose licensing and compliance regimes on companies that manufacture or distribute prescription devices into or within the state.
Some of our products are also subject to the Radiation Control for Health and Safety Act, administered by the FDA, which imposes performance standards and record keeping, reporting, product testing and product labeling requirements for electronic products that emit radiation, such as magnetic resonance imaging systems.
In addition, the commercialization and use of our devices in the United States is subject to regulation by the U.S. Department of Health and Human Services (“HHS”) and state agencies responsible for reimbursement and regulation of payment for healthcare items and services. Federal laws and regulations apply primarily in connection with government payer programs such as the Medicare and Medicaid programs, but state laws apply more broadly, encompassing healthcare items and services covered by private payers. At the state and federal level, the government’s interest is in regulating the quality and cost of healthcare and protecting the independent clinical judgment of licensed healthcare providers.
The Federal Trade Commission (“FTC”) also oversees the advertising and promotion of our products pursuant to broad authority to police deceptive advertising for goods or services within the United States. Under the Federal Trade Commission Act, the FTC is empowered, among other things, to (a) prevent unfair methods of competition and unfair or deceptive acts or practices in or affecting commerce; (b) seek monetary redress and other relief for conduct injurious to consumers; and (c) gather and compile information and conduct investigations relating to the organization, business, practices, and management of entities engaged in commerce. In the context of performance claims for products such as our goods and services, compliance with the FTC Act includes ensuring that there is scientific data to substantiate the claims being made, that the advertising is neither false nor misleading, and that any user or expert testimonials or endorsements we or our agents disseminate related to the goods or services comply with applicable disclosure rules and other regulatory requirements. In addition, with respect to our commercial products and any future products that are marketed as clinical products, FDA’s regulations applicable to medical device products prohibit them from being promoted for uses not within the scope of a given product’s authorized intended use(s), among other promotional and labeling rules applicable to products subject to the FDCA.
Further, medical device systems that include wireless radio frequency transmitters and/or receivers are subject to equipment authorization requirements in the United States. The Federal Communications Commission (“FCC”) requires advance clearance of all radio frequency devices before they can be sold or marketed in the United States. These clearances ensure that the proposed products comply with FCC radio frequency emission and power level standards and will not cause interference.
21
FDA Regulation of Medical Devices
Medical devices must undergo pre-market review by and receive clearance, authorization, or approval from the FDA prior to commercialization, unless the device is of a type exempted from such review by statute, regulation, or an FDA exercise of enforcement discretion. The FDA classifies medical devices into three classes based on risk. Regulatory control increases from Class I (lowest risk) to Class III (highest risk). The FDA generally must clear or approve the commercial sale of most new medical devices that fall within product categories designated as Class II and III. Commercial sales of most Class II and III medical devices within the United States must be preceded either by pre-market notification and FDA clearance pursuant to Section 510(k) of the FDCA (Class II) or by the granting of a pre-market approval (“PMA”) (Class III), after a pre-market application is submitted. Both 510(k) notifications and PMA applications must be submitted to FDA with significant user fees, although reduced fees for small businesses are available. Class I devices are generally exempt from pre-market review and notification, as are some moderate-risk Class II devices. Manufacturers of all classes of devices must comply with FDA’s Quality System Regulation (“QSR”), establishment registration, medical device listing, labeling requirements, and medical device reporting (“MDR”) regulations, which are collectively referred to as medical device general controls. Class II devices may also be subject to special controls such as performance standards, post-market surveillance, FDA guidelines, or particularized labeling. Some Class I and Class II devices may be exempted by regulation from the requirement of compliance with substantially all of the QSR.
510(k) Clearance Pathway
A 510(k) pre-market notification must contain information sufficient to demonstrate that the new device is substantially equivalent to a predictive device. A predicate device is a legally marketed device that is not subject to a PMA, meaning, (i) a device that was legally marketed prior to May 28, 1976 (“preamendments device”) and for which a PMA is not required, (ii) a device that has been reclassified from Class III to Class II or I, or (iii) a device that was found substantially equivalent through the 510(k) process. To obtain 510(k) clearance for a non-exempt Class II device, the FDA must determine that the device is substantially equivalent to such a predicate device based on the information submitted by the product developer in the pre-market notification. The FDA’s 510(k) clearance process generally takes from three to 12 months from the date the application is submitted, but it may take significantly longer if FDA has significant questions or needs more information about the new device or its manufacturing or quality controls.
As part of the 510(k) notification process for Class II devices that have an existing classification regulation available for purposes of the regulatory filing, the FDA may require the following:
Assuming successful completion of all required testing, a detailed 510(k) notification is submitted to the FDA requesting clearance to market the product. This premarket notification includes all relevant data from pertinent nonclinical studies and clinical trials (if applicable), together with detailed information relating to the product’s proposed labeling, and other relevant documentation. The FDA evaluates all 510(k) submissions prior to filing for substantive review based on specific acceptance criteria and may issue a refuse-to-accept notification if the submission is deficient with respect to any of the established criteria. If the FDA determines that the applicant’s device is substantially equivalent to the identified predicate device(s), the agency will issue a 510(k) clearance letter that authorizes commercial marketing of the device for one or more specific indications for use. If the FDA determines that the applicant’s device is not substantially equivalent to the predicate device(s), the agency will issue a not-substantially-equivalent letter stating that the new device may not be commercially distributed.
After a new medical device receives 510(k) clearance from the FDA, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change in its intended use, requires a new 510(k) clearance or could require the submission of a PMA application. The FDA requires each manufacturer to make the determination of whether a device modification requires a new 510(k) notification or PMA application in the first instance, but the FDA may review any such decision. If the FDA disagrees with a manufacturer’s decision not to seek a new 510(k) clearance or PMA for a particular change, the FDA may retroactively require the manufacturer to submit a 510(k) pre-market notification or a PMA application. The FDA may also require the manufacturer to cease U.S. marketing and/or recall any distributed units of the modified device until 510(k) clearance or a PMA for the modification is obtained.
22
De Novo Classification
If a previously unclassified new medical device does not qualify for the 510(k) pre-market notification process because no predicate device to which it is substantially equivalent can be identified, the device is automatically classified into Class III. However, if such a device would be considered low or moderate risk (in other words, it does not rise to the level of requiring a PMA), it may be eligible for the De Novo classification process. The De Novo classification process allows a device developer to request that the novel medical device be reclassified as either a Class I or Class II device, rather than having it regulated as a high-risk Class III device subject to the PMA requirements. If the manufacturer seeks reclassification into Class II, the classification request must include a draft proposal for special controls that are necessary to provide a reasonable assurance of the safety and effectiveness of the medical device.
Under the FDCA, the FDA is required to classify a device within 120 days following receipt of the De Novo classification request from an applicant; however, the most recent FDA performance review goals for fiscal years 2023 through 2027, FDA will attempt to issue a decision within 150 days of receipt on 70% of De Novo requests received during such period. De Novo classification requests are subject to user fees, unless a specific exemption applies.
As with the 510(k) pre-market notification process described above, any modification to a device authorized through the De Novo process that could significantly affect the safety or effectiveness of such device, or that would constitute a major change in its intended use, requires a new 510(k) clearance or could require the submission of a PMA application.
As an alternative to the De Novo classification process, a company could also file a reclassification petition seeking to change the automatic Class III designation of a novel post-amendment device under Section 513(f)(3) of the FDCA. FDA can also initiate reclassification of an existing device type on its own initiative. In December 2018, FDA issued a final rule to clarify the administrative process through which the FDA reclassifies a medical device. To reclassify a device under Section 513(e) of the FDCA, the FDA must first publish a proposed reclassification order that includes a summary of the valid scientific evidence that supports the reclassification; convene a device classification panel meeting; and consider comments to the public docket before it then publishes a final reclassification order in the Federal Register.
Pre-market Approval Pathway
Our point-of-care MRI systems have been classified and are regulated as Class II devices, although future products that we develop may be classified as Class III devices. Products classified by FDA as Class III generally require marketing approval via the PMA process. A PMA application must be supported by valid scientific evidence, which typically requires extensive data, including technical, nonclinical, clinical, manufacturing and labeling data, to demonstrate to the FDA’s satisfaction the safety and efficacy of the device for its intended use(s). A PMA application also must include a complete description of the device and its components, a detailed description of the methods, facilities and controls used to manufacture the device, and proposed labeling. After a PMA application is submitted and found to be sufficiently complete, it is considered “filed” and the FDA begins an in-depth review of the submitted information. During this substantive review period, the FDA may request additional information or clarification of information already provided. Also, during the review period, an advisory panel of experts from outside the FDA may be convened to review and evaluate the application and provide recommendations to the FDA. In addition, the FDA generally will conduct a pre-approval inspection of the manufacturing facility to evaluate compliance with the QSR, which requires manufacturers to implement and follow design, testing, control, documentation and other quality assurance procedures.
FDA review of a PMA application is required to be completed within 180 days of the application’s filing date although the process generally takes between one and three years, but may take significantly longer. The FDA can delay, limit or deny approval of a PMA application for many reasons, including:
If an FDA evaluation of a PMA application or manufacturing facilities is favorable, the FDA will either issue an approval letter, which authorizes commercial marketing of the device for specific indications for use, or an approvable letter, which usually contains a number of conditions which must be met in order to secure final approval of the PMA application.
23
When and if the conditions of the approvable letter have been fulfilled to the satisfaction of the FDA, the agency will issue a PMA approval letter authorizing commercial marketing of a device, subject to the conditions of approval and the limitations established in the approval letter. If the FDA’s evaluation of a PMA application or manufacturing facilities is not favorable, the FDA will deny approval of the PMA or issue a not approvable letter. The FDA may also determine that additional clinical trials are necessary, in which case the PMA approval may be delayed for several months or years while the trials are conducted, and data is submitted in an amendment to the PMA. The PMA process can be expensive, uncertain and lengthy. PMA approval may also be granted with post-approval requirements such as the need for additional clinical trials or patient follow-up for an indefinite period of time.
New PMA applications or PMA supplements may be required for modifications to the manufacturing process, labeling, device specifications, materials or design of a device that is approved through the PMA process. PMA supplements often require submission of the same type of information as an initial PMA application, except that the supplement is limited to information needed to support any changes from the device covered by the approved PMA application and may or may not require as extensive clinical data or the convening of an advisory panel.
Clinical Investigations Using Devices in Development
Clinical trials are almost always required to support a PMA application and are sometimes required for a De Novo classification request or 510(k) pre-market notification. In order to conduct a clinical investigation involving human subjects for the purpose of demonstrating the safety and effectiveness of a medical device, an investigator acting on behalf of the company must, among other things, apply for and obtain institutional review board (“IRB”) approval of the proposed investigation. In addition, if the clinical trial involves a “significant risk” (as defined by the FDA) to human health, the company sponsoring the trial (referred to as the “sponsor”) must also submit and obtain FDA approval of an investigational device exemption (“IDE”) application. An IDE application must be supported by appropriate data, such as animal and laboratory testing results, showing that it is safe to test the device in humans and that the testing protocol is scientifically sound. The IDE application must be approved in advance by the FDA for a specified number of trial participants, unless the product is deemed a non-significant risk device and eligible for abbreviated IDE requirements. Generally, clinical trials for a significant risk device may begin once the IDE application is approved by the FDA and the trial protocol and informed consent are approved by a duly-appointed IRB for each site. FDA’s IDE regulations govern investigational device labeling, prohibit promotion, and specify an array of good clinical practice (“GCP”) requirements, which include, among other things, recordkeeping, reporting and monitoring responsibilities of trial sponsors and study investigators. Clinical trials must further comply with the FDA’s regulations for IRB approval and for informed consent and other human subject protections, all of which are considered part of GCP requirements. Required records and reports are subject to inspection by the FDA. The results of clinical testing may be unfavorable or, even if the intended safety and efficacy success criteria are achieved, may not be considered sufficient for the FDA to grant approval or clearance of a product.
Information about certain clinical trials, including details of the protocol and eventually results, also must be submitted within specific timeframes to the National Institutes of Health, or NIH, for public dissemination on the ClinicalTrials.gov data registry. Information related to the product, patient population, phase of investigation, trial sites and investigators and other aspects of the clinical trial is made public as part of the registration process. Sponsors are obligated to disclose the results of their clinical trials after completion. Disclosure of results can be delayed in some cases for up to two years after the date of completion of the trial. Failure to timely register a covered clinical trial or to submit results as provided for in the law can give rise to civil monetary penalties and also prevent the non-compliant party from receiving future grant funds from the federal government. The NIH Final Rule on ClinicalTrials.gov registration and reporting requirements became effective in 2017, and both NIH and FDA have brought enforcement actions against non-compliant sponsors.
The commencement or completion of any of our clinical trials may be delayed or halted, or be inadequate to support approval of a PMA application (or FDA’s grant of a De Novo classification request or clearance of a 510(k) notification, as applicable), for numerous reasons, including, but not limited to, the following:
24
In the Consolidated Appropriations Act for 2023, Congress amended the FDCA to require the sponsor of any pivotal clinical trial to support marketing authorization of a medical device to develop a diversity action plan for such trial, and if submission of an IDE application is required, to submit such diversity action plan to the FDA. The action plan must include the sponsor’s diversity goals for enrollment, as well as a rationale for the goals and a description of how the sponsor will meet them. The FDA may grant a waiver for some or all of the requirements for a diversity action plan. It is unknown at this time how the diversity action plan may affect device clinical trial planning and timing or what specific information FDA will expect in such plans, but if FDA objects to a sponsor’s diversity action plan and requires the sponsor to amend the plan or take other actions, it may delay trial initiation.
Ongoing Post-Market Regulatory Requirements and FDA Enforcement
In 2020, Legacy Hyperfine received 510(k) clearance from the FDA for its portable MRI system. In February and October 2023, the Company received additional 510(k) clearances from the FDA for the latest updates to its Swoop® system AI-powered software. The combination of these two software updates significantly improved diffusion-weighted imaging (“DWI”), incorporated deep-learning based denoising in the post-processing of DWI images for crisper images, and improved image quality for all Swoop® system sequences. The Swoop® system has received marketing authorization for brain imaging in several countries, including the European Union (CE), the United Kingdom UKCA, Canada, Australia and New Zealand. All of the Company’s revenue to date has been generated from sales of the Swoop® system and related services.
After a medical device is authorized for marketing and placed in commercial distribution (or, for 510(k)-exempt products, placed into commerce without first obtaining FDA clearance or approval), numerous regulatory requirements apply. These general controls that must be met for all device classes include:
25
FDA’s MDR requirements also extend to healthcare facilities that use medical devices in providing care to patients, or “device user facilities,” which include hospitals, ambulatory surgical facilities, nursing homes, outpatient diagnostic facilities, or outpatient treatment facilities, but not physician offices. A device user facility must report any device-related death to both the FDA and the device manufacturer, or any device-related serious injury to the manufacturer (or, if the manufacturer is unknown, to the FDA) within 10 days of the event. Device user facilities are not required to report device malfunctions that would likely cause or contribute to death or serious injury if the malfunction were to recur but may voluntarily report such malfunctions through MedWatch, the FDA’s Safety Information and Adverse Event Reporting Program.
To ensure compliance with regulatory requirements, medical device manufacturers are subject to market surveillance and periodic, pre-scheduled and unannounced inspections by the FDA and certain state authorities. Failure to comply with applicable regulatory requirements can result in enforcement action by the FDA, which may lead to any of the following sanctions:
We and any of our contract manufacturers, and some suppliers of components or device accessories, are required to manufacture medical device products in compliance with current good manufacturing practice requirements set forth in the QSR, unless explicitly exempted by regulation. The QSR requires a quality system for the design, manufacture, packaging, labeling, storage, installation and servicing of marketed devices, and includes extensive requirements with respect to quality management and organization, device design, buildings, equipment, purchase and handling of components or services, production and process controls, packaging and labeling controls, device evaluation, distribution, installation, complaint handling, servicing, and record keeping. The FDA evaluates compliance with the QSR through periodic pre-scheduled or unannounced inspections that may include registered manufacturing facilities of our manufacturer. Following such inspections, FDA may issue reports known as Forms FDA 483 or Notices of Inspectional Observations, which list instances where the FDA inspector believes the manufacturer has failed to comply with applicable regulations and/or procedures. If the observations are sufficiently serious or the manufacturer fails to respond appropriately, the FDA may issue Warning Letters, which are notices of intended enforcement actions against the manufacturer. For less serious violations that may not rise to the level of regulatory significance, FDA may issue Untitled Letters. FDA may take more significant administrative or legal action if a manufacturer continues to be in substantial noncompliance with applicable regulations.
For example, if the FDA believes we or any of our contract manufacturers or regulated suppliers are not in compliance with these requirements and patients are being subjected to serious risks, it can shut down manufacturing operations, require recalls of our medical device products, refuse to approve new marketing applications, initiate legal proceedings to detain or seize products, enjoin future violations, or assess civil and criminal penalties against us or our officers or other employees. Any such action by the FDA would have a material adverse effect on our business. We may be unable to comply with all applicable FDA regulations.
U.S. Fraud and Abuse Laws and Other Compliance Requirements
Successfully commercializing a medical device or technology depends not on only FDA approval, but also on broad health insurance or third-party payor coverage. Government and private payors institute coverage criteria to ensure the appropriate utilization of
26
products and services and to control costs. Limited third-party payor coverage for a technology or procedure may limit adoption and commercial viability, while broader coverage supports optimal market uptake. Favorable coverage decisions by government payors like Medicare or Medicaid is critical because private payors typically follow the government’s lead regarding reimbursement. However, manufacturers whose technology is reimbursed by government payors are subject to various U.S. federal and state laws pertaining to healthcare fraud and abuse. These laws can be implicated by inappropriate sales and marketing arrangements with healthcare providers. Many commonly accepted commercial practices are illegal in the healthcare industry and violations of these laws are punishable by criminal and civil sanctions, including, in some instances, exclusion from participation in U.S. federal and state healthcare programs, including Medicare and Medicaid.
Anti-kickback Laws. The federal Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, receiving, offering or providing remuneration directly or indirectly to induce either the referral of an individual, or the furnishing, recommending, or arranging of a good or service, for which payment may be made under a government healthcare program such as Medicare and Medicaid. The definition of “remuneration” has been broadly interpreted to include anything of value, including such items as gifts, discounts, the furnishing of supplies or equipment, credit arrangements, waiver of payments, and providing anything at less than its fair market value. The Office of the Inspector General for the U.S. Department of Health and Human Services, has issued regulations, commonly known as safe harbors, which set forth certain provisions that, if satisfied in their entirety, will assure healthcare providers and other parties that they will not be prosecuted under the federal Anti-Kickback Statute. The failure of a transaction or arrangement to fit precisely within one or more safe harbors does not necessarily mean that it is illegal or that prosecution will be pursued. However, conduct and business arrangements that do not fully satisfy each applicable safe harbor element may result in increased scrutiny by government enforcement authorities or invite litigation by private citizens under federal whistleblower laws. The Anti-Kickback law is broadly interpreted and aggressively enforced with the result that beneficial commercial arrangements can be criminalized in the healthcare industry because of the Anti-Kickback law.
The penalties for violating the federal Anti-Kickback Statute include imprisonment for up to 10 years, fines of up to $100,000 per violation and possible exclusion from government healthcare programs such as Medicare and Medicaid. Many states have adopted prohibitions similar to the federal Anti-Kickback Statute, some of which apply to the referral of patients for healthcare services reimbursed by any source, not only by the government programs such as Medicare and Medicaid.
Federal False Claims Act. The federal False Claims Act prohibits knowingly presenting, or causing to be presented, a false or fraudulent claim for payment or approval by a federal healthcare program, knowingly making or using, or causing to be made or used, a false statement or record to obtain payment from the federal government, or avoiding, decreasing or concealing an obligation to pay money to the federal government. When an entity is determined to have violated the False Claims Act, it must pay three times the actual damages sustained by the government, plus mandatory per-claim civil penalties and attorneys’ fees. Violation of the False Claims Act also can result in exclusion from government healthcare programs.. Suits filed under the False Claims Act, known as “qui tam” actions, can be brought by any individual on behalf of the government and such individuals (known as “relators” or, more commonly, as “whistleblowers”) may share in any amounts paid by the entity to the government in fines or settlement. In addition, certain states have enacted laws modeled after the federal False Claims Act. Qui tam actions have increased significantly in recent years, causing greater numbers of healthcare companies to have to defend a false claim action, even before the validity of the claim is established and even if the government decides not to intervene in the lawsuit. Healthcare companies may decide to agree to large settlements with the government and/or whistleblowers to avoid the cost and negative publicity associated with litigation. In addition, the Patient Protection and Affordable Care Act of 2010 amended federal law to provide that a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act. Criminal prosecution is possible for knowingly making or presenting a false or fictitious or fraudulent claim to the federal government.
Various states have enacted false claims laws analogous to the False Claims Act. Some of these state laws apply where a claim is submitted to any commercial payor, and not merely a government healthcare program, and some authorize private citizens to bring suit on the state’s behalf.
Federal Physician Self-Referral Law. The Federal Physician Self-Referral Law, also referred to as the Stark Law, prohibits a physician (or an immediate family member of a physician) who has a financial relationship with an entity from referring patients to that entity for certain designated health services, including durable medical equipment and supplies, payable by Medicare, unless an exception applies. The Stark Law also prohibits such an entity from presenting or causing to be presented a claim to the Medicare program for such designated health services provided pursuant to a prohibited referral, and provides that certain collections related to any such claims must be refunded in a timely manner. Exceptions to the Stark Law include, among other things, exceptions for certain financial relationships, including both ownership and compensation arrangements. The Stark Law is a strict liability statute: to the extent that the statute is implicated and an exception does not apply, the statute is violated. In addition to the Stark Law, many states have implemented similar physician self-referral prohibitions that may extend to Medicaid, third-party payors, and self-pay patients. Violations of the Stark Law must be reported and unauthorized claims must be refunded to Medicare in order to avoid potential liability under the federal False Claims Act for avoiding a known obligation to return identified overpayments. Violations of the Stark Law can also form the basis for exclusion from participation in federal and state healthcare programs.
27
Civil Monetary Penalties Law. The Civil Monetary Penalties Law (“CMPL”) authorizes the imposition of substantial civil money penalties against an entity that engages in certain prohibited activities including but not limited to violations of the Stark Law or Anti-Kickback Statute, knowing submission of a false or fraudulent claim, employment of an excluded individual, and the provision or offer of anything of value to a Medicare or Medicaid beneficiary that the transferring party knows or should know is likely to influence beneficiary selection of a particular provider for which payment may be made in whole or part by a government healthcare program, unless an exception applies. Sanctions for violations of the CMPL include exclusion from participation in government healthcare programs, substantial fines, and payment of up to three times the amount billed, depending upon the nature of the offense.
State Analogs of Federal Fraud and Abuse Laws. Many U.S. states have their own laws intended to protect against fraud and abuse in the healthcare industry and more broadly. In some cases these laws prohibit or regulate additional conduct beyond what federal law affects. Penalties for violating these laws can range from substantial fines to criminal sanctions.
HIPAA. The Health Insurance Portability and Accountability Act of 1996 and implementing regulations (“HIPAA”), created two new federal crimes: healthcare fraud and false statements relating to healthcare matters. The healthcare fraud statute prohibits knowingly and willfully executing a scheme to defraud any healthcare benefit program, including private payors. A violation of this statute is a felony and may result in fines, imprisonment or exclusion from government healthcare programs. The false statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services.
FCPA and Other Anti-Bribery and Anti-Corruption Laws. The U.S. Foreign Corrupt Practices Act (“FCPA”) prohibits U.S. corporations and their representatives from offering, promising, authorizing or making payments to any foreign government official, government staff member, political party or political candidate in an attempt to obtain or retain business abroad. The scope of the FCPA would include interactions with certain healthcare professionals or organizations in many countries. Our present and future business has been and will continue to be subject to various other U.S. and foreign laws, rules and/or regulations.
Physician Payments Sunshine Act. Manufacturers of U.S. FDA-regulated devices reimbursable by government healthcare programs are subject to the Physician Payments Sunshine Act, which requires manufacturers to track and annually report certain payments and other transfers of value made to U.S.-licensed physicians, certain advanced non-physician healthcare practitioners, or U.S. teaching hospitals. As a manufacturer of U.S. FDA-regulated devices reimbursable by government healthcare programs, we are subject to this law. We are also required to report certain ownership interests held by physicians and their immediate family members. The law authorizes significant monetary penalties for violations, depending on the circumstances, and payments reported also have the potential to draw scrutiny on payments to and relationships with physicians, which may have implications under the Anti-Kickback Statute, Stark Law and other healthcare laws.
Similar to the federal law, certain states also have adopted marketing and/or transparency laws relevant to device manufacturers, some of which are broader in scope. Some state laws require medical device companies to comply with the relevant industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government in addition to requiring device manufacturers to report information related to payments to physicians and other healthcare providers or marketing expenditures. The need to build and maintain a robust compliance program with different compliance and/or reporting requirements increases the possibility that a healthcare company may violate one or more of the requirements, resulting in fines and penalties.
U.S. and European Data Security and Data Privacy Laws
HIPAA’s administrative simplification provisions established comprehensive federal standards for the privacy and security of health information. In 2009, Congress enacted Subtitle D of the Health Information Technology for Economic and Clinical Health Act, or HITECH, provisions of the American Recovery and Reinvestment Act of 2009, which expanded and strengthened HIPAA, created new targets for enforcement, imposed new penalties for noncompliance and established new breach notification requirements. HIPAA applies to health plans, healthcare clearing houses, and healthcare providers that conduct certain healthcare transactions electronically, which are referred to collectively as Covered Entities, as well as individuals or entities that perform services for Covered Entities involving the use, or disclosure of, individually identifiable health information or protected health information (“PHI”) under HIPAA. Such service providers are called “Business Associates.” Under HIPAA, as amended by the HITECH Act, HHS has issued regulations to protect the privacy and security of PHI used or disclosed by Covered Entities and Business Associates. HIPAA also regulates and standardizes the codes, formats and identifiers used in certain healthcare transactions and standardization of identifiers for health plans and providers, for example insurance billing. We are a Business Associate of our Covered Entity Customers in connection with the use of data and images to train AI algorithms as well as the provision of product maintenance and support services. Accordingly, we execute and must comply with HIPAA Business Associate Agreements and with HIPAA regulations applicable to Business Associates. Any non-compliance with HIPAA and HITECH and related penalties, could adversely impact our business.
The HIPAA security standards require the adoption of administrative, physical, and technical safeguards and the adoption of written security policies and procedures to maintain the security of protected health information.
28
The HIPAA privacy regulations address the privacy of PHI by limiting the use and release of such information. They also set forth certain rights that an individual has with respect to his or her PHI maintained by a covered entity, including the right to access or amend certain records containing PHI, request an accounting of disclosures of PHI or to request restrictions on the use or disclosure of PHI. Proposed modifications to the privacy regulations were published in January 2021 with a public comment period that ended in May 2021. The content of the final rules has not yet been issued but may result in changes to HIPAA that could impact us and our business. The HIPAA breach notification regulations impose certain reporting requirements on Covered Entities and their Business Associates in the event of a breach of PHI.
Significant civil and criminal fines and other penalties may be imposed for violating HIPAA directly, and in connection with acts or omissions of any agents, including a downstream Business Associate, as determined according to the federal common law of agency. Civil penalties are adjusted for inflation on an annual basis and can exceed one million dollars per year for failure to comply with a HIPAA requirement. A single breach incident can violate multiple requirements. Additionally, a person who knowingly obtains or discloses PHI in violation of HIPAA may face criminal penalties (including fines and imprisonment), which increase if the wrongful conduct involves false pretenses or the intent to sell, transfer or use PHI for commercial advantage, personal gain or malicious harm. Covered Entities are also subject to enforcement by state Attorneys General who were given authority to enforce HIPAA.
Additionally, while HIPAA does not create a private right of action allowing individuals to file suit against us in civil court for violations of HIPAA, its standards have been used as the basis for duty of care cases in state civil suits such as those for negligence or recklessness in the misuse or breach of PHI.
Even when HIPAA does not apply, according to the FTC, failing to take appropriate steps to keep consumers’ personal information secure could constitute unfair acts or practices in or affecting commerce in violation of Section 5(a) of the Federal Trade Commission Act. The FTC expects a company’s data security measures to be reasonable and appropriate in light of the sensitivity and volume of consumer information it holds, the size and complexity of its business, and the cost of available tools to improve security and reduce vulnerabilities. Individually identifiable health information is considered sensitive data that merits stronger safeguards. The FTC and states' Attorneys General have also brought enforcement actions and prosecuted some data breach cases as unfair and/or deceptive acts or practices under the FTC Act and comparable state laws.
The HIPAA privacy and security regulations establish a uniform federal "floor" and do not preempt state laws that are more stringent or provide individuals with greater rights with respect to the privacy or security of, and access to, their records containing PHI. These laws overlap with HIPAA and may differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. Failure to comply with these laws, where applicable, can result in the imposition of significant civil and/or criminal penalties and private litigation. The State of California, for example, has implemented comprehensive laws and regulations concerning health information and other personally identifiable information. The California Confidentiality of Medical Information Act (CMIA), imposes restrictive requirements regulating the use and disclosure of health information and other personally identifiable information. Separately, the California Consumer Privacy Act of 2018 (the CCPA) went into effect January 1, 2020. The CCPA, among other things, creates new data privacy obligations for covered businesses and provides new privacy rights to California residents, including the right to opt out of certain disclosures of their information. It also creates individual privacy rights for California consumers and increases the privacy and security obligations of entities handling certain personal information. The CCPA provides for civil penalties for violations, as well as a private right of action for data breaches, which may result in increased privacy and information security enforcement in California. Although the law includes limited exceptions, including for PHI maintained by a covered entity or business associate under HIPAA and medical information maintained by healthcare providers under the CMIA, it may regulate or impact our processing of personal information depending on the context. Further, the California Privacy Rights Act (CPRA) went into effect January 1, 2023, amending the CCPA. The CPRA imposes additional data protection obligations on covered businesses, including additional consumer rights processes, limitations on data uses, new audit requirements for higher risk data, and opt outs for certain uses of sensitive data and expands the application of the CCPA to personal information of our California-based employees. It also created a new regulatory entity, the California Privacy Protection Agency, which is authorized to issue substantive regulations under the CPRA and is expected to result in increased privacy and information security enforcement. Other states have implemented similar laws protecting identifiable health and personal information, and such laws may not be preempted by HIPAA, thus complicating compliance efforts. As of 2023, comprehensive privacy laws were in effect in California, Virginia, Colorado, Connecticut, and Utah. Additional consumer privacy laws have also been enacted in Delaware, Indiana, Iowa, Montana, New Jersey, Oregon, Tennessee, and Texas, which laws will take effect over the next three years.
In dealing with health information for the development of our technology or for commercial purposes, we are directly or indirectly affected by HIPAA and state-imposed health information privacy and security laws because these laws regulate the ability of our customers and research collaborators to share health information with us. Additionally, we must identify and comply with all applicable state laws for the protection of personal information with respect to employee information or other personal information that we collect.
29
In the European Union, increasingly stringent data protection and privacy rules have and will continue to have substantial impact on the use of personal and patient data across the healthcare industry. The EU General Data Protection Regulation, (“GDPR”), which took effect in May 2018, applies across the European Union and European Economic Area (“EEA”) and includes, among other things, a requirement for prompt notice of data breaches to data subjects and supervisory authorities in certain circumstances and significant fines for non-compliance. The GDPR establishes fines and penalties that reach up to 20 million euros, or up to 4% of total global turnover of the preceding fiscal year, whichever is higher. The GDPR sets out a number of requirements that must be complied with when handling the personal data of such EEA-based data subjects including: providing expanded disclosures about how their personal data will be used; higher standards for organizations to demonstrate that they have obtained valid consent or have another legal basis in place to justify their data processing activities; the obligation to appoint data protection officers in certain circumstances; new rights for individuals to be “forgotten” and rights to data portability, as well as enhanced current rights (e.g. access requests); the principal of accountability and demonstrating compliance through policies, procedures, training and audits; and a mandatory data breach regime. In particular, medical or health data, genetic data and biometric data where the latter is used to uniquely identify an individual are all classified as “special category” data under the GDPR and are afforded greater protection and require additional compliance measures. Noncompliance could result in the imposition of fines, penalties, data lockup or orders to stop noncompliant activities. We may be subject to the GDPR if we undertake operations in the EEA, offer products or services to individuals in the EU or monitor the behavior of individuals within the EEA. Our research activities in the EU currently implicate the GDPR and if we undertake commercial operations in the EU, offer products or services to individuals in the EEA or monitor the behavior of individuals within the EU, we will have additional compliance obligations.
We could be subject to evolving European Union laws on data export, for transfers of data outside the EEA to themselves, group companies or third parties. The GDPR only permits exports of data outside the EEA to jurisdictions that ensure an adequate level of data protection. On July 16, 2020, the Court of Justice of the European Union or the CJEU, issued a landmark opinion in the case Maximilian Schrems vs. Facebook (Case C-311/18), called Schrems II. This decision (a) called into question commonly relied upon data transfer mechanisms as between the European Union member states and the United States (such as the Standard Contractual Clauses) and (b) invalidated the EU-U.S. Privacy Shield on which many companies had relied as an acceptable mechanism for transferring such data from the EEA to the United States. However, on July 10, 2023, the European Commission adopted an adequacy decision for a new mechanism for transferring data from the EU to the United States – the EU-U.S. Data Privacy Framework, which provides EEA individuals with several new rights, including the right to obtain access to their data, or obtain correction or deletion of incorrect or unlawfully handled data. The adequacy decision followed the signing of an executive order introducing new binding safeguards to address the points raised in the Schrems II decision by the CJEU. The European Commission will continually review developments in the United States along with its adequacy decision. Adequacy decisions can be adapted or even withdrawn in the event of developments affecting the level of protection in the applicable jurisdiction. Future actions of data protection authorities in the EEA are difficult to predict. Some customers or other service providers may respond to these evolving laws and regulations by asking us to make certain privacy or data-related contractual commitments that we are unable or unwilling to make. This could lead to the loss of current or prospective customers or other business relationships.
Relatedly, following the United Kingdom’s withdrawal from the European Union, the GDPR has been implemented in the United Kingdom (and referred to as the UK GDPR). The UK GDPR sits alongside the UK Data Protection Act 2018, which implements certain derogations in the EU GDPR into United Kingdom law. Under the UK GDPR, companies not established in the United Kingdom but which process personal data in relation to the offering of goods or services to individuals in the United Kingdom, or monitor the behavior of individuals in the United Kingdom will be subject to the UK GDPR – the requirements of which are (at this time) largely aligned with those under the EU GDPR and as such, may lead to similar compliance and operational costs with potential fines of up to 17.5 million pounds sterling or 4% of global turnover, whichever is higher. In June 2021, the European Commission issued a decision, which will sunset on June 27, 2025 without further action, that the United Kingdom ensures an adequate level of protection for personal data transferred under the EU GDPR from the European Union to the United Kingdom. The U.K. Parliament continues to consider the Data Protection and Digital Information Bill to harmonize the 2018 Data Protection Act, U.K. GDPR, and the Privacy and Electronic Communications Regulations under one legislative framework.
International Regulation of Medical Devices
International marketing and distribution of medical devices are subject to regulation by foreign governments, and such regulations may vary substantially from country to country. The time required to obtain marketing authorization in a foreign country may be longer or shorter than that required for FDA clearance or approval, and the requirements may differ. There is a trend towards harmonization of quality system standards among the European Union, United States, Canada and various other industrialized countries. For example, on February 2, 2024, the U.S. FDA issued a Final Rule describing amendments to harmonize the QSR with ISO 13485:2016, which will become effective on February 2, 2026.
The primary regulatory environment in Europe is that of the European Economic Area (the “EEA”), which is comprised of the 27 Member States of the European Union (the “EU”), Iceland, Liechtenstein and Norway. In the EEA, medical devices were previously required to comply with the Essential Requirements defined in Annex I to the EU Medical Devices Directive (“MDD”) (applicable in
30
the non-EU EEA Member States via the Agreement on the European Economic Area), a coordinated system for the authorization of medical devices. The directives and standards outlined in the MDD regulate the design, manufacture, clinical trials, labeling and adverse event reporting for medical devices. Devices that comply with the requirements of a relevant directive are entitled to bear the CE conformity marking, indicating that the device conforms to the essential requirements of the applicable directives and, accordingly, can be commercially distributed throughout the EEA. The method of assessing conformity varies depending on the class of the product, but for most medical devices involves a combination of self-assessment by the manufacturer and a third-party assessment by a “Notified Body,” an organization designated by an EU country to assess a product’s conformity with the applicable legal requirements. This third-party assessment may consist of an audit of the manufacturer’s quality system and specific testing of the manufacturer’s product. An assessment by a Notified Body of one country within the EU is required in order for a manufacturer to commercially distribute the product throughout the EU.
In 2017, European Union regulatory bodies finalized a new Medical Device Regulation, which replaced the existing MDD framework and became effective on May 26, 2021. The Medical Device Regulation changes several aspects of the existing regulatory framework for medical device marketing in Europe in order to increase regulatory oversight of all medical devices marketed in the EU, which may, in turn, increase the costs, time and requirements that need to be met in order to place an innovative or high-risk medical device on the European market. In particular, the new regulations, among other things:
To demonstrate compliance with the General Safety and Performance Requirements (“GSPRs”), set forth in the Medical Device Regulation, device manufacturers must undergo a conformity assessment procedure, which varies according to the type of device and its risk classification. Conformity assessment procedures require an assessment of the technical documentation, including the device description, the design stages, the manufacturing process, available clinical evidence, literature data for the product, and post-market experience in respect of similar products already marketed. Except for low-risk medical devices (Class I non-sterile, non-measuring devices), where the manufacturer can self-declare the conformity of its products with the GSPRs (except for any parts which relate to sterility or metrology), the conformity assessment must be completed by a Notified Body. To that effect, device manufacturers must apply to a Notified Body to request a conformity assessment. Notified Bodies are independent organizations designated by EU member states to assess the conformity of devices before being placed on the market. A Notified Body would typically audit and examine a product’s technical dossiers and the manufacturer’s quality management system (which must, in particular, comply with ISO 13485). As a general rule, demonstration of a device’s conformity with the GSPRs must be based, among other things, on the evaluation of clinical data supporting the safety and performance of the device during normal conditions of use. If satisfied that the device conforms to the relevant GSPRs, the Notified Body issues a certificate of conformity, which the manufacturer uses as a basis for its own declaration of conformity. The manufacturer may then apply the CE-Mark to the device, allowing the device to be legally marketed throughout the EU. Notified Body certificates of conformity are valid for a fixed duration (to a maximum of five years).
Throughout the term of the certificate, the manufacturer is subject to periodic surveillance audits to verify continued compliance with applicable requirements. In particular, a new audit by the Notified Body is required before the manufacturer renews the relevant certificate.
Devices lawfully placed on the market pursuant to the Medical Device Directive prior to May 26, 2021 could initially continue to be made available on the market or put into service until May 26, 2025. Nevertheless, the European Parliament adopted legislation to extend this transitional period to give manufacturers more time to switch from the previously applicable provisions to the new certification requirements in the Medical Device Regulation. For high risk, class III and class IIb implantable devices the transitional period is extended until December 31, 2027. For medium and low risk, class IIb devices and class IIa, Im, Is and Ir devices the transition period is extended until December 31, 2028.
Outside of the EU, regulatory authorization must be sought on a country-by-country basis in order for the company to market their products. Some countries have adopted medical device regulatory regimes, such as the Classification Rules for Medical Devices published by the Hong Kong Department of Health, the Health Sciences Authority of Singapore regulation of medical devices under
31
the Health Products Act, and Health Canada’s risk classification system for invasive devices, among others. Each country may have its own processes and requirements for medical device licensing, approval/clearance, and regulation, therefore requiring the company to seek marketing authorizations on a country-by-country basis.
In addition, as previously noted, the UK left the EU on January 31, 2020 (referred to as “Brexit”), with a transitional period that expired on December 31, 2020. The United Kingdom and the European Union entered into a trade agreement known as the Trade and Cooperation Agreement (“TCA”), which came into effect on January 1, 2021. The TCA does not specifically refer to medical devices. However, as a result of Brexit, the Medical Device Regulation will not be implemented in the UK, and previous legislation that mirrored the Medical Device Regulation in the UK law has been revoked. The regulatory regime for medical devices in the UK will continue to be based on the requirements derived from EU legislation, and the UK may choose to retain regulatory flexibility or align with the Medical Device Regulation going forward. CE markings will continue to be recognized in the UK, and certificates issued by EU recognized Notified Bodies will be valid in the UK, until the earlier of June 30, 2028 or the expiration of the certificate for devices compliant with the Medical Device Directive or until June 30, 2030 for devices compliant with the Medical Device Regulation. For medical devices placed on the UK market after this period, the UK Conformity Assessed (“UKCA”) marking will be mandatory. In contrast, UKCA marking and certificates issued by UK Notified Bodies will not be recognized on the EU market. The TCA does provide for cooperation and exchange of information in the area of product safety and compliance, including market surveillance, enforcement activities and measures, standardization related activities, exchanges of officials, and coordinated product recalls (or other similar actions). Depending on which countries products will ultimately be sold in, manufacturers may start seeking alternative sources for components if this would allow them to benefit from no tariffs. The rules for placing medical devices on the Northern Ireland market will differ from those in the UK.
In addition, outside the United States, a range of anti-bribery and anti-corruption laws, as well as some industry-specific laws and codes of conduct, apply to the medical device industry and interactions with government officials and entities and healthcare professionals. Such laws include, but are not limited to the UK Bribery Act of 2010. Further, the European Union member countries have emphasized a greater focus on healthcare fraud and abuse and have indicated greater attention to the industry by the European Anti-Fraud Office. Countries in Asia have also become more active in their enforcement of anti-bribery laws and with respect to procurement and supply chain fraud.
Corporate Information
HealthCor Catalio Acquisition Corp. (“HealthCor”) was incorporated as a Cayman Islands exempted company on November 18, 2020 for the purpose of effecting a merger, share exchange, asset acquisition, share purchase, reorganization or other similar business combination with one or more businesses. Legacy Hyperfine was incorporated under the laws of the State of Delaware on February 25, 2014 under the name “Hyperfine Research, Inc.” On May 25, 2021, the name of Legacy Hyperfine was changed to “Hyperfine, Inc.” Liminal was incorporated under the laws of the State of Delaware on September 21, 2018 under the name “EpilepsyCo Inc.” On July 20, 2020, the name of Liminal was changed to “Liminal Sciences, Inc.” On December 21, 2021, HealthCor changed its jurisdiction of incorporation from the Cayman Islands to the State of Delaware by deregistering as an exempted company in the Cayman Islands and continuing and domesticating as a corporation, incorporated under the laws of the State of Delaware. On December 22, 2021, HealthCor, Legacy Hyperfine and Liminal completed the business combination (the “Business Combination”), pursuant to which each of Legacy Hyperfine and Liminal became a wholly owned subsidiary of HealthCor, HealthCor’s corporate name was changed to Hyperfine, Inc., Legacy Hyperfine’s corporate name was changed to Hyperfine Operations, Inc., Liminal’s corporate name was changed to Liminal Operations, Inc. (which was subsequently changed to Liminal Sciences, Inc.), and the business of Legacy Hyperfine and Liminal became the business of the Company. In December 2022, the Company suspended the Liminal program to develop a device to non-invasively measure key vital signs in the brain. Our principal executive offices are located at 351 New Whitfield Street, Guilford, Connecticut 06437, and our telephone number is (866) 796-6767.
Information Available on the Internet
Our internet address is https://hyperfine.io, to which we regularly post copies of our press releases as well as additional information about us. Our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and all amendments to those reports, will be available to you free of charge through the Investors section of our website as soon as reasonably practicable after such materials have been electronically filed with, or furnished to, the Securities and Exchange Commission (the “SEC”). The SEC maintains an internet site (http://www.sec.gov) that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC. We include our web site address in this report only as an inactive textual reference. Information contained in our website does not constitute a part of this report or our other filings with the SEC.
32
Item 1A. RISK FACTORS
Except for the historical information contained herein, this report contains forward-looking statements that involve risks and uncertainties. These statements include projections about our finances, plans and objectives for the future, future operating and economic performance and other statements regarding future performance. These statements are not guarantees of future performance or events. Our actual results could differ materially from those discussed in this report. Factors that could cause or contribute to these differences include, but are not limited to, those discussed in the following section, as well as those discussed in Part II, Item 7 entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and elsewhere throughout this report.
You should consider carefully the following risk factors, together with all of the other information included in this report. If any of the following risks, either alone or taken together, or other risks not presently known to us or that we currently believe to not be significant, develop into actual events, then our business, financial condition, results of operations or prospects could be materially adversely affected. If that happens, the market price of our Class A common stock could decline, and stockholders may lose all or part of their investment.
Unless the context otherwise requires, references in this section to “we,” “us,” “our” and the “Company” refer to Hyperfine, Inc. and its subsidiaries following the Business Combination, or to Legacy Hyperfine, Liminal, or HealthCor prior to the Business Combination, as the case may be.
Risks Related to Our Financial Condition and Capital Requirements
We are an early-stage health technology company with a history of net losses, which we expect to continue, and we may not be able to generate meaningful revenues or achieve and sustain profitability in the future.
We are an early-stage health technology company, and have incurred significant losses since Legacy Hyperfine and Liminal formed in 2014 and 2018, respectively, and expect to continue to incur losses in the future. We incurred net losses of $44.2 million and $73.2 million for the years ended December 31, 2023 and 2022, respectively. As of December 31, 2023, we had an accumulated deficit of $253.7 million. These losses and accumulated deficit were primarily due to the substantial investments made to develop and improve our technology and products. Over the next several years, we expect to continue to devote substantially all of our resources towards continuing development and commercialization of our products and research and development efforts for additional products. These efforts may prove more costly than we currently anticipate. We are generating product revenue but may never generate revenue sufficient to offset our expenses. In addition, as a public company, we will continue to incur significant legal, accounting, administrative, insurance and other expenses. Accordingly, we cannot assure you that we will achieve profitability in the future or that, if we become profitable, will sustain profitability.
We have a limited operating history, which may make it difficult to evaluate the prospects for our future viability and predict our future performance. As such, you cannot rely upon our historical operating performance to make an investment decision regarding us.
We have generated limited revenue from the sale of our products and services to date and have incurred significant losses. Our prospects must be considered in light of the uncertainties, risks, expenses, and difficulties frequently encountered by companies in their early stages of operations. We have not yet achieved wide market acceptance for our products, produced our products at scale, refined our sales model, or conducted at scale sales and marketing activities necessary for successful mass product adoption. Consequently, predictions about our future success or viability are highly uncertain and may not be as accurate as they could be if we had a longer operating history or a company history of successfully developing and commercializing products.
In addition, as a business with a limited operating history, we may encounter unforeseen expenses, difficulties, complications, delays and other known and unknown obstacles. We will need to transition from a company in the early commercialization stage to large scale commercialization, and we may not be successful in such a transition. We have encountered in the past, and will encounter in the future, risks and uncertainties frequently experienced by growing companies with limited operating histories in emerging and rapidly changing industries. If our assumptions regarding these risks and uncertainties, which we will use to plan and operate our business, are incorrect or change, or if we do not address these risks successfully, our results of operations could differ materially from our expectations, and our business, financial condition and results of operations could be adversely affected.
33
We may need to raise additional capital to fund commercialization plans for our products, including manufacturing, sales and marketing activities, expand our investments in research and development, develop clinical evidence, and commercialize new products and applications.
Our operations have consumed substantial amounts of cash since inception. We expect to use our cash resources to develop and further commercialize our products, develop new products, and for working capital and general corporate purposes. We may require additional capital to further develop and commercialize our products and to develop new products. In addition, our operating plans may change as a result of many factors that may currently be unknown to us, and we may need to seek additional funds sooner than planned.
We cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable to us, if at all. Moreover, the terms of any future financing may adversely affect the holdings or the rights of our stockholders and the issuance of additional securities, whether equity or debt, by us, or the possibility of such issuance, may cause the market price of our Class A common stock to decline. The incurrence of indebtedness could result in increased fixed payment obligations, and we may be required to agree to certain restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. We could also be required to seek funds through arrangements with collaborative partners or otherwise at an earlier stage than otherwise would be desirable, and we may be required to relinquish rights to some of our technologies or products or otherwise agree to terms that are unfavorable to us, any of which may have a material adverse effect on our business, operating results and prospects. In addition, raising additional capital through the issuance of equity or convertible debt securities would cause dilution to holders of our equity securities, and may affect the rights of then-existing holders of our equity securities. Even if we believe that we have sufficient funds for our current or future operating plans, we may seek additional capital if market conditions are favorable or if we have specific strategic considerations.
SEC regulations limit the funds we can raise during 12 months under our shelf registration statement on Form S-3.
In November 2023, we filed a shelf registration statement on Form S-3 with the SEC pursuant to which we registered for sale up to $150 million of any combination of our Class A common stock, preferred stock, debt securities, warrants, rights and/or units from time to time and at prices and on terms that we may determine. Our shelf registration statement on Form S-3 also included a prospectus covering up to an aggregate of $50.0 million in shares of Class A common stock that we may issue and sell from time to time, through B. Riley acting as our sales agent, pursuant to the Sales Agreement for our “at-the-market” equity program. There can be no assurance that B. Riley will be successful in consummating future sales based on prevailing market conditions or in the quantities or at the prices that we deem appropriate. In addition, under current SEC regulations, as of the filing of this Annual Report on Form 10-K, our public float is less than $75 million, and under SEC regulations for so long as our public float remains less than $75 million, the amount we can raise through primary public offerings of securities in any twelve-month period using shelf registration statements is limited to an aggregate of one-third of our public float, which is referred to as the baby shelf rules. As of March 5, 2024, our public float was approximately $68.5 million, based on 54,760,324 shares of outstanding common stock held by non-affiliates and at a price of $1.25 per share, which was the last reported sale price of our common stock on the Nasdaq Capital Market on March 5, 2024. As a result of our public float being below $75 million, we will be limited by the baby shelf rules until such time our public float exceeds $75 million, which means we only have the capacity to sell shares up to one-third of our public float in primary offerings under our shelf registration statement on Form S-3 in any twelve-month period.
Our operating results may fluctuate significantly in the future, which makes our future operating results difficult to predict and could cause our operating results to fall below expectations or any guidance we may provide.
Our quarterly and annual operating results may fluctuate significantly, which makes it difficult for us to predict our future operating results. These fluctuations may occur due to a variety of factors, many of which are outside our control, including, but not limited to:
34
The cumulative effects of the factors discussed above could result in large fluctuations and unpredictability in our quarterly and annual operating results. As a result, comparing our operating results on a period-to-period basis may not be meaningful.
This variability and unpredictability could also result in us failing to meet the expectations of industry or financial analysts or investors for any period. If we are unable to further commercialize products or generate revenue, or if our operating results fall below the expectations of analysts or investors or below any guidance we may provide, or if the guidance we provide is below the expectations of analysts or investors, the market price of our Class A common stock could decline.
We maintain our cash at financial institutions, often in balances that exceed federally insured limits.
The majority of our cash is held in accounts at U.S. banking institutions that we believe are of high quality. Cash held in non-interest-bearing and interest-bearing operating accounts may exceed the Federal Deposit Insurance Corporation (“FDIC”) insurance limits. If such banking institutions were to fail, we could lose all or a portion of those amounts held in excess of such insurance limitations. While the FDIC took control of one such banking institution, Silicon Valley Bank (“SVB”), on March 10, 2023, and the FDIC also took control of Signature Bank (“Signature Bank”) on March 12, 2023, we did not have any accounts with SVB or Signature Bank and therefore did not experience any specific risk of loss. The FDIC also announced that account holders would be made whole. Thus, we do not view the risk as material to our financial condition. However, the risk of loss in excess of insurance limitations has generally increased. Any material loss that we may experience in the future could have an adverse effect on our ability to pay our operational expenses or make other payments and may require us to move our accounts to other banks, which could cause a temporary delay in making payments to our vendors and employees and cause other operational inconveniences.
Risks Related to Our Businesses
Our success depends upon market acceptance of our products and services, our ability to develop and commercialize existing and new products and services and generate revenues, and our ability to identify new markets for our technology.
We have developed, and are engaged in the development of, MRI solutions. We are commercializing our Swoop® system to address limitations of current imaging technologies. Our success will depend on the acceptance of our products and services in the United States. and international healthcare markets. The marketplace may not be receptive to our products and services over competing products, including conventional MRI systems used in hospitals, imaging centers and physicians’ offices, and we may be unable to compete effectively. Factors that could affect our ability to successfully further commercialize our current products and services and to commercialize any potential future products and services include:
We cannot assure investors that our current products and services or any future products and services will gain broad market acceptance. If the market for our current products and services or any future products and services fails to develop or develops more slowly than expected, or if any of our products or services do not achieve or sustain market acceptance, our business and operating results would be materially and adversely affected.
35
Health technology development is costly and involves continual technological change, which may render our current or future products obsolete.
The market for point-of-care health technology and medical devices is characterized by rapid technological change, medical advances and evolving industry standards. Any one of these factors could reduce the demand for our devices or services or require substantial resources and expenditures for research, design and development to avoid technological or market obsolescence.
Our success will depend on our ability to enhance our current technology, products and services, develop or acquire and market new technologies and to continue to improve our technology, products and services to meet customer needs. A failure to adequately develop or acquire technology that will address customer requirements adequately, or to introduce such improved products and services on a timely basis, may have a material adverse effect on our business, financial condition and results of operations.
We might have insufficient financial resources to improve existing devices, advance technologies and develop new products and services at competitive prices. Technological advances by one or more competitors or future entrants into the field may result in our current products and services becoming non-competitive or obsolete, which may decrease revenues and profits and adversely affect our business and results of operations.
We may encounter significant competition across our existing and future planned products and services and in each market in which we sell or plan to sell our products and services from various companies, many of which have greater financial and marketing resources than us. Our primary competitors include several large companies which currently dominate the medical imaging market, including General Electric, Siemens, Philips, Hologic, Fuji, Toshiba, Canon and Hitachi.
In addition, our competitors, some of which are well-established manufacturers with significant resources, may engage in aggressive marketing tactics. Competitors may also possess the ability to commercialize additional lines of products, bundle products or offer higher discounts and incentives to customers in order to gain a competitive advantage. If the prices of competing products are lowered as a result, we may not be able to compete effectively.
We will be dependent upon the success of our sales and customer acquisition and retention strategies.
Our business is dependent upon the success of our sales and customer acquisition and retention strategies, and our marketing efforts are focused on developing a strong reputation with healthcare providers and increasing awareness of our products and services. If we fail to maintain a high quality of service or a high quality of device technology, we may fail to retain existing customers or add new customers. If we do not successfully continue our sales efforts and promotional activities, particularly to health systems and large institutions, or if existing customers decrease their level of engagement, our revenue, financial results and business may be significantly harmed. Our future success depends upon continued expansion of our commercial operations in the United States and internationally, as well as entering additional markets to commercialize our products and services. We believe that our growth will depend on the further development and commercialization of our current products and services, regulatory authorization of our current products and services in additional markets, and development, regulatory authorization and commercialization of our future products and services. If we fail to expand the use of our products and services in a timely manner, we may not be able to expand our market share or to grow our revenue. Our financial performance will be substantially dictated by our success in increasing and maintaining the volume of use of our products by our customers. If customers do not perceive our products or services to be useful, reliable and trustworthy, we may not be able to attract or retain customers or otherwise maintain or increase the frequency and duration of their engagement. As our business model is predicated on device hardware sales, and service and support agreements for use of device hardware and services, there is risk that any decline in sales, service and support renewal rates will adversely impact our business. A decrease in customer retention, growth or engagement with our products and services may have a material and adverse impact on our revenue, business, financial condition and results of operations.
Any number of factors could negatively affect customer retention, growth and engagement, including:
36
We expect to generate an increasing portion of our revenue internationally in the future and may become subject to various additional risks relating to our international activities, which could adversely affect our business, operating results and financial condition.
Revenue from non-U.S. countries was 40% of total revenue for the year ended December 31, 2023. We believe that a substantial percentage of our future revenue will come from international sources as we continue to commercialize our products and services, having received marketing authorization for brain imaging in several countries, including the European Union (CE), the United Kingdom (UKCA), Canada, Australia and New Zealand, and as we seek regulatory authorization for our products in additional jurisdictions, and we seek to expand our sales and marketing opportunities internationally. Our success will depend, in part, upon our ability to succeed in differing legal, regulatory, economic, social and political conditions by developing, implementing and maintaining policies and strategies that are effective in each location where we do business. We have limited experience operating internationally and engaging in international business involves a number of difficulties and risks, including:
For example, our business may continue to be impacted by the conflicts in Ukraine and the Middle East, any economic or other sanctions imposed on Russia and others for aggression in Ukraine, and any economic or other countermeasures by affected countries. Any such conflict may also impact our ability to secure raw materials and finished products and create supply chain disruptions. For example, we have incurred increased costs of the magnet, a key custom-made component in our Swoop® system, which is manufactured by a single source supplier in Europe. In the event of further increased costs or interruption from any of our suppliers or manufacturers, we may not be able to obtain capacity from other sources or develop alternate or secondary sources without incurring material additional costs and substantial delays. As we seek to expand into international markets, the conflict in Ukraine and the Middle East and any related economic or other sanctions or related countermeasures could limit our ability to expand our business and have a material adverse impact on demand for our products and sales in affected markets. In addition, sanctions imposed on Russia and others in response to the conflict in Ukraine may also continue to adversely impact the financial markets and the global economy, and any economic countermeasures by Russia and others could exacerbate market and economic instability.
If we dedicate significant resources to our international operations and are unable to manage these risks effectively, our business, operating results and financial condition may be adversely affected.
37
We are subject to export and import control laws and regulations that could impair our ability to compete in international markets or subject us to liability if we violate such laws and regulations.
We are required to comply with export and import control laws, which may affect our ability to enter into or complete transactions with certain customers, business partners, and other persons. In certain circumstances, export control regulations may prohibit the export of certain products, services, and technologies. We may be required to obtain an export license before exporting a controlled item, and granting of a required license cannot be assured. Compliance with the import laws that apply to our businesses may restrict our access to, and may increase the cost of obtaining, certain products and could interrupt our supply of imported inventory.
Exported technologies necessary to develop and manufacture certain products are subject to U.S. export control laws and similar laws of other jurisdictions. We may be subject to adverse regulatory consequences, including government oversight of facilities and export transactions, monetary penalties, and other sanctions for violations of these laws. In certain instances, these regulations may prohibit us from developing or manufacturing certain of our products for specific applications outside the United States. Failure to comply with any of these laws and regulations could result in civil and criminal, monetary, and nonmonetary penalties; disruptions to our business; limitations on our ability to import and export products and services; or damage to our reputation.
If we experience decreasing prices for our products and are unable to reduce our expenses, including the per unit cost of producing our products, there may be a material adverse effect on our business, results of operations, financial condition and cash flows.
We may experience decreasing prices for our products due to pricing pressure from managed care organizations and other third-party payors and suppliers, increased market power of our payors as the medical device industry consolidates, and increased competition among suppliers, including manufacturing services providers. If the prices for our products and services decrease and we are unable to reduce our expenses, including the cost of sourcing materials, logistics and the cost to manufacture our products, our business, results of operations, financial condition and cash flows may be adversely affected. To the extent that we engage in enterprise sales, we may be subject to procurement discounts, which could have a negative impact on the prices of our products.
We have undertaken internal restructuring activities that could result in disruptions to our business or otherwise materially harm our results of operations or financial condition.
In December 2022, we announced that we committed to an organizational restructuring designed to decrease our costs and create a more streamlined organization to support our business. As a result, we terminated approximately 13% of our global workforce including, among others, the employees of our subsidiary, Liminal. We believe this re-prioritized strategic focus was the best way to optimize our financial and other resources to advance our goal of developing and commercializing our products and services. There can be no assurance that our restructuring will achieve the cost savings, operating efficiencies or other benefits that we may have initially expected. If our restructuring fails to achieve some or all of the expected benefits therefrom, our cash resources may not last as long as estimated and our business, results of operations and financial condition could be materially and adversely affected.
If we are unable to attract, recruit, train, retain, motivate and integrate key personnel and expand our organization, our operations may be disrupted and we may not achieve our goals.
Our future success depends on our ability to attract, recruit, train, retain, motivate and integrate key personnel, including the Founder of Legacy Hyperfine and Liminal and our director, Dr. Jonathan Rothberg, our Chairperson, R. Scott Huennekens, and our President and Chief Executive Officer, Maria Sainz, as well as other members of our management team and our research and development, manufacturing, software engineering and sales and marketing personnel. As our development and commercialization plans and strategies develop, we will need additional managerial, operational, sales, marketing, financial, legal and other resources. Competition for qualified personnel in the health technology and medical device industry is intense. Due to this intense competition, we may be unable to attract and retain the qualified personnel necessary for the development of our business or to recruit suitable replacement personnel.
We believe that our management team must be able to act decisively to apply and adapt our business model in the rapidly changing markets in which we compete. In addition, we rely upon technical and scientific employees or third-party contractors to effectively establish, manage and grow our business. Consequently, we believe that our future viability will depend largely on our ability to attract and retain highly skilled managerial, sales, scientific and technical personnel. In order to do so, we may need to pay higher compensation or fees to our employees or consultants than we currently expect, and such higher compensation payments may have a negative effect on our operating results. Competition for experienced, high-quality personnel is intense, and we cannot assure investors that we will be able to recruit and retain such personnel. Our growth depends, in particular, on attracting and retaining highly-trained sales personnel with the necessary technical background and ability to understand our products and services at a technical level to effectively identify and sell to potential new customers and develop new products. Because of the technical nature of
38
our products and the dynamic market in which we compete, any failure to attract, recruit, train, retain, motivate and integrate qualified personnel could materially harm our operating results and growth prospects.
Our management may need to divert a disproportionate amount of our attention away from our day-to-day activities and devote a substantial amount of time to managing these growth activities. We may not be able to effectively manage the expansion of our operations, which may result in weaknesses in our infrastructure, operational mistakes, loss of business opportunities, loss of employees and reduced productivity among remaining employees. Our expected growth could require significant capital expenditures and may divert financial resources from other projects, such as the development of additional products. If our management is unable to effectively manage our growth, our expenses may increase more than expected. Our ability to generate and/or grow revenue could be reduced and we may not be able to implement our business strategy. Our future financial performance and our ability to commercialize products and services and compete effectively will depend, in part, on our ability to effectively manage any future growth.
We have limited experience in marketing and selling our products and related services, and if we are unable to successfully commercialize our products and related services, our business and operating results will be adversely affected.
We have limited experience marketing and selling our products and related services. We began selling our Swoop® system in 2020 and currently sell the device primarily to customers through direct sales. Future sales of our products will depend in large part on our ability to effectively market and sell our products and services, successfully manage and expand our sales force, and increase the scope of our marketing efforts. We have entered into distribution arrangements and may enter into additional distribution arrangements in the future. Because we have limited experience in marketing and selling our products, our ability to forecast demand, the infrastructure required to support such demand and the sales cycle to customers is unproven. If we do not build an efficient and effective marketing and sales force, our business and operating results will be adversely affected.
We rely on a single contract manufacturer, Benchmark Electronics, Inc. (“Benchmark”), to test, assemble and supply our finished products. If Benchmark fails to fulfill its obligations under its existing contractual arrangements with us or does not perform satisfactorily, our ability to source our devices could be negatively and adversely affected.
In October 2018, Legacy Hyperfine entered into a Manufacture and Supply Agreement with Benchmark (the “MSA”). Under the MSA, Benchmark agreed to manufacture our products pursuant to binding purchase orders. The parties have agreed to meet periodically regarding any minimum order quantities of components under the MSA. We also have certain inventory-related obligations, including the obligation to purchase excess and obsolete components from Benchmark. See “Item 1. Business - Key Agreements - Manufacture and Supply Agreement with Benchmark Electronics, Inc.”
In the event it becomes necessary to utilize a different contract manufacturer for our component products, we would experience additional costs, delays and difficulties in obtaining such components as a result of identifying and entering into an agreement with a new contract manufacturer as well as preparing such new manufacturer to meet the logistical requirements associated with manufacturing our devices, and our business would suffer.
We rely on a limited number of suppliers for our products. A loss of any of these suppliers could negatively affect our business.
We rely on a limited number of suppliers to manufacture components for our products, including in some cases only a single supplier for some of our components. In addition, we rely on Benchmark to purchase the magnet used in our Swoop® system, which is a key custom-made component manufactured by a single source supplier in Europe. Our reliance on a limited number of suppliers increases our risks, since we do not currently have alternative or replacement suppliers beyond these key parties. In the event of interruption from any of our suppliers, we may not be able to increase capacity from other sources or develop alternate or secondary sources without incurring material additional costs and substantial delays.
If we experience a significant increase in demand for our products, or if we need to replace an existing supplier or manufacturer, we may be unable to supplement or replace them on terms that are acceptable to us, which may undermine our ability to deliver our products to customers in a timely manner. Identifying suitable suppliers and manufacturers is an extensive process that requires us to become satisfied with their quality control, technical capabilities, responsiveness and service, financial stability, regulatory compliance, and labor and other ethical practices. Accordingly, a loss of any of our suppliers or our device manufacturer could have an adverse effect on our business, financial condition and operating results.
We have experienced and may continue to experience pricing pressures from contract suppliers or manufacturers on which we rely.
Third-party suppliers utilized by our manufacturer such as Benchmark have and may continue to impose pricing pressures. Because we currently also rely on Benchmark to manufacture, test and ship all of the Swoop® systems and on a limited number of suppliers to
39
supply our components, including Benchmark to purchase the magnet used in the scanner from a single source supplier, such pricing pressures from a third party such as Benchmark have and could increase our costs and could force us to increase the prices of our products if we are unable to enter into alternative arrangements with other suppliers or manufacturers, potentially leading to decreased customer demand.
If we do not successfully optimize and operate our sales and potential future distribution channels or we do not effectively expand and update our infrastructure, our operating results and customer experience may be negatively impacted.
If we do not adequately predict market demand or otherwise optimize and operate our sales and potential future distribution channels successfully, it could result in excess or insufficient inventory or fulfillment capacity, increased costs, or immediate shortages in product or component supply, or harm our business in other ways. In addition, if we do not maintain adequate infrastructure to enable us to, among other things, manage our purchasing and inventory, it could negatively impact our operating results and customer experience.
The market for our products and services is new, rapidly evolving, and increasingly competitive, as the healthcare industry in the United States is undergoing significant structural change, which makes it difficult to forecast demand for our products and services.
The market for our products and services is new and rapidly evolving, and it is uncertain whether we will achieve and sustain high levels of demand and market adoption. Our future financial performance will depend in part on growth in this market and on our ability to adapt to the changing demands of customers. It is difficult to predict the future growth rate and size of our target market. As a result, our commercial expectations may not be achieved. Negative publicity concerning our products could limit market acceptance of our products and services. If our customers do not perceive the benefits of our products and services, or if our products and services do not attract new customers, then our market may not develop at all, or we may develop more slowly than we expect. Our success will depend to a substantial extent on the willingness of healthcare organizations to increase their use of our technology and our ability to demonstrate the value of our technology relative to competing products and services to existing and potential customers. If healthcare organizations do not recognize or acknowledge the benefits of our products and services or if we are unable to reduce healthcare costs or drive positive health outcomes, then the market for our solutions might not develop at all, or might develop more slowly than we expect. Similarly, negative publicity regarding patient confidentiality and privacy in the context of technology-enabled healthcare or concerns experienced by competitors could limit market acceptance of our products and services.
The healthcare industry in the United States is undergoing significant structural change and is rapidly evolving. Our products and services are offered on a business model, through the sale of device and service and support agreements which is still relatively new in the healthcare industry. If companies do not adopt to this business model and our device does not achieve widespread adoption, or if there is a reduction in demand for the purchase of our device and service and support agreements, our business, financial condition, and results of operations could be adversely affected.
Quality problems could lead to recalls or safety alerts and/or reputational harm and could have a material adverse effect on our business, results of operations, financial condition and cash flows.
Quality of our products is very important to us and our customers due to the serious and costly consequences of product failure. Our business exposes us to potential product liability risks that are inherent in the design, manufacture, and marketing of medical devices. Product or component failures, manufacturing nonconformities, design defects, off-label use, or inadequate disclosure of product-related risks or product-related information with respect to our products, if they were to occur, could result in inaccurate imaging and safety risks. These problems could lead to the recall of, or the issuance of a safety alert relating to, our products, and could result in product liability claims and lawsuits.
Additionally, the manufacture and production of our products must occur in a highly controlled and clean environment to minimize particles and other yield- and quality-limiting contaminants. Weaknesses in process control or minute impurities in materials may cause defective products. If we are not able to maintain stringent quality controls, or if contamination problems arise, our development and commercialization efforts could be delayed, which would harm our business and results of operations.
If we fail to meet any applicable product quality standards and our products are the subject of recalls or safety alerts, our reputation could be damaged, we could lose customers, and our revenue and results of operations could decline.
40
If we are not able to develop and release new products and services, or successful enhancements, new features and modifications to our existing products and services, to successfully implement our software solutions or to achieve adequate clinical feasibility, our business, financial condition and results of operations could be adversely affected.
The markets in which we operate are characterized by rapid technological change, frequent new product and service introductions and enhancements, changing customer demands, and evolving industry standards. The introduction of products and services embodying new technologies can quickly make existing products and services, including the software that comes with the service and support agreement to be obsolete and unmarketable. Additionally, changes in laws and regulations could impact the usefulness of our products and could necessitate changes or modifications to our products to accommodate such changes. We invest substantial resources in researching and developing new products and enhancing existing products by incorporating additional features, improving functionality, and adding other improvements to meet customers’ evolving needs. The success of any enhancements or improvements to our existing products or any new products depends on several factors, including timely completion, competitive pricing, adequate quality testing, integration with new and existing technologies and third-party partners’ technologies and overall market acceptance. We may not succeed in developing, marketing and delivering on a timely and cost-effective basis enhancements or improvements to our existing products or any new products that respond to continued changes in market demands or new customer requirements, and any enhancements or improvements to our products or any new solutions may not achieve market acceptance. Since developing our products is complex, the timetable for the release of new products and enhancements to existing products is difficult to predict, and we may not offer new products and updates as rapidly as our customers require or expect. Any new products that we develop may not be introduced in a timely or cost-effective manner, may contain errors or defects, or may not achieve the broad market acceptance necessary to generate sufficient revenue. Moreover, even if we introduce new products, we may experience a decline in revenue from our existing products that is not offset by revenue from the new products. For example, customers may delay making purchases of new products to permit them to make a more thorough evaluation of these products or until industry and marketplace reviews become widely available. Customers may also delay purchasing a new product because their existing product or other device continues to meet their needs. Some customers may hesitate to migrate to a new product due to concerns regarding the performance of the new product. In addition, we may lose existing customers who choose a competitor’s products and services or decide that a traditional MRI system is sufficient to meet their needs. This could result in a temporary or permanent revenue shortfall and adversely affect our business, financial condition and results of operations.
The introduction of new products and solutions by competitors, the development of entirely new technologies to replace existing offerings or shifts in healthcare benefits trends could make our products obsolete or adversely affect our business, financial condition and results of operations. We may experience difficulties with software development, industry standards, design or marketing that could delay or prevent our development, introduction or implementation of new products, enhancements, additional features or capabilities. If customers do not widely purchase and adopt our products, we may not be able to realize a return on our investment. If we do not accurately anticipate customer demand or if we are unable to develop, license or acquire new features and capabilities on a timely and cost-effective basis, or if such enhancements do not achieve market acceptance, it could result in adverse publicity, loss of revenue or market acceptance or claims by customers brought against us, each of which could have a material and adverse effect on our reputation, business, results of operations and financial condition.
We are party to Technology and Services Exchange Agreements with certain affiliated companies, pursuant to which the parties agreed to share personnel and certain non-core technologies. The sharing arrangements under the agreements may prevent us from fully utilizing our personnel and/or the technologies shared under the agreements. Furthermore, if these agreements were to terminate, or if we were to lose access to these technologies and services, our business could be adversely affected.
We entered into Technology and Services Exchange Agreements (each, a “TSEA” and collectively, the “TSEA”) with other participant companies controlled by the Rothbergs. A TSEA by and among Butterfly Network, Inc., AI Therapeutics, Inc., Quantum-Si Incorporated, 4Bionics LLC, identifeye Health Inc. (f/k/a Tesseract Health, Inc.), Detect, Inc. (f/k/a Homodeus Inc.), Legacy Hyperfine and Liminal was signed in November 2020; a TSEA by and among Quantum-Si Incorporated, AI Therapeutics, Inc., 4Bionics LLC, identifeye Health Inc., Detect, Inc., Legacy Hyperfine and Liminal was signed in February 2021 (and which Protein Evolution, Inc. joined in August 2021); and a TSEA by and among Legacy Hyperfine, Liminal, AI Therapeutics, Inc., identifeye Health Inc. and Detect, Inc. was signed in July 2021 and became effective upon the closing of our December 2021 business combination. Under the TSEA, we and the other participant companies may, in their discretion, permit the use of certain non-core technologies, which include any technologies, information or equipment owned or otherwise controlled by the participant company that are not specifically related to the core business area of the participant, such as software, hardware, electronics, fabrication and supplier information, vendor lists and contractor lists, with the other participant companies. The TSEA provides that ownership of each non-core technology shared by us or another participant company will remain with the company that originally shared the non-core technology. In addition, any participant company (including us) may, in its discretion, permit its personnel to be engaged by another participant company to perform professional, technical or consulting services for such participant. Unless otherwise agreed to by us and the other participant company, all rights, title and interest in and to any inventions, works-of-authorship, idea, data or know-how invented, made, created or developed by the personnel (employees, contractors or consultants) in the course of conducting services for a participant company (“Created IP”) will be owned by the participant company for which the work was performed, and
41
the recipient participant company grants to the party that had its personnel provide the services that resulted in the creation of the Created IP a royalty-free, perpetual, limited, worldwide, non-exclusive, sub-licensable (and with respect to software, sub-licensable in object code only) license to utilize the Created IP only in the core business field of the originating participant company, including a license to create and use derivative works based on the Created IP in the originating participant’s core business field, subject to any agreed upon restrictions.
The technology and personnel-sharing arrangements under the TSEA may prevent us from fully utilizing our personnel if such personnel are also being used by the other participant companies and may also cause our personnel to enter into agreements with or provide services to other companies that interfere with their obligations to us. Created IP under the TSEA may be relevant to our business and created by our personnel but owned by the other participant companies. Furthermore, if the TSEA were to terminate, or if we were to lose access to the technologies and services available pursuant to the TSEA, our business could be adversely affected.
We may acquire other companies or technologies, which could fail to result in a commercial product or net sales, divert our management’s attention, result in additional dilution to our stockholders and otherwise disrupt our operations and harm our operating results.
We may in the future seek to acquire or invest in businesses, applications or technologies that we believe could complement or expand our portfolio, enhance our technical capabilities or otherwise offer growth opportunities. However, we cannot assure you that we would be able to successfully complete any acquisition we choose to pursue, or that we would be able to successfully integrate any acquired business, product or technology in a cost-effective and non-disruptive manner. The pursuit of potential acquisitions may divert the attention of management and cause us to incur various costs and expenses in identifying, investigating and pursuing suitable acquisitions, whether or not they are consummated. We may not be able to identify desirable acquisition targets or be successful in entering into an agreement with any particular target or obtain the expected benefits of any acquisition or investment.
To date, the growth of our operations has been largely organic, and we have limited experience in acquiring other businesses or technologies. We may not be able to successfully integrate any acquired personnel, operations and technologies, or effectively manage the combined business following an acquisition. Acquisitions could also result in dilutive issuances of equity securities, the use of our available cash, or the incurrence of debt, which could harm our operating results. In addition, if an acquired business fails to meet our expectations, our operating results, business and financial condition may suffer.
As international expansion of our business occurs, it will expose us to market, regulatory, political, operational, financial and economic risks associated with doing business outside of the United States.
Our long term strategy is to increase our international presence. We received marketing authorization for brain imaging in several countries, including the European Union (CE), the United Kingdom (UKCA), Canada, Australia and New Zealand. This strategy may include establishing and maintaining physician outreach and education capabilities outside of the United States and expanding our relationships with international customers. Doing business internationally involves a number of risks, including:
42
Any of these factors could significantly harm our future international expansion and operations and, consequently, have a material adverse effect on our business, financial condition and results of operations.
Unfavorable global economic conditions could adversely affect our business, financial condition or results of operations.
Our results of operations could be adversely affected by general conditions in the global economy and in the global financial markets, including changes in inflation, interest rates and overall economic conditions and uncertainties. For instance, if inflation or other factors were to significantly increase our business costs, it may not be feasible to pass price increases on to our customers. Inflation could also adversely affect the ability of our customers to purchase our products. An economic downturn could result in a variety of risks to our business, including weakened demand for our products and our inability to raise additional capital when needed on acceptable terms, if at all. A weak or declining economy could also strain our manufacturers and suppliers, possibly resulting in supply disruption, or cause future customers to delay making payments for our products. Any of the foregoing could harm our business and we cannot anticipate all of the ways in which the current economic climate and financial market conditions could adversely impact our business.
The enactment of legislation implementing changes in the U.S. Taxation of international business activities, the adoption of other tax reform policies or changes in tax legislation or policies in jurisdictions outside of the United States could materially impact our results of operations and financial condition.
We are subject to income tax in the numerous jurisdictions in which we operate. Reforming the taxation of international businesses has been a priority for politicians, and a wide variety of potential changes have been proposed. Some proposals, several of which have been enacted, impose incremental taxes on gross revenue, regardless of profitability. Furthermore, it is reasonable to expect that global taxing authorities will be reviewing current legislation for potential modifications in reaction to the implementation of the 2017 Tax Cuts and Jobs Act (the “Tax Act”) in the United States. Due to the expanding scale of our international business activities, changes in the taxation of such activities may increase our worldwide effective tax rate and the amount of taxes we pay and harm our business.
U.S. taxation of international business activities or the adoption of tax reform policies could materially impact our future financial position and results of operations.
Limitations on the ability of taxpayers to claim and utilize foreign tax credits and the deferral of certain tax deductions until earnings outside of the United States are repatriated to the United States, as well as changes to U.S. Tax laws that may be enacted in the future, could impact the tax treatment of future foreign earnings. Should the scale of our international business activities expand, any changes in the U.S. taxation of such activities could increase our worldwide effective tax rate and harm our future financial position and results of operations.
We may face exposure to foreign currency exchange rate fluctuations.
While we have historically transacted in U.S. Dollars with the majority of our customers and suppliers, we have transacted in some foreign currencies and may transact in more foreign currencies in the future. Accordingly, changes in the value of foreign currencies relative to the U.S. Dollar may affect our revenue and operating results. As a result of such foreign currency exchange rate fluctuations, it could be more difficult to detect underlying trends in our business and operating results. In addition, to the extent that fluctuations in currency exchange rates cause our operating results to differ from our expectations or the expectations of our investors, the trading price of our stock could be adversely affected.
Our ability to use net operating losses to offset future taxable income may be subject to certain limitations.
As of December 31, 2023, we had federal net operating loss carryforwards (“NOLs”) to offset future taxable income of approximately $170 million, of which $12.1 million will begin to expire in 2034 if not utilized. As of December 31, 2023, Liminal had federal NOLs to offset future taxable income of approximately $14.7 million. A lack of future taxable income would adversely affect our ability to utilize these NOLs. In addition, under Section 382 of the Internal Revenue Code of 1986, as amended (the “Code”), a corporation that undergoes an “ownership change” is subject to limitations on its ability to utilize its pre-change NOLs and other pre-change tax attributes (such as research tax credits) to offset post-change taxable income. For these purposes, an ownership change generally
43
occurs where the equity ownership of one or more stockholders or groups of stockholders who owns at least 5% of a corporation’s stock increases its ownership by more than 50 percentage points over its lowest ownership percentage within a three-year period (calculated on a rolling basis). Our existing NOLs may be subject to limitations arising out of previous ownership changes and we may be limited as to the amount that can be utilized each year as a result of such previous ownership changes, including our December 2021 business combination and related transactions. In addition, future changes in our stock ownership, including future offerings, as well as other changes that may be outside of our control, could result in additional ownership changes under Section 382 of the Code. Our NOLs may also be impaired under similar provisions of state law. We have recorded a full valuation allowance related to our NOLs and other deferred tax assets due to the uncertainty of the ultimate realization of the future benefits of those assets.
In addition to the limitations discussed above under Sections 382 of the Code, the utilization of NOLs incurred in taxable years beginning after December 31, 2017, are subject to limitations adopted by the Tax Cuts and Jobs Act, as modified by the Coronavirus Aid, Relief, and Economic Security Act (“CARES Act”). Under the Tax Act, in general, NOLs generated in taxable years beginning after December 31, 2017 may offset no more than 80 percent of such year’s taxable income and there is no ability for such NOLs to be carried back to a prior taxable year. The CARES Act modifies the Tax Act with respect to the Tax Act’s limitation on the deduction of NOLs and provides that NOLs arising in taxable years beginning after December 31, 2017 and before January 1, 2021, may be carried back to each of the five taxable years preceding the tax year of such loss, but NOLs arising in taxable years beginning after December 31, 2020 may not be carried back. In addition, the CARES Act eliminates the limitation on the deduction of NOLs to 80 percent of current year taxable income for taxable years beginning before January 1, 2021. As a result of such limitation, we may be required to pay federal income tax in some future year notwithstanding that we had a net loss for all years in the aggregate.
Risks Related to Healthcare Industry Shifts and Changing Regulations
We are subject to extensive government regulation, which could restrict the development, marketing, sale and distribution of our products and could cause us to incur significant costs.
Our medical devices and associated services are subject to extensive pre-market and post-market regulation by the FDA and various other federal, state, local and foreign government authorities. Government regulation of medical devices is meant to assure their safety and effectiveness, and includes requirements for, among other things:
The laws and regulations to which we and our products are subject are complex and subject to periodic changes. Regulatory changes could result in restrictions on our ability to carry on or expand our operations, higher than anticipated costs or lower than anticipated sales.
44
Before a new medical device, or a significant modification of a medical device, including a new use of or claim for an existing product, can be marketed in the United States, we must first receive either 510(k) clearance or premarket approval (“PMA”) from the FDA, unless an exemption applies. In the 510(k) clearance process, the FDA must determine that a proposed device is “substantially equivalent” to a device legally on the market, known as a “predicate” device, with respect to intended use, technology and safety and effectiveness, in order to clear the proposed device for marketing. Clinical data is sometimes required to support substantial equivalence. Legacy Hyperfine received 510(k) clearance from the FDA for its portable MRI system in 2020. In February and October 2023, we received additional 510(k) clearances from the FDA of the latest updates to our Swoop® system AI -powered software. The combination of these two software updates significantly improved diffusion-weighted imaging (“DWI”), incorporated deep-learning based denoising in the post-processing of DWI images for crisper images, and improved image quality for all Swoop® system sequences. The Swoop® system has received marketing authorization for brain imaging in several countries, including the European Union (CE), the United Kingdom, Canada, Australia and New Zealand. All of our revenue to date has been generated from sales of the Swoop® system and related services.
We may be required to obtain a new 510(k) clearance or PMA approval for significant post-market modifications to our products, including any modifications made to our commercially marketed devices.
Obtaining 510(k) clearance or PMA approval for medical devices can be expensive and time-consuming, and entails significant user fees, unless an exemption is available. The FDA’s process for obtaining 510(k) clearance usually takes three to 12 months, but it can last longer. In the PMA approval process, the FDA must determine that a proposed device is safe and effective for its intended use based, in part, on extensive data, including but not limited to, technical, nonclinical, clinical trial, manufacturing and labeling data. The process for obtaining a PMA is more costly and uncertain and approval can take anywhere from at least one year to, in some cases, multiple years from the time the application is initially filed with the FDA. Modifications to products that are approved through a PMA application generally require further FDA approval. Some of our future products may require PMA approval. In addition, the FDA may demand that we obtain a PMA prior to marketing future changes of our existing products. Further, we may not be able to obtain additional 510(k) clearances or PMAs for new products or for modifications to, or additional indications for, our products in a timely fashion or at all. Delays in obtaining future clearances or approvals could adversely affect our ability to introduce new or enhanced products in a timely manner, which in turn could harm our revenue and future profitability.
In order to conduct a clinical investigation involving human subjects for the purpose of demonstrating the safety and effectiveness of a medical device, if necessary, for a PMA application or 510(k) notification, a company must, among other things, apply for and obtain institutional review board (“IRB”) approval of the proposed investigation. In addition, if the clinical study involves a “significant risk” (as defined by the FDA) to human health, the sponsor of the investigation must also submit and obtain FDA approval of an investigational device exemption (“IDE”) application and follow applicable IDE regulations. Unless IDE-exempt, nonsignificant risk devices are still subject to certain abbreviated IDE requirements, but an IDE application is not required if such abbreviated requirements are met. We may not be able to obtain any necessary FDA and/or IRB approval to undertake clinical trials in the United States for future devices we develop and intend to market in the United States. If we do obtain such approvals, the FDA may find that our studies do not comply with the IDE or other regulations governing clinical investigations or the data from any such trials may not support clearance or approval of the investigational device. Moreover, certainty that clinical trials will meet desired endpoints, produce meaningful or useful data and be free of unexpected adverse effects, or that the FDA will accept the validity of foreign clinical study data (if applicable) cannot be assured, and such uncertainty could preclude or delay market clearance or authorizations resulting in significant financial costs and reduced revenue.
We are also subject to numerous post-marketing regulatory requirements, which include quality system regulations related to the manufacture of our devices, labeling regulations and medical device reporting (“MDR”) regulations. The last of these regulations requires us to report to the FDA if our devices cause or contribute to a death or serious injury, or malfunction in a way that would likely cause or contribute to a death or serious injury if the malfunction recurred. If we fail to comply with present or future regulatory requirements that are applicable to us, we may be subject to enforcement action by the FDA, which may include any of the following sanctions:
45
The occurrence of any of these events may have a material adverse effect on our business, financial condition and results of operations.
Our employees, independent contractors, consultants, manufacturers and suppliers may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
We are exposed to the risk that our employees, independent contractors, consultants, manufacturers and suppliers may engage in fraudulent or illegal activity. Misconduct by these parties could include intentional, reckless and/or negligent conduct or disclosure of unauthorized activities to us that violates: (i) the laws of the FDA and other similar foreign regulatory bodies, including those laws requiring the reporting of true, complete and accurate information to such regulators; (ii) manufacturing standards; (iii) healthcare fraud and abuse laws in the United States and similar foreign fraudulent misconduct laws; or (iv) laws that require the true, complete and accurate reporting of financial information or data. These laws may impact, among other things, future sales, marketing and education programs. In particular, the promotion, sales and marketing of healthcare items and services, as well as certain business arrangements in the healthcare industry, are subject to extensive laws designed to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, structuring and commissions, certain customer incentive programs and other business arrangements generally. Activities subject to these laws also involve the improper use of information obtained in the course of patient recruitment for clinical trials.
Although we have a code of business conduct and ethics, it is not always possible to identify and deter misconduct by our employees and other third parties, and the precautions we take to detect and prevent these activities may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations. If any such actions are instituted against us and we are not successful in defending ourselves or asserting our rights, those actions could result in the imposition of significant fines or other sanctions, including the imposition of civil, criminal and administrative penalties, damages, monetary fines, disgorgement, individual imprisonment, additional integrity reporting and oversight obligations, possible exclusion from participation in Medicare, Medicaid and other government healthcare programs, contractual damages, reputational harm, diminished profits and future earnings and curtailment of operations, any of which could adversely affect our ability to operate our business and our results of operations. Whether or not we are successful in defending against any such actions or investigations, we could incur substantial costs, including legal fees, and divert the attention of management in defending ourselves against any of these claims or investigations, which could have a material adverse effect on our business, financial condition and results of operations.
There is no guarantee that the FDA will grant 510(k) clearance or premarket approval of our future products, and failure to obtain necessary clearances or approvals for our future products would adversely affect our ability to grow our business.
Some of our new or modified products will require FDA clearance of a 510(k) notification or FDA approval of a PMA application. The FDA may refuse our requests for 510(k) clearance or PMA of new products or may not clear or approve these products for the indications that are necessary or desirable for successful commercialization. Early stage review may also result in delays or other issues. For example, the FDA has issued guidance intended to explain the procedures and criteria used in assessing whether 510(k) and PMA submissions should be accepted for substantive review. Under the “Refuse to Accept” guidance, the FDA conducts an early review against specific acceptance criteria to inform 510(k) and PMA submitters if the submission is administratively complete, or if not, to identify the missing element(s). Submitters are given the opportunity to provide the FDA with any information identified as missing. If the information is not provided within a specified time, the submission will not be accepted for FDA review. The FDA may also change its clearance and approval policies, adopt additional regulations or revise existing regulations, or take other actions that may prevent or delay approval or clearance of our products under development or impact our ability to gain clearance or approval for modifications to our currently approved or cleared products in a timely manner. Significant delays in receiving clearance or approval, or the failure to receive clearance or approval for our new products would have an adverse effect on our ability to expand our business.
Recent initiatives by the FDA to enhance and modernize various regulatory pathways for device products and its overall approach to safety and innovation in the medical technology industry creates the possibility of changing product development costs, requirements, and other factors and additional uncertainty for our future products and business.
Regulatory requirements may change in the future in a way that adversely affects us. Any change in the laws or regulations that govern the clearance and approval processes or the post-market compliance requirements relating to our current and future products could make it more difficult and costly to obtain clearance or approval for new products, or to produce, market and distribute existing products.
46
For example, the FDA and other government agencies have been focusing on the cybersecurity risks associated with certain medical devices and encouraging device manufacturers to take a more proactive approach to assessing the cybersecurity risks of their devices both during development and on a periodic basis after the devices are in commercial distribution. These regulatory efforts could lead to new FDA requirements in the future or additional product liability or other litigation risks if any of our products is considered to be susceptible to third-party tampering. In December 2016, Congress passed the 21st Century Cures Act, which made multiple changes to the FDA’s rules for medical devices as well as for clinical trials, and in September 2022, Congress passed the Medical Device User Fee reauthorization package, which affects medical device regulation both pre- and post-approval and could have certain impacts on our business. In recent years, the FDA has also considered a series of efforts to modernize and streamline the 510(k) notification and regulatory review process and monitoring post-market safety. For example, in October 2022, FDA announced that 510(k) applications may be submitted electronically using the electronic submission template and resource, or eSTAR. Further, changes in the FDA 510(k) process could make clearance more difficult to obtain, increase delay, add uncertainty and have other significant adverse effects on our ability to obtain and maintain clearance for our products.
It is unclear at this time whether and how various activities initiated or announced by the FDA to modernize the U.S. medical device regulatory system could affect our business, as some of the FDA’s new medical device safety and innovation initiatives have not been formalized and remain subject to change.
If we fail to obtain regulatory authorizations in other countries for existing products or products under development, we will not be able to commercialize these products in those countries.
In order for us to market our products in countries outside of the United States, we must comply with extensive safety and quality regulations in other countries regarding the quality, safety and efficacy of our products. These regulations, including the requirements for marketing authorizations, and the time required for regulatory review, vary from country to country. Failure to obtain regulatory authorization in any foreign country in which we plan to market our products may harm our ability to generate revenue and harm our business. Marketing authorization requirements vary between countries and can involve additional product testing and additional administrative review periods. The time required to obtain marketing authorization in other countries might differ from that required to obtain FDA clearance or other marketing authorization. The regulatory process in other countries may include all of the risks detailed above regarding FDA clearance in the United States. Regulatory authorization of a product in one country does not ensure regulatory approval in another, but a failure or delay in obtaining regulatory authorization in one country may negatively impact the regulatory process in others. Failure to obtain regulatory authorization in other countries or any delay or setback in obtaining such authorization could have the same adverse effects described above regarding FDA clearance in the United States.
The primary regulatory environment in Europe is that of the European Economic Area (“EEA”), which is comprised of the Member States of the European Union, Iceland, Liechtenstein and Norway. In 2023, our Swoop® system received CE certification. The Medical Device Regulation became fully effective on May 26, 2021.The Medical Device Regulation includes elements intended to strengthen the conformity assessment procedures, assert greater control over notified bodies and their standards, increase overall system transparency, and impose more robust device vigilance requirements on manufacturers and distributors.
The new rules and procedures that have been created under the overhauled EU regulations will likely result in increased regulatory oversight of all medical devices marketed in the EU, and this may, in turn, increase the costs, time and requirements that need to be met in order to place an innovative or high-risk medical device on the EEA market.
If we, our current or future contract manufacturers, or our current or future component suppliers are unable to manufacture our products in sufficient quantities, on a timely basis, at acceptable costs and in compliance with regulatory and quality requirements, the manufacturing and distribution of our devices could be interrupted, and our product sales and operating results could suffer.
When producing and distributing commercial medical device products, we, our contract manufacturer, and certain of our component suppliers are required to comply with the FDA’s Quality System Regulation (“QSR”), which is a complex regulatory framework that covers the procedures and documentation of the design, testing, production, control, quality assurance, labeling, packaging, sterilization, storage, shipping and servicing of our devices. Compliance with applicable regulatory requirements is subject to continual review and is monitored rigorously through periodic, sometimes unannounced, inspections by the FDA. We cannot assure investors that our facilities or our third-party manufacturers’ or suppliers’ facilities would pass any future quality system inspection. Failure of us or our third-party manufacturers and component suppliers to adhere to QSR requirements or take adequate and timely corrective action in response to an adverse quality system inspection finding could delay production of our products and lead to fines, difficulties in obtaining marketing authorizations for our products, recalls, or enforcement actions, including but not limited to injunctive relief or consent decrees, or other consequences, which could have a material adverse effect on our business, financial condition or results of operations. Any such failure, including the failure of our current or any future contract manufacturers to achieve and maintain the required high manufacturing standards, could result in further delays or failures in product testing or delivery, cost overruns, increased warranty costs or other problems that could harm our business and prospects.
47
In addition, any of our products shipped internationally are also required to comply with applicable quality standards and regulatory requirements, including the International Organization for Standardization (“ISO”) quality system standards as well as European Directives and norms in order to produce products for sale in the EU. In addition, many countries such as Canada and Japan have very specific additional regulatory requirements for quality assurance and manufacturing. If we fail to continue to comply with current good manufacturing requirements, as well as ISO or other regulatory standards, we may be required to cease all or part of our operations until we comply with these regulations. Maintaining compliance with multiple regulators adds complexity and cost to our manufacturing and compliance processes.
Our current or future products may be subject to product recalls even after receiving FDA clearance or approval. A recall of our products, either voluntarily or at the direction of the FDA, or the discovery of serious safety issues with our products, could have a significant adverse impact on us.
The FDA and similar governmental bodies in other countries have the authority to require the recall of our products if we or our third-party manufacturers fail to comply with relevant regulations pertaining to, among other things, manufacturing practices, labeling, advertising or promotional activities, or if new information is obtained concerning the safety or efficacy of these products. For example, under the FDA’s Medical Device Reporting regulations, we are required to report to the FDA any incident in which our marketed products may have caused or contributed to a death or serious injury or in which our marketed products malfunctioned in a manner likely to cause or contribute to death or serious injury if that malfunction were to recur. Repeated adverse events or product malfunctions may result in a voluntary or involuntary product recall, or administrative or judicial seizure or injunction, when warranted. A government-mandated recall may be ordered if the FDA finds that there is a reasonable probability that the device would cause serious, adverse health consequences or death. A voluntary recall by us could occur as a result of any material deficiency in a device, such as manufacturing defects, labeling deficiencies, packaging defects or other failures to comply with applicable regulations. In general, if we decide to make a change to our marketed product, we are responsible for determining whether to classify the change as a recall. It is possible that the FDA could disagree with our initial classification. The FDA requires that certain classifications of recalls be reported to the FDA within 10 working days after the recall is initiated. If a change to a device addresses a violation of the federal Food, Drug, and Cosmetic Act (“FDCA”), that change would generally constitute a medical device recall and require submission of a recall report to the FDA.
Recalls of any of our products would divert managerial and financial resources and have an adverse effect on our reputation, results of operations and financial condition, which could impair our ability to produce our products in a cost-effective and timely manner in order to meet our customers’ demands. We may also be subject to product liability claims, be required to bear other costs, or be required to take other actions that may have a negative impact on our future sales and our ability to generate profits. Companies are required to maintain certain records of recalls, even if they are not reportable to the FDA. We may initiate voluntary recalls, field corrections, or removals involving our products in the future that we determine do not require notification to the FDA. If the FDA disagrees with our determinations, the FDA could require us to report those actions as recalls. A future recall, withdrawal, or seizure of any product could materially and adversely affect consumer confidence in our brands, lead to decreased demand for our products and negatively affect our sales. In addition, the FDA could take enforcement action for failing to report recalls when they were conducted by us or one of our agents.
We may be subject to enforcement action if we engage in improper or off-label marketing or promotion of our commercial medical device products, including fines, penalties and injunctions.
Our promotional materials and training methods must comply with FDA and other applicable laws and regulations, including the prohibition of the promotion of unapproved, or off-label, uses of lawfully marketed medical device products. Physicians may, however, use our commercial products off-label, as the FDA does not restrict or regulate a physician’s practice of medicine. Medical device manufacturers and distributors are only permitted to promote their products in a way that is consistent with the FDA-authorized labeling and indications for use. If the FDA determines that our promotional materials or training materials promote a cleared or approved medical device in a manner inconsistent with our labeling, the agency could request that we modify our training or promotional materials or subject us to regulatory or enforcement actions, including the issuance of an Untitled Letter or a Warning Letter or seeking injunction, seizure, civil fines or criminal penalties. In addition to ensuring that the claims we make are consistent with our regulatory clearances or approvals, the FDA also ensures that promotional labeling for all regulated medical devices is neither false nor misleading.
It is also possible that other federal, state or foreign enforcement authorities might take action if they consider our promotional or training materials to constitute promotion of an off-label use, which could result in significant fines or penalties under other statutory authorities, such as laws prohibiting false claims for reimbursement. In that event, our reputation could be damaged, and adoption of our products could be impaired. Although our policy is to refrain from making statements or from disseminating promotional material that could be considered off-label promotion of our commercial medical device products, the FDA or another regulatory agency could disagree and conclude that we have engaged in off-label promotion. In addition, the off-label use of our products may increase the risk of product liability claims. Product liability claims are expensive to defend and could divert our management’s attention, result in substantial damage awards against us, and harm our reputation. Recent court decisions have impacted the FDA’s enforcement activity
48
regarding off-label promotion in light of First Amendment considerations, although there are still significant risks in this area in part due to potential False Claims Act exposure. Further, this area is subject to ongoing policy changes at the federal level, resulting in some degree of uncertainty for regulated businesses. For example, in August 2021 the FDA issued a final rule revising the agency’s regulation governing the types of evidence relevant to determining the “intended use” of a drug or device under the FDCA, which has significant implications for when a manufacturer or distributor has engaged in off-label marketing.
Digital marketing and social media efforts may expose us to additional regulatory scrutiny, including from the Federal Trade Commission (the “FTC”) and other consumer protection agencies and regulators.
In addition to the laws and regulations enforced by the FDA, advertising for various services and for non-restricted medical devices is subject to federal truth-in-advertising laws enforced by the FTC, as well as comparable state consumer protection laws. Our efforts to promote prescription medical device products via social media initiatives may subject us to additional scrutiny of our practices. For example, the FTC and other consumer protection agencies scrutinize all forms of advertising (whether in digital or traditional formats) for business services, consumer-directed products, and non-restricted medical devices to ensure that advertisers are not making false, misleading or unsubstantiated claims or failing to disclose material relationships between the advertiser and its products’ endorsers, among other potential issues. The FDA oversees the advertising and promotional labeling for restricted medical devices and ensures, among other things, that there is effective communication of, and a fair and balanced presentation of, the risks and benefits of such high-risk medical devices.
Under the Federal Trade Commission Act (“FTC Act”), the FTC is empowered, among other things, to (a) prevent unfair methods of competition and unfair or deceptive acts or practices in or affecting commerce; (b) seek monetary redress and other relief for conduct injurious to consumers; and (c) gather and compile information and conduct investigations relating to the organization, business, practices, and management of entities engaged in commerce. The FTC has very broad enforcement authority, and failure to abide by the substantive requirements of the FTC Act and other consumer protection laws can result in administrative or judicial penalties, including injunctions affecting the manner in which we would be able to market services or products in the future, or criminal prosecution. We plan to increase our advertising activities that may be subject to these federal and state truth-in-advertising laws. Any actual or perceived non-compliance with those laws could lead to an investigation by the FTC or a comparable state agency or could lead to allegations of misleading advertising by private plaintiffs. Any such action against us could disrupt our business operations, cause damage to our reputation, and have a material adverse effect on our business.
Because we do not require extensive training for users of our current products, although they are limited under the FDA’s marketing clearances to use by, and that images generated from the scanner be interpreted by, trained healthcare practitioners, there exists a potential for misuse of these products, misinterpretation of images by untrained professionals or misuse of these products by untrained professionals, which could ultimately harm our reputation and business.
Federal regulations allow us to sell our medical device products to or on the order of practitioners licensed by law to use or order the use of a prescription device. The definition of “licensed practitioners” varies from state to state. As a result, our current products may be purchased or operated by physicians with varying levels of training and, in many states, by non-physicians, including nurse practitioners, chiropractors and technicians. The FDA clearances of the products require interpretation of images by trained physicians and use of that information in determining a diagnosis. Outside the United States, many jurisdictions do not require specific qualifications or training for purchasers, or operators or interpreters, of medical device products. We do not supervise the procedures performed with our products, nor can we require that direct medical supervision occur. Although product training is offered, we do not require purchasers or operators of our non-invasive products to attend training sessions. The lack of required training and the purchase and use of our non-invasive products by non-physicians may result in product misuse and adverse treatment outcomes, which could harm our reputation and expose us to costly product liability litigation.
We are subject to federal, state and foreign laws prohibiting “kickbacks” and false or fraudulent claims, and other fraud and abuse laws, transparency laws, and other healthcare laws and regulations, which, if violated, could subject us to substantial penalties. Additionally, any challenge to or investigation into our practices under these laws could cause adverse publicity and be costly to respond to, and thus could harm our business.
Our relationships with customers and third-party payors are subject to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain our sales, marketing and other promotional activities by limiting the kinds of financial arrangements, including sales programs and certain customer and product support programs, we may have with hospitals, physicians or other purchasers of medical devices. Other federal and state laws generally prohibit individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid, or other third-party payors that are false or fraudulent, or are for items or services that were not provided as claimed. These laws include, among others, the federal Anti-Kickback Statute, the federal civil False Claims Act, other federal healthcare false statement and fraud statutes, the Open Payments program, the Civil Monetary Penalties Law, and analogous fraud and abuse and transparency laws in most states, as described in “Item
49
1. Business - Government Regulation.” Although the federal laws generally apply only to products or services for which payment may be made by a government healthcare program, state laws often apply regardless of whether federal funds may be involved.
While we believe and strive to ensure that our business arrangements with third parties and other activities and programs comply with all applicable laws, these laws are complex, and our activities may be found not to be compliant with one or more of these laws, which may result in significant civil, criminal and/or administrative penalties, fines, damages and exclusion from participation in government healthcare programs. Even an unsuccessful challenge or investigation into our practices could cause adverse publicity, and be costly to respond to, and thus could have a material adverse effect on our business, financial condition and results of operations. Our compliance with Medicare and Medicaid regulations may be reviewed by federal or state agencies, including the Office of Inspector General for the U.S. Department of Health and Human Services (“HHS-OIG”), Centers for Medicare & Medicaid Services (“CMS”), and the Department of Justice, or may be subject to whistleblower lawsuits under federal and state false claims laws. To ensure compliance with Medicare, Medicaid and other regulations, government agencies conduct periodic audits of the Company to ensure compliance with various supplier standards and billing requirements.
Similarly, our international operations are subject to the provisions of the FCPA, which prohibits U.S. companies and their intermediaries from making payments in violation of law to non-U.S. government officials for the purpose of obtaining or retaining business or securing any other improper advantage. In many countries, the healthcare professionals that medical device distributors regularly interact with may meet the definition of a foreign official for purposes of the FCPA. International business operations are also subject to various other international anti-bribery laws such as the U.K. Anti-Bribery Act. Despite meaningful measures that we undertake to facilitate lawful conduct, which include training and compliance programs and internal policies and procedures, we may not always prevent unauthorized, reckless or criminal acts by our employees or agents, or employees or agents of businesses or operations we may acquire. Violations of these laws, or allegations of such violations, could disrupt operations, involve significant management distraction and have a material adverse effect on our business, financial condition and results of operations, among other adverse consequences.
If we are found to have violated laws protecting the confidentiality of health information, we could be subject to civil or criminal penalties, which could increase our liabilities and harm our reputation or our business.
There are a number of federal and state laws protecting the confidentiality of certain health information and restricting the use and disclosure of that protected information. In particular, the U.S. Department of Health and Human Services promulgated privacy rules and security standards and breach notification rules under the Health and Insurance Portability and Accountability Act (“HIPAA”). These rules protect medical records and other identifiable health information by limiting their use and disclosure, giving individuals the right to access, amend and seek accounting of the uses and disclosures of their own health information and limiting most use and disclosures of health information to the minimum amount reasonably necessary to accomplish the intended purpose of the use or disclosure. When we provide services to our customers involving access to information protected under HIPAA, such as troubleshooting and maintenance of our products, we are functioning as a “business associate” under HIPAA, obligated to comply with much of HIPAA’s privacy rule, all of HIPAA’s security standards and also HIPAA breach notification requirements. As a business associate, we are subject to direct enforcement by the HHS Office for Civil Rights and state attorneys general, and we are also subject to audit and investigation. If we are found to be in violation of applicable HIPAA requirements, we could subject our customers or healthcare provider partners to civil or criminal penalties, which could increase our liabilities, harm our reputation and have a material adverse effect on our business, financial condition and results of operations.
We are subject to complex and evolving U.S. and foreign laws and regulations regarding privacy, data protection, and other matters. Many of these laws and regulations are subject to change and uncertain interpretation, and could result in claims, changes to our business practices, monetary penalties, increased cost of operations, or declines in customer growth or engagement, or otherwise harm our business.
We are subject to a variety of laws and regulations in the United States and abroad that involve matters central to our business, including laws and regulations relating to privacy, data sharing and data protection, artificial intelligence and use of machine learning, rights of publicity, content, intellectual property, advertising, marketing, distribution, data security, data retention and deletion, personal information, electronic contracts and other communications, competition, protection of minors, consumer protection, telecommunications, product liability, taxation, economic or other trade prohibitions or sanctions, corrupt practices, fraud, waste and abuse restrictions, and securities law compliance. The introduction of new products or expansion of our activities in certain jurisdictions may subject us to additional laws and regulations. For example, in addition to data protection laws passed by the federal government, many states and foreign countries have implemented their own data protection laws, some of which may apply simultaneously and conflict with federal law. Many of these laws create consumer rights including the right to know what personal information is collected, the right to know whether the data is sold or disclosed and to whom, the right to request that a company delete personal information collected, the right to opt-out of the sale of personal information and the right to non-discrimination in terms of price or service when a consumer exercises a privacy right. If we fail to comply with these regulations, we could be subject to civil sanctions, including fines and penalties for noncompliance.
50
In addition, foreign data protection, privacy, and other laws and regulations can be more restrictive than those in the United States. Data localization laws in some countries generally mandate that certain types of data collected in a particular country be stored and/or processed within that country. We could be subject to audits in Europe and around the world, particularly in the areas of consumer and data protection, as we continue to grow and expand our operations. Legislators and regulators may make legal and regulatory changes, or interpret and apply existing laws, in ways that make our products less useful to customers, require us to incur substantial costs, expose us to unanticipated civil or criminal liability, or cause us to change our business practices. These changes or increased costs could negatively impact our business and results of operations in material ways. For example, the GDPR imposes requirements in the EEA relating to, among other things, consent to process personal data of individuals, the information provided to individuals regarding the processing of their personal data, the security and confidentiality of personal data, notifications in the event of data breaches and use of third-party processors. GDPR also imposes restrictions on the transfer of personal data from the EEA to third countries like the United States. If we fail to comply with these standards, we could be subject to criminal penalties and civil sanctions, including fines and penalties and amounts could be significant.
Security breaches, loss of data and other disruptions could compromise sensitive information related to our business or prevent us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation.
In the ordinary course of our business, we collect and store sensitive data, personally identifiable information of individuals, and intellectual property and proprietary business information owned or controlled by us, our customers and other third parties. This data encompasses a wide variety of business-critical information, including research and development information, commercial information, and business and financial information. We face four primary risks relative to protecting this critical information: loss of access; inappropriate disclosure; inappropriate modification; and inadequate monitoring of our controls over the first three risks.
The secure processing, storage, maintenance, and transmission of this critical information is vital to our operations and business strategy, and we devote significant resources to protecting such information. We have implemented multiple layers of security measures to protect the confidentiality, integrity and availability of data and the systems and devices that store and transmit data. We utilize current security technologies, including encryption and data depersonalization, and our defenses are monitored and routinely tested. Although we take measures to protect sensitive information from unauthorized access or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers or viruses, breaches, interruptions due to employee error, malfeasance, lapses in compliance with privacy and security mandates, or other disruptions. Any such breach or interruption could compromise our networks and the information stored there could be accessed by unauthorized parties, publicly disclosed, lost, or stolen. Because the techniques used to obtain unauthorized access, disable or degrade service, or sabotage systems change frequently, and may not immediately produce signs of intrusion, we may be unable to anticipate these incidents or techniques, timely discover them, or implement adequate preventative measures.
Any such security breach or interruption, as well as any action by us or our employees or contractors that might be inconsistent with the rapidly evolving data privacy and security laws and regulations applicable within the United States and elsewhere where we conduct business, could result in the loss, misappropriation, corruption or unauthorized access of data, enforcement actions by U.S. states, the U.S. federal government or foreign governments, liability or sanctions under data privacy laws that protect personally identifiable information, regulatory penalties, litigation, including potential class action litigation,, the incurrence of significant remediation costs, increases to insurance premiums, disruptions to our development programs, business operations and collaborations, diversion of management efforts and damage to us and our brands’ reputation, any of which could harm or have an adverse effect on our financial position and results of our business and operations. Because of the rapidly moving nature of technology and the increasing sophistication of cybersecurity threats, our measures to prevent, respond to and minimize such risks may be unsuccessful.
As a HIPAA business associate, we comply with HIPAA security standards. Whenever possible, we work with de-identified information and employ additional measures such as encryption tools to protect the privacy of individuals, including our customers, and patient data and employee data. However, hackers may attempt to penetrate our computer systems, and, if successful, misappropriate personal or confidential business information. In addition, contractors or other third parties with whom we do business may attempt to circumvent our security measures or inadvertently cause a breach involving such information. While we continue to implement additional protective measures to reduce the risk of and detect cyber incidents, cyber-attacks are becoming more sophisticated and frequent, and the techniques used in such attacks change rapidly.
In addition, the GDPR, which took effect in May 2018, governs the collection and use of personal data of EEA residents. The GDPR, and its equivalents in the United Kingdom and Switzerland, are wide-ranging in scope, impose requirements relating to the consent of the individuals to whom the personal data relates, the information provided to the individuals regarding the processing of their personal data, the security and confidentiality of the personal data, data breach notification and the use of third-party processors in connection with the processing of the personal data. The GDPR also imposes strict rules on the transfer of personal data out of the EEA to third countries like the United States, enhances enforcement authority and imposes large penalties for noncompliance, including the potential for fines of up to 20 million euros or 4% of the annual global revenues of the infringer, whichever is greater.
51
While we strive to comply with the GDPR and its UK and Swiss equivalents, as applicable, there can be no assurance that as our operations evolve, our efforts to comply or to remain in compliance will be fully successful.
Further, unauthorized access, loss or dissemination of sensitive personal data, such as health information, could also disrupt our operations, including our ability to conduct research and development activities, process and prepare company financial information, manage various general and administrative aspects of our business and damage our reputation, any of which could adversely affect our business and reputation. In addition, there can be no assurance that we will promptly detect any such disruption or security breach, if at all. To the extent that any disruption or security breach were to result in a loss of or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development of our products could be delayed.
There can be no assurance that we will not be subject to cybersecurity incidents that bypass our security measures, impact the integrity, availability or privacy of health information or other data subject to privacy laws, or disrupt our information systems, devices or business, including our ability to deliver services to our customers. As a result, cybersecurity, physical security and the continued development and enhancement of our controls, processes and practices designed to protect our enterprise, information systems and data from attack, damage or unauthorized access remain a priority for us. As cyber threats continue to evolve, we may be required to expend significant additional resources to continue to modify or enhance our protective measures or to investigate and remediate any cybersecurity vulnerabilities. In addition to risks affecting our own systems, we could also be negatively impacted by a security breach impacting a third party’s network and affecting us, such as our third-party vendors and service providers. In the event that these third parties do not adequately safeguard our data, cybersecurity incidents could result and negatively impact our business, operations, and financial results.
Broad-based domestic and international government initiatives to reduce spending, particularly those related to healthcare costs, may reduce reimbursement rates for medical procedures, or make it more difficult for customers to purchase our products and services, all of which could adversely affect our business.
Healthcare reforms, changes in healthcare policies and changes to third-party coverage and reimbursements, including legislation enacted reforming the U.S. healthcare system and both domestic and foreign healthcare cost containment legislation, and any future changes to such legislation, may affect demand for our products and services and may have a material adverse effect on our financial condition and results of operations. The ongoing implementation of the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, (the “ACA”) in the United States, as well as state-level healthcare reform proposals, could reduce medical procedure volumes and impact the demand for medical device products or the prices at which we can sell products. The impact of healthcare reform legislation, and practices including price regulation, competitive pricing, comparative effectiveness of therapies, technology assessments, and managed care arrangements are uncertain. There can be no assurance that current levels of reimbursement will not be decreased in the future, or that future legislation, regulation, or reimbursement policies of third parties will not adversely affect the demand for our products and services or our ability to sell products and provide services on a profitable basis. The adoption of significant changes to the healthcare system in the United States, the EEA or other jurisdictions in which we may market our products and services, could limit the prices we are able to charge for our products and services or the amounts of reimbursement available for our products and services, could limit the acceptance and availability of our products and services, reduce medical procedure volumes and increase operational and other costs.
In addition, as a result of various judicial and Congressional challenges to certain aspects of the ACA, certain sections of the ACA have not been fully implemented or were effectively repealed. Following several years of litigation in the federal courts, in June 2021, the U.S. Supreme Court upheld the ACA when it dismissed a legal challenge to its constitutionality. Further legislative and regulatory changes under the ACA remain possible, but it is unknown what form any such changes or any law would take, and how or whether it may affect the medical device industry as a whole or our business in the future. In addition to the ACA, there have been and will likely continue to be other federal and state changes that affect the provision of healthcare goods and services in the United States. While we are unable to predict what changes may ultimately be enacted, to the extent that future changes affect how our products and services are paid for and reimbursed by government and private payers, our business could be adversely impacted. Moreover, complying with any new legislation or reversing changes implemented under the ACA could be time-intensive and expensive, resulting in a material adverse effect on the business.
Furthermore, many of our customers are healthcare facilities that are subject to state Determination of Need, or DoN, laws. The purpose of DoN laws is generally to promote competition and cost containment in healthcare. Under state DoN laws, healthcare facilities are required to complete an extensive review and approval process before making substantial capital expenditures. While our Swoop® system is generally more affordable than traditional MRI systems, in some states healthcare facilities are required to complete such processes in connection with their potential acquisition of a Swoop® system, which can result in delays in or decisions not to complete the sale process, resulting in an adverse impact on our business.
52
Risks Related to our Intellectual Property
If we are unable to obtain and maintain and enforce sufficient intellectual property protection for our products and technology, or if the scope of the intellectual property protection obtained is not sufficiently broad, our competitors could develop and commercialize products similar or identical to ours, and our ability to successfully commercialize our products may be impaired.
We rely on patent protection as well as trademark, copyright, trade secret and other intellectual property right protection and contractual restrictions to protect our proprietary products and technologies, all of which provide limited protection and may not adequately protect our rights or permit us to gain or keep any competitive advantage. If we fail to obtain, maintain and sufficiently enforce our intellectual property, third parties may be able to compete more effectively against us. In addition, we may incur substantial litigation costs in our attempts to recover damages or restrict use of our intellectual property.
To the extent our intellectual property offers inadequate protection, or is found to be invalid or unenforceable, we would be exposed to a greater risk of direct competition. If our intellectual property does not provide adequate coverage against our competitors’ products, our competitive position could be adversely affected, as could our business, financial condition, results of operations and prospects. Both the patent application process and the process of managing patent and other intellectual property disputes can be time-consuming and expensive.
Our success depends in large part on our ability to obtain and maintain protection of the intellectual property we may own solely or jointly with, or license from, third parties, particularly patents, in the United States and other countries directed to our products and technologies. We apply for patents covering our products and technologies and uses thereof, as we deem appropriate. However, obtaining and enforcing patents is costly, time-consuming and complex, and we may fail to apply for patents on important products and technologies in a timely fashion or at all, or we may fail to apply for patents in potentially relevant jurisdictions. We may not be able to file and prosecute all necessary or desirable patent applications, or maintain, enforce and license any patents that may issue from such patent applications, at a reasonable cost or in a timely manner or in all jurisdictions. It is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. Moreover, we may not develop additional proprietary products, methods and technologies that are patentable. We may not have the right to control the preparation, filing and prosecution of patent applications, or to maintain the rights to patents licensed from or to third parties. Therefore, these patents and applications may not be prosecuted, obtained and enforced by such third parties in a manner consistent with the best interests of our business.
In addition, the patent position of life sciences and medical technology companies generally is highly uncertain, involves complex legal and factual questions, and has been the subject of much litigation in recent years. Changes in either the patent laws or in interpretations of patent laws in the United States or other countries or regions may diminish the value of our intellectual property. As a result, the issuance, scope, validity, enforceability, and commercial value of our patent rights presents some degree of uncertainty. It is possible that some of our pending patent applications will not result in issued patents in a timely fashion or at all, and even if patents are granted, they may not provide a basis for intellectual property protection of commercially viable products or services, may not provide any competitive advantages, or may be challenged, narrowed and/or invalidated by third parties. There exists some degree of uncertainty over the breadth of claims that may be allowed or enforced in our patents or in third-party patents. It is possible that third parties will attempt to design around our current or future patents such that we cannot prevent such third parties from using similar technologies and commercializing similar products to compete with us. Some of our owned or licensed patents or patent applications may be challenged at a future point in time and we may not be successful in defending any such challenges made against our patents or patent applications. Any successful third-party challenge to our patents could result in the narrowing, unenforceability or invalidity of such patents and increased competition to our business. The outcome of patent litigation or other proceedings can be uncertain, and any attempt by us to enforce our patent rights against others or to challenge the patent rights of others may not be successful, or, regardless of success, may take substantial time and result in substantial cost, and may divert our efforts and attention from other aspects of our business. Any of the foregoing events could have a material adverse effect on our business, financial condition and results of operations.
The U.S. law relating to the patentability of certain inventions in the life sciences and medical technology industry is uncertain and rapidly changing, which may adversely impact our existing patents or our ability to obtain patents in the future.
Changes in either the patent laws or interpretation of the patent laws in the United States or in other jurisdictions could increase the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of issued patents. For instance, under the Leahy-Smith America Invents Act (the “America Invents Act”), enacted in September 2011, the United States transitioned to a first inventor to file system in which, assuming that other requirements for patentability are met, the first inventor to file a patent application is entitled to the patent on an invention regardless of whether a third party was the first to invent the claimed invention. These changes include allowing third-party submission of prior art to the United States Patent and Trademark Office (“USPTO”) during patent prosecution and additional procedures to challenge the validity of a patent through USPTO administered post-grant proceedings, including post-grant review, inter partes review, and derivation proceedings. The America Invents Act and its
53
implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business, financial condition, results of operations and prospects.
Various courts, including the U.S. Supreme Court, have rendered decisions that impact the scope of patentability of certain inventions or discoveries relating to life sciences and medical technology. Specifically, these decisions stand for the proposition that patent claims that recite laws of nature, natural phenomena, and abstract ideas are not themselves patentable unless those patent claims have sufficient additional features that provide practical assurance that the processes are genuine inventive applications of those laws, phenomena, and abstract ideas rather than patent drafting efforts designed to monopolize the law of nature, natural phenomenon, or abstract idea itself. What constitutes a “sufficient” additional feature is somewhat uncertain. Furthermore, in view of these decisions, since December 2014, the USPTO has published and continues to publish revised guidelines for patent examiners to apply when examining process claims for patent eligibility.
In addition, U.S. Supreme Court rulings have narrowed the scope of patent protection available in certain circumstances and weakened the rights of patent owners in certain situations. In addition to some degree of uncertainty with regard to the Company’s ability to obtain patents in the future, this combination of events has created a degree of uncertainty with respect to the value of patents, once obtained. Depending on relevant laws enacted by the U.S. Congress, and decisions by the federal courts and the USPTO, the laws and regulations governing patents could change in unpredictable ways that may have a material adverse effect on our ability to obtain new patents and to defend and enforce our existing patents and patents that we might obtain in the future.
Our patent portfolio may be negatively impacted by current uncertainties in the state of the law, new court rulings or changes in guidance or procedures issued by the USPTO or other similar patent offices around the world. From time to time, the U.S. Supreme Court, other federal courts, the U.S. Congress or the USPTO may change the standards of patentability, scope and validity of patents within the life sciences and medical technology and any such changes, or any similar adverse changes in the patent laws of other jurisdictions, could have a negative impact on our business, financial condition, prospects and results of operations.
We may not be able to protect our intellectual property rights throughout the world.
The laws of some foreign countries do not offer intellectual property rights to the same extent as the laws of the United States, and we and our licensors may encounter difficulties in obtaining, enforcing and defending such rights in foreign jurisdictions. Consequently, we and our licensors may not be able to prevent third parties from practicing our or our licensors’ inventions in some or all countries outside the United States, or from selling or importing products made using our or our licensors’ inventions in other jurisdictions. Competitors and other third parties may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and technologies and may also export infringing products to territories where we have patent protection, but enforcement practices or laws are not as strong as those in the United States. These products may compete with our products. Our and our licensors’ patents or other intellectual property rights may not be effective or sufficient to prevent them from competing. In addition, certain countries have compulsory licensing laws under which a patent owner may be compelled to grant licenses to other parties. Furthermore, many countries limit the enforceability of patents against other parties, including government agencies or government contractors. In these countries, the patent owner may have limited remedies, which could materially diminish the value of any patents.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain other countries are not as developed or as favorable as the United States in the enforcement of patents and other intellectual property rights, which could make it difficult for us to stop the misappropriation or other violations of our intellectual property rights including infringement of our patents in such countries. The legal systems in certain countries may also favor state-sponsored companies or companies headquartered in particular jurisdictions over our first-in-time patents and other intellectual property rights. The absence of harmonized intellectual property protection laws and effective enforcement makes it difficult to ensure consistent respect for patent, trade secret, and other intellectual property rights on a worldwide basis. As a result, it is possible that we will not be able to enforce our rights against third parties that misappropriate our proprietary technology in those countries.
Proceedings to enforce our or our licensors’ patent rights in foreign jurisdictions could result in substantial cost and divert our efforts and attention from other aspects of our business, could put us and our licensors’ patents at risk of being invalidated or interpreted narrowly and our and our licensors’ patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We and our licensors may not prevail in any lawsuits that we or our licensors initiate, or that are initiated against us or our licensors, and the damages or other remedies awarded, if any, may not be commercially meaningful. In addition, changes in the law and legal decisions by courts in the United States and foreign countries may affect our ability to obtain adequate protection for our products, services and other technologies and the enforcement of intellectual property. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license. Any of the foregoing events could have a material adverse effect on our business, financial condition, results of operations and prospects.
54
Issued patents covering our products could be found invalid or unenforceable if challenged.
Our owned and licensed patents and patent applications may be subject to validity, enforceability and priority disputes. The issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability. Some of our patents or patent applications (including licensed patents and patent applications) may be challenged at a future point in time in opposition, derivation, reexamination, inter partes review, post-grant review or interference or other similar proceedings, as applicable. Any successful third-party challenge to our patents in this or any other proceeding could result in the unenforceability or invalidity of such patents, which may lead to increased competition to our business, which could have a material adverse effect on our business, financial condition, results of operations and prospects. In addition, if we or our licensors initiate legal proceedings against a third party to enforce a patent covering our products, the defendant could counterclaim that such patent covering our products, as applicable, is invalid and/or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity or unenforceability are commonplace. There are numerous grounds upon which a third party can assert invalidity or unenforceability of a patent. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness or non-enablement. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent intentionally withheld relevant information from the relevant patent office, or knowingly made a misleading statement, during prosecution. Third parties may also raise similar claims before administrative bodies in the United States or abroad, even outside the context of litigation. Such mechanisms include ex parte re-examination, inter partes review, post-grant review, derivation and equivalent proceedings in non-U.S. jurisdictions, such as opposition proceedings. Such proceedings could result in revocation of or amendment to our patents in such a way that they no longer cover and protect our products. With respect to the validity of our patents, for example, we cannot be certain that there is no invalidating prior art of which we, our licensors, our patent counsel and the patent examiner were unaware during prosecution. The outcome following legal assertions of invalidity and unenforceability during patent litigation is unpredictable. If a defendant or other third party were to prevail on a legal assertion of invalidity or unenforceability, we would lose at least part, and perhaps all, of the patent protection on certain aspects of our products and technologies, which could have a material adverse effect on our business, financial condition, results of operations and prospects. In addition, if the breadth or strength of protection provided by our patents and patent applications is threatened, regardless of the outcome, it could dissuade companies from collaborating with us to license intellectual property, or develop or commercialize current or future products.
We may not be aware of all third-party intellectual property rights potentially relating to our products. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until approximately 18 months after filing or, in some cases, not until such patent applications issue as patents. We might not have been the first to make the inventions covered by each of our pending patent applications and we might not have been the first to file patent applications for these inventions. To determine the priority of these inventions, we may have to participate, as applicable, in interference proceedings, derivation proceedings or other post-grant proceedings declared by the USPTO, or other similar proceedings in non-U.S. jurisdictions that could result in substantial cost to us and the loss of valuable patent protection. The outcome of such proceedings is uncertain. No assurance can be given that other patent applications will not have priority over our patent applications. In addition, changes to the patent laws of the United States allow for various post-grant opposition proceedings that have not been extensively tested, and their outcome is therefore uncertain. Furthermore, if third parties bring these proceedings against our patents, regardless of the merit of such proceedings and regardless of whether we are successful, we could experience significant costs and our management may be distracted.
Any of the foregoing events could have a material adverse effect on our business, financial condition, results of operations and prospects.
If we are unable to protect the confidentiality of our trade secrets, the value of our technology could be materially adversely affected and our business could be harmed.
We rely heavily on trade secrets and confidentiality agreements to protect our unpatented know-how, technology and other confidential proprietary information, and to maintain our competitive position. However, trade secrets and know-how can be difficult to protect. In particular, we anticipate that with respect to our technologies, these trade secrets and know-how will over time be disseminated within the industry through independent development, the publication of journal articles describing the methodology, and the movement of personnel from academic to industry scientific positions.
In addition to pursuing patents on our technology, we take steps to protect our intellectual property and proprietary technology by entering into agreements, including confidentiality agreements, non-disclosure agreements and intellectual property assignment agreements, with our employees, consultants, academic institutions, corporate partners and, when needed, our advisers. However, we cannot be certain that such agreements have been entered into with all relevant parties, and we cannot be certain that our trade secrets and other confidential proprietary information will not be disclosed or that competitors or other third parties will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. For example, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Such agreements may not be enforceable or may not provide meaningful protection for
55
our trade secrets or other proprietary information in the event of unauthorized use or disclosure or other breaches of the agreements, and we may not be able to prevent such unauthorized disclosure, which could materially and adversely impact our ability to establish or maintain a competitive advantage in the market, and our business, financial condition, results of operations and prospects.
Monitoring unauthorized disclosure is difficult, and we do not know whether the steps we have taken to prevent such disclosure are, or will be, adequate. If we were to enforce a claim that a third party had wrongfully obtained and was using our trade secrets, it would be expensive and time-consuming, it could distract our personnel, and the outcome would be unpredictable. In addition, courts outside the United States may be less willing to protect trade secrets or may not recognize certain claims of intellectual property infringement.
We also seek to preserve the integrity and confidentiality of our confidential proprietary information by maintaining physical security of our premises and physical and electronic security of our information technology systems, but it is possible that these security measures could be breached. If any of our confidential proprietary information were to be lawfully obtained or independently developed by a competitor or other third party, absent patent and copyright protection, we would have no right to prevent such competitor from using that technology or information to compete with us, which could harm our competitive position. Competitors or third parties could purchase our products and attempt to replicate some or all of the competitive advantages we derive from our development efforts, design around our protected technology, develop their own competitive technologies that fall outside the scope of our intellectual property rights or independently develop our technologies without reference to our trade secrets. If any of our trade secrets were to be disclosed to or independently discovered by a competitor or other third party, it could materially and adversely affect our business, financial condition, results of operations and prospects.
We may be subject to claims challenging the inventorship and ownership of our patents and other intellectual property.
We or our licensors may be subject to claims that former employees, collaborators or other third parties have an interest in our owned or in-licensed patents, trade secrets or other intellectual property as an inventor or co-inventor. For example, we or our licensors may have inventorship disputes arise from alleged inventors such as employees, consultants, advisors or others who are involved in developing our products, some of whom may have conflicting intellectual property ownership obligations. In addition, counterparties to our consulting, sponsored research, software development and other agreements may assert that they have an ownership interest in intellectual property developed under such arrangements. In particular, certain software development agreements pursuant to which certain third parties have developed parts of our proprietary software may not include provisions that expressly assign to us ownership of all intellectual property developed for us by such third parties. Furthermore, certain of our sponsored research agreements pursuant to which we provide certain research services for third parties do not assign to us all intellectual property developed under such agreements. As such, we may not have the right to use all such developed intellectual property under such agreements, we may be required to obtain licenses from third parties and such licenses may not be available on commercially reasonable terms or at all, or may be non-exclusive. If we are unable to obtain such licenses and such licenses are necessary for the development, manufacture and commercialization of our products and technologies, we may need to cease the development, manufacture or commercialization of our products and technologies. Litigation may be necessary to defend against these and other claims challenging inventorship of our or our licensors’ ownership of our owned or in-licensed patents, trade secrets or other intellectual property. If we or our licensors fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights. In such an event, we may be required to obtain licenses from third parties and such licenses may not be available on commercially reasonable terms or at all, or may be non-exclusive. If we are unable to obtain and maintain such licenses, we may need to cease the development, manufacture or commercialization of our products and technologies. Even if we are successful in defending against such claims, litigation could result in substantial costs and loss of time and be a distraction to management and other employees, and certain customers or partners may defer engaging with us until the particular dispute is resolved. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operations and prospects.
We may not be able to protect and enforce our trademarks and trade names or build name recognition in our markets of interest thereby harming our competitive position.
The registered or unregistered trademarks or trade names that we own may be challenged, infringed, circumvented, declared generic or otherwise fail to function as a mark, lapsed or determined to be confusingly similar to or dilutive of other marks. We may not be able to protect our rights in these trademarks and trade names, which we need in order to build name recognition. In addition, third parties have filed, and may in the future file, for registration of trademarks similar or identical to our trademarks, thereby impeding our ability to build brand identity and possibly leading to consumer confusion. If such third parties were to succeed in registering or developing common law rights in any other trademarks that are similar or identical to our trademarks, and if we are not successful in challenging such rights and defending against challenges to Company’s trademarks, we may not be able to use such trademarks to develop brand recognition of our technologies, products or services. In addition, there could be potential trade name or trademark infringement claims brought by owners of other registered trademarks or trademarks that incorporate variations of our registered or unregistered trademarks or trade names. Further, we have and may in the future enter into agreements with owners of such third-party trade names or trademarks to avoid potential trademark litigation which may limit our ability to use our trade names or trademarks in certain fields of business. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names,
56
then we may not be able to compete effectively, and our business, financial condition, results of operations and prospects may be adversely affected. Our efforts to enforce or protect our proprietary rights related to trademarks, trade secrets, domain names, copyrights or other intellectual property may be ineffective and could result in substantial costs and diversion of resources. Any of the foregoing events could have a material adverse effect on our business, financial condition and results of operations.
Patent terms may be inadequate to protect our competitive position on our products for an adequate amount of time.
Patents have a limited lifespan. In the United States, if all maintenance fees are timely paid, the natural expiration of a utility patent is generally 20 years from its earliest U.S. non-provisional filing date. While extensions may be available, the life of a patent, and the protection it affords, is limited. In the United States, a patent’s term may, in certain cases, be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the USPTO in examining and granting a patent, or may be shortened if a patent is terminally disclaimed over a commonly owned patent or a patent naming a common inventor and having an earlier expiration date. Even if patents covering our products are obtained, once the patent life has expired, we may be open to competition from competitive products. If one of our products requires extended development, testing and/or regulatory review, patents protecting such products might expire before or shortly after such products are commercialized. As a result, our owned and licensed patent portfolio may not provide us with sufficient rights to exclude others from commercializing products similar or identical to our products, which could have a material adverse effect on our business, financial condition and results of operations.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed to us alleged trade secrets of their other clients or former employers, which could subject us to costly litigation.
As is common in the life sciences and medical industry, we engage the services of consultants and independent contractors to assist us in the development of our products. Many of these consultants and independent contractors were previously employed at or may have previously or may be currently providing consulting or other services to, universities or other technology, medical device, biotechnology or pharmaceutical companies, including our competitors or potential competitors. We may become subject to claims that we, a consultant or an independent contractor inadvertently or otherwise used or disclosed trade secrets or other information proprietary to their former employers or their former or current clients. We may similarly be subject to claims stemming from similar actions of an employee, such as one who was previously employed by another company, including a competitor or potential competitor. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to our management team. If we are not successful, we could lose access or exclusive access to valuable intellectual property.
We may become involved in lawsuits to defend against third-party claims of infringement, misappropriation or other violations of intellectual property rights or to protect or enforce our intellectual property, any of which could be expensive, time consuming and unsuccessful, and may prevent or delay our development and commercialization efforts.
Our commercial success depends in part on our ability and the ability of future collaborators to develop, manufacture, market and sell our product and use our products and technologies without infringing, misappropriating or otherwise violating the intellectual property rights of third parties. There is a substantial amount of litigation involving patents and other intellectual property rights in the life sciences and medical technology sector, as well as administrative proceedings for challenging patents, including interference, derivation, inter partes review, post grant review, and reexamination proceedings before the USPTO, or oppositions and other comparable proceedings in foreign jurisdictions. We may be exposed to, or threatened with, future litigation by third parties having patent or other intellectual property rights alleging that our products, manufacturing methods, software and/or technologies infringe, misappropriate or otherwise violate their intellectual property rights. Numerous issued patents and pending patent applications that are owned by third parties exist in the fields in which we are developing our products and technologies. It is not always clear to industry participants, including us, the claim scope that may issue from pending patent applications owned by third parties or which patents cover various types of products, technologies or their methods of use or manufacture. Thus, because of the large number of patents issued and patent applications filed in our fields, there may be a risk that third parties, including our competitors, may allege they have patent rights encompassing our products, technologies or methods and that we are employing technology protected by such patent rights without authorization.
If third parties, including our competitors, believe that our products or technologies infringe, misappropriate or otherwise violate their intellectual property rights, such third parties may seek to enforce against us their intellectual property rights, including patent rights, by filing against us an intellectual property-related lawsuit, including a patent infringement lawsuit. Even if we believe third-party intellectual property claims are without merit, there is no assurance that a court would find in our favor on questions of infringement, validity, enforceability, or priority. If any third parties were to assert these or any other patents against us and we are unable to successfully defend against any such assertions, we may be required, including by court order, to cease the development and commercialization of the infringing products or technology and we may be required to redesign such products and technologies so they do not infringe such patents, which may not be possible or may require substantial monetary expenditures and time. We could also be required to pay damages, which could be significant, including treble damages and attorneys’ fees if we are found to have
57
willfully infringed such patents. We could also be required to obtain a license to such patents in order to continue the development and commercialization of the infringing product or technology. However, such a license may not be available on commercially reasonable terms or at all, including because certain of these patents may be held by or exclusively licensed to our competitors. Even if such a license were available, it may require substantial payments or cross-licenses under our intellectual property rights, and it may only be available on a nonexclusive basis, in which case third parties, including our competitors, could use the same licensed intellectual property to compete with us. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operation and prospects.
We may choose to challenge, including in connection with any allegation of patent infringement by a third party, the patentability, validity, ownership or enforceability of any third-party patent that we believe may have applicability in our field, and any other third-party patent that may at some future time possibly be asserted against us. Such challenges may be brought either in court or by requesting that the USPTO, European Patent Office (“EPO”), or other foreign patent offices review the patent claims, such as in an ex-parte reexamination, inter partes review, post-grant review proceeding or opposition proceeding or other similar proceedings. However, there can be no assurance that any such challenge by us or any third party will be successful. Even if such proceedings are successful, these proceedings are expensive and may consume our time or other resources, distract our management and technical personnel, and the costs of these opposition proceedings could be substantial. There can be no assurance that our defenses of non-infringement, invalidity or unenforceability will succeed.
Third parties, including our competitors, could be infringing, misappropriating or otherwise violating our solely owned and/or in-licensed intellectual property rights. Monitoring unauthorized use of intellectual property is difficult and costly. We may not be able to detect unauthorized use of, or take appropriate steps to enforce, our intellectual property rights. From time to time, we seek to analyze our competitors’ products and services, and may in the future seek to enforce our rights based on potential infringement, misappropriation or violation of our intellectual property. However, the steps we take to protect our intellectual property rights may not be adequate to enforce our rights against such infringement, misappropriation or violation of our intellectual property. Any inability to meaningfully enforce our intellectual property rights could harm our ability to compete and reduce demand for our products and technologies.
Litigation proceedings may be necessary for us to enforce our patent and other intellectual property rights. In any such proceeding, a court may refuse to stop the other party from using the technology at issue on the grounds that our owned and in-licensed patents do not cover the technology in question. Further, in such a proceeding, the defendant could counterclaim that our intellectual property is invalid or unenforceable and the court may agree, in which case we could lose valuable intellectual property rights, which could allow third parties to commercialize technology or products similar to ours and compete directly with us, without payment to us. Alternatively or additionally, such a proceeding could result in requiring us to license rights from the prevailing party in order to be able to manufacture or commercialize our products without infringing such party’s intellectual property rights, and if we are unable to obtain such a license, we may be required to cease commercialization of our products and technologies, any of which could have a material adverse effect on our business, financial condition, results of operations and prospects. The outcome in any such proceeding is somewhat unpredictable.
Regardless of whether we are defending against or asserting an intellectual property-related claim in an intellectual property-related proceeding that may be necessary in the future, and regardless of outcome, substantial costs and diversion of resources may result which could have a material adverse effect on our business, financial condition, results of operations and prospects. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, there could be public announcements of the results of hearings, motions, or other interim proceedings or developments, and if securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on the price of our Class A common stock. Some of our competitors and other third parties may be able to sustain the costs of such litigation or proceedings more effectively than we can because of their greater financial resources and more mature and developed intellectual property portfolios. We may not have sufficient financial or other resources to adequately conduct these types of litigation or proceedings. Any of the foregoing, or any uncertainties resulting from the initiation and continuation of any litigation, could have a material adverse effect on our business, financial condition, results of operations and prospects. Claims that we have misappropriated the confidential information or trade secrets of third parties could have a similar adverse effect on our business, financial condition, results of operations and prospects.
Obtaining and maintaining our patent protection depends on compliance with various required procedures, document submissions, fee payments and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance fees, renewal fees, annuity fees and various other governmental fees on patents and/or applications will be due to be paid to the USPTO and various governmental patent agencies outside of the United States at several stages over the lifetime of the patents and/or applications. The USPTO and various non-U.S. governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. In certain circumstances, we
58
rely on our licensors to pay these fees due to the U.S. and non-U.S. patent agencies and to take the necessary action to comply with these requirements with respect to our licensed intellectual property. In many cases, an inadvertent lapse can be cured by payment of a late fee or by other means in accordance with the applicable rules. However, there are situations in which non-compliance can result in an irrevocable abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, our competitors may be able to enter the market without infringing our patents and this circumstance could have a material adverse effect on our business, financial condition, results of operations and prospects.
We currently rely on licenses from third parties, and in the future may rely on additional licenses from other third parties, and if we lose any of these licenses, then we may be subjected to future litigation.
We are, and may in the future become, a party to license agreements that grant us rights to use certain intellectual property, including patents and patent applications, typically in certain specified fields of use. We may need to obtain additional licenses from others to advance our research, development and commercialization activities.
Our success may depend in part on the ability of our licensors and any future licensors to obtain, maintain and enforce patent protection for our licensed intellectual property. Without protection for the intellectual property we license, other companies might be able to offer substantially identical products and technologies for sale, which could materially adversely affect our competitive business position and harm our business, financial condition, results of operations and prospects.
Our current license agreements impose, and future agreements may impose, various diligence, commercialization, milestone payment, royalty, insurance and other obligations on us and require us to meet development timelines, or to exercise commercially reasonable efforts to develop and commercialize licensed products, in order to maintain the licenses. If we fail to comply with these obligations, our licensor(s) may have the right to terminate our license, in which event we would not be able to develop or market products or technology covered by the licensed intellectual property. Any of the foregoing could have a material adverse effect on our competitive position, business, financial conditions, results of operations and prospects.
Moreover, disputes may also arise between us and our licensors regarding intellectual property subject to a license agreement, including:
If we do not prevail in such disputes, we may lose any or all of our rights under such license agreements, experience significant delays in the development and commercialization of our products and technologies, or incur liability for damages, any of which could have a material adverse effect on our business, financial condition, results of operations, and prospects. In addition, we may seek to obtain additional licenses from our licensor(s) and, in connection with obtaining such licenses, we may agree to amend our existing licenses in a manner that may be more favorable to the licensor(s), including by agreeing to terms that could enable third parties, including our competitors, to receive licenses to a portion of the intellectual property that is subject to our existing licenses and to compete with our products.
In addition, the agreements under which we currently and in the future license intellectual property or technology from third parties are complex and certain provisions in such agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could narrow what we believe to be the scope of our rights to the relevant intellectual property or technology or increase what we believe to be our financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business, financial condition, results of operations and prospects. Moreover, if disputes over intellectual property that we have licensed prevent or impair our ability to maintain our current licensing arrangements on commercially acceptable terms, we may be unable to successfully develop and commercialize any affected products or services, which could have a material adverse effect on our business, financial condition, results of operations and prospects.
59
Absent the license agreements, we may infringe patents subject to those agreements, and if the license agreements are terminated, we may be subject to litigation by the licensor. Litigation could result in substantial costs and distract our management. If we do not prevail, we may be required to pay damages, including treble damages, attorneys’ fees or costs and expenses and royalties, which could adversely affect our ability to offer products or services, our ability to continue operations and our business, financial condition, results of operations and prospects.
If we cannot license rights to use technologies on reasonable terms, we may not be able to commercialize new products in the future.
We may identify third-party technology that we may need to license or acquire in order to develop or commercialize our products or technologies. However, we may be unable to secure such licenses or acquisitions. The licensing or acquisition of third-party intellectual property rights is a competitive area, and several more established companies may pursue strategies to license or acquire third-party intellectual property rights that we may consider attractive or necessary. These established companies may have a competitive advantage over us due to their size, capital resources and greater clinical development and commercialization capabilities. In addition, companies that perceive us to be a competitor may be unwilling to assign or license rights to us.
We also may be unable to license or acquire third-party intellectual property rights on terms that would allow us to make an appropriate return on our investment or at all. In return for the use of a third party’s technology, we may agree to pay the licensor royalties based on sales of our products or services. Royalties are a component of cost of products or technologies and affect the margins on our products. We may also need to negotiate licenses to patents or patent applications before or after introducing a commercial product. We may not be able to obtain necessary licenses to patents or patent applications, and our business may suffer if we are unable to enter into the necessary licenses on acceptable terms or at all, if any necessary licenses are subsequently terminated, if the licensor fails to abide by the terms of the license or fails to prevent infringement by third parties, or if the licensed intellectual property rights are found to be invalid or unenforceable.
Certain of our future owned and in-licensed patents may be, subject to a reservation of rights by one or more third parties, including government march-in rights, that may limit our ability to exclude third parties from commercializing products similar or identical to ours.
In addition, our owned and in-licensed patents may be subject to a reservation of rights by one or more third parties. When new technologies are developed with government funding, in order to secure ownership of such patent rights, the recipient of such funding is required to comply with certain government regulations, including timely disclosing the inventions claimed in such patent rights to the U.S. government and timely electing title to such inventions. Any failure to timely elect title to such inventions may permit the U.S. government to, at any time, take title to such inventions. Additionally, the U.S. government generally obtains certain rights in any resulting patents, including a non-exclusive royalty-free license authorizing the government to use the invention or to have others use the invention on its behalf. If the government decides to exercise these rights, it is not required to engage us as its contractor in connection with doing so. In addition, these rights may permit the U.S. government to disclose our confidential information to third parties and to exercise march-in rights to use or allow third parties to use our licensed technology free of charge. The U.S. government can exercise its march-in rights if it determines that action is necessary because we fail to achieve practical application of the government-funded technology, because action is necessary to alleviate health or safety needs, to meet requirements of federal regulations, or to give preference to U.S. industry. In addition, our rights in such inventions may be subject to certain requirements to manufacture products embodying such inventions in the United States. Any exercise by the government of any of the foregoing rights could have a material adverse effect on our business, financial condition, results of operations and prospects.
Our products contain third-party open source software components and failure to comply with the terms of the underlying open source software licenses could restrict our ability to sell our products and provide third parties access to our proprietary software.
Our products may contain software licensed by third parties under open source software licenses. Use and distribution of open source software may entail greater risks than use of third-party commercial software, as open source software licensors generally do not provide warranties or other contractual protections regarding infringement claims or the quality of the code. Some open source software licenses contain requirements that the licensee make its source code publicly available if the licensee creates modifications or derivative works using the open source software, depending on the type of open source software the licensee uses and how the licensee uses it. If we combine our proprietary software with open source software in a certain manner, we could, under certain open source software licenses, be required to release the source code of our proprietary software to the public for free. This would allow our competitors and other third parties to create similar products with less development effort and time and ultimately could result in a loss of our product sales and revenue, which could have a material adverse effect on our business, financial condition, results of operations and prospects. In addition, some companies that use third-party open source software have faced claims challenging their use of such open source software and their compliance with the terms of the applicable open source license. We may be subject to suits by third parties claiming ownership of what we believe to be open source software, or claiming non-compliance with the applicable open source licensing terms. Use of open source software may also present additional security risks because the public availability of such software may make it easier for hackers and other third parties to compromise or attempt to compromise our technology and systems.
60
Although we review our use of open source software to avoid subjecting our proprietary software to conditions we do not intend, the terms of many open source software licenses have not been interpreted by U.S. courts, and there is a risk that these licenses could be construed in a way that could impose unanticipated conditions or restrictions on our ability to commercialize our products and proprietary software. Moreover, our processes for monitoring and controlling our use of open source software in our products may not be effective. If we are held to have breached the terms of an open source software license, we could be subject to damages or be required to seek licenses from third parties to continue offering our products on terms that are not economically feasible, to re-engineer our products, to discontinue the sale of our products if re-engineering could not be accomplished on a timely basis, or to make generally available, in source code form, our proprietary code, any of which could adversely affect our business, financial condition, results of operations and prospects.
Intellectual property rights do not necessarily address all potential threats.
The degree of future protection afforded by our intellectual property rights is uncertain because intellectual property rights have limitations and may not adequately protect our business or permit us to maintain our competitive advantage. For example:
Should any of these events occur, they could materially adversely affect our business, financial condition, results of operations and prospects.
Litigation Risks
We face the risk of product liability claims and may be subject to damages, fines, penalties and injunctions, among other things.
Our business exposes us to the risk of product liability claims that are inherent in the testing, manufacturing and marketing of medical devices, including those which may arise from the misuse (including system hacking or other unauthorized access by third parties to our systems) or malfunction of, or design flaws in, our hardware and software products. This liability may vary based on the FDA classification associated with our devices and with the state law governing product liability standards applied to specification developers and/or manufacturers in a given negligence or strict liability lawsuit. We may be subject to product liability claims if our products cause, or merely appear to have caused, an injury. Claims may be made by patients, healthcare providers or others selling our products. The risk of product liability claims may also increase if our products are subject to a product recall or seizure. Product liability claims may be brought by individuals or by groups seeking to represent a class.
61
Although we have insurance at levels that we believe to be appropriate, this insurance is subject to deductibles and coverage limitations. Our current product liability insurance may not continue to be available to us on acceptable terms, if at all, and, if available, the coverage may not be adequate to protect us against any future product liability claims. Further, if additional medical device products are approved or cleared for marketing, or if we launch additional 510(k)-exempt device products or products that are not FDA-regulated medical devices, we may seek additional insurance coverage. If we are unable to obtain insurance at an acceptable cost or on acceptable terms with adequate coverage or otherwise protect against potential product liability claims, we will be exposed to significant liabilities, which may harm our business. A product liability claim, recall or other claim with respect to uninsured liabilities or for amounts in excess of insured liabilities could result in significant costs and significant harm to our business.
We may be subject to claims against us even if the apparent injury is due to the actions of others or misuse of the device or a partner device. Healthcare providers may use our products in a manner inconsistent with the products’ labeling and that differs from the manner in which they were used in clinical studies and authorized by the FDA. Off-label use of products by healthcare providers is common, and any such off-label use of our products could subject us to additional liability, or require design changes to limit this potential off-label use once discovered. Defending a suit, regardless of merit, could be costly, could divert management attention and might result in adverse publicity, which could result in the withdrawal of, or result in reduced acceptance of, our products in the market.
Additionally, we have entered into various agreements where we indemnify third parties for certain claims relating to our products. These indemnification obligations may require us to pay significant sums of money for claims that are covered by these indemnification obligations. We are not currently subject to any product liability claims; however, any future product liability claims against us, regardless of their merit, may result in negative publicity about us that could ultimately harm our reputation and could have a material adverse effect on our business, financial condition, results of operations and prospects.
Risks Related to Our Securities and to Being a Public Company
If we experience material weaknesses in our internal control over financial reporting or otherwise fail to maintain an effective system of internal controls in the future, we may not be able to report our financial condition or results of operations accurately or in a timely manner and we may be unable to maintain compliance with applicable stock exchange listing requirements, which may adversely affect investor confidence in us and, as a result, materially and adversely affect our business and the value of our Class A common stock.
In connection with Legacy Hyperfine’s and Liminal’s combined financial statement close process for the years ended December 31, 2020 and 2019, we identified a material weakness in our internal control over financial reporting. We outsourced our accounting and financial reporting to 4Catalyzer Corporation (“4Catalyzer”) and as of and during the years ended December 31, 2020 and 2019, did not have our own finance function to appropriately perform the supervision and review of the information received from 4Catalyzer and assess its reasonableness and accuracy.
In addition, HealthCor previously recorded a portion of its Class A ordinary shares subject to possible redemption in permanent equity. Notwithstanding the presence of maximum redemption thresholds or charter provisions common in special purpose acquisition companies ("SPACs") that provide a limitation on redemptions that would cause a SPAC’s net tangible assets to be less than $5,000,001, in accordance with SEC Staff guidance on redeemable equity instruments, ASC 480-10-S99, “Distinguishing Liabilities from Equity”, and EITF Topic D-98, “Classification and Measurement of Redeemable Securities”, and, according to the SEC Staff communications with certain independent auditors, redemption provisions not solely within the control of the issuing company require ordinary shares subject to redemption to be classified outside of permanent equity. Although HealthCor did not specify a maximum redemption threshold in its Articles and Restated Memorandum and Articles of Association (the “HealthCor Articles”), the HealthCor Articles provided that HealthCor could not redeem its public shares in an amount that would cause its net tangible assets to be less than $5,000,001. In light of the recent SEC Staff communications with certain independent auditors, HealthCor’s management re-evaluated the effectiveness of its disclosure controls and procedures as of September 30, 2021. Based upon that evaluation, HealthCor concluded that the misclassification of the Class A ordinary shares was quantitatively material to individual line items within the balance sheet. This resulted in a restatement of the initial carrying value of the Class A ordinary shares subject to possible redemption, with the offset recorded to additional paid-in capital (to the extent available), accumulated deficit and ordinary shares.
The foregoing represented material weaknesses in our internal controls over financial reporting. A “material weakness” is a deficiency, or a combination of deficiencies, in internal control over financial reporting such that there is a reasonable possibility that a material misstatement of our annual or interim financial statements will not be prevented, or detected and corrected on a timely basis.
During the years ended December 31, 2023 and 2022, we undertook remediation measures related to the foregoing material weakness in internal control over financial reporting. In response to these material weaknesses, our management has expended a substantial amount of effort and resources for the remediation of material weaknesses in internal control over financial reporting. Our management developed a remediation plan, which included the hiring of accounting and finance resources, including the Chief
62
Administrative Officer and Chief Financial Officer and Vice President, Controller with technical public company accounting and financial reporting experience, as well as other team members. We also have access to accounting training, literature, research materials and increased communication among our personnel and outsourced third-party professionals with whom we may consult regarding the application of complex accounting transactions. Our remediation plan has been accomplished over time to achieve our objectives. We believe these actions are sufficient to remediate the identified material weaknesses and strengthen our internal control over financial reporting. During the second quarter of 2023, we completed testing of the operating effectiveness of the controls and have concluded that the material weaknesses have been remediated as of June 30, 2023. Based on these measures, we believe that the previously reported material weaknesses have been remediated. However, completion of remediation procedures for the material weaknesses does not provide assurance that our process and controls will continue to operate properly or that our financial statements will be free from error.
If other material weaknesses or control deficiencies occur in the future, we may be unable to report our financial results accurately on a timely basis or help prevent fraud, which could cause our reported financial results to be materially misstated and result in the loss of investor confidence or delisting and cause the market price of our shares of Class A common stock to decline. We cannot assure you that the initiatives we have taken to date, or any initiatives we may take in the future, will be sufficient to avoid potential future material weaknesses.
We could fail to maintain the listing of our Class A common stock on Nasdaq, which could seriously harm the liquidity of our shares and our ability to raise capital or complete a strategic transaction.
The Nasdaq Stock Market has established continued listing requirements, including a requirement to maintain a minimum closing bid price of at least $1.00 per share. In December 2022, we received written notice from Nasdaq notifying us that, because the closing bid price for our Class A common stock has fallen below $1.00 per share for 30 consecutive business days, we no longer met the minimum bid price requirement for continued inclusion on The Nasdaq Global Market. In February 2023, we received a letter from Nasdaq indicating that we had regained compliance with the bid price requirement. However, there can be no assurance that we will be able to maintain compliance with the bid price requirement or other Nasdaq requirements in the future. If we are not able to maintain compliance with Nasdaq requirements, our Class A common stock may be delisted from Nasdaq, which could have a material adverse effect on us and our stockholders, including by reducing the liquidity of our shares and having a material adverse effect on our ability to raise capital or complete a strategic transaction.
Because we are a “controlled company” within the meaning of the Nasdaq listing rules, our stockholders may not have certain corporate governance protections that are available to stockholders of companies that are not controlled companies.
So long as more than 50% of the voting power for the election of our directors is held by an individual, a group or another company, we will qualify as a “controlled company” under the Nasdaq listing rules. As of February 15, 2024, Dr. Rothberg controls approximately 85% of the voting power of our outstanding capital stock. As a result, we are a “controlled company” under the Nasdaq rules and are not subject to the requirements that would otherwise require us to have: (i) a majority of our board of directors consist of independent directors; (ii) director nominees selected, or recommended for our board of directors’ selection, either by a majority of the independent directors or a nominating committee comprised solely of independent directors; and (iii) a compensation committee comprised solely of independent directors.
Dr. Rothberg may have his interest in the Company diluted due to future equity issuances or his own actions in selling shares of our Class B common stock, in each case, which could result in a loss of the “controlled company” exemption under the Nasdaq listing rules. We would then be required to comply with those provisions of the Nasdaq listing rules.
The dual class structure of our common stock has the effect of concentrating voting power with Jonathan M. Rothberg, Ph.D., the Founder of Legacy Hyperfine and Liminal and a member of our board of directors, which will limit an investor’s ability to influence the outcome of important transactions, including a change in control.
Shares of our Class B common stock have 20 votes per share, while shares of our Class A common stock have one vote per share. Dr. Rothberg and his permitted transferees hold all of the issued and outstanding shares of our Class B common stock, and as of February 15, 2024, Dr. Rothberg holds approximately 85% of the voting power of our capital stock and is able to control matters submitted to our stockholders for approval, including the election of directors, amendments of our organizational documents and any merger, consolidation, sale of all or substantially all of our assets or other major corporate transactions. Dr. Rothberg may have interests that differ from yours and may vote in a way with which you disagree and which may be adverse to your interests. This concentrated control may have the effect of delaying, preventing or deterring a change in control of the Company, could deprive our stockholders of an opportunity to receive a premium for their capital stock as part of a sale of the Company, and might ultimately affect the market price of shares of our Class A common stock. If additional shares of our Class B common stock are issued, your shares of Class A common stock and your votes may be significantly diluted.
63
Potential conflicts of interest may arise among the holders of our Class B common stock and the holders of our Class A common stock.
Dr. Rothberg and his permitted transferees hold all of our Class B common stock. As a result, conflicts of interest may arise among Dr. Rothberg, on the one hand, and the Company and holders of our Class A common stock on the other hand. Dr. Rothberg has the ability to influence our business and affairs through his ownership of the high vote shares of our common stock, his general ability to elect our board of directors, and provisions in our certificate of incorporation, as amended (the "Charter"), requiring his approval for certain corporate actions (in addition to approval by our board of directors). If the holders of our Class A common stock are dissatisfied with the performance of our board of directors, they have no ability to remove any of our directors, with or without cause.
Further, through his ability to elect our board of directors and as well as his service on our board of directors, Dr. Rothberg has the ability to influence the determination of the amount and timing of our investments and dispositions, cash expenditures, indebtedness, issuances of shares of common stock, tax liabilities and amounts of reserves.
Delaware law and provisions in our Charter and bylaws could make a takeover proposal more difficult.
Our organizational documents are governed by Delaware law. Certain provisions of Delaware law and of our Charter and bylaws could discourage, delay, defer or prevent a merger, tender offer, proxy contest or other change of control transaction that a stockholder might consider in its best interest, including those attempts that might result in a premium over the market price for the shares of our Class A common stock held by our stockholders. These provisions provide for, among other things:
These anti-takeover provisions as well as certain provisions of Delaware law could make it more difficult for a third party to acquire the Company, even if the third party’s offer may be considered beneficial by many of our stockholders. As a result, our stockholders may be limited in their ability to obtain a premium for their shares. If prospective takeovers are not consummated for any reason, we may experience negative reactions from the financial markets, including negative impacts on the price of our Class A common stock. These provisions could also discourage proxy contests and make it more difficult for our stockholders to elect directors of their choosing and to cause us to take other corporate actions that our stockholders desire.
Our Charter designates the Court of Chancery of the State of Delaware as the sole and exclusive forum for certain types of actions and proceedings and the federal district courts as the sole and exclusive forum for other types of actions and proceedings, in each case, that may be initiated by our stockholders, which could limit our stockholders’ ability to obtain what such stockholders believe to be a favorable judicial forum for disputes with the Company or our directors, officers or other employees.
Our Charter provides that, unless we consent to the selection of an alternative forum, any (i) derivative action or proceeding brought on behalf of us; (ii) action asserting a claim of breach of a fiduciary duty owed by, or any other wrongdoing by, any current or former director, officer, other employee or stockholder of us; (iii) action asserting a claim against us arising pursuant to any provision of the Delaware General Corporation Law ("DGCL") or our Charter or our bylaws or as to which the DGCL confers jurisdiction on the Court of Chancery; (iv) action to interpret, apply, enforce, or determine the validity of any provisions in the certificate of incorporation of bylaws; or (v) action asserting a claim governed by the internal affairs doctrine, shall, to the fullest extent permitted
64
by law, be exclusively brought in the Court of Chancery of the State of Delaware or, if such court does not have subject matter jurisdiction thereof, the federal district court of the State of Delaware. Subject to the foregoing, the federal district courts of the United States are the exclusive forum for the resolution of any action, suit or proceeding asserting a cause of action under the Securities Act. The exclusive forum provision does not apply to suits brought to enforce any liability or duty created by the Exchange Act. Any person or entity purchasing or otherwise acquiring or holding an interest in any shares of our capital stock shall be deemed to have notice of and to have consented to the forum provisions in our Charter. These choice-of-forum provisions may limit a stockholder’s ability to bring a claim in a judicial forum that he, she or it believes to be favorable for disputes with the Company or our directors, officers or other employees or stockholders, which may discourage such lawsuits. We note that there is uncertainty as to whether a court would enforce these provisions and that investors cannot waive compliance with the federal securities laws and the rules and regulations thereunder. Section 22 of the Securities Act creates concurrent jurisdiction for state and federal courts over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder.
Alternatively, if a court were to find these provisions of our Charter inapplicable or unenforceable with respect to one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters in other jurisdictions, which could materially adversely affect our business, financial condition and results of operations and result in a diversion of the time and resources of our management and board of directors.
Changes in laws or regulations, or a failure to comply with any laws and regulations, or any litigation that we may be subject to or involved in may adversely affect our business, investments and results of operations.
We are subject to laws, regulations and rules enacted by national, regional and local governments and the Nasdaq Stock Market on which our securities are listed. In particular, we are required to comply with certain SEC, Nasdaq, Delaware and other legal and regulatory requirements. Compliance with, and monitoring of, applicable laws, regulations and rules may be difficult, time-consuming and costly.
Those laws, regulations and rules and their interpretation and application may also change from time to time and those changes could have a material adverse effect on our business, investments and results of operations. For example, it is difficult to predict what impact, if any, changes in federal laws and policies, including those relating to tax, environmental, labor and employment, will have on our business and industry, the economy as a whole, consumer confidence and discretionary spending. Further, a prior ruling by the Court of Chancery in Delaware introduced uncertainty as to whether Section 242(b)(2) of the Delaware General Corporation Law (“DGCL”) required a separate vote in favor of at least a majority of the outstanding shares of Class A common stock, in addition to a vote in favor of at least a majority of the outstanding shares of Class A and Class B common stock, voting together as a single class, to properly authorize shares of Class A common stock. In connection with the Business Combination, our stockholders authorized an increase in the number of shares of Class A common stock under Cayman Islands law, our jurisdiction at the time of the stockholder vote. Accordingly, we do not believe that the Delaware ruling applies to us. However, any failure to comply with applicable laws, regulations or rules, as interpreted and applied, could have a material adverse effect on our business and results of operations. Claims alleging that a portion of our Class A common stock was not authorized could lead to shares of our Class A common stock being voidable and have a material adverse effect on us and our prospects. In addition, uncertainty with respect to our capitalization resulting from the Court of Chancery’s ruling referenced above could have a material adverse impact on us, including on our ability to complete equity financing transactions or issue stock-based compensation to our employees, directors and officers until the underlying issues are definitively resolved. This uncertainty could impair our ability to execute our business plan, attract and retain employees, management and directors and adversely affect our commercial relationships.
We are an emerging growth company and a smaller reporting company within the meaning of the Securities Act, and we may take or continue to take advantage of certain exemptions from disclosure requirements available to emerging growth companies or smaller reporting companies, which could make our securities less attractive to investors and may make it more difficult to compare our performance with other public companies.
We are an “emerging growth company” within the meaning of the Securities Act, as modified by the JOBS Act, and we may take or continue to take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. As a result, our stockholders may not have access to certain information they may deem important. We could be an emerging growth company for up to five years, although circumstances could cause us to lose that status earlier, including if the market value of our Class A common stock held by non-affiliates is $700 million or more as of the last business day of the most recently completed second fiscal quarter, in which case we would no longer be an emerging growth company as of the end of that fiscal year. We cannot predict whether investors will find our securities less attractive because we will rely on these exemptions. If some investors find our securities less attractive as a result of our reliance on these exemptions, the trading prices of our securities may be lower than they otherwise would be, there may be a less active trading market for our securities and the trading prices of our securities may be more volatile.
65
Further, Section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that a company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such election to opt out is irrevocable. We have elected not to opt out of such extended transition period which means that when a standard is issued or revised and it has different application dates for public or private companies, we, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard. This may make comparison of our financial statements with another public company which is not an emerging growth company or is an emerging growth company which has opted out of using the extended transition period difficult or impossible because of the potential differences in accountant standards used.
Additionally, we are a “smaller reporting company” as defined in Item 10(f)(1) of Regulation S-K. Smaller reporting companies may take advantage of certain reduced disclosure obligations, including, among other things, providing only two years of audited financial statements. We are required to reflect a determination that we are no longer a smaller reporting company in our quarterly report on Form 10-Q for the first fiscal quarter of the next fiscal year after the fiscal year in which (i) the market value of our common stock held by non-affiliates is greater than or equal to $250 million as of the end of that fiscal year’s second fiscal quarter, and (ii) if our annual revenues are not greater than or equal to $100 million during the last completed fiscal year, the market value of our common stock held by non-affiliates is $700 million or more as of the end of that fiscal year’s second fiscal quarter. To the extent we take advantage of such reduced disclosure obligations, it may also make comparison of our financial statements with other public companies difficult or impossible.
Reports published by analysts, including projections in those reports that differ from our actual results, could adversely affect the price and trading volume of our Class A common stock.
Securities research analysts may establish and publish their own periodic projections for us. Those projections may vary widely and may not accurately predict the results we actually achieve. Our share price may decline if our actual results do not match the projections of these securities research analysts. Similarly, if one or more of the analysts who write reports on us downgrades our stock or publishes inaccurate or unfavorable research about our business, our share price could decline. In addition, securities research analysts may compare us to companies that are not appropriately comparable, which could lead to lower than expected valuations. If one or more analysts cease coverage of us or fail to publish reports on us regularly, our share price or trading volume could decline.
Our business and operations could be negatively affected if we become subject to any securities litigation or shareholder activism, which could cause us to incur significant expense, hinder execution of our business and growth strategy and impact our stock price.
In the past, following periods of volatility in the market price of a company’s securities, securities class action litigation has often been brought against that company. Shareholder activism, which could take many forms or arise in a variety of situations, has been increasing recently. Volatility in the stock price of our Class A common stock or other reasons may in the future cause us to become the target of securities litigation or shareholder activism. Securities litigation and shareholder activism, including potential proxy contests, could result in substantial costs and divert management’s and the board of directors’ attention and resources from our business. Additionally, such securities litigation and shareholder activism could give rise to perceived uncertainties as to our future, adversely affect our relationships with service providers and make it more difficult to attract and retain qualified personnel. Also, we may be required to incur significant legal fees and other expenses related to any securities litigation and activist shareholder matters. Further, our stock price could be subject to significant fluctuation or otherwise be adversely affected by the events, risks and uncertainties of any securities litigation and shareholder activism.
The grant of registration rights pursuant to the Registration Rights Agreement, the PIPE Subscription Agreements and the Letter Agreement, and the future exercise of such rights, may adversely affect the market price of our Class A common stock.
At the Closing, we, HC Sponsor LLC (the "Sponsor"), certain affiliates of the Sponsor and certain stockholders of Legacy Hyperfine and Liminal entered into the Registration Rights Agreement, pursuant to which, among other things, the parties to the Registration Rights Agreement were granted certain registration rights (including demand and piggy-back rights, subject to cooperation and cut-back provisions) with respect to their respective shares of our common stock, in each case, on the terms and subject to the conditions therein. In particular, the Registration Rights Agreement requires that we use our commercially reasonable efforts to file a registration statement under the Securities Act to permit the public resale of all registrable securities as permitted by Rule 415 of the Securities Act and to cause such registration statement to be declared effective. The registration statement was initially filed on January 24, 2022 and declared effective by the SEC on February 1, 2022. The post-effective amendment to the registration statement filed on January 24, 2023 was declared effective by the SEC on January 30, 2023.
66
Further, pursuant to the Subscription Agreements entered into in connection with the Business Combination and the Letter Agreement entered into in connection with the Business Combination, we agreed (i) to file a registration statement with the SEC for the resale of the registrable securities under such agreements and to use commercially reasonable efforts to cause such registration statement to be declared effective and (ii) to maintain the effectiveness of such registration statement until the earlier of (a) five years from the date of effectiveness of the initial registration statement, (b) the date on which investors under the subscription agreements cease to hold the securities covered thereby, and (c) the date all of the securities covered thereby can be sold publicly without restriction or limitation under Rule 144 under the Securities Act. We will bear the cost of registering these securities. The registration statement was initially filed on January 24, 2022 and declared effective by the SEC on February 1, 2022. The post-effective amendment to the registration statement filed on January 24, 2023 was declared effective by the SEC on January 30, 2023. The registration and availability of such a significant number of securities for trading in the public market may have an adverse effect on the market price of our Class A common stock.
The obligations associated with being a public company involve significant expenses and require significant resources and management attention, which may divert from our business operations.
As a public company, we are subject to the reporting requirements of the Exchange Act and the Sarbanes-Oxley Act. The Exchange Act requires the filing of annual, quarterly and current reports with respect to a public company’s business and financial condition. The Sarbanes-Oxley Act requires, among other things, that a public company establish and maintain effective internal control over financial reporting. As a result, we are incurring, and will continue to incur significant legal, accounting and other expenses. Our management team and many of our other employees will need to devote substantial time to compliance and may not effectively or efficiently manage our transition as a public company.
We do not intend to pay cash dividends for the foreseeable future.
We currently intend to retain our future earnings, if any, to finance the further development and expansion of our business and do not intend to pay cash dividends in the foreseeable future. Any future determination to pay dividends will be at the discretion of our board of directors and will depend on our financial condition, results of operations, capital requirements, and future agreements and financing instruments, business prospects and such other factors as our board of directors deems relevant.
Item 1B. UNRESOLVED STAFF COMMENTS
None.
Item 1C. CYBERSECURITY
Our current digital age has seen a remarkable growth in information technologies that allows for greater connectivity capabilities and faster sharing of data and information than ever before. These rapid advances in technology have been of great benefit to organizations like our own, which seek to deliver cutting-edge and life-enhancing solutions for our customers. In our pursuit of these objectives, we acknowledge and take seriously our responsibility to maintain the highest level of confidentiality, integrity, and availability of data belonging to our customers, prospects, external partners, and internal workforce members, and to protect our critical information technology systems and infrastructure against current and ever evolving cybersecurity threats and attacks.
It is our organization’s aim to comply with all cybersecurity and data privacy laws and regulations in applicable jurisdictions. Additionally, we are committed to meeting national and international standards and best practices for our industry for effective cybersecurity risk management of the organization’s confidential information and critical IT infrastructure. Our cybersecurity and data protection policies, standards, processes and practices are based on recognized frameworks established by the National Institute of Standards and Technology, or “NIST”, and other applicable industry standards and frameworks such as HITRUST and SOC 2. In general, we seek to address cybersecurity risks through a comprehensive, cross-functional approach that is focused on preserving the confidentiality, integrity, and availability of the data that we collect and store, and information systems and technologies through which that data is processed, by identifying, preventing and mitigating cybersecurity threats, and effectively responding to cybersecurity incidents when they occur. Our organization’s internal administrative, technical, and physical security measures (controls) are tested for compliance to these frameworks and reviewed annually. The domains and controls associated with these frameworks are tested and reviewed annually. Certifications of compliance, and/or Auditor’s ‘Letters of Opinion’ Attestation of Compliance from the applicable auditors are provided once the audits are completed each year.
The status of our cybersecurity program is regularly reported to our organization’s board of directors. Our board of directors is actively involved in oversight of our cybersecurity risk management activities, and cybersecurity represents an important element of our organization’s overall approach to risk management in the pursuit of its business goals and objectives.
67
Cybersecurity Risk Management and Strategy; Effect of Risk
We face risks related to cybersecurity such as unauthorized access, cybersecurity attacks and other security incidents, including as perpetrated by hackers and unintentional damage or disruption to hardware and software systems, loss of data, and misappropriation of confidential information. To identify and assess material risks from cybersecurity threats, we maintain a comprehensive cybersecurity program to ensure our data and information systems are effectively secured and prepared for information security risks by internal and external threat actors. Our program involves regular monitoring of our internal IT assets, data inventories, and potential security exploits and/or threats to those assets’ confidentiality, integrity, and availability. We employ a range of technical security tools and external security services, including regular network and endpoint monitoring, compliance audits, vulnerability assessments, penetration testing, threat modeling and tabletop exercises to inform our risk posture, remediation plans and cybersecurity investments.
We consider risks from cybersecurity threats alongside other company risks as part of our overall risk assessment process. As discussed in more detail under “Cybersecurity Governance” below, our board of directors and our audit committee provide oversight of our cybersecurity risk management and strategy processes, which are led by our Chief Compliance Officer, Chief Administrative Officer, Chief Operating Officer, Security Officer, Data Protection Officer, Senior Director of Cybersecurity and IT, and Governance Risk and Compliance Manager.
We also identify our cybersecurity threat risks by comparing our processes to standards established by the HITRUST and SOC2 frameworks and any findings resulting from penetration testing and threat modeling conducted by third party service providers. To provide for the confidentiality, integrity, and availability of critical data and information systems, maintain regulatory compliance, manage our material risks from cybersecurity threats, and protect against and respond to cybersecurity incidents, we undertake the following internal security measures:
68
As part of the above processes, we regularly engage with consultants, auditors and other third parties, including annually having a third-party independent Risk Assessor review our cybersecurity program to help identify areas for continued focus, improvement, and compliance.
Our processes also address cybersecurity threat risks associated with our use of third-party service providers, including our suppliers, manufacturers and other third parties who have access to patient and employee data or our IT systems. In addition, cybersecurity considerations affect the selection and oversight of our third-party service providers. We perform diligence on third parties that have access to our systems, data or facilities that house such systems or data, and we continually monitor cybersecurity threat risks identified through such diligence. Additionally, we generally require those third parties that could introduce significant cybersecurity risk to us to agree by contract to manage their cybersecurity risks in specified ways, and to agree to be subject to cybersecurity audits, which we conduct as appropriate.
We describe whether and how risks from identified cybersecurity threats have materially affected or are reasonably likely to materially affect us, including our business strategy, results of operations, or financial condition, under the heading under Risk Factors titled “Security breaches, loss of data and other disruptions could compromise sensitive information related to our business or prevent us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation”, which disclosures are incorporated by reference herein.
In the last two fiscal years, we have not experienced any material cybersecurity incidents and any expenses we have incurred from cybersecurity incidents were immaterial. This includes penalties and settlements, of which there were none. As needed, we will contract a professional cybersecurity investigation firm to conduct a full forensic analysis of any suspected material incident. To date, we have concluded that there has not been any evidence of material concern involving malware, persistence mechanisms or other compromised exchange of on-premises accounts within the Company’s environment.
Cybersecurity Governance; Management
Cybersecurity is an important part of our risk management processes and an area of focus for our board of directors and management. In general, our board of directors oversees risk management activities designed and implemented by our management, and considers specific risks, including, for example, risks associated with our strategic plan, business operations, and capital structure. Our board of directors executes its oversight responsibility for risk management both directly and through delegating oversight of certain of these risks to its committees, and our board of directors has authorized our audit committee to oversee risks from cybersecurity threats.
At least quarterly, our audit committee receives an update from management of our cybersecurity threat risk management and strategy processes covering topics such as data security posture, results from third-party assessments, progress towards pre-determined risk-mitigation-related goals, our incident response plan, and material cybersecurity threat risks or incidents and developments, as well as the steps management has taken to respond to such risks. Our audit committee generally receives materials that include a cybersecurity scorecard and other materials discussing current and emerging material cybersecurity threat risks, and describing our ability to mitigate those risks, as well as recent developments, evolving standards, technological developments and information security considerations arising with respect to our peers and third parties, and discusses such matters with our Chief Compliance Officer, Chief Administrative Officer, Chief Operating Officer, Security Officer, Data Protection Officer, Senior Director of Cybersecurity and IT, and Governance Risk and Compliance Manager. Our audit committee also receives prompt and timely information regarding any material cybersecurity incident that meets reporting thresholds, as well as ongoing updates regarding any such incident until it has been addressed.
Members of our audit committee are also encouraged to regularly engage in conversations with management on cybersecurity-related news events and discuss any updates to our cybersecurity risk management and strategy programs. Material cybersecurity threat risks are also considered during separate board meeting discussions of important matters like enterprise risk management, operational budgeting, business continuity planning, mergers and acquisitions, brand management, and other relevant matters.
69
Item 2. PROPERTIES
We currently maintain our principal executive offices at 351 New Whitfield Street, Guilford, Connecticut 06437. We also occupy office and laboratory space in Palo Alto, California. We lease office space under operating leases. We consider our current office space adequate for our current operations.
Item 3. LEGAL PROCEEDINGS
We are not currently a party to any material legal proceedings.
Item 4. MINE SAFETY DISCLOSURES
Not applicable.
70
PART II
Item 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our Class A common stock is traded on The Nasdaq Global Market under the symbol “HYPR”.
Stockholders
As of March 1, 2024, we had 56,895,145 outstanding shares of Class A common stock held by approximately 117 holders of record, 15,055,288 outstanding shares of Class B common stock held by approximately six holders of record, and no outstanding shares of preferred stock.
Unregistered Sales of Securities
Not applicable.
Issuer Purchases of Equity Securities
Not applicable.
Item 6. [RESERVED]
Not applicable.
71
Item 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS OF HYPERFINE
The following discussion and analysis of the financial condition and results of operations Hyperfine, Inc. and its subsidiaries (for purposes of this section, collectively referred as the “Company”, “we,” “us” and “our”) should be read together with the audited consolidated financial statements as of and for the years ended December 31, 2023 and 2022, together with the related notes thereto, included elsewhere in this Annual Report on Form 10-K. This discussion contains forward-looking statements and involves numerous risks and uncertainties, including, but not limited to, those described under the heading “Risk Factors.” Actual results may differ materially from those contained in any forward-looking statements.
Overview
We are an innovative health technology business with a mission to revolutionize patient care globally through accessible, affordable, clinically relevant ultra-low-field (“ULF”) magnetic resonance (“MR”) brain imaging. Our Swoop® Portable MR Imaging® System (“Swoop® system”) produces high-quality images at a lower magnetic field strength than conventional magnetic resonance imaging (“MRI”) scanners. Our Swoop® system is designed to transform brain MR for the patient, the clinician and the provider, and to provide a highly differentiated experience for patients, timely imaging to clinicians, and favorable economics for hospital administrators. The Swoop® system is a portable, ULF MRI device for producing images that display the internal structures of the head where full diagnostic examination is not clinically practical. When interpreted by a trained physician, these images provide information that can be useful in determining a diagnosis. Healthcare professionals can use the Swoop® system to make effective clinical diagnoses and decisions in various care settings where conventional MRI devices are inaccessible. The easy-to-use interface and portable design of our Swoop® system make it easily and readily accessible anywhere in a hospital, clinic, or patient care site and it does not require any special facilities accommodations nor specialized personnel to operate safely. ULF MR does not expose patients to harmful ionizing radiation and compares favorably in this regard to X-ray computed tomography (“CT”) or positron emission tomography (“PET”).
The demand for MR imaging has been increasing due to the aging population and the rising prevalence of neurological, neurodegenerative, and cardiovascular conditions, as well as the trends towards decentralized healthcare in mature as well as low- and middle-income countries. Healthcare professionals and insurers recognize imaging as an effective, non-invasive diagnostic tool for evaluation and ongoing monitoring. The Swoop® system is the next generation brain imaging device designed to increase access to MRI in a cost-effective manner and expand the current $35 billion imaging market.
Despite its advantages, many healthcare institutions worldwide lack the facilities, specialized operators, and capital necessary to acquire and maintain expensive conventional MRI devices. The Swoop® system is the first U.S. Food and Drug Administration (“FDA”)-cleared, portable, ULF, MR brain imaging system and is capable of providing imaging at multiple sites of care, such as intensive care units, clinics, emergency departments or physicians’ offices, and can inform the timely detection, diagnosis, monitoring, and treatment of acute and chronic conditions inside and outside the hospital. We designed the Swoop® system to address the limitations of conventional imaging technologies and make MR brain imaging accessible nearly anytime and anywhere across professional healthcare settings. We believe the adoption of the Swoop® system by healthcare professionals has clinical and economic benefits throughout healthcare communities in both high and low resource settings.
The Swoop® system is AI-powered and integrates deep learning, a form of AI, for the reconstruction and denoising of T1, T2, and fluid-attenuated inversion recovery (“FLAIR”) sequences. The Swoop® system also incorporates deep learning denoising in the diffusion-weighted imaging (“DWI”) sequences for image post-processing. The integration of deep learning does not require any additional steps from the user. As a result, deep learning can enhance the image quality and, consequently, the diagnostic value of images generated at ULF. The algorithms are designed to improve ULF image quality, while reducing the impact of scan artifacts. The images created with these algorithms were validated by expert radiologists. Furthermore, the Swoop® system is used clinically every day as the first mover in the field of portable ULF MRI, and with an install base that continues to expand. The learnings from this field experience has served to improve our software, AI, and denoising algorithms resulting in the image quality and performance boosts of our product over the eight software releases since our initial clearance. As we move forward, we are continuously investing in improving our AI-powered image quality and leveraging each imaging-focused software release to further improve the Swoop® system performance.
Legacy Hyperfine received initial 510(k) clearance for brain imaging of patients of all ages from the FDA in 2020 and has now received eight subsequent clearances from the FDA after the initial clearance. We received our most recent clearance from the FDA in October 2023 for the latest update of our Swoop® system AI-powered software. This updated software expanded the Swoop® system’s AI denoising capabilities by incorporating deep learning denoising in the DWI sequence for image post-processing. The Swoop® system has also received marketing authorization for brain imaging in several countries, including the European Union (CE), the United Kingdom (UKCA), Canada, Australia and New Zealand.
72
In December 2022, we suspended our Liminal program to develop a device to non-invasively measure key vital signs in the brain. Also, in December 2022, we announced an organizational restructuring designed to decrease our operating costs and create a more streamlined organization to support our business. As a result, we terminated approximately 13% of our global workforce including, among others, the employees of our Liminal subsidiary. In connection with the restructuring, we incurred $1.0 million of costs consisting primarily of cash severance costs, other severance benefits, fixed asset impairment costs and other related restructuring costs. We completed the restructuring as of the first quarter of 2023.
Key Performance Measures
Management reviews and analyzes several key performance measures including Total revenues, and Total Swoop® system units sold. These measures are reviewed and analyzed to evaluate our business performance, identify trends affecting our business, allocate capital, and make strategic decisions.
Total revenues were $11.0 million for the year ended December 31, 2023, an increase of $4.2 million, or 61.9% from the year ended December 31, 2022, primarily driven by increases in Swoop® system average selling price, units sold and service revenues. See "Results of Operations - Sales" below for further information. Total Swoop® system units sold were 37 units for the year ended December 31, 2023, an increase of 2 units, or 5.7% from the year ended December 31, 2022.
Factors Affecting Results of Operations
The following factors have been important to our business and we expect them to impact our results of operations and financial condition in future periods:
Technical innovation
We have developed our Swoop® system through extensive research and development activities. Moreover, our team is dedicated to clinical support programs designed to integrate the Swoop® system into an array of diverse healthcare environments and applications. We believe that, from our commercial and clinical experience, we are gaining invaluable insights into the Swoop® system’s feasibility. We believe these learnings will enable us to further improve our product and develop new services and tools in the future. We are continuously improving our image quality and imaging capabilities. Building upon this foundation and our expertise in ULF brain imaging, we plan to develop new imaging applications, broadening the range of clinical uses for our technology. Additionally, we are leveraging our strengths in AI and cloud technology to explore the Swoop® system’s role as a brain imaging clinical decision support platform. While these technical innovations may increase our research and development expenses, we expect them to have a positive impact on our results of operations and profitability in the future.
Commercialization efforts of the Swoop® system
While our results have included revenue from outside the United States, our primary commercial focus is on the United States. Legacy Hyperfine received initial 510(k) clearance from the FDA in 2020. We are focused on building relationships and executing contracts with U.S. hospital systems. We are building a direct sales and field support organization in the United States and have recently made changes within our sales and clinical support teams who are working in strong collaboration to increase adoption, support successful implementations and support routine use at customer sites.
Expand sales in international markets
The countries in which we have begun commercializing our Swoop® system include Canada, Australia, and New Zealand. We obtained a Medical Device License issued by Health Canada, UKCA certification in the United Kingdom, CE certification in the EU and regulatory authorization in Australia and New Zealand.
While we will maintain our commercial focus in the United States in 2024, our commitment to the vision of providing affordable and accessible imaging that enables earlier detection and timely management of health conditions worldwide is currently made possible by grant funding from the BMGF. Through our engagement with the BMGF, we have deployed and continue to deploy the Swoop® system in low-middle income settings without readily-accessible MRI technology. The multiple grants provided by our research partnership with the BMGF, which commenced funding in the spring of 2020, support the deployment of 25 Swoop® system and accessories to investigators. As of December 31, 2023, 22 Swoop® system units were provisioned and delivered to the BMGF and the majority of the milestones for service deliverables were also met. The ongoing investigation is designed to provide data to validate the potential use of the Swoop® system in measuring the impact of maternal anemia, malnutrition, infection, and birth-related injury. In May 2023, we were awarded an additional 3-year grant from the BMGF to continue to develop a scalable approach to measuring neurodevelopment via ULF brain imaging in neonates, infants, and young children in low-to-middle income countries. As of February 15, 2024, we completed the delivery and deployment of a total of 25 Swoop® system devices related to BMGF grants.
73
Description of Certain Components of Financial Data
Sales
We derive our sales from the following sources: device sales and service sales as described in more detail below. Our revenue recognition policies are discussed in more detail under “Summary of Significant Accounting Policies” in Note 2 to our consolidated financial statements and notes thereto for the years ended December 31, 2023 and 2022 included elsewhere in this Annual Report on Form 10-K.
Device: Device sales primarily consist of sales of our MRI devices.
Service: Service sales primarily consist of sales service and support including annual maintenance and cloud hosting.
During each of the first quarters of 2023 and 2022, we have taken pricing actions by increasing the price of the device while lowering the price of the annual service and support charges. This has resulted in higher device revenue per unit and lower service revenue per unit for sales under this business model.
Cost of sales
Cost of sales consists of product and service costs including personnel cost and benefits including stock-based compensation, product costs, production setup expenses, depreciation and amortization expenses, inventory excess and obsolescence expenses.
Research and development
Research and development costs consist of production costs for prototype, test and pre-production units, lab supplies, clinical study cost, consulting and personnel costs, including salaries, stock-based compensation, bonuses and benefit costs. Most of our research and development expenses are related to developing new products and services as well as to enhancing our current product and software capabilities. Consulting expenses are related to research and development activities as well as clinical and regulatory activities. Fabrication services include certain third-party engineering costs. Research and development expenses are expensed as incurred. We expect to continue to make substantial investments in research and development.
General and administrative
General and administrative expenses primarily consist of personnel costs and benefits, including stock-based compensation, patent and filing fees, office expenses, technology expenses and outside services. Outside services consist of professional services, legal and other professional fees. Other related costs include additional facilities expenses and general corporate overhead to support the employee base.
Sales and marketing
Sales and marketing costs primarily consist of personnel costs and benefits including stock-based compensation, advertising and promotional costs, as well as costs for conferences, meetings, and other events. We will seek to control sales and marketing expenses while continuing to promote our brand through marketing and advertising initiatives and expand our market presence and awareness.
Interest income
Interest income primarily consists of interest earned on our cash equivalents invested in money market securities.
Other expense, net
Other expense, net primarily relates to foreign exchange gain or loss.
Provision for income taxes
We utilize the asset and liability method of accounting for income taxes, as set forth in Accounting Standards Codification (“ASC”) 740, Income Taxes. Under this method, deferred tax assets and liabilities are recognized for the expected future tax consequences of temporary differences between the carrying amounts and the tax basis of assets and liabilities using the enacted statutory tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. A valuation allowance is established against net deferred tax assets if, based on the weight of available evidence, it is more-likely-than-not that some or all of the net deferred tax assets will not be realized. We recorded a full valuation allowance as of December 31, 2023 and 2022. Based on available evidence, we believe that it is more-likely-than-not that we will be unable to utilize all of our deferred tax assets in the future.
74
Results of Operations
The following is a discussion of our results of operations for the periods shown below, and our accounting policies are described under "Summary of Significant Accounting Policies" in Note 2 in our consolidated financial statements for the years ended December 31, 2023 and 2022 included elsewhere in this Annual Report on Form 10-K.
Comparison of the Years Ended December 31, 2023 and 2022 ($ Amounts in thousands)
|
|
Year Ended December 31, |
|
|
Change |
|
||||||
($ Amounts in thousands) |
|
2023 |
|
|
2022 |
|
|
% |
|
|||
Sales |
|
|
|
|
|
|
|
|
|
|||
Device |
|
$ |
8,746 |
|
|
$ |
5,246 |
|
|
|
66.7 |
% |
Service |
|
|
2,286 |
|
|
|
1,568 |
|
|
|
45.8 |
% |
Total sales |
|
$ |
11,032 |
|
|
$ |
6,814 |
|
|
|
61.9 |
% |
Cost of Sales |
|
|
|
|
|
|
|
|
|
|||
Device |
|
$ |
4,463 |
|
|
$ |
4,231 |
|
|
|
5.5 |
% |
Service |
|
|
1,812 |
|
|
|
1,676 |
|
|
|
8.1 |
% |
Total cost of sales |
|
$ |
6,275 |
|
|
$ |
5,907 |
|
|
|
6.2 |
% |
Gross margin |
|
|
4,757 |
|
|
|
907 |
|
|
|
424.5 |
% |
Operating expenses: |
|
|
|
|
|
|
|
|
|
|||
Research and development |
|
$ |
22,493 |
|
|
$ |
28,156 |
|
|
|
(20.1 |
)% |
General and administrative |
|
|
20,276 |
|
|
|
32,406 |
|
|
|
(37.4 |
)% |
Sales and marketing |
|
|
10,103 |
|
|
|
14,219 |
|
|
|
(28.9 |
)% |
Total operating expenses |
|
|
52,872 |
|
|
|
74,781 |
|
|
|
(29.3 |
)% |
Loss from operations |
|
$ |
(48,115 |
) |
|
$ |
(73,874 |
) |
|
|
(34.9 |
)% |
Interest income |
|
$ |
3,842 |
|
|
$ |
761 |
|
|
|
404.9 |
% |
Other expense, net |
|
|
35 |
|
|
|
(51 |
) |
|
|
(168.6 |
)% |
Loss before provision for income taxes |
|
$ |
(44,238 |
) |
|
$ |
(73,164 |
) |
|
|
(39.5 |
)% |
Provision for income taxes |
|
|
— |
|
|
|
— |
|
|
|
|
|
Net loss and comprehensive loss |
|
$ |
(44,238 |
) |
|
$ |
(73,164 |
) |
|
|
(39.5 |
)% |
Sales
|
|
Year Ended December 31, |
|
|
Change |
|
||||||||||
|
|
2023 |
|
|
2022 |
|
|
Amount |
|
|
% |
|
||||
Device |
|
$ |
8,746 |
|
|
$ |
5,246 |
|
|
$ |
3,500 |
|
|
|
66.7 |
% |
Service |
|
|
2,286 |
|
|
|
1,568 |
|
|
|
718 |
|
|
|
45.8 |
% |
Total sales |
|
$ |
11,032 |
|
|
$ |
6,814 |
|
|
$ |
4,218 |
|
|
|
61.9 |
% |
Device sales increased by $3.5 million, or 66.7%, for the year ended December 31, 2023 compared to the year ended December 31, 2022. The increase in device revenue is mainly due to an increase in unit average selling price as well as an increase in units sold.
Service sales increased by $0.7 million, or 45.8%, for the year ended December 31, 2023 compared to the year ended December 31, 2022. The increase is mainly due to the increased unit install-base.
Cost of sales
|
|
Year Ended December 31, |
|
|
Change |
|
||||||||||
|
|
2023 |
|
|
2022 |
|
|
Amount |
|
|
% |
|
||||
Device |
|
$ |
4,463 |
|
|
$ |
4,231 |
|
|
$ |
232 |
|
|
|
5.5 |
% |
Service |
|
|
1,812 |
|
|
|
1,676 |
|
|
|
136 |
|
|
|
8.1 |
% |
Total cost of sales |
|
$ |
6,275 |
|
|
$ |
5,907 |
|
|
$ |
368 |
|
|
|
6.2 |
% |
Percentage of revenue |
|
|
56.9 |
% |
|
|
86.7 |
% |
|
|
|
|
|
|
||
75
Cost of device sales increased by $0.2 million, or 5.5%, for the year ended December 31, 2023 compared to the year ended December 31, 2022. This increase was driven primarily by an increase in overhead and product costs of $0.1 million due to an increase of units sold, and an increase of $0.1 million in personnel and related costs.
Cost of service sales increased by $0.1 million, or 8.2%, for the year ended December 31, 2023 compared to the year ended December 31, 2022. This increase is due to an increased install base.
Research and development
|
|
2023 |
|
|
2022 |
|
|
Amount |
|
|
% |
|
||||
Research and development |
|
$ |
22,493 |
|
|
$ |
28,156 |
|
|
$ |
(5,663 |
) |
|
|
(20.1 |
)% |
Research and development expenses decreased by $5.7 million, or 20.1%, for the year ended December 31, 2023 compared to the year ended December 31, 2022. This decrease was driven primarily by a decrease in personnel related costs and stock-based compensation expenses of $5.3 million as a result of decreased headcount and a decrease in consulting costs of $1.6 million partially offset by $1.3 million of lower grant fulfilments recorded as credits to research and development expenses.
General and administrative
|
|
Year Ended December 31, |
|
|
Change |
|
||||||||||
|
|
2023 |
|
|
2022 |
|
|
Amount |
|
|
% |
|
||||
General and administrative |
|
$ |
20,276 |
|
|
$ |
32,406 |
|
|
$ |
(12,130 |
) |
|
|
(37.4 |
)% |
General and administrative expenses decreased by $12.1 million, or 37.4%, for the year ended December 31, 2023 compared to the year ended December 31, 2022. This decrease was driven primarily by a decrease in personnel related costs and stock-based compensation expenses of $6.8 million as a result of a lean executive team and decreased headcount, a decrease in accounting, auditing and SEC expenses of $1.7 million, a decrease in insurance expenses of $1.3 million, a decrease in restructuring expenses of $0.7 million which was completed in the first quarter of 2023, a decrease in legal expenses and patent fees of $0.7 million, and a decrease in recruitment expenses of $0.4 million.
Sales and marketing
|
|
Year Ended December 31, |
|
|
Change |
|
||||||||||
|
|
2023 |
|
|
2022 |
|
|
Amount |
|
|
% |
|
||||
Sales and marketing |
|
$ |
10,103 |
|
|
$ |
14,219 |
|
|
$ |
(4,116 |
) |
|
|
(28.9 |
)% |
Sales and marketing expenses decreased by $4.1 million, or 28.9%, for the year ended December 31, 2023 compared to the year ended December 31, 2022. This decrease was driven primarily by a decrease in personnel related costs and stock-based compensation expenses of $2.1 million as a result of decreased headcount, a decrease in digital marketing and marketing events costs of $1.3 million, a decrease in consulting costs of $0.4 million, and a decrease in recruitment costs of $0.3 million.
Interest income
|
|
Year Ended December 31, |
|
|
Change |
|
||||||||||
|
|
2023 |
|
|
2022 |
|
|
Amount |
|
|
% |
|
||||
Interest income |
|
$ |
3,842 |
|
|
$ |
761 |
|
|
$ |
3,081 |
|
|
|
404.9 |
% |
Interest income increased by $3.1 million, for the year ended December 31, 2023 compared to the year ended December 31, 2022. The increase was driven primarily by a higher interest rates and higher cash balances in money market funds and demand deposit accounts during the year ended December 31, 2023 compared to the year ended December 31, 2022.
Other income (expense), net
|
|
Year Ended December 31, |
|
|
Change |
|
||||||||||
|
|
2023 |
|
|
2022 |
|
|
Amount |
|
|
% |
|
||||
Other income (expense), net |
|
$ |
35 |
|
|
$ |
(51 |
) |
|
$ |
86 |
|
|
|
(168.6 |
)% |
76
Other income (expense), net had a favorable increase in other income of $86 thousand for the year ended December 31, 2023 compared to the year ended December 31, 2022. This favorable increase in other income was driven primarily by an increase in net realized gain on foreign currencies of approximately $47 thousand and an increase in interest income from customer financing of approximately $38 thousand.
Liquidity and Capital Resources
We have funded our operations primarily with proceeds from the issuance of common and preferred stock. We have incurred significant cash burn and recurring net losses, which includes a net loss of $44.2 million for the year ended December 31, 2023, and an accumulated deficit of $253.7 million as of December 31, 2023. As of December 31, 2023, we had cash and cash equivalents of $75.2 million. As we continue to invest in research and development of our products and sales and marketing, we expect to continue to incur a significant cash burn and recurring net losses for the foreseeable future until such time that our product and services sales generate enough gross profit to cover our operating expenses. However, we can provide no assurance that our product and service sales will generate a net profit in the future or that our cash resources will be sufficient to continue our commercialization and development activities.
In November 2023, we filed a shelf registration statement on Form S-3 with the SEC pursuant to which we registered for sale up to $150 million of any combination of our Class A common stock, preferred stock, debt securities, warrants, rights and/or units from time to time and at prices and on terms that we may determine. Our shelf registration statement on Form S-3 also included a prospectus covering up to an aggregate of $50.0 million in shares of Class A common stock that we may issue and sell from time to time, through B. Riley Securities, Inc. (“B. Riley”) acting as our sales agent, pursuant to the sales agreement that we entered into with B. Riley in November 2023 (the “Sales Agreement”), for our “at-the-market” equity program. We are not obligated to make any sales of Class A common stock under the Sales Agreement. As of December 31, 2023, the Company had not sold any Class A common stock pursuant to the Sales Agreement. As of the filing of this Annual Report on Form 10-K, we will be subject to the SEC general instructions of Form S-3 known as the “baby shelf rules.” Under these instructions, the amount of funds we can raise through primary public offerings of securities in any 12-month period using our registration statement on Form S-3 is limited to one-third of the aggregate market value of the shares of our common stock held by non-affiliates. Therefore, we will be limited in the amount of proceeds we are able to raise by selling shares of our common stock using our Form S-3, including under the Sales Agreement, until such time as our public float exceeds $75 million.
Our ability to access capital when needed is not assured and, if capital is not available when, and in the amounts needed, we could be required to delay, scale back or abandon some or all of our development programs, commercialization of our products, and other operations which could materially harm our operations, financial condition and operating results. We expect that our existing cash and cash equivalents, together with proceeds from the sales of our products and services, will enable us to conduct our planned operations for at least the next 12 months. Factors that could accelerate cash needs include: (i) delays in achieving scientific and technical milestones; (ii) unforeseen capital expenditures and fabrication costs related to manufacturing; (iii) changes we may make in our business or commercialization and hiring strategy; (iv) costs of running a public company; (v) higher inflation and increases in product transportation and labor costs; and (vi) other items affecting our forecasted level of expenditures and use of cash resources including potential acquisitions. As part of the prioritization of our projects and expenditures, in December 2022, we suspended and ceased business operations for the development of our Liminal brain sensing platform, which was the focus of Liminal and was in the early stages of development to non-invasively measure key vital signs in the brain.
We expect to use our funds to further invest in the development of our products and services, commercial expansion, and for working capital and general corporate purposes.
Our future cash requirements will depend on many factors, including market adoption of our products; the cost and timing of establishing additional sales, marketing and distribution capabilities; the cost of our research and development activities; our ability to enter into and maintain collaborations; the cost and timing of potential future regulatory clearances or approvals for our products; and the effect of competing technological and market developments. We cannot assure you that we will be able to obtain additional funds on acceptable terms, or at all. If we raise additional funds by issuing equity or equity-linked securities, our stockholders may experience dilution. Future debt financing, if available, may involve covenants restricting our operations or our ability to incur additional debt. Any debt or equity financing that we raise may contain terms that are not favorable to us or our stockholders. If we raise additional funds through collaboration and licensing arrangements with third parties, it may be necessary to relinquish some rights to our technologies or our products, or grant licenses on terms that are not favorable to us. If we do not have or are not able to obtain sufficient funds, we may have to delay development or commercialization of our products. We also may have to reduce marketing, customer support or other resources devoted to our products and services or cease operations.
77
Cash
As of December 31, 2023, we had cash and cash equivalents of $75.2 million. Our future capital requirements may vary from those currently planned and will depend on various factors including further development costs, commercialization strategy, regulatory developments, supply constraints, manufacturing costs and international expansion. If we need additional funds and are unable to obtain funding on a timely basis, we may need to curtail significantly our product development and commercialization efforts to provide sufficient funds to continue our operations, which could adversely affect our business prospects.
Cash flows
The following table summarizes our cash flows for the periods indicated:
|
|
Year Ended December 31, |
|||||||
(In thousands) |
|
2023 |
|
|
2022 |
|
|
||
Net cash used in operating activities |
|
$ |
(41,809 |
) |
|
$ |
(72,339 |
) |
|
Net cash used in investing activities |
|
|
(804 |
) |
|
|
(585 |
) |
|
Net cash provided by financing activities |
|
|
174 |
|
|
|
7 |
|
|
Net decrease in cash and cash equivalents and restricted cash |
|
$ |
(42,439 |
) |
|
$ |
(72,917 |
) |
|
Net cash used in operating activities
For the year ended December 31, 2023, net cash used in operating activities of $41.8 million was due primarily to a net loss of $44.2 million, non-cash items of $6.0 million and changes in operating assets and liabilities of $3.6 million. Non-cash items were primarily stock based compensation expense of $4.7 million and depreciation expense of $1.1 million. Changes in operating assets and liabilities were driven primarily by an increase in inventory of $2.2 million due to increased inventory units-on-hand, an increase in accounts receivable of $1.1 million due to increased revenue, a decrease in accrued expenses and other liabilities of $0.7 million, an increase in unbilled receivables of $0.5 million, an increase in prepaid inventory of $0.4 million and an increase of other long term assets of $0.2 million, partially offset by a decrease in prepaid expenses and other current assets of $1.5 million mainly due to the timing of directors and officers liability insurance prepayment.
For the year ended December 31, 2022, net cash used in operating activities of $72.3 million was due primarily to a net loss of $73.2 million, non-cash items of $11.8 million and changes in operating assets and liabilities of $10.9 million. Non-cash items were primarily stock based compensation expense of $10.7 million, including $0.1 million of stock-based compensation related to severance, and $0.1 million fixed asset impairment costs and other related restructuring costs. Changes in operating assets and liabilities were driven primarily by a decrease in accrued expenses and other current liabilities of $2.2 million, a decrease in due to related parties of $2.0 million, a decrease in deferred grant funding of $1.9 million, an increase in prepaid expenses and other current assets of $1.8 million, a decrease in accounts payable of $1.6 million, an increase in accounts receivables of $1.6 million, an increase in other long term assets of $0.6 million, an increase in unbilled receivables of $0.4 million, an increase in inventory of $0.3 million, and an increase in prepaid inventory of $0.3 million, partially offset by an increase in deferred revenue of $1.7 million.
Net cash used for investing activities
For the year ended December 31, 2023, net cash used in investing activities of $0.8 million was from fixed assets purchased.
For the year ended December 31, 2022, net cash used in investing activities of $0.6 million was from fixed assets purchased.
Net cash provided by financing activities
For the year ended December 31, 2023, net cash provided by financing activities of $0.2 million was proceeds from option exercises.
For the year ended December 31, 2022, net cash provided by financing activities of $7 thousand was proceeds from option exercises.
Contractual obligations
We sponsor a 401(k) defined contribution plan covering all eligible U.S. employees. Contributions to the 401(k) plan are discretionary. We did not make any matching contributions to the 401(k) plan for the years ended December 31, 2023 and 2022.
78
During 2020 and 2021 we received multiple grants totaling $4.9 million from the BMGF for the provision and equipping of sites with our portable MR brain imaging system to enable the performance of a multi-site study focused on optimizing diagnostic image quality. These grants are designed to provide data to validate the use of our Swoop® system in measuring the impact of maternal anemia, malnutrition, infection and birth related injury. All of these grants are designed to support the deployment of a total of 25 Swoop® system devices and other services to investigators, which commenced in the spring of 2021. In March 2023 the term of the BMGF grant agreement was extended to February 2024. As of December 31, 2023, 22 Swoop® system units have been provisioned and delivered to the BMGF and certain milestones for service deliverables were met. All system units and grant project deliverables have been completed as of February 2024.
In May 2023, we were awarded an additional $3.4 million grant from the BMGF to continue to develop a scalable approach to measuring neurodevelopment via low-field MRI in neonates, infants, and young children in low-to-middle income countries through February 2026.
Any grant funds, plus any income, that have not been used for, or committed to, the project must be returned promptly to the BMGF upon expiration of or termination of the agreement. Refer to Note 13 in the notes to our consolidated financial statements for the years ended December 31, 2023, and 2022 included elsewhere in this Annual Report on Form 10-K for a discussion of the BMGF grants.
Our purchase commitments and obligations include all open purchase orders and contractual obligations in the ordinary course of business, including commitments with contract manufacturers and suppliers, for which we have not received the goods or services. A majority of these purchase obligations are due within a year. Although open purchase orders are considered enforceable and legally binding, the terms generally allow us the option to cancel, reschedule, and adjust our requirements based on our business needs prior to the delivery of goods or performance of services.
We had no other significant contractual obligations as of December 31, 2023.
For information on contingencies, refer to Note 13 to our consolidated financial statements and notes thereto for the years ended December 31, 2023 and 2022 included elsewhere in this Annual Report on Form 10-K.
Critical Accounting Estimates
Our financial statements are prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”). The preparation of our financial statements in conformity with U.S. GAAP requires us to make estimates and assumptions that affect certain reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the reporting period. While our significant accounting policies are described in more detail in Note 2 in our consolidated financial statements for the years ended December 31, 2023 and 2022 included elsewhere in this Annual Report on Form 10-K, we believe that the following accounting policies are those most critical to the judgments and estimates used in the preparation of our financial statements.
Revenue recognition
We make judgments including determination of the timing and pattern of satisfaction of performance obligations and determination of the standalone selling price (“SSP”) of performance obligations. We offer alternative payment structures and “as-a-service” offerings that are assessed to determine whether an embedded lease arrangement exists. Revenue from Device-as-a-Service ("DaaS") contracts are within the scope of ASC 842 and ASC 606. Upon adoption of ASC 842, for contracts in which we act as a lessor and in which the lease component is an operating lease, we apply the practical expedient in ASC 842 to combine the lease component (the device itself in DaaS contracts) and non-lease (maintenance and SaaS) components, and to account for the combined components as a single lease component. Accordingly, we account for the monthly payments as lease revenue. For contracts in which the lease component is a sales-type lease, we derecognize the asset (the MRI device) and recognize a lease receivable in an amount that represents the present value of the lease payments.
Inventories
Inventories primarily consist of finished goods which are produced by our third-party contract manufacturer and raw materials ordered in advance by the third-party contract manufacturer due to long delivery-lead time and were billed to the Company. Inventories are stated at the lower of actual cost, determined using the average cost method, or net realizable value (“NRV”). We routinely evaluate quantities and value of our inventories in light of current market conditions and market trends and record a write-down against the cost of inventories for NRV below cost. NRV is based upon an estimated average selling price reduced by the estimated costs of disposal and transportation. The determination of NRV involves numerous judgments including estimating selling prices, existing customer orders, and estimated costs of disposal and transportation. If actual market conditions differ from our estimates, future results of operations could be materially affected.
79
The valuation of inventory also requires us to estimate excess and obsolete inventory. We periodically review the age, condition and turnover of our inventory to determine whether any inventory has become obsolete or has declined in value and incur a charge to operations for known and anticipated inventory obsolescence. We also consider how quickly customers will transition from older products to newer products, including whether older products can be re-manufactured into new products. The evaluation takes into consideration the effect that new products might have on the sale of existing products, product obsolescence, product merchantability and other factors. Market conditions are subject to change and if actual market conditions are less favorable than those projected by management, additional inventory write-downs may be required, which would have a negative impact on gross margin.
Stock-based compensation
Our stock-based compensation program includes restricted stock unit and stock option grants to our employees, directors and consultants. Stock options are granted at exercise prices not less than the estimated fair market value of our common stock at the dates of grant. For purposes of restricted stock unit grants, the grant date fair value is calculated as the fair market value of the stock on the date of grant.
The fair values of stock option grants are estimated using a Black-Scholes option-pricing model. Key inputs and assumptions include the expected term of the option, stock price volatility, risk free interest rate, dividend yield, stock price and exercise price. Many of the assumptions require significant judgment and changes in assumptions could have a significant impact in the determination of stock-based compensation expense.
Key assumptions include:
Generally, stock options granted to employees fully vest four years from the grant date and have a contractual term of 10 years and stock options granted to non-employees fully vest one year from the grant date or upon performance of a service and have a contractual term of 10 years.
Recently Issued Accounting Pronouncements
A description of recently issued accounting pronouncements that may potentially impact our financial position and results of operations is disclosed in Note 2 to our consolidated financial statements and notes thereto for the years ended December 31, 2023 and 2022 included elsewhere in this Annual Report on Form 10-K.
80
Item 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
We are exposed to market risk in the ordinary course of business. Market risk represents the risk of loss that may impact our results of operations or financial position due to adverse changes in financial market prices and rates. Our market risk exposure is primarily a result of fluctuations in interest rates, inflation risk, and foreign exchange risk. We do not hold, issue or enter into any financial instruments for speculative or trading purposes. We do not have significant exposure to foreign currencies.
Interest rate risk
Our cash, cash equivalents and restricted cash as of December 31, 2023 consisted of $75.7 million in money market funds, demand deposit and savings accounts. Such interest-earning instruments carry a degree of interest rate risk. The goals of our investment policy are liquidity and capital preservation. We believe that we do not have any material exposure to changes in the fair value of these assets as a result of changes in interest rates due to the short-term nature of our cash equivalents. Based on our balance sheet position at December 31, 2023, the annualized effect of a 0.5 percentage point decrease in interest rates would be to decrease earnings before income taxes by $0.4 million.
Inflation Risk
We do not believe that inflation has had a material effect on our business, financial condition, or results of operations, other than its impact on the general economy. Nonetheless, if our costs were to become subject to inflationary pressures, we might not be able to fully offset such higher costs through price increases. Our inability or failure to do so could harm our business, financial condition, and results of operations.
Foreign Exchange Risk
We operate our business primarily within the United States. With respect to our sales outside the U.S., the majority of our transactions are executed in U.S. dollars and, to a lesser extent, in foreign currency. We have not utilized hedging strategies with respect to such foreign exchange exposure. This limited foreign currency translation risk is not expected to have a material impact on our consolidated financial statements.
Item 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
See financial statements included in Item 15 "Exhibits and Financial Statement Schedules" of this Annual Report on Form 10-K.
Item 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
Not applicable.
Item 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
Our principal executive officer and principal financial officer evaluated the effectiveness of our disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) as of December 31, 2023. Based upon that evaluation, our principal executive officer and principal financial officer concluded that, as of December 31, 2023, our disclosure controls and procedures were effective at the reasonable assurance level to ensure that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the SEC’s rules and forms, and is accumulated and communicated to our management, including our principal executive and principal financial officers, or persons performing similar functions, as appropriate, to allow timely decisions regarding required disclosure.
81
Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting. Internal control over financial reporting is defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act, as a process designed by, or under the supervision of, our principal executive and principal financial officers and effected by our board of directors, management and other personnel to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Our internal control over financial reporting includes those policies and procedures that:
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Our management assessed the effectiveness of our internal control over financial reporting as of December 31, 2023 and, in making this assessment, used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework (2013 framework).
Based on our assessment, our management believes that, as of December 31, 2023, our internal control over financial reporting is effective based on those criteria.
Changes in Internal Controls
There were no changes in our internal control over financial reporting, identified in connection with the evaluation of such internal control that occurred during the fourth quarter ended December 31, 2023, that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. OTHER INFORMATION
Not applicable.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable.
82
PART III
Item 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The response to this item is incorporated by reference from the discussion responsive thereto under the headings “Management and Corporate Governance,” “Delinquent Section 16(a) Reports” and “Code of Conduct and Ethics” in our proxy statement for the 2024 annual meeting of stockholders (the “2024 Proxy Statement”).
Item 11. EXECUTIVE COMPENSATION
The response to this item is incorporated by reference from the discussion responsive thereto under the heading “Executive Officer and Director Compensation” in our 2024 Proxy Statement.
Item 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The response to this item is incorporated by reference from the discussion responsive thereto under the headings “Security Ownership of Certain Beneficial Owners and Management” and “Equity Compensation Plan Information” in our 2024 Proxy Statement.
The response to this item is incorporated by reference from the discussion responsive thereto under the headings “Certain Relationships and Related Person Transactions” and “Management and Corporate Governance” in our 2024 Proxy Statement.
Item 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The response to this item is incorporated by reference from the discussion responsive thereto under the heading “Ratification of Appointment of Independent Registered Public Accounting Firm” in our 2024 Proxy Statement.
83
PART IV
Item 15. |
EXHIBITS, FINANCIAL STATEMENT SCHEDULES |
Item 15(a)(1) |
Index to Audited Consolidated Financial Statements as of December 31, 2023 and 2022 and for the years ended December 31, 2023 and 2022 |
Item 15(a)(2) |
Financial statement schedules have not been included because they are not applicable or the information is included in the financial statements or notes thereto. |
Item 15(a)(3) |
Exhibits |
The following is a list of exhibits filed as part of this Annual Report on Form 10-K.
Exhibit |
Exhibit Description |
Filed Herewith |
Incorporated by |
Filing Date |
SEC File/ |
2.1† |
|
Form 8-K |
7/8/2021 |
001-39949 |
|
3.1 |
|
Form 8-K |
12/28/2021 |
001-39949 |
|
3.2 |
Certificate of Amendment of Certificate of Incorporation of Hyperfine, Inc. |
|
Form 8-K (Exhibit 3.2) |
6/12/2023 |
001-39949 |
3.3 |
|
Form 8-K |
6/12/2023 |
001-39949 |
|
4.1 |
|
Form S-4/A |
9/29/2021 |
333-259148 |
|
4.2 |
X |
|
|
|
|
10.1 |
|
Form 8-K |
7/8/2021 |
001-39949 |
|
10.2 |
|
Form 8-K |
7/8/2021 |
001-39949 |
84
85
|
Inc.) and The General Hospital Corporation, d/b/a Massachusetts General Hospital. |
|
|
|
|
10.18@ |
|
Form S-4 |
8/30/2021 |
333-259148 |
|
10.19.1+ |
|
Form 8-K |
12/28/2021 |
001-39949 |
|
10.19.2+ |
Form of Stock Option Agreement under 2021 Equity Incentive Plan |
|
Form 8-K |
12/28/2021 |
001-39949 |
10.19.3+ |
Form of Restricted Stock Unit Agreement under 2021 Equity Incentive Plan |
|
Form S-8 |
3/28/2022 |
333-263897 |
10.20.1+ |
|
Form 8-K |
12/28/2021 |
001-39949 |
|
10.20.2+ |
|
Form 8-K |
12/28/2021 |
001-39949 |
|
10.21.1+ |
Liminal Sciences, Inc. 2021 Employee, Director and Consultant Equity Incentive Plan, as amended |
|
Form 8-K |
12/28/2021 |
001-39949 |
10.21.2+ |
|
Form 8-K |
12/28/2021 |
001-39949 |
|
10.22+ |
|
Form 8-K |
6/12/2023 |
001-39949 |
|
10.23+ |
|
Form 8-K |
12/28/2021 |
001-39949 |
|
10.24+ |
|
Form S-1 |
1/8/2021 |
333-252002 |
|
10.25 |
|
Form 10-Q |
8/11/2022 |
001-39949 |
|
10.26 |
|
Form 8-K |
12/28/2021 |
001-39949 |
|
10.27+ |
|
Form 10-K |
3/22/2023 |
001-39949 |
|
10.28 |
|
Form S-3 |
11/9/2023 |
333-275449 |
|
21.1 |
|
Form 10-K |
3/25/2022 |
001-39949 |
|
23.1 |
X |
|
|
|
86
23.2 |
X |
|
|
|
|
31.1 |
X |
|
|
|
|
31.2 |
X |
|
|
|
|
32 |
Certification pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
X |
|
|
|
97 |
X |
|
|
|
|
101.INS |
Inline XBRL Instance Document |
X |
|
|
|
101.SCH |
Inline XBRL Taxonomy Extension Schema Document |
X |
|
|
|
104 |
Cover Page Interactive Date File (formatted as Inline XBRL and contained in Exhibit 101) |
X |
|
|
|
† Certain of the exhibits and schedules to this Exhibit have been omitted in accordance with Regulation S-K Item 601(a)(5). The Registrant agrees to furnish a copy of all omitted exhibits and schedules to the SEC upon its request.
+ Management contract or compensatory plan or arrangement.
@ Certain confidential portions of this Exhibit were omitted by means of marking such portions with brackets (“[***]”) because the identified confidential portions (i) are not material and (ii) is the type of information that the Company treats as private or confidential.
* The certification attached as Exhibit 32 that accompanies this Annual Report on Form 10-K is not deemed filed with the SEC and is not to be incorporated by reference into any filing of the Company under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date of this Form 10-K, irrespective of any general incorporation language contained in such filing.
Item 16. FORM 10-K SUMMARY
Not applicable.
87
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
HYPERFINE, INC. |
|
Date: March 22, 2024 |
|
By: |
/s/ Maria Sainz |
|
|
|
Maria Sainz |
|
|
|
President and Chief Executive Officer |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
Signatures
|
Title
|
Date
|
|
By: |
/s/ Maria Sainz
Maria Sainz |
President, Chief Executive Officer and Director (principal executive officer) |
March 22, 2024 |
|
|
|
|
By: |
/s/ Brett Hale
Brett Hale |
Chief Administrative Officer, Chief Financial Officer, Treasurer and Corporate Secretary (principal financial officer and principal accounting officer) |
March 22, 2024 |
|
|
|
|
By: |
/s/ R. Scott Huennekens
R. Scott Huennekens |
Chairperson of the Board |
March 22, 2024 |
|
|
|
|
By: |
/s/ Jonathan M. Rothberg, Ph.D.
Jonathan M. Rothberg, Ph.D.
|
Director |
March 22, 2024 |
By: |
/s/ John Dahldorf
John Dahldorf |
Director |
March 22, 2024 |
|
|
|
|
By: |
/s/ Ruth Fattori
Ruth Fattori |
Director |
March 22, 2024 |
|
|
|
|
By: |
/s/ Daniel J. Wolterman
Daniel J. Wolterman |
Director |
March 22, 2024 |
88
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Board of Directors and Stockholders
Hyperfine, Inc.
Opinion on the financial statements
We have audited the accompanying consolidated balance sheet of Hyperfine, Inc. (a Delaware corporation) and subsidiaries (the “Company”) as of December 31, 2023, the related consolidated statements of operations and comprehensive loss, changes in stockholders’ equity and cash flows for year ended December 31, 2023, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2023, and the results of its operations and its cash flows for the year ended December 31, 2023, in conformity with accounting principles generally accepted in the United States of America.
Basis for opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audit. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audit we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audit included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides a reasonable basis for our opinion.
/s/
We have served as the Company’s auditor since 2023
March 22, 2024
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the stockholders and the Board of Directors of Hyperfine, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheet of Hyperfine, Inc. and subsidiaries, (the "Company") as of December 31, 2022, the related consolidated statements of operations and comprehensive loss, changes in stockholders’ equity, and cash flows for the year ended December 31, 2022, and the related notes (collectively referred to as the "financial statements"). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2022, and the results of its operations and its cash flows for the year ended December 31, 2022, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/
March 22, 2023
We began serving as the Company’s auditor in 2021. In 2023 we became the predecessor auditor.
F-2
HYPERFINE, INC. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
AS OF DECEMBER 31, 2023 AND 2022
(in thousands, except share and per share amounts)
|
|
December 31, |
|
|||||
|
|
2023 |
|
|
2022 |
|
||
ASSETS |
|
|
|
|
|
|
||
CURRENT ASSETS: |
|
|
|
|
|
|
||
Cash and cash equivalents |
|
$ |
|
|
$ |
|
||
Restricted cash |
|
|
|
|
|
|
||
Accounts receivable, less allowance of $ |
|
|
|
|
|
|
||
Unbilled receivables |
|
|
|
|
|
|
||
Inventory |
|
|
|
|
|
|
||
Prepaid expenses and other current assets |
|
|
|
|
|
|
||
Due from related parties |
|
|
— |
|
|
|
|
|
Total current assets |
|
$ |
|
|
$ |
|
||
Property and equipment, net |
|
|
|
|
|
|
||
Other long term assets |
|
|
|
|
|
|
||
Total assets |
|
$ |
|
|
$ |
|
||
LIABILITIES AND STOCKHOLDERS' EQUITY |
|
|
|
|
|
|
||
CURRENT LIABILITIES: |
|
|
|
|
|
|
||
Accounts payable |
|
$ |
|
|
$ |
|
||
Deferred grant funding |
|
|
|
|
|
|
||
Deferred revenue |
|
|
|
|
|
|
||
Due to related parties |
|
|
|
|
|
— |
|
|
Accrued expenses and other current liabilities |
|
|
|
|
|
|
||
Total current liabilities |
|
$ |
|
|
$ |
|
||
Long term deferred revenue |
|
|
|
|
|
|
||
Other noncurrent liabilities |
|
|
|
|
|
— |
|
|
Total liabilities |
|
$ |
|
|
$ |
|
||
|
|
|
|
|
|
|||
STOCKHOLDERS' EQUITY: |
|
|
|
|
|
|
||
Class A Common stock, $ |
|
|
|
|
|
|
||
Class B Common stock, $ |
|
|
|
|
|
|
||
Additional paid-in capital |
|
|
|
|
|
|
||
Accumulated deficit |
|
|
( |
) |
|
|
( |
) |
Total stockholders' equity |
|
$ |
|
|
$ |
|
||
TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY |
|
$ |
|
|
$ |
|
||
The accompanying notes are an integral part of these consolidated financial statements.
F-3
HYPERFINE, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(in thousands, except share and per share amounts)
|
|
Year ended December 31, |
|
|||||
|
|
2023 |
|
|
2022 |
|
||
Sales |
|
|
|
|
|
|
||
Device |
|
$ |
|
|
$ |
|
||
Service |
|
|
|
|
|
|
||
Total sales |
|
$ |
|
|
$ |
|
||
Cost of sales |
|
|
|
|
|
|
||
Device |
|
$ |
|
|
$ |
|
||
Service |
|
|
|
|
|
|
||
Total cost of sales |
|
$ |
|
|
$ |
|
||
Gross margin |
|
|
|
|
|
|
||
Operating Expenses: |
|
|
|
|
|
|
||
Research and development |
|
$ |
|
|
$ |
|
||
General and administrative |
|
|
|
|
|
|
||
Sales and marketing |
|
|
|
|
|
|
||
Total operating expenses |
|
|
|
|
|
|
||
Loss from operations |
|
$ |
( |
) |
|
$ |
( |
) |
Interest income |
|
$ |
|
|
$ |
|
||
Other income (expense), net |
|
|
|
|
|
( |
) |
|
Loss before provision for income taxes |
|
$ |
( |
) |
|
$ |
( |
) |
Provision for income taxes |
|
|
|
|
|
|
||
Net loss and comprehensive loss |
|
$ |
( |
) |
|
$ |
( |
) |
Net loss per common share attributable to common stockholders, basic and diluted |
|
$ |
( |
) |
|
$ |
( |
) |
Weighted-average shares used to compute net loss per share attributable to common stockholders, basic and diluted |
|
|
|
|
|
|
||
The accompanying notes are an integral part of these consolidated financial statements.
F-4
HYPERFINE, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(in thousands, except share amounts)
|
|
Class A Common Stock |
|
|
Class B Common |
|
|
Additional |
|
|
Accumulated |
|
|
Total |
|
|||||||||||||
|
|
Shares |
|
|
Amount |
|
|
Shares |
|
|
Amount |
|
|
Paid-in Capital |
|
|
Deficit |
|
|
Equity |
|
|||||||
Balance, December 31, 2021 |
|
|
|
|
$ |
|
|
|
|
|
$ |
|
|
$ |
|
|
$ |
( |
) |
|
$ |
|
||||||
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
( |
) |
|
|
( |
) |
Issuance of restricted stock |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Exercise of stock options |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
— |
|
|
|
|
|||
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
— |
|
|
|
|
||
Balance, December 31, 2022 |
|
|
|
|
$ |
|
|
|
|
|
$ |
|
|
$ |
|
|
$ |
( |
) |
|
$ |
|
||||||
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
( |
) |
|
|
( |
) |
Issuance of restricted stock |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Exercise of stock options |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
— |
|
|
|
|
|||
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
— |
|
|
|
|
||
Balance, December 31, 2023 |
|
|
|
|
$ |
|
|
|
|
|
$ |
|
|
$ |
|
|
$ |
( |
) |
|
$ |
|
||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-5
HYPERFINE, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(in thousands)
|
|
Year ended December 31, |
|
|||||
|
|
2023 |
|
|
2022 |
|
||
Cash flows from operating activities: |
|
|
|
|
|
|
||
Net loss |
|
$ |
( |
) |
|
$ |
( |
) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
||
Depreciation |
|
|
|
|
|
|
||
Stock-based compensation expense |
|
|
|
|
|
|
||
Write-off of equipment |
|
|
|
|
|
— |
|
|
Other |
|
|
|
|
|
|
||
Changes in assets and liabilities |
|
|
|
|
|
|
||
Accounts receivable |
|
|
( |
) |
|
|
( |
) |
Unbilled receivables |
|
|
( |
) |
|
|
( |
) |
Inventory |
|
|
( |
) |
|
|
( |
) |
Prepaid expenses and other current assets |
|
|
|
|
|
( |
) |
|
Due from related parties |
|
|
|
|
|
( |
) |
|
Prepaid inventory |
|
|
( |
) |
|
|
( |
) |
Other long term assets |
|
|
( |
) |
|
|
( |
) |
Accounts payable |
|
|
|
|
|
( |
) |
|
Deferred grant funding |
|
|
( |
) |
|
|
( |
) |
Deferred revenue |
|
|
( |
) |
|
|
|
|
Due to related parties |
|
|
|
|
|
( |
) |
|
Accrued expenses and other current liabilities |
|
|
( |
) |
|
|
( |
) |
Operating lease liabilities, net |
|
|
|
|
|
— |
|
|
Net cash used in operating activities |
|
$ |
( |
) |
|
$ |
( |
) |
Cash flows from investing activities: |
|
|
|
|
|
|
||
Purchases of property and equipment |
|
|
( |
) |
|
|
( |
) |
Net cash used in investing activities |
|
$ |
( |
) |
|
$ |
( |
) |
Cash flows from financing activities: |
|
|
|
|
|
|
||
Proceeds from exercise of stock options |
|
|
|
|
|
|
||
Net cash provided by financing activities |
|
$ |
|
|
$ |
|
||
Net decrease in cash and cash equivalents and restricted cash |
|
|
( |
) |
|
|
( |
) |
Cash, cash equivalents and restricted cash, beginning of year |
|
|
|
|
|
|
||
Cash, cash equivalents and restricted cash, end of year |
|
$ |
|
|
$ |
|
||
Reconciliation of cash, cash equivalents, and restricted cash reported in the balance sheets |
|
|
|
|
|
|
||
Cash and cash equivalents |
|
$ |
|
|
$ |
|
||
Restricted cash |
|
|
|
|
|
|
||
Total cash, cash equivalents and restricted cash |
|
$ |
|
|
$ |
|
||
Supplemental disclosure of cash flow information: |
|
|
|
|
|
|
||
Cash received from exchange of research and development tax credits |
|
$ |
|
|
$ |
|
||
Supplemental disclosure of noncash information: |
|
|
|
|
|
|
||
Write-off of notes receivable |
|
$ |
— |
|
|
$ |
|
|
Noncash acquisition of fixed assets |
|
$ |
|
|
$ |
— |
|
|
The accompanying notes are an integral part of these consolidated financial statements.
F-6
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
1. ORGANIZATION AND DESCRIPTION OF BUSINESS
Hyperfine, Inc. (together with its subsidiaries, as applicable, “Hyperfine” or the “Company”), formerly known as HealthCor Catalio Acquisition Corp. (“HealthCor”), was incorporated as a Cayman Islands exempted company on November 18, 2020. The Company’s legal name became Hyperfine, Inc. in connection with the closing (the “Closing”) of the business combination with HealthCor (the “Business Combination”) on December 22, 2021 (the “Closing Date”). In connection with the Closing, Hyperfine, Inc., a Delaware corporation (“Legacy Hyperfine”), and Liminal Sciences, Inc., a Delaware corporation (“Liminal”), merged with and into separate wholly owned subsidiaries of HealthCor and became wholly-owned subsidiaries of the Company (the “Mergers”), and changed their names to Hyperfine Operations, Inc. and Liminal Operations, Inc., respectively. Liminal subsequently changed its name to Liminal Sciences, Inc.
The Company is an innovative health technology business with a mission to revolutionize patient care globally through accessible, affordable, clinically relevant ultra-low-field (“ULF”) magnetic resonance (“MR”) brain imaging. The Company's Swoop® Portable MR Imaging® System (“Swoop® system”) produces high-quality images at a lower magnetic field strength than conventional magnetic resonance imaging (“MRI”) scanners. The Swoop® system is designed to transform brain MR for the patient, the clinician and the provider, and to provide a highly differentiated experience for patients, timely imaging to clinicians, and favorable economics for hospital administrators. The Swoop® system is a portable, ULF MRI for producing images that display the internal structures of the head where full diagnostic examination is not clinically practical. When interpreted by a trained physician, these images provide information that can be useful in determining a diagnosis. Healthcare professionals can use the Swoop® system to make effective clinical diagnoses and decisions in various care settings where conventional MRI devices are inaccessible. The easy-to-use interface and portable design of the Company's Swoop® system make it easily and readily accessible anywhere in a hospital, clinic, or patient care site and it does not require any special facilities accommodations nor specialized personnel to operate safely. ULF MR does not expose patients to harmful ionizing radiation and compares favorable in this regard to X-ray computed tomography (“CT”) or positron emission tomography (“PET”). The Company's Swoop® system received initial 510(k) clearance for brain imaging from the U.S. Food and Drug Administration (“FDA”) in 2020. In February and October 2023, the Company received additional 510(k) clearances from the FDA of the latest updates to its Swoop® system AI-powered software. The combination of these two software updates significantly improved diffusion-weighted imaging (“DWI”), incorporated deep-learning based denoising in the post-processing of DWI images for crisper images, and improved image quality for all Swoop® system sequences. The Swoop® system has also received marketing authorization for brain imaging in several countries, including the European Union (CE certification), the United Kingdom (UK Conformity Assessment (“UKCA”)), Canada, Australia and New Zealand. All of the Company’s revenue to date has been generated from sales of the Swoop® system and related services. In December 2022, the Company suspended its Liminal program to develop a device to non-invasively measure key vital signs in the brain. In addition to Legacy Hyperfine and Liminal, the Company has an indirect wholly-owned subsidiary in the United Kingdom that did not have any significant operations during 2023 nor 2022.
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation and Principles of Consolidation
The accompanying consolidated financial statements include the accounts of the Company and have been prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and pursuant to the accounting disclosure rules and regulations of the Securities and Exchange Commission (the “SEC”). All intercompany transactions and balances have been eliminated.
Risks and Uncertainties
The Company is subject to risks and uncertainties caused by events with significant geopolitical and macroeconomic impacts, including, but not limited to, the conflicts in Ukraine and the Middle East, inflation and actions taken to counter such impacts.
F-7
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
The Company relies on single source manufacturers and suppliers for the supply of its products. Disruption from these manufacturers or suppliers has and would have a negative impact on the Company’s business, financial position and results of operations in its consolidated financial statements. The Company continues to critically review its liquidity and anticipated capital requirements in light of the significant uncertainty created by geopolitical and macroeconomic conditions. Based on the Company’s cash and cash equivalents balance, current business plan and revenue prospects, the Company believes that it will have sufficient cash resources and anticipated cash flows to fund its operations for at least the next 12 months.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to concentration of credit risk consist principally of cash and cash equivalents. At December 31, 2023 and 2022, substantially all the Company’s cash and cash equivalents were invested in three financial institutions, respectively. The Company also maintains balances in various operating accounts above federally insured limits. The Company has not experienced any losses on such accounts and does not believe it is exposed to any significant credit risk on cash and cash equivalents.
For the years ended December 31, 2023 and 2022 there were
The Company utilizes a single exclusive manufacturer for its Swoop® system. Additionally, the Company purchases raw materials from this manufacturer.
Segment Information
The Company’s Chief Operating Decision Maker (“CODM”) is its Chief Executive Officer (“CEO”). The Company has
Use of Estimates
The preparation of the consolidated financial statements in conformity with U.S. GAAP requires the Company to make estimates and assumptions about future events that affect the amounts reported in its consolidated financial statements and accompanying notes. Future events and their effects cannot be determined with certainty. On an ongoing basis, management evaluates these estimates and assumptions. Significant estimates and assumptions included:
F-8
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
The Company bases these estimates on historical and anticipated results and trends and on various other assumptions that the Company believes are reasonable under the circumstances, including assumptions as to future events. Changes in estimates are recorded in the period in which they become known. Actual results could differ from those estimates, and any such differences may be material to the Company’s consolidated financial statements.
Cash and Cash Equivalents
All highly liquid investments purchased with a maturity of three months or less are cash equivalents. As of December 31, 2023 and 2022, cash and cash equivalents consist principally of cash, money market funds, and demand deposits. The Company funded its operations primarily with proceeds from the issuance of common stock. The Company incurred significant cash burn and recurring net losses, which includes a net loss of $
Restricted Cash
Restricted cash balance represents funds received as part of grant funding and restricted in use to the purpose of the funding. For details, see the Note 2. Summary of Significant Accounting Policies - Grant Funding and Note 13. Commitments and Contingencies.
Accounts Receivable
Accounts receivable are stated as the amount the Company expects to collect. The Company maintains an allowance for credit losses for estimated losses based upon the expected collectability of all accounts receivable. As of December 31, 2023 and 2022, the allowance for credit losses was $
Inventories
Inventories primarily consist of finished goods which are produced by the Company’s third-party contract manufacturer as well as raw materials ordered in advance by the third-party contract manufacturer due to long delivery-lead time and which were billed to the Company. Inventories are stated at the lower of actual cost, determined using the average cost method, or net realizable value. Cost includes an allocation of wages, taxes and benefits for employees involved in warehousing, logistics coordination, material sourcing, and production planning activities. Net realizable value is based upon an estimated average selling price reduced by the estimated costs of disposal and transportation.
The valuation of inventory also requires the Company to estimate excess and obsolete inventory. The Company considers sales forecasts and historical experience to identify excess, close out, or slow-moving items as well as new product development schedules, product obsolescence and product merchantability, including whether older products can be remanufactured into new products, among other factors. The Company reduces the value of inventory for estimated obsolescence or lack of marketability by the difference between the cost of the affected inventory and the net realizable value.
Prepaid Expenses and Other Current Assets
Prepaid expenses and other current assets include amounts paid in advance for operating expenses as well as monies to be received from the State of Connecticut for research and development tax credits. These research and development tax credits are exchanged for a cash refund and are typically collected within one year from the date the tax return is filed with the state. The credits are recognized as an offset to research and development expenses in the consolidated statements of operations and comprehensive loss in the annual period in which the corresponding expenses were incurred.
F-9
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
Property and Equipment, Net
Property and equipment are stated at cost less accumulated depreciation and amortization. Depreciation expense is computed using the straight-line method over the estimated useful lives of the related assets. MRI devices purchased from a third-party manufacturer that were intended for use as research units, for customer demonstration purposes to support sales efforts and training and as leased units are classified as Property and Equipment.
Useful lives of property and equipment are as follows:
Property and equipment |
|
Estimated useful life |
|
|
Laboratory equipment |
|
|
|
|
Research devices |
|
|
|
|
Sales and marketing devices |
|
|
|
|
Computer equipment |
|
|
|
|
Tooling |
|
|
|
|
Trade show assets |
|
|
|
|
Leased devices |
|
|
|
|
Other |
|
|
||
Other property and equipment include furniture and fixtures, software, vehicles, and machinery and equipment.
Expenditures for major renewals and improvements are capitalized. Expenditures for repairs and maintenance are expensed as incurred. When assets are retired or otherwise disposed of, the cost of these assets and related accumulated depreciation is eliminated from the balance sheet, and any resulting gains or losses are included in the consolidated statements of operations and comprehensive loss in the period of disposal.
Impairment of Long-Lived Assets
The Company reviews its long-lived assets for impairment at least annually or whenever events or changes in business circumstances indicate that the carrying amount of the assets may not be fully recoverable using pretax undiscounted cash flows. If the recorded value of the asset is less than the undiscounted cash flow, the asset is written down to its estimated fair value. There was
Right-of-use (“ROU”) Assets and Leases Liabilities
The Company
The adoption of ASC 842 did not result in recording ROU assets and lease liabilities as the Company has applied ASC 842 practical expedient for short-term leases, which allows entities with leases of
F-10
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
Capitalized Software Development Costs
For the costs incurred in developing the firmware embedded in the hardware devices that the Company sells and leases to its customers, the Company applies the principles of FASB Accounting Standards Codification (“ASC”) 985-20, Accounting for the Costs of Computer Software to Be Sold, Leased, or Otherwise Marketed (“ASC 985-20”). ASC 985-20 requires that software development costs incurred in conjunction with product development be charged to research and development expense until technological feasibility is established. Thereafter, until the product is released for sale, software development costs must be capitalized and reported at the lower of unamortized cost or net realizable value of the related product. The Company has adopted the “tested working model” approach to establishing technological feasibility for its software products. Under this approach, the Company does not consider a product in development to have passed the technological feasibility milestone until the Company has completed a model of the product that contains essentially all the functionality and features of the final product and has tested the model to ensure that it works as expected. The Company’s hardware device, with the embedded firmware, was released for sale during the fourth quarter of the year ended December 31, 2020, when the Company had completed all of the research and development activity to establish the technological feasibility of the product. As of December 31, 2023 and 2022, the Company had not incurred significant costs between the establishment of technological feasibility and the release of a product for sale; thus, the Company had expensed all software development costs as incurred.
For software developed or acquired for internal use, including software used in the provision of service and support to the Company’s customers, the Company applies the principles of ASC 350-40, Accounting for the Cost of Computer Software Developed or Obtained for Internal Use (“ASC 350-40”). ASC 350-40 requires that software development costs incurred before the preliminary project stage be expensed as incurred. The Company capitalizes development costs related to these software applications once the preliminary project stage is complete and it is probable that the project will be completed, and the software will be used to perform the function intended. Costs incurred during the preliminary project and post-implementation stages, including training and maintenance, are expensed as incurred. Capitalized costs are amortized on a project-by-project basis using the
Revenue Recognition
The Company recognizes revenue in accordance with ASC Topic 606, “Revenue from Contracts with Customers.”
Revenue is recognized when or as a customer obtains control of the promised goods and services. The amount of revenue recognized reflects the consideration to which the Company expects to be entitled in exchange for these goods and services. To achieve this core principle, the Company applies the following 5 steps:
• Step 1: Identify Contracts with Customers: The Company executes signed contracts with its customers for the sale of hardware devices and services.
• Step 2: Identify Performance Obligations: The Company’s contracts with customers primarily include two performance obligations, namely the hardware device and services, which include access to the Company’s hosted cloud-based software applications and hardware maintenance and support on an ongoing basis throughout the service and support period.
• Step 3: Determine Transaction Price: The Company’s contracts with customers may include variable consideration in the form of discounts and price concessions. The Company estimates variable consideration using the expected value method based on the data available as of the end of each reporting period.
F-11
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
• Step 4: Allocate Transaction Price to Performance Obligations: The Company allocates transaction price to the performance obligations in a contract with a customer, based on the relative standalone selling prices of the goods and services. The standalone selling prices of the hardware devices and services are determined based on market assessment of the standalone selling prices for which the Company sells the respective goods and services on a standalone basis, including renewals of services.
• Step 5: Recognize Revenue as Performance Obligations are Satisfied: Each unit of hardware devices is a performance obligation satisfied at a point in time, when control of the good transfers from the Company to the customer, which is typically upon shipment of the good to the customer. For sales of hardware where control of the product transfers to the customer upon shipment, the Company has made an accounting policy election to account for shipping and handling as fulfillment activities rather than a performance obligation. The services are stand-ready obligations that are satisfied over time by providing the customer with ongoing access to the Company’s resources throughout the service period. The Company uses the time elapsed (straight-line) measure of progress to recognize revenue as these performance obligations are satisfied evenly over the respective service period.
The Company offers alternative payment structures and “as-a-service” offerings that are assessed to determine whether an embedded lease arrangement exists. The Company accounts for those contracts as a lease arrangement under ASC 842. The Company identifies certain Device-as-a-Service contracts to be within the scope of ASC 842 and ASC 606. For contracts that are in the scope of both ASC 842 and ASC 606, and in which the lease component is an operating lease, the Company applies the practical expedient in ASC 842 to combine the lease component (the device itself in device as a service, ("DaaS") contracts) and non-lease (maintenance and SaaS) components, and to account for the combined components as a single lease component. Accordingly, the Company accounts for the monthly payments as lease revenue. For contracts in which the lease component is a sales-type lease, the Company derecognizes the asset (the MRI device) and recognizes a lease receivable in an amount that represents the present value of the lease payments. At times, the Company may enter into arrangements with payment terms which exceed one year from the transfer of control of the product or service. In such cases, the Company assesses whether the arrangement contains a significant financing component. If a significant financing component exists, the transaction price is adjusted for the financing portion of the arrangement, which is recorded as interest income over the payment term using the effective interest method. The Company does not assess whether a significant financing component exists when, at contract inception, the period between the transfer of control to a customer and final payment is one year or less.
Deferred Revenue
Deferred revenue primarily consists of billings or payments received in advance of revenue recognition from services described above and is reduced as the revenue recognition criteria are met. Deferred revenue is classified as current or noncurrent based on expected revenue recognition timing. Specifically, deferred revenue that will be recognized as revenue within the succeeding 12 month period is recorded as deferred revenue as part of current liabilities and the remaining portion is recorded as long term deferred revenue in the Company’s consolidated balance sheets.
Warranties
The Company offers a device warranty to customers usually for 12 months from delivery of the device as well as for devices obtained through a capital purchase. The Company’s services include hardware maintenance and support. As noted in the accounting policy for revenue recognition, the Company recognizes revenue for service over time using the time elapsed measure of progress. The costs of hardware maintenance are recognized in costs of revenue as they are incurred.
Research and Development
Research and development costs consist of production costs for prototype, test and pre-production units, lab supplies, consulting and personnel costs, including salaries, stock-based compensation, bonuses, benefit costs and depreciation. Certain research and development grant funding is recognized as a reduction to research and development costs (see Note 2. Summary of Significant Accounting Policies - Grant Funding). The Company recognizes these costs as they are incurred.
F-12
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
Grant Funding
The Company received certain research and development funding through a grant issued by the Bill & Melinda Gates Foundation (“BMGF”). Funding is recorded on the consolidated balance sheet as restricted cash upon receipt. The funding is recognized in the consolidated statements of operations and comprehensive loss as a reduction to research and development expense in the period when milestone deliverables are fulfilled and met. Grant funding payments received in advance of research and development expenses incurred are recorded as deferred grant funding as a current liability in the Company’s consolidated balance sheets.
Cost of Sales
Cost of sales consists of product and service costs including personnel cost and benefits including stock-based compensation, product costs, production setup expenses, depreciation expenses, inventory excess and obsolescence expenses.
Patent Costs
Patent costs have been charged to operations as incurred, as their realization is uncertain. These costs are included in general and administrative expenses in the consolidated statements of operations and comprehensive loss.
General and Administrative
General and administrative expenses primarily consist of personnel costs and benefits including stock-based compensation, patent and filing fees, office expenses and outside services. Outside services consist of professional services, legal and other professional fees.
Sales and Marketing
Sales and marketing costs primarily consist of personnel costs and benefits including stock-based compensation, advertising, promotional, as well as conferences, meetings, and other events. Advertising costs are expensed as incurred. For the years ended December 31, 2023 and 2022, advertising expenses were $
Net Loss per Common Share
Basic net loss per common share is calculated by dividing the net loss attributed to common stockholders by the weighted average number of common shares outstanding during the period, without consideration of potentially dilutive securities.
Diluted net loss per common share is computed by dividing the net loss attributable to common stockholders by the weighted average number of common shares plus the common equivalent shares of the period, including any dilutive effect from such shares. The Company’s diluted net loss per common share is the same as basic net loss per common share for all periods presented since the effect of potentially dilutive securities is anti-dilutive.
Stock-Based Compensation
The measurement of stock-based compensation expense for all stock-based payment awards, including stock options granted to employees, directors, and consultants, is based on the estimated fair value of the awards on the date of grant.
The Company recognizes stock-based compensation expense for stock option grants and incentive unit grants with only service conditions on a straight-line basis over the requisite service period of the individual grants, which is generally the vesting period, based on the estimated grant date fair values. Generally, stock option grants and incentive unit grants fully vest
F-13
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
The Company recognizes the effect of forfeiture in compensation costs based on actual forfeitures when they occur.
The Company's stock-based compensation program includes stock option grants to its employees, directors, and consultants. Stock options are granted at exercise prices not less than the estimated fair market value of the Company’s common stock at the date of grant.
The fair values of stock option grants are estimated using a Black-Scholes option-pricing model. Key inputs and assumptions include the expected term of the option, stock price volatility, risk free interest rate, dividend yield, stock price and exercise price. Many of the assumptions require significant judgment and any changes could have a material impact in the determination of stock-based compensation expense.
Earn-Out Shares
Earn-Out Shares, as defined in Note 10. Net Loss Per Share, to which the Companies’ pre-closing equity holders are entitled, fall within the scope of ASC 815, Derivatives and Hedging (“ASC 815”) pursuant to which such Earn-Out Shares are equity classified and are to be recognized upon achievement of the market price milestone.
Earn-Out Shares to which certain employees are entitled to fall within the scope of ASC 718, Compensation – Stock Compensation (“ASC 718”), pursuant to which such Earn-Out Shares are equity classified and their grant date fair value will be recognized as compensation expense over the vesting period.
Commitments and Contingencies
The Company periodically evaluates all pending or threatened contingencies and any commitments, if any, that are reasonably likely to have a material adverse effect on its results of operations, financial position or cash flows. The Company assesses the probability of an adverse outcome and determines if it is remote, reasonably possible or probable. If information available prior to the issuance of the Company’s financial statements indicates that it is probable that an asset had been impaired or a liability had been incurred at the date of the Company’s financial statements, and the amount of the loss, or the range of probable loss can be reasonably estimated, then such loss is accrued and charged to operating expenses. If no accrual is made for a loss contingency because one or both of the conditions pursuant to the accounting guidance are not met, but the probability of an adverse outcome is at least reasonably possible, the Company discloses the nature of the contingency and provides an estimate of the possible loss or range of loss, or states that such an estimate cannot be made.
Research and Development Expenses and Research and Development Tax Credits
The Company recognizes research and development tax credits as a reduction of research and development expense as earned. For State of Connecticut research and development tax credits, which are exchanged for a cash refund from the State of Connecticut, such exchanged credits are recognized as earned as a reduction of research and development expense in the consolidated statements of operations and comprehensive loss.
Income Taxes
The Company utilizes the asset and liability method of accounting for income taxes, as set forth in ASC Topic 740, Income Taxes. Under this method, deferred tax assets and liabilities are recognized for the expected future tax consequences of temporary differences between the carrying amounts and the tax basis of assets and liabilities using the enacted statutory tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. A valuation allowance is established against net deferred tax assets if, based on the weight of available evidence, it is more likely than not that some or all of the net deferred tax assets will not be realized. The Company has recorded a full valuation allowance as of December 31, 2023 and 2022. Based on the available evidence, the Company believes that it is more likely than not that it will be unable to utilize all of its deferred tax assets in the future.
F-14
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
In accordance with the provisions of ASC Topic 740, the Company accrues for the estimated amount of taxes for uncertain tax positions if it is more likely than not that the Company would be required to pay such additional taxes. An uncertain tax position will not be recognized if it has a less than 50% likelihood of being sustained. The Company’s policy is to recognize any interest and penalties related to income taxes in income tax expense in the consolidated statements of operations and comprehensive loss. The Company’s open tax years subject to examination by the relevant taxing authorities are . As of December 31, 2023 and 2022, the Company had
Recent Accounting Pronouncements
Accounting pronouncements adopted
In June 2016, the FASB issued ASU 2016-13, Financial Instruments – Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments, which replaces the current incurred loss impairment methodology with a methodology that reflects expected credit losses and requires consideration of a broader range of reasonable and supportable information to inform credit loss estimates. The standard also requires that financial assets measured at amortized cost be presented at the net amount anticipated to be collected via an allowance for credit losses that is deducted from the amortized cost basis. Pursuant to ASU 2016-13, the Company is required to measure all expected credit losses based upon historical experience, current conditions, and reasonable (and supportable) forecasts that affect the collectability of the financial asset. The Company
Accounting pronouncements issued but not yet adopted
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures. The ASU requires that an entity disclose specific categories in the effective tax rate reconciliation as well as reconciling items that meet a quantitative threshold. Further, the ASU requires additional disclosures on income tax expense and taxes paid, net of refunds received, by jurisdiction. The new standard is effective for annual periods beginning after December 15, 2024 on a prospective basis with the option to apply it retrospectively. Early adoption is permitted. The adoption of this guidance will result in the Company being required to include enhanced income tax related disclosures.
In November 2023, the FASB issued ASU 2023-07, Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures. This ASU includes amendments that expand the existing reportable segment disclosure requirements and requires disclosure of (i) significant expense categories and amounts by reportable segment as well as the segment’s profit or loss measure(s) that are regularly provided to the chief operating decision maker (the “CODM”) to allocate resources and assess performance; (ii) how the CODM uses each reported segment profit or loss measure to allocate resources and assess performance; (iii) the nature of other segment balances contributing to reported segment profit or loss that are not captured within segment revenues or expenses; and (iv) the title and position of the individual or name of the group or committee identified as the CODM. This guidance requires retrospective application to all prior periods presented in the financial statements and is effective for fiscal years beginning after December 15, 2023 and interim periods within fiscal years beginning after December 15, 2024. Early adoption is permitted. The adoption of this guidance will result in the Company being required to include enhanced disclosures relating to its reportable segments.
F-15
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
3. REVENUE RECOGNITION
Disaggregation of Revenue
The Company disaggregates revenue from contracts with customers by product type. The Company believes that these categories aggregate the payor types by nature, amount, timing and uncertainty of its revenue streams.
|
|
Pattern of Recognition |
|
2023 |
|
|
2022 |
|
|
||
Device |
|
Point in time |
|
$ |
|
|
$ |
|
|
||
Service |
|
Over time |
|
|
|
|
|
|
|
||
Total revenue |
|
|
|
$ |
|
|
$ |
|
|
||
Contract Balances
Contract balances represent amounts presented in the consolidated balance sheets when either the Company has transferred goods or services to the customer, or the customer has paid consideration to the Company under the contract. These contract balances include trade accounts receivable and deferred revenue. Deferred revenue represents consideration received from customers at the beginning of the service and support period for services that are transferred to the customer over the respective service and support period. The accounts receivable balances represent amounts billed to customers for goods and services where the Company has an unconditional right to payment of the amount billed.
The following table provides information about receivables and deferred revenue from contracts with customers:
|
|
2023 |
|
|
2022 |
|
||
Accounts receivable |
|
$ |
|
|
$ |
|
||
Unbilled receivables - current |
|
|
|
|
|
|
||
Unbilled receivables - non-current (1) |
|
|
|
|
|
|
||
Deferred revenue |
|
|
|
|
|
|
||
Long term deferred revenue |
|
|
|
|
|
|
||
_________________________
(1)
The Company recognizes a receivable when it has an unconditional right to payment, and payment terms range from
Accounts Receivable, Unbilled Services, and Deferred Revenue
Accounts receivable are recorded at net realizable value. Unbilled receivables arise when performance obligations are satisfied for which revenue has been recognized but the customers have not been billed. Contractual provisions and payment schedules may or may not correspond to the timing of the performance of services under the contract.
Deferred revenue is a contact liability that consists of customer payments received in advance of performance and billings in excess of revenue recognized, net of revenue recognized from the balance at the beginning of the period.
The amount of revenue recognized during the years ended December 31, 2023 and 2022 that was included in the deferred revenue balance at the beginning of the period was $
F-16
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
Timing of Billing and Performance
Difference in the timing of revenue recognition and associated billings and cash collections result in recording of billed accounts receivable, unbilled accounts receivable (including contract assets), and deferred revenue on the consolidated balance sheet. Amounts are billed in accordance with the agreed-upon contractual terms, resulting in recording unbilled accounts receivable in instances where the right to bill is contingent solely on the passage of time, and contract assets in instances where the right to consideration is conditional on something other than the passage of time.
Revenue from Leasing Arrangements
Revenue from leasing arrangements is not subject to the revenue standard for contracts with customers and remains separately accounted for under ASC 842, Leases for the years ended December 31, 2023 and 2022. The Company records operating lease rental revenue as service revenue on a straight-line basis over the lease term. The Company records revenue from the sale of hardware devices under sales-type leases as device revenue in an amount equal to the present value of minimum lease payments at the inception of the lease. Sales-type leases also produce financing income, which is included in device revenue in the consolidated statements of operations and comprehensive loss and is recognized at effective rates of return over the lease term.
Costs of Obtaining or Fulfilling Contracts
The Company incurs incremental costs of obtaining contracts with customers. Incremental costs of obtaining contracts, which include commissions paid as a result of obtaining contracts with customers, are capitalized to the extent that the Company expects to recover such costs. Capitalized costs are amortized in a pattern that is consistent with the Company’s transfer to the customer of the related goods and services. Such costs are recorded in Other long term assets and were $
Transaction price allocated to remaining performance obligations
As of December 31, 2023 and 2022, the Company had remaining performance obligations amounting to $
Significant Judgements
The Company makes significant judgments applying the guidance related to the determination of the timing and pattern of satisfaction of performance obligations, determination of the standalone selling price of performance obligations and estimation of variable consideration if any.
The Company’s products are generally sold without a right of return, and the Company’s contracts generally provide a fixed transaction price. The Company reviews payment terms extending beyond one year. If it is determined that a material financing component exists, the Company recognize this as interest income over the financing term. The Company applies the practical expedient to not adjust for a material financing component if the gap between payment and delivery was expected, at the contract inception, to be less than one year.
The SSP of performance obligations is determined based on market assessment of the SSPs for which the Company expects to sell the respective goods and services on a standalone basis, including renewals of services.
The contract consideration allocation is based on the SSP at contract inception. The consideration (net of any discounts) is allocated among separate products and services based on their relative SSPs. Contract modifications typically add additional goods or services or change pricing. For such modifications, the most recent SSP is used for reallocation to the remaining performance obligations.
F-17
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
The Company recognizes revenue for certain performance obligations at the point in time when control is transferred, such as the delivery of products. Service revenue is recognized over the term of the service period as the customer benefits from the services throughout the service period.
Practical Expedients and Accounting Policy Elections
As a practical expedient, the Company does not adjust transaction price for the effects of a significant financing component in contracts in which the period between when the Company transfers the promised good or service to the customer and when the customer pays for that good or service is one year or less.
The Company has made an accounting policy election to exclude all sales taxes from the transaction price of its contracts with customers. Accordingly, sales taxes collected from customers and remitted to government authorities are not included in revenue and are accounted for as a liability until they have been remitted to the respective government authority.
4. FAIR VALUE OF FINANCIAL INSTRUMENTS
Fair value estimates of financial instruments are made at a specific point in time, based on relevant information about financial markets and specific financial instruments. As these estimates are subjective in nature, involving uncertainties and matters of significant judgment, they cannot be determined with precision. Changes in assumptions can significantly affect estimated fair value.
The Company measures fair value as the price that would be received to sell an asset or paid to transfer a liability (an exit price) in an orderly transaction between market participants at the reporting date. The Company utilizes a three-tier hierarchy, which prioritizes the inputs used in the valuation methodologies in measuring fair value:
Level 1 — Valuations based on quoted prices in active markets for identical assets or liabilities that an entity has the ability to access.
Level 2 — Valuations based on quoted prices for similar assets or liabilities, quoted prices for identical assets or liabilities in markets that are not active, or other inputs that are observable or can be corroborated by observable data for substantially the full term of the assets or liabilities.
Level 3 — Valuations based on inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities. The Company has no assets or liabilities valued with Level 3 inputs.
The carrying value of cash and cash equivalents, accounts payable and accrued expenses and other current liabilities approximates their fair values due to the short-term or on demand nature of these instruments.
There were
The Company had $
5. INVENTORIES
A summary of inventories are as follows at December 31:
|
|
2023 |
|
|
2022 |
|
||
Raw materials |
|
$ |
|
|
$ |
|
||
Finished goods |
|
|
|
|
|
|
||
Total inventories |
|
$ |
|
|
$ |
|
||
F-18
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
Manufacturing overhead costs primarily include management’s best estimate and allocation of the labor costs incurred related to acquiring finished goods from the Company’s contract manufacturer. Labor costs include wages, taxes and benefits for employees involved in warehousing, logistics coordination, material sourcing, and production planning activities. The majority of these costs have been written off based on the Company’s analysis of net realizable value.
6. PROPERTY AND EQUIPMENT, NET
Property and equipment, net, are recorded at historical cost and consist of the following at December 31:
|
|
2023 |
|
|
2022 |
|
||
Laboratory equipment |
|
$ |
|
|
$ |
|
||
Research devices |
|
|
|
|
|
|
||
Sales and marketing devices |
|
|
|
|
|
|
||
Computer equipment |
|
|
|
|
|
|
||
Construction in progress |
|
|
|
|
|
|
||
Tooling |
|
|
|
|
|
|
||
Trade show assets |
|
|
|
|
|
|
||
Leased devices |
|
|
|
|
|
|
||
Other |
|
|
|
|
|
|
||
Gross property and equipment |
|
|
|
|
|
|
||
Less: Accumulated depreciation and amortization |
|
|
( |
) |
|
|
( |
) |
Property and equipment, net |
|
$ |
|
|
$ |
|
||
Depreciation expense amounted to $
7. ROU ASSETS AND LEASES LIABILITIES
The Company has operating leases for its corporate offices. The Palo Alto, California lease agreement includes an option to renew the lease, which was not reasonably certain to be exercised, and therefore is not factored into the determination of lease payments. At the lease inception, the Company applied ASC 842 practical expedient for short-term leases, which allows entities with leases of
The weighted-average remaining lease term associated with the measurement of our operating lease obligations is
The Company recorded short-term operating lease cost during the year ended December 31, 2023 of $
Future minimum commitments due under the lease agreement as of December 31, 2023, are $
F-19
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
8. ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
Accrued expenses and other current liabilities consist of the following at December 31:
|
|
2023 |
|
|
2022 |
|
||
Bonuses |
|
$ |
|
|
$ |
|
||
Contracted services |
|
|
|
|
|
|
||
Legal fees |
|
|
|
|
|
|
||
Payroll and related benefits |
|
|
|
|
|
|
||
Operating lease liabilities |
|
|
|
|
|
— |
|
|
Other |
|
|
|
|
|
|
||
Total accrued expenses and other current liabilities |
|
$ |
|
|
$ |
|
||
9. STOCKHOLDERS' EQUITY
Common Stock
In November 2023, the Company filed a shelf registration statement on Form S-3 with the SEC pursuant to which the Company registered for sale up to $
Equity Incentive Plans
Hyperfine Inc. 2021 Equity Incentive Plan and Inducement Option Grant
The Company’s equity incentive plans include the Company’s 2021 Equity Incentive Plan (the “Hyperfine Plan”) and the Company has made an inducement option grant outside of the Hyperfine Plan in accordance with Nasdaq Listing Rule 5635(c)(4). The Hyperfine Plan is administered by the Company's board of directors. The board of directors may grant restricted stock and options to purchase shares either as incentive stock options or non-qualified stock options. The option grants are subject to certain terms and conditions, option periods and conditions, exercise rights and privileges as set forth in the Hyperfine Plan. During the year ended December 31, 2023, the number of shares available for grant increased by
Prior to the Business Combination, Legacy Hyperfine and Liminal were distinct entities with separate equity incentive plans for their employees and nonemployees. Both plans were subsequently adopted and assumed by the Company as a consequence of the Business Combination.
Stock option activity
Each stock option grant carries varying vesting schedules whereby the options become exercisable at the participant’s sole discretion provided they are an employee, director or consultant of the Company on the applicable vesting date. Each option shall terminate not more than ten years from the date of the grant.
F-20
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
During the first quarter of 2023, the Company granted inducement stock options to purchase
During the fourth quarter of 2022, the Company granted certain equity awards to the newly hired Chief Executive Officer. These awards include an option award to purchase
During the fourth quarter of 2022, the Company initiated and carried out certain restructuring actions in order to reduce costs, including termination of employees. As part of the employees’ severance, for certain employees the Company accelerated the vesting of unvested awards including
During the years ended December 31, 2022 and 2021, the Company granted certain equity awards to the Chief Executive Officer hired during 2021. These awards include (1) an option award to purchase
Certain equity awards were also granted to the Chairperson of the Legacy Hyperfine board of directors. The equity compensation included (1) an option award to purchase
As of December 31, 2023, there was an outstanding award of non-qualified stock options to purchase
F-21
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
The Company’s nonemployee directors were each granted non-qualified stock options to purchase
All options granted by the Company during the years ended December 31, 2023 and 2022 were granted with exercise prices equal to the estimated fair value of the Company's common stock at the date of grant, as determined by the Company's board of directors.
A summary of the stock option activity under the Hyperfine Plan is presented in the table below:
|
|
Number of Options |
|
|
Weighted Average Exercise Price |
|
|
Weighted Average Remaining Contractual Term |
|
|
Aggregate Intrinsic Value |
|
||||
Outstanding at January 1, 2022 |
|
|
|
|
$ |
|
|
|
|
|
$ |
|
||||
Granted |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Exercised |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|||
Forfeited |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|||
Outstanding at December 31, 2022 |
|
|
|
|
$ |
|
|
|
|
|
$ |
|
||||
Granted |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Exercised |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|||
Forfeited / Expired |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|||
Outstanding at December 31, 2023 |
|
|
|
|
$ |
|
|
|
|
|
|
|
||||
Options exercisable at December 31, 2023 |
|
|
|
|
$ |
|
|
|
|
|
$ |
|
||||
Vested or expected to vest at December 31, 2023 |
|
|
|
|
$ |
|
|
|
|
|
$ |
|
||||
The Company received cash proceeds from the exercise of stock options of $
The total intrinsic value (the amount by which the stock price exceeds the exercise price of the option on the date of exercise) of the stock options exercised during the years ended December 31, 2023 and 2022, was $
During the years ended December 31, 2023 and 2022, the Company recognized $
Tax benefits from tax deductions for exercised options and disqualifying dispositions in excess of the deferred tax asset attributable to stock compensation costs for such options are credited to additional paid‑in capital. The benefits are recognized against income taxes. Realized excess tax benefits related to stock options exercises was
As of December 31, 2023, there was approximately $
Stock Option Repricing
On September 26, 2022, stockholders controlled by Jonathan M. Rothberg, Ph.D., a member of the board of directors of the Company, representing a majority in voting power with respect to the Company’s Class A common stock and the Company’s Class B common stock, approved a one-time stock option repricing (the “Option Repricing”). The Board of Directors of the Company previously approved the Option Repricing on
F-22
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
stockholder approval of the Option Repricing. Stock options held by Dr. Rothberg were not repriced in the Option Repricing. The Option Repricing was effected on
The Company treated the repricing as a modification of the original awards and calculated additional compensation costs for the difference between the fair value of the modified award and the fair value of the original award on the modification date. The repricing of the awards with service conditions resulted in incremental stock-based compensation expense of $
The repricing of the awards with market and performance-based conditions resulted in incremental stock-based compensation expense of $
Stock option valuation inputs
The Company utilized the Black-Scholes option pricing model for determining the estimated fair value for service awards. The Black-Scholes model requires the use of subjective assumptions which determine the fair value of stock-based awards.
|
|
2023 |
|
2022 |
|
Risk Free interest rate |
|
|
|
||
Expected dividend yield |
|
|
|
||
Expected term |
|
|
|
||
Expected volatility |
|
|
|
Risk free interest rate
The risk free interest rate for periods within the expected term of the awards is based on the U.S. Treasury yield curve in effect at the time of the grant.
Expected dividend yield
The Company has never declared or paid any cash dividends and does not expect to pay any cash dividends in the foreseeable future.
Expected term
For employee awards, the Company calculates the expected term using the “simplified” method, which is the simple average of the vesting period and the contractual term. The simplified method is applied as the Company does not have sufficient historical data to provide a reasonable basis for an estimate of the expected term. The Company calculates expected term for employee awards that take into account the effects of employee’s expected exercise and post-vesting employment termination behavior.
Expected volatility
During the years ended December 31, 2023 and 2022, the expected volatility was determined using the historical volatilities of several publicly listed peer companies over a period equivalent to the expected term of the awards, as the Company does not have sufficient historical data to provide a reasonable basis for an estimate of the expected volatility.
F-23
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
Exercise price
The exercise price is taken directly from the grant notice issued to employees and nonemployees.
The stock options granted to the Company’s employees and nonemployees for the periods presented were as follows:
|
|
2023 |
|
|
2022 |
|
|
||
Stock options granted to employees |
|
|
|
|
|
|
|
||
Stock options granted to nonemployee directors |
|
|
|
|
|
|
|
||
Total stock options granted |
|
|
|
|
|
|
|
||
Restricted Stock Units
|
|
Number of |
|
|
Outstanding at January 1, 2022 |
|
|
|
|
Granted |
|
|
|
|
Released |
|
|
( |
) |
Forfeited |
|
|
( |
) |
Expired |
|
|
( |
) |
Outstanding at December 31, 2022 |
|
|
|
|
Granted |
|
|
|
|
Released |
|
|
( |
) |
Forfeited |
|
|
( |
) |
Expired |
|
|
— |
|
Outstanding at December 31, 2023 |
|
|
|
|
During the year ended December 31, 2023, the Company granted
The Company recognized $
On June 27, 2022, David Scott delivered his resignation as the Company’s President, Chief Executive Officer and member of the Board of Directors, effective as July 29, 2022. Pursuant to the terms of the Restricted Stock Unit Award Grant Notice and Agreement dated April 26, 2022 with Mr. Scott under the Hyperfine Plan, the
F-24
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
Earn-Out Shares
During the years ended December 31, 2023 and 2022,
During the years ended December 31, 2023 and 2022, the Company recorded $
Earn-out valuation inputs
The Company utilized a Monte Carlo Simulation pricing model for determining the estimated fair value for Earn-Out Shares. The fair value is based on the simulated price of the Company over the maturity date of the Earn-Out Shares.
|
|
2021 |
|
|
|
Stock Price |
|
|
|
||
Risk Free interest rate |
|
|
% |
|
|
Expected dividend yield |
|
|
% |
|
|
Term (years) |
|
|
|
||
Expected volatility |
|
|
% |
|
|
Risk free interest rate
The risk free interest rate for periods within the expected term of the awards is based on the U.S. Treasury yield curve in effect at the time of the grant.
Expected dividend yield
The Company has never declared or paid any cash dividends and does not expect to pay any cash dividends in the foreseeable future.
Expected term
For Earn-Out Shares, the expected term is determined to be
Expected volatility
As Legacy Hyperfine was privately held from inception through the Closing, there was no specific historical or implied volatility information available.
Accordingly, the Company estimates the expected volatility on the historical stock volatility of a group of similar companies that are publicly traded over a period equivalent to the expected term of the earn-out awards. A point estimate of expected annual equity volatility of
F-25
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
Stock-Based Compensation Expense
The Company’s stock-based compensation expense for the periods presented was as follows:
|
|
2023 |
|
|
2022 |
|
|
||
Cost of sales |
|
|
|
|
|
|
|
||
Research and development |
|
|
|
|
|
|
|
||
General and administrative |
|
|
|
|
|
|
|
||
Sales and marketing |
|
|
|
|
|
|
|
||
Total stock-based compensation expense |
|
$ |
|
|
$ |
|
|
||
Total unrecognized stock-based compensation expense as of December 31, 2023 was $
10. NET LOSS PER SHARE
Basic net loss per share is computed by dividing the net loss by the weighted-average number of shares of common stock of the Company outstanding during the period. Diluted net loss per share is computed by giving effect to all common equivalent shares of the Company, including outstanding stock options, RSUs and Earn-Out Shares, to the extent dilutive. Basic and diluted net loss per share was the same for each period presented as the inclusion of all common equivalent shares of the Company outstanding would have been anti-dilutive.
The following table presents the calculation of basic and diluted net loss per share for the Company’s common stock:
|
|
2023 |
|
|
2022 |
|
|
||
Numerator: |
|
|
|
|
|
|
|
||
Net Loss |
|
$ |
( |
) |
|
$ |
( |
) |
|
Numerator for Basic and Dilutive net loss per share – loss available to common |
|
$ |
( |
) |
|
$ |
( |
) |
|
Denominator: |
|
|
|
|
|
|
|
||
Common Stock |
|
|
|
|
|
|
|
||
Denominator for Basic and Dilutive net loss per share weighted – average common stock |
|
|
|
|
|
|
|
||
Basic and dilutive net loss per share |
|
$ |
( |
) |
|
$ |
( |
) |
|
Net loss per share attributable to Class A and Class B common stockholders was the same on a basic and diluted basis, as the inclusion of all common equivalent shares outstanding would have been anti-dilutive.
|
|
2023 |
|
|
2022 |
|
|
||
Outstanding options to purchase common stock |
|
|
|
|
|
|
|
||
Outstanding RSUs |
|
|
|
|
|
|
|
||
Earn-Out Shares (1) |
|
|
|
|
|
|
|
||
Total anti-dilutive common equivalent shares |
|
|
|
|
|
|
|
||
_________________________
(1) The Company will issue to holders of Legacy Hyperfine and Liminal securities as of immediately prior to the effective time of the Mergers, in accordance with their pro rata share, up to
F-26
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
11. INCOME TAXES
Significant components of the Company’s deferred tax assets (liabilities) are as follows:
|
|
As of December 31, |
|
|||||
|
|
2023 |
|
|
2022 |
|
||
Gross deferred tax assets (liabilities): |
|
|
|
|
|
|
||
Net operating loss carryforwards |
|
$ |
|
|
$ |
|
||
Tax credit carryforwards |
|
|
|
|
|
|
||
Fixed assets |
|
|
( |
) |
|
|
( |
) |
Stock-based compensation |
|
|
|
|
|
|
||
Capitalized R&D |
|
|
|
|
|
|
||
Deferred revenue |
|
|
|
|
|
|
||
Accrued bonuses |
|
|
— |
|
|
|
— |
|
Other |
|
|
|
|
|
|
||
Total deferred tax assets |
|
|
|
|
|
|
||
Valuation allowance |
|
|
( |
) |
|
|
( |
) |
Net deferred tax assets (liabilities) |
|
$ |
— |
|
|
$ |
— |
|
The Company had
|
|
As of December 31, |
|
|||||
|
|
2023 |
|
|
2022 |
|
||
Statutory tax rate |
|
|
% |
|
|
% |
||
State taxes, net of federal benefit |
|
|
% |
|
|
% |
||
Federal research and development credit |
|
|
( |
)% |
|
|
% |
|
Stock-based compensation |
|
|
( |
)% |
|
|
( |
)% |
Deferred tax adjustment resulting from tax rate change |
|
|
( |
)% |
|
|
( |
)% |
Other |
|
|
( |
)% |
|
|
( |
)% |
Valuation allowance |
|
|
( |
)% |
|
|
( |
)% |
Effective tax rate |
|
|
% |
|
|
% |
||
The Company’s effective tax rate for December 31, 2023 and 2022 differs from the federal statutory tax rate of
The Company has established a full valuation allowance against its net deferred tax assets due to the uncertainty of the Company’s ability to generate sufficient taxable income to realize the deferred tax assets, and therefore has not recognized any benefits from the net operating losses, tax credits and other deferred tax assets. The Company’s valuation allowance increased $
F-27
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
As of December 31, 2023, the Company had the following tax net operating loss carryforwards available to reduce future federal and state taxable income, and tax credit carryforwards available to offset future federal and state income taxes:
|
|
Hyperfine |
||||
|
|
Amount |
|
|
Begin to Expire in |
|
Hyperfine tax net operating loss carryforwards: |
|
|
|
|
|
|
Federal (pre-2018 NOLs) |
|
$ |
|
|
||
Federal (post-2017 NOLs) |
|
|
|
|
No Expiration |
|
States |
|
|
|
|
||
Tax credit carryforwards: |
|
|
|
|
|
|
Federal research and development |
|
|
|
|
||
Connecticut research and development |
|
|
|
|
No Expiration |
|
Connecticut others |
|
|
|
|
||
Federal others |
|
|
|
|
||
|
|
Liminal |
||||
|
|
Amount |
|
|
Begin to Expire in |
|
Liminal tax net operating loss carryforwards: |
|
|
|
|
|
|
Federal (pre-2018 NOLs) |
|
$ |
— |
|
|
|
Federal (post-2017 NOLs) |
|
|
|
|
No Expiration |
|
States |
|
|
|
|
||
Tax credit carryforwards: |
|
|
|
|
|
|
Federal research and development |
|
|
|
|
||
Connecticut research and development |
|
|
|
|
No Expiration |
|
Federal and state other |
|
|
|
|
||
Under Internal Revenue Code Section 382, if a corporation undergoes an “ownership change,” the corporation’s ability to use its pre-change net operating loss and tax credit carryforwards to offset its post-change income and tax liabilities may be limited. Generally,
The Company has adopted the accounting guidance within ASC Topic 740 on uncertainties in income taxes. ASC Topic 740 prescribes a recognition threshold and measurement attribute for the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return.
As of December 31, 2023 and 2022, the Company did
F-28
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
The Company files income tax returns in the U.S. federal and various state jurisdictions. As a result of the Company’s net operating loss carryforwards, the Company’s federal and state statutes of limitations generally remain open for all tax years until its net operating loss and tax credit carryforwards are utilized or expire prior to utilization. The Company does not currently have any federal or state income tax examinations in progress.
Additionally, as a result of legislation in the state of Connecticut, companies have the opportunity to exchange certain research and development tax credit carryforwards for a cash payment of
12. RELATED PARTY TRANSACTIONS
The Company utilizes and subleases office and lab space in Connecticut which is being leased from an unrelated landlord by 4Catalyzer Corporation, (“4C”), which is owned by a related party. The Company pays rent to 4C on a month-to-month basis. A total of approximately $
Legacy Hyperfine and Liminal each entered into a Master Services Agreement (the “Master Services Agreements”) with 4C effective as of July 7, 2021 pursuant to which Legacy Hyperfine and Liminal may engage 4C to provide services such as general administration, facilities, information technology, financing, legal, human resources and other services, through future statements of work and under terms and conditions to be determined by the parties with respect to any services to be provided. The Company incurred expenses from 4C of $
In January 2018, the Company entered into a Promissory Note (the “Note”) with one of its employees (the “Borrower”) in the amount of $
Legacy Hyperfine and Liminal entered into Technology and Services Exchange Agreements (each, a “TSEA” and collectively, the “TSEA”) with other participant companies controlled by the Rothbergs. A TSEA by and among Butterfly Network, Inc., AI Therapeutics, Inc., Quantum-Si Incorporated, 4Bionics, identifeye Health Inc. (f/k/a Tesseract Health, Inc.) /, Detect, Inc. (f/k/a Homodeus Inc.), Legacy Hyperfine and Liminal was signed in November 2020; a TSEA by and among Quantum-Si Incorporated, AI Therapeutics, Inc., 4Bionics, identifeye Health Inc., Detect, Inc., Legacy Hyperfine and Liminal was signed in February 2021 (and which Protein Evolution, Inc. joined in August 2021); and a TSEA by and among Legacy Hyperfine, Liminal, AI Therapeutics, Inc., identifeye Health Inc. and Detect, Inc. was signed in July 2021 and became effective upon the Closing. Under the TSEA, Legacy Hyperfine, Liminal and other participant companies may, in their discretion, permit the use of non-core technologies, which include any technologies, information or equipment owned or otherwise controlled by the participant company that are not specifically related to the core business area of the participant, such as software, hardware, electronics, fabrication and supplier information, vendor lists and contractor lists, by other participant companies. There were
F-29
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
13. COMMITMENTS AND CONTINGENCIES
Commitments
The Company sponsors a 401(k) defined contribution plan covering all eligible U.S. employees. Contributions to the 401(k) plan are discretionary. The Company did not make any matching contributions to the 401(k) plan for the years ended December 31, 2023 or 2022.
During 2020 and 2021, the Company was awarded multiple grants totaling $
In March 2023, the term of the BMGF grant agreement was extended to February 2024. As of December 31, 2023, 22 Swoop® system units have been provisioned and delivered to the BMGF and certain milestones for service deliverables were met. In May 2023, the Company was awarded an additional $
Purchase Commitments
The Company’s purchase commitments and obligations include all open purchase orders and contractual obligations in the ordinary course of business, including commitments with contract manufacturers and suppliers, for which the Company has not received the goods or services. A majority of these purchase obligations are due within a year. Although open purchase orders are considered enforceable and legally binding, the terms generally allow the Company the option to cancel, reschedule, and adjust its requirements based on the Company’s business needs prior to the delivery of goods or performance of services.
Contingencies
The Company does not have any outstanding or ongoing litigation and legal matters where, based on present information, including its assessment of the merits of the particular claims, the Company believes it is reasonably possible that any asserted or unasserted legal claims or proceedings, individually or in aggregate, will have a material adverse effect on its results of operations or financial condition. The ultimate outcome of any legal matter cannot be predicted with certainty.
The Company has indemnification obligations under some agreements that the Company enters into with other parties in the ordinary course of business, including business partners, investors, contractors, and the Company’s officers, directors and certain employees. The Company has agreed to indemnify and defend the indemnified party claims and related losses suffered or incurred by the indemnified party from actual or threatened third-party claim because of the Company’s activities or non-compliance with certain representations and warranties made by the Company. It is not possible to determine the maximum potential loss under these indemnification provisions due to the Company’s limited history of prior indemnification claims and the unique facts and circumstances involved in any particular case. To date, losses recorded in the consolidated statements of operations and comprehensive loss in connection with the indemnification provisions have not been material.
F-30
HYPERFINE, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS OF AND FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
(all amounts are in thousands, except share and per share data)
The Company agreed to pay $
14. RESTRUCTURING
During the quarter ended December 31, 2022, the Company initiated and carried out certain restructuring actions in order to reduce costs and improve efficiency. As a result, the Company recognized $
15. SUBSEQUENT EVENTS
The Company has evaluated subsequent events through the date the financial statements were issued and has determined that there were no subsequent events required to be disclosed.
F-31