UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
___________________________
Form 20-F
___________________________
| REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR (g) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
OR
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the fiscal year ended December 31 , 2023
OR
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
OR
| SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
___________________________
Commission File Number: 001-38455
___________________________
(Exact name of registrant as specified in its charter)
___________________________
| 2834 | Not Applicable | |||||||
| (State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification No.) | ||||||
Chief Financial Officer
Telephone: +49 89-89927-0
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
___________________________
Securities registered or to be registered pursuant to Section 12(b) of the Act:
| Title of class | Trading Symbol(s) | Name of each exchange on which registered | ||||||||||||
* Not for trading, but only in connection with the registration of the American Depositary Shares.
Securities registered or to be registered pursuant to Section 12(g) of the Act:
None
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act:
None
Indicate the number of outstanding shares of each of the issuer’s classes of capital or common stock as of the close of the period covered by the annual report:
Ordinary shares, no-par-value per share: 37,655,137 as of December 31, 2023
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
If this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934.
Yes £ No S
Indicate by check mark whether the registrant (1) has filed all reports required to be filed pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is an accelerated filer, a large accelerated filer, a non-accelerated filer, or an emerging growth company. See definition of “large accelerated filer,” “accelerated filer,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| S | Accelerated filer | £ | Non-accelerated filer | £ | Emerging growth company | £ | |||||||||||||||||
If an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards † provided pursuant to Section 13(a) of the Exchange Act. £
† The term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification after April 5, 2012.
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. S
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. S
For the description of the correction of the presentation error, refer to section 3.1 Revenues and Revenues Recognition in the Notes to the Consolidated Financial Statements as included in PART III of the Form 20-F.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b).£
Indicate by check mark which basis of accounting the registrant has used to prepare the financial statements included in this filing:
U.S. GAAP £ | by the International Accounting Standards Board S | Other £ | ||||||||||||
If “Other” has been checked in response to the previous question, indicate by check mark which financial statement item the registrant has elected to follow:
Item 17 £ Item 18 £
If this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes £ No S
TABLE OF CONTENTS
| Page | ||||||||
2
3
Introduction
Unless otherwise indicated or unless the context requires otherwise, “MOR,” “the company,” “our company,” “we,” “us,” and “our” refer to MorphoSys AG and its consolidated subsidiaries.
We own various trademark registrations and applications, and unregistered trademarks, including MorphoSys and our corporate logo. All other trade names, trademarks and service marks referred to in this annual report on Form 20-F, or this annual report, are the property of their respective owners. Trade names, trademarks and service marks of other companies appearing in this annual report are the property of their respective holders. Solely for convenience, the trademarks and trade names in this annual report may be referred to without the ® and ™ symbols, but such references should not be construed as an indicator that their respective owners will not assert, their rights thereto to the fullest extent under applicable law. We do not intend to use or display other companies’ trademarks or trade names to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
Our audited consolidated financial statements were prepared in accordance with International Financial Reporting Standards, or IFRS, as issued by the International Accounting Standards Board, or IASB. Our consolidated financial statements are presented in euros. All references in this annual report to “$,” “US$,” “U.S.$,” “U.S. dollars,” “dollars,” and “USD” mean U.S. dollars and all references to “€”, "EUR" and “euros” mean euros, unless otherwise noted. Throughout this annual report, references to “ADSs” mean American Depositary Shares or ordinary shares represented by American Depositary Shares, as the case may be.
Business Combination with Novartis
On February 5, 2024 MorphoSys announced that it entered into a business combination agreement (the "Business Combination Agreement") with Novartis BidCo AG (formerly known as Novartis data42 AG) and Novartis AG (hereinafter collectively referred to as “Novartis”) based on Novartis’ intention to submit a voluntary public takeover offer (the “Novartis Takeover Offer”) for all of MorphoSys’ outstanding common shares in exchange for payment of € 68.00 per share in cash. Separately, MorphoSys entered into a purchase agreement (the "Purchase Agreement") with Incyte Corporation ("Incyte") to sell and transfer to Incyte all rights worldwide related to tafasitamab for a purchase price of $ 25.0 million. MorphoSys and Incyte have been collaborating on the development and commercialization of tafasitamab since 2020. Prior to this agreement, tafasitamab was co-marketed in the U.S. by MorphoSys and Incyte as Monjuvi® (tafasitamab-cxix) and outside the U.S. by Incyte as Minjuvi®. MorphoSys’ Management Board and Supervisory Board unanimously approved both agreements.
In the Business Combination Agreement, MorphoSys and Novartis agreed on the terms of the Novartis Takeover Offer.
Novartis intends to offer MorphoSys’ shareholders € 68.00 per share in cash, for a total equity value of € 2.7 billion. The offer price corresponds to a premium of 94% and 142% on the volume-weighted average price during the last month and three months as of the unaffected January 25, 2024 close, respectively – the day before rumors about a transaction first surfaced. It also represents a premium of 89% percent to the closing share price of January 25, 2024.
Subject to a careful review of the offer document to be published by Novartis BidCo AG, MorphoSys’ Management Board and Supervisory Board intend to recommend the acceptance of the Novartis Takeover Offer. The Novartis Takeover Offer will contain customary closing conditions, in particular a minimum acceptance threshold of 65% of MorphoSys’ share capital and regulatory clearances. The closing is currently expected to take place in the first half of 2024. MorphoSys and Novartis agreed to take MorphoSys private promptly after the Novartis Takeover Offer has been settled. There is no assurance that the business combination will be consummated on the proposed terms, timing or at all.
The offer document of the Novartis Takeover Offer will be published by Novartis BidCo AG at a later date in accordance with the provisions of the German Securities Acquisition and Takeover Act, after the German Federal Financial Supervisory Authority (“BaFin”) has approved the publication. Promptly after the offer document is published, MorphoSys’ Management Board and Supervisory Board will issue a joint reasoned statement in accordance with sec. 27 of the German Securities Acquisition and Takeover Act. In accordance with U.S. securities laws, Novartis BidCo AG and Novartis AG will file a Tender Offer Statement which will include the offer document on Schedule TO and MorphoSys will file a Solicitation/Recommendation Statement on Schedule with the U.S. Securities and Exchange Commission.
Special Note Regarding Forward-Looking Statements
This report contains forward-looking statements concerning our business, operations and financial performance and condition as well as our plans, objectives and expectations for our business operations and financial performance and condition. Any
4
statements that are not of historical facts may be deemed to be forward-looking statements. You can identify these forward-looking statements by words such as “believes”, “estimates”, “anticipates”, “expects”, “plans”, “intends”, “may”, “could”, “might”, “will”, “should”, “aims” and other similar expressions that convey the uncertainty of future events or outcomes, although not all forward-looking statements contain these identifying words. Forward-looking statements appear in a number of places throughout this report and include statements regarding our intentions, beliefs, assumptions, projections, outlook, analyses and current expectations concerning, among other things, our intellectual property position, results of operations, cash needs, financial condition, liquidity, prospects, growth and strategies, the industry in which we operate and the trends that may affect the industry or us.
By their nature, forward-looking statements involve risks and uncertainties because they relate to events, competitive dynamics and industry change, and depend on economic circumstances that may or may not occur in the future or may occur on longer or shorter timelines than anticipated. Although we believe that we have a reasonable basis for each forward-looking statement contained in this report, we caution you that forward-looking statements are not guarantees of future performance and involve known and unknown risks, uncertainties and other factors that are in some cases beyond our control. All of our forward-looking statements are subject to risks and uncertainties that may cause our actual results or events to differ materially from our expectations. These forward-looking statements include, without limitation, statements about the following:
•the timing, progress and results of preclinical studies and clinical trials for our product candidates, including statements regarding the timing of initiation and completion of studies or trials and related preparatory work, the period during which the results of the trials and of our research and development programs will become available;
•the timing of and our ability to obtain and maintain regulatory approval for our product candidates;
•the proposed clinical development pathway for our product candidates, and the acceptability of the results of such trials for regulatory approval of such product candidates by the U.S. Food and Drug Administration, or U.S. FDA, the European Medicines Agency, or EMA1), or comparable foreign regulatory authorities;
•our expectations regarding the size of the patient populations for our product candidates, if approved for commercial use;
•our expectations regarding the timing for meetings with regulatory agencies;
•our intent regarding the commercialization of our product candidates;
•our estimates of our expenses, ongoing losses, future revenue, cash burn, cash runway, capital requirements and our needs for or ability to obtain additional financing;
•our ability to identify and develop new product candidates;
•our ability to identify new collaboration partners and successfully enter into new collaboration arrangements;
•our ability to identify, recruit and retain key personnel;
•our ability to protect and enforce our intellectual property protection for our proprietary and partnered product candidates, as well as the scope of such protection;
•our expectations with regard to our future revenues and our future financial condition;
•our expectations regarding pelabresib's ability to enhance the standard care for myelofibrosis, the further clinical development of pelabresib, including ongoing interactions with regulatory authorities and expectations regarding future regulatory filings and possible additional approvals for pelabresib;
•our expectations regarding Monjuvi® (tafasitamab-cxix)’s ability to treat, in combination with lenalidomide, adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) not otherwise specified, including DLBCL arising from low grade lymphoma, and who are not eligible for autologous stem cell treatment (ASCT);
1) For clarification purposes applying to whole document: the European Commission approves a product based on the recommendation of the European Medicines Agency (EMA) / Committee for Medicinal Products for Human Use (CHMP).
•our expectations regarding the future development of tulmimetostat;
•our ability to monetize and receive economic benefit from our partnered assets; and
•the development of and projections relating to our competitors or our industry.
5
In addition, even if our results, performance, financial condition and liquidity, and the development of the industry in which we operate are consistent with such forward-looking statements, they may not be predictive of results or developments in future periods. Among the factors that may result in differences are our expectations regarding risks and uncertainties related to our business, operations, strategy, goals and anticipated milestones, including our ongoing and planned research activities, ability to conduct ongoing and planned clinical trials, clinical supply of current or future drug candidates, commercial supply of current or future approved products, and launching, marketing and selling current or future approved products, our ability to obtain and maintain requisite regulatory approvals and to enroll patients in our planned clinical trials, additional interactions with regulatory authorities and expectations regarding future regulatory filings, our reliance on collaborations with third parties, estimating the commercial potential of our development programs and other risks indicated in the risk factors included in this report and other filings with the U.S. Securities and Exchange Commission.
Actual results could differ materially from our forward-looking statements due to a number of factors, including, the risks set forth under the section “Risk Factors” of this report and elsewhere in this report.
Any forward-looking statements that we make in this report are valid only as of the date of such statements, and we undertake no obligation to update such statements to reflect events or circumstances after the date of this report or to reflect the occurrence of unanticipated events.
6
Summary of the Material and Other Risks Associated with our Business
Below is a summary of the material risks to our business, operations and the investment in our ADSs. This summary does not address all of the risks that we face. Risks and uncertainties not presently known to us or that we presently deem less significant may also impair our business operations. Additional discussion of the risks summarized in this risk factor summary, and other risks that we face, can be found below under the heading “Risk Factors” and should be carefully considered, together with other information in this annual report on Form 20-F in its entirety before making investment decisions regarding our ADSs.
•The consummation of the Novartis Takeover Offer is subject to certain conditions.
•We and Novartis must obtain antitrust and merger control clearances to consummate the Novartis Takeover Offer, which, if delayed or not granted, may delay or jeopardize the Novartis Takeover Offer. In addition, conditions imposed by the competent merger control agencies in connection with their approvals may adversely impact our business, financial condition, or results of operation, including the loss of value of assets or businesses that may be required to be divested in connection with obtaining approvals under merger control or competition laws.
•If, following completion of the Novartis Takeover Offer, MorphoSys ordinary shares remain outstanding, the liquidity and market value of those ordinary shares could decline significantly, and our ordinary shares could be removed from certain stock indices. In addition, the liquidity of our ADSs could be negatively affected.
•Following closing of the Novartis Takeover Offer, we and Novartis intend to pursue a delisting of our ordinary shares and a termination of our ADS program. As a result, the liquidity and market value of our ordinary shares could decline significantly.
•Following completion of the Novartis Takeover Offer, we could enter into a domination and/or a profit and loss transfer agreement with Novartis, which could be disadvantageous to our shareholders who have not tendered their MorphoSys ordinary shares into the Novartis Takeover Offer.
•If our shareholders do not tender their ordinary shares in the Novartis Takeover Offer, they may receive consideration in a post-completion reorganization that is substantially lower in value than the offer price they would have received in the Novartis Takeover Offer.
•The announcement and pendency of the Novartis Takeover Offer, during which we are subject to certain interim operating restrictions, could have an adverse effect on our business and cash flows, financial condition and results of operations.
•We will incur significant transaction fees and costs in connection with the Novartis Takeover Offer.
•We may not be able to close-out the transition activities related to the sale of tafasitamab to Incyte in the planned timeframe resulting in opportunity costs and additional liabilities.
•We may fail to realize the anticipated strategic and financial benefits sought from the Novartis Takeover Offer.
•We cannot assure the adequacy of our capital resources to successfully complete the development and commercialization of our product candidates, and a failure to obtain additional capital, if needed, could force us to delay, limit, reduce or terminate one or more of our product development programs or commercialization efforts.
•We cannot assure to secure additional proceeds needed for business purposes from external refinancing.
•We cannot assure that we will obtain approval for pelabresib or that the approval (if obtained) will be as broad as intended with regards to indications or patient populations.
•We have incurred significant losses since inception and anticipate that we will continue to incur losses in the future.
•Our operating results may fluctuate significantly in the future.
•From time to time, we will need to raise additional funding, which may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us to delay, limit or terminate our product development efforts or other operations.
•We may lose liquidity reserves if banking institutions in whose funds or products we invest become insolvent.
•We may incur substantial expenses as a result of the limited nature of our disaster recovery and business continuity plan.
•The commercial success of our products, will depend upon the degree of market acceptance by physicians, patients, third-party payers and others in the medical community.
•If we are unable to maintain sales and marketing capabilities or enter into agreements with third parties to sell and market our product candidates, we may not be successful in commercializing our product candidates if and when they are approved.
7
•If we are unable to advance our proprietary product candidates in clinical development, obtain regulatory approval for our product candidates, including for pelabresib and tulmimetostat, and ultimately successfully commercialize them or experience significant delays in doing so, our business will be materially harmed.
•We currently rely on third-party suppliers and CMOs for the manufacturing and distribution of our product candidates, and our dependence on these third-parties may impair the development of our product candidates. Moreover, we rely on third-parties to produce commercial supplies of approved product candidate and our commercialization of any of our product candidates could be stopped, delayed or made less profitable if those third-parties fail to provide us with sufficient quantities of product or fail to do so at acceptable quality levels or prices or fail to otherwise complete their duties in compliance with their obligations to us or in compliance with applicable laws. Service or supply failures, or other failures, business interruptions, or other disasters affecting the manufacturing facilities of any party participating in the supply chain, would adversely affect our ability to supply our product candidates and products.
•We are exposed to potential product liability and professional indemnity risks that are inherent in the research, development, manufacturing, marketing and use of pharmaceutical products.
•We may be unable to acquire and retain talent which might adversely affect our ability to generate value.
•We may be subject to tax audits or disputes or changes in tax laws.
•Our internal computer systems and physical premises, or those of our strategic collaborators or other contractors or consultants, may fail or suffer security breaches, which could result in a material disruption of our product development programs and our manufacturing operations.
•Price controls may be imposed in certain markets, which may adversely affect our future profitability.
•We do not currently intend to pay dividends on our securities, and, consequently, your ability to achieve a return on your investment will depend on appreciation in the price of our shares.
•Holders of our ADSs may not be able to participate in any future preemptive subscription rights issues or to elect to receive dividends in shares, which may cause dilution to their holdings.
•As a foreign private issuer, we are exempt from a number of rules under the U.S. securities laws and are permitted to file less information with the SEC than a U.S. company. This may limit the information available to holders of ADSs or our ordinary shares.
•As a foreign private issuer, we are permitted to adopt certain home country practices in relation to corporate governance matters that differ significantly from Nasdaq corporate governance listing standards. These practices may afford less protection to shareholders than they would enjoy if we complied fully with corporate governance listing standards.
•U.S. holders of ADSs may suffer adverse tax consequences if we are characterized as a passive foreign investment company.
•We may lose our foreign private issuer status in the future, which could result in significant additional cost and expense.
•U.S. investors may have difficulty enforcing civil liabilities against our company and members of our Supervisory Board and Management Board and the experts named in this report.
•The rights of shareholders in a stock corporation subject to German law differ in material respects from the rights of shareholders of corporations incorporated in the United States.
•If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results or prevent fraud. As a result, shareholders could lose confidence in our financial and other public reporting, which would harm our business and the trading price of our shares.
Material non-financial aspects are taken into account in a separate non-financial group report, which is available on our website.
8
PART I
Item 1. Identity of Directors, Senior Management and Advisors
Not applicable.
Item 2.Offer Statistics and Expected Timetable.
Not applicable.
Item 3.Key Information
A.[Reserved]
B.Capitalization and Indebtedness
Not applicable
C.Reasons for the Offer and Use of Proceeds.
Not applicable.
D.Risk Factors
Our business faces significant risks. You should carefully consider all of the information set forth in this annual report and in our other filings with the U.S. Securities and Exchange Commission, or the SEC, including the following risk factors which we face and which are faced by our industry, any of which could materially adversely affect our business, financial condition, results of operations, or market price of our securities. The risks and uncertainties summarized and described below are not intended to be exhaustive and are not the only ones we face. Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also impair our business, prospects, financial condition and results of operations.
Risks Relating to the Novartis Takeover Offer
The consummation of the Novartis Takeover Offer is subject to certain conditions.
On February 5, 2024, we entered into a business combination agreement (the “Business Combination Agreement”) with Novartis BidCo AG (formerly known as Novartis data42 AG) and Novartis AG (hereinafter collectively referred to as “Novartis”) based on Novartis’ intention to submit a voluntary public takeover offer for all our ordinary shares at an offer price of € 68.00 per share in cash (the “Novartis Takeover Offer”). The Novartis Takeover Offer will contain customary closing conditions, in particular a minimum acceptance threshold of 65% of our share capital and regulatory clearances. The closing is currently expected to take place in the first half of 2024. The timing for the closing of the Novartis Takeover Offer will depend on the satisfaction of such conditions. Under the terms of the Business Combination Agreement, all conditions to the Novartis Takeover Offer must be satisfied by the end of the acceptance period, except for the regulatory clearances, i.e. the expiration of certain merger control waiting periods applicable to the Novartis Takeover Offer and the clearance by the competent merger control authorities. Under the terms of the Business Combination Agreement, the regulatory condition must be satisfied within twelve months following the date of the Business Combination Agreement, i.e., by February 5, 2025, 11:59 p.m. German time. If the regulatory condition is not satisfied by that date, the Novartis Takeover Offer will terminate and closing of the Novartis Takeover Offer will not occur. Furthermore, pursuant to the Business Combination Agreement, Novartis or we may terminate the Business Combination Agreement or the covenants therein under certain circumstances. No assurance can be given that all of the conditions to the Novartis Takeover Offer will be satisfied or, if they are, as to the timing of the closing of the Novartis Takeover Offer. If the conditions to the Novartis Takeover Offer are not satisfied or waived, the Novartis Takeover Offer will terminate and closing of the Novartis Takeover Offer will not occur. For more information on the Business Combination Agreement please refer to Item 10.C—Material Contracts—Business Combination Agreement with Novartis.
We and Novartis must obtain antitrust and merger control clearances to consummate the Novartis Takeover Offer, which, if delayed or not granted, may delay or jeopardize the Novartis Takeover Offer. In addition, conditions imposed by the competent merger control agencies in connection with their approvals may adversely impact our business, financial
9
condition, or results of operation, including the loss of value of assets or businesses that may be required to be divested in connection with obtaining approvals under merger control or competition laws.
Completion of the Novartis Takeover Offer is conditioned upon, among other things, regulatory clearances, i.e. the expiration of certain merger control waiting periods applicable to the Novartis Takeover Offer and the clearance by the competent merger control authorities. The governmental and regulatory agencies from which we and Novartis will seek these approvals have broad discretion in administering the applicable governing regulations. As a condition to their approval of the transactions contemplated by the Business Combination Agreement, those agencies may impose requirements, limitations or costs or require divestitures or place restrictions on the conduct of our and Novartis' respective businesses. Pursuant to the Business Combination Agreement, Novartis will use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to resolve such objections, subject to the limitations set forth in the Business Combination Agreement. No assurance can be given that the required approvals will be obtained or that the required conditions to the Novartis Takeover Offer will be satisfied. If the regulatory conditions are not satisfied within twelve months following the date of the Business Combination Agreement, i.e., by February 5, 2025, 11:59 p.m. German time, the Novartis Takeover Offer will terminate and closing of the Novartis Takeover Offer will not occur. Any delay in the completion of the Novartis Takeover Offer for regulatory reasons could diminish the anticipated benefits of the business combination or result in additional transaction costs.
Conditions imposed by regulatory agencies in connection with their approval of the Novartis Takeover Offer may require changes to our operations, restrict our ability to operate in certain jurisdictions following the Novartis Takeover Offer, restrict the combination of our and Novartis' operations in certain jurisdictions or require other commitments regarding ongoing operations.
If, following completion of the Novartis Takeover Offer, MorphoSys ordinary shares remain outstanding, the liquidity and market value of those ordinary shares could decline significantly, and our ordinary shares could be removed from certain stock indices. In addition, the liquidity of our ADSs could be negatively affected.
If the Novartis Takeover Offer is completed but not all outstanding MorphoSys ordinary shares have been tendered in the Novartis Takeover Offer, the free float in our ordinary shares will be significantly lower than the current free float, which may adversely affect the liquidity of the remaining MorphoSys ordinary shares. Reduced liquidity could make it more difficult for the remaining minority shareholders to sell their MorphoSys ordinary shares and could adversely affect the price of those remaining ordinary shares. In addition, reduced liquidity could result in increased volatility and the price for our ordinary shares may vary significantly in the future.
Our ordinary shares are included in the German SDAX & TecDAX stock indices. A significant reduction in free float as a result of the Novartis Takeover Offer may result in our ordinary shares being removed from those stock indices. Consequently, index funds and other investors who seek to mirror stock indices such as SDAX & TecDAX stock indices may sell or reduce their holdings of MorphoSys ordinary shares. This could result in reduced liquidity and an oversupply of our ordinary shares, which may adversely affect and cause significant variations in the price of those remaining ordinary shares.
During the acceptance period and thereafter until satisfaction of or failure to satisfy the regulatory condition, the MorphoSys ordinary shares tendered into the Novartis Takeover Offer shall be included in the stock market trading on the regulated market of the Frankfurt Stock Exchange and its sub-segment with additional post-admission obligations (Prime Standard) under a new and separate ISIN. During the acceptance period or thereafter any relevant body competent for the composition of a stock index, such as Deutsche Boerse AG, may decide to replace the MorphoSys ordinary shares not tendered in the SDAX & TecDAX or any other stock indices with the tendered MorphoSys ordinary shares; it may also reverse any such decision at any time. Consequently, index funds and other investors who seek to mirror indices such as the SDAX & TecDAX stock indices may sell or reduce their holdings of MorphoSys (tendered or non-tendered) ordinary shares.
In addition, the free float in our ADSs, which are listed on Nasdaq, will be significantly lower than the current free float, which may adversely affect the liquidity of our ADSs. Reduced liquidity could make it more difficult for the remaining ADS holders to sell their ADSs and could adversely affect the price of those remaining ADSs. In addition, reduced liquidity could result in increased volatility and the price for our ADSs may vary significantly in the future, which could require Nasdaq to temporarily suspend trading in our ADSs and ultimately result in our ADSs being delisted by Nasdaq.
The prices of our ordinary shares may be adversely affected if the Novartis Takeover Offer is not completed.
If the Novartis Takeover Offer is not completed, the price of our ordinary shares may decline for various reasons, including to the extent that the current market prices of our ordinary shares reflect a market premium based on the assumption that the Novartis Takeover Offer will be completed.
10
Following closing of the Novartis Takeover Offer, we and Novartis intend to pursue a delisting of our ordinary shares and a termination of our ADS program. As a result, the liquidity and market value of our ordinary shares could decline significantly.
Following closing of the Novartis Takeover Offer, we and Novartis intend to pursue a delisting of the MorphoSys ordinary shares from the regulated market of the Frankfurt Stock Exchange and its sub-segment with additional post-admission obligations (Prime Standard), a delisting of the ADSs from Nasdaq, a termination of the MorphoSys ADS program, and, when eligible, a deregistration of our ADSs and the MorphoSys ordinary shares under sections 12 and 15(d) of the Securities and Exchange Act of 1934, as amended. The measures by which the delisting and deregistration of our ordinary shares and ADSs will be accomplished will depend on the percentage of MorphoSys ordinary shares held by Novartis following closing of the Novartis Takeover Offer and could be either a squeeze-out of the remaining minority shareholders or the launch of a public delisting offer by Novartis. As a result, shareholders not tendering their MorphoSys ordinary shares into the delisting offer would end up holding non-listed ordinary shares with a highly reduced liquidity.
Following a termination of our ADS program, ADS holders that do not tender the underlying ordinary shares might end up holding the non-listed ordinary shares instead of ADSs. Some of the ADS holders might not be able to hold our (non-listed) ordinary shares due to their internal policies.
Following completion of the Novartis Takeover Offer, we could enter into a domination and/or a profit and loss transfer agreement with Novartis, which could be disadvantageous to our shareholders who have not tendered their MorphoSys ordinary shares into the Novartis Takeover Offer.
Following completion of the Novartis Takeover Offer, Novartis may control 75% or more of our ordinary shares represented at our general meeting, which would enable Novartis to initiate the conclusion of a domination and/or a profit and loss transfer agreement and profit and loss transfer agreement with MorphoSys (a "DPLTA"). Such agreement would allow Novartis to issue binding instructions to our Management Board, and, thus, to exercise control over MorphoSys' management. MorphoSys would be required to transfer to Novartis all annual net profits that would accrue without such transfer of profits, less any losses carried forward and any amounts appropriated to the legal reserves and Novartis would be required to compensate MorphoSys for any annual net losses that would be incurred by MorphoSys in the absence of a DPLTA and that would not be offset by any withdrawals from other revenue reserves created during the term of the DPLTA. The DPLTA could be disadvantageous to MorphoSys shareholders who have not tendered their ordinary shares into the Novartis Takeover Offer.
If our shareholders do not tender their ordinary shares in the Novartis Takeover Offer, they may receive consideration in a post-completion reorganization that is substantially lower in value than the offer price they would have received in the Novartis Takeover Offer.
Novartis might pursue a post-completion reorganization with respect to MorphoSys after completion of the Novartis Takeover Offer if the relevant ownership threshold for such a post-completion reorganization has been reached as a result of or following the Novartis Takeover Offer. A post-completion reorganization could eliminate any minority shareholder interests in MorphoSys remaining after the closing of the Novartis Takeover Offer or allow Novartis to control MorphoSys to the greatest extent permissible despite any remaining minority shareholder interests. If the Novartis Takeover Offer is consummated and Novartis (directly or indirectly) holds at least 75% of the ordinary shares represented in the shareholder meeting of MorphoSys, Novartis could decide to enter (directly or indirectly) into a domination and/or a profit and loss transfer agreement and/or a profit and loss transfer agreement with MorphoSys. If Novartis (directly or indirectly) holds at least 95% of the ordinary shares in MorphoSys, Novartis could initiate a squeeze-out of the minority shareholders of MorphoSys. If Novartis (directly or indirectly) holds more than 90% of the ordinary shares in MorphoSys, a squeeze-out would be possible in connection with a merger of MorphoSys into an entity of the Novartis group. The type of such post-completion reorganization transaction will depend on the percentage of MorphoSys ordinary shares acquired in the Novartis Takeover Offer and, to the extent legally permissible, in the open market or otherwise.
Due to the statutory legal framework applicable to such post-completion reorganization transactions, holders of our ordinary shares who do not tender their MorphoSys ordinary shares in the Novartis Takeover Offer may receive a different (including a lower) amount or a different form of consideration than they would have received had they tendered their MorphoSys ordinary shares in the Novartis Takeover Offer.
The announcement and pendency of the Novartis Takeover Offer, during which we are subject to certain interim operating restrictions, could have an adverse effect on our business and cash flows, financial condition and results of operations.
The announcement and pendency of the Novartis Takeover Offer could disrupt our business, and uncertainty about the effect of the Novartis Takeover Offer may have an adverse effect on us. These uncertainties could cause suppliers, vendors, partners and others that deal with us to defer entering into contracts with, or making other decisions concerning us or to seek to change or cancel existing business relationships with us. In addition, our employees may experience uncertainty regarding their roles after
11
the Novartis Takeover Offer. Employees may depart either before or after the completion of the Novartis Takeover Offer because of uncertainty and issues relating to the difficulty of coordination or because of a desire not to remain following the Novartis Takeover Offer. Therefore, the pendency of the Novartis Takeover Offer may adversely affect our ability to retain, recruit and motivate key personnel. Additionally, the attention of our management may be directed towards the completion of the Novartis Takeover Offer, including obtaining regulatory approvals, and may be diverted from the day-to-day business operations of MorphoSys. Matters related to the Novartis Takeover Offer may require commitments of time and resources that could otherwise have been devoted to other opportunities that might have been beneficial to us. Further, the business combination may give rise to potential liabilities, including those that may result from pending and future shareholder lawsuits relating to the Novartis Takeover Offer or a potential post-completion reorganization. Any of these matters could adversely affect our businesses or harm our results of operations, financial condition or cash flows.
We may be materially adversely affected by negative publicity related to the Novartis Takeover Offer.
From time to time, political and public sentiment in connection with a proposed public takeover may result in a significant amount of adverse press coverage and other adverse public statements affecting the parties to the public takeover. Adverse press coverage and public statements, whether or not driven by political or popular sentiment, may also result in legal claims or in investigations by regulators, legislators and law enforcement officials. Responding to these investigations and lawsuits, regardless of the ultimate outcome of the proceedings, can divert the time and effort of senior management from operating their businesses. Addressing any adverse publicity, governmental scrutiny or enforcement or other legal proceedings could be time-consuming and expensive and, regardless of the factual basis for the assertions being made, could have a negative impact on our reputation, on the morale and performance of our employees and on our relationships with regulators, suppliers and business partners. It may also have a negative impact on our ability to take timely advantage of various business and market opportunities. The direct and indirect effects of negative publicity, and the demands of responding to and addressing it, may have a material adverse effect on our business and cash flows, financial condition and results of operations.
Following the completion of the Novartis Takeover Offer, we will be majority owned by Novartis and our shareholders who do not tender their MorphoSys ordinary shares into the Novartis Takeover Offer will have less influence over MorphoSys.
After the completion of the Novartis Takeover Offer, Novartis will be our majority shareholder. Our shareholders who do not tender their ordinary shares into the Novartis Takeover Offer will have less influence over MorphoSys than prior to the Novartis Takeover Offer. As a majority shareholder, Novartis will be in a position to pass shareholder resolutions on their own, depending on their actual shareholding in MorphoSys, this could also be the case for resolutions requiring any kind of qualified majority. In addition, the Business Combination Agreement provides that MorphoSys will use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to ensure that Novartis will be adequately represented in our Supervisory Board following the completion of the Novartis Takeover Offer.
Upon completion of the Novartis Takeover Offer, certain change-of-control rights under material agreements may be triggered.
We are party to agreements that contain change-of-control provisions that may be triggered upon completion of the Novartis Takeover Offer. Upon the triggering of these change-of-control provisions, the counterparties to the agreement may be able to exercise certain rights that have a negative effect on us. For example, the terms and conditions of our convertible bonds issued in October 2020 provide the respective bondholders with the right to conditionally convert their bonds for MorphoSys ordinary shares upon publication of a respective offer document by Novartis and subsequently tender them into the Novartis Takeover Offer within any additional acceptance period. Following closing of the Novartis Takeover Offer, the holders of the convertible bonds may also put their bonds to us for payment of the principal amount plus accrued interest. Under the Business Combination Agreement, Novartis agreed to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to provide us following closing of the Novartis Takeover Offer with the financial resources required to pay any obligations arising from the implementation of the Novartis Takeover Offer. Failure to obtain such financial resources from Novartis could have a material adverse effect on our business and cash flows, financial condition and results of operations.
We will incur significant transaction fees and costs in connection with the Novartis Takeover Offer.
We have and will incur significant advisory, legal, accounting and other transaction fees and costs related to the Novartis Takeover Offer. Under the Business Combination Agreement, Novartis agreed to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to provide us following closing of the Novartis Takeover Offer with the financial resources required to pay any obligations arising from the implementation of the Novartis Takeover Offer. Failure to obtain such financial resources from Novartis could have a material adverse effect on our business and cash flows, financial condition and results of operations.
12
We may not be able to close-out the transition activities related to the sale of tafasitamab to Incyte in the planned timeframe resulting in opportunity costs and additional liabilities.
Separately to the Novartis Takeover Offer, MorphoSys entered into a Purchase Agreement with Incyte to sell and transfer all rights worldwide related to tafasitamab for $25.0 million. MorphoSys and Incyte have been collaborating on the development and commercialization of tafasitamab since 2020. Prior to this agreement, tafasitamab was co-marketed in the U.S. by MorphoSys and Incyte as Monjuvi® (tafasitamab-cxix) and outside the U.S. by Incyte as Minjuvi®. MorphoSys’ Management Board and Supervisory Board unanimously approved the Purchase Agreement. Per the Purchase Agreement, MorphoSys is obliged to support the transition of tafasitamab to Incyte which comprises - beside other items - the transfer of IP, transfer of contracts and the transfer of other documents. A transition plan as well as transition project team is in place, designed to ensure that the overall transition timeline is met. However, due to events and circumstances outside of MorphoSys' control there is a risk that the transition takes longer than expected, or that more than anticipated resources are required to fulfill the obligations as set forth by the Purchase Agreement which could result in opportunity costs and create additional liabilities for MorphoSys.
Risks Relating to our Business After Completion of the Novartis Takeover Offer
We may fail to realize the anticipated strategic and financial benefits sought from the Novartis Takeover Offer.
We may not realize all of the anticipated benefits of the Novartis Takeover Offer. In particular, we believe that Novartis has the necessary financial resources, additional scientific expertise and global footprint to maximize the full scope of pelabresib’s potential. However, Novartis may not provide the resources that are currently unavailable to us as a standalone biotech company and that are required to help accelerate the development opportunities and maximize the commercialization potential of pelabresib at a greater speed and scale.
We may be unable to retain and motivate our personnel successfully during or following the Novartis Takeover Offer.
As an innovation-driven and patient-centric company, we are highly dependent on the expertise of the members of our research and development team, as well as the other key functions such as commercial and supply to ensure that we can bring our medicines to our patients with the highest quality and compliance with required standards. Such employees may decide not to remain with us while the Novartis Takeover Offer is pending or after the Novartis Takeover Offer is completed. If key employees terminate their employment, or if an insufficient number of employees are retained to maintain effective operations, our business activities may be adversely affected. Recruiting and retaining qualified management, scientific, clinical, manufacturing, sales and marketing personnel is also critical to our success. We may not be able to locate suitable replacements for any key employees who leave us or offer employment to potential replacements on reasonable terms. In addition, we may not be able to motivate certain key employees following the completion of the Novartis Takeover Offer due to organizational changes, reassignments of responsibilities, the perceived lack of appropriate opportunities for advancement or other reasons.
Risks Related to Our Financial Condition
We cannot assure the adequacy of our capital resources to successfully complete the development and commercialization of our product candidates, and a failure to obtain additional capital, if needed, could force us to delay, limit, reduce or terminate one or more of our product development programs or commercialization efforts.
As of December 31, 2023, we had cash and cash equivalents and current and non-current other financial assets of € 680.5 million. We believe that we will continue to spend substantial resources for the foreseeable future developing our proprietary product candidates, including pelabresib. These expenditures will include costs associated with development, conducting clinical trials, seeking regulatory approvals, and launching and commercializing any products approved for sale and potentially acquiring new products. In addition, other unanticipated costs may arise. Because the outcome of our anticipated clinical trials is highly uncertain, we cannot reasonably estimate the actual amounts necessary to successfully complete the development and commercialization of our proprietary product candidates.
Our future funding requirements will depend on many factors, including but not limited to:
•the timing of, and the costs involved in, obtaining regulatory approvals for our product candidates if clinical trials are successful, including for obtaining regulatory approvals for pelabresib in the desired indications
•the success of our commercialization efforts and market acceptance for any of our product candidates for which we receive marketing approval;
•the costs of maintaining, expanding or contracting for sales, marketing and distribution capabilities in connection with commercialization of any of our product candidates for which we receive marketing approval;
13
•the numerous risks and uncertainties associated with developing therapeutic product candidates;
•the number and characteristics of product candidates that we pursue;
•the rate of enrollment, the need to expand, the progress, the costs and the outcomes of our clinical trials, which may or may not meet their intended endpoints;
•the timing of, and cost involved in, conducting non-clinical studies that are regulatory prerequisites to conducting clinical trials of sufficient duration for successful product registration;
•the cost of manufacturing clinical supply and establishing a commercial supply of our product candidates;
•the timing of, and costs involved in, conducting post-approval studies that may be required by regulatory authorities;
•the terms and timing of any collaborative, licensing, or other arrangements that we may establish, including any required milestone and royalty payments thereunder and any non-dilutive funding that we may receive;
•the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent claims, including litigation costs, if any, and the outcome of any such litigation;
•the timing, receipt, and amount of sales of, or royalties or milestones on, our existing products and future products, if any; and
•the costs to maintain the commercial organization including key executives.
In addition, our operating plan may change as a result of many factors currently unknown to us. As a result of these factors, we may need additional funds sooner than planned. We expect to finance future cash needs primarily through a combination of public or private equity offerings, strategic collaborations, debt financings, and non-dilutive funding. If sufficient funds on acceptable terms are not available when needed, or at all, we could be forced to significantly reduce operating expenses and delay, limit, reduce or terminate one or more of our product development programs or commercialization efforts.
We have sufficient liquid funds to ensure business operations until early 2026, including the convertible debt repayment, without requiring additional proceeds from external refinancing. However, in the current capital market environment, opportunities for external financing are limited compared to the prior year. In order to determine the medium and long-term liquidity requirements, we maintain a comprehensive liquidity plan based on our corporate planning that includes the simulated effects of various scenarios. To further reduce our financial risk, we take the outcome of the liquidity plan into account when prioritizing research and development projects and determining the financing requirements. Whilst the opportunity for equity financing may be limited due to the capital markets environment and/or the level of the share price, we also have access to other non-dilutive financing options, such as opportunistic out-licensing of (pre)clinical assets or the sale of potential future royalties.
We have incurred significant losses since inception and anticipate that we will continue to incur losses in the future.
We are a global biopharmaceutical company. We have incurred significant losses since our inception and our consolidated net loss for the year ended December 31, 2023 was € 189.7 million. As of December 31, 2023, our accumulated deficit was approximately € 1,013.1 million. The probability of being profitable strongly depends on the successful development of our other product candidates (namely pelabresib), and we may continue to incur losses in the coming years as we continue our research and development of, and seek regulatory approvals for, our product candidates, prepare for and begin to commercialize any approved product candidates and add infrastructure and personnel to support our product development efforts and operations as a public company in the United States. These activities will continue and will therefore impact significantly our profit or loss and our working capital in the foreseeable future.
Because of the numerous risks and uncertainties associated with biopharmaceutical product development, we are unable to accurately predict the timing or amount of expenses or when, or if, we will be able to achieve profitability. For example, our expenses could increase if we are required by the U.S. FDA or the EMA to perform trials in addition to those that we currently expect to perform, such as post-approval trials or if there are any delays in completing our currently planned clinical trials, delays in the regulatory approval process, the partnering process for our proprietary product candidates or in the development of any of our proprietary product candidates.
Our ability to generate revenue and achieve profitability in the future depends in large part on our ability, alone or with our collaborators and partners, and our current and any future approved products, and successfully complete the development of and obtain the necessary regulatory approvals for our current and any future product candidates. We may never succeed in these activities and may never generate revenue from product sales that is significant enough to achieve profitability. In addition, our revenues depend in part on the activities of our partners, over which we have no control, in respect of pursuing translational research and clinical trial activities and, where marketing approval has been granted and we have not retained commercialization rights, commercialization of our product candidates. Even if we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods. Our failure to become or remain profitable would depress our market
14
value and could impair our ability to raise capital, expand our business, develop other product candidates, or continue our operations. A decline in the value of our company could also cause our investors to lose all or part of their investment.
Our operating results may fluctuate significantly in the future.
Our results of operations may fluctuate significantly in the future due to a variety of factors, many of which are outside of our control. The revenues we generate and our operating results will be affected by numerous factors, including, but not limited to:
•the development status of our product candidates and, particularly, the timing of any milestone payments to be paid or received by us under our collaboration agreements;
•the incurrence of clinical expenses that could fluctuate significantly from period to period;
•the commercial success of the products marketed by our partners and the amount of royalties to us associated therewith;
•our ability to successfully commercialize products marketed by ourselves;
•foreign exchange fluctuations;
•the unpredictable effects of collaborations during these periods;
•the timing of our satisfaction of applicable regulatory requirements;
•the rate of expansion of our clinical development and other development efforts;
•the effect of competing technologies and products and market developments; and
•general and industry-specific economic conditions.
If our operating results fall below the expectations of investors or securities analysts, the price of our ordinary shares could decline substantially and any fluctuations in our operating results and cash flows may, in turn, cause the price of our shares to fluctuate substantially.
From time to time, we will need to raise additional funding, which may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us to delay, limit or terminate our product development efforts or other operations.
Identifying and acquiring rights to develop potential product candidates and conducting preclinical testing and clinical trials is a time-consuming, expensive and uncertain process that may take years to complete. We may never generate the necessary data or results required to obtain regulatory approval and achieve product sales, and even if one or more of our product candidates is approved, they may not achieve commercial success. Accordingly, we will need to continue to rely on additional financing to achieve our business objectives. Adequate additional financing may not be available to us on acceptable terms, or at all.
We may seek additional funding through a combination of equity offerings, debt financings, including convertible bond offerings, collaborations, licensing arrangements, strategic alliances, monetization of royalty streams and marketing or distribution arrangements. As a German public company, certain capital structure decisions will require shareholder approval, which may limit our flexibility to manage our capital structure. To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms may include liquidation or other preferences that adversely affect your rights as a holder of our shares. The incurrence of indebtedness or the issuance of certain debt securities could result in increased fixed payment obligations and could also result in certain additional restrictive covenants, such as limitations on our ability to incur additional debt or issue additional equity, limitations on our ability to acquire or license intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. In addition, issuance of additional equity securities, or the possibility of such issuance, may cause the market price of our shares to decline. In the event that we enter into collaborations or licensing arrangements or sell rights to receive royalties to third-parties to raise capital, we may be required to accept unfavorable terms, including relinquishing or licensing to a third-party on unfavorable terms our rights to technologies or product candidates that we otherwise would seek to develop or commercialize ourselves or potentially reserve for future potential arrangements when we might be able to achieve more favorable terms.
If we are unable to obtain funding on a timely basis, or if revenues from collaboration arrangements or product sales are less than we have projected, we may be required to significantly curtail, delay or discontinue one or more of our development programs or the commercialization of any product candidates or be unable to expand our operations or otherwise capitalize on our business opportunities, as desired, which could materially affect our business, financial condition and results of operations.
15
We may lose liquidity reserves if banking institutions in whose funds or products we invest become insolvent.
As a result of the ongoing, tense economic situation, it is possible that banking institutions in whose funds or products we invest may become insolvent. We are therefore continuing to invest, when possible, only in funds and products of banks that are considered safe and have a high rating or are backed by a strong partner. However, there is a possibility that at times there are no funds and products from banks that are considered safe, and thus we may have to invest in other, riskier funds or products. We diversify and invest in lower-risk money market funds in order to limit our exposure to individual financial institutions. However, a strategy that excludes all risks of potential bank insolvencies would be too expensive and impractical. This means that the risk of bank insolvency cannot be completely ruled out. Additionally, safe investments, e.g., German government bonds, are associated with relatively low interest yield. If we were to lose liquidity reserves due to the insolvency of banking institutions in whose funds or products we invest, we may be required to curtail, delay or discontinue one or more of our development programs or the commercialization of any product candidates or be unable to expand our operations or otherwise capitalize on our business opportunities, as desired, which could materially affect our business, financial condition and results of operations.
We may incur substantial expenses as a result of the limited nature of our disaster recovery and business continuity plan.
MorphoSys AG has implemented a business continuity plan to prevent the collapse of critical business processes to a large extent or to enable the resumption of critical business processes in case a natural disaster, public health emergency or other serious event occurs. However, depending on the severity of the situation, it may be difficult or in certain cases impossible for us to continue our business for a significant period of time. Our contingency plans for disaster recovery and business continuity may prove inadequate in the event of a serious disaster or similar event and we may incur substantial costs that could have a material adverse effect on our business.
Risks Related to Commercialization
The commercial success of our products, will depend upon the degree of market acceptance by physicians, patients, third-party payers and others in the medical community.
The commercial success of our products will depend in part on the medical community, patients, and third-party or governmental payers accepting such product(s) as medically useful, cost-effective, and safe. Any other products that we may bring to the market may not gain market acceptance by physicians, patients, third-party payers and others in the medical community. If these products do not achieve an adequate level of acceptance, we may not generate significant product revenue. See the section of this annual report titled "Item 4.B. Business Overview Coverage, Reimbursement and Pricing" below. The degree of market acceptance of our products will depend on a number of factors, including:
•the breadth of the approved clinical indications for our product candidates;
•the potential efficacy and potential advantages over alternative treatments;
•the prevalence and severity of any side effects, including any limitations or warnings contained in a product’s approved labeling;
•relative convenience and ease of administration;
•the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies;
•the strength of marketing and distribution support and timing of market introduction of competitive products;
•the pricing of our product and of any future products;
•publicity concerning our product, any future products, or competing products and treatments;
•sufficient third-party insurance coverage or reimbursement; and
•potential product liability claims.
Even if a potential product displays a favorable efficacy and safety profile in preclinical and clinical studies, market acceptance of the product will not be known until after it is launched. Our efforts to educate the medical community and payers on the benefits of our products may require significant resources and may never be successful. Our efforts to educate the marketplace may require more resources than are required by the conventional technologies marketed by our competitors. Any of these factors may cause our products, to be unsuccessful or less successful than anticipated.
16
If we are unable to maintain sales and marketing capabilities or enter into agreements with third parties to sell and market our product candidates, we may not be successful in commercializing our product candidates if and when they are approved.
To achieve commercial success for any approved medicine for which we retain sales and marketing responsibilities, we must maintain a sales and marketing organization or outsource these functions to other third parties. We will need to further build our sales and marketing infrastructure to sell, or participate in sales activities with our collaborators for, our product candidates if and when they are approved, including, for example, to support the potential approval of pelabresib and/or one or more product candidates in key markets.
There are risks involved with both holding our own sales and marketing capabilities or entering into arrangements with third parties to perform these services. For example, recruiting and training a sales force is expensive and time consuming and could delay any product launch. If the commercial launch of a product candidate for which we recruit a sales force and establish marketing capabilities is delayed or does not occur for any reason, we would have prematurely or unnecessarily incurred these commercialization expenses. This may be costly, and our investment would be lost if we cannot retain or reposition our sales and marketing personnel.
Factors that may inhibit our efforts to commercialize our medicines on our own include:
•our inability to recruit and retain adequate numbers of effective sales and marketing personnel;
•the inability of sales personnel to obtain access to physicians or persuade adequate numbers of physicians to prescribe any future medicines;
•the lack of complementary medicines to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies with more extensive product lines; and
•unforeseen costs and expenses associated with creating an independent sales and marketing organization.
If we enter into arrangements with third parties to perform sales, marketing and distribution services, our product revenue or the profitability of product revenue to us are likely to be lower than if we were to market and sell any medicines that we develop ourselves. In addition, we may not be successful in entering into arrangements with third parties to sell and market our product candidates or may be unable to do so on terms that are favorable to us. We likely will have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market our medicines effectively. If we do not maintain sales and marketing capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our product candidates.
Risks Related to the Development, Clinical Testing and Regulatory Approval of Our Product Candidates
If we are unable to advance our proprietary product candidates in clinical development, obtain regulatory approval for our product candidates, including for pelabresib and tulmimetostat, and ultimately successfully commercialize them or experience significant delays in doing so, our business will be materially harmed.
We have several product candidates in clinical development, including pelabresib and tulmimetostat. Although we may receive certain payments from our collaboration partners, including upfront payments, payments for achieving certain development, regulatory or commercial milestones and royalties, our ability to generate revenue from our product candidates’ sales is dependent on receipt of regulatory approval for, and successful commercialization of, such product candidates, which may never occur, be delayed or not be as broad as intended with regards to indications or patient population. Our business and future success is particularly dependent on our ability to develop, either alone or in partnership, successfully, receive regulatory approval for, and then successfully commercialize our proprietary product candidates, including pelabresib and tulmimetostat. Each of our product candidates will require additional preclinical and/or clinical development, regulatory approval in multiple jurisdictions, manufacturing supply, substantial investment and significant marketing efforts before we generate any revenue from product sales or royalties. We are not permitted to market or promote any of our product candidates before we receive regulatory approval from applicable regulatory authorities. The success of our product candidates will depend on several factors, including the following:
•successful completion of preclinical studies (including safety studies) required to obtain regulatory approval in the United States, the European Union and other jurisdictions for our product candidates;
•successful enrollment of patients in, and completion of, clinical studies (including safety studies) required to obtain regulatory approval in the United States, the European Union and other jurisdictions for our product candidates;
•successful demonstration of reproducibility in the production process and ability for market supply;
•strategic commitment to particular product candidates and indications by us and our collaborators;
17
•receipt of product approvals, including marketing approvals, from applicable regulatory authorities;
•successful local and regional pricing and reimbursement negotiations with third-party payors to enable patients’ access to our products;
•successful validation of biomarkers and development of biomarker assays in those studies or programs where biomarkers are part of the development plan;
•obtaining and maintaining patent and trade secret protection or regulatory exclusivity for our product candidates and brands;
•securing market supply and distribution network;
•securing quality raw material supplies;
•the successful launch and marketing of approved products and/or brands, whether alone or in collaboration with others;
•acceptance of our approved products and/or brands by patients, the medical community and third-party payors;
•effectively competing with other therapies and ability to demonstrate clinically meaningful benefits;
•enforcing and defending intellectual property rights and claims;
•maintaining a continued acceptable safety and efficacy profile of the products following approval;
•fulfillment of post-marketing commitments and requirements from applicable regulatory authorities; and
•maintaining a continued, sufficient supply of drug product in acceptable quality.
If we do not achieve one or more of these factors in a complete and timely manner or at all, we could experience significant delays or an inability to successfully commercialize our product candidates, which would materially adversely affect our business, financial condition, results of operations and prospects and, in case of product candidates, technologies and licenses we have acquired, may result in a significant impairment of assets.
Further, our product candidates may not receive regulatory approval even if we are successful in conducting clinical trials, non-clinical studies and assembling required CMC (chemistry, manufacturing and controls) information. If we do not receive regulatory approvals for our product candidates, we may not be able to continue our operations and partnership. Even if we successfully obtain regulatory approvals to market one or more of our product candidates, our revenues will be dependent, in part, upon the commercialization potential, our ability to supply sufficient amounts of product candidates, the uptake of our product candidates and the size of the markets in the territories for which we gain regulatory approval and have commercial rights. If the market potential that we are targeting are not as significant as we estimate, we may not generate significant revenues from sales of such products, if approved.
We plan to seek regulatory approval to commercialize future product candidates both in the United States and potentially in the European Union, and additional foreign jurisdictions, such as, for example, pelabresib for which MorphoSys is currently preparing the regulatory filings for approval in the U.S. and the EU on the basis of the MANIFEST-2 study. While the scope of regulatory approval is similar in other countries, to obtain separate regulatory approval in many other countries, we must comply with numerous and varying regulatory requirements of such countries regarding safety and efficacy and governing, among other things, clinical trials and commercial sales, pricing and distribution of our product candidates, and we cannot predict success in these jurisdictions.
Clinical trials are very expensive, resource intensive and complex to design and implement and involve uncertain outcomes. If clinical trials or production of our product candidates are prolonged, delayed or terminated, we may be unable to obtain required regulatory approvals, and therefore be unable to commercialize our product candidates on a timely basis or at all, which may materially adversely affect our business, financial condition, results of operations and prospects.
We are currently conducting clinical trials for pelabresib and tulmimetostat in various indications. Each of our clinical trials requires the investment of substantial resources and time and the timing of the commencement, continuation and completion of these clinical trials may be subject to significant delays or termination relating to various causes, including, among other things:
•difficulties in identifying and enrolling patients who meet trial eligibility criteria;
•failure of patients to complete the clinical trials or return for post-treatment follow-up;
•delays in accumulating the required number of clinical events for data analyses;
•clinical investigators or sites deviating from trial protocol or failing to comply with regulatory requirements or meet their contractual obligations;
18
•delay or failure to obtain required regulatory approvals to start the clinical trial in the participating countries;
•delays in or failure to reach agreement on acceptable terms with prospective contract research organizations, or CROs, third party vendors, clinical trial sites and contract manufacturing organizations, or CMOs, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs, trial sites and CMOs;
•delays in or failure to obtain ethics committee, or EC, institutional review board, or IRB, approval at participating trial sites;
•failure of third-party contractors used in our clinical trials or contract manufacturing organizations, or CMOs, to comply with regulatory requirements or meet their contractual obligations in a timely manner, or not at all;
•changes in regulatory requirements;
•the development and approval of competitive products;
•results from clinical trials of competing compounds, which may give rise to concerns about the target, the envisioned mode of action, the compound class or the commercial potential of the product candidate we are evaluating;
•higher-than-expected costs of clinical trials of our product candidates;
•insufficient or inadequate supply or quality of our product candidates or other materials necessary to conduct clinical trials of our product candidate;
•scheduling conflicts with participating investigators/trial sites due to competing trials;
•outbreak of pandemic and natural disaster;
•urgent safety measures;
•pre-defined interim analysis of clinical trial data (futility analysis) or unfavorable and unforeseen non-clinical or clinical information that reveals that the product candidate has an unfavorable risk-benefit ratio;
•suspension/termination of approval of clinical trial conduct by ECs, IRBs, the U.S. FDA, or other competent authorities (CA);
•strategic decision to stop the clinical trial or the clinical development program; and
•recommendations of the data safety monitoring board/data monitoring committee, or DSMB/DMC, based on provided clinical safety data.
It is uncertain whether any of our clinical trials will begin as planned, will need to be redesigned or amended or will be completed on schedule. Clinical testing is expensive and can take many years to complete, and its outcome is inherently uncertain. Failure can occur at any time during the clinical trial conduct. We could encounter delays if a clinical trial is suspended or terminated by us, by the ECs/IRBs of the institutions in which such trials are being conducted, by a DSMB/DMC for such trial or by the U.S. FDA or other regulatory authorities. Such authorities may impose such a suspension or termination due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical trial or trial site by the U.S. FDA or other regulatory authorities resulting in the imposition of a clinical hold, safety issues or adverse side effects, failure to demonstrate a benefit from using a drug, changes in governmental regulations or administrative actions or lack of adequate funding to continue the clinical trial. If we experience delays in the completion of, or termination of, any clinical trial of our product candidates, the commercial prospects of our product candidates will be harmed, and our ability to generate product revenues from any of these product candidates will be delayed. In addition, any delays in completing our clinical trials will increase our costs, slow down our product candidate development and approval process and jeopardize our ability to commence product sales and generate revenues. Significant clinical trial delays could also allow our competitors to bring products to market before we do or shorten any periods during which we have the exclusive right to commercialize our product candidates and impair our ability to commercialize our product candidates, and may harm our business and results of operations. In addition, some of the factors that cause, or lead to, a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of regulatory approval of our product candidates.
Clinical trials must be conducted with supplies of our product candidates produced under current good manufacturing practice, or cGMP, and supplied accordingly under good distribution practice, or GDP, requirements and other regulations. We rely on CROs, third party vendors, CMOs, and clinical trial sites to ensure the proper and timely conduct of our clinical trials. Even though we have agreements governing their committed activities, we depend on our collaborators, on clinical trial sites, CROs, and third party vendors to conduct and monitor our clinical trials in compliance with good clinical practice, or GCP, requirements. To the extent our collaborators, third party vendors, or CROs fail to enroll participants for our clinical trials, fail to conduct and monitor the study to GCP standards or are delayed for a significant time or fail in the execution of trials,
19
including achieving full enrollment, we may be affected by increased costs, program delays or both, which may harm our business.
If we are unable to successfully complete clinical trials of our product candidates or other testing, if the results of these trials or tests are unfavorable or are only modestly favorable, if there are safety concerns associated with our product candidates, or if we are required to conduct additional clinical trials or other testing of our product candidates beyond the trials and testing that we contemplate, we may:
•be delayed in obtaining marketing approval for our product candidates;
•not obtain marketing approval at all;
•obtain approval for indications or patient populations that are not as broad as intended or desired;
•obtain approval with product labeling that includes significant use or distribution restrictions or significant safety warnings, including boxed warnings;
•face higher pricing and reimbursement hurdles;
•be subject to additional post-marketing testing or other requirements; or
•remove the product from the market after obtaining marketing approval.
The occurrence of any such events may materially adversely affect our business, financial condition, results of operations and prospects.
The incidence and prevalence for target patient populations of our product candidates are based on estimates and third-party sources. If the market opportunities for our products or any future products are smaller than we estimate or if any approval that we obtain for a product is for a smaller patient population than anticipated, our business, financial condition, results of operations and prospects may be materially adversely affected.
Periodically, we make estimates regarding the incidence and prevalence of target patient populations for particular diseases based on various third-party sources and internally generated analysis and use such estimates in making decisions regarding our product development strategy, including determining indications on which to focus in preclinical or clinical trials.
These estimates may be inaccurate or based on imprecise data. For example, the total addressable market opportunity will depend on, among other things, any limitations on populations and indications in approved product labeling, patient access, product pricing and reimbursement as well as the approval of new or competing medicines. The number of patients in the addressable markets may turn out to be lower than expected or new patients may become increasingly difficult to identify or gain access to. Additionally, even if we obtain significant market share for a product within an approved indication, if the potential target populations for the product is small, it may be difficult to achieve profitability without obtaining marketing approval for additional indications. Any of these factors could materially adversely affect our business, financial condition, results of operations and prospects.
The speed at which we complete our clinical trials depends on many factors, including, but not limited to, patient enrollment. If we are unable to enroll patients in our clinical trials, our development efforts and business, financial condition, results of operations and prospects could be materially adversely affected.
Patient enrollment and accumulation of clinical events are significant factors in the timing and successful completion of clinical trials. Patient enrollment is affected by many factors, including the size and nature of the patient population, the proximity of patients to clinical sites, the eligibility criteria for the trial, the design of the clinical trial, competing clinical trials and clinicians’ and patients’ perceptions as to the potential advantages of the product being studied in relation to other available therapies, including any new products that may be approved for the indications we are investigating. Because there is a relatively limited number of patients worldwide, patient enrollment may be challenging. Trials may be subject to delays as a result of patient enrollment taking longer than anticipated or patient withdrawal or delays in accumulating the required number of clinical events for data analyses (as applicable). Delays in the completion of any clinical trial of our product candidates will increase our costs, slow down our product candidate development and delay or potentially jeopardize our ability to receive regulatory approval, commence product sales and generate revenue. Any of these occurrences may harm our clinical trials, which could materially adversely affect our business, financial condition, results of operations and prospects.
20
Results of previous preclinical studies and clinical trials may not be predictive of future results, and the results of our current and planned clinical trials may not satisfy the requirements of the U.S. FDA, the EMA or comparable foreign regulatory authorities.
Positive or timely results from preclinical or early-stage trials do not ensure positive or timely results in late-stage clinical trials or product approval by the U.S. FDA, the EMA, or comparable foreign regulatory authorities. We will generally be required to demonstrate with substantial evidence through well-conducted, possibly controlled clinical trials that our product candidates are safe and effective for use in a well-defined patient population before we can seek regulatory approvals for their commercial sale. Our ongoing and planned clinical trials may produce negative or inconclusive results, and we or any of our current and future collaborators may decide, or regulators may require us, to conduct additional clinical or preclinical testing. Success in preclinical studies or early-stage clinical trials does not mean that future clinical trials or registration clinical trials will be successful because product candidates in later-stage clinical trials may fail to demonstrate sufficient safety or efficacy to the satisfaction of the U.S. FDA, the EMA and comparable foreign regulatory authorities, despite having progressed through preclinical studies and initial clinical trials. Product candidates that have shown promising results in early clinical trials may still suffer significant setbacks in subsequent clinical trials. For example, a number of companies in the pharmaceutical industry, including those with greater resources and experience than us, have suffered significant setbacks in advanced clinical trials, even after obtaining promising results in earlier clinical trials. Similarly, interim results of a clinical trial do not necessarily predict final results.
Open-label clinical trials are subject to various limitations that may exaggerate any therapeutic effect on patients in open-label clinical trials receiving treatment. Open-label clinical trials may be subject to a “patient bias” where patients perceive their symptoms to have improved merely due to their awareness of receiving an experimental treatment. In addition, open-label clinical trials may be subject to an “investigator bias” where those assessing and reviewing the physiological outcomes of the clinical trials are aware of which patients have received treatment and may interpret the information of the treated group more favorably given this knowledge. The results from an open-label trial may not be predictive of future clinical trial results with any of our product candidates for which we include an open-label clinical trial when studied in a controlled environment with a placebo or active control.
The regulatory approval processes of the U.S. FDA and comparable foreign regulatory authorities are lengthy, time-consuming and inherently unpredictable and if we fail to obtain regulatory approval in any jurisdiction, we will not be able to commercialize our products in that jurisdiction, and our business, results of operations, financial condition and prospects may be materially adversely affected.
The time required to obtain approval by the U.S. FDA and comparable foreign regulatory authorities is unpredictable but typically takes many years following the commencement of clinical trials and depends upon numerous factors, including clinical trial designs, their outcome and the substantial discretion of the regulatory authorities. In addition, approval laws, regulations, policies or the type and amount of clinical data or other information necessary to gain approval may change during the course of a product candidate’s clinical development and may vary among jurisdictions. Our existing product candidates or any product candidates we may seek to develop in the future may never obtain regulatory approval.
Our future product candidates could fail to receive regulatory approval for many reasons, including the following:
•the U.S. FDA or comparable foreign regulatory authorities may disagree with the design or implementation of our clinical trials or the results of our clinical trials may not meet the level of statistical significance required by the U.S. FDA or comparable foreign regulatory authorities for approval;
•we may be unable to demonstrate to the satisfaction of the U.S. FDA or comparable foreign regulatory authorities that a product candidate is safe and effective for its proposed indication(s) in the proposed population;
•we may be unable to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks;
•the U.S. FDA or comparable foreign regulatory authorities may disagree with our interpretation of data from preclinical studies or clinical trials;
•the data collected may not be sufficient to support the submission of a BLA, NDA or other submission, or to obtain regulatory approval in the United States, the European Union or elsewhere;
•the U.S. FDA or comparable foreign regulatory authorities may fail to approve the manufacturing processes or facilities of third-party manufacturers with which we contract for clinical and commercial supplies;
•the laws, regulations or policies of the U.S. FDA or comparable foreign regulatory authorities may significantly change in a manner rendering our clinical data or other regulatory submissions insufficient for approval; and
21
•MorphoSys’ critical business operations, including but not limited to the Company’s supply chain, clinical trial conduct, as well as timelines for regulatory and commercial execution may be influenced negatively in case the implemented disaster recovery and business continuity plan may prove inadequate.
The approval process may result in failing to obtain regulatory approval to market any of our future product candidates, such as, for example, pelabresib for which MorphoSys is currently preparing the regulatory filings for approval in the U.S. and the EU on the basis of the MANIFEST-2 study, which would significantly harm our business, results of operations and prospects. The U.S. FDA, the EMA and other regulatory authorities have substantial discretion in the approval process and determining when or whether regulatory approval will be obtained for any of our product candidates. Even if we believe the data collected from clinical trials of our product candidates are promising, such data may not be sufficient to support approval by the U.S. FDA, the EMA or any other regulatory authority. These authorities could require additional clinical data, including clinical trials designed with internal controls, in order to support regulatory approvals.
In addition, even if we were to obtain approval, regulatory authorities may approve any of our future product candidates for fewer or more limited indications than we request, may grant approval contingent on the performance of costly post-marketing clinical trials, or may approve a future product candidate with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that product candidate, including, for example, pelabresib for which MorphoSys is currently preparing the regulatory filings for approval in the U.S. and the EU on the basis of the MANIFEST-2 study. Any of the foregoing scenarios could materially harm the commercial prospects for our product candidates.
Commercialization of our products in more than one jurisdiction requires separate regulatory approval in each jurisdiction and compliance with the numerous and varying regulatory requirements of each jurisdiction. The approval procedures vary from country to country and may require additional testing or other steps. Satisfying these and other regulatory requirements is costly, time-consuming, uncertain, and subject to unanticipated delays. In addition, in many countries outside the United States and in particular in many of the Member States of the European Union, a product must undergo health economic assessments to agree on pricing and/or be approved for reimbursement before it can be approved for sale in that country, or before it becomes commercially viable. The U.S. FDA and the EMA may come to different conclusions regarding approval of a marketing application. Approval by the U.S. FDA or the EMA does not ensure approval by regulatory authorities in other countries or jurisdictions, and approval by one foreign regulatory authority does not ensure approval by regulatory authorities in other foreign countries or by the U.S. FDA or the EMA. In addition, failure to obtain regulatory approval in any country may delay or have negative effects on the process for regulatory approval in other countries. Clinical trials conducted in one country may not be accepted by regulatory authorities in other countries, including as a result of population and other demographic difference across countries. We may not obtain regulatory approvals on a timely basis, if at all. We may not be able to file for regulatory approvals and may not receive necessary approvals to commercialize a product in any market. We may be required to conduct additional preclinical studies or clinical trials, which would be costly and time-consuming. If we or any future partner are unable to obtain regulatory approval for our product candidates in one or more significant jurisdictions, then the commercial opportunity for our product candidates, and our business, results of operations, financial condition and prospects, may be materially adversely affected.
Disruptions at the U.S. FDA and other government agencies caused by funding shortages or global health concerns could hinder their ability to hire, retain or deploy key leadership and other personnel, or otherwise prevent new or modified products from being developed, approved, or commercialized in a timely manner or at all, which could negatively impact our business.
The ability of the U.S. FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, statutory, regulatory, and policy changes, the U.S. FDA’s ability to hire and retain key personnel and accept the payment of user fees, and other events that may otherwise affect the U.S. FDA’s ability to perform routine functions. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable. Disruptions at the U.S. FDA and other agencies may also slow the time necessary for drugs or biologics or modifications to approved drugs or biologics to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, over the last several years, including for 35 days beginning on December 22, 2018, the U.S. government has shut down several times and certain regulatory agencies, such as the U.S. FDA, have had to furlough critical U.S. FDA employees and stop critical activities.
22
Our product or product candidates may cause undesirable side effects that could delay or prevent their regulatory approval, limit the commercial profile of an approved label, or result in significant negative consequences following regulatory approval, if any.
Undesirable side effects that may be caused by our product or product candidates could cause us, our collaboration partners or the regulatory authorities to interrupt, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of regulatory approval by the U.S. FDA, the EMA or comparable foreign regulatory authorities. The results of our trials could reveal a high and unacceptable severity and prevalence of side effects. In such an event, our trials could be suspended or terminated and the U.S. FDA, the EMA or comparable foreign regulatory authorities could order us to cease further development of or deny approval of our product candidates for any or all targeted indications. The product-related side effects could affect patient recruitment or the ability of enrolled patients to complete the trial or result in potential product liability claims.
Clinical trials assess a sample of the potential patient population. With a limited number of patients and duration of exposure, rare and severe side effects of our product candidates may only be uncovered with a significantly larger number of patients exposed to the product candidate. If our product candidates receive regulatory approval and we or others identify undesirable side effects caused by such approved products (or any other similar products), a number of potentially significant negative consequences could result, including:
•regulatory authorities may withdraw or limit their approval of such products and require us or our collaborators to take such products off the market;
•regulatory authorities may require the addition of labeling statements, such as a “boxed” warning or a contra-indication, or correspondence to alert physicians and other healthcare providers about new or updated information regarding the approved product, such as Dear Health Care Provider letters;
•we or our collaborators may be required to create a medication guide outlining the risks of such side effects for distribution to patients;
•we or our collaborators may be required to change the way such product candidates are distributed or administered, conduct additional clinical trials or change the labeling of the product candidates;
•regulatory authorities may require a Risk Evaluation and Mitigation Strategy, or REMS, plan to mitigate risks, which could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools;
•we or our collaborators may be subject to regulatory investigations and government enforcement actions;
•we or our collaborators may decide or be required to remove such product candidates from the marketplace;
•we or our collaborators could be sued and potentially held liable for injury caused to individuals exposed to or taking our product candidates;
•sales of the product(s) may decrease substantially; and
•our reputation and the reputation of our collaborators may suffer.
Any of these events could prevent us or our collaborators from achieving or maintaining market acceptance of the affected product candidates and could substantially increase the costs of commercializing our product candidates, if approved, and therefore could have a material adverse effect on our business, financial condition, results of operations and prospects.
We and our collaboration partners have conducted and intend to conduct additional clinical trials for selected product candidates at sites outside the United States, and the U.S. FDA may not accept data from trials conducted in such locations or may require additional U.S.-based trials.
We and our collaboration partners have conducted, are currently conducting and intend in the future to conduct, clinical trials outside the United States, particularly in, but not limited to, the European Union, where we are headquartered.
Although the U.S. FDA may accept data from clinical trials conducted outside the United States, acceptance of this data is subject to certain conditions imposed by the U.S. FDA. For example, the clinical trial must be well-designed and conducted by qualified investigators in accordance with GCP, including review and approval by an independent ethics committee and receipt of informed consent from trial patients. The trial population must also adequately represent the U.S. population, and the data must be applicable to the U.S. population and U.S. medical practice in ways that the U.S. FDA deems clinically meaningful. Generally, the patient population for any clinical trial conducted outside of the United States must be representative of the population for which we intend to seek approval in the United States. In addition, while these clinical trials are subject to applicable local laws, U.S. FDA acceptance of the data will be dependent upon its determination that the trials also comply with
23
all applicable U.S. laws and regulations. There can be no assurance that the U.S. FDA will accept data from trials conducted outside of the United States. If the U.S. FDA does not accept the data from any clinical trials that we or our collaboration partners conduct outside the United States, it would likely result in the need for additional clinical trials, which would be costly and time-consuming and delay or permanently halt our ability to develop and market these or other product candidates in the United States. In other jurisdictions, for instance, in Japan, there is a similar risk regarding the acceptability of clinical trial data conducted outside of that jurisdiction.
In addition, there are risks inherent in conducting clinical trials in multiple jurisdictions, inside and outside of the United States, such as:
•regulatory and administrative requirements of the jurisdiction where the trial is conducted that could burden or limit our ability to conduct our clinical trials;
•foreign exchange fluctuations;
•manufacturing, customs, shipment and storage requirements;
•cultural differences in medical practice and clinical research; and
•the risk that the patient populations in such trials are not considered representative as compared to the patient population in the target markets where approval is being sought.
If we receive regulatory approval for our product candidates, we are required to comply with comprehensive and ongoing regulatory requirements, including conducting confirmatory clinical trials of any drug that receives accelerated approval. In addition our product candidates, if approved, could be subject to labeling and other restrictions and market withdrawal and we may be subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with our drugs, which may materially adversely affect our business, prospects, financial condition and results of operations.
Any current or future product candidate for which we receive accelerated approval from the U.S. FDA, or similar conditional approval from the EMA or comparable regulatory authorities in other jurisdictions may be required to undergo one or more confirmatory clinical trials. If such drug fails to meet its safety and efficacy endpoints in such confirmatory clinical trials, the regulatory authority may withdraw its approval. There is no assurance that any such drug will successfully advance through its confirmatory clinical trial(s). Therefore, even if a drug receives accelerated approval from the U.S. FDA or similar conditional approval from the EMA or comparable regulatory authorities, such approval may be withdrawn at a later date, and under the Food and Drug Omnibus Reform Act of 2022 (FDORA), the FDA has increased authority for expedited procedures to withdraw approval of a drug or indication approved under accelerated approval if, for example, the confirmatory trial fails to verify the predicted clinical benefit of the product.
If the U.S. FDA, the EMA or a comparable foreign regulatory authority approves any of our product candidates, the manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, marketing, promotion and recordkeeping for the product will be subject to extensive and ongoing regulatory requirements. These requirements include submissions of safety and other post-marketing information and reports, registration, continued compliance with cGMPs and GCPs for any clinical trials that we conduct post-approval, as well as applicable product tracking and tracing requirements, all of which may result in significant expense and limit our ability to commercialize such products. Any regulatory approvals that we receive for our product candidates may also be subject to limitations on the approved indicated uses for which the product may be marketed or to the conditions of approval, or contain requirements for potentially costly post-marketing testing and surveillance to monitor the safety and efficacy of the product.
Further, on March 27, 2020, the Coronavirus Aid, Relief, and Economic Security (CARES) Act became law in response to the U.S. COVID-19 pandemic. Throughout the COVID-19 outbreak, there has been public concern over the availability and accessibility of critical medical products, and the CARES Act enhances U.S. FDA’s existing authority with respect to drug shortage measures. Under the CARES Act, we must have in place a risk management plan that identifies and evaluates the risks to the supply of approved drugs for certain serious diseases or conditions for each establishment where the drug or API is manufactured. The risk management plan will be subject to U.S. FDA review during an inspection. If we experience shortages in the supply of our marketed products, our results could be materially impacted.
In addition, regulatory policies may change or additional government regulations or legislation may be enacted that could prevent, limit or delay regulatory approval of our product candidates. If we fail to comply with existing requirements, are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any regulatory approval that we may have obtained or face regulatory or enforcement actions, which may materially adversely affect our business, prospects, financial condition and results of operations.
24
We may be subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with any of our products that receive regulatory approval, which may materially adversely affect our business, prospects, financial condition and results of operations.
Once a product is approved by the U.S. FDA, the EMA or a comparable foreign regulatory authority for marketing, it is possible that previously unknown problems may occur with the product, including problems with third-party manufacturers or manufacturing processes, packaging or failure to comply with regulatory requirements. If any of the foregoing occurs with respect to our products, it may result in, among other things:
•restrictions on the marketing or manufacturing or packaging of the product, withdrawal of the product from the market, or voluntary or mandatory product recalls;
•fines, warning letters or holds on clinical trials;
•refusal by the U.S. FDA, the EMA or comparable foreign regulatory authority to approve pending applications or supplements to approved applications filed by us, or suspension or revocation of product license approvals;
•requirements to conduct additional clinical trials, change our product labeling or submit additional applications or application supplements;
•product seizure or detention, or refusal to permit the import or export of products; and
•injunctions or the imposition of civil or criminal penalties.
The occurrence of any of these events, or any government investigation of alleged violations of law could require us to expend significant time and resources, could generate negative publicity, and may impair our ability to sell such product. If we or our collaborators are not able to maintain regulatory compliance, regulatory approval that has been obtained may be lost and we may not achieve or sustain profitability, which may materially adversely affect our business, prospects, financial condition and results of operations.
We may allocate our limited resources to pursue a particular product candidate or indication and fail to capitalize on product candidates or indications that may later prove to be more profitable or for which there is a greater likelihood of success, which may materially adversely affect our business, prospects, financial condition and results of operations.
Because we have limited financial and managerial resources, we must limit our licensing, development programs to specific product candidates that we identify for specific indications. As a result, we may forego or delay pursuit of opportunities with other product candidates or for other indications that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable commercial product candidates or profitable market opportunities, and our decisions concerning the allocation of development, collaboration, management and financial resources towards particular product candidates may not lead to the development of viable commercial products.
A breakthrough therapy designation or fast track designation by the U.S. FDA for our product candidates may not lead to a faster development or regulatory review or approval process, and either designation does not increase the likelihood that our product candidates will receive marketing approval.
We may seek breakthrough therapy designation for some of our product candidates. A breakthrough therapy is defined as a drug that is intended, alone or in combination with one or more other drugs, to treat a serious or life threatening disease or condition, and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. For drugs that have been designated as breakthrough therapies, interaction and communication between the U.S. FDA and the sponsor of the trial can help to identify the most efficient path for clinical development while minimizing the number of patients placed in ineffective control regimens.
We may also seek fast track designation for some of our product candidates. If a product candidate is intended for the treatment of a serious or life-threatening condition and the product demonstrates the potential to address unmet medical needs for this condition, the sponsor may apply for U.S. FDA fast track designation for a particular indication. Marketing applications filed by sponsors of products in fast track development may qualify for priority review under the policies and procedures offered by the U.S. FDA.
The U.S. FDA has broad discretion whether or not to grant breakthrough therapy designation or fast track designation. Accordingly, even if we believe one of our product candidates meets the criteria for breakthrough therapy designation or fast track designation, the U.S. FDA may disagree and instead determine not to make such designation. In any event, the receipt of a breakthrough therapy designation or fast track designation for a product candidate may not result in a faster development process, review or approval compared to other drugs and does not assure ultimate approval by the U.S. FDA. In addition, even
25
if one or more of our product candidates qualify for breakthrough designation or fast track designation, the U.S. FDA may later decide that the drugs no longer meet the conditions for qualification.
We may seek orphan drug designation for some of our product candidates. However, we may be unsuccessful in obtaining or may be unable to maintain the benefits associated with orphan drug designation, including the potential for market exclusivity.
As part of our business strategy, we may seek orphan drug designation for some of our product candidates, and we may be unsuccessful. Regulatory authorities in some jurisdictions, including the U.S. and the European Union, may designate drugs for relatively small patient populations as orphan drugs. Under the Orphan Drug Act, the U.S. FDA may designate a drug as an orphan drug if it is a drug intended to treat a rare disease or condition, which is generally defined as a patient population of fewer than 200,000 individuals annually in the U.S., or a patient population greater than 200,000 in the U.S. where there is no reasonable expectation that the cost of developing the drug will be recovered from sales in the U.S. In the U.S., orphan drug designation entitles a party to financial incentives such as opportunities for grant funding towards clinical trial costs, tax advantages and user-fee waivers.
Similarly, in the European Union, the European Commission grants orphan medicinal product designation after receiving the opinion of the European Medicines Agency's, or EMA's, Committee for Orphan Medicinal Products on an orphan medicinal product designation application. Orphan medicinal product designation may be granted in respect of products that are intended for the diagnosis, prevention or treatment of life-threatening or chronically debilitating conditions affecting not more than 5 in 10,000 persons in the European Union and for which no satisfactory method of diagnosis, prevention, or treatment has been authorized for marketing in the European Union (or, if such a method exists, the product would be of a significant benefit to those affected by the condition). In addition, designation is granted for products intended for the diagnosis, prevention, or treatment of a life-threatening, seriously debilitating or serious and chronic condition when, without incentives, it is unlikely that sales of the drug in the European Union would generate sufficient return to justify the necessary investment in developing the product. In the European Union, orphan medicinal product designation entitles a party to financial incentives such as reduction of fees or fee waivers.
Generally, if a drug with an orphan drug designation subsequently receives the first marketing approval for the indication for which it has such designation, the drug is entitled to a period of marketing exclusivity, which precludes the EMA or the U.S. FDA from approving another marketing application for the same drug and indication for that time period, except in limited circumstances. The applicable period is seven years in the U.S. and ten years in the European Union. The European Union exclusivity period can be reduced to six years if a product no longer meets the criteria for orphan drug designation or if the product is sufficiently profitable so that market exclusivity is no longer justified.
Even if we obtain orphan drug exclusivity for a drug, that exclusivity may not effectively protect the designated drug from competition because different drugs can be approved for the same condition. Even after an orphan drug is approved, the U.S. FDA can subsequently approve another drug for the same condition if the U.S. FDA concludes that the later drug is clinically superior in that it is shown to be safer, more effective or makes a major contribution to patient care. In addition, a designated orphan drug may not receive orphan drug exclusivity if it is approved for a use that is broader than the indication for which it received orphan designation. Moreover, orphan drug exclusive marketing rights in the U.S. may be lost if the U.S. FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the drug to meet the needs of patients with the rare disease or condition. Orphan drug designation neither shortens the development time or regulatory review time of a drug nor gives the drug any advantage in the regulatory review or approval process. While we intend to continue seek orphan drug designation for our product candidates, we may never receive such designations. Even if we receive orphan drug designation for any of our product candidates, there is no guarantee that we will enjoy the benefits of those designations.
We face substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than we do.
The development and commercialization of new drug products is highly competitive. We face competition with respect to our current products and product candidates, and we and our collaborators will face competition with respect to any product candidates that we or they may seek to develop or commercialize in the future, from major pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide. There are a number of large pharmaceutical and biotechnology companies that currently market and sell products or are pursuing the development of products for the treatment of the disease indications for which we are developing our product candidates in the hematology/oncology area. Some competitive products and therapies are based on scientific approaches that are the same as or similar to our approach, and others are based on entirely different approaches. Potential competitors also include academic institutions, government agencies and other public and private research organizations that conduct research, seek patent protection and establish collaborative
26
arrangements for research, development, manufacturing and commercialization. For additional information regarding our competition, see the section of this annual report titled “Item 4.B. Business Overview.”
We are developing our product candidates for the treatment of cancer. There are a variety of available drug therapies marketed for cancer. In many cases, these drugs are administered in combination to enhance efficacy, and cancer drugs are frequently prescribed off-label by healthcare professionals. Some of the currently approved drug therapies are branded and subject to patent protection, and others are available on a generic basis. Many of these approved drugs are well established therapies and are widely accepted by physicians, patients and third-party payors. Insurers and other third-party payors may also encourage the use of generic products. We expect that our product candidates, if approved, will be priced at a significant premium over competitive generic products. This may make it difficult for us to achieve our business strategy of using our product candidates in combination with existing therapies or replacing existing therapies with our product candidates.
Our competitors may develop products that are more effective, safer, more convenient or less costly than any that we are developing or that would render our product candidates obsolete or non-competitive. In addition, our competitors may discover biomarkers that more efficiently measure metabolic pathways than our methods, which may give them a competitive advantage in developing potential products. Our competitors may also obtain marketing approval from the U.S. FDA or other regulatory authorities for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market.
Additionally, even if we are successful in achieving marketing approval to commercialize a product candidate faster than our competitors, we may face competition due to the changing regulatory environment. In the United States, for small molecule drugs, the FDCA provides three years of marketing exclusivity for an NDA, or supplement to an existing NDA, if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by the FDA to be essential to the approval of the application, for example for new indications, dosages, or strengths of an existing drug. This three-year exclusivity covers only the modification for which the drug received approval on the basis of the new clinical investigations and does not prohibit the FDA from approving abbreviated new drug applications, or ANDAs, for drugs containing the active agent for the original indication or condition of use. The FDCA also provides a five-year period of non-patent marketing exclusivity within the United States to the first applicant to obtain approval of an NDA for a new chemical entity. A drug is a new chemical entity if the FDA has not previously approved any other new drug containing the same active moiety, which is the molecule or ion responsible for the action of the drug substance. During the exclusivity period, the FDA may not accept for review an ANDA or a 505(b)(2) NDA submitted by another company for another drug based on the same active moiety, regardless of whether the drug is intended for the same indication as the original innovator drug or for another indication, where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity or non-infringement to one of the patents listed with the FDA by the innovator NDA holder. Three-year and five-year exclusivity will not delay the submission or approval of a full NDA. However, an applicant submitting a full NDA would be required to conduct or obtain a right of reference to all of the nonclinical studies and adequate and well-controlled clinical trials necessary to demonstrate safety and efficacy.
Further, in the United States, the Biologics Price Competition and Innovation Act of 2009 created an abbreviated approval pathway for biological products that are demonstrated to be “highly similar,” or biosimilar, to or “interchangeable” with an U.S. FDA-approved biological product. This pathway could allow competitors to reference data from biological products already approved after 12 years from the time of approval. In Europe, the European Commission has granted marketing authorizations for several biosimilars pursuant to a set of general and product class-specific guidelines for biosimilar approvals issued over the past few years. In the European Union, a competitor may reference data from biological products already approved after the expiry of an eight year data exclusivity period, but will not be able to get on the market until 10 years after the time of approval of the reference product. This 10-year period will be extended to 11 years if, during the first eight of those 10 years, the marketing authorization holder obtains an approval for one or more new therapeutic indications that bring significant clinical benefits compared with existing therapies. In addition, companies may be developing biosimilars in other countries that could compete with our products.
If competitors are able to obtain marketing approval for drugs or biosimilars referencing our products, our products may become subject to competition from such drugs or biosimilars, with the attendant competitive pressure and consequences. Expiration or successful challenge of our applicable patent rights could also trigger competition from other products, assuming any relevant exclusivity period has expired.
Many of our competitors have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller and other clinical stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific and management personnel, establishing clinical trial
27
sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Risks Related to Our Reliance on Partnerships and Other Third-Parties
Collaborations on products and product candidates are important to our business, and future collaboration and license agreements may also be important to us. If we are unable to maintain any of these partnerships or if these partnerships are not successful, our business could be materially adversely affected.
We have in the past entered into, and intend to continue to enter into on a case-by-case basis, collaborations with other companies that we believe provide us with valuable funding and other benefits. However, we cannot ensure that any such collaboration will continue or be successful. Our inability to find a partner for any of our product candidates may result in our termination of that specific product candidate program or evaluation of a product candidate in a particular indication. For example, in March 2015, we and Celgene Corporation (now part of Bristol-Myers Squibb) agreed to end the existing co-development and co-promotion agreement for felzartamab (MOR202), following which we regained the rights to felzartamab. We have subsequently partnered Chinese regional rights to felzartamab (in November 2017), and our partner I-Mab will further develop felzartamab in multiple myeloma, or MM, and potentially also for additional indications, for China, Hong Kong, Macao and Taiwan. In June 2022, Human Immunology Biosciences, Inc. (HI-Bio) obtained exclusive rights to develop and commercialize felzartamab across all indications worldwide, with the exception of Greater China. In November 2018, we entered into a collaboration and licensing agreement with I-Mab for an additional proprietary program, MOR210. Our partner I-Mab will perform certain preclinical and clinical development activities (China, Hong Kong, Macao, Taiwan and South Korea). In June 2022 HI-Bio obtained exclusive worldwide rights to develop and commercialize MOR210 across all indications worldwide, with the exception of Greater China and South Korea. In June 2022, we announced with Pfizer and Incyte a clinical trial collaboration and supply agreement to investigate the immunotherapeutic combination of Pfizer’s maplirpacept (TTI-622), a novel SIRPα-Fc fusion protein, and Monjuvi® (tafasitamab-cxix) plus lenalidomide in patients with relapsed or refractory DLBCL who are not eligible for autologous stem cell transplant (ASCT). In January 2020, we entered into a collaboration and license agreement with Incyte Corporation, or Incyte, to further develop and commercialize our proprietary antibody tafasitamab globally. This agreement received clearance by the U.S. antitrust authorities under the Hart-Scott-Rodino Act as well as by the German and Austrian antitrust authorities on or before March 2, 2020, and became effective on March 3, 2020. Under the terms of the agreement, we and Incyte were co-commercializing tafasitamab in the U.S., while Incyte had exclusive commercialization rights outside of the U.S. In addition, we and Incyte agreed to co-develop tafasitamab broadly in r/r DLBCL, frontline DLBCL, as well as additional indications beyond DLBCL, such as follicular lymphoma (FL) and marginal zone lymphoma (MZL). In February 2024, Incyte obtained exclusive rights worldwide for tafasitamab and assumed full responsibility and agreed to cover all costs going forward for the development and commercialization of tafasitamab.
We cannot ensure that any such collaboration or license agreement or further clinical development or the commercialization will be successful.
In the future, we may enter into additional collaborations and license agreements to fund our development programs or to gain access to sales, marketing or distribution capabilities and we may also enter into collaborations and licensing or purchasing agreements under which we provide funding and gain access to targets, technologies or compounds.
Under the collaboration agreements where we grant our partners an exclusive license to certain therapeutic antibodies for specific targets we receive license fees, research and development funding, milestone payments and/or, if a product is approved for marketing, royalties in return. Following the discovery and preclinical testing phase, these partners are typically solely responsible for the further development of the product candidate and therefore exercise full control over its further development and potential commercialization. In other collaborations we also rely on both parties’ capabilities to co-develop and co-commercialize. In clinical trial collaborations, for example the one signed with Pfizer, we rely on the partners’ capabilities and diligence for performance of the agreed trial. Our existing collaborations, and any future collaborations and licensing or purchasing agreements we enter into, therefore may pose a number of risks, including the following:
•collaborators, licensees or licensors may have significant discretion in determining the efforts and resources that they will apply to these collaborations or license agreements;
•collaborators, licensees or licensors may not perform their obligations as expected by us or by health authorities, such as the U.S. FDA, the EMA or comparable foreign regulatory authorities;
•collaborators, licensees or licensors may dissolve, merge, be bought, or may otherwise become unwilling to fulfill the initial terms of the collaboration or license agreement with us;
•collaborators, licensees or licensors may not pursue development and commercialization of any product candidates that achieve regulatory approval or may elect not to continue or renew development or commercialization programs based on
28
clinical trial results, changes in the partners’ strategic focus or available funding, or external factors, such as an acquisition, that divert resources or create competing priorities or the actual or perceived competitive situation in a specific indication;
•collaborators, licensees or licensors may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or may require a new formulation of a product candidate for clinical testing;
•collaborators, licensees or licensors may not put sufficient resources or may delay or underperform in their activities to seek regulatory approval, pricing approval and perform commercial and medical affairs activities to market and sell the product;
•collaborators, licensees or licensors may not be compliant with applicable laws and regulations;
•collaborators, licensees or licensors could independently develop, or develop with third-parties, products that compete directly or indirectly with our products or product candidates if the partner believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours;
•product candidates discovered in collaboration with us may be viewed by our collaborators as competitive with their own product candidates or products, which may cause collaborators to cease to devote resources to the commercialization of our product candidates;
•a collaborator or licensee with marketing and distribution rights to one or more of our product candidates that achieve regulatory approval may not commit sufficient resources to the marketing and distribution of such product or products;
•a target, technology or compound we in-license, collaborate/co-develop or acquire could be determined to not perform in any pre-clinical, clinical, supply or commercialization activities as expected, including but not limited to showing deficiencies in pharmacology, pharmacokinetics, toxicology, safety, efficacy or manufacturing data, or such data may not be competitive with other projects from third parties, which may cause us to devote additional resources to the research, development, manufacturing and commercialization, may cause a delay or failure of regulatory approval or may cause us to stop the project and write off the investment already taken;
•disagreements with collaborators, licensees or licensors, including disagreements over proprietary rights, contract interpretation and breach of contract claims, payment obligations or the preferred course of development, supply and commercialization, might cause delays or termination of the research, development or commercialization of products or product candidates, might lead to additional responsibilities, including financial obligations for us with respect to products or product candidates, or delays or withholding of any payments due or might result in litigation or arbitration, any of which would be time-consuming and expensive;
•collaborators, licensees or licensors may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation;
•collaborators, licensees or licensors may infringe the intellectual property rights of third-parties, which may expose us to litigation and potential liability; and
•collaborations, or license agreements may be terminated for the convenience of the collaborator or licensee and, if terminated, we could be required to raise additional capital to pursue further development or commercialization of the applicable product candidates.
If our research, development and commercial partnerships do not result in the successful development and commercialization, as applicable, of products or if one of our partners terminates its agreement with us, we may not receive any future research funding or milestone or royalty payments under the agreement. If any commercial partner underperforms or terminates the agreement with us, we may generate less revenues or less profits / more losses. If we do not receive the funding, or do not generate the revenues or profits, we expect under these agreements, the development and commercialization of our product candidates and products could be delayed, and we may need additional resources to develop and commercialize our proprietary product candidates. All of the risks relating to product development, regulatory approval and commercialization described in this report also apply to the activities of our program collaborators and licensees. If our in-licensing or acquisition activities do not result in the successful development and commercialization, we may generate less revenues or less profits / more losses.
Additionally, subject to its contractual obligations to us, if one of our partners is involved in a business combination, the partners might de-emphasize or terminate the development or commercialization of any product candidate licensed to it by us. If one of our collaborators terminates its agreement with us, we may find it more difficult to attract new partners in a timely manner.
29
We face significant competition in seeking new partnerships.
For all our proprietary product candidates, we face significant competition. This may negatively impact our ability to enter into potential partnerships or licensing agreements for our compounds. Also for any in-licensing and acquisition activities, we face significant competition. Our ability to reach definitive agreements for partnerships will depend, among other things, upon our assessment of the partner’s resources and expertise, the terms and conditions of the proposed partnership and the proposed partner’s evaluation of a number of factors. The factors, depending on the type of partnership we or the partner would consider, may include the design or results of clinical trials, the likelihood of approval by the U.S. FDA, the EMA or comparable foreign regulatory authorities, the potential market for the subject product candidate, market access and pricing considerations in the respective territory, the costs and complexities of manufacturing and delivering such product candidate to patients, the potential of competing products, incidence and prevalence of the respective disease, and industry and market conditions generally. The partner may also consider alternative product candidates for similar indications that may be available to collaborate on and whether such collaboration could be more attractive than the one with us for our product candidate. In the case of in-licensing or acquisition partnerships, the partner may also consider alternative offers or partners other than MorphoSys to be more attractive, or keeping all rights to themselves.
Collaborations and commercialization partnerships are complex and time-consuming to negotiate and document. If we are unable to reach agreements with suitable partners on a timely basis, on acceptable terms, or at all, we may have to curtail or even stop the development of a product candidate in one or all indications, in one or all territories in the world, reduce or delay one or more of our development programs, delay its potential commercialization, or increase our expenditures and undertake development or commercialization activities at our own expense. If we elect to fund and undertake development or commercialization activities on our own, we may need to obtain additional expertise and additional capital, which may not be available to us on acceptable terms or at all. If we fail to enter into collaborations and other partnerships and do not have sufficient funds or expertise to undertake the necessary development and commercialization activities, we may not be able to further develop our product candidates in any or all indications or bring them to market in any or all territories in the world and our business may be materially and adversely affected. If we fail to identify additional targets, technologies or compounds for in-licensing, collaboration, co-development or commercialization, we may not be able to further expand our pipeline. If we succeed to identify further targets, technologies or compounds for in-licensing, collaboration, co-development or commercialization, we may need to increase our expenditures.
Our reliance on third-party suppliers could harm our ability to commercialize our drugs or any other drug candidates that may be approved in the future.
We do not currently own or operate manufacturing facilities for the production of our drugs or any other drug candidates that may be approved in the future. Our third-party suppliers may not be required to provide us with any guaranteed minimum production levels or have dedicated capacity for our drugs. As a result, there can be no assurances that we will be able to obtain sufficient quantities of our drugs or any other drug candidates that may be approved in the future, which could have a material adverse effect on our business as a whole. We are not certain, however, that our suppliers will be able to meet our demand, either because of the nature of our agreements with those suppliers, our limited experience with those suppliers or our relative importance as a customer to those suppliers. It may be difficult for us to assess their ability to timely meet our demand in the future based on past performance. While our suppliers have generally met our demand for their products on a timely basis in the past, they may subordinate our needs in the future to their other customers.
Establishing additional or replacement suppliers for the drug substance or drug product used in our drug candidates or approved drugs, if required, may not be accomplished quickly. If we are able to find a replacement supplier, such replacement supplier would need to be qualified and may require additional regulatory approval, which could result in further delay. While we seek to maintain adequate inventory, any interruption or delay in the supply of components or materials, or our inability to obtain such drug substance and drug product from alternate sources at acceptable prices in a timely manner could impede, delay, limit or prevent our development efforts, which could harm our business, results of operations, financial condition and prospects.
We rely and expect to continue to rely on third-parties, including research/medical institutions, clinical investigators, CROs and/or other service providers, to conduct our development activities (preclinical studies, quality testing and clinical trials) and perform data collection, analysis and reporting, which may result in costs and delays in the development of our product candidates. If these third-parties do not successfully carry out their contractual duties or meet expected deadlines, we may not be able to obtain regulatory approval for or commercialize our product candidates, and our business could be materially adversely affected.
We rely and expect to continue to rely on public and private medical/research institutions, clinical investigators, CROs, service providers and collaboration partners to conduct our early phase and late phase product development activities including the conduct of preclinical studies and clinical trials. Our development activities conducted in reliance on third-parties may be delayed, suspended or terminated, including for the following reasons:
30
•the third-parties do not devote a sufficient amount of resources, time or effort to our activities or otherwise fail to successfully carry out their contractual duties or to meet regulatory obligations or expected deadlines;
•we replace a third-party; or
•the quality or accuracy of the data obtained by third-parties is compromised due to their failure to adhere to the study plans/protocols, GxP, regulatory requirements or for other reasons.
Although we perform sponsor oversight and audits using risk-based approaches, we do not have the ability to control every action of third-parties in their conduct of development activities. Nevertheless, we are responsible for ensuring that each of our development activities is conducted in accordance with the applicable study plan/protocol, GxP, legal, regulatory, intellectual property and scientific standards, and our reliance on these third-parties does not relieve us of our sponsor responsibilities. We and our third-parties are required to comply with GxP standards, which are regulations and guidelines enforced by the U.S. FDA, the competent authorities of the member states of the European Economic Area, or EEA, and comparable foreign regulatory authorities for all of our products in clinical development. Regulatory authorities enforce these GxPs through periodic inspections of trial sponsors, principal investigators and trial sites, CROs and/or other involved service providers. If we or any of our third-parties fail to comply with applicable GxP standards, the study data generated in our preclinical studies and/or clinical trials may be deemed unreliable and the U.S. FDA, the EMA or comparable foreign regulatory authorities may require us to perform additional studies before potentially approving our marketing authorization applications. We cannot ensure that upon inspection by a given regulatory authority, such regulatory authority will determine that all of our development activities comply with GxP regulations. If third-parties do not successfully carry out their contractual duties or obligations or meet expected deadlines, if they need to be replaced or if the quality or accuracy of the data they obtain is compromised due to the failure to adhere to our study plans/protocols, GxP and other regulatory requirements or for other reasons, our preclinical studies or clinical trials may be extended, delayed or terminated and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates.
Third-party performance failures may increase our development costs, delay our ability to obtain regulatory approval and delay or prevent the commercialization of our product candidates. While we believe that there are alternative sources to provide these services, in the event that we seek such alternative sources, we may not be able to enter into replacement arrangements without incurring delays or additional costs.
We currently rely on third-party suppliers and CMOs for the manufacturing and distribution of our product candidates, and our dependence on these third-parties may impair the development of our product candidates. Moreover, we rely on third-parties to produce commercial supplies of approved product candidate and our commercialization of any of our product candidates could be stopped, delayed or made less profitable if those third-parties fail to provide us with sufficient quantities of product or fail to do so at acceptable quality levels or prices or fail to otherwise complete their duties in compliance with their obligations to us or in compliance with applicable laws. Service or supply failures, or other failures, business interruptions, or other disasters affecting the manufacturing facilities of any party participating in the supply chain, would adversely affect our ability to supply our product candidates and products.
We do not currently have, nor do we plan to acquire, the infrastructure or capability internally to manufacture our preclinical (with the exclusion of non-GLP testing), clinical product supplies, our commercial product, and we lack the resources and the capability to manufacture any of our product candidates on a clinical or commercial scale under GMP. We therefore rely on and expect to continue to rely on third-party contract manufacturing organizations, or CMOs, for the supply of cGMP-grade, clinical trial materials and commercial quantities of our product candidates and our approved products. The facilities used by our CMOs or other third-party manufacturers to manufacture our product candidates are subject to the U.S. FDA’s, the EMA’s and other comparable regulatory authorities’ preapproval inspections and routine inspections that will be conducted after we submit our BLA or NDA to the U.S. FDA or the required approval documents to any other relevant regulatory authority or after approval. Although we perform oversight of the manufacturing and testing activities by involvement in e.g. the Change Control and Deviation management of the CMO and qualification audits prior to contracting a CMO and subsequent regular audits of such facilities and GMP procedures, we are completely dependent on our contract manufacturers or other third-party manufacturers for compliance with cGMPs for manufacture of both active drug substances and finished drug products. If our contract manufacturers or other third-party manufacturers cannot successfully manufacture sufficient amounts of material that conforms to applicable specifications and the strict regulatory requirements of the U.S. FDA, the EMA or another comparable regulatory authority, we may not be able to secure and/or maintain regulatory approvals for our products manufactured at these facilities. In addition (except for our oversight obligations described above), we have no control over the ability of our contract manufacturers or other third-party manufacturers to maintain adequate quality control and quality assurance procedures and qualified personnel. If the U.S. FDA, the EMA or another comparable regulatory authority finds deficiencies at these facilities for the manufacture of our product candidates or products for commercial sale, or if it withdraws any approval because of deficiencies at these facilities in the future, we may need to find alternative manufacturing facilities, which would significantly impact our ability to develop, obtain regulatory approval for or market our product candidates, once approved. If, for any
31
reason, we were to experience an unexpected loss of supply of our product candidates, combination drug, or placebo or comparator product used in certain of our clinical trials, whether as a result of manufacturing, supply or storage issues or otherwise, we could experience delays, disruptions, suspensions or terminations of, or be required to restart or repeat, any pending or ongoing clinical trials. If market demand increases, our current planning assumptions the CMO might not be willing or able to supply this additional material, leading to supply shortage on the market.
We rely on our manufacturers to purchase from third-party suppliers the materials necessary to produce our product candidates for our clinical trials and our approved product for commercial sale. For certain items, there are a limited number of suppliers for raw materials that we use to manufacture our products and appropriate lead times for ordering such materials are factored into the manufacturing plans. However, there may be a need to assess alternate suppliers to prevent a possible disruption of the manufacture of the materials necessary to produce our product candidates for our clinical trials, and if approved, for commercial sale. Moreover, we currently do not have any agreements in place for the commercial production of these raw materials. Although we generally do not begin a clinical trial unless we believe we have access to a sufficient supply of a product candidate to complete the clinical trial, any significant delay in the supply of a product candidate, or the raw material components thereof, could considerably delay the completion of our clinical trials, product testing and potential regulatory approval of our product candidates. Such delays could for example be caused by the implementation of corrective actions at the supplier, or even replacement of a contract manufacturer or other involved third-parties. If we or our manufacturers are unable to purchase these raw materials after regulatory approval has been obtained for our product candidates, the commercial launch of our product candidates would be delayed or there would be a shortage in supply, which would impair our ability to generate revenue from the sale of our product candidates. Additionally, we may experience unforeseen difficulties or challenges in the manufacture of our product candidates on a commercial scale compared to the manufacture for clinical purposes. We currently rely on CMOs for the manufacturing of each of our proprietary product candidates. Thus any regulatory action, service failure, business interruptions, or other disasters affecting the facilities of our CMOs for our proprietary product candidates could result in a significant delay in the production and supply and could, as a result, have a material adverse effect on our business, results of operations, financial condition and prospects.
The manufacture of our product candidates approved product is complex. Our third-party manufacturers may encounter difficulties in production. If we encounter any such difficulties, our ability to supply our product candidates for clinical trials or, if approved, for commercial sale could be delayed or halted entirely.
The manufacture of our product candidates is complex and requires significant expertise and capital investment, including the development of advanced manufacturing techniques and process controls. The processes for manufacturing our approved product and our product candidates is susceptible to product loss due to contamination, equipment failure or improper installation or operation of equipment, vendor or operator error, contamination and inconsistency in yields, variability in product characteristics and difficulties in scaling the production process or product loss during fill and finishing. Even minor deviations from normal manufacturing processes could result in reduced production yields, product defects and other supply disruptions.
If microbial, viral or other contaminations are discovered in our product candidates or in the manufacturing facilities in which our product candidates are made, such manufacturing facilities may need to be closed for an extended period of time to investigate and remedy the contamination. Any adverse developments affecting manufacturing operations for our approved product or other product candidates may result in shipment delays, inventory shortages, lot failures, product withdrawals or recalls, or other interruptions in the supply of our products. We may also have to take inventory write-offs and incur other charges and expenses for products that fail to meet specifications, undertake costly remediation efforts or seek more costly manufacturing alternatives.
Risks Related To Our Intellectual Property Rights
If we are unable to obtain and maintain sufficient intellectual property protection for our products or product candidates, or if the scope of our intellectual property protection is not sufficiently broad, our ability to commercialize our products or product candidates successfully and to compete effectively may be materially adversely affected.
Our success depends in large part on our ability to obtain and maintain protection with respect to our intellectual property and proprietary technology. We rely upon a combination of patents, trade secret protection and confidentiality agreements to protect the intellectual property related to our products and product candidates. The patent position of pharmaceutical companies is generally uncertain because it involves complex legal and factual considerations. The standards applied by the United States Patent and Trademark Office, or USPTO, and foreign patent offices in granting patents are not always applied uniformly or predictably and can change. The patent applications that we own or in-license may fail to result in issued patents, and if they do, such patents may not cover our products or product candidates in the United States or in other countries. Accordingly, we cannot predict whether additional patents protecting our technology or our product candidates will issue in the United States or
32
in non-U.S. jurisdictions, or whether any patents that do issue will have claims of adequate scope to provide us with a competitive advantage. Additionally, the laws of some foreign countries do not protect intellectual property rights to the same extent as the laws of the United States, and many companies have encountered significant problems in protecting and defending such rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property rights, particularly those relating to biotechnology, which could make it difficult for us to stop the infringement of our licensed and owned patents, the reproduction of our manufacturing or other know-how or marketing of competing products in violation of our proprietary rights generally. Any of these outcomes could impair our ability to prevent competition from third-parties, which may have a material adverse effect on our business.
Competitors may use our technologies in jurisdictions where we have not obtained or are unable to adequately enforce patent protection to develop their own products and further, may export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States and Europe. These products may compete with our products, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing. Proceedings to enforce our patent rights, whether or not successful, could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing, and could provoke third-parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
The patent prosecution process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we may fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. In addition, we or our licensors may only pursue, obtain or maintain patent protection in a limited number of countries. There is no assurance that all potentially relevant prior art relating to our patents and patent applications has been found. We may be unaware of prior art or other documents or experiments that could be used to invalidate an issued patent or prevent our pending patent applications from issuing as patents. Even if patents do successfully issue and even if such patents cover our products or product candidates, third-parties (including our licensees) may challenge their validity, enforceability or scope, which may result in such patents being narrowed or invalidated. Further, the existence of issued patents does not guarantee our right to practice the patented technology or commercialize the patented product. Third-parties may have or obtain rights to patents which they may use to prevent or attempt to prevent us from commercializing any of our patented product candidates, or which might require us to take license to such patents in order to be able to commercialize the respective product candidates. If these other parties are successful in obtaining valid and enforceable patents, and establishing our infringement of those patents, we could be prevented from selling our products unless we were able to obtain a license under such third-party patents. In addition, third-parties may seek approval to market their own products similar to or otherwise competitive with our products. In these circumstances, we may need to defend and/or assert our patents, including by filing lawsuits alleging patent infringement. In any of these types of proceedings, a court or agency of competent jurisdiction may find our patents invalid and/or unenforceable.
Furthermore, even if they are unchallenged, our patents and patent applications may not adequately protect our intellectual property, provide exclusivity for our products or product candidates, prevent others from designing around our claims or otherwise provide us with a competitive advantage. Additionally, our confidentiality agreements and other contractual protections may not be adequate to protect our intellectual property from unauthorized disclosure, third-party infringement or misappropriation. We may not have adequate remedies in the case of a breach of any such agreements, and our trade secrets and other proprietary information could be disclosed to our competitors or others may independently develop substantially equivalent or superior proprietary information and techniques or otherwise gain access to our trade secrets or disclose such technologies. In addition, the research resulting in certain of our licensed patent rights and technology has been, and may in the future be, funded by the government or other institutional organizations that may have certain rights, including march-in rights, to such patent rights and technology.
If the patent applications we own or have in-licensed with respect to our product candidates fail to issue as patents, if their breadth or strength of protection is narrowed or threatened, or if they fail to provide meaningful exclusivity, it could dissuade companies from collaborating with us and adversely affect our competitive position. We cannot offer any assurances about which, if any, patents will issue, the breadth of any such patents or whether any issued patents will be found invalid or unenforceable or will be threatened by third-parties. Any successful challenge to any patents owned by or licensed to us could deprive us of rights necessary for the successful commercialization of any product or product candidate that we may develop and could impair or eliminate our ability to collect future revenues and royalties with respect to such products or product candidates. Since patent applications in the United States and most other countries are confidential for a period of time after filing, and some remain so until issued, we cannot be certain that we were the first to file any patent application related to a product or product candidate. In addition, patents have a limited lifespan. In the United States and most foreign jurisdictions, the natural expiration of a patent is generally 20 years from the earliest filing date of a non-provisional patent application to which the patent claims priority. Various extensions may be available; however, the life of a patent and the protection it affords
33
is limited. If we encounter delays in obtaining regulatory approvals, the period of time during which we could market a product under patent protection could be reduced. Even if patents covering our product candidates are obtained, once such patents expire, we may be vulnerable to competition from generic, similar or biosimilar products. The launch of a generic version or biosimilar version of one of our products in particular would be likely to result in an immediate and substantial reduction in the demand for our product, which could have a material adverse effect on our business, financial condition, results of operations or prospects.
We do not know if, when, or how the U.S. FDA may change the orphan drug regulations and policies in the future, and it is uncertain how any changes might affect our business.
On August 3, 2017, the Congress passed the U.S. FDA Reauthorization Act of 2017 (“FDARA”). FDARA, among other things, codified the U.S. FDA’s preexisting regulatory interpretation, to require that a drug Sponsor demonstrate the clinical superiority of an orphan drug that is otherwise the same as a previously approved drug for the same rare disease in order to receive orphan drug exclusivity. The law reverses prior precedent holding that the Orphan Drug Act unambiguously requires that the U.S. FDA recognize the orphan exclusivity period regardless of a showing of clinical superiority. Moreover, in the Consolidated Appropriations Act of 2021, Congress did not further change this interpretation when it clarified that the interpretation codified in FDARA would apply in cases where U.S. FDA issued an orphan designation before the enactment of FDARA but where product approval came after the enactment of FDARA. The U.S. FDA may further reevaluate the Orphan Drug Act and its regulations and policies. We do not know if, when, or how the U.S. FDA may change the orphan drug regulations and policies in the future, and it is uncertain how any changes might affect our business. Depending on what changes the U.S. FDA may make to its orphan drug regulations and policies, our business could be adversely impacted.
Obtaining and maintaining our patent protection, including patents licensed from third-parties, depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
The USPTO and foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other provisions during the patent process. There are situations in which non-compliance can result in abandonment or lapse of a patent or patent application, resulting in partial loss, complete loss or unenforceability of patent rights in the relevant jurisdiction. In such an event, competitors might be able to enter the market earlier than would otherwise have been the case.
If we or our licensors fail to maintain the patents and patent applications covering or otherwise protecting our product candidates, it could materially harm our business. In addition, to the extent that we have responsibility for taking any action related to the prosecution or maintenance of patents or patent applications in-licensed from a third-party, any failure on our part to maintain the in-licensed intellectual property could jeopardize our rights under the relevant license and may expose us to liability.
Third-parties might claim that we have not complied with the provisions of the respective governmental patent agencies. For example, third-parties might claim that not all prior art documents, or not all other documents or experiments, were submitted to the respective agencies under appropriate law. Such claims could lead to proceedings that are time-consuming and expensive. Such proceedings can result in abandonment or lapse of a patent or patent application, resulting in partial loss, complete loss or unenforceability of patent rights in the relevant jurisdiction. If such third-party claims are raised in the context of a pending litigation, then such proceedings can also result in a judgment that would require us to pay the other parties’ litigation expenses.
We may be involved in lawsuits to protect or enforce our patents, which could be expensive, time-consuming and unsuccessful.
Even if the patent applications we own or license are issued, competitors may infringe these patents. To counter infringement or unauthorized use, we may be required to file infringement claims, which can be expensive and time-consuming.
In an infringement proceeding, a court may decide that a patent of ours or our licensors is not valid, is unenforceable and/or is not infringed, or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. An adverse result in any litigation or defense proceedings could put our patents or our licensors’ patents at risk of being invalidated or interpreted narrowly and could put our patent applications at risk of not issuing.
Interference proceedings, or other similar enforcement and revocation proceedings, provoked by third-parties or brought by us may be necessary to determine the priority of inventions with respect to our patents or patent applications or those of our licensors. An unfavorable outcome could require us to cease using the related technology or to attempt to license rights to it from the prevailing party. Our business could be harmed if the prevailing party does not offer us a license on commercially reasonable terms. Our defense of litigation or interference proceedings may fail and, even if successful, may result in substantial
34
costs and distract our management and other employees. We may not be able to prevent, alone or with our licensors, misappropriation of our intellectual property rights, particularly in countries where the laws may not protect those rights as fully as in the United States.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our common shares.
Even if resolved in our favor, litigation or other legal proceedings relating to our, our licensor’s or other third-parties’ intellectual property claims may cause us to incur significant expenses and could distract our personnel from their normal responsibilities. In addition, there could be public announcements of the results of hearings, motions, or other interim proceedings or developments, and if securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on the price of our ordinary shares. If not resolved in our favor, litigation may require us to pay any portion of our opponents’ legal fees. Such litigation or proceedings could substantially increase our operating losses and reduce the resources available for development activities or any future sales, marketing, or distribution activities. We may not have sufficient financial or other resources to conduct such litigation or proceedings adequately. Some of our competitors may be able to sustain the costs of such litigation or proceedings more effectively than we can because of their greater financial resources and more mature and developed intellectual property portfolios. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have a material adverse effect on our ability to compete in the marketplace.
By letter dated June 10, 2021, MorphoSys was notified by a licensor of the initiation of arbitration proceedings in the United States. The licensor alleged breach of contract and claimed damages for the licensor’s argued loss of revenues. Despite the patent expiry in 2018 confirmed by the licensor at the time, this was disputed and a significantly longer patent term was assumed. A final and binding award was provided by the arbitration court on March 21, 2023. The arbitration was decided in favor of MorphoSys with respect to all claims imposed by the licensor. MorphoSys was granted full reimbursement of its arbitration costs.
Developments in patent law could have a negative impact on our business.
From time to time, authorities in the United States, the European Union and other government authorities may change the standards of patentability, and any such changes could have a negative impact on our business.
For example, in the United States, the Leahy-Smith America Invents Act, or the America Invents Act, which was signed into law in 2011, includes a number of significant changes to U.S. patent law. These changes include a transition from a “first-to-invent” system to a “first-to-file” system, changes to the way issued patents are challenged, and changes to the way patent applications are disputed during the examination process. As a result of these changes, patent law in the United States may favor larger and more established companies that have greater resources to devote to patent application filing and prosecution. The USPTO has developed new and untested regulations and procedures to govern the full implementation of the America Invents Act, and many of the substantive changes to patent law associated with the America Invents Act, and, in particular, the first-to-file provisions became effective on March 16, 2013. Substantive changes to patent law associated with the America Invents Act may affect our ability to obtain patents, and if obtained, to enforce or defend them.
Also, case law may have a substantial impact on the way patents are prosecuted, examined and litigated. This also affects the scope of protection that is available in a specific jurisdiction. In the United States, Amgen Inc. v. Sanofi 872 F.3d 1367 (2017) had an impact on the way antibody claims are examined and litigated.
Developments of patent law in other jurisdictions may impact our business. For example, the Unified Patent Court in the European Union was introduced in June 2023. Patents that are valid and enforceable under national law may be considered invalid and/or unenforceable under the new unified system. Also the Unified Patent Court may invalidate patents not just in one single jurisdiction, but across all 17 participating member states of the European Union in one single trial.
Our commercial success depends significantly on our ability to operate without infringing the patents and other proprietary rights of third-parties.
Our success will depend in part on our ability to operate without infringing the proprietary rights of third-parties. Other entities may have or obtain patents or proprietary rights that could limit our ability to make, use, sell, offer for sale or import our products and future approved products or impair our competitive position.
Patents could be issued to third-parties that we may ultimately be found to infringe. Third-parties may have or may obtain valid and enforceable patents or proprietary rights that could block us from developing product candidates using our technology. Our
35
failure to identify or correctly interpret third-party patents, or to obtain or maintain a license to any technology that we require may materially harm our business, financial condition, results of operations or prospects. Furthermore, we could be exposed to a threat of litigation.
In the pharmaceutical and biotechnology industry, significant litigation and other proceedings regarding patents, patent applications, trademarks and other intellectual property rights have become commonplace. The types of situations in which we may become a party to such litigation or proceedings include:
•we or our collaborators may initiate litigation or other proceedings against third-parties seeking to invalidate the patents held by those third-parties or to obtain a judgment that our products or processes do not infringe those third-parties’ patents;
•if our competitors file patent applications that claim technology also claimed by us or our licensors, we or our licensors may be required to participate in interference, derivation, inter partes review or opposition proceedings to determine the priority of invention, inventorship or validity of the applicable patent rights which could jeopardize our patent rights and potentially provide a third-party with a dominant patent position;
•if third-parties initiate litigation claiming that our processes or the processes of our CMOs or CROs, products or uses thereof infringe their patent or other intellectual property rights, we and our collaborators will need to defend against such proceedings; and
•if a license to necessary technology is terminated, the licensor may initiate litigation claiming that our processes or products infringe or misappropriate their patent or other intellectual property rights and/or that we breached our obligations under the license agreement, and we and our collaborators would need to defend against such proceedings.
Any such lawsuit would be costly and could affect our results of operations and divert the attention of our management and scientific personnel. There is a risk that a court would decide that we or our collaborators are infringing the third-party’s patents and would order us or our collaborators to stop the activities covered by the patents. In that event, we or our collaborators may not have a viable alternative to the technology protected by the patent and may need to halt work on the affected product candidate or cease commercialization of an approved product. In addition, there is a risk that a court may order us or our collaborators to pay the other party damages. An adverse outcome in any litigation or other proceeding could subject us to significant liabilities to third-parties and require us to cease using the technology that is at issue or to license the technology from third-parties. We may not be able to obtain any required licenses on commercially acceptable terms or at all. Any of these outcomes could have a material adverse effect on our business, financial condition, results of operations or prospects.
The pharmaceutical and biotechnology industries have produced a significant number of patents, and it may not always be clear to industry participants, including us, which patents cover various types of products or methods of use.
The coverage of patents is subject to interpretation by the courts, and the interpretation is not always uniform or predictable. If we are sued for patent infringement, we would need to demonstrate that our products, methods or uses thereof either do not infringe the patent claims of the relevant patent or that the patent claims are invalid or unenforceable, and we may not be able to do this. Proving invalidity is difficult. For example, in the United States, proving invalidity requires a showing of clear and convincing evidence to overcome the presumption of validity enjoyed by issued patents. Even if we are successful in these proceedings, we may incur substantial costs and divert management’s time and attention in pursuing these proceedings, which could have a material adverse effect on our business. If we are unable to avoid infringing the patent rights of others, we may be required to seek a license, defend an infringement action or challenge the validity or enforceability of the patents in court. We may not have sufficient resources to bring these actions to a successful conclusion and there is no assurance that such a license would be available or that a court would find in our favor. In addition, if we do not obtain a license, do not develop or obtain non-infringing technology, or fail to defend an infringement action successfully or have infringed patents declared invalid or unenforceable, we may incur substantial monetary damages, encounter significant delays in bringing our product candidates to market and be precluded from manufacturing or selling our product candidates.
The cost of any patent litigation or other proceeding, even if resolved in our favor, could be substantial. Some of our competitors may be able to sustain the cost of such litigation and proceedings more effectively than we can because of their substantially greater resources. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have a material adverse effect on our business, financial condition, results of operations or prospects.
We are dependent on third-parties for the prosecution, protection, and enforcement of intellectual property rights relating to some of our products and product candidates.
While we normally seek to obtain the right to control the prosecution, maintenance, enforcement and defense of intellectual property rights related to our products and product candidates, there may be times when our licensors or collaborators control, or have a first right to control, the filing, prosecution, enforcement and defense of such rights. For instance, pursuant to the 2nd
36
amended and restated collaboration and license agreement with Novartis Pharma AG, or Novartis, Novartis has a first right to file, prosecute and enforce all patent rights related to products generated under this agreement. We cannot be certain that our licensors or collaborators will prosecute, maintain, enforce and defend such intellectual property rights in a manner consistent with the best interests of our business, including by taking reasonable measures to protect the confidentiality of know-how and trade secrets, or the payment of all applicable prosecution and maintenance fees related to our technologies or any of our product candidates. We also cannot be certain that the drafting or prosecution of the licensed patents by our licensors have been conducted accurately and in compliance with applicable laws and regulations, and will result in valid and enforceable patents and other intellectual property rights. If they fail to do so, we could lose our rights to the intellectual property, our ability to develop and commercialize those products or product candidates may be adversely affected and we may not be able to prevent competitors from making, using and selling competing products.
If trademarks and trade names related to our products or product candidates are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be materially adversely affected.
Our registered or unregistered trademarks or trade names, as well as the registered or unregistered trademarks or trade names used by our licensees or distributors in relation with our products or product candidates, may be challenged, infringed, circumvented or declared generic or determined to be infringing on other trademarks. We may not be able to protect our rights to these trademarks and trade names, which we need to build name recognition by potential partners or customers. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be materially adversely affected.
If we are unable to protect the confidentiality of our proprietary information, the value of our technology and products could be materially adversely affected.
In addition to patent protection, we also rely on other proprietary rights, including protection of trade secrets, and other proprietary information. To maintain the confidentiality of trade secrets and proprietary information, we enter into confidentiality agreements with our employees, consultants, collaborators, CMOs, CROs and others upon the commencement of their relationships with us. These agreements require that all confidential information developed by the individual or made known to the individual by us during the course of the individual’s relationship with us be kept confidential and not disclosed to third-parties. Our agreements with employees as well as our personnel policies also generally provide that any inventions conceived by the individual in the course of rendering services to us shall be our exclusive property or that we may obtain full rights to such inventions at our election. However, we may not obtain these agreements in all circumstances, and individuals with whom we have these agreements may not comply with their terms. We also face the risk that present or former employees could continue to hold rights to intellectual property used by us, may demand the registration of intellectual property rights in their name and demand damages pursuant to the German Employee Invention Act. In the event of unauthorized use or disclosure of our trade secrets or proprietary information, these agreements, even if obtained, may not provide meaningful protection, particularly for our trade secrets or other confidential information. To the extent that our employees, consultants or contractors use technology or know-how owned by third-parties in their work for us, disputes may arise between us and those third-parties as to the rights in related inventions. To the extent that an individual who is not obligated to assign rights in intellectual property to us is rightfully an inventor of intellectual property, we may need to obtain an assignment or a license to that intellectual property from that individual, or a third-party or from that individual’s assignee. Such assignment or license may not be available on commercially reasonable terms or at all.
Adequate remedies may not exist in the event of unauthorized use or disclosure of our proprietary information. The disclosure of our trade secrets would impair our competitive position and may materially harm our business, financial condition and results of operations. Costly and time-consuming litigation could be necessary to enforce and determine the scope of our proprietary rights, and failure to maintain trade secret protection could adversely affect our competitive business position. In addition, others may independently discover or develop our trade secrets and proprietary information, and the existence of our own trade secrets affords no protection against such independent discovery.
As is common in the biotechnology and pharmaceutical industries, we employ individuals who were previously or concurrently employed at research institutions and/or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. We may be subject to claims that these employees, or we, have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers, or that patents and applications we have filed to protect inventions of these employees, even those related to one or more of our product candidates, are rightfully owned by their former or concurrent employer. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
37
We may not be successful in obtaining necessary intellectual property rights to product candidates for our development pipeline through acquisitions and in-licenses.
We may seek to acquire or in-license product candidates to expand our product candidate pipeline. However, we may be unable to acquire or in-license intellectual property rights relating to, or necessary for, any such product candidates from third-parties on commercially reasonable terms or at all. In that event, we may be unable to develop or commercialize such product candidates. We may also be unable to identify product candidates that we believe are an appropriate strategic fit for our company and intellectual property relating to, or necessary for, such product candidates.
The in-licensing and acquisition of third-party intellectual property rights for product candidates is a competitive area, and a number of more established companies are also pursuing strategies to in-license or acquire third-party intellectual property rights for product candidates that we may consider attractive or necessary. These established companies may have a competitive advantage over us due to their size, cash resources and greater clinical development and commercialization capabilities. Furthermore, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. If we are unable to successfully obtain rights to suitable product candidates, our business, financial condition, results of operations and prospects for growth could suffer.
In addition, we expect that competition for the in-licensing or acquisition of third-party intellectual property rights for product candidates that are attractive to us may increase in the future, which may mean fewer suitable opportunities for us as well as higher acquisition or licensing costs. We may be unable to in-license or acquire third-party intellectual property rights for product candidates on terms that would allow us to make an appropriate return on our investment.
We may not be able to adequately protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on our products in all countries throughout the world would be prohibitively expensive. The requirements for patentability may differ in certain countries, particularly developing countries, and the breadth of patent claims allowed can be inconsistent. In addition, the laws of some foreign countries may not protect our intellectual property rights to the same extent as laws in the U.S. Consequently, we may not be able to prevent third-parties from practicing our inventions in all countries outside the U.S. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, furthermore, may export otherwise infringing products to territories in which we have patent protection that may not be sufficient to terminate infringing activities.
We do not have patent rights in certain foreign countries in which a market may exist. Moreover, in foreign jurisdictions where we do have patent rights, proceedings to enforce such rights could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly, and our patent applications at risk of not issuing. Additionally, such proceedings could provoke third-parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Thus, we may not be able to stop a competitor from marketing and selling in foreign countries products that are the same as or similar to our products, and our competitive position in the international market would be harmed.
Our intellectual property agreements with third-parties may be subject to disagreements over contract interpretation, which could narrow the scope of our rights to the relevant intellectual property or technology or increase our financial or other obligations to our licensors.
Certain provisions in our intellectual property agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could affect the scope of our rights to the relevant intellectual property or technology, or affect financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business, financial condition, results of operations and prospects.
In addition, while it is our policy to require our employees and contractors who may be involved in the conception or development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact conceives or develops intellectual property that we regard as our own. Our assignment agreements may not be self-executing or may be breached, and we may be forced to bring claims against third-parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property.
Risks Related to Employee Matters
Our future success depends on our ability to attract, motivate and retain key executives and qualified personnel.
As an innovation-driven and patient-centric company, we are highly dependent on the expertise of the members of our research and development team, as well as the other key functions such as commercial and supply to ensure that we can bring our
38
medicines to our patients with the highest quality and compliance with required standards. In addition, the members of our Management Board are key in developing our long-term strategy and steering all areas of the company. They currently include Jean-Paul Kress, M.D., our Chief Executive Officer, and Lucinda Crabtree, Ph.D., who was appointed as member of the Management Board and Chief Financial Officer as of August 8, 2023. Our Management Board members have fixed-term contracts typically of three years.
Recruiting and retaining qualified management, scientific, clinical, manufacturing, sales and marketing personnel is also critical to our success. The loss of the services of our executive officers or other key employees could impede the achievement of our research, development and commercialization objectives and seriously harm our ability to successfully implement our business strategy. Furthermore, replacing executive officers and key employees may be difficult and may take an extended period of time because of the limited number of individuals in our industry with the breadth of skills and experience required to successfully develop, gain regulatory approval of and commercialize drugs. Competition to hire from this limited pool is intense, and we may be unable to hire, train, retain or motivate these key personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. We also experience competition for the hiring of scientific and clinical personnel. Failure to succeed in clinical trials may make it more challenging to recruit and retain qualified scientific personnel.
Risks Related to Tax Matters
We may be subject to tax audits or disputes or changes in tax laws.
Pending and future tax audits within our group, disputes with tax authorities and changes in tax law or fiscal regulations could lead to additional tax liabilities. We are subject to routine tax audits by the respective local tax authorities. Any additional tax liability could have an adverse effect on our business, financial condition, results of operations or prospects.
The accounting treatment of the payment that MorphoSys AG received from Royalty Pharma in the third quarter of 2021 could be examined by the tax authorities under German tax law in the context of a future tax audit. This examination is considered standard given the amount of the payment. Based on the Company’s knowledge of German tax law and supported by tax experts, the Company has concluded that the tax risk assessment is medium in accordance with the Company's internal risk valuation system. Consequently, due to the remaining uncertainty and the significance, a contingent income tax liability in the amount of € 226.8 million is reported (refer to Note 6.2).
Risks Related to our Business and Industry
If we are unable to comply, or have not fully complied, with healthcare fraud and abuse, false claims, marketing expenditure tracking and disclosure, government price reporting, transparency and health-information privacy and security laws in our relationships with healthcare professionals, institutional providers, principal investigators, consultants, customers (actual and potential), patients and third-party payors, we could face penalties, damages, fines, exclusion from government-funded healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations.
Our relationships with healthcare professionals, institutional providers, principal investigators, consultants, customers (actual and potential), patients and third-party payors are, and will continue to be, subject, directly and indirectly, to healthcare fraud and abuse, false claims, marketing expenditure tracking and disclosure, government price reporting, transparency and information privacy and security laws. If we are unable to comply, or have not fully complied, with such laws, we could face penalties, including, without limitation, civil, criminal, and administrative penalties, damages, fines, exclusion from government-funded healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations. See the section of this annual report titled "Item 4.B. Business Overview Healthcare Law and Regulation."
Our business operations and activities may be directly or indirectly subject to various fraud and abuse laws, including, without limitation, the federal Anti-Kickback Statute and the federal False Claims Act. As we obtained U.S. FDA approval for one of our proprietary product candidates (which we sold to Incyte in February 2024) in 2020 and commercialized this product in the United States, our exposure under such laws increased significantly, and our costs associated with compliance with such laws increased. If we obtain U.S. FDA approval for proprietary product candidates and commercialize proprietary product candidates in the United States in the future, our potential exposure and the costs associated with compliance with such laws will significantly increase These laws may impact, among other things, our current activities with principal investigators and research subjects, as well as proposed and future sales, marketing and education programs. In addition, we may be subject to patient privacy regulation by the federal government and state governments in which we conduct our business.
In the U.S. third party patient assistance programs that receive financial support from companies have also become the subject of enhanced government and regulatory scrutiny. The OIG has established guidelines that suggest that it is lawful for pharmaceutical manufacturers to make donations to charitable organizations who provide co-pay assistance to Medicare
39
patients, provided that such organizations, among other things, are bona fide charities, are entirely independent of and not controlled by the manufacturer, provide aid to applicants on a first-come basis according to consistent financial criteria and do not link aid to use of a donor's product. However, donations to patient assistance programs have received some negative publicity and have been the subject of multiple government enforcement actions, related to allegations regarding their use to promote branded pharmaceutical products over other less costly alternatives. Specifically, in recent years, there have been multiple settlements resulting out of government claims challenging the legality of their patient assistance programs and it is possible that we may make grants to independent charitable foundations that help financially needy patients with their premium, co-pay and co-insurance obligations. If we choose to do so, and if we or our vendors or donation recipients are deemed to fail to comply with relevant laws, regulations or evolving government guidance in the operation of these programs, we could be subject to damages, fines, penalties or other criminal, civil or administrative sanctions or enforcement actions. We cannot ensure that our compliance controls, policies, and procedures will be sufficient to protect against acts of our employees, business partners, or vendors that may violate the laws or regulations of the jurisdictions in which we operate. Regardless of whether we have complied with the law, a government investigation could impact our business practices, harm our reputation, divert the attention of management, increase our expenses and reduce the availability of foundation support for our patients who need assistance.
Some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government in addition to requiring manufacturers to report information related to payments to physicians and other healthcare providers or marketing expenditures and pricing information. State and foreign laws also govern the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts.
The scope and enforcement of each of these laws is uncertain and subject to rapid change in the current environment of healthcare reform, especially in light of the lack of applicable precedent and regulations. Federal and state enforcement bodies have recently increased their scrutiny of interactions between healthcare companies and healthcare providers, which has led to a number of investigations, prosecutions, convictions and settlements in the healthcare industry.
Ensuring that our internal operations and future business arrangements with third parties comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations, agency guidance or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of the laws described above or any other governmental laws and regulations that may apply to us, we may be subject to significant penalties, including administrative, civil and criminal penalties, damages, fines, disgorgement, the exclusion from participation in federal and state healthcare programs, individual imprisonment, reputational harm, and the curtailment or restructuring of our operations, as well as additional reporting obligations and oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of non-compliance with these laws. Further, defending against any such actions can be costly and time consuming, and may require significant financial and personnel resources. Therefore, even if we are successful in defending against any such actions that may be brought against us, our business may be impaired. If any of the physicians or other providers or entities with whom we expect to do business are found to not be in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs and imprisonment. If any of the above occur, our ability to operate our business and our results of operations could be adversely affected.
In addition, the regulatory approval and commercialization of any of our product candidates outside the United States will also likely subject us to foreign equivalents of the information privacy laws mentioned above, among other foreign laws.
The collection, processing, storing, sharing, use, disclosure and protection of personally identifiable information are subject to federal, state, local and foreign laws. The scope of these laws is changing, they are subject to differing interpretations and they may be costly to comply with and may be inconsistent between countries and jurisdictions or conflict with other rules. Numerous jurisdictions are currently considering, or have recently enacted, data protection legislation. In addition, many states in which we operate have laws that protect the privacy and security of sensitive and personally identifiable information (“personal information”). In the United States, certain state laws may be more stringent or broader in scope, or offer greater individual rights, with respect to sensitive and personal information than federal or other state laws, and such laws may differ from each other, which may complicate compliance efforts.
Globally, we are also subject to stringent privacy and data protection requirements, such as the General Data Protection Regulation (“GDPR”). The GDPR, effective since May 2018, imposes strict regulations regarding the collection, storage and all other processing of personal data including special protections for “sensitive information” which includes health and genetic information of data processes in the EU. The GDPR also imposes strict rules on the transfer of personal data out of the EEA to the United States or other regions that have not been deemed to offer “adequate” privacy protections. Compliance with the GDPR will be a rigorous and time-intensive process that may increase our cost of doing business or require us to change our business practices, and despite those efforts, there is a risk that we may be subject to fines and penalties, litigation, and
40
reputational harm in connection with any future European activities. We could be adversely affected if we fail to comply fully with all of these requirements. Non-compliance with the GDPR can trigger significant fines of up to € 20 million or 4% of total worldwide annual turnover, whichever is higher.
In addition, further to the United Kingdom’s (UK) exit from the EU on January 31, 2020, the GDPR ceased to apply in the UK at the end of the transition period on December 31, 2020. However, as of January 1, 2021, the UK’s European Union (Withdrawal) Act 2018 incorporated the GDPR (as it existed on December 31, 2020 but subject to certain UK specific amendments) into UK law (referred to as the ‘UK GDPR’). The UK GDPR and the UK Data Protection Act 2018 set out the UK’s data protection regime, which is independent from but aligned to the EU’s data protection regime. Non-compliance with the UK GDPR may result in monetary penalties of up to £ 17.5 million or 4% of worldwide revenue, whichever is higher. Although the UK is regarded as a third country under the EU’s GDPR, the European Commission (“EC”) has now issued a decision recognizing the UK as providing adequate protection under the EU GDPR and, therefore, transfers of personal data originating in the EU to the UK remain unrestricted. Like the EU GDPR, the UK GDPR restricts personal data transfers outside the UK to countries not regarded by the UK as providing adequate protection. The UK government has confirmed that personal data transfers from the UK to the EEA remain free flowing.
Significantly, to enable the transfer of personal data outside of the EEA or the UK, adequate safeguards must be implemented in compliance with EEA and UK data protection laws. On June 4, 2021, the European Commission issued new forms of standard contractual clauses for data transfers from controllers or processors in the EU/EEA (or otherwise subject to the GDPR) to controllers or processors established outside the EU/EEA (and not subject to the GDPR). The new standard contractual clauses replace the standard contractual clauses that were adopted previously under the EU Data Protection Directive. The UK is not subject to the European Commission’s new standard contractual clauses but has issued a new version of a UK-specific transfer mechanism (i.e. the UK International Data Transfer Agreement or the UK International Transfer Addendum to accompany the European Commission’s standard contractual clauses), to enable transfers from the UK. We will be required to implement these new safeguards when conducting restricted data transfers under the EU and UK GDPR and doing so will require significant effort and cost.
There may be further divergence between the EEA and the UK. The United Kingdom has announced plans to reform the country’s data protection legal framework in its Data Reform Bill, which will introduce significant changes from the EU GDPR. This may lead to additional compliance costs and could increase our overall risk exposure as we may no longer be able to take a unified approach across the EEA and the UK, and we will need to amend our processes and procedures to align with the new framework.
In the United States, there has been considerable legislative activity at the state level. California recently enacted the California Consumer Privacy Act, or CCPA, which creates new individual privacy rights for California consumers (as defined in the law) and places increased privacy and security obligations on entities handling personal data of consumers or households. The CCPA requires covered companies to provide certain disclosures to consumers about its data collection, use and sharing practices, and to provide affected California residents with ways to opt-out of certain sales or transfers of personal information. The CCPA went into effect on January 1, 2020, and became enforceable by the California Attorney General on July 1, 2020. The CCPA provides for civil penalties for violations, as well as a private right of action for certain data breaches that result in the loss of personal information. This private right of action may increase the likelihood of, and risks associated with, data breach litigation. While there is currently an exception for protected health information that is subject to the Health Insurance Portability and Accountability Act of 1996, or HIPAA, and clinical trial regulations, as currently written, the CCPA may impact our business activities. There continues to be uncertainty surrounding the enforcement and implementation of the CCPA exemplifying the vulnerability of our business to the evolving regulatory environment related to personal data and protected health information.
Additionally, a California ballot initiative, the California Privacy Rights Act, or “CPRA,” was passed in November 2020 and became effective on January 1, 2023. The CPRA imposes additional obligations on companies covered by the legislation and will significantly modify the CCPA, including by expanding consumers’ rights with respect to certain sensitive personal information. The CPRA also creates a new state agency that will be vested with authority to implement and enforce the CCPA, as modified by the CPRA.
While we are currently not subject to the CCPA and will likely not fall under the purview of the CPRA, certain other state laws impose similar privacy obligations. Colorado, Connecticut, Utah and Virginia all passed laws that are similar to the CCPA and we also expect anticipate that more states may enact legislation similar to the CCPA. The CCPA has prompted a number of proposals for new federal and state-level privacy legislation which, if enacted, may add additional complexity, variation in requirements, restrictions and potential legal risk, require additional investment of resources in compliance programs, impact strategies and the availability of previously useful data and could result in increased compliance costs and/or changes in business practices and policies.
41
Furthermore, the use and disclosure of personal health and other private information are subject to regulation in other jurisdictions in which we do business or expect to do business in the future.
Efforts to ensure that our business arrangements will comply with applicable information privacy laws may involve substantial costs. Various jurisdictions around the world continue to propose new laws that regulate the privacy and/or security of certain types of personal data. Complying with these laws, if enacted, would require significant resources and leave us vulnerable to possible fines and penalties if we are unable to comply. It is possible that governmental and enforcement authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law interpreting applicable fraud and abuse or other healthcare laws and regulations. If we are unable to comply, or have not fully complied, with such laws, we could face penalties, including, without limitation, civil, criminal, and administrative penalties, damages, fines, individual imprisonment, exclusion from government-funded healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations. Additionally, if our collaborators’ operations or relationships with healthcare providers, customers, patients and third-party payors are found to be non-compliant with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government-funded healthcare programs, which could also have a negative impact on us. Even if successful, defending against any such actions can be costly, time-consuming and may require significant financial and personnel resources. We may become exposed to costly and damaging liability claims, either when testing our product candidates in the clinic or at the commercial stage; and our product liability insurance may not cover all damages from such claims.
We are exposed to potential product liability and professional indemnity risks that are inherent in the research, development, manufacturing, marketing and use of pharmaceutical products.
The use of our investigational medicinal products in clinical trials and the sale of any approved products in the future may expose us to liability claims. These claims might be made by patients who use the product, healthcare providers, pharmaceutical companies or others selling such products. Any claims against us, regardless of their merit, could be difficult and costly to defend and could materially adversely affect the market for our product candidates or any prospects for commercialization of our product candidates.
Although the clinical trial process is designed to identify and assess potential side effects, it is always possible that a product, even after regulatory approval, may exhibit unforeseen side effects. If any of our product candidates were to cause adverse side effects during clinical trials or after approval of the product candidate, we may be exposed to substantial liabilities. Physicians and patients may not comply with any warnings that identify known potential adverse effects and patients who should not use our product candidates.
To cover such liability claims, we purchase clinical trial insurances in the conduct of each of our clinical trials. It is possible that our liabilities could exceed our insurance coverage or that our insurance will not cover all situations in which a claim against us could be made. We expanded our insurance coverage to include the sale of commercial products once we receive marketing approval for any of our proprietary products. However, we may not be able to maintain insurance coverage at a reasonable cost or obtain insurance coverage that will be adequate to satisfy any liability that may arise. If a successful product liability claim or series of claims is brought against us for uninsured liabilities or in excess of insured liabilities, our assets may not be sufficient to cover such claims and our business operations could be impaired. Should any of the events described above occur, this could have a material adverse effect on our business, prospects, financial condition and results of operations, including, but not limited to:
•decreased demand for our future product candidates;
•adverse publicity and injury to our reputation;
•withdrawal of clinical trial participants;
•initiation of investigations by regulators;
•costs to defend the related litigation;
•a diversion of management’s time and our resources;
•compensation in response to a liability claim;
•product recalls, withdrawals or labeling, marketing or promotional restrictions;
•loss of revenue;
•exhaustion of any available insurance and our capital resources; and
•the inability to commercialize our products or product candidates.
42
We could be adversely affected if we are subject to negative publicity. We could also be adversely affected if any of our products or any similar products distributed by other companies prove to be, or are asserted to be, harmful to patients. Any adverse publicity associated with illness or other adverse effects resulting from patients’ use or misuse of our products or any similar products distributed by other companies could have a material adverse impact on our business, financial condition, results of operations or prospects.
Price controls may be imposed in certain markets, which may adversely affect our future profitability.
In some countries, particularly Member States of the European Union, the pricing of prescription medicinal products is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after receipt of marketing approval for a product. In addition, there can be considerable pressure by governments and other stakeholders on prices and reimbursement levels, including as part of cost containment measures. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference pricing used by various countries and parallel distribution, or arbitrage between low-priced and high-priced countries, can further reduce prices. In some countries, in particular, in many Member States of the European Union, we may be required to conduct a clinical trial or other studies that compare the cost-effectiveness of our product candidates to other available therapies in order to obtain or maintain reimbursement or pricing approval. Publication of discounts by third-party payors or authorities may lead to further pressure on the prices or reimbursement levels within the country of publication and other countries. If reimbursement of our products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business, financial condition, results of operations or prospects could be materially adversely affected.
Current and future legislation may increase the difficulty and cost for us and any collaborators to obtain marketing approval of and commercialize our product candidates and affect the prices we, or they, may obtain.
In the United States and foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of our product candidates, restrict or regulate post-approval activities and affect our ability to profitably sell any product candidates for which we obtain marketing approval. We expect that current laws, as well as other healthcare reform measures that may be adopted in the future, may result in additional reductions in Medicare and other healthcare funding, more rigorous coverage criteria, new payment methodologies and in additional downward pressure on the price that we, or any collaborators, may receive for any approved products. See the section of this annual report titled "Item 4.B. Business Overview Healthcare Reform."
The costs of prescription pharmaceuticals in the United States has also been the subject of considerable discussion in the United States, and members of Congress and the Administration have stated that they will address such costs through new legislative and administrative measures. There have been several U.S. congressional inquiries, Administrative actions, and proposed bills designed to, among other things, bring more transparency to drug pricing, reduce the cost of prescription drugs under Medicare, review the relationship between pricing and manufacturer patient support programs, and reform government program reimbursement methodologies for drugs. Further, various regulatory proposals and policies have been issued to address the reimportation of drugs. For example, in October 2020, the U.S. FDA issued a final guidance document outlining a pathway for manufacturers to obtain an additional National Drug Code, or NDC, for an U.S. FDA-approved drug or biological product that was originally intended to be marketed in a foreign country and that was authorized for sale in that foreign country. Individual states have also become increasingly active in passing legislation and implementing regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access, and to encourage importation from other countries and bulk purchasing. Although a number of these and other proposed measures may require authorization through additional legislation to become effective, and the Biden administration may reverse or otherwise change these measures, Congress has indicated that it will continue to seek new legislative measures to control drug costs. To the extent that products that MorphoSys commercializes are subject to the legislative, regulatory, or other measures that promote or allow the reimportation of drugs the prices we receive for our products could decreases, which would adversely affect our future revenues and prospects for profitability.
In addition, individual states have also become increasingly active in passing legislation and implementing regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access, and to encourage importation from other countries and bulk purchasing.
The policies of the U.S. FDA or similar regulatory authorities may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. If we or our collaborators are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we or our collaborators are not able to maintain regulatory compliance, our product candidates may lose any regulatory approval that may have been obtained and we may not achieve or sustain profitability, which would adversely affect our business.
43
We cannot predict whether future healthcare legislative or policy changes will be implemented at the federal or state level or in countries outside of the United States in which we may do business, or the effect any future legislation or regulation will have on us.
Additionally, in the case of any United States federal government shutdown, now or in the future, that continued for a prolonged period of time, processes related to coverage and reimbursement determinations could be delayed. Resolving such delays could force us or our collaborators to incur significant costs, could limit our allowed activities or the allowed activities of our collaborators, could diminish any competitive advantages that we or our collaborators may attain or could adversely affect our business, financial condition, results of operations and prospects, the value of our common stock and our ability to bring new products to market as forecasted. Even without such delay, there is no guarantee we will receive approval or reimbursement for our product candidates on a timely basis, or at all.
If we fail to comply with our reporting and payment obligations under the Medicaid Drug Rebate Program or other governmental pricing programs, we could be subject to additional reimbursement requirements, penalties, sanctions and fines, which could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
We participate in the Medicaid Drug Rebate Program, the 340B program, the U.S. Department of Veterans Affairs, Federal Supply Schedule, or FSS, pricing program, and the Tricare Retail Pharmacy program, and have obligations to report the average sales price for certain of our drugs to the Medicare program. For calendar quarters beginning January 1, 2022, manufacturers will need to start reporting the average sales price for drugs under the Medicare program regardless of whether they are enrolled in the Medicaid Drug Rebate Program. Currently, only manufacturers participating in the Medicaid Drug Rebate Program are obligated to do so.
Pricing and rebate calculations vary across products and programs, are complex, and are often subject to interpretation by us, governmental or regulatory agencies and the courts, which can change and evolve over time. In the case of our Medicaid pricing data, if we become aware that our reporting for a prior quarter was incorrect, or has changed as a result of recalculation of the pricing data, we are generally obligated to resubmit the corrected data for up to three years after those data originally were due. Such restatements and recalculations increase our costs for complying with the laws and regulations governing the Medicaid Drug Rebate Program and could result in an overage or underage in our rebate liability for past quarters. Price recalculations also may affect the ceiling price at which we are required to offer our products under the 340B program and give rise to an obligation to refund entities participating in the 340B program for overcharges during past quarters impacted by a price recalculation.
Civil monetary penalties can be applied if we are found to have knowingly submitted any false price or product information to the government, if we are found to have made a misrepresentation in the reporting of our average sales price, if we fail to submit the required price data on a timely basis, or if we are found to have charged 340B covered entities more than the statutorily mandated ceiling price. The Centers for Medicare & Medicaid Services, or CMS, could also decide to terminate our Medicaid drug rebate agreement, in which case federal payments may not be available under Medicaid or Medicare Part B for our covered outpatient drugs. We cannot assure you that our submissions will not be found by CMS to be incomplete or incorrect.
Our failure to comply with our reporting and payment obligations under the Medicaid Drug Rebate Program and other governmental programs could negatively impact our financial results. CMS issued a final regulation, which became effective in April 2016, to implement the changes to the Medicaid Drug Rebate Program under the Affordable Care Act. In December 2020, CMS issued a final regulation that modified prior Medicaid Drug Rebate Program regulations to permit reporting multiple best price figures with regard to value-based purchasing arrangements (beginning in 2022); and provided definitions for “line extension,” “new formulation,” and related terms, with the practical effect of expanding the scope of drugs considered to be line extensions that are subject to an alternative rebate formula (beginning in 2022). Regulatory and legislative changes, and judicial rulings relating to the Medicaid Drug Rebate Program and related policies (including coverage expansion), have increased and will continue to increase our costs and the complexity of compliance, have been and will continue to be time-consuming to implement, and could have a material adverse effect on our results of operations, particularly if CMS or another agency challenges the approach we take in our implementation.
The HRSA issued a final regulation regarding the calculation of the 340B ceiling price and the imposition of civil monetary penalties on manufacturers that knowingly and intentionally overcharge covered entities, which became effective in January 2019. Implementation of this regulation could affect our obligations and potential liability under the 340B program in ways we cannot anticipate. We are also required to report the 340B ceiling prices for our covered outpatient drugs to HRSA, which then publishes them to 340B covered entities. Any charge by HRSA that we have violated this regulation or other requirements of the program could negatively impact our financial results. Moreover, HRSA newly established an administrative dispute resolution, or ADR, process under a final regulation effective January 2021, for claims by covered entities that a manufacturer
44
engaged in overcharging, including claims that a manufacturer limited the ability of a covered entity to purchase the manufacturer’s drugs at the 340B ceiling price, and by manufacturers that a covered entity violated the prohibitions against diversion or duplicate discounts. Such claims are to be resolved through an ADR panel of government officials rendering a decision that could be appealed only in federal court. This ADR regulation has been challenged in separate litigation instituted by PhRMA and by pharmaceutical manufacturers in multiple federal courts. Under the ADR final rule which became effective in January 2021, an ADR proceeding could potentially subject us to discovery by covered entities and other onerous procedural requirements and could result in additional liability. HRSA could also decide to terminate a manufacturer’s agreement to participate in the 340B program for a violation of that agreement or other good cause shown, in which case the manufacturer’s covered outpatient drugs may no longer be eligible for federal payment under the Medicaid or Medicare Part B program. In November 2022, HRSA issued a proposed rule to revise the ADR procedures contained in its January 2021 final regulation for disputes arising under the 340B drug pricing program between covered entities and manufacturers.
Further, legislation may be introduced that, if passed, would, among other things, further expand the 340B program to additional covered entities or would require participating manufacturers to agree to provide 340B discounted pricing on drugs used in an inpatient setting, and any additional future changes to the definition of average manufacturer price or the Medicaid rebate amount could affect our 340B ceiling price calculations and negatively impact our results of operations. Additionally, certain pharmaceutical manufacturers are involved in ongoing litigation regarding contract pharmacy arrangements under the 340B Program. The outcome of those judicial proceedings and the potential impact on the way in which manufacturers extend discounts to covered entities through contract pharmacies remain uncertain.
We have obligations to report the average sales price for certain of our drugs to the Medicare program. Statutory or regulatory changes or CMS guidance could affect the average sales price calculations for our products and the resulting Medicare payment rate, and could negatively impact our results of operations.
Pursuant to applicable law, knowing provision of false information in connection with price reporting under the U.S. Department of Veterans Affairs, FSS or Tricare Retail Pharmacy, or Tricare, programs can subject a manufacturer to civil monetary penalties. These program obligations also contain extensive disclosure and certification requirements. If we overcharge the government in connection with our arrangements with FSS or Tricare, we are required to refund the difference to the government. Failure to make necessary disclosures and/or to identify contract overcharges can result in allegations against us under the False Claims Act and other laws and regulations. Unexpected refunds to the government, and responding to a government investigation or enforcement action, would be expensive and time-consuming, and could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
We and our contract manufacturers and our suppliers could be subject to liabilities, fines, penalties or other sanctions under environmental, health and safety laws and regulations if we or they fail to comply with such laws or regulations or otherwise incur costs that could have a material adverse effect on our business.
We currently rely on and expect to continue to rely on third-parties for the manufacturing and supply of active pharmaceutical ingredients, or API, and drug products of our product and product candidates. These third-parties are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, transportation, use, storage, treatment and disposal of hazardous materials and wastes. Although we have auditing rights and obligations with all our CMOs for production of API and drug products and finished drug product, we do not have control over a manufacturer’s or supplier’s compliance with environmental, health and safety laws and regulations. Liabilities they incur pursuant to these laws and regulations could result in significant costs or in certain circumstances, an interruption in operations, any of which could adversely affect our business and financial condition if delayed manufacturing activities impact our clinical development activities and/or our commercial supply chain.
With respect to any hazardous materials or waste which we are currently, or in the future will be, handling, using, storing or disposing of, we cannot eliminate the risk of contamination or injury from these materials or waste, including at third-party disposal sites. In the event of such contamination or injury, we could be held liable for any resulting damages and liability. We also could incur significant costs associated with civil or criminal fines and penalties for failure to comply with applicable environmental, health and safety laws. In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair our research, development or production efforts. Failure to comply with these laws and regulations may also result in substantial fines, penalties or other sanctions.
45
Our internal computer systems and physical premises, or those of our strategic collaborators or other contractors or consultants, may fail or suffer security breaches, which could result in a material disruption of our product development programs and our manufacturing operations.
Our internal computer systems and those of our current and any future strategic collaborators, vendors, and other contractors or consultants are vulnerable to damage from cyber-attacks, computer viruses, unauthorized access, natural disasters, cybersecurity threats, terrorism, war and telecommunication and electrical failures. Cyber incidents have been increasing in sophistication and frequency and can include third parties gaining access to our data using stolen or inferred credentials, computer malware, viruses, spamming, phishing attacks, ransomware, card skimming code, and other deliberate attacks and attempts to gain unauthorized access. Because the techniques used by computer programmers who may attempt to penetrate and sabotage our network security or our website change frequently and may not be recognized until launched against a target, we may be unable to anticipate these techniques.
While we have not experienced any material computer system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations, whether due to a loss of our trade secrets or other proprietary information or other disruptions. These cyber-attacks could be carried out by threat actors of all types (including but not limited to nation states, organized crime, other criminal enterprises, individual actors and/or advanced persistent threat groups). In addition, we may experience intrusions on our physical premises by any of these threat actors. For example, the loss of clinical trial data from completed or future clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability, our competitive position could be harmed and the further development and commercialization of our product candidates could be delayed. Any breach, loss, or compromise of clinical trial participant personal data may also subject us to civil fines and penalties, or claims for damages either under the GDPR and relevant member state law in the EU, other foreign laws, and the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, and other relevant state and federal privacy laws in the United States.
Our product and future product candidates for which we intend to seek approval as biologic products may face competition sooner than anticipated.
The ACA includes a subtitle called the Biologics Price Competition and Innovation Act of 2009, or BPCIA, which created an abbreviated approval pathway for biological products that are biosimilar to or interchangeable with an U.S. FDA-licensed reference biological product. Under the BPCIA, an application for a biosimilar product may not be submitted to the U.S. FDA until four years following the date that the referenced product was first licensed by the U.S. FDA. In addition, the approval of a biosimilar product may not be made effective by the U.S. FDA until 12 years from the date on which the reference product was first licensed. During this 12-year period of exclusivity, another company may still market a competing version of the reference product if the U.S. FDA approves a full BLA for the competing product containing the sponsor’s own preclinical data and data from adequate and well-controlled clinical trials to demonstrate the safety, purity and potency of their product. The law is complex and is still being interpreted and implemented by the U.S. FDA. On July 9, 2021, President Biden issued an executive order directing the U.S. FDA to, among other things, continue to clarify and improve the approval framework for biosimilars, including the standards for interchangeability of biological products, facilitate the development and approval of biosimilar and interchangeable products, clarify existing requirements and procedures related to the review and submission of BLAs, and identify and address any efforts to impede biosimilar competition. As a result, the ultimate impact, implementation, and meaning of the BPCIA are subject to uncertainty and evolving interpretation. While it is uncertain when such processes intended to implement BPCIA may be fully adopted by the U.S. FDA, any such processes could have a material adverse effect on the future commercial prospects for our biological products.
One or more of our product candidates approved as a biological product under a BLA may qualify for the 12-year period of exclusivity. However, there is a risk that this exclusivity could be shortened due to congressional action or otherwise, or that the U.S. FDA will not consider our product candidates to be reference products for competing products, potentially creating the opportunity for generic competition sooner than anticipated. Other aspects of the BPCIA, some of which may impact the BPCIA exclusivity provisions, have also been the subject of recent litigation. Moreover, the extent to which a biosimilar, once approved, will be substituted for any one of our reference products in a way that is similar to traditional generic substitution for non-biological products is not yet clear, and will depend on a number of marketplace and regulatory factors that are still developing.
We may acquire businesses or products, or form strategic alliances, in the future, and we may not realize the benefits of such acquisitions.
On July 15, 2021, MorphoSys completed its acquisition of Constellation, adding two clinical stage assets pelabresib and tulmimetostat to its pipeline. We may acquire additional businesses or products, form strategic alliances or create joint ventures
46
with third parties that we believe will complement or augment our existing business. We may encounter numerous difficulties in developing, manufacturing and marketing any new products resulting from a strategic alliance or acquisition, including the Constellation acquisition, that delays or prevents us from realizing their expected benefits or enhancing our business. When acquiring businesses with promising products or technologies, we may not be able to realize the benefit of acquiring such businesses if, for instance, we are unable to successfully integrate them with our existing operations and company culture. We cannot assure you that, following any such acquisition, including the Constellation acquisition, we will achieve the expected synergies to justify the transaction. If we are unsuccessful in realizing any of the benefits following an acquisition, we may incur impairment charges in respect of the assets acquired, which could adversely affect our results of operations.
We are subject to currency exchange rate fluctuations.
Due to the international scope of our operations, our assets, earnings and cash flows are influenced by movements in exchange rates of several currencies, particularly the U.S. dollar and the euro. Our functional currency is the euro and a substantial proportion of our revenues and operating expenses is paid in U.S. Dollars. We also receive payments from our collaboration partners in U.S. dollars and we regularly acquire services, consumables and materials in U.S. dollars. As a result, our business may be affected by fluctuations in foreign exchange rates between the euro and the U.S. dollar, which may also have a significant impact on our reported results of operations and cash flows from period to period.
Risks Related to Ownership of Our Securities
We do not currently intend to pay dividends on our securities, and, consequently, your ability to achieve a return on your investment will depend on appreciation in the price of our shares.
We have never declared or paid any dividends on our ordinary shares and do not intend to do so in the foreseeable future. You are not likely to receive any dividends on our shares, and the success of an investment in our shares will depend upon any future appreciation in its value. Investors may need to sell all or part of their holdings of our shares after price appreciation, which may never occur, to realize any future gains on their investment. There is no guarantee that our shares will appreciate in value or even maintain the price at which our shareholders have purchased our shares.
Holders of our ADSs may not be able to participate in any future preemptive subscription rights issues or to elect to receive dividends in shares, which may cause dilution to their holdings.
Under German law, the existing shareholders have a preemptive right to subscribe for shares offered in proportion to the number of shares they hold in connection with any offering of shares. However, a shareholders’ meeting may vote, by a majority, which represents at least three quarters of the share capital represented at the meeting, to waive this preemptive right provided that, from the company’s perspective, there exists good and objective cause for such waiver.
Certain non-German shareholders may not be able to exercise their preemptive subscription rights in our future offerings due to the legislation and regulations of their home country. For example, ADS holders in the United States will not be entitled to exercise or sell such rights unless we register the rights and the securities to which the rights relate under the Securities Act or an exemption from the registration requirements is available. In addition, the deposit agreement provides that the depositary need not make rights available to you unless the distribution to ADS holders of both the rights and any related securities are either registered under the Securities Act or exempted from registration under the Securities Act. We are under no obligation to file a registration statement with respect to any such rights or securities or to endeavor to cause such a registration statement to be declared effective. Moreover, we may not be able to establish an exemption from registration under the Securities Act. Accordingly, ADS holders may be unable to participate in our rights offerings and may experience dilution in their holdings. In addition, if the depositary is unable to sell rights that are not exercised or not distributed or if the sale is not lawful or reasonably practicable, it will allow the rights to lapse, in which case you will receive no value for these rights.
As a foreign private issuer, we are exempt from a number of rules under the U.S. securities laws and are permitted to file less information with the SEC than a U.S. company. This may limit the information available to holders of ADSs or our ordinary shares.
We are a “foreign private issuer,” as defined in the SEC’s rules and regulations and, consequently, we are not subject to all of the disclosure requirements applicable to public companies organized within the United States. For example, we are exempt from certain rules under the Exchange Act, that regulate disclosure obligations and procedural requirements related to the solicitation of proxies, consents or authorizations applicable to a security registered under the Exchange Act, including the U.S. proxy rules under Section 14 of the Exchange Act. In addition, our officers and directors are exempt from the reporting and “short-swing” profit recovery provisions of Section 16 of the Exchange Act and related rules with respect to their purchases and sales of our securities. Moreover, while we currently make annual and semi-annual filings with respect to our listing on the
47
Frankfurt Stock Exchange and report our results of operations on a quarterly basis, we are not required to file periodic reports and financial statements with the SEC as frequently or as promptly as U.S. domestic issuers and are not required to file quarterly reports on Form 10-Q or current reports on Form 8-K under the Exchange Act. Accordingly, there is and will continue to be less publicly available information concerning our company than there would be if we were not a foreign private issuer.
As a foreign private issuer, we are permitted to adopt certain home country practices in relation to corporate governance matters that differ significantly from Nasdaq corporate governance listing standards. These practices may afford less protection to shareholders than they would enjoy if we complied fully with corporate governance listing standards.
The Nasdaq Listing Rules include certain accommodations in the corporate governance requirements that allow foreign private issuers to follow “home country” corporate governance practices in lieu of the otherwise applicable corporate governance standards of Nasdaq. The application of such exceptions requires that we disclose the Nasdaq Listing Rules that we do not follow and describe the German corporate governance practices we do follow in lieu of the relevant Nasdaq corporate governance standard. We continue to follow German corporate governance practices in lieu of the corporate governance requirements of Nasdaq in certain respects. In particular, we follow German corporate governance practices in connection with the distribution of annual and interim reports to shareholders, the application of our code of conduct to our Supervisory Board, proxy solicitation in connection with shareholders’ meetings, and obtaining shareholder approval in connection with the issuance of shares in connection with an acquisition, change of control transactions, the establishment of or material amendment to any equity-based compensation plans and the issuance of shares in a private placement in excess of 20% of the outstanding share capital at less than the greater of book or market value. To this extent, our practice varies from the requirements of Nasdaq. In addition, as a German public company, certain capital structure decisions will require shareholder approval, which may limit our flexibility to manage our capital structure. See the sections of this annual report titled “Item 6 —Directors, Senior Management and Employees” and “Item 16G —Corporate Governance.”
U.S. holders of ADSs may suffer adverse tax consequences if we are characterized as a passive foreign investment company.
A non-U.S. corporation will be classified as a passive foreign investment company, or PFIC, for U.S. federal income tax purposes for any taxable year, if either (i) 75% or more of its gross income for such year consists of certain types of “passive” income or (ii) 50% or more of the value of its assets (determined on the basis of a quarterly average) during such year produce or are held for the production of passive income. Passive income generally includes dividends, interest, royalties, rents, annuities, net gains from the sale or exchange of property producing such income and net foreign currency gains. In addition, a non-U.S. corporation will be treated as owning its proportionate share of the assets and earning its proportionate share of the income of any other corporation in which it owns, directly or indirectly, more than 25% (by value) of the stock.
The determination of whether we are a PFIC is a fact-intensive determination that must be made on an annual basis applying principles and methodologies that are in some circumstances unclear. Based on the composition of our income and valuation of our assets, we do not believe that we should be treated as a PFIC for the 2023 taxable year. However, a separate determination must be made after the close of each taxable year as to whether we are a PFIC for that year. As a result, our PFIC status may change in a future taxable year. In particular, the total value of our assets for purposes of the asset test generally will be calculated taking into account the market price of our ADSs or ordinary shares, which may fluctuate considerably. Fluctuations in the market price of the ADSs and ordinary shares may result in our being a PFIC for any taxable year. Even if we determine that we are not a PFIC for a taxable year, there can be no assurance that the U.S. Internal Revenue Services ("IRS") or courts will agree with our conclusion and that the IRS would not successfully challenge our position.
If we were to be or become a PFIC for any taxable year during which a U.S. holder (defined below in “Taxation—U.S. Taxation”) holds ADSs, certain adverse U.S. federal income tax consequences could apply to such U.S. holder. See the section of this annual report titled “Item 10 E. Taxation—U.S. Taxation—PFIC Rules.”
We may lose our foreign private issuer status in the future, which could result in significant additional cost and expense.
While we currently qualify as a foreign private issuer, the determination of foreign private issuer status is made annually on the last business day of an issuer’s most recently completed second fiscal quarter and, accordingly, the next determination will be made with respect to us on June 30, 2024.
In the future, we would lose our foreign private issuer status if we fail to meet the requirements necessary to maintain our foreign private issuer status as of the relevant determination date. In order to maintain our current status as a foreign private issuer, either (a) a majority of our ordinary shares are either directly or indirectly owned of record by non-residents of the United States or (b), (i) a majority of our executive officers or directors cannot be U.S. citizens or residents, (ii) more than 50% of our assets cannot be located in the United States and (iii) our business must be administered principally outside the United States.
48
The regulatory and compliance costs to us under U.S. securities laws as a U.S. domestic issuer may be significantly higher than the costs we incur as a foreign private issuer. If we are not a foreign private issuer, we will be required to file periodic reports and registration statements on U.S. domestic issuer forms with the SEC, which are more detailed and extensive in certain respects than the forms available to a foreign private issuer. We would be required under current SEC rules to prepare our financial statements in accordance with U.S. GAAP rather than IFRS. Such conversion of our financial statements to U.S. GAAP would involve significant time and cost, and we would still be required to prepare financial statements in accordance with IFRS under the rules of the Frankfurt Stock Exchange. In addition, we may lose our ability to rely upon exemptions from certain corporate governance requirements on United States stock exchanges that are available to foreign private issuers such as the ones described above and exemptions from procedural requirements related to the solicitation of proxies.
We will continue to incur increased costs as result of being a public company.
As a public company with ADSs listed on the Nasdaq Global Market, we incur significant legal, accounting and other expenses that we did not incur as a private company. The Sarbanes-Oxley Act of 2002, as well as rules subsequently implemented by the SEC and the Nasdaq Global Market, impose various requirements on the corporate governance practices of public companies. These and other rules and requirements may increase or change, resulting in an increase of our legal and financial compliance costs. Operating as a public company also makes it more difficult and more expensive for us to obtain director and officer liability insurance, and we may be required to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. It may also be more difficult for us to attract qualified persons to serve on our board of directors or as executive officers.
U.S. investors may have difficulty enforcing civil liabilities against our company and members of our Supervisory Board and Management Board and the experts named in this report.
We are incorporated under the laws of Germany. The majority of our assets are located outside the United States and currently one of the two members of our Management Board and three out of six Supervisory Board members reside outside of the United States. As a result, effecting service of process upon such persons may require compliance with international treaty procedures that could cause delay and in some case interfere with establishing personal jurisdiction in front of U.S. courts. The United States and Germany do not currently have a treaty providing for reciprocal recognition and enforceability of judgments rendered in connection with civil and commercial disputes and, accordingly, a final judgment rendered by a U.S. court based on civil liability would not automatically be recognized or enforceable in Germany. Therefore enforcing against members of our Management Board or Supervisory Board or against us, judgments obtained in U.S. courts’ that are predicated upon the civil liability provisions of the U.S. federal securities laws may be impossible under German law as a result of public policy or jurisprudence providing defenses for German nationals. Foreign courts may refuse to consider claims brought under U.S. securities laws on either procedural grounds or substantive grounds. Even if a foreign court is willing to decide the merits of such a claim, it may decide to apply the law of the jurisdiction in which the foreign court is located, rather than U.S. law.
Further, if a foreign court applies U.S. law, the burden of proving applicable U.S. law will fall on the party making the claims, a process that may be time-consuming and costly. Procedural matters are typically governed by the law of the jurisdiction in which the foreign court is located.
The rights of shareholders in a stock corporation subject to German law differ in material respects from the rights of shareholders of corporations incorporated in the United States.
We are a German stock corporation with our registered office in Germany. Our corporate affairs are governed by the laws governing stock corporations incorporated in Germany and our articles of association. The rights of shareholders and the responsibilities of members of our Management Board (Vorstand) and Supervisory Board (Aufsichtsrat) may be different from the rights and obligations of shareholders in companies governed by the laws of U.S. jurisdictions. In the performance of their duties, our Management Board and Supervisory Board may take into account a broad range of considerations, including our interests, the interests of our shareholders, employees, creditors and, to a limited extent, the general public. It is possible that some of these parties will have interests that are different from, or in addition to, your interests as a holder of ADSs. See the section of this annual report titled “Item 16G—Corporate Governance”.
If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results or prevent fraud. As a result, shareholders could lose confidence in our financial and other public reporting, which would harm our business and the trading price of our shares.
Effective internal controls over financial reporting are necessary for us to provide reliable financial reports and, together with adequate disclosure controls and procedures, are designed to prevent fraud. Any failure to implement required new or improved controls, or difficulties encountered in their implementation could cause us to fail to meet our reporting obligations. In addition,
49
any testing conducted by us in connection with Section 404 of the Sarbanes-Oxley Act of 2002, or any subsequent testing conducted by our independent registered public accounting firm, may reveal deficiencies in our internal controls over financial reporting that are deemed to be material weaknesses or that may require prospective or retroactive changes to our financial statements, or identify other areas for further attention or improvement. Inferior internal controls could also cause investors to lose confidence in our reported financial information, which could have a negative effect on the trading price of our shares.
Unfavorable U.S. or global economic conditions could adversely affect our business, financial condition, or results of operations.
Our results of operations could be adversely affected by general conditions in the global economy and financial markets, including by the recent global political and military events or any health epidemic. The most recent global financial crisis caused extreme volatility and disruptions in the capital and credit markets. A severe or prolonged economic downturn, such as the most recent global financial crisis, could result in a variety of risks to our business, including weakened demand for our product candidates and our ability to raise additional capital when needed on favorable terms, if at all. A weak or declining economy could strain our suppliers, possibly resulting in supply disruption, or cause delays in payments for our services by third-party payors or our collaborators. Any of the foregoing could harm our business and we cannot anticipate all of the ways in which the current economic climate and financial market conditions could adversely impact our business.
Item 4.Information on the Company.
A.History and Development of the company
MorphoSys AG was founded in 1992 in Martinsried, Germany, and was converted to a stock corporation on March 3, 1998 under the laws of Germany with an indefinite duration. Our legal and commercial name is MorphoSys AG. We were registered in the commercial register of the local court of Munich under number HRB 121023 on June 30, 1998. In 1999, MorphoSys was listed on the Frankfurt Stock Exchange, trading under the ticker symbol “MOR”. In 2014, MorphoSys joined the TecDAX index, and since September 2021, MorphoSys is part of the SDAX Index. In April 2018, following a U.S. initial public offering, American Depositary Shares of MorphoSys began trading on the Nasdaq, also under the symbol “MOR”. In July 2018, we established a wholly owned subsidiary, MorphoSys US Inc., to build our commercial infrastructure in the United States. MorphoSys US Inc. is the company’s agent in the United States and is located at 470 Atlantic Avenue, 14th Floor, Boston, Massachusetts 02210.
Our registered office is located at Semmelweisstrasse 7, 82152 Planegg, Germany, and our telephone number is +49 89-89927-0. Our website is www.morphosys.com. Information contained on our website is not incorporated by reference into this annual report, and you should not consider information contained on our website to be part of this annual report or in any other filings we make with the SEC, or in deciding whether to purchase or sell our ADSs. Material non-financial aspects are taken into account in a separate "Non-Financial Group Report," which is available on our website.
The SEC maintains an internet site at http://www.sec.gov that contains reports, information statements, and other information regarding issuers that file electronically with the SEC.
Subsequent Events: Business Combination with Novartis
On February 5, 2024 MorphoSys announced that it entered into a Business Combination Agreement with Novartis based on Novartis’ intention to submit the Novartis Takeover Offer for all of MorphoSys’ outstanding common shares in exchange for payment of € 68.00 per share in cash. Separately, MorphoSys entered into a Purchase Agreement with Incyte Corporation to sell and transfer all rights worldwide related to tafasitamab for $ 25.0 million. MorphoSys and Incyte have been collaborating on the development and commercialization of tafasitamab since 2020. Prior to this agreement, tafasitamab was co-marketed in the U.S. by MorphoSys and Incyte as Monjuvi® (tafasitamab-cxix) and outside the U.S. by Incyte as Minjuvi®. MorphoSys’ Management Board and Supervisory Board unanimously approved both agreements.
In the Business Combination Agreement, MorphoSys and Novartis agreed on the terms of the Novartis Takeover Offer.
Novartis intends to offer MorphoSys’ shareholders € 68.00 per share in cash, for a total equity value of € 2.7 billion. The offer price corresponds to a premium of 94% and 142% on the volume-weighted average price during the last month and three months as of the unaffected January 25, 2024 close, respectively – the day before rumors about a transaction first surfaced. It also represents a premium of 89% percent to the closing share price of January 25, 2024.
Subject to a careful review of the offer document to be published by Novartis, MorphoSys’ Management Board and Supervisory Board intend to recommend the acceptance of the Novartis Takeover Offer. The Novartis Takeover Offer will contain customary closing conditions, in particular a minimum acceptance threshold of 65% of MorphoSys’ share capital and regulatory clearances. The closing is currently expected to take place in the first half of 2024. MorphoSys and Novartis agreed
50
to take MorphoSys private promptly after the Novartis Takeover Offer has been settled. There is no assurance that the business combination will be consummated on the proposed terms, timing or at all. For risks associated with the Novartis Takeover Offer please refer to Item 3 Key Information—Risk Factors—Risks Related to the Novartis Takeover Offer.
The offer document of the Novartis Takeover Offer will be published by Novartis at a later date in accordance with the provisions of the German Securities Acquisition and Takeover Act, after the German Federal Financial Supervisory Authority (“BaFin”) has approved the publication. Promptly after the offer document is published, MorphoSys’ Management Board and Supervisory Board will issue a joint reasoned statement in accordance with sec. 27 of the German Securities Acquisition and Takeover Act. In accordance with U.S. securities laws, Novartis will file a Tender Offer Statement which will include the offer document on Schedule TO and MorphoSys will file a Solicitation/Recommendation Statement on Schedule with the U.S. Securities and Exchange Commission.
Principal Capital Expenditures:
In the years ended December 31, 2023, 2022 and 2021, our expenditures for property, plant and equipment were € 0.4 million, € 1.9 million, and € 4.3 million, respectively. In the years ended December 31, 2023, 2022 and 2021, our expenditures for intangible assets were € 2.5 million, € 13.3 million, and € 22.3 million, respectively.
In the course of the acquisition of Constellation in 2021, MorphoSys acquired € 719.4 million in intangible assets and € 1.6 million in property, plant and equipment.
For our commitments for capital expenditures, we refer to Item 5.B.
B.Business Overview
MorphoSys is a global biotechnology company focused on the development and commercialization of innovative cancer medicines.
In 2023, we advanced our mid-to-late-stage oncology pipeline, focusing our efforts on pelabresib, an investigational BET inhibitor, in first-line myelofibrosis – a debilitating and deadly blood cancer.
Our current clinical programs are:
•Pelabresib: An investigational selective small-molecule therapy aimed at promoting anti-tumor activity. It is designed to inhibit bromodomain and extra-terminal domain (BET) proteins, which may downregulate genes implicated in blood cancers. The Phase 3 MANIFEST-2 study is currently investigating pelabresib in combination with the JAK inhibitor ruxolitinib, the current standard of care, compared with placebo plus ruxolitinib in JAK inhibitor-naïve patients with myelofibrosis (those who have not been previously treated with a JAK inhibitor). Additionally, it is being explored in the Phase 2 MANIFEST trial in patients with myelofibrosis and high-risk essential thrombocythemia.
•Tulmimetostat: An investigational next-generation dual inhibitor of Enhancer of Zeste Homolog 2 (EZH2) and EZH1 designed to counteract abnormal gene activity that may contribute to cancer development and progression, as well as drug resistance. Tulmimetostat is being investigated in a Phase 1/2 study in patients with advanced solid tumors and lymphomas, including ARID1A-mutated endometrial carcinoma and ovarian clear cell carcinoma, diffuse large B-cell lymphoma, peripheral T-cell lymphoma, BAP1-mutated mesothelioma and metastatic castration-resistant prostate cancer.
51

Additionally, these are the most advanced clinical programs developed via MorphoSys’ legacy antibody technology platform by our partners:
•Ianalumab (VAY736) – a fully human IgG1/k antibody with a dual mode of action targeting B-cell lysis and BAFF-R blockade, developed by Novartis and being investigated in several Phase 3 studies for Sjögren’s disease, lupus nephritis, and other autoimmune diseases.
•Abelacimab (MAA868) – an antibody directed against Factor XI, developed by Anthos Therapeutics and being investigated in three Phase 3 studies for venous thromboembolism prevention and cancer-associated thrombosis.
•Setrusumab (BPS804/UX143) – an antibody directed against sclerostin, developed by Ultragenyx and Mereo BioPharma and being investigated in a pivotal Phase 2/3 study for osteogenesis imperfecta.
•Bimagrumab - an antibody binding to activin type II receptors, developed by Lilly and being investigated in a Phase 2b study for adult obesity.
•Felzartamab – a therapeutic human monoclonal antibody directed against CD38, developed by HI-Bio and I-Mab Biopharma and being investigated in clinical studies for renal autoimmune diseases and relapsed/refractory multiple myeloma.
•MOR210/TJ210/HIB210 – a human antibody directed against C5aR1, the receptor of the complement factor C5a and, being investigated by I-Mab Biopharma in a Phase 1 study for relapsed or refractory advanced solid tumors and by HI-Bio in healthy volunteers.
52
We have a world-class team of multi-disciplinary experts based in Munich and Boston, and our Management Board and senior experts have deep experience and capabilities in biology, chemistry, product discovery, clinical development and commercialization.
In February 2024, MorphoSys entered into a Business Combination Agreement with Novartis. As a result of the successful completion of the Novartis Takeover Offer, Novartis would obtain exclusive, worldwide rights to develop and commercialize pelabresib and tulmimetostat across all indications. Separately, we also entered into a Purchase Agreement to sell and transfer all exclusive rights worldwide related to tafasitamab to Incyte. Prior to this agreement, tafasitamab was co-marketed in the U.S. by MorphoSys and Incyte as Monjuvi® (tafasitamab-cxix) and outside the U.S. by Incyte as Minjuvi®.
Our Strategy
MorphoSys is focused on the development and commercialization of innovative cancer medicines, advancing its mid-to-late-stage oncology pipeline in areas where there is a dire need for more effective and well-tolerated therapies.
Advancing Clinical Development Programs
We believe that a focus on proprietary drug development and commercialization offers the best path to delivering growth and long-term success. We are currently furthering the clinical development of our investigational medicines pelabresib and tulmimetostat. To advance these clinical development programs, we invest a significant portion of our financial resources.
•Accelerating Pelabresib Clinical Development Program
Pelabresib, our investigational BET inhibitor, is potentially a foundational first-line treatment for myelofibrosis. Comprehensive Phase 3 MANIFEST-2 results, presented at the American Society of Hematology (ASH) 2023 Annual Meeting and Exposition in December 2023, demonstrated that the combination of pelabresib and the JAK inhibitor ruxolitinib improves all four hallmarks of myelofibrosis – spleen size, anemia, bone marrow fibrosis and disease-associated symptoms – versus placebo plus ruxolitinib, which is the standard of care in myelofibrosis. We are confident that the MANIFEST-2 data package – complemented by long-term Phase 2 results – will provide impactful evidence to regulatory agencies as we pursue approval for patients with myelofibrosis. As myelofibrosis represents a multi-billion-dollar market opportunity, this endeavor remains our number one priority. Beyond myelofibrosis, pelabresib has demonstrated potential clinical benefit in early-stage trials in other myeloid diseases – providing additional growth opportunities.
•Advancing Tulmimetostat Clinical Development Program
Tulmimetostat was designed to improve on first-generation EZH2 inhibitors through increased potency, longer residence time on target and a longer half-life, offering the potential for enhanced anti-tumor activity. It is currently being evaluated in a Phase 1/2 trial for advanced solid tumors and lymphomas. Updated results from the Phase 2 portion of the study were presented at American Society of Clinical Oncology (ASCO) 2023 Annual Meeting. The data suggest responses or disease stabilization across all solid tumor cohorts, including those with heavily pre-treated patients. Notably, complete and partial responses were also observed in the lymphoma cohort. In September 2023, the U.S. Food and Drug Administration (FDA) granted Fast Track designation for tulmimetostat for the treatment of patients with advanced, recurrent or metastatic ARID1A-mutated endometrial cancer who have progressed on at least one prior line of treatment.
Leveraging Strong Partnerships
Our key partner programs, developed via our legacy antibody technology platform, continue to mature and have the potential to generate significant value. These include ianalumab (Sjögren’s disease, lupus nephritis and other autoimmune diseases), abelacimab (venous thromboembolism prevention), setrusumab (osteogenesis imperfecta), bimagrumab (adult obesity) and felzartamab (autoimmune diseases, multiple myeloma). If approved by regulatory authorities, MorphoSys would receive milestone payments and royalties from these programs.
Our Strengths
Strong Mid- to Late-Stage Oncology Pipeline
We have a pipeline offering best- and first-in-class opportunities in oncology, both hematologic malignancies and solid tumors:
•Pelabresib
Pelabresib represents an opportunity to substantially improve the standard of care for myelofibrosis, a debilitating and deadly disease. Comprehensive Phase 3 MANIFEST-2 results presented at the ASH 2023 Annual Meeting and Exposition in December 2023 point to a paradigm shift in myelofibrosis treatment, a field in dire need of innovation. Moreover, proof-of-concept data provide evidence that pelabresib has potential clinical benefit in essential thrombocythemia.
53
•Tulmimetostat
Initial data showed encouraging response rates to monotherapy treatment with tulmimetostat, across different tumor types, in heavily pre-treated patients. Physicians have expressed excitement about the deep responses seen in these heavily pre-treated patients, the majority of whom currently have limited or no treatment options. These preliminary results are promising and showcase the therapy’s best-in-class potential in an array of cancer types. In September 2023, the FDA granted Fast Track designation for tulmimetostat for the treatment of patients with advanced, recurrent or metastatic ARID1A-mutated endometrial cancer who have progressed on at least one prior line of treatment.
Strong Cash Runway with Cash Available into early 2026
We have a strong cash runway with cash available into early 2026 (including the convertible debt repayment). As of December 31, 2023, we had € 680.5 million in cash and other financial assets.
Additionally, while not a core focus of our business strategy, our partner programs – developed via our legacy antibody technology platform – offer potential upside and provide us with options for non-dilutive financing.
We also have a broad intellectual property portfolio to protect our product candidates. For more information on our intellectual property, see “Item 4.B. Intellectual Property”.
World-Class Team of Clinical Development and Commercial Experts
We have a world-class team of experts based in Munich and Boston, with experience across disciplines, including clinical development, medical affairs, commercial and more. Our accomplished clinical development team has a proven track record of operational excellence.
As a result of our previous work with Monjuvi® until the sale of the asset to Incyte in February 2024, we have a strong U.S. commercial infrastructure that includes a medical affairs team with expertise in hematology-oncology.
Proprietary Clinical Development
MorphoSys is primarily advancing the clinical development of its own compounds, with further antibody candidates being clinically developed by partners. During the clinical phases, decisions are made on a case-by-case basis as to whether and at what point a partnership for further development and commercialization should be pursued. Drug candidates can be either fully out-licensed, developed on a proprietary basis, or developed with a partner (co-development).
Pelabresib
Overview
Pelabresib (formerly known as CPI-0610; was acquired through the Constellation acquisition) is an investigational selective small molecule BET inhibitor designed to promote anti-tumor activity by specifically inhibiting the function of BET proteins.
BET proteins are epigenetic readers that regulate specific genes by binding unique regions of the genome through their ability to read specific chemical tags on chromatin. In some instances, BET proteins regulate genes that are abnormally expressed in a variety of human cancers. BET inhibitors have the potential to downregulate the expression of oncogenes, which are key genes that have the potential to cause cancer, such as MYC or NF-κB target genes, and effectively kill many cancer cell lines in in vitro models. These observations led to the generation and clinical investigation of BET inhibitors in several cancer types. Preclinical activity was observed in cancer that are driven by NF-κB signaling.
The combination of Constellation's preclinical studies, as well as translational insights from the first-in-human study of pelabresib, led to prioritizing the clinical development of pelabresib in patients with myelofibrosis (MF).
Two major clinical trials of pelabresib in patients with MF - the Phase 2 MANIFEST trial and the Phase 3 MANIFEST-2 trial are being conducted. Following the acquisition of Constellation, we took measures to optimize the design and that were intended to improve the speed of enrollment of the MANIFEST-2 trial.
The FDA granted Fast Track Designation to pelabresib in October 2018 for the treatment of myelofibrosis (MF). The FDA grants Fast Track designation to facilitate the development and expedite the review of drugs to treat serious or life-threatening diseases and fill unmet medical needs. A drug that receives Fast Track designation is eligible for more frequent meetings with the FDA to discuss the drug’s development plan and ensure collection of appropriate data needed to support drug approval, more frequent written communication about the design of the proposed clinical trials and use of biomarkers, eligibility for accelerated approval and priority review, and rolling review. The FDA and the European Commission also granted orphan drug designation to pelabresib for the treatment of myelofibrosis in November 2019 and February 2020 respectively.
54
Description of Myelofibrosis and Current Treatment
MF is a form of blood cancer that disrupts the body's normal production of blood cells. It causes fibrosis (scarring) of the bone marrow that may lead to severe anemia as well as thrombocytopenia. Patients suffering from MF can have enlarged spleens as well as many other physical symptoms, including abdominal discomfort, bone pain, and extreme fatigue.
The megakaryocytes produce inflammatory molecules responsible for fibrosis via the excessive deposition of collagen, which scars the bone marrow and displaces hematopoietic cells. In response, the hematopoietic cells migrate to the spleen (liver and lung to a lesser extend), which is called extramedullary hematopoesis. The spleen gets enlarged and sometimes becomes dysfunctional. The extramedullary hematopoiesis is often not able to fully compensate for the loss of bone marrow hematopoiesis, leading to cytopenia. Additionally, compromised erythroid differentiation (the process by which immature bone marrow derived cells specialize and become red blood cells) leads to anemia in patients with MF.
Approximately 4–6 per 100,000 people in the U.S. are diagnosed with MF, 90% are intermediate or high-risk patients. There are limited treatment options for patients with MF. We believe there are approximately 18,000 intermediate or high-risk MF patients in the United States that are eligible for systemic treatment, including ruxolitinib. Only about 50% of patients achieve initial adequate disease control with JAK inhibitors.
Currently the JAK inhibitors ruxolitinib, fedratinib, momelotinib and pacritinib are the only approved products for MF, and there is no other approved class of drugs for patients whose MF progresses after treatment with these products. Ruxolitinib is the standard of care for patients with intermediate to high risk MF with platelet count over 50 x 10^9/L, while pacritinib has been approved for patients with intermediate to high risk MF with platelet count below 50 x10^9/L and momelotinib has been approved for patients with intermediate or high-risk myelofibrosis with anaemia. Ruxolitinib inhibits dysregulated janus kinase 1 and 2 (collectively referred to herein as “JAK”), signaling that is associated with MF. Ruxolitinib treatment leads to spleen volume reduction and symptom improvement in patients with MF. However, its side effects include anemia and thromobocytopenia. Fedratinib, is a JAK2 inhibitor, and has similar efficacy and its side effects include both anemia and thrombocytopenia. Ruxolitinib has not been shown to significantly reverse bone marrow fibrosis, a condition that has been documented as a primary cause of morbidity and mortality in MF. Many patients may not tolerate treatment with, or will have an insufficient response to, ruxolitinib. Patients generally have a poor survival prognosis following discontinuation of therapy with ruxolitinib. Current approved drugs for the treatment of patients experiencing MF offer symptomatic improvement and better treatment options are needed.
Pelabresib—Potential Mechanism of Action
Abnormal BET function has been implicated in cancer through several means, including chromosomal translocation and gene amplification and overexpression whereby oncogenic and inflammatory signals are turned on in cancer cells through altered BET activity.
BET proteins control the expression of the target genes of NF-κB, a key immune signaling pathway that is abnormally activated in various diseases, including cancer and immune disorders. NF-κB signaling has been shown to be abnormally high in some hematological malignancies, such as MF and activated B-cell-like diffuse large B-cell lymphoma (ABC-DLBCL). In preclinical studies in MF, animals treated with BET inhibitors alone or in combination with a JAK inhibitor displayed a reduction in NF-κB target gene expression, improvement in bone marrow fibrosis, and reduced disease burden.
In addition, BET proteins promote the generation of megakaryocytes from hematopoietic stem cells. Hematopoietic stem cells have mutation(s) leading to aberrant, highly proliferative, megakaryocytes, which represent key drivers of MF.
By blocking BET protein function with pelabresib, we believe that we promote erythroid cell differentiation which may result in improved hemoglobin levels and ameliorate anemia in patients with MF. As the result of the effect on both erythroid and megakaryocyte differentiation, we believe that pelabresib has the potential to improve the health of the bone marrow of patients with MF.
Development of Pelabresib
Results from preclinical studies, as well as translational insights from the first-in-human study of pelabresib, led to prioritizing the clinical development of pelabresib in MF. There are currently two ongoing trials evaluating pelabresib in this indication, the Phase 2 MANIFEST trial and the Phase 3 MANIFEST-2 trial.
Active Clinical Trials
MANIFEST: The MANIFEST is a global, multicenter, open-label Phase 2 study that evaluates pelabresib as a monotherapy or in combination with ruxolitinib (marketed as Jakafi®/Jakavi®), the current standard of care. In Arm 3 of this study, pelabresib is being evaluated in combination with ruxolitinib in JAK-inhibitor-naïve MF patients, with a primary endpoint of the proportion of patients with a ≥35% spleen volume reduction from baseline (SVR35) at 24 weeks of treatment. Pelabresib is also being
55
evaluated in a second-line setting (2L) either as a monotherapy in patients who are resistant to, intolerant of, or ineligible for ruxolitinib and no longer on the drug (Arm 1), or as an add-on therapy to ruxolitinib in patients with a suboptimal response to ruxolitinib or MF progression (Arm 2). Patients in Arms 1 and 2 are being stratified based on transfusion-dependent (TD) status. The primary endpoint for the patients in cohorts 1A and 2A, who were TD at baseline, is conversion to transfusion independence for 12 consecutive weeks. The primary endpoint for patients in cohorts 1B and 2B, who were not TD at baseline, is the proportion of patients with an SVR35 at 24 weeks of treatment. In Arm 4 of this study, pelabresib is being evaluated as a monotherapy in high-risk patients with essential thrombocythemia (ET) who are resistant or intolerant to hydroxyurea (HU). The primary endpoint for patients in Arm 4 is complete hematological response rate after one cycle, or 21 days, of treatment.
In December 2021, updated data from MANIFEST were presented at the 2021 ASH Annual Meeting. At this meeting, the interim data from Arm 3 of MANIFEST evaluating pelabresib as a first-line combination with ruxolitinib for patients with MF who had not previously been treated with a JAK inhibitor (JAK inhibitor-naïve) were presented.
In addition, data from Arm 1 of the ongoing MANIFEST trial were also presented in an oral presentation at the 2021 ASH Annual Meeting: pelabresib is being evaluated as a monotherapy in patients with advanced MF who are ineligible to receive, intolerant of, or refractory to JAK inhibitors, a population with very limited therapeutic options. Patients were divided into two cohorts, TD and non-TD. For the TD cohort, the primary endpoint was conversion to transfusion independence for 12 consecutive weeks. In the non-TD cohort, the primary endpoint was SVR35 at week 24. At week 24, 11% (n = 7) of patients reached SVR35. In addition, 31% of patients had a spleen volume reduction of 25% or more (n = 20) at week 24. Across all cohorts, 28% (n = 18) of patients achieved TSS50. No new safety signals were identified in the study. The most common hematologic adverse events were thrombocytopenia (23%, grade 3/4) and anemia (15%, grade 3). Non-hematological events included diarrhea (6%, grade 3) and respiratory tract infections (5%, grade 3).
Additionally, analyses from an exploratory endpoint presented at ASH 2021 showed a reduction of megakaryocyte clustering in bone marrow and correlation with spleen volume reduction. Megakaryocytes are the cells in the bone marrow responsible for making platelets, and the clustering of these cells are one of the signs of myelofibrosis. The exploratory data, which require further evaluation and confirmation, suggest the potential pelabresib may have in changing the course of myelofibrosis treatment, if approved.
In June 2022, MorphoSys presented data from multiple analyses of the ongoing MANIFEST study during oral and poster sessions at the European Hematology Association 2022 (EHA 2022) Hybrid Congress. A study was presented in an oral session that analyzed cells derived from blood of patients who enrolled in the MANIFEST trial and from healthy volunteers. The findings indicated that pelabresib alone or in combination with the JAK inhibitor ruxolitinib may have the potential to improve the typical imbalance in the two white blood cell populations, the myeloid and lymphoid cells, and help restore normal blood cell development. These improvements also correlated with decreases in spleen volume, a key clinical measure of treatment success. Additionally, pelabresib alone or in combination with ruxolitinib decreased pro-inflammatory and pro-fibrotic signaling in monocytes, suggested a potential attenuation of disease processes.
A second oral presentation highlighted positive interim data from the MANIFEST trial regarding the safety and efficacy of pelabresib in combination with ruxolitinib in patients who were not previously treated with a JAK inhibitor and in those with suboptimal response to ruxolitinib (Arm 2 of MANIFEST study). The findings showed that the combination led to reductions in spleen volume and symptom burden, with potential disease-modifying effect as measured by reduced levels of pro-inflammatory cytokines and improved bone marrow morphology. Over two-thirds (68%; n=57) of JAK inhibitor-naïve patients treated with the combination achieved at least a 35% reduction in spleen volume (SVR35) from baseline at week 24. Notably, 80% of patients achieved SVR35 at any time on study. Most patients also saw their symptoms reduced, with 56% (n=46) achieving at least a 50% reduction in total symptom score (TSS50) from baseline at week 24. No new safety signals were identified in the study. The most common hematologic adverse events were thrombocytopenia (12%, grade 3/4) and anemia (34%, grade 3/4). Non-hematological events included dyspnea (5%, grade 3) and respiratory tract infections (8%, grade 3/4).
In a poster presentation at EHA 2022, matching-adjusted indirect comparisons were used to compare findings for the combination of pelabresib plus ruxolitinib in treatment-naïve patients with intermediate- or high-risk disease in one arm of the MANIFEST trial with findings from historical clinical trials examining the use of JAK inhibitor monotherapy in myelofibrosis. Adjusting for cross-trial differences, the estimated response rate ratios favored pelabresib plus ruxolitinib combination therapy over ruxolitinib, fedratinib, or momelotinib monotherapy for SVR35 and for TSS50, suggesting improved efficacy versus the JAK inhibitors alone.
In December 2022, we presented new longer-term Phase 2 results on pelabresib in myelofibrosis from the ongoing MANIFEST study at ASH 2022. The latest analyses include longer-term data showing durable improvements in both spleen volume and symptom score beyond 24 weeks (data cutoff July 29, 2022), with pelabresib plus ruxolitinib in JAK inhibitor-naïve patients (Arm 3 of the study). Translational data from MANIFEST were also presented that suggest an association of biomarkers with potential disease-modifying activity of pelabresib.
56
At 24, 48, and 60 weeks, 68% (57/84), 61% (51/84), and 54% (45/84), respectively, of JAK inhibitor-naïve patients treated with pelabresib in combination with ruxolitinib achieved at least a 35% reduction in spleen volume (SVR35) from baseline. SVR35 was achieved by 80% (67/84) of patients at any time on study. Also at 24 weeks, 56% (46/82) of patients had at least a 50% reduction in their total symptom score (TSS50) from baseline, suggesting a reduction in symptom burden. At 48 and 60 weeks, 44% (36/82) and 43% (35/82) of patients, respectively, achieved TSS50. An exploratory analysis demonstrated that bone marrow fibrosis improved by one grade or more in 27% (17/63) of evaluable patients at week 24, and 59% (10/17) of those patients maintained that improvement at week 48 or beyond. An improvement of one grade or more at any time was achieved by 40% (25/63) of patients. The most common hematologic treatment-emergent adverse event (AE) of any grade was thrombocytopenia, which was reported in 55% (grade ≥3: 18%) of patients. Anemia was reported in 43% (grade ≥3: 34%) of patients. The most common (≥25%) non-hematologic treatment-emergent AEs of any grade were diarrhea (43%), respiratory tract infection (41%), asthenic conditions (38%), musculoskeletal pain (32%), constipation (30%), nausea (29%), dizziness (27%), and abdominal pain (26%).
In the MANIFEST study, changes in biomarkers correlated with improvements in clinical measures of treatment success (SVR35, TSS50, and hemoglobin increases indicative of improved anemia), suggesting a potential disease-modifying effect of pelabresib. Examined biomarkers included bone marrow scarring, known as fibrosis, and the frequency of a Janus Kinase 2 allele (V617F) that is known to drive disease activity. Across the three MF arms of MANIFEST, 40% (33/82) of patients who achieved SVR35 at week 24 also had at least a one-grade improvement in bone marrow fibrosis and/or a 20% or greater reduction in the frequency of the variant allele. Of TSS50 responders at week 24, 28% (28/100) also showed at least a one-grade improvement in bone marrow fibrosis and/or a 20% or greater reduction in the frequency of the variant allele. Furthermore, 29% (24/84) of patients who had hemoglobin improvements (any level of increase from baseline) also had at least a one-grade improvement in bone marrow fibrosis and/or a 20% or greater reduction in the frequency of the variant allele. All patients who had clinical responses (SVR35, TSS50, and hemoglobin improvement) plus reduced variant allele frequency and improvement in bone marrow fibrosis were naïve to JAK inhibitors.
At the European Hematology Association (EHA) Hybrid Congress in June 2023, we presented a poster on Arm 3 of the MANIFEST study, which examines the combination of pelabresib and ruxolitinib in JAK-inhibitor-naïve patients with myelofibrosis. This treatment resulted in deep and durable spleen and symptom responses at and beyond week 24. The findings demonstrated clinically meaningful improvements in anemia, including the need for fewer transfusions, which may positively impact patients’ quality of life. No new safety signals were observed with a longer follow-up of 11 additional months. A second poster on MANIFEST Arm 2 showed pelabresib as an add-on to ongoing ruxolitinib therapy in patients with a suboptimal/lost response to ruxolitinib monotherapy resulted in durable and deepening splenic and symptom responses at and beyond week 24. The findings suggested improvements in anemia, including the need for fewer transfusions, which may positively impact patients’ quality of life. No new safety signals were observed with a longer follow-up of 11 additional months. The most common treatment-emergent adverse events (TEAEs) were low grade.
During an oral presentation at the EHA and a poster discussion at the American Society of Clinical Oncology (ASCO) Annual Meeting in June 2023, we presented positive results from Arm 4 of the Phase 2 MANIFEST study, which is investigating pelabresib as a monotherapy in patients with high-risk essential thrombocythemia (ET), whose disease is refractory or intolerant to hydroxyurea. These robust proof-of-concept results support pelabresib’s expansion into other myeloid diseases. As such, we will continue our ongoing evaluation of pelabresib in ET in the MANIFEST study. We are also considering initiating a Phase 2 study in lower-risk myelodysplastic syndrome (MDS). Patients with MDS experience progressive anemia that can require regular blood transfusions or subcutaneous injections, often diminishing quality of life. Furthermore, patients have low long-term response rates to currently available treatments, reflecting a need for new therapeutic options.
MANIFEST‑2: MANIFEST‑2, a global, multicenter, double-blind, randomized Phase 3 clinical study, is evaluating pelabresib plus ruxolitinib versus placebo plus ruxolitinib in JAK-inhibitor-naïve patients with primary MF or post-essential thrombocythemia (post-ET) or post-polycythemia vera (post-PV) MF who have splenomegaly and symptoms requiring therapy. Since the acquisition of Constellation, MorphoSys has optimized the study’s design by increasing the number of trial participants. Measures were also taken to improve the speed of enrollment, including adding new contract research organizations (CROs), improving the interaction with investigators, and expanding the number of countries and sites. On April 4, 2023, MorphoSys announced that enrollment was completed for the MANIFEST-2 study.
On November 20, 2023, we announced topline results from the Phase 3 MANIFEST-2 study. MANIFEST-2 met its primary endpoint, as the combination therapy demonstrated a statistically significant and clinically meaningful improvement in the proportion of patients achieving at least a 35% reduction in spleen volume (SVR35) at week 24. The key secondary endpoints assessing symptom improvement – proportion of patients achieving at least a 50% reduction in total symptom score (TSS50) and absolute change in total symptom score (TSS) from baseline at week 24 – showed a strong positive trend favoring the pelabresib and ruxolitinib combination. In an analysis of patients classified as intermediate risk (Dynamic International Prognostic Scoring System [DIPSS] Int-1 and Int-2) – constituting more than 90% of patients in MANIFEST-2 – the combination therapy demonstrated significant improvements in both key secondary endpoints. DIPSS was a pre-defined
57
stratification factor in the MANIFEST-2 study protocol. 430 JAK inhibitor-naïve adult patients with myelofibrosis were randomized for this study.
On December 10, 2023, detailed findings of the MANIFEST-2 study were presented during an oral presentation at the 65th American Society for Hematology (ASH) Annual Meeting and Exposition:
•Strong Reductions in Spleen Size and Symptoms
In the MANIFEST-2 study, pelabresib and ruxolitinib demonstrated a near doubling in the proportion of patients achieving a ≥35% reduction in spleen volume (SVR35) at 24 weeks, the primary endpoint, versus placebo plus ruxolitinib (p<0.001). For the first key secondary endpoint assessing symptom reduction, absolute change in total symptom score (TSS) at 24 weeks, there was a strong numerical improvement for patients receiving pelabresib and ruxolitinib versus placebo plus ruxolitinib. The response rate for the second key secondary endpoint, proportion of patients achieving ≥50% reduction in symptom score (TSS50) at 24 weeks, was also numerically greater for patients receiving pelabresib and ruxolitinib. Significant improvements in both key secondary endpoints were observed with the pelabresib combination for patients classified as intermediate-risk (Dynamic International Prognostic Scoring System [DIPSS] Int-1 and Int-2), who account for over 90% of the MANIFEST-2 population. The proportion of patients achieving both SVR35 and TSS50 at 24 weeks was doubled with pelabresib and ruxolitinib versus placebo plus ruxolitinib (40.2% vs. 18.5%, respectively).
Details are included in the table below:
| Endpoint | Pelabresib + Ruxolitinib (N = 214) | Placebo +Ruxolitinib (N = 216) | Difference | ||||||||||||||||||||
| SVR35 | 65.9% | 35.2% | 30.4%* | ||||||||||||||||||||
| P-value: p<0.001 | |||||||||||||||||||||||
| Absolute Change in TSS | -15.99 | -14.05 | -1.94** | ||||||||||||||||||||
| (Mean Baseline: 28.26) | (Mean Baseline: 27.36) | P-value: 0.0545 | |||||||||||||||||||||
| TSS50 | 52.3% | 46.3% | 6.0%* | ||||||||||||||||||||
| P-value: 0.216 | |||||||||||||||||||||||
| *Difference calculated using Cochran–Mantel–Haenszel (CMH) common risk difference | |||||||||||||||||||||||
| **Least square mean estimate | |||||||||||||||||||||||
•Improvement in Anemia
Patients receiving pelabresib in combination with ruxolitinib reported fewer anemia adverse events (43.9%, grade ≥3: 23.1%) compared with placebo plus ruxolitinib (55.6%, grade ≥3: 36.4%). Additionally, by week 24, fewer patients in the pelabresib and ruxolitinib arm required red blood cell transfusions compared with the placebo arm (30.8% vs. 41.2%, respectively). A greater proportion of patients achieved a hemoglobin response — defined as a ≥1.5 g/dL mean increase in hemoglobin levels over baseline in the absence of transfusions during the previous 12 weeks — with pelabresib and ruxolitinib versus placebo plus ruxolitinib (9.3% vs. 5.6%, respectively). Average hemoglobin levels were greater in patients receiving pelabresib and ruxolitinib than in those receiving placebo plus ruxolitinib, starting at week 9 and continuing to week 24. Improvement in anemia was observed across all studied patient risk groups.
•Improvement in Bone Marrow Fibrosis
Bone marrow fibrosis, or the replacement of bone marrow with fibrous scar tissue, is a central pathological feature of myelofibrosis. In MANIFEST-2, fibrosis was improved by at least one grade in a greater proportion of patients receiving pelabresib and ruxolitinib (38.5% vs. 24.2% with placebo plus ruxolitinib) and worsened by at least one grade in a smaller proportion of patients receiving pelabresib and ruxolitinib (16.3% vs. 28.3% with placebo plus ruxolitinib) at 24 weeks. Bone marrow fibrosis is graded on a scale from 0 (normal) to 3 (most severe) based on fiber density; studies suggest a correlation between the grade of bone marrow fibrosis and patient prognosis.
•Biomarker Analysis Suggests Disease Modification
In a biomarker analysis, average plasma levels of inflammatory cytokines (IL-8, IL-6, TNF-α, and other NF-κB-regulated cytokines) were reduced in patients receiving pelabresib and ruxolitinib compared with placebo plus ruxolitinib at 24 weeks. Increased cytokine levels are associated with all four disease hallmarks; increased IL-8 levels are also associated with worse survival outcomes. These biomolecular improvements suggest early evidence of a disease-modifying effect.
•Safety Profile
Overall, grade ≥3 treatment-emergent adverse events (TEAEs) were reported less frequently with pelabresib and ruxolitinib than with placebo plus ruxolitinib (49.1% vs. 57.5%, respectively). In the pelabresib and ruxolitinib arm, the most common
58
(≥10%) hematologic TEAEs were anemia (43.9%; grade ≥3: 23.1%), thrombocytopenia (32.1%; grade ≥3: 9.0%), and platelet count decrease (20.8%; grade ≥3: 4.2%). In the placebo plus ruxolitinib arm, the most common hematologic TEAEs were anemia (55.6%; grade ≥3: 36.4%), thrombocytopenia (23.4%; grade ≥3: 5.6%), and platelet count decrease (15.9%; grade ≥3: 0.9%). The most common (≥10%) non-hematologic TEAEs in the pelabresib and ruxolitinib arm were diarrhea (23.1%; grade ≥3: 0.5%), dysgeusia (18.4%; grade ≥3: 0.5%), constipation (18.4%; grade ≥3: 0%), nausea (14.2%; grade ≥3: 0.5%), cough (12.7%; grade ≥3: 0), asthenia (11.8%; grade ≥3: 0.5%), fatigue (11.8%; grade ≥3: 0.5%), dizziness (11.3%; grade ≥3: 0%), headache (11.3%; grade ≥3: 0.5%), and COVID-19 (11.3%; grade ≥3: 0%). The most common non-hematologic TEAEs in the placebo plus ruxolitinib arm were constipation (24.3%; grade ≥3: 0%), diarrhea (18.7%; grade ≥3: 1.4%), fatigue (16.8%; grade ≥3: 0.9%), COVID-19 (15.9%; grade ≥3: 1.9%), nausea (15.0%; grade ≥3: 0%), asthenia (13.6%; grade ≥3: 0%), dyspnea (13.1%; grade ≥3: 0.9%), cough (11.2%; grade ≥3: 0%), and headache (10.7%; grade ≥3: 0%). Discontinuation rates due to adverse events were 10.7% with pelabresib and ruxolitinib and 6.5% with placebo plus ruxolitinib. The safety profile of the pelabresib and ruxolitinib combination therapy was in line with assessments from previous clinical studies.
•Planned Regulatory Next Steps
MorphoSys will continue conversations with regulatory agencies, with intention to submit a New Drug Application (NDA) for pelabresib in combination with ruxolitinib in myelofibrosis to the FDA and a Marketing Authorization Application (MAA) to the European Medicines Agency in the middle of 2024. The combination therapy received Fast Track designation for this disease from the FDA in 2018.
Tafasitamab
Overview
Tafasitamab (formerly known as MOR208, XmAb5574) is a humanized Fc-modified CD19-targeting immunotherapy. CD19 is selectively expressed on the surface of B-cells, which belong to a group of white blood cells. CD19 enhances B-cell receptor signaling, which is an important factor in B-cell survival and growth. CD19 is a validated target structure for the treatment of B-cell malignancies.
Tafasitamab was being developed pursuant to a collaboration and license agreement that we entered into in June 2010 with Xencor. For more information on this agreement, refer to Collaboration and License Agreements—Collaboration and License Agreement with Xencor.
In addition, at the beginning of 2020, we signed a global collaboration and license agreement to further develop and commercialize tafasitamab granting Incyte U.S. co-commercialization and ex-U.S. commercialization rights for tafasitamab. Under the terms of the agreement, we and Incyte were responsible for the further clinical development of tafasitamab. Tafasitamab was co-marketed by Incyte and MorphoSys in the United States under the trade name Monjuvi® and by Incyte in Europe, Canada, and other jurisdictions under the trade name Minjuvi®. For more information on this agreement, refer to Collaboration and License Agreements—Collaboration and License Agreement with Incyte.
In February 2024, Incyte obtained exclusive rights worldwide to tafasitamab. Incyte will assume full responsibility and cover all costs going forward for the development and commercialization of the asset. As part of the sale and transfer of tafasitamab to Incyte, Incyte also assumed the Collaboration and License Agreement with Xencor.
Tafasitamab is currently being investigated for the treatment of various B-cell malignancies, namely first-line DLBCL, relapsed/refractory follicular lymphoma (r/r FL), and relapsed/refractory marginal zone lymphoma (r/r MZL).
Development of Tafasitamab up to the Regulatory Approvals of Monjuvi® and Minjuvi® and Further Regulatory Progress
On July 31, 2020, the U.S. FDA granted accelerated approval to Monjuvi® in combination with lenalidomide for the treatment of adult patients with r/r DLBCL not otherwise specified, including DLBCL arising from low grade lymphoma, and who are not eligible for autologous stem cell transplant (ASCT).
On August 26, 2021, we and Incyte announced that the European Commission had granted conditional marketing authorization for Minjuvi® (tafasitamab) in combination with lenalidomide, followed by Minjuvi® monotherapy, for the treatment of adult patients with r/r DLBCL who are not eligible for ASCT. The approval was based on data from the L-MIND study, supported by RE-MIND.
On August 24, 2021, we announced that Health Canada had granted conditional marketing authorization to Incyte for Minjuvi® in combination with lenalidomide for the treatment of adults with r/r DLBCL not otherwise specified, including DLBCL arising from low grade lymphoma, who are not eligible for ASCT.
On March 22, 2022, we and Incyte announced that the Swiss agency for therapeutic products (Swissmedic) had granted temporary approval for Minjuvi® (tafasitamab) in combination with lenalidomide, followed by Minjuvi® monotherapy, for the
59
treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), after at least one prior line of systemic therapy including an anti-CD20 antibody, who are not eligible for autologous stem cell transplant (ASCT). Incyte holds exclusive commercialization rights for Minjuvi® in Switzerland.
In 2023 Minjuvi® (tafasitamab) in combination with lenalidomide has been approved in other jurisdictions including Australia and Brazil.
In February 2024, Incyte obtained exclusive rights worldwide to tafasitamab. Incyte will assume full responsibility and cover all costs going forward for the development and commercialization of the asset.
Ongoing Clinical Combination Trials with Tafasitamab
The clinical development of tafasitamab is focused on non-Hodgkin’s lymphoma (NHL). Treatment options for patients with r/r DLBCL who are not candidates for HDC and ASCT were limited prior to the U.S. approval of tafasitamab.
The clinical studies frontMIND and firstMIND may support the potential use of tafasitamab in the first-line treatment of DLBCL. Tafasitamab is also being examined with inMIND, a Phase 3 study in patients with r/r follicular lymphoma (FL) Grade 1 to 3a or r/r nodal, splenic, or extranodal marginal zone lymphoma (MZL).
More details on each study are given below:
frontMIND: In addition to clinical development in r/r DLBCL, on May 11, 2021 we announced that the first patient had been dosed in frontMIND, a pivotal Phase 3 trial of tafasitamab in first-line DLBCL: frontMIND is evaluating tafasitamab and lenalidomide in combination with R-CHOP compared to R-CHOP alone as first-line treatment for high-intermediate and high-risk patients with untreated DLBCL. On April 4, 2023, MorphoSys announced that the enrollment of the frontMIND study with 899 patients was completed. The topline data from this study are expected in the second half of 2025.
firstMIND: The study included patients with newly diagnosed DLBCL and paved the way for the frontMIND study. On December 10, 2022, we presented updated results from the firstMIND trial at ASH 2022. The final analysis from this Phase 1b trial showed no new safety signals and provided additional information on progression-free and overall survival at 24 months for patients with newly diagnosed diffuse large B-cell lymphoma treated with tafasitamab plus lenalidomide and R-CHOP. The Phase 1b study firstMIND is an open-label, randomized safety study combining tafasitamab or tafasitamab plus lenalidomide with standard R-CHOP for patients with newly diagnosed DLBCL. Additional analyses highlighted the prognostic potential of sensitive circulating tumor (ct) DNA minimal residual disease (MRD) assays in patients with DLBCL after first-line therapy.
Additionally, Incyte is responsible for conducting inMIND, a Phase 3 study in patients with r/r follicular lymphoma (FL) Grade 1-3a or r/r nodal, splenic, or extranodal marginal zone lymphoma (MZL). On August 1, 2023, Incyte announced that the inMIND study is fully enrolled. The inMIND study evaluates whether tafasitamab and lenalidomide as an add-on to rituximab provides improved clinical benefit compared with lenalidomide alone as an add-on to rituximab in patients with r/r FL or r/r MZL. The primary endpoint of the study is PFS in the FL population, and the key secondary endpoints are PFS and OS in the overall population as well as PET-CR at the end of treatment in the FL population. Topline data are expected in the second half of 2024.
L-MIND: In April 2023, we and Incyte presented at the American Association for Cancer Research (AACR) Annual Meeting 2023 final five-year follow-up data from the Phase 2 L-MIND study showing that Monjuvi® (tafasitamab-cxix) plus lenalidomide followed by Monjuvi® monotherapy provided prolonged, durable responses in adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL).
At the data cut-off (Nov. 14, 2022) for the full analysis set (80 patients), the best overall response rate (ORR) was 57.5% (95% CI = 45.9, 68.5; n = 80), and a complete response (CR) was observed in 41.3% of patients (95% CI = 30.4, 52.8; n = 33). A partial response (PR) was observed in 16.3% of patients (95% CI = 8.9, 26.2; n = 13). Additional results included:
• Median duration of response was not reached after a median follow-up of 44.0 months (95% CI = 29.9, 57.0).
• The median overall survival was 33.5 months (95% CI = 18.3, NR) and median progression-free survival was 11.6 months (95% CI = 5.7, 45.7).
• Of the 21 patients with >60 months of follow-up, 14 had received one prior line of therapy (pLoT), and seven patients had received ≥2 pLoTs.
• Patients with one pLoT (n = 40) had a higher ORR of 67.5% (CR = 52.5% and PR = 15%) compared to 47.5% of patients with two or more pLoTs (n = 40; CR = 30% and PR = 17.5%).
No new safety signals were identified. The majority of adverse events (AEs) were grade 1 or grade 2 during both combination and monotherapy treatment. Patients experienced a lower frequency of all-grade and grade 3 or higher adverse events during monotherapy. The most common adverse events with combination therapy were neutropenia (incidence per person per year, all-
60
grade/grade ≥3: 3.79/2.09) and thrombocytopenia (1.52/0.52), which declined after patients switched to monotherapy (all-grade/grade ≥3: 1.09/0.70 and 0.17/0.06, respectively, in the first two years of monotherapy). Neutropenia and diarrhea were the most common adverse events in the first two years of monotherapy. Monjuvi®, in combination with lenalidomide, was granted accelerated approval based on the one-year primary analysis of the L-MIND study. The data for the five-year analysis of the L-MIND study have not yet been submitted to, or reviewed by, the FDA.
During the American Society of Clinical Oncology (ASCO) Annual Meeting from June 2 to 6, 2023, the European Hematology Association (EHA) Hybrid Congress from June 8 to 11, 2023, the International Conference on Malignant Lymphoma (ICML) from June 13 to 17, 2023, and the Hybrid Annual Meeting of the Society of Hematologic Oncology (SOHO) from September 6 to 9, 2023, MorphoSys presented posters and e-publications of both the five-year L-MIND data overall and a new subgroup analysis. The new data showed that overall response rate was comparable across subgroups, numerically favoring patients with positive prognostic factors. Additionally, duration of response, progression-free survival, and overall survival highlighted long-term clinical efficacy across all subgroups.
B-MIND: The Phase 2/3 study B-MIND is evaluating the safety and efficacy of tafasitamab in combination with the chemotherapeutic agent bendamustine in comparison to rituximab plus bendamustine in patients with r/r DLBCL who are not candidates for HDC and ASCT. The study was fully recruited as of June 2021. The regulatory significance of the B-MIND study has decreased as only long-term safety data for B-MIND are required by the EMA as an obligation for the conditional marketing authorization. As such, all final analyses of primary and secondary endpoints will be performed in mid-2024.
In May 2022, Xencor announced the start of a Phase 2 combination study of the CD3xCD20 bispecific antibody plamotamab in combination with tafasitamab and lenalidomide in patients with relapsed or refractory DLBCL. Plamotamab is a tumor-targeted bispecific antibody that contains both a CD20 binding domain and a cytotoxic T-cell binding domain (CD3). In January 2023, Xencor announced that the company is winding down and ending enrollment in the Phase 2 study due to challenges with patient accrual in lymphoma. The study was terminated in February 2023. The early termination of this study was not based on clinical grounds, i.e., no safety concerns or lack of efficacy were observed.
In June 2022, MorphoSys, Incyte, and Pfizer announced a clinical trial collaboration and supply agreement to investigate the immunotherapeutic combination of Pfizer’s maplirpacept (TTI-622), a novel SIRPα-Fc fusion protein, and tafasitamab plus lenalidomide in patients with relapsed or refractory DLBCL who are not eligible for ASCT. Under the terms of the agreement, Pfizer initiated a multicenter, international Phase 1b/2 study of maplirpacept (TTI-622) with tafasitamab and lenalidomide. MorphoSys and Incyte provide tafasitamab for the study. The study is sponsored and funded by Pfizer and is conducted in North America, Europe, and Asia-Pacific.
In mid-2022, a first patient was treated in the MINDway study, a Phase 1b/2 study evaluating the safety of a modified tafasitamab IV dosing regimen in combination with lenalidomide in adult patients with r/r DLBCL in the same population as L-MIND to enable less frequent dosing in patients with r/r DLBCL.
In February 2024, Incyte obtained exclusive rights worldwide to tafasitamab. Incyte will assume full responsibility and cover all costs going forward for the development and commercialization of the asset.
Tulmimetostat
Overview
Tulmimetostat (formerly known as CPI-0209; also acquired as part of the Constellation acquisition) is an investigational small-molecule, next-generation dual EZH2 and EZH1 inhibitor with an epigenetic mechanism of action that has been designed to achieve comprehensive target coverage through increased on-target residence time. Data from in-vitro preclinical models of multiple cancer types suggested that tulmimetostat may bind to EZH2 more durably and with higher affinity than first-generation EZH2 inhibitors. Tulmimetostat designed to have minimal auto-induction of metabolism, which has been an issue with other EZH2 inhibitors. A Phase 1/2 clinical trial of tulmimetostat is currently ongoing, with patients enrolling. The trial evaluates tulmimetostat as a monotherapy in patients with advanced solid tumors or lymphomas. The preliminary results of Phase 2 results were presented at ASCO 2023 and at the International Gynecologic Cancer Society (IGCS) Meeting in November 2023.
Tulmimetostat - Potential Mechanism of Action
EZH2 as a drug target has a broad therapeutic potential in cancers with EZH2 gain of function mutations or overexpression. EZH2 genomic aberrations and overexpression are frequently associated with late-stage cancer and poor prognosis for a wide variety of cancers. Furthermore, EZH2 also synergizes with other cancer-promoting pathways, such as androgen receptor signaling and immune signaling. Therefore, EZH2 inhibition may have the potential to enhance the effectiveness of existing cancer therapies.
61
EZH2 acts as an epigenetic writer and regulates gene expression through methylation of histones or other proteins. While this effect of EZH2 on gene expression is a normal part of cellular development, some cancers depend on an altered pattern of gene expression and re-direct EZH2 to certain genes causing them to become abnormally repressed. Cancer cells with these aberrant gene expression programs may be more resistant to anti-cancer therapies. As a next generation EZH2 inhibitor, tulmimetostat also inhibits EZH1 that could partly compensate EZH2 inhibition as it has some overlapping activities with EZH2.
Abnormal EZH2 function has been implicated in cancer in a number of ways:
•Cancer genetics: mutations in the gene encoding EZH2 result in the altered enzymatic activity of EZH2, and cancer cells become dependent on this abnormal activity for tumor growth. Alternatively, mutations in other epigenetic regulators can change the gene expression pattern of cancer cells and indirectly create a dependence on EZH2 for cancer cell growth;
•Acquired drug resistance: therapeutic agents promote EZH2-mediated gene silencing that may lead to acquired resistance to these agents;
•Immune suppression: EZH2 mediates reprogramming of immune cells within the tumor, e.g., T cells, and tumor cells to create an immune-suppressive tumor microenvironment; and
•Oncogenic driver synergy: EZH2 suppresses the expression of certain gene sets and promotes tumor progression in the context of validated oncogenic drivers, such as the androgen receptor signaling pathway in prostate cancer. Combined inhibition of EZH2 and the oncogenic driver pathway may elicit a synergistic impact on tumor growth.
Development of Tulmimetostat
Active Clinical Trial
Phase 1/2 Trial in Solid Tumors
Patient enrollment in a Phase 1/2 clinical trial of tulmimetostat is ongoing. This Phase 1/2, open-label, multi-center, first-in-human study is designed to evaluate the safety and tolerability and preliminary clinical activity in patients with advanced solid tumors or lymphomas. The Phase 1 evaluated the dose escalation period in patients with advanced tumors and aimed to determine maximum tolerated dose (MTD) and/or recommended Phase 2 dose (RP2D) as a monotherapy in patients with advanced tumors or lymphomas. Patients are currently enrolled in the Phase 2 expansion cohorts in selected tumor indications: urothelial or other advanced/metastatic solid tumors (ARID1A mutated), ovarian clear-cell carcinoma (ARID1A mutated), endometrial carcinoma (ARID1A mutated), lymphoma, mesothelioma (BAP1 loss mutation), and metastatic castration-resistant prostate cancer.
Updated safety and efficacy data from the ongoing Phase 2 study of tulmimetostat monotherapy in multiple advanced malignancies were presented during the ASCO Annual Meeting in June 2023. The data demonstrated disease stabilization or better across all solid tumor cohorts studied, including those with heavily pre-treated patients: urothelial cancer or ARID1A-mutated advanced solid tumors, ARID1A-mutated ovarian clear-cell carcinoma and endometrial carcinoma, BAP1-mutated mesothelioma and, metastatic castration resistant prostate cancer. In addition, complete and partial responses were observed in the lymphoma cohort. Safety findings from the trial were consistent with the mechanism of EZH2 inhibition. At data cut-off (February 14, 2023), 81 patients enrolled in the Phase 2 expansion phase of the trial had received at least one dose of tulmimetostat in the cohorts listed above and 75 patients also had at least one post-baseline response assessment or discontinued the treatment prior to their first post-baseline assessment for any reason and hence included in the efficacy evaluable set. At trial entry, 86% of patients had been treated with at least two prior lines of therapy. Objective response was observed in patients with ovarian clear-cell carcinoma, endometrial cancer, mesothelioma, and peripheral T-cell lymphoma (PTCL). Of ten evaluable patients with urothelial cancer or ARID1A-mutated advanced solid tumors, one had a partial response as the best response and three had disease stabilization. Of the 14 evaluable patients with ovarian clear-cell carcinoma, four had a partial response as the best response and four had stable disease. Of the eight evaluable patients with endometrial carcinoma, three had partial responses as the best response and one had stable disease. Two of the eight evaluable patients with peripheral T-cell lymphoma had complete responses and one had a partial response. For the 21 evaluable patients with mesothelioma, there were three had partial responses as the best response and ten disease stabilizations. Of the ten evaluable patients with metastatic castration-resistant prostate cancer, six had stable disease. In the safety analysis set, 80 patients (98.8%) had at least one treatment-emergent AE (TEAE). The most frequent treatment-emergent adverse events (TEAEs) determined to be possibly related to tulmimetostat included thrombocytopenia (50.6%), diarrhea (45.7%), anemia (35.8%), nausea (33.3%), fatigue (32.1%), alopecia (27.2%), dysgeusia (24.7%), vomiting (22.2%), neutropenia (16.0%), decreased appetite (14.8%), and decreased weight (12.3%). TEAEs led to dose reductions in 31 patients (38.3%) and to dose interruptions in 57 patients (70.4%). Fourteen patients (17.3%) discontinued treatment due to AEs.
62
In September 2023, the FDA granted Fast Track designation for tulmimetostat, for the treatment of patients with advanced, recurrent, or metastatic endometrial cancer harboring AT-rich interacting domain-containing protein 1A (ARID1A) mutations and who have progressed on at least one prior line of treatment. The FDA grants Fast Track designation to facilitate the development and expedite the review of medicines intended to treat serious conditions and potentially address an unmet medical need, with the goal of getting these important, new therapies to patients earlier.
During the IGCS (International Gynecologic Cancer Society) 2023 Annual Global Meeting held in Seoul, South Korea, in November 2023, MorphoSys showcased in a featured poster abstract session, updated preliminary Phase 2 clinical data and first biomarker findings in a subset of patients with ARID1A-mutated ovarian clear-cell or endometrial carcinomas. At cutoff date (July 16, 2023), of the 89 patients enrolled in the Phase 2 study, efficacy data from 14 evaluable patients with ovarian clear-cell carcinoma and 11 evaluable patients with endometrial carcinoma were presented; >50% of each cohort have received ≥3 prior treatment lines. Of the 14 evaluable patients with ovarian clear-cell carcinoma, the best confirmed response was a partial response in one patient and stable disease in seven patients and of the 11 evaluable patients with endometrial carcinoma, four patients had a best confirmed response of PR and two patients had stable disease. The manageable safety profile across all 6 tumor cohorts (n = 89) was consistent with known class effects; Thrombocytopenia (in 50.6% patients) was the most frequent hematologic TEAE considered at least possibly related to tulmimetostat and diarrhea (in 51.7%) was the most frequent non-hematologic TEAE considered at least possibly related to tulmimetostat. Next generation sequencing did not reveal a specific hotspot for ARID1Amut locations impacting clinical outcome in patients with ovarian clear-cell or endometrial carcinoma. These efficacy, safety and biomarker data support further investigation of this dual inhibitor.
Clinical Development by Partners
The most advanced programs being developed by partners are outlined below.
Ianalumab
Ianalumab (VAY736) is a fully human IgG1/k mAb with a dual mode of action targeting B-cell lysis and BAFF-R blockade that is being investigated by Novartis in multiple indications within the immunology and hematology field. Ianalumab is currently in Phase 3 clinical development in lupus nephritis (LN), Sjögren’s disease, systemic lupus erythematosus (SLE), immune thrombocytopenia (1L and 2L ITP), and warm autoimmune hemolytic anemia (wAIHA). Ianalumab is also in Phase 2 clinical development in autoimmune hepatitis (AIH). MorphoSys is entitled to milestone payments and royalties upon approval and commercialization.
Abelacimab
Abelacimab (MAA868) is an antibody directed against Factor XI that is being investigated by Anthos Therapeutics in two complementary Phase 3 clinical studies in cancer-associated thrombosis (CAT) for the prevention of venous thromboembolism (VTE) and in one Phase 3 study in high-risk patients with atrial fibrillation (AF). The FDA granted Fast Track designation to abelacimab for both indications under study. In September 2023, Anthos Therapeutics announced that the AZALEA-TIMI 71 Phase 2 study in atrial fibrillation at moderate-to-high risk of stroke has been stopped early due to an overwhelming benefit (reduction in bleeding compared to standard-of-care direct oral anticoagulant. MorphoSys is entitled to milestone payments and royalties upon approval and commercialization.
Setrusumab
Setrusumab (BPS804/UX143) is a fully human monoclonal antibody inhibiting sclerostin that is currently being investigated by Ultragenyx and Mereo BioPharma in the Phase 3 portion of the pivotal Phase 2/3 clinical study and a Phase 3 study for the treatment of osteogenesis imperfecta. MorphoSys is entitled to milestone payments and royalties upon approval and commercialization.
Bimagrumab
Bimagrumab is a fully human monoclonal antibody against activin type II receptors that is currently in clinical development. Lilly is investigating bimagrumab in a global Phase 2b study in patients with obesity and announced completion of enrollment in June 2023. MorphoSys is entitled to milestone payments and royalties upon approval and commercialization.
Felzartamab
Felzartamab is a therapeutic human monoclonal antibody directed against CD38. Human Immunology Biosciences, Inc. (HI-Bio) obtained exclusive rights to develop and commercialize felzartamab across all indications worldwide, with the exception of Greater China. HI-Bio is evaluating felzartamab for patients with two renal autoimmune diseases, anti-PLA2R antibody-positive membranous nephropathy (M-PLACE and New-PLACE trials), and immunoglobulin A nephropathy (IGNAZ trial).
63
On May 25, 2023, HI-Bio announced that the FDA has granted orphan drug designation (ODD) for felzartamab in development for the treatment of membranous nephropathy (MN). On October 31, 2023, HI-Bio announced that the FDA has granted Breakthrough Therapy designation for felzartamab in primary membranous nephropathy (PMN). The FDA selectively grants Breakthrough Therapy designation to expedite the development and review of drugs that are intended to treat a serious or life-threatening condition and preliminary clinical evidence indicates the drug may demonstrate substantial improvement over available therapy on a clinically significant endpoint(s). HI-Bio initiated an open label Phase 1b study in patients with lupus nephritis (LN) end of 2023. In addition, felzartamab is also under investigation in a randomized, controlled, double-blind pilot Phase 2 trial for chronic Antibody Mediated Transplant Rejection (AMR), this is an investigator initiated trial (IIT).
I-Mab Biopharma holds the exclusive regional rights to develop and commercialize felzartamab in Greater China and is studying felzartamab in relapsed/refractory multiple myeloma. MorphoSys will be eligible to receive payments on achievement of development, regulatory, and commercial milestones in addition to royalties on net sales of felzartamab.
MOR210/TJ210/HIB210
MOR210/TJ210/HIB210 is a human antibody directed against C5aR1, the receptor of the complement factor C5a. HI-Bio obtained exclusive worldwide rights to develop and commercialize MOR210 across all indications worldwide, with the exception of Greater China and South Korea. In July 2023, HI-Bio announced that the first participants have been dosed in a Phase 1 healthy volunteer study of HIB210.
I-Mab Biopharma holds the exclusive rights for MOR210 in Greater China and South Korea and is currently investigating MOR210 for autoimmune diseases after Phase 1 trial in solid tumors completion. MorphoSys will be eligible to receive payments on achievement of development, regulatory, and commercial milestones in addition to royalties on net sales of MOR210/TJ210/HIB210.
Other Programs (Selection)
In addition to the partnered programs listed above, there are several additional partnered programs in early to mid-stage research and development, amongst others CMK389/NOV-8.
Collaboration and License Agreements
A core component of our business model is entering into collaboration and/or license agreements with leading global pharmaceutical and biotechnology companies to enable expanded development and commercialization of existing programs or gain access to external innovation via in-licensing. Many of these agreements are entered into in the ordinary course of our business and may or may not become significant or material to us, depending primarily on the development of the underlying product candidate(s).
Generally, our collaboration and license agreements may be for a specific therapeutic program or may be for multiple therapeutic programs across diseases. For programs that we out-license, we may participate in the generation and development of an antibody for a specified target and will have limited preclinical and clinical research and development obligations, with the licensee being primarily responsible for clinical development and commercialization. In general, pursuant to the collaboration agreements we enter into for programs that we out-license, most of our partners have the first right to prosecute, maintain and enforce patents for antibodies (and other patentable technology) developed with our technology. If our partners determine to relinquish any such patent right, we generally have a first right to obtain ownership of such patents. We are generally entitled to milestone payments during the course of development of the therapeutic product and to royalty payments upon the commercial sale of the products. The royalty term generally will be on a product-by-product and country-by-country basis starting on the first commercial sale and ending on the later of: (i) the expiration of certain specified patent rights, (ii) a certain defined period of years following the first commercial sale, or (iii) the expiration of regulatory exclusivity. The agreements will generally terminate or expire once the obligation of the licensee to pay royalties has ceased.
Below is a description of our current significant, or material, collaboration and license agreements.
Collaboration and License Agreement with Xencor
In June 2010, we entered into a collaboration and license agreement with Xencor which was subsequently amended (which we refer to, as amended, as the Xencor Collaboration Agreement). Under the Xencor Collaboration Agreement, Xencor granted us an exclusive, worldwide license, including the right to sublicense under certain conditions, for tafasitamab.
Under the terms of the agreement, Xencor initiated and sponsored a Phase 1 clinical trial for tafasitamab in patients with CLL which was completed in January 2013. Following the completion of such clinical trial, we were responsible for all additional clinical development and commercialization of tafasitamab until the sale and transfer of the asset to Incyte in February 2024.
64
Xencor already received an upfront payment of US$ 13.0 million (approximately € 10.5 million) and received US$ 65.5 million (approximately € 53.8 million) for development milestones under the Xencor Collaboration Agreement and was entitled to receive up to an additional US$ 236.5 million in aggregated milestone payments upon the achievement of certain development events including US$ 50 million in the aggregate with respect to sales of licensed antibody products. Furthermore, Xencor was also eligible to receive tiered royalty payments of tafasitamab in the mid single-digit to sub-teen double-digit percentage range based upon net sales of licensed antibody sold by us or our licensees.
Under the Xencor Collaboration Agreement, Xencor retained the rights to prosecute, maintain and enforce certain patents licensed to us, including those patents licensed to us that were already filed as of the effective date of the Xencor Collaboration Agreement and whose claims cover tafasitamab and certain other antibodies. We retained the right to prosecute, maintain and enforce patents that cover tafasitamab and no other antibody. Furthermore, Xencor retained the rights to prosecute, maintain and enforce certain patents directed to inventions developed under the Xencor Collaboration Agreement that were solely invented by or on behalf of Xencor.
In February 2024, Incyte obtained exclusive rights worldwide to tafasitamab. Incyte will assume full responsibility and cover all costs going forward for the development and commercialization of the asset. As part of the sale and transfer of tafasitamab to Incyte, Incyte also assumed the Collaboration and License Agreement with Xencor.
Collaboration and license agreement with Incyte
On January 13, 2020, we entered into a collaboration and licensing agreement with Incyte to further develop and commercialize our proprietary anti-CD19 antibody tafasitamab globally. Under the terms of the agreement, MorphoSys received an upfront payment of US$ 750 million. In addition, Incyte made an equity investment into MorphoSys of US$ 150 million in new American Depositary Shares (ADS) of MorphoSys at a premium to the share price at signing of the agreement. Depending on the achievement of certain developmental, regulatory and commercial milestones, MorphoSys is eligible to receive milestone payments amounting up to US$ 1.1 billion. MorphoSys also receives tiered royalties on ex-U.S. net sales of tafasitamab in a mid-teens to mid-twenties percentage range.
In the U.S., MorphoSys and Incyte co-commercialized Monjuvi®, with MorphoSys leading the commercialization strategy and booking all revenues from sales of tafasitamab. Incyte and MorphoSys were jointly responsible for commercialization activities in the U.S. and share profits and losses on a 50:50 basis. Outside the U.S., Incyte had exclusive commercialization rights, and lead the commercialization strategy and booked all revenues from sales of tafasitamab, paying MorphoSys royalties on ex-U.S. net sales.
Furthermore, the companies shared development costs associated with global and U.S.-specific trials at a rate of 55% (Incyte) and 45% (MorphoSys); Incyte covers 100% of the development costs for trials that are specific to ex-U.S.countries.
The agreement between MorphoSys and Incyte, including the equity investment, received clearance by the U.S. antitrust authorities under the Hart-Scott-Rodino Act as well as by the German and Austrian antitrust authorities on or before March 2, 2020, and became effective on March 3, 2020.
In February 2024, Incyte obtained exclusive rights worldwide to tafasitamab. Incyte will assume full responsibility and cover all costs going forward for the development and commercialization of the asset.
Equity participation agreement and license agreement with HI-Bio
In June 2022, MorphoSys entered into an equity participation agreement and license agreements with HI-Bio. Under the terms of the agreements, HI-Bio obtained exclusive rights to develop and commercialize felzartamab and MOR210 across all indications worldwide, with the exception of Greater China for felzartamab and Greater China and South Korea for MOR210. As part of the agreements, MorphoSys received a 15% equity stake in HI-Bio, along with certain equity earn-in provisions to mitigate dilution in further financing rounds and standard investment rights. MorphoSys is represented as a member of HI-Bio’s Board of Directors. On achievement of development, regulatory and commercial milestones, MorphoSys will be eligible to receive payments from HI-Bio of up to US$ 1 billion across both programs, in addition to tiered, single- to low double-digit royalties on net sales of felzartamab and MOR210 and will be compensated for ongoing program expenses. HI-Bio will assume full responsibility for future development and commercialization expenses. As of December 31, 2023, the equity stake in HI-Bio amounted to 12.1%, due to dilutive capital raises conducted in financial year 2023 and a partial sale of shares to a third party.
Intellectual property
Our commercial success depends in part on our ability to obtain and maintain proprietary or intellectual property protection for our product candidates and our core technologies and other know-how to operate without infringing, misappropriating or otherwise violating the proprietary rights of others and to prevent others from infringing, misappropriating or otherwise
65
violating our proprietary or intellectual property rights. We protect our proprietary and intellectual property position by, among other methods, licensing or filing of patent applications covering our proprietary technologies and products in our home country and all major markets, with a particular emphasis on the United States. We also rely on trade secrets, know-how and continuing technological innovation to develop and maintain our proprietary and intellectual property position, which we generally seek to protect through contractual obligations with third-parties.
Patents
Patents, patent applications and other intellectual property rights are important in the sector in which we operate. We consider on a case-by-case basis filing patent applications with a view to protecting certain innovative technologies, products, processes, and methods of treatment. We may also license or acquire rights to patents, patent applications or other intellectual property rights owned by third-parties, academic partners or commercial companies, which are of interest to us or necessary for our business.
As with other biotechnology and pharmaceutical companies, our ability to maintain and solidify our proprietary and intellectual property position for our product candidates and technologies will depend on our success in obtaining effective patent claims and enforcing those claims if granted. However, our pending patent applications, and any patent applications that we may in the future file or license from third-parties may not result in the issuance of patents. We also cannot predict the breadth of claims that may be allowed or enforced in our patents or whether the claims of any issued patent will provide sufficient proprietary protection from competitors. Any issued patents that we may receive or license in the future may be challenged, invalidated or circumvented. For example, we cannot be certain of the priority of our patents and patent applications over third-party patents and patent applications. In addition, because of the extensive time required for clinical development and regulatory review of a product candidate we may develop, it is possible that, before any of our product candidates can be commercialized, any related patent may expire or remain in force for only a short period following commercialization, thereby limiting the protection such patent would afford the respective product and any competitive advantage such patent may provide.
At the end of the financial year 2023, we maintained over 110 different proprietary patent families worldwide in addition to the numerous patent families we pursue with our partners.
HuCAL
Our HuCAL platform patent portfolio is wholly owned, and the platform is protected by several patent families. The basic HuCAL patents covering the composition of the library, methods to isolate antibodies from the library and methods to diversify antibodies isolated from the library expired in August 2016. Additional patent families protecting other technological aspects of the library, such as the specific CDR design (based on WO2008/053275) and certain improved display methods used with the library (based on WO2009/024593) are still in force in major jurisdictions, including Australia, Canada, China, the European Union (EP2190987), Israel, Japan, New Zealand, South Africa and the United States. The last U.S. patent (US9062097) expires on February 18, 2030. Patents in other jurisdictions expire in August 2028. The HuCAL library is also protected by considerable know-how proprietary to us.
Ylanthia
Our Ylanthia antibody library patent portfolio is wholly owned, and the platform is protected by two key patent families covering the composition of the library and nucleic acid collections encoding the library. Patent applications (based on WO2010/136598 and WO2012/066129) are filed in major jurisdictions, including Australia, Canada, China, the European Union, Hong Kong, India, Israel, Japan, Mexico, New Zealand, Russia, Singapore, South Africa, South Korea and the United States. Exemplary patents include EP2640742, US8367586, and US9541559. The patent term is expected to last at least until November 2031. One material U.S. patent, US9541559, expires on May 6, 2032. Additional patent families relate to ancillary technologies. The Ylanthia library also encompasses considerable know-how proprietary to us.
Tafasitamab
The original tafasitamab patent portfolio is exclusively licensed from Xencor. Over the years the portfolio emerged and now comprises over a dozen patent families covering various aspects of the molecule, its compositions, methods of use, combination treatments, and formulation, as well as other aspects. In January 2020, a collaboration and license agreement with Incyte Corporation was signed. The parties file, prosecute and, if necessary enforce and defend, the patent rights jointly. Xencor also has a first right to file, prosecute and enforce certain patent rights related to tafasitamab.
The basic composition-of-matter patent family was in-licensed from Xencor and applications were filed in Australia, Canada, the European Union, Hong Kong, India, Japan and the United States. The expiry date for the composition of matter patent is August 2029 for the United States and August 2027 for the other countries, not including any potential patent term extensions.
66
Respective patent term extension (PTE) in the U.S., and supplementary protection certificates (SPC) in Europe have been filed in response to respective approvals.
Other patent families were filed and are prosecuted in Australia, Brazil, Canada, China, the European Union, Israel, India, Japan, Mexico, New Zealand, Qatar, Russia, Singapore, South Africa, South Korea, the United States as well as in other additional countries.
As a consequence of the sale and transfer of tafasitamab to Incyte in February 2024, all patents related to tafasitamab have been transferred to Incyte.
Pelabresib
The main patents for pelabresib run until 2032 (U.S.) and 2031 (Europe), not including possible extension through supplemental protection certificates or term extensions. In addition, the use of pelabresib for the treatment of myelofibrosis is patent-protected in the U.S. until 2039.
Felzartamab
Our felzartamab patent portfolio is fully owned. Rights to the Greater Chinese territory were exclusively licensed to I-Mab and rights to the rest of the world were exclusively licensed to HI-Bio in 2022. The program is currently protected by about ten different patent families covering various aspects of the molecule, its compositions, combination treatments, dosage regimens, radioconjugates, as well as assays utilized in clinical development. The basic composition-of-matter patent expires in October 2026, outside the United States, and in January 2028, in the United States, in both cases not including any potential patent term extensions. Patent applications were filed and are prosecuted in Argentina, Australia, Brazil, Canada, China, the European Union, Hong Kong, Israel, India, Japan, Mexico, New Zealand, Russia, Singapore, South Africa, South Korea, Taiwan and the United States.
Tulmimetostat
The main patents for tulmimetostat have a term until 2039. Here, too, a possible extension through supplementary protection certificates or term extensions is not included.
Patent Term
The term of an individual patent depends upon the legal term for patents in the countries in which such patent is granted. In most countries, including the United States, the patent term is 20 years from the earliest filing date of a non-provisional patent application to which the patent claims priority. In the United States, a patent’s term may, in certain cases, be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the USPTO in examining and granting a patent, or may be shortened if a patent is terminally disclaimed over a commonly owned patent or a patent naming a common inventor and having an earlier expiration date. The Hatch-Waxman Act permits a patent term extension of up to five years beyond the expiration date of a U.S. patent as partial compensation for the length of time the product is under regulatory review while the patent is in force. The length of the patent term extension is related to the length of time the product is under regulatory review. Patent term extension cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval and only one patent applicable to an approved product may be extended. Similar provisions are available in other jurisdictions to extend the term of a patent that covers an approved product, or to offer similar protection for an extended period, as is the case in the European Union. In the future, if and when our product candidates receive approval from the U.S. FDA or other regulatory authorities, we expect to apply for patent term extensions on patents covering those products where such extensions are available; however there is no guarantee that the applicable authorities, including the U.S. FDA, will agree with our assessment of whether such extensions should be granted and, even if granted, the length of such extensions.
Trade Secrets
In addition to patents, we rely upon unpatented trade secrets and know-how and continuing technological innovation to develop and maintain our competitive position. However, trade secrets and know-how can be difficult to protect. We seek to protect our proprietary information, in part, by executing confidentiality agreements with our partners, collaborators, scientific advisors, employees, consultants and other third-parties, and invention assignment agreements with our consultants and employees. We have also executed agreements requiring assignment of inventions with selected scientific advisors and collaborators. The confidentiality agreements we enter into are designed to protect our proprietary information and the agreements or clauses requiring assignment of inventions to us are designed to provide mechanisms to grant us ownership of technologies that are developed through our relationship with the respective counterparty. We cannot guarantee that we have entered into such agreements with each party that may have or have had access to our trade secrets or proprietary technology and processes or that these agreements will afford us adequate protection of our intellectual property and proprietary information rights. If any of
67
the partners, collaborators, scientific advisors, employees and consultants who are parties to these agreements breaches or violates the terms of any of these agreements or otherwise discloses our proprietary information, we may not have adequate remedies for any such breach or violation, and we could lose our trade secrets as a result.
Trademarks and Domain Names
We conduct our business using trademarks with various forms of the “MorphoSys” brand and numerous additional trademarks, as well as domain names incorporating some or all of these trademarks. Key trademarks are protected in all major jurisdictions, including the United States, the European Union, Switzerland, Canada, Australia and Japan. Additionally we have protected the brand names of tafasitamab, Monjuvi® and Minjuvi® in all key jurisdictions worldwide. Such protection includes the filing of trademarks, as well as the registration of domain names. As a consequence of the sale and transfer of tafasitamab to Incyte in February 2024, all trademarks and domain names related to tafasitamab have been transferred to Incyte.
Manufacturing
We have adopted a manufacturing strategy of contracting with third-parties in accordance with cGMP for the manufacture of drug substances and products as well as active pharmaceutical ingredients, or API. Additional contract manufacturers are used to fill, label, package, and distribute investigational drug products and commercial product as well as tableting APIs. This allows us to maintain a more flexible infrastructure while focusing our expertise on developing our products. We will ultimately depend on contract manufacturers, or CMOs, for the manufacture of our products for commercial sale, as well as for clinical development and technical process development. CMOs are subject to extensive governmental regulation.
We have selected industry-leading partners for warehousing, distribution and logistics in the U.S.
U.S. Commercial Operations
MorphoSys’ commercial activities were focused on Monjuvi® in the United States; the Company was co-commercializing this product with Incyte.
On July 31, 2020, Monjuvi® (tafasitamab-cxix) in combination with lenalidomide was approved under accelerated approval by the U.S. FDA for the treatment of adult patients with relapsed or refractory (r/r) diffuse large B-cell lymphoma (DLBCL) not otherwise specified, including DLBCL arising from low-grade lymphoma, and who are not eligible for autologous stem cell transplant (ASCT). This was the first U.S. FDA approval of a second-line treatment for adult patients with r/r DLBCL in the U.S. Monjuvi® is accessible to patients in both community care and academic settings as an in-office outpatient targeted immunotherapy given as intravenous infusion that does not require hospitalization or heavy monitoring. Upon approval, MorphoSys and Incyte launched “My Mission Support,” a robust patient support program offering financial assistance, ongoing education, and other resources to eligible patients who are prescribed Monjuvi® in the U.S. The program was launched to support patients throughout their treatment journeys and to help lower patient access barriers.
Monjuvi® has been included in the National Comprehensive Cancer Network® Clinical Practice Guidelines (NCCN Guidelines®) in Oncology for B-cell Lymphomas since August 2020. The NCCN Guidelines were updated in the United States in March 2022 to include Monjuvi® in combination with lenalidomide as a preferred treatment option in the second-line setting (Category 2A designation). Inclusion in these guidelines increases awareness of a product within the oncology community and also drives certain formulary decisions. As of April 1, 2021, Monjuvi® was granted a J-code, further simplifying reimbursement for some treatment centers.
MorphoSys and Incyte saw a high penetration in the community setting; more than 70% of ordering sites have been in the community setting, with the balance coming from the academic setting. Since launch, the Company, along with its partner Incyte, has in aggregate received orders from more than 1,700 treatment sites. During the fourth quarter of 2023, more than 600 accounts ordered with approximately 90% of those accounts representing repeat orders. While we also saw positive trends year-over-year and sequentially, we saw intensifying competition, including recent approvals of new treatment options in second and later-line settings for relapsed or refractory diffuse large B-cell lymphoma.
In February 2024, Incyte obtained exclusive rights worldwide to tafasitamab. Incyte will assume full responsibility and cover all costs going forward for the development and commercialization of the asset.
Competition
We compete in an industry that is characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. Our competitors include pharmaceutical companies, biotechnology companies, academic institutions and other research organizations. There are many major pharmaceutical and biotechnology companies developing or marketing treatments for cancer disorders that may compete with us in the search and development of novel therapeutic targets.
68
Many of our competitors have significantly greater financial, manufacturing, marketing, drug development, technical and human resources than we do. Mergers and acquisitions in the pharmaceutical, biotechnology and diagnostic industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These companies also compete with us in recruiting and retaining top qualified scientific and management personnel and establishing clinical study sites and patient registration for clinical studies, as well as in acquiring technologies complementary to, or necessary for, our programs or in in-licensing or acquiring promising technologies or compounds.
The key competitive factors affecting the success of all of our therapeutic product candidates, if approved, are likely to be their efficacy, safety, dosing convenience, price, the effectiveness of companion diagnostics in guiding the use of related therapeutics, our marketing capabilities, the level of generic competition and the availability of reimbursement from government and other third-party payors. Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, have fewer or less severe side effects, are more convenient or are less expensive than any products that we may develop. Our competitors also may obtain U.S. FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market. In addition, our ability to compete may be affected in many cases by insurers or other third-party payors seeking to encourage the use of biosimilar or generic products.
The most common methods of treating patients with cancer are surgery, radiation and drug therapy. There are a variety of available drug therapies marketed for cancer. In many cases, these drugs are administered in combination to enhance efficacy. While our product candidates may compete with many existing drug and other therapies, to the extent they are ultimately used in combination with or as an adjunct to these therapies, our product candidates could not be competitive with them as such. Some of the currently approved drug therapies are branded and subject to patent protection, and others are available on a generic basis. Many of these approved drugs are well established therapies and are widely accepted by physicians, patients and third-party payors.
In addition to currently marketed therapies, there are also a number of products in late-stage clinical development to treat cancer. These product candidates in development may provide efficacy, safety, dosing convenience and other benefits that are not provided by currently marketed therapies or our drugs. As a result, they may provide significant competition for any of our product candidates for which we obtain marketing approval. If our lead product candidates are approved for the indications for which we are currently undertaking clinical studies, they will compete with the therapies and currently marketed drugs discussed elsewhere in this document.
Government Regulation
Government authorities in the United States, at the federal, state and local level, and in the European Union and other countries and jurisdictions, extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, post-approval monitoring and reporting, and import and export of drug and biological products. In addition, some jurisdictions regulate the pricing of pharmaceutical products. The processes for obtaining marketing approvals in the United States and in foreign countries and jurisdictions, along with subsequent compliance with applicable statutes and regulations and other requirements of regulatory authorities, require the expenditure of substantial time and financial resources.
Regulation and Procedures Governing Approval of Drug and Biological Products in the United States
In the United States, the U.S. FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act, or FDCA, and its implementing regulations and biologics under the FDCA, the Public Health Service Act, or PHSA, and their implementing regulations. Both drugs and biologics are also subject to other federal, state, and local statutes and regulations. The failure to comply with the applicable U.S. requirements at any time during the product development process, including during non-clinical testing, clinical testing or the approval process, may subject an applicant to delays in the conduct of a study or regulatory review and approval, and/or to administrative or judicial sanctions and adverse publicity. Sanctions may include, but are not limited to, the U.S. FDA’s refusal to allow an applicant to proceed with clinical testing, refusal to approve pending applications, withdrawal of an approval, warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, debarment, disgorgement of profits and civil or criminal investigations and penalties brought by the U.S. FDA or the Department of Justice or other governmental entities.
Our product candidates and any future product candidates must be approved by U.S. FDA through a biologics license application, or BLA, or new drug application, or NDA, process before they may be legally marketed in the United States. The process generally involves the following:
•Non-clinical laboratory tests, animal studies and formulation studies all performed in accordance with applicable regulations, including the U.S. FDA’s good laboratory practices, or GLP, regulations;
69
•submission to the U.S. FDA of an investigational new drug, or IND, application for human clinical testing, which must become effective before human clinical trials may begin;
•approval by an IRB representing each clinical site before each clinical trial may be initiated;
•performance of adequate and well-controlled human clinical trials to establish the safety, potency and purity of the product candidate for each proposed indication, in accordance with applicable regulations, including with good clinical practices, or GCP, regulations;
•preparation and submission to the U.S. FDA of a BLA or an NDA;
•review of the product by an U.S. FDA advisory committee, if applicable;
•satisfactory completion of one or more U.S. FDA inspections of the manufacturing facility or facilities, including those of third-parties, at which the product, or components thereof, are produced to assess compliance with current good manufacturing practices, or cGMP, requirements and to assure that the facilities, methods and controls are adequate to preserve the product’s identity, strength, quality and purity;
•satisfactory completion of any U.S. FDA audits of the clinical study sites to assure compliance with GCPs, and the integrity of clinical data in support of the BLA or NDA;
•satisfactory completion of one or more U.S. FDA inspections of the sponsor to demonstrate oversight of all relevant GxP activties outsourced to sites, CROs and CMOs.
•payment of user fees and securing U.S. FDA approval of the BLA or NDA;
•compliance with any post-approval requirements, including the potential requirement to implement a REMS and to conduct any post-approval studies required by the U.S. FDA; and
•compliance with post marketing requirements, or PMR, post marketing commitments, or PMC, and advertising and promotion regulation.
Non-clinical Studies and Investigational New Drug (IND) Application
Before testing any drug or biologic product candidate in humans, the product candidate must undergo non-clinical testing. Non-clinical tests include laboratory evaluations of product chemistry, formulation and stability, as well as animal studies to evaluate the potential for activity and toxicity. The conduct of the non-clinical tests and formulation of the compounds for testing must comply with federal regulations and requirements. The results of the non-clinical tests, together with manufacturing information, analytical data, any available clinical data or literature and a proposed clinical protocol, are submitted to the U.S. FDA as part of an IND application. The IND automatically becomes effective 30 days after receipt by the U.S. FDA, unless before that time the U.S. FDA raises concerns or questions about the product or conduct of the proposed clinical trial, including concerns that human research subjects will be exposed to unreasonable health risks, and places the trial on a clinical hold. In that case, the IND sponsor and the U.S. FDA must resolve any outstanding U.S. FDA concerns before the clinical trial can begin.
As a result, submission of the IND may result in the U.S. FDA not allowing the trial to commence or not be conducted on the terms originally specified by the sponsor in the IND. If the U.S. FDA raises concerns or questions either during this initial 30-day period, or at any time during the IND process, it may choose to impose a partial or complete clinical hold. If the U.S. FDA imposes a clinical hold, trials may not recommence without U.S. FDA authorization and then only under terms authorized by the U.S. FDA. A clinical hold issued by the U.S. FDA may therefore delay either the commencement of a proposed clinical study or cause suspension of an ongoing study, until all outstanding concerns have been adequately addressed and the U.S. FDA has notified the company that investigation may proceed. This could cause significant difficulties in completing planned clinical trials in a timely manner.
The U.S. FDA may impose clinical holds on a drug or biologic product candidate at any time before or during clinical trials due to safety concerns or non-compliance.
Clinical Trials
Clinical trials involve the administration of the investigational product candidate to healthy volunteers or patients with the disease to be treated under the supervision of a qualified principal investigator in accordance with GCP requirements. Clinical trials are conducted under study protocols detailing, among other things, the objectives of the study, inclusion and exclusion criteria, the parameters to be used in monitoring safety, dosing procedures and the effectiveness criteria to be evaluated. A protocol for each clinical trial and any subsequent protocol amendments must be submitted to the U.S. FDA as part of the IND.
70
A sponsor who wishes to conduct a clinical trial outside the United States may, but need not, obtain U.S. FDA authorization to conduct the clinical trial under an IND. If a foreign clinical trial is not conducted under an IND, the sponsor may submit data from the clinical trial to the U.S. FDA in support of the BLA or NDA so long as the clinical trial is well-designed and well-conducted in accordance with GCP, including review and approval by an independent ethics committee, and the U.S. FDA is able to validate the study data through an onsite inspection, if necessary.
Further, each clinical trial must be reviewed and approved by an IRB or ethics committee (EC), either centrally or individually at each institution at which the clinical trial will be conducted. The IRB will consider, among other things, clinical trial design, patient informed consent, ethical factors and the safety of human subjects. An IRB must operate in compliance with U.S. FDA regulations. The U.S. FDA, IRB, or the clinical trial sponsor may suspend or discontinue a clinical trial at any time for various reasons, including a finding that the clinical trial is not being conducted in accordance with U.S. FDA requirements or that the subjects or patients are being exposed to an unacceptable health risk. Clinical testing also must satisfy extensive GCP rules and the requirements for informed consent. The IRB also approves the form and content of the informed consent that must be signed by each clinical trial subject or his or her legal representative and must monitor the clinical trial until completed.
Additionally, some clinical trials are overseen by an independent group of qualified experts organized by the clinical trial sponsor, known as a data safety monitoring board or committee. This group may recommend the continuation of the study as planned, changes in study conduct, or cessation of the study at designated checkpoints based on access to certain data from the study.
Clinical trials typically are conducted in three sequential phases, but the phases may overlap or be combined. Additional studies may be required after approval.
Phase 1 clinical trials (or Phase 1) are initially conducted in a very limited population to test the product candidate for safety, including adverse effects, dose tolerance, absorption, metabolism, distribution, excretion and pharmacodynamics in healthy humans or, on occasion, in patients, such as in the case of some products for severe or life-threatening diseases, especially when the product may be too inherently toxic to ethically administer to healthy volunteers.
Phase 2 clinical trials (or Phase 2) are generally conducted in a limited patient population to identify possible adverse effects and safety risks, preliminarily evaluate the efficacy of the product candidate for specific targeted indications and determine dose tolerance and optimal dosage. Multiple Phase 2 clinical trials may be conducted by the sponsor to obtain information prior to beginning larger Phase 3 clinical trials.
Phase 3 clinical trials (or Phase 3) proceed if Phase 2 clinical trials demonstrate that a certain dose or dose range of the product candidate is potentially effective and has an acceptable safety profile. Phase 3 clinical trials are undertaken within an expanded patient population, often at geographically dispersed clinical trial sites, to gather additional information about safety and effectiveness necessary to evaluate the overall benefit-risk relationship of the product and to provide an adequate basis for physician labeling.
In some cases, the U.S. FDA may approve a BLA or NDA for a product candidate but require the sponsor to conduct additional clinical trials to further assess the product candidate’s safety and effectiveness after approval. Monjuvi® has been approved under accelerated approval by the U.S. FDA. Continued approval may be contingent upon verification and description of clinical benefit in a confirmatory trial. Such post-approval trials are typically referred to as Phase 4 clinical trials (or Phase 4). These studies may be used to gain additional experience from the treatment of patients in the intended therapeutic indication and to document a clinical benefit in the case of drugs or biologics approved under accelerated approval regulations. If the U.S. FDA approves a product while a company has ongoing clinical trials that were not necessary for approval, a company may be able to use the data from these clinical trials to meet all or part of any Phase 4 clinical trial requirement or to request a change in the product labeling. Failure to exhibit due diligence with regard to conducting required Phase 4 clinical trials could result in withdrawal of approval for products.
During all phases of clinical development, regulatory agencies require extensive monitoring and auditing of all clinical activities, clinical data, and clinical trial investigators. Annual progress reports detailing the results of the clinical trials must be submitted to the U.S. FDA. Written IND safety reports must be promptly submitted to the U.S. FDA and the investigators for serious and unexpected adverse events, any findings from other trials, tests in laboratory animals or in vitro testing that suggest a significant risk for human subjects, or any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator brochure. The sponsor must submit an IND safety report within 15 calendar days after the sponsor determines that the information qualifies for reporting. The sponsor also must notify the U.S. FDA of any unexpected fatal or life-threatening suspected adverse reaction within seven calendar days after the sponsor’s initial receipt of the information. The U.S. FDA or the sponsor or its data safety monitoring board may suspend a clinical trial at any time on various grounds, including a finding that the research subjects or patients are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s or GCP requirements or if the biological candidate has been associated with unexpected serious harm to patients.
71
There are also requirements governing the reporting of ongoing clinical trials and completed clinical trial results to public registries. Information about certain clinical trials including clinical trial results, must be submitted within specific timeframes for publication on the www.clinicaltrials.gov website.
Compliance with cGMP Requirements
Before approving a BLA or NDA, the U.S. FDA typically will inspect the facility or facilities where the product is manufactured. The U.S. FDA will not approve an application unless it determines that the manufacturing processes and facilities are in full compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. For example, for pharmaceutical products, among other things, the sponsor must develop methods for testing the identity, strength, quality, potency and purity of the final pharmaceutical product. Additionally, appropriate packaging must be selected and tested and stability studies must be conducted to demonstrate that the pharmaceutical product does not undergo unacceptable deterioration over its shelf life.
Manufacturers and others involved in the manufacture, packaging and distribution of drug or biological products, and those supplying products, ingredients and components of them, must also register their establishments with the U.S. FDA and certain state agencies. Both domestic and foreign manufacturing establishments must register and provide additional information to the U.S. FDA upon their initial participation in the manufacturing process.
Establishments are subject to periodic announced and unannounced inspections by government authorities to ensure compliance with cGMPs and other laws. Manufacturers may have to provide, on request, electronic or physical records regarding their establishments. Delaying, denying, limiting, or refusing inspection by the U.S. FDA or having non-compliance with applicable regulations may lead to a product being deemed to be adulterated.
FDA Review Process
The results of product candidate development, non-clinical testing and clinical trials, including negative or ambiguous results as well as positive findings, are submitted to the U.S. FDA as part of a BLA or NDA. The BLA or NDA must contain extensive manufacturing information and detailed information on the composition of the product and proposed labeling. The U.S. FDA adjusts the Prescription Drug User Fee Act, or PDUFA, user fees on an annual basis. Fee waivers or reductions are available in certain circumstances, including a waiver of the application fee for the first application filed by a small business. Additionally, no user fees are assessed on BLAs or NDAs for products designated as orphan drugs, unless the product also includes a non-orphan indication.
The U.S. FDA has 60 days after submission of the application to conduct an initial review to determine whether the BLA or NDA is sufficient to accept for filing based on the agency’s threshold determination that it is substantially complete so as to permit substantive review. Once the submission has been accepted for filing, the U.S. FDA begins an in-depth review of the application. Under the goals and policies agreed to by the U.S. FDA under PDUFA, the U.S. FDA aims to complete its initial review of a standard application and respond to the applicant within ten months of the 60-day filing date, and for a priority review application within six months. The U.S. FDA does not always meet its PDUFA goal dates and its review goals are subject to change from time to time. The review process may often be significantly extended by U.S. FDA requests for additional information or clarification. The review process and the PDUFA goal date may also be extended by three months if the applicant provides a major new clinical safety/efficacy study report, a major re-analysis of a previously submitted study, or other major amendment during the review cycle.
The U.S. FDA reviews a BLA or NDA to determine, among other things, whether the proposed product is safe and potent, or effective, for its intended use, and has an acceptable purity profile, and whether the product is being manufactured in accordance with cGMP requirements to assure and preserve the product’s identity, safety, strength, quality, potency and purity. Similarly, the U.S. FDA reviews an NDA to determine, among other things, whether the drug is safe and effective and whether the facility in which it is manufactured, processed, packaged or held meets standards designed to assure the product’s continued safety, quality and purity. On the basis of the U.S. FDA’s evaluation of the application and accompanying information, including the results of the inspection of the manufacturing facilities and any U.S. FDA audits of clinical trial sites to assure compliance with GCPs, the U.S. FDA may issue an approval letter, denial letter, or a complete response letter. An approval letter authorizes commercial marketing of the product with specific prescribing information for specific indications. If the application is not approved, the U.S. FDA may issue a complete response letter, which will contain the conditions that must be met in order to secure final approval of the application, and when possible will outline recommended actions the sponsor might take to obtain approval of the application. If a complete response letter is issued, the applicant may resubmit the BLA or NDA, addressing all of the deficiencies identified in the letter, or may withdraw the application.
Sponsors that receive a complete response letter who elect to address the deficiencies may submit to the U.S. FDA information that represents a complete response to the issues identified by the U.S. FDA in the response letter. Such resubmissions are classified under PDUFA as either Class 1 or Class 2, based on the information submitted by an applicant in response to an
72
action letter. Under the goals and policies agreed to by the U.S. FDA under PDUFA, the U.S. FDA aims to review and act on a Class 1 resubmission with two months of receipt and, with respect to a Class 2 resubmission, within six months of receipt. The U.S. FDA will not approve an application until issues identified in the complete response letter have been addressed.
The U.S. FDA may also refer the application to an advisory committee for review, evaluation and recommendation as to whether the application should be approved and under what conditions. In particular, the U.S. FDA may refer applications for novel drug or biologic products or drug or biologic products that present difficult questions of safety or efficacy to an advisory committee. Typically, an advisory committee is a panel of independent experts, including clinicians and other scientific experts. The U.S. FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
If the U.S. FDA approves a new product, it may limit the approved indications for use of the product, or limit the approval to specific dosages. It may also require that certain contraindications, warnings or precautions be included in the product labeling. In addition, the U.S. FDA may call for post-approval studies, including Phase 4 clinical trials, to further assess the product’s safety after approval. The agency may also require testing and surveillance programs to monitor the product after commercialization, or impose other conditions, including distribution restrictions or other risk management mechanisms, including REMS, to help ensure that the benefits of the product outweigh the potential risks. REMS can include medication guides, communication plans for healthcare professionals, and elements to assure safe use, or ETASU. ETASU can include but are not limited to special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring and the use of patent registries. If the U.S. FDA concludes a REMS is needed, the sponsor of the BLA or NDA must submit a proposed REMS; the U.S. FDA will not approve the BLA or NDA without a REMS, if required. The U.S. FDA may prevent or limit further marketing of a product based on the results of post-market studies or surveillance programs. After approval, many types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further testing requirements and U.S. FDA review and approval.
Fast Track, Breakthrough Therapy and Priority Review Designations
The U.S. FDA may designate certain products for expedited review if they are intended to address an unmet medical need in the treatment of a serious or life-threatening disease or condition. These programs include fast track designation, breakthrough therapy designation and priority review designation.
The U.S. FDA may designate a product for fast track review if it is intended, whether alone or in combination with one or more other products, for the treatment of a serious or life-threatening disease or condition, and if based on non-clinical or clinical data it demonstrates the potential to address unmet medical needs for such a disease or condition. Fast track designation applies to the combination of the product and the specific indication for which it is being studied. The sponsor of a new drug or biologic product may request fast track designation by U.S. FDA at any time during the clinical development of the product. For fast track products, sponsors may have greater interactions with the U.S. FDA and the U.S. FDA may initiate a review of sections of a fast track product’s application before the application is complete. This rolling review may be available if the U.S. FDA determines, after preliminary evaluation of clinical data submitted by the sponsor, that a fast track product may be effective. The sponsor must also provide, and the U.S. FDA must approve, a schedule for the submission of the remaining information and the sponsor must pay applicable user fees. However, the U.S. FDA’s time period goal for reviewing a fast track application does not begin until the last section of the application is submitted. Fast track designation may be withdrawn by the U.S. FDA if the U.S. FDA believes that the designation is no longer supported by data emerging in the clinical trial process.
Additionally, a product may be designated by the U.S. FDA as a breakthrough therapy if it is intended, either alone or in combination with one or more other products, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The U.S. FDA may take certain actions with respect to breakthrough therapies, including holding meetings with the sponsor throughout the development process; providing timely advice to the product sponsor regarding development and approval; involving more senior staff in the review process; assigning a cross-disciplinary project lead for the review team; and taking other steps to facilitate the design of clinical trials in an efficient manner.
The U.S. FDA may designate a product for priority review if it is a product that treats a serious condition and, if approved, would provide a significant improvement in safety or effectiveness. The U.S. FDA determines, on a case-by-case basis, whether the proposed product represents a significant improvement when compared with other available therapies. Significant improvement may be illustrated by evidence of increased effectiveness in the treatment of a condition, elimination or substantial reduction of a treatment-limiting product reaction, documented enhancement of patient compliance that may lead to an improvement in serious outcomes and evidence of safety and effectiveness in a new subpopulation. A priority designation is intended to direct overall attention and resources to the evaluation of such applications, and to shorten the U.S. FDA’s goal for taking action on a marketing application from ten months to six months.
73
Fast track designation, priority review and breakthrough therapy designation may expedite the development or approval process, but do not change the standards for approval.
Accelerated Approval Pathway
The U.S. FDA may grant accelerated approval to a product for a serious or life-threatening condition that provides a meaningful therapeutic advantage to patients over existing treatments based upon a determination that the product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit. The U.S. FDA may also grant accelerated approval for such a condition when the product has an effect on an intermediate clinical endpoint that can be measured earlier than an effect on irreversible morbidity or mortality, or IMM, and that is reasonably likely to predict an effect on IMM or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. Products granted accelerated approval must meet the same statutory standards for safety and effectiveness as those granted traditional approval.
For the purposes of accelerated approval, a surrogate endpoint is a marker, such as a laboratory measurement, radiographic image, physical sign, or other measure that is thought to predict clinical benefit but is not itself a measure of clinical benefit. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. An intermediate clinical endpoint is a measurement of a therapeutic effect that is considered reasonably likely to predict the clinical benefit of a product, such as an effect on IMM. The U.S. FDA has stated that although it has limited experience with accelerated approvals based on intermediate clinical endpoints, such endpoints generally may support accelerated approval where the therapeutic effect measured by the endpoint is not itself a clinical benefit and basis for traditional approval, if there is a basis for concluding that the therapeutic effect is reasonably likely to predict the ultimate clinical benefit of a product.
The accelerated approval pathway is most often used in settings in which the course of a disease is long and an extended period of time is required to measure the intended clinical benefit of a product. Thus, accelerated approval has been used extensively in the development and approval of products for treatment of a variety of cancers in which the goal of therapy is generally to improve survival or decrease morbidity and the duration of the typical disease course requires lengthy and sometimes large trials to demonstrate a clinical or survival benefit.
The accelerated approval pathway is usually contingent on a sponsor’s agreement to conduct, in a diligent manner, additional post-approval confirmatory studies to verify and describe the product’s clinical benefit. As a result, a product candidate approved on this basis is subject to rigorous post-marketing compliance requirements, including the completion of Phase 4 or post-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or confirm a clinical benefit during post-marketing studies, may lead the U.S. FDA to withdraw the product from the market, and under FDORA, the FDA has increased authority for expedited procedures to withdraw approval of a drug or indication approved under accelerated approval if, for example, the confirmatory trial fails to verify the predicted clinical benefit of the product. All promotional materials for product candidates approved under accelerated regulations are subject to prior review by the U.S. FDA.
Post-Approval Regulation
If regulatory approval for marketing of a product or for a new indication for an existing product is obtained, the sponsor will be required to comply with rigorous and extensive post-approval regulatory requirements as well as any post-approval requirements that the U.S. FDA has imposed on the particular product as part of the approval process. The sponsor will be required, among other things, to report certain adverse reactions and production problems to the U.S. FDA, provide updated safety and efficacy information and comply with requirements concerning advertising and promotional labeling. Manufacturers and certain of their subcontractors are required to register their establishments with the U.S. FDA and certain state agencies, and are subject to periodic unannounced inspections by the U.S. FDA and certain state agencies for compliance with ongoing regulatory requirements, including cGMP regulations, which impose certain procedural and documentation requirements upon manufacturers. For certain commercial prescription drug or biologic products, manufacturers and other parties involved in the supply chain must also meet chain of distribution requirements and build electronic, interoperable systems for product tracking and tracing and for notifying the FDA of counterfeit, diverted, stolen and intentionally adulterated products or other products that are otherwise unfit for distribution in the United States. Accordingly, the BLA or NDA holder and its third-party manufacturers must continue to expend time, money and effort in the areas of production and quality control to maintain compliance with cGMP regulations and other regulatory requirements. In addition, changes to the manufacturing process or facility generally require prior U.S. FDA approval before being implemented, and other types of changes to the approved product, such as adding new indications and additional labeling claims, are also subject to further U.S. FDA review and approval.
Once an approval is granted, the U.S. FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or
74
failure to comply with regulatory requirements, may result in revisions to the approved labeling to add new safety information; imposition of post-market study requirements or clinical trial requirements to assess new safety risks; or imposition of distribution restrictions or other restrictions under a REMS program. Other potential consequences include, among other things:
•restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
•fines, untitled letters or warning letters or holds on post-approval clinical trials;
•adverse publicity;
•refusal of the U.S. FDA to approve pending applications or supplements to approved applications, or suspension or revocation of product license approvals;
•product seizure or detention, or refusal to permit the import or export of products; or
•injunctions, fines, debarment, disgorgement of profits or the imposition of civil or criminal penalties.
The U.S. FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. Pharmaceutical products may be promoted only for the approved indications and in accordance with the provisions of the approved label. The U.S. FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability.
Orphan Drug Designation
Orphan drug designation in the United States is designed to encourage sponsors to develop products intended for rare diseases or conditions. In the United States, a rare disease or condition is statutorily defined as a disease or condition that affects fewer than 200,000 individuals in the United States or that affects more than 200,000 individuals in the United States but for which there is no reasonable expectation that the cost of developing and making available the product for the disease or condition will be recovered from sales of the product in the United States.
Orphan drug designation qualifies a company for certain financial incentives, including tax advantages and, if the product receives the first U.S. FDA approval for the indication for which it has orphan designation, market exclusivity for seven years following the date of the product’s marketing approval. An application for designation as an orphan product can be made any time prior to the filing of an application for approval to market the product. Once a product receives orphan drug designation from the Office of Orphan Products Development at the U.S. FDA, the product must then go through the review and approval process like any other product.
In addition, a sponsor of a product that is otherwise the same product as an already approved orphan drug may seek and obtain orphan drug designation for the subsequent product for the same rare disease or condition if it can present a plausible hypothesis that its product may be clinically superior to the first product. More than one sponsor may receive orphan drug designation for the same product for the same rare disease or condition, but each sponsor seeking orphan drug designation must file a complete request for designation.
The period of exclusivity begins on the date that the marketing application is approved by the U.S. FDA and applies only to the indication for which the product has been designated. The U.S. FDA may approve a second application for the same product for a different use or a second application for a clinically superior version of the product for the same use. The U.S. FDA cannot, however, approve the same product made by another manufacturer for the same indication during the market exclusivity period unless it has the consent of the sponsor, the manufacturer makes a showing of clinical superiority over the product with orphan exclusivity, or the sponsor is unable to provide sufficient quantities.
Orphan product designation does not convey any advantage in or shorten the duration of the regulatory review and approval process.
Pediatric Studies and Exclusivity
A BLA, NDA or supplement thereto must contain data that are adequate to assess the safety and effectiveness of the product for the claimed indications in all relevant pediatric subpopulations, and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. Sponsors who are planning to submit a marketing application for a drug or biological product that includes a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration must also submit pediatric study plans prior to the assessment data, and no later than 60 calendar days following an end-of-Phase 2 meeting with the U.S. FDA. Pediatric study plans must contain an outline of the proposed pediatric study or studies the sponsor plans to conduct, including study objectives and design, any deferral or waiver requests and other information required by regulation. The applicant, the U.S. FDA, and the U.S. FDA’s internal review committee must then
75
review the information submitted, consult with each other and agree upon a final plan. The U.S. FDA or the applicant may request an amendment to the plan at any time.
The U.S. FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults, or full or partial waivers from the pediatric data requirements. Unless otherwise required by regulation, the pediatric data requirements generally do not apply to products with orphan designation.
Pediatric exclusivity is another type of non-patent marketing exclusivity in the United States and, if granted, provides for the attachment of an additional six months of marketing protection to the term of any existing regulatory exclusivity, including the non-patent and orphan exclusivity. This six-month exclusivity may be granted if a BLA or NDA sponsor submits pediatric data that fairly respond to a written request from the U.S. FDA for such data. The data do not need to show the product to be effective in the pediatric population studied; rather, if the clinical trial is deemed to fairly respond to the U.S. FDA’s request, the additional protection is granted. If reports of requested pediatric studies are submitted to and accepted by the U.S. FDA within the statutory time limits, whatever statutory or regulatory periods of exclusivity, or for drugs patent protection cover the product are extended by six months. This is not a patent term extension, but it effectively extends the regulatory period during which the U.S. FDA cannot approve another application.
Regulation of Combination Products in the United States
Certain products may be comprised of components that would normally be regulated under different types of regulatory authorities and frequently by different centers at the U.S. FDA. These products are known as combination products. Specifically, under regulations issued by the U.S. FDA, a combination product may be:
•a product comprised of two or more regulated components that are physically, chemically, or otherwise combined or mixed and produced as a single entity;
•two or more separate products packaged together in a single package or as a unit and comprised of drug and device products, device and biological products, or biological and drug products;
•a drug, or device, or biological product packaged separately that according to its investigational plan or proposed labeling is intended for use only with an approved individually specified drug, or device, or biological product where both are required to achieve the intended use, indication, or effect and where upon approval of the proposed product the labeling of the approved product would need to be changed, e.g., to reflect a change in intended use, dosage form, strength, route of administration, or significant change in dose; or
•any investigational drug, device, or biological product packaged separately that according to its proposed labeling is for use only with another individually specified investigational drug, device, or biological product where both are required to achieve the intended use, indication, or effect.
Under the FDCA, as amended, the U.S. FDA is charged with assigning a center with primary jurisdiction, or a lead center, for review of a combination product. That determination is based on the “primary mode of action” of the combination product. Thus, if the primary mode of action of a device-biologic combination product is attributable to the biologic product, the U.S. FDA center responsible for premarket review of the biologic product would have primary jurisdiction for the combination product. The U.S. FDA has also established an Office of Combination Products to address issues surrounding combination products and provide more certainty to the regulatory review process. That office serves as a focal point for combination product issues for agency reviewers and industry. It is also responsible for developing guidance and regulations to clarify the regulation of combination products, and for assignment of the U.S. FDA center that has primary jurisdiction for review of combination products where the jurisdiction is unclear or in dispute.
Regulation and Procedures Governing Approval of Medicinal Products in the European Union
In order to market any product outside of the United States, a company also must comply with numerous and varying regulatory requirements of other countries and jurisdictions regarding quality, safety and efficacy and governing, among other things, clinical trials, marketing authorization, commercial sales and distribution of products. Whether or not it obtains U.S. FDA approval for a product, an applicant will need to obtain the necessary approvals by the comparable foreign regulatory authorities before it can initiate clinical trials or marketing of the product in those countries or jurisdictions. Specifically, the process governing approval of medicinal products in the European Union generally follows the same lines as in the United States although the approval of a medicinal product in the United States is no guarantee of approval of the same product in the European Union, either at all or within the same timescale as approval may be granted in the United States. It entails satisfactory completion of pharmaceutical development, non-clinical studies and adequate and well-controlled clinical trials to establish the safety and efficacy of the medicinal product for each proposed indication. It also requires the submission to relevant competent authorities for clinical trial authorization for a marketing authorization application, or MAA, and granting of
76
a marketing authorization by these authorities before the product can be marketed and sold in the European Union or its Member States.
The European Commission may also grant a “conditional marketing authorization” prior to obtaining the comprehensive clinical data required for an application for a full marketing authorization. Such conditional marketing authorizations may be granted for product candidates intended for treating, preventing or diagnosing seriously debilitating or life-threatening diseases (including medicines designated as orphan medicinal products), if (i) the risk-benefit balance of the product candidate is positive, (ii) it is likely that the applicant will be in a position to provide the required comprehensive clinical trial data post-authorization, (iii) the product fulfills an unmet medical need and (iv) the benefit to public health of the immediate availability on the market of the medicinal product concerned outweighs the risk inherent in the fact that additional data are still required. A conditional marketing authorization may contain specific obligations to be fulfilled by the marketing authorization holder, including obligations with respect to the completion of ongoing or new studies, and with respect to the collection of pharmacovigilance data. Conditional marketing authorizations are valid for one year, and may be renewed annually, if the risk-benefit balance remains positive, and after an assessment of the need for additional or modified conditions and/or specific obligations. The timelines for the centralized procedure described above also apply with respect to the review by the CHMP of applications for a conditional marketing authorization. A conditional marketing authorization can be converted into a standard centralized marketing authorization (no longer subject to specific obligations) once the marketing authorization holder fulfills the obligations imposed and the complete data confirm that the medicine’s benefits continue to outweigh its risks.
Clinical Trial Approval
In April 2014, the European Union adopted the new Clinical Trials Regulation (EU) No 536/2014, which replaced the Clinical Trials Directive 2001/20/EC on January 31, 2022. The transitory provisions of the new Regulation provide that, by January 31, 2025, all ongoing clinical trials must have transitioned to the new Regulation. The new Regulation overhauls the system of approvals for clinical trials in the European Union. Specifically, it is directly applicable in all Member States (meaning that no national implementing legislation in each Member State is required), and aims at simplifying and streamlining the approval of clinical trials in the European Union. The main characteristics of the new Regulation include: a streamlined application procedure via a single-entry point through the Clinical Trials Information System, or CTIS; a single set of documents to be prepared and submitted for the application as well as simplified reporting procedures for clinical trial sponsors; and a harmonized procedure for the assessment of applications for clinical trials, which is divided in two parts (Part I contains scientific and medicinal product documentation and Part II contains the national and patient-level documentation). Part I is assessed by a coordinated review by the competent authorities of all European Union Member States in which an application for authorization of a clinical trial has been submitted (Concerned Member States) of a draft report prepared by a Reference Member State. Part II is assessed separately by each Concerned Member State. Strict deadlines have been established for the assessment of clinical trial applications.
Marketing Authorization
To obtain a marketing authorization for a product in the European Union, an applicant must submit an MAA, either under a centralized procedure administered by the EMA or one of the procedures administered by competent authorities in the Member States (decentralized procedure, national procedure, or mutual recognition procedure). A marketing authorization may be granted only to an applicant established in the European Union. Regulation (EC) No. 1901/2006 on medicinal products for pediatric use provides that prior to obtaining a marketing authorization in the European Union in the centralized procedure, an applicant must demonstrate compliance with all measures included in an EMA-approved pediatric investigation plan ("PIP"), covering all subsets of the pediatric population, unless the EMA has granted a product-specific waiver, class waiver, or a deferral for one or more of the measures included in the PIP.
The centralized procedure provides for the grant of a single marketing authorization by the European Commission that is valid across the European Union and the additional Member States of the European Economic Area (Iceland, Norway and Liechtenstein) ("EEA"). Pursuant to Regulation (EC) No. 726/2004, the centralized procedure is compulsory for specific products, including for medicines produced by certain biotechnological processes, products designated as orphan medicinal products, advanced therapy medicinal products (gene-therapy, somatic-cell therapy and tissue-engineered products) and products with a new active substance indicated for the treatment of certain diseases, including products for the treatment of cancer, HIV, AIDS, neurodegenerative disorders, autoimmune and other immune dysfunctions, viral diseases or diabetes. For those products for which the use of the centralized procedure is not mandatory, applicants may elect to use the centralized procedure where either the product contains a new active substance indicated for the treatment of other diseases, or where the applicant can show that the product constitutes a significant therapeutic, scientific or technical innovation, or for which a centralized process is in the interest of public health in the European Union.
Under the centralized procedure, the Committee for Medicinal Products for Human use (“CHMP”), which is the EMA’s committee that is responsible for human medicines, is responsible for conducting the assessment of whether a medicine meets
77
the required quality, safety and efficacy requirements, and whether it has a positive benefit-risk profile. Under the centralized procedure, the maximum timeframe for the evaluation of an MAA is 210 days from the receipt of a valid MAA, excluding clock stops when additional information or written or oral explanation is to be provided by the applicant in response to questions of the CHMP. Clock stops may extend the timeframe of evaluation of an MAA considerably beyond 210 days. Where the CHMP gives a positive opinion, it provides the opinion together with supporting documentation to the European Commission, who makes the final decision to grant a marketing authorisation. Accelerated evaluation may be granted by the CHMP in exceptional cases, when a medicinal product is of major interest from the point of view of public health and, in particular, from the viewpoint of therapeutic innovation.
If the CHMP accepts such a request, the timeframe of 210 days for assessment will be reduced to 150 days (excluding clock stops), but it is possible that the CHMP may revert to the standard time limit for the centralized procedure if it determines that the application is no longer appropriate to conduct an accelerated assessment.
Now that the UK (which comprises Great Britain and Northern Ireland) has left the European Union, Great Britain is no longer covered by centralized marketing authorizations (under the Northern Ireland Protocol, centralized marketing authorizations continue to be recognized in Northern Ireland). All medicinal products with a current centralized marketing authorization were automatically converted to Great Britain marketing authorizations on January, 1 2021. For a period of three years from January 1, 2021, the Medicines and Healthcare products Regulatory Agency ("MHRA"), the United Kingdom's medicines regulator, may rely on a decision taken by the European Commission on the approval of a new marketing authorization in the centralized procedure, in order to more quickly grant a new Great Britain marketing authorization. A separate application will, however, still be required. On January 24, 2023, the MHRA announced that a new international recognition framework will be put in place from January 1, 2024. Under this new framework, the MHRA will have regard to decisions on the approval of a marketing authorization made by the EMA and certain other regulators when considering whether to grant a UK marketing authorization. The MHRA also has the power to have regard to marketing authorizations approved in EU Member States through decentralized or mutual recognition procedures with a view to more quickly granting a marketing authorization in the UK or Great Britain.
Periods of Authorization and Renewals
A marketing authorization is valid for five years, in principle, and it may be renewed after five years on the basis of a re-evaluation of the risk-benefit balance by the EMA or by the competent authority of the authorizing Member State for a nationally authorized product. Once renewed, the marketing authorization is valid for an unlimited period, unless the European Commission or the competent authority decides, on justified grounds relating to pharmacovigilance, to proceed with one additional five-year renewal period. Any authorization that is not followed by the placement of the product on the European Union market (in the case of the centralized procedure) or on the market of the authorizing Member State (for a nationally authorized product) within three years after authorization ceases to be valid (the so-called sunset clause.)
Regulatory Requirements after Marketing Authorization
Following approval, the holder of the marketing authorization is required to comply with a range of requirements applicable to the manufacturing, marketing, promotion and sale of the medicinal product. These include compliance with the European Union’s stringent pharmacovigilance or safety reporting rules, pursuant to which post-authorization studies and additional monitoring obligations can be imposed. In addition, the manufacturing of authorized products, for which a separate manufacturer’s license is mandatory, must also be conducted in strict compliance with the EMA’s cGMP requirements and comparable requirements of other regulatory bodies in the European Union, which mandate the methods, facilities and controls used in manufacturing, processing and packing of products to assure their safety and identity. Finally, the marketing and promotion of authorized products, including industry-sponsored continuing medical education and advertising directed toward the prescribers of products are strictly regulated in the European Union under Directive 2001/83EC, as amended. The advertising of prescription-only medicines to the general public is not permitted in the European Union.
Orphan Designation and Exclusivity
Regulation (EC) No. 141/2000 and Regulation (EC) No. 847/2000 provide that a product can be designated as an orphan medicinal product by the European Commission if its sponsor can establish: (1) that the product is intended for the diagnosis, prevention or treatment of a life-threatening or chronically debilitating condition; (2) either (i) such condition affects no more than five in ten thousand persons in the European Union when the application is made, or (ii) without incentives it is unlikely that the marketing of the product in the European Union would generate sufficient return to justify the necessary investment in its development; and (3) there exists no satisfactory method of diagnosis, prevention, or treatment of the condition in question that has been authorized in the European Union or, if such method exists, the product has to be of significant benefit compared to products available for the condition.
78
An orphan designation provides a number of benefits, including fee reductions, regulatory assistance and the possibility to apply for a centralized marketing authorization. Orphan designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process. The grant of a marketing authorization for an orphan medicinal products leads to a ten-year period of market exclusivity. During this market exclusivity period, neither the EMA nor the European Commission or the Member States can accept an application or grant a marketing authorization for the same therapeutic indication in respect of a “similar medicinal product”. A “similar medicinal product” is defined as a medicinal product containing a similar active substance or substances as contained in an authorized orphan medicinal product, and which is intended for the same therapeutic indication. The market exclusivity period for the authorized therapeutic indication may, however, be reduced to six years if, at the end of the fifth year, it is established that the product no longer meets the criteria for orphan designation because, for example, the product is sufficiently profitable not to justify market exclusivity. There are also limited derogations from the ten-year period of market exclusivity pursuant to which the European Commission may grant a marketing authorization for a similar medicinal product in the same therapeutic indication. These are where: (i) the second applicant can establish that although their product is similar to the orphan medicinal product already authorised, the second product is safer, more effective or otherwise clinically superior; (ii) the marketing authorization holder for the authorized orphan medicinal product consents to the second orphan medicinal product application; or (iii) the marketing authorization holder for the authorized orphan medicinal product cannot supply enough orphan medicinal product.
The aforementioned European Union rules are generally applicable in the EEA.
Coverage, Reimbursement and Pricing
Significant uncertainty exists as to the coverage and reimbursement status of our products and any product candidates for which we may obtain regulatory approval. Even if our product candidates are approved for marketing, sales of such products and product candidates will depend, in part, on the extent to which third-party payors, including government health programs in the United States (such as Medicare and Medicaid), commercial health insurers, and managed care organizations, provide coverage and establish adequate reimbursement levels for such product candidates. In the United States, the Member States of the European Union and markets in other countries, patients who are prescribed treatments for their conditions and providers performing the prescribed services generally rely on third-party payors to reimburse all or part of the associated healthcare costs. Reimbursement rules and levels are not harmonized. For example, in the United States, reimbursement decisions vary from payor to payor, including government health programs and commercial health insurers. Similarly, in the European Union policies vary from Member State to Member State. Patients are unlikely to use any product candidates we may develop unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of such product candidates. The process for determining whether a payor will provide coverage for a product may be separate from the process for setting the reimbursement rate that the payor will pay for the product once coverage is approved. Third-party payors are increasingly challenging the price and examining the medical necessity and cost-effectiveness of medical products and services and imposing controls to manage costs. Factors payors consider in determining reimbursement are based on whether the product is:
•a covered benefit under its health plan;
•safe, effective and medically necessary;
•appropriate for the specific patient;
•cost-effective; and
•neither experimental nor investigational.
In order to secure coverage and reimbursement for any product that might be approved for sale, a company may need to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost-effectiveness of the product, and the cost of these studies would be in addition to the costs required to obtain U.S. FDA or other comparable marketing approvals. Even after pharmacoeconomic studies are conducted, product candidates may not be considered medically necessary or cost-effective. A decision by a third-party payor not to cover any product candidates we may develop could reduce physician utilization of such product candidates once approved and have a material adverse effect on our sales, results of operations and financial condition. Additionally, a payor’s decision to provide coverage for a product does not imply that an adequate reimbursement rate will be approved. For example, the payor may require co-payments that patients find unacceptably high. Further, one payor’s determination to provide coverage for a product does not assure that such coverage will continue or that other payors will also provide coverage and reimbursement for the product, and the level of coverage and reimbursement can differ significantly from payor to payor. Third-party reimbursement and coverage may not be adequate to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development and generate revenue.
In the United States, the containment of healthcare costs also has become a priority of federal, and state governments as well as other third-party payors and the prices of pharmaceuticals have been a focus in this effort. Governments and other third-party payors have shown significant interest in implementing cost containment programs, including price controls, restrictions on
79
reimbursement and requirements for substitution of generic products. Adoption of price controls and cost containment measures, and adoption of more restrictive policies with third-party payors with existing controls and measures, could further limit a company’s revenue from the sale of any approved products. Coverage policies and third-party reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more products for which a company or its collaborators receive marketing approval, less favorable coverage policies and reimbursement rates may be implemented or coverage may be ended in the future.
Outside the United States, we will face challenges in ensuring obtaining adequate coverage and payment for any product candidates we may develop. Pricing of prescription pharmaceuticals is subject to governmental control in many countries. Pricing negotiations with governmental authorities or other third-party payors such as statutory health insurance funds can extend well beyond the receipt of regulatory marketing approval for a product and may require us to conduct a clinical trial that compares the cost-effectiveness of any product candidates we may develop to other available therapies. The conduct of such a clinical trial could be expensive and result in delays in our commercialization efforts.
In the European Union, pricing and reimbursement schemes vary widely from country to country. Some Member States provide that products may be marketed only after a reimbursement price has been agreed. Some Member States may require the completion of additional studies that compare the cost-effectiveness of a particular product candidate to currently available therapies (so-called health technology assessments) in order to obtain reimbursement or pricing approval. For example, the European Union provides options for its Member States to restrict the range of products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. European Union Member States may approve a specific price for a product or may instead adopt a system of direct or indirect controls on the profitability of the company placing the product on the market. Other Member States allow companies to fix their own prices for products, but monitor and control prescription volumes and issue guidance to physicians to limit prescriptions. Recently, many countries in the European Union have increased the amount of discounts required on pharmaceuticals and these efforts could continue as countries attempt to manage healthcare expenditures, especially in light of the severe fiscal and debt crises experienced by many countries in the European Union. The downward pressure on healthcare costs in general, particularly prescription products, has become intense. As a result, increasingly high barriers are being erected to the entry of new products. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference pricing used by various European Union Member States and parallel trade (arbitrage between low-priced and high-priced Member States) can further reduce prices. Special pricing and reimbursement rules may apply to orphan drugs. Inclusion of orphan drugs in reimbursement systems tends to focus on the medical usefulness, need, quality and economic benefits to patients and the healthcare system as for any product. Acceptance of any medicinal product for reimbursement may come with cost, use and often volume restrictions, which again can vary by country. In addition, results based rules of reimbursement may apply. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products, if approved in those countries.
Healthcare Law and Regulation
Healthcare providers and third-party payors play a primary role in the recommendation and prescription of pharmaceutical products that are granted marketing approval. Our current and future arrangements with providers, researchers, consultants, third-party payors and customers are subject to broadly applicable federal and state fraud and abuse, anti-kickback, false claims, transparency and patient privacy laws and regulations and other healthcare laws and regulations that may constrain our business and/or financial arrangements. Restrictions under applicable federal and state healthcare laws and regulations include, without limitation, the following:
•the U.S. federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, receiving, offering, or paying remuneration, directly or indirectly, in-cash or in kind, to induce or reward either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made, in whole or in part, under a federal healthcare program such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or a specific intent to violate it in order to have committed a violation. Moreover, the government may assert that a claim that includes items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act. On November 20, 2020, OIG finalized further modifications to the federal Anti-Kickback Statute. Under the final rules, OIG added safe harbor protections under the Anti-Kickback Statute for certain coordinated care and value-based arrangements among clinicians, providers, and others. This rule (with exceptions) became effective January 19, 2021. We continue to evaluate what effect, if any, these rules will have on our business;
•the federal civil and criminal false claims laws, including the civil False Claims Act, and civil monetary penalties laws, which prohibit individuals or entities from, among other things, knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false, fictitious, or fraudulent or knowingly making, using, or causing to be
80
made or used a false record or statement to avoid, decrease, or conceal an obligation to pay money to the federal government. Manufacturers can be held liable under the federal False Claims Act even when they do not submit claims directly to government payors if they deemed to "cause" the submission of false or fraudulent claims. The federal False Claims Act also permits a private individual acting as a "whistleblower" to bring actions on behalf of the federal government alleging violations of the federal False Claims Act and to share in any monetary recovery;
•the Health Insurance Portability and Accountability Act, or HIPAA, which created additional federal criminal laws that prohibit, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters. Similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or a specific intent to violate it in order to have committed a violation;
•HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act ("HITECH"), and their respective implementing regulations, including the Final Omnibus Rule published in January 2013, which impose obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information without the appropriate authorization by entities subject to the law, such as healthcare providers, health plans and healthcare clearinghouses and their respective business associates;
•the federal transparency requirements known as the federal Physician Payments Sunshine Act, under the ACA, which requires certain manufacturers of drugs, devices, biologics and medical supplies to report annually to the Centers for Medicare & Medicaid Services, or CMS, within the U.S. Department of Health and Human Services, information related to payments and other transfers of value made by that entity to physicians (currently defined to include doctors, dentists, optometrists, podiatrists and chiropractors) and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members; effective January 1, 2022, these reporting obligations extend to include transfers of value made to certain non-physician providers such as physician assistants and nurse practitioners;
•federal consumer protection and unfair competition laws, which broadly regulate marketplace activities and activities that potentially harm consumers;
•federal government price reporting laws, which require us to calculate and report complex pricing metrics to government programs and which may be used in the calculation of reimbursement and/or discounts on marketed products;
•the Foreign Corrupt Practices Act, a U.S. law which prohibits companies and their intermediaries from making, or offering or promising to make improper payments to non-U.S. officials for the purpose of obtaining or retaining business or otherwise seeking favorable treatment (which could include, for example, certain medical professionals); and
•analogous state and foreign laws and regulations, such as state anti-kickback and false claims laws and state price reporting and transparency laws, which may apply to healthcare items or services that are reimbursed by non-governmental third-party payors, including private insurers.
Some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government in addition to requiring pharmaceutical manufacturers to report information related to payments to physicians and other healthcare providers or marketing expenditures and pricing information. State and foreign laws also govern the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and often are not pre-empted by HIPAA, thus complicating compliance efforts.
Violations of these laws can subject us to criminal, civil and administrative sanctions including monetary penalties, damages, fines, disgorgement, individual imprisonment and exclusion from participation in government-funded healthcare programs, such as Medicare and Medicaid, additional reporting requirements and oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance with these laws, reputational harm, and we may be required to curtail or restructure our operations. Moreover, we expect that there will continue to be federal and state laws and regulations, proposed and implemented, that could impact our future operations and business.
In the U.S., to help patients afford our products, we utilize patient assistance programs and, may choose to use, co-pay coupon programs for eligible patients. Government enforcement agencies have shown increased interest in pharmaceutical companies' product and patient assistance programs, including reimbursement support services, and a number of investigations into these programs have resulted in significant civil and criminal settlements. In addition, at least one insurer has directed its network pharmacies to no longer accept co-pay coupons for certain specialty drugs the insurer identified. Our co-pay coupon programs could become the target of similar insurer actions. In November 2013, the CMS issued guidance to the issuers of qualified health plans sold through the ACA's marketplaces encouraging such plans to reject patient cost-sharing support from third parties and indicating that the CMS intends to monitor the provision of such support and may take regulatory action to limit it in the future. The CMS subsequently issued a rule requiring individual market qualified health plans to accept third-party premium and cost-sharing payments from certain government-related entities. In September 2014, the OIG of the HHS issued a Special
81
Advisory Bulletin warning manufacturers that they may be subject to sanctions under the federal anti-kickback statute and/or civil monetary penalty laws if they do not take appropriate steps to exclude Part D beneficiaries from using co-pay coupons. Accordingly, companies exclude these Part D beneficiaries from using co-pay coupons. It is possible that changes in insurer policies regarding co-pay coupons and/or the introduction and enactment of new legislation or regulatory action could restrict or otherwise negatively affect these patient support programs, which could result in fewer patients using affected products, and therefore could have a material adverse effect on our sales, business, and financial condition.
On December 2, 2020, the HHS published a regulation removing safe harbor protection for price reductions from pharmaceutical manufacturers to plan sponsors under Part D, either directly or through pharmacy benefit managers (PBMs), unless the price reduction is required by law. The rule also creates a new safe harbor for price reductions reflected at the point-of-sale, as well as a safe harbor for certain fixed fee arrangements between PBMs and manufacturers. Implementation of this change and new safe harbors for point-of-sale reductions in price for prescription pharmaceutical products and PBM service fees are currently under review by the current U.S. presidential administration and may be amended or repealed. Further, on December 31, 2020, CMS published a new rule, effective January 1, 2023, requiring manufacturers to ensure the full value of co-pay assistance is passed on to the patient or these dollars will count toward the Average Manufacturer Price and Best Price calculation of the drug ("Accumulator Rule"). On May 17, 2022, the U.S. District Court for the District of Columbia granted the Pharmaceutical Research and Manufacturers of America's (PhRMA) motion for summary judgement invalidating the Accumulator Rule. We cannot predict how the implementation of and any further changes to this rule will affect our business. Although a number of these and other proposed measures may require authorization through additional legislation to become effective, and the current U.S. presidential administration may reverse or otherwise change these measures, both the current U.S. presidential administration and Congress have indicated that they will continue to seek new legislative measures to control drug costs.
Healthcare Reform
A primary trend in the U.S. healthcare industry and elsewhere is cost containment. There have been a number of federal and state proposals during the last few years to reduce the cost of care through changes in the healthcare system, including limits on the pricing, coverage and reimbursement of pharmaceutical and biopharmaceutical products, especially under government-funded healthcare programs, and increased governmental control of drug pricing.
By way of example, in March 2010, the U.S. Congress enacted the Patient Protection and Affordable Care Act (ACA), which, among other things, includes changes to the coverage and payment for products under governmental and private insurance plans. Among the provisions of the ACA that may be of importance to our potential product candidates are:
•an annual, non-deductible fee on any entity that manufactures or imports specified branded prescription drugs and biologic products, apportioned among these entities according to their market share in certain government healthcare programs, although this fee would not apply to sales of certain products approved exclusively for orphan indications;
•expansion of eligibility criteria for Medicaid programs by, among other things, allowing states to offer Medicaid coverage to certain individuals with income at or below 133% of the federal poverty level, thereby potentially increasing a manufacturer’s Medicaid rebate liability;
•expansion of manufacturers’ rebate liability under the Medicaid Drug Rebate Program by increasing the minimum rebate for both branded and generic drugs and revising the definition of “average manufacturer price” for calculating and reporting Medicaid drug rebates on outpatient prescription drug prices and extending rebate liability to prescriptions for individuals enrolled in Medicare Advantage plans;
•a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for products that are inhaled, infused, instilled, implanted or injected;
•expanding the types of entities eligible for the 340B drug discount program;
•a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research; and
•establishment of the Center for Medicare and Medicaid Innovation within CMS to test innovative payment and service delivery models to lower Medicare and Medicaid spending, potentially including prescription product spending (funding has been allocated to support the mission of the Center for Medicare and Medicaid Innovation through 2019).
At this point, healthcare reform and its impacts on us are highly uncertain in many respects. For example, since its enactment, there have been judicial and Congressional challenges to numerous aspects of the ACA. The current Trump administration and U.S. Congress focused on additional executive and legislative changes, including in particular repeal and replacement of certain provisions of the ACA.
82
Since its enactment, there have been numerous judicial, administrative, executive, and legislative challenges to certain aspects of the ACA, and we expect there will be additional challenges and amendments to the ACA in the future. On June 17, 2021, the U.S. Supreme Court dismissed the most recent judicial challenge to the ACA brought by several states without specifically ruling on the constitutionality of the ACA. Prior to the Supreme Court’s decision, President Biden issued an Executive Order to initiate a special enrollment period from February 15, 2021 through August 15, 2021 for purposes of obtaining health insurance coverage through the ACA marketplace. The Executive Order also instructed certain governmental agencies to review and reconsider their existing policies and rules that limit access to healthcare, including among others, reexamining Medicaid demonstration projects and waiver programs that include work requirements, and policies that create unnecessary barriers to obtaining access to health insurance coverage through Medicaid or the ACA. It is unclear whether the ACA will be overturned, repealed, replaced, or further amended. We cannot predict what affect further changes to the ACA would have on our business.
Other legislative changes have been proposed and adopted in the United States since the ACA was enacted:
•On August 2, 2011, the U.S. Budget Control Act of 2011, among other things, led to aggregate reductions to Medicare payments to providers of up to 2% per fiscal year. These reductions went into effect in April 2013 and, due to subsequent legislative amendments to the statute, will remain in effect through 2030, with the exception of a temporary suspension from May 1, 2020 through March 31, 2022 due to the COVID-19 pandemic. Following the temporary suspension, a 1% payment reduction began April 1, 2022 and lasted through June 30, 2022. The 2% payment reduction resumed on July 1, 2022.
•On January 2, 2013, the American Taxpayer Relief Act of 2012, among other things, further reduced Medicare payments to several providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years.
•On April 13, 2017, CMS published a final rule that gives states greater flexibility in setting benchmarks for insurers in the individual and small group marketplaces, which may have the effect of relaxing the essential health benefits required under the ACA for plans sold through such marketplaces.
•On May 30, 2018, the Right to Try Act, was signed into law. The law, among other things, provides a federal framework for certain patients to access certain investigational new drug products that have completed a Phase 1 clinical trial and that are undergoing investigation for FDA approval. Under certain circumstances, eligible patients can seek treatment without enrolling in clinical trials and without obtaining FDA permission under the FDA expanded access program. There is no obligation for a pharmaceutical manufacturer to make its drug products available to eligible patients as a result of the Right to Try Act.
•On May 23, 2019, CMS published a final rule to allow Medicare Advantage Plans the option of using step therapy for Part B drugs beginning January 1, 2020.
•On December 20, 2019, former President Trump signed into law the Further Consolidated Appropriations Act (H.R. 1865), which repealed the Cadillac tax, the health insurance provider tax, and the medical device excise tax. It is impossible to determine whether similar taxes could be instated in the future.
These laws may result in additional reductions in Medicare and other healthcare funding and otherwise affect the prices we may obtain for any of our product candidates for which we may obtain regulatory licensing or the frequency with which any such product candidate is prescribed or used.
In addition, there have been several changes to the 340B drug pricing program, which imposes ceilings on prices that drug manufacturers can charge for medications sold to certain health care facilities. On December 27, 2018, the District Court for the District of Columbia invalidated a reimbursement formula change under the 340B drug pricing program, and CMS subsequently altered the FYs 2019 and 2018 reimbursement formula on specified covered outpatient drugs (“SCODs”). The court ruled this change was not an “adjustment” which was within the Secretary’s discretion to make but was instead a fundamental change in the reimbursement calculation. However, most recently, on July 31, 2020, the U.S. Court of Appeals for the District of Columbia Circuit overturned the district court’s decision and found that the changes were within the Secretary’s authority. On September 14, 2020, the plaintiffs-appellees filed a Petition for Rehearing En Banc (i.e., before the full court), but was denied on October 16, 2020. Plaintiffs-appellees filed a petition for a writ of certiorari at the Supreme Court on February 10, 2021 and the petition was granted on July 2, 2021. On June 15, 2022, the Supreme Court unanimously reversed the Court of Appeals' decision, holding that HHS's 2018 and 2019 reimbursement rates for 340B hospitals were contrary to the statute and unlawful. We continue to review developments impacting the 340B program.
Additionally, there has been increasing legislative and enforcement interest in the United States with respect to specialty drug pricing practices. Specifically, there have been several recent U.S. Congressional inquiries and proposed bills designed to, among other things, bring more transparency to drug pricing, reduce the cost of prescription drugs under Medicare, review the relationship between pricing and manufacturer patient programs, explore reimportation of drugs and reform government program reimbursement methodologies for drugs. At a federal level, President Biden signed an Executive Order on July 9, 2021 affirming the administration’s policy to (i) support legislative reforms that would lower the prices of prescription drug and
83
biologics, including by allowing Medicare to negotiate drug prices, by imposing inflation caps, and, by supporting the development and market entry of lower-cost generic drugs and biosimilars; and (ii) support the enactment of a public health insurance option. Among other things, the Executive Order also directs HHS to provide a report on actions to combat excessive pricing of prescription drugs, enhance the domestic drug supply chain, reduce the price that the Federal government pays for drugs, and address price gouging in the industry; and directs the FDA to work with states and Indian Tribes that propose to develop section 804 Importation Programs in accordance with the Medicare Prescription Drug, Improvement, and Modernization Act of 2003, and the FDA’s implementing regulations. FDA released such implementing regulations on September 24, 2020, which went into effect on November 30, 2020, providing guidance for states to build and submit importation plans for drugs from Canada. On September 25, 2020, CMS stated drugs imported by states under this rule will not be eligible for federal rebates under Section 1927 of the Social Security Act and manufacturers would not report these drugs for “best price” or Average Manufacturer Price purposes. Since these drugs are not considered covered outpatient drugs, CMS further stated it will not publish a National Average Drug Acquisition Cost for these drugs. If implemented, importation of drugs from Canada may materially and adversely affect the price we receive for any of our product candidates. Further, on November 20, 2020 CMS issued an Interim Final Rule implementing the Most Favored Nation, or MFN, Model under which Medicare Part B reimbursement rates would have been be calculated for certain drugs and biologicals based on the lowest price drug manufacturers receive in Organization for Economic Cooperation and Development countries with a similar gross domestic product per capita. However, on December 29, 2021 CMS rescinded the Most Favored Nations rule. Additionally, on November 30, 2020, HHS published a regulation removing safe harbor protection for price reductions from pharmaceutical manufacturers to plan sponsors under Part D, either directly or through pharmacy benefit managers, unless the price reduction is required by law. The rule also creates a new safe harbor for price reductions reflected at the point-of-sale, as well as a safe harbor for certain fixed fee arrangements between pharmacy benefit managers and manufacturers. Pursuant to court order, the removal and addition of the aforementioned safe harbors were delayed and recent legislation imposed a moratorium on implementation of the rule until January 1, 2026. The Inflation Reduction Act of 2022 (the "IRA") further delayed implementation of this rule to January 1, 2032. Although a number of these and other proposed measures may require authorization through additional legislation to become effective, and the Biden administration may reverse or otherwise change these measures, both the Biden administration and Congress have indicated that they will continue to seek new legislative measures to control drug costs.
In August 2022, the IRA was signed into law. The IRA includes several provisions that will impact our business to varying degrees, including provisions that reduce the out-of-pocket cap for Medicare Part D beneficiaries to $2,000 starting in 2025; impose new manufacturer financial liability on certain drugs in Medicare Part D, allow the U.S. government to negotiate Medicare Part B and Part D price caps for certain high-cost drugs and biologics without generic or biosimilar competition, require companies to pay rebates to Medicare for certain drug prices that increase faster than inflation, and delay the rebate rule that would limit the fees that pharmacy benefit managers can charge. Further, under the IRA, orphan drugs are exempted from the Medicare drug price negotiation program, but only if they have one rare disease designation and for which the only approved indication is for that disease or condition. If a product receives multiple rare disease designations or has multiple approved indications, it will not qualify for the orphan drug exemption. The effect of the IRA on our business and the healthcare industry in general is not yet known.
There have been, and likely will continue to be, legislative and regulatory proposals at the foreign, federal and state levels directed at broadening the availability of healthcare and containing or lowering the cost of healthcare. Such reforms could have an adverse effect on anticipated revenues from product candidates that we may successfully develop and for which we may obtain marketing approval and may affect our overall financial condition and ability to develop product candidates.
84
C.Organizational Structure
As of December 31, 2023, we had three subsidiaries. The following table sets out the country of incorporation and percentage ownership and voting interest held by us (directly or indirectly through subsidiaries) for each subsidiary.
| Company | Country of incorporation | Percentage ownership and voting interest | Main activity | |||||||||||||||||
| MorphoSys US Inc. | United States | 100.00 | % | Commercialization and selling of products in the field of medicines, pharmaceutical compounds and related intermediate products, as well as operation of all businesses necessary and measures related thereto | ||||||||||||||||
| Constellation Pharmaceuticals, Inc. | United States | 100.00 | % | Clinical-stage biopharmaceutical company developing novel therapeutics for patients with cancers associated with abnormal gene expression or drug resistance, as well as operation of all business necessary and measures related thereto | ||||||||||||||||
| Constellation Securities Corp. | United States | 100.00 | % | Engaged exclusively in buying, selling, dealing in, or holding securities on its own behalf for investment purposes | ||||||||||||||||
D.Property, Plants and Equipment
Our headquarters are in the suburbs of Munich, Germany, where we occupy office and laboratory space under a ten-year fixed term lease that started on January 1, 2017. In July 2018, we established a wholly owned subsidiary, MorphoSys US Inc., to commercialize Monjuvi® in the United States. MorphoSys US Inc. occupies office space in Boston, Massachusetts, USA, under a seven-year fixed term lease, which is also used by Constellation. In July 2021, we acquired a wholly owned subsidiary via a cash tender offer followed by an upstream merger, Constellation. Constellation occupied office space in Cambridge, Massachusetts, USA, for which the lease agreement was terminated and expired in February 2022.
Item 4A. Unresolved Staff Comments
None.
Item 5. Operating and Financial Review and Prospects
You should read the following discussion and analysis of the financial condition and results of operations of the Company in conjunction with the annual consolidated financial statements and the related notes thereto included elsewhere in this report. In addition to historical financial information, the following discussion contains forward-looking statements that reflect our plans, estimates and opinions. Our actual results could differ materially from those discussed in these forward-looking statements. Factors that could cause or contribute to these differences or cause our actual results or the timing of selected events to differ materially from those anticipated in these forward-looking statements include those set forth under “Risk Factors”, “Special Note Regarding Forward-Looking Statements” and elsewhere in this report.
Our consolidated financial statements comply with both the IFRSs published by the International Accounting Standards Board (IASB) and those adopted by the EU. The consolidated financial statements also take into account the supplementary provisions under commercial law, which must be applied in accordance with Section 315e (1) of the German Commercial Code (Handelsgesetzbuch—HGB).
MorphoSys has transformed from a research and technology platform focused business into a commercial biopharmaceutical company. As our business model has changed, we will adapt our guidance parameters. In 2023, Monjuvi® U.S. net product sales, the gross margin of Monjuvi® U.S. net product sales, research and development expenses, as well as total combined expenses for selling and general and administrative matters were used as key financial performance indicators, since these indicators were the most significant for steering MorphoSys Group. Monjuvi® U.S. net product sales and related gross margin enabled management and investors to track operational sales-related performance of our co-commercialized product. At the same time the expense split provides visibility to management and investors on commercialization and development efforts. These indicators are routinely analyzed and evaluated. The gross margin of Monjuvi® U.S. net product sales was defined as cost
85
of sales for Monjuvi® U.S. product sales divided by Monjuvi® U.S. net product sales. Going forward, the Management Board has decided to provide guidance on research and development expenses for selling and general and administrative matters since these indicators still provide visibility to the Company’s stakeholders with regards to future commercialization efforts for pelabresib and development efforts.
As other factors, cash and investments (presented in the following balance sheet items: "Cash and cash equivalents" as well as current "Other Financial Assets") are also regularly analyzed and evaluated. Cash and investments are not considered to be part of the key financial performance indicators.
In the years ahead, events such as the in-licensing and out-licensing of development candidates and significant milestone payments and royalties from the market maturity of HuCAL and Ylanthia antibodies could have an impact on the Company’s net assets and financial position. Such events could cause financial targets to change significantly. Similarly, failures in drug development could have negative consequences for the MorphoSys Group. Negative effects from a further pandemic similar to COVID-19 or from COVID-19 variants are also possible or cannot be excluded.
A.Operating Results
Revenues
Revenues in the reporting year decreased by 14% or € 40.0 million to € 238.3 million (2022: € 278.3 million). This decrease resulted first and foremost from prior year revenues stemming from the execution of out-licensing agreements with HI-Bio and Novartis. Group revenues included revenues of € 85.0 million (US$ 92.0 million) (2022: € 84.9 million (US$ 89.4 million)) from the recognition of Monjuvi® U.S. net product sales.
Success-based payments including royalties accounted for 50% or € 119.2 million (2022: 37% or € 103.1 million) of total revenues. On a regional basis, MorphoSys generated 89% or € 211.5 million of its commercial revenues from product sales and with biopharmaceutical companies in the U.S. and 11% or € 26.8 million from customers primarily located in Europe (excluding Germany) and Asia. In the same period last year, these percentages were 89% (€ 248.9 million) and 11% (€ 29.3 million), respectively. 78% of the Group's revenues were generated with customers Janssen, McKesson and Incyte (2022: 62% with Janssen, HI-Bio and McKesson).
In 2022, revenues increased by 55% or € 98.7 million to € 278.3 million (2021: € 179.6 million). This increase resulted mainly from higher revenues from licenses due to the out-licensing agreements with HI-Bio and Novartis. Group revenues included revenues of € 84.9 million (US$ 89.4 million) (2021: € 66.9 million (US$ 79.1 million)) from the recognition of Monjuvi® U.S. net product sales.
Success-based payments including royalties accounted for 37% or € 103.1 million (2021: 48% or € 85.5 million) of total revenues. On a regional basis, MorphoSys generated 89% or € 248.9 million of its commercial revenues from product sales and with biopharmaceutical companies in North America and 11% or € 29.3 million from customers primarily located in Europe and Asia. In 2021, these percentages were 87% (€ 156.3 million) and 13% (€ 23.3 million), respectively. 62% of the Group's revenues were generated with customers Janssen, HI-Bio and McKesson (2021: 59% with Janssen, Incyte and GSK).
Our revenues are subject to a number of uncertainties, including the variability in Monjuvi® product sales in the US market and the limited visibility that MorphoSys has on its royalty streams.
Following the approval and launch of Monjuvi® (tafasitamab-cxix) in the U.S., Incyte received conditional approvals for tafasitamab in the EU, Canada and other jurisdictions (brand name, Minjuvi®). In 2022, the Swiss agency for therapeutic products (Swissmedic), granted temporary approval for Minjuvi® (tafasitamab). Incyte has exclusive commercialization rights to tafasitamab outside the United States and MorphoSys is entitled to royalties. Therefore, revenues related to the commercialization in other regions are dependent from the approval of authorities. In case of non-approval the expected revenues will not be realizable.
Revenues from partnerships could also potentially influence our revenues, in case contractually agreed milestones and royalty thresholds are met.
The majority of our anticipated business transactions are conducted in euros and U.S. dollars and therefore are exposed to potential changes of foreign currency exchange rates. The assessment of the impacts on revenues is described in item 5.D. Trend information.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
86
Cost of Sales
Cost of sales increased from € 48.6 million in 2022 to € 58.4 million in 2023, mainly due to impairments in the amount of € 11.9 million (2022: € 0.0 million), relating to the recognition of the inventory obsolescence reserve and scrapping of inventories. Acquisition and production costs of inventories slightly increased to € 30.7 million in 2023 (2022: € 28.8 million), mainly for Monjuvi® and Minjuvi®. In addition, amortization and other expenses for intangible assets increased from € 9.8 million in 2022 to € 10.7 million in 2023. This was offset by a decrease in personnel costs from € 9.5 million in 2022 to € 8.2 million in 2023. The gross margin of Monjuvi® U.S. net product sales amounted to 69% (2022: 73%).
Cost of sales increased from € 32.2 million in 2021 to € 48.6 million in 2022, mainly due to higher acquisition and production costs of inventories of € 28.8 million in 2022 (2021: € 12.6 million), mainly for Monjuvi® and Minjuvi®. In addition, impairment, amortization and other expenses for intangible assets increased from € 7.4 million in 2021 to € 9.8 million in 2022. This was offset by a decrease in personnel costs from € 11.6 million in 2021 to € 9.5 million in 2022. The gross margin of Monjuvi® U.S. net product sales amounted to 73% (2021: 82%).
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
Gross Profit
Gross Profit amounted to € 179.9 million in 2023 (2022: € 229.6 million). This decrease resulted mainly from lower revenues from licenses due to the out-licensing agreements with HI-Bio and Novartis signed in 2022.
In 2022 gross profit amounted to € 229.6 million (2021: € 147.4 million). This increase resulted mainly from higher revenues from licenses due to the out-licensing agreements with HI-Bio and Novartis.
Operating Expenses
In 2023, operating expenses decreased by 4%, or € 18.0 million, to € 432.4 million compared to € 450.4 million in 2022. The year-over-year decrease resulted mainly from a decrease in expenses for external services and lower expenses for consumables. The decrease was partially offset by higher personnel expenses in 2023.
Research and development expenses decreased by 5%, or € 14.2 million, to € 283.6 million in the reporting year (2022: € 297.8 million). The year-over-year decrease mainly resulted from lower expenses for external laboratory and consulting services and lower expenses for consumables and were partially compensated by higher personnel related expenses.
The combined expenses for selling and general and administration amounted to € 147.2 million in 2023 (2022: € 152.5 million). This total mainly includes personnel expenses of € 83.0 million (2022: € 81.0 million) and expenses for external services of € 47.4 million (2022: € 54.4 million).
In 2023, selling expenses amounted to € 81.4 million compared to € 92.4 million in 2022. The decrease by 12%, or € 11.0 million, was due to streamlining and focusing of selling efforts. Selling expenses also included all of the expenses for services provided by Incyte as part of the joint U.S. marketing activities for Monjuvi®.
General and administrative (G&A) expenses increased by 9%, or € 5.7 million, from € 60.1 million in 2022 to € 65.8 million in 2023. The major driver for this increase were higher personnel expenses that were partly offset by lower expenses for external services.
In 2023, a goodwill impairment loss of € 1.6 million was recognized.
In 2022, operating expenses decreased by 31%, or € 205.4 million, to € 450.4 million compared to € 655.8 million in 2021. The year-over-year decrease resulted mainly from an impairment of goodwill in 2021 and offset by lower personnel expenses in 2022, partially offset by higher development activities due to the first-time recognition of Constellation's operating expenses for a full fiscal year in 2022.
Research and development expenses increased by 32%, or € 72.6 million, to € 297.8 million in 2022 (2021: € 225.2 million). The year-over-year increase mainly resulted from the recognition of research and development expenses of Constellation, whose research activities have been included in the MorphoSys consolidated financial statements since the third quarter of 2021.
The combined expenses for selling and general and administration amounted to € 152.5 million in 2022 (2021: € 199.8 million). This total mainly includes personnel expenses of € 81.0 million (2021: € 96.1 million) and expenses for external services of € 54.4 million (2021: € 87.2 million).
87
In 2022, selling expenses amounted to € 92.4 million compared to € 121.5 million in 2021. The decrease is due to streamlining and focusing of selling efforts. Selling expenses also included all of the expenses for services provided by Incyte as part of the joint U.S. marketing activities for Monjuvi®.
General and administrative (G&A) expenses decreased by 23%, or € 18.1 million, from € 78.3 million in 2021 to € 60.1 million in 2022. The major driver for this decline were one-time transaction costs for the Constellation acquisition in 2021 of € 19.7 million.
Furthermore, the decrease in operating expenses in 2022 resulted from the recognition of an impairment of goodwill amounting to € 230.7 million in 2021.
MorphoSys will invest a significant portion of its financial resources in the development and commercialization of its proprietary programs as it pursues its ambition to become a leader in oncology. This will require significant capital and result in significant net losses over the next several years.
A significant part of our future business activities will be denominated in U.S. dollars and therefore is exposed to potential fluctuations in foreign currency exchange rates.
Research and Development
Research and development expenses decreased by 5%, or € 14.2 million, to € 283.6 million in 2023 (2022: € 297.8 million). Expenses for external laboratory services and legal and scientific consulting services decreased from € 198.1 million in the previous year to € 170.9 million in the reporting year. This reflects our current clinical study progress as well as the prioritization activities relating to our R&D portfolio.
Personnel expenses increased from € 65.0 million in the previous year to € 80.2 million in the reporting year, mainly driven by the increase in share-based payment expenses due to the increase in share price of MorphoSys AG, which is the valuation basis for the share-based payment programs. Expenses for intangible assets increased to € 16.3 million in 2023 (2022: € 14.8 million). In 2023 the expense was impacted by an impairment loss amounting to € 8.9 million relating to the write-off of a license. In the previous year, these were influenced in particular by impairment losses of € 7.8 million in connection with an impairment of an internally generated intangible asset under development. Depreciation, amortization and other expenses for infrastructure increased from € 10.8 million in 2022 to € 11.0 million in 2023. In contrast, expenses for consumables decreased from € 3.8 million in the previous year to € 0.3 million in 2023. In addition, Other expenses decreased from € 5.4 million in 2022 to € 4.9 million in 2023.
Research and development expenses increased by 32%, or € 72.6 million, to € 297.8 million in 2022 (2021: € 225.2 million) mainly due to higher expenses for external laboratory services. Expenses for external laboratory services and legal and scientific consulting services increased from € 131.5 million in 2021 to € 198.1 million in 2022, mainly due to the recognition of research and development expenses of Constellation, whose research activities have been included in the MorphoSys consolidated financial statements since the third quarter of 2021.
Personnel expenses decreased from € 65.9 million in 2021 to € 65.0 million in 2022. Expenses for intangible assets amounted to € 14.8 million in 2022 (2021: € 7.9 million). In 2022, these were influenced in particular by impairment losses of € 7.8 million in connection with an impairment of an internally generated intangible asset under development. Depreciation, amortization and other expenses for infrastructure decreased from € 11.8 million in 2021 to € 10.8 million in 2022. Other expenses increased from € 4.1 million in 2021 to € 5.4 million in 2022. Expenses for consumables increased from € 4.1 million in 2021 to € 3.8 million in 2022.
Selling
Selling expenses decreased by 12%, or € 11.0 million, to € 81.4 million in 2023 (2022: € 92.4 million). This item mainly includes personnel expenses of € 39.8 million (2022: € 48.6 million) and expenses for external services of € 32.7 million (2022: € 35.8 million). The decrease in selling expenses is based on the ongoing measures to streamline and focus sales efforts. Selling expenses also included all expenses for services provided by Incyte as part of the joint U.S. sales activities for Monjuvi®.
Selling expenses decreased by 24%, or € 29.1 million, to € 92.4 million in 2022 (2021: € 121.5 million). This item mainly includes personnel expenses of € 48.6 million (2021: € 63.5 million) and expenses for external services of € 35.8 million (2021: € 51.3 million). The decrease in selling expenses is based on measures to streamline and focus sales efforts. Selling expenses also included all expenses for services provided by Incyte as part of the joint U.S. sales activities for Monjuvi®.
General and Administrative (G&A)
G&A expenses increased by 9%, or € 5.7 million, in 2023 and amounted to € 65.8 million (2022: € 60.1 million). The increase was mainly due to higher personnel expenses that amounted to € 43.2 million in 2023 (2022: € 32.5 million). In contrast,
88
expenses for external services declined to € 14.6 million (2022: € 18.6 million) and depreciation, amortization and other expenses for infrastructure decreased from € 5.0 million in the previous year to € 3.7 million in 2023.
G&A expenses decreased by 23%, or € 18.1 million in 2022 and amounted to € 60.1 million (2021: € 78.3 million). The decrease was mainly due to transaction costs for the Constellation acquisition in 2021 amounting to € 19.7 million. Personnel expenses amounted to € 32.5 million in 2022 (2021: € 32.6 million). Depreciation, amortization and other expenses for infrastructure decreased from € 6.9 million in 2021 year to € 5.0 million in 2022.
Impairment of Goodwill
In the reporting year of 2023, an impairment of goodwill in the amount of € 1.6 million was recorded, which initially resulted from an acquisition in financial year 2010 (2022: € 0.0 million; 2021: € 230.7 million).
Other Income
Other income decreased by 58%, or € 7.0 million, to € 5.0 million in the reporting year (2022: € 12.0 million) and mainly resulted from lower exchange rate gains of € 3.2 million (2022 : € 11.4 million).
In 2022, other income increased by 46%, or € 3.8 million, to € 12.0 million (2021: € 8.2 million) and mainly resulted from exchange rate gains of € 11.4 million (2021: € 7.6 million).
Other Expenses
In the 2023 reporting year, other expenses decreased by 54%, or € 8.5 million, from € 15.6 million in 2022 to € 7.1 million in 2023. This decrease was mainly the result of lower exchange rate losses of € 6.3 million (2022: € 15.0 million).
In 2022, other expenses increased by more than 100%, or € 9.2 million, from € 6.4 million in 2021 to € 15.6 million in 2022. This increase was mainly the result of exchange rate losses of € 15.0 million (2021: € 5.9 million).
Finance Income
Finance income decreased by 48%, or € 198.7 million, to € 213.4 million in the reporting year (2022: € 412.1 million) and mainly resulted from items amounting to € 115.6 million (2022: € 361.4 million) in connection with the changes in plan assumptions of finanical assets and financial liabilities from collaborations. The effect is mainly resulting from changes in internal planning assumptions in the fourth quarter 2023 regarding the expected net cash flows related to financial liabilities from collaborations in the amount of € 107.8 million and from income for foreign currency valuation in the amount of € 7.7 million (refer to Note 4.19 titled “Financial Assets and Liabilities from Collaborations” contained in the Notes to the Consolidated Financial Statements). In addition, finance income includes valuation income from the effect from differences between planning assumptions and actual figures from financial liabilities from future payments to Royalty Pharma in the amount of € 41.9 million (2022: € 31.2 million) (refer to Note 4.20 titled “Financial Liabilities from Future Payments to Royalty Pharma” contained in the Notes to the Consolidated Financial Statements). Also included is finance income from foreign exchange gains in the amount of € 9.8 million (2022: € 14.3 million), gains from measurement at fair value in the amount of € 7.1 million (2022: € 0.4 million) as well interest income in the amount of € 18.3 million (2022: € 4.6 million). Furthermore, € 16.4 million finance income resulted from the repurchase of own convertible bonds (2022: € 0 million) as well as from the partial sale of HI-Bio Shares of € 4.2 million (2022: € 0 million).
In 2022, Finance income increased by more than 100%, or € 315.5 million, to € 412.1 million (2021: € 96.6 million) and resulted from items amounting to € 361.4 million (2021: € 75.7 million) in connection with the changes in plan assumptions of financial assets and financial liabilities from collaborations. These items included effects from differences between planning assumptions and actual figures and the fair value measurement (refer to Note 4.19 titled “Financial Assets and Liabilities from Collaborations” contained in the Notes to the Consolidated Financial Statements). In addition, finance income includes valuation income from the effect from differences between planning assumptions and actual figures from financial liabilities from future payments to Royalty Pharma in the amount of € 31.2 million (2021: € 0.0 million) (refer to Note 4.20 titled “Financial Liabilities from Future Payments to Royalty Pharma” contained in the Notes to the Consolidated Financial Statements). Also included is finance income from the investment of cash and cash equivalents and exchange rate gains from investing of funds amounting to € 19.1 million (2021: € 20.9 million). In 2022 income from financial derivatives was recognized in the amount of € 0.2 million (2021: € 0.0 million).
Finance income could be positively and negatively affected by the changes in the planning assumptions of the financial assets and financial liabilities from collaborations and of financial liabilities from future payments to Royalty Pharma. Future changes in the foreign currency exchange rate might impact this profit or loss line item significantly, since a significant portion of our business transactions and investments in cash and cash equivalents are denominated in US dollars. Moreover, the amounts
89
presented under financial assets and financial liabilities from collaborations and to a certain extent also for liabilities from future payments to Royalty Pharma are denominated in US dollars, and are therefore sensitive to changes in the euros / US dollar exchange rate.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
Finance Expenses
Finance expenses decreased by 14%, or € 23.9 million, to € 142.0 million in the reporting year (2022: € 165.9 million). This decrease was mainly due to the effects from financial liabilities from future payments to Royalty Pharma of € 107.2 million (2022: € 81.2 million) resulting from differences between planning assumptions and actual figures, foreign currency effects and the application of the effective interest method (refer to Note 4.20 “Financial Liabilities from Future Payments to Royalty Pharma” contained in the Notes to the Consolidated Financial Statements). Furthermore, finance expenses include effects from financial liabilities from collaborations and decreased in 2023 to € 8.8 million (2022: € 60.4 million), specifically from the application of the effective interest method as well as the foreign currency revaluation (refer to Note 4.19 titled “Financial Assets and Liabilities from Collaborations” contained in the Notes to the Consolidated Financial Statements). In addition, this line item included finance expenses from the investment of cash and cash equivalents and exchange rate losses from financing activities of € 9.3 million (2022: € 8.5 million). Other finance expenses in the fiscal year 2023 amounted to € 16.7 million (2022: € 15.7 million), mainly relating to interest on the convertible bond issued in October 2020 in the amount of € 11.1 million (2022: € 12.5 million). Furthermore, a change in fair value of the Anti-Dilution Asset in the amount of € 4.3 million as well as € 0.9 million (2022: € 1.1 million) in interest expenses from the compounding of non-current lease liabilities were also recognized in the reporting year.
In 2022, Finance expenses decreased by 9%, or € 15.6 million, to € 165.9 million (2021: € 181.5 million). This decrease was mainly due to the effects from financial liabilities from future payments to Royalty Pharma of € 81.3 million (2021: € 94.7 million) resulting from differences between planning assumptions and actual figures, foreign currency effects and the application of the effective interest method (also refer to Note 4.20 “Financial Liabilities from Future Payments to Royalty Pharma” contained in the Notes to the Consolidated Financial Statements). Furthermore, finance expenses include effects from financial liabilities from collaborations of € 60.4 million (2021: € 59.7 million), specifically from the application of the effective interest method as well as the foreign currency revaluation (refer to Note 4.19 titled “Financial Assets and Liabilities from Collaborations” contained in the Notes to the Consolidated Financial Statements). Furthermore, this line item included finance expenses from the investment of cash and cash equivalents and exchange rate losses from financing activities of 8.5 Mio. € (2021: 11.4 Mio. €). Other finance expenses amounted to € 15.7 million (2021: € 15.6 million) in 2022, mainly relating to interest on the convertible bond issued in October 2020 in the amount of € 12.5 million (2021: € 12.1 million) as well as € 1.1 million (2021: € 1.2 million) in interest expenses from the compounding of non-current lease liabilities were also recognized in 2022.
Finance expenses could be positively and negatively affected by the changes in the planning assumptions of the financial assets and financial liabilities from collaborations and of financial liabilities from future payments to Royalty Pharma. Future changes in the foreign currency exchange rate might impact this profit or loss line item significantly, since a significant portion of our business transactions and investments in cash and cash equivalents are denominated in US Dollars. Moreover, the amounts presented under financial assets and financial liabilities from collaborations and to a certain extent also for liabilities from future payments to Royalty Pharma are denominated in US dollars and are therefore sensitive to changes in the euro / US Dollar exchange rate.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
Income Tax Benefits / Expenses
The Group recorded total income tax benefit of € 1.2 million in 2023 (2022: income tax expense of € 168.6 million; 2021: income tax benefit of € 76.6 million), which consisted of current tax income of € 1.5 million, (2022: current tax expense of € 0.6 million; 2021: current tax benefit of € 1.2 million) and deferred tax expense of € 0.3 million (2022: deferred tax expense of € 168.0 million; 2021: deferred tax benefit of € 75.4 million). The effective income tax rate equaled 0.6% in the reporting year (2022: 962.2%; 2021: 13.0%). No deferred taxes were recognized in the current financial year, as the conditions for not recognizing a surplus of assets as at 31. December 2023 are still met. In 2022 the existing deferred tax assets on loss carryforwards and temporary differences of MorphoSys AG were derecognized in the amount necessary due to a high probability of a history of losses occurring as of December 31, 2023. In 2021 the difference was primarily due to the permanent
90
difference on the impairment of goodwill as well as the effect of the non-recognition of deferred tax assets on temporary differences and current year tax losses for the U.S. tax group.
Consolidated Net Profit/Loss for the Period
In 2023, the consolidated net loss amounted to € 189.7 million (2022: consolidated net loss of € 151.1 million; 2021: consolidated net loss of € 514.5 million).
B.Liquidity and Capital Resources
Sources of Funding
We have funded our operations through cash proceeds from ongoing business operations, including upfront fees, milestone payments, license fees, royalties, and service fees from strategic partners.
Additionally, MorphoSys completed a private placement via an accelerated book building process in December 2023 raising gross proceeds of € 102.7 million. The proceeds of the transaction will be used to support the ongoing clinical development of key pipeline programs to regulatory approval and to accelerate launch preparations for pelabresib in first-line myelofibrosis.
Cash and investments are presented in the balance sheet items "Cash and Cash Equivalents" and current and non-current "Other Financial Assets."
On December 31, 2023, the Group had cash and investments of € 680.5 million, compared to € 907.2 million on December 31, 2022.
Cash in excess of immediate working capital requirements is invested in accordance with our investment policy, primarily with a view to liquidity and capital preservation. We pay particular attention to liquidity as well as capital preservation and invest mainly in money market funds, corporate bonds and fixed-term deposits with fixed or variable interest rates.
Our functional currency is the euro. Nevertheless, we have liquidity in U.S. dollars, which could lead to exchange rate gains or losses in our finance income/expenses depending on the fluctuation of the euro/U.S. dollar exchange rate.
As of December 31, 2023, we are not subject to any operating covenants or capital requirement covenants. The Business Combination Agreement with Novartis effective February 5, 2024, includes Interim Operating Covenants (IOC) MorphoSys is obliged to adhere to.
Based on the company's recent corporate planning, which also incorporates the additionally released positive cash impacts from the sale of tafasitamab to Incyte as announced on February 5, 2024, MorphoSys believes that its liquidity is sufficient to finance its operational activities until early 2026, including the convertible bonds repayment. Any potential cashflows resulting from the Novartis Business Combination Agreement as announced February 5, 2024, were not considered in this recent corporate planning.
Under the Business Combination Agreement, Novartis agreed to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to provide MorphoSys with the financial resources required following completion of the Novartis Takeover Offer to enable MorphoSys to pay any obligations of MorphoSys arising from the implementation of the Novartis Takeover Offer as and when due, for example, but not limited to, the obligation from the convertible bonds and the obligations arising form the long-term incentive plans.
For the unlikely case that Novartis would withdraw its takeover offer and MorphoSys consequently would remain a stand-alone company, management would need to assess different financing options to ensure the going-concern assumption beyond early 2026 according to regulatory requirements. Management would then consider both non-dilutive financing options, such as out-licensing of (pre-)clinical assets or the sale of potential future royalties, but also considers accessing the capital markets by way of issuance of new shares or share instruments (ADSs) and/or issuance or refinancing of convertible debt.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024 , please refer to the section "6.9 - Subsequent Events."
Uses of Funding
We primarily use cash and other financial assets to fund the research and development costs related to the development of our product candidates. Our primary future funding requirements include the development and commercialization of our proprietary clinical pipeline, particularly in relation to pelabresib and, to a lesser extent, tulmimetostat.
91
As outlined before, the Company's recent corporate planning, which also incorporates the additional cash impacts from the sale of tafasitamab to Incyte as announced on February 5, 2024, MorphoSys believes that its liquidity is sufficient to finance its operational activities until early 2026. Any potential cashflows resulting from the Novartis Business Combination Agreement as announced on February 5, 2024, were not considered in this recent corporate planning. For the unlikely case that Novartis would withdraw its takeover offer and MorphoSys consequently would remain a stand-alone company, management would need to assess different financing options to ensure the going-concern assumption beyond early 2026 according to regulatory requirements. Management would then consider both non-dilutive financing options, such as out-licensing of (pre-) clinical assets or the sale of potential future royalties, but also considers accessing the capital markets by way of issuance of new shares or share instruments (ADSs) and/ or issuance or refinancing of convertible debt.
Since Management believes that the voluntary takeover offer by Novartis, which was announced February 5, 2024, will obtain the minimum acceptance threshold of 65% of MorphoSys share capital, bondholders will receive the right to redeem their bonds at the point in time Novartis takes over control over MorphoSys. The redemption payment will comprise the nominal amount and the accrued interest by the date of control is taken over. Based on the Business Combination Agreement, Novartis undertakes to MorphoSys to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to provide the MorphoSys Group with the financial resources required following completion of the Novartis Takeover Offer to enable the relevant MorphoSys Group companies pay any obligations arising from the implementation of the Novartis Takeover Offer as and when due, including any obligations for example, but not limited to, from the convertible bond to the extent triggered by completion of the Novartis Takeover Offer.
We have based the estimate of the liquidity status on assumptions that may prove to be incorrect, and it is possible that we may utilize our capital resources more quickly than anticipated. The process of investigating product candidates in clinical trials and their commercialization is fundamentally an expensive process. Both the timing and progress of development trials as well as the success of commercialization cannot be predicted with certainty.
As our product candidates are in various stages of development and the outcome of our activities is uncertain, we cannot estimate the amounts required in their entirety to successfully complete the development and commercialization of our product candidates.
Contractual Obligations
The following table summarizes our contractual obligations at December 31, 2023.
| Payments due by period | ||||||||||||||||||||||||||
| (in € thousands) | Total | Less than 1 year | Between One and Five Years | More than 5 years | ||||||||||||||||||||||
| Leases | 13,360 | 4,124 | 9,237 | 0 | ||||||||||||||||||||||
| Other | 57,133 | 2,996 | 54,137 | 0 | ||||||||||||||||||||||
The item "Other" consists of future minimum payments under performance share unit programs and contracts for insurance and other services.
Lease Obligations
We enter into long-term leases for facilities, company cars and equipment. The majority of these leasing contracts can be renewed on a yearly or quarterly basis, and some agreements may be terminated prematurely.
Other Commitments
Other commitments may become due for future payments for outsourced studies. After December 31, 2023, future payments for outsourced studies of approximately € 276.7 million may become due, of which approximately € 133.7 million will be paid in the next 12 months.
If certain milestones are achieved by MorphoSys (for example, submitting an investigational new drug (IND) application for specific target molecules), this may trigger milestone payments to licensors of up to an aggregate of US$ 236.5 million (€ 214.0 million) related to regulatory events or the achievement of sales targets.
We do not currently have any off-balance-sheet arrangements and did not have such arrangements in the years 2023 or 2022 that have or are reasonably likely to have a material current or future effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, cash requirements or capital resources.
92
As of the date of this Annual Report, we do not have any off-balance sheet arrangements other than operating leases as described under this “Item 5. Operating and Financial Review and Prospects—E. Critical Accounting Estimates Disclosure” above.
Cash Flows
Net Cash Provided by/ (Used in) Operating Activities
In 2023, net cash used by operating activities amounted to € 295.8 million and was mainly attributable to the consolidated net loss of € 189.7 million (2022: consolidated net loss € 151.1 million) and changes in non-cash items. The consolidated net loss resulted both in 2023 and 2022 mainly from expenses incurred to finance MorphoSys’ ongoing operations, specifically cost of sales, research and development expenses, selling expenses, and general and administrative expenses. Changes in non-cash items included mainly income from the net change of financial assets and financial liabilities from collaborations amounting to € 106.7 million (2022: € 301.1 million), net change from future payments to Royalty Pharma of € 45.7 million (2022: € 46.8 million), € 24.5 million net gains from other financial assets (2022: € 3.2 million) as well as non-cash expenses for convertible bonds amounting to € 5.3 million (2022: € 12.5 million). These effects were offset by expenses for scheduled and non-scheduled depreciation and amortization of tangible and intangible assets and right-of-use assets amounting to € 32.8 million (2022: € 18.3 million), mainly related to impairments of licenses, inventory, and right-of-use assets. The expenses for shared based payments in the amount of € 27.4 million (2022: € 3.6 million), non-cash expense from the share of loss of associates accounted for using the at-equity method amounting to € 8.2 million (2022: € 4.3 million), as well as changes in the fair value of the anti-dilution right in the amount of € 4.3 million (2022: € 0.0 million) contributed to this offsetting effect.
Changes in operating assets and liabilities in 2023 including paid income taxes amounted to € 5.6 million (2022: € 51.9 million) in the reporting year. This results from a € 56.9 million decrease in trade receivables (2022: € 18.2 million increase) as well as from an increase of contract liabilities by € 19.4 million (2022: decrease by € 0.3 million). Offsetting effects arose from an increase in inventories, prepaid expenses and other assets of € 24.8 million (2022: increase of € 11.9 million), which was mainly due to higher prepayments for external laboratory services, and from a decrease in accounts payable and accrued liabilities by € 44.3 million (2022: decrease of € 21.1 million), mainly due to lower outstanding trade payables at year-end. Furthermore, MorphoSys paid income taxes in the amount of € 1.6 million (2022: € 0.5 million).
In 2022, net cash used by operating activities amounted to € 366.7 million and was mainly attributable to the consolidated net loss of € 151.1 million and changes in operating assets and liabilities, including income taxes paid, totaling € 51.9 million. This was offset by non-cash items totaling € 163.7 million. The consolidated net loss of € 151.1 million (2021: consolidated net loss € 514.5 million) in 2022 and 2021 resulted mainly from expenses incurred to finance MorphoSys’ ongoing operations, specifically cost of sales, research and development expenses, selling expenses, and general and administrative expenses. Non-cash items included mainly income tax expenses in the amount of € 168.6 million mainly from the reversal of deferred tax assets (2021: tax benefits in the amount of € 76.6 million), scheduled and non-scheduled depreciation and amortization of tangible and intangible assets and right-of-use assets amounting to € 18.3 million (2021: € 246.0 million mainly from impairment of goodwill) and non cash effective change of bonds amounting to € 12.5 million (2021: € 12.1 million). These were offset by income from the net change in financial assets or liabilities from collaborations of € 301.1 million (2021: € 16.0 million), net change in financial liabilities from future payments to Royalty Pharma of € 46.8 million (2021: € 42.8 million) and non cash income from the capitalization of investments in associates of € 19.9 million for HI-Bio (2021: € 0.0 million). Changes in operating assets and liabilities in 2022 mainly included an increase in inventories, prepaid expenses and other assets of € 11.9 million (2021: increase of € 30.3 million) and a decrease in accounts payable and accrued liabilities by € 21.1 million (2021: decrease of € 90.8 million). The increase in inventories, prepaid expenses and other assets is mainly due to higher prepayments for external laboratory services. The reason for the decrease in trade payables and accrued liabilities was mainly due to lower outstanding trade payables at year-end. Furthermore, MorphoSys paid income taxes in the amount of € 0.5 million (2021: € 64.6 million).
In 2021, net cash used by operating activities amounted to € 481.4 million and was mainly attributable to the consolidated net loss of € 514.5 million and changes in operating assets and liabilities, including income taxes paid, totaling € 177.6 million. This was offset by non-cash items totaling € 210.6 million. The consolidated net loss of € 514.5 million resulted mainly from expenses incurred to finance MorphoSys’ ongoing operations, specifically cost of sales, research and development expenses, selling expenses, and general and administrative expenses. Net profit in 2020 resulted mainly from revenues from the collaboration and license agreement with Incyte, which was not recurring in 2021. Non-cash items included mainly income tax benefits in the amount of € 76.6 million (2020: € 75.4 million) and the net change in financial assets / liabilities from collaborations in the amount of € 16.0 million (2020: € 36.6 million). These were offset by the net change in financial liabilities from future payments to Royalty Pharma in the amount of € 42.8 million (2020: € 0), scheduled depreciation and amortization as well as impairments of tangible and intangible assets and right-of-use assets amounting to € 246.0 million (2020: € 24.8 million) and the full year non cash effective change of bonds amounting to € 12.1 million (2020: € 2.5 million). Changes
93
in operating assets and liabilities in 2021 mainly included an increase in inventories, prepaid expenses and other assets of € 30.3 million (2020: increase of € 8.5 million), partially offset by a decrease in accounts receivable of € 10.5 million (2020: decrease of € 69.6 million). Accounts payable and accrued liabilities decreased by € 90.8 million (2020: increase of € 77.5 million). The main reason for this decline relates to accounts payable and accrued expenses of Constellation, which were included for the first time due to the acquisition on July 15, 2021. The accrued expenses and accounts payable of Constellation mainly comprised share-based payment obligations to Constellation's employees that became due on the date of the acquisition by MorphoSys as well as accrued transaction costs. Their subsequent payment in 2021 led to the decrease presented in this cash flow item. The year-on-year decrease in accounts receivable was mainly due to lower outstanding receivables at the end of the year 2021. The increase in inventories, prepaid expenses and other assets was due in particular to the higher inventories for the commercialization of Monjuvi® in the U.S. Furthermore, MorphoSys paid € 64.6 million of income taxes in financial year 2021 due to net profit in 2020 (2020: € 0.3 million).
Net Cash Provided by/ (Used in) Investing Activities
In 2023, net cash provided by investing activities amounted to € 15.4 million. This results from € 18.2 million of interest received, € 4.4 million cash receipts from sales of investments accounted at fair value through other comprehensive income, € 4.6 million cash receipts from sales of shares of investment in associates, as well as proceeds from the sale of other financial assets amounting to € 3,142.3 million. These were offset by payments to acquire other financial assets amounting to € 3,151.2 million and by payments to acquire tangible and intangible assets of € 2.9 million.
In 2022, net cash provided by investing activities amounted to € 345.0 million, primarily driven by proceeds from the sale of other financial assets amounting to € 2,240.7 million. These were offset by payments to acquire other financial assets amounting to € 1,884.9 million. This net cash inflow from investing activities was mainly due to a shift in the composition of our investment portfolio, as securities matured and were sold and new, comparable securities were acquired. In addition, € 13.3 million was used for the acquisition of intangible assets in 2022.
In 2021, net cash used in investing activities amounted to € 831.0 million, primarily driven by payments to acquire other financial assets amounting to € 2,188.3 million. These were offset by proceeds from the sale of other financial assets amounting to € 2,592.0 million. This net cash outflow from investing activities was mainly due to a shift in the composition of our investment portfolio, as securities matured and were sold and new, comparable securities were acquired. The cash outflow relating to the acquisition of 100% shares in Constellation, net of acquired cash, in 2021 amounted to € 1,206.6 million. In addition, € 22.3 million was used for the acquisition of intangible assets in 2021.
Net Cash Provided by/ (Used in) Financing Activities
Net cash provided by financing activities amounted to € 43.0 million in 2023 and was primarily driven by the capital increase in December 2023 that resulted in net cash inflows of € 96.0 million. This was partially compensated by cash payments for repurchases of own convertible bonds including transaction costs of € 40.8 million. Furthermore, cash outflows of € 2.4 million from financing collaborations from Incyte, interest paid in the amount of € 1.8 million, as well as cash payments for the principal element of lease payments in the amount of € 8.0 million were recorded.
Net cash provided by financing activities amounted to € 311.4 million in 2022 and consisted primarily of the cash receipts from the contracts with Royalty Pharma (development funding bond) in the amount of € 295.4 million as well as proceeds of € 23.8 million from financing collaborations from Incyte.
Net cash provided by financing activities amounted to € 1,322.9 million in 2021 and consisted primarily of the cash receipts from the contracts with Royalty Pharma in the amount of € 1,206.7 million and the proceeds from the issuance of shares of € 84.7 million to Royalty Pharma as well as proceeds of € 40.0 million from financing collaborations from Incyte.
Investments
In 2023, MorphoSys invested € 0.4 million in property, plant and equipment (2022: € 1.9 million), mainly office and laboratory equipment (i.e., machinery). Depreciation of property, plant and equipment in 2023 decreased to € 2.3 million (2022: € 2.9 million).
MorphoSys invested € 2.5 million in intangible assets in the reporting year (2022: € 13.3 million). Of this amount, € 2.4 million was spent on internally generated intangible assets. Amortization of intangible assets amounted to € 3.6 million in 2023 (2022: € 3.6 million). In 2023, impairment losses of € 9.6 million were recognized on intangible assets, thereof € 0.7 million on internally generated intangible assets.
94
Net Assets
Assets
At € 2,026.3 million, total assets as of December 31, 2023, were € 370.6 million lower compared to December 31, 2022 (€ 2,396.9 million).
Current assets decreased by € 275.0 million to € 814.0 million. As of December 31, 2023, Cash and Cash Equivalents amounted to € 158.5 million compared to € 402.4 million as of December 31, 2022. This change was mainly due to the consumption of cash for operations in 2023. The capital increase in December 2023 resulted in gross proceeds of € 102.7 million. Transaction cost were € 6.7 million, which led to a net cash inflow of € 96.0 million. In addition, the partial redemption of the convertible bond as of March 30, 2023, resulted in a cash outflow including transaction costs of € 40.8 million. Furthermore, accounts receivable decreased by € 59.1 million and prepaid expenses decreased by € 20.6 million. The decrease was partly offsett by an increase in inventories of € 37.8 million. Included within the Inventory value is drug substance owned by MorphoSys prepaid by the customer of € 19.4 million. Other current financial assets amounted to € 520.8 million (December 31, 2022: € 504.8 million), which were primarily invested in term deposits with fixed interest rates.
Non-current assets decreased by € 95.6 million from a balance of € 1,307.9 million as of December 31, 2022, to € 1,212.3 million as of December 31, 2023, mainly due to a € 42.5 million decrease in intangible assets as well as a decrease of "Goodwill" by € 13.9 million, resulting from the change in the euro/U.S. dollar exchange rate compared to December 31, 2022. The balance sheet position "Right-of-Use Asset" decreased mainly due to a revised assessment of an extension right that had an effect of € 25.3 million. In addition, the balance sheet item "Investments in associates" declined by € 2.9 million to € 2.4 million as of December 31, 2023 (December 31, 2022: € 5.4 million).
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
Liabilities
Current liabilities decreased by € 14.0 million from € 278.3 million in the prior year to € 264.3 million as of December 31, 2023, mainly as a result of € 47.5 million decrease in the line item “accounts payable and accruals” due to regular fluctuation and timing effects, specifically due to reduced accruals for research and development activities related to tafasitamab. Opposing effect was a € 19.4 million increase of the current contract liability as well as a € 17.6 million increase in the current portion of liabilities from future payments to Royalty Pharma (refer to Note 4.20 “Financial Liabilities from Future Payments to Royalty Pharma” of the Notes to the Consolidated Financial Statements).
Non-current liabilities (December 31, 2023: € 1,713.0 million; December 31, 2022: € 1,961.2 million) decreased mainly due to a reduction in the non-current portion of financial liabilities from collaborations from € 217.8 million as of December 31, 2022, to € 108.9 million as of December 31, 2023, mainly due to lower expected future revenues for Monjuvi® in the U.S.A. (refer to Note 4.19 "Financial assets and liabilities from collaborations" of the Notes to the Consolidated Financial Statements). In addition, the item "Financial liabilities from future payments to Royalty Pharma" decreased by € 82.0 million (see section 4.20 "Financial liabilities from future payments to Royalty Pharma" of the Notes to the Consolidated Financial Statements). The € 47.6 million decrease in the carrying amount of the convertible bond issued in October 2020 mainly results from a partial redemption. A decrease in the non-current lease liability by € 29.4 million was mainly due to a revised assessment of an extension right that had an effect of € 25.3 million. Decreases are offset in particular by a € 19.7 million increase in non-current provision related to shared based compensation.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
Stockholders’ Equity
As of December 31, 2023, Group equity totaled € 49.0 million compared to € 157.4 million on December 31, 2022. The Company’s equity ratio as of December 31, 2023, amounted to 2% compared to 7% on December 31, 2022. This decrease in the equity ratio resulted mainly from the consolidated net loss of the financial year 2023.
The number of shares issued totaled 37,655,137 as of December 31, 2023, of which 37,601,452 shares were outstanding (December 31, 2022: 34,231,943 shares issued and 34,165,963 shares outstanding).
On December 14, 2023, a total of 3,423,194 shares were issued in the context of a cash capital increase from Authorized Capital 2023-II and fully exhausted the Authorized Capital 2023-II. As a result, the number of authorized ordinary shares decreased by 286,134 from 9,195,696 as of December 2022 to 8,909,562 shares as of December 31, 2023.
95
On December 31, 2023, the Company held 53,685 treasury shares with a value of € 1,995,880 – a decrease of € 454,423 compared to December 31, 2022 (65,980 shares, € 2,450,303). The reason for this decrease was the transfer of 12,295 treasury shares amounting to € 454,423 to the Management Board and selected employees of the Company (beneficiaries) from the 2019 Long-Term Incentive Plan (LTI Plan). The vesting period for this LTI Plan expired on April 1, 2023, and offered beneficiaries a six-month period until November 3, 2023 to receive a total of 12,295 shares.
The development of the equity of the parent company MorphoSys AG (including the assessment with regard to the provision of Section 92 German Stock Corporation Act) as well as of MorphoSys Group is closely monitored by the Management Board. In addition, the company is closely monitoring the liquidity situation of MorphoSys Group and of MorphoSys AG, and believes that MorphoSys has sufficient liquid funds to ensure business operations for the forecast period (at least twelve months from the issuance date of the consolidated and statutory financial statements), which is subject to the going-concern assessment, without requiring additional proceeds from external refinancing. Any potential cashflows resulting from the Novartis Business Combination Agreement as announced on February 5, 2024, were not considered in the recent corporate planning.
Based on the company's recent corporate planning, which also incorporates the additionally released positive cash impacts from the sale of tafasitamab to Incyte as announced on February 5, 2024, MorphoSys believes that its liquidity is sufficient to finance its operational activities until early 2026, including the convertible bonds repayment. Any potential cashflows resulting from the Novartis Business Combination Agreement as announced February 5, 2024, were not considered in this recent corporate planning.
Under the Business Combination Agreement, Novartis agreed to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to provide MorphoSys with the financial resources required following completion of the Novartis Takeover Offer to enable MorphoSys to pay any obligations of MorphoSys arising from the implementation of the Novartis Takeover Offer as and when due, for example, but not limited to, the obligation from the convertible bonds and the obligations arising form the long-term incentive plans, each to the extent triggered by the completion of the Novartis Takeover Offer.
For the unlikely case that Novartis would withdraw its takeover offer and MorphoSys consequently would remain a stand-alone company, management would need to assess different financing options to ensure the going-concern assumption beyond early 2026 according to regulatory requirements. Management would then consider both non-dilutive financing options, such as out-licensing of (pre-) clinical assets or the sale of potential future royalties, but also considers accessing the capital markets by way of issuance of new shares or share instruments (ADSs) and/or issuance or refinancing of convertible debt.
At the time of this report, the Management Board is not aware of any imminent risks, neither individually nor collectively, that could affect the company as a going concern.
Financial Opportunities
Exchange rate and interest rate developments can positively or negatively affect our financial results. Interest rate and financial market developments are continuously monitored to promptly identify and take advantage of opportunities.
C.Research and Development; Patents and Licenses
See “Item 4.A. History and Development of the Company” and “Item 4.B. Business Overview.”
D.Trend Information
Other than as disclosed elsewhere in this annual report, we are not aware of any trends, uncertainties, demands, commitments or events for the period from January 1, 2023 to December 31, 2023 that are reasonably likely to have a material adverse effect on our net revenues, income, profitability, liquidity or capital resources, or that caused the disclosed financial information to be not necessarily indicative of future operating results or financial conditions.
Changes in the Business Environment
Global economy growth is projected to stay stable at an estimated 3.1% in 2023 and in 2024, then rise to 3.2% in 2025 (report: “World Economic Outlook Update January 2024,” published by the International Monetary Fund [IMF]). According to the IMF, the global economic recovery from the COVID-19 pandemic, the war in Ukraine, and the cost-of-living crisis is proving surprisingly resilient. Inflation is falling faster than expected from its 2022 peak.
The IMF’s growth forecast for the advanced economies in 2023 was +1.6%, compared to 2.6% in 2022, and the forecast for the emerging and developing economies was +4.1% (2022: +4.1%). The IMF’s estimate for growth in the euro area in 2023 was
96
+0.5% (2022: +3.4%), compared to -0.3% for Germany (2022: +1.8%); +2.5% for the U.S. (2022: +1.9%); and +5.2% for China (2022: +3.0%).
When managing its business activities, MorphoSys takes a number of potential macroeconomic risks and opportunities into consideration.
Lastly, MorphoSys AG has implemented a business continuity plan to prevent the collapse of critical business processes to a large extent or to enable the resumption of critical business processes in case a natural disaster, public health emergency or other serious event occurs. However, depending on the severity of the situation, it may be difficult or in certain cases impossible for us to continue our business for a significant period of time. Our contingency plans for disaster recovery and business continuity may prove inadequate in the event of a serious disaster or similar event and we may incur substantial costs that could have a material adverse effect on our business.
Currency Development
The EUR/USD exchange rate has fluctuated between 1.04 and 1.12 over the last year and stood at 1.10 on December 31, 2023, with inflation expectations and interest rate differences being the main drivers, in addition to trade conflicts and ongoing geopolitical tensions.
The majority of our business transactions are conducted in euros and U.S. dollars. With the acquisition of Constellation we have significantly expanded our footprint in the U.S. Primarily driven by the additional ongoing clinical studies, U.S. dollar expenses are expected to exceed U.S. dollar revenues for the next financial year. Therefore, strengthening of the U.S. dollar against the euro, all other things remaining equal, would have a negative impact on our operating result. We manage this risk through various mechanisms, such as optimizing our U.S. dollar assets against our U.S. dollar liabilities and maintaining an adequate (currently around 35%) amount of U.S. dollars in our bank accounts.
E.Critical Accounting Estimates
Not applicable.
Item 6.Directors, Senior Management and Employees
A.Directors and Senior Management
We are a German stock corporation and, in accordance with the German Stock Corporation Act, we have a two-tier board structure consisting of our Supervisory Board and a separate Management Board.
Our Supervisory Board supervises the policies of the Management Board and the general course of the affairs of our business. The Supervisory Board advises the Management Board and is guided by the interests of the business when performing its duties. The Management Board is in charge of managing us under the supervision of the Supervisory Board. The Management Board provides the Supervisory Board with such necessary information as the Supervisory Board requires to perform its duties.
Supervisory Board
The Supervisory Board consists of six professionally qualified members who represent our shareholders. The Chairman of the Supervisory Board (Marc Cluzel, M.D., Ph.D.), coordinates the Board’s activities, chairs the Supervisory Board meetings and represents the interests of the Supervisory Board externally. All Supervisory Board members are independent, as defined in the German Corporate Governance Code and the Nasdaq Listing Rules, and have many years of experience in the biotechnology and pharmaceutical industries. The Chairman of the Supervisory Board is not a former member of our Management Board. The members of the Supervisory Board and its committees are listed in the table below.
97
| Name | Year of birth | End of current period | Principal business activities performed outside of MorphoSys | |||||||||||||||||
| Marc Cluzel (Chairman), M.D., Ph.D. | 1955 | 2024 | Consultant and business professional; member of the board of directors of Moleac Pte. Ltd.; member of the board of directors of Griffon Pharmaceuticals Inc. | |||||||||||||||||
| George Golumbeski, Ph.D. (Deputy Chairman) | 1957 | 2024 | Business consultant in the life science and healthcare industries; chair of the board of directors of Carrick Therapeutics Ltd.; chair of the board of directors of Ananke Therapeutics Inc., member of the board of directors of Sage Therapeutics Inc.; chair of the board of directors of Shattuck Labs Inc.; chair of the Board of Directors of Actio Biosciences; member of the Board of Directors of Chroma Medicine | |||||||||||||||||
| Krisja Vermeylen | 1962 | 2024 | Business consultant in the life science and healthcare industries; member of the board of directors of Diaverum AB (until December 31, 2023) | |||||||||||||||||
| Michael Brosnan | 1955 | 2026 | Consultant in the life sciences and healthcare industries; member of the board of directors of Daimler Truck AG; member of the board of directors of Daimler Truck Holding AG; member of the board of directors of CureVac SE and CureVac N.V. | |||||||||||||||||
| Sharon Curran | 1968 | 2024 | Non-Executive Director in life sciences and healthcare industries; member of the board of directors of NIOX group plc., member of the board of directors of Spinnaker TopCo Ltd. / Norgine | |||||||||||||||||
| Andrew Cheng, M.D., Ph.D. | 1967 | 2025 | President and Chief Executive Officer of Akero Therapeutics, Inc.; member of the Board of Directors of Vera Therapeutics, Inc. | |||||||||||||||||
The following is a brief summary of the business experience of the members of our Supervisory Board:
Marc Cluzel, M.D., Ph.D., has been a member of our Supervisory Board since 2012 and Chairman of the Supervisory Board since the AGM 2018. He was Executive Vice President of Product Development at HÙYÀ Bioscience International, LLC from 2011 to 2012. Prior to that, between 1993 and 2010, he held several positions at Sanofi-Aventis, including Executive Vice President of Research and Development. Marc Cluzel received his Ph.D. in Biochemistry and his Doctor of Medicine from the University of Montpellier, France.
George Golumbeski, Ph.D., has been a member of our Supervisory Board since 2018. He currently serves as a Partner at DROIA Ventures and as a self-employed business consultant in the life science and healthcare industries. Prior to that he held the position as President of Grail Inc., and from April 2018 to March 2020 he served as the leader of Business Development, Collaboration and M&A activities at Celgene Corporation. Over the last 27 years, he has held leadership roles in business and corporate development, partnering and M&A with global pharmaceutical and life science companies, including Celgene Corporation, Novartis, Elan Corporation (today: Perrigo), and Schwarz Pharma (today: UCB). Dr. Golumbeski obtained his Doctorate in Genetics from the University of Wisconsin in Madison, USA and holds a degree in Biology from the University of Virginia, Charlottesville, USA.
Krisja Vermeylen has been a member of our Supervisory Board since 2017. From 1997 to October 2018, Mrs. Vermeylen held several positions at Novo Nordisk, including positions as General Manager of major EU markets and the position as Senior Vice President Corporate People & Organization. Prior to that, she held several positions at Pharmacia and Upjohn. Mrs. Vermeylen graduated with a Master in Pharmaceutical Sciences from the University of Antwerp, Belgium and is a certified Independent Director from INSEAD, France.
Michael Brosnan has been a member of our Supervisory Board since 2018. Currently he serves as a consultant in the life sciences and healthcare industries. Mr. Brosnan has over 40 years of experience in finance, controlling and auditing. From 2010 to 2019, he served as Chief Financial Officer of Fresenius Medical Care Management AG, a company with a dual listing in Germany (Frankfurt) and the United States (NYSE). Over the last 20 years, he has worked in various leadership and executive
98
positions for Fresenius Medical Care in the United States and Germany. Prior to joining Fresenius Medical Care, he held senior financial positions at Polaroid Corporation and was an audit partner at KPMG. Mr. Brosnan holds a degree in Business Administration and Accounting from Northeastern University, Boston, Massachusetts, USA.
Sharon Curran has been elected as a new member of our Supervisory Board during the AGM 2019. Ms. Curran currently serves as a Non-Executive Director in the life sciences and healthcare industries. Prior to that, Ms. Curran worked for AbbVie Inc., Illinois, USA as Vice President, Global Specialty Franchise and Customer Excellence and has also held a number of other senior positions in her career including Vice President Global Marketing Specialty, AbbVie; Global Brand and Commercial Director, Abbott MBO and Division Head, Eli Lilly UK & Ireland. Ms. Curran brings extensive commercial and specialty pharmaceutical experience to the Company. She holds an Executive Master of Science, Business Administration from Trinity College Dublin, Ireland, and a Bachelor of Science in Biotechnology from Dublin City University, Ireland.
Andrew Cheng, M.D., Ph.D., has been a member of our Supervisory Board since 2022. Currently he is a Director, President and Chief Executive Officer of Akero Therapeutics, Inc. Mr. Cheng has over 20 years of experience in the life sciences and healthcare industries. From 1999 to 2018, he has worked in various leadership and executive positions for Gilead Sciences in the United States including Chief Medical Officer and Executive Vice President, Clinical Research and Development Operations. Mr. Cheng received his Ph.D. in Integrated Program Genetics and Biochemistry and his M.D. degree from Columbia University, New York, USA.
Management Board
The following table sets forth the names and function of the current members of our Management Board and their year of birth and terms:
| Name | Year of birth | End of current period | Position | |||||||||||||||||
| Jean-Paul Kress, M.D. | 1965 | August 31, 2025 | Chief Executive Officer | |||||||||||||||||
| Sung Lee | 1970 | March 17, 2023 | Chief Financial Officer | |||||||||||||||||
Lucinda Crabtree, Ph.D. | 1979 | August 6, 2026 | Chief Financial Officer | |||||||||||||||||
| Charlotte Lohmann | 1970 | August 31, 2023 | Chief Legal Officer | |||||||||||||||||
A schedule of responsibilities currently defines the different areas of responsibility as follows:
•Jean-Paul Kress, M.D., Chief Executive Officer: Strategy & Planning, Business Development & Alliance Management, Human Resources, Legal, Compliance & Intellectual Property, Corporate Affairs and Investor Relations, Technical Operations, Facilities & Information Technology, Quality Assurance & Internal Audit, Research & Development, Global Oversight of Commercial Operations, as well as for coordinating the individual areas of responsibility for each Management Board member and representing the Management Board vis-à-vis the Supervisory Board and the public.
•Lucinda Crabtree, Ph.D., Chief Financial Officer: Accounting & Taxes, Global Controlling & Internal Controls, Corporate Development & M&A; Central Purchasing and Logistics, Environmental Social Governance (ESG).
Jean-Paul Kress, M.D.
As CEO, Jean-Paul Kress leads MorphoSys in its mission – More life for people with cancer – by focusing on translating scientific innovation into medicines that can redefine how cancer is treated. Since becoming CEO in 2019, Jean-Paul has led a strategic and cultural transformation to build a focused global biopharmaceutical company. As part of this evolution, Jean-Paul championed the company’s acquisition of Constellation Pharmaceuticals in 2021. This landmark achievement for the company enriched MorphoSys’ pipeline with promising investigational medicines – most importantly a potential breakthrough therapy to treat myelofibrosis. Today, MorphoSys is pursuing multiple opportunities to deliver safer and more effective medicines for people living with blood cancers and solid tumors. Prior to joining MorphoSys, Jean-Paul was the CEO of Syntimmune, where he sharpened the company’s focus on late-stage clinical development, resulting in its acquisition by Alexion. He has held several senior leadership roles at other pharmaceutical companies, including Biogen, Sanofi, Gilead and AbbVie. Jean-Paul also served as Chair of the Board of Directors at ERYTECH Pharma, where he ushered in the company’s merger with Pherecydes. Prior to that, he was a member of Sarepta Therapeutics’ Board of Directors. Jean-Paul received his M.D. from Faculté Necker-Enfants Malades in Paris and graduate and post-graduate degrees in biochemistry and in molecular and cellular pharmacology from Ecole Normale Supérieure in Paris.
99
Sung Lee (until March 17, 2023)
Sung Lee came to MorphoSys in February 2021 and leads all corporate finance functions of the company and information technology. He has more than 20 years of finance leadership experience in biopharmaceutical and technology businesses. He joined MorphoSys from Sangamo Therapeutics, Inc., where he served as Chief Financial Officer. Prior to that role, Mr. Lee spent nearly 14 years at Gilead Sciences where he most recently led the global Financial Planning & Analysis and Investor Relations functions. He started his career in the tax advisory business at PricewaterhouseCoopers LLP. He received a Master of Business Taxation from the University of Southern California and a B.A. in Economics from the University of California, Irvine.
In December 2022, Sung Lee resigned as a member of the Management Board with effect as of the end of March 17, 2023.
Lucinda Crabtree, Ph.D. (from August 8, 2023 onwards)
Lucinda Crabtree, Ph.D., joined MorphoSys in August 2023. She has broad biotech experience from various corporate roles and her time as an investment professional and board observer. Prior to joining MorphoSys, Lucinda served as the Chief Financial Officer at Autolus Therapeutics, a clinical-stage biopharmaceutical company developing T cell therapies. In her previous roles at Autolus, she led Finance, Business Strategy and Planning, as well as Investor Relations and Corporate Communications functions. Prior to her time at Autolus, Lucinda worked for several years as a senior investment professional on both the buy and the sell side, and she served as a board observer for several private healthcare companies. Firms she has worked at include Woodford Investment Management, Panmure Gordon, Goldman Sachs, J.P. Morgan and Jefferies. Lucinda started her career as a research scientist at a U.K.-based biotech company. She holds a first class Bachelor of Science degree in Physiology and Pharmacology from University College London and a Ph.D. in Pharmacology from University College London.
Charlotte Lohmann (until August 31, 2023)
Charlotte Lohmann joined MorphoSys in May 2012. She is a very experienced lawyer with more than 25 years of professional practice, having worked in both top tier international audit and law firms as well as in publicly listed biotech companies. She is an accomplished executive that has established solution-oriented legal departments with a focus on performance, effectiveness and making the legal department a true business partner. She has been successful in setting up and executing international projects as well as public listings on Frankfurt Stock Exchange and Nasdaq and has vast experience in advising management and supervisory boards (both as Chief Legal Officer and supervisory board member). Prior to joining MorphoSys, she worked for Wilex AG as Sr. VP Legal Affairs & Human Resources and for KPMG as Corporate Lawyer. Charlotte Lohmann holds a Law Degree from the University of Munich and is an accredited lawyer in Germany.
Charlotte Lohmann is also a member of the Board of Directors of Vivoryon Therapeutics N.V., Munich/Halle, Germany (a publicly listed company).
Board Diversity
Based on the diversity concepts for the Management Board and the Supervisory Board, our evaluation of nominees for the Management Board and the Supervisory Board includes consideration of their ability to contribute to the diversity of personal and professional experiences, opinions, perspectives and backgrounds on our Supervisory Board or Management Board. Nominees are not discriminated against based on race, color, religion, sex, ancestry, national origin, sexual orientation, disability or any other basis prescribed by law.
100
| Board Diversity Matrix (as of February 15, 2024) | ||||||||||||||
Country of Principal Executive Office | Germany | |||||||||||||
Foreign Private Issuer | Yes | |||||||||||||
Disclosure Prohibited Under Home Country Law | No | |||||||||||||
Total Number of Directors | 8 | |||||||||||||
| Part I: Gender Identity | Female | Male | Non-Binary | Did Not Disclose Gender | ||||||||||
Directors | 3 | 5 | 0 | 0 | ||||||||||
Part II: Demographic Background | ||||||||||||||
Asian | 1 | |||||||||||||
White | 7 | |||||||||||||
Underrepresented Individual in Home Country Jurisdiction | 0 | |||||||||||||
Did Not Disclose Demographic Background | 0 | |||||||||||||
Service Agreements
The service agreements with our Management Board members generally have a total term of three years. The current service agreement of our Chief Executive Officer Jean-Paul Kress, M.D., runs until August 31, 2025. Our Chief Financial Officer Lucinda Crabtree, Ph.D., has entered into a service agreement for a three year term until August 6, 2026.
In the event of (i) a change of control and (ii) a material reduction of his/her responsibilities within one year after the change of control, the Chief Executive Officer Jean-Paul Kress, M.D., as well as the Chief Financial Officer Lucinda Crabtree, Ph.D., are entitled to resign from their office as member of the Management Board and simultaneously terminate the service agreement against the payment of the outstanding fixed salary and annual bonus for the remainder of the fixed contract period, however, that such amount shall not exceed twice the annual remuneration.
B.Compensation
The following Remuneration Report provides an explanation and a clear and comprehensible presentation of the remuneration individually awarded and due to the current and former members of the Management Board and the Supervisory Board of MorphoSys AG (“the Company”) in the 2023 financial year.
Beyond the requirements of Section 162 (3) sentence 1 and 2 of the German Stock Corporation Act (Aktiengesetz, "AktG"), the Management Board and the Supervisory Board decided to have the Remuneration Report audited not only formally but also materially by the appointed auditor. The audit opinion is attached to the Remuneration Report 2023. The Remuneration Report will be submitted to the Annual General Meeting 2024 for approval.
The masculine form is used in this Remuneration Report for convenience purposes only, and refers equally to all genders.
a.Review of the 2023 Financial Year
I.Economic Environment in the 2023 Financial Year
In fiscal year 2023, MorphoSys AG and its employees focused on achieving its long-term goals and defined priorities. At the beginning of the year, the company announced the termination of its preclinical research programs in order to optimize its cost structure and focus on the most important strategic priorities. A repurchase of approximately 19% of the outstanding aggregate principal amount of MorphoSys's convertible bonds was completed on financially attractive terms in March 2023. Major
101
progress was made in the development of the clinical pipeline in 2023. In early April, the completion of patient recruitment for both the pivotal Phase 3 MANIFEST-2 trial and the Phase 3 trials frontMIND in DLBCL in first-line treatment was announced. In August 2023, the Phase 3 inMIND trial examining tafasitamab in relapsed or refractory follicular or marginal zone lymphoma, conducted by Incyte, also completed recruitment. At the end of November 2023, the first topline data from the MANIFEST-2 trial for the treatment of myelofibrosis became available, earlier than initially expected. Pelabresib in combination with ruxolitinib in the first-line treatment of myelofibrosis showed convincing results with clinical improvements in all key endpoints and a clinically meaningful improvement in anemia. Detailed results from the MANIFEST-2 study were presented in an oral presentation at the ASH Annual Meeting in December 2023. Throughout the year, the latest clinical data for pelabresib, tafasitamab and tulmimetostat were presented at various scientific conferences, including new preliminary results from an arm of the MANIFEST study evaluating pelabresib as monotherapy in patients with high-risk essential thrombocythemia (ET). In September, tulmimetostat received Fast Track designation from the FDA for the treatment of ARID1A-mutated endometrial cancer. On December 14, 2023, MorphoSys announced the completion of a share capital increase from € 34,231,943 by € 3,423,194 to € 37,655,137 through a full utilization of its authorized capital 2023-II, resulting in gross proceeds of approximately € 102.7 million. In the 2023 financial year, the company was able to slightly increase sales figures from Monjuvi® in the USA as planned despite a stronger competitive environment. The key partner programs, developed via MorphoSys' legacy antibody technology platform, also made good progress in 2023.
Thus, from the Company's perspective, important goals were achieved in 2023 to keep MorphoSys on the path to a leading position in the fields of hematology and oncology in order to create sustainable long-term value.
The intention announced by Novartis on February 5, 2024, to make a voluntary public takeover offer for all shares of MorphoSys at an equity value of € 2.7 billion and the Business Combination Agreement entered into with Novartis underscores the value created by the company and the progress made in MorphoSys's development pipeline.
The remuneration of the members of the Management Board of MorphoSys AG shall appropriately reward the performance of the Management Board members and decrease significantly if targets are not achieved (“Pay for Performance”). For this reason, the success and milestones achieved in the 2023 financial year were also reflected in the Management Board members’ variable remuneration.
For further detailed information on the business environment during the 2023 financial year, please refer to the MorphoSys AG Annual Report.
II.Resolution on the Remuneration Report for the 2022 Financial Year
For the 2022 financial year, a remuneration report was prepared in accordance with Section 162 AktG, audited also materially. The remuneration report for the 2022 financial year was approved by the 2023 Annual General Meeting with a majority of over 94%. Given the high approval rate of the remuneration report for the 2022 financial year, there was no reason to change the method of reporting.
III.Remuneration System for the Members of the Management Board
The currently applicable remuneration system for the members of the Management Board (the "Remuneration System 2022") has been approved by the Company’s Annual General Meeting on May 18, 2022 with a majority of 86.95%. The Remuneration System 2022 applies to the service agreements of the current members of the Management Board, Jean-Paul Kress, M.D., and Lucinda Crabtree, Ph.D. Further, the Remuneration System 2022 applied to the service agreement of the former member of the Management Board Charlotte Lohmann, who was a member of the Management Board from March 1, 2023, until the end of August 31, 2023. The service agreement of Sung Lee, who left the Company during the reporting year, did not fall under any remuneration system since it was already concluded before the implementation of the first remuneration system. To the extent the Remuneration System 2022 applied during the 2023 financial year, there were no deviations thereof within the meaning of Section 162 (1) no. 5 AktG.
The Remuneration System 2022 is published on the Company's website at www.morphosys.com/en/investors/corporate-governance. The main elements of the Remuneration System 2022 are summarized in this remuneration report.
The Supervisory Board has decided to further develop and revise the Remuneration System 2022 and to submit a new remuneration system 2024 to the Annual General Meeting 2024 for approval.
IV.Remuneration System for Members of the Supervisory Board
The remuneration system resolved for the members of the Company’s Supervisory Board by the Annual General Meeting of the Company on May 19, 2021, continued to apply unchanged in the 2023 financial year. The remuneration system for the members of the Supervisory Board is published on the Company's website at www.morphosys.com/en/investors/corporate-governance.
102
The Supervisory Board has decided to further develop and revise the Remuneration System 2022 and to submit a new remuneration system 2024 to the Annual General Meeting 2024 for approval.
V. Changes in the Composition of the Management Board and Supervisory Board
In the 2023 financial year, the following changes occurred in the composition of the Management Board: With effect as of the end of March 17, 2023, Sung Lee resigned from his position as member of the Management Board and Chief Financial Officer of the Company. Concurrently, his service agreement has been terminated with effect as of the end of March 31, 2023. Further, Charlotte Lohmann was appointed as member of the Management Board and Chief Legal Officer for the time period from March 1, 2023, to the end of August 31, 2023 on an interim basis. As of August 8, 2023, Lucinda Crabtree, Ph.D., was appointed as member of the Management Board and Chief Financial Officer for a term of three years until the end of August 6, 2026.
The following changes in the composition of the Supervisory Board occurred in the 2023 financial year: Michael Brosnan and George Golumbeski, Ph.D. were re-elected as members of the Supervisory Board. Michael Brosnan was re-elected until the end of the General Meeting that resolves upon the discharge of the Supervisory Board for the second financial year following the beginning of the term of office (i.e., presumably until the end of the Annual General Meeting 2026). George Golumbeski, Ph.D., was re-elected until the end of the General Meeting that resolves upon the discharge of the Supervisory Board for the 2023 financial year 2023 (i.e., presumably until the end of the Annual General Meeting 2024).
b.Remuneration of the Members of the Management Board of
MorphoSys AG
MorphoSys AG
I.Remuneration System 2022 and 2023
1.Overview
The Remuneration System 2022 consists of fixed remuneration components (annual base salary, fringe benefits and pension contributions), an annual bonus (short-term incentive (STI)) and a performance-related, long-term variable remuneration (long-term incentive, (LTI)) in the form of a Performance Share Unit Program ("PSUP").
The annual bonus depends on the achievement of a financial and a commercial target (each weighted at 25%), as well as on a development and business development and licensing (BD&L) target (weighted between 30% and 40%) and a research and BD&L target (weighted between 10% and 20%). The payout amount is capped at 160% of the annual base salary for the CEO and at 140% for all other Management Board members.
The long-term variable remuneration is granted in the form of performance share units under a performance share unit program. The performance share units will be settled after a four-year waiting period at the discretion of the Supervisory Board either in cash, treasury shares, or a combination of both. The payout amount depends on the achievement of a performance target linked to MorphoSys AG's share price performance, defined as the relative share price performance of MorphoSys AG's share price versus the performance of the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index, as well as on the achievement of a strategic development milestone and an ESG target during the four-year waiting period. The payout amount is capped at 250% of the original (individual) grant amount.
2. Target Total Remuneration
When determining the target total remuneration, the Supervisory Board ensures that the remuneration is commensurate with the tasks and performance of the respective Management Board member and the Company’s situation. The Supervisory Board also ensures that the remuneration is appropriate and in line with market practices. For this purpose, the Supervisory Board first compares the remuneration of the members of the Management Board of MorphoSys AG to the remuneration of management board members of comparable companies (horizontal comparison), generally taking a group of comparable European1 and American2 healthcare companies in addition to a group of German healthcare companies3.The Supervisory Board also conducts a comparison of the average
1 Abcam plc; ADC Therapeutics SA; Alkermes plc; Almirall S.A. ; Amarin Corporation plc; argenx SE; CureVac N.V. ; Dechra Pharmaceuticals plc; Galapagos N.V. ; Jazz Pharmaceuticals plc; and Orion Oyi; QIAGEN N.V. ; Swedish Orphan Biovitrum AB (publ);
2 Agios Pharmaceuticals, Inc; Alnylam Pharmaceuticals, Inc; Amicus Therapeutics, Inc; Biohaven Pharmaceutical Holding Company Ltd; bluebird bio, Inc; Denali Therapeutics Inc; FibroGen, Inc; Insmed, Inc; Ligand Pharmaceuticals, Inc; Sage Therapeutics, Inc; Sarepta Therapeutics, Inc; and Xencor, Inc.;
3 Carl Zeiss Meditec AG; CompuGroup Medical SE & Co KGaA; CureVac SE; Drägerwerk AG & Co. KGaA; Eckert- und Ziegler Strahlen- und Medizintechnik AG; Evotec SE; QIAGEN N.V.; Sartorius AG; Siemens Healthineers AG; Stratec SE; and SYNLAB AG.
103
remuneration of the first two management levels below the Company’s Management Board (vertical comparison).
The target total remuneration for all Management Board members is as follows:
The annual base salary corresponds to a share of approximately 17% to 30% of the target total remuneration. Short-term variable remuneration (annual bonus, STI) corresponds to approximately 15% to 20% of the target total remuneration, while long-term variable remuneration (LTI) corresponds to approximately 40% to 65% of the target total remuneration. Fringe benefits are granted in the average amount of approximately 3% to 9% of the target total remuneration, while the share of pension expenses is approximately 4% to 7% of the target total remuneration.
3. Caps and Maximum Remuneration
Both the annual bonus and the Performance Share Unit Program provide for maximum payout limits and were further designed in a manner that allows for a payout to fall to zero.
In accordance with Section 87a (1) sentence 2 no. 1 AktG, the Supervisory Board has further defined a maximum remuneration for the members of the Management Board. The maximum remuneration includes all fixed remuneration components, including fringe benefits and pension contributions, as well as the variable remuneration. In this regard, it is not decisive when the respective remuneration component is paid out, but for which financial year it has been promised. The maximum remuneration for the Chief Executive Officer for a financial year is € 9,000,000, and for each of the other members of the Management Board, it is € 3,700,000. The maximum remuneration does not represent the remuneration level targeted or deemed appropriate by the Supervisory Board but merely an absolute maximum limit that can be reached, if at all, if the targets were fully achieved and there was an increase in the MorphoSys AG share price. The maximum remuneration for a member of the Management Board may increase in exceptional cases in the year of appointment or in the second year after appointment, provided that the respective Management Board member has been granted remuneration payments, for example, as compensation for payments from a previous service agreement lost due to the acceptance of the new position at MorphoSys AG. In this case, the maximum remuneration may increase by up to 25% for the financial year in which the compensation payment is granted.
The Remuneration System 2022 is available on the Company's website at www.morphosys.com/de/investoren/corporate-governance.
104
| Fixed remuneration | Base salary | Annual fixed salary | |||||||||||||||
| Pension benefits | • Individual retirement contribution: Monthly contribution of 10% of the annual base salary • Monthly contribution to a pension plan | ||||||||||||||||
| Fringe benefits | Company Car, allowance for health, social, accident and occupational disability insurance policies, and reimbursement for tax advise and double housing, as well as other expense allowances | ||||||||||||||||
| Performance related remuneration | Annual Bonus (STI) | Structure | Annual Bonus payment depending on the achievement of financial and non-financial targets | ||||||||||||||
| Performance targets | Composition of one financial target (25%), one commercial target (25%), one development and BD&L-target (30% - 40%) and one research and BD&L-target (10% - 20%) | ||||||||||||||||
| Cap | Payout cap at 160% of the annual base salary (CEO) / 140% of the annual base salary (OBM) | ||||||||||||||||
| LTI | Structure | Share-based remuneration component consisting of performance share units with waiting period of four years, which will generally be settled by cash | |||||||||||||||
| Performance targets | • Relative share price performance • Development milestones target • ESG-target | ||||||||||||||||
| Cap | Payout cap at 250% of the initial grant amount | ||||||||||||||||
| Further provisions | Maximum remuneration | Maximum remuneration for one financial year of € 9.000.000 (CEO) / € 3.700.000 (OBM) | |||||||||||||||
| Malus and clawback | Right of the Supervisory Board to retain or reclaim variable remuneration in particular in case of breach of internal conduct policies of statutory duties by a member of the Management Board | ||||||||||||||||
| Severance cap | Severance payments shall not exceed twice the annual remuneration and shall not constitute remuneration for more than the remaining term of the service agreement | ||||||||||||||||
| Change of control | Termination right in case of (i) a change of control and (ii) a material reduction of the Management Board member's responsibilities | ||||||||||||||||
II.Remuneration of the Management Board in the 2023 Financial Year
1.Overview of the Main Remuneration Components
In the 2023 financial year, the remuneration of the Management Board members consisted of a fixed, non-performance-related remuneration, the annual base salary, market-standard fringe benefits and pension contributions, a short-term variable, performance-related remuneration, the annual bonus, and a long-term, share-based variable remuneration.
The level of remuneration of Management Board members depends to a large extent on the area of responsibility of the respective Management Board member, the member’s individual performance, the performance of the Management Board as a whole, as well as on the economic and financial success of MorphoSys AG. The intention is to provide an incentive for long-term and sustainable corporate governance, while at the same time linking the interests of the Management Board members with those of the Company's shareholders.
2. Non-Performance-Related Remuneration Components
Base Salary
The members of the Management Board receive a contractually agreed fixed base salary, which is paid out monthly. In May 2023, the Remuneration Committee reviewed the compensation package of the CEO against the market data for this role in Germany, Europe and the US, and recommended a new salary level effective 1 September 2023. The CEO's salary was increased by 5% to € 892,500. The annual base salary in the 2023 financial year for the individual members of the Management Board was as follows:
105
| Name of Management Board member | Role | Fixed base remuneration in €* | ||||||||||||
| Jean-Paul Kress, M.D. | Chief Executive Officer | 864,167 | ||||||||||||
| Lucinda Crabtree, Ph.D.** | Chief Financial Officer | 180,242 | ||||||||||||
| Sung Lee*** | Chief Financial Officer | 121,769 | ||||||||||||
| Charlotte Lohmann**** | Chief Legal Officer | 169,900 | ||||||||||||
| * The table shows the pro-rata base salary in case a Management Board member joins or leaves the Company during a financial year. | ||||||||||||||
| ** Lucinda Crabtree, Ph.D. was appointed as member of the Management Board of MorphoSys AG with effect as of August 8, 2023. | ||||||||||||||
*** Sung Lee resigned from his position as member of the Management Board with effect as of the end of March 17, 2023. The service agreement entered into between the Company and Sung Lee has been terminated with effect as of the end of March 31, 2023. | ||||||||||||||
| **** Charlotte Lohmann was appointed as member of the Management Board of the Company for the time period from March 1, 2023, to the end of August 31, 2023. | ||||||||||||||
Fringe Benefits
In addition to the fixed base salary, Management Board members receive market-standard fringe benefits, which mainly include the professional and private use of company cars, contributions to or reimbursement of costs for health, social and accident insurance, reimbursement of costs for legal advice related to the service agreement and dual residences.
In addition, in exceptional cases, a compensation payment (sign-on bonus) may be granted to Management Board members when they join the Management Board of MorphoSys AG (e.g., to compensate for lost benefits from a previous service relationship). In the 2023 financial year, no such compensation payment was granted to Management Board members.
Company Pension Scheme and other pension benefits
The Management Board members generally participate in a pension plan in form of a provident fund. The provident fund takes out a reinsurance policy that funds the pension benefits. In addition, the Management Board members also receive an amount equal to up to 10% of their fixed annual (gross) base salary, which is intended to be used by the Management Board members for their individual retirement plans. This amount may also be invested in a pension plan. Jean-Paul Kress, M.D., also has the option to use both payments, however, up to a maximum of 10% of his fixed annual (gross) base salary, for his individual retirement plans. The 10%-payment is not shown in the below table.
Management Board members who also have a company pension plan as part of their deferred remuneration (direct insurance) also receive an allowance for this Company pension plan. The pension scheme for individual Management Board members may be differently structured in exceptional cases, e.g., in case a Management Board member is resident abroad.
| Jean-Paul Kress, M.D. | Sung Lee* | ||||||||||||||||||||||||||||
| Chief Executive Officer | Chief Financial Officer | ||||||||||||||||||||||||||||
| 2023 | 2022 | 2023 | 2022 | ||||||||||||||||||||||||||
| in € thousands | in € thousands | in € thousands | in € thousands | ||||||||||||||||||||||||||
| Contribution to the provident fund | 130.34 | 127.73 | 18.14 | 83.46 | |||||||||||||||||||||||||
| Employer subsidy for deferred compensation | 0.48 | 0.48 | 0.12 | 0.48 | |||||||||||||||||||||||||
| Total | 130.82 | 128.21 | 18.26 | 83.94 | |||||||||||||||||||||||||
| Charlotte Lohman** | Lucinda Crabtree, Ph.D.*** | ||||||||||||||||||||||||||||
| Chief Legal Officer | Chief Financial Officer | ||||||||||||||||||||||||||||
| 2023 | 2022 | 2023 | 2022 | ||||||||||||||||||||||||||
| in € thousands | in € thousands | in € thousands | in € thousands | ||||||||||||||||||||||||||
| Contribution to the provident fund | 18.69 | — | 33.67 | — | |||||||||||||||||||||||||
| Employer subsidy for deferred compensation | 0.48 | — | — | ||||||||||||||||||||||||||
| Total | 19.17 | — | 33.67 | — | |||||||||||||||||||||||||
| * Sung Lee resigned from his position as member of the Management Board with effect as of the end of March 17, 2023 . The service agreement entered into between him and the Company has been terminated with effect as of end of March 31, 2023. | |||||||||||||||||||||||||||||
| ** Charlotte Lohmann was appointed as member of the management board of the Company for the time period from March 1, 2023, to August 31, 2023. | |||||||||||||||||||||||||||||
| ***Lucinda Crabtree, Ph.D. was appointed as member of the management board of MorphoSys AG with effect as of August 8, 2023. | |||||||||||||||||||||||||||||
106
3.Performance-Related Remuneration Components
Annual Bonus (Short-Term Incentive, STI)
The Management Board members receive a short-term variable remuneration in the form of an annual bonus (STI), which rewards the operational implementation of the Company's corporate strategy to support the long-term positive development of the Company and the MorphoSys Group in the respective financial year. The amount of the annual bonus is defined by the Supervisory Board for the upcoming financial year and measured on the basis of various financial and non-financial performance criteria (“Company Goals”) that are set uniformly for all Management Board members.
For this purpose, a target amount determining the payout amount in case of a 100% target achievement is set for each Management Board member. For the Chief Executive Officer, the target amount is 80% of the annual base salary, and for all other Management Board members, it is 70%.
At the beginning of the subsequent financial year, the Supervisory Board assesses the degree of achievement of the set targets and determines the amount of the annual bonus. The maximum payout amount is limited to 160% of the annual base salary for the Chief Executive Officer and to 140% for the other Management Board members.
The target achievement is measured as follows:
For each Company Goal, the Supervisory Board determines the percentage of target achievement, which may range between 0% and 125%. The percentage target achievement is converted into a target achievement level (the “Score”), which lies between 0% and 200%, with the target achievement and the corresponding Score increasing linearly between the defined percentage points.

On the basis of the so-calculated target achievement for each performance target and the relative weighting of each performance target as defined by the Supervisory Board, the Supervisory Board calculates the overall target achievement for the respective financial year as follows:
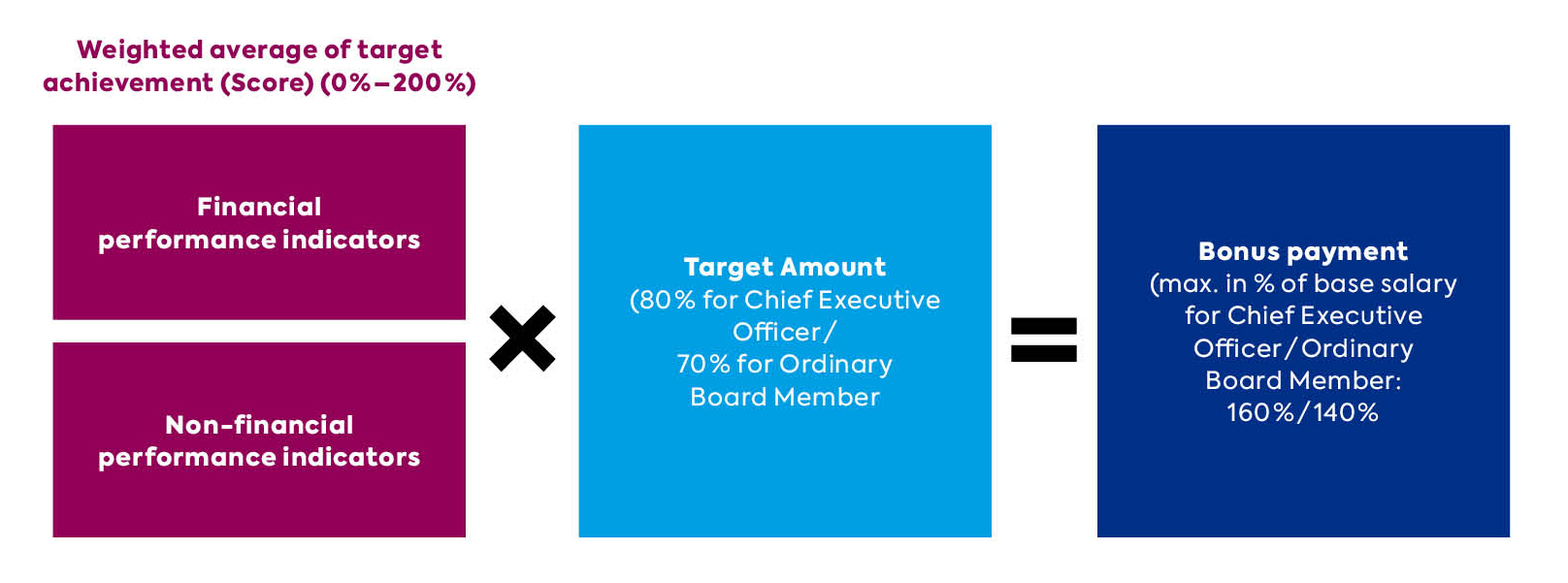
As the annual bonus 2022 was paid out to the members of the Management Board in the 2023 financial year, the annual bonus 2022 is attributed to the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in the 2023 financial year and thus disclosed in this remuneration report. Accordingly, the amount of the annual bonus (STI) for the 2023 financial year will not be determined and paid out until the 2024 financial year and will therefore be attributed to the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in the 2024 financial year. In order to
107
ensure the transparent and comprehensive disclosure of the remuneration granted to Management Board members for a financial year, MorphoSys has voluntarily disclosed the annual bonus for the 2023 financial year in this remuneration report.
Annual Bonus 2022
For the 2022 financial year, the Supervisory Board has defined the following performance criteria and their weighting uniformly for all Management Board members:
| Performance criteria | Evaluation criteria | Weighting | |||||||||||||||
| TARGET 1: COMMERCIAL TARGET | 25 | % | |||||||||||||||
| ● Achieve Monjuvi U.S. net product sales as communicated in Company's financial guidance | |||||||||||||||||
| TARGET 2: FINANCIAL TARGET | 25 | % | |||||||||||||||
| ● Manage operating expenses within budget as communicated in Company's financial guidance | |||||||||||||||||
| TARGET 3: DEVELOPMENT & BD&L- TARGET | 30 | % | |||||||||||||||
| ● Advance clinical development of phase 3 programs: | |||||||||||||||||
| •Pelabresib - Accelerate recruitment of MANIFEST-2: Achieve enrollment of 50% | |||||||||||||||||
| •Pelabresib - Complete recruitment of MF patients in MANIFEST-1: 100% | |||||||||||||||||
| •Tafasitamab - Achieve frontMIND enrollment of 75% | |||||||||||||||||
| •Tafasitamab - Achieve First Patient First Visit for MINDway study | |||||||||||||||||
| TARGET 4: RESEARCH & BD&L-TARGET | 20 | % | |||||||||||||||
| ● Execute at least one partnership for early or mid-stage MorphoSys program while advancing the pipeline | |||||||||||||||||
From the perspective of the Supervisory Board, the 2022 financial year was a successful and formative year for MorphoSys AG. The clinical development of Phase 3 programs continued successfully in 2022 and patient recruitment for the key clinical studies, MANIFEST-2 and FrontMIND, was accelerated. Monjuvi sales grew year-over-year despite the increasing competition, but the results did not meet the 2022 target. The out-licensing of products in earlier stages of development and collaborations the Company entered into with HI-Bio, Pfizer and Novartis helped to streamline the product pipeline, reduce costs and provide cash. The significant cost reduction efforts resulted in the Company achieving operating expenses below the financial guidance published at the beginning of the year.
In the 2022 financial year, the target achievement for the annual bonus was as follows:
| Performance criteria | Evaluation | Weighting | Target achievement | ||||||||||||||||||||
| TARGET 1: COMMERCIAL TARGET | ● Achieve Monjuvi U.S. net product sales as communicated in Company's financial guidance | 25 | % | 77.7 | % | ||||||||||||||||||
| TARGET 2: FINANCIAL TARGET | ● Manage operating expenses within budget as communicated in Company's financial guidance | 25 | % | 125 | % | ||||||||||||||||||
| TARGET 3: DEVELOPMENT & BD&L- TARGET | ● Advance clinical development of phase 3 programs: | 30 | % | 120 | % | ||||||||||||||||||
| TARGET 4: RESEARCH & BD&L-TARGET | ● Execute at least one partnership for early or mid-stage MorphoSys program while advancing the pipeline | 20 | % | 125 | % | ||||||||||||||||||
108
On the basis of the conversion table shown above, the target achievement of the individual performance criteria is converted into a respective target achievement level (Score). Taking into account the defined weighting of the individual performance criteria, the resulting overall target achievement amounts to 159.71%,
In the context of the payment amounts for the annual bonus 2022, the payment for the annual bonus 2021 has been corrected as follows:
On March 10, 2022, a one-time non-cash impairment charge was announced based on the Company's decision to consolidate all research activities on the most advanced programs following the acquisition of Constellation Pharmaceuticals and to centralize all laboratory activities at the German research site in Planegg. This negatively impacted the target achievement and the payout amounts of the annual bonus 2021. However, as the annual bonus 2021 had already been paid out at this time, the impact of this effect should be taken into account by means of a corresponding deduction when the annual bonus 2022 is paid out, as already explained in the remuneration report for the 2021 financial year.
The following table discloses the calculation of the payout amount for the annual bonus 2022 as well as the correction to be made to the payout of the annual bonus 2021 and the actual amounts paid out to the members of the Management Board:
| in € thousands | Target amount based on 100% target achievement (Score) | Maximum payout (160% of annual base salary) | Maximum payout (140% of annual base salary) | Total target achievement (Score) | STI payout | Correction STI payout 2021 | Final STI payout | |||||||||||||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | 637.3 | 1,274.7 | — | 159.7 | % | 1,017.9 | (77.0) | 940.9 | ||||||||||||||||||||||||||||||||||||
| Sung Lee | 333.6 | — | 667.2 | 159.7 | % | 532.8 | (37.3) | 495.5 | ||||||||||||||||||||||||||||||||||||
| Malte Peters, M.D.* | 360.5 | — | 721.1 | 159.7 | % | 575.8 | (44.2) | 531.6 | ||||||||||||||||||||||||||||||||||||
| *Malte Peters M.D. resigned from his position as member of the Management Board with effect as of the end of September 30, 2022, and was released from his activities for the Company for the remaining term of his service agreement until the end of December 31, 2022 under continued payment of his remuneration. | ||||||||||||||||||||||||||||||||||||||||||||
The annual bonus 2022 has been paid out to the Management Board members in March 2023 and is therefore attributed to the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in the 2023 financial year. Charlotte Lohmann and Lucinda Crabtree have not yet been members of the Management Board during the 2022 financial year and thus have not received an annual bonus for the 2022 financial year in this function.
Annual Bonus 2023
For the 2023 financial year, the Supervisory Board has defined the following performance criteria for the annual bonus and their weighting uniformly for all Management Board members:
| Performance criteria | Evaluation criteria | Weighting | ||||||||||||
| TARGET 1: COMMERCIAL TARGET | ● Achieve Monjuvi U.S. net product sales as communicated in the company’s financial guidance | 25 | % | |||||||||||
| TARGET 2: FINANCIAL TARGET | ● Manage operating expenses within budget as communicated in the company’s financial guidance | 25 | % | |||||||||||
| TARGET 3: DEVELOPMENT & BD&L-TARGET | ● Advance Pelabresib clinical program: | 40 | % | |||||||||||
| • Complete recruitment of MANIFEST-2 by Q2 2023 | ||||||||||||||
| • Explore additional indications | ||||||||||||||
| TARGET 4: RESEARCH & BD&L-TARGET | ● Advance further research and development programs: | 10 | % | |||||||||||
| • Tafasitamab – complete recruitment of frontMIND by Q2 2023 | ||||||||||||||
| • Evaluate strategic options for Tulmimetostat and discovery programs and execute accordingly | ||||||||||||||
109
From the Supervisory Board's perspective, the fiscal year 2023 was a very successful year for MorphoSys AG. Large progress was made in the development of the clinical pipeline. Patient recruitment was completed in all ongoing Phase 3 trials, in the case of MANIFEST-2 also earlier than initially planned. In addition, the first topline data from the MANIFEST-2 study for the treatment of myelofibrosis were announced at the end of November 2023. The pelabresib and ruxolitinib combination achieved its primary endpoint and the key secondary endpoints showed a strong positive trend in reducing symptom burden in the overall patient population by improved measures of anemia, improved bone marrow fibrosis and biomarker improvements that suggest disease modification. The study randomized 430 patients, making it one of the largest myelofibrosis studies conducted to date, and it delivered important data for patients with myelofibrosis – underscoring that pelabresib has the potential to shift the treatment paradigm for this debilitating disease. Detailed results from the MANIFEST-2 study were presented in an oral presentation at the ASH Annual Meeting in December 2023. New preliminary results from an arm of the MANIFEST study investigating pelabresib as monotherapy in patients with high-risk essential thrombocythemia (ET) open the possibility to further investigate pelabresib in this indication. Throughout the year, the latest clinical data for pelabresib, tafasitamab and tulmimetostat were presented at various scientific conferences. Tulmimetostat received fast track status from the FDA for the treatment of ARID1A-mutated endometrial cancer. During the year, several agreements were signed with academic institutions for further preclinical and clinical investigation of tulmimetostat. As planned, the company was able to slightly increase sales figures from the distribution of Monjuvi® in the USA despite a stronger competitive environment. Cost-cutting efforts at the beginning of the year helped to keep the company's operating costs in line with the financial guidance published at the beginning of the year.
In the 2023 financial year, the target achievement for the annual bonus was as follows:
| Performance criteria | Evaluation | Weighting | Target achievement | |||||||||||||||||
| TARGET 1: COMMERCIAL TARGET | ● Achieve Monjuvi U.S. net product sales as communicated in the company’s financial guidance | 25 | % | 114.83 | % | |||||||||||||||
| TARGET 2: FINANCIAL TARGET | ● Manage operating expenses within budget as communicated in the company’s financial guidance | 25 | % | 125 | % | |||||||||||||||
| TARGET 3: DEVELOPMENT & BD&L-TARGET | ● Advance Pelabresib clinical program: | 40 | % | 125 | % | |||||||||||||||
| • Complete recruitment of MANIFEST-2 by Q2 2023 | ||||||||||||||||||||
| • Explore additional indications | ||||||||||||||||||||
| TARGET 4: RESEARCH & BD&L-TARGET | ● Advance further research and development programs: | 10 | % | 125 | % | |||||||||||||||
| • Tafasitamab – complete recruitment of frontMIND by Q2 2023 | ||||||||||||||||||||
| • Evaluate strategic options for Tulmimetostat and discovery programs and execute accordingly | ||||||||||||||||||||
On the basis of the conversion table shown above, the target achievement of the individual performance criteria is converted into a respective target achievement level (Score). Taking into account the defined weighting of the individual performance criteria, the resulting overall target achievement amounts to 189.83%. The following table sets out the target achievement as well as the corresponding payout amounts:
| in € thousands | Target amount based on 100% target achievement (Score) | Maximum payout (160% of annual base salary) | Maximum payout (140% of annual base salary) | Total target achievement (Score)* | STI payout* | |||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | 691.3 | 1,382.7 | — | 189.83 | % | 1,312.4 | ||||||||||||||||||||||||||
| Lucinda Crabtree, Ph.D.** | 126.2 | — | 252.3 | 189.83 | % | 239.5 | ||||||||||||||||||||||||||
| Sung Lee*** | 85.2 | — | 170.5 | 189.83 | % | 121.3 | ||||||||||||||||||||||||||
| Charlotte Lohmann**** | 118.9 | — | 237.9 | 189.83 | % | 225.8 | ||||||||||||||||||||||||||
*This is an overall degree of target achievement (score) or a deferred amount based on a pre-final target evaluation. **With resolution of the supervisory board dated August 8, 2023, Lucinda Crabtree, Ph.D. was appointed as member of the management board of MorphoSys AG with effect as of the same day until August 6,2026 ***Sung Lee resigned from his position as member of the Management Board with effect as of the end of March 17, 2023. The service agreement between the Company and Sung Lee has been terminated with effect as of the end of March 31, 2023. The pro-rata STI payout for 2023 has been agreed upon in the corresponding termination agreement and paid out with the STI Bonus 2022. ****With resolution of the supervisory board dated February 28, 2023, Charlotte Lohmann was appointed as member of the management board of the Company for the time period from March 1, 2023, to August 31, 2023. | ||||||||||||||||||||||||||||||||
110
The annual bonus 2023 will be paid out to the Management Board members in March 2024 and is therefore attributed to the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in the 2024 financial year. For reasons of transparency and better comprehensibility, the annual bonus 2023 is however voluntarily disclosed in this remuneration report.
Long-Term Incentive (LTI)
The members of the Management Board also receive a long-term variable remuneration in the form of the participation in the Company’s various long-term remuneration programs. The participation in the long-term share-based remuneration programs provides an incentive to the respective Management Board members to contribute to the long-term sustainable development of the Company, while linking the interests of the Management Board members to those of the shareholders.
In the 2023 financial year, stock options granted in the 2019 financial year under the Stock Option Program 2019 (April) and the Stock Option Program 2019 (October) as well as performance shares granted under the Performance Share Plan 2019 became exercisable. The relevant performance criteria under both the Stock Option Program 2019 and the Performance Share Plan 2019 were the absolute and relative share price performance of MorphoSys AG. In addition, performance share units were granted to the Management Board members in the 2023 financial year under the Performance Share Unit Program 2023.
The inflow from the Stock Option Program 2019 and the Performance Share Plan 2019 in the 2023 financial year is allocated to the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in the 2023 financial year.
The performance share units are subject to a waiting period of four years and the achievement of the defined performance targets. The payout of the final number of performance share units granted to the members of the Management Board in the 2023 financial year thus depends on the achievement of the performance targets and will occur after the end of the waiting period. It will therefore only be disclosed in the remuneration report for the 2027 financial year.
Settlement of the Stock Option Programs 2019
In the 2023 financial year, stock options granted under the Stock Option Programs 2019 for the 2019 financial year to the members of the Management Board, at that time Simon Moroney, Ph.D., Jens Holstein, Markus Enzelberger, Ph.D., and Malte Peters, M.D., (each with effect as of April 1, 2019) (the "Stock Option Program 2019 (April)"), and Jean-Paul Kress, M.D., (with effect as of October 1, 2019) (the "Stock Option Program 2019 (October)") became exercisable for a duration of three years following both the approval of the Company’s consolidated annual financial statements for the financial year immediately prior to the expiry of the waiting period as well as the expiry of the waiting period
The performance targets for the Stock Option Plan 2019 were defined as the absolute share price performance of the share of MorphoSys AG and the relative share price performance of the share of MorphoSys AG compared to a benchmark index, consisting in equal parts of the NASDAQ Biotechnology Index and the TecDAX Index. Each performance target was weighted with 50% within the overall target achievement.
To determine the degree of target achievement for each performance target, the waiting period was divided into four equal periods of one year each. The arithmetic mean of the target achievement of each of the annual periods determines the percentage target achievement for each performance target. The percentage target achievements for each of the two performance targets were then added up and divided by two. The result forms the overall target achievement, which determines the final number of exercisable stock options, whereby, depending on the degree of target achievement, the final number of stock options may be up to 200% of the initial number of stock options, whereby one stock options grants one subscription right for one share in the Company.
111
Absolute Share Price Performance
The absolute share price performance of the share of MorphoSys AG during an annual period is measured by comparing the average closing price of MorphoSys AG's shares in Xetra trading on the Frankfurt Stock Exchange during the 30 trading days prior to the beginning and prior to the end of the respective annual period. If the share price decreases during the respective annual period, the degree of the absolute share price performance target achievement is 0%. If the share price performance of the share of MorphoSys AG is 0%, the performance target achievement is 50%. Thereafter, the performance target achievement increases linearly. If the MorphoSys AG share price increases by 8% during an annual period, the performance target achievement is 100%. If the share price increases by 16% during an annual period, the performance target achievement is 150%. If the share price increases by 24%, the performance target achievement is 200%. Any further increase in the performance target achievement is not possible (cap).
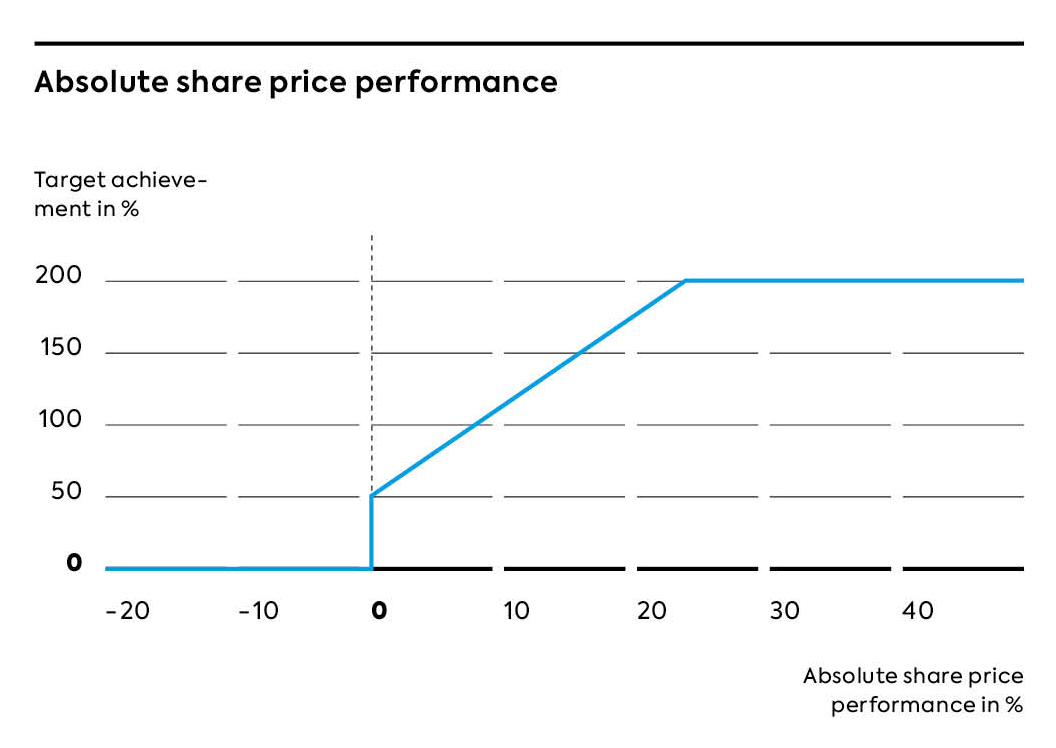
112
Relative Share Price Performance
To determine the performance target of relative share price performance, the performance of the MorphoSys share price is compared with and set into relation to the development of the NASDAQ Biotech Index and the TecDAX Index (collectively the "Benchmark Index") during each annual period. The Benchmark Index consists of the NASDAQ Biotech Index and the TecDAX Index, each weighted at 50%, i.e., the percentage share price performance of each index is added up and divided by two. The relevant MorphoSys share price is the average closing price of MorphoSys shares in Xetra trading during the last 30 trading days on the Frankfurt Stock Exchange prior to the beginning and the end of the respective annual period. The relevant price of the NASDAQ Biotech Index and the TecDAX Index is the average closing price of the NASDAQ Biotech Index and the TecDAX Index on the NASDAQ Stock Exchange and Frankfurt Stock Exchange, respectively, during the last 30 trading days prior to the beginning and the end of the respective annual period. If the MorphoSys share price decreases compared to the Benchmark Index during an annual period, the degree of performance target achievement of the relative share price performance is 0%. If the MorphoSys share price performance is 0% compared to the Benchmark Index, the performance target achievement is 100%. Thereafter, the performance target achievement increases linearly. If the MorphoSys share price increases by 8% compared to the Benchmark Index, the performance target achievement is 150%, if it increases by 16% compared to the Benchmark Index, the performance target is 200%. Any further increase in the performance target achievement is not possible (cap).
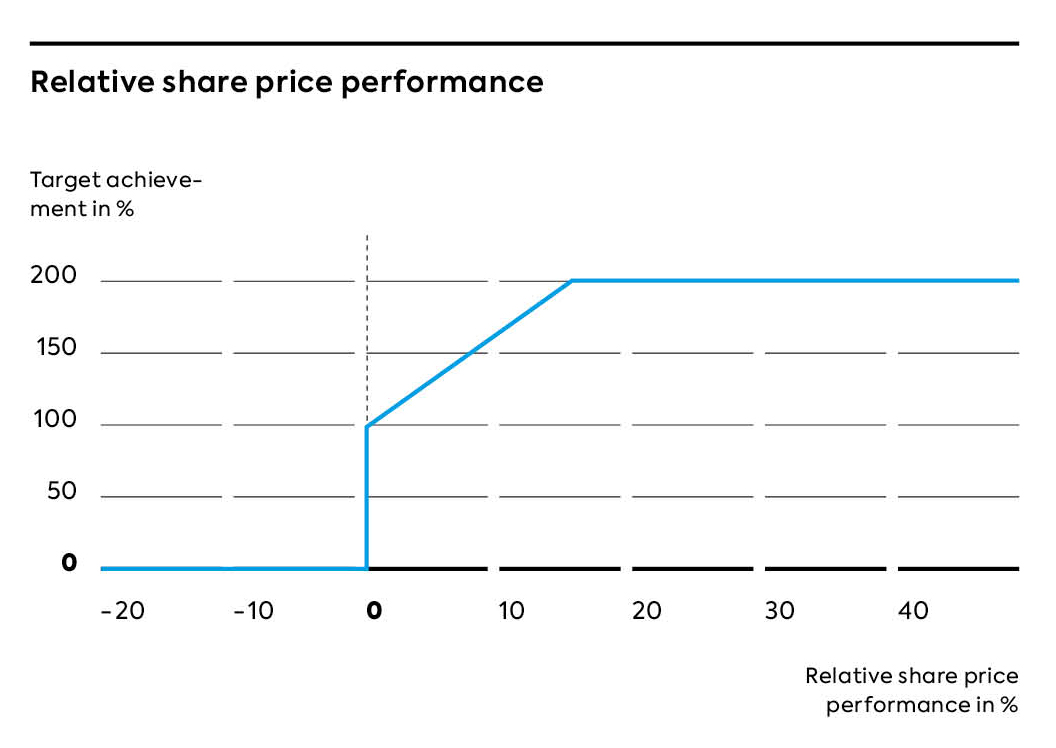
113
The performance target achievement during the waiting period of the Stock Option Program 2019 (April) was as follows:
| Average price in € at the beginning of the annual period* | Average price in € at the end of the annual period* | Share price development | Target achievement after the end of the waiting period | |||||||||||||||||||||||
| Absolute share price performance** | ||||||||||||||||||||||||||
| MorphoSys AG | +22.75 | % | ||||||||||||||||||||||||
| First annual period | 87.86 | 93.66 | +6.61 | % | ||||||||||||||||||||||
| Second annual period | 93.66 | 81.02 | (13.49 | %) | ||||||||||||||||||||||
| Third annual period | 81.02 | 23.84 | (70.57 | %) | ||||||||||||||||||||||
| Fourth annual period | 23.84 | 15.55 | (34.78 | %) | ||||||||||||||||||||||
| Relative share price performance*** | ||||||||||||||||||||||||||
| MorphoSys AG | +35.25 | % | ||||||||||||||||||||||||
| First annual period | 87.86 | 93.66 | +6.61 | % | ||||||||||||||||||||||
| Second annual period | 93.66 | 81.02 | (13.49 | %) | ||||||||||||||||||||||
| Third annual period | 81.02 | 23.84 | (70.57 | %) | ||||||||||||||||||||||
| Fourth annual period | 23.84 | 15.55 | (34.78 | %) | ||||||||||||||||||||||
| TecDAX Index | ||||||||||||||||||||||||||
| First annual period | 2,644.62 | 2,689.41 | +1.69 | % | ||||||||||||||||||||||
| Second annual period | 2,689.41 | 3,368.42 | +25.25 | % | ||||||||||||||||||||||
| Third annual period | 3,368.42 | 3,183.70 | (5.48 | %) | ||||||||||||||||||||||
| Fourth annual period | 3,183.70 | 3,242.39 | +1.84 | % | ||||||||||||||||||||||
| NASDAQ Biotechnology Index | ||||||||||||||||||||||||||
| First annual period | 3,509.60 | 3,458.14 | (1.47 | %) | ||||||||||||||||||||||
| Second annual period | 3,458.14 | 4,853.42 | +40.35 | % | ||||||||||||||||||||||
| Third annual period | 4,853.42 | 4,016.63 | (17.24 | %) | ||||||||||||||||||||||
| Fourth annual period | 4,016.63 | 4,055.68 | +0.97 | % | ||||||||||||||||||||||
| Overall target achievement | +29.00 | % | ||||||||||||||||||||||||
* Average price of the MorphoSys AG share in Xetra trading on the Frankfurt Stock Exchange during the 30 trading days prior to the beginning or end of the respective annual period (data source: Bloomberg). | ||||||||||||||||||||||||||
** The target achievement for the absolute share price performance on the basis of the above values amounted during the respective annual periods as follows: +91,00 % during the first annual period, +0,00 % during the second annual period, +0,00 % during the third annual period and +0,00 % during the fourth annual period. | ||||||||||||||||||||||||||
*** The target achievement for the relative share price performance on the basis of the above values amounted during the respective annual periods as follows: +141,00 % during the first annual period, +0,00 % during the second annual period, +0,00 % during the third annual period and +0,00 % during the fourth annual period. | ||||||||||||||||||||||||||
114
The performance target achievement during the waiting period of the Stock Option Program 2019 (October) was as follows:
| Average price in € at the beginning of the annual period* | Average price in € at the end of the annual period* | Share price development | Target achievement after the end of the waiting period | |||||||||||||||||||||||
| Absolute share price performance** | ||||||||||||||||||||||||||
| MorphoSys AG | 64.25 | % | ||||||||||||||||||||||||
| First annual period | 106.16 | 107.40 | 1.17 | % | ||||||||||||||||||||||
| Second annual period | 107.40 | 44.90 | (58.19 | %) | ||||||||||||||||||||||
| Third annual period | 44.90 | 18.78 | (58.17 | %) | ||||||||||||||||||||||
| Fourth annual period | 18.78 | 28.70 | 52.81 | % | ||||||||||||||||||||||
| Relative share price performance*** | ||||||||||||||||||||||||||
| MorphoSys AG | 50.00 | % | ||||||||||||||||||||||||
| First annual period | 106.16 | 107.40 | 1.17 | % | ||||||||||||||||||||||
| Second annual period | 107.40 | 44.90 | (58.19 | %) | ||||||||||||||||||||||
| Third annual period | 44.90 | 18.78 | (58.17 | %) | ||||||||||||||||||||||
| Fourth annual period | 18.78 | 28.70 | 52.81 | % | ||||||||||||||||||||||
| TecDAX Index | ||||||||||||||||||||||||||
| First annual period | 2,754.02 | 3,087.34 | 12.10 | % | ||||||||||||||||||||||
| Second annual period | 3,087.34 | 3,900.16 | 26.33 | % | ||||||||||||||||||||||
| Third annual period | 3,900.16 | 2,873.70 | (26.32 | %) | ||||||||||||||||||||||
| Fourth annual period | 2,873.70 | 3,085.08 | 7.36 | % | ||||||||||||||||||||||
| NASDAQ Biotechnology Index | ||||||||||||||||||||||||||
| First annual period | 3,337.17 | 4,171.00 | 24.99 | % | ||||||||||||||||||||||
| Second annual period | 4,171.00 | 5,307.74 | 27.25 | % | ||||||||||||||||||||||
| Third annual period | 5,307.74 | 3,893.65 | (26.64 | %) | ||||||||||||||||||||||
| Fourth annual period | 3,893.65 | 4,054.45 | 4.13 | % | ||||||||||||||||||||||
| Overall target achievement | 57.13 | % | ||||||||||||||||||||||||
| * Average price of the MorphoSys AG share in Xetra trading on the Frankfurt Stock Exchange during the 30 trading days prior to the beginning or end of the respective annual period (data source: Bloomberg). | ||||||||||||||||||||||||||
| ** The target achievement for the absolute share price performance on the basis of the above values amounted during the respective annual periods as follows: +57,00 % during the first annual period, +0,00 % during the second annual period, +0,00 % during the third annual period and +200,00 % during the fourth annual period. | ||||||||||||||||||||||||||
| *** The target achievement for the relative share price performance on the basis of the above values amounted during the respective annual periods as follows: +0,00 % during the first annual period, +0,00 % during the second annual period, +0,00 % during the third annual period and +200,00 % during the fourth annual period. | ||||||||||||||||||||||||||
The following is the final number of exercisable stock options based on the overall degree of target achievement of 29% achieved under the Stock Option Program 2019 (April) and of 57% achieved under the Stock Option Program 2019 (October), whereby the final number is calculated by multiplying the overall degree of target achievement with the initial number of stock options:
| Management Board member | Exercise price* (in €) | Initial number of stock options | Final number of stock options | Quantitative Change | ||||||||||||||||||||||
| Simon Moroney, Ph.D. | 87.86 | 10,587 | 3,071 | (7,516) | ||||||||||||||||||||||
| Malte Peters, M.D. | 87.86 | 6,936 | 2,012 | (4,924) | ||||||||||||||||||||||
| Jens Holstein | 87.86 | 6,936 | 2,012 | (4,924) | ||||||||||||||||||||||
| Markus Enzelberger, Ph.D. | 87.86 | 6,936 | 2,012 | (4,924) | ||||||||||||||||||||||
| Jean-Paul Kress, M.D. | 106.16 | 57,078 | 32,535 | (24,543) | ||||||||||||||||||||||
| *The exercise price differs based on the actual grant date, i.e. €87.86 on April 1, 2019 and €106.16 on October 1, 2019, respectively. | ||||||||||||||||||||||||||
In the 2023 financial year, neither Jean-Paul Kress, M.D., nor any former member of the Management Board who was granted stock options under the Stock Option Plan 2019 exercised these stock options. Charlotte Lohmann has received stock options
115
under the Stock Option Plan 2019 only in her function as employee of the Company. Lucinda Crabtree, Ph.D., has not been a member of the Company's Management Board at the time the stock options under the Stock Option Program 2019 were issued.
Settlement of the Performance Share Plan 2019
In the 2023 financial year, performance shares granted under the Performance Share Plan 2019 for the 2019 financial year to the members of the Management Board, at that time Simon Moroney, Ph.D., Jens Holstein, Markus Enzelberger, Ph.D., and Malte Peters, M.D., became exercisable for a period of six months following the expiry of the waiting period.
The performance targets for the Performance Share Plan 2019 are the absolute MorphoSys share price performance and the relative MorphoSys share price performance compared to a benchmark index, consisting in equal parts of the NASDAQ Biotechnology Index and the TecDAX Index. Each performance target has a weighting of 50% for the overall degree of target achievement.
To determine the degree of the performance target achievement for each performance target, the waiting period is divided into four equal periods of one year each. The arithmetic mean of the target achievement of each of the annual periods determines the percentage performance target achievement for each performance target. The percentage performance target achievements for each of the two performance targets is then added together and divided by two. The result forms the overall degree of performance target achievement, which determines the number of performance shares that can be exercised. Depending on the performance target achievement, the final number of performance shares may be up to 200% of the initial number of performance shares, whereby one performance share entitles to one share in the Company.
The number of exercisable performance shares is then multiplied by a company factor between 0 and 2 as determined by the Supervisory Board. For the Performance Share Plan 2019, the Supervisory Board set the company factor at 1.
At the discretion of the Company, performance shares are settled either in cash or in treasury shares of the Company.
Absolute Share Price Performance
The absolute MorphoSys AG's share price performance during an annual period is measured by comparing the average closing prices of MorphoSys AG's shares in Xetra trading on the Frankfurt Stock Exchange during the 30 trading days before the beginning and before the end of the respective annual period. If the share price decreases during an annual period, the degree of performance target achievement of the absolute share price performance target is 0%. If the MorphoSys share price increases by 0% to less than 1% during an annual period, the performance target achievement of the absolute share price performance target for that annual period is 10%. For each full percentage hurdle that the share price development during an annual period meets or exceeds, the performance target achievement increases by another 10%. If the share price increases 29% in the respective annual period, the performance target achievement is 300%. Any further increase of the performance target achievement is not possible (cap).
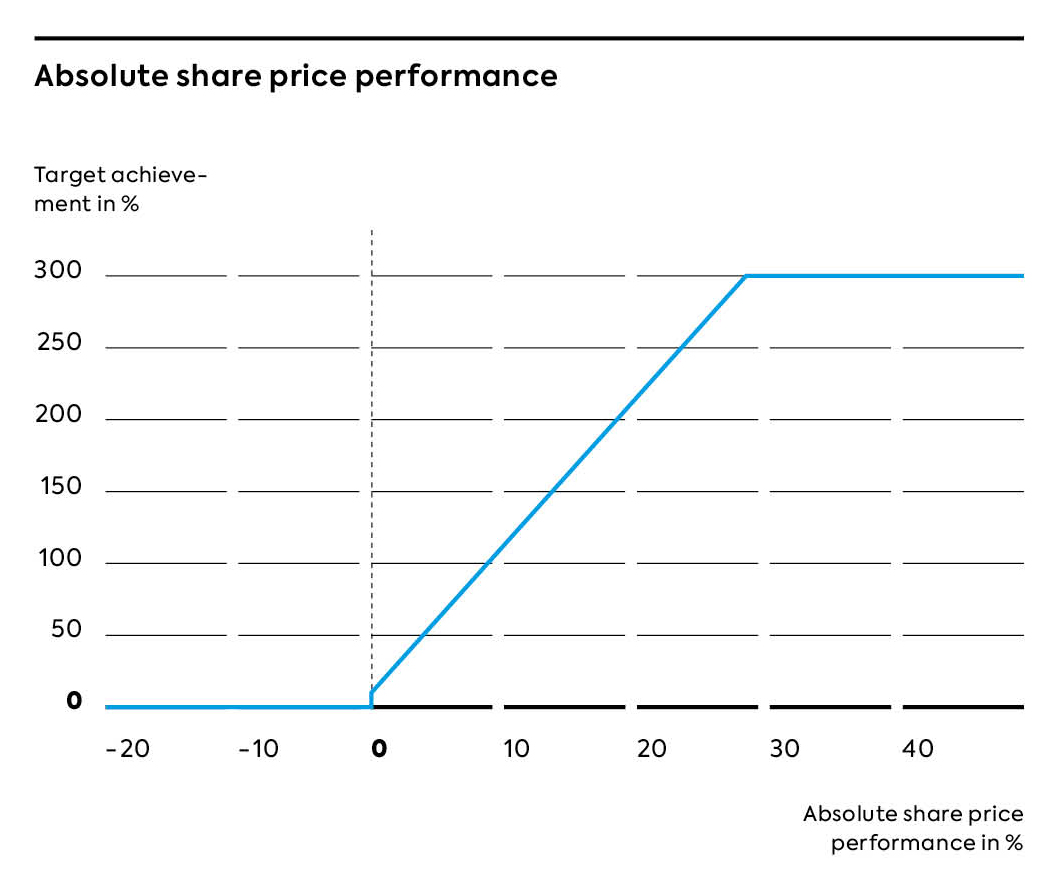
116
Relative Share Price Performance
For the relative performance target, for each annual period, the MorphoSys share price at the beginning of the annual period is compared to the performance of the NASDAQ Biotech Index and the TecDAX Index (collectively the “Benchmark Index”) and the respective values are put into relation. The Benchmark Index consists of the NASDAQ Biotech Index and the TecDAX Index, each weighted at 50%, i.e., the percentage share price performance of each index is added up and divided by two. The relevant MorphoSys share price is the average closing price of the MorphoSys shares in Xetra trading during the last 30 trading days on the Frankfurt Stock Exchange prior to the beginning and the end of the respective annual period. The relevant closing price of the NASDAQ Biotech Index and the TecDAX Index is the average closing price of the NASDAQ Biotech Index and the TecDAX Index on the NASDAQ Stock Exchange and Frankfurt Stock Exchange, respectively, during the last 30 trading days prior to the beginning and the end of the respective annual period. If during an annual period the share price declines compared to the Benchmark Index, the achievement of the performance target is 0%. If the MorphoSys share price increases by 0% to less than 0.5 % compared to the Benchmark Index, the performance target achievement of the relative share price performance target for the respective annual period is 10%. For each half-percentage hurdle that the share price development during an annual period meets or exceeds, the performance target achievement increases by another 10%. If the MorphoSys share price increases 14.5% during an annual period compared to the Benchmark Index, the performance target achievement is 300%. Any further increase in the MorphoSys share price compared to the Benchmark Index does not result in a further increase of the performance target achievement (cap).
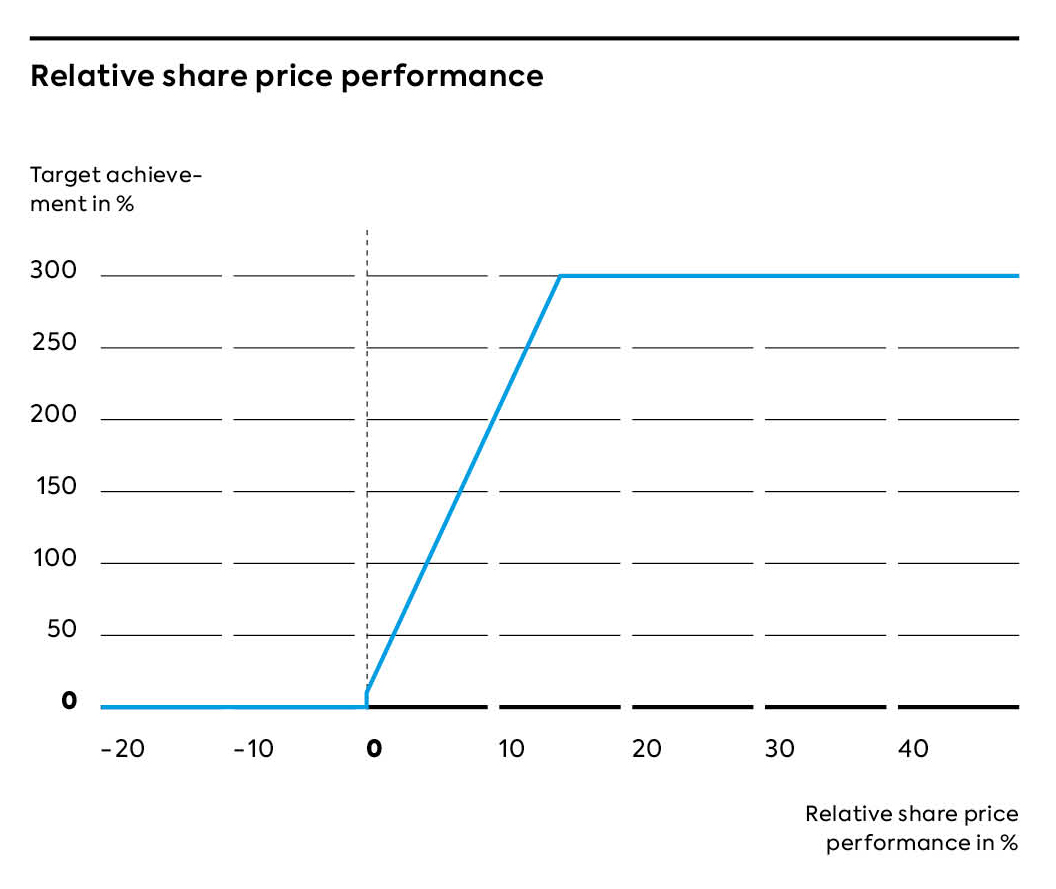
117
The following performance targets were achieved during the waiting period:
| Average price in € at the beginning of the annual period* | Average price in € at the end of the annual period* | Share price development | Target achievement after the end of the waiting period | |||||||||||||||||||||||
| Absolute share price performance** | ||||||||||||||||||||||||||
| MorphoSys AG | ||||||||||||||||||||||||||
| First annual period | 87.86 | 93.66 | +6.60 | % | +17.50 | % | ||||||||||||||||||||
| Second annual period | 93.66 | 81.02 | (13.50 | %) | ||||||||||||||||||||||
| Third annual period | 81.02 | 23.84 | (70.58 | %) | ||||||||||||||||||||||
| Fourth annual period | 23.84 | 15.55 | (34.77 | %) | ||||||||||||||||||||||
| Relative share price performance*** | ||||||||||||||||||||||||||
| MorphoSys AG | +32.50 | % | ||||||||||||||||||||||||
| First annual period | 87.86 | 93.66 | +6.60 | % | ||||||||||||||||||||||
| Second annual period | 93.66 | 81.02 | (13.50 | %) | ||||||||||||||||||||||
| Third annual period | 81.02 | 23.84 | (70.58 | %) | ||||||||||||||||||||||
| Fourth annual period | 23.84 | 15.55 | (34.77 | %) | ||||||||||||||||||||||
| TecDAX Index | ||||||||||||||||||||||||||
| First annual period | 2,644.62 | 2,689.41 | +1.69 | % | ||||||||||||||||||||||
| Second annual period | 2,689.41 | 3,368.42 | +25.25 | % | ||||||||||||||||||||||
| Third annual period | 3,368.42 | 3,183.70 | (5.48 | %) | ||||||||||||||||||||||
| Fourth annual period | 3,183.70 | 3,242.38 | +1.84 | % | ||||||||||||||||||||||
| NASDAQ Biotechnology Index | ||||||||||||||||||||||||||
| First annual period | 3,509.60 | 3,458.14 | (1.47 | %) | ||||||||||||||||||||||
| Second annual period | 3,458.14 | 4,853.42 | +40.35 | % | ||||||||||||||||||||||
| Third annual period | 4,853.42 | 4,016.63 | (17.24 | %) | ||||||||||||||||||||||
| Fourth annual period | 4,016.63 | 4,055.68 | +0.97 | % | ||||||||||||||||||||||
| Overall target achievement | +25.00 | % | ||||||||||||||||||||||||
* Average price of the MorphoSys AG share in Xetra trading on the Frankfurt Stock Exchange during the 30 trading days prior to the beginning or end of the respective annual period (data source: Bloomberg). | ||||||||||||||||||||||||||
** The target achievement for the absolute share price performance on the basis of the above values amounted during the respective annual periods as follows: +70,00% during the first annual period, +70,00% during the second annual period, +0,00% during the third annual period and +0,00% during the fourth annual period. | ||||||||||||||||||||||||||
*** The target achievement for the relative share price performance on the basis of the above values amounted during the respective annual periods as follows: +130,00% during the first annual period, +0,00% during the second annual period, +0,00% during the third annual period and +0,00% during the fourth annual period. | ||||||||||||||||||||||||||
In accordance with the terms & conditions of the Performance Share Plan 2019, the overall degree of target achievement for Management Board members who left the Company prematurely is calculated on the basis of the target achievement of annual periods completed prior to the departure of such Management Board members. Since all Management Board members who have received performance shares under the Performance Share Plan 2019 have left the Company prior to the expiry of the waiting period, the final number of performance shares is in each case calculated based on the individual target achievement as follows (whereby the final number of performance shares is calculated by multiplying the initial number of performance shares with the overall degree of target achievement):
118
| Management Board member | Initial number of performance shares | Target achievement* | Final number of performance shares | Quantitative change | ||||||||||||||||||||||
| Malte Peters, M.D. | 2,065 | 33.33 | % | 688 | 1,377 | |||||||||||||||||||||
| Simon Moroney, Ph.D. | 3,152 | 100.00 | % | 3,152 | 0 | |||||||||||||||||||||
| Jens Holstein | 2,065 | 100.00 | % | 2,065 | 0 | |||||||||||||||||||||
| Markus Enzelberger, Ph.D. | 2,065 | 100.00 | % | 2,065 | 0 | |||||||||||||||||||||
| *Target achievement is dependent on date of exit. | ||||||||||||||||||||||||||
All former members of the Management Board who were granted performance shares under the Performance Share Plan 2019, exercised the performance shares granted to them within the mandatory six-month exercise period following the expiry of the waiting period from May 4, 2023, to November 3, 2023. The performance shares were settled in treasury shares. Jean-Paul Kress, M.D., Lucinda Crabtree, Ph.D., and Charlotte Lohmann were not yet a member of the Company's Management Board at the time the performance shares under the Performance Share Plan 2019 were issued.
Performance Share Unit Program 2023
With effect as of April 1, 2023, Jean-Paul Kress, M.D., as well as the then-current member of the Management Board Charlotte Lohmann have been granted performance share units under the Company's Performance Share Unit Program 2023. Further, with effect as of October 1, 2023, Lucinda Crabtree has also been granted performance share units under the Performance Share Unit Program 2023. After the expiry of the four-year waiting period and subject to the achievement of the defined performance criteria, the performance share units will be settled at the Company's discretion in cash, by transfer of Company treasury shares, or a combination of both, whereby the payout is limited to 250% of the initial grant amount granted to each Management Board member.
The following table lists the performance share units granted to Management Board members in the 2023 financial year.
| Management Board member | Allocation amount (in € thousands) | Allocation price (in €) | Number of allocated PSUs | Maximum number of final PSUs | ||||||||||||||||||||||
| Jean-Paul Kress, M.D. | 3,000.0 | 15.00 | 200,000 | 400,000 | ||||||||||||||||||||||
| Lucinda Crabtree, Ph.D.* | 800.0 | 28.00 | 28,571 | 57,142 | ||||||||||||||||||||||
| Charlotte Lohmann** | 625.0 | 15.00 | 20,833 | 41,666 | ||||||||||||||||||||||
| * Lucinda Crabtree, Ph.D. was appointed as member of the management board of MorphoSys AG with effect as of August 8, 2023. ** Charlotte Lohmann was appointed as member of the management board for the time period from March 1, 2023, to August 31, 2023. € 312.5 of the allocation amount have been granted to her for her activity as member of the Management Board. Further € 312.5k have been granted to her for her activity as employee of the Company and will not count towards the maximum remuneration. | ||||||||||||||||||||||||||
In accordance with the provisions of the Remuneration System 2022, the performance targets for the Performance Share Unit Program 2023 are the relative performance of the MorphoSys share price compared to the performance of the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index as well as an ESG target, the performance of the employee engagement within the MorphoSys Group compared to a benchmark set by the Supervisory Board. Development milestones have been defined as a further performance target, the achievement of which depends on the number of NDA/BLA or SBLA approvals received. For the overall target achievement after the end of the waiting period, the relative share price performance target is weighted at 40% and the ESG target at 20%. The performance milestones are also weighted at 40%.
In the event of a premature departure of a Management Board member, the waiting period and the assessment of the overall performance target achievement remain unaffected.
Relative Share Price Performance
The relative performance of the MorphoSys AG share is measured by comparing the performance of the MorphoSys AG share price during the waiting period with the performance of the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index as the benchmark index. The relevant MorphoSys AG share price is the average closing price of the MorphoSys AG share in Xetra trading on the Frankfurt Stock Exchange during the 30 trading days prior to the beginning of the four-year waiting period and during the three months prior to the end of the four-year waiting period. The relevant share price of the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index is its average closing price during the 30 trading days prior to the beginning of the four-year waiting period and during the three months prior to the end of the four-year waiting period. If the share price decreases compared to the benchmark index during the four-year waiting period, the degree of achievement of the relative share price performance target is 0%. If the MorphoSys AG share price performance is 0%
119
compared to the benchmark index, the performance target achievement is 100%. If the MorphoSys AG share price increases by 32% compared to the benchmark index, the performance target achievement is 150%. If the share price of MorphoSys AG increases by 64% compared to the benchmark index, the performance target achievement is 200%. Any further increase in the performance target achievement is not possible (cap). Between the percentage points, the share price development and the corresponding performance target achievement increase linearly. Within the overall performance target achievement, the relative share price performance is weighted at 40%.
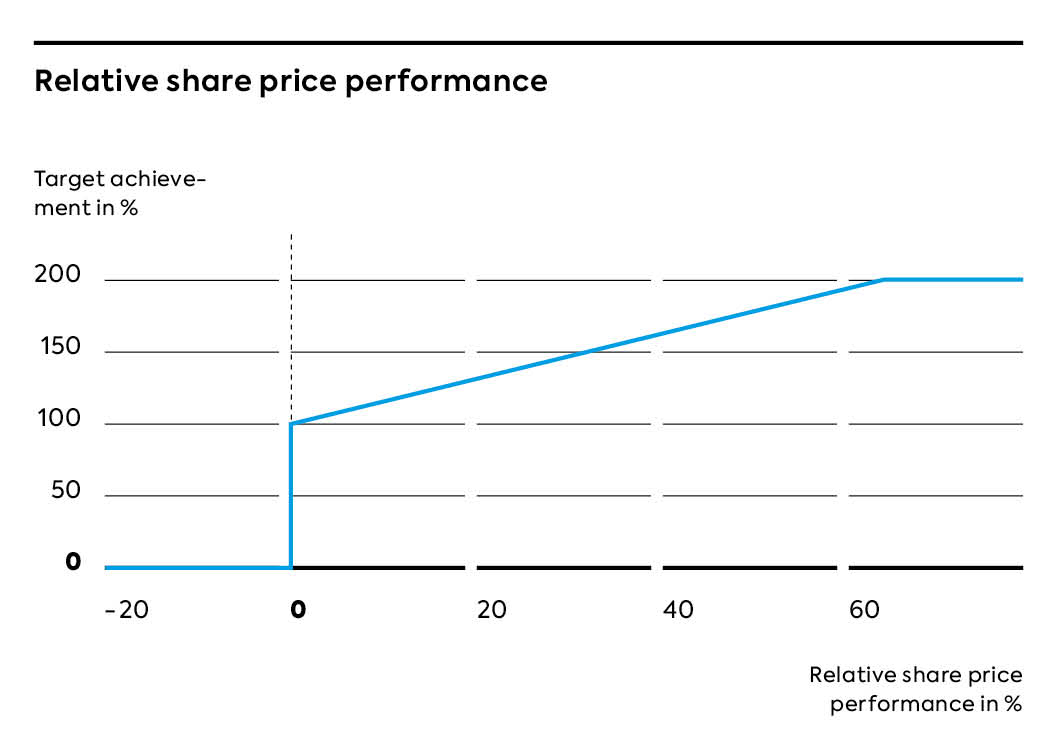
Development of the Workforce Engagement within the MorphoSys Group
In addition to the performance target relative share price performance, the Supervisory Board has also defined the development of the workforce engagement within the MorphoSys Group during the waiting period as a non-financial (ESG) target. This target has a weighting of 20% within the overall degree of target achievement. The target achievement is evaluated as follows:
For the workforce engagement performance target, the workforce engagement at the MorphoSys Group at the end of the four-year waiting period is compared to a benchmark of 55% set by the Supervisory Board. If the workforce engagement at the end of the waiting period is below 55%, the target achievement for the workforce engagement is 0%. If the workforce engagement at the end of the waiting period is 55%, the target achievement for the workforce engagement is 50%. If the workforce engagement at the end of the waiting period is 65%, the target achievement for the workforce engagement is 100%. If the workforce engagement at the end of the waiting period is 75%, the target achievement for the workforce engagement is 200%. Any further increase in the degree of the performance target achievement is not possible (cap). Between the percentage points, the target achievement increases linearly.
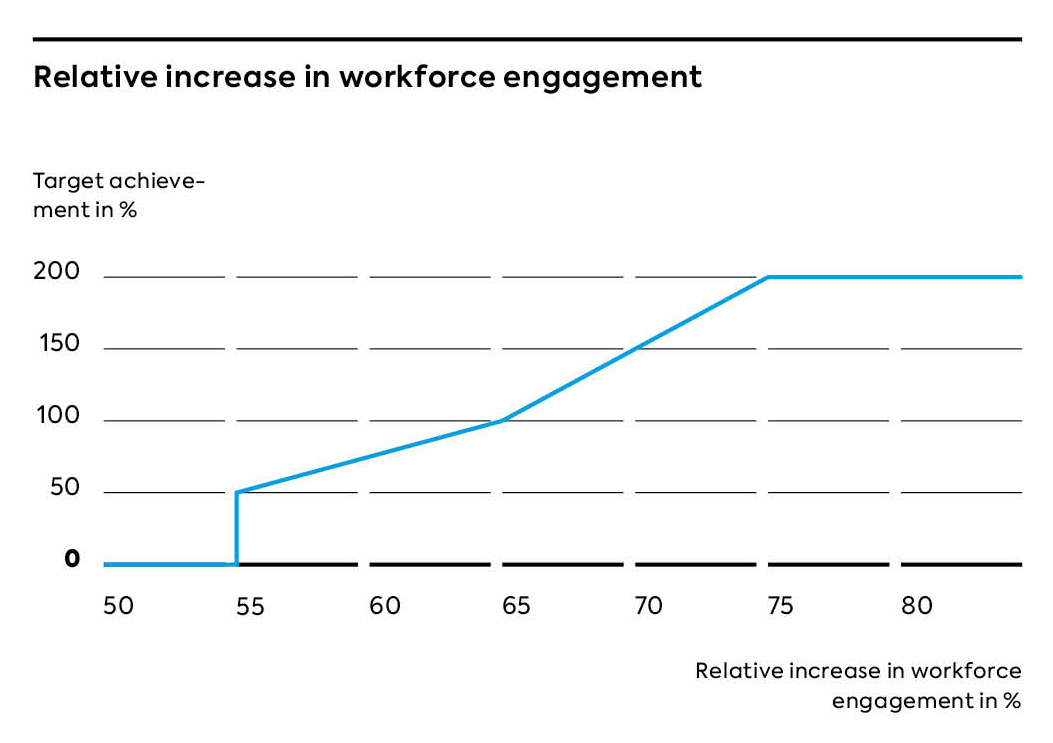
120
Development Milestones
The performance target of development milestones is based on the number of regulatory approvals of one or more NDAs/BLAs and/or SBLAs submitted by the Company in the U.S. If no application is approved, the target achievement for the development milestones performance target is 0%. If one application is approved, the target achievement for this performance target is 100%. Two approved applications correspond to a target achievement of 150%. Three approved applications correspond to a target achievement of 200%. Any further increase in the degree of performance target achievement is not possible (cap). The performance target of the development milestones is weighted at 40% within the overall target achievement.
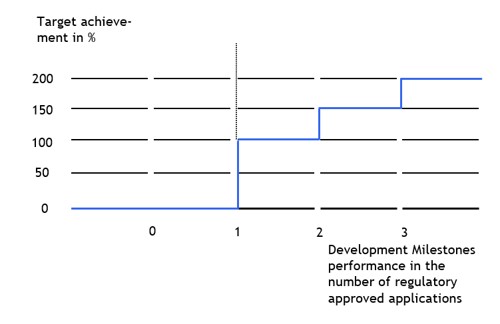
The overall target achievement of the Performance Share Unit Program 2023, as well as the resulting final number of performance share units and the payout amount, will be disclosed in the remuneration report for the 2027 financial year.
4.Further Remuneration Provisions
Compliance with the Maximum Remuneration
The service agreement of the former Management Board member Sung Lee did not fall under any remuneration system and thus, no maximum remuneration applied to the service agreement of Sung Lee. For the Chief Executive Officer, Jean-Paul Kress, M.D., the Chief Legal Officer, Charlotte Lohmann, who left the Management Board in the reporting period, as well as the Chief Financial Officer Lucinda Crabtree, Ph.D., the maximum remuneration as set out in the Remuneration System 2022 applies. If a Management Board member joins or leaves during the year, the maximum remuneration applies pro rata.
As the inflow from the Performance Share Unit Program allocated in the 2023 financial year can only be determined after the end of the four-year waiting period, i.e., in the 2027 financial year, compliance with the maximum remuneration for the 2023 financial year can be conclusively reported only as part of the remuneration report for the 2027 financial year.
Malus and Clawback Provisions
The service agreement of the Chief Executive Officer, Jean-Paul Kress, M.D., the service agreement of the Chief Legal Officer Charlotte Lohmann, who has left the Management Board in the reporting period, as well as the service agreement of the Chief Financial Officer Lucinda Crabtree, Ph.D., contain and contained, respectively, malus and clawback provisions entitling the Company to withhold or reclaim variable remuneration, particularly in the event of compliance violations or breaches of statutory obligations. Malus and clawback provisions are further included in all Performance Share Unit Programs since 2021. Further, the Company has implemented a Clawback Policy for the members of the Management Board in accordance with the corresponding provisions of the SEC, on the basis of which the Company has the right to reclaim incorrectly granted variable remuneration in case of a subsequent adjustment to the annual financial statements.
The Company had no reason to make use of these rights in the 2023 financial year
Benefits upon Termination of the Service Agreements
Severance Provisions
The service agreements of the Management Board members contain severance payment provisions that comply with the requirements of the German Corporate Governance Code. In the event of the premature termination of a Management Board
121
member’s service agreement, payments made by the Company to the Management Board member, including fringe benefits, shall not exceed the value of two years' remuneration (severance cap) and shall compensate no more than the remaining term of the service agreement. If the service agreement is terminated for good cause for which the Management Board member is responsible, no payments will be made to the Management Board member. The severance cap is calculated on the basis of the total remuneration for the previous full financial year and, where appropriate, is also based on the expected total remuneration for the current financial year.
From the time of his resignation from his position as member of the Management Board with effect as of the end of March 17, 2023, Sung Lee was released from his duties as a member of the Management Board until the end of March 31, 2023, with continued payment of his remuneration. In addition, all performance share units allocated to him became fully vested.
Change of Control
The service agreements of the current members of the Management Board provide for the following provisions in case of a change of control:
The service agreements of Jean-Paul Kress, M.D., and Lucinda Crabtree, Ph.D., provide for the following: In the event of a change of control and in the event that the area of responsibility of the respective member of the Management Board is significantly reduced within one year following the occurrence of the change of control, such Management Board member has the right to resign from the position as a member of the Management Board within three months of the reduction of the area of responsibility, by providing three months' notice to the end of a calendar month. The service agreement will also end at the same time. In this case, the respective Management Board member would be entitled to a severance payment in the amount of the annual base remuneration and the annual bonus until the regular expiry of the service agreement, not exceeding the value of two years' remuneration and compensating no more than the remaining term of the service agreement. The service agreement of Charlotte Lohmann, who has left the Management Board during the reporting year, provided for a corresponding provision.
The service agreement of Sung Lee, who left the Company during the reporting year, provided for a right of the Management Board member for the change of control to resign from the position as a member of the Management Board within three months following the change of control, by providing three months' notice to the end of a calendar month. The service agreement will also end at the same time. In this case, the respective Management Board member would have been entitled to a severance payment in the amount of the annual base remuneration and the annual bonus until the regular expiry of his service agreement, not exceeding the value of two years' remuneration and compensating no more than the remaining term of the service agreement.
In the event of a change of control in connection with a public takeover offer, the terms and conditions of the Performance Share Unit Program 2022 and the Performance Share Unit Program 2023 provide for a right of the Management Board and the Company, respectively, to cancel all performance share units in return for a compensation payment, with the remuneration payment corresponding to the amount of the offer price of the takeover offer. The maximum payout amount of 250% of the individual grant amount as well as the maximum remuneration remain unaffected.
Non-Compete Clause
The service agreements of the members of the Management Board provide for a non-compete clause for a period of six months after their departure. In return, MorphoSys AG will make a compensation payment in the amount of 100% of the annual base salary after termination of the service agreement for the duration of the non-competition clause. The service agreement of Sung Lee originally also provided for a non-compete clause, which however has been waived by the Company. The service agreement of the former Management Board member Charlotte Lohmann, entered into with effect as of March 1, 2023, did not provide for a post-contractual non-compete provision, as Charlotte Lohmann remained as employee of the Company after having left the Management Board.
5.Individual Disclosure of Management Board Remuneration for the 2023 Financial Year
Target Remuneration of the Current Management Board Members for the 2023 Financial Year
The following table shows the respective target total remuneration for Management Board members for the 2023 financial year. This includes the target total remuneration defined for the 2023 financial year, which will be granted in the case of a 100% target achievement. The target total remuneration is based on the assumption of continued service of all members of the Management Board throughout the entire 2023 financial year.
122
| Jean-Paul Kress, M.D. | Sung Lee* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chief Executive Officer | Chief Financial Officer | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2023 | 2022 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| in € thou-sands | share of total | in € thou-sands | share of total | in € thou-sands | share of total | in € thou-sands | share of total | |||||||||||||||||||||||||||||||||||||||||||||||||
| Fixed remuneration | Base compensation | 864.2 | 18.1 | % | 796.7 | 17.1 | % | 121.8 | 48.0 | % | 476.6 | 22.2 | % | |||||||||||||||||||||||||||||||||||||||||||
| Fringe benefits**** | 190.1 | 4.0 | % | 211.6 | 4.6 | % | 46.9 | 18.5 | % | 536.0 | 25.0 | % | ||||||||||||||||||||||||||||||||||||||||||||
| Private provision | 19.3 | 0.4 | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Total | 1,073.6 | 22.5 | % | 1,008.2 | 21.7 | % | 168.7 | 66.4 | % | 1,012.6 | 47.2 | % | ||||||||||||||||||||||||||||||||||||||||||||
| Variable remuneration | Short-term incentive (STI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bonus | 691.3 | 14.5 | % | 637.3 | 13.7 | % | 85.2 | 33.6 | % | 333.6 | 15.5 | % | ||||||||||||||||||||||||||||||||||||||||||||
| Long-term incentive (LTI) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PSUP | 3,000.0 | 63.0 | % | 3,000.0 | 64.6 | % | 0.0 | — | % | 800.0 | 37.3 | % | ||||||||||||||||||||||||||||||||||||||||||||
| SOP | — | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Total | 3,691.3 | 77.5 | % | 3,637.3 | 78.3 | % | 85.2 | 33.6 | % | 1,133.6 | 52.8 | % | ||||||||||||||||||||||||||||||||||||||||||||
| Total target remuneration | Total remuneration | 4,765.0 | 100.0 | % | 4,645.6 | 100.0 | % | 253.9 | 100.0 | % | 2,146.2 | 100.0 | % | |||||||||||||||||||||||||||||||||||||||||||
| Charlotte Lohmann** | Lucinda Crabtree, Ph.D.*** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chief Legal Officer | Chief Financial Officer | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2023 | 2022 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| in € thou-sands | share of total | in € thou-sands | share of total | in € thou-sands | share of total | in € thou-sands | share of total | |||||||||||||||||||||||||||||||||||||||||||||||||
| Fixed remuneration | Base compensation | 169.9 | 18.3 | % | — | — | % | 180.2 | 14.6 | % | — | — | % | |||||||||||||||||||||||||||||||||||||||||||
| Fringe benefits**** | 15.4 | 1.7 | % | — | — | % | 126.6 | 10.3 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Private provision | 0.0 | — | % | — | — | % | 0.0 | 0.0 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Total | 185.3 | 19.9 | % | — | — | % | 306.9 | 24.9 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Variable remuneration | Short-term incentive (STI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bonus | 118.9 | 12.8 | % | — | — | % | 126.2 | 10.2 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Long-term incentive (LTI) | — | — | % | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| PSUP | 625.0 | 67.3 | % | — | — | % | 800.0 | 64.9 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| SOP | — | — | % | — | — | % | — | — | — | — | % | |||||||||||||||||||||||||||||||||||||||||||||
| Total | 743.9 | 80.1 | % | — | — | % | 926,169 | 1 | — | — | % | |||||||||||||||||||||||||||||||||||||||||||||
| Total target remuneration | Total remuneration | 929.2 | 100.0 | % | — | — | % | 1,233.0 | 100.0 | % | — | — | % | |||||||||||||||||||||||||||||||||||||||||||
| * Sung Lee resigned from his position as member of the Management Board with effect as of the end of March 17, 2023. The service agreement entered into between the Company and Sung Lee has been terminated with effect as of the end of March 31, 2023. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ** Charlotte Lohmann was appointed as member of the management board of the Company for the time period from March 1, 2023, to August 31, 2023. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *** Lucinda Crabtree, Ph.D. was appointed as member of the management board of MorphoSys AG with effect as of August 8, 2023. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ****In addition to the fringe benefits mentioned here, contributions were also made to the pension scheme. For details see table Company pension plan. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Remuneration Awarded and Due to Current Management Board Members in the 2023 Financial Year Pursuant to Section 162 AktG
The following tables present the fixed and variable remuneration components awarded and due within the meaning of Section 162 (1) sentence 1 AktG to the in the 2023 financial year current Management Board members in the 2022 and 2023 financial years. The tables include all remuneration amounts actually received by the individual Management Board members in these financial years (“awarded”) and all remuneration amounts legally due but not yet received (“due”). To present the remuneration awarded for the 2023 fiscal year in a complete and transparent manner, the table further also voluntarily discloses the individual grant amount granted under the PSUP 2023 to each member of the Management Board. The actual inflow value under the PSUP 2023 will be disclosed in the remuneration report for the financial year 2027.
The amount of the annual bonus (STI) for the 2023 financial year will be determined and paid out during the 2024 financial year and will therefore be included in the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in the 2024 financial year. In contrast, the remuneration awarded and due pursuant to Section 162 (1) sentence 1 AktG in
123
the 2023 financial year also includes the annual bonus for the 2022 financial year, which was paid out in March 2023. The previous year's figures have been adjusted in accordance with this definition.
Furthermore, the stock options and performance shares granted in the 2019 financial year vested in the 2023 financial year. The value (in €) of the change in quantity of the stock options, i.e., the difference between the final and the originally granted number of stock options, is thereby attributed to the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in the 2023 financial year, based on the fair value of the stock options at the time they were granted in the 2019 financial year. Furthermore, the value of the MorphoSys shares transferred to fulfill the performance shares exercisable and exercised in the 2023 financial year shall be attributed to the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in the 2023 financial year, based on the share price of the MorphoSys AG share at the time of exercise.
In addition to the remuneration amounts, the relative proportion of all fixed and variable remuneration components in total remuneration is also disclosed. For the sake of transparency and comprehensibility, the relative proportions are not linked to the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG but to the actual remuneration received during the 2023 financial year including the value of the performance share units issued during the 2023 financial year.
124
| Jean-Paul Kress, M.D. | Sung Lee | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chief Executive Officer | Chief Financial Officer | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2023 | 2022 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| in € thou-sands | share of total acc. to §162 AktG | in € thou-sands | share of total acc. to §162 AktG | in € thou-sands | share of total acc. to §162 AktG | in € thou-sands | share of total acc. to §162 AktG | |||||||||||||||||||||||||||||||||||||||||||||||||
| Fixed remuneration | Base compensation | 864.2 | -168.1 | % | 796.7 | 39.8 | % | 121.8 | 17.4 | % | 476.6 | 47.1 | % | |||||||||||||||||||||||||||||||||||||||||||
| Fringe benefits**** | 190.1 | -37.0 | % | 211.6 | 10.6 | % | 46.9 | 6.7 | % | 536.0 | 52.9 | % | ||||||||||||||||||||||||||||||||||||||||||||
| Private provision | 19.3 | -3.8 | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Total | 1,073.6 | -208.9 | % | 1,008.2 | 50.3 | % | 168.7 | 24.0 | % | 1,012.6 | 100.0 | % | ||||||||||||||||||||||||||||||||||||||||||||
| Variable remuneration | Short-term incentive (STI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bonus | 1,017.9 | -198.1 | % | 995.3 | 49.7 | % | 532.8 | 76.0 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Long-term incentive (LTI) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PSUP****** | 3,000.0 | 3,000.0 | — | 800,000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOP | — | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| PSP (close-out value) | — | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| SOP (close-out value)***** | (2,605.5) | 506.9 | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Total | 1,412.4 | 308.9 | % | 3,995.3 | 49.7 | % | 532.8 | 76.0 | % | 800,000 | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Others | Severance payment | — | — | % | — | — | % | — | — | % | — | — | % | |||||||||||||||||||||||||||||||||||||||||||
| Total remuneration | Total remuneration | 2,486.0 | 5,003.5 | 701.5 | 1,812.6 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Total remuneration as defined by Section 162 AktG****** | (514.0) | 100.0 | % | 2,003.5 | 100.0 | % | 701.5 | 100.0 | % | 1,012.6 | 100.0 | % | ||||||||||||||||||||||||||||||||||||||||||||
| Charlotte Lohmann** | Lucinda Crabtree, Ph.D.*** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chief Legal Officer | Chief Financial Officer | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2023 | 2022 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| in € thou-sands | share of total acc. to §162 AktG | in € thou-sands | share of total acc. to §162 AktG | in € thou-sands | share of total acc. to §162 AktG | in € thou-sands | share of total acc. to §162 AktG | |||||||||||||||||||||||||||||||||||||||||||||||||
| Fixed remuneration | Base compensation | 169.9 | 91.7 | % | — | — | % | 180.2 | 58.7 | % | — | — | % | |||||||||||||||||||||||||||||||||||||||||||
| Fringe benefits**** | 15.4 | 8.3 | % | — | — | % | 126.6 | 41.3 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| private provision | — | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Total | 185.3 | 100.0 | % | — | — | % | 306.9 | 100.0 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Variable remuneration | Short-term incentive (STI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bonus | 0.0 | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Long-term incentive (LTI) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PSUP****** | 625.0 | — | 800.0 | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOP | — | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| PSP (close-out value) | — | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| SOP (close-out value)***** | — | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Total | 625.0 | — | % | — | — | % | 800.0 | — | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
| Others | Severance payment | — | — | % | — | — | % | — | — | % | — | — | % | |||||||||||||||||||||||||||||||||||||||||||
| Total remuneration | 810.3 | — | 1,106.9 | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total remuneration as defined by Section 162 AktG***** | 185.3 | 100.0 | % | — | — | % | 306.9 | 100.0 | % | — | — | % | ||||||||||||||||||||||||||||||||||||||||||||
*Sung Lee resigned from his position as member of the Management Board with effect as of the end of March 17, 2023. The service agreement entered into between the Company and Sung Lee has been terminated with effect as of the end of March 31, 2023. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| **Charlotte Lohmann was appointed as member of the management board of the Company for the time period from March 1, 2023, to August 31, 2023. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ***Lucinda Crabtree, Ph.D. was appointed as member of the management board of MorphoSys AG with effect as of August 8, 2023. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
****In addition to the fringe benefits mentioned here, contributions were also made to the pension scheme. For details see table Company pension plan. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
125
| *****The CEO Jean-Paul Kress, M.D., was granted 57,078 stock options (initial number of stock options) under the Stock Option Program 2019 at a fair value of € 106.16. At the end of the four-year term and based on a target achievement of 57%, the final number was 32,535 stock options. The specified value represents the quantitative development of the stock options on the basis of the fair value of € 106.16, since the stock options have not yet been exercised. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ******This amount corresponds to the remuneration awarded and due within the meaning of Section 162 AktG to the members of the Management Board during the financial year 2023. The disclosed amounts amongst others include the quantitative development of the stock options compared to the financial year during which they have been granted, which can also be negative. The amount does not include the value of the Performance Share Units granted to the members of the Management Board during the financial year 2023 under the Company's Performance Share Unit Program, since this value will be allocated to the remuneration awarded and due within the meaning of section 162 AktG of the financial year during which the waiting period expires. Against this background, the remuneration value disclosed for individual Management Board members can also be negative as it is only awarded and due once the waiting period has expired. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Remuneration Awarded and Due to Former Management Board Members in the 2023 Financial Year Pursuant to Section 162 AktG
The following table shows the remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG to former Management Board members in the 2023 financial year. In accordance with Section 162 (5) AktG, personal details are omitted for former Management Board members if they left the Management Board prior to December 31, 2013
| Malte Peters, M.D. (until December 31, 2022) | Roland Wandeler Ph.D. (until 12/31/2021) | Simon Moroney Ph.D. (until 8/31/2019) | Jens Holstein (until 11/13/2020) | Markus Enzelberger, Ph.D. (until 2/29/2020) | Marlies Sproll, Ph.D.* (until 10/31/2017) | Arndt Schottelius M.D. Ph.D. (until 2/15/2017) | ||||||||||||||||||||||||||||||||||||||||||||
| In € thou-sands | share of total acc. to §162 AktG | in € thou- sands | in % of total | in € thou- sands | share of total acc. to §162 AktG | in € thou- sands | share of total acc. to §162 AktG | in € thou- sands | share of total acc. to §162 AktG | in € thou- sands | share of total acc. to §162 AktG | in € thou- sands | share of total acc. to §162 AktG | |||||||||||||||||||||||||||||||||||||
| Fixed and variable remuneration | Bonus | 575.8 | 367.5 | % | — | — | % | — | — | % | — | — | % | — | — | % | — | — | % | — | — | % | ||||||||||||||||||||||||||||
| Other | — | — | % | 290.4 | 100.0 | % | — | — | % | — | — | % | — | — | % | — | — | % | — | — | % | |||||||||||||||||||||||||||||
| LTI | PSP | 13.5 | 8.6 | % | — | — | % | 78.8 | (13.5 | %) | 38.5 | (9.8 | %) | 50.9 | (13.3 | %) | — | — | % | — | — | % | ||||||||||||||||||||||||||||
| SOP** | (432.6) | (276.1 | %) | — | — | % | (660.4) | 113.5 | % | (432.6) | 113.3 | % | (432.6) | 113.3 | % | — | — | % | — | — | % | |||||||||||||||||||||||||||||
| Sub Total | 156.7 | 100.0 | % | 290.4 | 100.0 | % | (581.6) | 100.0 | % | (394.1) | 100.0 | % | (381.8) | 100.0 | % | — | — | % | — | — | % | |||||||||||||||||||||||||||||
| Total | 156.7 | 100.0 | % | 290.4 | 100.0 | % | (581.6) | 100.0 | % | (394.1) | 100.0 | % | (381.8) | 100.0 | % | 258,963 | 100.0 | % | — | — | % | |||||||||||||||||||||||||||||
| *The position as Management Board member of Marlies Sproll, Ph.D., was suspended during the period 04/15/2017 to 10/31/2017. **Former CEO Simon Moroney, Ph.D., was granted 10,987 stock options under the Stock Option Program 2019 at a fair value of € 87.86. At the end of the four-year waiting period and based on a target achievement of 29%, the final number of stock options amounted to 10,987. The former Management Board members Dr. Malte Peters, Dr. Markus Enzelberger and Jens Holstein were each granted 6,936 stock options under the Stock Option Program 2019 at a fair value of € 87.86. At the end of the four-year waiting period and based on a target achievement of 29%, the final number of stock options for each of them amounted to 6.936. The specified values represent the quantitative development of the stock options on the basis of the fair value of € 87.86, since the stock options have not yet been exercised. | ||||||||||||||||||||||||||||||||||||||||||||||||||
c.Remuneration of the Members of the Supervisory Board
The Company’s Annual General Meeting on May 19, 2021, adopted a remuneration system for the Supervisory Board.
In addition to reimbursement of their expenses, Supervisory Board members receive an annual fixed base remuneration amounting to € 98,210.00 for the chair of the Supervisory Board, € 58,926.00 for the deputy chair, and € 39,284.00 for all other members of the Supervisory Board.
In addition, the chair of the Supervisory Board receives € 4,000.00 for each Supervisory Board meeting chaired, and the other Supervisory Board members receive € 2,000.00 for each Supervisory Board meeting attended. For committee work, the chair of the Audit Committee receives € 18,000.00, the chair of another committee receives € 12,000.00, and the other committee members each receive € 6,000.00. Committee members also receive € 1,200.00 for each committee meeting attended. Depending on the domicile of the Supervisory Board member and the location of the Supervisory Board meeting, a lump-sum expense allowance of € 2,000.00 may be paid in addition.
In the 2023 financial year, the members of the Supervisory Board received a total of € 510,272.
The fixed annual base remuneration and the remuneration for work on committees are due and payable to Supervisory Board members in equal quarterly installments. Attendance fees and expense allowances for participation in Supervisory Board meetings are due and payable at the end of each calendar quarter in which the respective meetings took place.
126
| Base compensation | Committee compensation | Attendance fee | Total remuneration | |||||||||||||||||||||||||||||||||||||||||||||||
| in € thousands | share of total | in € thousands | share of total | in € thousands | share of total | in € thousands | ||||||||||||||||||||||||||||||||||||||||||||
| Marc Cluzel, M.D., Ph.D | 2023 | 98.2 | 61.3 | % | 6.0 | 3.7 | % | 56.0 | 35.0 | % | 160.2 | |||||||||||||||||||||||||||||||||||||||
| 2022 | 98.2 | 65.7 | % | 6.0 | 4.0 | % | 45.2 | 30.3 | % | 149.4 | ||||||||||||||||||||||||||||||||||||||||
| George Golumbeski, Ph.D.* | 2023 | 57.3 | 57.2 | % | 12.0 | 12.0 | % | 30.8 | 30.8 | % | 100.1 | |||||||||||||||||||||||||||||||||||||||
| 2022 | 58.9 | 58.8 | % | 12.0 | 12.0 | % | 29.2 | 29.2 | % | 100.1 | ||||||||||||||||||||||||||||||||||||||||
| Krisja Vermeylen | 2023 | 39.3 | 40.7 | % | 18.0 | 18.7 | % | 39.2 | 40.6 | % | 96.5 | |||||||||||||||||||||||||||||||||||||||
| 2022 | 39.3 | 44.0 | % | 18.0 | 20.2 | % | 32.0 | 35.8 | % | 89.3 | ||||||||||||||||||||||||||||||||||||||||
| Michael Brosnan | 2023 | 39.3 | 33.6 | % | 27.7 | 23.7 | % | 49.9 | 42.7 | % | 117.0 | |||||||||||||||||||||||||||||||||||||||
| 2022 | 39.3 | 43.0 | % | 18.0 | 19.7 | % | 34.0 | 37.3 | % | 91.3 | ||||||||||||||||||||||||||||||||||||||||
| Sharon Curran* | 2023 | 40.9 | 44.2 | % | 15.7 | 17.0 | % | 36.0 | 38.9 | % | 92.7 | |||||||||||||||||||||||||||||||||||||||
| 2022 | 39.3 | 54.2 | % | 6.0 | 8.3 | % | 27.2 | 37.5 | % | 72.5 | ||||||||||||||||||||||||||||||||||||||||
| Andrew Cheng, M.D., Ph.D. | 2023 | 39.3 | 49.1 | % | 6.0 | 7.5 | % | 34.8 | 43.5 | % | 80.1 | |||||||||||||||||||||||||||||||||||||||
| 2022 | 24.5 | 60.3 | % | 3.7 | 9.2 | % | 12.4 | 30.5 | % | 40.6 | ||||||||||||||||||||||||||||||||||||||||
| *Sharon Curran has been elected as Vice-Chair of the Supervisory Board for the month of December 2023 on an interim basis. | ||||||||||||||||||||||||||||||||||||||||||||||||||
d.Comparison of Remuneration and Earnings Development
Pursuant to Section 162 (1) sentence 2 no. 2 AktG, the following table presents the earnings development of MorphoSys AG, the annual change in the remuneration of the members of the Management Board and the Supervisory Board, and the annual change in the average remuneration of the employees of MorphoSys AG on a full-time equivalent basis over the last five financial years. With regard to the financial years 2018 through 2020, the average remuneration of the Management Board and the Supervisory Board members is based on the remuneration disclosed in the remuneration report for the respective financial year, whereas for the financial years 2022 to 2023, the remuneration awarded and due pursuant to Section 162 (1) sentence 1 AktG in the respective financial year was used. The value of the performance share units issued to the members of the Management Board in the 2021, 2022 and 2023 financial years under the Company's respective Performance Share Unit Programs is not included in the table, as the performance share units issued in the respective financial year are not attributable to the remuneration awarded or due in the respective financial year within the meaning of Section 162 AktG.
The development of earnings is presented by using the net profit/loss of MorphoSys AG for the year as performance indicator.
The average employee remuneration is calculated based on MorphoSys AG’s workforce in Germany, which had an average of 371 active employees (headcount, excluding trainees) in the 2023 financial year.
The average employee remuneration includes personnel expenses for wages and salaries, fringe benefits, employer contributions to social security, any short-term variable remuneration components attributable to the financial year, as well as amounts of share-based remuneration.
The employee remuneration therefore corresponds, in principle, to remuneration awarded and due within the meaning of Section 162 (1) sentence 1 AktG in accordance with the remuneration of the Management Board and the Supervisory Board.
127
| Financial year | 2018 | change | 2019 | change | 2020 | change | 2021 | change | 2022 | change | 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Company earnings performance (in € thousands) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net profit/loss* | (67,033.8) | (23.9 | %) | (83,078.5) | (30.7 | %) | (108,622.3) | (185.8 | %) | (310,482.2) | 232.4 | % | 411,013.3 | (97.1 | %) | 11,805.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average employee remuneration (in € thousands) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average remuneration | 105.7 | (3.3 | %) | 102.3 | 10.8 | % | 113.3 | 17.6 | % | 133.2 | (2.0 | %) | 130.6 | 7.3 | % | 140.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Management Board remuneration (in € thousands)** | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | — | — | % | 3,567.9 | (14.9 | %) | 3,035.6 | (36.2 | %) | 1,935.3 | 3.5 | % | 2,003.5 | (125.7 | %) | -514.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sung Lee | — | — | % | — | — | % | — | **** | 488.6 | 107.2 | % | 1,012.6 | (30.7 | %) | 701.5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charlotte Lohmann | — | — | % | — | — | % | — | — | % | — | — | — | % | — | — | **** | **** | 185.3 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Lucinda Crabtree, Ph.D. | — | — | % | — | — | % | — | — | % | — | — | — | % | — | — | **** | **** | 306.9 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Former Management Board members (in € thousands)** | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dr. Malte Peters (bis 31. 12. 2022) | 1,592.2 | 13.9 | % | 1,813.2 | (0.3 | %) | 1,808.0 | (16.5 | %) | 1,510.2 | (24.4 | %) | 1,141.3 | (86.3 | %) | 156.7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Roland Wandeler, Ph.D. (until December 31, 2021) | — | — | % | — | — | % | 676.0 | 64.9 | % | 1,115.0 | 35.7 | % | 290.4 | (80.8 | %) | 290.4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Simon Moroney, Ph.D. (until August 31, 2019) | 2,280.8 | 13.9 | % | 2,596.7 | (14.9 | %) | 2,209.8 | (65.2 | %) | - | 768.3 | (108.2 | %) | (581.6) | 826.0 | % | (581.6) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Jens Holstein (until November 13, 2020) | 1,655.5 | 12.9 | % | 1,869.3 | 83.6 | % | 3,432.7 | (2.8 | %) | - | 3,335.8 | (101.0 | %) | (394.1) | 1052.5 | % | (394.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Markus Enzelberger, Ph.D. (until February 29, 2020) | 1,376.1 | 6.2 | % | 1,461.8 | (55.7 | %) | 647.6 | (58.5 | %) | - | 268.6 | (115.8 | %) | (381.8) | 798.3 | % | (381.8) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Marlies Sproll, Ph.D. (until October 31, 2017)*** | 280.8 | 129.8 | % | 645.3 | 107.9 | % | 1,341.6 | (83.3 | %) | - | 224.4 | (100.0 | %) | 259.0 | - | 259.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Arndt Schottelius M.D., Ph.D. (until February 28, 2017) | 46.5 | 377.3 | % | 222.1 | (16.9 | %) | 184.5 | (100.0 | %) | - | — | — | % | — | - | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| * The improvement in results of operations in 2022 was mainly due to the reduction in other provisions to Incyte due to updated planning assumptions regarding expected net cash flows from future Monjuvi sales and the pro rata reversal of deferred income related to Royalty Pharma for otilimab and gantenerumab due to the updates published in Q4 2022 by GlaxoSmithKline and Roche, respectively. ** The disclosed values include, amongst others, the quantitative development of the stock options compared to the financial year during which they have been granted. The value of the performance share units issued to the members of the Management Board during the financial years 2021, 2022 and 2023 is not included, since this value will be alllocated to the remuneration awarded and due within the meaning of section 162 AktG of the financial year during which the waiting period expires. Against this background, the remuneration values shown in this table may be negative in case of individual Management Board members. For a detailed disclosure of the remuneration components granted to the Management Board members during the respective financial year reference is made to the remuneration report for such financial year. *** The Management Board activities of Dr. Marlies Sproll were suspended in the period from April 15, 2017 to October 31, 2017. **** The display of a change to the previous fiscal year is not possible due to the entry date of the respective member of the Management Board. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Financial year | 2018 | change | 2019 | change | 2020 | change | 2021 | change | 2022 | change | 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Company earnings development (in € thousands) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net profit/loss* | (67,033.8) | (23.9 | %) | (83,078.5) | (30.7 | %) | (108,622.3) | (185.8 | %) | (310,482.2) | 232.4 | % | 411,013.3 | (97.1 | %) | 11,805.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average employee remuneration (in € thousands) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average remuneration | 105.7 | (3.3 | %) | 102.3 | 10.8 | % | 113.3 | 17.6 | % | 133.2 | (2.0 | %) | 130.6 | 7.3 | % | 140.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Supervisory Board remuneration (in € thousands) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marc Cluzel, M.D., Ph.D. | 109.1 | 36.2 | % | 148.6 | 8.1 | % | 160.6 | 2.7 | % | 165.0 | (9.5 | %) | 149.4 | 7.2 | % | 160.2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| George Golumbeski, Ph.D. | 54.2 | 53.0 | % | 82.9 | 16.0 | % | 96.1 | 6.2 | % | 102.1 | (2.0 | %) | 100.1 | — | % | 100.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Krisja Vermeylen | 74.3 | 20.7 | % | 89.7 | 6.7 | % | 95.7 | 3.3 | % | 98.9 | (9.7 | %) | 89.3 | 8.1 | % | 96.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Michael Brosnan | 47.6 | 79.3 | % | 85.3 | 0.5 | % | 85.7 | 4.0 | % | 89.1 | 2.5 | % | 91.3 | 28.1 | % | 117.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sharon Curran | — | — | % | 39.4 | 91.1 | % | 75.3 | (0.8 | %) | 74.7 | (2.9 | %) | 72.5 | 27.8 | % | 92.7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Andrew Cheng M.D., Ph.D. | — | — | % | — | — | % | — | — | % | — | — | % | 40.6 | 97.1 | % | 80.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Former Supervisory Board members (in € thousands) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wendy Johnson (until 05/2022) | 83.6 | (0.4 | %) | 83.2 | 6.7 | % | 88.8 | 8.2 | % | 96.1 | (58.7 | %) | 39.7 | (100.0 | %) | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dr. Frank Morich (until 04/2020) | 84.2 | 24.1 | % | 104.5 | (68.8 | %) | 32.6 | (100.0 | %) | — | — | % | — | — | % | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dr. Gerald Möller (until 05/2018) | 48.4 | (100.0 | %) | — | — | % | — | — | % | — | — | % | — | — | % | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Karin Eastham (bis 05/2017) | 24.1 | (100.0 | %) | — | — | % | — | — | % | — | — | % | — | — | % | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Klaus Kühn (bis 05/2018) | — | — | % | — | — | % | — | — | % | — | — | % | — | — | % | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dr. Walter Blättler (until 08/2015) | — | — | % | — | — | % | — | — | % | — | — | % | — | — | % | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dr. Daniel Camus (until 08/2015) | — | — | % | — | — | % | — | — | % | — | — | % | — | — | % | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dr. Geoffrey Vernon (until 08/2015) | — | — | % | — | — | % | — | — | % | — | — | % | — | — | % | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| * The improvement in results of operations in 2022 was mainly due to the reduction in other provisions to Incyte due to updated planning assumptions regarding expected net cash flows from future Monjuvi sales and the pro rata reversal of deferred income related to Royalty Pharma for otilimab and gantenerumab due to the updates published in Q4 2022 by GlaxoSmithKline and Roche, respectively. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
128
e.Other Disclosures
MorphoSys maintains directors and officers liability insurance for Management Board members. This insurance covers the personal liability risk in the event that claims are made against members of the Management Board for pecuniary loss in the course of their duties. The insurance includes a deductible for Management Board members that complies with the requirements of the German Stock Corporation Act.
f.Supplementary Notes
This report is also available in German. In the event of any discrepancies, the German version shall be the authoritative version.
C. Board Practices
To ensure good corporate governance, a guiding principle of the cooperation between our Management Board and Supervisory Board is the open, comprehensive and regular communication of information. The dual board system prescribed by the German Stock Corporation Act clearly differentiates between a company’s management and supervision. The responsibility of both boards is clearly stipulated by law, by the Articles of Association and by the boards’ bylaws. The boards work closely together to make decisions and take actions for our benefit. Their stated objective is to sustainably increase our value.
Management Board members each have their own area of responsibility as defined in the schedule of responsibilities. They regularly report to their Management Board colleagues, their cooperation being governed by the bylaws. The Supervisory Board ratifies both the schedule of responsibilities and the bylaws.
Further, the Company has established the so-called Executive Committee. Under the leadership of the Chief Executive Officer of the Company, the Executive Committee is responsible for strategy development, the operational management of the Company and the achievement of its objectives and results. It shall prepare decisions for the resolutions of the Management Board and adopt decisions together with the Management Board, unless they fall within the sole competence of the Management Board by virtue of the law or a resolution of the Supervisory Board. The Executive Committee consists of the members of the Management Board and senior leadership persons of the Company from the core areas of the Company such as Business Development & Licensing and Alliance Management, Technical Operations, Human Resources, Legal, Compliance & Intellectual Property as well as Corporate Affairs. Currently, besides the members of the Management Board, the members of the Executive Committee are Charlotte Lohmann (Chief Legal and Human Resources Officer), Barbara Krebs-Pohl, Ph.D. (Chief Business Officer), Joe Horvart (US General Manager), Tim Demuth, M.D., Ph.D. (Chief Research and Development Officer), Luisa Ciccarelli (SVP, Global Head of Technical Operations) and Thomas Biegi (SVP, Head of Corporate Affairs).
Meetings of the Executive Committee in general take place weekly and at least every two weeks and at any time required in the interest of the Company. Separate Management Board meetings shall in general take place if this is required in the interest of the Company. During these meetings, resolutions are passed concerning dealings and transactions that, under the bylaws, require the approval of the entire Management Board. For material events, each Management Board or Supervisory Board member can call an extraordinary meeting of the entire Management Board. Management Board resolutions can also be passed outside of meetings by an agreement made orally, by telephone or in writing (also by e-mail). Generally, minutes are taken of the meetings of Executive Committee and of the full Management Board.
In addition to the regularly scheduled meetings, Management Board strategy workshops are also held for developing and prioritizing the Group-wide strategic objectives.
The Management Board promptly and comprehensively informs the Supervisory Board in writing and at Supervisory Board meetings about planning, business development, the Group’s position, risk management and other compliance issues. Extraordinary meetings of the Supervisory Board are also called for material events. The Management Board involves the Supervisory Board in the strategy, planning and all fundamental Company issues. In addition to regular Supervisory Board meetings, a strategy meeting generally takes place between the Management Board and Supervisory Board once annually to discuss our strategic direction. The Management Board’s bylaws specify that material business transactions require the approval of the Supervisory Board. Detailed information on the cooperation of the Management Board and Supervisory Board and important items of discussion during the 2023 financial year can be found in the Report of the Supervisory Board.
The Supervisory Board holds a minimum of two meetings per calendar half-year. The Supervisory Board has supplemented the Articles of Association with bylaws that apply to its duties. In accordance with these bylaws, the Chairperson of the Supervisory Board coordinates the activities of the Supervisory Board, chairs the Supervisory Board meetings and represents
129
the interests of the Supervisory Board externally. The Supervisory Board typically passes its resolutions in meetings, but resolutions may also be passed outside of meetings in writing (also by e-mail), by telephone or video conference.
The Supervisory Board has a quorum when at least two-thirds of its members take part in the vote. Resolutions of the Supervisory Board are generally passed with a simple majority. In the event of a tied vote, the vote of the Chairperson of the Supervisory Board is decisive.
Minutes are completed for Supervisory Board meetings and resolutions passed outside of meetings. A copy of the Supervisory Board’s minutes is made available to all Supervisory Board members. The Supervisory Board regularly conducts an evaluation in accordance with the recommendations of the German Corporate Governance Code on how effective the Supervisory Board and its committees are. The members of the Management Board also participate in this evaluation.
Composition and Working Practices of the Management Board and Supervisory Board Committees
The Management Board has not formed any committees.
The Supervisory Board has established three permanent committees: The Audit Committee, the Remuneration and Nomination Committee and the Science and Technology Committee. The members of the three committees formed by the Supervisory Board are professionally qualified.
The members of the Science and Technology Committee also serve as members of the Ad-Hoc Committee, which meets in this capacity if necessary.
Audit Committee
The main task of the Audit Committee is to support the Supervisory Board in fulfilling its supervisory duties with respect to the accuracy of the annual statutory and consolidated financial statements, the activities of the auditor and internal control functions, such as risk management, compliance and internal auditing. The Audit Committee submits a recommendation to the Supervisory Board for the election at the Annual General Meeting of an independent auditor. The members of the Audit Committee are Michael Brosnan (Chairperson), Sharon Curran and Krisja Vermeylen. Currently, Michael Brosnan meets the prerequisite of an independent expert for accounting and audit and Krisja Vermeylen and Sharron Curran meet the prerequisite of an independent expert for audit respectively.
Remuneration and Nomination Committee
The Remuneration and Nomination Committee is responsible for preparing and reviewing the Management Board’s compensation system. When necessary, the Remuneration and Nomination Committee searches for suitable candidates to appoint to the Management Board and Supervisory Board and submits appointment proposals to the Supervisory Board. The Committee also prepares the contracts made with Management Board members. The members of the Remuneration and Nomination Committee are Krisja Vermeylen (Chairperson), Marc Cluzel, M.D., Ph.D., and Michael Brosnan.
Science and Technology Committee
The Science and Technology Committee advises the Supervisory Board on matters concerning proprietary drug and technology development and prepares the relevant Supervisory Board resolutions. The members of the Science and Technology Committee are George Golumbeski, Ph.D. (Chairperson), Sharon Curran, and Andrew Cheng, M.D., Ph.D.
In line with Section C.14 of the German Corporate Governance Code, the Supervisory Board members’ biographies are published on our website under About us—Leadership—Supervisory Board.
Corporate Governance Practices
At MorphoSys, responsible, sustainable and value-oriented corporate governance is a high priority. Good corporate governance is an essential aspect of our corporate management and forms the framework for the Group’s management and supervision, which includes the Group’s organization, commercial principles and tools for its guidance and control.
The German Corporate Governance Code (the “Code”) provides a standard for the transparent monitoring and management of companies that strongly emphasizes shareholder interests. The German Federal Ministry of Justice originally published the Code in 2002; it was last amended on April 28, 2022, (Code “2022”), which came into force on June 27, 2022. In particular, the Code contains principles, recommendations and suggestions for the Management Board and the Supervisory Board that are intended to ensure that the company is managed in the enterprise’s best interests. Further, the objective of the Code is to make the dual German corporate governance system transparent and understandable. Against this background, the Code aims to promote confidence in the management and supervision of German listed companies by investors, customers, employees and the general public.
130
There is no obligation to comply with the recommendations or suggestions of the Code. The German Stock Corporation Act requires only that the Management Board and Supervisory Board of a German listed company issue an annual declaration that either (i) states that the company has complied with the recommendations of the Code or (ii) lists the recommendations that the company has not complied with and explains its reasons for deviating from the recommendations of the Code. In addition, a listed company is also required to state in this annual declaration whether it intends to comply with the recommendations or list the recommendations it does not plan to comply with in the future. These declarations have to be published permanently on the company’s website. If the company changes its policy on certain recommendations between such annual declarations, it must disclose this fact and explain its reasons for deviating from the recommendations. Non-compliance with suggestions contained in the Code need not be disclosed.
Many of the corporate governance principles contained in the Code have been practiced at MorphoSys for many years. Our corporate governance is detailed in the Statement on Corporate Governance pursuant to Section 289f HGB and the Group Statement on Corporate Governance pursuant to 315d HGB. The statement also contains the annual Declaration of Conformity, relevant information on corporate governance practices and a description of the Management Board and Supervisory Board’s working practices. Additional information can be found in the Corporate Governance Report of the 2023 Annual Report.
Independence
The Supervisory Board considers it appropriate that at least four of its members are independent (Section C.6 of the German Corporate Governance Code and the Nasdaq listing rules). Members of the Supervisory Board are considered independent when they have no personal or business relationship with MorphoSys, its management, a controlling shareholder or an affiliate that may give rise to a material and more than temporary conflict of interest. All six current members of the Supervisory Board meet the criteria to be classified as independent. Therefore, the Supervisory Board currently meets the quota of four independent members.
Significant and more than temporary conflicts of interest should be avoided, especially when it involves work for major competitors. It should be noted, however, that conflicts of interest in certain cases cannot be excluded. Any potential conflicts of interest must be disclosed to the Chairperson of the Supervisory Board and remedied appropriately. There are currently no conflicts of interest.
D.Employees
On December 31, 2023, the MorphoSys Group had 524 employees (December 31, 2022: 629). The MorphoSys Group employed an average of 564 employees in 2023 (2022: 647).
Of the average 564 employees, 9 worked in production, 365 in research and development, 127 in general and administrative positions, and 63 in sales and marketing. All of these employees are based at our locations in Germany and the United States. We do not have collective wage agreements with our employees, and there were no employee strikes during the reporting year.
At the end of the 2023 reporting year, our workforce comprised employees representing 37 different nationalities (2022: 43).
E.Share Ownership
The members of the Management Board and the Supervisory Board hold less than 1% of the shares issued by the Company. All shares, performance shares and stock options held by each member of the Management Board and the Supervisory Board are listed below.
131
Directors’ Holdings
Ordinary Shares
| 1/1/2023 | Additions | Sales | 12/31/2023 | |||||||||||||||||||||||
| Management Board | ||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | 0 | 0 | 0 | 0 | ||||||||||||||||||||||
Sung Lee1 | 2,250 | — | — | — | ||||||||||||||||||||||
Charlotte Lohmann2 | 1,168 | 157 | 0 | 1,325 | ||||||||||||||||||||||
Lucinda Crabtree, Ph.D.3 | — | 0 | 0 | 0 | ||||||||||||||||||||||
| Total | 3,418 | 157 | 0 | 1,325 | ||||||||||||||||||||||
| Supervisory Board | ||||||||||||||||||||||||||
| Marc Cluzel, M.D., Ph.D. | 4,500 | 4,025 | 0 | 8,525 | ||||||||||||||||||||||
| Michael Brosnan** | 5,000 | 0 | 0 | 5,000 | ||||||||||||||||||||||
| Sharon Curran | 0 | 0 | 0 | 0 | ||||||||||||||||||||||
| George Golumbeski, Ph.D. | 0 | 0 | 0 | 0 | ||||||||||||||||||||||
| Krisja Vermeylen | 2,000 | 1,000 | 0 | 3,000 | ||||||||||||||||||||||
Andrew Cheng, M.D., Ph.D. | 0 | 0 | 0 | 0 | ||||||||||||||||||||||
| Total | 11,500 | 5,025 | 0 | 16,525 | ||||||||||||||||||||||
**Michael Brosnan holds 20,000 ADSs, i.e. 5,000 shares converted in ordinary shares.
Stock Options
| 1/1/2023 | Additions | Adjustment due to Performance Criteria4 | Forfeitures | Exercises | 12/31/2023 | |||||||||||||||||||||||||||||||||
| Management Board | ||||||||||||||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | 81,989 | 0 | (24,543) | 0 | 0 | 57,446 | ||||||||||||||||||||||||||||||||
Sung Lee1 | 0 | — | — | — | — | — | ||||||||||||||||||||||||||||||||
Charlotte Lohmann2 | 4,595 | 0 | (1,493) | 0 | 0 | 3,102 | ||||||||||||||||||||||||||||||||
Lucinda Crabtree, Ph.D.3 | — | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||
| Total | 86,584 | 0 | (26,036) | 0 | 0 | 60,548 | ||||||||||||||||||||||||||||||||
Performance Shares
| 1/1/2023 | Additions | Adjustment due to Performance Criteria 4 | Forfeitures | Conversion to Shares | 12/31/2023 | |||||||||||||||||||||||||||||||||
| Management Board | ||||||||||||||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||
Sung Lee1 | 0 | — | — | — | — | — | ||||||||||||||||||||||||||||||||
Charlotte Lohmann2 | 626 | 0 | (469) | 0 | (157) | 0 | ||||||||||||||||||||||||||||||||
Lucinda Crabtree, Ph.D.3 | — | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||
| Total | 626 | 0 | (469) | 0 | (157) | 0 | ||||||||||||||||||||||||||||||||
1 Sung Lee resigned as a member of the Management Board with effect from the end of March 17, 2023. Changes after his departure from the Management Board are not presented.
2 With effect as of March 1, 2023, Charlotte Lohmann has been appointed as a member of the Management Board and Chief Legal Officer until the end August 31, 2023. Opening and closing balances presented in the tables were held by Charlotte Lohmann correspondingly before and after she was appointed as a member of the Management Board.
3 Lucinda Crabtree joined the Management Board of MorphoSys AG effective August 8, 2023.
4 Adjustment due to established performance criteria. For performance criteria that have not been met, a target achievement of 100% is assumed.
The members of our Supervisory Board do not hold stock options, convertible bonds or performance shares.
A detailed description of the stock option plans and long-term-incentive programs granted to members of our Management Board can be found in the Notes (sections 5.1 and 5.2).
132
F. Disclosure of a registrant’s action to recover erroneously awarded compensation
In November 2023, we adopted a Compensation Recovery Policy in compliance with the SEC rules and the NASDAQ listing standards to recover any excess incentive-based compensation from current and former executive officers after an accounting restatement. The Compensation Recovery Policy is included in this Form 20-F as Exhibit 97.1.
During or after fiscal year 2023, we were not required to prepare an accounting restatement that required recovery of erroneously awarded compensation pursuant to the Compensation Recovery Policy. As of December 31, 2023, there was no outstanding balance of erroneously awarded compensation to be recovered from the application of the Compensation Recovery Policy to a prior restatement.
133
Item 7. Major Shareholders and Related Party Transactions
A.Major Shareholders
The following table sets forth information, as of February 16, 2024, regarding the beneficial ownership of our ordinary shares for:
•members of our Supervisory Board;
•members of our Management Board;
•members of our supervisory and Management Boards as a group; and
•each person who has reported to us that such person beneficially owns 3% or more of our outstanding ordinary shares pursuant to applicable German law or 5% or more of our outstanding shares pursuant to applicable U.S. law.
Beneficial ownership is determined in accordance with the rules of the SEC. These rules generally attribute beneficial ownership of securities to persons who possess sole or shared voting power or investment power with respect to those securities and include ordinary shares that can be acquired within 60 days of February 16, 2024. The percentage of shares beneficially owned is computed on the basis of 37,655,137 issued shares as of February 16, 2024. Except as otherwise indicated, all of the shares reflected in the table are ordinary shares and all persons listed below have sole voting and investment power with respect to the shares beneficially owned by them, subject to applicable community property laws. The information is not necessarily indicative of beneficial ownership for any other purpose.
Except as otherwise indicated in the table below, the address of each of the directors, executive officers and named beneficial owners is Semmelweisstrasse 7, 82152 Planegg, Germany.
| Shareholders with 3% or more | Numbers | % | ||||||||||||
UBS Group AG (1) | 4,120,473 | 10.94 | % | |||||||||||
Kynam Capital Management, LP (2) | 3,690,331 | 9.80 | % | |||||||||||
Morgan Stanley (3) | 2,643,833 | 7.02 | % | |||||||||||
JP Morgan Chase & Co (4) | 2,286,262 | 6.07 | % | |||||||||||
The Goldman Sachs Group, Inc. (5) | 1,340,031 | 3.56 | % | |||||||||||
Royalty Pharma Investments 2019 ICAV (6) | 1,337,552 | 3.55 | % | |||||||||||
| Members of Supervisory Board and Management Board | Numbers | % | ||||||||||||
| Jean-Paul Kress, M.D. | 0 | * | ||||||||||||
| Sung Lee | 0 | * | ||||||||||||
| Marc Cluzel, M.D., Ph.D. | 8,525 | * | ||||||||||||
| Michael Brosnan** | 5,000 | * | ||||||||||||
| Sharon Curran | 0 | * | ||||||||||||
| George Golumbeski, Ph.D. | 0 | * | ||||||||||||
| Andrew Cheng, M.D., Ph.D. | 0 | * | ||||||||||||
| Krisja Vermeylen | 3,000 | * | ||||||||||||
| Members of Supervisory Board and Management Board (as a group) | 16,525 | * | ||||||||||||
* Indicates holdings of less than 1%
(1)The information is based solely on a notification provided by UBS Group AG pursuant to the German Securities Trading Act (Wertpapierhandelsgesetz). MorphoSys issued a respective voting rights announcement on February 16, 2024. The principal business address of UBS Group AG is Bahnhofstrasse 45, 8001 Zürich, Switzerland.
(2)The information is based solely on a notification provided by Kynam Capital Management, LP pursuant to the German Securities Trading Act (Wertpapierhandelsgesetz). MorphoSys issued a respective voting rights announcement on February 14, 2024. The principal business address of Kynam Capital Management, LP is 221 Elm Road Princeton, NJ 08540, United States.
(3)The information is based solely on a notification provided by Morgan Stanley & Co. LLC pursuant to the German Securities Trading Act (Wertpapierhandelsgesetz). MorphoSys issued a respective voting rights announcement on
134
February 16, 2024. The principal business address of Morgan Stanley & Co. LLC is 1585 Broadway New York, NY 10036, United States.
(4)The information is based solely on a notification provided by JPMorgan Chase & Co. pursuant to the German Securities Trading Act (Wertpapierhandelsgesetz). MorphoSys issued a respective voting rights announcement on February 15, 2024. The principal business address of JPMorgan Chase & Co. is 880 Powder Mill Rd., Wilmington, Delaware 19807, United States.
(5)The information is based solely on a notification provided by The Goldman Sachs Group, Inc. pursuant to the German Securities Trading Act (Wertpapierhandelsgesetz). MorphoSys issued a respective voting rights announcement on February 16, 2024. The principal business address of The Goldman Sachs Group, Inc. is Corporation Trust Center, 1209 Orange Street, Wilmington, United States.
(6)The information is based solely on a notification provided by Royalty Pharma plc pursuant to the German Securities Trading Act (Wertpapierhandelsgesetz). MorphoSys issued a respective voting rights announcement on August 2, 2021. The principal business address of Royalty Pharma plc is The Pavilions, Bridgewater Road, Bristol, England, BS13 8AE.
Our ordinary shares are issued only in bearer form. Accordingly, we cannot determine the identity of our shareholders or how many shares a particular shareholder owns and the number of ordinary shares directly held by persons with U.S. addresses.
All of our shareholders have the same voting rights. We are not aware of any arrangement that may, at a subsequent date, result in a change of control of our company.
B.Related Party Transactions
Since January 1, 2023, there has not been, nor is there currently proposed, any material transaction or series of similar material transactions to which we were or are a party in which any of the members of our Supervisory or Management Boards, executive officers, holders of more than 10% of any class of our voting securities, or any member of the immediate family of any of the foregoing persons, had or will have a direct or indirect material interest, other than the compensation and shareholding arrangements we describe in the “Directors, Senior Management and Employees” and “Major Shareholders” sections of this report.
C.Interests of Experts and Counsel
Not applicable.
Item 8.Financial Information
A.Consolidated Statements and Other Financial Information
Consolidated Financial Statements
Our consolidated financial statements are appended at the end of this annual report on Form 20-F, starting at page F-1, and incorporated herein by reference.
Legal Proceedings
From time to time, we are subject to various legal proceedings and claims that arise in the ordinary course of our business activities. Although the results of litigation and claims cannot be predicted with certainty, as of the date of this report, we do not believe we are party to any claim or litigation, the outcome of which would individually or in the aggregate be reasonably expected to have a material adverse effect on our business. Regardless of the outcome, litigation can have an adverse impact on us because of defense and settlement costs, diversion of management resources and other factors.
Within the past twelve months, we have not been party to any litigation, arbitration proceedings or administrative proceedings that we believe may have a material effect on our financial condition or profitability, and we are not aware of any such proceedings being pending or threatened. For further details also refer to Notes (section 6.2 Contingent Liabilities)
Dividend Distribution Policy
We have not paid any dividends on our ordinary shares since our inception, and we currently intend to retain any future earnings to finance the growth and development of our business. Therefore, we do not anticipate that we will declare or pay any
135
cash dividends in the foreseeable future. Except as required by law, any future determination to pay cash dividends will be at the discretion of our Management Board and Supervisory Board and will be dependent upon our financial condition, results of operations, capital requirements, and other factors our Management Board and Supervisory Board deem relevant.
B.Significant Changes
A detailed description of the significant changes can be found in the Notes (section 6.9).
Item 9.The Offer and Listing
A.Offer and Listing Details
The ADS have been listed on Nasdaq Global Market under the symbol “MOR” since April 23, 2018. Prior to that date, there was no public trading market for ADSs. Our ordinary shares have been trading on the Frankfurt Stock Exchange under the symbol “MOR” since March 1999. Prior to that date, there was no public trading market for ADSs or our ordinary shares.
B.Plan of Distribution
Not applicable.
C.Markets
The ADS have been listed on Nasdaq Global Market under the symbol “MOR” since April 23, 2018 and our ordinary shares have been listed on the Frankfurt Stock Exchange under the symbol “MOR” since March 1999.
D.Selling Shareholders
Not applicable.
E.Dilution
Not applicable.
F.Expenses of the Issue
Not applicable.
Item 10. Additional Information
A.Share Capital
Not applicable.
B.Memorandum and Articles of Association
The information set forth in our Registration Statement on Form F-1 (File No. 333-223843), automatically effective upon filing with the SEC on March 22, 2018, under the heading “Description of Share Capital” as supplemented by the section titled “Description of Share Capital” in the final prospectus supplement on Form 424(b)(4) dated April 18, 2018 filed with the SEC on April 19, 2018 is incorporated herein by reference.
C.Material Contracts
Business Combination Agreement with Novartis
On February 5, 2024 MorphoSys entered into the Business Combination Agreement with Novartis. Under the Business Combination Agreement, Novartis agreed to submit the Novartis Takeover Offer (as defined elsewhere herein). The Novartis Takeover Offer will contain customary closing conditions, in particular a minimum acceptance threshold of 65% of
136
MorphoSys’ share capital and regulatory clearances. MorphoSys agreed to publicly support the Novartis Takeover Offer, provided the Novartis Takeover Offer is launched in accordance with the terms provided for under the Business Combination Agreement.
Under the Business Combination Agreement, Novartis agreed to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to ensure sufficient financing for MorphoSys’ ordinary course of business after closing of the Novartis Takeover Offer, including the payment of any obligations of MorphoSys arising from the implementation of the Novartis Takeover Offer. The Business Combination Agreement provides that, immediately following the consummation of the Novartis Takeover Offer, Novartis will take all necessary steps to achieve, as soon as is reasonably possible, a delisting of the MorphoSys shares from all regulated markets in the European Economic Area, and MorphoSys will take all necessary steps under applicable laws to enable a delisting of the MorphoSys shares and the MorphoSys ADSs from Nasdaq as well as the deregistration of the MorphoSys Shares under the U.S. Exchange Act. In addition, MorphoSys and Novartis committed to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to ensure uncapped payouts of any long-term incentive plans active prior to signing of the Business Combination Agreement (the “Pre-2024 Long-Term Incentive Plans”) to management board members and all employees affected by any caps under German law. It will be offered to all employees to fully close out their Pre-2024 Long-Term Incentive Plans against payment of the offer price after the Novartis Takeover Offer. Uncapped payouts and the settlement of the Pre-2024 Long-Term Incentive Plans are planned to occur after the delisting.
Until closing of the Novartis Takeover Offer, MorphoSys will be subject to certain customary interim operating covenants. The strategy of MorphoSys will continue to be determined by the MorphoSys management board post-closing of the Novartis Takeover Offer. However, MorphoSys agreed, following closing of the Novartis Takeover Offer, to use best efforts to ensure that Novartis is appropriately represented on the MorphoSys supervisory board.
The Business Combination Agreement contains other customary covenants and agreements, including, but not limited to, covenants related to cooperation in the preparation of certain public filings and required documentation, public announcements, access to information and further assurances.
The Business Combination Agreement has a fixed term of three years and may be terminated by either party only for the reasons set out in the Business Combination Agreement. The Business Combination Agreement is governed by the laws of the Federal Republic of Germany.
A copy of the Business Combination Agreement is filed with this annual report on Form 20-F as Exhibit 4.14 and is incorporated herein by reference, and the foregoing description of the Business Combination Agreement is qualified in its entirety by reference thereto.
For a summary of other material contracts please refer to “Item 4. Business Overview” - section "Collaboration and License Agreements".
D.Exchange Controls
There are currently no legal restrictions in Germany on international capital movements and foreign exchange transactions, except in limited embargo circumstances (Teilembargo) relating to certain areas, entities or persons as a result of applicable resolutions adopted by the United Nations and the EU. Restrictions currently exist with respect to, among others, Belarus, Congo, Egypt, Eritrea, Guinea, Guinea-Bissau, Iran, Iraq, Lebanon, Libya, North Korea, Somalia, South Sudan, Sudan, Syria, Tunisia and Zimbabwe. In this regard, far reaching financial restrictions have also been introduced with regards to Russia. The regulatory environment regarding such legal restrictions is rapidly changing, reflective of evolving geopolitical environments.
For statistical purposes, there are, however, limited notification requirements regarding transactions involving cross-border monetary transfers. With some exceptions, every corporation or individual residing in Germany must report to the German Central Bank (Deutsche Bundesbank) (i) any payment received from, or made to, a non-resident corporation or individual that exceeds € 12,500 (or the equivalent in a foreign currency) and (ii) in case the sum of claims against, or liabilities payable to, non-resident individuals or corporations exceeds € 5,000,000 (or the equivalent in a foreign currency) in the aggregate at the end of any calendar month. Payments include cash payments made by means of direct debit, checks and bills, remittances denominated in euros and other currencies made through financial institutions, as well as netting and clearing arrangements.
E.Taxation
The following discussion is a summary of certain U.S. and German tax consequences of owning and disposing of the ADSs.
137
German Taxation
The following discussion addresses certain German tax consequences of acquiring, owning or disposing of the ADSs. With the exception of the subsection “Taxation of Holders Tax Resident in Germany” below, which provides an overview of dividend taxation to holders that are residents of Germany, this discussion applies only to U.S. treaty beneficiaries (defined below) that acquire ADSs.
This discussion is based on domestic German tax laws, including, but not limited to, circulars issued by German tax authorities, which are not binding on the German courts, and the Treaty (defined below). It is based upon tax laws in effect at the time of filing of this report. These laws are subject to change, possibly with retroactive effect. For example, certain member states of the European Union are considering introducing a financial transaction tax (Finanztransaktionssteuer) which, if and when introduced, may also be applicable on sales and/or transfer of ADSs. In addition, in Germany, for example, there are currently ongoing discussions on the raise of the top tax rate, which may also have an effect on the German tax consequences of acquiring, owning and disposing of the ADSs.
In addition, this discussion is based upon the assumption that each obligation in the deposit agreement and any related agreement will be performed in accordance with its terms. It does not purport to be a comprehensive or exhaustive description of all German tax considerations that may be of relevance in the context of acquiring, owning and disposing of ADSs.
The tax information presented in this report is not a substitute for tax advice. Prospective holders of ADSs should consult their own tax advisors regarding the German tax consequences of the purchase, ownership, disposition, donation or inheritance of ADSs in light of their particular circumstances, including the effect of any state, local, or other foreign or domestic laws or changes in tax law or interpretation. The same applies with respect to the rules governing the refund of any German dividend withholding tax (Kapitalertragsteuer) withheld. Only an individual tax consultation can appropriately account for the particular tax situation of each investor.
MorphoSys does not assume any responsibility for withholding tax at source.
Taxation of MorphoSys
MorphoSys’ taxable income, whether distributed or retained, is generally subject to corporate income tax (Körperschaftsteuer) at a uniform rate of 15% plus the solidarity surcharge (Solidaritätszuschlag) of 5.5% thereon, resulting in a total corporate income tax liability of 15.825%.
Dividends (Gewinnanteile) and other distributions received by MorphoSys from domestic or foreign corporations are exempt from corporate income tax, inter alia, if MorphoSys held at the beginning of the calendar year at least 10% of the registered share capital (Grundkapital or Stammkapital) of the distributing corporation which did not deduct the distributions from its own tax base; however, 5% of such revenue is treated as a non-deductible business expense and, as such, is subject to corporate income tax plus the solidarity surcharge. The acquisition of a participation of at least 10% in the course of a calendar year is deemed to have occurred at the beginning of such calendar year for the purpose of this rule. Participations in the share capital of other corporations which MorphoSys holds through a partnership, including co-entrepreneurships (Mitunternehmerschaften), are attributable to MorphoSys only on a pro rata basis at its entitlement to the profits of the relevant partnership. Subject to the above-mentioned requirements, 95% of the amount of dividends and other distributions that MorphoSys receives from corporations are exempt from corporate income tax. The same applies, in general and irrespective of the size of the shareholding, to profits earned by MorphoSys from the sale of shares in another domestic or foreign corporation. Losses incurred from the sale of such shares are not deductible for tax purposes.
In addition, MorphoSys is subject to trade tax (Gewerbesteuer) with respect to its taxable trade profit (Gewerbeertrag) from its permanent establishments in Germany (inländische gewerbesteuerliche Betriebsstätten). Trade tax is generally based on the taxable income as determined for corporate income tax purposes taking into account, however, certain add-backs and deductions.
The trade tax rate depends on the local municipalities in which MorphoSys maintains its permanent establishments. Dividends received from other corporations and capital gains from the sale of shares in other corporations are treated in principle in the same manner for trade tax purposes as for corporate income tax purposes. However, dividends received from domestic and foreign corporations are effectively 95% exempt from trade tax only if MorphoSys held at least 15% of the registered share capital (Grundkapital or Stammkapital) - in the event of foreign corporations - of the nominal capital (Nennkapital) of the distributing corporation at the beginning of the relevant tax assessment period.
Tax-loss carryforwards can be used to fully offset taxable income for corporate income tax and trade tax purposes up to an amount of EUR 1 million. If the taxable profit for the year or taxable profit subject to trade taxation exceeds this threshold, only up to 60% of the amount exceeding the threshold may be offset by tax-loss carryforwards. The remaining 40% is subject to tax (minimum taxation) (Mindestbesteuerung). The rules also provide for a tax carryback to the previous year with regard to corporate income tax. As a response to the COVID-19 pandemic tax carryback options for corporate income tax purposes were
138
extended for fiscal years 2020 and 2021 and as well for 2022. Unused tax-loss carryforwards may be generally carried forward indefinitely and used in subsequent assessment periods to offset future taxable income in accordance with this rule. However, unused losses, loss carryforwards and interest carryforwards are forfeited in full if within five years more than 50% of the subscribed capital, membership interests, equity interests or voting rights of MorphoSys are transferred, whether directly or indirectly, to an acquiring party or affiliated individuals/entities, or a similar change of ownership occurs (harmful acquisition) (schädlicher Beteiligungserwerb). A group of acquirers with aligned interests is also considered to be an acquiring party for these purposes. In addition, any current year losses incurred prior to the acquisition will not be deductible. A capital increase shall be deemed as equivalent to a transfer of the subscribed capital to the extent that it causes a change of the interest ratio in the capital of the corporation. Unused losses, loss carryforwards, and interest carryforwards are not forfeited (i) in the event of certain intra-group transactions, (ii) or to the extent that they are covered at the time of the harmful acquisition by certain built-in gains (stille Reserven) which are subject to tax in Germany. Alternatively to (i) and (ii), MorphoSys may, under certain requirements, opt for the continuity of business exemption (Fortführungsgebundener Verlustvortrag) to preserve unused losses, loss carryforwards and interest carryforwards.
German Taxation of ADS Holders
General
Based on the circular issued by the German Federal Ministry of Finance (BMF-Schreiben), dated May 24, 2013, reference number IV C 1 – S 2204/12/10003, as amended by the circular dated December 18, 2018 (reference number IV C 1 – S 2204/12/10003), in respect of the taxation of American Depositary Receipts, or ADRs, on domestic shares, or the ADR Tax Circular, for German tax purposes, the ADSs should, in light of the ADR Tax Circular, represent a beneficial ownership interest in the underlying shares of MorphoSys and qualify as ADRs for the purpose of the ADR Tax Circular. If the ADSs qualify as ADRs under the ADR Tax Circular, dividends would accordingly be attributable to holders of the ADSs for German tax purposes, and not to the legal owner of the ordinary shares (i.e., the financial institution on behalf of which the ordinary shares are stored at a domestic depository for the ADS holders). Furthermore, holders of the ADSs should be treated as beneficial owners of the capital of MorphoSys with respect to capital gains. However, investors should note that circulars published by the German tax authorities (including the ADR Tax Circular) are not binding on German courts, and it is unclear whether a German court would follow the ADR Tax Circular in determining the German tax treatment of the ADSs. For the purpose of this German tax section, it is assumed that the ADSs qualify as ADRs within the meaning of the ADR Tax Circular.
Taxation of Holders Not Tax Resident in Germany
The following discussion describes the material German tax consequences for a holder that is a U.S. treaty beneficiary of acquiring, owning and disposing of the ADSs. For purposes of this discussion, a “U.S. treaty beneficiary” is a resident of the United States for purposes of the Agreement between the Federal Republic of Germany and United States of America for the Avoidance of Double Taxation and the Prevention of Fiscal Evasion with respect to Taxes on Income and on Capital as of June 4, 2008 (Abkommen zwischen der Bundesrepublik Deutschland und den Vereinigten Staaten von Amerika zur Vermeidung der Doppelbesteuerung und zur Verhinderung der Steuerverkürzung auf dem Gebiet der Steuern vom Einkommen und vom Vermögen und einiger anderer Steuern in der Fassung vom 4. Juni 2008), hereinafter referred to as the “Treaty”, who is fully eligible for benefits under the Treaty.
A holder will be a U.S. treaty beneficiary entitled to full Treaty benefits in respect of the ADSs if it is, inter alia:
•the beneficial owner of the ADSs (and the dividends paid with respect thereto);
•a U.S. holder;
•not also a resident of Germany for German tax purposes; and
•not subject to the limitation on benefits (i.e., anti-treaty shopping) article of the Treaty that applies in limited circumstances.
Special rules apply to pension funds and certain other tax-exempt investors.
This discussion does not address the treatment of ADSs that are (i) held in connection with a permanent establishment or fixed base through which a U.S. treaty beneficiary carries on business or performs personal services in Germany or (ii) part of business assets for which a permanent representative in Germany has been appointed.
General Rules for the Taxation of Holders Not Tax Resident in Germany
Non-German resident holders of ADSs are subject to German taxation with respect to German sourced income (beschränkte Steuerpflicht). According to the ADR Tax Circular, income from the shares should be attributed to the holder of the ADSs for German tax purposes. As a consequence, income from the ADSs should be treated as German source income.
139
The full amount of a dividend distributed by MorphoSys to a non-German resident holder which does not maintain a permanent establishment or other taxable presence in Germany is subject to (final) German withholding tax at an aggregate rate of 26.375% (25% income tax plus 5.5% solidarity surcharge thereon) plus church tax (Kirchensteuer), if applicable. German withholding tax is withheld and remitted to the German tax authorities (i) by the disbursing agent (i.e., the German credit institution, financial services institution, securities trading enterprise or securities trading bank (each as defined in the German Banking Act (Kreditwesengesetz) and in each case including a German branch of a foreign enterprise, but excluding a foreign branch of a German enterprise)) that holds or administers the underlying shares in custody and disburses or credits the dividend income from the underlying shares or disburses or credits the dividend income from the underlying shares on delivery of the dividend coupons or disburses such dividend income to a foreign agent or (ii) the central securities depository (Wertpapiersammelbank) in terms of the German Depositary Act (Depotgesetz) holding the underlying shares in a collective deposit, if such central securities depository disburses the dividend income from the underlying shares to a foreign agent, regardless of whether a holder must report the dividend for tax purposes and regardless of whether or not a holder is a resident of Germany.
Pursuant to the Treaty, the German withholding tax may not exceed 15% of the gross amount of the dividends received by U.S. treaty beneficiaries. The excess of the total withholding tax, including the solidarity surcharge, over the maximum rate of withholding tax permitted by the Treaty is refunded to U.S. treaty beneficiaries upon application (subject to certain requirements). For example, for a declared dividend of 100, a U.S. treaty beneficiary initially receives 73.625 (100 minus the 26.375% withholding tax including solidarity surcharge). The U.S. treaty beneficiary is entitled to a partial refund from the German tax authorities in the amount of 11.375% of the gross dividend (of 100). As a result, the U.S. treaty beneficiary ultimately receives a total of 85 (85% of the declared dividend) following the refund of the excess withholding. However, investors should note that it is unclear how the German tax authorities will apply the refund process to dividends on the ADSs with respect to non-German resident holders of the ADSs. Further, such refund is subject to the German anti-avoidance treaty shopping rule (as described below in the section “—Withholding Tax Refund for U.S. Treaty Beneficiaries”).
German Taxation of Capital Gains of the U.S. Treaty Beneficiaries of the ADSs
The capital gains from the disposition of the ADSs realized by a non-German resident holder which does not maintain a permanent establishment or other taxable presence in Germany would be treated as German source income and be subject to German tax if such holder at any time during the five years preceding the disposition, directly or indirectly, owned 1% or more of MorphoSys’ share capital, irrespective of whether through the ADSs or shares of MorphoSys. If such holder had acquired the ADSs without consideration, the previous owner’s holding period and quota would be taken into account when calculating the above holding period and the participation threshold.
Pursuant to the Treaty, U.S. treaty beneficiaries are not subject to German tax even under the circumstances described in the preceding paragraph and therefore should not be taxed on capital gains from the disposition of the ADSs.
German statutory law requires the disbursing agent to levy withholding tax on capital gains from the sale of ADSs or other securities held in a custodial account in Germany. With regard to the German taxation of capital gains, disbursing agent means a German credit institution, a financial services institution, a securities trading enterprise or a securities trading bank (each as defined in the German Banking Act and, in each case including a German branch of a foreign enterprise, but excluding a foreign branch of a German enterprise) that holds the ADSs in custody or administers the ADSs for the investor or conducts sales or other dispositions and disburses or credits the income from the ADSs to the holder of the ADSs. The German statutory law does not explicitly condition the obligation to withhold taxes on capital gains being subject to taxation in Germany under German statutory law or on an applicable income tax treaty permitting Germany to tax such capital gains.
However, a circular issued by the German Federal Ministry of Finance provides that taxes on capital gains need not be withheld when the holder of the custody account is not a resident of Germany for tax purposes and the income is not subject to German taxation. The circular further states that there is no obligation to withhold such tax even if the non-German resident holder owns 1% or more of the share capital of a German company in a custodial account in Germany. While circulars issued by the German Federal Ministry of Finance are only binding on the German tax authorities but not on the German courts, in practice, the disbursing agents nevertheless typically rely on guidance contained in such circulars. Therefore, a disbursing agent would only withhold tax at 26.375% on capital gains derived by a U.S. treaty beneficiary from the sale of ADSs held in a custodial account in Germany in the event that the disbursing agent did not follow the above-mentioned guidance. In this case, the U.S. treaty beneficiary may be entitled to claim a refund of the withholding tax from the German tax authorities under the Treaty, as described below in the section “—Withholding Tax Refund for U.S. Treaty Beneficiaries”.
Withholding Tax Refund for U.S. Treaty Beneficiaries
U.S. treaty beneficiaries are generally eligible for treaty benefits under the Treaty, as described above in Section “—Taxation of Holders Not Tax Resident in Germany”. Accordingly, U.S. treaty beneficiaries are in general entitled to claim a refund of the
140
portion of the otherwise applicable 26.375% German withholding tax (corporate income tax including solidarity surcharge) on dividends that exceeds the applicable Treaty rate. However, such refund is only possible, provided that pursuant to special rules on the restriction of withholding tax credit, the following three cumulative requirements are met: (i) the shareholder must qualify as beneficial owner of the ADSs for an uninterrupted minimum holding period of 45 days within a period starting 45 days prior to and ending 45 days after the due date of the dividends, (ii) the shareholder has to bear at least 70% of the change in value risk related to the ADSs during the minimum holding period as described under (i) of this paragraph and has not entered into (acting by itself or through a related party) hedging transactions which lower the change in value risk by more than 30%, and (iii) the shareholder must not be obliged to fully or largely compensate directly or indirectly the dividends to third-parties. If these requirements are not met, then for a shareholder not being a tax-resident in Germany who applied for a full or partial refund of the withholding tax pursuant to a double taxation treaty, no refund is available. This restriction generally does only apply, if (i) the tax underlying the refund application is below a tax rate of 15% based on the gross amount of the dividends or capital gains and (ii) the shareholder does not directly own 10% or more in the shares of the company and is subject to income taxes in its state of residence, without being tax exempt. In addition to the aforementioned restrictions, the withholding tax refund may also be denied based on German anti-avoidance treaty shopping rules.
In general, as previously discussed, investors should note that it is unclear how the German tax administration will apply the refund process to dividends on the ADSs.
Due to the legal structure of the ADSs, only limited guidance from the German tax authorities exists on the practical application of this procedure with respect to the ADSs.
Taxation of Holders Tax Resident in Germany
This subsection provides an overview of dividend taxation with regard to the general principles applicable to MorphoSys’ holders that are tax resident in Germany. A holder is a German tax resident if, in case of an individual, he or she maintains a domicile (Wohnsitz) or a usual residence (gewöhnlicher Aufenthalt) in Germany or if, in case of a corporation, it has its place of management (Geschäftsleitung) or registered office (Sitz) in Germany.
The German dividend and capital gains taxation rules applicable to German tax residents require a distinction between ADSs held as private assets (Privatvermögen) and ADSs held as business assets (Betriebsvermögen).
ADSs as Private Assets (Privatvermögen)
If the ADSs are held as private assets by a German tax resident, dividends and capital gains are taxed as investment income and are principally subject to 25% German flat income tax on capital income (Abgeltungsteuer) (plus a 5.5% solidarity surcharge (Solidaritätszuschlag) thereon, resulting in an aggregate rate of 26.375% plus church tax (Kirchensteuer), if applicable), which is generally levied in the form of withholding tax (Kapitalertragsteuer). In other words, once deducted, the shareholder’s income tax liability on the dividends will be settled.
Shareholders may apply to have their capital investment income assessed in accordance with the general rules and with an individual’s personal income tax rate if this would result in a lower tax burden in which case actually incurred expenses are not deductible. The holder would be taxed on gross personal investment income (including dividends or gains with respect to ADSs), less the saver’s allowance of €801 for an individual or €1,602 for a married couple and a registered civil union (eingetragene Lebenspartnerschaft) filing taxes jointly. The deduction of expenses related to the investment income (including dividends or gains with respect to ADSs) is generally not possible for private investors.
Losses resulting from the disposal of ADSs can only be offset by capital gains from the sale of any shares (Aktien) and other ADSs. If, however, a holder directly or indirectly held at least 1% of the share capital of the company at any time during the five years preceding the sale, 60% of any capital gains resulting from the sale are taxable at the holder’s personal income tax rate (plus 5.5% solidarity surcharge thereon). Conversely, 60% of any capital losses are recognized for tax purposes.
Since 2021, the basis for the calculation of the solidarity surcharge (Solidaritätszuschlag) has been reduced for certain individual persons being subject to tax assessments (other than withholding taxes), and in certain cases, the solidarity surcharge has been abolished. However, the abolition or reduction of the solidarity surcharge is not applicable to corporations. In addition, the abolition or reduction of the solidarity surcharge will not affect withholding taxes. Solidarity surcharge will still be levied at 5.5% on the full withholding tax amount and withheld accordingly. There will not be any separate refund of such withheld solidarity surcharge (regardless of the aforementioned exemption limits) in case the withholding tax cannot be refunded either.
Church tax generally has to be withheld, if applicable, based on an automatic data access procedure, unless the shareholder has filed a blocking notice (Sperrvermerk) with the Federal Central Tax Office. Where church tax is not levied by way of withholding, it is determined by means of income tax assessment.
141
ADSs as Business Assets (Betriebsvermögen)
In case the ADSs are held as business assets, the taxation depends on the legal form of the holder (i.e., whether the holder is a corporation or an individual). Irrespective of the legal form of the holder, dividends are subject to the aggregate withholding tax rate of 26.375%. The withholding tax is credited against the respective holder’s income tax liability, provided that pursuant to special rules on the restriction of withholding tax credit, the following three cumulative requirements are met: (i) the shareholder must qualify as beneficial owner of the ADSs for an uninterrupted minimum holding period of 45 days occurring within a period starting 45 days prior to and ending 45 days after the due date of the dividends, (ii) the shareholder has to bear at least 70% of the change in value risk related to the ADSs during the minimum holding period as described under (i) of this paragraph and has not entered into (acting by itself or through a related party) hedging transactions which lower the change in value risk for more than 30%, and (iii) the shareholder must not be obliged to fully or largely compensate directly or indirectly the dividends to third-parties. If these requirements are not met, three-fifths of the withholding tax imposed on the dividends must not be credited against the shareholder’s (corporate) income tax liability, but may, upon application, be deducted from the shareholder’s tax base for the relevant tax assessment period. Such requirements also apply to ADSs, which lead to domestic income in Germany and which are held by a non-German depositary bank. A shareholder that is generally subject to German income tax or corporate income tax and that has received gross dividends without any deduction of withholding tax or that has received a refund of withholding tax, in particular due to a tax exemption without qualifying for a full tax credit under the aforementioned requirements has to notify the competent local tax office accordingly, has to file a withholding tax return (Steueranmeldung) and has to make a payment in the amount of the omitted withholding tax deduction. The special rules on the restriction of withholding tax credit do not apply to a shareholder whose overall dividend earnings within an assessment period do not exceed €20,000 or that has been the beneficial owner of the ADSs in the company for at least one uninterrupted year upon receipt of the dividends.
To the extent the amount withheld exceeds the income tax liability, the withholding tax will be refunded, provided that certain requirements are met (including the aforementioned requirements).
Special rules apply to credit institutions (Kreditinstitute), financial services institutions (Finanzdienstleistungsinstitute), financial enterprises (Finanzunternehmen), life insurance and health insurance companies, pension funds, and investment funds (Investmentfonds).
With regard to holders in the legal form of a corporation, dividends and capital gains are in general 95% tax exempt from corporate income tax (including solidarity surcharge), inter alia, if the shareholder held at least 10% of the registered share capital (Grundkapital oder Stammkapital) of MorphoSys at the beginning of the calendar year. The remaining 5% is treated as non-deductible business expense and, as such, is subject to corporate income tax (including solidarity surcharge). The acquisition of a participation of at least 10% in the course of a calendar year is deemed to have occurred at the beginning of such calendar year for the purpose of this rule. Participations in the share capital of other corporations held through a partnership, including co-entrepreneurships (Mitunternehmerschaften), are attributable to the holder only on a pro rata basis at the ratio of its entitlement to the profits of the relevant partnership. Moreover, actual business expenses incurred to generate the dividends may be deducted.
However, the amount of any dividends after deducting business expenses related to the dividends is subject to the trade tax, unless the corporation held at least 15% of MorphoSys’ registered share capital at the beginning of the relevant tax assessment period. In the latter case, the aforementioned exemption of 95% of the dividend income also applies for trade tax purposes.
Losses from the sale of ADSs are generally not tax deductible for corporate income tax and trade tax purposes.
With regard to individuals holding ADSs as business assets, 60% of dividends and capital gains are taxed at the individual’s personal income tax rate (plus 5.5% solidarity surcharge and church tax, if applicable, thereon). Correspondingly, only 60% of business expenses related to the dividends and capital gains as well as losses from the sale of ADSs are principally deductible for income tax purposes. Furthermore, trade tax may generally apply, provided the ADSs are held through a permanent establishment in Germany. However, the trade tax might be (partly) credited against the income tax liability of the holder pursuant to a lump sum procedure.
German Inheritance and Gift Tax (Erbschaft- und Schenkungsteuer)
The transfer of ADSs to another person by inheritance or gift should be generally subject to German inheritance and gift tax only if:
(1) the decedent or donor or heir, beneficiary or other transferee maintained his or her domicile or a usual residence in Germany or had its place of management or registered office in Germany at the time of the transfer, or is a German citizen who has spent no more than five consecutive years outside of Germany without maintaining a domicile in Germany or is a German citizen who serves for a German entity established under public law and is remunerated for his or her service
142
from German public funds (including family members who form part of such person’s household, if they are German citizens) and is only subject to estate or inheritance tax in his or her country of domicile or usual residence with respect to assets located in such country (special rules apply to certain former German citizens who neither maintain a domicile nor have their usual residence in Germany);
(2) at the time of the transfer, the ADSs are part of the business assets of a permanent establishment in Germany or for which a permanent representative in Germany has been appointed; or
(3) the ADSs subject to such transfer are part of a portfolio that represents at the time of the transfer 10% or more of the registered share capital of the company and that has been held directly or indirectly by the decedent or donor, either alone or together with related persons.
The Agreement between the Federal Republic of Germany and the United States of America for the avoidance of double taxation with respect to taxes on inheritances and gifts as of December 21, 2000 (Abkommen zwischen der Bundesrepublik Deutschland und den Vereinigten Staaten von Amerika zur Vermeidung der Doppelbesteuerung auf dem Gebiet der Nachlass-, Erbschaft- und Schenkungssteuern in der Fassung vom 21. Dezember 2000), hereinafter referred to as the “United States-Germany Inheritance and Gifts Tax Treaty”, provides that the German inheritance tax or gift tax can, with certain restrictions, only be levied in the cases of (1) and (2) above. Special provisions apply to certain German citizens living outside of Germany and former German citizens.
Other Taxes
No German transfer tax, value-added tax, stamp duty or similar taxes are assessed on dividend payments neither on the purchase, sale or other transfer of ADSs.
Due to the Tax Haven Defense Act (Steueroasen-Abwehrgesetz) special rules apply for business relationships or shareholdings in or with reference to one or more non-cooperative tax jurisdiction(s) (tax jurisdictions in the EU list of non-cooperative countries and territories for tax purposes). In particular, dividends and capital gains received by a resident in a non-cooperative tax jurisdiction would be fully taxable.
U.S. Taxation
The following discussion is a summary of certain material U.S. federal income tax considerations to U.S. holders (as defined below) of owning and disposing of the ADSs.
The information provided below is based on the Internal Revenue Code of 1986, as amended, Treasury Regulations, the Treaty, Internal Revenue Service, or IRS, rulings and pronouncements, and judicial decisions all as now in effect and all of which are subject to change or differing interpretations, possibly with retroactive effect. There can be no assurance that the IRS or a court will not take a contrary position with respect to any U.S. federal income tax considerations described below.
This discussion does not provide a complete analysis of all potential U.S. tax considerations that may be relevant to a decision to purchase ADSs by any particular investor. In particular, this discussion does not address tax considerations applicable to a U.S. holder (as defined in “—U.S. Taxation” below) that may be subject to special tax rules, including, without limitation, dealers or traders in securities, notional principal contracts or currencies, financial institutions, insurance companies, U.S. expatriates and inverted companies, certain stapled companies, tax-exempt organizations, tax-deferred or other retirement accounts, regulated investment companies, real estate investment trusts, a person that holds ADSs as part of a hedge, straddle, conversion or other integrated transaction for tax purposes, a person that purchases or sells ADSs as part of a wash sale for tax purposes, a person whose functional currency for tax purposes is not the U.S. dollar, a person who does not hold the ADSs as capital assets for tax purposes, a person who acquired the ADSs pursuant to the exercise of any employee stock option or otherwise as compensation, a person subject to special tax accounting rules as a result of any item of gross income with respect to the ADSs being taken into account in an applicable financial statement; or a person that owns or is deemed to own 10% or more of the company’s shares by vote or value. In addition, the summary does not address the 3.8% Medicare tax imposed on certain net investment income, the alternative minimum tax or any aspect of U.S. federal estate and gift tax laws or any foreign, state or local laws that may be applicable to a holder.
For purposes of this summary, a “U.S. holder” is a beneficial owner of ADSs that for U.S. federal income tax purposes is (1) an individual who is a citizen or resident of the United States, (2) a corporation, or an entity treated as a corporation for U.S. federal income tax purposes, created or organized in or under the laws of the United States or any state of the United States, including the District of Columbia, (3) an estate, the income of which is subject to U.S. federal income taxation regardless of its source, or (4) a trust (i) the administration of which is subject to the primary supervision of a U.S. court and which has one or more U.S. persons who have the authority to control all substantial decisions of the trust or (ii) that has otherwise elected to be treated as a U.S. person under the applicable regulations.
143
If a partnership (including an entity or arrangement, domestic or foreign, treated as a partnership for U.S. federal income tax purposes) holds ADSs, the tax treatment of a partner in the partnership will depend upon the status of the partner and the activities of the partnership. A holder of ADSs that is a partnership, and partners in such partnership, should consult their own tax advisors about the U.S. federal income tax consequences of owning and disposing of the ADSs.
In general, a holder of ADSs should be treated as the owner of our ordinary shares for U.S. federal income tax purposes. Holders should consult their own tax advisors concerning the tax consequences of converting ADSs to ordinary shares.
Each prospective holder of ADSs should consult its own tax advisors regarding the U.S. federal, state and local or other tax consequences of acquiring, owning and disposing of the company’s ADSs in light of their particular circumstances. U.S. holders should also review the discussion under “—German Taxation” for the German tax consequences to a U.S. holder of the ownership of the ADSs.
Distributions
Subject to the discussion below under “PFIC Rules,” the gross amount of any distribution that is actually or constructively received by a U.S. holder with respect to its ADSs without reduction for any German taxes withheld will be a dividend to the extent the amount of such distribution is paid out of our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. To the extent the amount of such distribution exceeds our current or accumulated earnings and profits, such amount will be treated first as a non-taxable return of capital to the extent of such U.S. holder’s adjusted tax basis in its ADSs, and to the extent the amount of such distribution exceeds such adjusted tax basis, will be treated as capital gain from the sale of the ADSs. Because we do not intend to determine our earnings and profits on the basis of U.S. federal income tax principles, any distribution we pay will generally be reported as dividend income for U.S. federal income tax purposes. If you are a non-corporate U.S. holder, dividends paid to you that constitute “qualified dividend income” (discussed below) should be taxable to you at a preferential rate (rather than the higher rates of tax generally applicable to items of ordinary income).
Dividends paid to a non-corporate U.S. holder generally will constitute qualified dividend income if (i) we are a “qualified foreign corporation” (discussed below), (ii) you are not under any obligation to make related payments with respect to positions in substantially similar or related property, and (iii) you hold our ADSs for more than 60 days during the 121-day period beginning 60 days before the ex-dividend date and your risk of loss with respect to the ADSs is not otherwise diminished. A non-U.S. corporation (other than a corporation that is classified as a PFIC for the taxable year in which the dividend is paid or the preceding taxable year) generally will be considered to be a qualified foreign corporation (i) if it is eligible for the benefits of a comprehensive tax treaty with the United States which the Secretary of Treasury of the United States determines is satisfactory for purposes of this provision and which includes an exchange of information provision, or (ii) with respect to any dividend it pays on ADSs that are readily tradable on an established securities market in the United States. We are incorporated under the laws of Germany, and we believe that we qualify as a resident of Germany for purposes of, and are eligible for the benefits of, the Treaty, although there can be no assurance in this regard. Further, the IRS has determined that the Treaty is satisfactory for purposes of the qualified dividend rules and that it includes an exchange-of-information program. In addition, our ADSs are listed on the Nasdaq Global Market, which is an established securities market in the United States, and we expect the ADSs to be readily tradable on the Nasdaq Global Market. However, there can be no assurance that the ADSs will be considered readily tradable on an established securities market in the United States. Accordingly, subject to the discussion below with respect to the PFIC rules, any dividends paid on our ADSs to non-corporate U.S. holders will generally be expected to be “qualified dividend income.” If we are a PFIC (as discussed below under—PFIC Rules”) during the year of a distribution or the year preceding a distribution, such distributions paid by us with respect to our ADSs will not be eligible for the preferential income tax rate. Prospective investors should consult their own tax advisors regarding the taxation of distributions under these rules.
Dividends paid on our ADSs will not be eligible for the dividends-received deduction generally available to corporate U.S. holders.
Subject to applicable limitations, non-refundable German taxes withheld from dividends on the ADSs can be generally claimed as a credit against the U.S. holder’s U.S. federal income tax liability. For purposes of the U.S. foreign tax credit rules, dividends with respect to our ADSs should constitute income from sources outside of the United States and should generally be passive income for purposes of computing the foreign tax credit allowable to the U.S. holder. The amount of the qualified dividend income, if any, paid to a U.S. holder that is subject to the reduced dividend income tax rate and that is taken into account for purposes of calculating the U.S. holder’s U.S. foreign tax credit limitation must be reduced by the rate differential portion of the dividend. In lieu of claiming a foreign tax credit, U.S. holders may, at their election, deduct foreign taxes, including any German income tax, in computing their taxable income, subject to generally applicable limitations under U.S. law. An election to deduct foreign taxes instead of claiming foreign tax credits applies to all foreign taxes paid or accrued in the taxable year.
144
The rules applicable to foreign tax credits are complex. Prospective investors should consult their tax advisors regarding the implications of the foreign tax credit provisions for them, in light of their particular situation.
The gross amount of any dividend paid in foreign currency will be included in the gross income of a U.S. holder in an amount equal to the U.S. dollar value of the foreign currency calculated by reference to the exchange rate in effect on the date the dividend distribution is includable in the U.S. holder’s income, regardless of whether the payment is in fact converted into U.S. dollars. If the foreign currency is converted into U.S. dollars on the date of receipt by the depositary, a U.S. holder generally should not be required to recognize foreign currency gain or loss in respect of the dividend. If the foreign currency received is not converted into U.S. dollars on the date of receipt, a U.S. holder will have a basis in the foreign currency equal to its U.S. dollar value on the date of receipt. Any gain or loss on a subsequent conversion or other disposition of the foreign currency will be treated as ordinary income or loss, and will generally be income or loss from sources within the United States for foreign tax credit limitation purposes.
Sales or Other Taxable Dispositions
A U.S. holder will generally recognize a gain or loss for U.S. federal income tax purposes upon the sale or other disposition of ADSs in an amount equal to the difference between the U.S. dollar value of the amount realized from such sale or other disposition and the U.S. holder’s tax basis in such ADSs. Subject to the discussion below under “—PFIC Rules,” such gain or loss generally will be capital gain or loss. Capital gains of individuals and certain other non-corporate U.S. holders recognized on the sale or other disposition of ADSs held for more than one year are generally eligible for a reduced rate of taxation. The gain or loss will generally be income or loss from sources within the United States for foreign tax credit limitation purposes. The deductibility of capital losses is subject to limitations.
A U.S. holder’s adjusted tax basis in the ADSs will generally equal the U.S. dollar value of the purchase price for the ADSs, based on the prevailing exchange rate on the date of such purchase. The amount realized on a disposition of the ADSs in exchange for foreign currency, will generally equal the U.S. dollar value of such currency translated at the spot exchange rate in effect on the date of the disposition. If, however, the ADSs are treated as traded on an “established securities market” for U.S. federal income tax purposes, a cash basis U.S. holder (or, if it elects, an accrual basis U.S. holder) will determine the U.S. dollar value of the purchase price for the ADSs or the amount realized on a disposition of the ADSs in exchange for non-U.S. currency, as the case may be, by translating the amount paid or received at the spot exchange rate in effect on the settlement date of the purchase or disposition, as the case may be. Any such election by an accrual basis U.S. holder must be applied consistently from year to year and cannot be changed without the consent of the IRS. A U.S. holder’s tax basis in any non-U.S. currency received on a disposition of the ADSs will generally equal the U.S. dollar value of such currency on the date of receipt. Any gain or loss realized by a U.S. holder on a subsequent conversion or other disposition of the non-U.S. dollar currency will generally be foreign currency gain or loss and treated as U.S. source ordinary income or loss. U.S. holders should consult their tax advisors regarding the sale or other taxable disposition of the ADSs under their particular circumstances.
PFIC Rules
A non-U.S. corporation will be classified as a passive foreign investment company, or PFIC, for U.S. federal income tax purposes for any taxable year, if either (i) 75% or more of its gross income for such year consists of certain types of “passive” income or (ii) 50% or more of the value of its assets (determined on the basis of a quarterly average) during such year produce or are held for the production of passive income. Passive income generally includes dividends, interest, royalties, rents, annuities, net gains from the sale or exchange of property producing such income and net foreign currency gains. In addition, a non-U.S. corporation will be treated as owning its proportionate share of the assets and earning its proportionate share of the income of any other corporation in which it owns, directly or indirectly, more than 25% (by value) of the stock.
The determination of whether we are a PFIC is a fact-intensive determination that must be made on an annual basis applying principles and methodologies that are in some circumstances unclear. Based on the composition of our income and valuation of our assets, we do not believe that we should be treated as a PFIC for the 2023 taxable year. However, a separate determination must be made after the close of each taxable year as to whether we are a PFIC for that year. As a result, our PFIC status may change in a future taxable year. In particular, the total value of our assets for purposes of the asset test generally will be calculated taking into account the market price of our ADSs or ordinary shares, which may fluctuate considerably. Fluctuations in the market price of the ADSs and ordinary shares may result in our being a PFIC for any taxable year. Even if we determine that we are not a PFIC for a taxable year, there can be no assurance that the U.S. Internal Revenue Services ("IRS") or courts will agree with our conclusion and that the IRS would not successfully challenge our position.
If we are classified as a PFIC for any taxable year during which a U.S. holder holds ADSs, unless the U.S. holder makes a “mark-to-market” election or a “qualified electing fund” election (as described below), the U.S. holder will generally be subject to special tax rules that have a generally penalizing effect, regardless of whether we remain a PFIC, on (i) any excess distribution that we make to the U.S. holder (which generally means any distribution paid during a taxable year to a U.S. holder
145
that is greater than 125% of the average annual distributions paid in the three preceding taxable years or, if shorter, the U.S. holder’s holding period for its ADSs), and (ii) any gain realized on the sale or other disposition of its ADSs.
If we are a PFIC for any taxable year during which a U.S. holder holds ADSs and any of our subsidiaries is also a PFIC, such U.S. holder will be treated as owning a proportionate amount (by value) of the shares of the lower-tier PFIC. U.S. holders should consult their tax advisors regarding the application of the PFIC rules to any of our subsidiaries.
If we were to be classified as a PFIC, a U.S. holder may make a mark-to-market election with respect to its ADSs provided the ADSs are treated as regularly traded on a qualified exchange or other market as defined in applicable Treasury Regulations. The Nasdaq Stock Market is a qualified exchange for this purpose and, consequently, if the ADSs are regularly traded, the mark-to-market election will be available to a U.S. holder if we are classified as a PFIC. Because, as a technical matter, a mark-to-market election cannot be made for any lower-tier PFICs that we may own, however, a U.S. holder may continue to be subject to the PFIC rules with respect to such holder’s indirect interest in any investments held by us that are treated as an equity interest in a PFIC for U.S. federal income tax purposes. U.S. holders should consult their tax advisors regarding the potential availability and consequences of a mark-to-market election in case we are classified as a PFIC in any taxable year.
[We do not intend to make available the information necessary for a U.S. holder to make a “qualified electing fund” election.] If a U.S. holder makes an effective qualified electing fund election, the U.S. holder will be required to include in gross income each year, whether or not we make distributions, as capital gains, such U.S. holder’s pro rata share of our net capital gains and, as ordinary income, such U.S. holder’s pro rata share of our earnings in excess of our net capital gains. However, a U.S. holder can only make a qualified electing fund election with respect to ADSs in a PFIC if such company agrees to furnish such U.S. holder with certain tax information annually. We cannot offer any assurance that we will make available the information necessary for a U.S. holder to make a “qualified electing fund” election.
If a U.S. holder holds ADSs in any year in which we are treated as a PFIC with respect to such U.S. holder, such U.S. holder will generally be required to file IRS Form 8621 and such other forms as may be required by the U.S. Treasury Department.
U.S. holders should consult their own tax advisors regarding the application of the PFIC rules to their investment in our ADSs and the elections discussed above.
Information with Respect to Foreign Financial Assets
Owners of “specified foreign financial assets” with an aggregate value in excess of US$50,000 (and in some circumstances, a higher threshold) may be required to file IRS Form 8938 (Statement of Specified Foreign Financial Assets) with respect to such assets on their tax returns. “Specified foreign financial assets” may include financial accounts maintained by foreign financial institutions, as well as any of the following, if they are held for investment and not held in accounts maintained by financial institutions: (i) stocks and securities issued by non-U.S. persons, (ii) financial instruments and contracts held for investment that have non-U.S. issuers or counterparties, and (iii) interests in foreign entities. U.S. holders are urged to consult their tax advisors regarding the application of these rules to their ownership of the ADSs.
F.Dividends and Paying Agents
Not applicable.
G.Statement by Experts
Not applicable.
H.Documents on Display
We are subject to certain reporting requirements of the Exchange Act. As a “foreign private issuer”, we are exempt from the rules under the Exchange Act prescribing certain disclosure and procedural requirements for proxy solicitations, and our officers, directors and principal shareholders are exempt from the reporting and “short-swing” profit recovery provisions contained in Section 16 of the Exchange Act, with respect to their purchases and sales of shares. In addition, we are not required to file reports and Financial Statements with the SEC as frequently or as promptly as companies that are not foreign private issuers whose securities are registered under the Exchange Act. However, we are required to file with the SEC, within 4 months after the end of each fiscal year, an Annual Report on Form 20-F containing Financial Statements audited by an independent accounting firm and interactive data comprising Financial Statements in extensible business reporting language. We publish unaudited interim financial information after the end of each quarter. We furnish this quarterly financial information to the SEC under cover of a Form 6-K.
146
The SEC maintains a website at http://www.sec.gov that contains reports and other information regarding registrants that are required to file electronically with the SEC.
I.Subsidiary Information
Not applicable.
Item 11. Quantitative and Qualitative Disclosures about Market Risk
Market risk represents the risk that changes in market prices, such as foreign exchange rates, interest rates or equity prices, will affect the Group‘s results of operations or the value of the financial instruments held. The Group is exposed to both currency and interest rate risks.
Currency Risk
The consolidated financial statements are prepared in euros. Both revenues and expenses of the Group are incurred in euros and US dollars. Throughout the year, the Group monitors the necessity to hedge foreign exchange rates to minimize currency risk and addresses this risk by using derivative financial instruments.
The use of derivatives is subject to a Group guideline approved by the Management Board, which represents a written guideline for dealing with derivatives. In accordance with the Group's hedging policy, only highly probable future cash flows and clearly determinable receivables that can be realized within a period of twelve months are hedged. MorphoSys enters into foreign exchange option and forward exchange contracts to hedge its foreign exchange exposure arising from U.S. dollar cash flows.
The financial liabilities from future payments to Royalty Pharma are dependent on future royalty income, which is determined on the basis of sales in U.S. dollars. The transfer of assigned license revenues is settled in euros. Refer to Note 4.20 for a sensitivity analysis on the impact of a change in the foreign exchange rate.
Different foreign exchange rates and their impact on financial assets and liabilities were simulated in a sensitivity analysis to determine the effects on profit or loss. Positive amounts would increase a consolidated net profit or decrease a consolidated net loss. Negative amounts would decrease a consolidated net profit or increase a consolidated net loss.
| in million € | 2023 | 2022 | 2021 | |||||||||||||||||
| Increase of the Euro by 10% | 7.3 | 15.6 | 39.3 | |||||||||||||||||
| Decrease of the Euro by 10% | (9.3) | (19.7) | (48.0) | |||||||||||||||||
Interest Rate Risk
The Group’s risk exposure to changes in interest rates mainly relates to fixed-term deposits and corporate bonds. Changes in the general level of interest rates may lead to an increase or decrease in the fair value of these securities. The Group’s investment focus places the safety of an investment ahead of its return and the ability to plan future cash flows. Interest rate risks are limited because all securities can be liquidated within a maximum of two years and due to the mostly fixed interest rates during the term in order to ensure that planning is possible. In addition, changes in interest rates may affect the fair value of financial assets from collaborations.
Different interest rates and their effect on existing other financial assets with variable interest rates and on financial assets from collaborations were simulated in a sensitivity analysis in order to determine the effect on profit or loss. Positive amounts would increase a consolidated net profit or decrease a consolidated net loss. Negative amounts would decrease a consolidated net profit or increase a consolidated net loss.
| in million € | 2023 | 2022 | 2021 | |||||||||||||||||
| Increase of the variable Interest Rate by 0.5% | 2.4 | 2.4 | 0.8 | |||||||||||||||||
| Decrease of the variable Interest Rate by 0.5% | (2.4) | (2.4) | (0.8) | |||||||||||||||||
The Group is currently not subject to significant interest rate risks from the account payables reported on the balance sheet.
147
Item 12.Description of Securities Other than Equity Securities
A.Debt Securities
On October 16, 2020, we placed unsubordinated, unsecured convertible bonds maturing on October 16, 2025 for a nominal amount of € 325.0 million, divided into 3,250 bonds with a par value of € 100,000 each. The convertible bonds were issued at 100% of their nominal amount and carry a semi-annual coupon of 0.625% per year.
On March 30, 2023, MorphoSys repurchased outstanding convertible bonds via a modified reverse Dutch auction procedure. At the close of the modified reverse Dutch auction procedure, MorphoSys had agreed to repurchase bonds representing € 62.9 million in aggregate principal amount (approximately 19.35% of the outstanding principal amount). The purchase price per € 100,000 nominal was € 64,000. The settlement procedure finished on March 30, 2023. Following the repurchase the bonds have been cancelled and deleted from the global certificate. As of December 31, 2023, there were 2,621 convertible bonds outstanding each with a notional amount of € 100,000. Upon repurchase MorphoSys realized a gain of € 16.4 million as the difference of the carrying amount as of the date of the repurchase and the fair value for the redeemed bonds. There were no bond conversions in the most recent fiscal year.
On September 12, 2022, we drew US$ 300.0 million (€ 295.4 million) from the development funding bond agreement with Royalty Pharma (concluded in 2021). Repayment will be made at 2.2 times the amount drawn according to a fixed payment schedule within ten years and nine months after drawdown without any repayment in the first two years after drawdown. This corresponds to a nominal interest rate of 13.3%.
B.Warrants and Rights
Not applicable.
C.Other Securities
Not applicable.
D.American Depository Shares
The Bank of New York Mellon, as depositary, registers and delivers ADSs. Each ADS represents one-quarter (1/4) of a deposited share with The Bank of New York Mellon SA/N.V., as custodian for the depositary in Frankfurt. Each ADS also represents any other securities, cash or other property which may be held by the depositary. The depositary’s office at which the ADSs will be administered is located at 240 Greenwich Street, New York, New York 10286, United States. The Bank of New York Mellon’s principal executive office is located at 240 Greenwich Street, New York, New York 10286, United States.
A deposit agreement among us, the depositary and the ADS holders sets out the ADS holder rights as well as the rights and obligations of the depositary. New York law governs the deposit agreement and the ADSs. A copy of the deposit agreement is incorporated by reference as an exhibit to this annual report.
148
Fees and Expenses
| Persons depositing or withdrawing shares or ADS holders must pay: | For: | |||||||
| $5.00 (or less) per 100 ADSs (or portion of 100 ADSs) | Issuance of ADSs, including issuances resulting from a distribution of shares or rights or other property | |||||||
| Cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates | ||||||||
| $0.5 (or less) per ADS | Any cash distribution to ADS holders | |||||||
| A fee equivalent to the fee that would be payable if securities distributed to you had been shares and the shares had been deposited for issuance of ADSs | Distribution of securities distributed to holders of deposited securities (including rights) that are distributed by the depositary to ADS holders | |||||||
| $0.5 (or less) per ADS per calendar year | Depositary services | |||||||
| Registration or transfer fees | Transfer and registration of shares on our share register to or from the name of the depositary or its agent when you deposit or withdraw shares | |||||||
| Expenses of the depositary | Cable and facsimile transmissions (when expressly provided in the deposit agreement) | |||||||
| Converting foreign currency to U.S. dollars | ||||||||
| Taxes and other governmental charges the depositary or the custodian has to pay on any ADSs or shares underlying ADSs, such as stock transfer taxes, stamp duty or withholding taxes | As necessary | |||||||
| Any charges incurred by the depositary or its agents for servicing the deposited securities | As necessary | |||||||
The depositary collects its fees for delivery and surrender of ADSs directly from investors depositing ordinary shares or surrendering ADSs for the purpose of withdrawal or from intermediaries acting for them. The depositary collects fees for making distributions to investors by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees. The depositary may collect its annual fee for depositary services by deduction from cash distributions or by directly billing investors or by charging the book-entry system accounts of participants acting for them. The depositary may collect any of its fees by deduction from any cash distribution payable (or by selling a portion of securities or other property distributable) to ADS holders that are obligated to pay those fees. The depositary may generally refuse to provide fee-attracting services until its fees for those services are paid.
From time to time, the depositary may make payments to us to reimburse us for costs and expenses generally arising out of establishment and maintenance of the ADS program, waive fees and expenses for services provided to us by the depositary or share revenue from the fees collected from ADS holders. In performing its duties under the deposit agreement, the depositary may use brokers, dealers, foreign currency dealers or other service providers that are owned by or affiliated with the depositary and that may earn or share fees, spreads or commissions.
The depositary may convert currency itself or through any of its affiliates and, in those cases, acts as principal for its own account and not as agent, advisor, broker or fiduciary on behalf of any other person and earns revenue, including, without limitation, transaction spreads, that it will retain for its own account. The revenue is based on, among other things, the difference between the exchange rate assigned to the currency conversion made under the deposit agreement and the rate that the depositary or its affiliate receives when buying or selling foreign currency for its own account. The depositary makes no representation that the exchange rate used or obtained in any currency conversion under the deposit agreement will be the most favorable rate that could be obtained at the time or that the method by which that rate will be determined will be the most favorable to ADS holders, subject to the depositary’s obligations under the deposit agreement. The methodology used to determine exchange rates used in currency conversions is available upon request.
149
PART II
Item 13. Defaults. Dividend Arrearages and Delinquencies
None.
Item 14. Material Modifications to the Rights of Security Holders and Use of Proceeds
None.
Item 15. Controls and Procedures
Item 15A. Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, has performed an evaluation of the effectiveness of our disclosure controls and procedures (as defined in Rule 13a-15(e) under the Exchange Act) as of the end of the period covered by this report, as required by Rule 13a-15(b) under the Exchange Act. Based upon this evaluation, our management, with the participation of our Chief Executive Officer and Chief Financial Officer, has concluded that, as of the end of the period covered by this annual report, our disclosure controls and procedures were effective in ensuring that the information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in by the SEC’s rules and forms, and that the information required to be disclosed by us in the reports that we file or submit under the Exchange Act is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely decisions regarding required disclosure.
Item 15B. Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rules 13a-15(f). Our internal control over financial reporting is a process designed by or under the supervision of the Chief Executive Officer and Chief Financial Officer, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of our financial statements for external reporting purposes in accordance with International Financial Reporting Standards.
As of December 31, 2023, our management conducted an assessment of the effectiveness of the Company’s internal control over financial reporting based on the criteria established in Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”). Based on this assessment, our management has determined that the Company’s internal control over financial reporting as of December 31, 2023 is effective.
Our internal control over financial reporting includes policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect transactions and dispositions of our assets; (2) provide reasonable assurances that our transactions are recorded as necessary to permit preparation of financial statements in accordance with International Financial Reporting Standards as issued by the IASB, and that our receipts and expenditures are being made only in accordance with authorizations of management; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on our financial statements.
Because of its inherent limitation, internal control over financial reporting, no matter how well designed, cannot provide absolute assurance of achieving financial reporting objectives and may not prevent or detect misstatements. Therefore, even if the internal control over financial reporting is determined to be effective it can provide only reasonable assurance with respect to financial statement preparation and presentation. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Item 15C. Attestation Report of the Registered Public Accounting Firm
The effectiveness of our internal control over financial reporting as of December 31, 2023 has been audited by PricewaterhouseCoopers GmbH Wirtschaftsprüfungsgesellschaft, an independent registered public accounting firm. Their report is included on page F-2. PricewaterhouseCoopers GmbH Wirtschaftsprüfungsgesellschaft, Frankfurt am Main, Zweigniederlassung München, is a member of the Chamber of Public Accountants (Wirtschaftsprüferkammer), Berlin, Germany.
150
Item 15D. Changes in Internal Control over Financial Reporting
We regularly review our system of internal control over financial reporting and make changes to our processes and systems to further strengthen controls and increase efficiency, while ensuring that we maintain an effective internal control environment.
There were no changes in our internal control over financial reporting (as required by Rules 13a 15(d) and 15d 15(d) of the Exchange Act) that occurred during the fiscal year ended December 31, 2023, which have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 16. [Reserved]
Item 16A. Audit Committee Financial Expert
Our board of directors has determined that Michael Brosnan is an audit committee financial expert as defined by SEC rules and has the requisite financial sophistication under the applicable rules and regulations of the Nasdaq Stock Market. Michael Brosnan is independent as such term is defined in Rule 10A-3 under the Exchange Act and under the listing standards of the Nasdaq Stock Market.
Item 16B. Code of Ethics
The Code of Conduct is available on the MorphoSys website at www.morphosys.com. It sets forth the key principles and ethical standards for how we work together to develop and deliver innovative therapies, how we protect the value and integrity of MorphoSys worldwide, and how we work with our customers, suppliers, public authorities, and other partners and stakeholders in conducting our business. The Code of Conduct applies to all employees and Management Board members (including officers and directors) of MorphoSys. Certain business partners, such as agents, consultants and vendors serve as an extension of MorphoSys. They are expected to follow the spirit of the Code of Conduct, as well as any applicable regulations and contractual provisions, when working on behalf of MorphoSys. We expect all of us to act with honesty and integrity, to comply with applicable laws and industry practices, and to exercise reasonable judgment when conducting business on behalf of MorphoSys. If we make any amendment to our Code of Conduct or grant any waivers, including any implicit waiver, from a provision of the Code of Conduct, we will disclose the nature of such amendment or waiver on our website to the extent required by the rules and regulations of the SEC.
Item 16C. Principal and Accountant Fees and Services
PricewaterhouseCoopers GmbH Wirtschaftsprüfungsgesellschaft has served as our independent registered public accounting firm for the years ending December 31, 2023 and 2022. The following table sets out the aggregate fees for professional audit services and other services rendered by PricewaterhouseCoopers and their member firms and / or affiliates in 2023 and 2022:
| Year ended December 31 (in 000' €) | 2023 | 2022 | ||||||||||||
| Audit Fees | 2,472 | 2,335 | ||||||||||||
| Fees for Other Assurance Services | 700 | 112 | ||||||||||||
| Other Fees for Other Services | 5 | 11 | ||||||||||||
| Total | 3,178 | 2,458 | ||||||||||||
Audit fees relate to the audit of the financial statements as set out in this Annual Report, certain procedures on our half year results, audit of our internal control over financial reporting and services related to our statutory and regulatory filings.
Other assurance services comprise fees in connection with the non-financial group report, services in connection with the issue of a comfort letter, as well as the audit of the content of the remuneration report.
Out of total fee, an amount of € 5k relates to a license fee for the use of a digital information platform and relate to PwC Product Sales LLC, USA and is included in other services. All remaining fees relate to PwC GmbH.
The Audit Committee has approved the audit fees and all of the fees for other assurance services and other fees for other services for the years 2023 and 2022. The Audit Committee monitors compliance with the German and U.S. rules on non-audit services provided by an independent registered public accounting firm. On a yearly basis, the Audit Committee pre-approves non-audit services performed by the independent registered public accounting firm up to a limit in line with EU regulation.
151
Item 16D. Exemptions from the Listing Standards for Audit Committees
Not applicable.
Item16E. Purchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
Item 16F. Change in Registrant's Certifying Accountant
PricewaterhouseCoopers GmbH Wirtschaftsprüfungsgesellschaft (PwC) has been the auditor of the consolidated and individual financial statements of MorphoSys AG, Planegg, without interruption since the 2011 financial year.
Due to the provisions of the German Act to Strengthen Financial Market Integrity (FISG), which came into force on July 21, 2021, the option to extend the maximum term of mandates to audit the financial statements of publicly traded corporations to up to 24 years was removed. Accordingly, the maximum term of ten years prescribed in Article 17 of the European Regulation on specific requirements regarding statutory audit of public-interest entities (EU Directive 2014/537) must be observed.
In accordance with EU Directive 2014/537 and the provisions of the FISG, the Company conducted a tender process in fiscal year 2024 – under the supervision of the Audit Committee – for the mandate to audit the consolidated and separate financial statements of MorphoSys AG and to review the condensed interim consolidated financial statements and the interim Group management report for the first six months of the fiscal year 2024.
In accordance with German stock corporation law, the Annual General Meeting of the company appoints the auditor for the consolidated and separate financial statements of the company based on the proposal of the Supervisory Board. The Audit Committee provides the Supervisory Board with a reasoned nomination based on the results of the tendering process.
Based on the submission of the Audit Committee, the Supervisory Board resolved on October 4, 2023, to propose KPMG AG Wirtschaftsprüfungsgesellschaft (KPMG) as the Company's independent registered public accounting firm for the consolidated and separate financial statements of MorphoSys AG for the fiscal year 2024, as well as the review of condensed interim financial statements for the first six months of the fiscal year ended 2024 to the Annual General Meeting that will be held in 2024. Accordingly, on October 4, 2023, the Supervisory Board dismissed PwC as the company's independent registered public accounting firm, effective immediately after the Company filed this Annual Report on Form 20-F.
The reports of PwC on the Company’s consolidated financial statements for the fiscal years ended December 31, 2023 and 2022, contained no adverse opinion or disclaimer of opinion, and were not qualified or modified as to uncertainty, audit scope or accounting principle.
During our fiscal years ended December 31, 2023 and 2022, and the subsequent interim period through the date of this filing, there have been no: (1) disagreements with PwC on any matter of accounting principles or practices, financial statement disclosure, or auditing scope or procedure, which disagreements, if not resolved to their satisfaction, would have caused them to make reference thereto in their reports on the consolidated financial statements of such years, or (2) reportable events as defined by Item 16F (a)(1)(v) of Form 20-F
We have provided PwC with a copy of this disclosure and requested that PwC furnish us with a letter addressed to the SEC stating whether or not it agrees with the above statements. The letter furnished by PwC is attached as Exhibit 15.2 to this Form 20-F.
During the fiscal years ended December 31, 2023 and 2022, and the subsequent interim period through the date of this filing, we did not consult KPMG regarding (i) the application of accounting principles to any specified transaction, either completed or proposed; (ii) the type of audit opinion that might be rendered on our financial statements, and either a written report was provided to us or oral advice was provided that KPMG concluded was an important factor considered by us in reaching a decision as to the accounting, auditing or financial reporting issue; or (iii) any matter that was either the subject of a disagreement (as defined in Item 16F(a)(1)(iv)) of Form 20-F or a reportable event (as defined in Item 16F(a)(1)(v)) of Form 20-F.
Item 16G. Corporate Governance
In general, Nasdaq Stock Market Rule 5615(a)(3) permits foreign private issuers such as us, to follow home country corporate governance practices instead of certain provisions of the Nasdaq Stock Market Rules. In addition, we also may qualify for certain exemptions under the Nasdaq Stock Market Rules as a foreign private issuer that may affect our corporate governance practices.
152
The significant differences between the corporate governance practices that we follow and those set forth in the Nasdaq Stock Market Rules are described below:
Distribution of Annual and Interim Reports. Nasdaq Listing Rule 5250(d) requires that our annual and interim reports be distributed or made available to shareholders within a reasonable period of time following filing with the SEC. Consistent with applicable rules and regulations in Germany, we do not distribute annual and interim reports automatically to shareholders. Instead, our annual and interim reports are available to shareholders on our website and delivery of printed versions thereof can be requested online. Furthermore, our annual and interim reports are also filed with the German Company Register (Unternehmensregister).
Code of Conduct. Nasdaq Listing Rule 5610 requires companies to adopt and make publicly available a code of conduct applicable to all directors, officers and employees. Although there is no requirement under German law for a company to have a code of conduct, we nevertheless have one in place applying to our Management Board and employees but not to our Supervisory Board.
Proxy Solicitation. Nasdaq Listing Rule 5620(b) requires companies that are not a limited partnership to solicit proxies and provide proxy statements for all meetings of shareholders and to provide copies of such proxy solicitation to Nasdaq. Under German law, there is no requirement for companies to solicit proxies in connection with a meeting of shareholders. Shareholders have the right to exercise their voting rights in the shareholders’ meeting through proxies appointed by them in writing. The proxies appointed by us are obligated to vote only in accordance with the instructions of the represented shareholder.
Shareholder Approval Requirements. Nasdaq Listing Rule 5635 requires companies to obtain shareholder approval before the issuance of securities to undertake any of the following transactions:
•acquiring the stock or assets of another company, where such acquisition results in the issuance of 20% or more of our outstanding share capital or voting power;
•entering into any change of control transaction;
•establishing or materially amending any equity compensation arrangement; and
•entering into any transaction other than a public offering involving the sale, issuance or potential issuance by us of shares (or securities convertible into or exercisable for shares) equal to 20% or more of our outstanding share capital or 20% or more of the voting power outstanding before the issuance for less than the greater of book or market value of the stock.
Consistent with the German Stock Corporation Act (Aktiengesetz), approval by the shareholders’ meeting is generally required for the issuance of any shares as well as any securities granting the respective holder the right to acquire shares (including options and convertibles).
Item 16H. Mine Safety Disclosure
Not applicable.
Item 16I. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable.
Item 16J. Insider Trading Policies
Pursuant to applicable SEC transition guidance, the disclosure required by Item 16J will be applicable to the Company from the fiscal year ending December 31, 2023.
Item 16K. Cybersecurity
Risk Management and Strategy
MorphoSys has a corporate risk management program in place that is designed to identify, assess, manage, and respond to risks affecting our enterprise. This program accounts for risks related to cybersecurity, and as part of managing cybersecurity risk, we conduct a cybersecurity risk assessment on an annual basis. This assessment follows a structured approach and is informed by the framework provided by the National Institute of Standards and Technology (NIST).
153
Supporting our cybersecurity risk management efforts are policies and procedures addressing information security, mandatory cybersecurity trainings for employees, and penetration testing of our systems that are periodically performed by both internal and external resources.
Furthermore, we recently engaged a third-party managed detection and response vendor to perform security operations on our behalf. This vendor is tasked with detecting and responding to cybersecurity threats to both our systems and applications and those managed by third-party providers.
Additionally, the Company performs assessments of certain third-party vendors prior to establishing a business relationship as part of our efforts to determine whether such vendors demonstrate appropriate commitments related to data security, availability, and confidentiality. This process is calibrated to the risk level associated with each vendor. Our vendor evaluation procedures include, as appropriate, the completion of a vendor security questionnaire.
Governance
With over 20 years’ experience as an information technology professional, the Company’s Head of Global IT is responsible for the Company’s cybersecurity program. The program is developed by the Company’s Head of IT Security with the Head of Global IT’s approval, and is informed by the cybersecurity risk assessment. The Head of Global IT reports to Head of Technical Operations, who provides strategic guidance as necessary on behalf of the Company’s Management Board. Additionally, the Head of Global IT provides updates to the Executive Committee, comprising the Company’ CEO and CFO along with other senior management members, on matters related to cybersecurity as needed.
The Company has established a Computer Emergency Response Team (CERT) that is charged with addressing cybersecurity incidents pursuant to the Company’s Cyber Security Incident Response Plan. The Head of IT is responsible for reporting cybersecurity incidents to key decision makers on the Executive Committee and Management Board.
At the Supervisory Board level, the Audit Committee is tasked with exercising oversight of the Company’s cybersecurity program. To permit the Audit Committee to perform its oversight duties, the Head of Global IT provides a structured reporting to the Audit Committee on a semi-annual basis. This reporting includes updates resulting from the cybersecurity risk assessment, the status of forecasted and already implemented security controls, as well as cybersecurity incidents (if any) in the reporting period. The chairman of the Audit Committee provides the Supervisory Board with a summary of the discussions held within the Audit Committee. Management considers the advice of the Audit Committee and/or Supervisory Board when updating the Company’s cybersecurity strategy and the respective tactical and operational execution of the cybersecurity program.
PART III
Item 17.Financial Statements
Not applicable.
Item 18.Financial Statements
Item 19.Exhibits
154
INDEX TO FINANCIAL STATEMENTS
F-1
Report of Independent Registered Public Accounting Firm
Opinions on the Financial Statements and Internal Control over Financial Reporting
We have audited the accompanying consolidated balance sheet of MorphoSys AG and its subsidiaries (the “Company”) as of December 31, 2023 and 2022, and the related consolidated statements of profit or loss, comprehensive income, changes in stockholders’ equity and cash flows for each of the three years in the period ended December 31, 2023, including the related notes (collectively referred to as the “consolidated financial statements”). We also have audited the Company’s internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO).
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2023 in conformity with International Financial Reporting Standards as issued by the International Accounting Standards Board and International Financial Reporting Standards as adopted by the European Union. Also in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control - Integrated Framework (2013) issued by the COSO.
Basis for Opinions
The Company's management is responsible for these consolidated financial statements, for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting, included in Management's Annual Report on Internal Control over Financial Reporting appearing under Item 15. Our responsibility is to express opinions on the Company’s consolidated financial statements and on the Company's internal control over financial reporting based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud, and whether effective internal control over financial reporting was maintained in all material respects.
Our audits of the consolidated financial statements included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
Definition and Limitations of Internal Control over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
F-2
Critical Audit Matters
The critical audit matters communicated below are matters arising from the current period audit of the consolidated financial statements that were communicated or required to be communicated to the audit committee and that (i) relate to accounts or disclosures that are material to the consolidated financial statements and (ii) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
Subsequent measurement of the financial liability arising from the Incyte collaboration and license agreement
As described in Notes 2.6.1 and 4.19 to the consolidated financial statements, as of December 31, 2023, the Company has recorded a current and non-current financial liability of € 114.4 million related to the collaboration and license agreement with Incyte Corporation, USA (hereinafter “Incyte”). The current and non-current financial liability represent Incyte’s prepaid entitlement to future profit sharing on sales of Monjuvi® (tafasitamab-cxix) in the USA. The financial liability is subsequently measured at amortized cost using the effective interest method. The basis for the valuation is the corporate planning and its shared profits and losses thereof in connection with the commercialization activities of MorphoSys and Incyte in the USA for the years ahead. Management’s significant assumptions include the forecasted number of patients and the expectations on selling price and costs associated with the sale of Monjuvi® (tafasitamab-cxix).
The principal considerations for our determination that performing procedures relating to the subsequent measurement of the financial liability from the Incyte collaboration and license agreement is a critical audit matter are that the outcome of the subsequent measurement of the financial liability is dependent to a large extent on the assumptions made by management with respect to the future cash outflows and inflows, the forecasted number of patients, the expectations on selling price and the costs associated with the sale of Monjuvi® (tafasitamab-cxix) and is therefore subject to significant judgement by management and considerable uncertainty. This in turn led to a high degree of auditor judgment, subjectivity, and effort in performing procedures and evaluating audit evidence related to management’s cash flow projections and significant assumptions regarding the forecasted number of patients, the expectations on selling price and the costs associated with the sale of Monjuvi® (tafasitamab-cxix). In addition, the audit effort involved the use of professionals with specialized skill and knowledge.
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included testing the effectiveness of controls relating to the subsequent measurement of the financial liability from the Incyte collaboration and license agreement. Our procedures also included, among others, testing management's process for the subsequent measurement of the financial liability, including evaluating the reasonableness of management’s significant assumptions regarding the cash outflows and inflows, the forecasted number of patients, the expectations on selling price and the costs associated with the sale of Monjuvi® (tafasitamab-cxix), and testing the completeness, accuracy, and relevance of underlying data used in the model. Professionals with specialized skill and knowledge were involved to assist in evaluating the reasonableness of the assumptions.
Recoverability of the goodwill of the group of CGUs Constellation and the intangible asset pelabresib
As described in Notes 2.6.9, 4.10 and 4.11 to the consolidated financial statements, as of December 31, 2023, the goodwill of € 342.3 million and the pelabresib intangible asset not yet available for use of € 766.7 million related to the acquisition of Constellation were subject to an annual impairment test. The recoverable amount of the group of cash-generating units (CGUs) Constellation and the pelabresib intangible asset not yet available for use was determined on the basis of value-in-use calculations. The cash flow projections included expected payments from the commercialization of pelabresib and other compounds, the cash outflows for anticipated research and development, and the costs for pelabresib’s and the other compounds' commercialization. The calculation showed that the value-in-use was higher than the carrying amount of the CGUs Constellation and the pelabresib intangible asset not available for use.
The principal considerations for our determination that performing procedures relating to the assessment of impairment of the goodwill related to the group of CGUs Constellation and the pelabresib intangible asset not available for use is a critical audit matter are that the outcome of the impairment test of the goodwill of the group of CGUs Constellation and the pelabresib intangible asset not available for use is dependent to a large extent on the assumptions made by management with respect to the future cash flows, the expected payments from the commercialization of pelabresib and other compounds as well as the costs for commercialization of pelabresib and other compounds, the forecasted number of patients, the expectation on selling price, the probability of successful product development and the discount rate and is therefore subject to significant judgement by management and considerable uncertainty. This in turn led to a high degree of auditor judgment, subjectivity, and effort in performing procedures and evaluating audit evidence related to management’s cash flow projections and significant assumptions regarding the forecasted number of patients, the expectation on selling price, the probability of successful product development and the discount rate. In addition, the audit effort involved the use of professionals with specialized skill and knowledge.
F-3
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included testing the effectiveness of controls relating to the assessment of impairment of the goodwill of the group of CGUs Constellation and the pelabresib intangible asset not available for use. Our procedures also included, among others, testing management’s process for determining the recoverable amounts, evaluating the completeness, accuracy and relevance of the underlying data used in the models and assessing the reasonableness of the key assumptions used by management, relating to the forecasted number of patients, the expectation on selling price, the probability of successful product development and the discount rate. Professionals with specialized skill and knowledge were involved to assist in evaluating the reasonableness of the assumptions.
Subsequent measurement of the financial liabilities arising from the agreements with Royalty Pharma on the sale of future license income and revenues
As described in Notes 2.6.1 and 4.20 to the consolidated financial statements, as of December 31, 2023, financial liabilities amounting to € 1,058.3 million were recognized due to the royalty purchase agreement and the revenue participation agreement with Royalty Pharma plc, USA, and Royalty Pharma USA Inc., USA, (hereinafter jointly “Royalty Pharma”) and Constellation. The financial liabilities represent Royalty Pharma’s right to receive certain future license income in the form of royalties of Tremfya and future revenues from sales of the product candidates pelabresib and tulmimetostat. The planning assumptions are influenced by estimates and mainly relate to the probability of successful product development, the expected license income and revenues from sales of Tremfya, pelabresib and tulmimetostat. Revenues are influenced by variable factors such as forecasted number of patients and the expectations on selling price. The financial liabilities are subsequently measured at amortized cost using the effective interest method.
The principal considerations for our determination that performing procedures relating to the subsequent measurement of the financial liabilities from the agreements with Royalty Pharma is a critical audit matter are that the outcome of the subsequent measurement of the financial liabilities is dependent to a large extent on the assumptions made by management with respect to the future license income in the form of royalties of Tremfya and future revenues from sales of pelabresib and tulmimetostat, including i) forecasted number of patients, ii) expectations on selling price, iii) probability of successful product development and is therefore subject to significant judgement by management and considerable uncertainty. This in turn led to a high degree of auditor judgment, subjectivity, and effort in performing procedures and evaluating audit evidence related to management’s cash flow projections and significant assumptions regarding the probability of successful product development, the expected license income and revenues from sales of Tremfya, pelabresib and tulmimetostat, forecasted number of patients and expectations on selling price. In addition, the audit effort involved the use of professionals with specialized skill and knowledge.
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included testing the effectiveness of controls relating to the measurement of the financial liabilities arising from the agreements with Royalty Pharma. Our procedures also included, among others, testing management's process for the subsequent measurement of the financial liabilities evaluating the completeness, accuracy and relevance of the underlying data used in the models and assessing the reasonableness of the key assumptions used by management, relating to the probability of successful product development, the expected license income and revenues from sales of Tremfya, pelabresib and tulmimetostat, forecasted number of patients and expectations on selling price. Professionals with specialized skill and knowledge were involved to assist in evaluating the reasonableness of the assumptions.
March 12, 2024
Wirtschaftsprüfungsgesellschaft
/s/Susanne Riedel /s/Sebastian Stroner
Wirtschaftsprüferin Wirtschaftsprüfer
(German Public Auditor) (German Public Auditor)
We have served as the Company’s auditor since 2011.
F-4
MorphoSys-Group:
Consolidated Financial Statements
F-8 | |||||||||||
F-8 | |||||||||||
F-9 | |||||||||||
F-11 | |||||||||||
F-12 | |||||||||||
F-14 | |||||||||||
F-14 | |||||||||||
F-14 | |||||||||||
F-14 | |||||||||||
F-14 | |||||||||||
F-14 | |||||||||||
F-15 | |||||||||||
F-15 | |||||||||||
F-16 | |||||||||||
F-17 | |||||||||||
F-18 | |||||||||||
F-18 | |||||||||||
F-20 | |||||||||||
F-20 | |||||||||||
F-21 | |||||||||||
F-21 | |||||||||||
F-21 | |||||||||||
F-22 | |||||||||||
F-24 | |||||||||||
F-24 | |||||||||||
F-25 | |||||||||||
F-25 | |||||||||||
F-25 | |||||||||||
F-26 | |||||||||||
F-27 | |||||||||||
F-27 | |||||||||||
F-27 | |||||||||||
F-28 | |||||||||||
F-28 | |||||||||||
F-28 | |||||||||||
F-28 | |||||||||||
F-29 | |||||||||||
F-29 | |||||||||||
F-30 | |||||||||||
F-30 | |||||||||||
F-30 | |||||||||||
F-30 | |||||||||||
F-31 | |||||||||||
F-5
F-31 | |||||||||||
F-31 | |||||||||||
F-31 | |||||||||||
F-32 | |||||||||||
F-35 | |||||||||||
F-35 | |||||||||||
F-35 | |||||||||||
F-36 | |||||||||||
F-36 | |||||||||||
F-37 | |||||||||||
F-37 | |||||||||||
F-37 | |||||||||||
F-38 | |||||||||||
F-39 | |||||||||||
F-40 | |||||||||||
F-41 | |||||||||||
F-43 | |||||||||||
F-43 | |||||||||||
F-45 | |||||||||||
F-45 | |||||||||||
F-46 | |||||||||||
F-46 | |||||||||||
F-46 | |||||||||||
F-46 | |||||||||||
F-47 | |||||||||||
F-49 | |||||||||||
F-51 | |||||||||||
F-51 | |||||||||||
F-52 | |||||||||||
F-52 | |||||||||||
F-52 | |||||||||||
F-53 | |||||||||||
F-53 | |||||||||||
F-53 | |||||||||||
F-53 | |||||||||||
F-53 | |||||||||||
F-53 | |||||||||||
F-56 | |||||||||||
F-57 | |||||||||||
F-60 | |||||||||||
F-65 | |||||||||||
F-69 | |||||||||||
F-69 | |||||||||||
F-69 | |||||||||||
F-70 | |||||||||||
F-75 | |||||||||||
F-75 | |||||||||||
F-6
F-7
Consolidated Statement of Profit or Loss (IFRS)
| in € | Note | 2023 | 2022 | 2021 | ||||||||||||||||||||||
| Product Sales | ||||||||||||||||||||||||||
| Royalties | ||||||||||||||||||||||||||
| Licenses, Milestones and Other | ||||||||||||||||||||||||||
| Revenues | 2.5.1, 3.1 | |||||||||||||||||||||||||
| Cost of Sales | 2.5.2, 3.2 | ( | ( | ( | ||||||||||||||||||||||
| Gross Profit | ||||||||||||||||||||||||||
| Operating Expenses | ||||||||||||||||||||||||||
| Research and Development | 2.5.3, 3.3.1 | ( | ( | ( | ||||||||||||||||||||||
| Selling | 2.5.3, 3.3.2 | ( | ( | ( | ||||||||||||||||||||||
| General and Administrative | 2.5.3, 3.3.3 | ( | ( | ( | ||||||||||||||||||||||
| Impairment of Goodwill | 2.6.9, 3.3.5, 4.11 | ( | ( | |||||||||||||||||||||||
| Total Operating Expenses | ( | ( | ( | |||||||||||||||||||||||
| Operating Profit / (Loss) | ( | ( | ( | |||||||||||||||||||||||
| Other Income | 3.4 | |||||||||||||||||||||||||
| Other Expenses | 3.4 | ( | ( | ( | ||||||||||||||||||||||
| Finance Income | 3.4 | |||||||||||||||||||||||||
| Finance Expenses | 3.4 | ( | ( | ( | ||||||||||||||||||||||
| Income from Reversals of Impairment Losses / (Impairment Losses) on Financial Assets | 6.4.1 | ( | ||||||||||||||||||||||||
| Share of Loss of Associates accounted for using the Equity Method | 2.2.2, 4.12 | ( | ( | |||||||||||||||||||||||
| Income Tax Benefit / (Expenses) | 2.5.4, 3.5 | ( | ||||||||||||||||||||||||
| Consolidated Net Profit / (Loss) | ( | ( | ( | |||||||||||||||||||||||
| Earnings per Share, Basic and Diluted (in €) | 2.5.5, 3.6 | ( | ( | ( | ||||||||||||||||||||||
| Shares Used in Computing Earnings per Share, Basic and Diluted | 2.5.5, 3.6 | |||||||||||||||||||||||||
The Notes are an integral part of these consolidated financial statements.
Consolidated Statement of Comprehensive Income (IFRS)
| in € | 2023 | 2022 | 2021 | |||||||||||||||||
| Consolidated Net Profit / (Loss) | ( | ( | ( | |||||||||||||||||
| Items that will not be reclassified to Profit or Loss | ||||||||||||||||||||
| Change in Fair Value of Shares through Other Comprehensive Income | ||||||||||||||||||||
| Items that may be reclassified to Profit or Loss | ||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | |||||||||||||||||||
| Other Comprehensive Income | ( | |||||||||||||||||||
| Total Comprehensive Income | ( | ( | ( | |||||||||||||||||
The Notes are an integral part of these consolidated financial statements.
F-8
Consolidated Balance Sheet (IFRS)
| in € | Note | 12/31/2023 | 12/31/2022 | |||||||||||||||||
| ASSETS | ||||||||||||||||||||
| Current Assets | ||||||||||||||||||||
| Cash and Cash Equivalents | 2.6.1, 4.1 | |||||||||||||||||||
| Other Financial Assets | 2.6.1, 4.2 | |||||||||||||||||||
| Accounts Receivable | 2.6.1, 4.3 | |||||||||||||||||||
| Financial Assets from Collaborations | 2.6.1, 4.19 | |||||||||||||||||||
| Income Tax Receivables | 2.6.2, 4.4 | |||||||||||||||||||
| Other Receivables | 4.5 | |||||||||||||||||||
| Inventories | 2.6.3, 4.6 | |||||||||||||||||||
| Prepaid Expenses and Other Assets | 2.6.4, 4.7 | |||||||||||||||||||
| Total Current Assets | ||||||||||||||||||||
| Non-Current Assets | ||||||||||||||||||||
| Property, Plant and Equipment | 2.6.5, 4.8 | |||||||||||||||||||
| Right-of-Use Assets | 2.6.6, 4.9 | |||||||||||||||||||
| Intangible Assets | 2.6.7, 4.10 | |||||||||||||||||||
| Goodwill | 2.6.8, 4.11 | |||||||||||||||||||
| Other Financial Assets | 2.6.1, 4.2 | |||||||||||||||||||
| Investment in Associates | 2.2.2, 4.12 | |||||||||||||||||||
| Prepaid Expenses and Other Assets | 2.6.4, 4.7 | |||||||||||||||||||
| Total Non-Current Assets | ||||||||||||||||||||
| TOTAL ASSETS | ||||||||||||||||||||
The Notes are an integral part of these consolidated financial statements.
F-9
| in € | Note | 12/31/2023 | 12/31/2022 | |||||||||||||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||||||||||||||
| Current Liabilities | ||||||||||||||||||||
| Accounts Payable and Accruals | 2.6.1, 2.6.10, 4.14 | |||||||||||||||||||
| Lease Liabilities | 2.6.6, 4.9 | |||||||||||||||||||
| Tax Liabilities | 2.6.12, 4.15 | |||||||||||||||||||
| Provisions | 2.6.10, 4.15 | |||||||||||||||||||
| Contract Liability | 2.6.11, 4.16 | |||||||||||||||||||
| Bonds | 2.6.1, 4.18 | |||||||||||||||||||
| Financial Liabilities from Collaborations | 2.6.1, 4.19 | |||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | 2.6.1, 4.20 | |||||||||||||||||||
| Total Current Liabilities | ||||||||||||||||||||
| Non-Current Liabilities | ||||||||||||||||||||
| Lease Liabilities | 2.6.6, 4.9 | |||||||||||||||||||
| Provisions | 2.6.10, 4.15 | |||||||||||||||||||
| Deferred Tax Liability | 2.6.13, 3.5, 4.17 | |||||||||||||||||||
| Bonds | 2.6.1, 4.18 | |||||||||||||||||||
| Financial Liabilities from Collaborations | 2.6.1, 4.19 | |||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | 2.6.1, 4.20 | |||||||||||||||||||
| Total Non-Current Liabilities | ||||||||||||||||||||
| Total Liabilities | ||||||||||||||||||||
| Stockholders’ Equity | ||||||||||||||||||||
| Common Stock | 2.6.14, 4.21.1 | |||||||||||||||||||
Treasury Stock ( | 2.6.14, 4.21.4 | ( | ( | |||||||||||||||||
| Additional Paid-in Capital | 2.6.14, 4.21.5 | |||||||||||||||||||
| Other Comprehensive Income Reserve | 2.6.14, 4.21.6 | |||||||||||||||||||
| Accumulated Deficit | 2.6.14, 4.21.7 | ( | ( | |||||||||||||||||
| Total Stockholders’ Equity | ||||||||||||||||||||
| TOTAL LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||||||||||||||
The Notes are an integral part of these consolidated financial statements.
F-10
Consolidated Statement of Changes in Stockholders’ Equity (IFRS)
| Note | Common Stock | Treasury Stock | Additional Paid-in Capital | Other Comprehensive Income Reserve | Accumulated Deficit | Total Stockholders' Equity | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | € | Shares | € | € | € | € | € | |||||||||||||||||||||||||||||||||||||||||||||||||
| Balance as of January 1, 2021 | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Capital Increase, Net of Issuance Cost | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expenses through Share-based Payment Transactions and Issue of Convertible Instruments | 5.1 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Adjustment Balance Carryforward | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of Stock Options Issued | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transfer of Treasury Stock for Long-Term Incentive Programs | 0 | 0 | ( | ( | 0 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Reserves: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change in Fair Value of Shares through Other Comprehensive Income | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Consolidated Net Loss | 0 | 0 | 0 | 0 | 0 | 0 | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Total Comprehensive Income | 0 | 0 | 0 | 0 | 0 | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Balance as of December 31, 2021 | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance as of January 1, 2022 | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Capital Increase, Net of Issuance Cost | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Expenses through Share-based Payment Transactions and Issue of Convertible Instruments | 5.1 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Adjustment Balance Carryforward | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of Stock Options Issued | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Transfer of Treasury Stock for Long-Term Incentive Programs | 5.1 | 0 | 0 | ( | ( | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Reserves: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change in Fair Value of Shares through Other Comprehensive Income | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Consolidated Net Loss | 0 | 0 | 0 | 0 | 0 | 0 | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Total Comprehensive Income | 0 | 0 | 0 | 0 | 0 | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Balance as of December 31, 2022 | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance as of January 1, 2023 | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Capital Increase, Net of Issuance Cost | 2.6.14, 4.21.1, 4.21.5 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Expenses through Share-based Payment Transactions and Issue of Convertible Instruments | 5.1 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Adjustment Balance Carryforward | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise of Stock Options Issued | 2.6.14, 5.1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Transfer of Treasury Stock for Long-Term Incentive Programs | 2.6.14, 5.1 | 0 | 0 | ( | ( | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Gain on the Disposal of an Investment | 4.21.5 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Reserves: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change in Fair Value of Shares through Other Comprehensive Income | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | 4.21.6 | 0 | 0 | 0 | 0 | 0 | ( | 0 | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Consolidated Net Loss | 4.21.7 | 0 | 0 | 0 | 0 | 0 | 0 | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Total Comprehensive Income | 0 | 0 | 0 | 0 | 0 | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Balance as of December 31, 2023 | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
The Notes are an integral part of these consolidated financial statements.
F-11
Consolidated Statement of Cash Flows (IFRS)
| in € | Note | 2023 | 2022 | 2021 | ||||||||||||||||||||||
| Operating Activities: | ||||||||||||||||||||||||||
| Consolidated Net Profit / (Loss) | ( | ( | ( | |||||||||||||||||||||||
| Adjustments to Reconcile Consolidated Net Profit / (Loss) to Net Cash Provided by / (Used in) Operating Activities: | ||||||||||||||||||||||||||
| Impairments of Assets | 4.7, 4.8, 4.10, 4.11 | |||||||||||||||||||||||||
| Depreciation and Amortization of Tangible and Intangible Assets and of Right-of-Use Assets | 4.8, 4.9, 4.10 | |||||||||||||||||||||||||
| Net (Gain) / Loss of Other Financial Assets | 4.2 | ( | ( | ( | ||||||||||||||||||||||
| (Income) from Reversals of Impairments / Impairments on Financial Assets | 4.1, 4.2, 6.4.1 | ( | ( | |||||||||||||||||||||||
| Net (Gain) / Loss on Derivative Financial Instruments | ( | |||||||||||||||||||||||||
| Non Cash Effective Net Change in Financial Assets / Liabilities from Collaborations | 4.19 | ( | ( | ( | ||||||||||||||||||||||
| Non Cash Effective Net Change in Financial Liabilities from Future Payments to Royalty Pharma | 4.20 | ( | ( | |||||||||||||||||||||||
| Gain on Repurchase and interest expense from Convertible Bond | 4.18 | ( | ||||||||||||||||||||||||
| Share-based Payment | 3.3.4, 5.1 | |||||||||||||||||||||||||
| Non Cash Income from Capitalization of Investment in Associates | 4.12 | ( | ||||||||||||||||||||||||
| Cash Income from Sales of Shares of Investment in Associates | 4.5 | ( | ||||||||||||||||||||||||
| Share of Loss of Associates accounted for using the Equity Method | 4.12 | |||||||||||||||||||||||||
| Change in Fair Value Anti-Dilution Asset | ||||||||||||||||||||||||||
| Other Cash and Non-Cash Expenses (+) / Income (-) | ||||||||||||||||||||||||||
| Income Tax (Benefit) / Expenses | 3.5 | ( | ( | |||||||||||||||||||||||
| Changes in Operating Assets and Liabilities: | ||||||||||||||||||||||||||
| Accounts Receivable | 4.3 | ( | ||||||||||||||||||||||||
| Income Tax Receivables, Other Receivables, Inventories and Prepaid Expenses and Other Assets | 4.4, 4.5, 4.6, 4.7 | ( | ( | ( | ||||||||||||||||||||||
| Accounts Payable and Accruals, Lease Liabilities, Tax Liabilities and Provisions | 4.9, 4.14, 4.15 | ( | ( | ( | ||||||||||||||||||||||
| Contract Liability | 4.16 | ( | ( | |||||||||||||||||||||||
| Income Taxes Paid (-) / Received (+) | ( | ( | ( | |||||||||||||||||||||||
| Net Cash Provided by / (Used in) Operating Activities | ( | ( | ( | |||||||||||||||||||||||
The Notes are an integral part of these consolidated financial statements.
F-12
| in € | Note | 2023 | 2022 | 2021 | ||||||||||||||||||||||
| Investing Activities: | ||||||||||||||||||||||||||
| Cash Payments to Acquire Other Financial Assets | ( | ( | ( | |||||||||||||||||||||||
| Cash Receipts from Sales of Other Financial Assets | ||||||||||||||||||||||||||
| Cash Payments for Derivative Financial Instruments | ( | ( | ||||||||||||||||||||||||
| Cash Receipts from Derivative Financial Instruments | ||||||||||||||||||||||||||
| Acquisitions, Net of Cash Acquired | ( | |||||||||||||||||||||||||
| Cash Payments to Acquire Property, Plant and Equipment | 4.8 | ( | ( | ( | ||||||||||||||||||||||
| Cash Receipts from Sales of Property, Plant and Equipment | ||||||||||||||||||||||||||
| Cash Payments to Acquire Intangible Assets | 4.10 | ( | ( | ( | ||||||||||||||||||||||
| Cash Receipts from Sales of Shares at Fair Value through Other Comprehensive Income | ||||||||||||||||||||||||||
| Cash Receipts from Sales of Shares of Investment in Associates | ||||||||||||||||||||||||||
| Cash Receipts from Sales of Subsidiaries | ||||||||||||||||||||||||||
| Interest Received | ||||||||||||||||||||||||||
| Net Cash Provided by / (Used in) Investing Activities | ( | |||||||||||||||||||||||||
| Financing Activities: | ||||||||||||||||||||||||||
| Cash Proceeds from Issuing Shares | 4.21.1, 4.21.5 | |||||||||||||||||||||||||
| Cash Payments for Costs from Issuing Shares | 4.21.5 | ( | ( | |||||||||||||||||||||||
| Cash Proceeds in Connection with Exercised Stock (2021) and Convertible Bonds (2020) | 4.21.1, 4.21.5 | |||||||||||||||||||||||||
| Cash Payments for Repurchases of own Convertible Bonds | 4.18 | ( | ||||||||||||||||||||||||
| Payment for transaction costs for repurchases of own convertible bonds | 4.18 | ( | ||||||||||||||||||||||||
| Cash Receipts (+) / Cash Payments (-) from Financing from Collaborations | 4.19 | ( | ||||||||||||||||||||||||
| Cash Receipts from Contracts with Royalty Pharma | 4.20 | |||||||||||||||||||||||||
| Cash Payments for Costs in Connection with Contracts with Royalty Pharma | 4.20 | ( | ||||||||||||||||||||||||
| Cash Payments for Principal Elements of Lease Payments | 4.9 | ( | ( | ( | ||||||||||||||||||||||
| Interest Paid | ( | ( | ( | |||||||||||||||||||||||
| Net Cash Provided by / (Used in) Financing Activities | ||||||||||||||||||||||||||
| Effect of Exchange Rate Differences on Cash | ( | ( | ||||||||||||||||||||||||
| Increase / (Decrease) in Cash and Cash Equivalents | ( | |||||||||||||||||||||||||
| Cash and Cash Equivalents at the Beginning of the Period | ||||||||||||||||||||||||||
| Cash and Cash Equivalents at the End of the Period | ||||||||||||||||||||||||||
The Notes are an integral part of these consolidated financial statements.
F-13
Notes
1 General Information
Business Activities and the Company
MorphoSys AG ("the Company" or "MorphoSys") is a biopharmaceutical company dedicated to development and commercialization of therapeutics for patients suffering from various cancers. The Company has a proprietary portfolio of compounds and a pipeline of compounds developed with partners from the pharmaceutical and biotechnology industry. MorphoSys was founded as a German limited liability company in July 1992. In June 1998, MorphoSys became a German stock corporation. In March 1999, the Company completed its initial public offering on Germany’s "Neuer Markt": the segment of the Deutsche Börse designated, at that time, for high-growth companies. On January 15, 2003, MorphoSys AG was admitted to the Prime Standard segment of the Frankfurt Stock Exchange. On April 18, 2018, MorphoSys completed an IPO on the Nasdaq Global Market through the issue of American Depositary Shares (ADS). Each ADS represents 1/4 of a MorphoSys ordinary share. MorphoSys AG’s registered office is located in Planegg (district of Munich), and the registered business address is Semmelweisstrasse 7, 82152 Planegg, Germany. The MorphoSys AG consolidated and separate financial statements can be viewed at this address. The Company is registered in the Commercial Register B of the District Court of Munich under the number HRB 121023.
2 Summary of Material Accounting Policies
2.1 Basis of Application and Changes in Accounting Standards
2.1.1 Basis of Application
These consolidated financial statements were prepared in accordance with the International Financial Reporting Standards (“IFRS”), taking into account the recommendations of the International Financial Reporting Standards Interpretations Committee (IFRS IC). All standards and interpretations were applied that were in force as of December 31, 2023 and adopted by the European Union (EU). As of December 31, 2023, there were no standards or interpretations that affected the consolidated financial statements for the years ended December 31, 2023, 2022 and 2021 that were in effect, but not yet endorsed into European law. As a result, the consolidated financial statements comply with both the IFRSs published by the International Accounting Standards Board (IASB) and those adopted by the EU. These consolidated financial statements also take into account the supplementary provisions under commercial law, which must be applied in accordance with Section 315e (1) of the German Commercial Code (Handelsgesetzbuch – HGB). In accordance with the regulations of the United States Securities and Exchange Commission, the statement of profit or loss is presented for a comparative period of three years. This extends beyond the comparative period of two years in accordance with the requirements of IFRS as adopted by the EU.
The consolidated financial statements as of the reporting dates of December 31, 2023 and 2022, as well as the periods from January 1 through December 31 for the years 2023, 2022 and 2021, comprise MorphoSys AG and its subsidiaries (collectively, the “MorphoSys Group” or the “Group”). MorphoSys AG prepares the consolidated financial statements for the largest and the smallest consolidated group.
All figures in this report were rounded to the nearest euro, thousand euros or million euros.
Due to the war in Ukraine, clinical trials in Ukraine and Russia were stopped and moved to other countries. Neither this nor the conflict in the Middle East had a significant negative impact on the business activities of MorphoSys AG. The same applies to the Company's net assets, financial position and results of operations. Indirect effects such as rising energy prices, inflation and fluctuating exchange rates also have no material impact on business activities in the past fiscal year.
According to the Corporate Sustainability Reporting Directive Implementation Act (CSR-RUG) on the disclosure of non-financial information, companies must, in addition to reporting on material aspects, also disclose related risks that are linked to their own business activities, business relationships, products and services, and that are very likely to have or will have serious negative effects on the material aspects. The Group has not identified any such risks in the financial year under review on a net basis.
Unless stated otherwise, the accounting policies set out below, were applied consistently to all periods presented in these consolidated financial statements.
2.1.2 Changes in Accounting Policies and Disclosures
The accounting standards applied generally correspond to the policies used in the prior year.
F-14
New or Revised Standards and Interpretations Adopted for the first Time in the Financial Year
| Standard / Interpretation | Mandatory Application for financial years starting on | Adopted by the European Union | Impact on MorphoSys | |||||||||||||||||||||||
| IFRS 17 and IFRS 17 (A) | Insurance Contracts and Amendments to IFRS 17 | 01/01/2023 | yes | none | ||||||||||||||||||||||
| IFRS 17 (A) | Initial Application of IFRS 17 and IFRS 9 — Comparative Information | 01/01/2023 | yes | none | ||||||||||||||||||||||
| IAS 1 (A) | Disclosure of Accounting Policies and IFRS Practice Statement 2 | 01/01/2023 | yes | yes | ||||||||||||||||||||||
| IAS 8 (A) | Definition of Accounting Estimates | 01/01/2023 | yes | yes | ||||||||||||||||||||||
| IAS 12 (A) | Deferred Tax related to Assets and Liabilities arising from a Single Transaction | 01/01/2023 | yes | yes | ||||||||||||||||||||||
| IAS 12 (A) | International Tax Reform – Pillar Two Model Rules | 01/01/2023 | yes | none | ||||||||||||||||||||||
(A) Amendments
Standards with the remark "none" do not have an impact on the consolidated financial statements. The impact on the consolidated financial statements of the amendments to IAS 1, IAS 8 and IAS 12 are not considered material and, therefore, not explained separately.
New or Revised Standards and Interpretations not yet Mandatorily Applicable
The following new or revised standards that were not yet mandatory in the reporting period and have not yet been adopted by the European Union, have not been applied prematurely. The effects on the consolidated financial statements of standards marked with “yes” are considered probable and are currently being examined by the Group. Only significant effects are described in more detail. Standards with the comment “none” are not expected to have a material impact on the consolidated financial statements.
| Standard / Interpretation | Mandatory Application for financial years starting on | Adopted by the European Union | Possible Impact on MorphoSys | |||||||||||||||||||||||
| IAS 1 (A) | Classification of Liabilities as Current or Non-current, Non-current Liabilities with Covenants | 1/1/2024 | yes | yes | ||||||||||||||||||||||
IAS 7 (A) and IFRS 7 (A) | Supplier Finance Arrangements | 1/1/2024 | no | none | ||||||||||||||||||||||
| IFRS 16 (A) | Lease Liability in a Sale and Leaseback | 1/1/2024 | yes | none | ||||||||||||||||||||||
| IAS 21 (A) | The Effects of Changes in Foreign Exchange Rates - Lack of Exchangeability | 1/1/2025 | no | none | ||||||||||||||||||||||
(A) Amendments
2.2 Consolidation Principles
2.2.1 Consolidated Companies and Scope of Consolidation
MorphoSys AG, as the ultimate parent company, is located in Planegg, near Munich. MorphoSys AG has one wholly owned subsidiary, MorphoSys US Inc. (Boston, Massachusetts, USA). MorphoSys US Inc. in turn has a wholly owned subsidiary - Constellation Pharmaceuticals, Inc. (Boston, Massachusetts, USA). Constellation Pharmaceuticals, Inc. also has a wholly owned subsidiary, Constellation Securities Corp. (Boston, Massachusetts, USA). Constellation Pharmaceuticals, Inc. and Constellation Securities Corp. are collectively referred to as “Constellation,” and all entities constitute the “MorphoSys Group” or “Group.”
The consolidated financial statements as of December 31, 2023, were prepared by the Management Board on March 12, 2024, by resolution of the Management Board, authorized for issue, and forwarded to the Supervisory Board for review and approval. The members of the Group’s Management Board are Jean-Paul Kress, M.D., as Chief Executive Officer (Chair of the Management Board) and Lucinda Crabtree, Ph.D., as Chief Financial Officer. On March 14, 2023, MorphoSys announced that Lucinda Crabtree, Ph.D., will join as Chief Financial Officer. She started on August 8, 2023. On March 17, 2023, Sung Lee left
F-15
the company and resigned as a member of the Management Board. With effect as of March 1, 2023, Charlotte Lohmann has been appointed as a member of the Management Board until August 31, 2023.
2.2.2 Consolidation Methods
Subsidiaries
The following Group subsidiaries are included in the scope of consolidation, as shown in the table below.
| Company | Purchase of Shares / Establishment | Included in Basis of Consolidation since | ||||||||||||
| Constellation Pharmaceuticals, Inc., Boston, Massachusetts, USA | July 2021 | 07/15/2021 | ||||||||||||
| Constellation Securities Corp., Boston, Massachusetts, USA | July 2021 | 07/15/2021 | ||||||||||||
| MorphoSys US Inc., Boston, Massachusetts, USA | July 2018 | 07/02/2018 | ||||||||||||
These subsidiaries are fully consolidated as they are direct or indirect wholly owned subsidiaries. MorphoSys controls the subsidiaries due to its full power over the investees. Additionally, MorphoSys is subject to risk exposure and has rights to variable returns from its involvement with the investees. MorphoSys also has unlimited capacity to exert power over the investees to influence its returns.
The Group does not have any entities consolidated as joint ventures using the equity method.
The assets and liabilities of the fully consolidated international entities are recognized using Group-wide uniform accounting and valuation methods. The consolidation methods applied have not changed from the previous year.
Upon consolidation, the carrying amounts of the parent company’s investments in each subsidiary are offset against the parent’s share in the equity of each subsidiary. Inter-company assets and liabilities, income and expenses, and profits or losses arising from transactions between Group companies are eliminated in full.
Associates
Associates are all entities over which the Group has significant influence but not control or joint control. This is generally the case where the Group holds between 20% and 50% of the voting rights. Investments in associates are accounted for using the equity method of accounting, after initially being recognized at cost.
Under the equity method of accounting, the investments are initially recognized at cost and adjusted thereafter to recognize the Group's share of post-acquisition profits or losses of the investee in profit or loss, and the Group's share of movements in other comprehensive income of the investee in other comprehensive income. Dividends received or receivable from associates are recognized as a reduction in the carrying amount of the investment.
Where the Groups' share of losses in an equity-accounted investment equals or exceeds its interest in the entity, including any other long-term interest that is attributable to the net investment in the investee in substance, the Group does not recognize further losses, unless it has incurred legal and constructive obligations or made payments on behalf of the investee.
Unrealized gains on transactions between the group and its associates are eliminated to the extent of the Group's interest in these entities. Unrealized losses are also eliminated unless the transaction provides evidence of an impairment of the asset transferred. Accounting policies of equity-accounted investees have been changed where necessary to ensure consistency with the policies adopted by the Group.
The carrying amount of equity-accounted investments is tested for impairment in accordance with the impairment method described in Note 2.6.9 "Impairment of Non-Financial Assets" in the consolidated financial statements as of December 31, 2023. The net investment in an associate is impaired and impairment losses are incurred if there is objective evidence of impairment as a result of events that occurred after the initial recognition of the net investment and that loss events have an impact on the estimated future cash flows from the net investment that can be reliably estimated. A significant or prolonged decline in the fair value of an investment in an equity instrument below its cost is an objective evidence of impairment.
2.3 Principles of Foreign Currency Translation
The Group’s consolidated financial statements are presented in euros, which is also the parent company’s functional currency. For each entity, the Group determines the functional currency. The items included in the financial statements of each entity are measured using that functional currency.
F-16
Transactions and Balances
Transactions in foreign currencies are initially recorded by the Group’s entities at their respective functional currency spot rates at the date the transaction first qualifies for recognition. Monetary assets and liabilities denominated in foreign currencies are translated at the functional currency spot rates of exchange at the reporting date. Differences arising on settlement or translation of monetary items are recognized in other income or expenses. For monetary items relating to investing and financing activities, differences are recognized in finance income or finance expenses.
Non-monetary items that are measured in terms of historical cost in a foreign currency are translated using the exchange rates at the dates of the initial transactions.
Group Companies
On consolidation, the assets and liabilities of foreign operations are translated into euros at the rate of exchange prevailing at the reporting date and their statements of profit or loss are translated at average exchange rates. The exchange differences arising on translation for consolidation are recognized in “Other Comprehensive Income Reserve” (equity).
2.4 Key Estimates and Assumptions
In preparing the consolidated financial statements, it is necessary to make estimates and assumptions that affect the carrying amounts of assets, liabilities and contingent liabilities at the balance sheet date and the amounts of income and expense recognized in the period under report. The actual results may differ from these estimates. The estimates and underlying assumptions are subject to continuous review and are based on historical experience and other factors, including the expectation of future events that are believed to be realistic under the prevailing circumstances. Any changes in estimates are recognized in the period in which the changes are made and in all relevant future periods. The resulting accounting-related estimates will, by definition, seldom correspond to the actual results.
The estimates and assumptions that carry a significant risk of causing material adjustments to the carrying amounts of assets and liabilities in the next financial year are addressed below.
Revenues
Revenues from product sales, royalties, license fees, milestones are subject to assumptions regarding variable consideration components, probabilities of occurrence and individual selling prices within the scope of the accounting and measurement principles explained in Note 2.5.1. Accruals in connection with revenues from product sales are also affected by estimates and assumptions.
Impairment of Financial Assets
Impairment losses on financial assets in the form of debt instruments and accounts receivable are based on assumptions about credit risk. The Group exercises discretion in making these assumptions and in selecting the inputs to calculate the impairment based on past experience, current market conditions and forward-looking estimates at the end of each reporting period.
Financial Liabilities from Collaborations
For details on estimates and assumptions in connection with financial liabilities from collaborations refer to Note 4.19.
Leases
In determining the lease term, all facts and circumstances are considered that create an economic incentive to exercise an extension option. Extension options are only included in the lease term if the lease is reasonably certain to be extended.
Licenses for Marketed Products
The acquired licenses are amortized over their estimated useful life. An impairment loss is recognized when events or changes in circumstances indicate that the licenses are impaired.
Intangible assets not yet available for use and Goodwill
The Group performs an annual review to determine whether in-process R&D programs (intangible assets not yet available for use) or goodwill are subject to impairment in accordance with the accounting policies discussed in Note 2.6.9. The recoverable amounts from in-process R&D programs and cash-generating units have been determined using value-in-use calculations and are subjected to a sensitivity analysis. These calculations require the use of estimates (see Notes 4.10 and 4.11).
F-17
Accruals
The Group has entered into various research and development contracts with research institutions and other companies. These agreements are generally cancellable, and related costs are recorded as research and development expenses as incurred. The Group recognizes accruals for estimated ongoing research costs that have been incurred. When evaluating the appropriateness of the accrued expenses, the Group analyzes the progress of the studies, including the phase and completion of events, invoices received and contractually agreed costs. Significant judgments and estimates are made in determining the deferred balances at the end of any reporting period. Actual results may differ from the Group’s estimates. The Group’s historical accrual estimates have not been materially different from the actual costs.
Financial Liabilities from Future Payments to Royalty Pharma
For details on estimates and assumptions in connection with the financial liabilities from future payment to Royalty Pharma refer to Note 4.20.
Income Taxes
Income taxes comprise taxes levied in the individual countries on taxable profit and changes in deferred taxes. The income taxes reported are recognized on the basis of the statutory regulations in force or enacted as of the reporting date in the amount in which they are expected to be paid or refunded. Deferred taxes are recognized for tax-deductible or temporary taxable differences between the carrying amounts of assets and liabilities in the IFRS balance sheet and the tax base, as well as for tax effects arising from consolidation measures and tax reduction claims arising from loss carryforwards that are likely to be realized in subsequent years. Goodwill is excluded.
The assessment of the recoverability of deferred tax assets considers the currently achieved total results of a legal entity as well as the expected future taxable results, derived from the corporate planning. The recognition of deferred tax assets on tax loss carryforwards requires management to make estimates and judgments about the amount of future taxable profit available against which the tax loss carryforwards can be utilized. Deferred tax assets on loss carryforwards are only recognized to the extent that sufficient taxable income is expected in the future.
Uncertain tax positions are analyzed on an ongoing basis and, if taxes are sufficiently probable, risk provisions are recognized in an appropriate amount in each case. Uncertainties arise, among other things, from matters that are being discussed in ongoing tax audits but have not yet resulted in final findings or are under discussion due to disputed legal situations or new case law.
As the estimates can change over time, for example, as a result of findings in the course of the tax audit or current case law, there will also be a corresponding effect on the amount of the required assessment of the risk provision. The amount of the expected tax liability or tax receivable reflects the amount representing the best estimate or the expected value, taking into account any existing tax uncertainties.
For the assessment of the impairment of deferred tax assets, the planning assumptions are influenced by key estimates and these include the profit forecasts of the respective legal entities and the assessment of convincing evidence in the context of IAS 12.35 to overcome a history of losses.
2.5 Accounting Policies applied to Line Items of the Statement of Profit or Loss
2.5.1 Revenues and Revenues Recognition
Recognizing revenue from contracts with customers requires the following five-stage approach:
•Identification of the contract
•Identification of performance obligations
•Determination of the transaction price
•Allocation of the transaction price
•Revenue recognition
The Group’s revenues typically include revenue from product sales, royalties, license fees, milestone payments and service fees.
Revenues from Product Sales
Revenues from the sale of MorphoSys products are recognized at the transaction price at the time the customer obtains control of the product. This is defined as the point at which the customer receives the product. As a result, revenues are recognized
F-18
based on a specific point in time. The transaction price represents the consideration expected by MorphoSys in exchange for the product and takes into account variable components. The variable consideration is only included in the transaction price if it is highly probable that there will not be a subsequent material adjustment to the transaction price.
The most common elements of variable consideration related to product sales at MorphoSys are listed below and are determined according to the expected value approach.
•Rebates and discounts agreed with government agencies, buying groups, specialty distributors and specialty pharmacies are accrued and deducted from revenues at the time the related revenues are recognized. They are calculated based on actual discounts and rebates granted, specific regulatory requirements, specific terms in individual agreements, product pricing and/or the anticipated sales channel mix. Because the Company recognizes revenue upon transfer of control of the product to specialty distributors and specialty pharmacies, and not upon transfer to the end-user (patient), for certain rebates the Company is required to estimate the mix of product sales between its sales channels in determining the amount of rebate that will ultimately be paid.
•Discounts offered to customers are intended to encourage prompt payment and are deferred and recognized as revenue deductions at the time the related revenues are recognized.
•Accruals for product returns are recognized as revenue deductions at the time the corresponding revenues are recognized.
Variable consideration is deducted from trade receivables, in case these are directly paid to the direct customer. In case payments are to be made to another party, these are presented as accruals. Accruals for revenue deductions are adjusted to the actual amounts when rebates and discounts and cash discounts are realized. The accruals represent estimates of the related obligations, meaning that management’s judgment is required in estimating the impact of these revenue deductions.
Royalties
Revenue recognition for royalties (income based on a percentage of sales of a marketed product) is based on the same revenue recognition principles that apply to sales-based milestones, as described below.
License Fees and Milestone Payments
The Group recognizes revenues from license fees for intellectual property (IP) both at a point in time and over a period of time. The Group must make an assessment as to whether such a license represents a right-to-use the IP (at a point in time) or a right to access the IP (over time). Revenue for a right-to-use license is recognized by the Group when the licensee can use and benefit from the IP after the license term begins, e.g., the Group has no further obligations in the context of the out-licensing of a drug candidate or technology. A license is considered a right to access the intellectual property when the Group undertakes activities during the license term that significantly affect the IP, the customer is directly exposed to any positive or negative effects of these activities, and these activities do not result in the transfer of a good or service to the customer. Revenues from the right to access the IP are recognized on a straight-line basis over the license term.
Milestone payments for research and development are contingent upon the occurrence of a future event and represent variable consideration. The Group’s management estimates at the contract’s inception that the most likely amount for milestone payments is zero. The most likely amount method of estimation is considered the most predictive for the outcome since the outcome is binary; e.g. achieving a specific success in clinical development (or not). The Group includes milestone payments in the total transaction price only to the extent that it is highly probable that a significant reversal of accumulated revenue will not occur when the uncertainty associated with the variable consideration is subsequently resolved.
Sales-based milestone payments included in contracts for IP licenses are considered by the Group to be sales-based license fees because they are solely determined by the sales of an approved drug. Accordingly, such milestones are recognized as revenue once the sales of such drugs occur or at a later point if the performance obligation has not been fulfilled.
Service Fees
Service fees for the assignment of personnel to research and development collaborations are recognized as revenues in the period the services were provided. If a Group company acts as an agent, revenues are recognized on a net basis.
Agreements with multiple Performance Obligations
A Group company may enter into agreements with multiple performance obligations that include both licenses and services. In such cases, an assessment must be made as to whether the license is distinct from the services (or other performance obligations) provided under the same agreement. The transaction price is allocated to separate performance obligations based on the relative stand-alone selling price of the performance obligations in the agreement. The Group company estimates stand-alone selling prices for goods and services not sold separately on the basis of comparable transactions with other customers.
F-19
The residual approach is the method used to estimate a stand-alone selling price when the selling price for a good or service is highly variable or uncertain.
Principal-Agent Relationships
In agreements involving two or more independent parties who contribute to the provision of a specific good or service to a customer, the Group company assesses whether it has promised to provide the specific good or service itself (the company acting as a principal) or to arrange for this specific good or service to be provided by another party (the company acting as an agent). Depending on the result of this assessment, the Group company recognizes revenues on a gross (principal) or net (agent) basis. A Group company is an agent and recognizes revenue on a net basis if its obligation is to arrange for another party to provide goods or services, i.e., the Group company does not control the specified good or service before it is transferred to the customer. Indicators to assist a company in determining whether it does not control the good or service before it is provided to a customer and is, therefore, an agent, include, but are not limited to, the following criteria:
•Another party is primarily responsible for fulfilling the contract.
•The company does not have inventory risk.
•The company does not have discretion in establishing the price.
No single indicator is determinative or weighted more heavily than other indicators. However, some indicators may provide stronger evidence than others, depending on the individual facts and circumstances. A Group company’s control needs to be substantive; obtaining the legal title to a good or service only momentarily before it is transferred to the customer does not necessarily indicate that a Group company is a principal. Generally, an assessment as to whether a Group company is acting as a principal or an agent in a transaction requires a considerable degree of judgment.
Based on the relevant facts and circumstances, the assessment of an agreement may lead to the conclusion that the counterparty is a cooperation partner or partner rather than a customer because the contract parties share equally in the risk of co-developing a drug and in the future profits from the marketing of the approved drug.
2.5.2 Cost of Sales
The cost of sales includes the acquisition and production cost of inventories recognized as an expense, personnel expenses, inventory write-downs, reversals of inventory write-downs, impairments and scheduled depreciation and other expenses for intangible assets, costs for external services as well as other costs. Cost of sales are recognized as an expense as incurred.
2.5.3 Operating Expenses
Operating expenses are allocated to the functional costs on the basis of cost centers or percentage allocation keys.
Research and Development Expenses
Research costs are expensed in the period in which they occur. Development costs are generally expensed as incurred. Development costs are recognized as an intangible asset when the criteria such as the probability of expected future economic benefits, as well as the reliability of cost measurement, are met. Development costs previously recognized as an expense are not recognized as an asset in a subsequent period.
This line item contains personnel expenses, consumable supplies, impairment charges, impairment reversals, amortization and other costs related to intangible assets (additional information can be found in Note 4.10), costs for external services, infrastructure costs and depreciation as well as other costs.
Selling Expenses
The line item includes personnel costs, consumable supplies, amortization of intangible assets (software; additional information can be found in Note 4.10), costs for external services, infrastructure costs and depreciation as well as other costs. This item also includes all expenses for services provided by Incyte in connection with the joint US sales activities.
General and Administrative Expenses
The line item includes personnel costs, consumable supplies, amortization of intangible assets (software; additional information can be found in Note 4.10), costs for external services, infrastructure costs and depreciation as well as other costs.
F-20
Expenses through Share-based Payment Transactions and Issue of Convertible Instruments
The Group spreads the compensation expenses from the estimated fair values of share-based payments on the reporting date over the period in which the beneficiaries provide the services that triggered the granting of the share-based payments. Personnel expense is recognized in the respective functional area to which the beneficiary is allocated.
Share-based compensation is considered when the Group acquires goods or services in exchange for shares or stock options (“settlement in equity instruments”) or other assets that represent the value of a specific number of shares or stock options (“cash settlement”). Additional information can be found in Note 5.
2.5.4 Income Tax Benefit / Expenses
Current income taxes are calculated based on the respective local taxable income and local tax rules for the period. In addition, current income taxes presented for the period include adjustments for uncertain tax payments or tax refunds for periods not yet finally assessed, excluding interest expenses and penalties on the underpayment of taxes. In the event that amounts included in the tax returns are considered unlikely to be accepted by the tax authorities (uncertain tax positions), a provision for income taxes is recognized. Tax refund claims from uncertain tax positions are recognized when it is probable that they can be realized. Current taxes reflect the expected tax liability on the taxable income for the year, based on the enacted or substantially enacted tax rates, as well as adjustments to the tax liability for previous years.
Deferred tax assets or liabilities are calculated for temporary differences between the tax bases and the financial statement carrying amounts, including differences from consolidation, unused tax loss carryforwards, and unused tax credits. Measurement is based on enacted or substantively enacted tax rates and tax rules.
Deferred tax assets are offset against deferred tax liabilities when the taxes are levied by the same taxation authority, and the entity has a legally enforceable right to offset current tax assets against current tax liabilities according to their maturity.
Assessments as to the recoverability of deferred tax assets require the use of judgment regarding assumptions related to estimated future taxable profits. This includes the amounts of taxable future profits, the periods in which those profits are expected to occur, and the availability of tax planning opportunities. The Group records a deferred tax asset only when it is probable that a corresponding amount of taxable profit will be available against which the deductible temporary differences relating to the same taxation authority and the same taxable entity can be utilized.
The analysis and forecasting required in this process are performed for individual jurisdictions by qualified local tax and financial professionals. Given the potential significance surrounding the underlying estimates and assumptions, group-wide policies and procedures have been designed to ensure consistency and reliability around the recoverability assessment process. Forecast operating results are based upon approved business plans, which are themselves subject to a well-defined process of control. As a matter of policy, especially strong evidence supporting the recognition of deferred tax assets is required if an entity has suffered a loss in either the current or the preceding period.
Changes in deferred tax assets and liabilities are generally recognized through profit and loss in the consolidated statement of profit or loss, except for changes recognized directly in equity, and changes recognized in connection with a business combination, where the purchase price allocation results in deferred tax assets and liabilities being recognized as an offset against goodwill. Deferred tax assets are recognized only to the extent that it is likely that there will be future taxable income to offset. Deferred tax assets are reduced by the amount that the related tax benefit is no longer expected to be realized.
2.5.5 Earnings per Share
The Group reports basic and diluted earnings per share. Basic earnings per share are computed by dividing the net profit or loss attributable to parent company shareholders by the weighted-average number of ordinary shares outstanding for the reporting period. Diluted earnings per share are calculated in the same manner with the exception that the net profit or loss attributable to parent company shareholders and the weighted-average number of ordinary shares outstanding are adjusted for any dilutive effects resulting from stock options and restricted stock units granted to the Management Board and employees and convertible bonds. The potentially dilutive shares are excluded from the calculation of the dilutive earnings per share, if the dilutive effect would result in a decline in the loss per share for the respective year.
2.6 Accounting Policies applied to Line Items of the Balance Sheet
The balance sheet is presented on the basis of the current/non-current distinction. Current assets and liabilities are those that are due within a period of one year. Regardless of their maturity, accounts receivable, accounts payable and inventories are also deemed to be current if they are due or sold within the normal course of a business cycle, which can be longer than one year. Deferred taxes are presented as non-current assets and liabilities.
F-21
2.6.1 Financial Instruments
A financial instrument is a contract that gives rise to a financial asset of one entity and a financial liability or equity instrument of another entity. Financial assets and liabilities comprise non-derivative and derivative receivables and payables.
The Group recognizes financial instruments at the point in time when it becomes the contractual party of the instrument. A normal market purchase or sale of financial assets is recognized on the trade date, i.e. the date on which the obligation to buy or sell the asset was entered into.
On initial recognition, the Group measures financial assets and financial liabilities at fair value, with the exception of trade receivables without a significant financing component, which are measured at the transaction price specified in Note 2.5.1.
When the financial asset is not subsequently measured at fair value in profit or loss, transaction costs directly attributable to the acquisition of that asset will be added to the fair value. Transaction costs of financial assets measured at fair value through profit or loss are recognized as expenses in profit or loss.
Direct attributable transaction costs are deducted from the fair value if they are attributable to financial liabilities measured at amortized cost. Transaction costs are recognized directly in profit or loss if they are related to the issue of financial liabilities measured at fair value.
Financial assets and liabilities are offset only when it is currently legally enforceable to offset the amounts and there is an intention to do so. The Group does not perform offsetting.
Financial Assets
Classification, Measurement and Disclosure
The Group's financial assets include both debt instruments and equity instruments. A debt instrument is a contractual right to receive cash or another financial asset from another entity or to exchange financial assets or financial liabilities with another entity under conditions that are potentially favorable to the entity. An equity instrument is any contract that evidences a residual interest in the assets of an entity after deducting all of its liabilities.
The classification of financial assets (debt instruments) for subsequent measurement depends on the Group’s business model for managing the financial assets and the asset’s cash flow characteristics. The business model reflects how the Group manages its financial assets to generate cash flows. The business model determines whether cash flows will result from collecting contractual cash flows, selling the financial assets, or both. A financial asset can give rise to cash flows that are ‘solely payments of principal and interest (SPPI)’ on the principal amount outstanding. This SPPI test involves an assessment of whether the cash flows of the instrument consist solely of payments of interest and principal. Interest is typically consideration for the time value of money and credit risk. Payments of principal are payments on the principal amount outstanding.
Assets that are held in order to collect the contractual cash flows and for which these cash flows represent interest and principal payments only are measured at amortized cost (AC). Interest income from these financial assets is recognized in finance income using the effective interest method. Negative interests are recognized in Finance Expense. Gains and losses upon derecognition are recognized directly in profit or loss and recorded in the finance result. Impairment losses are recognized as a separate line item in profit or loss. The Group's financial assets at amortized cost comprise the balance sheet item "Cash and Cash Equivalents", part of the balance sheet item "Other Financial Assets" (term deposits), the balance sheet item "Accounts Receivable" and part of the balance sheet item "Prepaid Expenses and Other Assets" (restricted cash for e.g. rental deposits).
The Group considers all balances on bank accounts and cash in hand to be cash and cash equivalents. Cash equivalents are held for the purpose of meeting short-term cash commitments rather than for investment.
Assets that are held to collect the contractual cash flows and to sell the financial assets and where the cash flows represent principal and interest payments only are measured at fair value through other comprehensive income (FVOCI). The Group does not hold any financial assets that are measured at fair value through other comprehensive income.
Assets that do not meet the criteria of the categories “at amortized cost” or “at fair value through other comprehensive income” are allocated to the category “at fair value through profit or loss” (FVTPL). Gains and losses on debt instruments that are subsequently measured at fair value through profit or loss are recognized in other income/expenses or the finance result in the period in which they occur. The Group's financial assets measured at fair value through profit or loss include part of the balance sheet item "Other Financial Assets" (money market funds) and the balance sheet item "Financial Assets from Collaborations". Derivatives with a positive fair value are recorded in the balance sheet item "Other Receivables" and derivatives with a negative fair value are recorded in the balance sheet item "Other Liabilities."
MorphoSys does not apply hedge accounting.
The Group reclassifies debt instruments only in case when there is a change in the business model for managing such assets.
F-22
For investments in equity instruments that are not held for trading, classification depends on whether the Group has irrevocably elected, at the time of initial recognition when the instrument is acquired, to measure the equity instruments at fair value through other comprehensive income. If this option is not exercised, equity instruments are measured at fair value through profit or loss. The Group has exercised the option to measure all equity instruments held at fair value through other comprehensive income. As a result, after derecognition of such an instrument, no subsequent reclassification of these gains and losses to the consolidated income statement takes place. Dividends from such instruments continue to be recognized in profit or loss under other income when the Group's right to receive payment is established. Equity instruments include the equity investments made by the Group. As of December 31, 2023, MOR Group does not account for any equity instruments.
Impairment and Reversal of Impairment
Financial assets in the categories measured at amortized cost (AC) and at fair value through other comprehensive income (FVOCI) require the calculation of an impairment loss, which is recognized on the basis of expected credit losses. A distinction is made between a general and a simplified impairment model.
Impairment losses on financial instruments are reported under impairment losses on financial assets. Reversals of impairment are recognized in income from reversals of impairment losses.
Impairment losses on trade receivables are reported in other expenses. Amounts, which were written off previously, but are received in subsequent periods, are recognized in other income.
Financial Instruments according to General Expected Credit Loss Model
The Group assesses on a forward-looking basis the expected credit losses associated with its debt instruments carried at amortized cost. When a debt instrument is recognized for the first time, an impairment loss is recognized in the amount of the expected loss for twelve months. The impairment method applied depends on whether there has been a significant increase in credit risk. If at the reporting date, the credit risk of a financial instrument has not increased significantly since initial recognition, the Group measures the loss allowance for that financial instrument at an amount equal to twelve-month expected credit losses (Level 1). Where the expected lifetime of an asset is less than twelve months, expected losses are measured at its expected lifetime. Expected credit losses are based on the contractual cash flows multiplied by the premium of a credit default swap according to the expected maturity of the contracting party (Level 1). In case the credit risk of a financial instrument has increased significantly since initial recognition, the Group measures impairment for that financial instrument at an amount equal to the lifetime expected credit losses. The Group currently classifies an increase in credit risk on debt instruments as significant when the premium on a counterparty credit default swap has increased by 100 basis points since the initial recognition of the instrument or if the amount is more than 30 days overdue (Level 2). If there is an objective indication of impairment, the interest received must also be adjusted so that the interest as of this date is accrued based on the net carrying amount (carrying amount less risk provisions) of the financial instrument (Level 3).
Financial Instruments according to Simplified Expected Credit Loss Model
In the case of accounts receivable, the Group applies the simplified approach, which requires expected lifetime losses to be recognized from the initial recognition of the receivables (Level 2). In the event of objective evidence of impairment of trade receivables, such assets are reported as credit-impaired and the expected loss is calculated as the difference between the gross carrying amount and the present value of the expected cash flows discounted at the original effective interest rate (Level 3).
All accounts receivable were aggregated to measure the expected credit losses. All accounts receivable are currently due from customers in the pharmaceutical industry with similar credit risk profiles. The impairment is determined on the basis of the premium for an industry credit default swap. In the event that accounts receivable cannot be grouped together, they are measured individually.
Objective indications of the impairment of financial instruments may result from an overdue period of more than 90 days, significant financial difficulties on the part of the issuer or debtor, a breach of contract such as a default or delay in interest or principal payments, an increased probability of insolvency or other reorganization proceedings, or the disappearance of an active market for a financial asset due to financial difficulties.
Financial instruments are impaired if, based on a reasonable estimate, they are not expected to be realized and one of the objective indications occurs. An indicator that there is no reasonable expectation of recovery is, among other things, when internal or external information indicates that the Group will not receive the outstanding contractual amounts in full. This is generally assumed if financial instruments are more than two years overdue.
Derecognition
Financial assets are derecognized when the rights to receive cash flows from the financial assets have expired or have been transferred and the Group has transferred substantially all risks and rewards of ownership.
F-23
Financial Liabilities
Classification, Measurement and Disclosure
Contracts for liabilities are examined to determine whether they are only equity or only debt in nature or contain components of both. If the economic substance of the contractual agreement contains both components, they are recognized separately as equity instruments and as financial liabilities.
Financial liabilities are classified in the following categories:
•Financial liabilities at fair value through profit or loss
•Financial liabilities at amortized cost
Subsequent measurement at fair value through profit or loss (FVTPL) can be irrevocably designated upon initial recognition or is performed for derivatives with a negative fair value. Gains or losses arising from changes in fair value are recognized in profit or loss in finance income/expenses. The Group does not make a designation for measurement at fair value.
Financial liabilities measured at amortized cost (FLAC) are measured using the effective interest method. Gains and losses are recognized in the income statement in other income/expenses or in finance income/expenses using the effective interest method. For financial liabilities measured at amortized cost, an assessment is made at initial recognition as to whether separable embedded derivatives have been agreed in the contract. Embedded derivatives must be separated and recognized separately at fair value through profit or loss unless their terms are closely related to the host contract. The Group's financial liabilities measured at amortized cost include trade payables (part of the balance sheet item "Accounts Payable and Accruals"), the balance sheet items "Financial Liabilities from Collaborations" and the balance sheet items "Financial Liabilities from Future Payments to Royalty Pharma".
For contracts with equity and liability components, the fair value of the liability component is determined at the time of initial recognition using the market interest rate applicable to comparable instruments. This amount is recognized as a financial liability measured at amortized cost until the contract is settled or becomes due. The component classified as equity is determined by the difference between the total value of the contract and the fair value of the liability component. The resulting amount, net of income tax effects, is recognized as part of equity in additional paid-in capital and is not adjusted in subsequent periods. Transaction costs associated with the instrument are allocated between the two components based on the allocation of proceeds. Transaction costs attributable to the debt component are deducted from the carrying amount of the debt component and are amortized over the life of the contract using the effective interest method. Such a contract includes the convertible bond in the balance sheet item "Bonds". The exercise of the conversion option does not give rise to a gain or loss, but rather to a derecognition of the liability and a recognition of equity.
All amounts on financial liabilities at amortized cost that are payable within the next twelve months, are reported as a current liability. For bonds the undiscounted cash flows within the next twelve are considered as current. For the financial liabilities from collaborations and the financial liabilities from future payments to Royalty Pharma the planned payments in the next twelve months are discounted to determine the current liability.
Derecognition
A financial liability is derecognized when the underlying obligation is discharged, cancelled or expires.
2.6.2 Income Tax Receivables
Income tax receivables mainly include receivables from the tax authorities in connection with tax receivables from tax allowances and withheld capital gains tax, which were recognized at nominal value.
2.6.3 Inventories
Inventories are measured at the lower value of production or acquisition cost and net realizable value under the first-in, first-out method. Acquisition costs comprise all purchase costs, including those incurred in bringing the inventories into operating condition, and take purchase price reductions into account, such as bonuses and discounts. Manufacturing costs comprise all directly attributable costs as well as reasonably allocated overhead. Net realizable value is the estimated selling price less the estimated expenses necessary for completion and sale. Inventories are divided into the categories of raw materials and supplies, as well as unfinished and finished goods.
Material used in clinical trials (investigational medicinal product or IMP) is not capitalized since recognition criteria for inventory are not met. IMP is therefore expensed when incurred and recognized in the income statement under research and development expenses.
F-24
2.6.4 Prepaid Expenses and Other Assets
Prepaid expenses include expenses resulting from an outflow of liquid assets prior to the reporting date that are only recognized as expenses in the subsequent financial year. Such expenses usually involve maintenance contracts, sublicenses and upfront payments for external laboratory services not yet performed. Measurement is at nominal value or acquisition cost less impairments.
2.6.5 Property, Plant and Equipment
Property, plant and equipment are recorded at historical cost less accumulated depreciation (see Note 4.8) and any impairment losses (see Note 2.6.9). Historical cost includes expenditures directly related to the purchase at the time of the acquisition. Replacement purchases, building alterations and improvements are capitalized, whereas repair and maintenance expenses are recognized as expenses as they are incurred. Property, plant and equipment are depreciated on a straight-line basis over its estimated useful life (see table below). Leasehold improvements are depreciated on a straight-line basis over the shorter of either the asset’s estimated useful life or the remaining term of the lease.
| Asset Class | Useful Life | Depreciation Rates | ||||||||||||
| Office Equipment | ||||||||||||||
| Laboratory Equipment | ||||||||||||||
| Low-value Office and Laboratory Equipment | Immediately | % | ||||||||||||
| Computer Hardware | ||||||||||||||
| Permanent Improvements to Property/Buildings | % | |||||||||||||
The residual values and useful lives of assets are reviewed at the end of each reporting period and adjusted when necessary.
Borrowing costs that can be directly attributed to the acquisition, construction or production of a qualifying asset are not included in the acquisition or production costs.
2.6.6 Leases
For lessees, a uniform approach is applied to the recognition of leases, according to which assets for the right-of-use assets of the leased assets and liabilities for the payment obligations entered into are required to be recognized in the balance sheet for all leases. At the time a leased asset becomes available for the Group’s use, a right-of-use asset and corresponding lease liability are recognized in the balance sheet. Lease accounting is applied at the lowest component and the Group analyzes whether the lease contains more than one component.
In determining the term of the lease, all facts and circumstances are taken into account that provide an economic incentive to exercise extension options. If extension options are exercised with sufficient certainty, they are taken into account when determining the term of the contract.
Right-of-use assets are measured at cost, which is calculated as the lease liability plus lease payments made at or before the date on which the asset is made available for use, less lease incentives received and additional initial direct costs and dismantling obligations. Subsequent measurement of right-of-use assets is at amortized cost. The right-of-use assets are amortized on a straight-line basis over the shorter of either the useful life or the term of the lease agreement and the amortization is recognized in profit or loss. The useful economic lives of the right-of-use assets are regularly revised.
The lease liability is the present value of the fixed and variable lease payments that are paid during the term of the lease less any lease incentives receivable. The discounting is carried out based on the implied interest rate underlying the lease contract if the rate can be determined. If not, discounting is carried out based on the lessee’s incremental borrowing rate, i.e., the interest rate a lessee would need to pay to borrow over a similar term, and with a similar security, the funds necessary to obtain an asset of similar value and condition to the right-of-use asset in a similar economic environment.
In subsequent measurement, the carrying amount of the lease liability is increased to reflect the interest expense on the lease liability and reduced to reflect the lease payments made. Each lease installment is separated into a repayment portion and a financing expense portion. Finance expenses are recognized in profit or loss over the term of the lease.
The Group is exposed to potential future increases in variable lease payments based on an index or rate, which are not included in the lease liability until they take effect. When adjustments to lease payments based on an index or rate take effect, the lease liability is reassessed and adjusted against the right-of-use asset.
F-25
The payments for the redemption of lease liabilities and the payments attributable to the interest portion of the lease liabilities are allocated to cash flow from financing activities.
For low-value leases and short-term leases (terms of less than twelve months), mainly technical equipment, use is made of the simplified application. Accordingly, no right-of-use assets or lease liabilities are recognized; instead, the lease payments are recognized as an expense over the term of the lease.
Impairment losses are recognized in accordance with the principles described in Note 2.6.9.
2.6.7 Intangible Assets
Purchased intangible assets are capitalized at acquisition cost and exclusively amortized on a straight-line basis over their useful lives. Internally generated intangible assets are recognized to the degree the corresponding recognition criteria are met.
Development costs are capitalized as intangible assets when the corresponding capitalization criteria have been met, namely, clear specification of the product or procedure, technical feasibility, intention of completion, use, commercialization, coverage of development costs through future free cash flows, reliable determination of these free cash flows and availability of sufficient resources for completion of development and sale. Amortization of intangible assets is recorded in cost of sales or research and development expenses.
Expenses to be classified as research expenses are allocated to research and development expenses.
Subsequent expenditures for capitalized intangible assets are capitalized only when they substantially increase the future economic benefit of the specific asset to which they relate. All other expenditures are expensed as incurred.
Patents
Patents obtained by the Group are recorded at acquisition cost less accumulated amortization (see below) and any impairment (see Note 2.6.9). Patent costs are amortized on a straight-line basis over the lower of the estimated useful life of the patent (ten years) or the remaining patent term. Amortization starts when the patent is issued.
Licenses
The Group has acquired license rights from third parties by making upfront license payments, paying annual fees to maintain the license and paying fees for sublicenses. The Group amortizes upfront license payments on a straight-line basis over the estimated useful life of the acquired license (8 to 13 years). The amortization period and method are reviewed at the end of each financial year. Sublicense fees are amortized on a straight-line basis over the term of the contract or the estimated useful life of the collaboration for contracts without a set duration.
Licenses for Marketed Products
The balance sheet item contains prepaid license fees and milestone payments for Monjuvi® that are subsequently paid after the milestones have been reached. The Group amortizes those payments over the estimated useful life of the acquired license. The duration and method of amortization are reviewed at the end of each financial year. In the case of triggering events, the asset is tested for any impairment. Because the Group applies the cost accumulation approach, milestones in the near future are not taken into account.
In-Process R&D Programs
This line item contains capitalized payments from the in-licensing of compounds, as well as milestone payments for these compounds subsequently paid as milestones were achieved. Additionally, intangible assets identified in a business combination are included in this balance sheet item. No market approvals have been granted for those compounds.
Internally Generated Intangible Assets
Certain development costs related to tafasitamab and Monjuvi® have been capitalized as internally generated intangible assets, as the recognition criteria, as stated above, are met. The development of these assets is currently not yet completed and therefore they are not yet subject to amortization. Until the development activities are completed, the capitalized assets will undergo an annual impairment test.
F-26
Software
Software is recorded at acquisition cost less accumulated amortization (see below) and any impairment (see Note 2.6.9). Amortization is recognized in profit or loss on a straight-line basis over the estimated useful life of three to five years. Software is amortized from the date the software is operational.
| Intangible Asset Class | Useful Life | Amortization Rates | ||||||||||||
| Patents | % | |||||||||||||
| Licenses and Licenses for Marketed Products | ||||||||||||||
| In-process R&D Programs and Internally Generated Intangible Assets | Not yet amortized, Impairment Only | — | ||||||||||||
| Software | ||||||||||||||
2.6.8 Goodwill
Goodwill is recognized from business combinations. Goodwill is tested annually for impairment (see Notes 2.6.9 and 4.11).
2.6.9 Impairment of Non-Financial Assets
The carrying amounts of the Group’s non-financial assets and inventories are reviewed at each reporting date for any indication of impairment. The non-financial asset’s recoverable amount and the inventory’s net realizable value are estimated if such indication exists. For goodwill and intangible assets that have indefinite useful lives or are not yet available for use, the recoverable amount is estimated at the same time each year or determined on an interim basis, if required. Impairment is recognized if the carrying amount of an asset or the cash-generating unit (CGU) exceeds its estimated recoverable amount.
The recoverable amount of an asset or CGU is the greater of its value-in-use or its fair value less the cost of disposal. In assessing value-in-use, the estimated future pre-tax cash flows are discounted to their present value using a pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset or CGU. For the purposes of impairment testing, assets that cannot be tested individually are grouped into the smallest group of assets that generates cash flows from ongoing use that are largely independent of the cash flows of other assets or CGUs. Goodwill acquired in a business combination may be allocated to groups of CGUs that are expected to benefit from the combination’s synergies.
The Group’s corporate assets do not generate separate cash flows and are utilized by more than one CGU. Corporate assets are allocated to CGUs on a reasonable and consistent basis and are tested for impairment as part of the impairment testing of the CGU that was allocated the corporate asset.
2.6.10 Accounts Payable, Accruals and Provisions
Accounts payable are presented in Note 2.6.1 under financial liabilities at amortized cost.
Accruals and provisions are recognized for obligations to third parties arising from past events that are uncertain in their timing or amount. Furthermore, accruals and provisions are only recognized for legal or factual obligations to third parties if the event’s occurrence is more likely than not. Accruals and provisions are recognized in the amount required to settle the respective obligation and discounted to the reporting date when the interest effect is material. The amount required to meet the obligation also includes expected price and cost increases. The interest portion of the addition to accruals and provisions is recorded in the finance result. The measurement of accruals and provisions is based on past experience and considers the circumstances in existence on the reporting date. These non-financial liabilities with a maturity of more than one year are discounted to their present value. The difference between accruals and provisions is generally due to significantly less uncertainty in the amount and timing of the accrued liabilities.
F-27
2.6.11 Contract Liabilities
Upfront payments from customers for services to be rendered by the Group and revenue that must be recognized over a period of time are deferred and measured at the nominal amount of cash received. For current contract liabilities, the corresponding rendering of services and revenue recognition is expected to occur within a twelve-month period following the reporting date.
2.6.12 Tax Liabilities
Tax liabilities are recognized and measured at their nominal value. Tax liabilities contain obligations from current taxes, excluding deferred taxes. Liabilities for trade taxes, corporate taxes and similar taxes on income are determined based on the taxable income of the consolidated entities less any prepayments made.
2.6.13 Deferred Taxes
Deferred tax assets and liabilities are calculated using the liability method, which is commonly used internationally. Under this method, taxes expected to be paid or recovered in subsequent financial years are based on the applicable tax rate at the time of recognition.
Deferred tax assets and liabilities are recorded separately in the balance sheet and take into account the future tax effect resulting from temporary differences between carrying amounts in the balance sheet for assets and liabilities and tax loss carryforwards.
Deferred tax assets are offset against deferred tax liabilities when the taxes are levied by the same taxation authority and their maturity and the entity has a legally enforceable right to offset current tax assets against current tax liabilities. Deferred tax assets and liabilities may not be discounted.
Deferred tax assets on loss carryforwards and temporary differences are recognized and measured on the basis of projected future taxable income. They are only recognized if sufficient taxable income is available in the future to utilize the deferred tax assets.
In assessing the recoverability of deferred tax assets, only the effects on earnings of the reversal of temporary differences arising from deferred tax liabilities and the planned results from operating activities are taken into account. The planned results are based on internal forecasts of the future earnings situation of the respective Group company for the assessment of recoverability in the case of loss carryforwards and the long-term planning of the respective company for the assessment of recoverability in the case of temporary differences. If there are doubts about the realizability of the loss carryforwards, no corresponding deferred tax assets are recognized in individual cases, or deferred tax assets already recognized are impaired. The tax deferrals recognized are subject to ongoing reviews of the underlying assumptions. Changes in assumptions or circumstances may necessitate adjustments, which may result in additional tax deferrals or their reversal. Deferred tax assets and liabilities are offset if they relate to the same tax authority, and the right to offset current tax assets and liabilities is legally enforceable. Deferred tax assets and liabilities are recognized on an undiscounted basis. If the items underlying the temporary differences, or tax expenses and income respectively, are recognized directly in equity respectively in other comprehensive income, this also applies to the current taxes or deferred tax assets and liabilities attributable thereto.
2.6.14 Stockholders' Equity
Common Stock
Ordinary shares are classified as stockholders’ equity. Incremental costs directly attributable to the issue of ordinary shares are recognized as a deduction from stockholders’ equity.
Treasury Stock
Repurchases of the Company’s own shares at prices quoted on an exchange or at market value are recorded in this line item as a deduction from common stock.
When common stock recorded as stockholders’ equity is repurchased, the amount of consideration paid, including directly attributable costs, is recognized as a deduction from stockholders’ equity net of taxes and classified as treasury shares. When treasury shares are subsequently sold or reissued, the proceeds are recognized as an increase in stockholders’ equity, and any difference between the proceeds from the transaction and the initial acquisition costs is recognized in additional paid-in capital.
The allocation of treasury shares to beneficiaries under long-term incentive plans (in this case: performance shares) is reflected in this line item based on the set number of shares to be allocated after the expiration of the four-year vesting period (quantity structure) and multiplied by the weighted-average purchase price of the treasury shares (value structure). The adjustment is
F-28
carried out directly in equity through a reduction in the line item “treasury stock,” which is a deduction from common stock, while simultaneously reducing additional paid-in capital. Further information can be found in Note 5.1.
Additional Paid-In Capital
Additional paid-in capital mainly consists of personnel expenses resulting from the grant of share-based payments, the conversion option of the convertible bonds classified as equity, as well as the proceeds from newly created shares in excess of their nominal value.
Other Comprehensive Income Reserve
The line item “Other Comprehensive Income Reserve” includes changes in the fair value of equity instruments that are recognized in other comprehensive income and currency exchange differences that are not recognized in profit or loss.
Accumulated Deficit
The “Accumulated Deficit” line item consists of the Group’s accumulated consolidated net profits/losses. A separate measurement of this item is not made.
3 Notes to the Statement of Profit or Loss
3.1 Revenues and Revenues Recognition
| in 000' € | 2023 | 2022 | 2021 | ||||||||||||||
| Product Sales, Net | |||||||||||||||||
| Royalties | |||||||||||||||||
| License Fees | |||||||||||||||||
| Milestone Payments | |||||||||||||||||
| Service Fees | |||||||||||||||||
| Other | |||||||||||||||||
| Licenses, Milestones and Other | |||||||||||||||||
| Total | |||||||||||||||||
The following overview shows the Group’s regional distribution of revenue on the basis of the customer location:
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Germany | ||||||||||||||||||||
| Europe (excluding Germany) | ||||||||||||||||||||
| Asia | ||||||||||||||||||||
| USA | ||||||||||||||||||||
| Total | ||||||||||||||||||||
Of the sales generated in Europe in 2023, a total of € 25.6 million is attributable to Switzerland (2022: € 28.0 million to Switzerland; 2021: € 16.0 million to the United Kingdom).
The following overview shows the timing of the satisfaction of performance obligations:
| in 000' € | 2023 | 2022 | 2021 | ||||||||||||||
| At a Point in Time | |||||||||||||||||
| Over Time | |||||||||||||||||
| Total | |||||||||||||||||
Of the total revenues generated in 2023, a total of € 119.2 million were recognized from performance obligations that were fulfilled in previous periods and related to milestone payments and royalties (2022: € 103.1 million; 2021: € 85.5 million).
In 2023 the revenues from Service Fees are presented in the category "Over Time" for the satisfaction of the performance obligation. In 2022 and 2021 the revenue recognition criteria relating to the Service Fees was also "Over Time", however were shown in our notes disclosures as "At a Point in Time", which has now been corrected in 2023.
F-29
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
3.2 Cost of Sales
Cost of sales consisted of the following:
| in 000' € | 2023 | 2022 | 2021 | ||||||||||||||
| Expensed Acquisition or Production Cost of Inventories | |||||||||||||||||
| Personnel Expenses | |||||||||||||||||
| Impairment (+) and Reversals of Impairment (-) on Inventories | |||||||||||||||||
| Impairment, Amortization and Other Costs of Intangible Assets | |||||||||||||||||
| External Services | |||||||||||||||||
| Depreciation and Other Costs for Infrastructure | |||||||||||||||||
| Other Costs | |||||||||||||||||
| Total | |||||||||||||||||
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
3.3 Operating Expenses
3.3.1 Research and Development Expenses
Research and development expenses consisted of the following:
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Personnel Expenses | ||||||||||||||||||||
| Consumable Supplies | ||||||||||||||||||||
| Impairment, Amortization and Other Costs of Intangible Assets | ||||||||||||||||||||
| External Services | ||||||||||||||||||||
| Depreciation and Other Costs for Infrastructure | ||||||||||||||||||||
| Other Costs | ||||||||||||||||||||
| Total | ||||||||||||||||||||
3.3.2 Selling Expenses
Selling expenses consisted of the following:
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Personnel Expenses | ||||||||||||||||||||
| Consumable Supplies | ||||||||||||||||||||
| Amortization of Intangible Assets | ||||||||||||||||||||
| External Services | ||||||||||||||||||||
| Depreciation and Other Costs for Infrastructure | ||||||||||||||||||||
| Other Costs | ||||||||||||||||||||
| Total | ||||||||||||||||||||
F-30
3.3.3 General and Administrative Expenses
General and administrative expenses consisted of the following:
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Personnel Expenses | ||||||||||||||||||||
| Consumable Supplies | ( | |||||||||||||||||||
| Amortization of Intangible Assets | ||||||||||||||||||||
| External Services | ||||||||||||||||||||
| Depreciation and Other Costs for Infrastructure | ||||||||||||||||||||
| Other Costs | ||||||||||||||||||||
| Total | ||||||||||||||||||||
3.3.4 Personnel Expenses
Personnel expenses consisted of the following:
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Wages and Salaries | ||||||||||||||||||||
| Social Security Contributions | ||||||||||||||||||||
| Share-based Payment Expense | ||||||||||||||||||||
| Other | ||||||||||||||||||||
| Total | ||||||||||||||||||||
The increase in share-based payment expenses is mainly due to the increase in the share price of MorphoSys AG as of December 31, 2023, compared to the previous year (December 31, 2023: €34.00 ; December 31, 2022: €13.21 ), which is used for the valuation of the share-based payment programs.
The cost of defined contribution plans amounted to € 3.2 million in 2023 (2022: € 4.3 million; 2021: € 2.8 million).
The following average number of employees were employed in the various functions in recent fiscal years.
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Production | ||||||||||||||||||||
| Research and Development | ||||||||||||||||||||
| Selling | ||||||||||||||||||||
| General and Administrative | ||||||||||||||||||||
| Total | ||||||||||||||||||||
At December 31, 2023 the number of employees amounted to 524 (December 31, 2022: 629 ; December 31, 2021: 732 ).
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
3.3.5 Impairment of Goodwill
In the financial year 2023, an impairment of € 1.6 million was recognized on goodwill, which initially resulted from an acquisition in financial year 2010 (2022 € 0.0 million ; 2021: € 230.7 million).
F-31
3.4 Other Income and Expenses, Finance Income and Finance Expenses
The other income is shown in the following overview.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Gain on Foreign Exchange | ||||||||||||||||||||
| Grant Income | ||||||||||||||||||||
| Income from Other Items | ||||||||||||||||||||
| Other Income | ||||||||||||||||||||
The other expenses are shown in the following overview.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Loss on Foreign Exchange | ( | ( | ( | |||||||||||||||||
| Expenses from Other Items | ( | ( | ( | |||||||||||||||||
| Other Expenses | ( | ( | ( | |||||||||||||||||
The finance income is shown in the following overview.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Foreign Exchange Gains | ||||||||||||||||||||
| Gains from Measurement at Fair Value | ||||||||||||||||||||
| Income from Carrying Amount Adjustments of Financial Liabilities at Amortized cost | ||||||||||||||||||||
| Income from Sale of Shares | ||||||||||||||||||||
| Interest Income | ||||||||||||||||||||
| Gain from Repurchase of own Convertible Bonds | ||||||||||||||||||||
| Finance Income | ||||||||||||||||||||
The finance expenses are shown in the following overview.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Foreign Exchange Losses | ( | ( | ( | |||||||||||||||||
| Losses from Measurement at Fair Value | ( | ( | ( | |||||||||||||||||
| Effective Interest Expenses from Financial Liabilities at Amortized Cost | ( | ( | ( | |||||||||||||||||
| Expenses from Carrying Amount Adjustments of Financial Liabilities at Amortized cost | ( | ( | ( | |||||||||||||||||
| Other Interest Expenses | ( | ( | ( | |||||||||||||||||
| Interest Expenses on Lease Liabilities | ( | ( | ( | |||||||||||||||||
| Bank Fees | ( | ( | ( | |||||||||||||||||
| Finance Expenses | ( | ( | ( | |||||||||||||||||
The explanation of the main components of financial income and financial expenses can be found in Note 4.5, 4.19 and 4.20 of these notes.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
3.5 Income Tax Benefit / Expenses
MorphoSys AG is subject to corporate taxes, the solidarity surcharge and trade taxes. The Company’s corporate income tax rate in the reporting year remained unchanged (15.0 %), as did the solidarity surcharge (5.5 %) and the effective trade tax rate (10.85 %), resulting in a combined tax rate of 26.675 %.
F-32
The U.S. tax group, comprising of MorphoSys US Inc. and Constellation is subject to Federal Corporate Income Tax of 21.0 % and State Income Tax. State Income Taxes reflected a mix of various state tax rates and resulted in an average state tax rate of 6.38 %.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
Current Tax Benefit / (Expense) (Thereof Regarding Prior Years: kEUR | ( | |||||||||||||||||||
| Deferred Tax Benefit / (Expenses) | ( | ( | ||||||||||||||||||
| Total Income Tax Benefit / (Expenses) | ( | |||||||||||||||||||
The Group recognized a total income tax benefit of € 1.2 million in the reporting year 2023. This consisted of a deferred tax expense of € 0.3 million , as well as € 1.5 million current tax benefit, which mainly result from tax allowances recognized for prior years.
The following table reconciles the expected income tax expense to the actual income tax expense as presented in the consolidated financial statements. The combined income tax rate of 26.675 % in the 2023 financial year (2022: 26.675 %; 2021: 26.675 %) was applied to profit before taxes to calculate the statutory income tax expense. This rate consisted of a corporate income tax of 15.0 %, a solidarity surcharge of 5.5 % on the corporate tax, and an average trade tax of 10.85 % applicable to the Group.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Earnings Before Income Taxes | ( | ( | ||||||||||||||||||
| Expected Tax Rate | % | % | % | |||||||||||||||||
| Expected Income Tax | ( | |||||||||||||||||||
| Tax Effects Resulting from: | ||||||||||||||||||||
| Share-based Payment | ( | ( | ( | |||||||||||||||||
| Permanent Differences | ( | ( | ||||||||||||||||||
| Non-Tax-Deductible Items | ( | ( | ( | |||||||||||||||||
| Non-taxable income | ||||||||||||||||||||
| Derecognition of Deferred Tax Assets on Temporary Differences | ( | ( | ( | |||||||||||||||||
| Derecognition of Deferred Tax Assets on Tax Losses | ( | ( | ( | |||||||||||||||||
| Tax Rate Differences to Local Tax Rates | ( | ( | ( | |||||||||||||||||
| Prior Year Taxes | ||||||||||||||||||||
| Other Effects | ( | ( | ||||||||||||||||||
| Actual Income Tax | ( | |||||||||||||||||||
| Effective Tax Rate | % | % | % | |||||||||||||||||
The permanent differences as of December 31, 2021, related exclusively to the impairment of goodwill.
As of December 31, 2023, the group companies are still in a history of losses. Therefore, the increased requirement for the impairment test according to IAS 12.35 was applied unchanged as in the previous year. In this context, the existence of other substantial indications is required that in the future the availability of corresponding taxable income is no longer only probable, but sufficiently certain. Taking into account these increased recognition requirements, it could not be demonstrated with certainty for the long-term planning period of the Company that corresponding positive tax planning results will be available to ensure the recoverability of the deferred tax assets on temporary differences or tax loss carryforwards. For this reason, deferred tax assets were only capitalized to the extent that they will be offset against the scheduled reversal of deferred tax liabilities in the future and otherwise not recognized or impaired.
Due to the history of losses and the current uncertainties regarding the realization of planned taxable income, corresponding deferred tax assets on loss carry forwards were only recognized as outlined in the following table.
F-33
| in 000' € | Carry-Forward of Tax Losses | |||||||
| Tax Losses from Prior Years | ||||||||
| Tax Losses from Current Year | ||||||||
| Foreign Currency Translation Differences | ( | |||||||
| Total Tax Losses as of December 31, 2023 | ||||||||
| Expected Deferred Tax Assets on Total Tax Losses | ||||||||
| Derecognition of Deferred Tax Assets on Tax Losses | ( | |||||||
| Deferred Tax Assets on Tax Losses | ||||||||
The tax losses as of December 31, 2023, include losses of € 150.0 million with a limited utilization period, which relate to the U.S. tax group and forfeit from 2027 until 2036. The deferred tax assets on temporary differences, which have not been capitalized in the period, amount to € 15.8 million.
Deferred tax liabilities are presented as non-current items in the consolidated statements of financial position. Deferred tax assets and deferred tax liabilities consisted of the following:
| in 000’s €, as of December 31 | Deferred Tax Asset 2023 | Deferred Tax Asset 2022 | Deferred Tax Liability 2023 | Deferred Tax Liability 2022 | ||||||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | ||||||||||||||||||||||||||
| Bonds | ||||||||||||||||||||||||||
| Leases | ||||||||||||||||||||||||||
| Intangible Assets | ||||||||||||||||||||||||||
| Receivables and Other Assets | ||||||||||||||||||||||||||
| Property, Plant and Equipment | ||||||||||||||||||||||||||
| Provisions | ||||||||||||||||||||||||||
| Other Liabilities | ||||||||||||||||||||||||||
| Tax Losses | ||||||||||||||||||||||||||
| Offsetting | ( | ( | ( | ( | ||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||
| Changes in Deferred Taxes in 2023 | ||||||||||||||
| in 000' € | Income / (Expense) | Direct Recognition in Equity | ||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | ||||||||||||||
| Bonds | ||||||||||||||
| Leases | ( | |||||||||||||
| Intangible Assets | ||||||||||||||
| Receivables and Other Assets | ||||||||||||||
| Property, Plant and Equipment | ||||||||||||||
| Provisions | ( | |||||||||||||
| Other Liabilities | ( | |||||||||||||
| Tax Losses | ( | |||||||||||||
| Foreign Currency Translation Differences | ( | |||||||||||||
| Total | ( | |||||||||||||
As of December 31, 2023 there were no deferred tax items recognized against equity (2022: € 0.0 million ; 2021: € 0.0 million ).
F-34
3.6 Earnings per Share
Basic earnings per share are calculated by dividing the 2023 consolidated net loss of € 189,734,199 (2022: consolidated net loss of € 151,058,190 ; 2021: consolidated net loss of € 514,460,016 ) by the weighted-average number of ordinary shares outstanding during the respective year (2023: 34,312,744 ; 2022: 34,155,650 ; 2021: 33,401,069 ).
Diluted earnings per share is calculated by taking into account the potential increase in the Group’s ordinary shares as the result of granted stock options, restricted stock units and convertible bonds.
The following table shows the reconciliation of basic earnings per share to diluted earnings per share (in €, except for disclosures in shares).
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Numerator (in €) | ||||||||||||||||||||
| Consolidated Net Profit / (Loss) - used in calculating Basic Earnings per Share | ( | ( | ( | |||||||||||||||||
| Profit used in calculating Diluted Earnings per Share | ( | ( | ( | |||||||||||||||||
| Denominator (in Shares) | ||||||||||||||||||||
| Weighted average Ordinary Shares Used in Calculating Basic Earnings per Share | ||||||||||||||||||||
| Weighted average Ordinary Shares and potential Ordinary Shares Used in Calculating Diluted Earnings per Share | ||||||||||||||||||||
| Earnings per Share (in €) | ||||||||||||||||||||
| Basic | ( | ( | ( | |||||||||||||||||
| Diluted | ( | ( | ( | |||||||||||||||||
The 630,104 restricted stock units are still unvested as of December 31, 2023 and the 1,996,344 shares from the convertible bonds are potentially dilutive shares for 2023, but excluded from the calculation of dilutive earnings per share as it would result in a decline in the loss per share.
4 Notes to the Balance Sheet
4.1 Cash and Cash Equivalents
| in 000' € | 12/31/2023 | 12/31/2022 | |||||||||
| Bank Balances and Cash in Hand | |||||||||||
| Impairment | ( | ( | |||||||||
| Cash and Cash Equivalents | |||||||||||
The presentation of the development of the expected twelve-month loss for cash and cash equivalents can be found in Note 6.4.1.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
F-35
4.2 Other Financial Assets
Other Financial Assets include, on the one hand, money market funds classified as FVTPL and on the other hand term deposits and bonds classified as AC.
The financial assets at fair value, with changes recognized in profit or loss, are following.
| Unrealized | ||||||||||||||||||||||||||||||||
| in 000' € | Maturity | Cost | Gross Profit | Losses | Market Value | |||||||||||||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||||||||||||||
| Money Market Funds | daily | |||||||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||||||||||||||
| Money Market Funds | daily | |||||||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||||||||
Details on the fair value hierarchy and the measurement methods for Financial Assets from Escrow Accounts can be found in Note 6.3. As of December 31, 2023, the escrow account amounted to € 0.8 million (December 31. 2022: € 0.0 ).
Realized and unrealized gains and losses on money market funds were recognized in the finance result in profit or loss. The valuation of money market funds resulted in a net gain of € 6.7 million in 2023 (2022: net gain of € 0.2 million; 2021: net gain of € 0.6 million).
The financial assets at amortized cost are shown in the following overview.
| in 000’ € | Maturity | Cost | Effective Interest Income (+) / Expense (-) | Impairment | Carrying Amount | |||||||||||||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||||||||||||||
| Term Deposits, Current Portion | 1 to 12 months | ( | ||||||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||||||||||||||
| Term Deposits, Current Portion | 1 to 12 months | ( | ||||||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||||||||
As of December 31, 2023, these assets mainly consisted of term deposits with fixed or variable interest rates.
Net interest income from financial assets classified as “at amortized cost” amounted to € 18.3 million in 2023 (2022: € 3.0 million net interest expense; 2021: € 1.7 million net interest expense) and was recognized in the finance result.
The risk associated with these financial instruments results primarily from bank credit risks. Further information on the credit risk for term deposits and corporate bonds can be found in Note 6.4.1.
4.3 Accounts Receivable
All accounts receivable are non-interest-bearing and generally have payment terms of between 20 and 66 days. As of December 31, 2023, and as of December 31, 2022, accounts receivable mainly consisted of receivables against Incyte from shared development costs as well as receivables from Monjuvi® product sales.
As of December 31, 2023, a total of € 14.4 million of the carrying amount of accounts receivables was attributable to the single Customer Incyte (December 31, 2022: € 51.4 million), or 45 % of the Group‘s total accounts receivable at the end of 2023 (December 31, 2022: 56 %).
F-36
The table below shows the accounts receivable by region as of the reporting date.
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Germany | ||||||||||||||
| Europe (excluding Germany) | ||||||||||||||
| Asia | ||||||||||||||
| USA | ||||||||||||||
| Impairment | ( | ( | ||||||||||||
| Total | ||||||||||||||
The presentation of the development of the risk provisions in the 2023 and 2022 financial years for accounts receivable using the simplified impairment model can be found in Note 6.4.1.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
4.4 Income Tax Receivables
As of December 31, 2023, income tax receivables amounted to € 5.3 million (December 31, 2022: € 2.6 million). These mainly comprised tax refund claims from tax allowances and withheld capital gains tax, which were recognized at nominal value.
4.5 Other Receivables
Other receivables as of December 31, 2023, mainly consisted of receivables from creditors with debit accounts in the amount of € 1.0 million (December 31, 2022: € 2.0 million). After the recognition of the change in fair value in the amount of € 4.3 million of the anti-dilution right from the HI-Bio acquisition and the executed financing round in December 2023, the anti-dilution right was fully utilized and hence the balance of the anti-dilution right as of December 31, 2023, was reduced to € 0.0 million (December 31, 2022: € 9.8 million). Further details can be found in Note 4.12.
The anti-dilution right was measured FVTPL and its measurement was based in part on unobservable parameters. This resulted in a fair value classification in the Level 3 valuation hierarchy. The planning assumptions underlying the valuation was influenced by estimates derived from the business valuation of HI-Bio. As of December 31, 2023, the anti-dilution right was fully used and hence balance sheet item was 0 €.
The anti-dilution right changed in 2023 and 2022 as follows.
| in 000' € | 2023 | 2022 | ||||||||||||
| Balance as of January 1 | ||||||||||||||
| Additions | ||||||||||||||
| Gains/(losses) recognized in profit or loss statement | ( | ( | ||||||||||||
| Reclassification to investment in associates | ( | ( | ||||||||||||
| Balance as of December 31 | ||||||||||||||
As of December 31, 2023 and December 31, 2022, there were no
4.6 Inventories
The table below shows inventories as of the reporting date.
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Raw materials, Supplies and Production Materials | ||||||||||||||
| Finished Goods | ||||||||||||||
| Impairment | ( | |||||||||||||
| Total | ||||||||||||||
As part of the assessment of the net realizable value test for the inventory, the Company performed an assessment of the shelf-life of the product on stock benchmarked against the most recent demand forecast. This analysis led to an impairment and
F-37
respective losses are presented in cost of sales in the income statement. An impairment loss of € 7.4 million had to be recognized in 2023 (2022: € 0.0 million ). Included in the value of the "Finished Goods" is an amount of € 19.4 million of drug substance which has already been prepaid by the customer. For details refer to 4.16 "Contract Liabilities."
The increase in "Raw materials and supplies" is mainly due to the purchase of drug substance during the year, which is the basis for further drug product production.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
4.7 Prepaid Expenses and Other Assets
The current prepaid expenses and other assets are shown in the following table.
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Receivables due from Tax Authorities from Input Tax Surplus | ||||||||||||||
| Prepayments for External Laboratory Services | ||||||||||||||
| Prepayments for Sublicenses | ||||||||||||||
| Other Prepayments | ||||||||||||||
| Total | ||||||||||||||
"Other Prepayments" mainly include payments made in advance for raw materials and supplies required for the production of tafasistamab as well as for maintenance contracts, insurances and sublicenses. The decrease compared to the previous year is mainly due to lower prepayments for external laboratory services and consumables in connection with the production of tafasitamab.
The non-current prepaid expenses and other assets are shown in the following table.
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Prepaid Expenses | ||||||||||||||
| Other Assets | ||||||||||||||
| Total | ||||||||||||||
The non-current prepaid expenses mainly include prepayments for external services that will be utilized from 2025 onwards.
The Group has classified certain items within other assets as “restricted cash” that is not available for operational purposes of the Group. As of December 31, 2023, the Group had non-current restricted cash of € 1.0 million for rental deposits issued (December 31, 2022: € 1.1 million). As of December 31, 2023, € 0.2 million were deposited as collateral for credit cards by MorphoSys US Inc. (December 31, 2022: € 0.2 million).
F-38
4.8 Property, Plant and Equipment
| in 000' € | Office and Laboratory Equipment | Furniture and Fixtures | Total | |||||||||||||||||
| Cost | ||||||||||||||||||||
| January 1, 2022 | ||||||||||||||||||||
| Additions | ||||||||||||||||||||
| Disposals | ( | ( | ( | |||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||
| Accumulated Depreciation and Impairment | ||||||||||||||||||||
| January 1, 2022 | ||||||||||||||||||||
| Depreciation Charge for the Year | ||||||||||||||||||||
| Impairment | ||||||||||||||||||||
| Disposals | ( | ( | ( | |||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||
| Carrying Amount | ||||||||||||||||||||
| January 1, 2022 | ||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||
| Cost | ||||||||||||||||||||
| January 1, 2023 | ||||||||||||||||||||
| Additions | ||||||||||||||||||||
| Disposals | ( | ( | ( | |||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | ( | ( | |||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||
| Accumulated Depreciation and Impairment | ||||||||||||||||||||
| January 1, 2023 | ||||||||||||||||||||
| Depreciation Charge for the Year | ||||||||||||||||||||
| Disposals | ( | ( | ||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | ( | ( | |||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||
| Carrying Amount | ||||||||||||||||||||
| January 1, 2023 | ||||||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||
No borrowing costs were capitalized during the reporting period, and there were neither restrictions on the retention of title nor property, plant and equipment pledged as security for liabilities. There were no material contractual commitments for the purchase of property, plant and equipment as of the reporting date.
Depreciation is contained in the following line items of profit or loss.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Research and Development | ||||||||||||||||||||
| Research and Development (Impairment) | ||||||||||||||||||||
| Selling | ||||||||||||||||||||
| General and Administrative | ||||||||||||||||||||
| Total | ||||||||||||||||||||
F-39
4.9 Leases
The development of the right-of-use assets and lease liabilities is shown below.
Right-of-Use Assets | Lease Liabilities | |||||||||||||||||||||||||||||||
| in 000' € | Building | Cars | Technical Equipment | Total | ||||||||||||||||||||||||||||
| Balance as of January 1, 2022 | ||||||||||||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||||||||
| Depreciation of Right-of-Use Assets | ( | ( | ( | ( | 0 | |||||||||||||||||||||||||||
| Interest Expenses on Lease Liabilities | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||
| Lease Payments | 0 | 0 | 0 | 0 | ( | |||||||||||||||||||||||||||
| Disposals | ||||||||||||||||||||||||||||||||
| Value adjustment | ||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ||||||||||||||||||||||||||||||||
| Balance as of December 31, 2022 | ||||||||||||||||||||||||||||||||
| Balance as of January 1, 2023 | ||||||||||||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||||||||
| Depreciation of Right-of-Use Assets | ( | ( | ( | ( | 0 | |||||||||||||||||||||||||||
| Interest Expenses on Lease Liabilities | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||
| Lease Payments | 0 | 0 | 0 | 0 | ( | |||||||||||||||||||||||||||
| Disposals | ( | ( | ||||||||||||||||||||||||||||||
| Value adjustment | ( | ( | ( | ( | ||||||||||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | ( | ( | ( | ||||||||||||||||||||||||||||
| Balance as of December 31, 2023 | ||||||||||||||||||||||||||||||||
Lease agreements had the following effects on the statement of profit or loss.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Depreciation of Right-of-Use Assets | ||||||||||||||||||||
| Depreciation of Right-of-Use Assets (Change of Useful Life) | ||||||||||||||||||||
| Interest Expenses on Lease Liabilities | ||||||||||||||||||||
| Expenses for Short Term Leases | ||||||||||||||||||||
| Expenses for Leases of Low Value Assets | ||||||||||||||||||||
| Total | ||||||||||||||||||||
Depreciation of right-of-use assets is contained in the following line items of profit or loss.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Cost of Sales | ||||||||||||||||||||
| Research and Development | ||||||||||||||||||||
| Selling | ||||||||||||||||||||
| General and Administrative | ||||||||||||||||||||
| Total | ||||||||||||||||||||
The maturity analysis of the lease liabilities as of December 31, 2023, is as follows.
| December 31, 2023; in 000’ € | ||||||||||||||||||||||||||||||||
| Contractual Maturities of Financial Liabilities | Less than 1 Year | Between One and Five Years | More than 5 Years | Total Contractual Cash Flows | Carrying Amount Liabilities | |||||||||||||||||||||||||||
| Lease Liabilities | ||||||||||||||||||||||||||||||||
The rental conditions for leases are negotiated individually and include different terms. Leases are generally concluded for fixed periods but may include extension options. Such contractual conditions offer the Group the greatest possible operational flexibility. In determining the term of the lease, all facts and circumstances are taken into account that provide an economic
F-40
incentive to exercise extension options. If extension options are exercised with sufficient certainty, they are taken into account when determining the term of the contract. The leases contain fixed and variable lease payments linked to an index. As of December 31, 2023, potential future lease payments of € 25.3 million (discounted) were no longer included in the lease liabilities, as the company assumes that the option to extend the lease for an office building beyond the minimum lease term will no longer be exercised. The capitalized right-of-use asset was reduced accordingly.
4.10 Intangible Assets
| in 000' € | Patents | Licenses | Licenses for Marketed Products | In-process R&D Programs | Internally Generated Intangible Assets | Software | Total | |||||||||||||||||||||||||||||||||||||
| Cost | ||||||||||||||||||||||||||||||||||||||||||||
| January 1, 2022 | ||||||||||||||||||||||||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||||||||||||||||||||
| Disposals | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||||||||||||||||||||||||||
| Accumulated Amortization and Impairment | ||||||||||||||||||||||||||||||||||||||||||||
| January 1, 2022 | ||||||||||||||||||||||||||||||||||||||||||||
| Amortization Charge for the Year | ||||||||||||||||||||||||||||||||||||||||||||
| Impairment | ||||||||||||||||||||||||||||||||||||||||||||
| Disposals | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||
| Reclassification | ||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||||||||||||||||||||||||||
| Carrying Amount | ||||||||||||||||||||||||||||||||||||||||||||
| January 1, 2022 | ||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||||||||||||||||||||||||||
| Cost | ||||||||||||||||||||||||||||||||||||||||||||
| January 1, 2023 | ||||||||||||||||||||||||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||||||||||||||||||||
| Disposals | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||
| Accumulated Amortization and Impairment | ||||||||||||||||||||||||||||||||||||||||||||
| January 1, 2023 | ||||||||||||||||||||||||||||||||||||||||||||
| Amortization Charge for the Year | ||||||||||||||||||||||||||||||||||||||||||||
| Impairment | ||||||||||||||||||||||||||||||||||||||||||||
| Disposals | ||||||||||||||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||
| Carrying Amount | ||||||||||||||||||||||||||||||||||||||||||||
| January 1, 2023 | ||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||
There were no material contractual commitments for the purchase of intangible assets as of the reporting date.
In the financial year 2023 an impairment of the full amount was recorded for a license since it was decided to no longer pursue the underlying technology and also all efforts to materialize the value through out-licensing activities have not been successful.
F-41
Consequently, the license had to be impaired. In 2022 an impairment loss of € 7.8 million was recognized due to a management decision not to utilize production capacities at a manufacturer in the future.
Amortization was included in the following line items of profit or loss.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Cost of Sales | ||||||||||||||||||||
| Cost of Sales (Impairment) | ||||||||||||||||||||
| Research and Development | ||||||||||||||||||||
| Research and Development (Impairment) | ||||||||||||||||||||
| Selling | ||||||||||||||||||||
| General and Administrative | ||||||||||||||||||||
| Total | ||||||||||||||||||||
In-Process R&D Programs
Tafasitamab
As an intangible asset not yet available for use and a carrying amount of € 10.4 million, tafasitamab was subject to an annual impairment test on December 31, 2023, as required by IAS 36. This intangible asset represents a milestone payment for tafasitamab that was capitalized in 2021. This payment was made for an indication for which marketing authorization has not yet been granted.
The recoverable amount of the tafasitamab cash-generating unit was determined on the basis of value-in-use calculations, which concluded that the recoverable amount exceeded its carrying amount. The cash flow forecasts took into account expected cash inflows from the potential commercialization of tafasitamab, the cash outflows for anticipated research and development, and the costs for tafasitamab’s commercialization. The cash flow forecasts are based on the period of patent protection for tafasitamab. For this reason, a planning horizon of approximately 21 years is considered appropriate for the value-in-use calculation. The values of the underlying assumptions were determined using both internal (past experience) and external sources of information (market information). Based on the updated cash flow forecast, the value-in-use was determined as follows: A beta factor of 1.6 (2022: 1.0 ) and WACC before taxes of 15.9 % (2022: 11.4 %). A sensitivity analysis was performed for the discount rate. A sensitivity analysis for changes in the cash flows was not performed since the cash flows from research and development and the commercialization of the compound have already been probability adjusted in the value-in-use calculations so as to reflect the probabilities of success in phases of clinical trials. The analysis did not reveal any need for impairment. The values ascribed to the assumptions correspond to the Management Board’s forecasts for future development and are based on internal planning scenarios, as well as external sources of information.
No indicators of impairment were identified on December 31, 2023.
Pelabresib and tulmimetostat
As intangible assets not yet available for use and a carrying amount of together € 768.8 million, pelabresib (carrying amount € 766.7 million) and tulmimetostat (carrying amount € 2.1 million) were subject to an annual impairment test on December 31, 2023, as required by IAS 36. Pelabresib and tulmimetostat each constitute a cash-generating unit. The recoverable amount was determined on the basis of value-in-use calculations, which concluded that the recoverable amount exceeded its carrying amount. The cash flow forecasts took into account expected cash inflows (revenues based on patient numbers and the price obtained in the market) from the potential commercialization of pelabresib and tulmimetostat, the cash outflows for anticipated research and development, and the costs for the commercialization of pelabresib and tulmimetostat. The cash flow forecasts are based on the period of patent protection for pelabresib and tulmimetostat. For this reason, a planning horizon of approximately 21 years is considered appropriate for the value-in-use calculation. The values of the underlying assumptions were determined using both internal (past experience) and external sources of information (market information). Based on the updated cash flow forecast, the value-in-use was determined as follows: A beta factor of 1.6 (2022: 1.5 ) and WACC before taxes of 14.0 % (2022: 13.7 %).
A sensitivity analysis of the determined value-in-use was performed. This included the underlying estimates for the cash flow forecasts and for the discount rate. In each case, one planning assumption is changed and all other estimates are kept constant. The value-in-use would correspond to the carrying amount if the cash flow forecasts were reduced by 49 % or the discount rate were increased by 9.4 %. The values attributed to the assumptions correspond to the Management Board's assessment with regard to future developments and are based on internal planning scenarios as well as external sources.
F-42
Licenses for Marketed Products
Tafasitamab
Since the market approval of Monjuvi®, the compound is classified as an intangible asset with a finite useful life and amortized as of that date. The Group amortizes the intangible asset on a straight-line basis over the estimated useful life of the acquired license until 2044 and recognizes the amortization in cost of sales. The duration and method of amortization are reviewed at the end of each financial year. In the event of triggering events, the asset is tested for impairment, if any. As of December 31, 2023, no indications of impairment were identified.
Internally generated intangible assets
In 2021, it was decided to contract new manufacturers of tafasitamab. Related costs, including FTE and external costs, were capitalized as internally generated intangible assets. As of December 31, 2023, the carrying amount was € 14.5 million (December 31, 2022: € 16.9 million). As soon as the know-how transfer is successful and an associated certification has been obtained, amortization will commence.
There was an impairment in 2023 amounting to € 0.7 million (2022: € 7.8 million).The impairment in the prior year was based on a management decision not to utilize production capacities at a manufacturer in the future.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
4.11 Goodwill
Slonomics Technology
The goodwill from the acquisition of Sloning BioTechnology GmbH in 2010 was written off in full in financial year 2023, as management believes that the future cash flows from the contribution of Slonomics technology can no longer be realized. Accordingly, an impairment of goodwill in the amount of € 1.6 million was recognized in the income statement.
Constellation
As of December 31, 2023, goodwill of € 342.3 million from the acquisition of Constellation was subject to an impairment test. Goodwill was allocated to the group of cash-generating units Constellation, as goodwill is monitored at this level. In addition, future potential cash flows of this group of cash-generating units will only be generated by Constellation's own compounds, which are also recognized by these companies.
The recoverable amount of the group of cash-generating units Constellation was determined on the basis of value-in-use calculations. The calculation showed that the value-in-use was higher than the carrying amount of this group of cash-generating units.The cash flow projections included expected payments from the commercialization of pelabresib and other compounds, the cash outflows for anticipated research and development, and the costs for pelabresib’s and the other compounds' commercialization. The cash flow forecasts are based on the period of patent protection for pelabresib and the other compounds. For this reason, a planning horizon of approximately 21 years is considered appropriate for the value-in-use calculation. The values of the underlying assumptions were determined using both internal (past experience) and external sources of information (market information). Based on the cash flow forecast, the value-in-use was determined as follows: A beta factor of 1.6 (2022: 1.5 ) and WACC before taxes of 15.5 % (2022: 14.7 %).
A sensitivity analysis of the determined value-in-use was performed. This included the underlying estimates for the cash flow forecasts and for the discount rate. In each case, one planning assumption is changed and all other estimates are kept constant. The value-in-use would correspond to the carrying amount if the cash flow forecasts were reduced by 34 % or the discount rate were increased by 5.4 %. The values attributed to the assumptions correspond to the Management Board's assessment with regard to future developments and are based on internal planning scenarios as well as external sources.
4.12 Investment in Associates
As of December 31, 2023, MorphoSys AG holds a 12.1 % stake in Human Immunology Biosciences, Inc. ("HI-Bio"), based in San Francisco, California, USA (2022: 15.0 %). HI-Bio is a biotechnology company focused on the discovery and development of precision medicines for autoimmune and inflammatory diseases. HI-Bio is not publicly traded. MorphoSys obtained a 15.0% share in HI-Bio by making a contribution in kind of a license for felzartamab (MOR202) back in financial year 2022. The shareholding represents both the capital and the voting rights and takes into account the pro rata sale of shares in 2023 from
F-43
which € 4.6 million was received in cash. In addition to the shareholding, MorphoSys AG had the right to receive further shares (anti-dilution right). The right to receive further shares was recognized as fair value as a financial asset (refer to Note 4.5).
HI-Bio is accounted for in the consolidated financial statements using the equity method, as described in the Group's accounting policies (refer to Note 2.2.2 of these notes). This accounting treatment is due to the fact that, despite a shareholding of less than 20%, MorphoSys AG can exercise significant influence over HI-Bio. The relevant criteria for this are: representation of MorphoSys on the Board of Directors of HI-Bio and consequently participation in decision-making processes of HI-Bio, MorphoSys entered into significant transactions with HI-Bio, and MorphoSys has provided significant technical information to HI-Bio.
The following tables provide summarized financial information of the balance sheet and comprehensive income about the Group's investment in HI-Bio (including modifications due to differences in accounting policies). This reflects the status as of September 30, 2023, as this is the last available financial statement from HI-Bio as of the date of preparation of the MorphoSys consolidated financial statements.
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Current Assets | ||||||||||||||
| thereof Cash and Cash Equivalents | ||||||||||||||
| thereof Other Assets | ||||||||||||||
| Non-Current Assets | ||||||||||||||
| Current Liabilities | ||||||||||||||
| thereof Financial Liabilities (excluding Accounts Payable) | ||||||||||||||
| thereof Other Financial Liabilities | ||||||||||||||
| Non-Current Liabilities | ||||||||||||||
| Stockholders’ Equity | ||||||||||||||
Group Share in Equity (2023: | ||||||||||||||
| in 000' € | 2023 | 2022 | ||||||||||||
| Revenues | ||||||||||||||
| Interest Income | ( | |||||||||||||
| Depreciation and Amortization | ( | ( | ||||||||||||
| Interest Expenses | ( | |||||||||||||
| Income Tax Benefit / (Expenses) | ||||||||||||||
| Loss | ( | ( | ||||||||||||
| Other Comprehensive Income | ||||||||||||||
| Total Comprehensive Income | ( | ( | ||||||||||||
| Dividends Received | ||||||||||||||
The following table reconciles the summarized financial information presented to the carrying amount of the investment in the associates in the consolidated financial statements. The carrying amount of HI-Bio does not reconcile to the group share in equity in the associate. This is due to a fair value adjustments, a goodwill allocation, made at the time of acquisition and also due to timing differences (HI-Bio figures from the previous quarter are utilized) as well as the transfer of the dilution asset.
| in 000' € | 2023 | 2022 | ||||||||||||
| Balance as of January 1 / June 14 | ||||||||||||||
| Group Share of Total comprehensive Loss | ( | ( | ||||||||||||
| Anti-Dilution Asset | ||||||||||||||
| Sale of Shares of Investment in Associates | ( | |||||||||||||
| Balance as of December 31 | ||||||||||||||
In 2023 HI-Bio was able to close certain capital raising rounds which resulted in the reclassification of € 5.6 million from the Anti-Dilution asset to the carrying-amount of the share in associates.
F-44
License agreements will enable HI-Bio to develop and commercialize MorphoSys' anti-CD38 antibody felzartamab and anti-C5aR1 antibody MOR210. HI-Bio will receive worldwide commercialization rights for felzartamab and MOR210 except for the territories for felzartamab and MOR210 licensed to I-Mab Biopharma in 2017 and 2018.
Upon the achievement of certain milestone events for Felzartamab, MorphoSys receives additional shares of up to US$ 67.5 million (€ 61.1 million) and payments of up to US$ 500.0 million (€ 452.5 million). In addition, MorphoSys is eligible to receive tiered royalties on future net sales of felzartamab.
During the period from June 14, 2022 to June 30, 2023, all of MorphoSys's expenses related to the clinical development of felzartamab, which include personnel costs, costs for external services and material expenses, have been fully compensated or reimbursed by HI-Bio.
As consideration for the licensing of MOR210, MorphoSys received a payment of US$ 15.0 million (€ 14.4 million) in 2022. Upon achievement of certain events, MorphoSys may receive further payments of up to US$ 500.0 million (€ 452.5 million). In addition, MorphoSys is eligible to receive tiered royalties on future net sales of MOR210.
4.13 Deferred Tax Assets
At group level no deferred tax assets were recognized after netting with deferred tax liability in the 2023 financial year (December 31, 2022: € 0.0 million ).
4.14 Accounts Payable and Accruals
Accounts payable and licenses payable were non-interest-bearing and, under normal circumstances, have payment terms of no more than 30 days.
Accounts payable and accruals are listed in the following table.
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Accounts Payable | ||||||||||||||
| Accruals | ||||||||||||||
| Other Liabilities | ||||||||||||||
| Total | ||||||||||||||
Accruals are shown in the following overview:
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Accruals for External Laboratory Services | ||||||||||||||
| Accrued Personnel Expenses for Payments to Employees and Management | ||||||||||||||
| Accruals for Outstanding Invoices | ||||||||||||||
| Accruals for Revenue Deductions from Product Sales | ||||||||||||||
| Accruals for Legal Fees | ||||||||||||||
| Accruals for Audit Fees and other related Costs | ||||||||||||||
| Accruals for License Payments | ||||||||||||||
| Total | ||||||||||||||
At the Company’s Annual General Meeting in May 2023, PricewaterhouseCoopers GmbH Wirtschaftsprüfungsgesellschaft (PwC GmbH), Munich, was appointed as the auditor. The Supervisory Board engaged PwC GmbH to audit the financial statements.
The table below shows the total fees PwC Network received in the 2023 financial year.
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Audit Fees | ||||||||||||||
| Fees for Other Assurance Services | ||||||||||||||
| Other Fees for Other Services | ||||||||||||||
| Total | ||||||||||||||
The Audit Fees relate to the audit of the consolidated financial statements and the audit of the annual financial statements as well as all related services, including the review of the interim consolidated financial statements.
F-45
Other assurance services comprise fees in connection with the non-financial group report, services in connection with the issue of a comfort letter, as well as the audit of the content of the remuneration report.
Out of total fee, an amount of € 5 k relates to a license fee for the use of a digital information platform and relate to PwC Product Sales LLC, USA and is included in other services. All remaining fees relate to PwC GmbH.
4.15 Tax Liabilities and Provisions
As of December 31, 2023, the Group recorded tax liabilities of € 0.3 million (December 31, 2022: € 0.8 million) and provisions of € 32.5 million (December 31, 2022: € 14.7 million). Provisions included mainly expenses for share-based payments when these are settled by other assets equivalent to the value of a certain number of shares or stock options (“cash settlement”), as well as present obligations for onerous contracts.
The table below shows the development of tax liabilities and current and non-current provisions in the 2023 financial year.
| in 000' € | 01/01/2023 | Additions | Utilization | Release | 12/31/2023 | |||||||||||||||||||||||||||
| Tax Liabilities | ( | |||||||||||||||||||||||||||||||
| Provisions, current | ( | ( | ||||||||||||||||||||||||||||||
| Provisions, non-current | ( | ( | ||||||||||||||||||||||||||||||
| Total | ( | ( | ||||||||||||||||||||||||||||||
Provisions mainly include provisions for share-based payments in the amount of € 28.2 million.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
4.16 Contract Liabilities
Contract liabilities relate to transaction prices paid by customers that are allocated to unfulfilled performance obligations. The changes in this item are shown in the table below.
| in 000' € | 2023 | 2022 | ||||||||||||
| Balance as of January 1 | ||||||||||||||
| Prepayments Received in the Financial Year | ||||||||||||||
| Revenues Recognized in the Reporting Period that was included in the Contract Liability at the Beginning of the Period | ( | |||||||||||||
| Revenues Recognized for Received Prepayments and Services Performed in the Financial Year | ( | |||||||||||||
| Balance as of December 31 | ||||||||||||||
| thereof short-term | ||||||||||||||
| thereof long-term | ||||||||||||||
4.17 Deferred Tax Liabilities
4.18 Bonds
MorphoSys AG placed non-subordinated, unsecured convertible bonds in 2020 for a nominal amount of € 325.0 million, equal to 3,250 bonds with a nominal amount of € 100,000 each, and maturing on October 16, 2025.
The convertible bonds were issued at 100 % of their nominal amount and carry a coupon of 0.625 % p.a. payable semi-annually. The conversion price is € 131.29 . The convertible bonds are traded on the Open Market Segment (Freiverkehr) of the Frankfurt Stock Exchange.
The convertible bonds are convertible between November 26, 2020 and the fortieth trading day prior to maturity. As of the maturity date, MorphoSys has the right to either pay the full amount in cash or to settle a certain amount through the delivery of shares. The convertible bonds are convertible into approximately 2,475,436 new or existing bearer ordinary shares MorphoSys.
F-46
MorphoSys is entitled to redeem the convertible bonds at any time the market price of MorphoSys shares reaches at least 130 % of the then applicable conversion price over a period of twenty trading days or when only 20 % or less of the original total nominal amount of the convertible bond is still outstanding. Repayment is then made in the amount of the nominal value plus accrued interest.
The holders of the convertible bonds have a conditional call right should an investor directly or indirectly acquire at least 30 % of the voting rights in MorphoSys (representing a change of control). In the event of such a change of control, each convertible bondholder has the right to call the bonds that have not yet been converted or redeemed. Repayment is then made in the amount of the nominal value plus accrued interest.
The conversion right securitized in the convertible bond represents an equity instrument and was recognized in equity (additional paid in capital) for an amount of € 49.2 million net of deferred taxes and issuance costs attributable to the equity component. The equity component is not adjusted over time, and the liability component is classified as a financial liability at amortized cost. As of the date of initial recognition, the liability component amounted to € 270.7 million after the deduction of issuance costs. The difference between this amount and the nominal value of € 325.0 million is recognized as an interest expense over the term of the financial liability using the effective interest method.
The early termination rights from MorphoSys (issuer call and clean-up call) and the put option of the convertible bondholders in the case of change of control all represent embedded derivatives that, however, have not been separated in accordance with IFRS 9, as they are considered to be closely related to the base contract. Accordingly, these components are included in the financial liability.
On March 30, 2023, MorphoSys repurchased outstanding convertible bonds via a modified reverse Dutch auction procedure. At the close of the modified reverse Dutch auction procedure, MorphoSys had agreed to repurchase bonds representing € 62.9 million in aggregate principal amount (approximately 19.35 % of the outstanding principal amount). The purchase price per € 100,000 nominal was € 64,000 . The settlement procedure finished on March 30, 2023. Following the repurchase the bonds have been cancelled and deleted from the global certificate. As of December 31, 2023, there were 2,621 convertible bonds outstanding each with a notional amount of € 100,000. Upon repurchase MorphoSys realized a gain of € 16.4 million as the difference of the carrying amount as of the date of the repurchase and the fair value for the redeemed bonds. There were no bond conversions in the most recent fiscal year.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
4.19 Financial Assets and Liabilities from Collaborations
MorphoSys AG and Incyte Corporation signed a collaboration and license agreement in 2020 for the further global development and commercialization of MorphoSys’s proprietary anti-CD19 antibody tafasitamab. Under the terms of this agreement, MorphoSys could, among other things, pending on the achievement of certain developmental, regulatory, and commercial milestones, receive milestone payments amounting to up to US$ 1.1 billion (€ 995.5 million). MorphoSys also receives tiered royalties in a mid-teen to mid-twenties percentage of net sales of Monjuvi® outside the U.S. In the U.S., MorphoSys and Incyte co-commercialize Monjuvi®, with MorphoSys being responsible for the commercial relationship with the end customer, which also comprises the deliveries of the drug and the collection of the related cash inflows. The revenues from product sales of Monjuvi® are, therefore, recognized by MorphoSys, as it is the principal of the transaction. Incyte and MorphoSys are jointly responsible for the commercialization activities in the U.S. and will equally share any profits and losses (50 /50 basis). Outside the U.S., Incyte has received exclusive commercialization rights, determines the commercialization strategy and is responsible for the commercial relationship with the end customer, including the deliveries of the drug and the collection of the related cash inflows. Therefore, Incyte will recognize all revenues generated from sales of tafasitamab outside the U.S. and will pay royalties to MorphoSys on these sales.
As part of the agreement, MorphoSys recorded the balance sheet items "Financial Assets from Collaborations" and "Financial Liabilities from Collaborations." The financial asset represents MorphoSys’s current reimbursement claim against Incyte from the expected future losses associated with the U.S. commercialization activities (as Incyte has agreed to compensate MorphoSys for 50 % of said losses) measured at fair value. The financial liability, measured initially at fair value, represents Incyte’s prepaid entitlement to future profit sharing on sales of Monjuvi® in the U.S. (as MorphoSys will share 50 % of these profits with Incyte). Incyte has already acquired this right with the payments made in 2020; therefore, a liability had to be recognized at that time. The basis for the initial valuation at fair value was the corporate planning and its shared profits and losses thereof in connection with the commercialization activities of MorphoSys and Incyte in the United States for the years ahead.
The financial asset is subsequently measured at fair value through profit or loss and the financial liability at amortized cost using the effective interest method. Any resulting effective interest is recognized in the finance result. The basis for the
F-47
valuation at fair value is the corporate planning and its shared profits and losses thereof in connection with the commercialization activities of MorphoSys and Incyte in the U.S. for the years ahead. Cash flows from the profits and losses shared equally between the two parties are generally recognized directly against the financial asset or financial liability. Differences between the planned and actual cash flows from the financial asset or financial liability are recorded in the finance result. Effects resulting from changes in planning estimates regarding the expected net cash flows from financial assets and financial liabilities are also recognized in the finance result. The initial effective interest rate continues to be applied for the subsequent measurement of the financial liability, whereas the current yield curve is used for the financial assets. Foreign currency translation effects from the financial asset or financial liability are also recognized in the finance result.
The planning assumptions are influenced by estimates and mainly comprise revenues and costs for the production and sale of Monjuvi® in the U.S., the discount rate and the expected term of cash flows. Revenues are affected by variable influencing factors such as patient numbers and the number of doses of Monjuvi® administered, as well as the price that can be obtained in the market. Costs include the manufacturing costs for these doses of Monjuvi® and other cost components for e.g. sale, transport, insurance, and packaging. To determine the fair value of financial assets from collaborations, expected cash inflows from Incyte‘s planned losses resulting from the co-promotion activities of Monjuvi® in the U.S. are discounted using market interest rates of financial instruments with comparable currencies and maturities, taking into account Incyte‘s credit risk. The expected cash outflows are discounted using market interest rates of financial instruments with comparable currencies and maturities, taking into account the credit risk of MorphoSys. The term is the estimated time period over which Monjuvi® will generate benefits in the approved indication and therefore the expected term of product sales in the U.S. These estimates are based on assumptions that are jointly arrived at and approved quarterly by the responsible departments at MorphoSys and Incyte. Financial assets and financial liabilities from collaborations are furthermore subject to significant uncertainties from currency exchange rate developments.
As of December 31, 2023, US$ 3.8 million (€ 3.4 million) were recognized as a current financial asset and US$ 6.1 million (€ 5.5 million) as a current financial liability and US$ 120.3 million (€ 108.9 million) as a non-current financial liability as result of the collaboration with Incyte. As of December 31, 2022, € 0.0 million of current financial assets, € 2.5 million of current financial liabilities and € 217.8 million of non-current financial liabilities were recognized. The change is mainly resulting from changes in internal planning assumptions in the fourth quarter 2023 regarding the expected net cash flows related to financial liabilities from collaborations. For this purpose, an amount of € 107.8 million was recognized in financial income. Changes resulted mainly from lower expected future sales revenues for Monjuvi® in the USA. Additionally, income for foreign currency valuation in the amount of € 7.7 million as well as effects from cash payments of € 2.4 million were recognized. This was offset by expense from the application of the effective interest method in the amount of € 8.8 million.
MorphoSys and Incyte will also share the development costs for the jointly initiated worldwide and U.S.-specific clinical trials at a ratio of 55 % (Incyte) to 45 % (MorphoSys). This 45 % share of development costs borne by MorphoSys is included in research and development costs. Should MorphoSys provide services in excess of this 45 % share, MorphoSys will be entitled to a compensation claim against Incyte, which will qualify as revenue in accordance with IFRS 15. Related expenses for the provision of the service are recognized as cost of sales. Conversely, MorphoSys has to bear additional research and development expenses if Incyte performs more than 55 % of the total clinical trial services. In addition, Incyte will assume 100 % of future development costs for clinical trials in countries outside the United States, which are conducted in Incyte’s own responsibility. Incyte has the option to obtain development services from MorphoSys for this purpose. If this option is exercised, the related income will be recognized as revenue.
The financial assets from collaborations are classified at FVTPL and their measurement is based on the above-mentioned partly unobservable parameters. This results in a fair value classification in the Level 3 measurement hierarchy. The assets changed in 2023 as follows:
| in 000' € | 2023 | 2022 | ||||||||||||
| Balance as of January 1 | ||||||||||||||
| Additions | ||||||||||||||
| Cash Receipts | ( | |||||||||||||
| Through Profit or Loss (in Finance Income/Expenses) | ||||||||||||||
| Balance as of December 31 | ||||||||||||||
The estimates underlying the financial liabilities from collaborations are subject to a sensitivity analysis below. This would have resulted in the following effects on the carrying amount of the financial liabilities from collaborations as of December 31, 2023 and 2022. In each case, one planning assumption is changed and all other estimates are kept constant.
F-48
| 12/31/2023 | 12/31/2022 | |||||||||||||||||||||||||
| in million € | +1% | (1)% | +1% | (1)% | ||||||||||||||||||||||
| Change in Price obtained in the Market (revenue related) | ( | ( | ||||||||||||||||||||||||
| Change in Patient Numbers and Number of Doses administered (revenue related) | ( | ( | ||||||||||||||||||||||||
| Change in Manufacturing Costs and other Cost Components (cost related) | ( | ( | ||||||||||||||||||||||||
| Change in Patient Numbers and Number of Doses administered (cost related) | ( | ( | ||||||||||||||||||||||||
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
4.20 Financial Liabilities from Future Payments to Royalty Pharma
In 2021, a royalty purchase agreement and a revenue participation agreement were concluded with Royalty Pharma. In addition, a development funding bond was agreed, which was issued during fiscal year 2022. These agreements are summarized in the balance sheet item "Financial Liabilities from Future Payments to Royalty Pharma" (hereinafter referred to as "Royalty Pharma - Financial Liability").
| in 000' € | 2023 | 2022 | ||||||||||||
| Royalty Pharma - Financial Liability | ||||||||||||||
| thereof short-term | ||||||||||||||
| thereof long-term | ||||||||||||||
| Development Funding Bond | ||||||||||||||
| thereof short-term | ||||||||||||||
| thereof long-term | ||||||||||||||
| Balance as of Financial Liabilities from Future Payments to Royalty Pharma | ||||||||||||||
| thereof short-term | ||||||||||||||
| thereof long-term | ||||||||||||||
Royalty Pharma - Financial Liability
The "Royalty Pharma - Financial Liability" changed as follows in 2023 and 2022:
| in 000' € | 2023 | 2022 | ||||||||||||
| Balance as of January 1 | ||||||||||||||
| Addition | ||||||||||||||
| Amortizations from Effective Interest Method | ||||||||||||||
| Changes from Adjustments to Planning Assumptions | ( | ( | ||||||||||||
| Transfer of Assigned License Revenues to Royalty Pharma | ( | ( | ||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | |||||||||||||
| Balance as of December 31 | ||||||||||||||
This financial liability represents MorphoSys' (and Royalty Pharma's) obligation under the royalty purchase agreement to pass on certain future royalty revenues to Royalty Pharma in the form of royalties and milestones. This includes 100 % of MorphoSys' entitlement since April 1, 2021 for royalties from net sales of Tremfya from Janssen passed on to Royalty Pharma. Also included in the financial liability is Constellation's obligation to transfer 3 % of future net sales of clinical-stage compounds (pelabresib and tulmimetostat) to Royalty Pharma under the revenue participation agreement. If net sales of pelabresib exceed US$ 30.0 million (€ 27.1 million) in any fiscal year, an additional payment of US$ 50.0 million (€ 45.2 million) will be due. However, the rights to the underlying intellectual property of pelabresib and tulmimetostat remain with MorphoSys.
In addition, a contingent payment from Royalty Pharma to MorphoSys of up to US$ 100.0 million (€ 90.5 million) was agreed, which is subject to the achievement of certain clinical, regulatory and commercial milestones for otilimab from GSK, gantenumerab from Roche and pelabresib from Constellation.
F-49
The financial liability was measured at fair value at the date of inception (July 15, 2021). The initial measurement at fair value was based on corporate planning and the resulting net sales for the coming years, reduced by the market inequity in fiscal year 2021 described under "Development Financing Bond" (see below). There is no cash inflow and outflow at MorphoSys, as the agreed royalty percentages are paid directly by Janssen to Royalty Pharma. The cash flows from the transfer of assigned license revenues are generally recognized directly against the financial liability with no effect on profit or loss. Deviations of the actual cash flows from the original planning are recognized in finance income/expenses. Effects resulting from changes in the planning assumptions regarding the expected net cash flows are also recognized in finance income/expenses. The initial effective interest rate continues to be used for the subsequent measurement of the financial liability, as the financial liability is measured at amortized cost using the effective interest method. Royalty revenue from any product sales will continue to be recognized in profit or loss by MorphoSys, which acts as the principal.
The planning assumptions are influenced by estimates and mainly relate to the expected revenues from Tremfya, pelabresib and tulmimetostat and the expected term of the cash flows. Revenues are influenced by variable factors such as patient numbers and the number of doses administered as well as the price that can be achieved in the market. The estimated figures are also subject to exchange rate fluctuations, as the planning is made in USD, but payment has been agreed in euros. The term represents the estimated period over which Tremfya in the approved indication and pelabresib and tulmimetostat will generate future cash inflows and thus the expected duration of product sales. The above estimates are weighted with an expected probability of obtaining regulatory approval. The cash inflows and outflows represent an estimate of future revenues and costs from the out-licensed products and are subject to a significant degree of judgment. These estimates are based on assumptions that are developed and approved by the responsible departments of MorphoSys on a quarterly basis.
The estimates underlying the "Royalty Pharma - Financial Liability" are subject to a sensitivity analysis below. This would have resulted in the following effects on the carrying amount of the Royalty Pharma financial liability measured at amortized cost as of December 31, 2023, and December 31, 2022. In each case, one planning assumption is changed and all other estimates are kept constant.
| 12/31/2023 | 12/31/2022 | |||||||||||||||||||||||||
| in million € | +1% | (1)% | +1% | (1)% | ||||||||||||||||||||||
| Change in variable Factors on Revenues | ( | ( | ||||||||||||||||||||||||
| Change in Foreign Exchange Rate for future Royalties and Net Sales | ( | |||||||||||||||||||||||||
Development Funding Bond
The development funding bond changed as follows in 2023 and 2022:
| in 000' € | 2023 | 2022 | ||||||||||||
| Balance as of January 1 | ||||||||||||||
| Cash Receipts | ||||||||||||||
| Amortizations from Effective Interest Method | ||||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | ( | ||||||||||||
| Balance as of December 31 | ||||||||||||||
As all of the agreements with Royalty Pharma in 2021 were entered into on an arm's length basis, it can be assumed that the consideration paid by Royalty Pharma corresponds in total to the fair value of the liabilities entered into. However, as the implied interest rate on the development funding bond individually is 13.3 %, which is higher than the market interest rate of 6.3 % (as of 2021), it can be assumed that part of the consideration is to be considered as compensation for the market inequity (in the amount of the present value of the interest rate differential) of the development funding bond. Accordingly, for the agreed minimum amount of US$ 150.0 million (equivalent to € 147.7 million), the "Royalty Pharma - Financial Liability" was reduced by US$ 69.0 million (€ 58.4 million), and this amount was allocated to the development funding bond as compensation for the market inequity. The development funding bond is measured using the effective interest method.
Due to the issue amount exceeding the agreed minimum amount of US$ 150.0 million (equivalent to € 147.7 million), there is a difference in the transaction price and the fair value at initial recognition of the development funding bond at the time of payment in 2022. This is determined using the present value of the interest rate difference between the nominal interest rate of 13.3% and a market interest rate of 7.5% (as of 2022) and was measured in the amount of US$ 57.6 million (equivalent to € 56.7 million). The resulting fair value is higher than the amount paid out, so that the difference is to be regarded as a loss on initial recognition of the financial liability and recognized as a deferral. This results from the fact that the fair value of this financial liability is not evidenced by a quoted market price in an active market for an identical liability, nor by a valuation technique that uses only data from observable markets. The deferral of the initial measurement loss is recorded in the same
F-50
balance sheet line item as the development funding bond. The deferral is amortized over the life of the bond based on the performance of the bond.
The development of the deferral of the initial measurement loss can be seen in the following table. The initial measurement loss is included as a deferral with a debit amount in the development funding bond.
| in 000' € | 2023 | 2022 | ||||||||||||
| Balance as of January 1 | ||||||||||||||
| Addition | ||||||||||||||
| Amortization | ( | ( | ||||||||||||
| Foreign Currency Translation Differences from Consolidation | ( | ( | ||||||||||||
| December 31 | ||||||||||||||
4.21 Stockholders' Equity
4.21.1 Common Stock
As of December 31, 2023, the Company had common stock in the amount of € 37,655,137 or 37,655,137 shares (December 31, 2022: € 34,231,943 or 34,231,943 shares), divided into 37,655,137 no-par-value bearer shares (December 31, 2022: € 34,231,943 or 34,231,943 shares). The increase in common stock resulted entirely from the new shares created in the context of the capital increase in December 2023.
With the exception of the 53,685 treasury shares (€ 53,685 ) held by the Company (December 31, 2022: 65,980 treasury shares or € 65,980 ), the shares concerned are bearer shares with dividend entitlements and voting rights, with each share carrying one vote at the Annual General Meeting.
The development of the equity of the parent company MorphoSys AG (including the assessment with regard to the provision of Section 92 German Stock Corporation Act) as well as of MorphoSys Group is closely monitored by the Management Board. In addition, the company is closely monitoring the liquidity situation of MorphoSys Group and of MorphoSys AG, and believes that MorphoSys has sufficient liquid funds to ensure business operations for the forecast period (at least twelve months from the issuance date of the consolidated and statutory financial statements), which is subject to the going-concern assessment, without requiring additional proceeds from external refinancing. Any potential cashflows resulting from the Novartis Business Combination Agreement as announced on February 5, 2024, were not considered in the recent corporate planning.
Based on the company's recent corporate planning, which also incorporates the additionally released positive cash impacts from the sale of tafasitamab to Incyte as announced on February 5, 2024, MorphoSys believes that its liquidity is sufficient to finance its operational activities until early 2026, including the convertible bonds repayment. Any potential cashflows resulting from the Novartis Business Combination Agreement as announced February 5, 2024, were not considered in this recent corporate planning.
Under the Business Combination Agreement, Novartis agreed to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to provide MorphoSys with the financial resources required following completion of the Novartis Takeover Offer to enable MorphoSys to pay any obligations of MorphoSys arising from the implementation of the Novartis Takeover Offer as and when due, for example, but not limited to, the obligation from the convertible bonds and the obligations arising form the long-term incentive plans, each to the extent triggered by the completion of the Novartis Takeover Offer.
For the unlikely case that Novartis would withdraw its takeover offer and MorphoSys consequently would remain a stand-alone company, management would need to assess different financing options to ensure the going-concern assumption beyond early 2026 according to regulatory requirements. Management would then consider both non-dilutive financing options, such as out-licensing of (pre-) clinical assets or the sale of potential future royalties, but also considers accessing the capital markets by way of issuance of new shares or share instruments (ADSs) and/or issuance or refinancing of convertible debt.
At the time of this report, the Management Board is not aware of any imminent risks, neither individually nor collectively, that could affect the company as a going concern.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
F-51
4.21.2 Authorized Capital
In comparison to December 31, 2022, the number of authorized ordinary shares decreased from 9,195,696 (€ 9,195,696 ) to 8,909,562 (€ 8,909,562 ). At the Annual General Meeting on May 17, 2023, Authorized Capital 2023-I in the amount of € 6,846,388 and Authorized Capital 2023-II in the amount of € 3,423,194 , was newly created. The reduction of Authorized Capital 2019-I in the amount of € 46,246 , the reduction of Authorized Capital 2021-I in the amount of € 4,861,376 , the reduction of Authorized Capital 2021-II in the amount of € 1,951,452 and the reduction of Authorized Capital 2021-III in the amount of € 273,448 had an offsetting effect.
Under the Authorized Capital 2023-I, the Management Board is authorized, with the consent of the Supervisory Board, to increase the Company’s share capital on one or several occasions until and including May 16, 2028 against cash and/or non-cash contributions by a total of up to € 6,846,388 by issuing up to 6,846,388 new no-par-value bearer shares.
Under the Authorized Capital 2023-II, the Management Board is authorized, with the consent of the Supervisory Board, to increase the Company’s share capital on one or several occasions until and including May 16, 2028 against cash and/or non-cash contributions by a total of up to € 3,423,194 by issuing up to 3,423,194 new no-par-value bearer shares.
On December 14, 2023, a total of 3,423,194 shares were issued from Authorized Capital 2023-II. The Authorized Capital 2023-II was thus fully utilized. The cash increase was recorded in the commercial register on December 15, 2023.
Pursuant to the Company’s Articles of Association, the shareholders may authorize the Management Board to increase the share capital with the consent of the Supervisory Board within a period of five years by issuing shares for a specific total amount referred to as authorized capital (Genehmigtes Kapital), which is a concept under German law that enables the company to issue shares without going through the process of obtaining an additional shareholders’ resolution. The aggregate nominal amount of the authorized capital created by the shareholders may not exceed half of the share capital existing at the time of registration of the authorized capital in the commercial register.
4.21.3 Conditional Capital
In comparison to December 31, 2022, the number of ordinary shares of conditional capital decreased from 6,804,134 (€ 6,804,134 ) to 6,688,406 (€ 6,688,406 ). In the course of this General Meeting on May 17, 2023, the Conditional Capital 2016-III was reduced by € 115,728 .
Although shareholders may resolve to amend or create conditional capital (Bedingtes Kapital), they may do so only to issue conversion or subscription rights to holders of convertible bonds in preparation for a merger with another company or to issue subscription rights to employees and members of the Management Board of the Company or of an affiliated company by way of consent or authorizing resolution. According to German law, the aggregate nominal amount of the conditional capital created at the shareholders’ meeting may not exceed half of the share capital existing at the time of the shareholders’ meeting adopting such resolution. The aggregate nominal amount of the conditional capital created for the purpose of granting subscription rights to employees and members of the management of our Company or of an affiliated company may not exceed 10 % of the share capital existing at the time of the shareholders’ meeting adopting such resolution.
4.21.4 Treasury Stock
In the years 2023, 2022 and 2021, the Group did not repurchase any of its own shares. The composition and development of this line item are listed in the table below.
| Number of Shares | Value | |||||||||||||
| Balance as of December 31, 2020 | ||||||||||||||
| Transfer in 2021 | ( | ( | ||||||||||||
| Balance as of December 31, 2021 | ||||||||||||||
| Transfer in 2022 | ( | ( | ||||||||||||
| Balance as of December 31, 2022 | ||||||||||||||
| Transfer in 2023 | ( | ( | ||||||||||||
| Balance as of December 31, 2023 | ||||||||||||||
On December 31, 2023, the Company held 53,685 treasury shares with a value of € 1,995,880 – a decrease of € 454,423 compared to December 31, 2022 (65,980 shares, € 2,450,303 ). The reason for this decrease was the transfer of 12,295 treasury shares amounting to € 454,423 to the Management Board and selected employees of the Company (beneficiaries) from the 2019
F-52
Long-Term Incentive Plan (LTI Plan). The vesting period for this LTI Plan expired on April 1, 2023, and offered beneficiaries a six-month period until November 3, 2023, to receive a total of 12,295 shares.
Consequently, the number of MorphoSys shares owned by the Company as of December 31, 2023, was 53,685 (December 31, 2022: 65,980 ) and the number of outstanding shares amounted to 37,601,452 (December 31, 2022: 34,165,963 ). The repurchased shares may be used for all of the purposes named in the authorization granted by the Annual General Meeting on May 23, 2014, particularly for existing and future employee stock option programs and/or to finance acquisitions. The shares may also be redeemed.
4.21.5 Additional Paid-in Capital
As of December 31, 2023, the capital reserve amounted to € 938,088,474 (December 31, 2022: € 833,708,724 ). The increase by a total of € 104,379,750 resulted mainly from the capital increase in December 2023 (€ 99,272,626 , before costs for raising equity totaling € 6,650,567 ). In addition, additional paid-in-capital increased due to the sale of the investment in adivo GmbH on June 7, 2023. The gain on the disposal amounted to € 6,271,775 and was recognized in equity due to the recycling from other comprehensive income. Furthermore, the increase is attributable to the allocation of personnel expenses from share-based payments in the amount of € 5,940,339 . Part of the increase was offset by a decline that resulted from the reclassification of treasury shares related to share allocations from the 2019 Long-Term Incentive Plan in the amount of € 454,423 .
4.21.6 Other Comprehensive Income Reserve
On December 31, 2023, this reserve included changes in the fair value of equity instruments of € 331,972 (December 31, 2022: € (27,486 )) recognized directly in equity, as well as currency translation differences from consolidation of € 88,103,480 (December 31, 2022: € 115,354,088 ). The currency translation differences from consolidation included exchange rate differences from the revaluation of the financial statements of Group companies prepared in foreign currencies and differences between the exchange rates used in the balance sheet and income statement.
4.21.7 Accumulated Deficit
The consolidated net loss for the year of € 189,734,199 is reported under “accumulated deficit.” As a result, the accumulated deficit increased from € 823,407,416 in 2022 to € 1,013,133,943 in 2023.
5 Remuneration System for the Management Board and Employees of the Group
5.1 Equity-Settled Share-Based Payment Transactions
5.1.1 Stock Option Plans
2017 Stock Option Plan
On April 1, 2017, MorphoSys AG established a stock option plan (SOP) for the Management Board and selected employees of the Company (beneficiaries). The program is considered an equity-settled share-based payment and is accounted for accordingly. The vesting/performance period has ended on March 31, 2021. The performance criteria were set at 110 %. Each stock option thus grants 1.1 subscription rights to shares in the Company. The number of subscription rights vested per year were calculated based on the key performance criteria of the absolute and relative MorphoSys share price performance compared to the Nasdaq Biotech Index and the TecDAX Index.
The exercise price is € 55.52 . The exercise period is years after the end of the -year vesting period/performance period, which is March 31, 2024.
Based on the performance criteria achieved, 72,650 stock options can be exercised; this corresponds to 79,935 shares. Of these, the Management Board can exercise 0 stock options (0 shares), the members of the Executive Committee can exercise 4,018 stock options (4,421 shares) and other current and former employees of the Company can exercise 68,632 stock options (75,514 shares). As of December 31, 2023, 0 stock options have been exercised, representing 0 shares.
In 2023, personnel expenses from stock options under the Group’s 2017 SOP amounted to € 0 based on the fair value on the grant date (2022: € 0 ; 2021: € 2,757 ).
2018 Stock Option Plan
On April 1, 2018, MorphoSys AG established a stock option plan (SOP) for the Management Board and selected employees of the Company (beneficiaries). The program is considered an equity-settled share-based payment and is accounted for
F-53
accordingly. The vesting/performance period has ended March 31, 2022. The program’s performance criteria were set at 60 %. Each stock option grants up to 0.6 subscription rights to shares in the Company. The number of subscription rights vested per year is calculated based on the key performance criteria of the absolute and relative MorphoSys share price performance compared to the Nasdaq Biotech Index and the TecDAX Index.
The exercise price is € 81.04 . The exercise period is three years after the end of the -year vesting period/performance period, which is March 31, 2025.
Based on the performance criteria achieved, 63,127 stock options can be exercised; this corresponds to 37,901 shares. Of these, a member of the Management Board can exercise 0 stock options (0 shares), members of the Executive Committee can exercise 3,854 stock options (2,314 shares) and other current and former employees of the Company can exercise 63,924 stock options (35,587 shares). As of December 31, 2023, 0 stock options have been exercised, representing 0 shares.
In 2023, personnel expenses from stock options under the Group’s 2018 SOP amounted to € 0 based on the fair value on the grant date (2022: € (14,267 ); 2021: € 52,795 ).
2019 Stock Option Plan
On April 1, 2019, MorphoSys AG established a stock option plan (SOP) for the Management Board and selected employees of the Company (beneficiaries). The program is considered an equity-settled share-based payment and is accounted for accordingly. The vesting/performance period period has ended on March 31, 2023. The performance criteria were set at 29%. Based on this target achievement, each stock option leads to the same amount of subscription rights to shares in the Company. The number of subscription rights vested per year is calculated based on the key performance criteria of the absolute and relative MorphoSys share price performance compared to the Nasdaq Biotech Index and the TecDAX Index.
The exercise price is € 87.86 . The exercise period is years after the end of the -year vesting period/performance period, which is March 31, 2026.
Based on the performance criteria achieved, the Management Board and selected employees of the Company (beneficiaries) can receive in total 19,935 shares (19,935 stock options). Thereof, 0 shares can be transferred to a member of the Management Board, 1,220 shares to other members of the Executive Committee and 18,715 shares to other current and former employees of the Company. As of December 31, 2023, 0 shares were transferred to the beneficiaries.
On October 1, 2019, MorphoSys established a further stock option plan (SOP plan) for one member of the Management Board. The terms and conditions were identical to those of the April 1, 2019 program. The vesting period/performance period has ended on September 30, 2023. The performance criteria were set at 57 %. Based on this target achievement, each stock option leads to the same amount of subscription rights to shares in the Company. The number of subscription rights vested per year is calculated based on the key performance criteria of the absolute and relative MorphoSys share price performance compared to the Nasdaq Biotech Index and the TecDAX Index.
The exercise price is € 106.16 . The exercise period is years after the end of the -year vesting period/performance period, which is September 30, 2026.
Based on the performance criteria achieved, one member of the Management Board can receive in total 32,535 shares. As of December 31, 2023, 0 stock options have been exercised by the beneficiaries.
In 2023, personnel expenses from stock options under the Group’s 2019 SOP amounted to € 51,358 based on the fair value on the grant date (2022: € 218,126 ; 2021: € 625,806 ).
2020 Stock Option Plan
On April 1, 2020, MorphoSys AG established a stock option plan (SOP) for the Management Board and selected employees of the Company (beneficiaries). The program is considered an equity-settled share-based payment and is accounted for accordingly. The grant date was April 21, 2020, and the vesting/performance period is years. Each stock option grants up to two subscription rights to shares in the Company. The subscription rights vest each year by 25 % within the -year vesting period, provided that the performance criteria specified for the respective period have been 100 % fulfilled. The number of subscription rights vested per year is calculated based on the key performance criteria of the absolute and relative MorphoSys share price performance compared to the Nasdaq Biotech Index and the TecDAX Index. The program’s performance criteria can be met annually up to a maximum of 200 %. If the share price development falls short of the program’s performance parameters, the target achievement for that year is 0 %.
The exercise price, derived from the average market price of the Company’s shares in the XETRA closing auction on the Frankfurt Stock Exchange from the 30 trading days prior to the issue of the stock options, is € 93.66 .
F-54
MorphoSys reserves the right to settle the exercise of stock options using either newly created shares from Conditional Capital 2016-III, through the issue of treasury shares, or in cash should the exercise from Conditional Capital 2016-III not be possible. The exercise period is three years after the end of the -year vesting period/performance period, which is March 31, 2027.
In the event of a departure from the Company, the beneficiaries generally retain the stock options that have vested by the time of their departure.
In the event of a termination of a beneficiary for reasons of conduct or a revocation of the appointment of a member of the Management Board for reasons constituting good cause within the meaning of Section 626 (2) of the German Civil Code (BGB), all unexercised stock options forfeit without entitlement to compensation.
If an accumulated period of absence of more than 90 days occurs during the -year vesting period/performance period, 1/48 of the stock options granted are forfeited for each up to 30 days of absence. A period of absence is defined as absence due to illness, continued payment of remuneration in the event of illness or a suspended service or employment relationship without continued payment of remuneration.
If a change of control occurs during the -year vesting period, the stock options will become fully vested. In this case, however, the right to exercise the stock options arises only at the end of the -year vesting period.
In 2023, personnel expenses from stock options under the Group’s 2020 SOP amounted to € 110,703 based on the fair value on the grant date (2022: € 481,879 ; 2021: € 1,033,944 ).
2021 Stock Option Plan
On October 1, 2021, MorphoSys AG established a stock option plan (SOP) for selected employees of Constellation (beneficiaries). The program is considered an equity-settled share-based payment and is accounted for accordingly. The grant date was October 29, 2021, and the vesting/performance period is years. Each stock option grants up to two subscription rights to shares in the Company. The subscription rights vest each year by 25 % within the -year vesting period, provided that the performance criteria specified for the respective period have been 100 % fulfilled. The number of subscription rights vested per year is calculated based on the key performance criteria of the absolute and relative MorphoSys share price performance compared to the Nasdaq Biotech Index and the TecDAX Index. The program’s performance criteria can be met annually up to a maximum of 200 %. If the share price development falls short of the program’s performance parameters, the target achievement for that year is 0 %.
The exercise price, derived from the average market price of the Company’s shares in the XETRA closing auction on the Frankfurt Stock Exchange from the 30 trading days prior to the issue of the stock options, is € 44.91 .
MorphoSys reserves the right to settle the exercise of stock options using either newly created shares from Conditional Capital 2020-I, through the issue of treasury shares, or in cash should the exercise from Conditional Capital 2020-I not be possible. The exercise period is three years after the end of the -year vesting period/performance period, which is September 30, 2028.
In the event of a departure from the Company, the beneficiaries generally retain the stock options that have vested by the time of their departure.
In the event of a termination of a beneficiary for reasons of conduct or a revocation of the appointment of a member of the Management Board for reasons constituting good cause within the meaning of Section 626 (2) of the German Civil Code (BGB), all unexercised stock options forfeit without entitlement to compensation.
If a change of control occurs during the -year vesting period, the stock options will become fully vested. In this case, however, the right to exercise the stock options arises only at the end of the four-year vesting period.
In 2023, personnel expenses from stock options under the Group’s 2021 SOP amounted to € 124,064 based on the fair value on the grant date (2022: € 796,616 ; 2021:€ 711,223 ).
F-55
Development of Stock Option Plans and Fair Value
The table below shows the development of the stock option plans in the financial year 2023.
| April 2017 Stock Option Plan | April 2018 Stock Option Plan | April 2019 Stock Option Plan | October 2019 Stock Option Plan | April 2020 Stock Option Plan | October 2021 Stock Option Plan | |||||||||||||||||||||||||||||||||
| Outstanding on January 1, 2023 | ||||||||||||||||||||||||||||||||||||||
| Granted | ||||||||||||||||||||||||||||||||||||||
| Adjustment due to Performance Criteria | ( | ( | ||||||||||||||||||||||||||||||||||||
| Exercised | ||||||||||||||||||||||||||||||||||||||
| Forfeited | ( | ( | ||||||||||||||||||||||||||||||||||||
| Expired | ||||||||||||||||||||||||||||||||||||||
| Outstanding on December 31, 2023 | ||||||||||||||||||||||||||||||||||||||
| Exercisable on December 31, 2023 | ||||||||||||||||||||||||||||||||||||||
| Weighted-average Exercise Price (€) | ||||||||||||||||||||||||||||||||||||||
The fair value of the stock options from the 2019, 2020 and 2021 stock option plans was determined using a Monte Carlo simulation. The expected volatility is based on the development of the share volatility of the last four years. Furthermore, the calculation of fair value equally considered the performance criteria of the absolute and relative performance of MorphoSys shares compared to the development of the Nasdaq Biotech Index and the TecDAX Index. The parameters and fair value of each program are listed in the table below.
| April 2019 Stock Option Plan | October 2019 Stock Option Plan | April 2020 Stock Option Plan | October 2021 Stock Option Plan | |||||||||||||||||||||||
| Share Price on Grant Date in € | ||||||||||||||||||||||||||
| Exercise Price in € | ||||||||||||||||||||||||||
| Expected Volatility of the MorphoSys share in % | ||||||||||||||||||||||||||
| Expected Volatility of the Nasdaq Biotech Index in % | ||||||||||||||||||||||||||
| Expected Volatility of the TecDAX Index in % | ||||||||||||||||||||||||||
| Performance Term of Program in Years | ||||||||||||||||||||||||||
| Dividend Yield in % | n/a | n/a | n/a | n/a | ||||||||||||||||||||||
| Risk-free Interest Rate in % | between and | between and | between ( and ( | between ( | ||||||||||||||||||||||
| Fair Value on Grant Date in € | ||||||||||||||||||||||||||
5.1.2 Long-Term Incentive Programs
2019 Long-Term Incentive Plan
On April 1, 2019, MorphoSys AG established Long-Term Incentive Plan (Performance Share Plan) for the Management Board and selected employees of the Company (beneficiaries). The vesting period for this LTI Plan expired on November 3, 2023. The program is considered a share-based payment program with settlement in equity instruments and is accounted for accordingly. The LTI Plan is a performance-related share plan and will be paid out in ordinary shares (performance shares) of MorphoSys AG if predefined key performance criteria are achieved. These criteria are evaluated annually by the Supervisory Board. The performance criteria were based on a mathematical comparison of the absolute and relative performance of the MorphoSys share price against the Nasdaq Biotech Index and the TecDAX Index. Achievement of these criteria was set at 25 %. In addition, the Supervisory Board set a “company factor” as 1 , which determines the number of performance shares to be issued. Based on these conditions and the set factor, 12,295 performance shares of MorphoSys AG were transferred to the beneficiaries after the four-year vesting period in the period ending November 3, 2023. A member of the Management Board
F-56
received 157 performance shares (for further information, see the tables entitled “Shares” and “Performance Shares” in Note 5.3 “Related Parties”), and members of the Executive Committee received 157 performance shares. A total of 11,981 performance shares were granted to other current and former employees of the Company.
In 2023, personnel expenses resulting from performance shares under the Group’s 2019 LTI Plan amounted to € 2,325 based on the fair value on the grant date (2022: € 25,278 ; 2021: € 190,767 ).
Development of Long-Term Incentive Plans and Fair Value
The table below shows the development of the LTI plans in the financial year 2023.
| April 2019 Long-Term Incentive Program | ||||||||
| Outstanding on January 1, 2023 | ||||||||
| Granted | ||||||||
| Adjustment due to Performance Criteria | ( | |||||||
| Exercised | ( | |||||||
| Forfeited | ||||||||
| Expired | ||||||||
| Outstanding on December 31, 2023 | ||||||||
| Exercisable on December 31, 2023 | ||||||||
| Weighted-average Exercise Price (€) | n/a | |||||||
The fair value of the performance shares from the Long-Term Incentive Plan from 2019 has been determined using a Monte Carlo simulation. The expected volatility is based on the development of the share volatility of the last four years. Furthermore, the calculation of fair value equally considered the performance criteria of the absolute and relative performance of MorphoSys shares compared to the development of the Nasdaq Biotech Index and the TecDAX Index. The parameters and the fair value of each program are listed in the table below.
| April 2019 Long-Term Incentive Program | ||||||||
| Share Price on Grant Date in € | ||||||||
| Exercise Price in € | n/a | |||||||
| Expected Volatility of the MorphoSys share in % | ||||||||
| Expected Volatility of the Nasdaq Biotech Index in % | ||||||||
| Expected Volatility of the TecDAX Index in % | ||||||||
| Performance Term of Program in Years | ||||||||
| Dividend Yield in % | n/a | |||||||
| Risk-free Interest Rate in % | between | |||||||
| Fair Value on Grant Date in € | ||||||||
5.1.3 Restricted Stock Unit Plan (RSUP)
2021 Restricted Stock Unit Plan (RSUP)
On April 1, 2021, MorphoSys AG established a Long-Term Incentive Plan (LTI Plan) for selected employees of MorphoSys US Inc. (beneficiaries). The program is considered a share-based payment program with settlement in equity instruments and is accounted for accordingly. The LTI Plan is a performance-related share plan (Restricted Stock Unit Plan – RSUP) and is paid out in shares of MorphoSys AG that are to be created from authorized capital provided predefined performance criteria have been fulfilled. The term of the plan is years and includes one-year performance periods. If the predefined performance criteria for the respective period are 100 % met, 33.3 % of the performance shares become vested in each year. The number of performance shares vested per year is calculated based on the key performance criteria of MorphoSys US Inc. and the MorphoSys share price performance during the annual performance period. The performance criteria can be met up to a
F-57
maximum of 125 % per year. If less than 0 % of the defined performance criteria are met in any one year, no shares will be vested for that year. At the end of the total -year performance period, the corresponding number of shares eventually vested is calculated, and the shares created from authorized capital are transferred from the Company to the beneficiaries.
MorphoSys reserves the right to pay a specific amount of the LTI Plan in cash at the end of the performance period, equal to the value of the performance shares granted.
If a beneficiary loses his office or terminates his employment with MorphoSys US Inc. prior to the end of a performance period, the beneficiary will generally be entitled to all vested restricted stock units for already completed one-year performance periods. All remaining restricted stock units are forfeited without entitlement to compensation.
The fair value of the restricted shares granted on April 1, 2021, in accordance with the grant dates or measurement dates for each of the performance periods were € 44.63 per share on August 6, 2021, € 18.46 per share on June 15, 2022, and € 18.96 per share as of April 18, 2023.
On October 1, 2021, MorphoSys established a Long-Term Incentive Plan in the form of a restricted stock unit plan (RSUP) for certain employees of MorphoSys US Inc. (beneficiaries). The terms and conditions were identical to those of the April 1, 2021 program, except that the performance criteria can be met up to a maximum of 175 % per year.
The fair value of the restricted shares granted on October 1, 2021, in accordance with the grant dates or measurement dates for each of the performance periods were € 40.50 per share on October 1, 2021, € 18.46 per share on June 15, 2022, and € 18.96 per share on April 18, 2023.
In 2023, personnel expenses of the Group from the MorphoSys US Inc. 2021 RSU Plan amounted to € (27,091 ) based on the fair values (2022: € (219,040 ); 2021: € 1,260,750 ).
2022 Restricted Stock Unit Plan (RSUP)
On June 1, 2022, MorphoSys established a Long-Term Incentive Plan (LTI Plan) for certain employees of MorphoSys US Inc. and the Constellation Pharmaceuticals, Inc. (beneficiaries). This program is considered a share-based payment program with settlement in equity instruments and is accounted for accordingly. The LTI Plan is a performance-related share plan (Restricted Stock Unit Plan – RSUP) and is paid out in shares of MorphoSys AG created from authorized capital when predefined key performance criteria are achieved. The plan has a term of three years and comprises three performance periods with a term of one year each. If the predefined performance criteria for the respective period are 100 % met, 33 % of the performance shares become vested in each year. The number of shares vested per year is calculated based on key performance criteria of MorphoSys US entities during the annual performance period. The performance criteria can be met annually up to a maximum of 175 %. If the specified performance criteria are met by less than 0 % in one year, no shares will be earned for that year. After the end of the total three-year performance period, the final number of shares vested is calculated, and the shares created through authorized capital are transferred from the Company to the beneficiaries.
MorphoSys reserves the right to pay a certain amount of the LTI Plan in cash equal to the amount of the performance shares at the end of the performance period.
If a beneficiary ceases to hold office or is no longer employed at MorphoSys US Inc. or at Constellation Pharmaceuticals, Inc. before the end of a performance period, the beneficiary is generally entitled to all restricted stock units that have vested for previously completed one-year performance periods. All other restricted stock units will be forfeited without compensation.
The fair value of the restricted stock units granted on June 1, 2022, according to the reporting date for the three performance periods amounted to € 18.46 per share on June 15, 2022, € 18.96 on April 18, 2023, and € 34.00 per share on December 31, 2023.
As of June 1, 2022, U.S. beneficiaries had been granted 408,956 restricted shares. For the 2022 LTI Plan, the calculation of personnel expenses from share-based compensation was based on the assumption that beneficiaries would leave the Company during the three-year period, for which 40 % of the shares granted are designated.
On October 1, 2022, MorphoSys established a Long-Term Incentive Plan in the form of a restricted stock unit plan (RSUP) for certain employees of MorphoSys US Inc. and the Constellation Pharmaceuticals, Inc. (beneficiaries). The terms and conditions were identical to those of the June 1, 2022 program. For the calculation of the personnel expenses from share-based compensation, it was assumed for the 2022 LTI Plan that 20 % of beneficiaries would leave the Company during the -year period.
The fair value of the restricted shares granted on October 1, 2022, in accordance with the grant dates or measurement dates for each of the performance periods were € 22.22 per share on October 18, 2022, € 18.96 on April 18, 2023 and € 34.00 per share on December 31, 2023.
F-58
In 2023, personnel expenses of the Group from the MorphoSys US entities 2022 RSU Plan amounted to € 2,549,992 based on the fair values (2022: € 444,718 ; 2021: € — ).
2023 Restricted Stock Unit Plan (RSUP)
On April 1, 2023, MorphoSys established a Long-Term Incentive Plan (LTI Plan) for certain employees of MorphoSys US Inc. and the Constellation Pharmaceuticals, Inc. (beneficiaries). This program is considered a share-based payment program with settlement in equity instruments and is accounted for accordingly. The LTI Plan is a performance-related share plan (Restricted Stock Unit Plan – RSUP) and is paid out in shares of MorphoSys AG created from authorized capital when predefined key performance criteria are achieved. The plan has a term of three years and comprises three performance periods with a term of one year each. If the predefined performance criteria for the respective period are 100 % met, 33 % of the performance shares become vested in each year. The number of shares vested per year is calculated based on key performance criteria of MorphoSys US entities during the annual performance period. The performance criteria can be met annually up to a maximum of 175 %. If the specified performance criteria are met by less than 0 % in one year, no shares will be earned for that year. After the end of the total three-year performance period, the final number of shares vested is calculated, and the shares created through authorized capital are transferred from the Company to the beneficiaries.
MorphoSys reserves the right to pay a certain amount of the LTI Plan in cash equal to the amount of the performance shares at the end of the performance period.
If a beneficiary ceases to hold office or is no longer employed at MorphoSys US Inc. or at Constellation Pharmaceuticals, Inc. before the end of a performance period, the beneficiary is generally entitled to all restricted stock units that have vested for previously completed one-year performance periods. All other restricted stock units will be forfeited without compensation.
The fair value of the restricted stock units granted on April 1, 2023, according to the reporting date for the three performance periods amounted to € 18.96 per share on April 18, 2023, (fair value and grant date for first performance period) and € 34.00 per share on December 31, 2023. Targets have not yet been set for the second and third performance periods, and thus a grant date is not yet available.
As of April 1, 2023, U.S. beneficiaries had been granted 494,979 restricted shares. In the period from April 1, 2023, to December 31, 2023, U.S. beneficiaries have left MorphoSys US Inc. and Constellation Pharmaceuticals, Inc., and therefore 53,646 restricted shares have expired.
For the 2023 LTI Plan, the calculation of personnel expenses from share-based compensation was based on the assumption that beneficiaries would leave the Company during the three-year period, for which 40 % of the shares granted are designated.
On October 1, 2023, MorphoSys established a Long-Term Incentive Plan in the form of a restricted stock unit plan (RSUP) for certain employees of MorphoSys US Inc. and the Constellation Pharmaceuticals, Inc. (beneficiaries). The terms and conditions were identical to those of the April 1, 2023, program. 26,606 restricted shares were granted. For the calculation of the personnel expenses from share-based compensation, it was assumed for the 2023 LTI Plan that 40 % of beneficiaries would leave the Company during the three-year period.
The fair value of the restricted shares granted on October 1, 2023, in accordance with the grant dates or measurement dates for each of the three performance periods were € 26.60 per share as of October 24, 2023, and € 34.00 per share as of December 31, 2023. Targets have not yet been set for the second and third performance periods, and thus a grant date is not yet available.
In 2023, personnel expenses of the Group from the MorphoSys US entities 2023 RSU Plan amounted to € 3,349,318 based on the fair values (2022: € — ; 2021: € — ).
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
F-59
Development of RSUP with Equity-Settled Share-Based Payment
The table below shows the development of the performance shares under the MorphoSys RSU Plans in the financial year 2023.
| MorphoSys US Inc. – April 2021 Restricted Stock Unit Plan | MorphoSys US Inc. – October 2021 Restricted Stock Unit Plan | MorphoSys US – June 2022 Restricted Stock Unit Plan | MorphoSys US - October 2022 Restricted Stock Unit Plan | MorphoSys US – April 2023 Restricted Stock Unit Plan | MorphoSys US Inc. – October 2023 Restricted Stock Unit Plan | |||||||||||||||||||||||||||||||||
| Outstanding on January 1, 2023 | ||||||||||||||||||||||||||||||||||||||
| Granted | ||||||||||||||||||||||||||||||||||||||
| Exercised | ||||||||||||||||||||||||||||||||||||||
| Forfeited | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||
| Expired | ||||||||||||||||||||||||||||||||||||||
| Outstanding on December 31, 2023 | ||||||||||||||||||||||||||||||||||||||
| Exercisable on December 31, 2023 | ||||||||||||||||||||||||||||||||||||||
| Weighted-average Exercise Price (€) | n/a | n/a | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||
5.2 Cash-Settled Share-Based Payment Transactions
2020 Restricted Stock Unit Plan (RSUP)
On April 1, 2020, MorphoSys AG established a Long-Term Incentive Plan (LTI Plan) for selected employees of MorphoSys US Inc. (beneficiaries). The program was originally considered an equity-settled share-based payment transaction and was accounted for accordingly. As of December 31, 2022, it was decided to settle this program in cash.
The holding period/performance period expired on March 31, 2023. The performance criteria were based on the performance of MorphoSys US Inc. and the share price performance of MorphoSys AG during the annual performance period. The fulfillment of these performance criteria was set at 49 %. Taking these conditions into account, a payout amount of € 290,378 resulted. This obligation was fulfilled in 2023.
On October 1, 2020, MorphoSys AG established a Long-Term Incentive Plan (LTI Plan) for selected employees of MorphoSys US Inc. (beneficiaries). The program was originally considered an equity-settled share-based payment transaction and was accounted for accordingly. As of September 30, 2023, it was decided to settle this program in cash. This resulted in a reclassification of €246,265 from Equity to Provisions. The holding period/performance period expired on September 30, 2023. The performance criteria were based on the performance of MorphoSys US Inc. and the share price performance of MorphoSys AG during the annual performance period. The fulfillment of these performance criteria was set at 71 %. Taking these conditions into account, a payout amount of € 61,364 resulted. This obligation was fulfilled in 2023.
In 2023, personnel expenses of the Group from the MorphoSys US Inc. 2020 RSU Plan amounted to € (28,242 ) based on the fair values (2022: € (1,074,075 ); 2021: € (462,243 )).
2020 Performance Share Unit Program
On April 1, 2020, MorphoSys established a Performance Share unit Program (PSU Program) for the Management Board and certain employees of the Company (beneficiaries). The program is considered a cash-settled, share-based payment and is accounted for accordingly. The PSU program is a performance-based program and is paid out in cash subject to the fulfillment of predefined performance criteria. The grant date was April 21, 2020; the vesting period/performance period is four years. If the predefined performance criteria for the respective period are 100 % met, 25 % of the performance share units become vested in each year of the four-year vesting period. The number of performance share units vested per year is calculated on the basis of the performance criteria of the absolute and relative development of the MorphoSys share price compared to the development of the Nasdaq Biotech Index and the TecDAX Index. The performance criteria can be met each year up to a maximum of 200 %. If the defined performance criteria are met by less than 0 % in any one year, no performance share units will be earned for that year. However, the right to receive a certain cash settlement from the PSU program does not arise until the end of the four-year vesting period/performance period. After the end of the four-year vesting period, there is a -month period during which the performance shares can be transferred from the Company to the beneficiaries.
MorphoSys reserves the right to settle the PSU program at the end of the vesting period in MorphoSys AG’s own ordinary shares equal to the amount of the performance share units earned. The currently available treasury stock is not sufficient to settle the vested awards. MorphoSys therefore accounts for the plan only as a cash-settled share-based payment.
F-60
In the event of a departure from the Company, the beneficiaries generally retain the performance share units that have vested by the time of their departure.
In the event of a termination of a beneficiary for reasons of conduct or a revocation of the appointment of a member of the Management Board for reasons constituting good cause within the meaning of Section 626 (2) of the German Civil Code (BGB), all performance share units forfeit without entitlement to compensation.
If an accumulated period of absence of more than months occurs during the four-year vesting period/performance period, 1/48 of the performance share units are forfeited for each month of absence. A period of absence is defined as an absence due to illness or a period of inactive service or employment without continued payment of remuneration.
If a change of control occurs during the four-year vesting period, all performance share units will become fully vested. In this case, the right to receive a specific allocation of performance share units under the PSU Program occurs only at the end of the four-year vesting period.
On June 1, 2020, MorphoSys established a Performance Share Unit Program (PSU Program) for one member of the Management Board. The terms and conditions were identical to those of the April 1, 2020 program.
In March 2021, the terms of the Performance Share Unit Programs (PSU Programs) of April 1, 2020, and June 1, 2020, for the Management Board and certain employees of the Company (beneficiaries) were amended so that the number of performance share units still to be vested for the remaining three years is calculated on the basis of the performance criteria of the absolute performance of the MorphoSys share price and the relative performance of the MorphoSys share price compared to the performance of the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index. Previously, the number of performance share units earned in the first year was calculated on the basis of the performance criteria of the absolute and relative performance of the MorphoSys share price compared to the performance of the Nasdaq Biotech Index and the TecDAX Index. If the predefined performance criteria for the respective period are 100 % met, 25 % of the performance share units become vested in the first year, and 75 % become vested during the remaining three-year vesting period. The modification of the program’s terms concerns the respective remaining vesting periods/performance periods of the programs for the subsequent three years as of April 1, 2021 and June 1, 2021. The approval of the Management Board and certain employees of the Company (beneficiaries) to the modified program terms was obtained by April 17, 2021. The modification of the programs had no material impact on the fair values of the performance shares or on the period over which the personnel expenses are allocated.
In 2023, personnel expenses under the Group’s 2020 Performance Share Unit Program amounted to € 9,863 (2022: € (81,677 ); 2021: € 1,083,058 ).
2021 Performance Share Unit Program
On April 1, 2021, MorphoSys established a Performance Share Unit Program (PSU Program) for the Management Board and certain employees of the Company (beneficiaries). The program is considered a cash-settled, share-based payment and is accounted for accordingly. The PSU program is a performance-based program and is paid out in cash subject to the fulfillment of predefined performance criteria. The grant date was April 19, 2021; the vesting period/performance period is four years. If the predefined performance criteria for the respective period are 100 % met, 25 % of the performance share units become vested in each year of the four-year vesting period. The number of performance share units to be vested is calculated on the basis of the performance criteria of the absolute share price development of the MorphoSys share, the relative development of the MorphoSys share price compared to the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index and an assessment of the employee engagement. The performance criteria can be met each year up to a maximum of 200 %. If the defined performance criteria are met by less than 0 % in any one year, no performance share units will be earned for that year. However, the right to receive a certain cash settlement from the PSU program does not arise until the end of the four-year vesting period/performance period. After the end of the four-year vesting period, there is a -month period during which the performance shares can be transferred from the Company to the beneficiaries.
MorphoSys reserves the right to settle the PSU program at the end of the vesting period in MorphoSys AG’s own ordinary shares equal to the amount of the performance share units earned. The currently available treasury stock is not sufficient to settle the vested awards. MorphoSys therefore accounts for the plan only as a cash-settled share-based payment.
In the event of a departure from the Company, the beneficiaries generally retain the performance share units that have vested by the time of their departure.
In the event of a termination of a beneficiary for reasons of conduct or a revocation of the appointment of a member of the Management Board for reasons constituting good cause within the meaning of Section 626 (2) of the German Civil Code (BGB), all performance share units forfeit without entitlement to compensation.
F-61
If an accumulated period of absence of more than months occurs during the four-year vesting period/performance period, 1/48 of the performance share units are forfeited for each month of absence. A period of absence is defined as an absence due to illness or a period of inactive service or employment without continued payment of remuneration.
If a change of control occurs during the four-year vesting period, all performance share units will become fully vested. In this case, the right to receive a specific allocation of performance share units under the PSU Program occurs only at the end of the four-year vesting period.
On October 1, 2021, MorphoSys established a Performance Share Unit Program (PSU Program) for certain employees of the Company who are not members of the Executive Committee. The terms and conditions were identical to those of the April 1, 2021 program. The grant date was October 20, 2021.
In 2023, personnel expenses under the Group’s 2021 Performance Share Unit Program amounted to € 1,302,782 (2022: € (444,524 ); 2021: € 701,136 ).
2022 Performance Share Unit Program
On June 1, 2022, MorphoSys established a Performance Share Unit Program (PSU Program) for the Management Board and certain employees of the Company (beneficiaries). The program is considered a cash-settled, share-based payment and is accounted for accordingly. The PSU program is a performance-based program and is paid out in cash subject to the fulfillment of predefined performance criteria. The grant date was June 15, 2022. The vesting period/performance period is four years. If the predefined performance criteria for the four-year period are 100 % met, 100 % of the performance share units become vested in the four-year vesting period. The number of performance share units to be vested is calculated on the basis of the performance criteria of the relative development of the MorphoSys share price compared to the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index, the achievement of Development Milestones and an assessment of the employee engagement. The performance criteria can be met up to a maximum of 200 %. If the defined performance criteria are met by less than 0 %, no performance share units will be earned for the four-year assessment period. The right to receive a certain cash settlement from the PSU program does not arise until the end of the four-year vesting period/performance period. After the end of the four-year vesting period, there is a three-month period during which the earned performance shares are transferred from the Company to the beneficiaries by means of a cash settlement.
MorphoSys reserves the right to settle the PSU program at the end of the vesting period in MorphoSys AG’s ordinary shares equal to the amount of the performance share units earned. The currently available treasury stocks are likely not sufficient to settle the vested awards. MorphoSys therefore accounts for the plan as a cash-settled share-based payment in accordance with IFRS 2.
In the event of a departure from the Company, beneficiaries generally retain the performance share units that have vested by the time of their departure.
In the event of the termination of a beneficiary’s employment for reasons of conduct, or a revocation of the appointment of a member of the Management Board for reasons constituting good cause as defined by Section 626 (2) of the German Civil Code (BGB), all performance share units are forfeited without entitlement to compensation.
If a change of control occurs during the four-year vesting period, all performance share units will become fully vested. In this case, the right to receive a specific allocation of performance share units under the PSU program occurs only at the end of the four-year vesting period.
As of June 1, 2022, a total of 696,622 performance share units were granted to beneficiaries, of which 242,104 performance share units to the Management Board, 84,208 performance share units to other members of the Executive Committee and 370,310 performance share units to certain employees of the Company who are not members of the Management Board or Executive Committee. For the calculation of the personnel expenses from share-based compensation, it was assumed for the PSU program 2022 that 25 % of beneficiaries would leave the Company during the four-year period.
On October 1, 2022, MorphoSys established a Performance Share unit Program (PSU Program) for certain employees of the Company and for members of the Executive Committee. The terms and conditions were identical to those of the June 1, 2022 program. A total of 40,414 performance share units were granted to beneficiaries, of which 16,666 performance share units to members of the Executive Committee and 23,748 performance share units to certain employees of the Company who are not members of the Management Board or Executive Committee. The grant date was October 18, 2022.
In 2023, personnel expenses under the Group’s 2022 Performance Share Unit Program amounted to € 9,720,070 (2022: € 2,946,000 ; 2021: € — ).
F-62
2023 Performance Share Unit Program
On April 1, 2023, MorphoSys established a Performance Share Unit Program (PSU Program) for the Management Board and certain employees of the Company (beneficiaries). The program is considered a cash-settled, share-based payment and is accounted for accordingly. The PSU program is a performance-based program and is paid out in cash subject to the fulfillment of predefined performance criteria. The grant date was April 18, 2023. The vesting period/performance period is four years. If the predefined performance criteria for the four-year period are 100 % met, 100 % of the performance share units become vested in the four-year vesting period. The number of performance share units to be vested is calculated on the basis of the performance criteria of the relative development of the MorphoSys share price compared to the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index, the achievement of Development Milestones and an assessment of the employee engagement. The performance criteria can be met up to a maximum of 200 %. If the defined performance criteria are met by less than 0 %, no performance share units will be earned for the four-year assessment period. The right to receive a certain cash settlement from the PSU program does not arise until the end of the four-year vesting period/performance period. After the end of the four-year vesting period, there is a three-month period during which the earned performance shares are transferred from the Company to the beneficiaries by means of a cash settlement.
MorphoSys reserves the right to settle the PSU program at the end of the vesting period in MorphoSys AG’s ordinary shares equal to the amount of the performance share units earned. The currently available treasury stocks are likely not sufficient to settle the vested awards. MorphoSys therefore accounts for the plan as a cash-settled share-based payment in accordance with IFRS 2.
In the event of a departure from the Company, beneficiaries generally retain the performance share units that have vested by the time of their departure.
In the event of the termination of a beneficiary’s employment for reasons of conduct, or a revocation of the appointment of a member of the Management Board for reasons constituting good cause as defined by Section 626 (2) of the German Civil Code (BGB), all performance share units are forfeited without entitlement to compensation.
If a change of control occurs during the four-year vesting period, all performance share units will become fully vested. In this case, the right to receive a specific allocation of performance share units under the PSU Program occurs only at the end of the four-year vesting period.
As of April 1, 2023, a total of 982,783 performance share units were granted to beneficiaries, of which 241,666 performance share units to the Management Board, 130,000 performance share units to other members of the Executive Committee and 611,117 performance share units to certain employees of the Company who are not members of the Management Board or Executive Committee. For the calculation of the personnel expenses from share-based compensation, it was assumed for the PSU program 2023 that 25 % of beneficiaries would leave the Company during the four-year period.
On October 1, 2023, MorphoSys established a Performance Share Unit Program (PSU Program) for the Management Board and certain employees of the Company (beneficiaries). The terms and conditions were identical to those of the April 1, 2023 program. A total of 40,086 performance share units were granted to beneficiaries, of which 28,571 performance share units to the Management Board and 11,515 performance share units to certain employees of the Company who are not members of the Management Board or Executive Committee. The grant date was October 24, 2023.
In 2023, personnel expenses under the Group’s 2023 Performance Share Unit Program amounted to € 10,337,432 (2022: € — ; 2021: € — )
Long-Term Cash Incentive Plan (CLTI Plan)
On April 30, 2020, MorphoSys US Inc. established a long-term cash incentive plan (CLTI plan) for certain employees of MorphoSys US Inc. (beneficiaries). The holding period/performance period expired on March 31, 2023. The performance criteria were based on the performance of MorphoSys US Inc. and the share price performance of MorphoSys AG during the annual performance period. The fulfillment of these performance criteria was set at 49 %. Taking these conditions into account, a payout amount of € 178,790 resulted and the corresponding obligation was fulfilled in 2023.
In 2023, personnel expenses of the Group from the MorphoSys US Inc. 2020 CLTI plan amounted to € (131,585 ) (2022: € 42,585 ; 2021: € 117,395 ).
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
F-63
Development of Cash-Settled Programs and Fair Value
The table below shows the development of the Performance Share Unit Programs and the Restricted Stock Unit Plans in the financial year 2023.
| MorphoSys US Inc. – April 2020 Restricted Stock Unit Plan | MorphoSys US Inc. – October 2020 Restricted Stock Unit Plan | April 2020 Performance Share Unit Program | June 2020 Performance Share Unit Program | April 2021 Performance Share Unit Program | |||||||||||||||||||||||||||||||
| Outstanding on January 1, 2023 | |||||||||||||||||||||||||||||||||||
| Granted | |||||||||||||||||||||||||||||||||||
| Exercised | ( | ( | |||||||||||||||||||||||||||||||||
| Forfeited | ( | ( | ( | ( | |||||||||||||||||||||||||||||||
| Expired | |||||||||||||||||||||||||||||||||||
| Outstanding on December 31, 2023 | |||||||||||||||||||||||||||||||||||
| Exercisable on December 31, 2023 | |||||||||||||||||||||||||||||||||||
| Weighted-average Exercise Price (€) | n/a | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||
| October 2021 Performance Share Unit Program | June 2022 Performance Share Unit Program | October 2022 Performance Share Unit Program | April 2023 Performance Share Unit Program | October 2023 Performance Share Unit Program | ||||||||||||||||||||||||||||
| Outstanding on January 1, 2023 | ||||||||||||||||||||||||||||||||
| Granted | ||||||||||||||||||||||||||||||||
| Exercised | ||||||||||||||||||||||||||||||||
| Forfeited | ( | ( | ( | |||||||||||||||||||||||||||||
| Expired | ||||||||||||||||||||||||||||||||
| Outstanding on December 31, 2023 | ||||||||||||||||||||||||||||||||
| Exercisable on December 31, 2023 | ||||||||||||||||||||||||||||||||
| Weighted-average Exercise Price (€) | n/a | n/a | n/a | n/a | n/a | |||||||||||||||||||||||||||
The fair values of the performance share units of the 2020, 2021, 2022 and 2023 PSU Programs are determined using a Monte Carlo simulation. The expected volatility is based on the development of the share price volatility of the last years. The calculation of fair values equally considered the performance criteria of the absolute performance of MorphoSys shares, the relative performance compared to the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index, and an evaluation of employee engagement. The parameters and the fair value of each program are listed in the table below.
| April 2020 Performance Share Unit Program | June 2020 Performance Share Unit Program | April 2021 Performance Share Unit Program | October 2021 Performance Share Unit Program | ||||||||||||||||||||||||||
| Share Price in € on December 31, 2023 | |||||||||||||||||||||||||||||
| Exercise Price in € | n/a | n/a | n/a | n/a | |||||||||||||||||||||||||
| Expected Volatility of the MorphoSys share in % | |||||||||||||||||||||||||||||
| Expected Volatility of the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index in % | |||||||||||||||||||||||||||||
| Remaining Performance Term of Program in Years | 0.25 | 0.42 | 1.25 | 1.75 | |||||||||||||||||||||||||
| Dividend Yield in % | n/a | n/a | n/a | n/a | |||||||||||||||||||||||||
| Risk-free Interest Rate in % | |||||||||||||||||||||||||||||
| Fair Value on December 31, 2023, in € | |||||||||||||||||||||||||||||
F-64
| June 2022 Performance Share Unit Program | October 2022 Performance Share Unit Program | April 2023 Performance Share Unit Program | October 2023 Performance Share Unit Program | |||||||||||||||||||||||
| Share Price in € on December 31, 2023 | Share Price in € on | |||||||||||||||||||||||||
| Exercise Price in € | n/a | n/a | n/a | n/a | ||||||||||||||||||||||
| Expected Volatility of the MorphoSys share in % | ||||||||||||||||||||||||||
| Expected Volatility of the EURO STOXX Total Market Pharmaceuticals & Biotechnology Index in % | ||||||||||||||||||||||||||
| Remaining Performance Term of Program in Years | 2.42 | 2.75 | 3.25 | 3.75 | ||||||||||||||||||||||
| Dividend Yield in % | n/a | n/a | n/a | n/a | ||||||||||||||||||||||
| Risk-free Interest Rate in % | ||||||||||||||||||||||||||
| Fair Value on December 31, 2023, in € | Fair Value on , in € | |||||||||||||||||||||||||
5.3 Related Parties
Related parties are legal entities or individuals that can influence MorphoSys AG and its subsidiaries or are subject to control, joint control or significant influence by MorphoSys AG or its subsidiaries. These include, in particular, associates accounted for using the equity method. In addition to the members of the Management Board and the Supervisory Board, related parties who hold a key position in MorphoSys AG as the parent company of the Group also include all persons at the management level below. Key management personnel from the Group's perspective comprises those persons who direct and control the significant part of the Group's activities. Therefore, in addition to the Management Board and the Supervisory Board, the other members of the Executive Committee are considered to be key management personnel from the perspective of MorphoSys AG.
Balances and transactions between the Company and its fully consolidated subsidiaries, which constitute related parties, have been eliminated in the course of consolidation and are not commented on in this Note. Details of transactions between the Group and other related parties are disclosed below.
Related Entity
In 2023, revenues of € 3.8 million and cost reimbursements of € 5.8 million were recognized with the associated company under the underlying license agreements. As of December 31, 2023, trade receivables from associated companies amounted to € 0.5 million. For the terms and conditions related to these transactions, refer to Note 4.12.
Related Person
The Group engages in business relationships with members of the Management Board, the Supervisory Board and the other members of the Executive Committee as related parties responsible for the planning, management and monitoring of the Group. In addition to cash compensation, the Group has granted the Management Board performance shares. The tables below show the shares held and equity-settled stock options and performance shares from LTI plans that are part of share-based plans by the members of the Management Board and Supervisory Board (or by parties closely associated to them), as well as the changes in their ownership during the 2023 financial year.
Related parties that can be influenced by the Group or can have a significant influence on the Group can be divided into subsidiaries, members of the Supervisory Board, members of management in key positions and other related entities.
F-65
| Shares | ||||||||||||||||||||||||||
| 1/1/2023 | Additions | Sales | 12/31/2023 | |||||||||||||||||||||||
| Management Board | ||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | ||||||||||||||||||||||||||
Sung Lee1 | ||||||||||||||||||||||||||
Charlotte Lohmann2 | ||||||||||||||||||||||||||
Lucinda Crabtree, Ph.D.3 | ||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||
| Supervisory Board | ||||||||||||||||||||||||||
| Marc Cluzel, M.D., Ph.D. | ||||||||||||||||||||||||||
| Michael Brosnan** | ||||||||||||||||||||||||||
| Sharon Curran | ||||||||||||||||||||||||||
George Golumbeski, Ph.D. | ||||||||||||||||||||||||||
Andrew Cheng, M.D., Ph.D. | ||||||||||||||||||||||||||
| Krisja Vermeylen | ||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||
**Michael Brosnan holds 20,000 ADSs, i.e. 5,000 shares converted in ordinary shares.
| Stock Options | ||||||||||||||||||||||||||||||||||||||
| 1/1/2023 | Additions | Adjustment due to Performance Criteria4 | Forfeitures | Exercises | 12/31/2023 | |||||||||||||||||||||||||||||||||
| Management Board | ||||||||||||||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | ( | |||||||||||||||||||||||||||||||||||||
Sung Lee1 | ||||||||||||||||||||||||||||||||||||||
Charlotte Lohmann2 | ( | |||||||||||||||||||||||||||||||||||||
Lucinda Crabtree, Ph.D.3 | ||||||||||||||||||||||||||||||||||||||
| Total | ( | |||||||||||||||||||||||||||||||||||||
| Performance Shares from LTI plans | ||||||||||||||||||||||||||||||||||||||
| 1/1/2023 | Additions | Adjustment due to Performance Criteria4 | Forfeitures | Conversion to Shares | 12/31/2023 | |||||||||||||||||||||||||||||||||
| Management Board | ||||||||||||||||||||||||||||||||||||||
| Jean-Paul Kress, M.D. | ||||||||||||||||||||||||||||||||||||||
Sung Lee1 | ||||||||||||||||||||||||||||||||||||||
Charlotte Lohmann2 | ( | ( | ||||||||||||||||||||||||||||||||||||
Lucinda Crabtree, Ph.D.3 | ||||||||||||||||||||||||||||||||||||||
| Total | ( | ( | 0 | |||||||||||||||||||||||||||||||||||
1 Sung Lee resigned as a member of the Management Board with effect from the end of March 17, 2023. Changes after his departure from the Management Board are not presented.
2 With effect as of March 1, 2023, Charlotte Lohmann has been appointed as a member of the Management Board and Chief Legal Officer until the end August 31, 2023. Opening and closing balances presented in the tables were held by Charlotte Lohmann correspondingly before and after she was appointed as a member of the Management Board.
3 Lucinda Crabtree joined the Management Board of MorphoSys AG effective August 8, 2023.
4 Adjustment due to established performance criteria. For performance criteria that have not been met, a target achievement of 100% is assumed.
MorphoSys does not award any long-term variable remuneration component to the Supervisory Board.
F-66
The remuneration system for the Management Board meets the requirements of the German Stock Corporation Act and the German Corporate Governance Code and is intended to further a sustainable and long-term development of the Company and MorphoSys-Group. The Management Board’s total remuneration consists of several components, including fixed compensation, an annual cash bonus that is dependent upon the achievement of corporate targets (short-term incentives – STI), variable compensation components with long-term incentives (LTI) and other remuneration components. The variable remuneration components with long-term incentive consist of a long-term incentive plan (LTI Plan) in form of the performance share unit program. In previous years, stock options under the Company's stock option programs and performance shares under the Company's performance share plans have also been issued to Management Board members. In addition to fixed base remuneration, Management Board members receive standard fringe benefits, which mainly include the professional and private use of company cars, contributions to or reimbursement of costs for health, social and accident insurance, reimbursement of costs for legal advice related to service agreements, and dual residences. All total compensation packages are reviewed annually by the Compensation and Nomination Committee for scope and appropriateness and compared with the outcome of an annual Executive Board compensation analysis. The remuneration of the Management Board members is based largely on the duties of the respective Management Board member, the financial situation and the performance and business of the Company. All resolutions on the remuneration of the Management Board members are passed by the full Supervisory Board. The Management Board’s total remuneration package and the pension contracts were thoroughly reviewed and then adjusted by the Supervisory Board in 2022 and 2023.
The Management Board members generally participate in a pension plan in form of a provident fund. The provident fund takes out a reinsurance policy that funds the pension benefits. In addition, the Management Board members also receive an amount equal to up to 10% of their fixed annual (gross) base salary, which is intended to be used by the Management Board members for their individual retirement plans. This amount may also be invested in a pension plan. Jean-Paul Kress, M.D., also has the option to use both payments, however, up to a maximum of 10% of his fixed annual (gross) base salary, for his individual retirement plans.
Management Board members who also have a company pension plan as part of their deferred remuneration (direct insurance) also receive an allowance for this Company pension plan. The pension scheme for individual Management Board members may be differently structured in exceptional cases, e.g., in case a Management Board member is resident abroad.
If a Management Board member’s service contract terminates due to death, the member’s spouse or life partner is entitled to the fixed monthly salary for the month of death and the 12 months thereafter.
In the event of (i) a change of control and (ii) a material reduction of the area of responsibilities within one year after the change of control, the members of the Management Board, Jean-Paul Kress, M.D. and Lucinda Crabtree, Ph.D., are entitled to resign from the office as member of the Management Board and simultaneously terminate the service agreement against the payment of the outstanding fixed salary and annual bonus for the remainder of the fixed contract period, however, that such amount shall not exceed twice the annual remuneration.
The Performance Share Unit Programs also provide for the right of the Management Board members and/or the Company to forfeit all unexercised performance share units in return for a compensation payment in the amount of the respective offer price in the event of a voluntary takeover bid or a mandatory offer. In addition, in such a case all granted stock options, performance share units and performance shares will generally vest with immediate effect and can be exercised after expiry of the statutory waiting periods, whereby a change of control has occurred when (i) MorphoSys transfers assets or a substantial portion of its assets to unaffiliated third parties, (ii) MorphoSys merges with an unaffiliated company, (iii) an agreement pursuant to Section 291 AktG is entered into with MorphoSys as a dependent company, MorphoSys is integrated under Section 319 AktG or (iv) a shareholder or third party holds 30% or more of MorphoSys’s shares and/or voting rights.
In 2023, the STI 2022 was paid out. Financial and non-financial performance indicators were set for the STI 2022. The financial performance indicator included the financial performance indicators as presented in the management report. The non-financial ones included commercial, development and business development related targets. These performance indicators resulted in a weighted target achievement of 159.71%.
For the fiscal year 2023, the members of the Management Board were granted a total compensation (in accordance with HGB) of € 8,279,615 (2022: € 9,159,782 ), consisting of performance-unrelated remuneration of € 1,955,735 (2022: € 2,738,488 ), performance-related remuneration of € 1,898,880 (2022: € 1,821,294 ) as well as long-term incentive compensation of € 4,425,000 (2022: € 4,600,000 ) in the form of share-based compensation. The latter represents the fair value upon grant date. In 2022, termination benefits to members of the Management Board were recognized in the amount of € — (2022: € 320,248 ).
As of March 17, 2023, Sung Lee resigned from his position as CFO and as a member of the Management Board. The performance share units allocated to him will be granted in full, subject to the fulfillment of all other plan conditions.
F-67
Charlotte Lohmann was appointed as member of the Management Board and Chief Legal Officer with effect as of March 1, 2023, until the end of August 31, 2023.
On March 14, 2023, MorphoSys announced that Lucinda Crabtree, Ph.D., will join as Chief Financial Officer and member of the Management Board. She has been appointed as a Management Board member with effect as of August 8, 2023.
As of April 1, 2023, the Management Board was granted 241,666 Performance Share Units. The fair value as of December 31, 2023, amounts to € 21.03 . As of October 1, 2023, the Management Board was granted 28,571 Performance Share Units. The fair value as of December 31, 2023, amounts to € 25.28 .
For the individualized Management Board compensation, refer to the separately available remuneration report.
In the years 2023 and 2022, there were no other long-term benefits in accordance with IAS 24.17 (c) accruing to the Management Board or Supervisory Board. No benefits upon termination of service in accordance with IAS 24.17 (d) were accrued for the Supervisory Board in the years 2023 and 2022.
Compensation (in accordance with HGB) to former members of the Management Board, including bonus payments and other severance related items, amounted to € 0.9 million in 2023 (2022: € 1.4 million).
The compensation of the members of the Executive Committee consists of fixed compensation components (annual base compensation, customary fringe benefits and pension contributions), an annual bonus (STI) and a performance-based multi-year compensation (LTI), the Performance Share Unit Program ("PSUP") for members in Germany and the Restricted Stock Unit Program ("RSUP") for the member in the USA.
The total compensation for key management personnel (Management Board and members of the Executive Committee) in 2023 and 2022 were as follows.
| in € | 2023 | 2022 | ||||||||||||
| Total Short-Term Employee Benefits | ||||||||||||||
| Total Post-Employment Benefits | ||||||||||||||
| Total Termination Benefits | ||||||||||||||
| Total Share-Based Payment | ||||||||||||||
| Total Compensation | ||||||||||||||
As of December 31, 2023, there were accrued personnel expenses of € 3.7 million for payments to key management personnel for performance-related remuneration and non-current provisions of € 9.1 million for long-term incentive compensation (December 31, 2022: € 3.1 million and € 2.0 million, respectively).
The total remuneration for the Supervisory Board, excluding reimbursed travel costs, in 2023 and 2022 was as follows.
| Fixed Compensation | Attendance Fees1 | Total Compensation | ||||||||||||||||||||||||||||||||||||
| in € | 2023 | 2022 | 2023 | 2022 | 2023 | 2022 | ||||||||||||||||||||||||||||||||
| Marc Cluzel, M.D., Ph.D. | ||||||||||||||||||||||||||||||||||||||
| Michael Brosnan | ||||||||||||||||||||||||||||||||||||||
| Sharon Curran | ||||||||||||||||||||||||||||||||||||||
| George Golumbeski, Ph.D. | ||||||||||||||||||||||||||||||||||||||
Andrew Cheng, M.D., Ph.D. | ||||||||||||||||||||||||||||||||||||||
Wendy Johnson | ||||||||||||||||||||||||||||||||||||||
| Krisja Vermeylen | ||||||||||||||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||||||||||||||
1 The attendance fee contains expense allowances for the attendance at the Supervisory Board and the Committee meetings.
No other agreements currently exist with present or former members of the Supervisory Board.
As of December 31, 2023, the members of the Executive Committee (excluding the Management Board) held 10,589 stock options, 47,916 restricted stock units and 0 performance shares granted by the Company.
F-68
In 2023, performance share units under a new performance share unit program as well as restricted stock units under a new restricted stock unit plan were issued to the members of the Executive Committee (excluding the Management Board) (see Note 5.2).
Since April 1, 2023, the members of the Executive Committee (excluding the Board of Management) have a three-year period to exercise in total 1,220 stock options which have been granted to them under the 2019 SOP-Plan, which grant the same amount of subscription rights in shares of the Company. By December 31, 2023, no stock options have been exercised and thus, no shares were transferred.
On April 1, 2023, a total of 157 shares from the 2019 LTI Plan (performance share plan) were allocated to the members of the Executive Committee (excluding the Management Board), who were given the option to receive the shares within a six-month period. By December 31, 2023, 314 shares have been transferred.
6 Additional Notes
6.1 Obligations arising from Leases and Other Contracts
The future minimum payments under non-cancelable leases of low-value assets, performance share unit programs and contracts for insurance and other services on December 31, 2023 were as follows:
| in 000' € | Leases of Low-Value Assets and Short-Term Leases | Share-based payment programs | Other | Total | ||||||||||||||||||||||
| Less than 1 Year | ||||||||||||||||||||||||||
| Between One and Five Years | ||||||||||||||||||||||||||
| More than 5 Years | ||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||
As of December 31, 2022, these future minimum payments were as follows:
| in 000' € | Leases of Low-Value Assets and Short-Term Leases | Share-based payment programs | Other | Total | ||||||||||||||||||||||
| Less than 1 Year | ||||||||||||||||||||||||||
| Between One and Five Years | ||||||||||||||||||||||||||
| More than 5 Years | ||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||
Additionally, the Company has contracts for outsourced studies whereas the services have not been rendered as of December 31, 2023, and which could result in future payment obligations. These amounts could be shifted or substantially lower due to changes in the study timeline or premature study termination.
| in million € | 2023 | 2022 | ||||||||||||
| Less than 1 Year | ||||||||||||||
| Between One and Five Years | ||||||||||||||
| More than 5 Years | ||||||||||||||
| Total | ||||||||||||||
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
6.2 Contingent Liabilities
Contingent liabilities are potential obligations from past events that exist only when the occurrence of one or more uncertain future events – beyond the Company’s control – is confirmed. Current obligations can represent a contingent liability if it is not
F-69
probable enough that an outflow of resources justifies the recognition of a provision. Moreover, it is not possible to make a sufficiently reliable estimate of the sum of obligations.
The Management Board is unaware of any proceedings that may result in a significant obligation for the Group or lead to a material adverse effect on the Group’s net assets, financial position or results of operations.
If certain milestones are achieved by MorphoSys (for example, submitting an investigational new drug (IND) application for specific target molecules), this may trigger milestone payments to licensors of up to an aggregate of US$ 236.5 million (€ 214.0 million) related to regulatory events or the achievement of sales targets.
Monjuvi®’s product sales trigger percentage-based royalty payments.
Obligations may arise from enforcing the Company’s patent rights versus third parties. It is also conceivable that competitors may challenge the patents of the MorphoSys Group or that MorphoSys may come to the conclusion that its patents or patent families have been infringed upon by competitors. This could prompt MorphoSys to take legal action against competitors or lead competitors to file counterclaims against MorphoSys. Currently, there are no specific indications such obligations have arisen.
The assessment of potentially uncertain tax positions included the tax treatment of the financial liability from future payments to Royalty Pharma. In contrast to IFRS accounting, a deferred income item was recognized for tax purposes, which will be realized over the term of the underlying license agreements. The Company assumes that the tax authorities will share this assessment and that this will not be objected in a future tax audit. Due to the remaining uncertainty and the significance of the potential tax risk, we reported a contingent income tax liability in accordance with IFRIC 23.A5, IAS 12.88 and IAS 37. A different tax assessment would have a significant impact in the form of an additional tax payment. For tax purposes, deferred income for the obligations to Royalty Pharma amounted to € 786.4 million as of December 31, 2023 and the associated contingent tax liability upon non-acceptance of the deferral including interest effects amounts to € 226.8 million.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
6.3 Additional Disclosures for Financial Instruments
Fair Value Hierarchy and Measurement Methods
The fair value is the price that would be achieved for the sale of an asset in an arm’s length transaction between independent market participants or the price to be paid for the transfer of a liability (disposal or exit price).
Fair value is a market-based, not an entity-specific measurement. The fair value of non-financial assets is based on the best use of the asset by a market participant. For financial instruments, the use of bid prices for assets and ask prices for liabilities is permitted but not required if those prices best reflect the fair value in the respective circumstances. For simplification, mid rates are also permitted.
MorphoSys applies the following hierarchy in determining and disclosing the fair value of financial instruments:
Level 1: Quoted (unadjusted) prices in active markets for identical financial assets or liabilities to which the Company has access.
Level 2: Inputs other than quoted prices included within Level 1 that are observable for the financial asset or the financial liability, either directly (i.e., as prices) or indirectly (i.e., derived from prices).
Level 3: Inputs for the financial asset or the financial liability that are not based on observable market data (that is, unobservable inputs).
Hierarchy Level 1
The fair value of financial instruments traded in active markets is based on the quoted market prices on the reporting date. A market is considered active if quoted prices are available from an exchange, dealer, broker, industry group, pricing service, or regulatory body that is easily and regularly accessible, and prices reflect current and regularly occurring market transactions at arm’s length conditions. For assets held by the Group, the appropriate quoted market price is the buyer’s bid price.
Hierarchy Levels 2 and 3
The fair value of financial instruments not traded in active markets can be determined using valuation methods. In this case, fair value is determined using the results of a valuation method that makes maximum use of market data and relies as little as possible on not observable market data. If all significant inputs required for measuring fair value by using valuation methods
F-70
are observable, the instrument is allocated to Hierarchy Level 2. If significant inputs are not based on observable market data, the instrument is allocated to Hierarchy Level 3.
Hierarchy Level 2 contains foreign exchange forward agreements to hedge exchange rate fluctuations, term deposits as well as restricted cash. Future cash flows for these foreign exchange forward agreements are determined based on forward exchange rate curves. The fair value of these instruments corresponds to their discounted cash flows. The fair value of the term deposits and restricted cash is determined by discounting the expected cash flows using term-specific and risk-adjusted market interest rates.
Hierarchy Level 3 financial assets comprise equity investments, financial assets and financial liabilities from collaborations, financial assets which are part of other receivables (anti-dilution right HI-Bio), financial assets from Escrow accounts, the debt component of the convertible bond as well as financial liabilities from future payments to Royalty Pharma. For appraising the fair value of the financial assets from restricted escrow accounts (these are accounted for at fair value through profit or loss), the expected cash inflows were probability adjusted depending on the occurrence of certain conditions and discounted using market interest rates of the obligated contract party. The underlying valuations are generally carried out by employees in the finance department who report directly to the Chief Financial Officer. The valuation process and results are reviewed and discussed among the persons involved on a regular basis.
For the purpose of determining the fair value of financial assets from collaborations, expected cash inflows are discounted using market interest rates of financial instruments with comparable currencies and maturities, taking into account Incyte’s credit risk.
The fair value of the debt component of the convertible bond is determined based on the contractual cash flows (interest and principal), that are discounted using market interest rates of financial instruments with a comparable currency and maturities, taking into account MorphoSys' credit risk.
For further information on the assumptions and estimates used to derive the cash flows from the HI-Bio anti-dilution right, as well as a sensitivity analysis of the main estimates and assumptions, please refer to Note 4.5.
In order to determine the fair value of the non-current financial liabilities from collaborations for disclosure purposes (these are accounted for at amortized cost using the effective interest method), the expected cash outflows are discounted using market interest rates of financial instruments with comparable currencies and maturities, taking into account MorphoSys' credit risk.
For determining the fair value of the non-current financial liabilities for future payments to Royalty Pharma for disclosure purposes (these are accounted for at amortized cost using the effective interest method), the expected cash outflows from the planned royalty and milestone payments as well as the payments on the development funding bond to Royalty Pharma are discounted using market interest rates of financial instruments with comparable currencies and maturities, taking into account MorphoSys' credit risk.
For further information on the assumptions and estimates made to derive the cash flows from the financial assets and liabilities from collaborations and the financial liabilities from future payments to Royalty Pharma, as well as a sensitivity analysis of the significant estimates and assumptions of the financial liabilities recognized at amortized cost whose fair value is assigned to hierarchy level 3, please refer to Note 4.19 and 4.20.
Reclassifications between the hierarchy levels are generally taken into account as of the reporting dates. In 2023 and 2022, no transfers were made between the fair value hierarchy levels.
The carrying amounts of current financial assets and liabilities at amortized cost approximate their fair values given their short maturities.
F-71
The table below shows the fair values of financial assets and liabilities and the carrying amounts presented in the consolidated balance sheet.
| December 31, 2023; in 000' € | Classification Financial Instrument | Carrying Amount | Fair Value | Hierarchy Level | ||||||||||||||||||||||
| Cash and Cash Equivalents | AC | * | * | |||||||||||||||||||||||
| Other Financial Assets | ||||||||||||||||||||||||||
| thereof Money Market Funds | FVTPL | 1 | ||||||||||||||||||||||||
| thereof Fixed Term Deposits | AC | * | * | |||||||||||||||||||||||
| thereof Financial Asset from Escrow Account | FVTPL | 3 | ||||||||||||||||||||||||
| Accounts Receivable | AC | * | * | |||||||||||||||||||||||
| Financial Assets from Collaborations | FVTPL | 3 | ||||||||||||||||||||||||
| Other Receivables | ||||||||||||||||||||||||||
| thereof Anti-Dilution Right HI-Bio | FVTPL | 3 | ||||||||||||||||||||||||
| thereof Non-Financial Assets | n/a | n/a | n/a | |||||||||||||||||||||||
| Prepaid Expenses and Other Assets | ||||||||||||||||||||||||||
| thereof Non-Financial Assets | n/a | n/a | n/a | |||||||||||||||||||||||
| Current Financial Asset | ||||||||||||||||||||||||||
| Other Financial Assets | ||||||||||||||||||||||||||
| thereof Financial Asset from Escrow Account | FVTPL | 3 | ||||||||||||||||||||||||
| Prepaid Expenses and Other Assets | ||||||||||||||||||||||||||
| thereof Restricted Cash | AC | 2 | ||||||||||||||||||||||||
| thereof Non-Financial Assets | n/a | n/a | n/a | |||||||||||||||||||||||
| Non-Current Financial Asset | ||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||
| Accounts Payable and Accruals | ( | |||||||||||||||||||||||||
| thereof Accounts Payable | FLAC | ( | * | * | ||||||||||||||||||||||
| thereof Non-Financial Liabilities | n/a | ( | n/a | n/a | ||||||||||||||||||||||
| Bonds | FLAC | ( | * | * | ||||||||||||||||||||||
| Financial Liabilities from Collaborations | FLAC | ( | * | * | ||||||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | FLAC | ( | * | * | ||||||||||||||||||||||
| Current Financial Liabilities | ( | |||||||||||||||||||||||||
| Bonds | FLAC | ( | ( | 3 | ||||||||||||||||||||||
| Financial Liabilities from Collaborations | FLAC | ( | ( | 3 | ||||||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | FLAC | ( | ( | 3 | ||||||||||||||||||||||
| Non-Current Financial Liabilities | ( | |||||||||||||||||||||||||
| Total | ( | |||||||||||||||||||||||||
* For these instruments the carrying amount is a reasonable approximation of fair value.
F-72
| December 31, 2022; in 000' € | Classification Financial Instrument | Carrying Amount | Fair Value | Hierarchy Level | ||||||||||||||||||||||
| Cash and Cash Equivalents | AC | * | * | |||||||||||||||||||||||
| Other Financial Assets | ||||||||||||||||||||||||||
| thereof Money Market Funds | FVTPL | 1 | ||||||||||||||||||||||||
| thereof Fixed Term Deposits | AC | * | * | |||||||||||||||||||||||
| Accounts Receivable | AC | * | * | |||||||||||||||||||||||
| Financial Assets from Collaborations | FVTPL | 3 | ||||||||||||||||||||||||
| Other Receivables | ||||||||||||||||||||||||||
| thereof Anti-Dilution Right HI-Bio | FVTPL | 3 | ||||||||||||||||||||||||
| thereof Non-Financial Assets | n/a | n/a | n/a | |||||||||||||||||||||||
| Current Financial Asset | ||||||||||||||||||||||||||
| Prepaid Expenses and Other Assets | ||||||||||||||||||||||||||
| thereof Restricted Cash | AC | 2 | ||||||||||||||||||||||||
| thereof Non-Financial Assets | n/a | n/a | n/a | |||||||||||||||||||||||
| Non-Current Financial Asset | ||||||||||||||||||||||||||
| Total | ||||||||||||||||||||||||||
| Accounts Payable and Accruals | ( | |||||||||||||||||||||||||
| thereof Accounts Payable | FLAC | ( | * | * | ||||||||||||||||||||||
| thereof Non-Financial Liabilities | n/a | ( | n/a | n/a | ||||||||||||||||||||||
| Bonds | FLAC | ( | * | * | ||||||||||||||||||||||
| Financial Liabilities from Collaborations | FLAC | ( | * | * | ||||||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | FLAC | ( | * | * | ||||||||||||||||||||||
| Current Financial Liabilities | ( | |||||||||||||||||||||||||
| Bonds | FLAC | ( | ( | 3 | ||||||||||||||||||||||
| Financial Liabilities from Collaborations | FLAC | ( | ( | 3 | ||||||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | FLAC | ( | ( | 3 | ||||||||||||||||||||||
| Non-Current Financial Liabilities | ( | |||||||||||||||||||||||||
| Total | ( | |||||||||||||||||||||||||
*For these instruments the carrying amount is a reasonable approximation of fair value.
The totals of the carrying amounts of the financial instruments per measurement category are shown in the following overview.
| in 000' € | Classification Financial Instrument | 12/31/2023 | 12/31/2022 | |||||||||||||||||
| Financial Assets FVTPL | FVTPL | |||||||||||||||||||
| Financial Assets AC | AC | |||||||||||||||||||
| Financial Liabilities FLAC | FLAC | ( | ( | |||||||||||||||||
F-73
The development of the fair values of financial assets measured at fair value and allocated to hierarchy level 3 is shown in the following reconciliation.
| in 000' € | Financial Asset from Escrow Account | Anti-Dilution Right HI-Bio | Shares in Affiliated Companies < 20 % at Fair Value | |||||||||||||||||
| Balance as of January 1, 2023 | ||||||||||||||||||||
| Additions | ||||||||||||||||||||
| Gains/(losses) recognized in other comprehensive income | ||||||||||||||||||||
| Gains/(losses) recognized in profit or loss statement | ( | |||||||||||||||||||
| Reclassification to investment in associates | ( | |||||||||||||||||||
| Reclassification hierarchy levels | ||||||||||||||||||||
| Disposals | ( | |||||||||||||||||||
| Balance as of December 31, 2023 | ||||||||||||||||||||
| in 000' € | Financial Asset from Escrow Account | Anti-Dilution Right HI-Bio | Shares in Affiliated Companies < 20 % at Fair Value | |||||||||||||||||
| Balance as of January 1, 2022 | ||||||||||||||||||||
| Additions | ||||||||||||||||||||
| Gains/(losses) recognized in other comprehensive income | ||||||||||||||||||||
| Gains/(losses) recognized in profit or loss statement | ( | |||||||||||||||||||
| Reclassification to investment in associates | ( | |||||||||||||||||||
| Reclassification hierarchy levels | ||||||||||||||||||||
| Disposals | ||||||||||||||||||||
| Balance as of December 31, 2022 | ||||||||||||||||||||
Equity Investments
Since July 2018, MorphoSys held an investment in adivo GmbH. During the 2023 financial year, all shares in this investment, which was accounted for at fair value through other comprehensive income, were sold. The gain on the disposal amounted to € 6.3 million and was recognized in equity. The amount was reclassified from Other Comprehensive Income to the capital reserve (2022: € 0.0 million). This corresponds to a fair value before sale of € 6.7 million. As of December 31, 2022, the fair value of the investment was measured at € 0.0 million.
In the 2023 and 2022 financial years, no
Net Result according to Measurement Categories
The following net gains or losses resulted from financial instruments in the financial year.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| FVTPL | ||||||||||||||||||||
| AC | ||||||||||||||||||||
| FLAC | ( | |||||||||||||||||||
| Total | ( | |||||||||||||||||||
The net gains on financial assets at fair value through profit or loss (FVTPL) resulted from valuation effects from changes in the fair value of financial assets from collaborations and money market funds. Net losses on financial assets at amortized cost (AC) resulted from the application of the effective interest method for the term deposits, exchange rate fluctuations and risk provisions. The category financial liabilities at amortized cost (FLAC) includes the gains and losses from fair value changes due to changes in planning estimates and the effective interest rate from the financial liabilities from collaborations as well as from the application of the effective interest rate method for the financial liabilities from future payments to Royalty Pharma and the convertible bonds.
F-74
The gross interest income and expenses from financial assets and liabilities measured at amortized cost are shown in the following table.
| in 000' € | 2023 | 2022 | 2021 | |||||||||||||||||
| Interest Income AC | ||||||||||||||||||||
| Interest Expenses AC | ( | ( | ( | |||||||||||||||||
| Interest Income FLAC | ||||||||||||||||||||
| Interest Expenses FLAC | ( | ( | ( | |||||||||||||||||
| Total | ( | ( | ( | |||||||||||||||||
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
6.4 Financial Risk Management
Due to its operating activities with regard to assets, liabilities and planned transactions, the Group is exposed in particular to risks from the default of a contractual party (credit risk), from the non-fulfilment of liabilities (liquidity risk) and from market risks, in particular from changes in exchange rates and interest rates. The aim of the risk management is to limit these risks through ongoing operational and finance-oriented activities.
6.4.1 Credit Risk
Financial instruments in which the Group may have a credit risk are mainly cash and cash equivalents, other financial assets, derivative financial instruments and accounts receivable. The Group’s cash, cash equivalents and other financial assets are mainly denominated in euros and US dollars. Other financial assets are high quality assets. Cash, cash equivalents and other financial assets are generally held at numerous reputable financial institutions in Europe and the United States. With respect to its positions, the Group continuously monitors the financial institutions that are its counterparties to the financial instruments, as well as their creditworthiness, and does not anticipate any risk of non-performance.
The changes in risk provisions (see Note 2.6.1) recognized in the statement of profit or loss for the financial years 2023, 2022 and 2021 under the item impairment losses on financial assets were determined based on the rationale that negative values represent additions and positive values represent reversals of risk provisions. There were no write-offs in the 2023 financial year. In the general impairment model, the risk provision is recognized for financial assets at amortized cost - cash and cash equivalents, parts of other financial assets (term deposits) - and in the simplified impairment model for accounts receivable.
General Impairment Model | Simplified Impairment Model | Total | ||||||||||||||||||||||||||||||||||||
| in 000' € | Stage 1 | Stage 2 | Stage 3 | Stage 2 | Stage 3 | |||||||||||||||||||||||||||||||||
| Balance as of January 1, 2022 | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Unused Amounts Reversed | ||||||||||||||||||||||||||||||||||||||
| Increase in Impairment Losses for Credit Risks recognized in Profit or Loss during the Year | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Change between Impairment Stages | ||||||||||||||||||||||||||||||||||||||
| Amounts written off during the Year as uncollectible | ||||||||||||||||||||||||||||||||||||||
| Balance as of December 31, 2022 | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Balance as of January 1, 2023 | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Unused Amounts Reversed | ||||||||||||||||||||||||||||||||||||||
| Increase in Impairment Losses for Credit Risks recognized in Profit or Loss during the Year | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Change between Impairment Stages | ||||||||||||||||||||||||||||||||||||||
| Amounts written off during the Year as uncollectible | ||||||||||||||||||||||||||||||||||||||
| Balance as of December 31, 2023 | ( | ( | ( | |||||||||||||||||||||||||||||||||||
The gross carrying amounts of the Group's financial assets by credit risk rating class are as follows.
F-75
| Financial Assets as of December 31, 2023 | Internal Credit Rating | Basis for Recognition of Expected Credit Loss Provision | Gross Carrying Amount (in 000’ €) | ||||||||||||||
| Cash and Cash Equivalents | low | Expected Twelve-Month Loss | |||||||||||||||
| Term Deposits | low | Expected Twelve-Month Loss | |||||||||||||||
| Accounts Receivable | low | Lifetime Expected Credit Losses | |||||||||||||||
| Financial Assets as of December 31, 2022 | Internal Credit Rating | Basis for Recognition of Expected Credit Loss Provision | Gross Carrying Amount (in 000’ €) | |||||||||||||||||
| Cash and Cash Equivalents | low | Expected Twelve-Month Loss | ||||||||||||||||||
| Term Deposits | low | Expected Twelve-Month Loss | ||||||||||||||||||
| Accounts Receivable | low | Lifetime Expected Credit Losses | ||||||||||||||||||
The Group is also exposed to credit risk from debt instruments that are measured at fair value through profit or loss. This includes the items “Financial Assets at Fair Value through Profit or Loss” and “Financial Assets from Collaborations.” As of December 31, 2023, the maximum credit risk corresponded to the carrying amounts of these items amounting to € 238.3 million (December 31, 2022: € 14.6 million).
One of the Group’s policies requires that all customers who wish to transact business on credit undergo a credit assessment based on external ratings. Nevertheless, the Group’s revenue and accounts receivable are still subject to credit risk from customer concentration. The Group’s single most significant customer accounted for € 14.4 million of accounts receivables as of December 31, 2023 (December 31, 2022: € 51.4 million), or 45 % of the Group’s total accounts receivable at the end of 2023. The Group’s top three customers individually accounted for 47 %, 16 % and 15 % of the total revenue in 2023.
As of December 31, 2022, 56 % of the Group’s accounts receivable balance related to a single customer; of the total revenue in 2022, three customers individually accounted for 35 %, 15 % and 12 %.
The maximum credit risk (equal to the carrying amount) for rent deposits and other deposits on the reporting date amounted to € 1.2 million (December 31, 2022: € 1.3 million).
6.4.2 Liquidity Risk
Liquidity risk arises primarily from accounts payable, lease liabilities (refer to Note 4.9), bonds, financial liabilities from collaborations and financial liabilities from future payments to Royalty Pharma. Liquidity risk is managed on the basis of balance sheet and profit and loss figures. This is done by means of liquidity planning for the current year on a monthly basis, for the three subsequent years on an annual basis and a monthly target/actual comparison. The top priority is always to ensure sufficient liquidity so that all payment obligations can be met.
The following table shows the maturities of the cash flows of accounts payable and bonds at the balance sheet date. For the financial liabilities from collaborations, the non-discounted, future planned half profit sharing payments from Incyte for the sales of Monjuvi® in the U.S. are presented. The financial liabilities from future payments to Royalty Pharma include the undiscounted, planned net sales in the coming years. There is no cash inflow and outflow at MorphoSys as the agreed percentage royalties and milestones are paid directly by Janssen, GSK and Roche to Royalty Pharma. Refer to Note 4.9 for the contractual cash flows of lease liabilities.
| in '000 €; due on December 31, 2023 in | Less than 1 Year | Between One and Five Years | More than 5 Years | Total | ||||||||||||||||||||||
| Accounts Payable | ||||||||||||||||||||||||||
| Bonds | ||||||||||||||||||||||||||
| Financial Liabilities from Collaborations | ||||||||||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | ||||||||||||||||||||||||||
F-76
| in 000' €; due on December 31, 2022 in | Less than 1 Year | Between One and Five Years | More than 5 Years | Total | ||||||||||||||||||||||
| Accounts Payable | ||||||||||||||||||||||||||
| Bonds | ||||||||||||||||||||||||||
| Financial Liabilities from Collaborations | ||||||||||||||||||||||||||
| Financial Liabilities from Future Payments to Royalty Pharma | ||||||||||||||||||||||||||
There were no financial instruments pledged as collateral as of December 31, 2023.
6.4.3 Market Risk
Market risk represents the risk that changes in market prices, such as foreign exchange rates, interest rates or equity prices, will affect the Group‘s results of operations or the value of the financial instruments held. The Group is exposed to both currency and interest rate risks.
Currency Risk
The consolidated financial statements are prepared in euros. Both revenues and expenses of the Group are incurred in euros and US dollars. Throughout the year, the Group monitors the necessity to hedge foreign exchange rates to minimize currency risk and addresses this risk by using derivative financial instruments.
The use of derivatives is subject to a Group guideline approved by the Management Board, which represents a written guideline for dealing with derivatives. In accordance with the Group's hedging policy, only highly probable future cash flows and clearly determinable receivables that can be realized within a period of twelve months are hedged. MorphoSys enters into foreign exchange option and forward exchange contracts to hedge its foreign exchange exposure arising from U.S. dollar cash flows.
The Group’s exposure to foreign currency risk based on the carrying amounts of the items is shown in the table below.
| as of December 31, 2023; in 000' € | US$ | Other | ||||||||||||
| Cash and Cash Equivalents | ||||||||||||||
| Accounts Receivable | ||||||||||||||
| Financial Assets from Collaborations | ||||||||||||||
| Accounts Payable and Accruals | ( | ( | ||||||||||||
| Financial Liabilities from Collaborations | ( | |||||||||||||
| Total | ( | ( | ||||||||||||
| as of December 31, 2022; in 000' € | US$ | Other | ||||||||||||
| Cash and Cash Equivalents | ||||||||||||||
| Accounts Receivable | ||||||||||||||
| Accounts Payable and Accruals | ( | ( | ||||||||||||
| Financial Liabilities from Collaborations | ( | |||||||||||||
| Total | ( | ( | ||||||||||||
The financial liabilities from future payments to Royalty Pharma are dependent on future royalty income, which is determined on the basis of sales in U.S. dollars. The transfer of assigned license revenues is settled in euros. Refer to Note 4.20 for a sensitivity analysis on the impact of a change in the foreign exchange rate.
Different foreign exchange rates and their impact on financial assets and liabilities were simulated in a sensitivity analysis to determine the effects on profit or loss. Positive amounts would increase a consolidated net profit or decrease a consolidated net loss. Negative amounts would decrease a consolidated net profit or increase a consolidated net loss.
| in million € | 2023 | 2022 | 2021 | |||||||||||||||||
| Increase of the Euro by 10% | ||||||||||||||||||||
| Decrease of the Euro by 10% | ( | ( | ( | |||||||||||||||||
F-77
Interest Rate Risk
The Group’s risk exposure to changes in interest rates mainly relates to fixed-term deposits and corporate bonds. Changes in the general level of interest rates may lead to an increase or decrease in the fair value of these securities. The Group’s investment focus places the safety of an investment ahead of its return and the ability to plan future cash flows. Interest rate risks are limited because all securities can be liquidated within a maximum of two years and due to the mostly fixed interest rates during the term in order to ensure that planning is possible. In addition, changes in interest rates may affect the fair value of financial assets from collaborations.
Different interest rates and their effect on existing other financial assets with variable interest rates and on financial assets from collaborations were simulated in a sensitivity analysis in order to determine the effect on profit or loss. Positive amounts would increase a consolidated net profit or decrease a consolidated net loss. Negative amounts would decrease a consolidated net profit or increase a consolidated net loss.
| in million € | 2023 | 2022 | 2021 | |||||||||||||||||
| Increase of the variable Interest Rate by 0.5% | ||||||||||||||||||||
| Decrease of the variable Interest Rate by 0.5% | ( | ( | ( | |||||||||||||||||
The Group is currently not subject to significant interest rate risks from the account payables reported on the balance sheet.
6.4.4 Capital Management
The Management Board’s policy for capital management is to preserve a strong and sustainable capital base in order to maintain the confidence of investors, business partners, and the capital market and to support future business development and to safeguard its ability to continue as a going concern. As of December 31, 2023, the equity ratio was 2.4 % (December 31, 2022: 6.6 %; see also the following overview).
| in 000' € | 12/31/2023 | 12/31/2022 | ||||||||||||
| Stockholders’ Equity | ||||||||||||||
| In % of Total Capital | ||||||||||||||
| Total Liabilities | ||||||||||||||
| In % of Total Capital | ||||||||||||||
| Total Capital | ||||||||||||||
MorphoSys actively manages its cash and investments to primarily ensure liquidity and principal preservation while seeking to maximize returns. MorphoSys' cash and short-term investments are located at several banks. Financial investments are made in investment instruments having at minimum a Standard & Poor's rating (or equivalent) of at least A-.
No minimum capital requirements are stipulated in MorphoSys' Articles of Association. The Company has obligations to issue shares out of conditional and authorized capital relating to the exercise of stock options and restricted stock units on the basis of share-based payment transactions (refer to Notes 5.1 and 5.2).
There are no liabilities to banks.
F-78
6.5 Disclosures to Statement of Cash Flows - Net Debt Reconciliation
The following overview contains the presentation and development of the liabilities from financing activities. “Amortizations from Effective Interest Method,” “Changes from Adjustments to Planning Assumptions” and “Transfer of Assigned License Revenues to Royalty Pharma” include non-cash movements, including accrued interest expense.
| in 000' € | Lease Liabilities | Bonds | Financial Liabilities from Collaborations | Financial Liabilities from Future Payments to Royalty Pharma | Total | |||||||||||||||||||||||||||
| Balance as of January 1, 2022 | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
| Cash Flows | ( | ( | ||||||||||||||||||||||||||||||
| New Leases | ( | ( | ||||||||||||||||||||||||||||||
| Disposal Leases | ||||||||||||||||||||||||||||||||
| Amortizations from Effective Interest Method | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
| Gain on Repurchase from Convertible Bond | ||||||||||||||||||||||||||||||||
| Changes from Adjustments to Planning Assumptions | ||||||||||||||||||||||||||||||||
| Transfer of Assigned License Revenues to Royalty Pharma | ||||||||||||||||||||||||||||||||
| Value adjustment | ||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences | ( | ( | ( | |||||||||||||||||||||||||||||
| Balance as of December 31, 2022 | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
| Balance as of January 1, 2023 | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
| Cash Flows | ||||||||||||||||||||||||||||||||
| New Leases | ( | ( | ||||||||||||||||||||||||||||||
| Disposal Leases | ||||||||||||||||||||||||||||||||
| Amortizations from Effective Interest Method | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
| Gain on Repurchase from Convertible Bond | ||||||||||||||||||||||||||||||||
| Changes from Adjustments to Planning Assumptions | ||||||||||||||||||||||||||||||||
| Transfer of Assigned License Revenues to Royalty Pharma | ||||||||||||||||||||||||||||||||
| Value adjustment | ||||||||||||||||||||||||||||||||
| Foreign Currency Translation Differences | ||||||||||||||||||||||||||||||||
| Balance as of December 31, 2023 | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
The "Transfer of Assigned License Revenues to Royalty Pharma" include transactions whereas Janssen directly transfers to Royalty Pharma the settlement amount without influence by MorphoSys on timing and/or amount. As MorphoSys has not received or paid cash for these assigned license revenues, the related amounts have neither been included in the operating nor in the financing cash flow, respectively.
6.6 Geographical Disclosures
A total of € 90.6 million (December 31, 2022: € 142.7 million) of the Group’s non-current assets, excluding deferred tax assets, are located in Germany and € 1,121.7 million in the USA (December 31, 2022: € 1,165.2 million). Of the Group’s investments, € 2.9 million (2022: € 15.2 million) were made in Germany and € 0.0 million (2022: € 0.0 million) in the USA. In accordance
F-79
with internal definitions, investments solely include additions to property, plant and equipment and intangible assets not related to leases and business combinations.
6.7 Corporate Governance
The Group has submitted the Declaration of Conformity with the recommendations of the Government Commission on the German Corporate Governance Code for the 2023 financial year under Section 161 of the German Stock Corporation Act (AktG). This declaration was published on the Group’s website (https://www.morphosys.com/en/investors/corporate-governance) on November 29, 2023, and made permanently available to the public.
6.8 Research and Development Agreements
The Group has entered some research and development agreements. The following information describes the agreements that have a material effect on the Group and the developments under the research and development agreements in the 2023 financial year.
6.8.1 Proprietary Clinical Development
Partnerships in the 2023 financial year existed with (in alphabetical order) Incyte, Pfizer and Xencor.
In January 2020, MorphoSys and Incyte announced that the companies had signed a collaboration and license agreement for the continued global development and commercialization of MorphoSys’s proprietary anti-CD19 antibody tafasitamab. A detailed description of the agreement can be found in Note 4.19.
In June 2010, MorphoSys and the U.S.-based biopharmaceutical company Xencor signed an exclusive global licensing and cooperation agreement under which MorphoSys received exclusive global licensing rights to tafasitamab, the antibody for the treatment of cancer and other indications. The companies jointly conducted a Phase 1/2a trial in the U.S. in patients with chronic lymphocytic leukemia. MorphoSys was solely responsible for the further clinical development after the successful completion of the Phase 1 clinical trial and commercialization. Upon signing the license and cooperation agreement, Xencor received a payment of US$ 13.0 million (€ 10.5 million) from MorphoSys. Xencor also received milestone payments from MorphoSys totaling US$ 65.5 million (€ 53.8 million). These payments were then capitalized under in-process R&D programs. Xencor was entitled to development, regulatory and commercially related milestone payments. Furthermore, Xencor was also eligible to receive tiered royalty payments of tafasitamab in the mid single-digit to sub-teen double-digit percentage range based upon net sales of licensed antibody sold by us or our licensees. Our royalty obligations were on country-by-country basis until the later to occur of the expiration of the last valid claim in the licensed patent covering tafasitamab in such country, or 11 years after the first sale thereof following marketing authorization in such country.
In November 2020, MorphoSys, Incyte and Xencor announced a clinical collaboration agreement to study the combination of tafasitamab, plamotamab and lenalidomide in patients with r/r DLBCL, first-line DLBCL and r/r FL. In May 2022, Xencor announced the start of a Phase 2 combination study of the CD3xCD20 bispecific antibody plamotamab in combination with tafasitamab and lenalidomide in patients with relapsed or refractory DLBCL. In January 2023, Xencor announced that the company is winding down and ending enrollment in the Phase 2 study due to challenges with patient accrual in lymphoma.
In June 2022, MorphoSys, Incyte, and Pfizer announced a clinical trial collaboration and supply agreement to investigate the immunotherapeutic combination of Pfizer’s maplirpacept (TTI-622), a novel SIRPα-Fc fusion protein, and tafasitamab plus lenalidomide in patients with relapsed or refractory DLBCL who are not eligible for ASCT. Under the terms of the agreement, Pfizer initiated a multicenter, international Phase 1b/2 study of maplirpacept (TTI-622) with tafasitamab and lenalidomide. MorphoSys and Incyte provide tafasitamab for the study. The study is sponsored and funded by Pfizer and is conducted in North America, Europe, and Asia-Pacific.
In February 2024, Incyte obtained exclusive rights worldwide to tafasitamab. Incyte will assume full responsibility and cover all costs going forward for the development and commercialization of the asset. As part of the sale and transfer of tafasitamab to Incyte, Incyte also assumed the Collaboration and License Agreement with Xencor.
For the effects from the agreements with Novartis regarding the potential business combination of MorphoSys via a voluntary takeover offer and with Incyte on the sale of tafasitamab on February 5, 2024, please refer to the section "6.9 - Subsequent events" of the notes.
6.8.2 Clinical Development Through Partners
Through some commercial partnerships, MorphoSys receives various types of payments that are spread over the duration of the agreements or recognized in full as revenue as predefined targets and milestones are reached. These payments include payments
F-80
upon signature, annual license fees in exchange for access to MorphoSys’s technologies and payments for funded research to be performed by MorphoSys on behalf of the partner. MorphoSys is also entitled to milestone payments and royalties on product sales for specific antibody programs.
Prior to the 2023 financial year, active collaborations with a number of partners had already ended. However, drug development programs initiated in the active phase are designed so that they can be continued by the partner and, therefore, still result in performance-based payments for the achievement of the defined milestones.
In November 2017, MorphoSys announced it had signed an exclusive regional licensing agreement with I-Mab to develop and commercialize felzartamab in mainland China, Taiwan, Hong Kong and Macao. Felzartamab is MorphoSys’s proprietary antibody targeting CD38. Under the terms of the agreement, I-Mab has the exclusive right for the later development and commercialization of felzartamab in the agreed regions. In November 2017, MorphoSys received a payment of US$ 20.0 million (€ 16.8 million) and until 2023 milestone payments of US$ 8.0 million (€ 7.1 million). MorphoSys is also entitled to receive additional success-based clinical and commercial milestone payments from I-Mab of up to roughly US$ 90.5 million (approximately € 81.9 million). In addition, MorphoSys will be entitled to receive double-digit, staggered royalties on the net sales of felzartamab in the agreed regions. I-Mab is investigating felzartamab in a Phase 3 clinical study in Greater China in combination with lenalidomide plus dexamethasone in r/r multiple myeloma. I-Mab is also evaluating felzartamab as a potential third-line therapy in r/r multiple myeloma in a Phase 2 trial. Both studies are considered pivotal in the agreed regions.
In June 2022, MorphoSys entered into an equity participation agreement and license agreement with HI-Bio to allow HI-Bio to develop and commercialize felzartamab. Under the terms of the agreements, HI-Bio will obtain exclusive rights to develop and commercialize felzartamab across all indications worldwide, with the exception of Greater China. As part of the agreements, MorphoSys will receive an equity stake in HI-Bio, along with certain equity earn-in provisions and standard investment rights. MorphoSys will also be represented as a member of HI-Bio’s Board of Directors. On achievement of development, regulatory and commercial milestones, MorphoSys will be eligible to receive payments from HI-Bio of up to US$ 500.0 million (€ 452.5 million), in addition to tiered, single- to low double-digit royalties on net sales of felzartamab. HI-Bio will assume full responsibility for future development and commercialization expenses.
In November 2018, MorphoSys announced the signing of an exclusive strategic development collaboration and regional licensing agreement with I-Mab for MOR210/TJ210. I-Mab has exclusive rights to develop and market MOR210/TJ210 in mainland China, Hong Kong, Macao, Taiwan and South Korea. MorphoSys received an upfront payment of US$ 3.5 million (€ 3.1 million) and until 2023 milestone payments of US$ 2.5 million (€ 2.1 million). MorphoSys is further eligible to receive performance-related clinical and sales-based milestone payments of up to US$ 99.0 million (€ 89.6 million). In addition, MorphoSys will receive tiered royalties in the mid-single-digit percentage range of net sales of MOR210/TJ210 in I-Mab’s territories. In return for conducting a successful clinical proof of concept trial, I-Mab is entitled to low-single-digit royalties on net sales of MOR210/TJ210 outside the I-Mab territory, as well as staggered shares of proceeds from the further out-licensing of MOR210.
In June 2022, MorphoSys entered into an equity participation agreement and license agreements with HI-Bio to allow HI-Bio to develop and commercialize MOR210/HIB210. Under the terms of the agreement, HI-Bio will obtain exclusive rights to develop and commercialize MOR210/HIB210 across all indications worldwide, with the exception of Greater China and South Korea. On achievement of development, regulatory and commercial milestones, MorphoSys will be eligible to receive payments from HI-Bio of up to US$ 500.0 million (€ 452.5 million), in addition to tiered, single- to low double-digit royalties on net sales of MOR210/HIB210. HI-Bio will assume full responsibility for future development and commercialization expenses. Upon signing, MorphoSys also received an upfront payment of US$ 15.0 million (€ 13.6 million) for MOR210/HIB210.
The Group’s alliance with Novartis AG for the research and development of biopharmaceuticals came to an end in November 2017. The collaboration began in 2004 and led to the creation of several ongoing therapeutic antibody programs against a number of diseases, amongst others, Ianalumab (VAY736) and CMK389/NOV-8. MorphoSys receives performance-based milestones contingent upon the successful clinical development and regulatory approval of several products. In addition to these payments, MorphoSys is also entitled to royalties on any future product sales.
In December 2022, MorphoSys announced that its fully owned subsidiary Constellation Pharmaceuticals, Inc. has entered into a global licensing agreement with Novartis to research, develop and commercialize its preclinical inhibitors of a novel cancer target. Novartis will assume full responsibility for all subsequent research, development and commercialization activities for the program. As part of the agreement, MorphoSys received an immediate upfront payment of US$ 23.0 million (€ 20.8 million). On achievement of development, regulatory and commercial milestones, MorphoSys will be eligible to receive milestone payments from Novartis in addition to mid-single- to low-double-digit royalties on program net sales.
F-81
6.9 Subsequent Events
In the first quarter of 2024, MorphoSys issued a further cash-settled share-based compensation program (Performance Share Unit Program - PSU program) for certain employees of the Company (beneficiaries). In addition, a new restricted stock unit plan was established in the first quarter of 2024 for certain employees of MorphoSys US Inc. and of Constellation Pharmaceuticals, Inc. (beneficiaries).
Novartis Business Combination Agreement
On February 5, 2024, MorphoSys announced that it entered into a Business Combination Agreement with Novartis BidCo AG (formerly known as Novartis data42 AG) and Novartis AG (hereinafter collectively referred to as “Novartis”) based on Novartis’ intention to submit a voluntary public takeover offer (the “Novartis Takeover Offer”) for all of MorphoSys’ outstanding common shares in exchange for payment of € 68.0 per share. Separately, MorphoSys entered into a purchase agreement (the "Purchase Agreement") with Incyte Corporation ("Incyte") to sell and transfer to Incyte all rights worldwide related to tafasitamab for a purchase price of $ 25.0 million. MorphoSys and Incyte have been collaborating on the development and commercialization of tafasitamab since 2020. Prior to this agreement, tafasitamab was co-marketed in the U.S. by MorphoSys and Incyte as Monjuvi® (tafasitamab-cxix) and outside the U.S. by Incyte as Minjuvi®. MorphoSys’ Management Board and Supervisory Board unanimously approved both agreements.
Novartis intends to offer MorphoSys’ shareholders € 68.0 per share in cash, for a total equity value of € 2.7 billion. The offer price corresponds to a premium of 94 % and 142 % on the volume-weighted average price during the last month and three months as of the unaffected January 25, 2024 close, respectively – the day before rumors about a transaction first surfaced. It also represents a premium of 89 % to the closing share price of January 25, 2024.
Subject to a careful review of the offer document to be published by Novartis BidCo AG, MorphoSys’ Management Board and Supervisory Board intend to recommend the acceptance of the Novartis Takeover Offer. The Novartis Takeover Offer will contain customary closing conditions, in particular a minimum acceptance threshold of 65 % of MorphoSys’ share capital and regulatory clearances. The closing is currently expected to take place in the first half of 2024. MorphoSys and Novartis agreed to take MorphoSys private promptly after the Novartis Takeover Offer has been settled. There is no assurance that the business combination will be consummated on the proposed terms, timing or at all.
The offer document of the Novartis Takeover Offer will be published by Novartis BidCo AG at a later date in accordance with the provisions of the German Securities Acquisition and Takeover Act, after the German Federal Financial Supervisory Authority (“BaFin”) has approved the publication. Promptly after the offer document is published, MorphoSys’ Management Board and Supervisory Board will issue a joint reasoned statement in accordance with sec. 27 of the German Securities Acquisition and Takeover Act. In accordance with U.S. securities laws, Novartis BidCo AG and Novartis AG will file a Tender Offer Statement, which will include the offer document on Schedule TO, and MorphoSys will file a Solicitation/Recommendation Statement on Schedule with the U.S. Securities and Exchange Commission.
The transaction would result in MorphoSys' common shares being acquired by Novartis in exchange for payment. Novartis offers a price per MorphoSys share which is significantly higher than the trading price of the MorphoSys shares prior to the announcement of the transaction. Management Board believes that the minimum acceptance threshold of 65 % will be obtained and the change of control by Novartis will take place.
Based on the Business Combination Agreement, Novartis undertakes to MorphoSys to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to provide the MorphoSys Group with the financial resources required following completion of the Novartis Takeover Offer to enable the relevant MorphoSys Group companies pay any obligations arising from the implementation of the Novartis Takeover Offer as and when due, including any obligations for example, but not limited to, from the convertible bond and the obligations arising from the long-term incentive plans, each to the extent triggered by completion of the Novartis Takeover Offer.
MorphoSys expects advisory fees triggered by completion of the Novartis Takeover Offer in a mid double-digit million Euro range.
Based on the underlying contractual provisions, this potential change of control will have the following significant effects on the balance sheet and income statement of MorphoSys.
F-82
Convertible Bond
The non-subordinated, unsecured convertible bond placed in 2020 and partially redeemed via the modified reverse Dutch auction procedure amounted to € 245.7 million (current and non-current portion) as of December 31, 2023.
After the publication of the official takeover bid by Novartis, the bondholders have two options:
a.Bondholders can exercise their conversion right by submitting a conditional conversion notice, which will become effective once Novartis obtains the acceptance by more than 65 % of MorphoSys’ share capital to receive ordinary shares based on an adjusted conversion price. Since the preliminary calculated adjusted conversion price is significantly in excess of the offered € 68.0 per share, there is no economic rationale to bondholders to exercise their conversion right.
b.Bondholders can exercise their put right in the event of an acquisition of control (i.e., point in time Novartis takes over control over MorphoSys). Bonds will then be redeemed on the control record date at their principal amount plus accrued interest.
Assuming June 30, 2024, as the date Novartis will takeover control over MorphoSys, the estimated cash payment of the notional amount and estimated accrued interest to bondholders will be approx. € 262.4 million.
Share-based payment programs
In the past, MorphoSys granted various share-based payment programs ("Long-Term Incentive Plans"), as presented in sections 5.1 and 5.2 of the notes to the financial statements, to selected beneficiaries. As outlined in the Business Combination Agreement, MorphoSys and Novartis commit to use all such efforts which are from the perspective of a prudent business person reasonable and appropriate to ensure uncapped payouts of any long-term incentive plans active prior to signing of the Business Combination Agreement (the “Pre-2024 Long-Term Incentive Plans”) to Management Board members and all employees affected by any caps under German law. It will be offered to all beneficiaries to fully close out their still active Pre-2024 Long-Term Incentive Plans against payment of the offer price after the Novartis Takeover Offer.
For certain Long-Term Incentive Plans for which MorphoSys assumed an equity settlement after the vesting period (refer to section 5.1 of the notes to the financial statements), this assumption will now need to be revised to a full cash-settlement, and the respective provisions for subsequent valuation are to be applied to these programs accordingly.
MorphoSys currently expects that the Novartis takeover would result in an estimated amount of approximately € 134 million of additional expenses until the assumed change-of control event, thereof, approximately € 36 million are attributable to key management personnel. This estimation may change in the future depending on the further development of the circumstances.
With regard to the obligations arising from other contracts as outlined in section 6.1, MorphoSys currently assumes that the payments of approximately € 168 million related to the still active "Pre-2024 Long-Term Incentive Plans" programs will be made with in calendar year 2024, after a successful change-of-control and delisting of MorphoSys.
In case Management Board members or Group employees leave the Group following a completion of the Takeover Offer, MorphoSys assumes additional payouts in a mid double-digit million Euro range could occur associated with the programs granted in the first quarter 2024.
The employment contracts of key management personnel include the option to terminate the employment relationship in the event of a transfer of control. In the event of termination of the employment contract, key management personnel are still entitled to salary and bonus payments. The company currently assumes that this could result in obligations of approx. € 7 million.
Purchase Agreement with Incyte on the sale of tafasitamab
As of February 5, 2024, Incyte obtained exclusive worldwide rights, assumed full responsibility and covers all costs going forward for the development and commercialization of tafasitamab for a total cash consideration (purchase price) of $ 25.0 million under the terms of the Purchase Agreement.
Based on the Purchase Agreement, MorphoSys and Incyte agreed to transfer all relevant intellectual property rights in connection with tafasitamab to Incyte. The intangible assets relating to the underlying intellectual property rights capitalized in MorphoSys balance sheet as of February 5, 2024, amounted to approximately € 75 million. Furthermore, it was agreed that all commercial and clinical inventories in the amount of approximately € 61 million held by MorphoSys as of February 5, 2024, will also be transferred to Incyte.
During the agreed transition period of 180 days, MorphoSys will provide certain transition services relating to the ongoing tafasitamab clinical and commercial activities to Incyte. Incyte will bear the cost associated with these transitional services as incurred.
F-83
MorphoSys and Incyte have been collaborating on tafasitamab since 2020 under the Collaboration and License Agreement (refer to section 4.19). The Purchase Agreement with Incyte terminates this agreement as of February 5, 2024. Consequently, the balance sheet item "Financial Liability from Collaborations" in the amount of approximately € 116 million will be released.
In total, MorphoSys expects a net profit of approximately € 8 million from this transaction, excluding the effects from the transition services to be rendered.
Due to the Purchase Agreement with Incyte on the sale of tafasitamab, the obligations from future payments in connection with contracts for outsourced studies (as presented in section 6.1) will reduce by approx. € 129 million.
Furthermore, MorphoSys will no longer be obliged to milestone payments to licensors in the amount of US$ 236.5 million (€ 214.0 million), which were presented as contingent liabilities as of December 31, 2023.
As a consequence, MorphoSys will terminate the contracts with all customer-facing field sales employees in the U.S., which relates to approximately 7 % of total MorphoSys Group's workforce. The financial impact of this decision mainly includes severance costs as decided by Management. The communication to the affected employees took place on February 22, 2024. The provision for the matter will amount to approximately € 5 million.
Planegg, March 12, 2024
| Jean-Paul Kress, M.D. | Lucinda Crabtree, Ph.D. | ||||
| Chief Executive Officer | Chief Financial Officer | ||||
F-84
Responsibility Statement
To the best of our knowledge, and in accordance with the applicable reporting principles, the consolidated financial statements give a true and fair view of the Group’s net assets, financial position and results of operations, and the group management report provides a fair review of the development and performance of the business and the position of the Group, together with a description of the principal opportunities and risks associated with the Group’s expected development.
Planegg, March 12, 2024
| Jean-Paul Kress, M.D. | Lucinda Crabtree, Ph.D. | ||||
| Chief Executive Officer | Chief Financial Officer | ||||
F-85
EXHIBIT INDEX
Exhibit Number | Description of Document | |||||||
| 1* | ||||||||
| 2* | ||||||||
| 4.1 | ||||||||
| 4.2 | ||||||||
| 4.3 | ||||||||
| 4.4 | ||||||||
| 4.5 | ||||||||
| 4.6 | ||||||||
| 4.7 | ||||||||
| 4.8 | ||||||||
| 4.9 | ||||||||
| 4.10 | ||||||||
| 4.11 | ||||||||
| 4.12 | ||||||||
| 4.13 | ||||||||
| 4.14 | ||||||||
| 8.1* | ||||||||
| 12.1* | ||||||||
| 13.1* | ||||||||
| 15.1* | ||||||||
| 15.2* | ||||||||
| 97.1* | ||||||||
| 101.INS* | XBRL Instance Document | |||||||
| 101.SCH* | XBRL Taxonomy Extension Schema Document | |||||||
| 101.CAL* | XBRL Taxonomy Extension Calculation Linkbase Document | |||||||
| 101.DEF* | XBRL Taxonomy Extension Definition Linkbase Document | |||||||
| 101.LAB* | XBRL Taxonomy Extension Label Linkbase Document | |||||||
| 101.PRE* | XBRL Taxonomy Extension Presentation Linkbase Document | |||||||
* Filed at the SEC herewith.
† Certain information omitted pursuant to a request for confidential treatment filed separately with the SEC.
Signatures
MorphoSys AG hereby certifies that it meets all of the requirements for filing on Form 20-F and that it has duly caused and authorized the undersigned to sign this Annual Report on Form 20-F on its behalf.
MorphoSys AG (Registrant)
/s/ Jean-Paul Kress, M.D. | ||
Name: Jean-Paul Kress, M.D. | ||
| Title: CEO and member of the Board of Management | ||
| Dated: | ||