UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
OR
For the fiscal year ended
OR
OR
Date of event requiring this shell company report ______________
For the transition period from ______________ to ______________
Commission file number:
(Exact name of Registrant as specified in its charter)
N/A
(Translation of Registrant’s name into English)
AUSTRALIA
(Jurisdiction of incorporation or organization)
+ 61 3 9826 0399
(Address of principal executive offices)
Chief Executive Officer
(Name, Telephone, E-mail and/or Facsimile number and Address of Company Contact Person)
Securities registered or to be registered pursuant to Section 12(b) of the Act.
Title of each class |
Trading Symbol |
Name of each exchange on which registered |
* |
||
* Not for trading, but only in connection with the registration of the American Depositary Shares. |
|
|
Securities registered or to be registered pursuant to Section 12(g) of the Act.
None
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act:
None
Indicate the number of outstanding shares of each of the issuer’s classes of capital or common stock as of the close of the period covered by the annual report.
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. ☐ Yes ☒
If this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934. ☐ Yes ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. ☒
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). ☒
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, or a non-accelerated filer, or an emerging growth company. See definition of “large accelerated filer,” “accelerated filer,” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
Large accelerated filer ☐ |
Accelerated filer ☐ |
Emerging growth company |
If an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards† provided pursuant to Section 13(a) of the Exchange Act.
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b)
Indicate by check mark which basis of accounting the registrant has used to prepare the financial statements included in this filing:
|
☐ |
☒ |
|
|
||
U.S. GAAP |
Accounting Standards Board |
Other |
☐ |
|
||
If “Other” has been checked in response to the previous question, indicate by check mark which financial statement item the registrant has elected to follow.
Item 17 ☐ Item 18 ☐
If this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
TABLE OF CONTENTS
4 |
||||
6 |
||||
7 |
||||
9 |
||||
|
Item 1. |
|
9 |
|
|
Item 2. |
|
9 |
|
|
Item 3. |
|
9 |
|
|
3A. |
|
9 |
|
|
3B. |
|
9 |
|
|
3C. |
|
9 |
|
|
3D. |
|
9 |
|
|
Item 4. |
|
68 |
|
|
4A. |
|
68 |
|
|
4B. |
|
68 |
|
|
4C. |
|
119 |
|
|
4D. |
|
119 |
|
|
Item 4A. |
|
119 |
|
|
Item 5. |
|
119 |
|
|
5A. |
|
122 |
|
|
5B. |
|
127 |
|
|
5C. |
|
134 |
|
|
5D. |
|
134 |
|
|
5E. |
|
134 |
|
|
Item 6. |
|
139 |
|
|
6A. |
|
139 |
|
|
6B. |
|
143 |
|
|
6C. |
|
147 |
|
|
6D. |
|
150 |
|
|
6E. |
|
150 |
|
|
6F. |
|
Disclosure of a Registrant's Action to Recover Erroneously Awarded Compensation |
151 |
|
Item 7. |
|
151 |
|
|
7A. |
|
151 |
|
|
7B. |
|
153 |
|
|
7C. |
|
154 |
|
|
Item 8. |
|
155 |
|
|
8A. |
|
155 |
|
|
8B. |
|
155 |
|
|
Item 9. |
|
155 |
|
|
9A. |
|
155 |
|
|
9B. |
|
155 |
|
|
9C. |
|
155 |
|
|
9D. |
|
155 |
|
|
9E. |
|
155 |
|
|
9F. |
|
155 |
|
|
Item 10. |
|
156 |
|
|
10A. |
|
156 |
|
|
10B. |
|
156 |
|
|
10C. |
|
156 |
|
|
10D. |
|
156 |
|
|
10E. |
|
156 |
|
|
10F. |
|
164 |
|
|
10G. |
|
164 |
|
|
10H. |
|
164 |
|
|
10I. |
|
165 |
|
|
10J. |
|
165 |
|
|
Item 11. |
|
165 |
|
|
Item 12. |
|
165 |
|
|
12A. |
|
165 |
|
|
12B. |
|
165 |
|
2
TABLE OF CONTENTS
|
12C. |
|
165 |
|
|
12D. |
|
165 |
|
168 |
||||
|
Item 13. |
|
168 |
|
|
Item 14. |
|
Material Modifications to the Rights of Security Holders and Use of Proceeds |
168 |
|
Item 15. |
|
168 |
|
|
Item 16 |
|
170 |
|
|
16A. |
|
170 |
|
|
16B. |
|
170 |
|
|
16C. |
|
171 |
|
|
16D. |
|
171 |
|
|
16E. |
|
Purchases of Equity Securities by the Issuer and Affiliated Purchasers |
171 |
|
16F. |
|
171 |
|
|
16G. |
|
172 |
|
|
16H. |
|
172 |
|
|
16I. |
|
Disclosure Regarding Foreign Jurisdictions that Prevent Inspections |
172 |
|
16J. |
|
172 |
|
173 |
||||
|
Item 17. |
|
173 |
|
|
Item 18. |
|
173 |
|
|
Item 19. |
|
173 |
|
3
presentation of financial and other information
Our reporting and functional currency is the U.S. dollar, and our financial statements included elsewhere in this Annual Report on Form 20-F, or annual report, are presented in U.S. dollars. The consolidated financial statements and related notes included elsewhere in this annual report have been prepared under the International Financial Reporting Standards, or IFRS, as issued by the International Accounting Standards Board, or IASB, which differs in certain significant respects from U.S. Generally Accepted Accounting Principles, or GAAP.
Except where the context requires otherwise and for purposes of this annual report only:
Except with respect to U.S. dollar amounts presented as contractual terms, amounts denominated in U.S. dollars when received or paid and unless otherwise indicated, certain Australian dollar amounts contained in this annual report have been translated into U.S. dollars at the rate published by the Reserve Bank of Australia as of June 30, 2023. These translations should not be considered representations that any such amounts have been, could have been or could be converted into U.S. dollars or Australian dollars at that or any other exchange rate as of that or any other rate. We have made rounding adjustments to some of the figures included in this annual report. Accordingly, numerical figures shown as totals in some tables may not be an arithmetic aggregation of the figures that precede them.
This annual report includes trademarks, tradenames and service marks, certain of which belong to us and others that are the property of other organizations. Solely for convenience, trademarks and tradenames referred to in this annual report appear without the ® and ™ symbols, but the absence of those references is not intended to indicate, in any way, that we will not assert our rights or that the applicable owner will not assert its rights to these trademarks and tradenames to the fullest extent under applicable law. We do not intend our use or display of other parties’ trademarks, trade names or service marks to imply, and such use or display should not be construed to imply, a relationship with, or endorsement or sponsorship of us by, these other parties.
In July 2023, Opthea announced “sozinibercept” as the non-proprietary drug name for OPT-302. The American Medical Association’s United States Adopted Names (USAN) Council, in consultation with the World Health Organization’s International Non-proprietary Names (INN) Expert Committee, approved and adopted the non-proprietary drug name. Opthea will use the name sozinibercept (formerly OPT-302) in upcoming publications, public statements, and in corporate materials moving forward.
This annual report contains estimates and information concerning our industry and our business, including estimated market size and projected growth rates of the markets for our product candidates. Unless otherwise expressly stated, we obtained this industry, business, market, medical and other information from reports, research surveys, studies and similar data prepared by third parties, industry, medical and general publications, government data and similar sources.
4
This information involves a number of assumptions and limitations. Although we are responsible for all of the disclosure contained in this annual report and we believe the third-party market position, market opportunity and market size data included in this annual report are reliable, we have not independently verified the accuracy or completeness of this third-party data. In addition, projections, assumptions and estimates of our future performance and the future performance of the industry in which we operate are necessarily subject to a high degree of uncertainty and risk due to a variety of factors, including those described in “Risk Factors” and in “Forward-Looking Statements.” These and other factors could cause results to differ materially from those expressed in the estimates made by the independent parties and by us.
Australian Disclosure Requirements
Our ordinary shares are primarily quoted on the Australian Securities Exchange (“ASX”) in addition to our listing of our ADSs on the Nasdaq Global Select Market, or Nasdaq. As part of our ASX listing, we are required to comply with various disclosure requirements as set out under the Australian Corporations Act 2001 and the ASX Listing Rules. Information furnished under the sub-heading “Australian Disclosure Requirements” is intended to comply with ASX listing and Corporations Act 2001 disclosure requirements and is not intended to fulfill information required by this annual report.
5
ENFORCEMENT OF CIVIL LIABILITIES
We are a public limited company incorporated under the laws of Australia. Certain of our directors are non-residents of the United States and substantially all of their assets are located outside the United States. As a result, it may not be possible or practicable for you to:
You may also have difficulties enforcing in courts outside the United States judgments that are obtained in U.S. courts against any of our non-U.S. resident directors or us, including actions under the civil liability provisions of the U.S. securities laws.
With that noted, there are no treaties between Australia and the United States that would affect the recognition or enforcement of foreign judgments in Australia. We also note that investors may be able to bring an original action in an Australian court against us to enforce liabilities based in part upon U.S. federal securities laws. The disclosure in this section is not based on the opinion of counsel.
We have appointed Corporation Service Company, located at 1180 Avenue of the Americas, Suite 210, New York, NY 10036, as our agent to receive service of process with respect to any action brought against us under the federal securities laws of the United States.
6
FORWARD-LOOKING STATEMENTS
This annual report contains forward-looking statements about us and our industry that involve substantial risks and uncertainties. All statements other than statements of historical facts contained in this annual report, including statements regarding our future results of operations, financial condition, business strategy and plans and objectives of management for future operations, are forward-looking statements. In some cases, you can identify forward-looking statements because they contain words such as “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” or “would,” or the negative of these words or other similar terms or expressions.
We have based these forward-looking statements largely on our current expectations and projections about future events and trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. These forward-looking statements are subject to a number of known and unknown risks, uncertainties, other factors and assumptions, including the risks described in “Risk Factors” and elsewhere in this annual report, regarding, among other things:
7
These risks are not exhaustive. Other sections of this annual report may include additional factors that could harm our business and financial performance. New risk factors may emerge from time to time, and it is not possible for our management to predict all risk factors, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in, or implied by, any forward-looking statements.
You should not rely on forward-looking statements as predictions of future events. We have based the forward-looking statements contained in this annual report primarily on our current expectations and projections about future events and trends that we believe may affect our business, financial condition and operating results. We undertake no obligation to update any forward-looking statements made in this annual report to reflect events or circumstances after the date of this annual report or to reflect new information or the occurrence of unanticipated events, except as required by law. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and you should not place undue reliance on our forward-looking statements. Our forward-looking statements do not reflect the potential impact of any future acquisitions, mergers, dispositions, joint ventures or investments.
In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based on information available to us as of the date of this annual report. While we believe that information provides a reasonable basis for these statements, that information may be limited or incomplete. Our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all relevant information. These statements are inherently uncertain, and investors are cautioned not to unduly rely on these statements.
You should read this annual report and the documents that we reference in this annual report and have filed as exhibits to this annual report with the understanding that our actual future results, levels of activity, performance and achievements may be different from what we expect. We qualify all of our forward-looking statements by these cautionary statements.
8
PART I
Item 1. Identity of Directors, Senior Management and Advisers
Not applicable.
Item 2. Offer Statistics and Expected Timetable
Not applicable.
Item 3. Key Information
3A. Reserved
3B. Capitalization and Indebtedness
Not applicable.
3C. Reasons for the offer and use of proceeds
Not applicable.
3D. Risk Factors
Investing in our securities involves a high degree of risk. You should consider and read carefully all of the risks and uncertainties described below, as well as other information included in this annual report, including our consolidated financial statements and related notes included elsewhere in this annual report, before making an investment decision. If any of the following risks actually occur, it could harm our business, prospects, results of operations and financial condition. In such event, the trading price of the ADSs could decline and you might lose all or part of your investment. You should not interpret our disclosure of any of the following risks to imply that such risks have not already materialized.
Risk Factors Summary
Our business is subject to a number of risks and uncertainties, including those risks discussed at-length below in this summary. These risks include, among others, the following:
Risks Related to the Development Funding Agreement with Ocelot SPV LP
9
Risks Related to our Financial Position and Need for Capital
Risks Related to Development and Commercialization of Our Product Candidate
10
Risks Related to Legal and Regulatory Compliance Matters
Risks Related to Our Reliance on Third Parties
Risks Related to Employee Matters and Managing Our Growth
Risks Related to Intellectual Property
Risks Related to Ownership of the ADSs
11
Risks Related to Our Financial Position and Need for Capital
We are a clinical-stage biopharmaceutical company with no products approved for commercial sale. We have incurred net losses since our inception, we expect to incur significant losses and increasing operating losses for the foreseeable future, and we may never be profitable.
We are a clinical-stage biopharmaceutical company with no products approved for commercial sale. To date, our operations have been limited to organizing and staffing our company, business planning, raising capital, developing our product candidate, Sozinibercept (OPT-302), and licensing certain related technology, conducting research and development activities, including preclinical studies and clinical trials, and providing general and administrative support for these operations. Investment in biopharmaceutical product development is highly speculative because it entails substantial upfront capital expenditures and significant risk that any potential product candidate will fail to demonstrate adequate effect and/or an acceptable safety profile, gain regulatory approval and become commercially viable. We have not generated any revenue from product sales to date, and we continue to incur significant research and development and other expenses related to our ongoing operations. We are not profitable and have incurred net losses since our inception. Our total comprehensive losses were US$92.8 million and US$142.4 million for the years ended June 30, 2022 and 2023. As of June 30, 2023, we had an accumulated loss of US$359.4 million. We have spent, and expect to continue to spend, significant resources to fund research and development of, and seek regulatory approvals for, Sozinibercept (OPT-302) and any future product candidates. The pivotal phase 3 studies for Sozinibercept (OPT-302), COAST and ShORe have been delayed and the cost to complete the studies is higher than anticipated. A new co-investor of Carlyle and Abingworth intends to participate in a funding under the Funding Agreement of US$50 million to increase total Funding Agreement funding from $120 million to $170 million which is subject to the co-investor’s final due diligence and receipt of regulatory and tax approvals, appropriate documentation and compliance with closing conditions. There can be no assurance that the due diligence will be completed to the satisfaction of the co-investor of Carlyle and Abingworth, that the closing terms and conditions will be satisfied or that we will ultimately receive the additional $50 million. If the additional US$50 million is not received by June 30, 2024, we will need to raise additional funds or reduce expenditures to continue as a going concern. In August 2023 Opthea announced a non-underwritten institutional placement (“Placement”) and accelerated non-renounceable entitlement offering (“ANREO”) offering of A$90 million,- (approximately US$58 million). We have received net proceeds of approximately $54 million from the Placement and ANREO (together, the "2023 Equity Offering") and we anticipate receiving $35 million from the third tranche of funding under the Funding Agreement by December 31, 2023.
While we expect that with our cash on hand at June 30, 2023 of $89 million, together with the net proceeds from the 2023 Equity Offering and the $85 million expected under the Funding Agreement, we will be able to fund our operations through the third calendar quarter of 2024, such proceeds will not be sufficient to fully fund all anticipated costs of the Phase 3 clinical trials to top-line data. We will need to raise additional funds to complete the efficacy and safety phase of both studies and to report top-line data. In addition, the forecast of our cash runway above, following receipt of the proceeds from the 2023 Equity Offering and under the Funding Agreement, is subject to a number of assumptions, including the timing of completion of Phase 3 clinical trial patient enrollment and Clinical Research Organization ("CRO") and labor costs. Estimated patient enrollment timing used for our forecast of our cash runway is based on our monthly enrollment rates for our Phase 3 clinical trials, which timing has in the past significantly fluctuated from prior estimates, including due to factors outside our control. We have in the past incurred significantly increased costs in connection with the activities conducted by third party CROs and other service providers to prepare for and progress our Phase 3 clinical trials, and may continue to incur higher than expected costs for such activities in the future. If patient enrollment continues to be delayed in the future, or if any additional factors cause the Phase 3 clinical trials to be further delayed or more costly, including higher than expected CRO and labor costs, then we will need to obtain additional financing earlier than our forecast. If we fail to obtain additional capital from other sources, prior to top-line data for our Phase 3 clinical trials, which may not be available on a timely basis or at all, we could be forced to delay, limit or terminate our operations, liquidate all or a portion of our assets and/or seek insolvency protection in the near term. Our failure to raise capital, if and when needed, could delay or suspend our business strategy and could have a material adverse effect on our activities.
If sufficient capital is not available, we may seek to modify the original trial design and protocol. We expect to incur substantial and increasing operating losses over the next several years as our research, development, manufacturing and clinical trial activities increase. Additionally, if Sozinibercept (OPT-302) is approved for commercial sale, our commercialization expenses will increase significantly as we seek a commercialization
12
partner or establish sales, marketing, distribution, manufacturing, supply chain and other commercial infrastructure. As a result, our accumulated losses will also increase significantly. We may encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may negatively affect our business. The size of our future net losses will depend, in part, on the rate of future growth of our expenses and our ability to generate revenue. Our prior losses and expected future losses have had and will continue to have a negative impact on our total (deficit) equity and working capital. The net losses we incur may fluctuate significantly from quarter-to-quarter such that a period-to-period comparison of our results of operations may not be a good indication of our future performance. Even if we eventually generate product revenue, we may never be profitable and, if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. If Sozinibercept is approved in a major market (as defined in the Funding Agreement), within 90 days of approval, we must make a fixed payment to Ocelot followed by six annual payments. Ocelot will also receive a variable payment of 7% of net sales. To date, Ocelot has invested $85 million and we anticipate the receipt of an additional $35 million before the end of calendar 2023. If Sozinibercept is approved, - Ocelot will receive four times invested capital or $680 million over approximately six years. We anticipate that profits generated by the sales of Sozinibercept should be able to fund this repayment, however there can be no assurances that we will have sufficient cash resources to repay this amount when it is due.
We currently have no source of product revenue and may never become profitable.
Sozinibercept (OPT-302) has not been approved for commercial sale, and we expect it to be several years before Sozinibercept (OPT-302) is approved, if ever, and we are able to commence sales of Sozinibercept (OPT-302). To date, we have not generated any revenue from the licensing or commercialization of Sozinibercept (OPT-302) and do not expect to receive revenue from it for a number of years, if ever. We will not be able to generate product revenue unless and until Sozinibercept (OPT-302) or any future product candidate, alone or with future partners, successfully completes clinical trials, receives regulatory approval and is successfully commercialized. Although we may seek to obtain revenue from collaboration or licensing agreements with third parties, we currently have no such agreements that could provide us with material, ongoing future revenue and we may never enter into any such agreements. Our ability to generate future product revenue from Sozinibercept (OPT-302) or any future product candidates also depends on a number of additional factors, including our or our future partners’ ability to:
13
In addition, because of the numerous risks and uncertainties associated with biologic product development, including that Sozinibercept (OPT-302) may not advance through development, achieve the endpoints of applicable clinical trials or receive approval for use in combination with one or more approved therapies, we are unable to predict the timing or amount of increased expenses, or if or when we will achieve or maintain profitability. In addition, our expenses could increase beyond expectations if we decide, or are required by the U.S. Food and Drug Administration, or the FDA, or comparable non-U.S. regulatory authorities, including the European Medicines Agency, or the EMA, to perform studies or trials in addition to those that we currently anticipate. Even if we complete the development and regulatory processes described above, we anticipate incurring significant costs associated with launching and commercializing these products.
Even if we generate revenue from the sale of any of our product candidates that may be approved, we may not become profitable and may need to obtain additional funding to continue operations. If we fail to become profitable or do not sustain profitability on a continuing basis, then we may be unable to continue our operations at planned levels and be forced to reduce our operations.
We will require substantial additional capital to finance our operations, which may not be available to us on acceptable terms, or at all. As a result, we may not complete the development and commercialization of Sozinibercept (OPT-302) or develop new product candidates.
As a clinical-stage biopharmaceutical company, our operations have consumed significant amounts of cash since our inception. We expect our research and development expenses to increase substantially in connection with our ongoing activities, particularly as we continue our Phase 3 clinical trials of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD, continue clinical development of Sozinibercept (OPT-302) in combination with aflibercept for the treatment of persistent diabetic macular edema, or DME, and other retinal diseases, and continue commercialization plans. Even if we are able to obtain regulatory approval for Sozinibercept (OPT-302) and any future product candidates that we may develop, we will require substantial additional capital to commercialize such product candidates.
14
We will require additional capital to develop, obtain regulatory approval for and commercialize Sozinibercept (OPT-302) and any future product candidates, including to complete our ongoing Phase 3 clinical trials for Sozinibercept (OPT-302) for the treatment of wet AMD. In particular, we will require additional capital to progress our ongoing and future planned clinical trials without delays, including payments to Ocelot in connection with the achievement of certain regulatory milestones. We may also require additional external funding to meet the minimum cash condition under our development funding agreement with Ocelot, including prior to the readout of top-line results for our Phase 3 clinical trials for Sozinibercept (OPT-302) for the treatment of wet AMD. A new co-investor of Carlyle and Abingworth intends to participate in a funding under the Funding Agreement of US$50 million to increase total Funding Agreement funding from $120 million to $170 million which is subject to the co-investor's final due diligence and receipt of regulatory and tax approvals, appropriate documentation and compliance with closing conditions. There can be no assurance that the due diligence will be completed to the satisfaction of the co-investor of Carlyle and Abingworth, the closing terms and conditions will be satisfied or that we will ultimately receive the additional US$50 million. We anticipate receiving the third tranche of the $35 million under the Funding Agreement by December 31, 2023. However, there can be no assurance that we will meet the conditions under such agreement to receive any future payments, some of which are payable at Ocelot's discretion. We do not have any other committed external source of funds. We expect to finance future cash needs through public or private equity or collaborations. However, our Development Funding Agreement with Ocelot limits the types of financing we may pursue in the future. We also intend to continue to apply for tax incentives under the Research and Development Tax Incentive scheme provided by the Australian government. See “—Risks
15
Related to Development and Commercialization of Our Product Candidates—We have received tax credits under the Research and Development Tax Incentive scheme in Australia that may become repayable if we did not or do not comply with the rules of the scheme, or we may become ineligible for tax credits in our current or future tax years, which could harm our business, financial condition and results of operations.” Additional capital may not be available in sufficient amounts or on reasonable terms, if at all. If we are not able to raise additional capital, we may not be able to complete the clinical trials or complete them as originally planned, manufacture materials in preparation for filing and if approved commercialization continue operations or otherwise capitalize on our business opportunities, and our business and financial condition will be negatively impacted.
Failure to remain in compliance with our obligations under the Development Funding Agreement with Ocelot could lead to reduced funding under the agreement and/or the acceleration of potentially significant payments to Ocelot.
On August 12, 2022, we entered into a Development Funding Agreement (the “Funding Agreement”) with Ocelot SPV LP ("Ocelot" or "Investor"), an affiliate of Carlyle and Abingworth, in collaboration with Carlyle and Abingworth’s recently formed development company Launch Therapeutics (“Launch Tx”), pursuant to which Investor agreed to provide funding to support our development of Sozinibercept (OPT-302) for the treatment of wet AMD. Pursuant to the Funding Agreement, Investor has committed to provide Opthea US$120 million in funding which may be increased up to $170 million at Investor’s option, of which $85 million has already been funded with the remainder being funded in an additional tranche of $35 million to be paid on December 31, 2023. In the event that third tranche of US$35 million is not paid by December 31, 2023 it would be considered a Fundamental Material Breach of the Funding Agreement by Carlyle and Abingworth. Under a Fundamental Material Breach of the Funding Agreement, we have limited recourse but would have the ability to terminate the Funding Agreement by Carlyle and Abingworth. Although termination by us for lack of payment by Carlyle and Abingworth of the US$35 million would relieve us from any repayments under the Funding Agreement, failure to receive the third tranche of US$35 million or the increased funding of US$50 million would have a negative impact on our expected cash runway and ability to complete enrollment in the ongoing trials.
A new co-investor of Carlyle and Abingworth intends to participate in a funding under the Funding Agreement of US$50 million to increase total Funding Agreement funding from $120 million to $170 million which is subject to the co-investor's final due diligence and receipt of regulatory and tax approvals, appropriate documentation and compliance with closing conditions. There can be no assurance that the due diligence will be completed to the satisfaction of the co-investor of Carlyle and Abingworth, the closing terms and conditions will be satisfied or that we will ultimately receive the additional US$50 million. Under the Funding Agreement, Opthea will pay to Investor (1) upon the first to occur of regulatory approval of Sozinibercept (OPT-302) for the treatment of wet AMD in the United States, United Kingdom or European Union (“Regulatory Approval”), fixed payments equal to a total of approximately two times the funding provided, consisting of seven payments, with the first payment due shortly after Regulatory Approval and the remaining six payments payable over a six-year period thereafter, and (2) variable payments equal to 7% of net sales of Sozinibercept (OPT-302) for the treatment of wet AMD for each calendar quarter. At the time that Investor receives an aggregate of four times the funding provided (US$680 million if Investor funds the full US$170 million under the Funding Agreement), Opthea’s payment obligations under the Funding Agreement will be fully satisfied.
The Funding Agreement terminates upon the payment of all payments owing to Investor, unless earlier terminated. The Funding Agreement may be earlier terminated by Investor if:
16
In certain instances which may result upon the termination of the Funding Agreement, we will be obligated to pay Investor several multiples of the amounts paid to us under the Funding Agreement.
The payments required under the Funding Agreement are significant. Failure to generate sufficient revenue to make such payments if and as they become due, or failure to otherwise finance such payments would have a material adverse effect on our business. In addition, if we are unable to comply with our obligations under the Funding Agreement and/or one of the termination events described above occurs, Investor may be relieved of their obligation to provide further funding under the Funding Agreement and our payment obligations thereunder may be accelerated. The acceleration of payments under the Funding Agreement would have a material impact on our business and we may not be able to make such payments at such time. We may also require additional external funding to meet the minimum cash condition under the Funding Agreement, including prior to the readout of top-line results for our Phase 3 clinical trials for Sozinibercept (OPT-302) for the treatment of wet AMD. Furthermore, the obligations under the Funding Agreement are secured by a lien on all of our assets (other than intellectual property not related to Sozinibercept (OPT-302)). The security interest will terminate when Investor receives payments and/or change of control acceleration payments equal to two times the funding provided or upon certain terminations of the Funding Agreement. A default under the Funding Agreement may result in a foreclosure on our intellectual property and seizure of all of our assets. In addition, we may need to implement further internal controls and processes to ensure compliance with all obligations under the Funding Agreement, otherwise we could inadvertently default under it. For additional details regarding the Funding Agreement, see Note 25 Financial Liabilities to the Consolidated Financial Statements in this annual report.
Raising additional capital may cause dilution to holders of our ordinary shares and ADSs, restrict our operations or require us to relinquish rights to our technologies or product candidates.
Holders of our ordinary shares and ADSs could suffer dilution or be negatively affected by fixed payment obligations we may incur if we raise additional funds through the issuance of additional equity securities or debt. In February 2022, we established an “at the market” program (the “ATM Program”) with Jefferies LLC (“Jefferies”). Pursuant to the ATM Program, we may offer and sell up to US$75 million of our ordinary shares in the form of ADSs, with each ADS representing eight ordinary shares, through Jefferies. Issuances of our ordinary shares sold pursuant to the sales agreement will have a dilutive effect on our existing stockholders. Further, these securities may have rights senior to those of our ordinary shares and could contain covenants or protective rights that would restrict our operations and potentially impair our competitiveness, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions that could negatively impact our ability to conduct our business. If we need to secure additional financing, such additional fundraising efforts may divert our management and research efforts from our day-to-day activities, which may negatively affect our ability to develop and commercialize Sozinibercept (OPT-302) and any future product candidates.
To the extent we obtain additional funding through product collaborations, these arrangements would generally require us to relinquish rights to some of our technologies, product candidates or products, and we may not be able to enter into such agreements, on acceptable terms, if at all. If we are unable to obtain additional funding on a timely basis, we may be required to curtail or terminate some or all of our development programs or product candidates.
Unstable market and economic conditions may have serious adverse consequences on our business and financial condition.
Global credit and financial markets have experienced extreme disruptions at various points over the last few decades, characterized by diminished liquidity and credit availability, declines in consumer confidence, declines in economic growth, rising inflation and interest rates, bank failures, supply chain disruptions, increases in unemployment rates and uncertainty about economic stability. If another such disruption in credit and financial
17
markets and deterioration of confidence in economic conditions occurs, our business may be harmed. If the equity and credit markets were to deteriorate significantly in the future, it may make any necessary debt or equity financing more difficult to complete, more costly and more dilutive. Failure to secure any necessary financing in a timely manner and on favorable terms could harm our growth strategy, financial performance and share price and could require us to delay or abandon development or commercialization plans. In addition, there is a risk that one or more of our service providers, manufacturers or other partners would not survive or be able to meet their commitments to us under such circumstances, which could directly affect our ability to attain our operating goals on schedule and on budget.
Risks Related to Development and Commercialization of Our Product Candidates
Our business substantially depends on the success of Sozinibercept (OPT-302), our only product candidate under clinical development, which has not completed a pivotal Phase 3 clinical trial. If we are unable to obtain regulatory approval for and successfully commercialize Sozinibercept (OPT-302) or any future product candidates, or we experience significant delays in doing so, our business will be harmed.
To date, the primary focus of our product development has been Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of patients with wet AMD and DME. Currently, Sozinibercept (OPT-302) is our only product candidate under clinical development. This may make an investment in our company riskier than similar companies that have multiple product candidates in active development and that therefore may be able to better sustain a failure of a lead candidate. Successful continued development and ultimate regulatory approval of Sozinibercept (OPT-302) combination therapy for the treatment of wet AMD, DME or other indications is critical to the future success of our business. We have invested, and will continue to invest, a significant portion of our time and financial resources in the clinical development of Sozinibercept (OPT-302). If we cannot successfully develop, obtain regulatory approval for and commercialize Sozinibercept (OPT-302), we may not be able to continue our operations. The future regulatory and commercial success of Sozinibercept (OPT-302) is subject to a number of risks, including the following:
18
Masked data from patients that have completed the week 52 visit in the ongoing Phase 3 clinical trials show greater mean best corrected visual acuity ("BCVA") increases from baseline than results with standard of care anti-VEGF-A monotherapy from our Phase 2b study. Masked data represent pooled data from both OPT-302 combination and standard of care monotherapy treatment arms. However, the Phase 3 clinical trial masked data are incomplete and subject to additional analysis once unmasked, and our Phase 3 clinical trials are not fully enrolled and the majority of patients enrolled in the trial have not completed the week 52 visit. There is no assurance that standard of care monotherapy in our Phase 3 clinical trials will yield similar results to our prior clinical trials or previously published clinical trials with anti-VEGF-A monotherapies. As a result, there can be no assurance that top-line results for Sozinibercept (OPT-302) from the Phase 3 clinical trial, if completed, will be consistent with results from masked data available to date.
Many of these risks as described above are beyond our control, including the risks related to clinical development, the regulatory submission process, potential threats to our intellectual property rights and the manufacturing, marketing and sales efforts of any future collaborator. Of the large number of drugs in development in the pharmaceutical industry, only a small percentage result in the submission of a new drug application or a biologics license application, or BLA, to the FDA and even fewer are approved for commercialization. Furthermore, even if we do receive regulatory approval to market Sozinibercept (OPT-302), any such approval may be subject to limitations on the indicated uses or patient populations for which we may market the products. Accordingly, even if we are able to obtain the requisite financing to continue to fund our development programs, we may be unable to successfully develop or commercialize Sozinibercept (OPT-302). If we or any of our future development collaborators are unable to develop, or obtain regulatory approval for, or, if approved, successfully commercialize Sozinibercept (OPT-302), we may not be able to generate sufficient revenue to continue our business.
Clinical drug development involves a lengthy and expensive process with uncertain timelines and uncertain outcomes. Our clinical trials may fail to adequately demonstrate the safety and efficacy of Sozinibercept (OPT-302) or any future product candidates.
Sozinibercept (OPT-302) and any future product candidates will be subject to rigorous and extensive clinical trials and extensive regulatory approval processes implemented by the FDA and comparable non-U.S. regulatory authorities before obtaining marketing approval from these regulatory authorities. The drug development and approval process is lengthy and expensive, and approval is never certain. For example, we expect to complete patient recruitment in the COAST study in the first calendar quarter of 2024 and in the ShORe study in the second calendar quarter of 2024. Top-line data is expected to be reported when all patients complete the 52-week treatment period for the primary analysis. Investigational new drugs, such as Sozinibercept (OPT-302), may not prove to be safe and effective in clinical trials. We may be unable to conduct clinical trials at preferred sites, enlist clinical investigators, enroll sufficient numbers of participants or begin or successfully complete clinical trials in a timely fashion, if at all. Our estimated patient enrollment timing is based on our monthly enrollment rates for our Phase 3 clinical trials, which timing has in the past significantly fluctuated from prior estimates, including due to factors outside our control. In particular, we have incurred and experienced, and may continue to incur and experience in the future, significantly increased costs and delays in connection with the activities conducted by third-party CROs and other third parties progress our Phase 3 clinical trials, including due to delays and difficulties in enrolling eligible participants and opening clinical trial sites. In addition, the design of a clinical trial can determine whether its results will support approval of a product, and flaws in the design of a clinical trial may not become apparent until the clinical trial is well advanced.
19
We have in the past experienced, and if we continue to experience delays in clinical testing, our commercial prospects will be harmed, our costs may increase and our business may be harmed.
Conducting clinical studies for any product candidates in the United States requires filing an investigational new drug application, or IND, and reaching agreement with the FDA on clinical protocols, finding appropriate clinical sites and clinical investigators, securing approvals for such studies from the institutional review board at each such site, manufacturing clinical quantities of product candidates and supplying drug product to clinical sites. Currently, we have an active IND with the FDA in the United States for Sozinibercept (OPT-302). If any such future IND is not cleared by the FDA, our clinical development timeline may be negatively impacted and any future clinical programs may be delayed or terminated.
We cannot guarantee that we will be able to successfully accomplish required regulatory activities or all of the other activities necessary to initiate and complete clinical trials. As a result, our clinical trials may be extended, delayed or terminated, and we may be unable to obtain regulatory approvals or successfully commercialize our products. We do not know whether any other clinical trials will begin as planned, will need to be restructured or will be completed on schedule, or at all. In particular, we have incurred and experienced, and may continue to incur and experience in the future, significantly increased costs and delays in connection with the activities conducted by third-party CROs and other third parties to progress our Phase 3 clinical trials including due to delays and difficulties in enrolling eligible participants and opening clinical trial sites. Our product development costs will increase if we continue to experience delays in clinical testing. Significant clinical trial delays also could shorten any periods during which we may have the exclusive right to commercialize Sozinibercept (OPT-302) and any future product candidates or allow our competitors to bring products to market before we do, which would impair our ability to successfully commercialize Sozinibercept (OPT-302) or any future product candidates and may harm our business, results of operations and prospects. Events that have in the past and may in the future result in a delay or unsuccessful completion of clinical development include:
20
Our or our future collaborators’ inability to timely complete clinical trials could result in additional costs to us as well as impair our ability to generate product revenue, continue development, commercialize Sozinibercept (OPT-302) and any future product candidates and receive royalties on product sales. In addition, if we make changes to a product candidate, we may need to conduct additional nonclinical studies or clinical trials to bridge or demonstrate the comparability of our modified product candidate to earlier versions, which could delay our clinical development plan or marketing approval for our current product candidate and any future product candidates.
We have in the past encountered difficulties in enrolling patients in our clinical trials and if we encounter such difficulties in the future, our clinical development activities could be delayed or otherwise negatively affected.
The timely completion of clinical trials largely depends on patient enrollment. Enrollment in our Phase 3 clinical trials has been challenged in part by the COVID-19 pandemic, supply chain issues, global and regional inflation, national and local recessions, challenges in hiring, qualified staff (at sites, our CRO and distribution locations), local regulatory approvals importation and custom requirements and administrative delays. We expect to complete patient recruitment in the COAST study in the first calendar quarter of 2024 and in the ShORe study in the second calendar quarter of 2024. Top-line data is expected to be reported when all patients complete the 52-week treatment period for the primary analysis. Our estimated patient enrollment timing is based on our monthly enrollment rates for our Phase 3 clinical trials, but we have encountered delays in the past and may in the future encounter delays in enrolling, or be unable to enroll, a sufficient number of patients to complete any of our clinical trials, and even once enrolled, we may be unable to retain a sufficient number of patients to complete any of our trials. Many factors have affected enrollment for our Phase 3 clinical trials, as described above, and additional factors may continue to affect patient enrollment, including:
21
In addition, other companies are conducting clinical trials for the same indications and seek to enroll patients in their trials that may otherwise be eligible for our clinical studies or trials, which could lead to slow recruitment and delays in our clinical programs. Further, since the number of qualified clinical investigators is limited, we expect to conduct some of our clinical trials at the same clinical trial sites that some of our competitors use, which could further reduce the number of patients who are available for our clinical trials in these sites.
Our inability to enroll a sufficient number of patients for our clinical trials would result in additional significant delays or may require us to abandon one or more clinical trials altogether. If we are unable to enroll a sufficient number of patients that will complete clinical testing, we will be unable to seek or gain marketing approval for Sozinibercept (OPT-302) and any future product candidates and our business will be harmed. Even if we are able to enroll a sufficient number of patients in our clinical studies or trials, delays in patient enrollment have in the past resulted, and may in the future result in increased costs or delays and other impacts on the timing or outcome of our clinical trials, which could prevent completion of these trials and negatively affect our ability to advance the development of Sozinibercept (OPT-302) and any future product candidates.
Sozinibercept (OPT-302) and any future product candidates may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval or limit the commercial profile of an approved label.
Undesirable side effects caused by Sozinibercept (OPT-302) combination therapy or any future product candidates could cause us or regulatory authorities to interrupt, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of regulatory approval by the FDA or other comparable non-U.S. regulatory authorities. Additional clinical studies may be required to evaluate the safety profile of Sozinibercept (OPT-302) combination therapy or any future product candidates. We have no clinical safety data on patient exposure to Sozinibercept (OPT-302) administered in combination with an anti-VEGF-A therapy for longer than 72 weeks.
Future results of our trials could reveal a high and unacceptable severity and prevalence of side effects or unexpected characteristics, including, for example, immunogenicity. In such an event, we could suspend or terminate our trials, or the FDA or comparable non-U.S. regulatory authorities could order us to cease clinical trials or deny approval of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy or any future product candidates for any or all targeted indications. Drug-related side effects could affect patient recruitment or the ability of enrolled subjects to complete the trial or result in potential product liability claims. Any of these occurrences could materially and negatively affect our business, financial condition, results of operations and prospects. While Sozinibercept (OPT-302) has been well tolerated in our completed clinical trials, dosed patients have experienced certain adverse events, including potentially treatment-related serious adverse events, or SAEs, of myocardial infarction, endophthalmitis and vitritis in our Phase 2b clinical trial of Sozinibercept (OPT-302) combination therapy for the treatment of wet AMD, and a potentially treatment-related SAE of stroke for one patient in our Phase 1b/2a clinical trial of Sozinibercept (OPT-302) combination therapy for the treatment of DME.
22
It may be difficult to discern whether certain events or symptoms observed during our clinical trials or by patients using our approved products are related to Sozinibercept (OPT-302) or any future product candidates or approved products, including anti-VEGF-A therapies used in combination with Sozinibercept (OPT-302), or some other factor. As a result, we and our development programs may be negatively affected even if such events or symptoms are ultimately determined to be unlikely related to Sozinibercept (OPT-302) or any future product candidates or approved products. We are developing Sozinibercept (OPT-302) to complement existing VEGF-A inhibitors, including ranibizumab and aflibercept. There are some potential side effects associated with intravitreal anti-VEGF-A therapies such as intraocular hemorrhage, intraocular pressure elevation, retinal detachment, inflammation, vasculitis, artery occlusion or infection inside the eye and over-inhibition of VEGF, as well as the potential for potential systemic side effects such as heart attack, stroke, wound-healing problems and high blood pressure. Further, Sozinibercept (OPT-302) in combination with anti-VEGF-A therapies for the treatment of wet AMD is administered as sequential intravitreal injections over several weeks. There are risks inherent in the intravitreal injection procedure of drugs such as existing anti-VEGF-A therapies in combination with Sozinibercept (OPT-302) which can cause injury to the eye and other complications including conjunctival hemorrhage, punctate keratitis, eye pain, conjunctival hyperemia, which results in a discharge, intraocular inflammation and inflammation of the interior of the eye. For example, in our completed clinical trials, patients dosed with Sozinibercept (OPT-302) have experienced potentially treatment-related ocular adverse events such as eye pain, vitreous floaters, eye irritation and raised intraocular pressure.
We cannot assure you that additional or more severe adverse side effects than those observed to date related to Sozinibercept (OPT-302) combination therapy or any future product candidates will not be observed in our clinical trials or in the commercial setting. If observed, such adverse side effects could delay or preclude regulatory approval of Sozinibercept (OPT-302) combination therapy or any future product candidates, limit commercial use or result in the withdrawal of previously granted marketing approvals. If we or others identify undesirable or unacceptable side effects caused by Sozinibercept (OPT-302) combination therapy or any future product candidates or products:
Any of these results could decrease or prevent any sales of our approved product or substantially increase the costs and expenses of commercializing and marketing our product.
Even if we complete the necessary Phase 3 pivotal clinical trials, the marketing approval process is expensive, time-consuming and uncertain and may prevent us or any future collaboration partners from obtaining
23
approvals for the commercialization of Sozinibercept (OPT-302) for the treatment of wet AMD or any other indication as well as for any other product candidate we develop.
Any product candidate we may develop and the activities associated with their development and commercialization, including their design, testing, manufacture, safety, efficacy, recordkeeping, labeling, storage, approval, advertising, promotion, sale and distribution, are subject to comprehensive regulation by the FDA and other regulatory authorities in the United States and by comparable non-U.S. regulatory authorities. Failure to obtain marketing approval for a product candidate will prevent us from commercializing the product candidate in a given jurisdiction. We have not received approval to market any product candidates from regulatory authorities in any jurisdiction and it is possible that none of the product candidates we may seek to develop in the future will ever obtain regulatory approval. While we expect to expand our internal regulatory function to support the marketing approval process for Sozinibercept (OPT-302), we have no prior experience in filing and supporting the applications necessary to gain marketing approvals and expect to rely in part on third-party CROs or regulatory consultants to assist us in this process. Securing regulatory approval requires the submission of extensive preclinical and clinical data and supporting information to the various regulatory authorities for each therapeutic indication to establish the biologic product candidate’s safety, purity, efficacy and potency. Securing regulatory approval also requires the submission of information about the product manufacturing process to, and inspection of manufacturing facilities by, the relevant regulatory authority. Any product candidates we develop may not be effective, may be only moderately effective or may prove to have undesirable or unintended side effects, toxicities or other characteristics that may preclude our obtaining marketing approval or prevent or limit commercial use.
The process of obtaining marketing approvals, both in the United States and in other jurisdictions, is expensive, may take many years if additional clinical trials are required, if approval is obtained at all, and can vary substantially based upon a variety of factors, including the type, complexity and novelty of the product candidates involved. Changes in marketing approval policies during the development period, changes in or the enactment of additional statutes or regulations, or changes in regulatory review for each submitted product application, may cause delays in the approval or rejection of an application. The FDA and comparable authorities in other countries have substantial discretion in the approval process and may refuse to accept any application or may decide that our data are insufficient for approval and require additional preclinical, clinical or other studies. In addition, varying interpretations of the data obtained from preclinical and clinical testing could delay, limit, or prevent marketing approval of a product candidate. Any marketing approval we ultimately obtain may be limited or subject to restrictions or post-approval commitments that render the approved product not commercially viable.
If we experience delays in obtaining approval or if we fail to obtain approval of any product candidates we may develop, the commercial prospects for those product candidates, including for Sozinibercept (OPT-302) in other indications, may be harmed, and our ability to generate revenues will be materially impaired.
Lack of efficacy, adverse events or undesirable side effects may emerge in clinical trials conducted by third parties with product candidates for wet AMD or DME, which could negatively affect our stock price, our ability to attract additional capital and our development program.
Lack of efficacy, adverse events or undesirable side effects may emerge in clinical trials conducted by third parties developing product candidates for wet AMD or DME. In addition, other companies have developed products for wet AMD and DME, including product candidates administered in combination with anti-VEGF-A therapies, and have suffered setbacks and clinical trial failures in the past, including failures of primary endpoints in Phase 3 pivotal clinical trials following positive data from Phase 1 and 2 trials. Lack of efficacy, adverse events or undesirable side effects experienced by subjects in third party clinical trials currently being conducted or previously conducted could negatively affect our stock price, our ability to attract additional capital and our development of Sozinibercept (OPT-302) or even the viability of Sozinibercept (OPT-302) as a product candidate. In addition, any such adverse events or undesirable side effects may lead to increased regulatory requirements for, or additional regulatory review of, Sozinibercept (OPT-302), which may result in delays in development and commercialization of Sozinibercept (OPT-302) and harm our business, financial condition and results of operations.
The results of completed clinical trials may not be predictive of future results. Data from our clinical trials to date may not be indicative of results obtained when these trials are completed or in later-stage trials.
There is a high failure rate for drugs and biologic products proceeding through clinical trials. Failure can occur at any time during the clinical trial process. The results of completed clinical trials of Sozinibercept (OPT-302) or any future product candidate may not be predictive of the results of later-stage clinical trials, including our Phase 3 trials of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet
24
AMD, and the results of trials in certain patients may not be predictive of those obtained in another. In fact, many companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in late-stage clinical trials even after achieving promising results in earlier stage clinical trials. In addition, data obtained from clinical activities is subject to varying interpretations, which may delay, limit or prevent regulatory approval.
The results of our Phase 2b clinical trial of Sozinibercept (OPT-302) combination therapy may not be predictive of the results of our Phase 3 clinical program due, in part, to the fact that we have no clinical data on Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy in any clinical trial longer than 72 weeks and that we are conducting our Phase 3 clinical trials at many clinical centers that were not included in our Phase 2b clinical trial. The number of patients exposed to product candidates and the average exposure time in prior clinical trials may be inadequate to detect rare adverse events or findings that may only be detected once a product candidate is administered to more patients and for greater periods of time. Any approved label for Sozinibercept (OPT-302) combination therapy may also be limited if our Phase 3 clinical trial results do not show long-term clinically significant efficacy results, including for over 12 months or in combination with either of the approved anti-VEGF-A therapies. In addition, if a combination of Sozinibercept (OPT-302) with an anti-VEGF-A therapy in our Phase 3 clinical program for the treatment of wet AMD does not achieve clinically significant superiority over anti-VEGF-A monotherapy with statistical significance on the primary endpoints of our Phase 3 clinical trials, or the FDA or a comparable non-U.S. regulatory authority requires additional clinical trials beyond our Phase 3 clinical program to support an approved label of Sozinibercept (OPT-302) used in combination with multiple anti-VEGF-A therapies, our ability to successfully commercialize Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD would be harmed.
For example, masked data from patients that have completed the week 52 visit in the ongoing Phase 3 clinical trials show greater mean BCVA increases from baseline than results with standard of care anti-VEGF-A monotherapy from our Phase 2b study. Masked data represent pooled data from both OPT-302 combination and standard of care monotherapy treatment arms. However, the Phase 3 clinical trial masked data are incomplete and subject to additional analysis once unmasked, and our Phase 3 clinical trials are not fully enrolled and the majority of patients enrolled in the trial have not completed the week 52 visit. There is no assurance that standard of care monotherapy in our Phase 3 clinical trials will yield similar results to our prior clinical trials or previously published clinical trials with anti-VEGF-A monotherapies. As a result, there can be no assurance that top-line results for Sozinibercept (OPT-302) from the Phase 3 clinical trial, if completed, will be consistent with results from masked data available to date.
Interim, top-line and preliminary data from our clinical trials that we announce or publish from time to time may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, we may publicly disclose preliminary or top-line data from our clinical trials, which are based on a preliminary analysis of then-available data, and the results and related findings and conclusions are subject to change following a more comprehensive review of the data related to the particular study or trial. We also make assumptions, estimations, calculations and conclusions as part of our analyses of data, and we may not have received or had the opportunity to fully and carefully evaluate all data. As a result, the top-line results that we report may differ from future results of the same studies or different conclusions or considerations may qualify such results, once additional data have been received and fully evaluated. Top-line data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, top-line data should be viewed with caution until the final data are available. From time to time, we may also disclose interim data from our clinical trials. In addition, we may report interim analyses of only certain endpoints rather than all endpoints. Interim data from clinical trials that we may complete are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data become available.
In addition, adverse changes between interim data and final data could significantly harm our business and prospects. Additional disclosure of interim data by us or by our competitors in the future could also result in volatility in the price of the ADSs and our ordinary shares. Further, others, including regulatory agencies, may not accept or agree with our assumptions, estimates, calculations, conclusions or analyses or may interpret or weigh the importance of data differently, which could impact the value of the particular program, the approvability or commercialization of the particular product candidate or product and our company in general. In addition, the information we choose to publicly disclose regarding a particular study or clinical trial is based on what is typically extensive information, and you or others may not agree with what we determine is the material or otherwise
25
appropriate information to include in our disclosure, and any information we determine not to disclose may ultimately be deemed significant with respect to future decisions, conclusions, views, activities or otherwise regarding a particular product candidate or our business. If the top-line data that we report differ from actual results, or if others, including regulatory authorities, disagree with the conclusions reached, our ability to obtain approval for, and commercialize, Sozinibercept (OPT-302) or any future product candidates may be harmed, which could harm our business, financial condition, results of operations and prospects.
We may face substantial competition, which may result in others discovering, developing or commercializing competing products before or more successfully than us.
The biopharmaceutical industry is intensely competitive and subject to rapid innovation and significant technological advancements. We believe the key competitive factors that will affect the development and commercial success of Sozinibercept (OPT-302) and any future product candidates are efficacy, safety and tolerability profile, reliability, convenience of dosing, price, the level of generic competition and reimbursement. Our competitors include multinational pharmaceutical companies, specialized biotechnology companies, universities and other research institutions. A number of biotechnology and pharmaceutical companies are pursuing the development or marketing of pharmaceuticals that target the same diseases that we are targeting. Smaller or earlier-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies.
There are a number of large pharmaceutical and biotechnology companies that are currently pursuing the development of products for the treatment of wet AMD and DME or have commercially approved products for the treatment of wet AMD or DME, including Roche, Regeneron and Novartis. The current standard of care for wet AMD is monotherapy administration of anti-VEGF-A therapies, including ranibizumab and aflibercept, as well as off-label use of bevacizumab. These drugs are well established therapies and are widely accepted by physicians, patients and third-party payors, which may make it difficult to convince these parties to switch to Sozinibercept (OPT-302) combination therapy. In addition to competition from other companies directly targeting wet AMD or DME, any products we may develop may also face competition from other types of therapies or patient and physician preferences. In addition, many companies are developing new therapeutics, and we cannot predict what the standard of care will be as Sozinibercept (OPT-302) or any future product candidates progress through clinical development.
If our competitors market products are more effective, safer or cheaper than our products, are more durable or have reduced injection burden compared to our products (including Sozinibercept (OPT-302)), or reach the market sooner than our products, we may not achieve commercial success. In addition, the biopharmaceutical industry is characterized by rapid technological change. If we fail to stay at the forefront of technological change, we may be unable to compete effectively. Technological advances or products developed by our competitors may render our technologies, products or product candidates obsolete, less competitive or not economical.
Many of our competitors have substantially greater financial, technical, human and other resources than we do and may be better equipped to develop, manufacture and market technologically superior products. In addition, many of these competitors have significantly longer operating histories and greater experience than we have in undertaking nonclinical studies and human clinical trials of new pharmaceutical products and in obtaining regulatory approvals of human therapeutic products. Clinical trials for the treatment of wet AMD and DME may be relatively costly and time-consuming. The requirements for approval by the FDA and comparable non-U.S. regulatory authorities may change over time and this may require changes to ongoing or future clinical trial designs that could impact timelines and cost. Further, many of our competitors have established distribution channels for the commercialization of their products, whereas we have no such channel or capabilities. In addition, many competitors have greater name recognition and more extensive collaborative relationships.
As a result, our competitors may obtain regulatory approval of their products more rapidly than we do or may obtain patent protection or other intellectual property rights that limit our ability to develop or commercialize our product candidate or any future product candidates. Our competitors may also develop and succeed in obtaining approval for drugs that are more effective, more convenient, more widely used and less costly or have a better safety profile than our products and these competitors may also be more successful than we are in manufacturing and marketing their products. If we are unable to compete effectively against these companies, then we may not be able to commercialize our product candidate or any future product candidates or achieve a competitive position in the market. This would negatively affect our ability to generate revenue. Our competitors also compete with us in recruiting and retaining qualified scientific, management and commercial personnel, establishing clinical trial sites and enrolling patients for clinical trials, as well as in acquiring technologies complementary to, or necessary
26
for, our programs. Our inability to compete effectively in any of these aspects of our business could harm our business, financial condition, results of operations and prospects.
A fast-track designation by the FDA may not lead to a faster development or regulatory review or approval process and does not increase the likelihood that our product candidates will receive marketing approval.
If a drug or biologic is intended for the treatment of a serious or life-threatening condition and the product demonstrates the potential to address unmet medical needs for this condition, the sponsor may apply for FDA fast-track designation for a particular indication. Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy was granted fast-track designation for the treatment of wet AMD in July 2021 and we may seek fast track designation for certain of our future product candidates, but there is no assurance that the FDA will grant this status to any of our future product candidates. If granted, fast-track designation makes a product eligible for more frequent interactions with FDA to discuss the development plan and clinical trial design, as well as rolling review of the application, which means that the company can submit completed sections of its marketing application for review prior to completion of the entire submission. Marketing applications of products candidates with fast-track designation may qualify for priority review under the policies and procedures offered by the FDA, but the fast-track designation does not assure any such qualification or ultimate marketing approval by the FDA. The FDA has broad discretion whether or not to grant fast track designation, so even if we believe a particular product candidate is eligible for this designation, there can be no assurance that the FDA would decide to grant it. Even if we do receive fast- track designation, we may not experience a faster development process, review or approval compared to conventional FDA procedures, and receiving a fast-track designation does not provide any assurance of ultimate FDA approval. In addition, the FDA may withdraw fast-track designation if it believes that the designation is no longer supported by data from our clinical development program. In addition, the FDA may withdraw any fast-track designation at any time.
Sozinibercept (OPT-302) and any future product candidates may not achieve adequate market acceptance among physicians, patients, healthcare payors and others in the medical community necessary for commercial success.
Even if our product candidates receive regulatory approval, they may not gain adequate market acceptance among physicians, patients, healthcare payors and others in the medical community. Given the number of drugs in development or currently approved for the treatment of wet AMD and DME, if we are unsuccessful in achieving a differentiated profile with Sozinibercept (OPT-302), including in combination with existing therapies, based on efficacy, safety and tolerability, dosing and administration, market acceptance will be limited. For example, current treatments for wet AMD, including ranibizumab, aflibercept and low-cost, off-label use of bevacizumab, are well established in the medical community and perceived as demonstrating meaningful clinical response in many cases. As a result, doctors may continue to rely on these treatments without Sozinibercept (OPT-302) or may continue to use such existing treatments as first-line therapies. The medical community may also resist adopting a combination therapy over monotherapy for any of our targeted indications. In particular, recent clinical development has focused on maintaining vision gains with a VEGF-A inhibitor while reducing the number of injections. While we plan to evaluate the potential for co-formulation of Sozinibercept (OPT-302) with approved and/or biosimilar forms of VEGF-A inhibitors to provide flexibility of treatment options for physicians and to reduce the frequency and number of injections for patients, there can be no assurance that we will be successful or that any co-formulated product will have a favorable safety profile. If we are unable to reduce the injection burden of Sozinibercept (OPT-302) combination therapy or demonstrate sufficient efficacy improvements with a comparatively higher frequency and number of injections over standard of care anti-VEGF-A therapies, develop a co-formulation of Sozinibercept (OPT-302) for patients or otherwise increase the duration of efficacy of Sozinibercept (OPT-302) doses, or if physicians determine that a more frequent regimen is necessary, the market acceptance of Sozinibercept (OPT-302) may be limited which would harm our business, financial condition and results of operations.
In addition, the potential market opportunity for Sozinibercept (OPT-302) is difficult to estimate precisely. If Sozinibercept (OPT-302) receives marketing approval for the treatment of wet AMD, it will be approved solely for use in combination with one or more anti-VEGF-A therapies, and may be limited to use with only one anti-VEGF-A therapy for the treatment of wet AMD depending on whether the results from each of our Phase 3 clinical trials support an approved label for use of Sozinibercept (OPT-302) in combination with more than one anti-VEGF-A therapy. The market opportunity for Sozinibercept (OPT-302) will be dependent upon the continued use of anti-VEGF-A therapies in the treatment of wet AMD and the market share of such anti-VEGF-A therapies for
27
which Sozinibercept (OPT-302) is approved as a combination therapy. The degree of market acceptance of any of our approved product candidates will depend on a number of factors, including:
Sales of medical products also depend on the willingness of physicians to prescribe the treatment, which is likely to be based on a determination by these physicians that the products are safe, therapeutically effective and cost-effective. In addition, the inclusion or exclusion of products from treatment guidelines established by various physician groups and the viewpoints of influential physicians can affect the willingness of other physicians to prescribe the treatment. We cannot predict whether physicians, physicians’ organizations, hospitals, other healthcare providers, government agencies or private insurers will determine that our products are safe, therapeutically effective and cost-effective as compared with competing treatments.
Efforts to educate the medical community and third-party payors on the benefits of Sozinibercept (OPT-302) combination therapy may require significant resources and may not be successful. Demonstrating the safety and efficacy of our product candidates and obtaining regulatory approvals will not guarantee future revenue. Our commercial success also depends on coverage and adequate reimbursement of our product candidates by third-party payors, including government payors, which may be difficult or time-consuming to obtain, may be limited in scope and may not be obtained in all jurisdictions in which we may seek to market our products.
If the market opportunities for any product that we or our strategic collaborators develop are smaller than we believe they are, our revenue may be negatively affected and our business may suffer.
We intend to focus our product candidate development on therapies for the treatment of wet AMD and additional retinal disease indications such as DME or retinal vein occlusion, or RVO. Our projections of addressable patient populations that have the potential to benefit from treatment with our product candidate are based on estimates. These estimates have been derived from a variety of sources, including the scientific literature, surveys of clinics, patient foundations or market research, and may prove to be incorrect. Further, new studies may change the estimated incidence or prevalence of these diseases. The number of patients may turn out to be lower than expected. Additionally, the potentially addressable patient population for our product candidates may
28
not ultimately be amenable to treatment with our product candidates. Our market opportunity may also be limited by future competitor treatments that enter the market. If any of our estimates are inaccurate, the market opportunities for any of our product candidates could be significantly diminished and have an adverse impact on our business.
If Sozinibercept (OPT-302) is approved by the FDA as a combination therapy for the treatment of wet AMD, the approval will be limited to this specific indication and, unless we seek regulatory approval for additional indications, we will be prohibited from marketing Sozinibercept (OPT-302) for other indications. We may be subject to fines, penalties or injunctions if we are determined to have promoted or be promoting the use of Sozinibercept (OPT-302) for unapproved or “off-label” uses, resulting in damage to our reputation and business.
If Sozinibercept (OPT-302) receives marketing approval for the treatment of wet AMD, it will be approved solely for use in combination with one or more anti-VEGF-A therapies, and may be limited to only one anti-VEGF-A therapy for the treatment of wet AMD depending on the results of our ongoing pivotal Phase 3 clinical trials. Although we are also developing Sozinibercept (OPT-302) for other retinal diseases, any regulatory approval of Sozinibercept (OPT-302) for wet AMD would not cover the treatment of any other indication. As a result, we would be prohibited from promoting Sozinibercept (OPT-302) for the treatment of DME unless we are granted FDA approval for such indication.
The FDA strictly regulates the promotional claims that may be made about prescription products. While physicians may choose to prescribe products for uses that are not described in the product’s labeling and for uses that differ from those tested in clinical trials and approved by the regulatory authorities, we are prohibited from marketing and promoting the products for indications that are not specifically approved by the FDA or comparable non-U.S. regulatory authorities. These “off-label” uses are common across medical specialties and may constitute an appropriate treatment for some patients in varied circumstances. Regulatory authorities in the United States generally do not restrict or regulate the behavior of physicians in their choice of treatment within the practice of medicine. Regulatory authorities do, however, restrict communications by biotechnology or pharmaceutical companies on off-label use. If the FDA determines that our promotional activities constitute promotion of an off-label use, it could request that we modify our promotional materials and subject us to FDA regulatory or enforcement actions as well as actions by other agencies, including issuance of warning letters or untitled letters, suspension or withdrawal of an approved product from the market, mandatory or voluntary recalls, civil fines, disgorgement of money, operating restrictions, additional reporting requirements and/or oversight if we become subject to a corporate integrity agreement or similar agreement, injunctions or criminal prosecution, any of which could significantly harm our business.
Sozinibercept (OPT-302) is being developed to be used as a combination therapy for use with anti-VEGF-A therapies, which exposes us to additional risks.
We are developing Sozinibercept (OPT-302) to be used in combination with currently approved VEGF-A inhibitors. Even if Sozinibercept (OPT-302) were to receive marketing approval or be commercialized, we would continue to be subject to the risks that the FDA or similar regulatory authorities could revoke approval of some or all approved anti-VEGF-A therapies for safety, efficacy, manufacturing or supply issues. This could result in Sozinibercept (OPT-302) being restricted from commercialization or being less commercially successful.
We may also evaluate Sozinibercept (OPT-302) or any other future product candidates in combination with one or more other product candidates that have not yet been approved for marketing by the FDA or similar foreign regulatory authorities. We will not be able to market and sell Sozinibercept (OPT-302) or any product candidate we develop in combination with any such unapproved therapies that do not ultimately obtain marketing approval.
If the FDA or similar foreign regulatory authorities do not approve these other product candidates or revoke their approval of, or if safety, efficacy, manufacturing, or supply issues arise with, the drugs we choose to evaluate in combination with Sozinibercept (OPT-302) or any product candidate we develop, we may be unable to obtain approval of or market Sozinibercept (OPT-302) or any product candidate we develop.
If we fail to develop and commercialize additional product candidates, we may be unable to grow our business.
Although the development and commercialization of Sozinibercept (OPT-302) is currently our primary focus, as part of our longer-term growth strategy, we plan to evaluate the development and commercialization of other therapies related to retinal diseases. The success of this strategy depends primarily upon our ability to identify and
29
validate new therapeutic candidates, and to identify, develop and commercialize new drugs and biologics. Our research efforts may initially show promise in discovering potential new drugs and biologics, yet fail to yield product candidates for clinical development for a number of reasons, including:
If any of these events occur, we may be forced to abandon our development efforts for one or more programs, which could harm our business, operating results and prospects and could potentially cause us to cease operations. Future research programs to identify new product candidates may require substantial technical, financial and human resources. We may focus our efforts and resources on potential programs or product candidates that ultimately prove to be unsuccessful.
Product candidates may require additional, time-consuming development efforts prior to commercial sale, including preclinical studies, clinical trials and approval by the FDA and/or comparable non-U.S. regulatory authorities. All product candidates are prone to the risks of failure that are inherent in pharmaceutical product development, including the possibility that the product candidate will not be shown to be sufficiently safe and effective for approval by regulatory authorities. In addition, we cannot assure you that any such products that are approved will be manufactured or produced economically, be successfully commercialized, be widely accepted in the marketplace or be more effective than other commercially available alternatives.
Our business was and may in the future be negatively affected by the effects of health epidemics, in regions where we or third parties on which we rely have significant manufacturing facilities, concentrations of clinical trial sites or other business operations.
Health epidemics in regions where we have concentrations of clinical trial sites or other business operations could negatively affect our business, including by causing significant disruption in the operations of third-party manufacturers and CROs upon whom we rely. For example, the novel coronavirus disease 2019, or COVID-19, negatively impacted our ability to initiate clinical trial sites, maintain patient enrollment and enroll new patients. Our ability to attract additional clinical trial sites and principal investigators to conduct our clinical trials and to conduct the necessary clinical trial site initiation procedures were negatively impacted by quarantines, shelter-in-place and similar restrictions imposed by federal, state and local governments.
Moreover, we rely on third-party CROs and other third parties to assist us with clinical development activities. We experienced significantly increased costs and delays in connection with the activities conducted by third-party CROs and other third parties to prepare for and progress our Phase 3 clinical trials, due in part to the COVID-19 pandemic and its related effects. The outbreak of health epidemics in the future could cause significant disruptions to our clinical development timelines, which would harm our business, financial condition, results of operations and growth prospects.
Risks Related to Legal and Regulatory Compliance Matters
Disruptions at the FDA and other government agencies caused by funding shortages or global health concerns could hinder their ability to hire and retain key leadership and other personnel, otherwise prevent new products from being developed, approved or commercialized in a timely manner or at all, or otherwise prevent those
30
agencies from performing normal business functions on which the operation of our business may rely, which could negatively impact our business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept the payment of user fees, statutory, regulatory and policy changes, and other events that may otherwise affect the FDA’s ability to perform routine functions. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies on which our operations may rely, including those that fund research and development activities, is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, in recent years, including in 2018 and 2019, the U.S. government shut down several times and certain regulatory agencies, such as the FDA, had to furlough critical employees and stop critical activities. Separately, in response to the COVID-19 pandemic, in March 2020, the FDA temporarily postponed inspections of manufacturing facilities. Regulatory authorities outside the United States may adopt similar restrictions or other policy measures or issue guidance materially affecting the conduct of clinical trials. If a prolonged government shutdown occurs, or if global health concerns prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews or other regulatory activities, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business. Further, in our operations as a public company, future government shutdowns or delays could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
Even if we commercialize Sozinibercept (OPT-302) or any future product candidate, we may face challenges to achieving profitability such as our products becoming subject to unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives, which could harm our business.
In the United States and in other countries, patients who are prescribed treatment for their conditions generally rely on third-party payors to reimburse all or part of the costs associated with their treatment. The availability of coverage and adequacy of reimbursement for our products by third-party payors, including government health care programs (e.g., Medicare, Medicaid, TRICARE), managed care providers, private health insurers, health maintenance organizations and other organizations are essential for most patients to be able to afford medical services and pharmaceutical products such as Sozinibercept (OPT-302) or any our product candidates. Third-party payors decide which medications they will pay for and establish reimbursement levels. The availability of coverage and extent of reimbursement by governmental and other third-party payors are essential for most patients to be able to afford treatments such as Sozinibercept (OPT-302).
In the United States, the principal decisions about reimbursement for new medicines are typically made by the Centers for Medicare & Medicaid Services, or CMS, an agency within the U.S. Department of Health and Human Services, or HHS. CMS decides whether and to what extent our products will be covered and reimbursed under Medicare and private payors tend to follow CMS to a substantial degree. Other countries have equivalent authorities who play a similar role. Factors payors consider in determining reimbursement are based on whether the product is:
Our ability to commercialize any products successfully will depend in part on the extent to which coverage and adequate reimbursement for these products and related treatments will be available from third-party payors, including government health care programs and private health insurers. Moreover, a payor’s decision to provide
31
coverage for a drug product does not imply that an adequate reimbursement rate will be approved. If coverage and adequate reimbursement are not available, or are available only to limited levels, we may not be able to successfully commercialize our product candidates. Even if coverage is provided, the approved reimbursement amount may not be high enough to allow us to establish or maintain pricing sufficient to realize a sufficient return on our investment.
In the United States, no uniform policy for coverage and reimbursement for products exists among third-party payors. Therefore, coverage and reimbursement for our products can differ significantly from payor to payor. The process for determining whether a payor will provide coverage for a product may be separate from the process for setting the reimbursement rate that the payor will pay for the product. One payor’s determination to provide coverage for a product does not assure that other payors will also provide coverage and reimbursement for the product. Third-party payors may also limit coverage to specific products on an approved list, or formulary, which might not include all of the FDA-approved products for a particular indication.
Government authorities and other third-party payors in the United States and abroad have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications and for newly approved products, and as a result, they may not cover or provide adequate reimbursement for Sozinibercept (OPT-302) and future product candidates. Increasingly, certain third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products. We cannot be sure that coverage and reimbursement will be available for any product that we or our future collaborators commercialize and, if reimbursement is available, what the level of reimbursement will be. Coverage and reimbursement may impact the demand for, or the price of, any product candidate for which we or our future collaborators obtain marketing approval. If coverage and reimbursement are not available or reimbursement is available only to limited levels, we and our future collaborators may not be able to successfully commercialize any product candidate for which marketing approval is obtained. A decision by a third-party payor not to cover or not to separately reimburse for our medical products or therapies using our products could reduce physician utilization of our products once approved. Assuming there is coverage for our product candidates, or therapies using our product candidates by a third-party payor, the resulting reimbursement payment rates may not be adequate or may require co-payments that patients find unacceptably high. We cannot be sure that coverage and reimbursement in the United States will be available for Sozinibercept (OPT-302) and any future product candidates and any reimbursement that may become available may not be adequate or may be decreased or eliminated in the future.
There may be significant delays in obtaining coverage and reimbursement for newly approved drugs, and coverage may be more limited than the purposes for which the drug is approved by the FDA or comparable non-U.S. regulatory authorities. Moreover, eligibility for coverage and reimbursement does not imply that a drug will be paid for in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim reimbursement levels for new drugs, if applicable, may also not be sufficient to cover our costs and may only be temporary. Reimbursement rates may vary according to the use of the drug and the clinical setting in which it is used, may be based on reimbursement levels already set for lower cost drugs and may be incorporated into existing payments for other services. Net prices for drugs may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and by any future relaxation of laws that presently restrict imports of drugs from countries where they may be sold at lower prices than in the United States. Our inability to promptly obtain coverage and profitable reimbursement rates from both government-funded and private payors for any approved products that we develop could harm our operating results, our ability to raise capital needed to commercialize products and our overall financial condition.
The regulations that govern marketing approvals, pricing and reimbursement for new drug products vary widely from country to country. Current and future legislation may significantly change the approval requirements in ways that could involve additional costs and cause delays in obtaining approvals. Some countries require approval of the sale price of a drug before it can be marketed. In many countries, the pricing review period begins after marketing or product-licensing approval is granted. In some non-U.S. markets, prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. As a result, we might obtain marketing approval for a product in a particular country, but then be subject to price regulations that delay or limit our commercial launch of the product, possibly for lengthy time periods, which could negatively impact the revenue we generate from the sale of the product in that particular country. Adverse pricing limitations may hinder our ability to recoup our investment in one or more product candidates, even if our product candidates obtain marketing approval.
32
To become and remain profitable, we or any potential future collaborator must develop and eventually commercialize Sozinibercept (OPT-302) or any future product candidates with significant market potential at an adequate profit margin after cost of goods sold and other expenses. Commercialization of Sozinibercept (OPT-302) or any future product candidates may entail a substantial cost of goods sold and there can be no assurance that we will be able to achieve a suitable gross margin with respect to sales of Sozinibercept (OPT-302) or any future product candidates.
Changes in U.S. healthcare law and implementing regulations, as well as changes in healthcare policy, and equivalent changes in the laws and policies in other countries may impact our business in ways that we cannot currently predict and may harm our business and results of operations.
There have been, and likely will continue to be, several executive, legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of product candidates, restrict or regulate post-approval activities and affect our ability to profitably sell any product candidates for which we obtain marketing approval. Among policy-makers and payors in the United States there is significant interest in promoting changes in healthcare systems with the stated goals of containing healthcare costs, improving quality and/or expanding access, and the pharmaceutical industry has been a particular focus of these efforts and has been significantly affected by major legislative initiatives.
The Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010, collectively referred to as the Affordable Care Act, substantially changed the way healthcare is financed by both the government and private insurers, and significantly impacts the U.S. pharmaceutical industry. The Affordable Care Act, among other things contains a number of provisions, including those governing enrollment in federal healthcare programs, reimbursement adjustments and changes to fraud and abuse laws.
Since its enactment, there have been executive, judicial and Congressional challenges to certain aspects of the Affordable Care Act. For example, the Tax Cuts and Jobs Act of 2017, or the TCJA, was enacted, which included a provision that repealed, effective January 1, 2019, the tax-based shared responsibility payment imposed by the Affordable Care Act on certain individuals who fail to maintain qualifying health coverage for all or part of a year that is commonly referred to as the “individual mandate.” On December 14, 2018, a U.S. District Court Judge in the Northern District of Texas ruled that the individual mandate is a critical and inseparable feature of the Affordable Care Act, and therefore, because it was repealed as part of the TCJA, the remaining provisions of the Affordable Care Act are invalid as well. June 17, 2021, the U.S. Supreme Court dismissed a challenge on procedural grounds that argued the Affordable Care Act is unconstitutional in its entirety because the “individual mandate” was repealed by Congress. Further on August 16, 2022, President Biden signed the Inflation Reduction Act of 2022 or the IRA, into law, which among other things, extends enhanced subsidies for individuals purchasing health insurance coverage in the Affordable Care Act marketplaces through plan year 2025. The IRA also eliminates the “donut hole” under the Medicare Part D program beginning in 2025 by significantly lowering the beneficiary maximum out-of-pocket cost through a newly established manufacturer discount program. It is possible that the Affordable Care Act will be subject to judicial or Congressional challenges in the future. It is unclear how any such challenges and the healthcare reform measures of the Biden administration will impact the Affordable Care Act.
Other legislative changes have been proposed and adopted since the Affordable Care Act was enacted. These changes include aggregate reductions to Medicare payments to providers of 2% per fiscal year pursuant to the Budget Control Act of 2011 and subsequent laws, which began in 2013 and will remain in effect until 2032, unless additional Congressional action is taken. In January 2013, the American Taxpayer Relief Act of 2012 was signed into law, which, among other things, further reduced Medicare payments to several types of providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three (3) to five (5) years. New laws may result in additional reductions in Medicare and other healthcare funding, which may materially negatively affect customer demand and affordability for our products and, accordingly, the results of our financial operations.
Also, there has been heightened governmental scrutiny recently over the manner in which pharmaceutical companies set prices for their marketed products, which have resulted in several U.S. presidential executive orders, Congressional inquiries and proposed and enacted federal legislation, as well as state efforts, designed to, among other things, bring more transparency to product-pricing, reduce the cost of prescription drugs under Medicare, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drug products. For example, in July 2021, the Biden administration expressed its intent to pursue certain policy initiatives to reduce drug prices. For example, the executive order expressed the
33
Biden administration’s support of legislative reforms to lower prescription drug prices, including by allowing Medicare’s negotiation of drug prices. In response to Biden’s executive order, on September 9, 2021, HHS released a Comprehensive Plan for Addressing High Drug Prices that outlines principles for drug-pricing reform and sets out a variety of potential legislative policies that Congress could pursue as well as potential administrative actions HHS can take to advance these principles. In addition, the IRA directs the HHS Secretary to establish a Drug Price Negotiation Program (the “Program”) to lower prices for certain high-expenditure, single-source prescription drugs and biologics covered under Medicare Part B and Part D that have been approved by the FDA for at least seven (7) years for prescription drugs and at least eleven (11) years for biologics. Under the Program, the HHS Secretary will publish a list of “selected drugs,” and will then negotiate maximum fair prices (“MFP”) with their manufacturers. The Program will be implemented in stages. Beginning in 2026, ten (10) Medicare Part D “selected drugs” will be subject to price negotiations. By 2029, and in subsequent years thereafter, the number will increase to twenty (20) drugs and biologics covered under Medicare Part B and Part D. Agreements between HHS and manufacturers will remain in place until a drug or biologic is no longer considered a “selected drug” for negotiation purposes. Manufacturers who do not comply with the negotiated prices set under the Program will be subject to an excise tax based on a percentage of total sales of a “selected drug” up to 95% and potential civil monetary penalties. Further, beginning in October 2023, the IRA will require manufacturers that increase prices of certain Medicare Part B and Part D drugs or biologics at a rate greater than inflation to pay rebates to CMS or be subject to civil monetary penalties. HHS has and will continue to issue and update guidance as these programs are implemented, although the IRA may be subject to legal challenges. It is currently unclear how the IRA will be implemented but is likely to have a significant impact on the pharmaceutical industry. In addition, in response to the Biden administration's October 2022 executive order on February 14, 2023, HHAS released a report outlining three new models for testing by the Center for Medicare and Medicaid Innovation which will be evaluated on their ability to lower the cost of drugs, promote accessibility, and improve quality of care. It is unclear whether the models will be utilized in any health reform measures in the future.
At the state level, individual states in the United States are increasingly active in passing legislation and implementing regulations designed to control pharmaceutical and biological product-pricing, including price or patient-reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk-purchasing. Legally mandated price controls on payment amounts by third-party payors or other restrictions on coverage or access could harm our business, results of operations, financial condition and prospects. In addition, regional healthcare authorities and individual hospitals are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription drug and other healthcare programs. This could reduce the ultimate demand for our product candidates that we successfully commercialize or put pressure on our product-pricing.
We expect that these and other healthcare reform measures that may be adopted in the future may result in more rigorous coverage criteria and lower reimbursement and put additional downward pressure on the price that we receive for any approved product. Any reduction in reimbursement from Medicare or other government-funded programs may result in a similar reduction in payments from private payors. The implementation of cost-containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our drugs once marketing approval is obtained. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action in the United States. If we or any third parties we may engage are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we or such third parties are not able to maintain regulatory compliance, our product candidates may lose any regulatory approval that may have been obtained and we may not achieve or sustain profitability.
As a company primarily based outside of the United States, our business is subject to economic, political, regulatory and other risks associated with international operations.
As a company with substantial operations in Australia and an international clinical trial program, our business is subject to risks associated with conducting business outside the United States. Many of our suppliers and clinical-trial relationships are located outside the United States. Accordingly, our future results could be harmed by a variety of factors, including:
34
If we fail to comply with non-U.S. regulatory requirements governing human clinical trials and marketing approval for drugs, we could be prevented from selling our product candidates in non-U.S. markets, which may negatively affect our operating results and financial condition.
The requirements governing the conduct of clinical trials, product-licensing, pricing and reimbursement for marketing our product candidates outside the United States vary greatly from country to country and may require additional testing. We expect that our future clinical development of our product candidates will involve a number of clinical trials in non-U.S. jurisdictions. We have no direct experience as a company in obtaining non-U.S. regulatory approvals. The time required to obtain approvals outside the United States may differ from that required to obtain FDA approval. We may not obtain non-U.S. regulatory approvals on a timely basis, if at all. Approval by the FDA does not guarantee approval by comparable non-U.S. regulatory authorities, and approval by one non-U.S. regulatory authority does not ensure approval by regulatory authorities in other countries or by the FDA. Failure to comply with these regulatory requirements or obtain required approvals could impair our ability to develop non-U.S. markets for our product candidates and may harm our results of operations and financial condition.
35
Price controls may be imposed in non-U.S. markets, which may negatively affect our future profitability.
In some countries, particularly EU member states, Japan, Australia and Canada, the pricing of prescription drugs is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after receipt of marketing approval for a product. In addition, there can be considerable pressure by governments and other stakeholders on prices and reimbursement levels, including as part of cost-containment measures. Political, economic, and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference-pricing used by various EU member states and parallel distribution, or arbitrage between low-priced and high-priced member states, can further reduce prices. In some countries, we or our collaborators may be required to conduct a clinical trial or other studies that compare the cost-effectiveness of our product candidates to other available therapies in order to obtain or maintain reimbursement or pricing approval. Publication of discounts by third-party payors or authorities may lead to further pressure on the prices or reimbursement levels within the country of publication and other countries. If reimbursement of our products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business, revenues, or profitability could be harmed.
We have received tax incentives under the Research and Development Tax Incentive scheme in Australia that may become repayable if we did not or do not comply with the rules of the scheme, or we may become ineligible for tax incentives in our current or future tax years, which could harm our business, financial condition and results of operations.
We have received cash incentives in the past under the Research and Development Tax Incentive scheme, or the R&D Scheme, to offset the costs of our clinical trials and other qualifying expenses incurred both in Australia and other jurisdictions. Certain research and development costs that we incur in the future may be ineligible for cash incentives under the R&D Scheme. For example, costs incurred outside Australia in connection with our future clinical trials are generally not eligible for cash incentives under the R&D Scheme. In addition, the federal government of Australia and the Australian Taxation Office, or ATO, could change the rules of the regulatory regime or amend past tax returns and, as a result, amounts paid to us may become repayable to the ATO including the amount of tax incentives in respect to our fiscal year ended June 30, 2023, included as current receivables in our consolidated financial statements. We have received an aggregate of US$35.6 million (A$50.3 million) in cash tax incentives during the five fiscal years ended June 30, 2023, under the R&D Scheme. As of June 30, 2023, our current tax receivable under the R&D Scheme was US$5.9 million. This receivable amount as of June 30, 2023, is based on Australian legislation as enacted as of June 30, 2023. Any proposed changes to the legislation, such as rate changes to eligibility requirements, may have a retrospective impact on our current tax receivable under the R&D Scheme - currently, no such legislative changes have occurred. Any rule changes made to reduce the amount we are able to claim under the R&D Scheme currently or in the future and any retrospective changes made to the R&D Scheme that reduce the incentives that we have claimed in past tax years could harm our business, financial condition and results of operations.
The withdrawal of the United Kingdom, or the U.K., from the European Union, or the EU, commonly referred to as “Brexit,” may adversely impact our ability to obtain regulatory approvals of our product candidates in the U.K. or the EU and may require us to incur additional expenses to develop and commercialize our product candidates in the U.K. or the EU or receive clinical supply of our product candidates from manufacturing partners in the U.K.
Following the result of a referendum in 2016, the U.K. left the EU on January 31, 2020, commonly referred to as Brexit. Pursuant to the formal withdrawal arrangements agreed between the U.K. and the EU, the U.K. was subject to a transition period until December 31, 2020 (the "Transition Period"), during which EU rules continued to apply. A trade and cooperation agreement (the "Trade and Cooperation Agreement") that outlines the future trading relationship between the United Kingdom and the European Union was agreed in December 2020.
Since a significant proportion of the regulatory framework in the U.K. applicable to our business and our product candidate is derived from EU directives and regulations, Brexit has had, and may continue to have, a material impact upon the regulatory regime with respect to the development, approval and commercialization of our product candidate in the U.K. or the EU. For example, Great Britain is no longer covered by the centralized procedures for obtaining EU-wide marketing authorization from the EMA, and a separate marketing authorization will be required to market our product candidate in Great Britain. It is currently unclear whether the Medicines & Healthcare products Regulatory Agency in the U.K. is sufficiently prepared to handle the increased volume of marketing authorization applications that it is likely to receive. Any delay in obtaining, or an inability to obtain,
36
any marketing approvals, would delay or prevent us from commercializing our product candidate in the U.K. or the EU and restrict our ability to generate revenue and achieve and sustain profitability.
While the Trade and Cooperation Agreement provides for the tariff-free trade of medicinal products between the U.K. and the E.U. there may be additional non-tariff costs to such trade which did not exist prior to the end of the Transition Period. Further, should the U.K. diverge from the EU from a regulatory perspective in relation to medicinal products, tariffs could be put into place in the future. We could therefore, both now and in the future, face significant additional expenses (when compared to the position prior to the end of the Transition Period) to operate our business, which could significantly and materially harm or delay our ability to generate revenues or achieve profitability of our business. Any further changes in international trade, tariff and import/export regulations as a result of Brexit or otherwise may impose unexpected duty costs or other non-tariff barriers on us. These developments, or the perception that any of them could occur, may significantly reduce global trade and, in particular, trade between the impacted nations and the U.K. It is also possible that Brexit may negatively affect our ability to attract and retain employees, particularly those from the EU.
Product liability lawsuits against us could cause us to incur substantial liabilities and to limit commercialization of any products that we may develop.
We face an inherent risk of product liability exposure related to the testing of our product candidates in human clinical trials and will face an even greater risk if we commercialize any resulting products. Product liability claims may be brought against us by subjects enrolled in our clinical trials, patients, or others using our products. If we cannot successfully defend ourselves against claims that our product candidates or products that we may develop caused injuries, we could incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
Our clinical trial liability insurance coverage may not adequately cover all liabilities that we may incur. We may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise. Our inability to obtain product liability insurance at an acceptable cost or to otherwise protect against potential product liability claims could prevent or delay the commercialization of any products or product candidates that we develop. We intend to expand our insurance coverage for products to include the sale of commercial products if we obtain marketing approval for our product candidates in development, but we may be unable to obtain commercially reasonable product liability insurance for any products approved for marketing. Large judgments have been awarded in class-action lawsuits based on drugs that had unanticipated side effects. If we are sued for any injury caused by our products, product candidates or processes, our liability could exceed our product liability insurance coverage and our total assets. Claims against us, regardless of their merit or potential outcome, may also generate negative publicity or harm our ability to obtain physician endorsement of our products or expand our business.
37
Our business operations and relationships with investigators, healthcare professionals, consultants, third-party payors, patient organizations and customers are subject to broadly applicable healthcare laws and regulations, which could expose us to civil penalties, criminal sanctions, contractual damages, reputational harm and diminished profits and future earnings.
Healthcare providers and third-party payors will play a primary role in the recommendation and prescription of any product candidate for which we obtain regulatory approval. Our current and future arrangements may expose us to broadly applicable fraud and abuse and other healthcare laws that may constrain the business or financial arrangements and relationships through which we would market, sell and distribute our products. In particular, the research of our product candidates, as well as the promotion, sales and marketing of healthcare items and services, as well as certain business arrangements in the healthcare industry, are subject to extensive laws designed to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, structuring and commission(s), certain customer incentive programs and other business or financial arrangements.
Such laws include, but are not limited to, the following:
38
Pharmaceutical and other healthcare companies have been prosecuted under these laws for a variety of promotional and marketing activities, such as providing free trips, free or discounted goods, improper consulting fees and grants and other monetary benefits to prescribers reporting to pricing services inflated average wholesale prices that were then used by federal programs to set reimbursement rates, engaging in off-label promotion; and submitting inflated best price information to the Medicaid Rebate Program to reduce liability for Medicaid rebates. Ensuring that our internal operations and future business arrangements with third parties comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations, agency guidance or case law involving applicable fraud and abuse or other healthcare laws and regulations. If any actions are instituted against us for violation of these laws or regulations, and we are not successful in defending ourselves, those actions could have a significant impact on our business, including the imposition of significant civil, criminal and administrative sanctions, damages, disgorgement, monetary fines, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, imprisonment, integrity oversight and reporting obligations, contractual damages, reputational harm, diminished profits and future earnings, and curtailment or restructuring of our operations, any of which could harm our ability to operate our business and our results of operations.
We are subject to stringent and evolving U.S. and foreign laws, regulations, rules, contractual obligations, policies and other obligations related to data privacy and security. Our actual or perceived failure to comply with such obligations could lead to regulatory investigations or actions; litigation; fines and penalties; a disruption of our business operations; reputational harm; loss of revenue or profits; and other adverse business consequences.
In the ordinary course of business, we collect, receive, store, process, generate, use, transfer, disclose, make accessible, protect, secure, dispose of, transmit, and share personal data and other sensitive information including proprietary and confidential business data. trade secrets. intellectual property, data we collect about trial participants in connection with clinical trials. and other sensitive third-party data. Our data processing activities may subject us to numerous data privacy and security obligations. such as various laws, regulations guidance, industry standards, external and internal privacy and security policies, contractual requirements, and other obligations relating to data privacy and security.
39
In the United States. federal, state. and local governments have enacted numerous data privacy and security laws, including data breach notification laws. personal data privacy laws, consumer protection laws (e.g. Section 5 of the Federal Trade Commission Act), and other similar laws (e.g., wiretapping laws). For example HIPAA, as amended by HITECH, imposes specific requirements relating to the privacy, security, and transmission of individually identifiable health information. Additionally, the California Consumer Privacy Act of 2018 ("CCPA") applies to personal data of consumers, business representatives, and employees, and requires businesses to provide specific disclosures in privacy notices and honor requests of California residents to exercise certain privacy rights. The CPA provides for civil penalties of up to $7,500 per violation and allows private litigants affected by certain data breaches to recover significant statutory damages. Although the CPA exempts some data processed in the context of clinical trials. the CPA may increase compliance costs and potential liability with respect to other personal data we maintain about California residents. In addition, the California Privacy Rights Act of 2020 ("CPRA") expands the CPA's requirements, including by adding a new right for individuals to correct their personal data and establishing a new regulatory agency to implement and enforce the law. Other states, such as Virginia and Colorado, have also passed comprehensive data privacy and security laws. and similar laws are being considered in several other states. as well as at the federal and local levels. While these states. like the CCPA, also exempt some data processed in the context of clinical trials. these developments further complicate compliance efforts. and increase legal risk and compliance costs for us and the third parties upon whom we rely.
Outside the United States, an increasing number of laws, regulations, and industry standards may govern data privacy and security. For example, the European Union's General Data Protection Regulation ("EU GDPR the United Kingdom's GDPR ("UK GDPR"), Brazil's General Data Protection Law (Lei Geral de Protecão de Dados Pessoais, or "LGPD") (Law No. 13,709/2018), and China's Personal Information Protection Law (PIPL?) impose strict requirements for processing personal data. For example, under the EU GDPR, companies may face temporary or definitive bans on data processing and other corrective actions, fines of up to 20 million Euros or 4% of annual global revenue, whichever is greater; or private litigation related to processing of personal data brought by classes of data subjects or consumer protection organizations authorized at law to represent their interests. In Canada, the Personal Information Protection and Electronic Documents Act (PIPEDA") and various related provincial laws, as well as Canada's Anti-Spam Legislation ("CASL*), may apply to our operations We may be subject to new and emerging data privacy and security regimes, including Australia's Privacy Act China's Personal Information Protection Law. Japan's Act on the Protection of Personal Information, and Singapore's Personal Data Protection Act 43
In the ordinary course of business. we may transfer personal data from Europe and other jurisdictions to the United States or other countries. Europe and other jurisdictions have enacted laws requiring data to be localized or limiting the transfer of personal data to other countries. In particular, the European Economic Area ("EA") and the United Kingdom (*UK" have significantly restricted the transfer of personal data to the United States and other countries whose privacy laws it believes are inadequate. Other jurisdictions may adopt similarly stringent interpretations of their data localization and cross-border data transfer laws. Although there are currently various mechanisms that may be used to transfer personal data from the EA and UK to the United States in compliance with law. such as the EA and UK's standard contractual clauses, these mechanisms are subject to legal challenges, and there is no assurance that we can satisfy or rely on these measures to lawfully transfer personal data to the United States. If there is no lawful manner for us to transfer personal data from the EA, the UK or other jurisdictions to the United States, or if the requirements for a legally-compliant transfer are too onerous, we could face significant adverse consequences, including the interruption or degradation of our operations. the need to relocate part of or all of our business or data processing activities to other jurisdictions at significant expense, increased exposure to regulatory actions, substantial fines and penalties, the inability to transfer data and work with partners. vendors and other third parties, and injunctions against our processing or transferring of personal data necessary to operate our business. Additionally, companies that transfer personal data out of the EA and UK to other jurisdictions, particularly to the United States, are subject to increased scrutiny from regulators, individual litigants, and activist groups. Some European regulators have ordered certain companies to suspend or permanently cease certain transfers out of Europe for allegedly violating the GDPR's cross-border data transfer limitations.
In addition to data privacy and security laws, we are or may become contractually subject to industry standards adopted by industry groups. We are also bound by contractual obligations related to data privacy and security. and our efforts to comply with such obligations may not be successful. For example, certain data privacy laws. such as the GDPR and the CCPA, require covered businesses to impose specific contractual restrictions on their service providers. We publish privacy policies, marketing materials and other statements,
40
such as compliance with certain certifications or self-regulatory principles, regarding data privacy and security. If these policies. materials or statements are found to be deficient, lacking in transparency. deceptive, unfair, or misrepresentative of our practices, we may be subject to investigation, enforcement actions by regulators or other adverse consequences.
Obligations related to data privacy and security are quickly changing, becoming increasingly stringent, and creating regulatory uncertainty. Additionally, these obligations may be subject to differing applications and interpretations, which may be inconsistent or conflict among jurisdictions. Preparing for and complying with these obligations requires us to devote significant resources, which may necessitate changes to our services. information technologies, systems, and practices and to those of any third parties that process personal data on our behalf In addition, these obligations may require us to change our business model.
We may at times fail (or be perceived to have failed) in our efforts to comply with our data privacy and security obligations. Moreover, despite our efforts, our personnel or third parties on whom we rely may fail to comply with such obligations, which could negatively impact our business operations. If we or the third parties on which we rely fail, or are perceived to have failed, to address or comply with applicable data privacy and security obligations, we could face significant consequences, including but not limited to: government enforcement actions (e.g., investigations, fines, penalties, audits, inspections, and similar); litigation (including class-action claims); additional reporting requirements and/or oversight; bans on processing personal data; orders to destroy or not use personal data; and imprisonment of company officials.
Any of these events could have a material adverse effect on our reputation, business, or financial condition including but not limited to: loss of customers; interruptions or stoppages in our business operations (including clinical trials); inability to process personal data or to operate in certain jurisdictions; limited ability to develop or commercialize our products; expenditure of time and resources to defend any claim or inquiry; adverse publicity, or substantial changes to our business model or operations.
If we are not able to obtain required regulatory approvals, we will not be able to commercialize Sozinibercept (OPT-302) or any future product candidate, and our ability to generate product revenue will be impaired.
Sozinibercept (OPT-302) and any future product candidate that we may develop, as well as the activities associated with their development and commercialization, including their design, research, testing, manufacture, safety, efficacy, recordkeeping, labeling, packaging, storage, approval, advertising, promotion, sale and distribution are subject to comprehensive regulation by the FDA and other regulatory agencies in the United States and by similar regulatory authorities outside the United States. Failure to obtain marketing approval for, and thus commercialize any product candidate, could negatively impact our ability to generate any revenue from product sales.
We have not received approval from regulatory authorities to market any product candidate in any jurisdiction, and it is possible that our product candidate will never obtain the appropriate regulatory approvals necessary for us to commence product sales. Neither we nor any collaborator is permitted to market our product candidate in the United States or any other jurisdiction until we receive regulatory approval of a BLA from the FDA or similar application from regulatory authorities outside of the United States.
The time required to obtain approval of a BLA by the FDA or similar application from regulatory authorities outside of the United States is unpredictable but typically takes many years following the commencement of clinical trials and depends upon numerous factors, including the substantial discretion of the regulatory authority. Prior to submitting a BLA to the FDA or any comparable application to any other non-U.S. regulatory authorities for approval of any product candidate, we will need to complete pivotal Phase 3 clinical trials and demonstrate favorable results with respect to safety, tolerability and efficacy. In addition, approval policies, regulations or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate’s clinical development and may vary among jurisdictions.
Securing marketing approvals requires the submission of extensive nonclinical and clinical data and supporting information to regulatory authorities for each therapeutic indication to establish the safety and efficacy of our product candidate for the specified indications. We expect to rely on third-party CROs, consultants and our collaborators to assist us in filing and supporting the applications necessary to gain marketing approvals. Securing marketing approval also requires the submission of information about the product manufacturing process to, and inspection of manufacturing facilities by, regulatory authorities. Errors in the submission of applications for marketing approval or issues, including those related to gathering the appropriate data and the inspection process,
41
may ultimately delay or affect our ability to obtain regulatory approval, commercialize our product candidate and generate product revenue.
Our failure to obtain regulatory approval in international jurisdictions would prevent us from marketing our product candidates outside the United States.
If we succeed in developing any products, we intend to market them in non-U.S. jurisdictions in addition to the United States. In order to market and sell our products in other jurisdictions, we must obtain separate marketing approvals and comply with numerous and varying regulatory requirements. We may not obtain non-U.S. regulatory approvals on a timely basis, if at all. Approval by the FDA does not ensure approval by regulatory authorities in other countries or jurisdictions. Approval by one regulatory authority outside the United States does not ensure approval by regulatory authorities in other countries or jurisdictions or by the FDA. If we fail to obtain approval of any of our product candidates by regulatory authorities in another country, we will be unable to commercialize our product in that country, and the commercial prospects of that product candidate and our business prospects could decline. The approval procedure varies among countries and can involve additional testing. The time required to obtain approval may differ substantially from that required to obtain FDA approval. The regulatory approval process outside the United States generally includes all of the risks associated with obtaining FDA approval. In addition, in many countries outside the United States, we must secure product reimbursement approvals before regulatory authorities will approve the product for sale in that country. Obtaining non-U.S. regulatory approvals and compliance with non-U.S. regulatory requirements could result in significant delays, difficulties and costs for us and could delay or prevent the introduction of our products in certain countries. Further, clinical trials conducted in one country may not be accepted by regulatory authorities in other countries and regulatory approval in one country does not ensure approval in any other country, while a failure or delay in obtaining regulatory approval in one country may have a negative effect on the regulatory approval process in others. Also, regulatory approval for any of our product candidates may be withdrawn if we fail to comply with regulatory requirements, if problems occur after the product candidate reaches the market or for other reasons. If we fail to comply with the regulatory requirements in international markets and fail to receive applicable marketing approvals, our target market will be reduced and our ability to realize the full market potential of our product candidates will be harmed and our business will be negatively affected.
Even if Sozinibercept (OPT-302) combination therapy or any future product candidate receives regulatory approval, it may still face future development and regulatory difficulties.
Even if we obtained regulatory approval for a product candidate, it would be subject to ongoing requirements by the FDA and comparable non-U.S. regulatory authorities governing the manufacture, quality control, further development, labeling, packaging, storage, distribution, safety surveillance, import, export, advertising, promotion, recordkeeping and reporting of safety and other post-market information. The FDA and comparable non-U.S. regulatory authorities will continue to closely monitor the safety profile of any product even after approval. If the FDA, or comparable non-U.S. regulatory authorities, become aware of new safety information after approval of any of our product candidates, it may require labeling changes or establishment of a risk evaluation and mitigation strategy or similar strategy, impose significant restrictions on a product’s indicated uses or marketing or impose ongoing requirements for potentially costly post-approval studies or post-market surveillance.
In addition, manufacturers of drug products and their facilities are subject to continual review and periodic inspections by the FDA and other regulatory authorities for compliance with current good manufacturing practices, or cGMP, regulations and standards. If we or a regulatory agency discovers previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, a regulatory agency may impose restrictions on that product, the manufacturing facility or us, including requiring recall or withdrawal of the product from the market or suspension of manufacturing. If we, our product candidates or the manufacturing facilities for our product candidates fail to comply with applicable regulatory requirements, or undesirable side effects caused by such products are identified, a regulatory agency may:
42
The occurrence of any event or penalty described above may inhibit our ability to commercialize our products and generate product revenue.
Advertising and promotion of any product candidate that obtains approval in the United States will be heavily scrutinized by the FDA, the Department of Justice, the Department of Health and Human Services’ Office of Inspector General, state attorneys general, members of Congress and the public. Violations, including promotion of our products for unapproved (or off-label) uses, are subject to enforcement letters, inquiries and investigations and significant civil and criminal sanctions by the government. In the United States, engaging in the impermissible promotion of our products for off-label uses can also subject us to false claims litigation under federal and state statutes, which can lead to significant civil and criminal penalties. Additionally, comparable non-U.S. regulatory authorities will heavily scrutinize advertising and promotion of any product candidate that obtains approval outside of the United States.
The FDA’s policies may change, and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained, which would negatively affect our business, prospects and ability to achieve or sustain profitability.
Risks Related to Our Reliance on Third Parties
We have relied on, and expect to continue to rely on, third-party manufacturers to produce Sozinibercept (OPT-302) or any future product candidates. Any failure by a third-party manufacturer to produce acceptable product candidates for us pursuant to our specifications and regulatory standards may delay or impair our ability to initiate or complete our clinical trials, obtain and maintain regulatory approvals or commercialize approved products.
The manufacturing of biologic drugs such as Sozinibercept (OPT-302) is complex and the process of identifying the qualifying suppliers takes a significant investment of time and money. We do not own or operate manufacturing facilities for the production of clinical or commercial quantities of our product candidates, and we lack the resources and the capabilities to do so. As a result, we currently rely, and expect to rely for the foreseeable future, on third-party manufacturers to supply us with Sozinibercept (OPT-302) and any future product candidates.
We currently have a sole-source relationship with Patheon N.V.("Patheon"), a division of Thermo Fisher Scientific Inc., pursuant to which it supplies us with Sozinibercept (OPT-302) drug substance and drug product. If there should be any disruption in our supply arrangement with Patheon, including any adverse events affecting
43
Patheon or Thermo Fisher Scientific, it could have a negative effect on the clinical development of Sozinibercept (OPT-302) and other operations while we work to identify and qualify an alternate supply source. In addition, we do not have a long-term supply arrangement to purchase anti-VEGF-A therapy for use in combination with Sozinibercept (OPT-302) in our clinical trials and acquire such drug product on a purchase-order basis. Any complications with our existing suppliers of anti-VEGF-A therapies could considerably delay our clinical trials for Sozinibercept (OPT-302), including our Phase 3 pivotal clinical program of Sozinibercept (OPT-302) for the treatment of wet AMD, or the regulatory approvals of Sozinibercept (OPT-302).
Reliance on third-party suppliers and manufacturers entails risks to which we would not be subject if we manufacture product candidates or products ourselves. For example, if we do not maintain our key manufacturing relationships, including with Patheon, we may fail to find replacement manufacturers or develop our own manufacturing capabilities in a timely manner or at all, which could delay or impair our ability to obtain regulatory approval for our products and substantially increase our costs or deplete profit margins, if any. If we do find replacement manufacturers, we may not be able to enter into agreements with them on terms and conditions favorable to us in a timely manner, if at all, and there could be a substantial delay before new facilities could be qualified and registered with or licensed by the FDA and other comparable non-U.S. regulatory authorities.
Even if we are able to establish agreements with third-party manufacturers, reliance on third-party manufacturers entails additional risks, including:
The FDA and other comparable non-U.S. regulatory authorities require manufacturers to register manufacturing facilities. The FDA and other comparable non-U.S. regulatory authorities also inspect these facilities to confirm compliance with cGMP. Contract manufacturers may face manufacturing or quality control problems causing drug-substance production and shipment delays or a situation where the contractor may not be able to maintain compliance with the applicable cGMP requirements. We may have little to no control regarding the occurrence of third-party manufacturer incidents. Failure by our third-party manufacturers and suppliers to pass such inspections and otherwise satisfactorily complete the FDA approval regimen with respect to our product candidate may result in regulatory actions such as the issuance of FDA Form 483 notices of observations, warning letters or injunctions or the loss of operating licenses. In addition, our third-party manufacturers and suppliers are subject to numerous environmental, health and safety laws and regulations, including those governing the handling, use, storage, treatment and disposal of waste products, and failure to comply with such laws and regulations could result in significant costs associated with civil or criminal fines and penalties for such third parties. Any failure to comply with cGMP requirements or other FDA or comparable non-U.S. regulatory
44
requirements could negatively impact our clinical research activities and our ability to develop Sozinibercept (OPT-302) or any future product candidates and market our products following approval.
If Sozinibercept (OPT-302) or any future product candidates are approved by the FDA or other comparable non-U.S. regulatory authorities for commercial sale, we may need to manufacture such product candidate in larger quantities. We intend to use third-party manufacturers for commercial quantities of Sozinibercept (OPT-302) to the extent we advance this product candidate and other product candidates. Our manufacturers may not be able to successfully increase the manufacturing capacity for any of our product candidates in a timely or efficient manner, or at all. If we are unable to successfully increase the manufacturing capacity for a product candidate, the regulatory approval or commercial launch of that product candidate may be delayed or there may be a shortage in the supply of the product candidate.
In addition, the operations of our third-party manufacturers may be subject to earthquakes, power shortages, telecommunications failures, failures or breaches of information technology systems, water shortages, floods, hurricanes, typhoons, fires, extreme weather conditions, medical epidemics, such as the COVID-19 pandemic, and other natural or man-made disasters or business interruptions. Damage or extended periods of interruption to our facilities due to fire, natural disaster, power loss, communications failure, unauthorized entry or other events could cause us to cease or delay development of some or all of our product candidates. Our ability to obtain clinical supplies of our product candidates could be disrupted if the operations of these suppliers are affected by a man-made or natural disaster or other business interruption.
Our current and anticipated future dependence upon others for the manufacture of our product candidates may negatively affect our future profit margins and our ability to develop our product candidates and commercialize any products that receive regulatory approval on a timely basis.
In some cases, the technical skills or technology required to manufacture our product candidates may be unique or proprietary to the original manufacturer, we may have difficulty transferring such skills or technology to another third party and a feasible alternative may not exist. These factors would increase our reliance on such manufacturer or require us to obtain a license from such manufacturer in order to have another third party manufacture our product candidates. If we are required to change manufacturers for any reason, we will be required to verify that the new manufacturer maintains facilities and procedures that comply with quality standards and with all applicable regulations and guidelines and the manufacturer may be required to obtain applicable licenses or approvals. The delays associated with the verification of a new manufacturer, if we are able to identify an alternative source, could negatively affect our ability to develop product candidates in a timely manner or within budget.
The manufacture of biologic products is complex and we are subject to many manufacturing risks, any of which could substantially increase our costs and limit supply of our products.
The manufacture of biologic products is complex and requires significant expertise and capital investment, including the development of advanced manufacturing techniques and process controls. Manufacturers of biologic products often encounter difficulties in production, particularly in scaling up and validating initial production and ensuring the avoidance of contamination. These problems include difficulties with production costs and yields, quality control, including stability of the product, quality assurance testing, operator error and shortages of qualified personnel, as well as compliance with strictly enforced federal, state and non-U.S. regulations. We cannot assure you that any stability or other issues relating to the manufacture of Sozinibercept (OPT-302) will not occur in the future.
The process of manufacturing Sozinibercept (OPT-302) is complex, highly regulated and subject to several risks, including:
45
To date, Sozinibercept (OPT-302) has been manufactured by a single third-party manufacturer, Patheon, solely for preclinical studies and Phase 1, 2 and 3 trials. Any such failure will require us to seek alternative manufacturing sources, which may result in considerable additional expense and delays in our planned clinical trials. We have limited process-development capabilities and have access only to external manufacturing capabilities. We do not have and we do not currently plan to acquire or develop the facilities or capabilities to manufacture bulk drug substance or filled drug product for use in human clinical trials or commercialization. Any delay or interruption in the supply of clinical trial materials, including as a result of breach by us or Patheon of our agreement with Patheon, or our inability to agree to the terms of supply or related services in any statement of work, could delay the completion of clinical trials, increase the costs associated with maintaining clinical trial programs and, depending upon the period of delay, require us to commence new clinical trials at additional expense or terminate clinical trials completely.
Changes in methods of product candidate manufacturing or formulation may result in additional costs or delay.
As product candidates proceed through nonclinical studies to late-stage clinical trials towards potential approval and commercialization, it is common that various aspects of the development program, such as manufacturing methods and formulation, are altered along the way in an effort to optimize processes and results. Such changes carry the risk that they will not achieve these intended objectives. Any of these changes could cause Sozinibercept (OPT-302) or any future product candidate to perform differently and affect the results of planned clinical trials or other future clinical trials conducted with the altered materials. Such changes may also require additional testing, FDA notification or FDA approval. This could delay completion of clinical trials, require the conduct of bridging clinical trials or the repetition of one or more clinical trials, increase clinical trial costs, delay approval of Sozinibercept (OPT-302) or any future product candidate or jeopardize our ability to commence sales and generate revenue.
We rely on third parties to conduct our clinical trials and some aspects of our research and development activities, and those third parties may not perform satisfactorily, including failing to meet deadlines for the completion of such trials, research or testing.
We currently rely on, and expect to continue to rely on, third parties, such as CROs, clinical data management organizations, medical institutions, consultants and clinical investigators, to conduct our clinical trials and certain aspects of our research and development activities. Any of these third parties may terminate their engagements with us at any time. If we need to enter into alternative arrangements, it would delay our product development activities and such alternative arrangements may not be available on terms acceptable to us.
Our reliance on these third parties for research and development activities will reduce our control over these activities but will not relieve us of our responsibilities. For example, we will remain responsible for ensuring that each of our clinical trials is conducted in accordance with the general investigational plan and clinical trial protocols. Moreover, the FDA requires us to comply with standards, commonly referred to as current Good Clinical Practices, for conducting, recording and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of trial participants are protected. We are also required to register ongoing clinical trials and post the results of completed clinical trials on a government-sponsored database within certain time frames. Failure to do so can result in fines, adverse publicity and civil and criminal sanctions.
Furthermore, these third parties may also have relationships with other entities, some of which may be our competitors. If these third parties do not successfully carry out their contractual duties, meet expected deadlines
46
or conduct our clinical trials in accordance with regulatory requirements, standard operating procedures or clinical trial protocols, we will not be able to obtain, or may be delayed in obtaining, marketing approvals for Sozinibercept (OPT-302) or any future product candidates and will not be able to, or may be delayed in our efforts to, successfully commercialize our product candidates.
We also expect to rely on other third parties to store and distribute drug supplies for our clinical trials. Any performance failure on the part of our distributors could delay clinical development, marketing approval and/or commercialization of Sozinibercept (OPT-302) or any future product candidates, producing additional losses and depriving us of potential revenue.
Disputes under key agreements or conflicts of interest with our scientific advisors or clinical investigators could delay or prevent development or commercialization of our product candidates.
Any agreements we have or may enter into with third parties, such as collaboration, license, formulation supplier, manufacturing, clinical research organization or clinical trial agreements, may give rise to disputes regarding the rights and obligations of the parties. Disagreements could develop over contract interpretation, rights to ownership or use of intellectual property, the scope and direction of research and development, the approach for regulatory approvals or commercialization strategy. We intend to conduct research programs in a range of therapeutic areas, but our pursuit of these opportunities could result in conflicts with the other parties to these agreements that may be developing or selling pharmaceuticals or conducting other activities in these same therapeutic areas. Any disputes or commercial conflicts could lead to the termination of our agreements, delay progress of our product development programs, compromise our ability to renew agreements or obtain future agreements, lead to the loss of intellectual property rights, result in increased financial obligations for us or result in costly litigation.
We work with outside scientific advisors and collaborators at academic and other institutions that assist us in our research and development efforts. Our scientific advisors are not our employees and may have other commitments that limit their availability to us. If a conflict of interest between their work for us and their work for another entity arises, we may lose their services.
We may seek to establish commercial collaborations for our product candidates, and, if we are not able to establish them on commercially reasonable terms, we may have to alter our development and commercialization plans.
Our drug development programs and the potential commercialization of our product candidates will require substantial additional cash to fund expenses. We may decide to collaborate with other pharmaceutical and biotechnology companies for the development and potential commercialization of our product candidates.
We face significant competition in seeking appropriate collaborators. Whether we reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of the collaborator’s resources and expertise, the terms and conditions of the proposed collaboration and the proposed collaborator’s evaluation of a number of factors. Those factors may include the design or results of clinical trials, the likelihood of approval by the FDA or comparable non-U.S. regulatory authorities, the potential market for the product candidate, the costs and complexities of manufacturing and delivering such product candidate to patients, the potential of competing products and the existence of uncertainty with respect to our ownership of technology, which can exist if there is a challenge to such ownership without regard to the merits of the challenge and industry and market conditions generally. The collaborators may also consider alternative product candidates or technologies for similar indications that may be available to collaborate on and whether such a collaboration could be more attractive than the one with us for our product candidate.
Collaborations are complex and time-consuming to negotiate and document. In addition, there have been a significant number of business combinations among large pharmaceutical companies that have resulted in a reduced number of potential future collaborators.
We may not be able to negotiate collaborations on a timely basis, on acceptable terms, or at all. If we are unable to do so, we may have to curtail the development of the product candidate for which we are seeking to collaborate, reduce or delay its development program or one or more of our other development programs, delay its potential commercialization or reduce the scope of any sales or marketing activities or increase our expenditures and undertake development or commercialization activities at our own expense. If we elect to increase our expenditures to fund development or commercialization activities on our own, we may need to obtain
47
additional capital, which may not be available to us on acceptable terms, or at all. If we do not have sufficient funds, we may not be able to further develop our product candidates or bring them to market and generate product revenue.
Risks Related to Employee Matters and Managing Our Growth
We must attract and retain highly skilled employees in order to succeed. If we are not able to retain our current senior management team and our scientific advisors or continue to attract and retain qualified scientific, technical and business personnel, our business will suffer.
We may not be able to attract or retain qualified personnel and consultants due to the intense competition for such individuals in the biotechnology and pharmaceutical industries. In particular, we in the future hire employees, including senior employees, in the United States as we continue clinical development of Sozinibercept (OPT-302) and prepare for potential commercialization. The hiring environment in the United States for such candidates is extremely competitive. Many of the other pharmaceutical companies that we compete against for qualified personnel have greater financial and other resources, different risk profiles and a longer history in the industry than we do. They also may provide more diverse opportunities and better chances for career advancement. Some of these characteristics may be more appealing to high-quality candidates than what we have to offer. If we are not able to attract and retain necessary personnel and consultants to accomplish our business objectives, we may experience constraints that will significantly impede the achievement of our development and commercial objectives, our ability to raise additional capital and our ability to implement our business strategy.
Our industry has experienced a high rate of turnover of management personnel in recent years. We are highly dependent on the development, regulatory, commercialization and business development expertise of the members of our executive team, as well as other key employees and consultants. If we lose one or more of our executive officers or other key employees or consultants, our ability to implement our business strategy successfully could be seriously harmed. Any of our executive officers or other key employees or consultants may terminate their employment at any time with three months’ notice, subject to certain exceptions, and replacing such individuals may be difficult and time-consuming because of the limited number of individuals in our industry with the necessary breadth of skills and experience. Competition to hire and retain employees and consultants from this limited pool is intense, and we may be unable to hire, train, retain or motivate such individuals. Additionally, we do not currently maintain “key person” life insurance on the lives of our executives or any of our employees. This lack of insurance means that we may not receive adequate compensation for the loss of the services of these individuals. If we are unable to continue to attract and retain high-quality personnel, the rate and success with which we can discover and develop product candidates and our business will be limited.
Our employees, contractors, vendors, principal investigators, consultants and future partners may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements and insider trading.
We are exposed to the risk of fraud or other misconduct by our employees, contractors, vendors, principal investigators, consultants or future partners. Misconduct by these parties could include failures to comply with FDA or comparable non-U.S. authority regulations, to provide accurate information to the FDA or comparable non-U.S. regulators, to comply with U.S. federal and state and non-U.S. healthcare fraud and abuse laws and regulations, to report financial information or data timely, completely or accurately, or to disclose unauthorized activities to us. In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Third-party misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. Although we have adopted a Code of Conduct, it is not always possible to identify and deter misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us resulting from this misconduct and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant fines or other sanctions. If we or our future partners market products in a manner that violates fraud and abuse and other healthcare laws, or if we or our future partners violate government price reporting laws, we or our future partners may be subject to administrative civil and/or criminal penalties, among other sanctions.
48
Most states also have statutes or regulations similar to these federal laws, which may apply to items such as pharmaceutical products and services reimbursed by private insurers. We and/or our future partners may be subject to administrative, civil and criminal sanctions for violations of any of these federal and state laws. Pharmaceutical and other healthcare companies have been prosecuted under these laws for a variety of promotional and marketing activities, such as: providing free trips, free goods, sham consulting fees and grants and other monetary benefits to prescribers; reporting to pricing services inflated average wholesale prices that were then used by federal programs to set reimbursement rates; engaging in off-label promotion; and submitting inflated best price information to the Medicaid Rebate Program to reduce liability for Medicaid rebates. Ensuring that our internal operations and future business arrangements with third parties comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations, agency guidance or case law involving applicable fraud and abuse or other healthcare laws and regulations.
We may encounter difficulties in managing our growth, which could negatively impact our operations.
As we advance our clinical development programs for product candidates, seek regulatory approval in the United States and elsewhere and increase the number of ongoing product development programs, we anticipate that we will need to increase our product development, scientific and administrative headcount. In particular, as we progress our Phase 3 clinical trials for Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD, we will require additional key staff for clinical development operations as well as additional key financial and administrative personnel. We will also need to establish commercial capabilities in order to commercialize any product candidates that may be approved. Such an evolution may impact our strategic focus and our deployment and allocation of resources.
Our ability to manage our operations and growth effectively depends upon the continual improvement of our procedures, reporting systems and operational, financial and management controls. We currently have no experience as a company in or infrastructure for sales, marketing and distribution, and our operations are currently limited to clinical development activities and as our operations expand, we likely will need to manage additional relationships with such third parties. We may not be able to implement administrative and operational improvements in an efficient or timely manner and may discover deficiencies in existing systems and controls. If we do not meet these challenges, we may be unable to execute our business strategies and may be forced to expend more resources than anticipated addressing these issues.
We may acquire additional technology and complementary businesses in the future. Acquisitions involve many risks, any of which could materially harm our business, including the diversion of management’s attention from core business concerns, failure to effectively exploit acquired technologies, failure to successfully integrate the acquired business or realize expected synergies or the loss of key employees from either our business or the acquired businesses.
If Sozinibercept (OPT-302) or any future product candidate is approved, we intend either to establish a sales and organization with technical expertise and supporting distribution capabilities to commercialize Sozinibercept (OPT-302) or any future product candidate or to outsource such functions to one or more third parties. Either of these options would be expensive and time-consuming. Some or all of these costs may be incurred in advance of any approval of Sozinibercept (OPT-302) or any future product candidate. In addition, we may not be able to hire a sales force that is sufficient in size or has adequate expertise in the medical markets that we intend to target.
If we are unable to successfully manage our growth and the increased complexity of our operations, our business, financial position, results of operations and prospects may be harmed.
Risks Related to Intellectual Property
Our success depends upon our ability to obtain and maintain intellectual property protection for our products and technologies.
Our success will depend in significant part on our current or future licensors’, licensees’ or collaborators’ ability to establish and maintain adequate protection of our owned and licensed intellectual property covering the product candidates we plan to develop, and the ability to develop these product candidates and commercialize the products resulting therefrom, without infringing the intellectual property rights of others. In addition to taking other steps to protect our intellectual property, we hold issued patents, we have applied for patents, and we intend to continue to apply for, patents with claims covering our technologies, processes and product candidates when
49
and where we deem it appropriate to do so. We have filed patent applications both in the United States and in certain non-U.S. jurisdictions to obtain patent rights to inventions we have developed, with claims directed to compositions of matter, methods of use and other technologies relating to our programs. There can be no assurance that any of these patent applications will issue as patents or, for those applications that do mature into patents, that the claims of the patents will exclude others from making, using or selling our product candidates or products that compete with or are similar to our product candidates. In countries where we have not sought and do not seek patent protection, third parties may be able to manufacture and sell our product candidates without our permission, and we may not be able to stop them from doing so.
With respect to patent rights, we do not know whether any of the pending patent applications for any of our product candidates will result in the issuance of patents that effectively protect our technologies, processes and product candidates, or if any of our issued patents or our current or future licensors’, licensees’ or collaborators’ issued patents will effectively prevent others from commercializing competitive technologies, processes and products. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing or in some cases not at all, until they are issued as a patent. Therefore, we cannot be certain that we or our current or future licensors, licensees or collaborators were the first to make the inventions claimed in our owned or licensed patents or pending patent applications, or that we or our current or future licensors, licensees or collaborators were the first to file for patent protection of such inventions. For a description of our patent portfolio, see “Item 4B Business Overview” of this annual report.
Any changes we make to Sozinibercept (OPT-302) or any future product candidates to cause them to have what we view as more advantageous properties may not be covered by our existing patents and patent applications, and we may be required to file new applications and/or seek other forms of protection for any such altered product candidates. The patent landscape surrounding the technology underlying our product candidates is potentially crowded, and there can be no assurance that we would be able to secure patent protection that would adequately cover an alternative to Sozinibercept (OPT-302) or any future product candidates.
The patent prosecution process is expensive and time-consuming, and we and our current or future licensors, licensees or collaborators may not be able to prepare, file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we or our current or future licensors, licensees or collaborators will fail to identify patentable aspects of inventions made in the course of development and commercialization activities before it is too late to obtain patent protection for them. Moreover, in some circumstances, we may not have the right to control the preparation, filing and prosecution of patent applications, or to maintain or enforce the patents, covering technology that we license from or license to third parties and may be reliant on our current or future licensors, licensees or collaborators to perform these activities, which means that these patent applications may not be prosecuted, and these patents enforced, in a manner consistent with the best interests of our business. If our current or future licensors, licensees or collaborators fail to establish, maintain, protect or enforce such patents and other intellectual property rights, such rights may be reduced or eliminated. If our current or future licensors, licensees or collaborators are not fully cooperative or disagree with us as to the prosecution, maintenance or enforcement of any patent rights, such patent rights could be compromised.
Similar to the patent rights of other biotechnology companies, the scope, validity and enforceability of our owned and licensed patent rights generally are highly uncertain and involve complex legal and factual questions. The issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and our patents may be challenged in the courts or patent offices in the United States and abroad. In recent years, these areas have been the subject of much litigation in the industry. As a result, the issuance, scope, validity, enforceability and commercial value of our and our current or future licensors’, licensees’ or collaborators’ patent rights are highly uncertain. Our and our current or future licensors’, licensees’ or collaborators’ pending and future patent applications may not result in patents being issued that protect our technology or product candidates, or products resulting therefrom, in whole or in part, or that effectively prevent others from commercializing competitive technologies and products. The patent examination process may require us or our current or future licensors, licensees or collaborators to narrow the scope of the claims of pending and future patent applications, which would limit the scope of patent protection that is obtained, if any. Our and our current or future licensors’, licensees’ or collaborators’ patent applications cannot be enforced against third parties practicing the technology that is currently claimed in such applications unless and until a patent issues from such applications, and then only to the extent the claims that issue are broad enough to cover the technology being practiced by those third parties.
Furthermore, given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates might expire before or shortly after the resulting products
50
are commercialized. As a result, our owned and in-licensed patents may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours. We expect to seek extensions of patent terms for our issued patents, where available. This includes in the United States under the Hatch-Waxman Act, which permits a patent term extension of up to five years beyond the original expiration date of the patent as compensation for regulatory delays. However, such a patent term extension cannot lengthen the remaining term of a patent beyond a total of 14 years from the product’s approval date. Only one patent applicable to each regulatory review period may be extended and only those claims covering the approved drug, a method for using it, or a method for manufacturing it may be extended, with extended rights limited to the approved product, its approved uses and/or its manufacture. During the period of patent term extension, the claims of a patent are not enforceable for their full scope, but are instead limited to the scope of the approved product. In addition, the applicable authorities, including the FDA in the United States, and any comparable non-U.S. regulatory authorities, may not agree with our assessment of whether such extensions are available, and may refuse to grant extensions to our patents, or may grant more limited extensions than we request. In addition, we may not be granted an extension because of, for example, failing to apply within applicable deadlines, failing to apply prior to the expiration of relevant patents or otherwise failing to satisfy applicable requirements. If this occurs, any period during which we have the right to exclusively market our product will be shorter than we would otherwise expect, and our competitors may obtain approval of and launch products earlier than might otherwise be the case.
We may not be able to protect our intellectual property rights throughout the world.
The legal protection afforded to inventors and owners of intellectual property in countries outside of the United States may not be as protective or effective as that in the United States and we may, therefore, be unable to acquire and enforce intellectual property rights outside the United States to the same extent as in the United States. Whether filed in the United States or abroad, our patent applications may be challenged or may fail to result in issued patents.
In addition, our existing patents and any future patents we obtain may not be sufficiently broad to prevent others from practicing our technologies or from developing or commercializing competing products. Furthermore, others may independently develop or commercialize similar or alternative technologies or drugs, or design around our patents. Our patents may be challenged, invalidated, circumvented or narrowed, or fail to provide us with any competitive advantages. In many non-U.S. countries, patent applications and/or issued patents, or parts thereof, must be translated into the native language. If our patent applications or issued patents are translated incorrectly, they may not adequately cover our technologies; in some countries, it may not be possible to rectify an incorrect translation, which may result in patent protection that does not adequately cover our technologies in those countries.
Filing, prosecuting, enforcing and defending patents on product candidates in all countries throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States may be less extensive than those in the United States. In addition, the laws of some non-U.S. countries do not protect intellectual property rights to the same extent as federal and certain state laws in the United States. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Competitors may use technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States. These products may compete with Sozinibercept (OPT-302) or any future product candidates and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in non-U.S. jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biopharmaceuticals. This could make it difficult for us to stop the infringement of our patents or the marketing of competing products in violation of our proprietary rights, generally. Proceedings to enforce our patent rights in non-U.S. jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly, could place our patent applications at risk of not issuing and could provoke third parties to assert claims against us or our collaborator. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful.
51
The requirements for patentability differ and certain countries have heightened requirements for patentability, requiring more disclosure in the patent application. In addition, certain countries have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties. In those countries, we may have limited remedies if patents are infringed or if we are compelled to grant a license to a third party, which could materially diminish the value of those patents. This could limit our potential revenue opportunities. In addition, many countries limit the enforceability of patents against government agencies or government contractors. In these countries, the patent owner may have limited remedies, which could materially diminish the value of such patent. Accordingly, our efforts to enforce intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we own or license.
Changes in patent law could diminish the value of patents in general, thereby impairing our ability to protect Sozinibercept (OPT-302) and any future product candidates.
As is the case with other biotechnology and pharmaceutical companies, our success is heavily dependent on intellectual property rights, particularly patents. Obtaining and enforcing patents in the biopharmaceutical industry involves technological and legal complexity, and obtaining and enforcing biopharmaceutical patents is costly, time-consuming and inherently uncertain. The U.S. Supreme Court in recent years has issued rulings either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations or ruling that certain subject matter is not eligible for patent protection. In addition to increasing uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by Congress, the federal courts, the USPTO and equivalent bodies in non-U.S. jurisdictions, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce existing patents and patents we may obtain in the future.
Patent reform laws, such as the Leahy-Smith America Invents Act, or the Leahy-Smith Act, as well as changes in how patent laws are interpreted, could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. The Leahy-Smith Act made a number of significant changes to U.S. patent law. These include provisions that affect the filing and prosecution strategies associated with patent applications, including a change from a “first-to-invent” to a “first-inventor-to-file” patent system, and a change allowing third-party submission of prior art to the USPTO during patent prosecution and additional procedures to attack the validity of a patent by USPTO-administered post-grant proceedings, including post-grant review, inter partes review and derivation proceedings. The USPTO has developed regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act and, in particular, the “first-inventor-to-file” provisions, became effective in 2013. The Leahy-Smith Act and its implementation may increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could harm our business, financial condition and results of operations.
We may be unable to obtain intellectual property rights or technology necessary to develop and commercialize Sozinibercept (OPT-302) and any future product candidates.
The patent landscape around our programs is complex, and there may be one or more third-party patents and patent applications containing subject matter that might be relevant to Sozinibercept (OPT-302). Depending on what claims may ultimately issue from these patent applications, and how courts construe the issued patent claims, as well as depending on the ultimate formulation and method of use of Sozinibercept (OPT-302) or any future product candidates, we may need to obtain a license to practice the technology claimed in such patents. There can be no assurance that such licenses will be available on commercially reasonable terms, or at all. If a third party does not offer us a necessary license or offers a license only on terms that are unattractive or unacceptable to us, we might be unable to develop and commercialize one or more of our product candidates, which would harm our business, financial condition and results of operations. Moreover, even if we obtain licenses to such intellectual property, but subsequently fail to meet our obligations under the relevant license agreements, or such license agreements are terminated for any other reasons, we may lose our rights to the technologies licensed under those agreements.
The licensing or acquisition of third-party intellectual property rights is an area in which many companies operate that have interests that are in conflict with ours, and several more established companies may pursue strategies to license or acquire third-party intellectual property rights that we may consider attractive or necessary. These established companies may have a competitive advantage over us due to their size, capital resources and greater clinical development and commercialization capabilities. In addition, companies that perceive us to be a
52
competitor may be unwilling to assign or license rights to us. We also may be unable to license or acquire third-party intellectual property rights on terms that would allow us to make an appropriate return on our investment, or at all. If we are unable to successfully obtain rights to required third-party intellectual property rights or maintain the existing intellectual property rights we have, we may have to abandon development of the relevant program or product candidate, which could harm our business, financial condition, results of operations and prospects.
We may become involved in lawsuits or other proceedings to protect or enforce our intellectual property, which could be expensive, time-consuming and unsuccessful and have a negative effect on the success of our business.
Third parties may infringe our patents or misappropriate or otherwise violate our intellectual property rights. In the future, we may initiate legal proceedings to enforce or defend our intellectual property rights, to protect our trade secrets or to determine the validity or scope of intellectual property rights we own or control. Also, third parties may initiate legal proceedings against us to challenge the validity or scope of intellectual property rights we own, control or to which we have rights. For example, generic or biosimilar drug manufacturers or other competitors or third parties may challenge the scope, validity or enforceability of our patents, requiring us to engage in complex, lengthy and costly litigation or other proceedings. These proceedings can be expensive and time-consuming and many of our adversaries in these proceedings may have the ability to dedicate substantially greater resources to prosecuting these legal actions than we can. Accordingly, despite our efforts, we may not be able to prevent third parties from infringing upon or misappropriating intellectual property rights we own, control or have rights to, particularly in countries where the laws may not protect those rights as fully as in the United States. Litigation could result in substantial costs and diversion of management resources, which could harm our business and financial results. In addition, if we initiated legal proceedings against a third party to enforce a patent covering a product candidate, the defendant could counterclaim that such patent is invalid or unenforceable. In patent litigation in the United States, defendants usually assert counterclaims alleging invalidity or unenforceability. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness or non-enablement. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant information from the USPTO, or made a misleading statement, during prosecution. In an infringement or declaratory judgment proceeding, a court may decide that a patent owned by or licensed to us is invalid or unenforceable, or may refuse to stop the other party from using the subject matter alleged to be infringing on the grounds that our patents do not cover that subject matter. An adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated, narrowed, held unenforceable or interpreted in such a manner that would not preclude third parties from entering the market with competing products.
Third-party pre-issuance submission of prior art to the USPTO, or opposition, derivation, revocation, reexamination, inter partes review or interference proceedings, or other pre-issuance or post-grant proceedings or other patent office proceedings or litigation in the United States or other jurisdictions provoked by third parties or brought by us, may be necessary to determine the inventorship, priority, patentability or validity of inventions with respect to our patents or patent applications. An unfavorable outcome could leave our technology or product candidates without patent protection, allow third parties to commercialize our technology or product candidates and compete directly with us, without payment to us, or could require us to obtain license rights from the prevailing party in order to be able to manufacture or commercialize our product candidates without infringing third-party patent rights. Our business could be harmed if the prevailing party in such a case does not offer us a license on commercially reasonable terms, or at all. Even if we obtain a license, it may be non-exclusive, thereby giving our competitors access to the same technologies licensed to us. In addition, if the breadth or strength of protection provided by our patents and patent applications is threatened, it could dissuade companies from collaborating with us to license, develop or commercialize current or future product candidates. Even if we successfully defend such litigation or proceeding, we may incur substantial costs and our defense may distract our management and other employees.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, many non-U.S. jurisdictions have rules of discovery that are different than those in the United States and that may make defending or enforcing our patents extremely difficult. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could negatively affect the price of the ADSs and our ordinary shares.
53
Third parties may initiate legal proceedings against us alleging that we infringe their intellectual property rights, the outcome of which would be uncertain and could harm our business.
Our commercial success depends upon our ability to develop, manufacture, market and sell Sozinibercept (OPT-302) and any future product candidates that we may develop and use our proprietary technologies without infringing, misappropriating or otherwise violating the intellectual property and proprietary rights of third parties. The biotechnology and pharmaceutical industries are characterized by extensive litigation regarding patents and other intellectual property rights. Third parties may initiate legal proceedings against us alleging that we infringe their intellectual property rights or we may initiate legal proceedings against third parties to challenge the validity or scope of intellectual property rights controlled by third parties, including in oppositions, interferences, revocations, reexaminations, inter partes review or derivation proceedings before the USPTO or its counterparts in other jurisdictions. These proceedings can be expensive and time-consuming and many of our adversaries in these proceedings may have the ability to dedicate substantially greater resources to prosecuting these legal actions than we can.
We could be found liable for monetary damages, including treble damages and attorneys’ fees, if we are found to have willfully infringed a patent of a third party. A finding of infringement could prevent us from commercializing Sozinibercept (OPT-302) or any future product candidates or force us to cease some of our business operations, which could materially harm our business.
We may not be aware of all third-party intellectual property rights potentially relating to Sozinibercept (OPT-302) or any future product candidates and technologies. We are not aware of any facts that would lead us to conclude that the valid and enforceable claims of any third-party patents would reasonably be interpreted to cover our product candidates. As to pending third-party applications, we cannot predict with any certainty which claims will issue, if any, or the scope of such issued claims. Even if we believe third-party intellectual property claims are without merit, there is no assurance that a court would find in our favor on questions of infringement, validity, enforceability or priority. A court of competent jurisdiction could hold that these third-party patents are valid, enforceable and infringed, which could materially and negatively affect our ability to commercialize any product candidates we may develop and any other product candidates or technologies covered by the asserted third-party patents. In order to successfully challenge the validity of any such U.S. patent in federal court, we would need to overcome a presumption of validity. As this burden is a high one requiring us to present clear and convincing evidence as to the invalidity of any such U.S. patent claim, there is no assurance that a court of competent jurisdiction would invalidate the claims of any such U.S. patent. If any such third-party patents (including those that may issue from such applications) were successfully asserted against us or other commercialization partners and we were unable to successfully challenge the validity or enforceability of any such asserted patents, then we and other commercialization partners may be prevented from commercializing our product candidates, or may be required to pay significant damages, including treble damages and attorneys’ fees if we are found to willfully infringe the asserted patents, or obtain a license to such patents, which may not be available on commercially reasonable terms, or at all. Even if we were able to obtain a license, it could be non-exclusive, thereby giving our competitors and other third parties access to the same technologies licensed to us, and it could require us to make substantial licensing and royalty payments. Any of the foregoing would harm our business, financial condition and operating results.
Although we have reviewed certain third-party patents and patent filings that we believe may be relevant to our therapeutic candidates or products, we have not conducted a freedom-to-operate search or analysis for any of our therapeutic candidates or products, and we may not be aware of patents or pending or future patent applications that, if issued, would block us from commercializing our therapeutic candidates or products. Thus, we cannot guarantee that our therapeutic candidates or products, or our commercialization thereof, do not and will not infringe any third party’s intellectual property.
We may be subject to claims by third parties asserting misappropriation of intellectual property, or claiming ownership of what we regard as our own intellectual property.
Although we seek to ensure that our employees do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that we or these employees have used or disclosed confidential information or intellectual property, including trade secrets or other proprietary information, of any such employee’s former employer, or that third parties have an interest in our patents as an inventor or co-inventor. Litigation may be necessary to defend against these claims. If we fail in prosecuting or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or the services of personnel or sustain other damages. Such intellectual property rights could be awarded to a third party, and we could be required to obtain a license from such third party to commercialize our technology or products. Such a
54
license may not be available on commercially reasonable terms, or at all. Even if we successfully prosecute or defend against such claims, litigation could result in substantial costs and distract management.
In addition, while it is our policy to require our employees and contractors who may be involved in the conception or development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who, in fact, conceives or develops intellectual property that we regard as our own. The assignment of intellectual property rights may not be self-executing, or the assignment agreements may be breached, and we may be forced to bring claims against third parties, or defend claims that they may bring against us, to determine the ownership of what we regard as our intellectual property. Such claims could harm our business, financial condition, results of operations and prospects.
Our inability to protect our confidential information and trade secrets would harm our business and competitive position.
In addition to seeking patents for some of our technology and product candidates, we also rely substantially on trade secrets, including unpatented know-how, technology and other proprietary materials and information, to maintain our competitive position. We seek to protect these trade secrets, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, corporate collaborators, outside scientific collaborators, contract manufacturers, consultants, advisors and other third parties. We also enter into confidentiality and invention or patent assignment agreements with our employees and consultants. However, these steps may be inadequate, we may fail to enter into agreements with all such parties or any of these parties may breach the agreements and disclose our trade secrets and there may be no adequate remedy available for such breach of an agreement. We cannot assure you that our trade secrets will not be disclosed or that we can meaningfully protect our trade secrets. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time consuming, and the outcome is unpredictable. In addition, some courts both within and outside the United States may be less willing, or unwilling, to protect trade secrets. If a competitor lawfully obtained or independently developed any technology or information that we protect as trade secret, we would have no right to prevent such competitor from using that technology or information to compete with us, which could harm our competitive position.
Intellectual property rights do not necessarily address all potential threats.
The degree of future protection afforded by our intellectual property rights is uncertain because intellectual property rights have limitations and may not adequately protect our business or permit us to maintain our competitive advantage. For example:
55
Should any of these events occur, they could harm our business, financial condition, results of operations and prospects.
If our trademarks and trade names are not maintained and adequately protected, we may not be able to build name recognition in our markets of interest, and our business may be negatively affected.
Failure to obtain trademark registrations in the future, could limit our ability to protect and enforce our trademarks and impede our marketing efforts in the countries in which we operate. We may not be able to protect our rights to trademarks and trade names which we may need to build name recognition with potential partners or customers in our markets of interest. As a means to enforce any future trademark rights and prevent infringement, we may be required to file trademark claims against third parties or initiate trademark opposition proceedings. This can be expensive and time consuming and can strain the financial resources of a company of our size, and we may not be successful in enforcing our trademark rights. In addition, our registered or unregistered trademarks or trade names may be challenged, infringed, circumvented, declared generic or determined to be infringing on other marks.
Future trademark applications in the United States and in other non-U.S. jurisdictions where we may file may not be allowed or may subsequently be opposed. Even if these applications result in registration of trademarks, third parties may challenge our use or registration of these trademarks in the future. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be negatively affected.
Our product candidates may face competition sooner than anticipated from biosimilar products.
Even if we are successful in achieving regulatory approval to commercialize a product candidate faster than our competitors, our product candidates may face competition from biosimilar products. In the United States, our product candidates are regulated by the FDA. The Biologics Price Competition and Innovation Act of 2009, or BPCIA, created an abbreviated pathway for the approval of biosimilar and interchangeable biologic products. The abbreviated regulatory pathway establishes legal authority for the FDA to review and approve biosimilar biologics, including the possible designation of a biosimilar as “interchangeable” based on its similarity to an existing brand product. Under the BPCIA, an application for a biosimilar product cannot be approved by the FDA until 12 years after the original branded product was approved under a BLA. The law is complex and is still being interpreted and implemented by the FDA. As a result, its ultimate impact, implementation, and meaning are subject to uncertainty. While it is uncertain when such processes intended to implement BPCIA may be fully adopted by the FDA, any such processes could have a material adverse effect on the future commercial prospects for our product candidates.
There is a risk that any exclusivity we may be afforded if any of our product candidates are approved as a biologic product under a BLA could be shortened due to congressional action or otherwise, or that the FDA will not consider our product candidates to be reference products for competing products, potentially creating the opportunity for generic or biosimilar competition sooner than anticipated. Moreover, the extent to which a biosimilar product, once approved, will be substituted for any one of our reference products in a way that is similar to traditional generic substitution for non-biologic products is not yet clear, and will depend on a number of marketplace and regulatory factors that are still developing. In addition, a competitor could decide to forego the biosimilar approval path and submit a full BLA after completing its own preclinical studies and clinical trials. In such cases, any exclusivity to which we may be eligible under the BPCIA would not prevent the competitor from marketing its product as soon as it is approved.
56
In Europe, the European Commission has granted marketing authorizations for several biosimilar products pursuant to a set of general and product class-specific guidelines for biosimilar approvals issued over the past few years. In Europe, a competitor may reference data supporting approval of an innovative biological product, but will not be able to market it until 10 years after the time of approval of the innovative product. This 10-year marketing exclusivity period may be extended to 11 years if, during the first eight of those 10 years, the marketing authorization holder obtains an approval for one or more new therapeutic indications that bring significant clinical benefits compared with existing therapies. In addition, companies may be developing biosimilar products in other countries that could compete with our products, if approved.
If competitors are able to obtain marketing approval for biosimilars referencing our product candidates, if approved, such products may become subject to competition from such biosimilars, with the attendant competitive pressure and potential adverse consequences. Such competitive products may be able to immediately compete with us in each indication for which our product candidates may have received approval.
Risks Related to Ownership of the ADSs
The trading price and volume of the ADSs may be volatile, and ADSs holders could incur substantial losses.
The price and trading volumes of our ordinary shares and ADSs may be significantly affected by events such as announcements regarding scientific and clinical results concerning product candidates currently being developed by us, our collaboration partners or our main competitors, changes in market conditions related to our sector of activity, announcements of new contracts, technological innovations and collaborations by us or our main competitors, developments concerning intellectual property rights, as well as the development, regulatory approval and commercialization of new products by us or our main competitors and changes in our financial results.
In addition, equity markets may be subject to considerable price and trading volume fluctuations, and often, these movements do not reflect the operational and financial performance of the listed companies concerned. In particular, biotechnology companies’ share prices have been highly volatile in the past and may continue to be highly volatile in the future. As we operate in a single industry, we are especially vulnerable to these factors to the extent that they affect our industry. Fluctuations in the stock market as well as the macroeconomic environment could significantly affect the price of the ADSs. As a result of this volatility, investors may not be able to sell their ADSs at or above the price originally paid for the security. The market price and trading volume for the ADSs may be influenced by many factors, including:
57
These and other market and industry factors may cause the market price and demand for the ADSs to fluctuate, regardless of our actual operating performance, which may limit or prevent investors from readily selling their ADSs and may otherwise negatively affect the liquidity of the trading market for the ADSs.
We do not currently intend to pay dividends on our securities and, consequently, your ability to achieve a return on your investment will depend on appreciation in the price of the ADSs.
We have not declared or paid any cash dividends on our ordinary shares since February 2005 and do not currently intend to do so for the foreseeable future. We currently intend to invest our future earnings, if any, to fund our operations and growth. Therefore, you are not likely to receive any dividends on your ADSs for the foreseeable future and the success of an investment in the ADSs will depend upon any future appreciation in its value. Consequently, investors may need to sell all or part of their holdings of the ADSs after price appreciation, which may never occur, as the only way to realize any future gains on their investment. There is no guarantee that the ADSs will appreciate in value or even maintain the price at which our shareholders have purchased them. Investors seeking cash dividends should consider not purchasing the ADSs.
While we do not anticipate paying any cash dividends on our ordinary shares in the foreseeable future, if such a dividend is declared, the depositary for the ADSs has agreed to pay to you the cash dividends or other distributions it or the custodian receives on our ordinary shares or other deposited securities after deducting its fees and expenses. You will receive these distributions in proportion to the number of our ordinary shares your ADSs represent. However, in accordance with the limitations set forth in the deposit agreement, it may be unlawful or impractical to make a distribution available to holders of ADSs. We have no obligation to take any other action to permit the distribution of the ADSs, ordinary shares, rights or anything else to holders of the ADSs. This means that you may not receive the distributions we make on our ordinary shares or any value from them if it is unlawful or impractical to make them available to you. These restrictions may negatively impact the value of your ADSs. In addition, exchange rate fluctuations may affect the amount of Australian dollars that we are able to distribute, and the amount in U.S. dollars that our shareholders receive upon the payment of cash dividends or other distributions we declare and pay in Australian dollars, if any. These factors could harm the value of the ADSs, and, in turn, the U.S. dollar proceeds that holders receive from the sale of the ADSs.
58
Future sales of ordinary shares or ADSs by existing holders could depress the market price of the ordinary shares or ADSs.
We had a total of 467,159,434 ordinary shares outstanding as of June 30, 2023. In August 2023 Opthea announced a non-underwritten institutional placement (“Placement”) and accelerated non-renounceable entitlement offering (“ANREO”) offering of A$90 million,- (approximately US$58 million) which was completed in September 2023. Existing shareholders were entitled to purchase new shares at a ratio of 1 for 3.07 pro-rata. The Placement and the institutional component (Institutional Entitlement Offer) of the fully underwritten ANREO together raised approximately A$73.7 million, (US$ 47.2 million) and the retail component raised approximately A$16.3 million (US$10.8 million). Total shares outstanding as of the date of this report is 662,806,891.
Participants in the Placement and ANREO will receive one option, each exercisable at A$0.80 per option and expiring on August 31, 2025 (New Options), for every two New Shares issued under the Placement and ANREO. A total of approximately 98 million New Options were issued in connection with the Placement and ANREO.
As of the date of this annual report, the exercise of all outstanding options/rights and ADS options exercisable for ordinary shares would enable the subscription of new ordinary shares representing approximately 20.0% of the diluted share capital. The ordinary shares subject to subscription under outstanding options exercisable for ordinary shares will become eligible for sale in the public market in the future, subject to certain legal and contractual limitations. Sales of a large number of the ordinary shares in the public market could depress the market price of the ADSs. If these additional ordinary shares are sold, or if it is perceived that they will be sold, in the public market, the trading price of the ordinary shares and ADSs could decline substantially, which could impair our ability to raise additional capital through the issuance of ordinary shares, ADSs or other securities in the future.
The dual listing of our ordinary shares and the ADSs may negatively impact the liquidity and value of the ADSs.
Our ADSs are listed on Nasdaq and our ordinary shares are listed on the ASX. We cannot predict the effect of this dual listing on the value of our ordinary shares and ADSs. However, the dual listing of our ordinary shares and ADSs may dilute the liquidity of these securities in one or both markets and may negatively impact the development of an active trading market for the ADSs in the United States. The price of the ADSs could also be negatively impacted by trading in our ordinary shares on the ASX.
We have incurred and will continue to incur significant increased costs as a result of operating as a company with ADSs that are publicly traded in the United States, and our management will be required to devote substantial time to new compliance initiatives.
As a company whose ADSs are publicly traded in the United States, we have incurred and will continue to incur significant legal, accounting, insurance and other expenses. In addition, the Sarbanes-Oxley Act, Dodd-Frank Wall Street Reform and Consumer Protection Act and related rules implemented by the United States Securities and Exchange Commission, or SEC, and Nasdaq have imposed various requirements on public companies listed in the United States including requiring establishment and maintenance of effective disclosure and financial controls. Our management and other personnel will need to devote a substantial amount of time to these compliance initiatives, and we will need to add additional personnel and build our internal compliance infrastructure. Moreover, these rules and regulations will increase our legal and financial compliance costs and will make some activities more time-consuming and costly. These laws and regulations could also make it more difficult and expensive for us to attract and retain qualified persons to serve on our board of directors, our board committees or as our senior management. Furthermore, if we are unable to satisfy our obligations as a public company listed in the United States, we could be subject to delisting of the ADSs, fines, sanctions and other regulatory action and potentially civil litigation.
U.S. investors may have difficulty enforcing civil liabilities against our company, our directors or members of senior management and the experts named in this annual report.
Certain members of our senior management and board of directors named in this annual report are non-residents of the United States, and a substantial portion of the assets of such persons are located outside the United States. As a result, it may be impracticable to serve process on such persons in the United States or to enforce judgments obtained in U.S. courts against them based on civil liability provisions of the securities laws of the
59
United States. Even if you are successful in bringing such an action, there is doubt as to whether Australian courts would enforce certain civil liabilities under U.S. securities laws in original actions or judgments of U.S. courts based upon these civil liability provisions. In addition, awards of punitive damages in actions brought in the United States or elsewhere may be unenforceable in Australia or elsewhere outside the United States. An award for monetary damages under U.S. securities laws would be considered punitive if it does not seek to compensate the claimant for loss or damage suffered and is intended to punish the defendant. The enforceability of any judgment in Australia will depend on the particular facts of the case as well as the laws and treaties in effect at the time. The United States and Australia do not currently have a treaty or statute providing for recognition and enforcement of the judgments of the other country (other than arbitration awards) in civil and commercial matters.
As a result, our public shareholders may have more difficulty in protecting their interests through actions against us, our management or our directors than would shareholders of a corporation incorporated in a jurisdiction in the United States. In addition, as a company incorporated in Australia, the provisions of the Corporations Act 2001(Cth), or the Corporations Act, regulate the circumstances in which shareholder derivative actions may be commenced which may be different, and in many ways less permissive, than for companies incorporated in the United States.
Australian takeover laws may discourage takeover offers being made for us or may discourage the acquisition of a significant position in our ordinary shares or ADSs.
We are incorporated in Australia and are subject to the takeover laws of Australia. Among other things, we are subject to the Corporations Act. Subject to a range of exceptions, the Corporations Act prohibits the acquisition of a direct or indirect interest in our issued voting shares if the acquisition of that interest will lead to a person’s voting power in us increasing to more than 20%, or increasing from a starting point that is above 20% and below 90%. Australian takeover laws may discourage takeover offers being made for us or may discourage the acquisition of a significant position in our ordinary shares. This may have the ancillary effect of entrenching our board of directors and may deprive or limit our shareholders’ opportunity to sell their ordinary shares and may further restrict the ability of our shareholders to obtain a premium from such transactions. See Exhibit 2.3 “Description of Securities” as well as our Constitution, which is included as an exhibit to this annual report.
Our Constitution and Australian laws and regulations applicable to us may adversely affect our ability to take actions that could be beneficial to our shareholders.
As an Australian company we are subject to different corporate requirements than a corporation organized under the laws of the United States. Our Constitution, as well as the Corporations Act, sets forth various rights and obligations that apply to us as an Australian company and which may not apply to a U.S. corporation. These requirements may operate differently than those of many U.S. companies. You should carefully review the summary of these matters set forth under Exhibit 2.3 “Description of Securities” as well as our Constitution, which is included as an exhibit to this annual report, prior to investing in our securities.
Your right as a holder of ADSs to participate in any future preferential subscription rights offering or to elect to receive dividends in ordinary shares may be limited, which may cause dilution to your holdings.
The deposit agreement provides that the depositary will not make rights available to you unless the distribution to ADS holders of both the rights and any related securities are either registered under the Securities Act of 1933, as amended, or the Securities Act or exempted from registration under the Securities Act. If we offer holders of our ordinary shares the option to receive dividends in either cash or shares, under the deposit agreement the depositary may require satisfactory assurances from us that extending the offer to holders of ADSs does not require registration of any securities under the Securities Act before making the option available to holders of ADSs. We are under no obligation to file a registration statement with respect to any such rights or securities or to endeavor to cause such a registration statement to be declared effective. Moreover, we may not be able to establish an exemption from registration under the Securities Act. Accordingly, ADS holders may be unable to participate in our rights offerings or to elect to receive dividends in shares and may experience dilution in their holdings. In addition, if the depositary is unable to sell rights that are not exercised or not distributed or if the sale is not lawful or reasonably practicable, it will allow the rights to lapse, in which case you will receive no value for these rights.
60
You may not be able to exercise your right to vote the ordinary shares underlying your ADSs.
Holders of ADSs may exercise voting rights with respect to the ordinary shares represented by the ADSs only in accordance with the provisions of the deposit agreement. The deposit agreement provides that, upon receipt of notice of any meeting of holders of our ordinary shares, the depositary will fix a record date for the determination of ADS holders who shall be entitled to give instructions for the exercise of voting rights. Upon timely receipt of notice from us, if we so request, the depositary shall distribute to the holders as of the record date (i) the notice of the meeting or solicitation of consent or proxy sent by us and (ii) a statement as to the manner in which instructions may be given by the holders.
You may instruct the depositary to vote the ordinary shares underlying your ADSs. Otherwise, you will not be able to exercise your right to vote, unless you withdraw the ordinary shares underlying the ADSs you hold. However, you may not know about the meeting far enough in advance to withdraw those ordinary shares. If we ask for your instructions, the depositary, upon timely notice from us, will notify you of the upcoming vote and arrange to deliver our voting materials to you and will try to vote ordinary shares as you instruct. We cannot guarantee you that you will receive the voting materials in time to ensure that you can instruct the depositary to vote your ordinary shares or to withdraw your ordinary shares so that you can vote them yourself. If we do not ask for your instructions, you can still send voting instructions to the depository and the depository may try to carry out those instructions, but it is not required to do so.
Under our Constitution, any resolution to be considered at a meeting of the shareholders shall be decided on a show of hands unless a poll is demanded in accordance with the terms of our Constitution. A poll may be demanded before a vote is taken, or, in the case of a vote taken on a show of hands, immediately before or immediately after, the declaration of the result of the show of hands. Under voting by a show of hands, multiple “yes” votes by ADS holders will only count as one “yes” vote and will be negated by a single “no” vote, unless a poll is demanded.
You may be subject to limitations on the transfer of your ADSs and the withdrawal of the underlying ordinary shares.
Your ADSs are transferable on the books of the depositary. However, the depositary may close its books at any time or from time to time when it deems expedient in connection with the performance of its duties. The depositary may refuse to deliver, transfer or register transfers of your ADSs generally when our books or the books of the depositary are closed, or at any time if we or the depositary think it is advisable to do so because of any requirement of law, government or governmental body, or under any provision of the deposit agreement, or for any other reason subject to your right to surrender your ADSs and receive the underlying ordinary shares. Temporary delays in the surrendering of your ADSs and receipt of the underlying ordinary shares may arise because the depositary has closed its transfer books or we have closed our transfer books, the transfer of ordinary shares is blocked to permit voting at a shareholders’ meeting or we are paying a dividend on our ordinary shares. In addition, you may not be able to surrender your ADSs and receive the underlying ordinary shares when you owe money for fees, taxes and similar charges and when it is necessary to prohibit withdrawals in order to comply with any laws or governmental regulations that apply to ADSs or to the withdrawal of ordinary shares or other deposited securities. See “Item 12D —Description of American Depositary Shares.”
Holders of ADSs are not treated as holders of our ordinary shares.
Holders of ADSs are not treated as holders of our ordinary shares, unless they surrender the ADSs to receive the ordinary shares underlying their ADSs in accordance with the deposit agreement and applicable laws and regulations. The depositary is the holder of the ordinary shares underlying the ADSs. Holders of ADSs therefore do not have any rights as holders of our ordinary shares, other than the rights that they have pursuant to the deposit agreement. See “Item 12D —Description of American Depositary Shares.”
ADS holders may not be entitled to a jury trial with respect to claims arising under the deposit agreement, which could result in less favorable outcomes to the plaintiff(s) in any such action.
The deposit agreement governing the ADSs provides that holders and beneficial owners of ADSs, including those holders and owners who acquired ADSs in secondary transactions, irrevocably waive the right to a trial by jury in any legal proceeding arising out of or relating to the deposit agreement or the ADSs, including in respect of claims under federal securities laws, against us or the depositary to the fullest extent permitted by applicable law. If this jury trial waiver provision is prohibited by applicable law, an action could nevertheless proceed under
61
the terms of the deposit agreement with a jury trial. To our knowledge, the enforceability of a jury trial waiver under the federal securities laws has not been finally adjudicated by a federal court. However, we believe that a jury trial waiver provision is generally enforceable under the laws of the State of New York, which govern the deposit agreement, by a court of the State of New York or a federal court, which have non-exclusive jurisdiction over matters arising under the deposit agreement, applying such law. In determining whether to enforce a jury trial waiver provision, New York courts and federal courts will consider whether the visibility of the jury trial waiver provision within the agreement is sufficiently prominent such that a party has knowingly waived any right to trial by jury. We believe that this is the case with respect to the deposit agreement and the ADSs. In addition, New York courts will not enforce a jury trial waiver provision in order to bar a viable setoff or counterclaim sounding in fraud or one which is based upon a creditor’s negligence in failing to liquidate collateral upon a guarantor’s demand, or in the case of an intentional tort claim (as opposed to a contract dispute), none of which we believe are applicable in the case of the deposit agreement or the ADSs.
No condition, stipulation or provision of the deposit agreement or ADSs serves as a waiver by any holder or beneficial owner of ADSs or by us or the depositary of compliance with any provision of the federal securities laws. If you or any other holder or beneficial owner of ADSs brings a claim against us or the depositary in connection with such matters, you or such other holder or beneficial owner may not be entitled to a jury trial with respect to such claims, which may have the effect of limiting and discouraging lawsuits against us and/or the depositary. If a lawsuit is brought against us and/or the depositary under the deposit agreement, it may be heard only by a judge or justice of the applicable trial court, which would be conducted according to different civil procedures and may result in different outcomes than a trial by jury would have had, including results that could be less favorable to the plaintiff(s) in any such action, depending on, among other things, the nature of the claims, the judge or justice hearing such claims, and the venue of the hearing.
As the jury trial waiver relates to claims arising out of or relating to the ADSs or the deposit agreement, we believe that the waiver would likely continue to apply to ADS holders or beneficial owners who withdraw the ordinary shares from the ADS facility with respect to claims arising before the cancellation of the ADSs and the withdrawal of the ordinary shares, and the waiver would likely not apply to ADS holders or beneficial owners who subsequently withdraw the ordinary shares represented by ADSs from the ADS facility with respect to claims arising after the withdrawal. However, to our knowledge, there has been no case law on the applicability of the jury trial waiver to ADS holders or beneficial owners who withdraw the ordinary shares represented by the ADSs from the ADS facility.
We currently report our financial results under IFRS, which differs in certain significant respect from U.S. generally accepted accounting principles, or U.S. GAAP.
Currently we report our financial statements under IFRS. There have been and there may in the future be certain significant differences between IFRS and U.S. GAAP, including differences related to intangible assets, share-based compensation expense, and income tax. As a result, our financial information and reported earnings for historical or future periods could be significantly different if they were prepared in accordance with U.S. GAAP. In addition, we do not intend to provide a reconciliation between IFRS and U.S. GAAP unless it is required under applicable law. As a result, you may not be able to meaningfully compare our financial statements under IFRS with those companies that prepare financial statements under U.S. GAAP.
As a foreign private issuer, we are exempt from a number of rules under the U.S. securities laws and are permitted to file less information with the SEC than a U.S. company.
We are a foreign private issuer, as defined in the SEC’s rules and regulations and, consequently, we are not subject to all of the disclosure requirements applicable to public companies organized within the United States. For example, we are exempt from certain rules under the Securities Exchange Act of 1934, as amended, or the Exchange Act, that regulate disclosure obligations and procedural requirements related to the solicitation of proxies, consents or authorizations applicable to a security registered under the Exchange Act, including the U.S. proxy rules under Section 14 of the Exchange Act. In addition, our senior management and directors are exempt from the reporting and “short-swing” profit recovery provisions of Section 16 of the Exchange Act and related rules with respect to their purchases and sales of our securities. Moreover, while we currently make annual and semi-annual filings with respect to our listing on the ASX and expect to file financial reports on an annual and semi-annual basis, we will not be required to file periodic reports and financial statements with the SEC as frequently or as promptly as U.S. public companies and will not be required to file quarterly reports on Form 10-Q or current reports on Form 8-K under the Exchange Act. In addition, foreign private issuers are not required to file their annual report on Form 20-F until four months after the end of each fiscal year. Accordingly, there may
62
be less publicly available information concerning our company than there would be if we were not a foreign private issuer.
As a foreign private issuer, we are permitted to adopt certain home country practices in relation to corporate governance matters that differ significantly from Nasdaq corporate governance listing standards and these practices may afford less protection to shareholders than they would enjoy if we complied fully with Nasdaq corporate governance listing standards.
As a foreign private issuer listed on Nasdaq, we are subject to its corporate governance listing standards. However, Nasdaq rules permit foreign private issuers to follow the corporate governance practices of their home country. Some corporate governance practices in Australia may differ from Nasdaq corporate governance listing standards. For example, we could include non-independent directors as members of our Remuneration and Nomination committees, and our independent directors may not necessarily hold regularly scheduled meetings at which only independent members of the board of directors are present. Currently, we follow home country practice to the maximum extent possible. Therefore, our shareholders may be afforded less protection than they otherwise would have under corporate governance listing standards applicable to U.S. domestic issuers. For an overview of our corporate governance practices, see “Item 16G—Corporate Governance.”
We may lose our foreign private issuer status in the future, which could result in significant additional cost and expense.
While we currently qualify as a foreign private issuer, the determination of foreign private issuer status is made annually on the last business day of an issuer’s most recently completed second fiscal quarter and, accordingly, our next determination will be made on December 31, 2023. In the future, we would lose our foreign private issuer status if we to fail to meet the requirements necessary to maintain our foreign private issuer status as of the relevant determination date. For example, if 50% or more of our securities are held by U.S. residents and more than 50% of our senior management or directors are residents or citizens of the United States, we could lose our foreign private issuer status.
The regulatory and compliance costs to us under U.S. securities laws as a U.S. domestic issuer may be significantly more than costs we incur as a foreign private issuer. If we are not a foreign private issuer, we will be required to file periodic reports and registration statements on U.S. domestic issuer forms with the SEC, which are more detailed and extensive in certain respects than the forms available to a foreign private issuer. We would be required under current SEC rules to prepare our financial statements in accordance with U.S. GAAP rather than IFRS, and modify certain of our policies to comply with corporate governance practices required of U.S. domestic issuers. Such conversion of our financial statements to U.S. GAAP would involve significant time and cost. In addition, we may lose our ability to rely upon exemptions from certain corporate governance requirements on U.S. stock exchanges that are available to foreign private issuers such as the ones described above and exemptions from procedural requirements related to the solicitation of proxies.
We are an “emerging growth company” under the JOBS Act and will be able to avail ourselves of reduced disclosure requirements applicable to emerging growth companies, which could make our ordinary shares ADSs less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act. For as long as we continue to be an emerging growth company, we may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not “emerging growth companies,” including not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act, and if we were to lose our foreign private issuer status, -exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved.
We cannot predict if investors will find the ordinary shares or ADSs less attractive because we may rely on these exemptions. If some investors find the ordinary shares or ADSs less attractive as a result, there may be a less active trading market for the ordinary shares or ADSs and the price of the ordinary shares or ADSs may be more volatile. We may take advantage of these exemptions until such time that we are no longer an emerging growth company. We would cease to be an emerging growth company upon the earliest to occur of (i) the last day of the fiscal year in which we have more than US$1.235 billion in annual revenue; (ii) the last day of the fiscal year in which we qualify as a “large accelerated filer”; (iii) the date on which we have, during the previous three-year period, issued more than US$1.0 billion in non-convertible debt securities; and (iv)June 30, 2026.
63
It is likely that we will be classified as a passive foreign investment company, which could result in adverse U.S. federal income tax consequences for U.S. holders.
In general, a non-U.S. company will be considered a passive foreign investment company, or PFIC, for U.S. federal income tax purposes for any taxable year in which (1) 75% or more of its gross income consists of passive income or (2) 50% or more of the average quarterly value of its assets is attributable to assets that produce, or are held for the production of, passive income. For purposes of these tests, passive income generally includes dividends, interest, gains from the sale or exchange of investment property and certain rents and royalties. In addition, for purposes of the above calculations, a non-U.S. corporation that directly or indirectly owns at least 25% by value of the shares of another corporation is treated as if it held its proportionate share of the assets and received directly its proportionate share of the income of such other corporation.
There can be no assurance that we will not be considered a PFIC in any past, current or future taxable year, and based on the nature and composition of our income, assets, activities and market capitalization, it is likely that we will be a PFIC for our taxable year ended June 30, 2023, and in future taxable years. However, our PFIC status is based on an annual determination and may change from year to year. Our status as a PFIC will depend on the composition of our income and the composition and value of our assets, which may be volatile, from time to time. Our status may also depend, in part, on how quickly we utilize the cash we raise in any offering of our securities. Our U.S. counsel expresses no opinion regarding our conclusions or our expectations regarding our PFIC status.
If we are a PFIC for any taxable year during which a U.S. holder (as defined below in the section titled “Item 10E – Taxation”) holds ADSs, the U.S. holder may be subject to adverse tax consequences regardless of whether we continue to qualify as a PFIC, including ineligibility for any preferred tax rates on capital gains or on actual or deemed dividends, interest charges on certain taxes treated as deferred, and additional reporting requirements. We will continue to be treated as a PFIC with respect to such U.S. holder in all succeeding years during which the U.S. holder owns the ADSs, regardless of whether we continue to meet the PFIC test described above, unless the U.S. holder makes a valid and timely qualified electing fund (QEF) or mark-to-market election, or makes a deemed sale election once we cease to be a PFIC; however, we do not currently intend to provide the information necessary for a U.S. holder to make a QEF election. For further discussion of the PFIC rules and the adverse U.S. federal income tax consequences in the event we are classified as a PFIC, see “Item 10E—Taxation.”
If a United States person is treated as owning at least 10% of our ordinary shares, such holder may be subject to adverse U.S. federal income tax consequences.
If a U.S. holder is treated as owning, (directly, indirectly or constructively,) at least 10% of the value or voting power of our ordinary shares or ADSs, such U.S. holder may be treated, for U.S. federal income tax purposes, as a “United States shareholder” with respect to each “controlled foreign corporation” in our group, if any. Because our group includes a U.S. subsidiary (Opthea US Inc.), certain of our current and future non-U.S. subsidiaries will be treated as controlled corporations, regardless of whether we are treated as a controlled foreign corporation. A United States shareholder of a controlled foreign corporation may be required to annually report and include in its U.S. taxable income its pro-rata share of “Subpart F income,” “global intangible low-taxed income” and investments in U.S. property by controlled foreign corporations, regardless of whether we make any distributions. An individual that is a United States shareholder with respect to a controlled foreign corporation generally would not be allowed certain tax deductions or foreign tax credits that would be allowed to a United States shareholder that is a U.S. corporation. We cannot provide any assurances that we will furnish to any United States shareholder information that may be necessary to comply with the reporting and payment obligations described above. Failure to comply with such obligations may subject a United States shareholder to significant monetary penalties and stall the beginning of the statute of limitations period for relevant U.S. federal income tax returns. U.S. holders should consult their tax advisors regarding the potential application of these rules to their investment in the ADSs.
Future changes to tax laws could materially adversely affect our company and reduce net returns to our shareholders.
Our tax treatment is subject to the enactment of, or changes in, tax laws, regulations and treaties, or the interpretation thereof, tax policy initiatives and reforms under consideration and the practices of tax authorities in jurisdictions in which we operate, including those related to the Organization for Economic Co-Operation and Development’s Base Erosion and Profit Shifting Project, the European Commission’s state aid investigations and other initiatives. Such changes may include (but are not limited to) the taxation of operating income, investment income, dividends received or (in the specific context of withholding tax) dividends paid. We are unable to predict what tax reform may be proposed or enacted in the future or what effect such changes would have on our business,
64
but such changes, to the extent they are brought into tax legislation, regulations, policies or practices, could affect our financial position and overall or effective tax rates in the future in countries where we have operations, reduce post-tax returns to our shareholders, and increase the complexity, burden and cost of tax compliance.
Tax authorities may disagree with our position and conclusions regarding certain tax positions, resulting in unanticipated costs, taxes or non-realization of expected benefits.
A tax authority may disagree with tax positions that we have taken, which could result in increased tax liabilities. For example, the U.S. Internal Revenue Service or another tax authority could challenge our allocation of income by tax jurisdiction and the amounts paid between our affiliated companies pursuant to our intercompany arrangement and transfer pricing policies, including amounts paid with respect to our intellectual property development. Similarly, a tax authority could assert that we are subject to tax in a jurisdiction where we believe we have not established a taxable connection, often referred to as a “permanent establishment” under international tax treaties, and such an assertion, if successful, could increase our expected tax liability in one or more jurisdictions. A tax authority may take the position that material income tax liabilities, interest and penalties are payable by us, in which case, we expect that we might contest such assessment. Contesting such an assessment may be lengthy and costly and if we were unsuccessful in disputing the assessment, the implications could increase our anticipated effective tax rate, where applicable.
General Risk Factors
We may be subject to securities litigation, which is expensive and could divert management’s attention.
The market price of the ordinary shares or ADSs may be volatile and, in the past, companies that have experienced volatility in the market price of their stock have been subject to securities class action litigation. We may be the target of this type of litigation in the future. Securities litigation against us could result in substantial costs and divert our management’s attention from other business concerns, which could seriously harm our business.
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, the price of the ADSs and their trading volume could decline.
The trading market for the ADSs depends in part on the research and reports that securities or industry analysts publish about us or our business. As a public listed company in Australia since 1985, our equity securities are currently subject to coverage by a number of analysts. If fewer securities or industry analysts cover our company, the trading price for the ADSs could be negatively impacted. If one or more of the analysts who covers us downgrades our equity securities or publishes incorrect or unfavorable research about our business, the price of the ADSs would likely decline. If one or more of these analysts ceases coverage of our company or fails to publish reports on us regularly, or downgrades our securities, demand for the ADSs could decrease, which could cause the price of the ADSs or their trading volume to decline.
If our information technology systems or data, or those of our third-parties, upon whom we rely, are or were compromised, we could experience adverse consequences resulting from such compromise, including but not limited to regulatory investigations or actions; litigation; fines and penalties; disruptions of our business operations' reputational harm' loss of revenue or profits; loss of customers or sales; and other adverse consequences.
In the ordinary course of our business, we process confidential sensitive, and/or proprietary information, including intellectual property, business information, personal data, and health information (collectively, sensitive information).
Cyber-attacks, malicious internet-based activity, online and offline fraud, and other similar activities threaten the confidentiality, integrity, and availability of our sensitive information and information technology systems, and those of the third parties upon which we rely. Such threats are prevalent and continue to rise, are increasingly difficult to detect, and come from a variety of sources, including traditional computer "hackers," threat actors, "hacktivists," organized criminal threat actors, personnel (such as through theft or misuse), sophisticated nation states, and nation-state-supported actors. Some actors now engage and are expected to continue to engage in cyber-attacks, including without limitations nation-state actors for geopolitical reasons and in conjunction with military conflicts and defense activities.
65
We and the third parties upon which we rely are subject to a variety of evolving threats, including but not limited to social-engineering attacks (including through phishing attacks), malicious code (such as viruses and worms), malware (including as a result of advanced persistent threat intrusions), denial-of-service attacks, credential stuffing attacks, credential harvesting, personnel misconduct or error, ransomware attacks, supply-chain attacks, software bugs, server malfunctions, software or hardware failures, loss of data or other information technology assets, adware, telecommunications failures, earthquakes, fires, floods and other similar threats. In particular, severe ransomware attacks are becoming increasingly prevalent and can lead to significant interruptions in our operations, loss of sensitive data and income, reputational harm, and diversion of funds. Extortion payments may alleviate the negative impact of a ransomware attack, but we may be unwilling or unable to make such payments due to, for example, applicable laws or regulations prohibiting such payments.
Remote work has become more common and has increased risks to our information technology systems and data, as more of our employees utilize network connections, computers and devices outside our premises or network, including working at home, while in transit and in public locations.
Future or past business transactions (such as acquisitions or integrations) could expose us to additional cyber security risks and vulnerabilities, as our systems could be negatively affected by vulnerabilities present in acquired or integrated entities' systems and technologies. Furthermore, we may discover security issues that were not found during due diligence of such acquired or integrated entities, as it may be difficult to integrate companies into our information technology environment and security program.
We rely on third-party service providers and technologies to operate critical business systems to process sensitive information in a variety of contexts, including, without limitations, cloud-based infrastructure, data center facilities, encryption and authentication technology, employee email, and other functions. Our ability to monitor these third parties' information security practices is limited, and these third parties may not have adequate information security measures in place. If our third-party service providers experience a security incident or other interruption, we could experience adverse consequences. While we may be entitled to damages if our third-party service providers fail to satisfy their privacy or security-related obligations to us, any award may be insufficient to cover our damages, or we may be unable to recover such award.
In addition, supply-chain attacks have increased in frequency and severity, and we cannot guarantee that third parties' infrastructure in our supply chain or our third-party partners' supply chains have not been compromised.
Any of the previously identified or similar threats could cause a security incident or other interruption that could result in unauthorized, unlawful, or accidental acquisition, modification, destruction, loss, alteration, encryption, disclosure of, or access to our sensitive information of our information technology systems, or those of the third parties upon whom we rely. A security incident or other interruptions could disrupt our ability (and that of third parties upon whom we rely) to develop and provide our products. For example, the loss of clinical trial data from completed, ongoing or planned clinical trials for our product candidates could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data.
We may expend significant resources or modify our business activities (including our clinical trial activities) to try to protect against security incidents. Certain data privacy and security obligations may require us to implement and maintain specific security measures or industry-standard or reasonable security measures to protect our information technology systems and sensitive information.
While we have implemented security measures designed to protect against security incidents, there can be no assurance that these measures will be effective.
We take steps to detect and remediate vulnerabilities, but we may not be able to detect and remediate all vulnerabilities because the threats and techniques used to exploit the vulnerability change frequently and are often sophisticated in nature. Therefore, such vulnerabilities could be exploited but may not be detected until after a security incident has occurred. These vulnerabilities pose material risks to our business. Further, we may experience delays in developing and deploying remedial measures designed to address any such identified vulnerabilities.
Applicable data privacy and security obligations may require us to notify relevant stakeholders of security incidents. Such disclosures are costly, and the disclosure or the failure to comply with such requirements could lead to adverse consequences.
66
If we (or a third party upon whom we rely) experience a security incident or are perceived to have experienced a security incident, we may experience adverse consequences, such as government enforcement actions (for example, investigations, fines, penalties, audits, and inspections); additional reporting requirements and/or oversight; restrictions on processing sensitive information (including personal data); litigation (including class claims); indemnification obligations; negative publicity; reputational harm; monetary fund diversions; interruptions in our operations (including availability of data); financial loss; and other similar harms. Security incidents and attendant consequences may cause customers to stop using our products, deter new customers from using our products, and negatively impact our ability to grow and operate our business.
Our contracts may not contain limitations of liability, and even where they do, there can be no assurance that limitations of liability in our contracts are sufficient to protect us from liabilities, damages, or claims related to our data privacy and security obligations. We cannot be sure that our insurance coverage will be adequate or sufficient to protect us from or to mitigate liabilities arising out of our privacy and security practices, that such coverage will continue to be available on commercially reasonable terms or at all, or that such coverage will pay future claims.
In addition to experiencing a security incident, third parties may gather, collect, or infer sensitive information about us from public sources, data brokers, or other means that reveals competitively sensitive details about our organization and could be used to undermine our competitive advantage or market position.
Our insurance policies are expensive and only protect us from some business risks, leaving us exposed to significant uninsured liabilities.
We do not carry insurance for all categories of risk that our business may encounter. We believe that we maintain insurance customary for businesses of our size and type, including clinical trial liability insurance. However, there are types of losses we may incur that cannot be insured against or that we believe are not economically reasonable to insure. Moreover, any loss incurred could exceed policy limits and policy payments made to us may not be made on a timely basis. Such losses could negatively affect our business prospects, results of operations, cash flows and financial condition. We do not know if our current levels of coverage are adequate or if we will be able to obtain insurance with adequate levels of coverage in the future, if at all. Any significant uninsured liability may require us to pay substantial amounts, which could negatively impact our financial position and results of operations.
We have identified a material weakness in our internal control over financial reporting and may identify additional material weaknesses in the future or fail to implement and maintain an effective system of internal control over financial reporting, which may result in us being unable to accurately report our results of operations, meet our reporting obligations or prevent fraud, and investor confidence in our company and the market price of the ADSs may be negatively impacted.
Section 404(a) of the Sarbanes-Oxley Act of 2002, or the Sarbanes-Oxley Act, requires that our management assess and report annually on the effectiveness of our internal controls over financial reporting and identify any material weaknesses in our internal controls over financial reporting. We identified a material weakness in our internal controls over financial reporting as of June 30, 2023 because we did not design and maintain effective controls in relation to accounting for non-routine transactions as the control failed to detect and prevent the incorrect remeasurement of the fair value of our Funding Agreement liability following changes in certain assumptions relating to the timing of our repayments to Ocelot under the Funding Agreement. Although we are taking certain measures to remediate this material weakness, this material weakness will not be considered remediated until management completes the design and implementation of the remediation measures and the controls operate for a sufficient period of time and management concludes, through testing, that these controls are effective. If we are unable to remediate this material weakness or if in the future, we fail to maintain effective internal controls, as such standards are modified, supplemented or amended from time to time, we may not be able to ensure that we can conclude on an ongoing basis that we have effective internal controls over financial reporting in accordance with Section 404(a).
Section 404(b) also requires an attestation report on the effectiveness of internal control over financial reporting be provided by our independent registered public accounting firm beginning with our annual report following the date on which we are no longer an “emerging growth company”, which may be up to five fiscal years from the initial public offering of our ADSs. If we are unable to attest to the effectiveness of our internal control over financial reporting, we could lose investor confidence in the accuracy and completeness of our financial reports, and the market price of our ordinary shares and ADSs could decline. Failure to maintain effective
67
internal control over financial reporting could also restrict our future access to the capital markets and subject each of us, our directors and our officers to both significant monetary and criminal liability. In addition, changing laws, regulations and standards relating to corporate governance and public disclosure are creating uncertainty for public companies, increasing legal and financial compliance costs and making some activities more time consuming. These laws, regulations and standards are subject to varying interpretations, in many cases due to their lack of specificity, and, as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies. This could result in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices. We intend to invest resources to comply with evolving laws, regulations and standards, and this investment may result in increased general and administrative expense and a diversion of management’s time and attention from revenue generating activities to compliance activities. If our efforts to comply with new laws, regulations and standards differ from the activities intended by regulatory or governing bodies due to ambiguities related to their application and practice, regulatory authorities may initiate legal proceedings against us and our business, financial position, results and prospects may be adversely affected.
The presence of material weaknesses could result in financial statement errors which, in turn, could lead to errors in our financial reports or delays in our financial reporting, which could require us to restate our operating results or result in our auditors issuing a qualified audit report. In order to establish and maintain effective disclosure controls and procedures and internal controls over financial reporting, we will need to expend significant resources and provide significant management oversight. Developing, implementing and testing changes to our internal controls may require specific compliance training of our directors and employees, entail substantial costs in order to modify our existing accounting systems, take a significant period of time to complete and divert management’s attention from other business concerns. These changes may not, however, be effective in establishing and maintaining adequate internal controls.
If either we are unable to remediate the material weakness that exists as of June 30, 2023 or in the future conclude that we have ineffective internal controls over financial reporting or when applicable, our independent auditors are unwilling or unable to provide us with an unqualified report on the effectiveness of our internal controls over financial reporting as required by Section 404(b) of the Sarbanes-Oxley Act, investors may lose confidence in our operating results, the price of the ADSs could decline and we may be subject to litigation or regulatory enforcement actions. In addition, if we are unable to meet the requirements of Section 404 of the Sarbanes-Oxley Act, we may not be able to remain listed on Nasdaq.
Item 4. Information on the Company
4A. History and Development of Opthea Limited
We were incorporated under the laws of Australia in 1984 under the name Circadian Technologies Limited. In 1985, we completed an initial public offering of our ordinary shares and the listing of our ordinary shares on the Australian Securities Exchange, or the ASX. In December 2015, we changed the name of our company to Opthea Limited. Our headquarters and registered offices are located at Suite 0403, Level 4, 650 Chapel Street, South Yarra, VIC 3141, Australia. Our telephone number is +61 3 9826 0399. Our agent for service of process in the United States is Corporation Service Company, located at 1180 Avenue of the Americas, Suite 210, New York, NY 10036. Our website address is www.opthea.com. The reference to our website is an inactive textual reference only and information contained in, or that can be assessed through, our website is not part of this annual report. The SEC maintains a website (www.sec.gov) that contains reports, proxy and information statements and other information regarding registrants, such as us, that file electronically with the SEC.
For a description of our principal capital expenditures and divestitures for the three years ended June 30, 2023, and for those currently in progress, see Item 5 “Operating and Financial Review and Prospects”.
4B. Business Overview
We are a clinical-stage biopharmaceutical company developing a novel therapy for the treatment of highly prevalent and progressive retinal diseases. We are developing our Phase 3-product candidate, Sozinibercept (OPT-302), a biologic designed to inhibit VEGF-C and VEGF-D, to complement VEGF-A inhibitors for the treatment of ophthalmic diseases. Anti-VEGF-A therapies represent the standard of care for wet age-related macular degeneration, or AMD, and other retinal diseases; however, there remains a significant unmet medical need as many patients do not adequately respond to these treatments. As the only biologic inhibitor of VEGF-C and VEGF-D in clinical development for ophthalmology, Sozinibercept (OPT-302) differs from standard of care therapies and when administered in combination with a VEGF-A inhibitor, is designed to achieve broader
68
inhibition of the vascular endothelial growth factor, or VEGF, family and target a mechanism of clinical resistance to improve visual acuity. Our lead indication for Sozinibercept (OPT-302) combination therapy is wet AMD, a chronic, progressive disease and the leading cause of vision loss for individuals over the age of 50. In a 366-patient Phase 2b clinical trial for the treatment of wet AMD, 2.0 mg Sozinibercept (OPT-302), in combination with a standard of care anti-VEGF-A therapy, ranibizumab (Lucentis), met the primary endpoint of a statistically significant superior mean gain in visual acuity over ranibizumab monotherapy at week 24. We initiated two pivotal Phase 3 clinical trials in treatment-naive patients with wet AMD to evaluate the efficacy and safety of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapies compared to anti-VEGF-A monotherapy in March 2021. We expect to complete patient recruitment in the COAST study in the first calendar quarter of 2024 and in the ShORe study in the second calendar quarter of 2024. Top-line data is expected to be reported when all patients complete the 52-week treatment period for the primary analysis. In addition to our clinical trials in wet AMD, we have observed evidence of improved clinical outcomes in a Phase 1b/2a clinical trial of Sozinibercept (OPT-302) in combination with another standard of care anti-VEGF-A therapy, aflibercept (Eylea), in patients with treatment-refractory diabetic macular edema, or DME. We retain worldwide rights to develop and commercialize Sozinibercept (OPT-302) for the treatment of wet AMD and DME and believe that the novel treatment mechanism of Sozinibercept (OPT-302) has the potential to provide therapeutic benefit for other progressive eye diseases.
Wet AMD is a rapidly progressing disease with loss of central vision developing over a period of weeks to months in which abnormal new blood vessels form in the back of the eye in a process called choroidal neovascularization, or CNV. These newly formed vessels are highly permeable, leaking exudate leading to fluid accumulation and retinal lesion formation. This, in turn, adversely affects sensory cells in the retina and if left untreated, results in rapid loss of visual acuity.
Wet AMD affects approximately one million people in the United States and 2.5 million people in Europe. The standard of care for wet AMD and other ocular neovascular diseases is the administration of monotherapies that primarily inhibit VEGF-A. These therapeutic agents, which include ranibizumab and aflibercept, prevent VEGF-A molecules from binding to, and activating, VEGF receptors and thereby inhibit the formation and permeability of blood vessels. As the risk of developing wet AMD increases with age, it is predicted that the overall aging of the population will result in a significant increase in the number of wet AMD cases, both in the United States and worldwide. Many wet AMD patients also experience suboptimal clinical responses despite receiving one or both of the leading standard of care treatments ranibizumab and aflibercept, which had combined annual worldwide sales approaching US$13 billion in 2022. In addition, nearly half of wet AMD patients are treated with off-label bevacizumab as a lower cost alternative anti-VEGF-A therapy. As a result, we believe there is a significant and expanding market opportunity for novel therapies that can improve vision in patients with wet AMD, which has the potential to lead to sales greater than the combined annual sales of ranibizumab and aflibercept.
Despite the widespread use and commercial success of VEGF-A inhibitors, at least 45% of wet AMD patients treated with a VEGF-A inhibitor experience some degree of suboptimal clinical response, with a majority of patients failing to achieve 20/40 vision after 12 months of treatment, providing further opportunity for visual acuity improvement. Furthermore, many patients have persistent retinal fluid and insufficient gains in visual acuity to resume routine daily activities such as driving and reading following regular treatment with a VEGF-A inhibitor. In addition, improvements in visual acuity following regular administration of VEGF-A monotherapy are often not sustained with long-term use.
Sozinibercept (OPT-302) is designed to address a deficiency in the treatment paradigm for wet AMD and other retinal diseases, such as DME, by targeting alternate members of the VEGF family, namely VEGF-C and VEGF-D, which are not targeted by current standard of care therapies. VEGF-C and VEGF-D function in parallel with VEGF-A to drive neovascularization and vascular leakage, which are key hallmarks of both wet AMD and DME. In addition, treatment with VEGF-A inhibitors leads to upregulation of VEGF-C and VEGF-D to compensate for VEGF-A inhibition, which may represent an important mechanism of clinical resistance to anti-VEGF-A monotherapy. We are developing Sozinibercept (OPT-302) to be used in combination with standard of care anti-VEGF-A monotherapies to achieve broader inhibition of the VEGF family, with the goal of improving overall efficacy and demonstrating superior vision gains over that which can be achieved by inhibiting VEGF-A alone.
In our completed Phase 2b wet AMD clinical trial, 2.0 mg Sozinibercept (OPT-302) in combination with ranibizumab demonstrated a statistically significant superior mean gain in visual acuity at week 24 compared to patients treated with ranibizumab with a sham injection, which we refer to as ranibizumab monotherapy. The trial
69
was an international, multi-center, double-masked trial in 366 treatment-naive patients with wet AMD. Patients were randomized into three groups and received intravitreal injections every four weeks of either 0.5 mg or 2.0 mg Sozinibercept (OPT-302) in combination with 0.5 mg ranibizumab or 0.5 mg ranibizumab monotherapy. Treatments were administered by intravitreal injections once every four weeks for 20 weeks (six treatments in total). The primary endpoint was the mean change at week 24 in best corrected visual acuity, or BCVA, from baseline on the Early Treatment of Diabetic Retinopathy Study, or ETDRS, standardized eye chart, which we refer to as visual acuity. Patients treated with 2.0 mg Sozinibercept (OPT-302) combination therapy demonstrated a statistically significant improvement in visual acuity compared to patients treated with ranibizumab monotherapy. In the patients that received 2.0 mg Sozinibercept (OPT-302) combination therapy, visual acuity improved at week 24 from baseline by a mean of +14.2 letters compared to +10.8 letters for those treated with ranibizumab monotherapy, a statistically significant benefit of +3.4 letters. Patients that received 2.0 mg Sozinibercept (OPT-302) combination therapy also demonstrated improvements in retinal anatomy which were consistent with the visual acuity gains observed in the trial, including reductions in retinal fluid and lesion size by week 24. In a pre-specified subgroup analysis, greater activity of Sozinibercept (OPT-302) was observed in lesion types that are considered more difficult to treat with anti-VEGF-A therapy. Our clinical experience to date, which includes administration of Sozinibercept (OPT-302) to patients enrolled in our Phase 3 clinical trials, as well as administration of over 1,800 doses of Sozinibercept (OPT-302) to 399 patients with retinal disease in our Phase 1 and Phase 2 clinical trials, indicates that Sozinibercept (OPT-302) intravitreal injections are well tolerated, with the incidence of treatment-emergent adverse events, or TEAEs, comparable to anti-VEGF-A monotherapy in our clinical trials.
In August 2020, we successfully completed End-of-Phase 2 meetings with the U.S. Food and Drug Administration (FDA), and a Scientific Advice meeting with the European Medicines Agency (EMA). The regulatory engagement provided us with guidance on our Phase 3 clinical program for Sozinibercept (OPT-302) in wet AMD and associated manufacturing processes that we believe will support the submission of a Biologics License Application in the U.S. and Marketing Authorization Application in Europe. Further regulatory milestones were achieved during the year, firstly with our successful application to the FDA for an initial Pediatric Study Plan (iPSP) waiver, which was received in March 2021. The receipt of the waiver means that we will not have to conduct an additional study of Sozinibercept (OPT-302) in the pediatric population for use of Sozinibercept (OPT-302) in this U.S. population. Furthermore, in July 2021, the FDA granted Fast Track designation for Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of patients with wet AMD. We believe the FDA’s Fast Track designation acknowledges the significant unmet medical need in the management of wet AMD, and the potential role that Sozinibercept (OPT-302) may have in addressing it. The FDA’s Fast Track Designation for Sozinibercept (OPT-302) offers benefits to expedite the Phase 3 clinical program and subsequent potential approval process, including more frequent communication and meetings with the FDA, and a Rolling Review of completed sections of its BLA.
We initiated two concurrent pivotal Phase 3 clinical trials for the treatment of wet AMD in March 2021. These double-masked, sham-controlled Phase 3 clinical trials enrolled treatment-naive patients and assessed the efficacy and safety of 2.0 mg of Sozinibercept (OPT-302) in combination with ranibizumab (Lucentis) (referred to as the ShORe trial) or aflibercept (Eylea) (referred to as the COAST trial), compared to ranibizumab or aflibercept monotherapy in each respective trial. In addition, to understand the durability of Sozinibercept (OPT-302) treatment effect with less frequent dosing, each trial compared the clinical efficacy of Sozinibercept (OPT-302) administered in combination with the applicable VEGF-A inhibitor on an every four-week and every eight-week dosing regimen. For consistency, the ShORe and COAST Phase 3 trials built upon and maintain key features of our Phase 2b clinical trial of Sozinibercept (OPT-302) combination therapy for the treatment of wet AMD, while evaluating the administration of Sozinibercept (OPT-302) combination therapy over a longer treatment period and in a greater number of patients. The primary endpoint of both trials will be the mean change in visual acuity from baseline at week 52. Patients will continue to be dosed until week 96 to further assess long-term safety at week 100. Since the first patients were treated in our Phase 3 pivotal program in March 2021, we have continued to activate additional clinical trial sites and recruit patients in the U.S., Europe and other regions of the world. In August 2021, the first sites opened enrollment in Canada, and in October 2021 the first sites opened enrollment in Europe and Asia Pacific. In total, we expected to enroll approximately 990 patients into each trial, with patients recruited from more than 20 countries worldwide. Our efforts to advance our Phase 3 clinical trials, including clinical trial activations and trial site engagements, have been challenged in part by the COVID-19 pandemic and administrative delays. In particular, we have incurred and experienced, and may continue to incur and experience in the future, significantly increased costs and delays in connection with the activities conducted by third-party CROs and other third parties to prepare for and progress our Phase 3 clinical trials, including trial site activation and patient enrollment. We continue to expect to complete patient recruitment in the COAST study in the first calendar quarter of 2024 and in the ShORe study in the second calendar quarter of 2024. Top-line data is expected
70
to be reported when all patients complete the 52-week treatment period for the primary analysis. If the results at the completion of the primary efficacy phase at week 52 of the Phase 3 clinical trials are favorable, we intend to submit Biologics License and Marketing Authorization Applications with the FDA and EMA respectively.
In addition to our pivotal Phase 3 clinical trials, we plan to develop a co-formulation of Sozinibercept (OPT-302) with an approved and/or biosimilar anti-VEGF-A therapy designed to achieve VEGF-A, VEGF-C and VEGF-D inhibition following the administration of a single intravitreal injection of the co-formulated product. Sozinibercept (OPT-302) is currently administered as a combination therapy consisting of a sequential injection of Sozinibercept (OPT-302) following intravitreal administration of a VEGF-A inhibitor. We believe that a co-formulated Sozinibercept (OPT-302) and VEGF-A inhibitor product could provide flexibility of treatment options for physicians and reduce the frequency and number of injections for patients. We intend to file an investigative new drug application, or IND, for the co-formulated product in a Phase 1 clinical trial for the treatment of wet AMD.
While we intend to focus our development efforts on seeking commercialization of Sozinibercept (OPT-302) for the treatment of wet AMD, we are also investigating the therapeutic potential of Sozinibercept (OPT-302) for DME. DME is a progressive eye disease and a complication of diabetic retinopathy, or DR, a condition caused by chronically elevated glucose levels in diabetics that damages the retina. DME can cause blurred vision, severe vision loss and blindness. Wet AMD and DME share a similar underlying pathophysiology, including retinal neovascularization and increased vascular permeability, and as a result, VEGF-A inhibitors are also considered the standard of care treatment for DME. Based on its mechanism of action and clinical results to date, we believe that Sozinibercept (OPT-302) also has the potential to deliver therapeutic benefit in DME patients. In our Phase 1b/2a clinical trial of Sozinibercept (OPT-302) in combination with aflibercept in patients with treatment-refractory DME, we observed evidence of improved clinical outcomes following Sozinibercept (OPT-302) combination therapy in this indication.
We also believe that our novel treatment mechanism has the potential to provide therapeutic benefit for other progressive retinal diseases beyond wet AMD and DME. We may further investigate the efficacy of Sozinibercept (OPT-302) to improve clinical outcomes in patients with polypoidal choroidal vasculopathy, or PCV, a form of wet AMD that is highly prevalent in Asian populations and less responsive to anti-VEGF-A therapy than other wet AMD subtypes. Beyond wet AMD and DME, we may explore applications of Sozinibercept (OPT-302) in other retinal diseases in which a VEGF-C or VEGF-D inhibitor could have therapeutic potential, such as retinal vein occlusion, or RVO.
Our Company
We are a public company listed on the Australian Securities Exchange and Nasdaq. We have assembled a team of experts with deep scientific, clinical and business expertise in biotechnology and specifically in neovascular disease. Megan Baldwin, Ph.D., our Chief Executive Officer and Managing Director, has over 20 years of research and development and biopharmaceutical industry experience on neovascularization and therapeutic strategies in ophthalmic indications and cancer. Prior to joining our company, she was a postdoctoral research fellow and an associate market planning manager at Genentech, where she conducted angiogenesis research before joining the anti-angiogenic therapy commercial group. Karen Adams, our Vice President Finance and Company Secretary, was previously the Chief Financial Officer of the Victor Smorgon Group. She has over 20 years’ experience of financial management in board-level positions for private and listed companies in Australia, UK, the US and Ireland. Judith Robertson, our Chief Commercial Officer, was previously Chief Commercial Officer of Eleusis Ltd and Chief Commercial Officer of Aerie Pharmaceuticals. Prior to Aerie, Ms. Robertson was Vice President Immunology and Ophthalmology Global Commercial Strategy Leader at Johnson and Johnson, Janssen Pharmaceutical and Vice President, Ophthalmology Global Business Franchise Head at Novartis (formerly Alcon). In October 2022, Timothy Morris joined Opthea as Chief Financial Officer, based in the US. Mr. Morris has over 25 years of experience as the chief financial officer for public biotechnology companies. We believe that the breadth of experience and successful track record of our senior management, combined with our established relationships with leaders in the industry and medical community, provide us with unique insights into biologic drug development for the treatment of neovascular disease.
71
Our Strategy
Our goal is to become a leader in developing and commercializing therapeutics for the treatment of retinal diseases. The key elements of our strategy are to:
72
Our Pipeline
The following table summarizes the stage of clinical development and status of our product candidate, Sozinibercept (OPT-302):
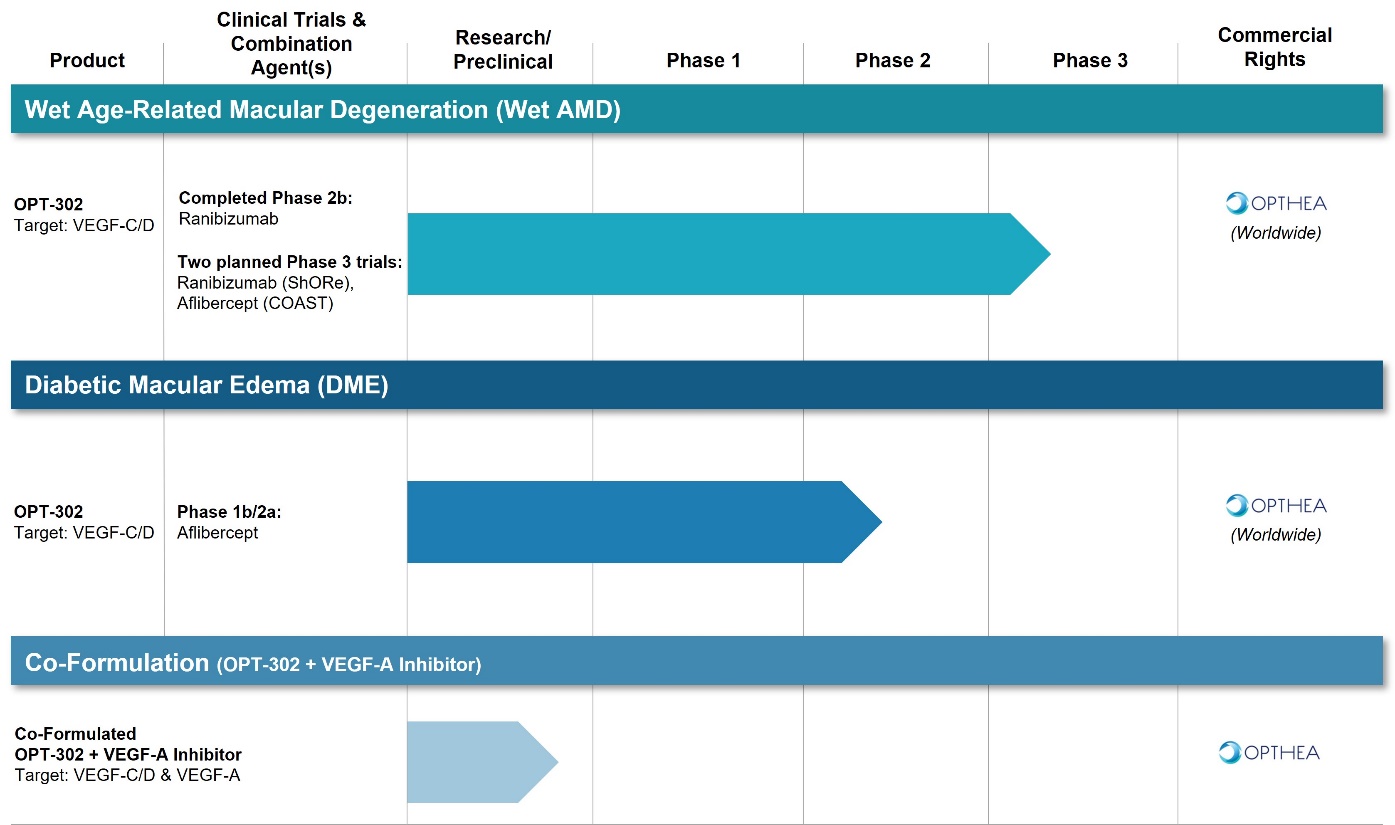
VEGFs in Ocular Diseases
Multiple ophthalmic diseases and conditions, including wet AMD and DME, involve aberrant blood vessel formation and growth, as well as vascular permeability and resulting leakage that contributes to disease progression. In wet AMD, lesions consist of newly formed blood vessels that are typically fragile and leak, leading to the accumulation of fluid in the retinal tissue at the back of the eye. As shown in the figure below, if left untreated, this fluid can cause retinal swelling that disrupts the local architecture and function of sensory cells and neurons in the eye, resulting in vision loss. In patients with DME, high blood glucose levels drive physiological changes resulting in vascular permeability that also result in fluid accumulation, or edema, in the macula, the central region of the retina, and loss of visual acuity.
73
Neovascularization and Vascular Permeability are Key Hallmarks of a Number of Retinal Diseases, Leading to Lesion Formation, Edema and Distortion of the Retina Photoreceptor Layer Causing Loss of Vision
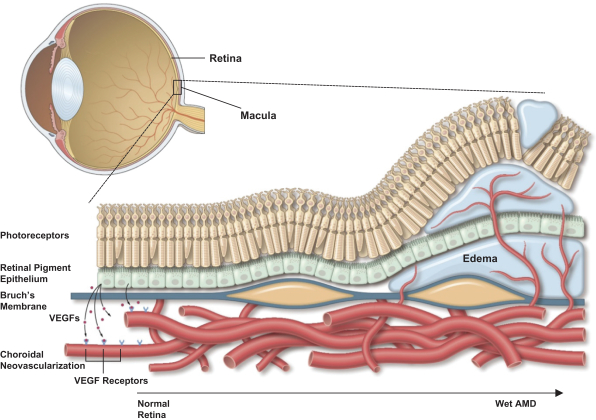
Neovascularization and vascular permeability associated with retinal disease progression are driven by a family of related growth factors known as VEGFs. Members of the VEGF family, including VEGF-A, VEGF-C and VEGF-D, exert their activity by binding and activating VEGF receptors referred to as VEGFR-1, VEGFR-2, and VEGFR-3. Receptor activation triggers signaling pathways that lead to the development of new blood vessels, a process known as angiogenesis, as well as vascular permeability.
74
Members of the VEGF Family of Growth Factors Have Overlapping but Distinct Specificities for VEGFRs
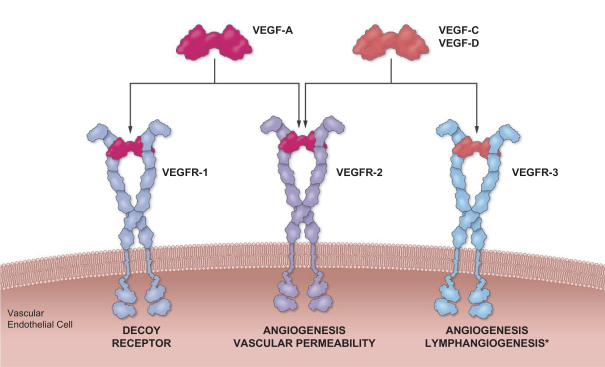
* Lymphangiogenesis refers to the proliferation of lymphatic vessels from pre-existing lymphatic vessels.
VEGF-A
VEGF-A is the most well-characterized member of the VEGF family of growth factors and was the first to be targeted for therapeutic intervention. VEGF-A is a potent growth factor and its relevance in ophthalmic neovascularization has been well-established. Overexpression of VEGF-A in animal models has shown VEGF-A to be a causal factor in the development of neovascularization and vascular permeability, which are key hallmarks in the progression of wet AMD. In wet AMD patients, VEGF-A levels are shown to be elevated in the aqueous humor fluid of the eye. VEGF-A binds to VEGFR-1, which acts to regulate VEGF-A activity, and VEGFR-2, which is a key driver of neovascularization and vascular permeability. Several inhibitors of VEGF-A have been approved to treat a number of neovascular ocular diseases. The two leading ocular VEGF-A inhibitors by revenue, ranibizumab and aflibercept, had combined annual worldwide sales approaching US$13 billion in 2022.
VEGF-C and VEGF-D
VEGF-C and VEGF-D contribute to the development and persistence of neovascular diseases, as evidenced by their elevated levels in multiple pathological conditions. Both VEGF-C and VEGF-D can stimulate neovascularization and VEGF-C can induce permeability by binding to VEGFR-2, independent of VEGF-A activity. Additionally, VEGF-C and VEGF-D bind to VEGFR-3, a receptor that is not activated by VEGF-A, which confers biological activities to both VEGF-C and VEGF-D that are distinct from those of VEGF-A. Activation of VEGFR-3 can stimulate vascular proliferation and modulate vascular permeability, vascular leakage and edema formation. The receptor binding profiles, and the distinct biological activities of VEGF-C and VEGF-D suggest that inhibiting VEGF-C and VEGF-D may have therapeutic potential in ocular diseases by acting independently of, and in tandem with, the activity of VEGF-A inhibitors.
Resistance to VEGF-A Therapies May be Driven by VEGF-C and VEGF-D
Standard of care treatments for wet AMD and DME inhibit VEGF-A activation of its receptors that are typically expressed on vascular endothelium. This can lead to inhibition of blood vessel growth and leakage which can stabilize disease and improve clinical outcomes, including visual acuity, in patients with retinal eye conditions. However, not all patients fully respond to VEGF-A inhibition. A substantial proportion of patients with wet AMD and DME have further opportunity for visual acuity gain and/or a need for resolution of persistent retinal fluid
75
following anti-VEGF-A treatment. Furthermore, gains in visual acuity are often not sustained over the long term, even when anti-VEGF-A therapies are administered regularly. This resistance may occur as anti-VEGF-A monotherapies do not fully address the multifactorial pathogenesis of wet AMD and DME, including having no activity to block VEGF-C and VEGF-D.
VEGF-C and VEGF-D are implicated in the development of resistance to clinical use of anti-VEGF-A therapies. Levels of VEGF-C and/or VEGF-D have been observed to be unregulated in response to anti-VEGF-A therapies, most notably in patients with wet AMD. In a study conducted by third-party researchers in wet AMD patients, a 66% increase in the level of VEGF-C in the aqueous humor fluid in eyes was observed following two monthly doses of the VEGF-A inhibitor, bevacizumab (Avastin). This is illustrated in the figure below.
VEGF-C Levels are Increased in Aqueous Fluid of the Eye in Wet AMD Patients Treated with Monthly Intravitreal Bevacizumab
Study conducted by Cabral et al. (2018)
Upregulation of VEGF-C and VEGF-D can continue to drive signaling through VEGFR-2, even in the presence of a VEGF-A inhibitor, as well as signal through VEGFR-3, both of which may contribute to ongoing angiogenesis and vascular permeability associated with persistent wet AMD. VEGF-C and VEGF-D mediated activation of VEGFR-2 and VEGFR-3, as well as their compensatory elevation following VEGF-A inhibition, may contribute to sub-optimal clinical response to anti-VEGF-A monotherapy. We believe Sozinibercept (OPT-302) in combination with a VEGF-A inhibitor can address a key mechanism of clinical sub-responsiveness to standard of care treatments for serious retinal eye diseases by broad blockade of the VEGF family of growth factors which is not achieved by anti-VEGF-A monotherapies.
Sozinibercept (OPT-302)
We are developing our Phase 3 product candidate, Sozinibercept (OPT-302), a biologic designed to inhibit VEGF-C and VEGF-D, to complement existing VEGF-A inhibitors, for the treatment of ophthalmic diseases, including wet AMD and DME. Anti-VEGF-A therapies represent the standard of care for wet AMD and other retinal diseases. However, there remains a significant unmet need as many patients do not adequately respond to these treatments. As the only biologic inhibitor of VEGF-C and VEGF-D in clinical development for ophthalmology, Sozinibercept (OPT-302) differs from standard of care VEGF-A inhibitors, and in combination with a VEGF-A inhibitor, is designed to address the sub-optimal clinical responses of anti-VEGF-A monotherapies by achieving broader inhibition of the VEGF family to improve visual acuity over standard of care anti-VEGF-A monotherapies.
In 2019, we completed a 366-patient Phase 2b clinical trial of Sozinibercept (OPT-302) in combination with ranibizumab for the treatment of wet AMD, which met the primary endpoint of a statistically significant superior mean gain in visual acuity over ranibizumab monotherapy at week 24. We initiated two pivotal Phase 3 clinical trials in treatment-naive patients with wet AMD to evaluate the efficacy and safety of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy compared to a standard of care monotherapy in March, 2021. We initiated these two pivotal Phase 3 clinical trials in treatment-naive patients with wet AMD to evaluate the efficacy and safety of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy compared to anti-VEGF-A
76
monotherapy. We expect to complete patient recruitment in COAST study in the first calendar quarter of 2024 and in the ShORe study in the second calendar quarter of 2024. Top-line data is expected to be reported when all patients complete the 52-week treatment period for the primary analysis. In addition, in our Phase 1b/2a clinical trial of Sozinibercept (OPT-302) in combination with aflibercept for the treatment of persistent DME, we observed evidence of clinical activity and improvements in visual acuity outcomes compared to aflibercept monotherapy. Sozinibercept (OPT-302) was observed to be well tolerated across Phase 1 and Phase 2 clinical studies in two disease indications following intravitreal administration of over 1,800 doses of Sozinibercept (OPT-302) to 399 patients either as monotherapy or in combination with standard of care VEGF-A inhibitors.
Sozinibercept (OPT-302) Mechanism of Action
We have designed Sozinibercept (OPT-302) to function as a ligand trap, capable of binding and sequestering VEGF-C and VEGF-D, thereby preventing these growth factors from activating VEGFR-2 and VEGFR-3. Sozinibercept (OPT-302) is comprised of the first three extracellular domains of human VEGFR-3, fused to the Fc domain, or the constant fragment of human immunoglobulin G1, or IgG1, as illustrated in the figure below. VEGF-C and VEGF-D function independent of, but in parallel with, VEGF-A to drive neovascularization and vascular leakage, key hallmarks of both wet AMD and DME. In addition, treatment with VEGF-A inhibitors leads to upregulation of VEGF-C and VEGF-D to compensate for VEGF-A inhibition, which may represent an important mechanism of clinical resistance to VEGF-A monotherapy.
Structure of Sozinibercept (OPT-302)
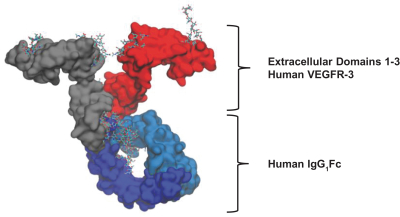
Ligand trap therapeutics that include the receptor-binding domains for other ligands have been approved for a number of indications. One such agent is aflibercept, marketed as Eylea, a ligand trap consisting of extracellular domains of VEGFR-1 and VEGFR-2, which primarily mediates its activity by binding and inhibiting VEGF-A. Aflibercept has marketing approval for the treatment of wet AMD, DME, macular edema secondary to RVO and DR. In rabbits, Sozinibercept (OPT-302) has been shown to have a comparable ocular biodistribution and intravitreal pharmacokinetic profile as aflibercept, with low systemic exposure.
Wet AMD
AMD is a chronic, progressive disease of the macula, a part of the retina containing the greatest concentration of light-sensing cells responsible for detailed, high visual acuity and central vision. The development of AMD is strongly associated with age, affecting up to 40% of individuals over the age of 75. There are two forms of AMD, dry AMD and wet AMD. Dry AMD is the most common form, representing approximately 85% to 90% of all AMD cases. However, dry AMD can develop into wet AMD, and wet AMD accounts for 90% of the severe vision loss associated with the disease.
Wet AMD is a rapidly progressing disease with loss of central vision developing over a period of weeks to months, and is the leading cause of vision loss for individuals over the age of 50. The most common symptoms of wet AMD are loss of central vision, distortion of objects or blurred vision. Peripheral vision usually remains intact. The disease typically affects patients initially in one eye, with a high likelihood of it occurring in the second eye over time. If left untreated, wet AMD can lead to rapid loss of visual acuity and blindness, adversely impacting the patient’s ability to conduct daily activities, such as driving and reading.
77
Wet AMD occurs when new blood vessels in the choroid, or the vascular layer in the eye just under the retina, intrude into the retinal layers and leak fluid. The formation of these new blood vessels is referred to as CNV. These newly formed vessels are highly permeable, which can lead to fluid accumulation and adversely affect sensory cells in the retina.
Wet AMD can be classified as occult or classic, based on the neovascular pattern within lesions. In occult lesions, all of the blood vessels are below the retinal pigment epithelium, or RPE, and the areas on angiograms have a stippled appearance. Classic lesions, by contrast, appear as well-demarcated areas on angiograms due to neovascular blood vessels being located above the RPE. Classic lesions are further subdivided into predominantly classic if 50% or more of the blood vessels are above the RPE, and minimally classic, if greater than 0% and less than 50% of the blood vessels are above the RPE. Classic-containing lesions tend to be the most responsive to anti-VEGF-A therapies.
Classifications of Wet AMD Based on Neovascular Lesion Pattern
There is a further sub-type of wet AMD lesion referred to as retinal angiomatous proliferation, or RAP. RAP lesions are a type of CNV in which neovascularization in the retina protrudes into the sub-retinal space and connects to the choroidal circulation. Between 10% and 21% of wet AMD patients have RAP lesions which are more commonly found in older Caucasian patients. Although there are no therapies specifically approved to treat RAP lesions, patients are typically treated with VEGF-A inhibitors.
PCV is a further sub-type of wet AMD. PCV is an abnormality of inner choroidal vessels that causes dilations in the blood vessels in the retina that resemble polyps, known as polypoidal protrusions. PCV typically does not respond well to VEGF-A inhibitor therapies and many patients are diagnosed with PCV only after they fail to respond to these therapies. There is a high prevalence of PCV in Asian countries, with between 23% and 54% of patients with presumed cases of wet AMD in Japan having PCV. Prevalence rates of between 4% and 10% have been reported in Caucasian patients with presumed cases of wet AMD.
Current Treatments for Wet AMD and Their Limitations
There are five VEGF-A inhibitors approved by the U.S. Food and Drug Administration, or FDA, for the treatment of wet AMD, all of which are administered by regular intravitreal injections as monotherapy. These VEGF-A inhibitors include: the VEGF-A specific antibody ranibizumab (Lucentis®), the antibody fragment brolucizumab (Beovu®), the bispecific antibody faricimab (Vabysmo®) and the VEGFR-based ligand trap aflibercept (Eylea®). The VEGF-A antibody, bevacizumab (Avastin®), an FDA approved therapy for colorectal and other cancers, is also used off-label by many physicians to treat wet AMD, comprising approximately half of anti-VEGF-A injections administered globally. Recent clinical development has focused on maintaining vision gains with a VEGF-A inhibitor whilst reducing the number of injections.
78
VEGF-A inhibitors stabilize loss vision in over 90% of wet AMD patients. However, the effectiveness of these therapies in many patients is limited. Improvements of ≥15 letters of visual acuity typically occur in less than 40% of treated patients. In addition, despite regular treatment with a VEGF-A inhibitor, many patients have insufficient gains in visual acuity to resume routine daily activities such as driving and reading. Chronic vision loss can also occur despite ongoing treatment with an anti-VEGF-A inhibitor. Retrospective and prospective analyses of patients treated with VEGF-A inhibitor therapies for five years have found that, after initial gains in visual function following one year of treatment, many patients then had a gradual decline in visual acuity in subsequent years, resulting in the eventual reversal of the majority of gains. In addition, in clinical settings, up to two-thirds of patients treated with VEGF-A inhibitor therapies continue to have retinal fluid following treatment and approximately 25% experience further vision loss 12 months following treatment. Treatment options are limited for patients who do not respond adequately or experience visual decline despite ongoing therapy with standard of care VEGF-A inhibitors, and typically involve switching treatment from one anti-VEGF-A monotherapy to another with minimal additional visual benefit achieved.
Market Opportunity for the Treatment of Wet AMD
Wet AMD affects approximately 1 million people in the United States and 2.5 million in Europe. As the risk of developing wet AMD increases with age, it is predicted that the overall aging of the population will result in a significant increase in the number of wet AMD cases, both in the United States and worldwide. Many wet AMD patients also experience suboptimal clinical responses despite receiving one or both of the leading standard of care treatments ranibizumab and aflibercept, which had combined annual worldwide sales approaching US$13 billion in 2022. In addition, nearly half of wet AMD patients are treated with off-label bevacizumab as a lower cost alternative anti-VEGF-A therapy. As a result, we believe there is a significant and expanding market opportunity for novel therapies that can improve vision in patients with wet AMD, which has the potential to lead to sales greater than the combined annual sales of ranibizumab and aflibercept.
We believe that Sozinibercept (OPT-302), used in combination with standard of care VEGF-A inhibitors, can address the significant unmet need for wet AMD patients by providing improved outcomes over that which can be achieved by inhibiting VEGF-A alone.
Phase 1/2a Clinical Trial Results in Wet AMD
In 2017, we completed a Phase 1/2a clinical trial of Sozinibercept (OPT-302) for the treatment of wet AMD patients under an investigational new drug, or IND, application accepted by the FDA in 2015. The trial was divided into two parts. Part 1 was a Phase 1 first-in-human 20-patient dose-escalation study in which Sozinibercept (OPT-302) was administered at three escalating doses (0.3 mg, 1.0 mg or 2.0 mg), either in combination with 0.5 mg ranibizumab or alone as a 2.0 mg monotherapy, once every four weeks for a total of three doses, with a follow-up visit at week 12. Part 2 was a Phase 2a 31-patient dose-expansion trial in which 2.0 mg Sozinibercept (OPT-302) was administered in combination with ranibizumab or as a monotherapy once every four weeks for a total of three doses, with a follow-up visit at week 12. Of the 51 patients dosed, 49% were treatment naive and 51% were previously treated with anti-VEGF-A therapy. The previously-treated patients received an average of 17 prior treatments, equating to prior treatment over an average of 1.3 years, of an intravitreal VEGF-A inhibitor prior to enrolling in the trial. The Phase 1/2a trial was conducted at 14 trial sites in the United States.
Sozinibercept (OPT-302) was well tolerated up to the highest dose tested both in combination with ranibizumab and as a monotherapy. No dose-limiting toxicities, or DLTs, were observed and the maximum tolerated dose, or MTD, was not reached. There were no treatment-related serious adverse events, or SAEs. The most common TEAEs were conjunctival hemorrhage, eye pain and corneal inflammation mainly related to the intravitreal injection procedure. TEAEs did not lead to permanent discontinuation of the study for any patient. The pharmacokinetic profile of Sozinibercept (OPT-302) was similar in the absence or presence of ranibizumab, and there was no evidence of Sozinibercept (OPT-302) immunogenicity in this clinical trial.
Although the focus of this trial was safety, we also observed preliminary signals of clinical benefit. Improvements in visual acuity were observed in treatment-naive patients as well as patients previously treated with anti-VEGF-A therapy, as measured by the number of letters that could be read on a standard eye chart following Sozinibercept (OPT-302) monotherapy and Sozinibercept (OPT-302) combination therapy. As illustrated in the figure below, after 12 weeks, in patients across all dose groups, the mean change in visual acuity from baseline increased by +10.8 letters in treatment-naive patients and +4.9 letters in previously treated patients who received Sozinibercept (OPT-302) combination therapy.
79
Mean Change in Visual Acuity from Baseline to Week 12 in Treatment-Naive and Previously-Treated Wet AMD Patients Administered Sozinibercept (OPT-302) in Combination with Ranibizumab
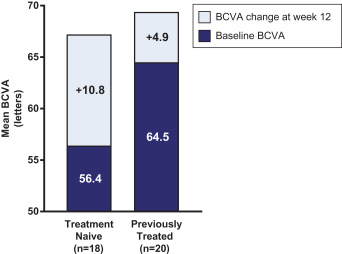
In addition, improvements in visual acuity following treatment with Sozinibercept (OPT-302) combination therapy were consistent with anatomical outcomes, such as reductions in intraretinal and subretinal fluid. Retinal thickness, which is assessed by spectral domain optical coherence tomography, or SD-OCT, using a standard criterion called central subfield thickness, or CST, was also reduced following Sozinibercept (OPT-302) combination therapy.
Phase 2b Clinical Trial Results in Wet AMD
Based on the positive results of the Phase 1/2a trial, we completed an international, multi-center, double-masked Phase 2b clinical trial of Sozinibercept (OPT-302) in combination with ranibizumab in a total of 366 treatment-naive patients in August 2019. As illustrated in the figure below, patients were randomized into three groups to receive either 0.5 mg or 2.0 mg Sozinibercept (OPT-302) with 0.5 mg ranibizumab or ranibizumab monotherapy, which included a sham injection. A sham intravitreal injection involves pressing a syringe hub against the surface of the eye to mimic an intravitreal injection so that the patient remains masked to the treatment group to which they have been randomized. Administration was by intravitreal injection once every four weeks for 20 weeks (six treatments in total). The primary endpoint of the clinical trial was the mean change in BCVA from baseline on the ETDRS standardized eye chart at week 24. Secondary outcome measures included the proportion of patients gaining ≥15 letters in BCVA, changes in retinal thickness, change in intraretinal and subretinal fluid and proportion of patients losing ≥15 letters in BCVA.
Design of the Phase 2b Clinical Trial of Sozinibercept (OPT-302) with Ranibizumab in Wet AMD

As illustrated in the figure below, 366 treatment-naive patients were randomized 1:1:1 to each of the three treatment groups. We used data from the 362 patients who had a baseline assessment of their visual acuity and completed at least one post-dose visit as the modified intent to treat population, or mITT, in all analyses. Key inclusion criteria for patients in the trial included CNV classified as either occult, minimally classic or
80
predominantly classic, and BCVA on the ETDRS standardized eye chart of ≥25 and ≥60 letters. The Phase 2b trial was conducted at 109 trial sites in the United States, Europe, Israel and the United Kingdom.
Patient Distribution in the Phase 2b Clinical Trial in Wet AMD
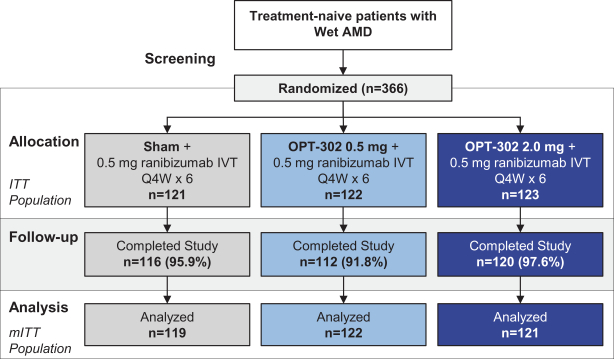
Q4W refers to administration every four weeks. IVT refers to administration by intravitreal injection.
In presentations of statistical results in this annual report, a p-value is a measure of statistical significance of the observed results, or the probability that the observed results were achieved purely by chance. By convention, a p-value of 0.05 or lower is commonly considered statistically significant. The FDA and comparable non-U.S. regulatory authorities utilize the reported statistical measures when evaluating the results of a clinical trial, including statistical significance as measured by p-value, to evaluate the reported evidence of a drug product’s safety and efficacy.
Improvements in Visual Acuity
The Phase 2b clinical trial met the primary endpoint, demonstrating a statistically significant superior mean gain in visual acuity at week 24 compared to baseline in the 2.0 mg Sozinibercept (OPT-302) combination therapy group compared to the ranibizumab monotherapy group. The figure below illustrates patients in the 2.0 mg Sozinibercept (OPT-302) combination therapy group had a mean visual acuity improvement of +14.22 letters relative to baseline, compared to +10.84 letters for those treated with ranibizumab monotherapy at week 24 (p=0.0107). This represents a statistically significant benefit of +3.4 letters and a greater than 30% relative improvement in vision outcomes in the Sozinibercept (OPT-302) combination group compared to ranibizumab monotherapy. Evidence of improved visual acuity was observed beginning as early as week 8 and continued throughout the course of the trial to week 24. Mean visual acuity in the lower dose 0.5 mg Sozinibercept (OPT-302) combination therapy group was not significantly different from the ranibizumab monotherapy group. However, evidence of a dose response was observed between the 2.0 mg and 0.5 mg Sozinibercept (OPT-302) combination therapy groups on several anatomical outcomes, such as retinal thickness and the proportion of patients with intraretinal and subretinal fluid at week 24.
81
Mean Change in Visual Acuity from Baseline to Week 24 in Treatment-Naive Wet AMD Patients Administered Ranibizumab Monotherapy or Sozinibercept (OPT-302) Combination Therapy
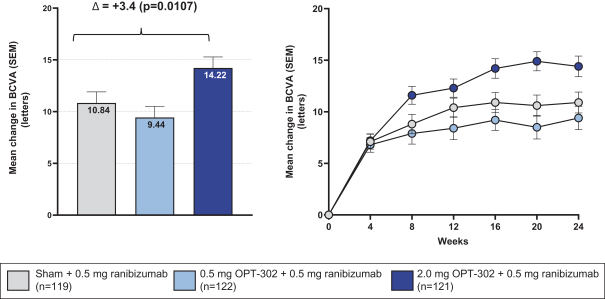
Error bars shown in all figures represent the SEM, or the standard error of the mean.
Secondary outcome measurements from the trial were also supportive of the primary endpoint. As illustrated in the figure below, we observed a greater proportion of patients gaining 15, 10 and 5 letters in the 2.0 mg Sozinibercept (OPT-302) combination therapy group compared to the ranibizumab monotherapy group. In addition, the proportion of patients losing 15, 10 and 5 letters was lower in the 2.0 mg Sozinibercept (OPT-302) combination therapy group compared to the ranibizumab monotherapy group. The Phase 2b trial was not designed for statistical significance on secondary and exploratory endpoints and was not powered to detect statistically significant differences in secondary outcome measurements.
82
Greater Proportion of Patients Gaining, and Fewer Patients Losing, 15, 10 and 5 Letters with Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
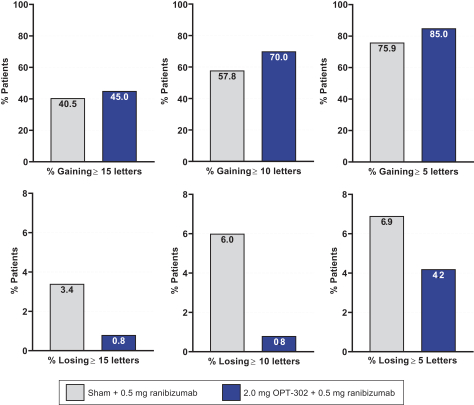
Reductions in Retinal Thickness and Fluid
In addition to the statistically significant improvement in visual acuity, treatment with Sozinibercept (OPT-302) combination therapy led to greater reductions in retinal thickness. The reductions in retinal thickness in patients is consistent with less fluid accumulation in the retina and reduced disease severity as increased fluid accumulation in the retina is associated with the loss of visual acuity in wet AMD patients. The figure below depicts the greater mean reduction in retinal thickness in the Sozinibercept (OPT-302) combination therapy group compared to the ranibizumab monotherapy group at week 24.
83
Greater Reduction in Retinal Thickness from Baseline to Week 24 following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
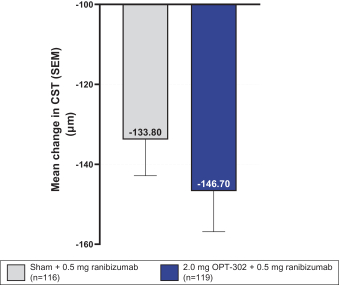
Fewer patients in the Sozinibercept (OPT-302) combination therapy group had subretinal fluid and intraretinal fluid present compared to the ranibizumab monotherapy group. The presence of subretinal fluid is a hallmark of wet AMD and its resolution is referred to as “drying” of the retina while intraretinal fluid is a prognostic biomarker for poor visual acuity and sub-optimal response to anti-VEGF-A therapies. As illustrated in the figure below, there were approximately 10% fewer patients with subretinal fluid and 5% fewer patients with intraretinal fluid following administration of Sozinibercept (OPT-302) combination therapy compared to ranibizumab monotherapy.
Fewer Patients with Subretinal and Intraretinal Fluid Present at Week 24 Following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
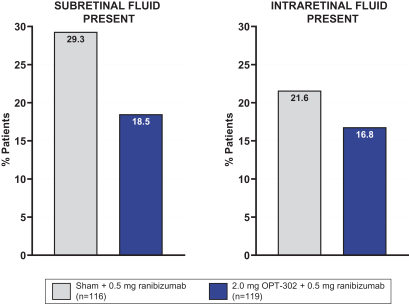
Greater improvements in anatomical indicators of disease severity, including on exploratory endpoints of mean reduction in total lesion area and CNV area, were observed in the Sozinibercept (OPT-302) combination therapy group compared to ranibizumab monotherapy. As illustrated in the figure below, the patients treated with Sozinibercept (OPT-302) combination therapy had an approximately 39% further reduction in both total lesion area and CNV area compared to ranibizumab monotherapy.
84
Greater Reduction in Total Lesion Area and CNV Area from Baseline to Week 24 following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
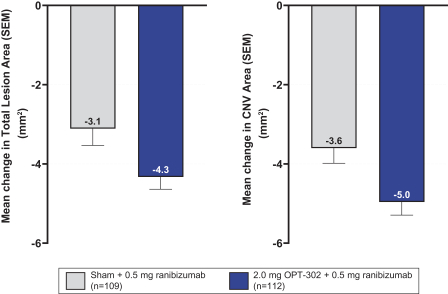
Improved Therapeutic Outcomes in Wet AMD Lesion Subtypes
A number of pre-specified subgroup and exploratory analyses were incorporated into the Phase 2b trial design in order to identify those wet AMD patients who may respond best to Sozinibercept (OPT-302). The Phase 2b trial randomized patients with a broad range of lesion morphologies including occult, minimally classic and predominantly classic lesions. In addition, the Phase 2b trial investigated efficacy in post-hoc analyses in other wet AMD subtypes, including PCV and RAP. This trial was not designed for statistical significance on these subgroup and exploratory analyses and was not powered to detect statistically significant differences in related measurements.
Patients enrolled in our Phase 2b trial consisted of 44% occult, 43% minimally classic and 13% predominantly classic lesion types, which is similar to the distribution reported in treatment-naive wet AMD patients. Predominantly classic patients typically respond well to VEGF-A inhibitor therapy and an additive benefit of Sozinibercept (OPT-302) combination therapy could not be discerned in this small patient group of only 15 patients per treatment arm. The majority of patients randomized in the Phase 2b trial had occult or minimally classic lesions. These patients did not respond as well to ranibizumab monotherapy as those with predominantly classic lesions. Patients with occult and minimally classic lesions treated with Sozinibercept (OPT-302) combination therapy experienced improvement in visual acuity beginning at week 8 and persisting through week 24 compared to ranibizumab monotherapy. The figure below illustrates the mean change in visual acuity over 24 weeks in each of the lesion classifications treated with Sozinibercept (OPT-302) combination therapy compared to ranibizumab monotherapy.
85
Mean Change in Visual Acuity from Baseline to Week 24 in Wet AMD Lesion Types Following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
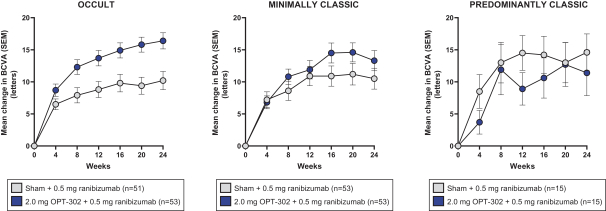
In a post-hoc analysis, in the occult lesion subgroup, the mean visual acuity gain from baseline to week 24 in the Sozinibercept (OPT-302) combination therapy group was +16.2 letters (n=53), compared to +10.2 letters in the ranibizumab monotherapy group (n=51), a benefit of +6.0 letters. In addition, in the minimally classic subgroup, the mean visual acuity gain from baseline to week 24 in the Sozinibercept (OPT-302) combination therapy group was +13.7 letters (n=53), compared to +11.1 letters in the ranibizumab monotherapy group (n=53), a benefit of +2.7 letters.
Gains in Visual Acuity from Baseline to Week 24 in Patients with Occult and Minimally Classic Lesions Following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
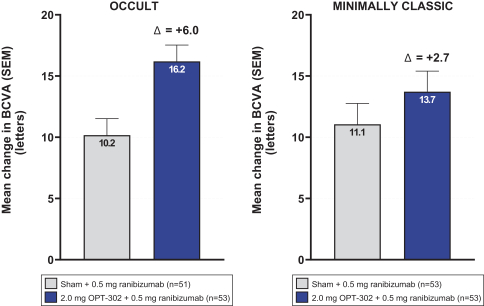
In particular, in the occult lesion subgroup, we observed a greater proportion of patients gaining 15 and 10 letters in the Sozinibercept (OPT-302) combination therapy group compared to the ranibizumab monotherapy group (approximately 22% and 19% over ranibizumab monotherapy, respectively). In the minimally classic subgroup, we observed a greater proportion of patients (approximately 10% over ranibizumab monotherapy) gaining 10 letters in the Sozinibercept (OPT-302) combination therapy group compared to the ranibizumab monotherapy group.
In patients with occult lesions, treatment with Sozinibercept (OPT-302) combination therapy also led to a greater mean reduction in retinal thickness of -134.6 µm from baseline compared to -103.5 µm for ranibizumab monotherapy. Sozinibercept (OPT-302) combination therapy did not have greater clinical benefit in the reduction of retinal thickness in minimally classic lesions, compared to the ranibizumab monotherapy group.
86
In patients with occult lesions, approximately 13% and 12% fewer patients in the Sozinibercept (OPT-302) combination group had subretinal fluid and intraretinal fluid, respectively, as compared to ranibizumab monotherapy at week 24. In patients with minimally classic lesions, approximately 2% fewer patients treated with Sozinibercept (OPT-302) combination therapy had subretinal fluid and intraretinal fluid compared to patients treated with ranibizumab monotherapy at week 24.
As shown in the figures below, by week 24, patients with occult and minimally classic lesions also had greater reductions in both wet AMD total lesion area and CNV area following Sozinibercept (OPT-302) combination therapy compared to patients receiving ranibizumab monotherapy.
Greater Reduction in Total Lesion Area from Baseline to Week 24 in Patients with Occult and Minimally Classic Lesions Following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
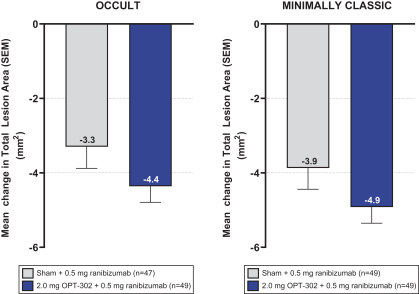
Greater Reduction in CNV Area from Baseline to Week 24 in Patients with Occult and Minimally Classic Lesions Following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
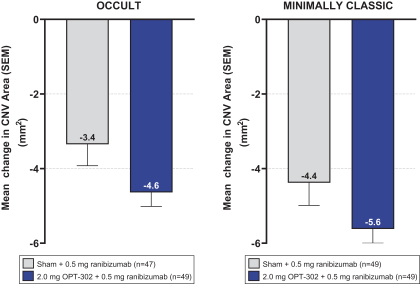
87
Sozinibercept (OPT-302) combination therapy improved visual acuity to the greatest extent in occult lesions compared to minimally classic or predominantly classic lesions, in each case compared to ranibizumab monotherapy. While these results were generated in treatment-naive wet AMD patients, classification of lesions following treatment with either Sozinibercept (OPT-302) combination therapy or ranibizumab monotherapy suggests that virtually all lesions shifted toward an occult morphology. This suggests that Sozinibercept (OPT-302) combination therapy also has the potential to provide benefit to patients who have been previously treated with anti-VEGF-A monotherapy, given the predominantly occult morphology of their lesions following treatment.
RAP and PCV Lesion Subtypes
In our Phase 2b clinical trial, over 85% of patients enrolled did not have RAP lesions detected at randomization and these patients responded better to Sozinibercept (OPT-302) than patients with RAP lesions. In patients without RAP lesions, the mean visual acuity gain from baseline to week 24 was +15.0 letters (n=103) with Sozinibercept (OPT-302) combination therapy, compared to +10.6 letters for those treated with ranibizumab monotherapy (n=102), a benefit of +4.4 letters.
Mean Change in Visual Acuity from Baseline to Week 24 in Patients Without RAP Lesions Following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
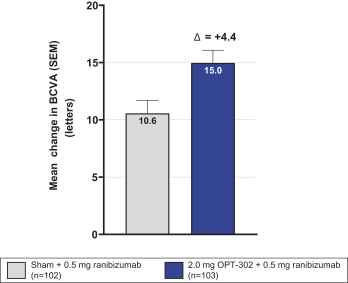
In patients without RAP lesions who had occult or minimally classic lesions, representing the majority of wet AMD patients, the mean visual acuity gain from baseline to week 24 was +16.1 letters with Sozinibercept (OPT-302) combination therapy (n=88) compared to +10.3 letters for those treated with ranibizumab monotherapy (n=87), a benefit of +5.7 letters. This represents the patient population for which the primary analysis of the primary endpoint of the planned Phase 3 trials of Sozinibercept (OPT-302) combination therapy for the treatment of wet AMD will be first conducted, followed by analysis of the total patient population and the every-eight week dosing groups.
88
Mean Change in Visual Acuity from Baseline to Week 24 in the combined group of RAP Absent Occult and Minimally Classic Lesions Following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
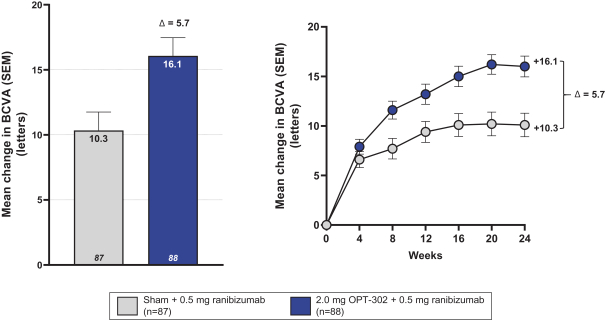
Patients with RAP absent occult lesions demonstrated a mean visual acuity gain at week 24 of +16.8 letters with Sozinibercept (OPT-302) combination therapy (n=49) compared to +10.2 letters for those treated with ranibizumab monotherapy (n=47), a gain of +6.5 letters. Patients with RAP absent minimally classic lesions demonstrated a mean visual acuity gain at week 24 of +14.9 letters with Sozinibercept (OPT-302) combination therapy (n=39) compared to +10.0 letters for those treated with ranibizumab monotherapy (n=40), a gain of +4.7 letters.
Mean Change in Visual Acuity from Baseline to Week 24 in RAP Absent Occult and Minimally Classic Lesions Following Sozinibercept (OPT-302) Combination Therapy Compared to Ranibizumab Monotherapy
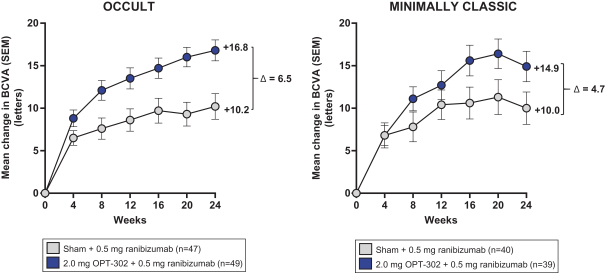
Improved Therapeutic Outcomes of Sozinibercept (OPT-302) in PCV Lesions
In patients with PCV lesions, the mean visual acuity gain from baseline to week 24 in the Sozinibercept (OPT-302) combination therapy group was +13.5 letters (n=22), compared to +6.9 letters in the ranibizumab
89
monotherapy group (n=20). This equates to a benefit of +6.7 letters and is almost a two-fold improvement in visual acuity gain observed from baseline following Sozinibercept (OPT-302) combination therapy. Ranibizumab monotherapy was not observed to be as effective in patients with PCV compared to other wet AMD subtypes.
Safety and Tolerability
Sozinibercept (OPT-302) was well tolerated in this Phase 2b trial with a very low incidence of ocular inflammation and no safety issues identified with addition of Sozinibercept (OPT-302) to ranibizumab intravitreal therapy. The incidence of ocular TEAEs was similar in Sozinibercept (OPT-302) combination groups compared to the ranibizumab monotherapy group. TEAEs were considered potentially treatment related in approximately 15% of patients. The most common treatment-related TEAEs were eye pain, vitreous floaters, eye irritation and raised intraocular pressure. One patient discontinued from the trial due to a TEAE, which was not considered treatment related. Three patients treated with Sozinibercept (OPT-302) combination therapy had potentially treatment-related SAEs: one case each of vitritis, endophthalmitis and myocardial infarction.
Pivotal Phase 3 Clinical Trials in Wet AMD
We initiated two concurrent pivotal Phase 3 clinical trials for the treatment of wet AMD in March 2021. These double-masked, sham-controlled Phase 3 clinical trials enroll treatment-naive patients and assess the efficacy and safety of 2.0 mg of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for treatment-naive patients with wet AMD compared to a standard of care anti-VEGF-A monotherapy. In addition, to understand the durability of Sozinibercept (OPT-302) treatment effect with less frequent dosing, each trial compares the clinical efficacy of Sozinibercept (OPT-302) administered in combination with the applicable VEGF-A inhibitor on an every 4-week and every 8-week dosing regimen. The primary endpoint of both trials will be the mean change in visual acuity from baseline at week 52. Patients will continue to be dosed until week 96 to further assess long-term safety at week 100. We expect to complete patient recruitment in the COAST study in the first calendar quarter of 2024 and in the ShORe study in the second calendar quarter of 2024. Top-line data is expected to be reported when all patients complete the 52-week treatment period for the primary analysis. If the results at the completion of the primary efficacy phase at week 52 of the Phase 3 clinical trials are favorable, we intend to file for marketing approval for Sozinibercept (OPT-302) in 2026 for the treatment of wet AMD in the United States, the European Union and certain other territories.
Study of Sozinibercept (OPT-302) in Combination with Ranibizumab (ShORe) Phase 3 Trial
In the Study of Sozinibercept (OPT-302) in combination with Ranibizumab, or ShORe, Phase 3 trial, treatment-naive patients with wet AMD will be randomized to one of three treatment groups. Patients randomized to the standard dosing arm will receive standard of care 0.5 mg ranibizumab every four weeks in combination with 2.0 mg Sozinibercept (OPT-302) on a standard every four weeks dosing regimen. In the extended dosing arm, 0.5 mg ranibizumab will be administered every four weeks to week 52, in combination with 2.0 mg Sozinibercept (OPT-302) administered every four weeks for three total doses over 12 weeks, followed by Sozinibercept (OPT-302) dosing every eight weeks to week 52, with a sham injection administered at visits where Sozinibercept (OPT-302) is not administered. Patients randomized to the control arm will receive 0.5 mg ranibizumab administered in combination with sham intravitreal injections administered every four weeks to week 52. The primary and secondary efficacy outcomes will be determined at the end of the efficacy phase at week 52. Each patient will then continue to be treated for an additional year in the safety phase to reach week 100 to evaluate safety and tolerability over a total two-year period.
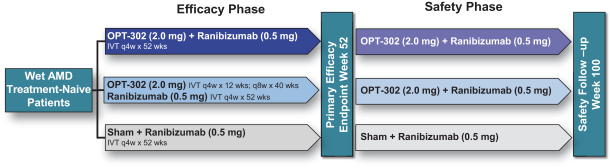
90
Combination of Sozinibercept (OPT-302) with Aflibercept STudy (COAST) Phase 3 Trial
In the Combination Sozinibercept (OPT-302) with Aflibercept STudy, or COAST, Phase 3 trial, treatment-naive wet AMD patients will be randomized to one of three treatment groups. Patients randomized to the standard dosing arm will receive 2.0 mg aflibercept administered every four weeks for three total doses over 12 weeks, followed by aflibercept dosing every eight weeks to week 52, in combination with 2.0 mg Sozinibercept (OPT-302) administered every four weeks to week 52. In the extended dosing arm, 2.0 mg aflibercept in combination with 2.0 mg Sozinibercept (OPT-302) will be administered every four weeks for three total doses over 12 weeks, followed by dosing every eight weeks to week 52, with a sham injection administered at visits where Sozinibercept (OPT-302) and aflibercept are not administered. Patients randomized to the control arm, will receive 2.0 mg aflibercept administered every four weeks for three total doses over 12 weeks, followed by dosing every eight weeks to week 52, in combination with sham intravitreal injections administered every four weeks to week 52. Similar to the ShORe trial, the primary and secondary efficacy outcomes of the COAST trial will be assessed at the end of the efficacy phase at week 52. Each patient will then continue to be treated for an additional year in the safety phase to reach week 100 to evaluate the safety and tolerability over a total two-year period.
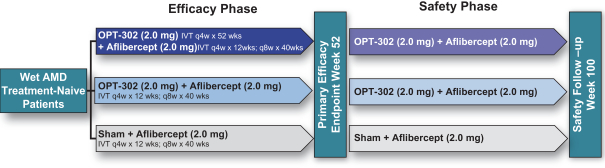
For consistency, the ShORe and COAST Phase 3 trials build upon and maintain key features of our Phase 2b clinical trial of Sozinibercept (OPT-302) combination therapy for the treatment of wet AMD, while evaluating the administration of Sozinibercept (OPT-302) combination therapy over a longer treatment period and in a greater number of patients. In addition, the results of our Phase 2b clinical trial has informed the design of the Phase 3 trials. Analysis of the Phase 2b trial demonstrated that Sozinibercept (OPT-302) combination therapy increased visual acuity by a further +5.7 letters over ranibizumab monotherapy in wet AMD patients with minimally classic and occult lesions, representing the majority of wet AMD patients. Based on these positive data, primary analysis of the primary endpoint of the Phase 3 trials will be first conducted in patients with minimally classic and occult lesions administered Sozinibercept (OPT-302) every four weeks and every eight weeks, followed by analysis on the total patient population.
Development of Co-Formulation
Sozinibercept (OPT-302) is currently administered as a combination therapy consisting of a sequential injection of Sozinibercept (OPT-302) following intravitreal administration of a VEGF-A inhibitor. We plan to develop a co-formulation of Sozinibercept (OPT-302) with an approved and/or biosimilar anti-VEGF-A therapy to achieve VEGF-A, VEGF-C and VEGF-D inhibition following the administration of a single intravitreal injection of the co-formulated product. We believe that a co-formulated Sozinibercept (OPT-302) and VEGF-A inhibitor product could provide flexibility of treatment options for physicians and reduce the frequency and number of injections for patients.
We are currently assessing opportunities with multiple third parties to in-license and/or generate a biosimilar anti-VEGF-A therapy, which we intend to co-formulate with Sozinibercept (OPT-302) and advance through non-clinical studies, including IND-enabling safety and tolerability studies. We intend to file an IND for the co-formulated product in in a Phase 1 clinical trial for the treatment of wet AMD.
Diabetic Macular Edema
Diabetic macular edema is a complication of DR, a disease affecting the blood vessels of the retina in diabetics. Chronically elevated blood glucose levels, or hyperglycemia, causes damage to the small blood vessels or capillaries in the retina in patients with diabetes. The consequent chronic decrease in oxygen supply to retinal cells results in tissue damage that is referred to as DR. Approximately one-third of patients with DR or up to 10% of diabetics develop DME, which is characterized by accumulation of fluid and retinal thickening within the macula and is responsible for most of the central visual loss experienced in the diabetic population.
91
Central-involved DME is diagnosed when swelling or edema occurs from fluid leaking into the central fovea region of the macula.
Current Treatments for DME and Their Limitations
VEGF-A inhibitor therapy is the first-line standard of care therapy for DME. Ranibizumab, aflibercept and faricimab-syoa are approved for the treatment of DME, and similarly to wet AMD, many patients receive bevacizumab as an off-label, lower cost alternative VEGF-A inhibitor therapy. Many patients with central-involved DME require near-monthly administration of intravitreal VEGF-A inhibitors during the first 12 months of treatment, with fewer injections needed in subsequent years to maintain clinical benefit. VEGF-A inhibitors have largely replaced the use of laser photocoagulation as a treatment for DME.
The anti-inflammatory corticosteroid implant therapies dexamethasone (Ozurdex®) and fluocinolone acetonide (Illuvien®) are also approved for use in central-involved DME. These agents, however, are rarely used as first-line therapy due to inferior visual acuity outcomes compared to anti-VEGF-A therapy. Patients with persistent DME and who are insufficiently responsive to anti-VEGF-A therapy have shown some treatment benefit with intravitreal corticosteroids. However, as intravitreal corticosteroids are associated with high rates of ocular adverse events including cataract progression and intraocular pressure elevation, switching to corticosteroids from an anti-VEGF-A therapy with a sub-optimal response needs to be carefully considered.
Despite the widespread use of treatments targeting VEGF-A in the management of DME, there is still a significant unmet need as many patients demonstrate a sub-optimal response, remain treatment refractory or require frequent injections for persistent leakage in the macula. Up to two-thirds of patients with central-involved DME treated with VEGF-A inhibitors do not show reductions in the fluid or clinically meaningful improvement in visual acuity. In addition, approximately 25% of DME patients treated with VEGF-A inhibitors continue to have macula thickening and swelling following treatment. This resistance may occur as treatment selective anti-VEGF-A monotherapies do not fully address all of the factors involved in the pathogenesis of DME. As such, combination therapies targeting alternative factors and pathways have the potential for improved clinical outcomes in DME patients.
Market Opportunity for the Treatment of DME
It is estimated that between 1.3 million and 2.0 million people worldwide, including 14% of Type 1 diabetics and 6% of Type 2 diabetics, have DME. The risk of developing DME increases with time. According to the Wisconsin Epidemiologic Study of Diabetic Retinopathy, after 10 years of follow-up, 20% of patients with Type 1 diabetes and 25% of those with Type 2 diabetes will have developed DME. Ranibizumab and aflibercept, two VEGF-A inhibitors approved for the treatment of DME, generated combined annual worldwide sales approaching US$13 billion in 2022. Historically, approximately 20% of these sales are attributable to the treatment of DME.
Potential for Sozinibercept (OPT-302) in DME
We believe that as a potent inhibitor of VEGF-C and VEGF-D, Sozinibercept (OPT-302) has the potential to provide significant therapeutic benefit to patients affected by DME. Although the underlying causes of wet AMD and DME differ, members of the VEGF family play a role in the progression of both diseases. VEGF-C and VEGF-D and their receptors are specifically implicated in the progression of diabetes. For example, patients with diabetes have higher levels of VEGF-C and VEGF-A and increased expression of both VEGFR-2 and VEGFR-3 in the retina compared to non-diabetics. The VEGF-A inhibitors ranibizumab and aflibercept, originally approved for treatment of wet AMD patients, have also been approved for treatment of DME patients. Similar to wet AMD, bevacizumab is also frequently used off-label as a treatment for DME. Faricimab-svoa was approved in 2022 for wet AMD and DME.
In our completed Phase 1b dose escalation clinical trial in patients with persistent DME, we observed what we believe to be promising evidence of a dose response of Sozinibercept (OPT-302) in combination with aflibercept, including further improvements in visual acuity despite patients’ having been previously treated with anti-VEGF-A therapy. In our completed Phase 2a clinical trial, we observed improved visual acuity outcomes and evidence of a reduction in retinal thickness in treatment-refractory DME patients following Sozinibercept (OPT-302) combination therapy.
92
Phase 1b Clinical Trial of Sozinibercept (OPT-302) in DME
In 2018, we completed a Phase 1b dose-escalation clinical trial of Sozinibercept (OPT-302) in combination with aflibercept in nine patients with persistent DME that were previously treated with anti-VEGF-A therapies. Patients were administered three escalating doses (0.3 mg, 1.0 mg or 2.0 mg) of Sozinibercept (OPT-302) in combination with 2.0 mg aflibercept by intravitreal injections once every four weeks for a total of three doses. The primary analysis was conducted at week 12, four weeks after the final dose.
Across all nine patients in the Phase 1b trial, a mean gain in visual acuity of +7.7 letters from baseline to week 12 was observed across all dose groups, with a clear dose response of improved visual acuity with increasing doses of Sozinibercept (OPT-302). There was a corresponding mean decrease in retinal thickness at week 12 of -71 µm from baseline and six of nine (67%) patients had a 50% reduction in excess foveal thickness.
Dose-dependent Increases in Visual Acuity and Reduction in Retinal Thickness at Week 12 Following Treatment with Sozinibercept (OPT-302) in Combination with Aflibercept
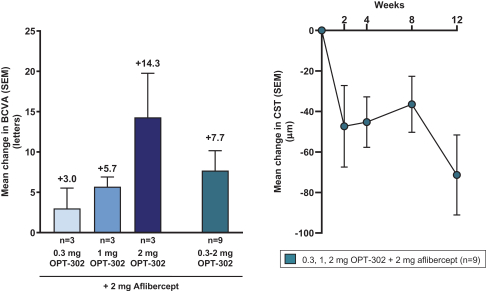
Five of the nine patients in the Phase 1b trial had bilateral, persistent treatment-refractory DME. In these patients, study eyes received Sozinibercept (OPT-302) in combination with aflibercept, and fellow eyes received standard of care anti-VEGF-A monotherapy. In these bilateral disease patients, the mean change in visual acuity from baseline to week 12 was +10.0 letters in the study eye and +2.6 letters in the fellow eye. The corresponding reduction in retinal thickness from baseline to week 12 was -80 µm for Sozinibercept (OPT-302) combination treated study eyes, compared to -6 µm in fellow eyes that received anti-VEGF-A monotherapy.
93
In Patients with Bilateral DME, Sozinibercept (OPT-302) Combination Therapy Improved Visual Acuity and Reduced Retinal Thickness Compared to Fellow Eyes Treated with Anti-VEGF-A Monotherapy
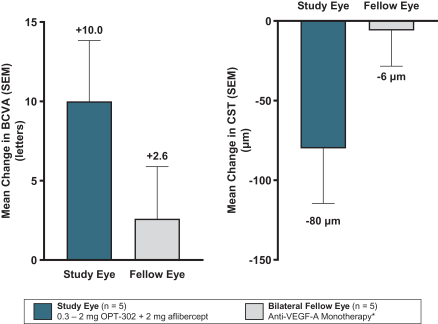
* Patients with bilateral disease and persistent DME in the fellow eye receiving anti-VEGF-A (ranibizumab or aflibercept) monotherapy. Prior anti-VEGF-A therapy in Fellow Eyes BL to Week 12 (5 patients): 3x Aflibercept, 3x Ranibizumab, 1x Ranibizumab, 4x Ranibizumab, 3x Aflibercept.
Sozinibercept (OPT-302) in combination with aflibercept was well tolerated at all dose levels, with no DLTs and the MTD was not reached. There were no treatment-related clinically significant changes in intraocular pressure, electrocardiograms or vital signs. The most common AEs were related to the intravitreal injection procedure.
Phase 2a Clinical Trial of Sozinibercept (OPT-302) in DME
Based on the positive results of our Phase 1b trial, we reported outcomes from a Phase 2a trial in persistent DME patients refractory to anti-VEGF-A therapy in June 2020. Similar to the Phase 1b trial, this proof-of-concept trial was designed to investigate the ability of Sozinibercept (OPT-302) to improve outcomes in persistent DME patients. The primary endpoints were clinical response rate in visual acuity as well as safety and tolerability.
Clinical Trial Design
This Phase 2a trial was a randomized, double-masked, dose expansion trial that enrolled patients diagnosed with persistent center-involved DME despite regular administration of prior anti-VEGF-A monotherapy. These patients are considered to be a difficult-to-treat patient population since they have received prior anti-VEGF-A therapy and experienced a suboptimal clinical response. In our trial, these patients were defined as having visual acuity between 20/40 and 20/320 Snellen equivalent, or 73 and 24 BCVA letters on the ETDRS standardized eye chart, and retinal thickness of 320 µm on SD-OCT. In this Phase 2a trial, the mean number of prior intravitreal anti-VEGF-A injections was eight in each of the treatment groups, reflecting that the patients recruited into this trial were heavily pre-treated, with a mean of 39 days since the immediate prior injection to the start of the trial.
The Phase 2a trial was conducted at 53 trial sites in the United States, Israel, Australia and Latvia. Of the 144 patients randomized in the trial, 115 patients conformed sufficiently with the trial protocol and were included in our analyses of clinical efficacy. Patients were randomized 2:1 to receive either 2.0 mg Sozinibercept (OPT-302) in combination with 2.0 mg aflibercept or a sham injection and 2.0 mg aflibercept. Patients received intravitreal injections once every four weeks for a total of three doses. The primary analysis was conducted at week 12, four weeks after the final dose.
94
Design of the Phase 2a Clinical Trial of Sozinibercept (OPT-302) in Combination with Aflibercept in Persistent Diabetic Macular Edema

PRN refers to pro re nata, or treatment on an as needed basis.
Improvements in Visual Acuity
The primary efficacy endpoint of the trial was the clinical response rate, defined as the proportion of patients receiving Sozinibercept (OPT-302) combination therapy that achieved a 5 letter gain in visual acuity at week 12 compared to baseline. Our predefined measure of success was a response rate of greater than or equal to 38%, based on historical observations that show limited ability to achieve a 5 letter improvement in DME patients on long-term anti-VEGF-A monotherapy. As an exploratory trial, this Phase 2a was not powered to detect statistical significance of Sozinibercept (OPT-302) combination therapy compared to aflibercept monotherapy.
We observed that 52.8% of patients treated with Sozinibercept (OPT-302) combination therapy achieved a 5 letter improvement in visual acuity at week 12 compared to baseline, meeting the pre-specified primary efficacy endpoint for this trial.
The mean change in visual acuity at week 12 compared to baseline was +5.9 letters in patients receiving Sozinibercept (OPT-302) combination therapy and +6.1 letters in the aflibercept monotherapy group. In the Sozinibercept (OPT-302) combination therapy group, the percentage of patients with visual acuity gains of 10 and 15 letters was higher, and the percentage of patients who lost 5 letters was lower than that in the aflibercept monotherapy group. These measures of visual function are shown in the figure below.
Measures of Visual Acuity at Week 12 Following Sozinibercept (OPT-302) Combination Therapy and Aflibercept Monotherapy
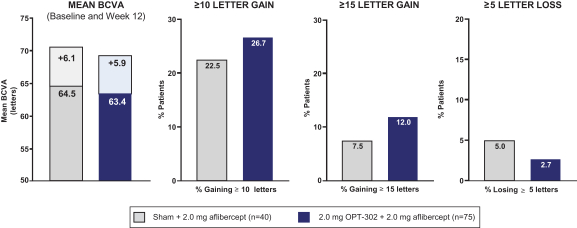
Patients treated with Sozinibercept (OPT-302) combination therapy also had decreased retinal thickness compared to aflibercept monotherapy, as shown in the figure below.
95
Greater Reduction in Retinal Thickness from Baseline to Week 12 Following Sozinibercept (OPT-302) Combination Therapy Compared to Aflibercept Monotherapy
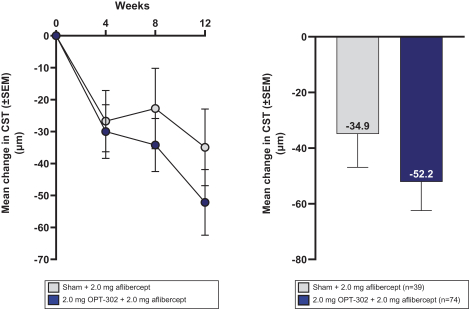
Prior Treatment History in Persistent DME Patients
Prior studies have shown that DME patients with mild visual acuity loss of 20/40 or better at baseline, show similar outcomes to any of the VEGF-A therapies: ranibizumab, aflibercept and bevacizumab. However, in patients with poorer baseline vision of 20/50 or worse, aflibercept has better outcomes compared to ranibizumab and bevacizumab over the first 12 months of treatment. It has also been shown that DME patients who experience inadequate responses to ranibizumab have achieved further anatomical and functional improvements upon switching to aflibercept.
Due to the challenge of enrolling a large group of patients with identical prior treatment histories, we designed our Phase 2a trial to accelerate enrollment by randomizing patients with variable prior treatment histories. This strategy allowed us to more broadly understand the prior treatment history in persistent DME patients, which will inform the design of our future trials in DME.
In order to explore the importance of differences in prior treatment history, we collected detailed anti-VEGF-A treatment histories for patients enrolled in our Phase 2a DME trial. Patients randomized into our Phase 2a trial had variable prior treatment histories which included infrequent or irregular dosing and/or therapy with aflibercept, ranibizumab and bevacizumab. Approximately one third of patients had a prior treatment history of having received only aflibercept, or aflibercept for their three anti-VEGF-A treatments immediately before trial enrollment. Approximately 11% of patients had a prior treatment history of having received only ranibizumab, or ranibizumab for their three anti-VEGF-A treatments immediately before trial enrolment, whereas approximately 44% of patients had received only bevacizumab prior to trial enrollment. Patients with a prior treatment history of bevacizumab were required to receive at least one injection of either aflibercept or ranibizumab immediately prior to randomization into the trial. Post-hoc analyses of the results from our Phase 2a trial suggest that some patients may have benefited from the increased efficacy of aflibercept and/or from the switch to aflibercept therapy as administered in our trial on an every four-week dosing cycle. In the subset of patients who had only received aflibercept, or received aflibercept for their three anti-VEGF-A treatments immediately before trial enrollment, referred to as treatment history of prior aflibercept, the mean improvement in visual acuity observed for the aflibercept monotherapy group was +3.4 letters (n=13), compared to a mean improvement of +7.4 letters for those patients with more variable prior treatment history who received aflibercept monotherapy (n=27) following randomization into the trial. This suggests that the majority of patients enrolled in the trial had not achieved a maximal response to all anti-VEGF-A therapies prior to enrolling in the trial.
96
Due to the observed increase in treatment benefit for patients with a variable treatment history who then received aflibercept every four weeks in the trial, the subset of patients with a treatment history of prior aflibercept may represent the most stringent and least variable patient population in which to test the ability of Sozinibercept (OPT-302) to provide additional benefit. In this more homogeneous patient population, as shown in the figure below, patients administered Sozinibercept (OPT-302) combination therapy demonstrated a mean improvement in visual acuity of +6.6 letters (n=22) from baseline to week 12, compared to +3.4 letters (n=13) in the aflibercept monotherapy group.
Greater Gains in Visual Acuity following Sozinibercept (OPT-302) Combination Therapy in Patients with a Treatment History of Prior Aflibercept
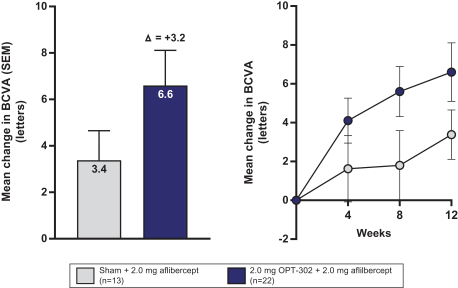
In addition, 27.3% of patients gained 10 letters and 9.1% gained 15 letters of visual acuity from baseline to week 12 following Sozinibercept (OPT-302) combination therapy. There were no patients with a treatment history of prior aflibercept that gained 10 letters of visual acuity in the aflibercept monotherapy group. Furthermore, the proportion of patients who lost 1 letters was 9.1% in the Sozinibercept (OPT-302) combination therapy group and 23.1% in the aflibercept monotherapy group. We believe that these results, as shown in the figures below, strongly support the potential of Sozinibercept (OPT-302) to improve the visual acuity in patients with persistent DME despite prior treatment with anti-VEGF-A monotherapy.
Proportion of Patients with a Treatment History of Prior Aflibercept Who Gained and Lost Visual Acuity from Baseline to Week 12
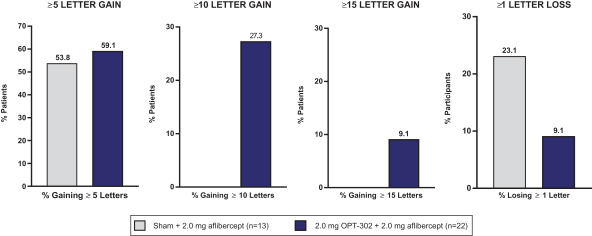
97
In this subgroup of patients with a treatment history of prior aflibercept, anatomical changes were consistent with functional visual acuity outcomes. As shown in the figure below, a greater mean reduction in retinal thickness was observed in the Sozinibercept (OPT-302) combination therapy group compared to the aflibercept monotherapy group at week 12. In particular, 22.7% of patients in the Sozinibercept (OPT-302) combination therapy group experienced at least a 300 µm reduction in retinal thickness at week 12, compared to 7.7% of patients in the aflibercept monotherapy group.
Greater Mean Reduction in Retinal Thickness following Sozinibercept (OPT-302) Combination Therapy in Patients with a Treatment History of Prior Aflibercept
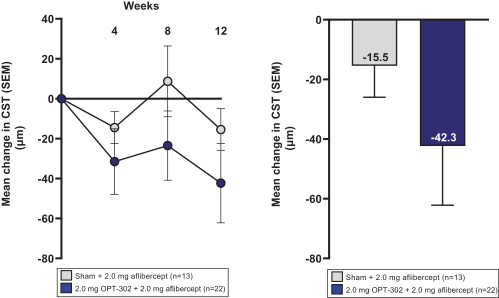
Safety and Tolerability
Sozinibercept (OPT-302) combination therapy was well tolerated. There was one potentially treatment-related SAE of cerebrovascular accident, or stroke, resulting in one patient discontinuing treatment and withdrawing from the trial. The most common TEAEs were conjunctival hemorrhage and increased intraocular pressure and were mainly related to the intravitreal injection procedure. TEAEs did not lead to discontinuation of the trial for any patient. The incidence of intra-ocular inflammation was low, occurring in one patient for each treatment group, and the observed events were manageable and able to be resolved.
We now have extensive global clinical dosing experience demonstrating a favorable tolerability profile following repeated intravitreal administration of Sozinibercept (OPT-302) in 399 patients, with over 1,800 doses of Sozinibercept (OPT-302) administered across three international clinical studies in two disease indications and in combination with the two leading standard of care anti-VEGF-A therapies, ranibizumab and aflibercept. In particular, across our clinical trials, the incidence of intra-ocular inflammation was similar across all treatment groups.
Retinal Vein Occlusion
Based on the positive clinical data from our clinical trials of Sozinibercept (OPT-302) in wet AMD and DME, we intend to prioritize future development in these two indications while exploring potential opportunities to develop Sozinibercept (OPT-302) in other ophthalmic indications such as RVO, DR and other diseases involving aberrant CNV.
RVO is a sight-threatening visual disorder resulting from blockage of one of the veins carrying blood out of the retina. This blocked vein can leak blood and fluid resulting in swelling that can cause macular edema. Persistent, inadequately-treated macular edema associated with RVO can blur vision, cause significant loss in visual acuity and eventually lead to blindness. Macular edema is the most common cause of vision loss in people who suffer from RVO.
98
Similar to wet AMD and DME, the first-line standard of care to treat macular edema associated with RVO is intravitreal anti-VEGF-A monotherapy. VEGF-A inhibitors however, are only effective in significantly improving vision in approximately 30% to 40% of patients with macular edema associated with RVO above sham control. The prevalence of RVO in people over the age of 50 has been reported to be 0.7%, or approximately 1.8 million people in the United States and Europe. Over 500,000 individuals in the United States and Europe have macular edema associated with RVO. We believe that Sozinibercept (OPT-302) has the potential to bring therapeutic benefit to patients suffering from macular edema secondary to RVO.
Competition
The biotechnology and pharmaceutical industries, and the ophthalmic disease subsector, are characterized by rapidly advancing technologies, evolving understanding of disease etiology, intense competition and a strong emphasis on intellectual property. While we believe that Sozinibercept (OPT-302) and our knowledge and experience provide us with certain competitive advantages, we face substantial potential competition from many different sources, including large and specialty pharmaceutical and biotechnology companies, academic research institutions and governmental agencies and public and private research institutions. Many of our current or potential competitors, either alone or with their collaboration partners, have significantly greater financial resources and expertise in research and development, manufacturing, pre-clinical studies, conducting clinical trials and marketing approved products than we do. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These competitors also compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs. Accordingly, our competitors may be more successful than we may be in developing, commercializing and achieving widespread market acceptance. In addition, our competitors’ products may be more effective or more effectively marketed and sold than any treatment we or our development partners may commercialize and may render our product candidates obsolete or noncompetitive before we can recover the expenses related to developing and commercializing our product candidates.
We are developing Sozinibercept (OPT-302) for the treatment of wet AMD and additional retinal disease indications, such as DME and RVO, together with certain combination agents. Companies that have products that may compete with Sozinibercept (OPT-302) include Roche Group, Regeneron Pharmaceuticals, Inc. and Novartis AG, which have marketed anti-VEGF-A therapies including ranibizumab (Lucentis) and aflibercept (Eylea), each a standard of care treatment for wet AMD, and brolucizumab (Beovu). Faricimab-svoa (Vabysmo) is approved for wet AMD and DME. A higher dose version (8 mg) of aflibercept was recently approved that allow some patients less frequent dosing. Companies with products in development for the treatment of wet AMD and DME - include Ocugen, Inc., Ocular Therapeutix, Inc., EyePoint Pharmaceuticals, Inc., Clearside Biomedical, Inc., and Outlook Therapeutics, Inc. We are also aware of other companies that are working on therapies for the whole eye, including Santen, Inc. and Ocular Therapeutix, Inc. In addition, bevacizumab (Avastin), marketed by Genentech, Inc., is used off-label to treat wet AMD.
It is possible that our competitors will succeed in developing technologies that are more effective than our product candidates or that would render our technology obsolete or noncompetitive, or will succeed in developing biosimilar or interchangeable products for our product candidates. We anticipate that we will continue to face increasing competition in the future as new companies enter our market and scientific developments surrounding biosimilars and other retinal therapies continue to accelerate, particularly once ranibizumab and aflibercept approach loss of exclusivity. We cannot predict to what extent the entry of biosimilars or other competing products will impact potential future sales of our products or our product candidates.
With respect to our current and potential future product candidates, we believe that our ability to compete effectively and develop products that can be manufactured cost-effectively and marketed successfully will depend on our ability to:
99
Our Commercial License Arrangement with Selexis SA
In October 2013, we entered into a commercial license agreement, or the Selexis Agreement, with Selexis SA, or Selexis, under which Selexis granted us a non-exclusive, worldwide, sublicensable license under certain patents, know-how and other intellectual property controlled by Selexis to use certain cell lines, deliverables and materials provide by Selexis to manufacture Sozinibercept (OPT-302) and related products and to use, sale and otherwise exploit such products.
We paid Selexis a nominal upfront payment upon entering into the Selexis Agreement. We are also required to make certain payments under the Selexis Agreement totaling approximately US$1.3 million upon the achievement of certain development and commercial milestones. We are also obligated to pay a low single-digit running royalty on worldwide net sales of the licensed products. Our royalty obligations will continue, on a product-by-product and country-by-country basis, until the expiration of the relevant patents, but will not extend beyond October 2024 in any event. After the expiration of the royalty term, our license will continue and become full paid, perpetual and irrevocable.
The Selexis Agreement will expire on the date of expiration of the last-to-expire of the license patents. Either party may terminate the Selexis Agreement for the other party’s uncured material breach or bankruptcy. We may also terminate the Selexis Agreement at any time upon prior notice to Selexis.
Intellectual Property
As of June 30, 2023, we have rights to 17 issued U.S. patents, six U.S. patent applications, 64 issued non-U.S. patents and four pending non-U.S. applications. Our current issued patents and patent applications began expiring in September 2022 all are projected to expire between September 2022 and November 2034 although our most recent provisional application, when completed, would not expire until 2043.
With respect to soluble forms of VEGFR-3, we own and have licensed rights to patent families including issued patents in the United States, Europe, Canada and Australia, which began expiring in 2022 and are expected to continue expiring until 2031. These patents cover composition of matter and/or method of use claims, including claims directed at the treatment of eye diseases associated with abnormal blood vessel growth, such as wet AMD.
With respect to Sozinibercept (OPT-302), we own a patent family with two issued U.S. patents, an issued European patent validated in 38 countries and non-U.S. patents granted in Australia, Canada, China, Colombia, Indonesia, Israel, India, South Korea, Mexico, Malaysia, New Zealand, Russia, Singapore and South Africa. Patent applications are pending in the United States and in Europe, Brazil, Malaysia and Philippines. The two issued U.S. patents have claims covering the composition of matter of Sozinibercept (OPT-302) and its use and/or nucleic acids, vectors, and host cells for producing it. These issued patents and pending patent applications, if issued, are expected to expire in 2034, without taking into account any patent term extension.
100
The actual protection afforded by a patent, which can vary from country to country, depends on the type of patent, the scope of its coverage as determined by the patent office or courts in the country, and the availability of legal remedies in the country. The information in the above list is based on our current assessment of patents that we own or control or have exclusively licensed. The information is subject to revision, for example, in the event of changes in the law or legal rulings affecting our patents or if we become aware of new information. Significant legal issues remain unresolved as to the extent and scope of available patent protection for biotechnology products and processes in the United States and other important markets outside the United States. We expect that litigation will likely be necessary to determine the term, validity, enforceability and/or scope of certain of our patents and other proprietary rights. An adverse decision or ruling with respect to one or more of our patents could result in the loss of patent protection for a product and, in turn, the introduction of competitor products or follow-on biologics to the market earlier than anticipated.
Patents expire, on a country by country basis, at various times depending on various factors, including the filing date of the corresponding patent application(s), the availability of patent term adjustment, patent term extension and supplemental protection certificates and requirements for terminal disclaimers. In most countries, including the United States, the patent term is 20 years from the earliest claimed filing date of a non-provisional patent application or its foreign equivalent in the applicable country. In the United States, a patent’s term may, in certain cases, be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the USPTO in examining and granting a patent, or may be shortened if a patent is terminally disclaimed over a commonly owned patent or a patent naming a common inventor and having an earlier expiration date. In the United States, a patent may also be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, or Hatch-Waxman Amendments. The Hatch-Waxman Amendments permit a patent extension term of up to five years as compensation for patent term lost during the FDA regulatory review process in the US. A patent term extension cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval, only one patent applicable to each regulatory review period may be extended and only those claims covering the approved drug, a method for using it, or a method for manufacturing it may be extended, with extended rights limited to the approved product, its approved uses, and/or its manufacture.
Although we believe our owned and licensed patents and patent applications provide us with a competitive advantage, the patent positions of biotechnology and pharmaceutical companies can be uncertain and involve complex legal and factual questions. We may not be able to develop patentable products or processes or obtain patents from pending patent applications. In the event of patent issuance, the patents may not be sufficient to protect the proprietary technology owned by or licensed to us or our partners. Our current patents, or patents that issue on pending applications, may be challenged, invalidated, infringed or circumvented. In addition, changes to patent laws in the United States or in other countries may limit our ability to defend or enforce our patents, or may apply retroactively to affect the term and/or scope of our patents. Our patents have been and may in the future be challenged by third parties in post-issuance administrative proceedings or in litigation as invalid, not infringed or unenforceable under U.S. or foreign laws, or they may be infringed by third parties. As a result, we have or may be from time to time involved in the defense and enforcement of our patent or other intellectual property rights in a court of law and administrative tribunals, such as in USPTO inter partes review or reexamination proceedings, foreign opposition proceedings or related legal and administrative proceedings in the United States and elsewhere. The costs of defending our patents or enforcing our proprietary rights in post-issuance administrative proceedings or litigation may be substantial and the outcome can be uncertain. An adverse outcome may allow third parties to use our proprietary technologies without a license from us.
Furthermore, we rely upon trade secrets and know-how and continuing technological innovation to develop and maintain our competitive position. We seek to protect our proprietary information, in part, by using confidentiality and invention assignment agreements with its commercial partners, collaborators, employees and consultants. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant it ownership of technologies that are developed through a relationship with a third party. These agreements may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our commercial partners, collaborators, employees and consultants use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
Our commercial success will also depend in part on not infringing upon the proprietary rights of third parties. It is uncertain whether the issuance of any third-party patent would require us to alter its development or commercial strategies for our product candidates or processes, or to obtain licenses or cease certain activities. Our breach of any license agreements or failure to obtain a license to proprietary rights that it may require to develop
101
or commercialize its future products may have an adverse impact on us. If third parties prepare and file patent applications in the United States that also claim technology to which we have rights, we may have to participate in interference or derivation proceedings in the USPTO to determine priority of invention.
As of June 30, 2023, we or our subsidiaries have registered and own “Opthea” as a trademark in ten jurisdictions, including the United States and Europe. Other than the registered trademark listed above, we currently rely on our unregistered trademarks, trade names and service marks, as well as our domain names and logos, as appropriate, to market our brands and to build and maintain brand recognition.
Government Regulation
The FDA and other regulatory authorities at federal, state, and local levels, as well as in non-U.S. countries, extensively regulate, among other things, the research, development, testing, manufacture, quality control, import, export, safety, effectiveness, labeling, packaging, storage, distribution, record-keeping, approval, advertising, promotion, marketing, post-approval monitoring, and post-approval reporting of biologics such as those we are developing. We, along with third-party contractors, will be required to navigate the various nonclinical, clinical and commercial approval requirements of the governing regulatory agencies of the countries in which we wish to conduct studies or seek approval or licensure of Sozinibercept (OPT-302) or any future product candidate.
U.S. Biological Product Development
In the United States, the FDA regulates biologics under both the Federal Food, Drug and Cosmetic Act and the Public Health Service Act and their implementing regulations. Both drugs and biologics also are subject to other federal, state and local statutes and regulations. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, and local statutes and regulations requires the expenditure of substantial time and financial resources. Failure to comply with the applicable U.S. requirements at any time during the product development process, approval process or post-market regulations may subject an applicant to administrative or judicial sanctions. These sanctions could include, among other actions, the FDA’s refusal to approve pending applications, withdrawal of an approval, license revocation, a clinical hold, untitled or warning letters, product recalls or market withdrawals, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement and civil or criminal penalties. Any agency or judicial enforcement action could have a material adverse effect on us.
Our product candidates and any future biological product candidates we develop must be approved by the FDA through a biologics license application, or BLA, process before they may be legally marketed in the United States. The BLA is a request for approval to market the biologic for one or more specified indications and must contain proof of safety, purity and potency. The FDA review and approval process generally involves the following:
102
Nonclinical Studies and IND
Preclinical studies include laboratory evaluation of product chemistry and formulation, as well as in vitro and animal studies to assess the potential for adverse events and in some cases to establish a rationale for therapeutic use. The conduct of preclinical studies is subject to federal regulations and requirements, including GLP regulations for safety/toxicology studies.
Prior to beginning the first clinical trial with a product candidate in the United States, we must submit an IND to the FDA. An IND is a request for authorization from the FDA to administer an investigational new drug product to humans. The central focus of an IND submission is on the general investigational plan and the protocol(s) for clinical studies. The IND also includes results of animal and in vitro studies assessing the toxicology, PK, pharmacology, and PD characteristics of the product candidate; chemistry, manufacturing, and controls information; and any available human data or literature to support the use of the investigational product. An IND must become effective before human clinical trials may begin. The IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, raises safety concerns or questions about the proposed clinical trial. In such a case, the IND may be placed on clinical hold until the IND sponsor and the FDA resolve the outstanding concerns or questions. Submission of an IND therefore may or may not result in FDA authorization to begin a clinical trial.
Clinical Trials
Clinical trials involve the administration of the investigational product to human subjects under the supervision of qualified investigators in accordance with GCP, which include the requirement that all research subjects provide their informed consent for their participation in any clinical study. Clinical trials are conducted under protocols detailing, among other things, the objectives of the study, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. A separate submission to the existing IND must be made for each successive clinical trial conducted during product development and for any subsequent protocol amendments. For new indications, a separate new IND may be required. Furthermore, an independent IRB for each site proposing to conduct the clinical trial must review and approve the plan for any clinical trial and its informed consent form before the clinical trial begins at that site and must monitor the study until completed. Regulatory authorities, the IRB or the sponsor may suspend a clinical trial at any time on various grounds, including a finding that the subjects are being exposed to an unacceptable health risk or that the trial is unlikely to meet its stated objectives. Some studies also include oversight by an independent group of qualified experts organized by the clinical study sponsor, known as a data safety monitoring board, which provides authorization for whether or not a study may move forward at designated check points based on access to certain data from the study and may halt the clinical trial if it determines that there is an unacceptable safety risk for subjects or other grounds, such as no demonstration of efficacy. There are also requirements governing the reporting of ongoing clinical studies and clinical study results to public registries. For purposes of BLA approval, human clinical trials are typically conducted in three sequential phases that may overlap.
A sponsor who wishes to conduct a clinical trial outside of the United States may, but need not, obtain FDA authorization to conduct the clinical trial under an IND. If a foreign clinical trial is not conducted under an IND, the sponsor may submit data from the clinical trial to the FDA in support of a BLA. The FDA will accept a well-designed and well-conducted foreign clinical study not conducted under an IND if the study was conducted in accordance with GCP requirements, and the FDA is able to validate the data through an onsite inspection if deemed necessary.
103
In some cases, the FDA may require, or companies may voluntarily pursue, additional clinical trials after a product is approved to gain more information about the product. These so-called Phase 4 studies may be made a condition to approval of the BLA. Concurrent with clinical trials, companies may complete additional animal studies and develop additional information about the biological characteristics of the product candidate and must finalize a process for manufacturing the product in commercial quantities in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the product candidate and, among other things, must develop methods for testing the identity, strength, quality and purity of the final product, or for biologics, the safety, purity and potency. Additionally, appropriate packaging must be selected and tested and stability studies must be conducted to demonstrate that the product candidate does not undergo unacceptable deterioration over its shelf life.
BLA Submission, Review and Approval
Assuming successful completion of the clinical trials, the results of product development, nonclinical studies and clinical trials are submitted to the FDA as part of a BLA requesting approval to market the product for one or more indications. The BLA must include all relevant data available from pertinent preclinical and clinical studies, including negative or ambiguous results as well as positive findings, together with detailed information relating to the product’s chemistry, manufacturing, controls, and proposed labeling, among other things. The submission of a BLA requires payment of a substantial application user fee to FDA, unless a waiver or exemption applies.
Once a BLA has been submitted, the FDA’s goal is to review standard applications within ten months after it accepts the application for filing (a 60-day process), or, if the application qualifies for priority review, six months after the FDA accepts the application for filing. In both standard and priority reviews, the review process can be significantly extended by FDA requests for additional information or clarification. The FDA reviews a BLA to determine, among other things, whether a product is safe, pure and potent and the facility in which it is manufactured, processed, packed, or held meets standards designed to assure the product’s continued safety, purity and potency. The FDA may convene an advisory committee to provide clinical insight on application review questions. Before approving a BLA, the FDA will typically inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving a BLA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. If the FDA determines that the application, manufacturing process or manufacturing facilities are not acceptable, it will outline the deficiencies in the submission and often will request additional testing or information. Notwithstanding the submission of any requested additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval.
After the FDA evaluates a BLA and conducts inspections of manufacturing facilities where the investigational product and/or its drug substance will be produced, the FDA may issue an approval letter or a Complete Response letter. An approval letter authorizes commercial marketing of the product with specific prescribing information for specific indications. A Complete Response letter will describe all of the deficiencies that the FDA has identified in the BLA, except that where the FDA determines that the data supporting the application are inadequate to support approval, the FDA may issue the Complete Response letter without first conducting required inspections, testing submitted product lots, and/or reviewing proposed labeling. In issuing the Complete Response letter, the FDA may recommend actions that the applicant might take to place the BLA in condition for approval, including requests for additional information or clarification. The FDA may delay or refuse approval of a BLA if
104
applicable regulatory criteria are not satisfied, require additional testing or information and/or require post-marketing testing and surveillance to monitor safety or efficacy of a product.
If regulatory approval of a product is granted, such approval will be granted for particular indications and may entail limitations on the indicated uses for which such product may be marketed. For example, the FDA may approve the BLA with a Risk Evaluation and Mitigation Strategy, or REMS, to ensure the benefits of the product outweigh its risks. A REMS is a safety strategy to manage a known or potential serious risk associated with a product and to enable patients to have continued access to such medicines by managing their safe use, and could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. The FDA also may condition approval on, among other things, changes to proposed labeling or the development of adequate controls and specifications. Once approved, the FDA may withdraw the product approval if compliance with pre- and post-marketing requirements is not maintained or if problems occur after the product reaches the marketplace. The FDA may require one or more Phase 4 post-market studies and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization, and may limit further marketing of the product based on the results of these post-marketing studies.
Expedited Development and Review Programs
Any marketing application for a biologic submitted to the FDA for approval may be eligible for FDA programs intended to expedite the FDA review and approval process, such as priority review, fast track designation, breakthrough therapy and accelerated approval.
A product is eligible for priority review if it has the potential to provide safe and effective therapy where no satisfactory alternative therapy exists or to provide a significant improvement in the treatment, diagnosis or prevention of a serious disease or condition compared to marketed products. For products containing new molecular entities, priority review designation means the FDA’s goal is to take action on the marketing application within six months of the 60-day filing date (compared with ten months under standard review). In July 2021, Sozinibercept (OPT-302) was granted Fast Track Designation by the FDA for the treatment of wet AMD.
In addition, a sponsor can request designation of a product candidate as a “breakthrough therapy.” A breakthrough therapy is defined as a drug or biologic that is intended, alone or in combination with one or more other drugs or biologics, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug or biologic may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. Drugs or biologics designated as breakthrough therapies are also eligible for accelerated approval. The FDA must take certain actions, such as holding timely meetings and providing advice, intended to expedite the development and review of an application for approval of a breakthrough therapy. The benefits of breakthrough therapy designation include the same benefits as fast track designation, plus intensive guidance from the FDA to ensure an efficient drug development program.
Additionally, products studied for their safety and effectiveness in treating serious or life-threatening diseases or conditions may receive accelerated approval upon a determination that the product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. As a condition of accelerated approval, the FDA will generally require the sponsor to perform adequate and well-controlled post-marketing clinical studies to verify and describe the anticipated effect on irreversible morbidity or mortality or other clinical benefit. In addition, the FDA currently requires as a condition for accelerated approval pre-approval of promotional materials, which could adversely impact the timing of the commercial launch of the product.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or decide that the time period for FDA review and approval will not be shortened. Furthermore, priority review, fast track designation, breakthrough therapy designation, and accelerated approval do not change the standards for approval but may expedite the development or approval process.
105
Orphan Drug Designation
Under the Orphan Drug Act, the FDA may grant orphan designation to a drug or biologic intended to treat a rare disease or condition, which is a disease or condition that affects fewer than 200,000 individuals in the United States, or 200,000 or more individuals in the United States for which there is no reasonable expectation that the cost of developing and making available in the United States a drug or biologic for this type of disease or condition will be recovered from sales in the United States for that drug or biologic. Orphan designation must be requested before submitting a BLA. After the FDA grants orphan designation, the generic identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. The orphan drug designation does not convey any advantage in, or automatically shorten the duration of, the regulatory review or approval process.
If a product that has orphan designation subsequently receives the first FDA approval for the disease for which it has such designation, the product is entitled to orphan exclusivity, which means that the FDA may not approve any other applications, including a full BLA, to market the same product for the same indication for seven years, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity. Orphan exclusivity does not prevent FDA from approving a different drug or biologic for the same disease or condition, or the same drug or biologic for a different disease or condition. Among the other benefits of orphan drug designation are tax credits for certain research and a waiver of the BLA application fee. A designated orphan product may not receive orphan exclusivity if it is approved for a use that is broader than the indication for which it received orphan designation. In addition, exclusive marketing rights in the United States may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition.
Post-Approval Requirements
Any products manufactured or distributed by us pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to quality control and quality assurance, record-keeping, reporting of adverse experiences, periodic reporting, product sampling and distribution, and advertising and promotion of the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims, are subject to prior FDA review and approval. There also are continuing user fee requirements, under which FDA assesses an annual program fee for each product identified in an approved BLA. Biologic manufacturers and their subcontractors are required to register their establishments with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with cGMP, which impose certain procedural and documentation requirements upon us and our third-party manufacturers. Changes to the manufacturing process are strictly regulated, and, depending on the significance of the change, may require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP and impose reporting requirements upon us and any third-party manufacturers that we may decide to use. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain compliance with cGMP and other aspects of regulatory compliance.
The FDA may withdraw approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including AEs of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical studies to assess new safety risks; or imposition of distribution restrictions or other restrictions under a REMS program. Other potential consequences include, among other things:
106
The FDA closely regulates the marketing, labeling, advertising and promotion of biologics. A company can make only those claims relating to safety and efficacy, purity and potency that are approved by the FDA and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses. Failure to comply with these requirements can result in, among other things, adverse publicity, warning letters, corrective advertising and potential civil and criminal penalties. Physicians may prescribe legally available products for uses that are not described in the product’s labeling and that differ from those tested by us and approved by the FDA. The FDA does not regulate the behavior of physicians in their choice of treatments. The FDA does, however, restrict manufacturer’s communications on the subject of off-label use of their products.
Biosimilars and Reference Product Exclusivity
The Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, or Affordable Care Act signed into law in 2010, includes a subtitle called the Biologics Price Competition and Innovation Act of 2009, which created an abbreviated approval pathway for biological products that are biosimilar to or interchangeable with an FDA-approved reference biological product. To date, a number of biosimilars have been licensed under the BPCIA, and numerous biosimilars have been approved in Europe. The FDA has issued several guidance documents outlining its approach to the review and approval of biosimilars.
Biosimilarity, which requires that there be no clinically meaningful differences between the biological product and the reference product in terms of safety, purity, and potency, can be shown through analytical studies, animal studies, and a clinical study or studies. Interchangeability requires that a product is biosimilar to the reference product and the product must demonstrate that it can be expected to produce the same clinical results as the reference product in any given patient and, for products that are administered multiple times to an individual, the biologic and the reference biologic may be alternated or switched after one has been previously administered without increasing safety risks or risks of diminished efficacy relative to exclusive use of the reference biologic. Complexities associated with the larger, and often more complex, structures of biological products, as well as the processes by which such products are manufactured, pose significant hurdles to implementation of the abbreviated approval pathway that are still being worked out by the FDA.
Under the BPCIA, an application for a biosimilar product may not be submitted to the FDA until four years following the date that the reference product was first licensed by the FDA. In addition, the approval of a biosimilar product may not be made effective by the FDA until 12 years from the date on which the reference product was first licensed. During this 12-year period of exclusivity, another company may still market a competing version of the reference product if the FDA approves a full BLA for the competing product containing that applicant’s own preclinical data and data from adequate and well-controlled clinical trials to demonstrate the safety, purity and potency of its product. The BPCIA also created certain exclusivity periods for biosimilars approved as interchangeable products. At this juncture, it is unclear whether products deemed “interchangeable” by the FDA will, in fact, be readily substituted by pharmacies, which are governed by state pharmacy law.
The BPCIA is complex and continues to be interpreted and implemented by the FDA. In addition, government proposals have sought to reduce the 12-year reference product exclusivity period. Other aspects of the BPCIA, some of which may impact the BPCIA exclusivity provisions, have also been the subject of recent litigation. As a result, the ultimate impact, implementation, and impact of the BPCIA is subject to significant uncertainty.
Other U.S. Healthcare Laws and Compliance Requirements
In the United States, our current and future operations are subject to regulation by various federal, state and local authorities in addition to the FDA, including but not limited to, the Centers for Medicare and Medicaid Services, or CMS other divisions of the U.S. Department of Health and Human Services, or HHS (such as the Office of Inspector General, Office for Civil Rights and the Health Resources and Service Administration), the U.S. Department of Justice, or DOJ, and individual U.S. Attorney offices within the DOJ, and state and local governments. For example, our clinical research, sales, marketing and scientific/educational grant programs may have to comply with the anti-fraud and abuse provisions of the Social Security Act, the false claims laws, transparency and price reporting laws, the privacy and security provisions of the Health Insurance Portability and Accountability Act, or HIPAA and similar state laws, each as amended, as applicable. Our business operations
107
and current and future arrangements with investigators, healthcare professionals, consultants, third-party payors and customers may be subject to healthcare laws, regulations and enforcement by the federal government and by authorities in the states and foreign jurisdictions in which we conduct our business. Such laws include, without limitation, state and federal anti-kickback, fraud and abuse, false claims, privacy and security, price reporting, and transparency laws. Some of our pre-commercial activities are subject to some of these laws.
The federal Anti-Kickback Statute prohibits, among other things, any person or entity, from knowingly and willfully offering, paying, soliciting or receiving any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or in return for purchasing, leasing, ordering or arranging for the purchase, lease or order of any item or service reimbursable, in whole or in part, under Medicare, Medicaid or other federal healthcare programs. The term remuneration has been interpreted broadly to include anything of value. The Anti-Kickback Statute has been interpreted to apply to arrangements between therapeutic product manufacturers on one hand and prescribers, purchasers, and formulary managers on the other. There are a number of statutory exceptions and regulatory safe harbors protecting some common activities from prosecution. The exceptions and safe harbors are drawn narrowly and practices that involve remuneration that may be alleged to be intended to induce prescribing, purchasing or recommending may be subject to scrutiny if they do not qualify for an exception or safe harbor. Failure to meet all of the requirements of a particular applicable statutory exception or regulatory safe harbor does not make the conduct per se illegal under the Anti-Kickback Statute. Instead, the legality of the arrangement will be evaluated on a case-by-case basis based on a cumulative review of all of its facts and circumstances. Our practices may not in all cases meet all of the criteria for protection under a statutory exception or regulatory safe harbor.
Additionally, the intent standard under the Anti-Kickback Statute was amended by the Affordable Care Act to a stricter standard such that a person or entity no longer needs to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. Further, courts have found that if “one purpose” of remuneration is to induce referrals, the federal Anti-Kickback statute is violated. Violations of the Anti-Kickback Statute can result in significant civil and criminal fines and penalties for each violation, imprisonment, and exclusion from federal healthcare programs. In addition, the Affordable Care Act codified case law that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal False Claims Act, or the FCA, as discussed below.
The federal false claims laws, including the FCA, which can be enforced by private citizens through civil qui tam actions, and civil monetary penalty laws, prohibit any person or entity from, among other things, knowingly presenting, or causing to be presented, a false or fraudulent claim for payment to, or approval by, the federal government, including federal healthcare programs, such as Medicare and Medicaid, knowingly making, using, or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government, or knowingly making a false statement to improperly avoid, decrease or conceal an obligation to pay money to the federal government. A claim includes “any request or demand” for money or property presented to the U.S. government. Pharmaceutical and other healthcare companies have been, and continue to be, prosecuted under these laws, among other things, for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product and for causing false claims to be submitted because of the companies’ marketing of the product for unapproved, off-label, and thus generally non-reimbursable, uses.
HIPAA created additional federal criminal statutes that prohibit, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud or to obtain, by means of false or fraudulent pretenses, representations or promises, any money or property owned by, or under the control or custody of, any healthcare benefit program, including private third-party payors, willfully obstructing a criminal investigation of a healthcare offense, and knowingly and willfully falsifying, concealing or covering up by trick, scheme or device, a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services. Like the Anti-Kickback Statute, the Affordable Care Act amended the intent standard for certain healthcare fraud statutes under HIPAA such that a person or entity no longer needs to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. When an entity is determined to have violated the FCA, the government may impose civil fines and penalties for each false claim, plus treble damages, and exclude the entity from participation in Medicare, Medicaid and other federal healthcare programs.
We may be subject to data privacy and security regulations by both the federal government and the states in which we conduct our business. HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and its implementing regulations, imposes requirements on certain covered healthcare providers, health plans, and healthcare clearinghouses as well as their respective business associates
108
and their covered subcontractors relating to the privacy, security and transmission of individually identifiable health information. Among other things, HITECH makes HIPAA’s privacy and security standards directly applicable to business associates, which are independent contractors or agents of covered entities that create, maintain, transmit, receive or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also created four new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce HIPAA and seek attorneys’ fees and costs associated with pursuing federal civil actions. In addition, many state laws govern the privacy and security of health information in specified circumstances, many of which differ from each other in significant ways, are often not pre-empted by HIPAA, and may have a more prohibitive effect than HIPAA, thus complicating compliance efforts.
Additionally, the federal Physician Payments Sunshine Act, or the Sunshine Act, within the Affordable Care Act, and its implementing regulations, require that certain manufacturers of drugs, devices, biological and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions) report annually to CMS information related to certain payments or other transfers of value made or distributed to physicians (defined to include (doctors, dentists, optometrists, podiatrists and chiropractors) certain other healthcare professionals (such as physicians assistants and nurse practitioners) and teaching hospitals, or to entities or individuals at the request of, or designated on behalf of, the physicians and teaching hospitals and to report annually certain ownership and investment interests held by physicians and their immediate family members. Failure to report accurately could result in penalties.
Many states have similar statutes or regulations to the above federal laws that may be broader in scope and may apply regardless of payor. We may also be subject to state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government, and/or state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers, drug pricing or marketing expenditures. Certain state and local laws also require the registration of pharmaceutical sales representatives. We may be subject to state and foreign laws governing the privacy and security of health information, some of which may be more stringent than those in the United States (such as the GDPR, which was adopted by the EU and subsequently became effective in May 2018). These laws may differ from each other in significant ways and may not have the same effect, further complicating compliance efforts.
In order to distribute products commercially, we must comply with state laws that require the registration of manufacturers and wholesale distributors of drug and biological products in a state, including, in certain states, manufacturers and distributors who ship products into the state even if such manufacturers or distributors have no place of business within the state. Some states also impose requirements on manufacturers and distributors to establish the pedigree of product in the chain of distribution, including some states that require manufacturers and others to adopt new technology capable of tracking and tracing product as it moves through the distribution chain. Several states have enacted legislation requiring pharmaceutical and biotechnology companies to establish marketing compliance programs, file periodic reports with the state, make periodic public disclosures on sales, marketing, pricing, clinical trials and other activities, and/or register their sales representatives, as well as to prohibit pharmacies and other healthcare entities from providing certain physician prescribing data to pharmaceutical and biotechnology companies for use in sales and marketing, and to prohibit certain other sales and marketing practices. All of our activities are potentially subject to federal and state consumer protection and unfair competition laws. Additionally, to the extent that we have business operations in foreign countries or sell any of our products in foreign countries and jurisdictions, including Canada or the EU, we may be subject to additional regulation.
Ensuring business arrangements with third parties comply with applicable healthcare laws and regulations is a costly endeavor. If our operations are found to be in violation of any of the federal and state healthcare laws described above or any other current or future governmental regulations that apply to us, we may be subject to penalties, including without limitation, significant civil, criminal and/or administrative penalties, damages, fines, disgorgement, imprisonment, exclusion from participation in government programs, such as Medicare and Medicaid, injunctions, private “qui tam” actions brought by individual whistleblowers in the name of the government, or refusal to allow us to enter into government contracts, contractual damages, reputational harm, administrative burdens, diminished profits and future earnings, additional reporting obligations and oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of non-compliance with these laws, and the curtailment or restructuring of our operations, any of which could adversely affect its ability to operate our business and results of operations. Any action against us for violation of these laws, even if
109
we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. The shifting compliance environment and the need to build and maintain robust and expandable systems to comply with multiple jurisdictions with different compliance or reporting requirements increases the possibility that a healthcare company may run afoul of one or more of the requirements.
Coverage, Pricing and Reimbursement in the U.S.
Significant uncertainty exists as to the coverage and reimbursement status of any product candidates for which we may obtain regulatory approval. In the United States and in foreign markets, sales of any products for which we receive regulatory approval for commercial sale will depend, in part, on the extent to which third-party payors provide coverage and establish adequate reimbursement levels for such products. In the United States, third-party payors include federal and state healthcare programs, private managed care providers, health insurers and other organizations. Third-party payors decide which medications they will pay for and establish reimbursement levels. Adequate coverage and reimbursement from governmental healthcare programs, such as Medicare and Medicaid in the United States, and commercial payors are critical to new product acceptance.
Our ability to commercialize any products successfully also will depend in part on the extent to which coverage and reimbursement for these products and related treatments will be available from third-party payors, which decide which therapeutics they will pay for and establish reimbursement levels. In the United States, the principal decisions about reimbursement for new medicines are typically made by CMS, an agency within HHS. CMS decides whether and to what extent our products will be covered and reimbursed under Medicare and private payors tend to follow CMS to a substantial degree. Coverage and reimbursement by a third-party payor may depend upon a number of factors, including the third-party payor’s determination that use of a therapeutic is:
We cannot be sure that coverage or reimbursement will be available for any product that we commercialize and, if coverage and reimbursement are available, what the level of reimbursement will be. Coverage may also be more limited than the purposes for which the product is approved by the FDA or comparable foreign regulatory authorities. Reimbursement may impact the demand for, or the price of, any product for which we obtain regulatory approval.
We may develop products that, once approved, may be administered by a physician. For products administered under the supervision of a physician, obtaining coverage and adequate reimbursement may be particularly difficult because of the higher prices often associated with such drugs. Additionally, separate reimbursement for the product itself or the treatment or procedure in which the product is used may not be available, which may impact physician utilization.
Third-party payors are increasingly challenging the price, examining the medical necessity, and reviewing the cost-effectiveness of medical products, therapies and services, in addition to questioning their safety and efficacy. Obtaining reimbursement for our products may be particularly difficult because of the higher prices often associated with branded drugs and drugs administered under the supervision of a physician. We may need to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost-effectiveness of our products, in addition to the costs required to obtain FDA approvals. Our product candidates may not be considered medically necessary or cost-effective. Obtaining coverage and reimbursement approval of a product from a government or other third-party payor is a time-consuming and costly process that could require us to provide to each payor supporting scientific, clinical and cost-effectiveness data for the use of our product on a payor-by-payor basis, with no assurance that coverage and adequate reimbursement will be obtained. A payor’s decision to provide coverage for a product does not imply that an adequate reimbursement rate will be approved. Further, one payor’s determination to provide coverage for a product does not assure that other payors will also
110
provide coverage for the product. Adequate third-party reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on its investment in product development. If reimbursement is not available or is available only at limited levels, we may not be able to successfully commercialize any product candidate that we successfully develop.
The marketability of any product candidates for which we receive regulatory approval for commercial sale may suffer if the government and third-party payors fail to provide adequate coverage and reimbursement. In addition, emphasis on managed care, the increasing influence of health maintenance organizations, and additional legislative changes in the United States has increased, and we expect will continue to increase, the pressure on healthcare pricing. The downward pressure on the rise in healthcare costs in general, particularly prescription medicines, medical devices and surgical procedures and other treatments, has become very intense. Coverage policies and third-party reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more products for which we receive regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
U.S. Healthcare Reform
In the United States and some foreign jurisdictions, there have been, and continue to be, several legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of product candidates, restrict or regulate post-approval activities, and affect the ability to profitably sell product candidates for which marketing approval is obtained. Among policy makers and payors in the United States and elsewhere, there is significant interest in promoting changes in healthcare systems with the stated goals of containing healthcare costs, improving quality and/or expanding access. In the United States, the pharmaceutical industry has been a particular focus of these efforts and has been significantly affected by major legislative initiatives.
For example, the Affordable Care Act has substantially changed healthcare financing and delivery by both governmental and private insurers. The Affordable Care Act among other things contains a number of provisions, including those governing enrollment in federal healthcare programs, reimbursement adjustments and changes to fraud and abuse laws.
Since its enactment, there have been executive, legal and political challenges to certain aspects of the Affordable Care Act. By way of example, the Tax Cuts and Jobs Act, or TCJA, included a provision that repealed, effective January 1, 2019, the tax-based shared responsibility payment imposed by the Affordable Care Act on certain individuals who fail to maintain qualifying health coverage for all or part of a year that is commonly referred to as the “individual mandate.” On June 17, 2021 the U.S. Supreme Court dismissed a challenge on procedural grounds that argued the Affordable Care Act is unconstitutional in its entirety because the “individual mandate” was repealed by Congress. On August 16, 2022, President Biden signed the IRA, into law, which among other things, extends enhanced subsidies for individuals purchasing health insurance coverage in Affordable Care Act marketplaces through plan year 2025. The IRA also eliminates the “donut hole” under the Medicare Part D program beginning in 2025 by significantly lowering the beneficiary maximum out-of-pocket cost through a newly established manufacturer discount program. It is possible that the Affordable Care Act will be subject to judicial or Congressional challenges in the future. It is unclear how any such challenges and the healthcare reform measures of the Biden administration will impact the Affordable Care Act.
Other legislative changes have been proposed and adopted since the Affordable Care Act was enacted. For example, in August 2011, President Obama signed into law the Budget Control Act of 2011, which, among other things, included aggregate reductions to Medicare payments to providers of 2% per fiscal year, which went into effect beginning on April 1, 2013 and will stay in effect until 2032 unless additional Congressional action is taken. In January 2013, the American Taxpayer Relief Act of 2012 was signed into law, which, among other things, further reduced Medicare payments to several types of providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years.
Additionally, there has been increasing legislative and enforcement interest in the United States with respect to specialty drug pricing practices. Specifically, there have been several recent U.S. presidential executive orders, Congressional inquiries and proposed and enacted federal legislation designed to, among other things, bring more transparency to drug pricing, reduce the cost of prescription drugs under Medicare, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drugs. At the federal level, in July 2021, the Biden administration released an executive order,
111
“Promoting Competition in the American Economy,” with multiple provisions aimed at prescription drugs. In response to Biden’s executive order, on September 9, 2021, HHS released a Comprehensive Plan for Addressing High Drug Prices that outlines principles for drug pricing reform and sets out a variety of potential legislative policies that Congress could pursue as well as potential administrative actions HHS can take to advance these principles. In addition, the IRA directs the HHS Secretary to establish a Drug Price Negotiation Program (the “Program”) to lower prices for certain high-expenditure, single-source prescription drugs and biologics covered under Medicare Part B and Part D that have been approved by the FDA for at least 7 years for prescription drugs and at least 11 years for biologics. Under the Program, the HHS Secretary will publish a list of “selected drugs,” and will then negotiate maximum fair prices (“MFP”) with their manufacturers. The Program will be implemented in stages. Beginning in 2026, 10 Medicare Part D “selected drugs” will be subject to price negotiations. By 2029, and in subsequent years thereafter, the number will increase to 20 drugs and biologics covered under Medicare Part B and Part D. Agreements between HHS and manufacturers will remain in place until a drug or biologic is no longer considered a “selected drug” for negotiation purposes. Manufacturers who do not comply with the negotiated prices set under the Program will be subject to an excise tax based on a percentage of total sales of a “selected drug” up to 95% and potential civil monetary penalties. Further, beginning in October 2023, the IRA will require manufacturers that increase prices of certain Medicare Part B and Part D drugs or biologics at a rate greater than inflation to pay rebates to CMS or be subject to civil monetary penalties. HHS has and will continue to issue and update guidance as these programs are implemented, although the IRA may be subject to legal challenges. It is currently unclear how the IRA will be implemented but is likely to have significant impact on the pharmaceutical industry. In addition, in response to the Biden administration's October 2022 executive order, on February 14, 2023, HHS released a report outlining three new models for testing by the Center for Medicare and Medicaid Innovation which will be evaluated on their ability to lower the cost of drugs, promote accessibility, and improve quality of care. It is unclear whether the models will be utilized in any health reform measures in the future.
Individual states in the United States have also become increasingly active in passing legislation and implementing regulations designed to control biopharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. Legally mandated price controls on payment amounts by third-party payors or other restrictions on coverage or access could harm our business, results of operations, financial condition and prospects. In addition, regional healthcare authorities and individual hospitals are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription drug and other healthcare programs. This could reduce the ultimate demand for our product candidates that we successfully commercialize or put pressure on our product pricing.
We expect that these and other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and lower reimbursement, and in additional downward pressure on the price that we receive for any approved product. Any reduction in reimbursement from Medicare and other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize our products. Such reforms could have an adverse effect on anticipated revenue from product candidates that we may successfully develop and for which we may obtain regulatory approval and may affect our overall financial condition and ability to develop product candidates.
European Union Product Approval Process
Non-Clinical Studies and Clinical Trials
Similar to the United States, the various phases of non-clinical research in the EU are subject to significant regulatory controls.
Non-clinical studies are performed to demonstrate the health or environmental safety of new chemical or biological substances. In particular, non-clinical studies, both in vitro and in vivo, must be planned, performed, monitored, recorded, reported, and archived in accordance with the GLP principles, as set forth in Directive 2004/10/EC, which define a set of rules and criteria for a quality system for the organizational process and the
112
conditions for non-clinical studies. These GLP standards reflect the Organization for Economic Co-operation and Development requirements.
The Clinical Trials Directive 2001/20/EC, the Directive 2005/28/EC on Good Clinical Practice, or GCP, and the related national implementing provisions of the individual EU member states currently govern the system for the approval of clinical trials in the EU.
Clinical trials of medicinal products in the EU must be conducted in accordance with EU and national regulations of EU members states and the International Conference on Harmonization, or ICH, guidelines on GCP, as well as the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki. If the sponsor of the clinical trial is not established within the EU, it must appoint an EU entity to act as its legal representative. The sponsor must take out a clinical trial insurance policy, and increasingly in EU member states, the sponsor is liable to provide “no fault” compensation to any study subject injured in the clinical trial.
Under the applicable regulatory system, an applicant must obtain prior approval from the competent national authority of the EU member states in which the clinical trial is to be conducted. Furthermore, the applicant may only start a clinical trial at a specific study site after the competent ethics committee has issued a related favorable opinion. The application for authorization of a clinical trial must be accompanied by, among other documents, a copy of the trial protocol and an investigational medicinal product dossier containing information about the manufacture and quality of the medicinal product under investigation as prescribed by Directive 2001/20/EC, Directive 2005/28/EC, where relevant the related implementing national provisions of the individual EU member states, and further detailed in applicable guidance documents. Any substantial changes to the trial protocol or to other information submitted with the clinical trial application must be notified to and approved by the relevant competent national authorities and ethics committees. Medicinal products used in clinical trials must be manufactured in accordance with good manufacturing practice (GMP).
In April 2014, the new Clinical Trials Regulation, (EU) No 536/2014 (Clinical Trials Regulation) was adopted. The Regulation came into effect in the EU on January 31, 2022. The Clinical Trials Regulation is directly applicable in all the EU member states, repealing the Clinical Trials Directive 2001/20/EC. A three-year transition period is in place to move from the previous European Communities (Clinical Trials on Medicinal Products for Human Use) Regulations, SI No 190 of 2004 which transposed into law the provision of Council Directive 2001/20/EC. The transition period sets out the following key timelines:
The new Clinical Trials Regulation aims to simplify and streamline the approval of clinical trials in the EU. The main characteristics of the regulation include: a streamlined application procedure via a single-entry point, the "EU portal"; a single set of documents to be prepared and submitted for the application as well as simplified reporting procedures for clinical trial sponsors; and a harmonized procedure for the assessment of applications for clinical trials, which is divided in two parts. Part I is assessed first by a single “reference” member state whose conclusions are then assessed by the competent authorities of all EU member states in which an application for authorization of a clinical trial has been submitted to “concerned” member states. Part II is assessed separately by each concerned EU member state. Strict deadlines have been established for the assessment of clinical trial applications. The role of the relevant ethics committees in the assessment procedure will continue to be governed by the national law of the concerned EU member state. However, overall related timelines will be defined by the
113
Clinical Trials Regulation, which provides Sponsors a reliable timeline to evaluate the approval of the clinical trial throughout Europe.
Marketing Authorizations
To obtain a marketing authorization, or MA, for a product in the EU, an applicant must submit a Marketing Authorization Application, or MAA, either under a centralized procedure administered by the European Medicines Agency, or EMA, or one of the procedures administered by competent authorities in the EU member states (decentralized procedure, national procedure, or mutual recognition procedure). An MA may be granted only to an applicant established in the EU.
The centralized procedure provides for the grant of a single MA by the European Commission that is valid for all EU member states. Pursuant to Regulation (EC) No 726/2004, the centralized procedure is compulsory for specific products, including for (i) medicinal products derived from biotechnological processes, (ii) products designated as orphan medicinal products, (iii) advanced therapy medicinal products (ATMPs), and (iv) products with a new active substance indicated for the treatment of HIV/AIDS, cancer, neurodegenerative diseases, diabetes, auto-immune and other immune dysfunctions, and viral diseases. For products with a new active substance indicated for the treatment of other diseases and products that are highly innovative or for which a centralized process is in the interest of patients, the centralized procedure may be optional on the related approval of the EMA.
Under the centralized procedure, the EMA’s Committee for Medicinal Products for Human Use (CHMP) is responsible for conducting the assessment of a product. The CHMP is also responsible for several post-authorization and maintenance activities, such as the assessment of modifications or extensions to an existing MA.
Under the centralized procedure in the EU, the maximum timeframe for the evaluation of an MAA is 210 days, excluding clock stops when additional information or written or oral explanation is to be provided by the applicant in response to questions of the CHMP. Accelerated assessment may be granted by the CHMP in exceptional cases, when a medicinal product targeting an unmet medical need is expected to be of major interest from the point of view of public health and in particular from the viewpoint of therapeutic innovation. If the CHMP accepts a request for accelerated assessment, the time limit of 210 days will be reduced to 150 days (not including clock stops). The CHMP can, however, revert to the standard time limit for the centralized procedure if it considers that it is no longer appropriate to conduct an accelerated assessment.
Unlike the centralized authorization procedure, the decentralized MA procedure requires a separate application to, and leads to separate approval by, the competent authorities of each EU member state in which the product is to be marketed. This application is identical to the application that would be submitted to the EMA for authorization through the centralized procedure. The reference EU member state prepares a draft assessment and drafts of the related materials within 120 days after receipt of a valid application. The resulting assessment report is submitted to the concerned EU member states who, within 90 days of receipt, must decide whether to approve the assessment report and related materials. If a concerned EU member state cannot approve the assessment report and related materials due to concerns relating to a potential serious risk to public health, disputed elements may be referred to the Heads of Medicines Agencies’ Coordination Group for Mutual Recognition and Decentralized Procedures – Human (CMDh) for review. The subsequent decision of the European Commission is binding on all EU member states.
The mutual recognition procedure allows companies that have a medicinal product already authorized in one EU member state to apply for this authorization to be recognized by the competent authorities in other EU member states. Like the decentralized procedure, the mutual recognition procedure is based on the acceptance by the competent authorities of the EU member states of the MA of a medicinal product by the competent authorities of other EU member states. The holder of a national MA may submit an application to the competent authority of an EU member state requesting that this authority recognize the MA delivered by the competent authority of another EU member state.
In principle, an MA has an initial validity of five years. The MA may be renewed after five years on the basis of a re-evaluation of the risk-benefit balance by the EMA or by the competent authority of the EU member state in which the original MA was granted. To support the application, the MA holder must provide the EMA or the competent authority with a consolidated version of the eCTD (Common Technical Document) providing up-to-date data concerning the quality, safety and efficacy of the product, including all variations introduced since the MA was granted, at least nine months before the MA ceases to be valid. The European Commission or the
114
competent authorities of the EU member states may decide on justified grounds relating to pharmacovigilance, to proceed with one further five-year renewal period for the MA. Once subsequently definitively renewed, the MA shall be valid for an unlimited period. Any authorization which is not followed by the actual placing of the medicinal product on the EU market (in case of centralized procedure) or on the market of the authorizing EU member state within three years after authorization ceases to be valid (the so-called sunset clause).
Innovative products that target an unmet medical need and are expected to be of major public health interest may be eligible for a number of expedited development and review programs, such as the Priority Medicines, or PRIME, scheme, which provides incentives similar to the breakthrough therapy designation in the U.S. PRIME is a voluntary scheme aimed at enhancing the EMA’s support for the development of medicinal products that target unmet medical needs. It permits increased interaction and early dialogue with companies developing promising medicinal products, to optimize their product development plans and speed up their evaluation to help the product reach patients earlier than normal. Product developers that benefit from PRIME designation are potentially eligible for accelerated assessment of their MAA although this is not guaranteed. Benefits accrue to sponsors of product candidates with PRIME designation, including but not limited to, early and proactive regulatory dialogue with the EMA, frequent discussions on clinical trial designs and other development program elements, and potentially accelerated MAA assessment once a dossier has been submitted.
In the EU, a “conditional” MA may be granted in cases where all the required safety and efficacy data are not yet available. The conditional MA is subject to conditions to be fulfilled for generating the missing data or ensuring increased safety measures. It is valid for one year and must be renewed annually until all related conditions have been fulfilled. Once any pending studies are provided, the conditional MA can be converted into a traditional MA. However, if the conditions are not fulfilled within the timeframe set by the EMA, the MA will cease to be renewed.
An MA may also be granted “under exceptional circumstances” where the applicant can show that it is unable to provide comprehensive data on efficacy and safety under normal conditions of use even after the product has been authorized and subject to specific procedures being introduced. These circumstances may arise in particular when the intended indications are very rare and, in the state of scientific knowledge at that time, it is not possible to provide comprehensive information, or when generating data may be contrary to generally accepted ethical principles. Like a conditional MA, an MA granted in exceptional circumstances is reserved to medicinal products intended to be authorized for treatment of rare diseases or unmet medical needs for which the applicable does not hold a complete data set that is required for the grant of a standard MA. However, unlike the conditional MA, an applicant for authorization in exceptional circumstances is not subsequently required to provide the missing data. Although the MA “under exceptional circumstances” is granted definitively, the risk-benefit balance of the medicinal product is reviewed annually, and the MA is withdrawn in case the risk-benefit ratio is no longer favorable.
In addition to an MA, various other requirements apply to the manufacturing and placing on the EU market of medicinal products. Manufacture of medicinal products in the EU requires a manufacturing authorization, and import of medicinal products into the EU requires a manufacturing authorization allowing for import. The manufacturing authorization holder must comply with various requirements set out in the applicable EU laws, regulations and guidance. These requirements include compliance with EU GMP standards when manufacturing medicinal products and APIs, including the manufacture of APIs outside of the EU with the intention to import the APIs into the Union. Similarly, the distribution of medicinal products within the EU is subject to compliance with the applicable EU laws, regulations and guidelines, including the requirement to hold appropriate authorizations for distribution granted by the competent authorities of the EU member states. MA holders and/or manufacturing and import authorization, or MIA holders and/or distribution authorization holders may be subject to civil, criminal or administrative sanctions, including suspension of manufacturing authorization, in case of non-compliance with the EU or EU member states’ requirements applicable to the manufacturing of medicinal products.
Data and Market Exclusivity
The EU provides opportunities for data and market exclusivity related to MAs. Upon receiving an MA, innovative medicinal products are generally entitled to receive eight years of data exclusivity and 10 years of market exclusivity. Data exclusivity, if granted, prevents regulatory authorities in the EU from referencing the innovator’s data to assess a generic application or biosimilar application for eight years from the date of authorization of the innovative product, after which a generic or biosimilar MAA can be submitted, and the innovator’s data may be referenced. The market exclusivity period prevents a successful generic or biosimilar
115
applicant from commercializing its product in the EU until 10 years have elapsed from the initial MA of the reference product in the EU. The overall ten-year period may, occasionally, be extended for a further year to a maximum of 11 years if, during the first eight years of those ten years, the MA holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are held to bring a significant clinical benefit in comparison with existing therapies. However, there is no guarantee that a product will be considered by the EU’s regulatory authorities to be a new chemical/biological entity, and products may not qualify for data exclusivity.
Orphan Medicinal Products
Regulation (EC) No. 141/2000, as implemented by Regulation (EC) No. 847/2000 provides that a medicinal product can be designated as an orphan medicinal product by the European Commission if its sponsor can establish that (1) the product is intended for the diagnosis, prevention or treatment of a life-threatening or chronically debilitating condition; (2) either (a) such condition affects no more than five in ten thousand persons in the EU when the application is made, or (b) the product, without the benefits derived from orphan status, would not generate sufficient return in the EU; and (3) there exists no satisfactory method of diagnosis, prevention or treatment of the condition in question that has been authorized in the EU or, if such method exists, the product will be of significant benefit to those affected by that condition.
In the EU, an application for designation as an orphan product can be made any time prior to the filing of the MAA. Orphan medicinal product designation entitles an applicant to incentives such fee reductions or fee waivers, protocol assistance, and access to the centralized MA procedure. Upon grant of an MA, orphan medicinal products are entitled to a ten-year period of exclusivity for the approved therapeutic indication, which means that the EMA cannot accept another MAA, or grant an MA, or accept an application to extend an MA for a similar product for the same indication for a period of ten years. The period of exclusivity is extended by two years for orphan medicinal products that have also complied with an agreed PIP. No extension to any supplementary protection certificate can be granted on the basis of pediatric studies for orphan indications. Orphan medicinal product designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process.
The period of exclusivity may, however, be reduced to six years if, at the end of the fifth year, it is established that the product no longer meets the criteria on the basis of which it received orphan medicinal product destination, including where it can be demonstrated on the basis of available evidence that the original orphan medicinal product is sufficiently profitable not to justify maintenance of market exclusivity or where the prevalence of the condition has increased above the threshold. Additionally, an MA may be granted to a similar medicinal product with the same orphan indication during the 10 year period if: (i) if the applicant consents to a second original orphan medicinal product application, (ii) if the manufacturer of the original orphan medicinal product is unable to supply sufficient quantities; or (iii) if the second applicant can establish that its product, although similar, is safer, more effective or otherwise clinically superior to the original orphan medicinal product. A company may voluntarily remove a product from the register of orphan products.
Post-Approval Requirements
Where an MA is granted in relation to a medicinal product in the EU, the holder of the MA is required to comply with a range of regulatory requirements applicable to the manufacturing, marketing, promotion and sale of medicinal products.
Similar to the United States, both MA holders and manufacturers of medicinal products are subject to comprehensive regulatory oversight by the EMA, the European Commission and/or the competent regulatory authorities of the individual EU member states. The holder of an MA must establish and maintain a pharmacovigilance system and appoint an individual qualified person for pharmacovigilance who is responsible for oversight of that system. Key obligations include expedited reporting of suspected serious adverse reactions and submission of periodic safety update reports, or PSURs.
All new MAAs must include a risk management plan, or RMP, describing the risk management system that the company will put in place and documenting measures to prevent or minimize the risks associated with the product. The regulatory authorities may also impose specific obligations as a condition of the MA. Such
116
risk-minimization measures or post-authorization obligations may include additional safety monitoring, more frequent submission of PSURs, or the conduct of additional clinical trials or post-authorization safety studies.
Coverage, Pricing and Reimbursement outside the United States
Outside the U.S., pharmaceutical companies, products and distributors are also generally subject to extensive governmental price controls and other market regulations. We believe the increasing emphasis on cost-containment initiatives in EEA and other countries has and will continue to put pressure on the pricing and usage of our products. In many countries, the prices of medical products are subject to varying price control mechanisms as part of national health systems that fund a large part of the cost of those products to consumers. Some countries operate positive and negative list systems under which products may only be marketed once a reimbursement price has been agreed. To obtain reimbursement or pricing approval, some of these countries may require the completion of comparative trials that compare the cost effectiveness of a particular product candidate to currently available therapies. Other countries allow companies to fix their own prices for medical products, but monitor and control company profits. Additional price controls in foreign countries or other changes in pricing regulation could restrict the amount that we are able to charge for our products. Accordingly, in markets outside the U.S., the reimbursement for our products may be reduced compared with the U.S. and may be insufficient to generate commercially reasonable revenue and profits.
Moreover, increasing efforts by governmental and third-party payors abroad, as in the U.S., to control healthcare costs may cause such organizations to limit both coverage and the level of reimbursement for new products approved and, as a result, they may not cover or provide adequate payment for our products. We expect to experience pricing pressures in connection with the sale of any of our products due to the trend toward managed healthcare, the increasing influence of health maintenance organizations and additional legislative changes.
If we are unable to establish or sustain coverage and adequate reimbursement for any of products from third-party payors, the adoption of those products and sales revenue will be adversely affected, which, in turn, could adversely affect the ability to market or sell those product candidates, if approved.
In various EEA countries, we expect to be subject to continuous cost-cutting measures, such as lower maximum prices, lower or lack of reimbursement coverage and incentives to use cheaper products as an alternative. Health Technology Assessment, or HTA, of medicinal products is becoming an increasingly common part of the pricing and reimbursement procedures in some EEA countries, including countries representing major markets. The HTA process, which is currently governed by the national laws of these countries, is the procedure according to which the assessment of the public health impact, therapeutic impact and the economic and societal impact of use of a given medicinal product in the national healthcare systems of the individual country is conducted. The outcome of HTA regarding specific medicinal products will often influence the pricing and reimbursement status granted to these medicinal products by the competent authorities of individual EEA Member States. On January 31, 2018, the European Commission adopted a proposal for a regulation on health technologies assessment. The proposed regulation is intended to boost cooperation among EEA Member States in assessing health technologies, including new medicinal products, and providing the basis for cooperation at the EEA level for joint clinical assessments in these areas. In June 2021, the European Parliament and Council reached a provisional agreement on the draft regulation. Entry into application of the Regulation could impose stricter and more detailed procedures to be followed by MAHs concerning conduct of HTA in relation to their products which may influence related pricing and reimbursement decisions.
Advertising Regulation
In the EU, the advertising and promotion of medicinal products are subject to both EU and EU member states’ laws governing promotion of medicinal products, interactions with physicians and other healthcare professionals, misleading and comparative advertising and unfair commercial practices. Although general requirements for advertising and promotion of medicinal products are established under EU rules, the details are governed by regulations in individual EU member states and can differ from one country to another. Applicable laws require that promotional materials and advertising in relation to medicinal products comply with the product’s Summary of Product Characteristics, or SmPC, as approved by the competent authorities in connection with an MA, therefore all off-label promotion is prohibited. The SmPC is the document that provides information to physicians concerning the safe and effective use of the product. Promotional activity that does not comply with the SmPC is considered off-label and is prohibited in the EU. Direct-to-consumer advertising of prescription medicinal products is also prohibited in the EU.
117
If we fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Rest of the World Regulation
For other countries outside of the EU and the United States, such as countries in Eastern Europe, Latin America or Asia, the requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary from country to country. Additionally, the clinical trials must be conducted in accordance with GCP requirements and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki.
If we fail to comply with applicable foreign regulatory requirements, we may be subject to, among other things, fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
The Foreign Corrupt Practices Act
The FCPA prohibits any U.S. individual or business from paying, offering, or authorizing payment or offering of anything of value, directly or indirectly, to any foreign official, political party or candidate for the purpose of influencing any act or decision of the foreign entity in order to assist the individual or business in obtaining or retaining business. The FCPA also obligates companies whose securities are listed in the United States to comply with accounting provisions requiring us to maintain books and records that accurately and fairly reflect all transactions of the corporation, including international subsidiaries, and to devise and maintain an adequate system of internal accounting controls for international operations.
Additional Regulation
In addition to the foregoing, state and federal laws regarding environmental protection and hazardous substances, including the Occupational Safety and Health Act, the Resource Conservancy and Recovery Act and the Toxic Substances Control Act, affect our business. These and other laws govern our use, handling and disposal of various biological, chemical and radioactive substances used in, and wastes generated by, our operations. If our operations result in contamination of the environment or expose individuals to hazardous substances, we could be liable for damages and governmental fines. We believe that we are in material compliance with applicable environmental laws and that continued compliance therewith will not have a material adverse effect on our business. We cannot predict, however, how changes in these laws may affect our future operations.
Other Regulations
We are also subject to numerous federal, state and local laws and foreign laws relating to such matters as safe working conditions, manufacturing practices, environmental protection, fire hazard control, and disposal of hazardous or potentially hazardous substances. We may incur significant costs to comply with such laws and regulations now or in the future.
In addition to regulation in the United States and the EEA, a variety of foreign regulations govern clinical trials, commercial sales and distribution of drugs. Pharmaceutical firms who wish to market their medicinal drugs outside the EEA and the United States must submit marketing authorization application to the national authorities of the concerned countries, such as the Pharmaceutical and Medical Device Agency, or PMDA in Japan. The approval process varies from jurisdiction to jurisdiction and the time to approval may be longer or shorter than that required by the FDA or European Commission.
Manufacturing and Supply
We are dependent on specialized third parties, who are subject to cGMP requirements and regulations, for the supply and manufacture of Sozinibercept (OPT-302) drug substance and drug product. We do not have any internal manufacturing and control capabilities. We source the drug substance for Sozinibercept (OPT-302) and our clinical trials on a purchase order basis. However, we believe that competitive pricing is achieved because there are a number of potential long-term replacements for our suppliers of drug substance.
118
In October 2013, we entered into a biopharmaceutical manufacturing agreement, or the Patheon Agreement, with Patheon Biologics Company, Australia Pty Ltd. and Patheon Biologics Company B.V., or collectively Patheon now part of ThermoFisher Scientific. The Patheon Agreement establishes the general terms and conditions pursuant to which Patheon or its affiliates will manufacture Sozinibercept (OPT-302) drug product for us in accordance with cGMP requirements. Under the Patheon Agreement, Patheon granted us a perpetual, royalty-free, fully paid-up, non-exclusive, worldwide, transferable and sublicensable license, under all of Patheon’s intellectual property rights embedded in the development and manufacture process to the extent necessary for developing, making, using and selling Sozinibercept (OPT-302).
The Patheon Agreement will expire on the date that all of the manufacturing services to be performed by Patheon are completed. We may terminate the Patheon Agreement for any reason upon prior written notice. Patheon may terminate the Patheon Agreement upon prior written notice if Patheon has not performed any activities under the Patheon Agreement for certain period of time or if, despite Patheon’s commercially reasonably best efforts, Patheon determines that the services cannot be completed according to specifications approved by us or within a reasonable time after the originally planned timeframe. Either party may terminate the Patheon Agreement for the other party’s uncured material breach or bankruptcy. In addition, the Patheon Agreement will terminate if the parties are unable to reach agreement regarding necessary changes to the services based on the results of individual stage of development work. Upon termination of the Patheon Agreement, we are required to pay Patheon for services properly performed, including non-cancelable costs. Based upon the timing of the termination, we may also be required to pay Patheon certain close out cost for canceled services.
Employees
As of June 30, 2023, we had 24 full-time employees, eight of whom had an M.D. or Ph.D. degree. None of our employees are represented by collective bargaining agreements. We believe that our management maintains good relations with our employees. As of June 30, 2023, our employees were based in Australia (8) and United States (16), with 17 employees in our research and development and commercialization department and seven employees in our general and administrative department.
Legal Proceedings
From time to time, we may become involved in legal proceedings arising in the ordinary course of our business. We are currently not a party to any material legal proceedings.
4C. Organizational Structure
Opthea Limited is the parent company of two wholly-owned subsidiaries for which it holds 100% of the voting power: Vegenics Pty Ltd, incorporated in Australia in January 2006 and Opthea US Inc, incorporated in the United States in May 2021.
4D. Property and Equipment
We occupy approximately 591 square feet of office space in South Yarra, Victoria, Australia under a lease that expires in July 2025. We believe that our existing facilities are adequate to meet our current needs and that suitable additional alternative facilities will be available in the future on commercially reasonable terms to meet our future needs.
Item 4A. Unresolved Staff Comments
Not applicable.
Item 5. Operating and Financial Review and Prospects
The following “Operating and Financial Review and Prospects” should be read together with our consolidated financial statements and the accompanying notes included elsewhere in this annual report. The following discussion is based on our financial information prepared in accordance with IFRS, as issued by the IASB, which might differ in material respects from accounting principles generally accepted in other jurisdictions, including U.S. GAAP. This discussion includes both historical information and forward-looking information based upon current expectations that involve risk, uncertainties and assumptions. Our actual results may differ materially from
119
management’s expectations as a result of various factors, including, but not limited to, those discussed in “Risk Factors” and elsewhere in this annual report.
Overview
We are a clinical-stage biopharmaceutical company developing a novel therapy for the treatment of highly prevalent and progressive retinal diseases. We are developing our Phase 3-ready product candidate, Sozinibercept (OPT-302), a biologic designed to inhibit VEGF-C and VEGF-D, to complement VEGF-A inhibitors for the treatment of ophthalmic diseases. Anti-VEGF-A therapies represent the standard of care for wet age-related macular degeneration, or AMD, and other retinal diseases; however, there remains a significant unmet medical need as many patients do not adequately respond to these treatments. As the only biologic inhibitor of VEGF-C and VEGF-D in clinical development, Sozinibercept (OPT-302) differs from standard of care therapies and when administered in combination with a VEGF-A inhibitor, is designed to achieve broader inhibition of the vascular endothelial growth factor, or VEGF, family and target a mechanism of clinical resistance to improve visual acuity. Our lead indication for Sozinibercept (OPT-302) combination therapy is wet AMD, a chronic, progressive disease and the leading cause of vision loss for individuals over the age of 50. In a 366-patient Phase 2b clinical trial for the treatment of wet AMD, 2.0 mg Sozinibercept (OPT-302), in combination with a standard of care anti-VEGF-A therapy, ranibizumab (Lucentis), met the primary endpoint of a statistically significant superior mean gain in visual acuity over ranibizumab monotherapy at week 24. We initiated two pivotal Phase 3 clinical trials in treatment-naive patients with wet AMD to evaluate the efficacy and safety of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapies compared to anti-VEGF-A monotherapy in March 2021. We expect to complete patient recruitment in the COAST study in the first calendar quarter of 2024 and in the ShORe study in the second calendar quarter of 2024. We anticipate reporting top-line data after the patients complete the 52 week treatment period. In addition to our clinical trials in wet AMD, we have observed evidence of improved clinical outcomes in a Phase 1b/2a clinical trial of Sozinibercept (OPT-302) in combination with another standard of care anti-VEGF-A therapy, aflibercept (Eylea), in patients with treatment-refractory diabetic macular edema, or DME. We retain worldwide rights to develop and commercialize Sozinibercept (OPT-302) for the treatment of wet AMD and DME and believe that the novel treatment mechanism of Sozinibercept (OPT-302) has the potential to provide therapeutic benefit for other progressive eye diseases.
We were founded in 1984 and completed our initial public offering and listing of ordinary shares on the Australian Securities Exchange in 1985. In October 2020, we completed a U.S, initial public offering of ADSs on Nasdaq. In April 2007, we acquired intellectual property relating to VEGF receptor 3 and subsequently developed the intellectual property for our lead product candidate, Sozinibercept (OPT-302). Our development focus on the treatment of retinal diseases began in 2013. Since then, we have devoted substantially all of our efforts to organizing and staffing our company, business planning, raising capital, developing and manufacturing our lead product candidate, Sozinibercept (OPT-302), conducting research and development activities, including preclinical studies and clinical trials, and providing general and administrative support for these operations. Our operations relating to the development of Sozinibercept (OPT-302) have been financed primarily through the issuance and sale of new ordinary shares totaling US$320.9 million through June 30, 2023. We have also received an aggregate of US$35.6 million (A$50.3 million) in cash tax incentives for the five fiscal years ended June 30, 2023 under the Research and Development, or R&D, Tax Incentive Scheme for the funding of the development of and clinical trials for Sozinibercept (OPT-302).
We have incurred operating losses since 2013. Our ability to generate product revenue sufficient to achieve profitability will be dependent on the successful development and eventual commercialization of Sozinibercept (OPT-302) and any future product candidates. Our total comprehensive loss was US$44.9 million, US$92.8 million, and US$142.4 million for the years ended June 30, 2021, 2022 and 2023, respectively. As of June 30, 2023, we had an accumulated loss of US$359.4 million. We expect to continue to incur significant expenses for at least the next several years as we advance Sozinibercept (OPT-302) through late-stage clinical development, including our planned pivotal Phase 3 trials of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD, and, if these results are favorable, seek regulatory approval for Sozinibercept (OPT-302). In addition, we may also pursue development of Sozinibercept (OPT-302) for the treatment of additional indications, including DME, retinal vein occlusion and other indications in which Sozinibercept (OPT-302) has the potential to provide therapeutic benefit. In addition, if we obtain marketing approval for Sozinibercept (OPT-302), we expect to incur significant commercialization expenses related to product manufacturing, marketing, sales and distribution. As a company whose ADSs are publicly traded in the United States, we have incurred and will continue to incur additional costs associated with operating as a public
120
company in the United States, including significant additional legal, accounting, investor relations, compliance and other expenses.
As a result, we will need substantial additional funding to complete the ongoing Phase 3 trials and report top-line data to support our continuing operations and pursue our growth strategy. Until such time as we can generate significant revenue from product sales, if ever, we expect to finance our operations through the sale of equity, debt financings, or other capital sources, which may include collaborations with other companies or other strategic transactions as well as Australian research and development tax incentives. We may be unable to raise additional funds or enter into such other agreements or arrangements when needed on favorable terms, or at all. If we fail to raise capital or enter into such agreements as and when needed, we may have to significantly delay, scale back or discontinue the development and commercialization of Sozinibercept (OPT-302).
Because of the numerous risks and uncertainties associated with the development of biopharmaceutical product candidates, we are unable to predict the timing or amount of increased expenses or when or if we will be able to achieve or maintain profitability. Even if we are able to generate product sales, we may never become profitable. If we fail to become profitable or are unable to sustain profitability on a continuing basis, then we may be unable to continue our operations at planned levels and be forced to scale back or discontinue our operations.
On August 12, 2022, we entered into a Development Funding Agreement (the “Funding Agreement”) with Ocelot SPV LP (“Investor”), an affiliate of Carlyle and Abingworth, in collaboration with Carlyle and Abingworth’s recently formed development company Launch Therapeutics (“Launch Tx”), pursuant to which Investor agreed to provide funding to support our development of Sozinibercept (OPT-302) for the treatment of wet AMD. Pursuant to the Funding Agreement, Investor has committed to provide Opthea US$120 million in funding which may be increased up to $170 million at Investor’s option, of which $85 million has already been funded with the remainder being funded in an additional tranche of US$35 million to be paid on December 31, 2023. If Sozinibercept (OPT-302) is approved in a major market Carlyle and Abingworth will be eligible to receive fixed success payments, as well as variable success payments of 7% on annual net sales, which collectively terminate after reaching four times the funded amount. In addition, in August 2022 we received commitments for the private placement of ordinary shares for aggregate gross proceeds of US$90 million consisting of two tranches (the “placement and entitlement offer”).
In addition to the above, on September 26, 2022 Opthea completed its Share Purchase Plan raising US$618 thousand (AU$940 thousand) and issuing 817,824 ordinary shares.
On August 24, 2023, we announced a A$80 million capital raise consisting of an A$10 million private placement ("Placement") and a A$70 million Accelereated Non Renounceble Entitlement Offer ("ANREO"). On August 28, 2023, Opthea announced an increase in the Placement by a further A$10 million to increase the overall raise to A$90 million (approximately US$58 million). The proceeds from the Placement and Entitlement (together, the "2023 Equity Offering") which was completed in September 2023 will be used to continue advancing the clinical development of OPT-302 for the treatment of we Age related macular Degeneration (wet AMD) including to progress our Phase 3 clinical trials and for general corporate purposes. In addition, subsequent to June 30, 2023, we were notified that a new co-investor of Carlyle and Abingworth intends to participate in a funding under the Funding Agreement of US$50 million (the "Incremental DFA Funding") to increase total Funding Agreement funding from US$120 million to US$170 million, which is subject to the co-investor’s final due diligence and receipt of regulatory and tax approvals, appropriate documentation and compliance with closing conditions. There can be no assurance that the due diligence will be completed to the satisfaction of the co-investor of Carlyle and Abingworth, the closing terms and conditions will be satisfied, or that we will ultimately receive the additional US$50 million. If the additional US$50 million is not received by June 30, 2024, we will need to raise additional funds or reduce expenditures to continue as a going concern. If we fail to obtain additional capital from other sources, prior to top-line data for out Phase 3 clinical trials, which may not be available on a timely basis or at all, we could be forced to delay, limit or terminate our operations, liquidate all or a portion of its assets and/or seek insolvency protection in the near term. Our failure to raise capital, if and when needed, could delay or suspend our business strategy and could have a material adverse effect on our activities.
As of June 30, 2023, we had cash and cash equivalents of US$89.2 million. We believe that our existing cash and cash equivalents as of June 30, 2023, as well as net proceeds of approximately US$54 million from the 2023 Equity Offering, - and the incremental DFA Funding and the remaining $35 million under the Funding Agreement, if received, will enable us to fund our operating and research and development expenses into the third calendar quarter of 2024. However, we will need to raise additional funds to complete the efficacy and safety phase of both studies and to report top-line data. In addition, the forecast of our cash runway, following receipt of the proceeds
121
from the 2023 Equity Offering and under the Funding Agreement, is subject to a number of assumptions, including the timing of completion of Phase 3 clinical trial patient enrollment and CRO and labor costs. Estimated patient enrollment timing used for our forecast of our cash runway is based on our monthly enrollment rates for our Phase 3 clinical trials, which timing has in the past significantly fluctuated from prior estimates, including due to factors outside our control.
The amounts and timings of our expenditures will depend upon and have been impacted in the past, and may continue to be impacted by, numerous factors, including historical or future delays in completing our clinical trials, particularly as it relates to enrollment, the timing of regulatory submissions, the performance and costs efficiency of CROs and any continuing impacts of the COVID-19 pandemic, the global supply chain and macroeconomic challenges. In particular, delays in patient enrollment have in the past resulted, and may in the future result in increased costs or delays and other impacts on the timing of our Phase 3 clinical trials. We have in the past incurred significantly increased costs in connection with the activities conducted by third party CROs and other service providers to prepare for and progress our Phase 3 clinical trials, and may continue to incur higher than expected costs for such activities in the future. We have based this estimate on assumptions that may prove to be wrong, and we could exhaust our available capital resources sooner than we expect. We may also experience future delays in our clinical development or commercialization of Sozinibercept (OPT-302) for an indication, including due to factors and conditions set forth above or other factors that we cannot presently anticipate. We intend to focus our development efforts on achieving commercialization of Sozinibercept (OPT-302) for the treatment of wet AMD, and we will require additional funding to reach commercialization of Sozinibercept (OPT-302) in any indication, including wet AMD. In addition, we may require additional external funding to meet the minimum cash condition under the Funding Agreement, including prior to the readout of top-line results for our Phase 3 clinical trials for Sozinibercept (OPT-302) for the treatment of wet AMD. See “—Liquidity and Capital Resources.”
5A. Operating Results
Components of Our Results of Operations
Revenue
Revenue consists of sales-based royalties in connection with the out-licensing of certain intellectual property assets that are unrelated to our core business and the development of Sozinibercept (OPT-302) and are not currently under development. These licenses are primarily used by third-party licensees for research purposes, and we expect revenue from these out-licensing arrangements to be nominal in future periods. These are variable consideration amounts and are recognized when the sales by our license partners to third parties occur, as the performance obligation to transfer the intellectual property to the license partner is already satisfied.
To date, we have not generated any revenue from sales of approved products. Because of the numerous risks and uncertainties associated with product development and regulatory approval, we are unable to predict the amount, timing or whether we will be able to obtain revenue from sales of approved products, and we may never succeed in obtaining regulatory approval for Sozinibercept (OPT-302) or any other product candidate. If our development efforts for Sozinibercept (OPT-302) are successful and result in an approved and marketed product, or if we enter into additional collaboration or license agreements with third parties, we may generate revenue in the future from a combination of product sales or payments from such collaboration or license agreements.
Other Income
Other income primarily comprises funding under a one-time Australian government grant and the Australian Tax Office and fees from BNY Mellon ADS holders.
122
Operating Expenses
Research and Development Expenses. Research and development expenses comprise the research project costs related to the development programs, including clinical trials, for Sozinibercept (OPT-302) for the treatment of wet AMD and DME. R&D expenses also include:
We expense R&D costs as incurred and have not capitalized any amounts of R&D costs as of June 30, 2022 and 2023. In the years ended June 30, 2021, 2022 and 2023, we have made advance payments for R&D activities that will be provided by our CROs in future periods for use in R&D activities.
Our direct R&D expenses are tracked on a program-by-program basis for our product candidate and consist primarily of external costs, such as fees paid to CROs, CDMOs, research laboratories and outside consultants in connection with our process development, manufacturing and clinical development activities. We do not allocate employee costs associated with our research efforts, laboratory supplies and facilities, including depreciation and other indirect costs, to specific programs because these costs are deployed across multiple development activities and indications for Sozinibercept (OPT-302) and, as such, are not separately classified. We use internal resources primarily to conduct our research activities as well as for managing our process development, manufacturing and clinical development activities. These employees work across multiple development programs and, therefore, we do not track these costs by program.
R&D expenses in fiscal years after June 30, 2023 are expected to comprise costs of a similar nature to that recorded to date. Product candidates in later stages of clinical development, such as Sozinibercept (OPT-302), generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of later-stage clinical trials. We expect that our R&D expenses will increase in connection with our planned clinical development, manufacturing and regulatory approval activities in the near term and in the future, including as we continue our pivotal Phase 3 clinical trials of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD.
At this time, we cannot accurately estimate or know the nature, timing and costs of the efforts that will be necessary to complete the clinical development of Sozinibercept (OPT-302) and any future product candidates. We may never succeed in achieving regulatory approval for any of our product candidates. The duration, costs and timing of clinical trials and development of our product candidates will depend on a variety of factors, including:
123
A change in the outcome of any of these factors with respect to the development of Sozinibercept (OPT-302) could significantly change the duration, costs and timing associated with clinical trials and development of Sozinibercept (OPT-302).
Patent and Intellectual Property Expenses. Patent and Intellectual Property expenses comprise the cost of outside patent attorneys to manage and prosecute our patent portfolio.- license and patent assignment costs in respect of our in-license agreements for certain technologies not currently under development and unrelated to our out-licensing arrangements under which we receive sales-based royalties.
Administrative Expenses. Administrative expenses comprise employee benefit expenses, including share-based payment expenses; investor relations expenses; insurance costs; audit, accountancy and legal fees; other personnel-related expenses; and depreciation expense. We anticipate that our administrative expenses will increase in the future as we increase our headcount to support development of Sozinibercept (OPT-302) and our continued research activities. We also anticipate that we will incur increased accounting, audit, legal, regulatory, compliance and director and officer insurance costs as well as investor and public relations expenses associated with being a public company in the United States.
Occupancy Expenses. Occupancy expenses represent the costs relating to our headquarters in Melbourne, Australia, including lease maintenance and incidental costs.
Finance Income – Interest income
Finance income primarily comprises of income from interest on bank deposits.
Net Foreign Exchange Gain (Loss)
Net foreign exchange gain (loss) represents the impact of the variance in exchange rates between the U.S. dollar and the Australian dollar, Euro, British Pound and Canadian dollar on our cash and cash equivalents, financial assets, financial liabilities and foreign currency denominated transactions.
Income Tax Benefit
Income tax benefit represents the cash incentive amount receivable under the R&D Tax Incentive Scheme, an Australian Federal Government program under which eligible companies with annual aggregated revenue of less than A$20.0 million can receive cash amounts equal to 43.5% of eligible R&D expenditures from the Australian Taxation Office, or the ATO. The ATO may also make other changes to the eligibility of R&D expenditures, including placing a cap on the amount of non-clinical trial R&D expenses claimed under the scheme.
The R&D Tax Incentive Scheme incentive relates to eligible expenditures incurred in Australia and, under certain circumstances, in other countries in connection with the development of Sozinibercept (OPT-302). The R&D tax incentive is applied annually to eligible expenditures incurred during the fiscal year following an annual application and subsequent filing of our income tax return subsequent to fiscal year end. We estimate the amount of R&D tax incentive after the completion of a fiscal year based on eligible Australia and overseas expenditures incurred during that year. We expect to continue applying for the R&D tax incentive as we further develop Sozinibercept (OPT-302). In particular, we intend to apply for the costs expected to be incurred in Australia related to our planned pivotal Phase 3 clinical trials of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD to be eligible for the R&D tax incentive for future fiscal years once incurred. However, there can be no assurance that the ATO will allow these costs to be eligible for the tax incentive.
124
Results of Operations for the Fiscal Years Ended June 30, 2021, 2022 and 2023
The following table sets forth a summary of our consolidated statement of profit or loss and other comprehensive income for the periods presented.
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
|
|
(in thousands) |
|
|||||||||
Revenue |
|
|
108 |
|
|
|
91 |
|
|
|
68 |
|
Other income |
|
|
277 |
|
|
|
108 |
|
|
|
27 |
|
Operating expenses |
|
|
|
|
|
|
|
|
|
|||
Research and development expenses |
|
|
(122,128 |
) |
|
|
(78,654 |
) |
|
|
(25,892 |
) |
Patent and intellectual property expenses |
|
|
(167 |
) |
|
|
(160 |
) |
|
|
(428 |
) |
Administrative expenses |
|
|
(28,116 |
) |
|
|
(17,922 |
) |
|
|
(13,418 |
) |
Interest expense on DFA |
|
|
(13,462 |
) |
|
|
— |
|
|
|
— |
|
Finance income |
|
|
3,228 |
|
|
|
235 |
|
|
|
372 |
|
Fair value adjustment gain on DFA |
|
|
12,302 |
|
|
|
— |
|
|
|
— |
|
Net foreign exchange gain (loss) |
|
|
(489 |
) |
|
|
(2,814 |
) |
|
|
(11,012 |
) |
Loss before income tax |
|
|
(148,447 |
) |
|
|
(99,116 |
) |
|
|
(50,283 |
) |
Income tax benefit |
|
|
5,926 |
|
|
|
6,299 |
|
|
|
4,939 |
|
Loss for the year |
|
|
(142,521 |
) |
|
|
(92,817 |
) |
|
|
(45,344 |
) |
Other comprehensive income: |
|
|
|
|
|
|
|
|
|
|||
Items that will not be reclassified subsequently to profit or loss: |
|
|
|
|
|
|
|
|
|
|||
Fair value gains on investments in financial assets |
|
|
— |
|
|
|
— |
|
|
|
469 |
|
Other comprehensive income for the period, net of tax |
|
|
— |
|
|
|
— |
|
|
|
469 |
|
Total comprehensive loss for the year |
|
|
(142,521 |
) |
|
|
(92,817 |
) |
|
|
(44,875 |
) |
Revenue
Revenue was US$91 thousand for the fiscal year ended June 30, 2022, compared to US$108 thousand for the fiscal year ended June 30, 2023. This increase was due to higher sales-based royalties received under our out-licensing arrangements. Revenue for the years ended June 30, 2022 and 2023 consisted of sales-based royalties in connection with the out-licensing of certain intellectual property assets that are unrelated to our core business and the development of Sozinibercept (OPT-302) which are not currently under development.
Revenue was US$68 thousand for the fiscal year ended June 30, 2021, compared to US$91 thousand for the fiscal year ended June 30, 2022. This increase was due to higher sales-based royalties received under our out-licensing arrangements. Revenue for the years ended June 30, 2021 and 2022 consisted of sales-based royalties in connection with the out-licensing of certain intellectual property assets that are unrelated to our core business and the development of Sozinibercept (OPT-302) which are not currently under development.
Other Income
Other income was US$108 thousand for the fiscal year ended June 30, 2022, compared to US$277 thousand for the fiscal year ended June 30, 2023. The increase in 2023 is due to the receipt of fees from BNY Mellon ADR holders.
Other income was US$27 thousand for the fiscal year ended June 30, 2021, compared to US$108 thousand for the fiscal year ended June 30, 2022. The increase in 2022 is due to the receipt of fees from BNY Mellon ADR holders which offsets the decrease in 2021 that was primarily due to a one-time Australian government grant.
Research and Development Expenses
Research and development expenses were US$78.7 million for the fiscal year ended June 30, 2022, compared to US$122.1 million for the fiscal year ended June 30, 2023. This increase was primarily due to costs relating to our Phase 3 clinical trial of Sozinibercept (OPT-302) for the treatment of wet AMD, chemistry manufacturing and controls costs incurred during the fiscal year ended June 30, 2022 related to manufacturing activities for our Phase
125
3 clinical trials of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD
Research and development expenses were US$25.9 million for the fiscal year ended June 30, 2021, compared to US$78.7 million for the fiscal year ended June 30, 2022. This increase was primarily due to costs relating to our Phase 3 clinical trial of Sozinibercept (OPT-302) for the treatment of wet AMD that started during the fiscal year ended June 30, 2021, chemistry manufacturing and controls costs incurred during the fiscal year ended June 30, 2022 related to manufacturing activities for our Phase 3 clinical trials of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD.
Our research and development expenses are broken down as set forth in the table below:
|
|
|
|
|
|
|
|
|||||||
|
|
2023 |
|
|
|
2022 |
|
|
2021 |
|
||||
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
US$ |
|
|
|
US$ |
|
|
US$ |
|
||||
|
|
(in thousands) |
|
|||||||||||
Costs related to the ShORe Phase 3 clinical trial of OPT-302 in wet AMD |
|
|
59,472 |
|
|
|
|
36,675 |
|
|
|
9,277 |
|
|
Costs related to the COAST Phase 3 clinical trial of OPT-302 in wet AMD |
|
|
50,544 |
|
|
|
|
27,520 |
|
|
|
7,886 |
|
|
Costs related to the Phase 2b clinical trial of OPT-302 in wet AMD |
|
|
18 |
|
|
|
|
— |
|
|
|
(148 |
) |
|
Costs related to the Phase 1b/2a clinical trial of OPT-302 in DME |
|
|
18 |
|
|
|
|
— |
|
|
|
795 |
|
|
Chemistry manufacturing and controls |
|
|
12,062 |
|
|
|
|
14,430 |
|
|
|
8,048 |
|
|
Other direct non-clinical expenses |
|
|
14 |
|
|
|
|
29 |
|
|
|
34 |
|
|
Total research and development expenses |
|
|
122,128 |
|
|
— |
|
|
78,654 |
|
|
|
25,892 |
|
Patent and Intellectual Property Expenses
Patent and intellectual property expenses were US$160 thousand for the fiscal year ended June 30, 2022, compared to US$167 thousand for the fiscal year ended June 30, 2023. This increase was primarily due to slightly higher expenses related to filing and prosecution of patents.
Patent and intellectual property expenses were US$137 thousand for the fiscal year ended June 30, 2021, compared to US$132 thousand for the fiscal year ended June 30, 2022. This decrease was primarily due to validation work carried out on our European patent application across multiple jurisdictions in 2020 and maintained in 2021 and 2022 as well as the decrease in royalty milestone payment on initiation of our Phase 3 clinical trials paid in 2021.
Administrative Expenses
Administrative expenses were US$17.9 million for the fiscal year ended June 30, 2022, compared to US$28.1 million for the year ended June 30, 2023. This increase was primarily due to a US$5.7 million increase in professional and advisory fees and expenses and a US$4.3 million increase in personnel-related expenses, each resulting from increased headcount as we built out the US team to aid continued clinical trial oversight and management and capital raising activities in the fiscal year ended June 30, 2023.
Administrative expenses were US$13.4 million for the fiscal year ended June 30, 2021, compared to US$17.9 million for the fiscal year ended June 30, 2022. This increase was primarily due to a US$1.3 million increase in share-based payments, a US$2.2 million increase in professional fees and expenses and a US$1.0 million increase in personnel-related expenses, each resulting from increased headcount in building out of the US team to aid continued research and development and capital raising activities in the fiscal year ended June 30, 2022.
Interest expense on DFA
Interest expense on the DFA was US$nil thousand for the fiscal year ended June 30, 2022, compared to US$13.5 million for the fiscal year ended June 30, 2023. This increase was due to the accretion of the financial liability recognized upon the receipt of payments under the DFA. The interest expense is a non-cash item.
126
There was no interest expense on DFA in each of the fiscal years ended June 30, 2021, and 2022 as the DFA funding was received in the fiscal year ended June 30 2023.
Fair value adjustment gain on DFA
There was no fair value adjustment gain on the DFA for the fiscal year ended June 30, 2022, compared to US$12.3 million for the fiscal year ended June 30, 2023. This increase was due to the revaluation of the financial liability regarding the assumptions on the anticipated timing of repayments under the DFA. The fair value adjustment on the DFA is a non-cash item.
There was no fair value adjustment on the DFA in each of the fiscal years ended June 30, 2021 and 2022 as the DFA funding was received in the fiscal year ended June 30, 2023.
Net Foreign Exchange Loss
Net foreign exchange differences were a loss of US2.8 million for the fiscal year ended June 30, 2022, compared to a loss of US0.5 million for the fiscal year ended June 30, 2023. The decrease was, primarily as a result of net variances of the exchange rate between the Australian dollar and U.S. dollar on Australian dollar-denominated cash and cash equivalents, financial assets, financial liabilities and foreign currency denominated transactions.
Net foreign exchange differences were a loss of US$11 million for the fiscal year ended June 30, 2021, compared to a loss of US$2.8 million for the fiscal year ended June 30, 2022. The decrease was, primarily as a result of net variances of the exchange rate between the Australian dollar and U.S. dollar on Australian dollar-denominated cash and cash equivalents, financial assets, financial liabilities and foreign currency denominated transactions.
Income Tax Benefit
Income tax benefit was US$6.3 million for the fiscal year ended June 30, 2022, compared to US$5.9 million for the fiscal year ended June 30, 2023. This decrease was due to lower R&D tax incentive receivable recognized during the fiscal year ended June 30, 2023 based on eligible spend.
Income tax benefit was US$4.9 million for the fiscal year ended June 30, 2021, compared to US$6.3 million for the fiscal year ended June 30, 2022. This increase was due to higher R&D tax incentive receivable recognized during the fiscal year ended June 30, 2022.
5B. Liquidity and Capital Resources
The liquidity and capital resources discussion that follows contains certain estimates as of the date of this annual report of our estimated future sources and uses of liquidity (including estimated future capital resources and capital expenditures) and future financial and operating results. These estimates reflect numerous assumptions made by us with respect to industry performance, general business, economic, regulatory, market and financial conditions and other future events, and matters specific to our businesses, all of which are difficult or impossible to predict and many of which are beyond our control.
Sources and Uses of Liquidity
Our operations relating to the development of Sozinibercept (OPT-302) have been financed primarily through the issuance and sale of new ordinary shares totaling US$241.1 million in the five years ended June 30, 2023. We have also received an aggregate of US$84.5 million in the five years ended June 30, 2023, under the Development Funding Agreement with Carlyle and Abingworth, pursuant to which Investor agreed to provide funding to support our development of Sozinibercept (OPT-302) for the treatment of wet AMD. We have also received an aggregate of US$35.6 million (A$50.3 million) in the five fiscal years ended June 30, 2023, under the R&D Tax Incentive Scheme for the funding of the development and clinical trials of Sozinibercept (OPT-302).
In February 2022, we established an “at the market” program (the “ATM Program”) with Jefferies LLC (“Jefferies”). Pursuant to the ATM Program, we may offer and sell up to US$75 million of our ordinary shares in the form of ADSs, with each ADS representing eight ordinary shares, through Jefferies.
127
Sales of ADSs under the ATM Program may be made from time to time, with the timing and amount of any sales to be determined by us based on a variety of factors. We may determine to sell some, all or none of the ADSs under the ATM Program and may terminate the ATM Program at our discretion. We, through Jefferies, may sell ADSs by any lawful method deemed to be an “at-the-market offering” defined by Rule 415(a)(4) under the Securities Act of 1933, as amended. Sales made through the ATM Program may be made at market prices prevailing at the time of a sale or at prices related to prevailing market prices. As a result, actual sales prices may vary.
To effectuate the establishment of the ATM Program, we have entered into a Sales Agreement with Jefferies, acting as sales agent, forward purchaser and forward seller (the “Sales Agreement”). The Sales Agreement provides that, in addition to the issuance and sale of ADSs through Jefferies as sales agent, we may, from time to time, enter into forward sale agreements (each, a “Forward Sale Agreement”) with Jefferies, in its capacity as forward purchaser. To hedge each such Forward Sale Agreement, Jefferies or its affiliate will, at our request, attempt to borrow from third parties and then sell a number of ADSs equal to the number of ADSs underlying each such forward purchase agreement. We will not initially receive any proceeds from any sale of ADSs borrowed by Jefferies, or its affiliate, and sold through Jefferies. Instead, we expect to fully physically settle each Forward Sale Agreement, if any, with Jefferies on one or more dates specified by us on or prior to the maturity date of such Forward Sale Agreement. On physical settlement, we will receive aggregate net cash proceeds equal to the number of ADSs specified in such Forward Sale Agreement multiplied by the relevant forward price per ADS, as adjusted pursuant to the terms of such Forward Sale Agreement. However, subject to certain exceptions, we may also elect to cash settle or net share settle a particular Forward Sale Agreement, in which case we may not receive any proceeds from the issuance of ADSs, and we will instead receive or pay cash (in the case of cash settlement) or receive or deliver ADSs (in the case of net share settlement). As a result, the timing of any issuances of ADSs will depend on a variety of factors, including timing of sales under the ATM Program and the method of settlement of any Forward Sale Agreements.
During the year ended June 30, 2023, no sales of our ordinary shares were made under the ATM Program
As of June 30, 2023, we had cash and cash equivalents of US$89.2 million to fund the manufacturing of sufficient clinical grade Sozinibercept (OPT-302) drug product and complete the regulatory and clinical preparation activities to initiate our planned pivotal Phase 3 clinical trials of Sozinibercept (OPT-302) in combination with anti-VEGF-A therapy for the treatment of wet AMD. As of June 30, 2023, we had an accumulated loss of US$359.4 million.
Subsequent to June 30, 2023, we were notified that a new co-investor of Carlyle and Abingworth intends to participate in a funding under the Funding Agreement of US$50 million to increase total Funding Agreement funding from US$120 million to US$170 million, which is subject to the co-investor’s final due diligence and receipt of regulatory and tax approvals, appropriate documentation and compliance with closing conditions. There can be no assurance that the due diligence will be completed to the satisfaction of the co-investor of Carlyle and Abingworth, the closing terms and conditions will be satisfied, or that we will ultimately receive the additional US$50 million. If the additional US$50 million is not received by June 30, 2024, we will need to raise additional funds or reduce expenditures to continue as a going concern.
Concurrently with the receipt of the notice from the co-investor to increase its investment, we entered into binding commitments for the 2023 Equity Financing, from which we have subsequently received gross proceeds of approximately A$90 million (approximately US$58 million).
Funding Requirements
We believe that our existing cash and cash equivalents as of June 30, 2023, as well as the additional $35 million expected to be available under the Funding Agreement, the Incremental DFA Funding and the net proceeds from the 2023 Equity Offering will enable us to fund our operating and research and development expenses into the third calendar quarter of 2024. The Incremental DFA Funding is subject to the co-investor’s final due diligence and receipt of regulatory and tax approvals, appropriate documentation and compliance with closing conditions. There can be no assurance that the due diligence will be completed to the satisfaction of the co-investor of Carlyle and Abingworth, the closing terms and conditions will be satisfied, or that we will ultimately receive the additional US$50 million. If the additional US$50 million is not received by June 30, 2024, we will need to raise additional funds or reduce expenditures to continue as a going concern. We have based these estimates on assumptions,
128
including the timing of completion of Phase 3 clinical trial patient enrollment and CRO and labor costs, that may prove to be wrong, and we have utilized, and in the future could utilize, our available capital resources sooner than we currently expect. Estimated patient enrollment timing used for our forecast of our cash runway is based on our monthly enrollment rates for our Phase 3 clinical trials, which timing has in the past significantly fluctuated from prior estimates, including due to factors outside our control. We have in the past incurred significantly increased costs in connection with the activities conducted by third party CROs and other service providers to prepare for and progress our Phase 3 clinical trials, and may continue to incur higher than expected costs for such activities in the future. We will require additional funding prior to top-line data for our Phase 3 clinical trials and may require additional funding prior to the third calendar quarter of 2024 if any assumptions regarding our cash runway prove to be wrong or incomplete, including due to factors that have resulted in higher than expected costs in the past such as delays in patient enrollment.
We expect our expenses to increase in connection with our ongoing activities, particularly as we continue the research and development of, and seek marketing approval for, Sozinibercept (OPT-302). In addition, if we obtain marketing approval for Sozinibercept (OPT-302) or any future product candidates, we expect to incur significant commercialization expenses related to sales, marketing, manufacturing and distribution to the extent that such sales, marketing and distribution are not the responsibility of any future collaborators. We have incurred and will continue to incur additional costs associated with operating as a public company in the United States. Accordingly, we will need to obtain substantial additional funding in connection with our continuing operations.
Conducting clinical trials is a time-consuming, expensive and uncertain process that takes many years to complete, and we may never generate the necessary data or results required to obtain marketing approval and achieve product sales. In addition, Sozinibercept (OPT-302) or any future product candidate, if approved, may not achieve commercial success. Our commercial revenues, if any, will be derived from sales of Sozinibercept (OPT-302) and any future product candidate that we do not expect to be commercially available for many years, if at all. Accordingly, we will need to continue to rely on additional financing to achieve our business objectives. Adequate additional financing may not be available to us on acceptable terms, or at all. We may also require external funding to meet the minimum cash condition under the Funding Agreement, including prior to the readout of top-line results for our Phase 3 clinical trials for Sozinibercept (OPT-302) for the treatment of wet AMD. Subsequent to June 30, 2023, we were notified that a new co-investor of Carlyle and Abingworth intends to participate in a funding under the Funding Agreement of US$50 million to increase total funding Agreement funding from US$120 million to US$170 million which is subject to the co-investor's final due diligence and receipt of regulatory and tax approvals, appropriate documentation and compliance with closing conditions. There can be no assurance that the due diligence will be completed to the satisfaction of the co-investor of Carlyle and Abingworth, the closing terms and conditions will be satisfied or that we will ultimately receive the additional US$50 million. We anticipate receiving the third tranche of $35 million under the Funding Agreement by December 31, 2023. However, there can be no assurance that we will meet the conditions under such agreement to receive any future payments, some of which are payable at Ocelot’s discretion. For more information as to the risks associated with our future funding needs, see “Item 3D—Risk Factors—Risks Related to Our Financial Position and Need for Capital.”
Until we can generate a sufficient amount of revenue from the sale of approved products, if ever, we expect to finance our operating activities through our existing liquidity, including with additional funding expected to be available to us under the Funding Agreement and future financing activities, including a combination of equity offerings, including through our ATM program, debt financings, collaborations, strategic alliances and licensing arrangements. However, the Funding Agreement limits the type of financings we may pursue in the future. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interest of our equity holders will be diluted, and the terms of these new securities may include liquidation or other preferences that adversely affect your rights as a holder of ADSs. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise funds through collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, intellectual property, future revenue streams or product candidates. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves.
We have in the past experienced delays in our Phase 3 clinical trials, including the establishment of trial sites and patient recruitment delays, and incurred significantly increased costs in connection with the activities conducted by third-parties that assist us with clinical development, including CROs, and other service providers
129
to prepare for and progress our Phase 3 clinical trials. We may also experience further delays in our clinical development of commercialization of Sozinibercept (OPT-302) for an indication, including due to factors and conditions set forth above or other factors that we cannot presently anticipate. Our present and future funding requirements, both short and long-term will depend on many factors, including, among other things:
Commercial License Agreement with Selexis SA
In October 2013, we entered into a commercial license agreement, or the Selexis Agreement, with Selexis SA, or Selexis, under which Selexis granted us a non-exclusive, worldwide, sublicensable license under certain patents, know-how and other intellectual property controlled by Selexis to use certain cell lines, deliverables and materials provided by Selexis to manufacture Sozinibercept (OPT-302) and related products and to use, sell and otherwise exploit such products.
We paid Selexis a nominal upfront payment upon entering into the Selexis Agreement. We are also required to make certain payments under the Selexis Agreement totaling approximately US$1.3 million upon the achievement of certain development and commercial milestones. We are also obligated to pay a low single-digit running royalty on worldwide net sales of the licensed products. Our royalty obligations will continue, on a product-by-product and country-by-country basis, until the expiration of the relevant patents, but will not extend beyond October 2024 in any event. After the expiration of the royalty term, our license will continue and become full paid, perpetual and irrevocable.
The Selexis Agreement will expire on the date of expiration of the last-to-expire of the license patents. Either party may terminate the Selexis Agreement for the other party’s uncured material breach or bankruptcy. We may also terminate the Selexis Agreement at any time upon prior notice to Selexis.
Development Funding Agreement with Carlyle and Abingworth
On August 12, 2022, we entered into a Development Funding Agreement (the “Funding Agreement”) with Ocelot SPV LP (“Investor”), an affiliate of Carlyle and Abingworth, in collaboration with Carlyle and Abingworth’s recently formed development company Launch Therapeutics, pursuant to which Investor agreed to provide funding to us to support our development of Sozinibercept (OPT-302) for the treatment of wet (neovascular) age-related macular degeneration (“wet AMD”). Pursuant to the Funding Agreement, Investor has committed to provide Opthea US$120 million in funding which may be increased up to US$170 million at Investor’s option, of which US$85 million has already been funded with the remainder being funded in one additional tranche of US$35 million which is expected to be paid on December 31, 2023. In the event the third
130
tranche of US$35 million is not paid by December 31, 2023 it would be considered a Fundamental Material Breach of the Funding Agreement by Carlyle and Abingworth. Under a Fundamental Material Breach of the Funding Agreement, we have limited recourse but would have the ability to terminate the Funding Agreement by Carlyle and Abingworth. Although termination by us for lack of payment by Carlyle and Abingworth of the US$35 million would relieve us from any repayments under the Funding Agreement, failure to receive the third tranche of US$35 million would have a negative impact on our expected cash runway and ability to complete enrollment in the ongoing trials.
Subsequent to June 30, 2023, we were notified that a new co-investor of Carlyle and Abingworth intends to participate in a funding under the Funding Agreement of US$50 million to increase total Funding Agreement funding from US$120 million to US$170 million, which is subject to the co-investor’s final due diligence and receipt of regulatory and tax approvals, appropriate documentation and compliance with closing conditions. There can be no assurance that the due diligence will be completed to the satisfaction of the co-investor, of Carlyle and Abingworth, the closing terms and conditions will be satisfied, or that the we will ultimately receive the additional US$50 million. Failure to receive the US$50 million would have a negative impact on our expected cash runway and ability to complete enrollment in the ongoing trials.
Pursuant to the Funding Agreement, we will be required to use commercially reasonable efforts to develop Sozinibercept (OPT-302) for the treatment of wet AMD in accordance with the Funding Agreement, including pursuant to certain development timelines set forth therein.
In return, Opthea will pay to Investor (1) upon the first to occur of regulatory approval of Sozinibercept (OPT-302) for the treatment of wet AMD in the United States, United Kingdom or European Union (“Regulatory Approval”), fixed payments equal to a total of approximately two times the funding provided, consisting of seven payments, with the first payment due shortly after Regulatory Approval and the remaining six payments payable over a six-year period thereafter, and (2) variable payments equal to 7% of net sales of Sozinibercept (OPT-302) for the treatment of wet AMD for each calendar quarter.
At the time that Investor receives an aggregate of four times the funding provided (US$680 million if Investor funds the full US$170 million under the Funding Agreement) (the “Cap”), Opthea’s payment obligations under the Funding Agreement will be fully satisfied.
We have the option to satisfy our payment obligations to Investor upon Regulatory Approval or a change of control of Opthea by paying an amount equal to the present value of the remaining payments payable to Investor subject to a mid-single-digit discount rate. We also have an option to buy out the remaining payments at any time by paying an amount equal to the remaining payments due subject to a proposed discount rate, which Investor may accept or reject. Upon a change of control of Opthea, an acceleration payment of a specified multiple of the funding provided is payable, net of payments already made to Investor and creditable against future payments to Investor.
We will grant Investor a security interest in all of our assets (other than intellectual property not related to Sozinibercept (OPT-302)). The security interest will terminate when Investor receives payments and/or change of control acceleration payments equal to two times the funding provided or upon certain terminations of the Funding Agreement (the “Release Date”). The Funding Agreement also includes customary representations and warranties and covenants, including certain negative covenants regarding limitations on incurrence of indebtedness, liens, investments, restricted payments, sales of assets, and royalty sales. The negative covenants will terminate upon the Release Date.
The Funding Agreement terminates upon the payment of all payments owing to Investor, unless earlier terminated by Investor if:
131
The Funding Agreement may also be earlier terminated by Opthea if Investor fails to fund as provided in the Funding Agreement. The Funding Agreement may be terminated by either party (i) if the other party materially breaches the Funding Agreement (“Material Breach”), (ii) if Sozinibercept (OPT-302) fails to receive regulatory approval in the United States or European Union, (iii) upon the bankruptcy of the other party, (iv) if a serious safety concern arises in an Sozinibercept (OPT-302) clinical trial or (v) upon a change of control of Opthea.
In certain instances, upon the termination of the Funding Agreement, we will be obligated to pay Investor a multiple of the amounts paid to us under the Funding Agreement, including specifically,
In addition, if following certain events of termination of the Funding Agreement, Opthea continues to develop Sozinibercept (OPT-302) for the treatment of wet AMD and obtains Regulatory Approval, it will make the payments to Investor as if the Funding Agreement had not been terminated, less any payments made upon termination.
The Funding Agreement also includes a minimum cash requirement, and Opthea may need to obtain additional funding to meet this requirement in the future, including prior to the expected readout of top-line results for its Phase 3 clinical trials. To the extent that Opthea raises additional capital through the sale of equity or convertible debt securities to meet this requirement, Opthea’s equity holders will be diluted.
The payments required under the Funding Agreement are significant. Failure to generate sufficient revenue to make such payments if and as they become due, or failure to otherwise finance such payments would have a material adverse effect on our business. In addition, if we are unable to comply with our obligations under the Funding Agreement and/or one of the termination events described above occurs, Investor may be relieved of their obligation to provide further funding under the Funding Agreement and our payment obligations thereunder may be accelerated. The acceleration of payments under the Funding Agreement would have a material impact on our business and we may not be able to make such payments at such time. We may also require additional external funding to meet the minimum cash condition under our development funding agreement with Investor, including prior to the readout of top-line results for our Phase 3 clinical trials for Sozinibercept (OPT-302) for the treatment of wet AMD. Furthermore, the obligations under the Funding Agreement are secured by a lien on all of our assets (other than intellectual property not related to Sozinibercept (OPT-302)). The security interest will terminate when Investor receives payments and/or change of control acceleration payments equal to two times the funding provided or upon certain terminations of the Funding Agreement. A default under the Funding Agreement may result in a foreclosure on our intellectual property. In addition, we may need to implement further internal controls
132
and processes to ensure compliance with all obligations under the Funding Agreement, otherwise we could inadvertently default under it.
Cash Flows
The following table summarizes our cash flows for the periods presented:
|
|
|
|
|
|
|
|
|||||||
|
|
2023 |
|
|
|
2022 |
|
|
2021 |
|
||||
|
|
|
|
|
|
|
|
|
Restated |
|
||||
|
|
US$ |
|
|
|
US$ |
|
|
US$ |
|
||||
|
|
(in thousands) |
|
|||||||||||
Net cash used in operating activities |
|
|
(120,608 |
) |
|
|
|
(71,335 |
) |
|
|
(45,546 |
) |
|
Net cash provided by investing activities |
|
|
(22 |
) |
|
|
|
(17 |
) |
|
|
656 |
|
|
Net cash provided by financing activities |
|
|
167,285 |
|
|
|
|
172 |
|
|
|
116,936 |
|
|
Net increase/(decrease) in cash and cash equivalents |
|
|
46,655 |
|
|
— |
|
|
(71,180 |
) |
|
|
72,046 |
|
Operating Activities
For the year ended June 30, 2023, net cash used in operating activities was US$120.6 million, attributable to a net loss of US$142.5 million adjusted for US$1.7 million in non-cash items as well as a net cash inflow from changes in operating assets and liabilities of US$13.9 million, partially offset by a R&D tax incentive of US$6.3 million. Non-cash adjustments of US$1.7 million consisted of US$5.9 million in income tax benefit recognized in profit or loss and the fair value adjustment gain on the DFA of US$12.3 offset by US$0.5 million in net exchange differences, US$5.8 million in share-based payments, US$13.5 million of interest expense on the DFA and US$101 thousand in depreciation expense.
For the year ended June 30, 2022, net cash used in operating activities was US$71.3 million, attributable to a net loss of US$92.8 million adjusted for US$1.8 million in non-cash items as well as a net cash inflow from changes in operating assets and liabilities of US$14.7 million, partially offset by a R&D tax incentive of US$4.9 million. Non-cash adjustments of US$1.8 million consisted of US$6.3 million in income tax benefit recognized in profit or loss offset by US$2.8 million in net exchange differences, US$5.2 million in share-based payments and US$78 thousand in depreciation expense.
For the year ended June 30, 2021, net cash used in operating activities was US$45.5 million, attributable to a net loss of US$45.3 million adjusted for US$6.9 million in non-cash items as well as a net cash outflow from changes in operating assets and liabilities of US$16.2 million, partially offset by a R&D tax incentive of US$5.8 million. Non-cash adjustments of US$10.1 million consisted of US$4.9 million in income tax benefit recognized in profit or loss offset by US$11.0 million in net exchange differences, US$3.9 million in share-based payments and US$107 thousand in depreciation expense.
Investing Activities
For the years ended June 30, 2021, 2022 and 2023, net cash provided by investing activities was US $656 thousand, cash outflow of US$17 thousand and US$22 thousand, respectively, attributable to US$669 thousand, US$nil and US$nil, respectively, in cash received on disposal of our financial asset of shares and options in Antisense Therapeutics Ltd in 2020 and Optiscan Imaging Limited in 2021, both ASX-listed companies, offset by cash payments of US$12 thousand, US$17 thousand and US$22 thousand, respectively, for the purchase of computer equipment.
Financing Activities
For the year ended June 30, 2023, net cash provided by financing activities was US$167.3 million, attributable to $81.8 million from capital placement, US$1.0 million received on the exercise of options granted to employees and US$84.5 million under the Development Funding agreement. Net cash provided by financing activities also included US$71 thousand in respect of the payment of lease liabilities.
For the year ended June 30, 2022, net cash provided by financing activities was US$171 thousand, attributable to US$257 thousand received on the exercise of options granted to employees. Net cash provided by financing activities also included US$86 thousand in respect of the payment of lease liabilities.
133
For the year ended June 30, 2021, net cash provided by financing activities was US$117 million, attributable to US$11 million received on the exercise of pre-funded warrants and US$105 million from the US IPO. Net cash provided by financing activities also included US$87 thousand in respect of the payment of lease liabilities.
5C. Research and Development, Patents and Licenses
For a description of the amount spent during each of the last three fiscal years on company-sponsored research and development activities, as well as the components of research and development expenses, see “Item 5A—Operating Results – Results of Operations.”
For a description of our research and development process, see “Item 4B—Business Overview.”
5D. Trend Information
For a discussion of trends, see “Item 4B—Business Overview,” “Item 5A—Operating Results” and “Item 5B—Liquidity and Capital Resources.”
5E. Critical Accounting Estimates
Critical Accounting Policies and Estimates
We believe that the following accounting policies involve a high degree of judgment and complexity. Accordingly, these are the policies we believe are the most critical to aid in fully understanding and evaluating our consolidated financial condition and results of our operations. See note 3 to our consolidated financial statements appearing elsewhere in this annual report for a description of our other significant accounting policies. The preparation of our consolidated financial statements in conformity with IFRS requires us to make estimates and judgments that affect the amounts reported in those financial statements and accompanying notes. Although we believe that the estimates we use are reasonable, due to the inherent uncertainty involved in making those estimates, actual results reported in future periods could differ from those estimates.
Research and Development Costs
The majority of our expenditures is incurred as a result of our clinical trials for Sozinibercept (OPT-302). During the years ended June 30, 2022 and 2023, we progressed Phase 3 wet age-related macular degeneration (wet AMD) clinical trial of Sozinibercept (OPT-302) combination therapy for the treatment. A key measure of our performance is the level of expenditures incurred on the research and development of Sozinibercept (OPT-302).
Judgment is required in relation to:
We have determined that we are still in a research phase, and accordingly, no development costs have been capitalized as of June 30, 2021, 2022 or June 30, 2023.
Financial liabilities
Financial liabilities are recognized in our statement of financial position when we become a party to the contractual provisions of the instrument. Financial liabilities are initially measured at fair value. Transaction costs that are directly attributable to the acquisitions or issue of financial liabilities (other than financial liabilities at fair value through profit or loss) are deducted from the fair value of the financial liabilities, as appropriate, on initial recognition. Subsequent measurement of the liability will be at its amortized cost, subject to any re-measurement of the obligation for changes in assumptions.
Amortized cost and effective interest method
134
The effective interest method is a method of calculating the amortized cost of an instrument and of allocating interest expense over the relevant period. The effective interest rate is the rate that exactly discounts estimated future cash payments through the expected life of the financial liability, or (where appropriate) a shorter period, to the amortized cost of the financial liability. Interest expense is recognized in profit and loss and is included in the “Interest expense on DFA” line item.
Revaluation
At every reporting period, we will review the expected approval and commercial launch dates. If the dates are delayed from those used at previous reporting period, it is expected that a revaluation will result in another non-cash gain. If the timelines for approval and launch are accelerated, the Company would anticipate a revaluation resulting in a non-cash charge to be recognized on the Profit and Loss statement. The gains or losses are unrealized.
Share-based Payment Transactions
We provide benefits to our directors and employees (including key management personnel) in the form of share-based payments, whereby employees render services in exchange for ordinary shares or rights over ordinary shares (equity-settled transactions). The cost of these equity-settled transactions with employees is measured by reference to the fair value at the date at which they are granted. Binomial models are used to value the options issued, with key assumptions being the listed price per ordinary share on the grant date, the option exercise price, expected volatility of the underlying ordinary shares based on the historical share price volatility and the risk-free interest rate.
The cost of the equity-settled transactions is recognized, together with a corresponding increase in equity, over the period in which the performance conditions are fulfilled (the vesting period), ending on the date on which the relevant employees become fully entitled to the award (the vesting date). The charge to profit or loss for the period is the cumulative amount less the amounts already charged in previous periods. There is a corresponding credit to equity.
Until an award has vested, any amounts recorded are contingent and will be adjusted if more or fewer awards vest than were originally anticipated to do so. Should one or more of the assumptions and estimates used in estimating the fair value of share‑based payments change, this could have a material impact on the amounts recognized in equity and employee‑related expenses.
Income Tax—Research and Development Tax Incentive
The R&D Tax Incentive Scheme is an Australian Federal Government program under which eligible companies with annual aggregated revenue of less than A$20 million can receive cash amounts equal to 43.5% of eligible research and development expenditures from the ATO. The R&D Tax Incentive Scheme incentive relates to eligible expenditures incurred in Australia and, under certain circumstances, overseas on the development of our lead candidate Sozinibercept (OPT-302). The R&D tax incentive is applied annually to eligible expenditures incurred during our financial year following an annual application and subsequent filing of our income tax return after the financial year end.
We estimate the amount of R&D tax incentive after the completion of the financial year based on eligible Australia and overseas expenditures incurred during that year.
We have presented incentives in respect of the R&D Tax Incentive Scheme within income tax benefit in the Statement of Profit or Loss and Other Comprehensive Income by analogizing with IAS 12 “Income Taxes.”
Judgment is required as to the eligibility for the R&D tax incentive in respect of:
135
For the years ended June 30, 2022 and 2023, we have recognized an R&D tax incentive receivable of US$6.3 million and US$5.9 million, respectively within our consolidated statement of financial position with a corresponding amount recognized within income tax benefit within the consolidated statement of profit or loss and other comprehensive income. The R&D tax incentive receivable as of June 30, 2022 and June 30, 2023 is based on the legislation as currently enacted as at June 30, 2022 and June 30, 2023 respectively. Any proposed changes to the legislation, such as rate changes or the eligibility requirements, may have a retrospective impact if the legislation is passed, currently no such legislative changes have occurred.
Investment tax credits such as the R&D tax incentive are outside of the scope of IAS 12 “Income Taxes” and IAS 20 “Accounting for Government Grants and Disclosure of Government Assistance.” Based on the guidance in IAS 8 “Accounting Policies, Changes in Accounting Estimates and Errors,” companies need to make an accounting policy choice on how to present these incentives, which in practice is done by either analogizing with IAS 12 or with IAS 20. We have determined that the R&D tax incentive should be presented by analogizing to IAS 12 because the nature of the incentive is considered to be more closely aligned to income taxes, based on the following considerations:
Income tax
The Group’s accounting policy for taxation requires judgments as to the differences between tax and accounting treatments of income and costs recognized in the Consolidated Statement of Profit or Loss and Other Comprehensive Income. Judgment is also required in assessing whether deferred tax assets and liabilities are recognized in the statement of financial position and if accumulated income tax losses can be used to offset potential future tax profits.
Recently Adopted Accounting Pronouncements relating to the Financial Statements
Amendments to Accounting Standards that are Mandatorily Effective for the Current Year
We have adopted all of the new and revised Standards and Interpretations issued by the IASB that are relevant to our operations and effective for the current year.
During the fiscal year ended June 30, 2023, we applied a number of amendments to IFRS and Interpretations issued by the IASB that are effective for an annual period that begins on or after January 1, 2022. Their adoption has not had any material impact on the disclosures or on the amounts reported in our consolidated financial statements.
New and Revised International Financial Reporting Standards and Interpretations on Issue But not Yet Effective
The new and revised International Financial Reporting Standards, Interpretations and amendments that have been issued but are not yet effective, are not expected to have a material impact on the amounts recognized or disclosures included in our consolidated financial statements.
136
Qualitative and Quantitative Disclosures about Market Risk
We are exposed to market risks in the ordinary course of our business. Market risk represents the risk of loss that may impact our financial position due to adverse changes in financial market prices and rates. Our market risk exposure is primarily the result of fluctuations in interest rates and foreign currency exchange rate risk.
Interest Rate Risk
As of June 30, 2023, we had cash and cash equivalents of US$89.2 million, including US$77.2 million in short term deposit accounts. We have limited exposure to interest rate risk. Our exposure to market interest rates relates primarily to the short-term deposits. The deposits are held with two of Australia’s largest banks. Our cash and cash equivalents are not locked into long-term deposits at fixed rates so as to mitigate the risk of earning interest below the current floating rate. We do not have any credit facilities bearing variable interest rates.
Foreign Currency Exchange Rate Risk
As a result of services provided by non-related entities in the United States, Canada, United Kingdom and Europe, part of our financial assets and liabilities and foreign currency denominated transactions are affected by movements in the applicable exchange rate. We do not enter into any hedging transactions. We enter into forward rate foreign exchange rate contracts in respect of the settlement of supplier invoices denominated in U.S. dollars to mitigate the risk of movements in the Australian dollar and U.S dollar exchange rates. As of June 30, 2022 and June 30, 2023, we had US$33.3 million and US$60.4 million, respectively, in net exposure to the Australian dollar and U.S. dollar, primarily in payables and cash. An increase or decrease of the Australian dollar to U.S. dollar exchange rate by 10% would increase our after-tax loss by US$3.8 million (2022: US$2.1 million) or decrease our after tax loss by US$4.7 million (2022: US$2.6 million), respectively. As we continue our clinical development activities, we expect to face continued exposure to exchange rate risk from the U.S. dollar. There was minimal or insignificant exposure to the British Pound, Euro and Canadian dollar during the years ended June 30, 2022 and 2023.
Emerging Growth Company Status
As a company with less than US$1.235 billion in revenue during our last fiscal year, we qualify as an “emerging growth company” as defined in the JOBS Act. As an emerging growth company, we may take advantage of specified reduced disclosure and other requirements that are otherwise applicable generally to public companies. These provisions include:
We may take advantage of these exemptions until such time that we are no longer an emerging growth company. Accordingly, the information that we provide shareholders and holders of the ADSs may be different than you might obtain from other public companies. We will cease to be an emerging growth company upon the earliest to occur of (i) the last day of the fiscal year in which we have more than US$1.235 billion in annual revenue; (ii) the last day of the fiscal year in which we qualify as a “large accelerated filer”; (iii) the date on which we have, during the previous three-year period, issued more than US$1.0 billion in non-convertible debt securities; and (iv) June 30, 2026.
Foreign Private Issuer Status
We are also considered a “foreign private issuer” under U.S. securities laws. In our capacity as a foreign private issuer, we are exempt from certain rules under the Securities Exchange Act of 1934, as amended, that impose certain disclosure obligations and procedural requirements for proxy solicitations under Section 14 of the Exchange Act. In addition, our senior management, the members of our board of directors and our principal shareholders are exempt from the reporting and “short-swing” profit recovery provisions of Section 16 of the Exchange Act and the rules under the Exchange Act with respect to their purchases and sales of our securities. Moreover, we are not required to file periodic reports and financial statements with the SEC as frequently or as
137
promptly as U.S. companies whose securities are registered under the Exchange Act. In addition, we are not required to comply with Regulation FD, which restricts the selective disclosure of material information.
We may take advantage of these exemptions until such time as we are no longer a foreign private issuer. We will remain a foreign private issuer until such time that 50% or more of our outstanding voting securities are held by U.S. residents and any of the following three circumstances applies: (i) the majority of the members of board of directors or our senior management are U.S. citizens or residents; (ii) more than 50% of our assets are located in the United States; or (iii) our business is administered principally in the United States.
We have taken advantage of certain reduced reporting and other requirements in this annual report. Accordingly, the information contained herein may be different from the information you receive from other public companies.
138
Item 6. Directors, Senior Management and Employees
6A. Directors and Senior Management
The following table sets forth information relating to our directors, senior management and key employees as of June 30, 2023.
Name |
|
|
Age |
|
|
Position |
Senior Management and Key Employees |
|
|
|
|
|
|
Megan Baldwin, Ph.D. |
|
|
48 |
|
|
Chief Executive Officer and Managing Director |
Timothy E Morris |
|
|
62 |
|
|
Chief Financial Officer |
Karen Adams |
|
|
52 |
|
|
Vice President Finance and Company Secretary |
Judith Robertson |
|
|
63 |
|
|
Chief Commercial Officer |
Joel Naor (1) |
|
|
60 |
|
|
Chief Medical Officer |
Mark O’Neill |
|
|
57 |
|
|
Vice President Chemistry, Manufacturing and Controls |
Michael Gerometta, Ph.D. |
|
|
58 |
|
|
Head of Chemistry, Manufacturing and Controls Development |
Bruno Gagnon |
|
|
55 |
|
|
Senior Vice President Global Clinical Operations |
Ian Leitch, Ph.D. |
|
|
59 |
|
|
Director of Clinical Research |
Non-Employee Directors |
|
|
|
|
|
|
Julia Haller, M.D. |
|
|
68 |
|
|
Director |
Jeremy Levin, D.Phil, MB BChir |
|
|
69 |
|
|
Chairperson |
Lawrence Gozlan |
|
|
44 |
|
|
Director |
Susan Orr |
|
|
61 |
|
|
Director |
Anshul Thakral |
|
|
45 |
|
|
Director |
Daniel Spiegelman |
|
|
65 |
|
|
Director |
Quinton Oswald |
|
|
72 |
|
|
Director |
(1) Dr. Naor resigned on July 15, 2023.
The business addresses for our senior management and board of directors is Opthea Limited, Suite 0403, Level 4, 650 Chapel Street, South Yarra, VIC 3141, Australia.
Senior Management and Key Employees
Megan Baldwin, Ph.D., has served as our Chief Executive Officer and Managing Director since February 2014. Since joining our company in 2008, Dr. Baldwin has held various positions, including Head of Preclinical R&D from February 2009 to November 2012 and Chief Executive Officer of Opthea Pty Ltd., previously a wholly-owned subsidiary, from November 2012 to December 2015. Dr. Baldwin has over 20 years of experience focusing on angiogenesis and therapeutic strategies for ophthalmic and cancer indications. Prior to joining our company, Dr. Baldwin was employed at Genentech, Inc. (now a subsidiary of the Roche Group), a leader in the field of angiogenesis-based therapies for cancer and other diseases. Dr. Baldwin earned a Bachelor of Science Honours and Ph.D. in Medicine from the University of Melbourne. We believe that Dr. Baldwin’s business expertise and her daily insight into corporate matters as our Chief Executive Officer qualify her to serve on our board of directors.
Timothy Morris, has served as our Chief Financial Officer since October 2022. Mr. Morris is a veteran pharmaceutical executive with extensive professional finance and accounting background in both public and private companies, including over 25 years in public biotechnology companies as CFO. Prior to joining Opthea, he served as Chief Operating Officer/Chief Financial Officer at Humanigen, Inc. since August 2020. Previously, Mr. Morris was Chief Financial Officer of Iovance Biotherapeutics which raised over US$1 billion in four offerings to fund expansion of the clinical development program, build manufacturing capability and to prepare for commercialization. Prior to IOVA, Mr. Morris was Chief Financial Officer of AcelRx Pharmaceuticals, VIVUS Inc., and Questcor Pharmaceuticals Inc. He is currently on the Board of Directors of DBV Technologies S.A., Aquestive Therapeutics, Humanetics Corporation and Univercells S.A. Mr. Morris earned his Bachelor of Science in Business from California State University, Chico.
139
Karen Adams, has served as our Vice President Finance since May 2021 and as our Secretary since June 2021. Prior to joining Opthea, Karen was the Chief Financial Officer of the Victor Smorgon Group, an investment management company, from December 2018 to May 2021. From February 2014 to August 2018, she served as the Director of Finance of Nexvet Biopharma Pty Ltd, a veterinary biologic therapeutics company. Karen holds a Graduate Degree in Business from Swinburne University and is a member of the Australian Society of Chartered Accountant, Graduate of Australian Institute of Company Directors and a Fellow of the Institute of Company Secretaries.
Judith Robertson has served as our Chief Commercial Officer since January 1, 2022. Ms. Robertson served as a member of our board of directors from June 2021 to January 1, 2022. Since 2019, Ms. Robertson has served as a member of the board of directors of Durect Corporation, a biotechnology company. From December 2020 to April 2021, Ms. Robertson served as the Chief Commercial Officer of Eleusis Ltd. From December 2016 she served as the Chief Commercial Officer of Aerie Pharmaceuticals Inc., a publicly held biotechnology company. Prior to Aerie, Ms. Robertson was Vice President Immunology and Ophthalmology Global Commercial Strategy Leader at Johnson and Johnson, Janssen Pharmaceuticals, and Vice President, Ophthalmology Global Business Franchise Head at Novartis (formerly Alcon). Ms. Robertson’s prior experience also includes sales and marketing roles at Novartis, Bristol Myers Squibb and Searle USA. Ms. Robertson received Bachelor of Arts from Ryerson University and a Master of Business Administration from Kellogg School of Management at Northwestern University.
Joel Naor, MD MBS MSc., has served as our Chief Medical Officer since March 2022 and resigned on July 15, 2023. Has over two decades of experience leading clinical development programs that target retina conditions encompassing biologics, small molecules, sustained release technologies, stem cells and photodynamic therapy. In his most recent role, he served as Vice President of Clinical Science and Development Operations at Kodiak Sciences Inc, a biopharmaceutical company. Previously, Dr. Naor was the Chief Medical Officer for Macusight, Inc. until the company was acquired by Santen Inc. in 2010, and subsequently served as Vice President and Head of Global Medical Affairs for Santen Inc. He has also held leadership positions at Allergan Inc. a health care company, QLT Inc. a biopharmaceutical company. and Stem Cells Inc., a biopharmaceutical company. Dr. Joel Naor received his Doctor of Medicine (M.D.) from the Technion – Israel Institute of Technology and completed training in Ophthalmology at the University of Toronto. He holds a Master of Science (MSc.) in Epidemiology from the University of Toronto and a Master of Business Administration (M.B.A.) from Simon Fraser University in Vancouver.
Mark O Neill MSc. B Chem Eng, has served as our Vice President of Chemistry, Manufacturing and Controls since January 2022. Mr. O’Neill was most recently head of Process Development for Avexis Gene therapies where he orchestrated all product development and technical operations activities pertaining to the startup and licensure of Zolgensma drug substance manufacturing at the Colorado site. Prior to Avexis, he was Vice President and General Manager of the Thermo Fisher Groningen Single Use Biologics Manufacturing Facility in Groningen, The Netherlands, where he oversaw all operations including startup of commercial manufacturing and initial commercial licensure at the facility. Mr. O’Neill has over 30 years of experience in the manufacturing of biopharmaceuticals including 20 years with Amgen where he gained extensive experience in all aspects of lifecycle management including Quality, Engineering, Production, Development, Supply Chain and Business Development. Mark holds a Master of Science Degree from Colorado School of Mines in Environmental and Chemical Engineering and a Bachelor of Science Degree in Chemical Engineering from the University of Colorado
Bruno Gagnon, has served as our Senior Vice President, Global Clinical Operations since July 2022. Mr Gagnon is former Sr. Vice President of Development Operations at Eidos Therapeutics, a BridgeBio company in San Francisco, CA. Mr Gagnon also served as Vice President of Clinical Operations at BioMarin Pharmaceutical. Previously, held positions of increasing responsibilities at Roche Diagnostics, Chiron and Hoechst Marion Roussel (now Sanofi). Over his 30-year career, functions under his leadership have included Global Clinical Trial Management, Patient Advocacy, Medical Writing, Outsourcing and Contracts, Supply Chain Management, Clinical Data Management, Clinical Systems, Document Management and Clinical Training. He has earned a Bachelor’s Degree from the School of Pharmacy, Laval University and a Master’s in Pharmaceutical Sciences from University of Montreal, both in Quebec, Canada.
140
Michael Gerometta, Ph.D., has served as our Head of Chemistry, Manufacturing and Controls Development since December 2008. Dr. Gerometta has over 30 years of experience in the Australian biotechnology industry, working with numerous contract manufacturing organizations overseas and locally in all facets of translational CMC from concept through to Phase 2 studies. Dr. Gerometta earned a Bachelor of Science in Chemistry from the University of Technology, Sydney, and a Ph.D. in Biotechnology from the Queensland University of Technology.
Ian Leitch, Ph.D., has served as one of our Directors of Clinical Research since September 2011. From September 2006 to September 2011, Dr. Leitch was a member of the Medical Sciences group at Amgen Inc., a biopharmaceutical company, and had responsibility for the design and management of clinical studies in oncology. From 1998 to 2006, Dr. Leitch was Senior Program Manager for Cardiovascular Research and Clinical Study Director for Ophthalmology at Miravant Medical Technologies Inc., a biopharmaceutical company. He earned a Bachelor of Science with Honours and a Ph.D. from the Department of Pharmacology, Faculty of Medicine, at Monash University and completed part of the doctoral studies at the University of California, Santa Barbara.
Non-Employee Directors
Julia Haller, M.D. has served as a member of our board of directors since June 2021. Since 2007, Dr. Haller has served as Ophthalmologist-in-Chief and William Tasman, ME Endowed Chair at Wills Eye Hospital in Philadelphia. Dr. Haller is Professor and Chair of the Department of Ophthalmology at the Sidney Kimmel Medical College at Thomas Jefferson University as well as a director of Bristol Myers Squibb, a biopharmaceutical company and Outlook Therapeutics, a biopharmaceutical company. She is also a member of the National Academy of Medicine, the Chair of the College of Physicians of Philadelphia, Chair of the Heed Ophthalmic Society, past president of the Women in Medicine Legacy Foundation, and serves on several prestigious boards including John Hopkins Medical and Surgical Association (immediate past president), the Association of University Professors of Ophthalmology, and the Society of Heed Fellows. Dr. Haller received a Bachelor of Arts from Princeton University and an M.D. from Harvard Medical School. We believe Dr. Haller’s extensive experience as an internationally recognized ophthalmologist and vitreoretinal surgeon qualify her to serve on our board of directors.
Lawrence Gozlan has served as a member of our board of directors since July 2020. Since 2007, Mr. Gozlan has served as the Life Sciences Investment Manager of Jagen Pty Ltd., an international investment organization. Mr. Gozlan has also served as a member of the board of directors of Alterity Therapeutics Ltd., a drug development company, since 2011. Mr. Gozlan earned a Bachelor of Science in Microbiology and Immunology from the University of Melbourne. We believe Mr. Gozlan’s extensive investment experience in biotechnology and life sciences companies qualify him to serve on our board of directors.
Jeremy Levin, D.Phil., MB BChir has served as the Chairperson of our board of directors since October 2020. Since March 2015, Dr. Levin has served as the Chief Executive Officer, and since April 2014, as the chairperson of the board of directors, of Ovid Therapeutics Inc., a biopharmaceutical company. From May 2012 to October 2013, Dr. Levin served as the President and Chief Executive Officer of Teva Pharmaceutical Industries Ltd., a publicly held pharmaceutical company. From September 2007 to December 2012, Dr. Levin held several roles at Bristol-Myers Squibb Company, a publicly held pharmaceutical company, ultimately serving as the Senior Vice President of Strategy, Alliances and Transactions. Dr. Levin also served as a member of the executive committee at Bristol-Myers Squibb Company. Dr. Levin earned a Bachelor of Arts in Zoology, Master of Arts in Cell Biology and D.Phil. in Chromatin Structure, all from University of Oxford, and a Bachelor of Medicine, Bachelor of Surgery from the University of Cambridge. We believe Dr. Levin’s extensive experience in the global biotechnology and pharmaceutical industry qualify him to serve on our board of directors.
Susan Orr, OD has served as a member of our board of directors since April 2022. Dr. Orr is an experienced medical and business leader with specialization in identifying, developing, and commercializing ophthalmic therapeutic product candidates. Since February 2020, Dr. Orr has served as the Chief Medical Officer at Claris Biotherapeutics, a biotechnology firm, and is a member of the Retina Global Board of Directors. From October 2018 to November 2019, Dr. Orr was the Chief Executive Officer at Notal Vision, an ophthalmic remote monitoring service provider. From July 2016 to September 2018, Dr. Orr served as Notal Vision’s Chief Medical Officer and Vice President of Medical Affairs. Prior to joining Notal Vision, Dr. held leadership roles at Alcon and Janssen across Marketing, Strategy, Business Development, and multiple Development functions, following 10 years in private practice in Canada. Dr. Orr earned a Bachelor of Science and a Doctor of Optometry from the University of Waterloo. We believe Dr. Orr’s extensive experience in ophthalmology and leadership qualifies her to serve on our board of directors.
141
Anshul Thakral has served as a member of our board of directors since June 2023. Mr. Thakral is Chief Executive Officer and Board Member of Launch Therapeutics, a clinical development company backed by funds managed by global investment firm Carlyle and its life sciences franchise, Abingworth. Mr. Thakral has worked for over 20 years in the pharmaceutical and biotechnology industry and is an experienced executive, management consultant and entrepreneur. Mr. Thakral was previously Chief Commercial Officer and Executive Vice President of Peri and Post-Approval Services at PPD, and prior to that was Global Head of PPD Biotech. Before PPD, Mr. Thakral ran the global life sciences business unit at Gerson Lehrman Group and worked at McKinsey & Company as an associate principal in the health care practice, where he provided strategic advice to global pharmaceutical and biotechnology companies on growth, research and development, business development and commercialization. He currently serves on the boards of TriNetX, Saama Technologies, Orsini Specialty Pharmacy, is an Operating Executive at Carlyle and is a Venture Partner at Abingworth. Mr. Thakral holds a Master’s degree in Biomedical Engineering from Johns Hopkins University and a Masters Business Administration (MBA) from the Wharton School at the University of Pennsylvania. We believe Mr. Thakral’s extensive experience in the global biotechnology and pharmaceutical industry qualifies him to serve on our board of directors.
Daniel Spiegelman has served as a member of our board of directors since September 2020. From May 2012 to January 2020, Mr. Spiegelman served as Executive Vice President, Chief Financial Officer of BioMarin Pharmaceutical Inc., a biotechnology company. Mr. Spiegelman currently serves as Board Chairman for Tizona Therapeutics, and Audit Committee Chairman and for Myriad Genetics (NASDAQ: MYRD), Spruce Biosciences (NASDAQ: SPRB), Maze Therapeutics and Kyverna Therapeutics, and has previously served as board director for several other companies. Mr. Spiegelman has also served as a Venture Partner with Samsara BioCapital Since May 2023. From October 2008 to March 2018, Mr. Spiegelman served as a member of the board of directors of Cascadian Therapeutics, Inc., a publicly held biotechnology company that was acquired by Seattle Genetics, Inc. in 2018. From May 2009 to May 2012, Mr. Spiegelman served as a consultant to provide strategic financial management support to a portfolio of public and private life science companies. Mr. Spiegelman has also served as a member of the board of directors of Myriad Genetics, a molecular diagnostic company, since May 2020. Mr. Spiegelman earned a Bachelor of Arts from Stanford University and a Master of Business Administration from the Stanford Graduate School of Business. We believe Mr. Spiegelman’s extensive leadership experience in biotechnology and pharmaceutical companies qualify him to serve on our board of directors.
Quinton Oswald has served as a member of our board of directors since April 2022. Mr. Oswald brings over 25 years of international general management experience, including onsite assignments in the U.S, Europe and South Africa. From April 2016 to December 2018, Mr. Oswald was the Chief Executive Officer of Notal Vision, a commercial stage ophthalmic home monitoring services provider with a focus on both wet and dry AMD. From April 2013 to April 2016, Mr Oswald served as the President and Chief Executive Officer of Neurotech Pharmaceuticals, a biotechnology research company, and from September 2010 to April 2013, as the Chief Executive Officer of SARcode Bioscience, an ophthalmic biopharmaceutical company. We believe Mr. Oswald's extensive leadership experience in biotechnology companies qualifies him to serve on our board of directors.
142
Family Relationships
There are no family relationships among any of the members of our board of directors and our senior management.
Board Diversity Matrix
The following board diversity matrix sets forth the information concerning the gender, demographic background and certain other characteristics of our board of directors as of the date of this annual report, as self-identified by its members, in accordance with Rule 5606 of the Nasdaq Listing Rules.
Board Diversity Matrix (As of September 12, 2023) |
||||
Country of Principal Executive Offices: |
Australia |
|||
Foreign Private Issuer |
Yes |
|||
Disclosure Prohibited under Home Country Law |
No |
|||
Total Number of Directors |
8 |
|||
|
Female |
Male |
Non- Binary |
Did Not Disclose Gender |
Part I: Gender Identity |
|
|||
Directors |
3 |
5 |
- |
- |
Part II: Demographic Background |
||||
Underrepresented Individual in Home Country Jurisdiction |
- |
|||
LGBTQ+ |
- |
|||
Did Not Disclose Demographic Background |
1 |
|||
6B. Compensation
Overview
Our remuneration policy is to align director and senior management objectives with shareholder and business objectives by providing a fixed remuneration component and typically offering long-term incentives based on key performance areas. Our board of directors believes the remuneration policy to be appropriate and effective in its ability to attract and retain the best executives and directors to run and manage the consolidated entity, as well as create goal congruence between directors, executives and shareholders. Our board of directors and the Remuneration Committee are responsible for determining the appropriate remuneration package for our senior management, including our Chief Executive Officer.
Australian executives and directors receive a superannuation guarantee contribution required under Australian law and do not receive any other retirement benefits.
Remuneration of Senior Management
For the fiscal year ended June 30, 2023, the aggregate cash remuneration paid to our senior management was US$2,291,360 (2022:US$1,216,106).
Our senior management receive fixed compensation and performance-linked remuneration. The level of fixed remuneration is set to provide a base level of compensation which is both appropriate to the applicable position and is competitive in the marketplace. The Remuneration Committee accesses external advice independent of senior management if required. Fixed compensation is comprised of base salary and superannuation contribution and is reviewed every 12 months by the Remuneration Committee.
143
Performance-linked remuneration consists of short-term and long-term incentives. The objective of short-term incentives is to link the achievement of our operational targets with the remuneration received by our senior management charged with meeting those targets. Total potential short-term incentives are set at a level that we believe provides sufficient incentive to our senior management to achieve the operational targets at a cost to us that is reasonable under the circumstances. Short-term incentives may include cash bonuses based on the extent to which specific targets set at the beginning of each fiscal year are met. The targets consist of a number of key performance indicators covering corporate objectives and individual measures of performance. Individual performance indicators are linked to our development plans. Our Remuneration Committee determines, on an annual basis and after consideration of performance against the key performance indicators, the amount, if any, of short-term incentives payable to our senior management. Payments of short-term incentive bonuses are made in the following reporting period.
We also provide long-term incentives through option grants under our Long-Term Incentive Plan. The objective of the Long-Term Incentive Plan is to reward our management and key employees in a manner that aligns this element of compensation with the creation of shareholder wealth. Long Term Incentive Plan grants are made to senior management and employees who are able to influence the generation of shareholder wealth and have a direct impact on our performance and development. Option vesting conditions are based on continued service to us.
In making remuneration determinations for our senior management, the Remuneration Committee considers operational contributions by our senior management as well as the following performance indicators: revenue, loss before tax, tax benefit, loss after tax, basic loss per ordinary share, net tangible assets per share and changes in prevailing trading prices of our ordinary shares on the ASX.
Remuneration of Non-Employee Directors
Our non-employee directors receive a fixed fee annually, which is reviewed by our board of directors on an annual basis. Dr. Orr and Mses. Haller, and Messrs. Oswald and Gozlan are each entitled to an annual fixed fee of US$50,000/A$65,700, and Mr. Thakral is also entitled to the same annual fee pro-rata for the period of service to our board. Mr. Spiegelman is entitled to an annual fixed fee of US$75,000 (inclusive of fees for his service as chairperson of the audit and risk committee and nominations committee member fee). Dr. Levin is entitled to an annual fixed fee of US$75,000 (for his service as chairperson of the board of directors). Board members are entitled to an annual fixed fee of US$10,000/A$13,140 for service as Chair of any other committees and US$5,000/A$6,570 for service on any other committee in a non-Chair role. Unless otherwise noted, the fixed fees cover both service on the board of directors and committees of the board of directors. The remuneration of our non-employee directors is reviewed by our board of directors on an annual basis. Non-employee directors are not provided with retirement benefits apart from statutory superannuation, which is only applicable to Australian resident directors. Non-employee directors are reimbursed for costs directly related to conducting business related to their service on our board of directors.
We implemented a non-executive director share and option plan, or the NED Plan, in 2014. Under the NED Plan, present and future non-executive directors may:
The NED Plan is designed to assist us in preserving our cash for use toward advancing the clinical development of our product candidate and provide our non-employee directors an opportunity to demonstrate their commitment and support for us through sacrificing some or all of their cash fees for ordinary shares or options. The NED Plan also provides us with further flexibility in the design of the directors’ remuneration packages and in turn assists us with retaining existing directors and attracting new additional directors with the relevant experience and expertise.
144
For the fiscal year ended June 30, 2023, the aggregate cash remuneration paid to our non-employee directors was US$615,492 (2022:US$395,656), including Nil in cash reimbursements.
Employment Agreements with Senior Management
The key provisions of the employment agreements are set out below for each member of our senior management. None of these employment agreements have termination dates. The base salary under the employment agreements may be increased by the board of directors from time to time, including by 3% for the fiscal year ended June 30, 2022.
Officer |
|
Date of |
|
Base Salary |
|
Termination without Cause |
|
Benefits upon Termination without |
Megan Baldwin, Ph.D. |
|
April 23, |
|
A$575,000 per year |
|
Not less than three months’ notice (if by employee) Not less than twelve months’ notice or payment in lieu of notice period (if by us) |
|
Upon notice of termination by us, any options that have vested or will vest during the notice period will be released; all other options will lapse at the discretion of our board of directors. |
Karen Adams |
|
June 15, |
|
A$333,500 per year |
|
Not less than three months’ notice (if by employee) Not less than three months’ notice or payment in lieu of notice period (if by us) |
|
Not applicable. |
Timothy Morris Chief Financial Officer |
|
October 24, 2022 |
|
US$475,000 per year |
|
Not less than twelve months’ notice or payment in lieu of notice period (if by us) |
|
Upon notice of termination by us, any options that have vested or will vest during the notice period will be released; all other options will lapse at the discretion of our board of directors |
Joel Naor Chief Medical Officer |
|
March 1, 2022 |
|
US$450,000 per year |
|
Not less than twelve months’ notice or payment in lieu of notice period (if by us) |
|
Upon notice of termination by us, any options that have vested or will vest during the notice period will be released; all other options will lapse at the discretion of our board of directors |
Judith Robertson Chief Commercial Officer |
|
January 1, 2022 |
|
US$390,000 per year |
|
Not less than twelve months’ notice or payment in lieu of notice period (if by us |
|
Upon notice of termination by us, any options that have vested or will vest during the notice period will be released; all other options will lapse at the discretion of our board of directors |
Upon termination of employment, our senior management are entitled to receive their statutory entitlements of accrued annual and long service leave, together with any superannuation benefits.
145
Remuneration of Our Non-Employee Directors and Senior Management During the Fiscal Year Ended June 30, 2023
Details of the remuneration of our non-employee directors and senior management for the fiscal year ended June 30, 2023 are set forth below.
|
|
|
|
|
Short- |
|
|
Post- |
|
|
|
|
|
|
|
|||||||||
|
|
Salary/ |
|
|
Cash |
|
|
Benefits |
|
|
Superannuation |
|
|
Share- |
|
|
Total |
|
||||||
Non-Employee Directors |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Julia Haller |
|
$ |
59,464 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
267,948 |
|
|
$ |
327,412 |
|
Laurence Gozlan(3) |
|
|
230,644 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
493,665 |
|
|
|
724,309 |
|
Jeremy Levin |
|
|
75,000 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
350,472 |
|
|
|
425,472 |
|
Susan Orr (4) |
|
|
58,928 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
147,267 |
|
|
|
206,195 |
|
Michael Sistenich (5) |
|
|
58,201 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
145,079 |
|
|
|
203,280 |
|
Daniel Spiegelman |
|
|
75,000 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
487,695 |
|
|
|
562,695 |
|
Quinton Oswald (4) |
|
|
55,096 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
147,267 |
|
|
|
202,363 |
|
Anshul Thakral |
|
|
3,159 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
3,159 |
|
Senior Management |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Megan Baldwin, Ph.D. |
|
|
386,036 |
|
|
|
133,634 |
|
|
|
— |
|
|
|
80,059 |
|
|
|
969,820 |
|
|
|
1,569,549 |
|
Timothy Morris (7) |
|
|
327,628 |
|
|
|
36,630 |
|
|
|
40,058 |
|
|
|
— |
|
|
|
370,630 |
|
|
|
774,946 |
|
Karen Adams |
|
|
224,126 |
|
|
|
44,290 |
|
|
|
— |
|
|
|
36,729 |
|
|
|
202,452 |
|
|
|
507,597 |
|
Judith Robertson (8) |
|
|
390,000 |
|
|
|
140,400 |
|
|
|
52,990 |
|
|
|
5,200 |
|
|
|
268,242 |
|
|
|
856,832 |
|
Joel Noar (9) |
|
|
450,000 |
|
|
|
— |
|
|
|
57,259 |
|
|
|
15,180 |
|
|
|
370,935 |
|
|
|
893,374 |
|
146
Details of options held by our non-employee directors and senior management as of June 30, 2023 are set forth below.
|
|
Number of |
|
|
Grant |
|
Exercise |
|
|
Percentage |
|
|
Last Vesting |
|
Expiration |
|||
Non-Employee Directors(2) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
Lawrence Gozlan |
|
|
2,000,000 |
|
|
10/12/2020 |
|
$ |
3.24 |
|
|
|
75 |
% |
|
10/11/2023 |
|
10/11/2024 |
Lawrence Gozlan |
|
|
500,000 |
|
|
11/16/2022 |
|
Nil |
|
|
|
0 |
% |
|
11/15/2032 |
|
11/16/2032 |
|
Lawrence Gozlan |
|
|
2,000,000 |
|
|
11/16/2022 |
|
|
0.658 |
|
|
|
21 |
% |
|
11/15/2032 |
|
11/16/2032 |
Julia Haller |
|
|
2,000,000 |
|
|
10/19/2021 |
|
|
0.948 |
|
|
|
50 |
% |
|
10/19/2024 |
|
10/18/2025 |
Jeremy Levin |
|
|
3,000,000 |
|
|
01/19/2021 |
|
|
1.56 |
|
|
|
75 |
% |
|
01/19/2024 |
|
01/18/2025 |
Michael Sistenich |
|
|
1,500,000 |
|
|
11/16/2022 |
|
|
0.658 |
|
|
|
21 |
% |
|
11/29/2019 |
|
11/29/2022 |
Daniel Spiegelman |
|
|
2,000,000 |
|
|
10/12/2020 |
|
|
2.16 |
|
|
|
75 |
% |
|
10/11/2023 |
|
10/11/2024 |
Daniel Spiegelman |
|
|
2,000,000 |
|
|
11/16/2022 |
|
|
0.672 |
|
|
|
21 |
% |
|
11/15/2032 |
|
11/16/2032 |
Daniel Spiegelman |
|
|
150,000 |
|
|
11/16/2022 |
|
Nil |
|
|
|
0 |
% |
|
11/15/2032 |
|
11/16/2032 |
|
Susan Orr |
|
|
1,000,000 |
|
|
04/21/2022 |
|
|
0.948 |
|
|
|
50 |
% |
|
04/21/2025 |
|
04/20/2026 |
Quinton Oswald |
|
|
1,000,000 |
|
|
04/21/2022 |
|
|
0.948 |
|
|
|
50 |
% |
|
04/21/2025 |
|
04/20/2026 |
Anshul Thakral |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
Senior Management |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
Megan Baldwin, Ph.D. |
|
|
3,000,000 |
|
|
11/16/2022 |
|
|
0.658 |
|
|
|
21 |
% |
|
11/29/2019 |
|
11/29/2022 |
Megan Baldwin, Ph.D. |
|
|
1,600,000 |
|
|
10/19/2021 |
|
Nil |
|
|
|
11 |
% |
|
09/30/2024 |
|
10/18/2031 |
|
Megan Baldwin, Ph.D. |
|
|
500,000 |
|
|
11/16/2022 |
|
Nil |
|
|
|
0 |
% |
|
11/15/2032 |
|
11/16/2032 |
|
Tim Morris |
|
|
300,000 |
|
|
10/24/2022 |
|
|
4.850 |
|
|
|
0 |
% |
|
10/23/2032 |
|
10/24/2032 |
Karen Adams |
|
|
800,000 |
|
|
06/06/2022 |
|
|
1.460 |
|
|
|
50 |
% |
|
06/06/2025 |
|
06/05/2032 |
Karen Adams |
|
|
150,000 |
|
|
11/16/2022 |
|
Nil |
|
|
|
0 |
% |
|
11/15/2032 |
|
11/16/2032 |
|
Judith Robertson |
|
|
2,000,000 |
|
|
10/19/2021 |
|
|
0.948 |
|
|
|
50 |
% |
|
10/19/2024 |
|
10/18/2025 |
Joel Noar |
|
|
300,000 |
|
|
03/01/2022 |
|
|
6.090 |
|
|
|
25 |
% |
|
02/28/2026 |
|
03/01/2032 |
.
6C. Board Practices
Board of Directors
Our board of directors currently consists of eight members, including Dr. Baldwin, our Chief Executive Officer and Managing Director. Directors are elected at our annual general meeting of shareholders. Under our Constitution, at the close of each annual general meeting one-third of the directors, other than the Managing Director, or if their number is not a multiple of three, then the number nearest to but not more than one-third of the directors must retire. In addition, a director, other than the managing director, must retire from office at the conclusion of the third annual general meeting of shareholders after the director was last elected, even if his or her retirement results in more than one-third of all directors retiring from office. A retiring director remains in office until the end of such shareholder meeting and will be eligible for re-election at that meeting.
The membership of our board of directors is directed by the following requirements as set forth in our Constitution and our Board Charter, as applicable:
147
Our board of directors has delegated responsibility for the management of our businesses to the Chief Executive Officer and Managing Director but remains responsible for overseeing the performance of management. The principal roles and responsibilities of our board of directors include the following:
Our board of directors has established delegated limits of authority, which define the matters that are delegated to management and those that require board of director approval. Under the Corporations Act, at least two of our directors must be resident Australians. None of our non-employee directors have any service contracts with us that provide for benefits upon termination of employment. Under our Board Charter, the board of directors is required to meet at least six times per year.
Board Committees
To assist with the effective discharge of its duties, the board of directors has established an Audit and Risk Committee, a Remuneration Committee and a Nomination Committee. Each committee operates under a charter approved by our board of directors, which sets forth the purposes and responsibilities of the committees as well as qualifications for committee membership, committee structure and operations and committee reporting to the board of directors.
Audit and Risk Committee
The members of our Audit and Risk Committee are Messrs. Spiegelman, Oswald and Dr Orr. Our board of directors has determined that each of Messrs. Spiegelman and Oswald and Dr Orr satisfies the independence requirements under Nasdaq listing standards and Rule 10A-3(b)(1) under the Exchange Act. The chairperson of our Audit and Risk Committee is Mr. Spiegelman. Our board of directors has determined that Mr. Spiegelman is an “audit committee financial expert” within the meaning of SEC regulations. Each member of our Audit and Risk Committee can read and understand fundamental financial statements in accordance with applicable requirements. In arriving at these determinations, our board of directors has examined each member’s scope of experience and the nature of his or her employment.
The charter for our Audit and Risk Committee requires the committee to consist of at least three directors, each of whom must be non-employee directors and a majority of which must be independent directors. The chairperson of our Audit and Risk Committee must be an independent director and cannot be the chairperson of our board of directors. The Audit and Risk Committee is required to hold at least one regular meeting per fiscal quarter and must review its charter at least annually.
148
The role of the Audit and Risk Committee is to advise our board of directors in discharging responsibilities of our board of directors with respect to our financial reporting including accounting standards, internal control integrity and compliance, external audit activities including auditor appointment, independence, terms of engagement and fees and business risk management. Specific responsibilities of our Audit and Risk Committee include:
Remuneration Committee
The members of our Remuneration Committee are Ms. Haller and Messrs. Gozlan and Oswald. The chairperson of our Remuneration Committee is Mr. Oswald. The objectives of the Remuneration Committee are to link remuneration to the creation of shareholder value, to offer competitive and appropriate remuneration for the business performance delivered and to put into place a remuneration framework that reflects the responsibilities of senior management while being sufficiently competitive to attract and retain high caliber performers. The charter of our Remuneration Committee requires the committee to consist of at least three directors, a majority of whom must be independent. The chairperson of our Remuneration Committee must be an independent director. The Remuneration Committee is required to hold at least one regular meeting each year. Specific responsibilities of our Remuneration Committee include:
149
Nomination Committee
The members of our Nomination Committee are Messrs. Gozlan, Oswald and Spiegelman. The chairperson of our Nomination Committee is Mr. Gozlan. The role of the Nomination Committee is to assist our board of directors by identifying, reviewing and evaluating individuals qualified to become members of our board of directors, reviewing and recommending the nomination of directors and assisting the board of directors with other related tasks. The charter of our Nomination Committee requires the committee to consist of at least three directors, the majority of whom must meet the independence recommendations of the ASX Corporate Governance Council (as well as all applicable laws and regulations) and each of whom must be free of any relationship that, in the opinion of the board of directors, would interfere with his or her exercise of independent judgment. The members of the Nomination Committee will be appointed annually by the board of directors. The Nomination Committee is required to hold at least one regular meeting each year. Specific responsibilities of our Nomination Committee include:
Code of Conduct
We have adopted a Code of Conduct applicable to all of our directors, officers and employees. Our Code of Conduct is available on our website at www.opthea.com. We post on our website all disclosures that are required by law or the listing standards of Nasdaq concerning any amendments to, or waivers from, any provision of the Code of Conduct. The reference to our website address does not constitute incorporation by reference of the information contained at or available through our website, and you should not consider it to be a part of, this annual report.
6D. Employees
As of June 30, 2023, we had twenty-four full-time employees, eight of whom had an M.D. or Ph.D. degree. None of our employees are represented by collective bargaining agreements. We believe that our management maintains good relations with our employees. As of June 30, 2023, our employees were equally based in Australia (8) and the United States (16), with seventeen (17) employees in our research and development and commercialization department and seven employees in our general and administrative department.
For information regarding the share ownership of our directors and executive officers, see “Item 6B—Compensation” and “Item 7A—Major Shareholders.”
150
6F. Disclosure of a Registrant's Action to Recover Erroneously Awarded Compensation
Not applicable
The following table sets forth information with respect to the beneficial ownership of our ordinary shares as of September 28, 2023, for:
To our knowledge, as of the date of this annual report, 92,910,105 ordinary shares were held by one record holder in the United States, representing approximately 14.02% of our total outstanding shares. The record holder is The Bank of New York Mellon, the depositary of our ADS program. The number of beneficial owners of our ADSs in the United States is likely to be much larger than the number of record holders of our ordinary shares in the United States.
Beneficial ownership is determined in accordance with the rules and regulations of the SEC. In computing the number of ordinary shares beneficially owned by a person and the percentage ownership of that person, we have included ordinary shares that the person has the right to acquire within 60 days of September 28, 2023, including through the exercise of any option, warrant or other right or the conversion of any other security. These ordinary shares, however, are not included in the computation of the percentage ownership of any other person.
The calculations of the percentage of beneficial ownership are based on 662,806,891 ordinary shares (including ordinary shares in the form of ADSs) issued and outstanding as of September 28, 2023.
Unless otherwise indicated, the address of each beneficial owner listed below is c/o Opthea Limited, Suite 0403, Level 4, 650 Chapel Street, South Yarra, VIC 3141, Australia.
|
|
Shares Beneficially Owned |
|
|||||
Name of Beneficial Owner |
|
Number |
|
|
Percent |
|
||
Principal Shareholders |
|
|
|
|
|
|
||
Funds affiliated with Regal Funds Management Pty Ltd.(1) |
|
|
93,246,599 |
|
|
|
14.1 |
% |
Funds affiliated with Baker Bros. Advisors L.P.(2) |
|
|
31,627,844 |
|
|
|
4.8 |
|
Directors, Director Nominees and Senior Management |
|
|
|
|
|
|
||
Megan Baldwin, Ph.D.(3) |
|
|
4,945,142 |
|
|
* |
|
|
Karen Adams(4) |
|
|
400,000 |
|
|
|
— |
|
Julia Haller(5) |
|
|
1,000,000 |
|
|
|
— |
|
Lawrence Gozlan(6) |
|
|
3,791,591 |
|
|
* |
|
|
Jeremy Levin, DPhil, MB BChir(7) |
|
|
2,281,496 |
|
|
* |
|
|
Timothy Morris |
|
|
— |
|
|
|
— |
|
Susan Orr (8) |
|
|
500,000 |
|
|
|
— |
|
Quinton Oswald (9) |
|
|
500,000 |
|
|
|
— |
|
Judith Robertson(10) |
|
|
1,000,000 |
|
|
|
— |
|
Daniel Spiegelman(11) |
|
|
1,914,234 |
|
|
|
|
* |
Anshul Thakral |
|
|
— |
|
|
|
— |
|
All directors and senior management as a group (eleven persons)(14) |
|
|
16,332,463 |
|
|
|
3.5 |
% |
* Represents beneficial ownership of less than 1%.
- Represents unexercised options
151
Mr. Anshul Thakral should receive options upon Shareholder approval at the 2023 AGM and Mr. Tim Morris has no exercisable options until post one full year of employment.
152
With the appointment of Mr. Anshul Thakral as a non-executive director of Opthea, who is the CEO of Launch Tx and Operations Executive of Carlyle on June 7, 2023, the following are considered related parties of Opthea, Launch Tx, Ocelot and Carlyle.
Trading transactions
During the year, the Company entered into the following transactions with related parties
|
|
Consolidated |
|
|||||||||
|
|
Purchase of Service |
|
|||||||||
|
|
|
2023 |
|
|
|
2022 |
|
|
|
2021 |
|
Launch Tx - Ocelot |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Launch |
|
|
900,000 |
|
|
|
— |
|
|
|
— |
|
Purchase of services assisting Opthea with the management and oversight of trials under the Service Agreement with Launch Tx which was entered into in Mach 2023.
|
|
Consolidated |
|
|||||||||
|
|
Amounts owed to related parties |
|
|||||||||
|
|
|
2023 |
|
|
|
2022 |
|
|
|
2021 |
|
Launch Tx - Ocelot |
|
|
85,660,000 |
|
|
|
— |
|
|
|
— |
|
Launch |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Amounts owed to Ocelot relate to the Development Funding agreement and carry an effective rate of 23.82%.
On August 28,2023 Mr. Lawrence Gozlan, a director of the Company, and the Company have entered into a Consultancy Agreement of up to US$300,000 in respect of the provision of services associated with managing, overseeing and coordinating the conduct and implementation of the Capital raising. The consultancy agreement is effective for the financial year June 30, 2024. In the opinion of the Directors, these duties are outside the scope of the ordinary duties of a Director.
Other than compensation arrangements which are described under “Item 6B—Compensation” or as disclosed below, from July 1, 2022 through the date of this annual report, we did not enter into any transactions or loans with any: (i) enterprises that directly or indirectly, through one or more intermediaries, control, are controlled by or are under common control with us; (ii) associates; (iii) individuals owning, directly or indirectly, an interest in our voting power that gives them significant influence over us, and close members of any such individual’s family; (iv) key management personnel and close members of such individuals’ families; or (v) enterprises in which a substantial interest in our voting power is owned, directly or indirectly, by any person described in (iii) or (iv) or over which such person is able to exercise significant influence.
Director and Senior Management Compensation
See “Item 6B—Compensation” for information regarding compensation of our senior management and directors.
Indemnification Agreements
Our Constitution provides that, except to the extent prohibited by law including under the Corporations Act and, to the extent that an officer is not otherwise indemnified by us pursuant to an indemnity, we will indemnify every person who is or has been an officer of the company against any liability (other than legal costs that are unreasonable) incurred by that person as an officer. This includes any liability incurred by that person in their capacity as an officer of our subsidiaries where we requested that person to accept that appointment.
153
We have entered into Deeds of Indemnity, Insurance and Access, or Indemnity Deeds, with Megan Baldwin, Ph.D., Timothy Morris, Karen Adams, Judith Robertson, Lawrence Gozlan, Julia Haller, Jeremy Levin, Susan Orr, Quinton Oswald, Anshul Thakral and Daniel Spiegelman, each a non-employee director, or executive officer. Under the Indemnity Deeds, we have agreed to indemnify (to the maximum extent permitted under Australian law and our Constitution, subject to certain specified exceptions) each director and executive officer against all liabilities incurred in their capacity as our or our subsidiaries’ director or officer and any and all costs and expenses relating to such a claim or to any notified event incurred by such director or executive officer, including costs and expenses reasonably and necessarily incurred to mitigate any liability for such a claim or any claim which may arise from such a notified event. The Indemnity Deeds provide that the indemnities are unlimited as to amount, continuous and irrevocable.
Separately, we have obtained insurance for our directors and executive officers, as required by the Indemnity Deeds.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers or persons controlling us pursuant to the foregoing provisions, we have been informed that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
Related Person Transaction Policy
We comply with Australian law and the rules and regulations of the ASX regarding approval of transactions with related parties. We have also adopted a related person transaction policy that sets forth our procedures for the identification, review, consideration and approval or ratification of related person transactions, which became effective in connection with the listing of our ADSs on Nasdaq. For purposes of our policy, a related person transaction is a transaction, arrangement or similar contractual relationship, or any series of similar transactions, arrangements or relationships, in which we and any related person are, were or will be participants and the amount involved in the transaction exceeds US$120,000, with the exception of usual transactions concluded under normal conditions. A related person is any member of our board of directors, our senior management or any beneficial owner of more than 5% of any class of our ordinary shares, including any of their immediate family members and any entity owned or controlled by such persons.
Under the policy, if a transaction has been identified as a related person transaction, including any transaction that was not a related person transaction when originally consummated or any transaction that was not initially identified as a related person transaction prior to consummation, our senior management must present information regarding the related person transaction to the board of directors for review, consideration and approval or ratification. The presentation must include a description of, among other things, the material facts, the interests, direct and indirect, of the related persons, the benefits to us of the transaction and whether the transaction is on terms that are comparable to the terms available to or from, as the case may be, an unrelated third party or to or from employees generally. Under the policy, we will collect information that we deem reasonably necessary from each member of our board of directors and, to the extent feasible, significant shareholder to enable us to identify any existing or potential related-person transactions and to effectuate the terms of the policy.
All of the transactions described above were entered into prior to the adoption of the written policy, but our board of directors evaluated and approved all transactions that were considered to be related party transactions under Australian law and the rules and regulations of the ASX at the time at which they were consummated.
7C. Interests of Experts and Counsel
Not applicable.
154
Item 8. Financial Information
8A. Consolidated Statements and Other Financial Information
Consolidated Financial Statements
See “Item 18—Financial Statements.”
Legal Proceedings
See “Item 4B—Business Overview—Legal Proceedings.”
Dividend Policy
We have not declared or paid any dividends on our ordinary shares since February 2005. We intend to retain any earnings for use in our business and do not currently intend to pay cash dividends on our ordinary shares. Dividends, if any, on our outstanding ordinary shares will be declared by and subject to the discretion of our board of directors, and subject to Australian law.
Any dividend we declare will be paid to the holders of ADSs, subject to the terms of the deposit agreement, to the same extent as holders of our ordinary shares, to the extent permitted by applicable law and regulations, less the fees and expenses payable under the deposit agreement. Any dividend we declare will be distributed by the depositary bank to the holders of the ADSs, subject to the terms of the deposit agreement.
8B. Significant Changes
No significant change, other than as otherwise described in this annual report, has occurred in our operations since the date of our financial statements included in this annual report.
Item 9. The Offer and Listing
9A. Offer and Listing Details
Our ADSs have been listed on the Nasdaq Global Select Market under the symbol “OPT” since October 16, 2020. Our ordinary shares are listed on the ASX under the symbol “OPT.”
9B. Plan of Distribution
Not applicable.
9C. Markets
See “—Offer and Listing Details.”
Not applicable.
9E. Dilution
Not applicable.
9F. Expenses of the Issue
Not applicable.
155
Item 10. Additional Information
Not applicable.
10B. Constitution
See Exhibit 2.3 “Description of Securities,” which is incorporated herein by reference.
10C. Material Contracts
For information on our material contracts, see “Item 4B—Business Overview—Our Commercial License Arrangement with Selexis SA,” “Item 5B—Liquidity and Capital Resources—Sources and Uses of Liquidity,” and “Item 5B–Liquidity and Capital Resources–Our Development Funding Agreement with Carlyle and Abingworth.”
10D. Exchange Controls
The Australian dollar is freely convertible into U.S. dollars. In addition, there are currently no specific rules or limitations regarding the export from Australia of profits, dividends, capital or similar funds belonging to foreign investors, except that certain payments to non-residents must be reported to the Australian Transaction Reports and Analysis Centre (“or AUSTRAC”), which monitors such transaction, and amounts on account of potential Australian tax liabilities may be required to be withheld unless a relevant taxation treaty can be shown to apply.
10E. Taxation
The following summary of the material Australian and U.S. federal income tax considerations relating to an investment in the ADSs or ordinary shares is based upon laws and relevant interpretations thereof in effect as of the date of this annual report, all of which are subject to change, possibly with retroactive effect. This summary does not deal with all possible tax consequences relating to an investment in the ADSs or ordinary shares, such as the tax consequences under U.S. state, local and other tax laws other than U.S. federal income tax laws and certain Australian tax laws.
Material United States Federal Income Tax Considerations
The following discussion is a summary of U.S. federal income tax considerations generally applicable to the ownership and disposition of the ADSs or ordinary shares by a U.S. holder (as defined below). This summary applies only to U.S. holders that hold such ADSs or ordinary shares as capital assets (generally, property held for investment) for U.S. federal income tax purposes. This summary does not address all U.S. federal income tax considerations that may be relevant to a particular U.S. holder and does not represent a detailed discussion of all of the U.S. federal income tax considerations applicable to a holder of our ordinary shares or ADSs that may be subject to special tax rules including, without limitation:
156
Holders of the ADSs or ordinary shares who fall within one of the categories above are advised to consult their tax advisor regarding the specific tax consequences which may apply to their particular situation.
If a partnership (or any other entity or arrangement treated as a partnership for U.S. federal income tax purposes) holds the ADSs or ordinary shares, the tax consequences relating to an investment in the ADSs or ordinary shares will depend in part upon the status of the partner and the activities of the partnership. Such a partner or partnership should consult its tax advisor regarding the U.S. federal income tax considerations of owning and disposing of the ADSs or ordinary shares in its particular circumstances.
The discussion in this section is based on the U.S. Internal Revenue Code of 1986, as amended, or the Code, existing, proposed and temporary U.S. Treasury Regulations promulgated thereunder, administrative and judicial interpretations thereof, and the Convention between the Government of the United States of America and the Government of Australia for the Avoidance of Double Taxation and the Prevention of Fiscal Evasion with Respect to Taxes on Income, signed on August 6, 1982, as amended and currently in force, or the Treaty, in each case as in effect and available on the date hereof. Such authorities are subject to change, which change could apply retroactively, and to differing interpretations, all of which could affect the tax considerations described below. There can be no assurances that the U.S. Internal Revenue Service, or the IRS, will not take a position concerning the tax consequences of the ownership and disposition of ADSs or ordinary shares or that such a position would not be sustained by a court. U.S. holders should consult their own tax advisors concerning the U.S. federal, state, local and non-U.S. tax consequences of owning and disposing of ADSs or ordinary shares in their particular circumstances.
This summary does not address the estate tax considerations, alternative minimum tax considerations, the potential application of the Medicare contribution tax on net investment income, the special tax accounting rules under Section 451(b) of the Code, or any U.S. state, local, or non-U.S. tax considerations applicable to the acquisition, ownership and disposition of ordinary shares or ADSs or ordinary shares.
For the purposes of this description, a “U.S. holder” is a beneficial owner of the ADSs or ordinary shares that is (or is treated as), for U.S. federal income tax purposes:
The discussion in this section is based in part upon the representations of the depositary and the assumption that each obligation in the amended and restated deposit agreement and any related agreement will be performed in accordance with its terms.
157
In general, and taking into account the earlier assumptions, for U.S. federal income tax purposes, a U.S. holder holding ADSs will be treated as the owner of the ordinary shares represented by the ADSs. Exchanges of ordinary shares for ADSs, and ADSs for ordinary shares, generally will not be subject to U.S. federal income tax.
ALL HOLDERS AND POTENTIAL HOLDERS OF THE ADSs SHOULD CONSULT THEIR OWN TAX ADVISORS AS TO THE PARTICULAR TAX CONSEQUENCES APPLICABLE TO THEM RELATING TO THE ACQUISITION, OWNERSHIP AND DISPOSITION OF THE ADSs OR ORDINARY SHARES, INCLUDING THE APPLICABILITY OF U.S. FEDERAL, STATE AND LOCAL TAX LAWS, AUSTRALIAN TAX LAWS AND OTHER NON-U.S. TAX LAWS.
Passive Foreign Investment Company Considerations.
If we are classified as a PFIC in any taxable year, during which a U.S. holder holds the ADSs or ordinary shares, such U.S. holder will be subject to special rules generally intended to reduce or eliminate any benefits from the deferral of U.S. federal income tax that a U.S. holder could derive from investing in a non-U.S. company that does not distribute all of its earnings on a current basis.
We will be classified as a PFIC for U.S. federal income tax purposes in any taxable year in which, after applying certain look-through rules with respect to the income and assets of our subsidiaries, either: (1) at least 75% of the gross income is “passive income” or (2) at least 50% of the average quarterly value of our total gross assets (which would generally be measured by fair market value of our assets, and for which purpose the total value of our assets may be determined in part by the market value of the ADSs and our ordinary shares, which are subject to change) is attributable to assets that produce “passive income” or are held for the production of “passive income.”
Passive income for this purpose generally includes dividends, interest, royalties, rents, gains from commodities and securities transactions, the excess of gains over losses from the disposition of assets which produce passive income, and includes amounts derived by reason of the temporary investment of funds raised in offerings of our securities. If a non-U.S. corporation owns directly or indirectly at least 25% by value of the stock of another corporation or the partnership interests in a partnership, the non-U.S. corporation is treated for purposes of the PFIC tests as owning its proportionate share of the assets of the other corporation or partnership and as receiving directly its proportionate share of the other corporation’s or partnership’s income.
There can be no assurance that we will not be considered a PFIC in any past, current or future taxable year, and based on the nature and composition of our income, assets, activities and market capitalization, it is likely that we will be a PFIC for our taxable year ended June 30, 2023 and in future taxable years. However, our PFIC status is based on an annual determination and may change from year to year. Our status as a PFIC will depend on the composition of our income and the composition and value of our assets, which may be determined in large part by reference to the market value of the ADSs and our ordinary shares, which may be volatile, from time to time. Our status may also depend, in part, on how quickly we utilize the cash we raise in any offering of our securities. Our U.S. counsel expresses no opinion regarding our conclusions or our expectations regarding our PFIC status.
If we are classified as a PFIC in any year with respect to which a U.S. holder owns the ADSs or ordinary shares, we will continue to be treated as a PFIC with respect to such U.S. holder in all succeeding years during which the U.S. holder owns the ADSs or ordinary shares, regardless of whether we continue to meet the tests described above unless we cease to be a PFIC and the U.S. holder has made a “deemed sale” election under the PFIC rules. If the “deemed sale” election is made, a U.S. holder will be deemed to have sold the securities the U.S. holder holds at their fair market value as of the date of such deemed sale and any gain from such deemed sale would be subject to the rules described below. After the deemed sale election, so long as we do not become a PFIC in a subsequent taxable year, the U.S. holder’s securities with respect to which such election was made will not be treated as shares in a PFIC and the U.S. holder will not be subject to the rules described below with respect to any “excess distribution” the U.S. holder receives from us or any gain from an actual sale or other disposition of the securities. U.S. holders should consult their tax advisors as to the possibility and consequences of making a deemed sale or other “purging” election if such election becomes available.
If we are a PFIC, and you are a U.S. holder that does not make one of the elections described herein, a special tax regime will apply to both (a) any “excess distribution” by us to you (generally, your ratable portion of distributions in any year, other than the taxable year in which your holding period in the shares or ADSs begins, which are greater than 125% of the average annual distribution received by you in the shorter of the three preceding years or the portion of your holding period for the ADSs or ordinary shares that preceded the year of
158
the distribution) and (b) any gain realized on the sale or other disposition of the ADSs or ordinary shares. Under this regime, any excess distribution and realized gain will be treated as ordinary income and will be subject to tax as if (a) the excess distribution or gain had been realized ratably over your holding period, (b) the amount deemed realized in each year had been subject to tax in each year of that holding period at the highest marginal rate for such year (other than income allocated to the current period or any taxable period before we became a PFIC, which would be subject to tax at the U.S. holder’s regular ordinary income rate for the current year and would not be subject to the interest charge discussed below) and (c) the interest charge generally applicable to underpayments of tax had been imposed on the taxes deemed to have been payable in those years. In addition, dividend distributions made to you will not qualify for the lower rates of taxation applicable to qualified dividends discussed above under “Distributions.”
Certain elections may alleviate some of the adverse consequences of PFIC status and would result in an alternative treatment of our ordinary shares or ADSs.
If a U.S. holder makes a mark-to-market election, with respect to our ordinary shares or ADSs, the U.S. holder generally will recognize as ordinary income any excess of the fair market value of our ordinary shares or ADSs at the end of each taxable year over their adjusted tax basis, and will recognize an ordinary loss in respect of any excess of the adjusted tax basis of our ordinary shares or ADSs over their fair market value at the end of the taxable year (but only to the extent of the net amount of income previously included as a result of the mark-to-market election). If a U.S. holder makes the election, the U.S. holder’s tax basis in our ordinary shares or ADSs will be adjusted to reflect these income or loss amounts. Any gain recognized on the sale or other disposition of our ordinary shares or ADSs in a year in which we are a PFIC will be treated as ordinary income and any loss will be treated as an ordinary loss (but only to the extent of the net amount of income previously included as a result of the mark-to-market election). The mark-to-market election is available only if we are a PFIC and our ordinary shares or ADSs are “regularly traded” on a “qualified exchange.” Our ordinary shares or ADSs will be treated as “regularly traded” in any calendar year in which more than a de minimis quantity of our ordinary shares or ADSs are traded on a qualified exchange on at least 15 days during each calendar quarter (subject to the rule that trades that have as one of their principal purposes the meeting of the trading requirement are disregarded). Nasdaq is a qualified exchange for this purpose and, consequently, if the ADSs are regularly traded, the mark-to-market election will be available to a U.S. holder. It should be noted that only the ADSs and not our ordinary shares are listed on Nasdaq. Consequently, our ordinary shares may not be marketable if the ASX (where our ordinary shares are currently listed) does not meet the applicable requirements. U.S. holders should consult their tax advisors regarding the availability of the mark-to-market election for ordinary shares that are not represented by ADSs.
However, a mark-to-market election generally cannot be made for equity interests in any lower-tier PFICs that we own, unless shares of such lower-tier PFIC are themselves “marketable.” As a result, even if a U.S. holder validly makes a mark-to-market election with respect to our ordinary shares or ADSs, the U.S. holder may continue to be subject to the PFIC rules (described above) with respect to its indirect interest in any of our investments that are treated as an equity interest in a PFIC for U.S. federal income tax purposes. U.S. holders should consult their tax advisors as to the availability and desirability of a mark-to-market election, as well as the impact of such election on interests in any lower-tier PFICs.
We do not currently intend to provide the information necessary for U.S. holders to make qualified electing fund elections if we were treated as a PFIC for any taxable year. U.S. holders should consult their tax advisors to determine whether any of the other elections described above would be available and if so, what the consequences of the alternative treatments would be in their particular circumstances.
If we are determined to be a PFIC, the general tax treatment for U.S. holders described in this section would apply to indirect distributions and gains deemed to be realized by U.S. holders in respect of any of our subsidiaries that also may be determined to be PFICs. U.S. holders should consult their tax advisors regarding the application of the PFIC rules to our subsidiaries.
If a U.S. holder owns ordinary shares or ADSs during any taxable year in which we are a PFIC, the U.S. holder may be required to file an IRS Form 8621 (Information Return by a Shareholder of a Passive Foreign Investment Company or Qualified Electing Fund) with respect to the company, generally with the U.S. holder’s federal income tax return for that year. You should consult your tax advisor concerning any filing requirements arising from the PFIC rules.
159
The U.S. federal income tax rules relating to PFICs are complex. Prospective U.S. investors are urged to consult their own tax advisors with respect to the acquisition, ownership and disposition of our ordinary shares or ADSs, the consequences to them of an investment in a PFIC, any elections available with respect to ordinary shares and ADSs and the IRS information reporting obligations with respect to the acquisition, ownership and disposition of ordinary shares and ADSs.
Distributions
We do not expect to make any distributions in respect of the ADSs or ordinary shares. Subject to the discussion under “—Passive Foreign Investment Company Considerations” above, the gross amount of any distribution (including any amounts withheld in respect of foreign tax) actually or constructively received by a U.S. holder with respect to the ADSs or ordinary shares will generally be taxable to the U.S. holder as a dividend to the extent of the U.S. holder’s pro rata share of our current or accumulated earnings and profits as determined under U.S. federal income tax principles. Distributions in excess of earnings and profits will generally be non-taxable to the U.S. holder to the extent of, and will be applied against and reduce (but not below zero), the U.S. holder’s adjusted tax basis in the ADSs or ordinary shares. Distributions in excess of earnings and profits and such adjusted tax basis will generally be taxable to the U.S. holder as either long-term or short-term capital gain depending upon whether the U.S. holder has held the ADSs or ordinary shares for more than one year as of the time such distribution is received. However, since we do not calculate our earnings and profits under U.S. federal income tax principles, it is expected that any distribution will be reported as a dividend, even if that distribution would otherwise be treated as a non-taxable return of capital or as capital gain under the rules described above.
Non-corporate U.S. holders may qualify for the preferential rates of taxation with respect to dividends on the ADSs or ordinary shares applicable to long-term capital gains (i.e., gains from the sale of capital assets held for more than one year) and “qualified dividend income” (as discussed below) if we are a “qualified foreign corporation” and certain other requirements (discussed below) are met. A non-U.S. corporation generally will be considered to be a qualified foreign corporation (a) if it is eligible for the benefits of a comprehensive tax treaty with the United States which the Secretary of Treasury of the United States determines is satisfactory for purposes of this provision and which includes an exchange of information provision, or (b) with respect to any dividend it pays on ADSs or ordinary shares which are readily tradable on an established securities market in the United States. The ADSs are listed on Nasdaq, which is an established securities market in the United States, and we expect the ADSs to be readily tradable on Nasdaq. However, there can be no assurance that the ADSs will be considered readily tradable on an established securities market in the United States in later years. The Company, which is incorporated under the laws of Australia, believes that it qualifies as a resident of Australia for purposes of, and is eligible for the benefits of, the Treaty, although there can be no assurance in this regard. Further, the IRS has determined that the Treaty is satisfactory for purposes of the qualified dividend rules and that it includes an exchange-of-information program. However, the preferential tax rates available for qualified dividend income do not apply if we are a PFIC in the taxable year in which such dividends are paid or in the preceding taxable year. Therefore, in light of the discussion under “—Passive Foreign Investment Company Considerations” above, you should assume that dividends generally will not constitute qualified dividend income eligible for reduced rates of taxation. In addition, the dividends will not be eligible for the dividends-received deduction generally allowed to corporate U.S. holders.
A U.S. holder generally may claim the amount of any Australian withholding tax as either a deduction from gross income or a credit against its U.S. federal income tax liability. The foreign tax credit is subject to numerous complex limitations that must be determined and applied on an individual basis. In addition, the creditability of foreign taxes could be affected by actions taken by intermediaries in the chain of ownership between the holders of the ADSs and our company if, as a result of such actions, the holders of the ADSs are not properly treated as beneficial owners of the underlying ordinary shares. Each U.S. holder should consult its own tax advisors regarding the foreign tax credit rules.
In general, the amount of a distribution paid to a U.S. holder in a foreign currency will be the U.S. dollar value of the foreign currency calculated by reference to the spot exchange rate on the day the depositary receives the distribution, in the case of the ADSs, or on the day the distribution is received by the U.S. holder, in the case of ordinary shares, regardless of whether the foreign currency is converted into U.S. dollars at that time. Any foreign currency gain or loss a U.S. holder realizes on a subsequent conversion of foreign currency into U.S. dollars will be U.S. source ordinary income or loss. If dividends received in a foreign currency are converted into U.S. dollars on the day they are received, a U.S. holder should not be required to recognize foreign currency gain or loss in respect of the dividend.
160
Sale, Exchange or Other Taxable Disposition
A U.S. holder will generally recognize gain or loss for U.S. federal income tax purposes upon the sale, exchange or other taxable disposition of the ADSs or ordinary shares in an amount equal to the difference between the U.S. dollar value of the amount realized from such sale or exchange and the U.S. holder’s adjusted tax basis in those securities, determined in U.S. dollars. Subject to the discussion under “—Passive Foreign Investment Company Considerations” above, this gain or loss will generally be a capital gain or loss. The adjusted tax basis in the ADSs or ordinary shares generally will be equal to the cost of such ADSs or ordinary shares. Capital gain from the sale, exchange or other taxable disposition of the ADSs or ordinary shares by a non-corporate U.S. holder is generally eligible for a preferential rate of taxation applicable to capital gains if the non-corporate U.S. holder’s holding period determined at the time of such sale, exchange or other taxable disposition for such securities exceeds one year (i.e., such gain is long-term taxable gain). The deductibility of capital losses for U.S. federal income tax purposes is subject to limitations. Any such gain or loss that a U.S. holder recognizes generally will be treated as U.S. source gain or loss for foreign tax credit limitation purposes.
For a cash basis taxpayer, units of foreign currency paid or received are translated into U.S. dollars at the spot rate on the settlement date of the purchase or sale. In that case, no foreign currency exchange gain or loss will result from currency fluctuations between the trade date and the settlement date of such a purchase or sale.
An accrual basis taxpayer, however, may elect the same treatment required of cash basis taxpayers with respect to purchases and sales of our ordinary shares or ADSs that are traded on an established securities market, provided the election is applied consistently from year to year. Such election may not be changed without the consent of the IRS. For an accrual basis taxpayer who does not make such election, units of foreign currency paid or received are translated into U.S. dollars at the spot rate on the trade date of the purchase or sale. Such an accrual basis taxpayer may recognize exchange gain or loss based on currency fluctuations between the trade date and the settlement date. Any foreign currency gain or loss a U.S. holder realizes will be U.S. source ordinary income or loss.
Backup Withholding and Information Reporting.
U.S. holders generally will be subject to information reporting requirements with respect to dividends on the ordinary shares or ADSs and on the proceeds from the sale, exchange, or disposition of the ordinary shares or ADSs that are paid within the United States or through U.S.-related financial intermediaries, unless the U.S. holder is an “exempt recipient.” In addition, U.S. holders may be subject to backup withholding on such payments, unless the U.S. holder provides a taxpayer identification number and a duly executed IRS Form W-9 or otherwise establishes an exemption. Backup withholding is not an additional tax, and the amount of any backup withholding will be allowed as a credit against a U.S. holder’s U.S. federal income tax liability and may entitle such holder to a refund, provided that the required information is timely furnished to the IRS.
Foreign Asset Reporting.
Certain individual U.S. holders are required to report information relating to an interest in the ordinary shares or ADSs, subject to certain exceptions (including an exception for shares held in accounts maintained by U.S. financial institutions) by filing IRS Form 8938 (Statement of Specified Foreign Financial Assets) with their federal income tax return. U.S. holders are urged to consult their tax advisors regarding their information reporting obligations, if any, with respect to their ownership and disposition of the ordinary shares or ADSs.
THE DISCUSSION ABOVE IS A SUMMARY OF THE U.S. FEDERAL INCOME TAX CONSEQUENCES OF AN INVESTMENT IN THE ADSs AND IS BASED UPON LAWS AND RELEVANT INTERPRETATIONS THEREOF IN EFFECT AS OF THE DATE OF THIS ANNUAL REPORT, ALL OF WHICH ARE SUBJECT TO CHANGE, POSSIBLY WITH RETROACTIVE EFFECT. EACH PROSPECTIVE INVESTOR IS URGED TO CONSULT ITS OWN TAX ADVISOR ABOUT THE TAX CONSEQUENCES TO IT OF AN INVESTMENT IN THE ADSs IN LIGHT OF THE INVESTOR’S OWN CIRCUMSTANCES.
161
Material Australian Tax Considerations
In this section, we discuss the material Australian income tax, stamp duty and goods and services tax considerations related to the acquisition, ownership and disposal by the absolute beneficial owners of the ADSs or ordinary shares represented by ADSs. It is based upon existing Australian tax law as of the date of this registration statement, which is subject to change, possibly retrospectively. This discussion does not address all aspects of Australian tax law which may be important to particular investors in light of their individual investment circumstances, such as ADSs or shares held by investors subject to special tax rules (for example, financial institutions, insurance companies or tax exempt organizations). In addition, this summary does not discuss any non-Australian or state tax considerations, other than stamp duty and goods and services tax.
Prospective investors are urged to consult their tax advisors regarding the Australian and non-Australian income and other tax considerations of the acquisition, ownership and disposition of the ADSs or shares. This summary is based upon the premise that the holder of an ADS is not an Australian tax resident and is not carrying on business in Australia through a permanent establishment (referred to as a “Non-Australian Holder” in this summary). This summary is also based on the assumption that a Non-Australian Holder is “absolutely entitled” to the ordinary shares represented by an ADS (see “—Nature of ADSs for Australian Taxation Purposes” below).
Nature of ADSs for Australian Taxation Purposes
Non-Australian Holders of ADSs should obtain specialist Australian tax advice regarding their rights and obligations under the deposit agreement with the depositary, including whether the deposit arrangement constitutes a “bare trust” resulting in the holders of an ADS being “absolutely entitled” to the underlying shares represented by the ADS for Australian taxation purposes. Apart from certain aspects of the Australian tax legislation (for example, the Australian capital gains tax and withholding tax provisions, which are discussed below), there is no express legislative basis for disregarding “bare trusts” for Australian tax purposes generally.
This summary proceeds on the assumption that the deposit arrangement constitutes a bare trust, which results in holders of ADSs being “absolutely entitled” to the underlying shares. On this basis, holders of ADSs can be treated as the owners of the underlying ordinary shares for Australian capital gains tax purposes. Dividends paid on the underlying ordinary shares will also be treated as dividends derived by the holders of ADSs as the persons presently entitled to those dividends.
Taxation of Dividends
Australia operates a dividend imputation system under which dividends may be declared to be “franked” to the extent they are paid out of company profits that have been subject to income tax. Fully franked dividends are not subject to dividend withholding tax. To the extent that they are unfranked, dividends payable to Non-Australian Holders will be subject to dividend withholding tax except to the extent they are declared to be “conduit foreign income”, or CFI. Dividend withholding tax will be imposed at 30%, unless a shareholder is a resident of a country with which Australia has a double taxation treaty and qualifies for the benefits of the treaty. Under the provisions of the current Double Taxation Convention between Australia and the United States, the Australian tax withheld on unfranked dividends that are not declared to be CFI paid by us to which a resident of the United States is beneficially entitled is limited to 15%.
Under the Double Taxation Convention between Australia and the United States, if a U.S. resident company that is a Non-Australian Holder directly owns a 10% or more voting interests, the Australian tax withheld on unfranked dividends that are not declared to be CFI paid by us to which the company is beneficially entitled is limited to 5%.
Character of ADSs or Shares for Australian Taxation Purposes
The Australian tax treatment of a sale or disposal of the ADSs or underlying shares will depend on whether they are held on revenue or capital account. ADSs may be held on revenue rather than capital account, for example, where they are held by share traders or any profit arises from a profit-making undertaking or scheme entered into by the holder. Non-Australian Holders of ADSs should obtain specialist Australian tax advice regarding the characterization of any gain or loss on a sale or disposal of the ADSs or underlying shares as revenue or capital in nature.
162
Tax on Sales or other Dispositions of Shares or ADSs—Capital Gains Tax
Non-Australian Holders who are treated as the owners of the underlying shares on the basis that they are absolutely entitled to those shares will not be subject to Australian capital gains tax on the gain made on a sale or other disposal of ordinary shares, provided the shares are not “taxable Australian property.” Taxable Australian property includes “indirect Australian real property interests,” which are interests in a company where:
Australian capital gains tax applies to net capital gains at a taxpayer’s marginal tax rates. Net capital gains are calculated after reduction for capital losses, which may only be offset against capital gains.
If a Non-Australian Holder of ADSs was not absolutely entitled to the underlying shares, and the ADSs were held on capital account, the same principles would apply in determining whether a gain on the sale or disposal of the ADSs would be subject to Australian capital gains tax. That is, a Non-Australian Holder should not be subject to Australian capital gains tax provided the ADSs are not taxable Australian property.
The 50% capital gains tax discount is not available to Non-Australian Holders on gains from assets acquired after May 8, 2012 where they were non-Australian residents during the entire holding period. Companies are not entitled to a capital gains tax discount.
Broadly, where there is a disposal of “taxable Australian property,” which includes indirect Australian real property interests, the purchaser will be required to withhold and remit to the Australian Taxation Office, or the ATO, 12.5% of the proceeds from the sale. A transaction is excluded from the withholding requirements in certain circumstances, including where the transaction is an on-market transaction conducted on an approved stock exchange, a securities lending arrangement, or the transaction is conducted using a broker operated crossing system. There may also be an exception to the requirement to withhold where a Non-Australian Holder provides a declaration that their ordinary shares are not “indirect Australian real property interests”. The Non-Australian Holder may be entitled to receive a tax credit for the tax withheld by the purchaser which they may claim in their Australian income tax return.
Tax on Sales or other Dispositions of ADSs—Revenue Account
Non-Australian Holders who hold their ADSs on revenue account may have the gains made on the sale or other disposal of the ADSs included in their assessable income under the ordinary income provisions of the income tax law, if the gains are sourced in Australia. In the case of gains which are ordinary income, there are no express provisions which treat holders of ADSs as the owners of the underlying shares where they are absolutely entitled to those shares under a bare trust.
Non-Australian Holders assessable under these ordinary income provisions in respect of gains made on ADSs held on revenue account would be assessed for such gains at the Australian tax rates for non-Australian residents, which start at a marginal rate of 32.5% for individuals and would be required to file an Australian tax return. Some relief from Australian income tax may be available to a Non-Australian Holder who is resident of a country with which Australia has a double taxation treaty, qualifies for the benefits of the treaty and does not, for example, derive the gain in carrying on business through a permanent establishment in Australia.
To the extent an amount would be included in a Non-Australian Holder’s assessable income under both the capital gains tax provisions and the ordinary income provisions, the capital gain amount may be reduced, so that the holder may not be subject to double Australian tax on any part of the gain.
The statements under “—Tax on Sales or Other Dispositions of Shares—Capital Gains Tax” regarding a purchaser being required to withhold 12.5% tax on the acquisition of certain taxable Australian property are also relevant where the disposal of the ADSs by a Non-Australian Holder is likely to generate gains on revenue account, rather than a capital gain.
163
Dual Residency
If a holder of ADSs is a resident of both Australia and the United States under those countries’ domestic taxation laws, that holder may be subject to tax as an Australian resident. If, however, the holder is determined to be a U.S. resident for the purposes of the Double Taxation Convention between the United States and Australia and qualifies for the benefit of that treaty, the Australian tax may be subject to limitation by the Double Taxation Convention. Holders should obtain specialist taxation advice in these circumstances.
Stamp Duty
No Australian stamp duty is payable by Australian residents or non-Australian residents on the issue, transfer and/or surrender of the ADSs or ordinary shares, provided that the securities issued, transferred and/or surrendered do not represent 90% or more of our issued shares.
Australian Death Duty
Australia does not have estate or death duties. As a general rule, no capital gains tax liability is realized upon the inheritance of a deceased person’s shares. The disposal of inherited shares by beneficiaries may, however, give rise to a capital gains tax liability if the gain falls within the scope of Australia’s jurisdiction to tax.
Goods and Services Tax
No Australian goods and services tax will be payable on the supply of the ADSs or ordinary shares.
THE DISCUSSION ABOVE IS A SUMMARY OF THE AUSTRALIAN TAX CONSEQUENCES OF AN INVESTMENT IN OUR ORDINARY SHARES OR ADSs AND IS BASED UPON LAWS AND RELEVANT INTERPRETATIONS THEREOF IN EFFECT AS OF THE DATE OF THIS ANNUAL REPORT, ALL OF WHICH ARE SUBJECT TO CHANGE, POSSIBLY WITH RETROACTIVE EFFECT. EACH PROSPECTIVE INVESTOR IS URGED TO CONSULT ITS OWN TAX ADVISOR ABOUT THE TAX CONSEQUENCES TO IT OF AN INVESTMENT IN OUR ORDINARY SHARES OR ADSs IN LIGHT OF THE INVESTOR’S OWN CIRCUMSTANCES.
10F. Dividends and Paying Agents
Not applicable.
10G. Statement by Experts
Not applicable.
10H. Documents on Display
We are subject to the information reporting requirements of the Exchange Act applicable to foreign private issuers and under those requirements will file reports with the SEC. Those reports may be inspected without charge at the locations described below. As a foreign private issuer, we are exempt from the rules under the Exchange Act related to the furnishing and content of proxy statements, and our officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. In addition, we are not required under the Exchange Act to file periodic reports and financial statements with the SEC as frequently or as promptly as United States companies whose securities are registered under the Exchange Act. Nevertheless, we will file with the SEC an Annual Report on Form 20-F containing financial statements that have been examined and reported on, with and opinion expressed by an independent registered public accounting firm.
The SEC maintains a website (www.sec.gov) that contains reports, proxy and information statements and other information regarding registrants, such as us, that file electronically with the SEC. With respect to references made in this annual report to any contract or other document of our company, such references are not necessarily complete and you should refer to the exhibits attached or incorporated by reference to this annual report for copies of the actual contract or document.
164
We maintain a corporate website at www.opthea.com. Information contained on, or that can be accessed through, our website does not constitute a part of this annual report and our website address is included herein as an inactive textual reference only.
10I. Subsidiary Information
Not required.
10J. Annual Report to Security Holders
Pursuant to Item 10J of Form 20-F, exhibit 15.2 to this annual report on Form 20-F includes our annual report to security holders. None of such annual report is incorporated by reference into this annual report on Form 20-F. Such annual report is not deemed to be filed as part of this annual report on Form 20-F.
Item 11. Quantitative and Qualitative Disclosures about Market Risk
For information about our exposure to market risk and how we manage this risk, see “Item 5E—Critical Accounting Estimates— Qualitative and Quantitative Disclosures about Market Risk.”
Item 12. Description of Securities Other than Equity Securities
12A. Debt Securities
Not applicable.
12B. Warrants and Rights
Not applicable.
12C. Other Securities
Not applicable.
The Bank of New York Mellon, as depositary, registers and delivers American Depositary Shares, or ADSs. Each ADS represents eight ordinary shares (or a right to receive eight ordinary shares) deposited with HSBC Bank Australia Limited, as custodian for the depositary in Australia. Each Ads also represents any other securities, cash or other property that may be held by the depositary. The deposited shares together with any other securities, cash or other property held by the depositary are referred to as the deposited securities. The depositary’s office at which the ADSs are administered, and its principal executive office are located at 240 Greenwich Street, New York, New York 10286.
You may hold ADSs either (A) directly (i) by having an ADR, which is a certificate evidencing a specific number of ADSs, registered in your name, or (ii) by having uncertificated ADSs registered in your name, or (B) indirectly by holding a security entitlement in ADSs through your broker or other financial institution that is a direct or indirect participant in The Depository Trust Company, or DTC. If you hold ADSs directly, you are a registered ADS holder, or an ADS holder. This description assumes you are an ADS holder. If you hold the ADSs indirectly, you must rely on the procedures of your broker or other financial institution to assert the rights of ADS holders described in this section. You should consult with your broker or financial institution to find out what those procedures are.
Registered holders of uncertificated ADSs receive statements from the depositary confirming their holdings.
165
As an ADS holder, you are not treated as one of our shareholders and you do not have shareholder rights. Australian law governs shareholder rights. The depositary is the holder of the shares underlying your ADSs. As a registered holder of ADSs, you have ADS holder rights. The amended and restated deposit agreement among us, the depositary, ADS holders and all other persons indirectly or beneficially holding ADSs sets out ADS holder rights as well as the rights and obligations of the depositary. New York law governs the deposit agreement and the ADSs.
Fees and Expenses
The following table shows the fees and charges that a holder of our ADSs may have to pay, either directly or indirectly. The majority of these costs are set by the depositary bank and are subject to change:
|
|
|
Persons depositing or withdrawing shares or ADS holders must pay: |
|
For: |
US$5.00 (or less) per 100 ADSs (or portion of 100 ADSs) |
|
Issuance of ADSs, including issuances resulting from a distribution of shares or rights or other property
Cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates |
US$0.05 (or less) per ADS |
|
Any cash distribution to ADS holders |
A fee equivalent to the fee that would be payable if securities distributed to you had been shares and the shares had been deposited for issuance of ADSs |
|
Distribution of securities distributed to holders of deposited securities (including rights) that are distributed by the depositary to ADS holders |
US$0.05 (or less) per ADS per calendar year |
|
Depositary services |
Registration or transfer fees |
|
Transfer and registration of shares on our share register to or from the name of the depositary or its agent when you deposit or withdraw shares |
Expenses of the depositary |
|
Cable (including SWIFT) and facsimile transmissions (when expressly provided in the deposit agreement) Converting foreign currency to U.S. dollars |
Taxes and other governmental charges the depositary or the custodian has to pay on any ADSs or shares underlying ADSs, such as stock transfer taxes, stamp duty or withholding taxes |
|
As necessary |
Any charges incurred by the depositary or its agents for servicing the deposited securities |
|
As necessary |
The depositary collects its fees for delivery and surrender of ADSs directly from investors depositing shares or surrendering ADSs for the purpose of withdrawal or from intermediaries acting for them. The depositary collects fees for making distributions to investors by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees. The depositary may collect its annual fee for depositary services by deduction from cash distributions or by directly billing investors or by charging the book-entry system accounts of participants acting for them. The depositary may collect any of its fees by deduction from any cash distribution payable (or by selling a portion of securities or other property distributable) to ADS holders that are obligated to pay those fees. The depositary may generally refuse to provide fee-attracting services until its fees for those services are paid.
From time to time, the depositary may make payments to us to reimburse us for costs and expenses generally arising out of establishment and maintenance of the ADS program, waive fees and expenses for services provided to us by the depositary or share revenue from the fees collected from ADS holders. In performing its duties under the deposit agreement, the depositary may use brokers, dealers, foreign currency dealers or other service providers that are owned by or affiliated with the depositary and that may earn or share fees, spreads or commissions.
166
The depositary may convert currency itself or through any of its affiliates, or the custodian or we may convert currency and pay U.S. dollars to the depositary. Where the depositary converts currency itself or through any of its affiliates, the depositary acts as principal for its own account and not as agent, advisor, broker or fiduciary on behalf of any other person and earns revenue, including, without limitation, transaction spreads, that it will retain for its own account. The revenue is based on, among other things, the difference between the exchange rate assigned to the currency conversion made under the deposit agreement and the rate that the depositary or its affiliate receives when buying or selling foreign currency for its own account. The depositary makes no representation that the exchange rate used or obtained by it or its affiliate in any currency conversion under the deposit agreement will be the most favorable rate that could be obtained at the time or that the method by which that rate will be determined will be the most favorable to ADS holders, subject to the depositary’s obligation to act without negligence or bad faith. The methodology used to determine exchange rates used in currency conversions made by the depositary is available upon request. Where the custodian converts currency, the custodian has no obligation to obtain the most favorable rate that could be obtained at the time or to ensure that the method by which that rate will be determined will be the most favorable to ADS holders, and the depositary makes no representation that the rate is the most favorable rate and will not be liable for any direct or indirect losses associated with the rate. In certain instances, the depositary may receive dividends or other distributions from the United States in U.S. dollars that represent the proceeds of a conversion of foreign currency or translation from foreign currency at a rate that was obtained or determined by us and, in such cases, the depositary will not engage in, or be responsible for, any foreign currency transactions and neither it nor we make any representation that the rate obtained or determined by us is the most favorable rate and neither it nor we will be liable for any direct or indirect losses associated with the rate.
Payment of Taxes
ADS holders are responsible for any taxes or other governmental charges payable on your ADSs or on the deposited securities represented by any of your ADSs. The depositary may refuse to register any transfer of your ADSs or allow you to withdraw the deposited securities represented by your ADSs until those taxes or other charges are paid. It may apply payments owed to you or sell deposited securities represented by your ADSs to pay any taxes owed and you will remain liable for any deficiency. If the depositary sells deposited securities, it will, if appropriate, reduce the number of ADSs to reflect the sale and pay to ADS holders any proceeds, or send to ADS holders any property, remaining after it has paid the taxes. Your obligation to pay taxes and indemnify us and the depository against any tax claims will survive the transfer or surrender of your ADSs, the withdrawal of the deposited ordinary shares as well as the termination of the deposit agreement.
See Exhibit 2.3:” Description of Securities” for additional information on the ADSs.
167
PART II
Item 13. Defaults, Dividend Arrearages and Delinquencies
Not applicable.
Item 14. Material Modifications to the Rights of Security Holders and Use of Proceeds
E. Use of Proceeds
Not applicable
Item 15. Controls and Procedures
A. Disclosure Controls and Procedures
We maintain disclosure controls and procedures designed to provide reasonable assurance that information required to be disclosed in reports filed under the Exchange Act is recorded, processed, summarized and reported within the specified time periods and accumulated and communicated to our management, including our Principal Executive Officer and Principal Financial Officer, as appropriate, to allow timely decisions regarding required disclosure. Our management, with the participation of our Principal Executive Officer and Principal Financial Officer, has evaluated the effectiveness of our disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) as of June 30, 2023. Based on such evaluation, our Principal Executive Officer and Principal Financial Officer have concluded that, as of June 30, 2023, our disclosure controls and procedures were not effective at the reasonable assurance level because of the material weakness in our internal control over financial reporting as described below.
B. Management’s Annual Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal controls over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) and for the assessment of the effectiveness of our internal control over financial reporting. Under the supervision and with the participation of our Chief Executive Officer (principal executive officer) and our Chief Financial Officer (principal financial officer), management assessed our internal control over financial reporting based upon the framework in Internal Control — Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). Based on this assessment, our management has concluded that our internal control over financial reporting was not effective at the reasonable assurance level as of June 30, 2023 because we did not design and maintain effective controls in relation to accounting for non-routine transactions as the control failed to detect and prevent the incorrect remeasurement of the fair value of our Funding Agreement liability following changes in certain assumptions relating to the timing of the repayments by the Company to Ocelot under the Funding Agreement.
We will continue to take certain measures to remediate the material weakness described above including implementing key mitigating controls to address the risk underlying assumptions used in relation to accounting for non-routine transactions, such as the Funding Agreement. The material weakness will not be considered remediated until management completes the design and implementation of the measures described above and the controls operate for a sufficient period of time and management has concluded, through testing, that these controls are effective. We are working to remediate the material weakness as efficiently and effectively as possible.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements, and can only provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
C. Attestation Report of the Registered Public Accounting Firm
This annual report does not include an attestation report of the company’s registered public accounting firm due to a transition period established by the SEC’s rules for emerging growth companies.
168
D. Changes in Internal Control Over Financial Reporting
Other than as discussed above in Management's Annual Report on Internal Control Over Financial Reporting, there have been no changes to our internal control over financial reporting during the fiscal year ended June 30, 2023, that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
169
Item 16. [Reserved]
Item 16A. Audit Committee Financial Expert
The members of our Audit and Risk Committee are Messrs. Quinton Oswald, Daniel Spiegelman and Dr Susan Orr Our board of directors has determined that each of Messrs.Oswald, Spiegelman and Dr Orr satisfies the independence requirements under Nasdaq listing standards and Rule 10A-3(b)(1) of the Exchange Act. The chairperson of our Audit and Risk Committee is Mr. Spiegelman. Our board of directors has determined that Mr. Spiegelman is an “audit committee financial expert” within the meaning of SEC regulations. Each member of our Audit and Risk Committee can read and understand fundamental financial statements in accordance with applicable requirements. In arriving at these determinations, our board of directors has examined each member’s scope of experience and the nature of his or her employment.
Item 16B. Code of Ethics
We have adopted a Code of Conduct applicable to all of our directors, officers and employees. Our Code of Conduct is available on our website at www.opthea.com. We post on our website all disclosures that are required by law or the listing standards of Nasdaq concerning any amendments to, or waivers from, any provision of the Code of Conduct. The reference to our website address does not constitute incorporation by reference of the information contained at or available through our website, and you should not consider it to be a part of, this annual report.
170
Item 16C. Principal Accountant Fees and Services
Fees Paid to Independent Public Accountants
The following table sets forth, for each of the years indicated, the fees billed by Deloitte Touche Tohmatsu.
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
(in thousands A$) |
|
|||||||||
Audit Fees |
|
|
A$357 |
|
|
|
A$295 |
|
|
|
A$409 |
|
Audit-Related Fees |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Tax Fees |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Other Fees |
|
|
— |
|
|
|
171 |
|
|
|
45 |
|
Total |
|
|
A$357 |
|
|
|
A$499 |
|
|
|
A$454 |
|
“Audit Fees” are the aggregate fees billed for the audit of our annual financial statements. This category also includes services that Deloitte Touche Tohmatsu provides, such as consents and assistance with and review of documents filed with the SEC.
“Audit-Related Fees” are the aggregate fees billed for assurance and related services that are reasonably related to the performance of the audit, including fees related to our public offering, and are not reported under Audit Fees.
“Tax Fees” are the aggregate fees billed for professional services rendered by Deloitte Touche Tohmatsu for tax compliance, tax advice and tax planning related services.
“Other Fees” are any additional amounts billed for products and services provided by Deloitte Touche Tohmatsu.
There were no “Audit-Related Fees,” or “Tax Fees” billed or paid during the fiscal years ended June 30, 2023, 2022 or 2021.
Pre-Approval of Audit and Non-Audit Services
The Audit and Risk Management Committee’s pre-approval is required for all services provided by Deloitte Touche Tohmatsu. These services may include audit services, audit-related services, tax services and permissible non-audit services, and are subject to a specific budget. The Audit and Risk Management Committee uses a combination of two approaches – general pre-approval and specific pre-approval – in considering whether particular services or categories of services are consistent with the SEC’s rules on auditor independence. Under general pre-approval proposed services may be pre-approved without consideration of specific case-by-case services.
Item 16D. Exemptions from the Listing Standards for Audit Committees
Not applicable.
Item 16E. Purchases of Equity Securities by the Issuer and Affiliated Purchasers
Not applicable.
Item 16F. Change in Registrant’s Certifying Accountant
Not applicable.
171
Item 16G. Corporate Governance
Under Nasdaq Stock Market Rule 5615(a)(3), foreign private issuers, such as our company, are permitted to follow certain home country corporate governance practices instead of certain provisions of the Nasdaq Stock Market Rules. In order to rely on this exception, we are required to disclose each Nasdaq Stock Market Rule that we do not follow and describe the home country practice we do follow in lieu thereof. In accordance with this exception, we intend to follow Australian corporate governance practices in lieu of the following Nasdaq corporate governance standards:
Item 16H. Mine Safety Disclosure
Not applicable.
Item 16I. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable.
Item 16J. Insider Trading Policies
Not applicable.
172
PART III
Item 17. Financial Statements
See “Item 18—Financial Statements.”
Item 18. Financial Statements
The consolidated financial statements and the related notes required by this Item are included in this annual report beginning on page F-1.
Item 19. Exhibits
|
|
|
|
Incorporated by Reference |
|
|
||||||||
Exhibit Number |
|
Exhibit Description |
|
Form |
|
File No. |
|
Exhibit |
|
|
Filing Date |
|
Filed Herewith |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1.1 |
|
|
F-1 |
|
333-249020 |
|
|
3.1 |
|
|
9/24/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1.2 |
|
|
F-1 |
|
333-249020 |
|
|
3.2 |
|
|
9/24/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2.1 |
|
|
F-1/A |
|
333-249020 |
|
|
4.1 |
|
|
10/9/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2.2 |
|
Form of American Depositary Receipt evidencing American Depositary Shares. |
|
F-1/A |
|
333-249020 |
|
|
4.1 |
|
|
10/9/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2.3 |
|
|
20-F |
|
001-39621 |
|
|
2.3 |
|
|
10/28/21 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.1* |
|
|
6-K |
|
001-39621 |
|
|
99.3 |
|
|
9/13/21 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.2* |
|
|
F-1 |
|
333-249020 |
|
|
10.6 |
|
|
9/24/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.3* |
|
Executive Employment Contract, dated April 23, 2014, between the Registrant and Megan Baldwin, Ph.D. |
|
F-1 |
|
333-249020 |
|
|
10.7 |
|
|
9/24/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.4* |
|
Executive Employment Contract, dated May 3, 2021, between the Registrant and Karen Adams. |
|
20-F |
|
001-39621 |
|
|
4.4 |
|
|
10/28/21 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.5* |
|
Executive Employment Contract, dated October 24, 2022, between the Opthea US Inc and Timothy Morris |
|
|
|
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.6* |
|
|
F-1/A |
|
333-249020 |
|
|
10.9 |
|
|
10/9/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.7# |
|
Commercial License Agreement, dated as of October 28, 2013, between the Registrant and Selexis SA. |
|
F-1 |
|
333-249020 |
|
|
10.1 |
|
|
9/24/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.8# |
|
|
F-1 |
|
333-249020 |
|
|
10.2 |
|
|
9/24/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.9 |
|
|
F-1 |
|
333-249020 |
|
|
10.3 |
|
|
9/24/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
173
4.10 |
|
|
F-1 |
|
333-249020 |
|
|
10.4 |
|
|
9/24/20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.11 |
|
|
20-F |
|
001-39621 |
|
|
4.10 |
|
|
9/29/22 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.12 * |
|
Executive Employment Contract, dated December 31, 2021, between Opthea US Inc and Judith Robertson. |
|
20-F |
|
001-39621 |
|
|
4.11 |
|
|
9/29/22 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.14 |
|
Sales Agreement by and between the Registrant and Jefferies LLC, dated February 1, 2022. |
|
F-3 |
|
333-262444 |
|
|
1.2 |
|
|
2/1/22 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.15 # |
|
Development Funding Agreement by and between Opthea Limited and Ocelot SPV LP dated August 12, 2022 |
|
20-F |
|
001-39621 |
|
|
4.14 |
|
|
9/29/22 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4.16 |
|
Consultancy Agreement by and between Opthea Limited and Lawrence Gozlan, dated as of August 28, 2023 |
|
|
|
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
8.1 |
|
|
20-F |
|
001-39621 |
|
|
8.1 |
|
|
10/28/21 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
12.1 |
|
|
|
|
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
12.2 |
|
|
|
|
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
13.1† |
|
|
|
|
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
15.1 |
|
Consent of Deloitte Touche Tohmatsu, independent registered public accounting firm. |
|
|
|
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
101 |
|
The following financial statements from the Company’s Annual Report on Form 20-F for the fiscal year ended June 30, 2023 were formatted in Inline XBRL: (i) Consolidated Statement of Profit or Loss and Comprehensive Income, (ii) Consolidated Statement of Financial Position, (iii) Consolidated Statement of Changes in Equity, (iv) Consolidated Statement of Cash Flows, and (v) the Notes to Consolidated Financial Statements, tagged as blocks of text and including detailed tags. |
|
|
|
|
|
|
|
|
|
|
|
X |
174
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
104 |
|
The cover page from the Company’s Annual Report on Form 20-F for the fiscal year ended June 30, 2023, formatted in Inline XBRL (Included as Exhibit 101). |
|
|
|
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
* Indicates management contract or compensatory plan or arrangement.
# Certain confidential portions of this exhibit were omitted by means of marking such portions with brackets (“[***]”) because the identified confidential portions are not material and are of the type that the Company treats as private or confidential. The registrant agrees to furnish supplementally a copy of any omitted schedule or exhibit to the SEC upon request.
† The certifications attached as Exhibits 13.1 and 13.2 accompanying this Annual Report on Form 20-F are not deemed filed with the SEC and are not to be incorporated by reference into any filing of Opthea Limited under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date of this Annual Report on Form 20-F, irrespective of any general incorporation language contained in such filing.
175
SIGNATURES
The registrant hereby certifies that it meets all of the requirements for filing on Form 20-F and that it has duly caused and authorized the undersigned to sign this annual report on its behalf.
|
Opthea Limited |
||
|
|
|
|
|
By: |
|
/s/ Megan Baldwin |
|
Name: |
|
Megan Baldwin, Ph.D. |
|
Title: |
|
Chief Executive Officer and Managing Director |
Dated: September 28, 2023
176
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
Report of Independent Registered Public Accounting Firm (PCAOB ID: 0 |
F-2 |
|
|
Consolidated Statements of Profit or Loss and Other Comprehensive Income |
F-3 |
|
|
F-4 |
|
|
|
F-5 |
|
|
|
F-6 |
|
|
|
F-7 |
F-1
Report of Independent Registered Public Accounting Firm
To the Shareholders and the Board of Directors of Opthea Limited
Opinion on the Financial Statements
We have audited the accompanying consolidated statements of financial position of Opthea Limited and subsidiaries (the “Company”) as of June 30, 2023 and 2022, the related consolidated statements of profit or loss and other comprehensive income, changes in equity, and cash flows for each of the three years in the period ended June 30, 2023, and the related notes (collectively referred to as the “financial statements”).
In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of June 30, 2023 and 2022, and the results of its operations and its cash flows for each of the three years in the period ended June 30, 2023, in conformity with International Financial Reporting Standards as issued by the International Accounting Standards Board.
Substantial Doubt related to Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the financial statements, the Group incurred a net loss of $142.5 million, has a net cash outflow from operating activities of $120.6 million during the year and, as of June 30, 2023, the Group had an equity deficit of $5.8 million that raise substantial doubt about its ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 2 and 36. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/
DELOITTE TOUCHE TOHMATSU
September 28, 2023
We have served as the Company’s auditors since 2012.
F-2
OPTHEA LIMITED
Consolidated Statements of Profit or Loss and Other Comprehensive Income
For the Years ended June 30, 2023, and 2022, and 2021
|
|
Years ended June 30, |
|
|||||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Revenue |
|
|
|
|
|
|
|
|
|
|||
Other income |
|
|
|
|
|
|
|
|
|
|||
Operating expenses: |
|
|
|
|
|
|
|
|
|
|||
Research and development |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Patent and intellectual property expenses |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Administration expenses |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Interest expense on DFA |
|
|
( |
) |
|
|
— |
|
|
|
— |
|
Finance income |
|
|
|
|
|
|
|
|
|
|||
Fair value adjustment gain on DFA |
|
|
|
|
|
— |
|
|
|
— |
|
|
Net foreign exchange (loss)/gain |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Loss before income tax |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Income tax benefit |
|
|
|
|
|
|
|
|
|
|||
Loss for the year |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|||
Other comprehensive income |
|
|
|
|
|
|
|
|
|
|||
Items that will not be reclassified subsequently to profit or loss: |
|
|
|
|
|
|
|
|
|
|||
Fair value gains on investments in financial assets |
|
|
— |
|
|
|
— |
|
|
|
|
|
Other comprehensive income for the period, net of tax |
|
|
— |
|
|
|
— |
|
|
|
|
|
Total comprehensive loss for the year |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|||
Loss for the year is attributable to: |
|
|
|
|
|
|
|
|
|
|||
Owners of the Company |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Net loss |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Total comprehensive loss for the year is attributable to: |
|
|
|
|
|
|
|
|
|
|||
Owners of the Company |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Comprehensive loss |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Loss per share attributable to the owners of the Company: |
|
|
|
|
|
|
|
|
|
|||
- Basic and diluted loss per share (cents) |
|
|
) |
|
|
) |
|
|
) |
|||
The above consolidated statements of profit or loss and other comprehensive income should be read in conjunction with the accompanying notes.
F-3
OPTHEA LIMITED
Consolidated Statements of Financial Position
as of June 30, 2023, and 2022
|
|
June 30, |
|
|
June 30, |
|
||
|
|
2023 |
|
|
2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
Assets |
|
|
|
|
|
|
||
Current assets: |
|
|
|
|
|
|
||
Cash and cash equivalents |
|
|
|
|
|
|
||
Current tax receivable |
|
|
|
|
|
|
||
Receivables |
|
|
|
|
|
|
||
Prepayments |
|
|
|
|
|
|
||
Total current assets |
|
|
|
|
|
|
||
Non-current assets: |
|
|
|
|
|
|
||
Property and equipment, net |
|
|
|
|
|
|
||
Right-of-use assets |
|
|
|
|
|
— |
|
|
Prepayments |
|
|
|
|
|
|
||
Total non-current assets |
|
|
|
|
|
|
||
Total assets |
|
|
|
|
|
|
||
Liabilities |
|
|
|
|
|
|
||
Current liabilities: |
|
|
|
|
|
|
||
Payables (includes amounts to a related party of US$ |
|
|
|
|
|
|
||
Lease liabilities |
|
|
|
|
|
— |
|
|
Provisions |
|
|
|
|
|
|
||
Total current liabilities |
|
|
|
|
|
|
||
Non-current liabilities: |
|
|
|
|
|
|
||
Lease liabilities |
|
|
|
|
|
— |
|
|
Financial liability due to a related party |
|
|
|
|
|
— |
|
|
Provisions |
|
|
|
|
|
|
||
Total non-current liabilities |
|
|
|
|
|
|
||
Total liabilities |
|
|
|
|
|
|
||
Net Assets |
|
|
( |
) |
|
|
|
|
Equity |
|
|
|
|
|
|
||
Contributed equity |
|
|
|
|
|
|
||
Accumulated Loss |
|
|
( |
) |
|
|
( |
) |
Reserves |
|
|
|
|
|
|
||
Total Equity |
|
|
( |
) |
|
|
|
|
The above consolidated statements of financial position should be read in conjunction with the accompanying notes.
F-4
OPTHEA LIMITED
Consolidated Statements of Changes in Equity
for the Years ended June 30, 2023, and 2022, and 2021
|
|
|
|
|||||||||||||||||||||||||
|
|
Contributed |
|
|
Pre-funded |
|
|
Share-based |
|
|
Fair value of |
|
|
FX translation |
|
|
Accumulated |
|
|
Total |
|
|||||||
|
|
equity |
|
|
warrants |
|
|
payment |
|
|
Investment |
|
|
Reserve |
|
|
Deficit |
|
|
Equity |
|
|||||||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||||||
Balance at June 30, 2020 (Restated) |
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
( |
) |
|
|
|
||||||
Fair value gains on investments in |
|
— |
|
|
— |
|
|
— |
|
|
|
|
|
— |
|
|
— |
|
|
|
|
|||||||
Net loss for the year |
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
|
( |
) |
|
|
( |
) |
|||||
Total Comprehensive income and expense for the period |
|
— |
|
|
— |
|
|
— |
|
|
|
|
|
— |
|
|
|
( |
) |
|
|
( |
) |
|||||
Issuance of ordinary shares and pre-funded warrants on NASDAQ listing net of issuance cost $ |
|
|
|
|
|
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
|
|
|||||||
Issuance of ordinary shares on exercise of pre-funded warrants net of issuance costs $ |
|
|
|
|
|
( |
) |
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
||||||
Exercise of options granted under LTIP and NED plan |
|
|
|
|
— |
|
|
|
( |
) |
|
— |
|
|
— |
|
|
— |
|
|
— |
|
||||||
Recognition of share-based payment |
|
— |
|
|
— |
|
|
|
|
|
— |
|
|
— |
|
|
— |
|
|
|
|
|||||||
Exchange on conversion |
|
— |
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
|
|||||||
Balance at June 30, 2021 |
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
( |
) |
|
|
|
||||||
Net loss for the year |
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
|
( |
) |
|
|
( |
) |
|||||
Total Comprehensive income and expense for the period |
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
|
( |
) |
|
|
( |
) |
|||||
Exercise of options granted under LTIP and NED plan |
|
|
|
|
— |
|
|
|
( |
) |
|
— |
|
|
— |
|
|
— |
|
|
|
|
||||||
Recognition of share-based payment |
|
— |
|
|
— |
|
|
|
|
|
— |
|
|
— |
|
|
— |
|
|
|
|
|||||||
Balance at June 30, 2022 |
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
( |
) |
|
|
|
||||||
Net loss for the year |
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
|
( |
) |
|
|
( |
) |
|||||
Total Comprehensive income and expense for the period |
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
|
( |
) |
|
|
( |
) |
|||||
Issuance of ordinary shares net of issuance costs $ |
|
|
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
— |
|
|
|
|
|||||||
Exercise of options granted under LTIP and NED plan |
|
|
|
|
— |
|
|
|
( |
) |
|
— |
|
|
— |
|
|
— |
|
|
|
|
||||||
Recognition of share-based payment |
|
— |
|
|
— |
|
|
|
|
|
— |
|
|
— |
|
|
— |
|
|
|
|
|||||||
Balance at June 30, 2023 |
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
( |
) |
|
|
( |
) |
|||||
The above consolidated statements of change in equity should be read in conjunction with the accompanying notes.
F-5
OPTHEA LIMITED
Consolidated Statements of Cash Flows
for the Years ended June 30, 2023, and 2022, and 2021
|
|
Year ended June 30, |
|
|||||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Cashflow from operating activities |
|
|
|
|
|
|
|
|
|
|||
Interest received |
|
|
|
|
|
|
|
|
|
|||
Royalty and license income received |
|
|
|
|
|
|
|
|
|
|||
Grant and other income |
|
|
|
|
|
|
|
|
|
|||
Payment of lease interest |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Payments to suppliers, employees and for research & development and intellectual property costs ( inclusive of GST) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Research and development tax incentive scheme credit received |
|
|
|
|
|
|
|
|
|
|||
Net cash flows used in operating activities |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Cash flow from investing activities: |
|
|
|
|
|
|
|
|
|
|||
Purchase of plant and equipment |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Proceeds from sale of financial assets |
|
|
|
|
|
— |
|
|
|
|
||
Net cash used in investing activities |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
Cash flows from financing activities: |
|
|
|
|
|
|
|
|
|
|||
Payment of lease liabilities |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Net proceeds on issue of shares |
|
|
|
|
|
— |
|
|
|
|
||
Net proceeds on issue of pre-funded warrants |
|
|
— |
|
|
|
— |
|
|
|
|
|
Net proceeds from DFA |
|
|
|
|
|
— |
|
|
|
— |
|
|
Cash received for ordinary shares issued on exercise of options |
|
|
|
|
|
|
|
— |
|
|||
Net cash provided by financing activities |
|
|
|
|
|
|
|
|
|
|||
Increase in cash and cash equivalents |
|
|
|
|
|
( |
) |
|
|
|
||
Effects of exchange rate changes on the balance of |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
Cash and cash equivalents at beginning of period |
|
|
|
|
|
|
|
|
|
|||
Cash and cash equivalents at end of period |
|
|
|
|
|
|
|
|
|
|||
The above consolidated statements of cash flows should be read in conjunction with the accompanying notes.
F-6
OPTHEA LIMITED
Notes to Consolidated Financial Statements
Note 1 Reporting Entity
Opthea Limited (the Company) is a listed public company incorporated in Australia. The address of its registered office and principal place of business is: Suite 0403, Level 4, 650 Chapel Street, South Yarra, VIC 3141, Australia. These consolidated financial statements comprise the Company and its subsidiaries (together referred to as the Group). The Group’s principal activity is the development of new drugs for the treatment of eye diseases.
Note 2. Basis of Accounting
These financial statements are general purpose financial statements which have been prepared in accordance with International Financial Reporting Standards (“or IFRS”) as issued by the International Accounting Standards Board (the “IASB”).
The financial statements comprise the consolidated financial statements of the Group. For the purposes of preparing the consolidated financial statements, the Company is a for‑profit entity.
The financial statements were authorized for issue by the directors on September 28, 2023.
Going Concern
For the year ended June 30, 2023, the Group incurred a loss after income tax of $
The consolidated financial statements have been prepared on a going concern basis, which contemplates continuity of normal activities and realization of assets and settlement of liabilities in the normal course of business. As the Group is still in the research and development phase, the ability of the Group to continue its development activities as a going concern is dependent upon it deriving sufficient cash from investors and from funding provided under the Development Funding Agreement (‘DFA or Agreement’) with Carlyle and Abingworth. Of the initial total funding of US$
Subsequent to June 30, 2023, the Group was notified that a new co-investor of Carlyle and Abingworth intends to participate in a funding under the DFA of US$
Concurrently with the receipt of the notice from the co-investor to increase its investment, the Group entered into binding commitments for the private placement of ordinary shares and entitlement rights and accompanying options for aggregate gross proceeds of approximately A$
F-7
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
occurred on September 20, 2023. The shares were issued and cash received on September 20, 2023. See Note 37, Events after the balance sheet date, for further information.
While we expect that with our cash on hand at June 30, 2023 of $
The Directors and management have considered the cash flow forecasts including the funding requirements of the business as well as the funding expected to be raised through the Agreement and Equity Offering. They have also considered the Group’s key risks and uncertainties affecting the likely development of the business, as well as the conditions set forth in the Agreement. Based on this assessment, the Directors and management believe that the conditions in the DFA can be met and that the Group has adequate resources to continue normal activities and realize its assets and settle its liabilities in the normal course of business. Accordingly, the directors have prepared the financial statements on the going concern basis.
Should the Group not be able meet the conditions in the DFA to increase the total funding from US$
Note 3. Summary of Accounting Policies
The consolidated financial statements have been prepared using the significant accounting policies and measurement bases summarized below.
Basis of measurement
The consolidated financial statements have been prepared on a historical cost basis, except for the investments classified as financial assets, which have been measured at fair value. All amounts are presented in United States dollars unless otherwise stated.
Functional currency
An entity’s functional currency is the currency of the primary economic environment in which the entity operates. During 2021, the Group’s operations continued to move further towards being US$ denominated and several other factors during the period have also contributed to the Group changing its functional currency during the year, such as the completion of U.S. initial public offering (IPO) and the Nasdaq listing in October 2020, opening a US subsidiary in May 2021 for a planned expansion into the US, and expanding the Board of Directors with the appointment of now five US based Directors. A significant element in the Group’s assessment to change the functional currency resulted from the significant increase in expenses denominated in US dollars relating to advanced clinical trials since the commencement of Phase 3 trials in March 2021. These changes, as well as the fact that the Group’s principal source of financing is now the U.S. capital market and all of the Group’s budgeting and planning is conducted solely in dollars led to the Company determining that the U.S. dollar (US$) best represents the currency of the primary economic environment in which the entity now operates. Accordingly, the Group changed its functional currency from Australian dollar (A$) to U.S. dollar (US$) effective January 1, 2021.
Presentation Currency
Following the change in functional currency, the Group changed its presentation currency from Australian dollars (A$) to US$. The change in presentation currency was made to better reflect the Group’s business activities
F-8
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
and to enhance access to U.S. capital markets. Prior to the change, the Group reported its financial statements in Australian dollars (A$).
A change in presentation currency is a change in accounting policy which is accounted for retrospectively during 2021. In making this change in presentation currency, the Group followed the requirements set out in IAS 21 The Effects of Changes in Foreign Exchange Rates. As required by IAS 21, the consolidated statement of profit or loss and other comprehensive income and the consolidated statement of cash flows for each period have been translated into the presentation currency using the average exchange rates prevailing during each reporting period. All assets and liabilities have been translated using the exchange rates prevailing at the consolidated statement of financial position dates. Shareholders’ equity transactions have been translated using the rates of exchange in effect as of the dates of various capital transactions. All resulting exchange differences arising from the translation are included as a separate component of other comprehensive income. All comparative financial information has been restated in fiscal year 2021 to reflect the Group’s results as if they had been historically reported in US$ and the effect on the consolidated financial statements resulted in an addition to the foreign currency translation reserve of US$
Change in Presentation of Other Income
The Group changed its presentation of Other income by reclassifying interest income out of Other income and into Finance Income – interest income to better reflect the nature of the related amounts as finance income. This reclassification had no effect on the reported results of operations.
Basis of consolidation
The consolidated financial statements incorporate the financial statements of the Company and its subsidiaries. Control is achieved when the Company:
Consolidation of a subsidiary begins when the Company obtains control over the subsidiary and ceases when the Company loses control of the subsidiary.
All intragroup assets and liabilities, equity, income, expenses and cash flows relating to transactions between members of the Group are eliminated in full on consolidation.
Foreign currency translation
i. Functional and presentation currency
As of January 1, 2021 the Group determined that its functional and presentation currency had changed from Australian Dollars to United States Dollars. Therefore, the functional and presentation currency of the Group is the United States dollars (US$).
ii. Transactions and balances
Transactions in foreign currencies are initially recorded in the functional currency by applying the exchange rates ruling in place at the date of the transaction. Monetary assets and liabilities denominated in foreign currencies are retranslated at the rate of exchange ruling in place at the reporting date.
Non‑monetary items that are measured in terms of historical cost in a foreign currency are translated using the exchange rate as of the date of the initial transaction. Non‑monetary items measured at fair value in a foreign currency are translated using the exchange rates at the date when the fair value was determined.
Financial assets and liabilities
F-9
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Recognition and derecognition of financial assets
Purchases and sales of financial assets that require delivery of assets within the time frame generally established by regulation or convention in the marketplace are recognized on the trade date, i.e., the date that the Group commits to purchase the asset. Financial assets are derecognized when the right to receive cash flows from the financial assets has expired or when the entity transfers substantially all the risks and rewards of the financial assets. If the entity neither retains nor transfers substantially all of the risks and rewards, it derecognizes the asset if it has transferred control of the assets.
When financial assets are recognized initially, they are measured at fair value, plus directly attributable transaction costs.
Cash and cash equivalents
Cash and cash equivalents in the statement of financial position comprise cash at bank and in hand and short‑term deposits with an original maturity of three months or less that are readily convertible to known amounts of cash and which are subject to an insignificant risk of changes in value.
For the purposes of the statement of cash flows, cash and cash equivalents consist of cash and cash equivalents as defined above.
Other receivables
Other receivables generally comprise bank interest receivable, other receivables from external parties and Goods and Services Tax (GST) credits receivable and are recognized and carried at original invoice amount less an allowance for any uncollectible amounts. The amounts are usually received within
The Group measures the loss allowance for receivables at an amount equal to lifetime expected credit losses (ECL). The ECL on receivables are estimated under the simplified approach as permitted under IFRS 9 “Financial Instruments.” This uses a provision matrix by reference to past experience of the debtor and an analysis of the debtor’s current financial position, adjusted for factors that are specific to the debtors and general economic conditions of the industry in which the debtors operate.
The Group writes off a receivable when there is information indicating that the debtor is in severe financial difficulty and there is no realistic prospect of recovery.
Investments
Investments in financial assets comprise of the Group’s non‑current investments in listed companies.
On initial recognition, the Group may make an irrevocable election (on an instrument‑by‑instrument basis) to designate investments in equity instruments as fair value through other comprehensive income (FVTOCI). Designation at FVTOCI is not permitted if the equity instrument is held for trading.
Investments in equity instruments at FVTOCI are initially measured at fair value plus transaction costs. Subsequently, they are measured at fair value with gains or losses arising from changes in the fair value recognized in other comprehensive income and accumulated in the fair value of investments reserve. The fair values of investments in financial assets that are actively traded in organized financial markets is determined by reference to quoted market bid prices at the close of business on the reporting date. The cumulative gain or loss is not reclassified to profit or loss on disposal of the equity instruments.
Dividends on these investments in equity instruments are recognized in profit or loss in accordance with International Financial Reporting Standards.
Finance income
Almost all of the Group’s finance income is earned on short‑term bank deposits, and as such, finance income is recognized when the Group’s right to receive the payment is established.
F-10
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Payables
Payables are carried at amortized cost and due to their short‑term nature, they are not discounted. They represent liabilities for goods and services provided to the Group prior to the end of the financial year that are unpaid and arise when the Group becomes obliged to make future payments in respect of the purchase of these goods and services.
The amounts are unsecured and are usually paid within
Financial liabilities
Financial liabilities are recognized in the Group’s statement of financial position when the Group becomes a party to the contractual provisions of the instrument. Financial liabilities are initially measured at fair value. Transaction costs that are directly attributable to the acquisitions or issue of financial liabilities (other than financial liabilities at fair value through profit or loss) are deducted from the fair value of the financial liabilities, as appropriate, on initial recognition. Subsequent measurement of the liability will be at its amortized cost, subject to any re-measurement of the obligation for changes in assumptions.
Amortized cost and effective interest method
The effective interest method is a method of calculating the amortized cost of an instrument and of allocating interest expense over the relevant period. The effective interest rate is the rate that exactly discounts estimated future cash payments through the expected life of the financial liability, or (where appropriate) a shorter period, to the amortized cost of the financial liability.
Interest expense is recognized in profit and loss and is included in the “Interest expense on DFA” line item.
Equipment
Equipment is stated at historical cost less accumulated depreciation and any accumulated impairment losses. Depreciation is calculated on a straight‑line basis over their useful economic lives as follows:
The assets’ residual values, useful lives and amortization methods are reviewed, and adjusted if appropriate, at each financial year end.
An item of equipment is derecognized upon disposal or when no further economic benefits are expected from its use or disposal.
Research and development costs
Research costs are expensed as incurred. An intangible asset arising from the development expenditure on an internal project will only be recognized when the Group can demonstrate the technical feasibility of completing the intangible asset so that it will be available for use or sale, its intention to complete and its ability to use or sell the asset, how the asset will generate future economic benefits, the availability of resources to complete the development and the ability to measure reliably the expenditure attributable to the intangible asset during its development.
As of June 30, 2023, 2022 and 2021, the Group is in the research phase and has not capitalized any development costs to date.
Provisions and employee benefits
i. Wages, salaries, annual leave and sick leave
F-11
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Liabilities for wages and salaries, including non‑monetary benefits and annual leave expected to be settled within 12 months of the reporting date are recognized in current provisions in respect of employees’ services up to the reporting date. They are measured at the amounts expected to be paid when the liabilities are settled. Expenses for non‑accumulating sick leave are recognized when the leave is taken and are measured at the rate paid or payable.
ii. Long service leave
The liability for long service leave is recognized in the provision for employee benefits and measured as the present value of expected future payments to be made in respect of services provided by employees up to the reporting date. Consideration is given to expected future wage and salary levels, experience of employee departures, and periods of service. Expected future payments are discounted using market yields at the reporting date on bonds with terms to maturity that match, as closely as possible, the estimated future cash outflows.
Share‑based payment transactions
The Group provides benefits to directors and employees (including key management personnel) of the Group in the form of share‑based payments, whereby employees render services in exchange for shares or rights over shares (equity‑settled transactions).
The cost of these equity‑settled transactions with employees is measured by reference to the fair value at the date at which they are granted. Binomial models are used to value the options issued.
The cost of the equity‑settled transactions is recognized, together with a corresponding increase in equity, over the period in which the performance conditions are fulfilled (the vesting period), ending on the date on which the relevant employees become fully entitled to the award (the vesting date).
The charge to profit or loss for the period is the cumulative amount less the amounts already charged in previous periods. There is a corresponding credit to equity.
Until an award has vested, any amounts recorded are contingent and will be adjusted if more or fewer awards vest than were originally anticipated to do so.
Contributed equity
Ordinary shares are classified as equity. Incremental costs directly attributable to the issue of new shares or options are shown in equity as a deduction, net of tax, from the proceeds.
Revenue recognition
License revenue in connection with licensing of the Group’s intellectual property (including patents) to customers is recognized as a right to use the Group’s intellectual property as it exists at the point in time in which the license is granted. This is because the contracts for the license of intellectual property are distinct and do not require, nor does the customer reasonably expect, that the Group will undertake further activities that significantly affect the intellectual property to which the customer has the rights. Although the Group is entitled to sales‑based royalties from the eventual sales of goods and services to third parties using the intellectual property licensed, these royalty arrangements do not in themselves indicate that the customer would reasonably expect the Group to undertake such activities, and no such activities are undertaken or contracted in practice. Accordingly, the promise to provide rights to the Group’s intellectual property is accounted for as a performance obligation satisfied at a point in time.
The following consideration is received in exchange for licenses of intellectual property:
F-12
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
During the years ended June 30, 2023, 2022 and 2021, the Group’s only revenue related to sales‑based royalties.
Income tax
Current tax
Current tax assets and liabilities for the current and prior periods are measured at the amount expected to be recovered from or paid to the taxation authorities based on the current period’s taxable income.
The tax rates and tax laws used to compute the amount are those that are enacted or substantively enacted by the reporting date.
Research and development tax incentive
The Research and Development (R&D) Tax Incentive Scheme is an Australian Federal Government program under which eligible companies with annual aggregated revenue of less than A$
The Group estimates the amount of R&D tax incentive after the completion of the financial year based on eligible Australia and overseas expenditures incurred during that year.
The Group has presented incentives in respect of the R&D Tax Incentive Scheme within income tax benefit in the Statements of Profit or Loss and Other Comprehensive Income by analogizing with IAS 12 “Income Taxes”.
Deferred tax
Deferred income tax is provided on all temporary differences at the reporting date between the tax bases of assets and liabilities and their carrying amounts for financial reporting purposes.
Deferred income tax liabilities are recognized for all taxable temporary differences except when the deferred income tax liability arises from the initial recognition of goodwill or of an asset or liability in a transaction that is not a business combination and that, at the time of the transaction, affects neither the accounting profit nor taxable profit or loss.
Deferred income tax assets are recognized for all deductible temporary differences, carry forward of unused tax assets (or credits) and unused tax losses, to the extent that it is probable that taxable profit will be available against which the deductible temporary differences, and the carry forward of unused tax credits and unused tax losses can be utilized, except when the deferred income tax asset relating to the deductible temporary differences arises from the initial recognition of an asset or liability in a transaction that is not a business combination and, at the time of the transaction, affects neither the accounting profit or taxable profit or loss.
The carrying amount of deferred income tax assets is reviewed at each reporting date and reduced to the extent that it is no longer probable that sufficient taxable profit will be available to allow all or part of the deferred income tax asset to be utilized.
Unrecognized deferred income tax assets are reassessed at each reporting date and are recognized to the extent that it has become probable that future taxable profit will allow the deferred tax asset to be recovered.
Deferred income tax assets and liabilities are measured at the tax rates that are expected to apply to the year when the asset is realized or the liability is settled, based on tax rates (and tax laws) that have been enacted or substantively enacted at balance date.
F-13
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Income taxes relating to items recognized directly in equity are recognized directly in equity and not in profit or loss.
Tax consolidation legislation
Tax consolidation is a system adopted by the ATO that treats a group of entities as a single entity for tax purposes. Opthea Limited and its
This method requires adjustments for transactions and events occurring within the tax consolidated group that do not give rise to a tax consequence for the Group or that have a different tax consequence at the level of the Group.
The head entity which is the parent entity, in assuming the net unused tax losses and unused relevant tax credits, has recognized reductions to investments in subsidiaries and where the amount of tax losses assumed is in excess of the carrying value of the investment, the parent has recognized the difference as a distribution from subsidiaries in profit or loss.
Other taxes
Revenues, expenses, assets and liabilities are recognized net of the amount of GST except:
The net amount of GST recoverable from, or payable to the taxation authority is included as part of receivables or payables in the statement of financial position.
Cash flows are included in the statement of cash flows on a gross basis and the GST component of cash flows arising from investing and financing activities, which is recoverable from, or payable to, the taxation authority is classified as part of operating cash flows.
Commitments and contingencies are disclosed net of the amount of GST recoverable from, or payable to, the taxation authority.
F-14
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 4. Critical Accounting Judgments and Key Sources of Estimation Uncertainty
In applying the Group’s accounting policies, management continually evaluates judgments, estimates and assumptions based on experience and other factors, including expectations of future events that may have an impact on the Group. All judgments, estimates and assumptions made are believed to be reasonable based on the most current set of circumstances available to management. Actual results may differ from the judgments, estimates and assumptions.
Significant judgments, estimates and assumptions made by management in the preparation of these financial statements are outlined below:
4.1 Critical judgments in applying accounting policies
Research and development costs
The majority of Opthea’s expenditure is incurred as a result of clinical trials for OPT‑302. During the years ended June 30, 2020 2021 and 2022, Opthea completed its Phase 2b wet age‑related macular degeneration (wet AMD) and Phase 1b/2a diabetic macular edema (DME) trials and Phase 3 clinical trials for Sozinibercept (OPT-302) was initiated during 2021. A key measure of Opthea’s performance is the level of expenditure incurred on the research of OPT‑302.
Judgment is required in relation to:
The directors have determined that the Group is still in a research phase and accordingly,
Taxation
Research and development tax incentive
The Research and Development (R&D) Tax Incentive Scheme is an Australian Federal Government program under which eligible companies can receive cash refunds of
F-15
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
For the years ended June 30, 2023, and 2022, the Group has recognized an R&D tax incentive receivable of US$
The R&D tax incentive receivable as of June 30, 2023 and 2022 is based on the legislation as currently enacted as of June 30, 2023 and 2022, respectively. Any proposed changes to the legislation, such as rate changes and eligibility requirements, may have a retrospective impact if the legislation is passed. During the year, no such legislative changes have occurred.
Investment tax credits such as the R&D tax incentive are outside of the scope of IAS 12 “Income Taxes” and IAS 20 “Accounting for Government Grants and Disclosure of Government Assistance.” Based on the guidance in IAS 8 “Accounting Policies, Changes in Accounting Estimates and Errors,” companies need to make an accounting policy choice on how to present these incentives, which in practice is done by either analogizing with IAS 12 or with IAS 20. In the Group’s opinion, the R&D tax incentive should be presented by analogizing to IAS 12 because the nature of the incentive is considered to be more closely aligned to income taxes, based on the following considerations:
Income tax
The Group’s accounting policy for taxation requires judgments as to the differences between tax and accounting treatments of income and costs recognized in the consolidated statements of profit or loss and other comprehensive income. Judgment is also required in assessing whether deferred tax assets and liabilities are recognized in the statements of financial position and if accumulated income tax losses can be used to offset potential future tax profits.
Functional currency
Effective January 1, 2021 the Group’s functional and presentation currency changed from Australian dollars to U.S. dollars as disclosed in Note 3.
The Group’s assets, liabilities and equity which were previously denominated in Australian dollars were translated into U.S. dollars on the date the functional currency changed.
Significant judgment is required in determining the currency of the primary economic environment in which the Group operates, which requires an evaluation of various indicators related to the Group’s underlying transactions, events and conditions as they relate to generating and expending cash.
F-16
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
4.2 Key sources of estimation uncertainty
Development funding - financial liability
The Group evaluated the Financing Agreement and determined it to be a research and development funding arrangement with the characteristics of a debt instrument, as the transfer of financial risk to Launch Tx was not considered substantive and genuine. Accordingly, the Group has recorded payments received under the Financing Agreement as part of a development financing liability in its consolidated balance sheet. The Group measures the overall development financing liability at amortized cost based on the estimated timing of regulatory approval and attainment of certain sales milestones and the contractual success fee payments expected to be due therefrom, as discounted using an imputed interest rate. The development financing liability will be accreted as interest expense to its expected future repayment amount over the expected life of the agreement using the effective interest rate method. If the dates are delayed from those used at reporting date, it is expected that a fair value adjustment will result in a non-cash gain. If the timelines for approval and launch are accelerated, the Group would anticipate a fair value adjustment resulting in a non-cash charge to be recognized in the Consolidated Statements of Profit or Loss.
Share‑based payment transactions
The Group measures the cost of equity‑settled transactions with employees by reference to the fair value of the equity instruments at the date at which they are granted. Fair values are determined internally using Binomial models. The related assumptions are detailed in note 34. The accounting estimates and assumptions relating to equity‑settled share‑based payments have no impact on the carrying amounts of assets and liabilities in future reporting periods but may impact expenses and equity. Should one or more of the assumptions and estimates used in estimating the fair value of share‑based payments change, this could have a material impact on the amounts recognized in equity and employee‑related expenses.
Note 5. Application of New and Revised Accounting Standards
New and amended Accounting Standards that are effective for the current year
The Group has adopted all of the new and revised Standards and Interpretations issued by the International Accounting Standards Board (the IASB) that are relevant to its operations and effective for the current year.
In the current year, the Group has applied a number of amendments to International Financial Reporting Standards and Interpretations issued by the International Accounting Standards Board (IASB) that are effective for an annual period that begins on or after July 1, 2022. Their adoption has not had any material impact on the disclosures or on the amounts reported in these financial statements.
New and revised International Accounting Standards and Interpretations on issue but not yet effective
At the date of authorization of the financial statements, the Group has not applied the new and revised International Accounting Standards, Interpretations and amendments that have been issued but are not yet effective.
The new and revised Accounting Standards, Interpretations and amendments are not expected to have a material impact on the amounts recognized or disclosures included in the Group’s financial statements.
Note 6. Segment Information
The Group operates in
The Group is focused primarily on developing a novel therapy for the treatment of highly prevalent and progressive retinal diseases.
The chief executive officer regularly reviews entity wide information that is compliant with International Financial Reporting Standards. There is only
F-17
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
The Group’s only revenue stream in the current and previous financial year is royalty income generated from licenses granted in respect of the Group’s intellectual property that are unrelated to the Group’s core business and the development of OPT‑302 and that are not under development. These licenses are primarily used by third‑party licensees for research purposes. All of the royalty income of US$
Note 7. Revenue
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Sales-based royalties |
|
|
|
|
|
|
|
|
|
|||
Total Revenue |
|
|
|
|
|
|
|
|
|
|||
Note 8. Other Income
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Grant and other income |
|
|
|
|
|
|
|
|
|
|||
Total other income |
|
|
|
|
|
|
|
|
|
|||
Note 9. Research and Development Expenses
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Research project costs |
|
|
|
|
|
|
|
|
|
|||
Total research and development expenses |
|
|
|
|
|
|
|
|
|
|||
The research project costs relate to the research programs in respect to the treatment of eye diseases by Sozinibercept (OPT‑302).
F-18
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 10. Expenses
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Administrative expenses |
|
|
|
|
|
|
|
|
|
|||
Employee benefits expenses: |
|
|
|
|
|
|
|
|
|
|||
Salaries and fees |
|
|
|
|
|
|
|
|
|
|||
Cash bonuses |
|
|
|
|
|
|
|
|
|
|||
Superannuation |
|
|
|
|
|
|
|
|
|
|||
Share-based payments expense |
|
|
|
|
|
|
|
|
|
|||
Total employee benefits expense |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
Other expenses: |
|
|
|
|
|
|
|
|
|
|||
Insurance |
|
|
|
|
|
|
|
|
|
|||
Investor relations costs |
|
|
|
|
|
|
|
|
|
|||
Audit and accounting |
|
|
|
|
|
|
|
|
|
|||
Travel expenses |
|
|
|
|
|
|
|
|
|
|||
Payroll tax |
|
|
|
|
|
|
|
|
|
|||
Legal fees |
|
|
|
|
|
|
|
|
|
|||
Advisory fees1 |
|
|
|
|
|
|
|
|
|
|||
Consultancy costs |
|
|
|
|
|
|
|
|
|
|||
Other expenses |
|
|
|
|
|
|
|
|
|
|||
Total other expenses |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
Depreciation of: |
|
|
|
|
|
|
|
|
|
|||
Equipment and furniture |
|
|
|
|
|
|
|
|
|
|||
Right-of-use assets |
|
|
|
|
|
|
|
|
|
|||
Total depreciation expense |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
Loss on disposal of non-current assets |
|
|
— |
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|||
Total administrative expenses |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
1. Advisory fees relates to a market assessment of potential financing alternatives and solutions.
F-19
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 11. Finance Income
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Interest income |
|
|
|
|
|
|
|
|
|
|||
Total finance income |
|
|
|
|
|
|
|
|
|
|||
Note 12. Interest expense on DFA
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Interest expense on DFA |
|
|
|
|
|
— |
|
|
|
— |
|
|
Total interest expense on DFA |
|
|
|
|
|
— |
|
|
|
— |
|
|
The interest expense on DFA is non-cash interest at the imputed rate of
Note 13. Fair value adjustment gain on DFA
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Fair value adjustment gain on DFA |
|
|
|
|
|
— |
|
|
|
— |
|
|
Total fair value adjustment gain on DFA |
|
|
|
|
|
— |
|
|
|
— |
|
|
There are several factors that could affect the estimated timing of regulatory approval and attainment of sales milestones, some of which are not entirely within the Company’s control. Therefore, at each reporting date, the Company reassesses the estimated timing of regulatory approval and attainment of sales milestones and the expected contractual success fee payments due therefrom. If the timing and/or amount of such expected payments is materially different than original estimates, the Company will prospectively adjust the accretion of the development financing liability and the imputed interest rate.
At June 30, 2023 the Company performed a fair value adjustment of the carrying amount of the Financial Liability. The expected timeline for approval and commercial launch have been delayed by twelve months, thus extending date of expected repayments. As the Company has more time to repay the amounts owed, the carrying value of the Financial Liability at June 30, 2023 was adjusted downward to reflect this delay. The fair value adjustment resulted in a non-cash gain on revaluation of $
At every reporting period, the Company will review the expected approval and commercial launch dates. If the dates are delayed from those used at June 30, 2023, it is expected that a fair value adjustment will result in another non-cash gain. If the timelines for approval and launch are accelerated, the Company would anticipate a fair value adjustment resulting in a non-cash charge to be recognized on the Profit and Loss statement.
Note 14. Net Foreign Exchange Loss
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Net foreign exchange (loss)/gain |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Total net foreign exchange (loss)/gain |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Exchange differences arising on the translation of monetary items are recognized in the Statements of Profit and Loss and other Comprehensive Income. After the Company’s US IPO where the Company raised US$
F-20
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
exchange contract under which the term deposit converted back to US dollars at effectively the same foreign exchange rate as when the term deposit was entered into. As the Group’s functional currency was the Australian dollar (A$) until December 31, 2020, the Group recorded a foreign exchange loss of US$
Note 15. Income Taxes
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
(a) Income tax benefit |
|
|
|
|
|
|
|
|
|
|||
The major components of income tax benefit are: |
|
|
|
|
|
|
|
|
|
|||
Statement of Profit or Loss and Other Comprehensive Income |
|
|
|
|
|
|
|
|
|
|||
Current tax |
|
|
|
|
|
|
|
|
|
|||
Current income tax credit |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
Deferred tax |
|
|
|
|
|
|
|
|
|
|||
In respect of the current year |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Total income tax benefit recognized in the Statement of Comprehensive Income |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
(b) Current tax receivable |
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Research and Development Tax Incentive Credit receivable |
|
|
|
|
|
|
|
|
|
|||
(c) Numerical reconciliation between aggregate income tax benefit recognized in the Statement of Profit of Loss and Other Comprehensive Income and benefit calculated per the statutory income tax rate
A reconciliation between income tax benefit and the product of accounting loss before income tax multiplied by the Group’s applicable income tax rate is as follows:
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Accounting loss before tax |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
At the Company's statutory income tax rate of |
|
|
|
|
|
|
|
|
|
|||
R&D tax incentive on eligible expenses |
|
|
|
|
|
|
|
|
|
|||
Non-deductible R&D expenditure |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Other non-deductible expenses - share-based payment expense |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Amount of temporary differences and carried forward tax losses not recognized |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|||
F-21
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
(d) Recognized deferred tax assets and liabilities in statement of financial position |
|
June 30, 2023 |
|
|
June 30,2022 |
|
|
June 30,2021 |
|
|||
Deferred income tax at June 30 relates to the following: |
|
|
|
|
|
|
|
|
|
|||
Deferred tax liabilities: |
|
|
|
|
|
|
|
|
|
|||
Interest and royalty income receivable (future assessable income) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|||
Deferred tax assets related to temporary differences: |
|
|
|
|
|
|
|
|
|
|||
Recognition of tax losses |
|
|
— |
|
|
|
— |
|
|
|
|
|
Accrued expenses and other liabilities |
|
|
|
|
|
|
|
|
|
|||
Employee provisions |
|
|
|
|
|
|
|
|
|
|||
Other miscellaneous items |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
Net deferred tax assets |
|
|
|
|
|
|
|
|
— |
|
||
Less: temporary differences not recognized |
|
|
( |
) |
|
|
( |
) |
|
|
— |
|
Net deferred tax recognized in the statement of financial position |
|
|
— |
|
|
|
— |
|
|
|
— |
|
(e) Unrecognized temporary differences
Temporary differences with respect to deferred tax assets associated with intellectual property and other miscellaneous items which have a low probability of realization are unrecognized. These amounted to US$
(f) Carry forward unrecognized tax losses
The Group had income tax losses of US$
(g) Franking credit balance
Franking credits are a type of tax credit in Australia that is available to the Group’s shareholder to reduce double taxation on any dividends paid by the Group. The franking account balance at the end of the financial year at
F-22
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 16. Earnings per Share
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
The following reflects the income used in the basic and diluted earnings per share computations: |
|
|
|
|
|
|
|
|
|
|||
(a) Earnings used in calculating earnings per share |
|
|
|
|
|
|
|
|
|
|||
Net loss attributable to ordinary equity holders of the parent |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|||
(b) Weighted average number of shares |
|
|
|
|
|
|
|
|
|
|||
Weighted average number of ordinary shares on issue for basic earnings per share |
|
|
|
|
|
|
|
|
|
|||
Effect of dilution: |
|
|
|
|
|
|
|
|
|
|||
Share options |
|
|
|
|
— |
|
|
— |
|
|||
Weighted average number of ordinary shares adjusted for the effect of dilution |
|
|
|
|
|
|
|
|
|
|||
Loss per share (basic and diluted in cents) |
|
|
) |
|
|
) |
|
|
) |
|||
On August 24 and 28, 2023 the company announced a capital raising which has involved
Diluted earnings per share is calculated as net loss divided by the weighted average number of ordinary shares and dilutive potential ordinary shares. Options granted under the Long Term Incentive (LTIP) and Non‑Executive Director Share and Option (NED Plan) plans would generally be included in the calculation due to the conditions of the issuance being satisfied. As the Group is in a loss position, the options are anti‑dilutive and, accordingly, the basic loss per share is the same as the diluted loss per share.
A total number of
Fully paid ordinary shares have
|
|
|
|
|||||||||
Options |
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
NED Plan |
|
|
|
|
|
|
|
|
|
|||
LTIP |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
Performance Rights |
|
|
|
|
|
|
|
|
|
|||
NED Plan |
|
|
|
|
|
— |
|
|
|
— |
|
|
LTIP |
|
|
|
|
|
|
|
— |
|
|||
|
|
|
|
|
|
|
|
|
— |
|
||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
ADS Options |
|
|
|
|
|
|
|
|
|
|||
NED Plan |
|
|
|
|
|
— |
|
|
|
— |
|
|
LTIP |
|
|
|
|
|
|
|
— |
|
|||
|
|
|
|
|
|
|
|
|
— |
|
||
F-23
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
As of June 30, 2023,
Note 17. Current Assets – Cash and Cash Equivalents
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
Cash at bank and in hand |
|
|
|
|
|
|
||
Short-term deposits |
|
|
|
|
|
|
||
Total cash and cash equivalents |
|
|
|
|
|
|
||
Cash at bank earns interest at floating rates based on daily bank deposit rates. The carrying amounts of cash and cash equivalents represent fair value.
Short term‑deposits are with
Note 18. Current Assets - Receivables
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
Interest receivable |
|
|
|
|
|
|
||
GST receivable1 |
|
|
|
|
|
|
||
Other receivable1 |
|
|
|
|
|
|
||
Total current receivables |
|
|
|
|
|
|
||
1
Note 19. Current Assets - Prepayments
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
R&D Contract Research Organization |
|
|
|
|
|
|
||
Insurance |
|
|
|
|
|
|
||
Other prepayments |
|
|
|
|
|
|
||
Total current prepayments |
|
|
|
|
|
|
||
The R&D Contract Research Organization prepayment consists of prepayments on the Phase 3 clinical trial for OPT‑302 in order to secure sites across the world and start patient recruitment. These prepayments covered the initial startup of the Phase 3 clinical trials and other key milestones and are expected to be consumed within the next 12 months. The insurance amount relates to specific Phase 3 clinical trial insurance in place for various sites around the world covering periods to 2024. The non‑current portion of the prepayments are recorded as non‑current assets. Refer Note 21.
F-24
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 20. Non-Current Assets - Right of Use Assets
The Group has a lease contract for its head office premises in Melbourne, Australia, which commenced on 15 July 2022. The agreement does not contain any extension options.
|
|
|
|
|
|
|
||
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
US$ |
|
||||
Right-of-Use Asset Cost |
|
|
|
|
|
|
||
Opening balance as at July 1 |
|
|
|
|
|
|
||
Additions |
|
|
|
|
|
— |
|
|
Exchange on translations |
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
||
Right-of-Use-Asset Depreciation |
|
|
|
|
|
|
||
Opening balance as at July 1 |
|
|
( |
) |
|
|
( |
) |
Charge to the period |
|
|
( |
) |
|
|
( |
) |
Exchange on translation |
|
|
— |
|
|
|
— |
|
|
|
|
( |
) |
|
|
( |
) |
Net carrying amount at June 30 |
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
US$ |
|
|
US$ |
|
|||||
Amounts recognized in profit or loss: |
|
|
|
|
|
|
|
|
|
|||
Depreciation expense on right-of-use asset |
|
|
|
|
|
|
|
|
|
|||
Lease finance costs |
|
|
|
|
|
|
|
|
|
|||
Expense relating to leases of low value assets |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
The Group did not have any short-term leases during the year ended June 30, 2023.
Note 21. Non-Current Assets - Prepayments
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
Insurance |
|
|
|
|
|
|
||
Total non-current prepayments |
|
|
|
|
|
|
||
The non‑current prepayment amount relates to specific Phase 3 Clinical trial insurance in place for various sites around the world covering periods to 2024.
Note 22. Current Liabilities – Payables
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
Accounts Payable (unsecured)1 |
|
|
|
|
|
|
||
Payroll related tax liability |
|
|
|
|
|
|
||
Total current payables |
|
|
|
|
|
|
||
1 Accounts Payable are non‑interest bearing and are normally settled on
Includes an amount of US$
F-25
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 23. Current Liabilities - Provisions
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
Annual leave |
|
|
|
|
|
|
||
Long service leave |
|
|
|
|
|
|
||
Total current provisions |
|
|
|
|
|
|
||
Note 24. Lease Liabilities
Lease liabilities are as indicated below.
At the commencement date of the lease of its office premises, the Group recognizes lease liabilities measured at the present value of lease payments to be made over the lease term ending on
|
|
|
|
|
|
|
||
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
US$ |
|
||||
Carrying amount at July 1 |
|
|
|
|
|
|
||
New lease |
|
|
|
|
|
— |
|
|
Payments |
|
|
( |
) |
|
|
( |
) |
Carrying amount at June 30 |
|
|
|
|
|
— |
|
|
Maturity analysis: |
|
|
|
|
|
|
||
Year 1 |
|
|
|
|
|
— |
|
|
Year 2 |
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
— |
|
|
Less: unearned interest |
|
|
( |
) |
|
|
— |
|
|
|
|
|
|
|
— |
|
|
Analyzed into: |
|
|
|
|
|
|
||
Current portion |
|
|
|
|
|
— |
|
|
Non-current portion |
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
— |
|
|
Note 25. Non-Current Liabilities - Financial Liabilities
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
Carrying amount at July 1 |
|
|
— |
|
|
|
— |
|
Funding at fair value |
|
|
|
|
|
— |
|
|
Interest expense on DFA |
|
|
|
|
|
|
||
Fair value adjustment gain on DFA |
|
|
( |
) |
|
|
— |
|
Total financial liabilities |
|
|
|
|
|
— |
|
|
Pursuant to the DFA, Launch Tx has committed to provide Opthea US$
In return, Opthea will pay to Launch Tx (1) upon the first to occur of regulatory approval of Sozinibercept (OPT-302) for the treatment of wet AMD in the United States, United Kingdom or European Union (“Regulatory Approval”),
F-26
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
fixed payments equal to a total of approximately two times the funding provided, consisting of seven payments, with the first payment due shortly after Regulatory Approval and the remaining six annual payments payable over a six-year period thereafter, and (2) variable payments equal to
In certain instances, upon the termination of the Funding Agreement, we will be obligated to pay Investor several multiples of the amounts paid to us under the Funding Agreement. The Group remains in compliance with the DFA and no such instances has occurred.
The Group evaluated the Financing Agreement and determined it to be a research and development funding arrangement with the characteristics of a debt instrument, as the transfer of financial risk to Launch Tx was not considered substantive and genuine. Accordingly, the Group has recorded payments received under the Financing Agreement as part of a development financing liability in its consolidated balance sheets. The Group accounts for the overall development financing liability at amortized cost based on the estimated timing of regulatory approval and attainment of certain sales milestones and the contractual success fee payments expected to be due therefrom, as discounted using an imputed interest rate. The development financing liability will be accreted as interest expense to its expected future repayment amount over the expected life of the agreement using the effective interest rate method. Certain legal and financial advisory fees incurred specifically to complete the Financing Agreement were capitalized and recorded as a reduction to the carrying amount of the development financing liability and will also be amortized to interest expense using the effective interest method.
Pursuant to the Financing Agreement, the Opthea granted Launch Tx a security interest in all its assets (other than intellectual property not related to Sozinibercept (OPT-302)), provided that the Group is permitted to incur certain indebtedness. The security interest will terminate when the Group has paid Launch Tx the funding provided or upon certain terminations of the Financing Agreement.
There are several factors that could affect the estimated timing of regulatory approval and attainment of sales milestones, some of which are not entirely within the Group’s control. Therefore, at each reporting date, the Group reassesses the estimated timing of regulatory approval and attainment of sales milestones and the expected contractual success fee payments due therefrom. If the timing and/or amount of such expected payments is materially different than original estimates, the Group will prospectively adjust the accretion of the development financing liability and the imputed interest rate.
As of June 30, 2023, the development financing liability was classified as a long-term liability, as the Group expects the related repayments to take place between
Note 26. Non-Current Liabilities - Provisions
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
Long service leave |
|
|
|
|
|
|
||
Total non-current provisions |
|
|
|
|
|
|
||
F-27
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 27. Contributed Equity
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
|
June 30, 2021 |
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
(a) Ordinary shares |
|
|
|
|
|
|
|
|
|
|||
Issued and fully paid at June 30 |
|
|
|
|
|
|
|
|
|
|||
Movement in ordinary shares: |
|
|
|
|
|
|
|
|
|
|||
Opening balance |
|
|
|
|
|
|
|
|
|
|||
Issue of shares on exercise of options granted under the LTIP |
|
|
|
|
|
|
|
|
|
|||
Issue of shares on Nasdaq listing net of issuance cost $ |
|
|
— |
|
|
|
— |
|
|
|
|
|
Issue of shares on exercise of pre-funded warrants net of issuance cost $ |
|
|
— |
|
|
|
— |
|
|
|
|
|
Issue of shares net of issuance costs $ |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|||
Ordinary shares on issue: |
|
No: |
|
|
No: |
|
|
No: |
|
|||
Opening balance |
|
|
|
|
|
|
|
|
|
|||
Issue of shares on exercise of options granted under the LTIP |
|
|
|
|
|
|
|
|
|
|||
Issue of shares on Nasdaq listing |
|
|
— |
|
|
|
— |
|
|
|
|
|
Issue of share on exercise of pre-funded warrants |
|
|
— |
|
|
|
— |
|
|
|
|
|
Issue of shares on ASX |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|||
Fully paid ordinary shares have
Issued capital at June 30, 2023 amounted to US$
During the year ended June 30, 2023 the Company issued
During the year ended June 30, 2021 the Company issued
At June 30, 2023, the company had
At June 30, 2022, the company had
At June 30, 2021, the company had
Options granted to directors and employees.
The company has
The company granted
F-28
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
of $
The company granted
The company granted
(c) Capital management
The Group is not subject to any externally imposed capital requirements. When managing share capital, management’s objective is to ensure the entity continues as a going concern as well as to provide benefits to shareholders and for other stakeholders. In order to maintain or achieve an appropriate capital structure, the Company may issue new shares or reduce its share capital, subject to the provisions of the Company’s constitution. The Group only commits to significant R&D expenditure when this is fully funded either by existing funds or further equity raises.
Note 28. Accumulated Losses and Reserves
|
|
2023 |
|
|
2022 |
|
||
|
|
|
|
|
|
|
||
|
|
US$ |
|
|
US$ |
|
||
(a) Movements in accumulated losses were as follows: |
|
|
|
|
|
|
||
Balance at July 1 |
|
|
( |
) |
|
|
( |
) |
Net loss for the period |
|
|
( |
) |
|
|
( |
) |
Balance at June 30 |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
||
(b) Reserves |
|
|
|
|
|
|
||
Fair value of Investments reserve (i) |
|
|
|
|
|
|
||
Share-based payments reserve (ii) |
|
|
|
|
|
|
||
Foreign translation reserve (iii) |
|
|
|
|
|
|
||
Total reserves |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
||
(i) Movement in fair value of investments reserve: |
|
|
|
|
|
|
||
Opening balance |
|
|
|
|
|
|
||
Fair value on gains on investments in financial assets |
|
|
— |
|
|
|
— |
|
Exchange on translation |
|
|
— |
|
|
|
— |
|
Closing balance |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
||
(ii) Movement in share-based payments reserve: |
|
|
|
|
|
|
||
Opening balance |
|
|
|
|
|
|
||
Share-based payments expense |
|
|
|
|
|
|
||
Exercise of options |
|
|
( |
) |
|
|
( |
) |
Exchange on translation |
|
|
— |
|
|
|
— |
|
Closing balance |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
||
(iii) Movement in foreign translation reserve: |
|
|
|
|
|
|
||
Opening balance |
|
|
|
|
|
|
||
(Gains)/loss on translation |
|
|
— |
|
|
|
— |
|
Closing balance |
|
|
|
|
|
|
||
F-29
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
(c) Nature and purpose of reserves
Fair value of investments reserve
This reserve records fair value changes on listed investments. As at June 30, 2023 no remaining investments are held by the Group. Management's accounting policy is to not reclassify the realized fair value to accumulated loss upon disposal.
Share‑based payment reserve
This reserve is used to record the value of equity benefits provided to executives and employees as part of their remuneration.
Foreign currency translation reserve
The reserve records the value of foreign currency movements on the initial translation of financial statements from A$ to US$ that was completed in 2021.
Note 29. Financial Risk Management Objectives and Policies
The Group’s principal financial assets comprise cash, receivables, short‑term deposits and investments in listed shares.
The Group manages its exposure to key financial risks, including interest rate and currency risk in accordance with the Group’s financial risk management practices. The objective is to support the delivery of the Group’s financial targets whilst protecting future financial security.
The Group’s other various financial assets and liabilities, such as receivables and payables, arise directly from its operations. The main risks arising from the Group’s financial assets and liabilities are interest rate risk, foreign currency risk, equity securities price risk and liquidity risk.
The Group uses different methods to measure and manage different types of risks to which it is exposed. These include monitoring levels of exposure to interest rate and foreign exchange risk and assessments of market forecasts for interest rates and foreign exchange rates. Liquidity risk is monitored through future rolling cash flow forecasts.
The board reviews and agrees policies for managing each of these risks as summarized below.
Risk exposures and responses
The Group has investigated the main financial risk areas which could impact on its financial assets and determined the impact on post tax (losses) or profits for a range of sensitivities. These can be seen in the post-tax (loss)/profit impact for each risk area.
For each risk area, the equity impact relates solely to reserve movements and excludes movements in accumulated losses as the impact of these can be seen within the post-tax (loss)/profit impact.
The Group’s exposure to market interest rates relates primarily to the short‑term deposits. The deposits are held with two of Australia’s largest banks.
The objective of managing interest rate risk is to minimize the Group’s exposure to fluctuations in interest rates that might impact its interest income and cash flow. To manage interest rate risk, the Group invests the majority of its cash in short‑term deposits for varying periods of between
F-30
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
The Group currently has borrowings under the DFA with Ocelot (2022:
At June 30, 2023, 2022 and 2021, if interest rates moved, with all variables held constant, post tax (loss)/profit and equity would have been affected as illustrated in the following table:
|
|
Post tax (loss)/profit impact |
|
|||||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Judgments of reasonably possible movements |
|
|
|
|
|
|
|
|
|
|||
+0.50% (50 basis points) (2022:+ |
|
|
|
|
|
|
|
|
|
|||
-0.50% (50 basis points) (2022:-0.50%) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
The post-tax figures include an offset for unrecognized tax losses (bringing the tax effect to
Significant assumptions used in the interest rate sensitivity analysis include:
As a result of services provided by non‑related entities in Australia, Canada, United Kingdom and Europe, part of the Group’s monetary assets and liabilities are affected by movements in the exchange rate.
The Group does not enter into any hedging transactions.
At the reporting date, the Group has the following exposure to foreign currencies. :
|
|
Consolidated |
|
|||||||||||||
2023 |
|
AUD |
|
|
EURO |
|
|
GBP |
|
|
CAD |
|
||||
Financial assets |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Cash |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Receivables |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Financial liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Payables |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Other financial liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Net exposure |
|
|
|
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
Consolidated |
|
|||||||||||||
2022 |
|
AUD |
|
|
EURO |
|
|
GBP |
|
|
CAD |
|
||||
Financial assets |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Cash |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Receivables |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Financial liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Payables |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Other financial liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Net exposure |
|
|
|
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
The following sensitivity is based on the foreign currency risk exposures in existence at June 30, 2023 and 2022.
At June 30, 2023, 2022 and 2021, had the United States dollar moved with all other variables held constant, post tax (loss) profit and equity would have been affected as illustrated in the table below:
F-31
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
|
|
Post tax (loss)/profit impact |
|
|||||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Judgments of reasonably possible movements |
|
|
|
|
|
|
|
|
|
|||
Consolidated |
|
|
|
|
|
|
|
|
|
|||
AUD/USD +10% (2022:+ |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
AUD/USD -10% (2022:-10%) |
|
|
|
|
|
|
|
|
|
|||
The reasonably possible movements at June 30, 2023 are higher than at June 30, 2022 and higher than June 30, 2021 due mainly to net exposure to the Australian dollar due to cash at bank deposits. There was minimum or insignificant exposure to the GBP, Euro and CAD during the current financial year.
Significant assumptions used in the foreign currency exposure sensitivity analysis include:
Credit risk is associated with those financial assets of the Group which comprise cash and cash equivalents, receivables and listed investments. The Group’s exposure to credit risk arises from default of the counter party, with a maximum exposure equal to the carrying amount of these investments. Credit risk is considered minimal as the Group transacts with reputable recognized Australian banks.
Liquidity risk arises from the financial liabilities of the Group and the Group’s subsequent ability to meet their obligations to repay their financial liabilities as and when they fall due.
The Group’s objective is to maintain an appropriate cash asset balance to fund its operations.
Note 30. Related Party Disclosures
The consolidated financial statements include the financial statements of Opthea Limited and its subsidiaries in the following table:
|
|
Parent entity % equity interest |
|
|||||||||
|
|
June 30, 2023 |
|
|
June 30, 2022 |
|
|
June 30, 2021 |
|
|||
|
|
% |
|
|
% |
|
|
% |
|
|||
Vegenics Pty Ltd1 |
|
|
|
|
|
|
|
|
|
|||
Opthea US Inc2 |
|
|
|
|
|
|
|
|
|
|||
F-32
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
(1) Opthea Limited is the ultimate parent entity. Vegenics Pty Ltd is incorporated in Australia and has the same financial year as Opthea Limited.
(2) Opthea Limited is the ultimate parent entity. Opthea US Inc was incorporated in the United States in May 2021 and has the same financial year as Opthea Limited.
Balances and transactions between the Company and its subsidiaries, a related party of the Company, have been eliminated on consolidation and are not disclosed in this note. Transactions between the Group and its associates are disclosed below:
With the appointment of Anshul Thakral (who is the CEO of Launch and Operation Executive of Carlyle) on June 7, 2023, as a Director of Opthea resulting in Launch, Ocelot and Carlyle being related parties of Opthea.
F-33
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Trading transactions
During the year, group entities entered into the following transactions with related parties who are not members of the Group.
|
|
Consolidated |
|
|||||||||
|
|
Purchase of Service |
|
|||||||||
|
|
|
2023 |
|
|
|
2022 |
|
|
|
2021 |
|
Launch Tx - Ocelot |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Launch |
|
|
|
|
|
— |
|
|
|
— |
|
|
Purchase of services assisting Opthea with the management and oversight of trials under the Service Agreement with Launch Tx.
|
|
Consolidated |
|
|||||||||
|
|
Amounts owed to related parties |
|
|||||||||
|
|
|
2023 |
|
|
|
2022 |
|
|
|
2021 |
|
Launch Tx - Ocelot |
|
|
|
|
|
— |
|
|
|
— |
|
|
Launch |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Amounts owed to Ocelot relate to the Development Funding agreement and carry an effective rate of
F-34
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 31. Commitments
The Group has entered into research and development contracts and intellectual property license agreements with various third parties in respect of services for the Phase 3 wet AMD clinical trial and the clinical grade manufacture of OPT‑302.
|
|
|
|
|
|
|
|
|
|
|||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Within one year |
|
|
|
|
|
|
|
|
|
|||
After one year but not more than five years |
|
|
|
|
|
|
|
|
|
|||
After more than five years |
|
|
|
|
|
|
|
|
— |
|
||
|
|
|
|
|
|
|
|
|
|
|||
Currently, the biggest research contract has a
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Within one year |
|
|
|
|
|
|
|
|
— |
|
||
After one year but not more than five years |
|
|
— |
|
|
|
— |
|
|
|
— |
|
After more than five years |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
— |
|
||
Currently, the biggest contract has a
Note 32. Contingencies
The Group is party to various research agreements with respect to which a commitment to pay is contingent on the achievement of research milestones. Assuming all milestones are achieved within the time‑frames stipulated in the contracts, those which could become payable in less than one year total US$
Under these license/collaboration agreements, payments are to be made only if certain research and clinical development milestones are achieved and royalties may become payable on any eventual sales of products developed under these agreements.
The group had a bank guarantee outstanding at June 30, 2023 in respect of a rental deposit for its office premises of US$
F-35
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 33. Cash Flow Statements Reconciliation
|
|
|
|||||||||||
|
|
2023 |
|
|
2022 |
|
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
Restated |
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
||||
(a) Reconciliation to cash at the end of the year |
|
|
|
|
|
|
|
|
|
|
|||
Cash at bank and in hand (note 17) |
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|||
(b) Reconciliation of net loss after tax to net cash flows from operations |
|
|
|
|
|
|
|
|
|
|
|||
Net loss for the year |
|
|
( |
) |
|
|
( |
) |
|
|
|
( |
) |
Adjustments for: |
|
|
|
|
|
|
|
|
|
|
|||
Income tax benefit recognized in profit or loss |
|
|
( |
) |
|
|
( |
) |
|
|
|
( |
) |
Net loss of disposal of non-current assets |
|
|
— |
|
|
|
|
|
|
|
— |
|
|
Depreciation of non-current assets |
|
|
|
|
|
|
|
|
|
|
|||
Depreciation of right-of-use assets |
|
|
|
|
|
|
|
|
|
|
|||
Interest expense on DFA |
|
|
|
|
|
— |
|
|
|
|
— |
|
|
Fair value adjustment gain on DFA |
|
|
( |
) |
|
|
— |
|
|
|
|
— |
|
Share-based payments |
|
|
|
|
|
|
|
|
|
|
|||
Net exchange differences |
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|||
Changes in: |
|
|
|
|
|
|
|
|
|
|
|||
Payables |
|
|
|
|
|
|
|
|
|
( |
) |
||
Receivables |
|
|
|
|
|
|
|
|
|
( |
) |
||
Prepayments |
|
|
|
|
|
|
|
|
|
( |
) |
||
Provisions |
|
|
|
|
|
|
|
|
|
|
|||
Net cash flows used in operating activities before tax |
|
|
( |
) |
|
|
( |
) |
|
|
|
( |
) |
R&D tax incentive received |
|
|
|
|
|
|
|
|
|
|
|||
Net cash flows used in operating activities |
|
|
( |
) |
|
|
( |
) |
|
|
|
( |
) |
F-36
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 34. Key Management Personnel
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Short-term employee benefits |
|
|
|
|
|
|
|
|
|
|||
Post-employment benefits |
|
|
|
|
|
|
|
|
|
|||
Share-based payments expense |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
There were
Note 35. Share-Based Payments
The expense recognized for share‑based payments during the year is shown in the table below:
|
|
|
|
|
|
|
||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||
|
|
|
|
|
|
|
|
|
|
|||
|
|
US$ |
|
|
US$ |
|
|
US$ |
|
|||
Expense arising from equity-settled share-based payment |
|
|
|
|
|
|
|
|
|
|||
Director and employee services received |
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|||
During the 2015 financial year, the Group introduced an ownership‑based compensation scheme for non‑executive directors, executives and senior employees, the Long-Term Incentive Plan (LTIP) and Non‑Executive Directors Share and Option Plan (NED Plan). In accordance with the terms of the plans, as approved by shareholders at the 2014 annual general meeting, eligible non‑executive directors, executives and senior employees with the Group may be granted options to purchase ordinary shares.
Each employee share option converts into
The number of options granted is subject to approval by the board and rewards executives and senior employees to the extent of the Group’s and the individual’s achievement judged against both qualitative and quantitative criteria as determined by the board on a case by case basis.
F-37
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
The vesting condition of options granted under the LTIP and NED Plan is continuous service.
Options/Rights services |
|
Grant date |
|
Grant date |
|
|
Exercise |
|
|
Expiry date |
|
Vesting date |
||
LTIP - director FY2019 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2018 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2019 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|
||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2023 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2023 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2023 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2023 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2023 |
|
|
|
|
|
|
|
|
|
|||||
LTIP - employees FY2023 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2016 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2019 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|||||
There has been no alteration of the terms and conditions of the above share‑based payment arrangements since the grant date.
F-38
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Where relevant, the expected life used in the model has been adjusted based on management’s best estimate for the effects of non‑transferability, exercise restrictions (including the probability of meeting conditions attached to the option), and behavioral considerations. Expected volatility is based on the historical share price volatility over the past
Options/Rights services |
|
Grant |
|
|
Exercise |
|
|
Fair |
|
|
Expected |
|
|
Option life |
|
Dividend |
|
|
Risk |
|
|
Model |
||||||
LTIP - director FY2019 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employees FY2018 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employees FY2019 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employees FY2022 |
|
|
|
|
nil |
|
|
|
|
|
na |
|
|
|
|
% |
|
n/a |
|
|
||||||||
LTIP - employees FY2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employees FY2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employees FY2023 |
|
|
|
|
nil |
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employees FY2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
NED Plan FY2016 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
NED Plan FY2019 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
NED Play FY2021 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
NED Plan FY2021 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
NED Plan FY2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
Fair value of ADS options granted
Where relevant, the expected life used in the model has been adjusted based on management’s best estimate for the effects of non‑transferability, exercise restrictions (including the probability of meeting conditions attached to the option), and behavioral considerations. Expected volatility is based on the historical share price volatility over the past
ADS Options |
|
Grant |
|
|
Exercise |
|
|
Fair |
|
|
Expected |
|
|
ADS Option life |
|
Dividend |
|
|
Risk |
|
|
Model |
||||||
LTIP - employee 2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2022 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
LTIP - employee 2023 |
|
|
|
|
|
|
|
|
|
|
|
% |
|
|
|
% |
|
|
% |
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
F-39
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
The following reconciles the share/rights options outstanding at the beginning and end of the year:
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||||||||||||||
|
|
Number of |
|
|
Weighted |
|
|
Number of |
|
|
Weighted |
|
|
Number of |
|
|
Weighted |
|
||||||
Balance at beginning of year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Granted during the year: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
To employees and directors under the LTIP and NED Plan |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
Exercised during the year |
|
|
( |
) |
|
|
|
|
|
( |
) |
|
|
|
|
|
( |
) |
|
|
|
|||
Expired during the year |
|
|
( |
) |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Balance at end of the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Exercisable at end of year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
The share options outstanding at the end of the year had a weighted average exercise price of US $
Movements in ADS options during the year
The following reconciles the ADS options outstanding at the beginning and end of the year:
|
|
2023 |
|
|
2022 |
|
|
2021 |
|
|||||||||||||||
|
|
Number of |
|
|
Weighted |
|
|
Number of |
|
|
Weighted |
|
|
Number of |
|
|
Weighted |
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Balance at beginning of year |
|
|
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
||
Granted during the year: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
To employees and directors under the LTIP and NED Plan |
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
— |
|
||||||
Exercised during the year |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Expired during the year |
|
|
( |
) |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Balance at end of the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
— |
|
||||
Exercisable at end of year |
|
|
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
||
F-40
OPTHEA LIMITED
Notes to Consolidated Financial Statements — Continued
Note 36. Events After the Balance Sheet Date
On August 24 , 2023, Opthea announced a A$
On August 28, 2023 Mr. Lawrence Gozlan, a director of the Company, and the Company have entered into a Consultancy Agreement of up to US$
Besides the above-mentioned subsequent events, no matters or circumstances have arisen since the end of the reporting period, which significantly affected, or may significantly affect, the operations of the Group, the results of those operations, or the state of affairs of the Group in future financial years.
F-41