UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
(Mark One) | |
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
For the fiscal year ended | |
OR | |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
For the transition period from to | |
Commission file number:
(Exact name of registrant as specified in its charter)
|
|
|
|
Registrant’s telephone number, including area code: (
Securities registered pursuant to Section 12(b) of the Exchange Act:
Title of each class |
| Trading Symbol(s) | Name of each exchange on which registered |
Securities registered pursuant to Section 12(g) of the Exchange Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes ◻
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Accelerated filer ◻ | ||
Non-accelerated filer ◻ | Smaller reporting company | ||
Emerging growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes
As of the last business day of the registrant’s most recently completed second fiscal quarter (June 30, 2023), the aggregate market value of the voting and non-voting common equity held by non-affiliates of the registrant, based on the last reported sales price for the registrant’s common stock, par value $0.001 per share, on The Nasdaq Global Market on such date, was approximately $
As of February 26, 2024, the registrant had
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s proxy statement for its 2024 Annual Stockholders’ Meeting, which the registrant intends to file with the Securities and Exchange Commission pursuant to Regulation 14A within 120 days after the end of the registrant’s fiscal year ended December 31, 2023, are incorporated by reference into Part III of this Annual Report on Form 10-K.
QUANTERIX CORPORATION
INDEX TO FORM 10-K
Note Regarding Forward-Looking Statements
i
Unless the context otherwise requires, the terms “Quanterix,” the “Company,” “we,” “it,” “us,” and “our” in this Annual Report on Form 10 K refer to Quanterix Corporation and its consolidated subsidiaries.
Note Regarding Forward-Looking Statements
This Annual Report on Form 10-K contains forward-looking statements (within the meaning of the U.S. Private Securities Litigation Reform Act of 1995) that involve risks and uncertainties. All statements other than statements of historical facts contained in this Annual Report on Form 10-K are forward-looking statements. In some cases, forward-looking statements can be identified by words such as “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “seek,” “should,” “target,” “will,” “would,” or the negative of these words or other comparable terminology. These forward-looking statements include, but are not limited to, statements about:
| ● | the implementation of our business model and strategic plans for our business, products and services, especially following the restructuring and strategic realignment we commenced in the third quarter of 2022; |
| ● | the potential size of the markets and fields addressable by our Simoa technology platforms; |
| ● | the commercialization and adoption of our existing products and services and the success of our new product and service offerings; |
| ● | our estimates regarding expenses, future revenues, capital requirements, and our needs for additional financing; |
| ● | the ability of our Simoa technology’s sensitivity to improve existing diagnostics and to enable the development of new diagnostic tests and tools; |
| ● | the potential of our Simoa technology in the field of screening/diagnostic tests linked to therapeutic drugs and adoption by healthcare professionals; |
| ● | the impact of our Simoa technology on proteomic research; |
| ● | our ability to realize the intended benefits of our assay redevelopment program; |
| ● | our ability to retain and expand our customer base and achieve sufficient market acceptance of our products; |
| ● | the ability of our contract manufacturers to reliably and consistently manufacture and supply our Simoa instruments; |
| ● | the usefulness of the data generated by our Simoa technology in the life science research and diagnostics fields; |
| ● | our ability to successfully penetrate the diagnostics market; and |
| ● | our future financial performance. |
These forward-looking statements are subject to a number of risks, uncertainties, and assumptions, including those further described in the section titled “Part I, Item 1A, Risk Factors” and elsewhere in this Annual Report on Form 10-K. Moreover, we operate in a very competitive and rapidly changing environment, and new risks emerge from time to time. It is not possible for us to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties, and assumptions, the forward-looking events and circumstances discussed in this Annual Report on Form 10-K may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements.
Readers should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in any forward-looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance, or events and circumstances reflected in forward-looking statements will be achieved or occur. We undertake no obligation to update publicly any forward-looking statements for any reason after the date of this Annual Report on Form 10-K to conform these statements to new information, actual results, or to changes in our expectations, except as required by law.
ii
Readers should read this Annual Report on Form 10-K and any documents referenced herein that we have filed with the Securities and Exchange Commission (“SEC”) as exhibits to this Annual Report on Form 10-K with the understanding that our actual future results, levels of activity, performance, and events and circumstances may be materially different from what we expect.
This Annual Report on Form 10-K includes statistical and other industry, and market data that we obtained from industry publications and research, surveys, and studies conducted by third parties. Industry publications and third-party research, surveys, and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. Such data involves a number of assumptions and limitations and contains projections, and estimates of the future performance of the markets in which we operate and intend to operate that are subject to a high degree of uncertainty. We caution not to give undue weight to such projections, assumptions and estimates.
Service Marks, Trademarks, and Trade Names
“Quanterix,” “Simoa,” “Simoa HD-X,” “Simoa HD-1,” “SR-X,” “SP-X”, “HD-X”, “LucentAD,” “Lucent Diagnostics,” and our logo are our trademarks. All other service marks, trademarks, and trade names appearing in this Annual Report on Form 10-K are the property of their respective owners. We do not intend our use or display of other companies’ trade names, trademarks, or service marks to imply a relationship with, or endorsement or sponsorship of us, by these other companies.
iii
PART I
ITEM 1. BUSINESS
Overview
We are a life sciences company that has developed next-generation, ultra-sensitive digital immunoassay platforms that advance life sciences research and diagnostics. Our platforms are based on our proprietary digital “Simoa” detection technology and enable customers to reliably detect protein biomarkers at ultra-low concentrations in blood, serum and other fluids that, in many cases, are undetectable using conventional, analog immunoassay technologies. The ability of our Simoa platforms to detect proteins in the femtomolar range is enabling the development of novel therapies and diagnostics and has the potential to facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis, and, ultimately, prevention. Our Simoa platforms have achieved significant scientific validation and commercial adoption, and our Simoa technology has been cited in more than 2,700 scientific publications in areas of high unmet medical need and research interest such as neurology, oncology, cardiology, infectious disease, and inflammation.
Industry Background
Proteins are versatile macromolecules and serve critical functions in nearly all biological processes. Proteins are analytes that are highly relevant physiologically, providing real-time pictures of disease, and researchers and clinicians rely extensively on protein biomarkers for use as research and clinical tools. However, normal physiological levels of many proteins are not detectable in easily accessible blood samples using conventional, analog immunoassay technologies, and many of these technologies can only detect proteins once they have reached levels that reflect more advanced disease or injury. For many other low abundance proteins, these technologies cannot detect proteins even at disease- or injury-elevated levels.
Enzyme-linked immunosorbent assay (“ELISA”) technology has been the most widely used method of sensitive detection of proteins for more than 50 years. In simple terms, ELISA involves using a plate coated with an antibody that binds to the target molecule. If the target is present in the sample, it attaches to the plate via the antibody. Then, a second molecule, often an enzyme-linked antibody, is added, binding to the target. The enzyme produces a detectable signal (like fluorescence or a color change) when a specific substrate is added, indicating the presence and quantity of the target molecule in the sample. Although ELISA is widely used in medical diagnostics and research, it has very significant limitations, including:
| ● | limited sensitivity due to dilution of colored or fluorescent product molecules in the large volume of liquid in traditional-sized wells; |
| ● | narrow dynamic range (i.e., the range of concentration of proteins being detected), that may require sample dilution, diluting molecules and increasing sample volume requiring additional enzymes to reach detection limit; |
| ● | low detection limit of readers restricts sensitivity and ability to detect low abundance proteins, particularly when proteins are at normal physiological levels; and |
| ● | limited success in increasing sensitivity of detection due to procedural complexity and length. |
Our Simoa Technology
Our Simoa bead-based and planar array technologies are based on traditional ELISA technology but significantly advance conventional ELISA technology and are capable of substantially greater protein detection sensitivity. We believe that our Simoa platforms are among the most sensitive commercially available multiplex protein detection platforms.
1
Simoa Bead-Based Technology
Simoa bead-based digital immunoassays utilize the basic principles of conventional bead-based sandwich ELISA. However, unlike ELISA, which runs the enzyme-substrate reaction on all molecules in one well, Simoa bead-based reactions are run on individual molecules in tiny microwells, 40 trillionths of a milliliter that are 2.5 billion times smaller than traditional ELISA wells. Traditional ELISA analog measurements increase in intensity only as the concentration of a sample increases. Simoa bead-based digital technology measurements, however, are independent of sample concentration intensity and rely on a binary signal/no signal readout, enabling detection sensitivity in the femtomolar range compared with nanomolar and picomolar levels of detection in conventional ELISA.
Our Simoa bead-based platform is highly flexible, designed to enable practical high-sensitivity protein analysis for academic researchers looking at novel proteins all the way through to high throughput analysis performed by large biopharmaceutical organizations.
Simoa Planar Array Technology
Simoa planar array immunoassays utilize the basic principles of conventional microplate-based sandwich ELISA. However, unlike ELISA, which runs the enzyme-substrate reaction on all molecules coating the entire bottom surface in one well, Simoa planar array reactions are run on spatially segregated micro-spots within the bottom of microtiter plate wells that concentrate the signal to a surface area 1,000 times smaller than a traditional ELISA. The small spot size and spatial segregation of each spot enables multiplexing up to 12 different assays within a single sample well.
Our Simoa planar array platform is highly flexible, designed to enable practical high-sensitivity multiplex protein analysis for drug discovery and development applications as well as translational biomarker research.
Restructuring Plan
In August 2022, we announced a restructuring and strategic realignment plan (the “Restructuring Plan”). The Restructuring Plan included the elimination of 119 positions across the Company in 2022 and other cost-saving measures that were substantially completed in 2022. The Restructuring Plan also included an assay redevelopment program with the objective of improving our ability to manufacture and deliver high-quality assays at scale. This six-quarter operational program was substantially completed in the fourth quarter of 2023, and we have now launched five new Simoa Advantage PLUS assays. The Simoa Advantage PLUS assays have been developed using improved protocols, by leveraging manufacturing efficiencies and reagent improvements to provide more consistent results and improved lot-to-lot consistency, and through enabling production of larger lot sizes with extended shelf lives. These assays began shipping to customers in the first quarter of 2024. We expect to continue to apply these improved protocols and manufacturing efficiencies to other existing assays and assays that we may develop in the future.
Our Strategy
Our commercial strategy is to pursue the application of our Simoa technology in life science research/pharma and diagnostics. Our strategy to achieve this includes:
| ● | Continue to drive adoption of our Simoa technology in life science research/pharma. We believe our Simoa technology has the potential to significantly expand the life science research market and is uniquely positioned for use by pharmaceutical companies in drug trials as biomarkers are being increasingly used as an adjunct to help increase the potential of regulatory approval. |
| ● | Expand our presence in diagnostics. We believe Simoa technology has the capability to enable the development of a new category of less-invasive diagnostic tests and tools based on blood, serum, saliva and other fluids that could replace current invasive, expensive, and inconvenient diagnostic methods, including spinal tap, diagnostic imaging and biopsy. |
2
| ● | Leverage the growing importance of neurological biomarkers to advance the development of therapeutics and diagnostics for neurodegenerative conditions. The importance of neurological biomarkers, such as neurofilament light chain (NfL), p-Tau 181, p-Tau 217 and glial fibrillary acidic protein (GFAP), has increased significantly in recent years, and our ultra-sensitive Simoa platforms have allowed research of neurological disorders, previously limited primarily to cerebrospinal fluid (CSF), to expand significantly. |
| ● | Expand further into indications beyond neurology. Although we believe that our ultra-sensitive Simoa platforms are particularly well suited for neurology indications, we also believe that the ability of our Simoa technology to detect and quantify low abundance proteins is a distinct advantage in detecting disease earlier in other indications, including oncology, infectious disease, inflammation and cardiology, and there are over 800 scientific publications that reference our Simoa technologies in non-neurology indications. |
Our Market Opportunities
Based on industry estimates, the life science research/pharma and diagnostics markets are believed to have an aggregate total addressable market (“TAM”) of approximately $75 billion.
Life Science Research/Pharma
We have targeted the large and growing life science research and pharma market because of the relatively low regulatory and reimbursement risk. We believe our Simoa platforms are well-positioned to capture a significant share of this market because of superior sensitivity, automated workflow capabilities, multiplexing, and the ability to work with a broader range of sample types.
Researchers study proteins to understand the biological basis for disease and how to improve diagnosis and treatment. The proteins detectable by conventional, analog immunoassay technologies represent a mere fraction of the proteins that can be detected by Simoa technology, and we believe that Simoa can inspire a new level of research into these previously undetectable proteins and their role in disease. By substantially lowering the limit of detection of protein biomarkers, our Simoa platforms hold significant potential to expand research into the diseases associated with the thousands of proteins that were previously undetectable, as well as into earlier detection of the proteins currently detectable by other technologies only after they have reached levels that reflect more advanced disease or injury.
In addition, as pharmaceutical companies look for ways to more efficiently and effectively develop and obtain regulatory approval for drugs, use of biomarkers in clinical drug trials is becoming more prevalent. Biomarkers are increasingly being used as clinical endpoints in clinical trials. Using Simoa’s unprecedented sensitivity to measure previously undetectable levels of target biomarkers prior to and following administration of a drug, drug developers can non-invasively and objectively determine whether a drug candidate is having a desired impact on the target biomarker. Drug developers can also use diagnostics to stratify patients and select only those patients for whom a drug is expected to be most effective and safe.
Based on industry estimates, we believe the proteomics life science research/pharma TAM is approximately $20 billion.
Diagnostics
Diagnostics represents a significant commercial opportunity for our Simoa technology. We believe existing biomarker diagnostics can be improved by Simoa’s sensitivity to enable earlier detection of diseases and injuries, and that new diagnostics may be developed using protein biomarkers that are not detectable using conventional, analog immunoassay technologies but are detectable using Simoa technology. We also believe that the ultra-sensitive protein detection provided by our Simoa platforms can enable the development of a new category of non-invasive diagnostic tests and tools based on blood, serum and other fluids that have the potential to replace current more invasive, expensive and inconvenient diagnostic methods, including spinal tap, diagnostic imaging and biopsy.
3
Based on industry estimates, we believe the proteomics diagnostics TAM is approximately $55 billion.
Our Products and Services
Instruments
We currently offer the following three Simoa instruments, which we believe are among the most sensitive multiplex protein detection platforms commercially available today:
HD-X | ||||
| We commercially launched our HD-X instrument in the second half of 2019. The HD-X is an upgraded version of the Simoa HD-1 (our first Simoa instrument, which was launched in January 2014) that was designed to deliver significant productivity and operational efficiency improvements, as well as greater user flexibility. The HD-X is based on our bead-based technology, and assays run on the HD-X are fully automated. By the end of 2023, approximately 82% of the HD installed base were HD-X instruments. | |||
SR-X
| We commercially launched our SR-X instrument in December 2017 as a compact benchtop instrument with a lower price point, more flexible assay preparation, and a wider range of applications. The SR-X utilizes the same Simoa bead-based technology and assay kits as the HD-X. In contrast to the fully automated workflow of the HD-X, the assay incubation and washing steps for the SR-X are performed outside of the instrument using conventional liquid handling methods. The offline sample prep provides additional flexibility to enable researchers to apply Simoa detection in an expanded range of applications. | |||
SP-X
| We commercially launched the SP-X instrument in April 2019. The SP-X uses the Simoa planar array technology developed initially by Aushon Biosystems (Aushon), which we acquired in 2018, for multiplex chemiluminescent immunoassay measurement, which we refined by leveraging our proprietary sophisticated Simoa image analysis and data analysis algorithms to provide sensitivity similar to that found in our Simoa bead-based platform. | |||
Simoa Assays and Consumables
Recurring revenue is derived through the sale of consumables used to run assays on our instruments and from our growing menu of Simoa digital biomarker assays. The current menu of approximately 36 analyte-specific single-plex and multi-plex assay kits for our bead-based instruments includes assays for biomarkers in the areas of neurology, infectious disease, immunology and oncology for both human and mouse samples. The current menu of assay kits for the
4
planar array instrument includes approximately 95 biomarkers ranging from 1-10 analytes per assay in the areas of immunology and oncology research.
In addition to the assays we have developed, both of the Simoa platforms allow ease and flexibility in assay design, enabling our customers to develop their own proprietary in-house assays, called homebrew assays, using our homebrew assay kits. These kits include all components required for customers to run tests using their own antibodies. Our consumables portfolio for our bead-based platform also includes our proprietary Simoa disks that are unique to our bead-based platform, as well as cuvettes and disposable tips.
NfL Antibodies and NfL ELISA Kits
In August 2019, we acquired UmanDiagnostics AB (“Uman”), a company located in Umeå, Sweden, that commercializes proprietary NfL antibodies and NfL ELISA kits. Uman’s NfL antibodies are used by researchers and biopharmaceutical and diagnostics companies to detect NfL to advance the development of therapeutics and diagnostics for certain neurodegenerative conditions. Since we commercially launched the first assay that could reliably measure NfL in blood using Uman’s antibodies and our Simoa technology in 2017, NfL has seen dramatic growth as a neurological biomarker. Through Uman we sell proprietary NfL capture and detection antibodies, as well as two NfL ELISA kits for CSF, one of which is CE-certified in Europe, and one research use only (“RUO”) NfL ELISA kit for serum.
Services
Through our Accelerator Laboratory, which includes a CLIA-certified laboratory, we provide customers a contract research option. The Accelerator Laboratory supports multiple projects and services, including:
| ● | Sample Testing. Utilizing commercially available Simoa kits, we have run large studies for customers with thousands of specimens and small experiments with just a few samples. We have extensive experience testing many different sample types where biomarkers may be present at very low levels. |
| ● | Homebrew Assay Development. Utilizing proprietary or commercially available reagents in combination with our homebrew assay kits, we can rapidly develop a prototype assay exhibiting improved sensitivity compared to traditional ELISA. The Accelerator Laboratory can also be used to screen reagents to identify the optimal assay format or expand prototype efforts for further assay optimization or validation to ultimately deliver the highest level of performance. |
| ● | Custom Assay Development. After identifying the optimal assay and conditions, the Accelerator Laboratory can be used to generate qualified bulk reagents or custom assay kits, providing customer access to validated kits for assays not yet commercially available on the Simoa platform. |
| ● | Lucent Diagnostics. In July 2023, we launched Lucent Diagnostics, our brand for clinical testing services for healthcare providers in the field of neurology, with testing services initially focused on Alzheimer’s disease. The first product offering was LucentAD, a blood test to aid in the evaluation of patients experiencing cognitive symptoms consistent with the early signs of Alzheimer’s disease. Subsequently, we launched an additional blood test for Alzheimer’s disease, LucentAD p-Tau 217, in October 2023. The process for testing involves a healthcare provider drawing a blood sample from an individual, shipping the sample to our Accelerator Laboratory, to analyze the sample and communicating the result back to the provider. To support a streamlined provider workflow, we have also launched a web portal where healthcare providers can order sample collection materials, order a test, track the status of a test and retrieve the test report. |
| ● | LDT Testing. We currently offer three laboratory developed tests (LDTs) through our CLIA-certified laboratory: (1) an LDT to quantitatively measure p-Tau 181 in plasma as an aid in diagnostic evaluation of Alzheimer’s disease; (2) an LDT to quantitatively measure NfL in serum as an aid in the evaluation of individuals for possible neurodegenerative conditions or other causes of neuronal or central nervous system |
5
| damage; and (3) an LDT to quantitatively measure p-Tau 217 in plasma to aid in the diagnosis of Alzheimer’s disease. |
We also generate revenues through extended-warranty and service contracts for our installed base of instruments.
Research and Development
We continue to seek to improve our platform and technology to enable more sensitive detection and measurement of biological molecules. This evaluation includes examining new assay formats and instrumentation improvements and upgrades to increase the performance of our Simoa assays and instruments. We intend to selectively expand our assay menu to extend the scope of applications for our platform to biomarkers of significant interest to the scientific community. We also intend to continue to develop and market new instruments with different and/or improved capabilities in order to further broaden our market reach.
Our Key Focus Areas
We have focused the application of our Simoa technology on areas of high growth and high unmet need and where existing platforms have significant shortcomings that our technology addresses, including neurology and oncology, as well as cardiology, infectious disease, and inflammation.
Neurology
The ability of Simoa technology to detect neurological biomarkers in blood at ultra-low levels, combined with our multiplexing capability, has significantly advanced neurology research, drug development, and diagnostics test development. Prior to the launch of our p-Tau 181 LDT for clinical use in July 2022, the brain was the only organ in the body for which there was not a blood-based diagnostic test. The challenge with developing blood-based tests for the brain is that the blood-brain barrier, which is formed by endothelial cells lining the cerebral microvasculature, is very tight and severely restricts the movement of proteins and other substances between these endothelial cells and into blood circulation. Accordingly, diagnosis of brain disease and injury has traditionally required either brain imaging or a spinal tap to collect CSF. The sensitivity of Simoa technology has enabled researchers to discover that extremely small amounts of critical neural biomarkers diffuse through the blood brain barrier and are released into the blood during injury and in connection with many neurodegenerative brain diseases. However, the concentrations of many of these neural biomarkers in the blood can be so low that they are difficult to detect by conventional, analog immunoassay technologies. Furthermore, neurological pathophysiology is complex, and it has become clear that no single biomarker is sufficient to serve as comprehensive indicator of these processes. For this reason, the capability of Simoa to multiplex blood-based tests for multiple neural biomarkers into a single test has emerged as enormously useful to facilitate biomarker profiling of neurological disorders. This capability has materially contributed to rapid research progress, notably in the Alzheimer’s disease landscape.
Developments on the Alzheimer’s Disease Landscape
The FDA approval of Leqembi as a disease-modifying treatment for Alzheimer’s disease, and the ongoing FDA review of donanemab, has underscored a need for a non-invasive widely available blood test to facilitate diagnosis in the early stages of the disease to identify patients for treatment when therapeutic intervention is most likely to deliver clinical benefit. Currently established biomarker-based approaches to diagnostic workup for Alzheimer’s include positron emission tomography (PET) imaging and CSF biomarkers for amyloid and phosphorylated tau, both of which are invasive, expensive, and may not be widely available. A Rand Corporation study on the readiness of the U.S. health care system infrastructure for an Alzheimer’s treatment highlighted a dire situation of unpreparedness and where key bottlenecks lie. The model assumed annual mild cognitive impairment (MCI) screening for adults leading to 7.5 million referrals to a diagnostic phase in a secondary specialist setting, which is well beyond the country’s capacity. Of these referrals, 6.7 million were assumed to require CSF biomarker or amyloid PET testing for biomarker-based diagnosis of Alzheimer’s disease at an estimated cost of $20.1 billion. Of the amyloid positive subjects, 80% (2.4 million) were assumed to fit the criteria for therapeutic intervention. Assessing the effectiveness of an anti-amyloid Alzheimer’s
6
therapy by testing for amyloid plaque reduction over the course of treatment would potentially involve serial testing of each subject with CSF analysis or PET imaging at a significant cost with currently available detection modalities. The availability of a simple blood test for amyloid positivity has the potential to alleviate bottleneck and cost burdens and facilitate the accessibility of Alzheimer’s therapeutic intervention for patients. We believe our Simoa technology is well-suited to meet this critical need.
High Accuracy Simoa p-Tau 217 Test
The Alzheimer’s field has converged to a consensus on the most desirable single blood-based biomarker at present: plasma p-Tau 217. This biomarker has been shown to out-perform all the other plasma biomarker candidates for Alzheimer’s diagnostics and disease progression monitoring. The biomarker also has the distinction of being very low abundance in blood and difficult to measure by conventional means. Quanterix is well-positioned to provide multiple assays for robustly measuring p-Tau 217 in plasma and helping to establish the clinical evidence through a body of peer reviewed publications demonstrating the superiority of this biomarker relative to other biomarkers. While p-Tau 217 has demonstrated superiority over other neural biomarkers, the strength of the data across all the biomarker candidates (p-Tau 217, p-Tau 181, amyloid b 42/40 ratio, GFAP, NfL) is such that all these biomarkers have been included in the recent updated criteria for diagnosis and staging of Alzheimer’s disease from the National Institute on Aging and Alzheimer’s Association (NIA-AA). However, only p-Tau 217 has been recommended by the NIA-AA as being sufficiently accurate as a single biomarker to enable a diagnostic use case. With this recent consensus on p-Tau 217, Quanterix has launched an LDT for p-Tau 217 to make the test available for clinical use. The test was extensively validated across two independent cohorts (873 samples), and the diagnostic accuracy of the test was shown to meet or exceed the recommendation of the NIA-AA (≥90% accuracy, sensitivity and specificity ≥90%). Thus, the Simoa p-Tau 217 test meets or exceeds the diagnostic performance criteria for the only plasma-based test for Alzheimer’s disease recommended by the NIA-AA.
Simoa Multi-Marker Algorithmic Test
Leveraging both the multiplexing and high sensitivity capability of Simoa, Quanterix has developed a multiplex test that combines four biomarkers into a single plasma test: amyloid b 42, amyloid b 40, GFAP, and NfL. This 4-plex assay, along with p-Tau 181 and p-Tau 217, has been employed by researchers to advance the understanding of how blood-based biomarkers reflect different aspects of Alzheimer’s pathology to both further the basic knowledge of the disease pathophysiology, and to explore the potential of these biomarkers to predict disease state, severity, and progression. This work has resulted in a large body of published evidence that has advanced the understanding of the disease and facilitated drug development through the use of these biomarkers as exploratory indicators of drug target engagement. From a diagnostic test standpoint, a central concept has emerged: since these biomarkers reflect different aspects of Alzheimer’s pathology, combining their signals through logistic regression can enhance overall test accuracy. Furthermore, profiling these biomarkers individually can help clinicians evaluate patients with cognitive symptoms with differential diagnostic information. This information, together with clinical evaluations and cognitive testing, can help determine whether cognitive symptoms are the result of Alzheimer’s disease, or another type of dementia. For example, a patient presenting with cognitive symptoms who has a normal p-Tau 181/217 level, but an abnormal NfL level, is likely to have frontal temporal dementia (FTD). For these reasons, Quanterix is partnering with the Alzheimer’s Drug Discovery Foundation to develop and clinically implement a multi-marker blood test to aid Alzheimer’s diagnosis and differential diagnosis of patients presenting with cognitive symptoms of uncertain origin. The validation of the test includes the 4-plexed biomarkers listed above together with p-Tau 217, and this test is expected to launch in 2024.
Other Neurological Conditions
Our ultra-sensitive Simoa technology has also been instrumental in advancing research into other neurological conditions, such as multiple sclerosis and traumatic brain injury (“TBI”). Evidence of the potential clinical utility of NfL as a biomarker in multiple sclerosis has progressed rapidly, and Simoa’s role in that progression has been foundational. In the past several years, Simoa technology has supported a number of scientific presentations at the 39th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (“ECTRIMS”), the world’s largest meeting dedicated to advancing research for multiple sclerosis. In April 2022, the FDA granted our Simoa NfL plasma test Breakthrough Device designation as a prognostic aid in assessing the risk of disease activity in patients diagnosed with
7
relapsing-remitting multiple sclerosis (“RRMS”). The test has shown promise to be used in conjunction with clinical, imaging and laboratory findings as an aid in identifying RRMS patients who are at lower or higher risk for relapse within four years. We believe this prognostic information could be clinically useful in tailoring the therapeutic approach to more effectively treat the disease.
Current methods of TBI diagnosis involve CT scans that fail to diagnose approximately 90% of mild TBI. Simoa technology has demonstrated the sensitivity to identify relevant neurological biomarkers, such as NfL, tau, GFAP and UCH-L1, to more adequately aid in diagnosis of TBIs and overall brain health. Researchers in neurology have used Simoa technology to study biomarkers in the blood of athletes after concussion in many high-impact sports. Simoa can measure critical neural biomarkers in blood that correlate repeated head trauma from both concussions and subconcussive events with poor patient outcomes, including the potential development of Chronic Traumatic Encephalopathy (CTE), which currently can only be diagnosed after death via a brain autopsy.
Oncology
Our ultra-sensitive Simoa technology has the potential to detect increased levels of oncology biomarkers during the very early stages in disease development. Biomarkers can be useful tools for diagnostics, prognostics and predictive cancer detection. However, many traditional assay technologies can only detect these biomarkers after the disease has progressed and the patient has become symptomatic. Simoa’s highly sensitive detection capability has the potential to enable earlier detection, better monitoring and treatment and improved prognoses for patients. Additionally, Simoa technology has shown early promise as a liquid biopsy alternative to more invasive diagnostic procedures.
Cancer immunotherapy is a promising new area that is significantly affecting cancer remission rates. One challenge of immunotherapy approaches is that the elicited immune responses are not always predictable and can vary from person to person and protocol to protocol. There exists a significant need to develop biomarker tools to monitor these drugs and their effects. Circulating (serum and plasma) protein biomarkers have the potential to be used in the field of health-oncology to stratify patients, predict response, predict recurrence, reveal mechanism of action and monitor for adverse effects. One technical challenge facing the health-oncology drug development process has been the availability of immunoassays with sufficient sensitivity to measure immunomodulatory biomarkers directly in serum and plasma. We have developed a number of tumor biomarker and immune modulation assays (cytokines and chemokines) that can be used to monitor tumor proliferation and host immune response. In particular key immune regulatory cells (T-regs, dendritic cells, macrophages) secrete very low amounts of the protein Interferon gamma (IFN-gamma) and these levels cannot be reliably measured in serum and plasma using conventional, immunoassay technology, however they can be tracked with our Simoa IFN-gamma assay. Additionally, we have developed an ultra-sensitive assay for IL-6, which is one of the cytokines commonly measured for monitoring cytokine release syndrome as an adverse effect in immunotherapies. Several studies have shown that our ultrasensitive assays can be valuable tools for monitoring health-oncology drugs and protocols.
Inflammation
Inflammation underlies the response of the body to injury in a variety of diseases. Simoa assays can measure inflammatory and anti-inflammatory molecules in serum and plasma with unprecedented sensitivity. This has the potential to enable new discoveries into the role of inflammation in the biology of health and disease. Our Simoa technology measures low levels of inflammatory proteins, including cytokines and chemokines, that characterize a range of inflammatory diseases, including Crohn’s disease, asthma, rheumatoid arthritis and neuro-inflammation. We believe the sensitivity of Simoa technology can provide a clearer picture of the underlying state of the immune response and disease progression.
Our Simoa technology also has the potential to be used by companies developing anti-inflammatory drugs to quantify the effect a drug has on a particular inflammatory cytokine and to monitor therapeutic efficacy. We believe that a better understanding of the inflammatory response will be critical to future opportunities for wellness screening and disease response monitoring. Anti-inflammatory drugs are expensive and can have serious side effects, such as increased risk of infection. By monitoring biomarkers indicative of response, clinicians may be able to adjust dose to reduce side effects or increase efficacy.
8
Infectious Disease
The ability to detect infectious disease biomarkers before the onset of an immune response, where a virus is most contagious and multiplying rapidly, is critical for controlling the spread of disease. We believe that our Simoa technology has the potential to impact the reduction of the spread of infectious diseases by making early stage detection more specific and widely available.
Today, early detection of infectious disease is conducted using nucleic acid testing to detect the nucleic acid of the viral or bacterial organism because the levels of infectious disease specific antigens are too low in the early stage of disease to be detected by traditional immunoassay technology. However, the sensitivity of our single molecule detection capabilities enables the detection of extremely low levels of infectious disease specific antigens with sensitivity that can rival the use of nucleic acid testing in this application, without the potential biases inherent in amplification technologies, such as PCR.
Cardiology
Heart disease and related cardiovascular ailments remain the leading cause of death in the United States, contributing to nearly one in four deaths in the United States, according to the CDC. A significant need remains for early prediction of heart attacks and other cardiac events. Simoa’s highly sensitive digital measurement capabilities have the potential to be used to predict early cardiac disease.
Sales and Marketing
We distribute our Simoa instruments and consumables via direct field sales and support organizations located in North America and Europe and through a combination of our own sales force and third-party distributors in additional countries, including Australia, Brazil, China, Czech Republic, India, Hong Kong, Israel, Japan, New Zealand, Qatar, Saudi Arabia, Singapore, South Africa, South Korea, Taiwan, and UAE. In addition, we sell Uman’s NfL antibodies and NfL ELISA kits directly and in conjunction with a distributor worldwide.
As of December 31, 2023, we had 141 employees in sales, sales support and marketing, including technical field application scientists and field service personnel. This staff is primarily located in North America and Europe. We expect we will expand our sales, support, and marketing efforts in the future by expanding our direct footprint in Europe as well as developing a comprehensive distribution and support network in China and other Asia-Pacific region countries where significant new opportunities exist.
Manufacturing and Supply
We outsource the development and manufacturing of our Simoa bead-based instruments to third-party manufacturers, and we develop and manufacture our planar array instrument and all assay kits in our own facilities.
Instruments
The HD-X instrument is manufactured by STRATEC Biomedical AG (“STRATEC”), based in Birkenfeld, Germany, and is manufactured and shipped from their Birkenfeld and Beringen, Switzerland facilities. The SR-X is manufactured by Paramit Corporation (“Paramit”), based in Morgan Hill, California, and is shipped to our global customers by Paramit. See “—Key Agreements” for a description of our agreements with STRATEC and Paramit. Installation of, and training on, our instruments is provided by our employees where we conduct direct sales, and by distributors where sales are conducted through distributors.
We believe this manufacturing strategy is efficient and conserves capital. However, in the event it becomes necessary to utilize a different contract manufacturer for the HD-X or the SR-X, we could experience additional costs, delays and difficulties in doing so, and our business could be harmed.
9
The SP-X instruments are manufactured, tested, shipped and supported by us from our Billerica, Massachusetts facility. All internal components are sourced domestically except one significant component that is sourced in Germany. These components are sourced from a limited number of suppliers, including certain single-source suppliers. Although we believe that alternatives would be available, it would take time to identify and validate replacement components, which could negatively affect our ability to supply SP-X instruments on a timely basis.
Consumables
We assemble our assay kits for our bead-based platform in our Billerica, Massachusetts facility. Our bead-based assays include all components required to run an enzyme-based immunoassay, such as beads, capture and detector reagents, enzyme reagents and enzyme substrate. These reagents are sourced from a limited number of suppliers, including certain single-source suppliers. Although we believe that alternatives would be available, it would take time to identify and validate replacement reagents for our assay kits, which could negatively affect our ability to supply assay kits on a timely basis. As part of our assay redevelopment program, we have increased the shelf life of our bead-based assays to 18 months and believe we are able to mitigate this supplier risk through inventory control.
Simoa disks for our bead-based platform are supplied through a single source supplier pursuant to a long-term supply agreement with STRATEC Consumables, a subsidiary of STRATEC. We believe that this agreement provides for a sufficient notification period to allow for supply continuity and the identification and tech transfer to a new supplier in the event either party wishes to terminate the relationship. Our cuvettes for our bead-based platform are single sourced through STRATEC, and the disposable tips used in our bead-based platform are commercially available.
We assemble our 96-well sample plate kits for our planar array platform in our Billerica, Massachusetts facility. Reagents for our planar array assays include all components required to run an enzyme-based chemiluminescent immunoassay, such as capture antibody printed plates and detector reagents, enzyme reagents and enzyme substrate. These reagents are sourced from a limited number of suppliers, including certain single-source suppliers. Although we believe that alternatives would be available, it would take time to identify and validate replacement reagents for our assay kits, which could negatively affect our ability to supply assay kits on a timely basis. Because our planar array assays have a shelf life of 12 months, we believe we are able to mitigate this risk through inventory control.
NfL antibodies and NfL ELISA Kits
The storage of Uman’s proprietary NfL antibody producing hybridomas, as well as the cultivation and purification of the antibodies, is outsourced to a contract manufacturer, and bulk material of purified antibodies is delivered to Uman’s site in Umeå, Sweden. Functional testing and verification of concentration are performed at Uman before the material is approved for use in production activities. The antibodies can be aliquoted and sold as single reagents or used for the production of Uman’s NfL ELISA kits. The contract manufacturer of antibodies is audited regularly, and we have entered into a written supply agreement with the contract manufacturer. The current shelf-life of the antibodies is 18 months.
All components in Uman’s NfL ELISA kits are manufactured in-house at Uman from starting materials sourced from suppliers that have been evaluated and approved. Uman has entered into supply agreements with critical suppliers. The kit components include buffers (sample diluents and wash solutions), ELISA 96-well plates coated with a capture antibody, detector antibodies, streptavidine conjugates, substrates (TMB) and stop reagents. The final ELISA kit products are subject to quality control procedures, which include testing of human CSF or human serum quality control samples to assure a high batch consistency. After testing and batch record review, the material is released to market. The current shelf-life of the kits is 18 months (NF-light ELISA (CSF)) or 13 months (NF-light Serum ELISA).
Key Agreements
Development Agreement and Supply Agreement with STRATEC
In August 2011, we entered into a Strategic Development Services and Equity Participation Agreement with STRATEC, pursuant to which STRATEC undertook the development of the Simoa HD instrument. In September 2011,
10
we also entered into a Supply and Manufacturing Agreement with STRATEC (the “STRATEC Supply Agreement”), pursuant to which STRATEC agreed to supply HD instruments to us, and we agreed to procure those instruments exclusively from STRATEC, subject to STRATEC’s ability to supply the instruments. We are responsible for obtaining any regulatory approval necessary to sell the instruments. The instrument price stipulated in the STRATEC Supply Agreement was established based on certain specified assumptions and is subject to certain adjustments.
The STRATEC Supply Agreement is terminable by either party on 12 months’ notice to the other party. The STRATEC Supply Agreement may also be terminated on the insolvency of a party or the uncured material breach of a party, or, by us, on a change of control of our company (subject to certain obligations to compensate STRATEC on such termination). On termination by us for STRATEC’s insolvency or uncured material breach or termination by STRATEC for convenience, we are granted a nonexclusive royalty free license of STRATEC intellectual property to manufacture the instruments. In certain of these circumstances, we could be obligated to issue warrants to purchase our common stock.
Paramit Manufacturing Services Agreement
In November 2016, we entered into a Manufacturing Services Agreement (the “Paramit Agreement”) with Paramit to produce and test our SR-X instrument on an as-ordered basis. We also engaged Paramit to supply spare parts for the SR-X instrument. Paramit has no obligation to maintain inventory in excess of any open purchase orders or materials in excess of the amount Paramit reasonably determines will be consumed within 90 days or within the lead time of manufacturing our instrument, whichever is greater. We have an obligation to purchase any material or instruments deemed in excess pursuant to the Paramit Agreement. The price is determined according to a mutually agreed-upon pricing formula. The parties agreed to review the pricing methodology yearly or upon a material change in cost.
The Paramit Agreement had an initial three-year term with automatic one year extensions. It is terminable by either party for convenience with written notice to the other party given at least nine months prior to the end of the then-current term. The agreement may also be terminated by us with three months’ notice to Paramit upon the occurrence of (i) a failure of Paramit to obtain any necessary governmental licenses, registrations or approvals required to manufacture our instrument or (ii) an assignment by Paramit of its rights or obligations under the agreement without our consent. The Paramit Agreement is terminable by Paramit with 30 days’ notice to us in the event of a material breach after written notice and a 60-day opportunity to cure the breach.
Competition
We compete with both established and development-stage life science companies that design, manufacture and market instruments for proteomics discovery and clinical research applications. For example, companies such as Bio-Techne, DiaSorin, MesoScale Discovery, Gyros, SEER, MilliporeSigma, Bio-Rad Laboratories, Thermo Fisher Scientific, Roche, C2N Diagnostics, and others, have products for protein measurements in biofluids that compete in certain segments of the market in which we sell our products. Our Accelerator Laboratory competes with other research laboratories such as LabCorp, Covance, Q2 Solutions, Rules Based Medicine, Monogram Biosciences, PPD Laboratories, and others, some of which are customers of ours. In addition, as we or our partners expand the applications for our products to include diagnostics, we expect to compete with companies such as Siemens, Abbott, Roche, Ortho Clinical Diagnostics and Thermo Fisher Scientific. Furthermore, our technology and products are showing promise for non-invasive early disease detection, and in the future, we could experience competition from companies that develop and market imaging and other molecular detection technologies. In addition, a number of other companies and academic groups are in the process of developing novel technologies for life science research and diagnostics. Many of the companies with which we compete or will compete have substantially greater resources than we have.
11
The life science instrumentation and lab services industries are highly competitive and expected to grow more competitive with the increasing knowledge gained from ongoing research and development. We believe the principal competitive factors for us include:
| ● | sensitivity; |
| ● | cost of instruments and consumables; |
| ● | reputation among customers and key opinion leaders; |
| ● | innovation in product offerings; |
| ● | accuracy and reproducibility of results; and |
| ● | customer support infrastructure. |
We believe that we are well positioned with respect to these competitive factors and expect to enhance our position through ongoing global expansion, innovative new product introductions and ongoing collaborations, and partnerships with key opinion leaders.
Intellectual Property
Our success depends in part on our ability to obtain and maintain intellectual property protection for our products and technology. We use a variety of intellectual property protection strategies, including patents, trademarks, trade secrets, and other methods of protecting proprietary information.
Our patent strategy is multilayered, providing coverage of aspects of the core technology as well as specific uses and applications. The first layer is based on protecting the fundamental methods for detecting single molecules independent of the specific analyte to be detected. The second layer covers embodiments of the core technology directed to the detection of specific analytes. The third layer protects novel instrumentation, consumables, and manufacturing processes used in applying the invention to certain commercial products or future product opportunities. The fourth layer is concerned with specific uses of the core technology (e.g., biomarkers and diagnostics). Our patent strategy is both offensive and defensive in nature, seeking to protect not only technology we currently practice but also alternative, related embodiments.
As of December 31, 2023, we owned or exclusively licensed 105 issued patents and patent applications worldwide. Our owned or exclusively licensed patents and patent applications, if issued, are expected to expire between 2025 and 2042, in each case without taking into account any possible patent term adjustments or extensions and assuming payment of all appropriate maintenance, renewal, annuity or other governmental fees.
Our core Simoa bead-based technology, directed to general methods and devices for single molecule detection, originated at Tufts University (Tufts), in the laboratory of Professor David Walt, who is the founder of Quanterix and a current member of our Board of Directors. Prof. Walt and his students pioneered the single molecule array technology, including technologies that enabled the detection of single enzyme labels in arrays of microwells, thereby facilitating ultra-sensitive detection. We have exclusively licensed from Tufts the relevant patent filings related to these technologies. (See “—License Agreement with Tufts University” below).
In addition to our reliance on patent protection for our inventions, products and technologies, we also rely on trade secrets, know-how, confidentiality agreements, and continuing technological innovation to develop and maintain our competitive position. For example, some elements of our manufacturing processes, analytics techniques and processes, as well as computational-biological algorithms, and related processes and software, are based on unpatented trade secrets and know-how that are not publicly disclosed. Although we take steps to protect our proprietary information and trade secrets, including through contractual means with our employees, advisors and consultants, these agreements may be breached or may be unenforceable and we may not have adequate remedies. In addition, third parties may independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets or disclose our technology. As a result, we may not be able to meaningfully protect our intellectual property. For further discussion of the risks relating to our intellectual property, see the section titled “Risk factors—Risks Related to our Intellectual Property.”
12
License Agreement with Tufts University
In June 2007, as amended in April 2013, August 2017, and September 2020, we entered into a license agreement with Tufts, pursuant to which we obtained an exclusive, worldwide license to all patent rights to the Simoa bead-based technology owned by Tufts, as well as a non-exclusive license to related know-how. The rights licensed to us are for all fields of use and are sublicensable.
Under the terms of the agreement, as amended, we paid a one-time, non-refundable upfront fee and issued Tufts shares of our stock. We are required to pay Tufts low single-digit royalties on all net sales of products and services that use the licensed technology, as well as a portion of any sublicensing revenues. We are also obligated to pay annual maintenance fees, which are fully creditable against any royalty payments made by us, and a milestone payment upon any sublicense by us. We were also required to reimburse Tufts for all patent prosecution costs.
The term of the license agreement will continue on a country-by-country basis so long as there is a valid claim of a licensed patent in such country. Tufts may terminate the agreement or convert to a non-exclusive license in the event (1) we fail to pay any undisputed amount when required and fail to cure such non-payment within 60 days after receipt of notice from Tufts, (2) we are in breach of any material provision of the agreement and fail to remedy such breach within 60 days after receipt of notice from Tufts, (3) we do not demonstrate diligent efforts to develop a product incorporating the licensed technology, (4) we are found on five separate audits to have underpaid pursuant to the terms of the agreement, (5) we cease to carry on the business related to the licensed technology either directly or indirectly, or (6) we are adjudged insolvent, make an assignment for the benefit of creditors or have a petition in bankruptcy filed for or against us that is not removed within 60 days. We may terminate the agreement at any time upon at least 60 days’ written notice. Upon termination of the agreement, all rights revert to Tufts.
Government Regulation
The majority of our products are currently intended for RUO applications, although our customers may use our products to develop their own products that are subject to regulation by the FDA or the Center for Medicare & Medicaid Services (“CMS”). Although most in vitro diagnostic products intended for RUO are not currently required to obtain premarket clearance or approval by the FDA, products labeled as RUO are subject to the FDA’s premarket review requirements if they are determined to be intended for use for clinical rather than non-clinical research purposes. Consequently, other than our three laboratory developed tests intended for clinical testing, our products are labeled and intended “For Research Use Only. Not for Diagnostic Procedures.”
The FDA has issued Final Guidance for Industry and Food and Drug Administration Staff on “Distribution of In Vitro Diagnostic Products Labeled for Research Use Only or Investigational Use Only” (the “RUO/IUO Guidance”). The purpose of this FDA guidance document is to provide the FDA’s current thinking on when IVD products are properly labeled for RUO or for investigational use only (“IUO”) and when products labeled RUO or IUO will be viewed by the FDA as intended for clinical use. The RUO/IUO Guidance explains that the FDA will review the totality of the circumstances when evaluating whether equipment and testing components are properly labeled as RUO. Merely including a labeling statement that a product is intended for research use only will not necessarily exempt the device from the FDA’s premarket notification and clearance process (510(k)), premarket approval, or other requirements, if the circumstances surrounding the distribution of the product indicate that the manufacturer intends its product to be used for clinical diagnostic use. These circumstances may include written or verbal marketing claims or links to articles regarding a product’s performance in clinical applications, a manufacturer’s provision of technical support for clinical validation or clinical applications, or solicitation of business from clinical laboratories, all of which could be considered evidence of intended uses that conflict with RUO labeling. We believe that our labeling and promotion of our products, including the custom assay RUO products developed by the Accelerator Laboratory, is consistent with the RUO/IUO Guidance because we have not promoted our products for clinical use in humans.
IVD tests intended for clinical diagnostic use are regulated by the FDA as medical devices, however, the FDA has traditionally not regulated certain laboratory tests referred to as laboratory developed tests (“LDTs”) (as discussed further below). The FDA defines a medical device in part as an instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article which is intended for the diagnosis of disease or other
13
conditions or in the cure, mitigation, treatment, or prevention of disease in man. The FDA will regulate the development, testing, manufacturing, marketing, post-market surveillance, distribution, advertising, and labeling of our clinical IVD devices (other than our LDTs) and we will be required to register as a medical device manufacturer and list our marketed products.
Clinical Laboratory Improvement Amendments of 1988, Regulation of LDTs and State Regulation
We own and operate a CLIA-certified laboratory. The Clinical Laboratory Improvement Amendments of 1988 (“CLIA”) are federal regulatory standards that apply to all clinical laboratory testing performed on humans in the United States (with the exception of research testing that does not report patient specific results). A clinical laboratory is defined by CLIA as any facility that performs examinations of specimens obtained from humans for the purpose of providing information for the diagnosis, prevention, or treatment of any disease or impairment of, or the assessment of health of, human beings. CLIA requires such laboratories to be certified by the federal government and mandates compliance with various operational, personnel, facilities administration, quality and proficiency testing requirements intended to ensure that testing services are accurate, reliable and timely. CLIA certification also is a prerequisite to be eligible to bill state and federal health care programs, as well as many private insurers, for laboratory testing services.
In addition, CLIA requires certified laboratories to enroll in an approved proficiency testing program for each of the specialties and subspecialties for which it is certified. If a laboratory fails to achieve a passing score on a proficiency test, then its CLIA certificate may be suspended, limited or revoked, or other sanctions may be imposed.
As a condition of CLIA certification, laboratories are subject to survey and inspection every other year (except laboratories with only a certificate of waiver or certificate of provider-performed microscopy procedures are not subject to biennial inspections), in addition to being subject to additional random inspections. The biennial survey is conducted by CMS, a CMS agent (typically a state agency), or a CMS-approved accreditation organization.
High complexity, CLIA-certified laboratories, such as ours, frequently develop testing procedures to provide diagnostic results to customers. These tests have been offered by high-complexity laboratories for the last few decades as LDTs, the validation and performance of which are subject to CMS oversight through its enforcement of CLIA. The FDA also has claimed that it has regulatory authority over LDTs under the agency’s medical device authorities, but historically has not exercised enforcement with respect to most LDTs when they meet FDA’s definition of an LDT. FDA defines an LDT as an in vitro diagnostic that is intended for clinical use and is designed, manufactured, and used within a single high-complexity, CLIA-certified laboratory. In 2010, the FDA had announced that it decided to exercise regulatory authority over LDTs, and that it planned to issue guidance to the industry regarding its regulatory approach. The FDA never finalized such guidance and later indicated that Congress should enact legislation to address improved oversight of diagnostics, including LDTs, rather than the FDA addressing the issue through administrative proposals. However, over the past several years, FDA has increased its oversight of what it perceived as especially high-risk LDTs, and in October 2018 the FDA issued a safety communication warning against the use of unapproved LDTs that describe relationships between gene variants and particular drugs. FDA also regulated LDTs for Covid-19 under its emergency use authorization (“EUA”) authority and related guidance. In August 2020, the Department of Health and Human Services (“HHS”) issued a policy stating that FDA could not require premarket review (including EUAs) for LDTs, but this policy was revoked by the HHS in November 2021 (accordingly, reinstating FDA’s EUA requirements for Covid-19 LDTs). Thus, while FDA generally exercises enforcement discretion for LDTs, it continues to exercise oversight over certain high-risk tests. In addition, over the past several years, several bills to establish a new FDA regulatory framework with similarities to the current medical device framework for “in vitro clinical tests,” which would include both in vitro diagnostics and LDTs, have been introduced but have not been enacted.
Most recently, however, in September 2023, FDA issued a proposed rule regarding LDTs that would establish that in vitro diagnostic products are devices under the Federal Food, Drug, and Cosmetic Act, including when the manufacturer is a laboratory. The proposed rule also describes a policy under which FDA would provide greater oversight of LDTs by phasing out its general enforcement discretion approach, and phase in medical device regulation, for LDTs over a period of four years. FDA requested comment on several aspects of its proposal and approach, including whether certain types of LDTs should remain under enforcement discretion. Currently FDA’s target date for final action on this rule is April 2024, but we cannot predict the ultimate timing or form of FDA guidance or regulation, legislative
14
action or their potential impact. Any new regulatory approach for LDTs by the FDA, including as described in the September 2023 proposed rule, would likely lead to an increased regulatory burden, including additional costs and delays in introducing new LDTs, and could result in tests being removed from the market if regulatory clearance or approval from FDA cannot be obtained.
In addition, some states require that any laboratory be licensed by the appropriate state agency in the state in which it operates. Laboratories must also hold state licenses or permits, as applicable, from various states including, but not limited to, California, New York, Pennsylvania, Rhode Island, and Maryland, to the extent that they accept specimens from one or more of these states, each of which requires out-of-state laboratories to obtain licensure. Laboratories also must receive approval from New York for any LDT that has not been reviewed by FDA, to the extent that LDT is offered to New York patients.
If a laboratory is out of compliance with state laws or regulations governing licensed laboratories or with CLIA, it may be subject to enforcement actions that may include suspension, limitation or revocation of the license or CLIA certificate, assessment of financial penalties or fines, or imprisonment. Loss of a laboratory’s CLIA certificate or state license may also result in the inability to receive payments from state and federal health care programs as well as private third-party payors.
When we perform clinical diagnostic testing, we may be subject to the Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), depending on the types of transactions we engage in, as well as additional federal and state laws that impose a variety of fraud and abuse prohibitions on healthcare providers, including clinical laboratories.
Europe/Rest of World Government Regulation
We must obtain the requisite approvals from regulatory authorities in non-U.S. countries prior to the commencement of clinical trials or marketing of our product for clinical diagnostic use in those countries. The regulations in other jurisdictions vary from those in the U.S. and may be easier or more difficult to satisfy and are subject to change. For example, in the European Union (the “E.U.”) new regulations recently entered into force that introduce greater regulation of medical devices and IVDs. The new IVD regulation (the “IVD Regulation”) is significantly different from the European directive for IVD medical devices (the “IVD Directive”) that it replaces in that it ensures that the new requirements apply uniformly and on the same schedule across the member states, includes a risk-based classification system and increases the requirements for conformity assessment.
The CE registration for the NfL ELISA assay kit of our subsidiary Uman was approved in March 2014 under the IVD Directive. Under the IVD Directive, the assay is classified as a general IVD product and required self-certification with no involvement of a notified body/authority. The IVD Regulation introduces a new classification system for IVDs and assessment by a notified body is required for class B, C and D products. Uman’s NfL ELISA assay kit is classified as a class B product and must fully comply with (and have a CE mark issued under) the IVD Regulation by May 2027 (subject to proposed extension of the transitional periods in the IVD Regulation). The new requirements include an ISO 13485 certification of the quality system (which Uman received in July 2018) and increased technical evidence and follow-up of performance of the specific product (e.g., clinical evidence and post-market activities). The work to evaluate and to meet the new technical requirements is on-going. When all requirements are met, a notified body will be contacted, and the certification initiated.
The NF-light Serum ELISA is currently sold only as a RUO product (not intended for diagnostic use). Work is on-going to prepare a technical file compliant with the IVD Regulation for this product as well.
Other Governmental Regulation
Privacy and Data Security Laws and Regulations
As a business with a global footprint, compliance with evolving regulations and standards in privacy and data security has resulted, and may continue to result, in increased costs, new compliance challenges, and the threat of
15
increased regulatory enforcement activity. Our business relies on various safeguards to secure electronic transmission, storage and hosting of sensitive information, including personal information, protected health information, financial information, intellectual property and other sensitive information related to our customers and workforce.
For example, in the U.S., the collection, maintenance, protection, use, transmission, disclosure, and disposal of certain personal information and the security of medical devices are regulated at the U.S. federal and state, international, and industry levels. U.S. federal and state laws protect the confidentiality of certain patient health information, including patient medical records, and restrict the use and disclosure of patient health information by health care providers. Privacy and Security Rules under HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (“HITECH”), govern the use, disclosure, and security of protected health information by “Covered Entities,” (which include health care providers that submit electronic claims, health plans, and health care clearinghouses) and by their “Business Associates” (which is anyone that performs a service on behalf of a Covered Entity involving the use or disclosure of protected health information and is not a member of the Covered Entity’s workforce). Rules under HIPAA and HITECH include specific security standards and breach notification requirements. HHS (through the Office for Civil Rights) has direct civil enforcement authority against Covered Entities and Business Associates with regard to both the Security and Privacy Rules. The U.S. Department of Justice has criminal enforcement authority against Covered Entities, Business Associates, and certain other entities and individuals. In addition, State Attorneys General may being enforcement actions under HIPAA. Generally Quanterix is not a Covered Entity, however, we may operate as a Business Associate to Covered Entities under certain circumstances.
In addition, a number of states have also adopted laws and regulations that may affect our privacy and data security practices for personal information, such as comprehensive state privacy laws that govern the use, disclosure and protection of personal information, such as certain health information, Social Security numbers, and credit card account data. State consumer protection laws and consumer health privacy laws also establish privacy and security standards for use and management of personal information, including information related to consumers and care providers.
Outside the U.S., we are impacted by the privacy and data security requirements at the international, national and regional level, and on an industry specific basis. Legal requirements in foreign countries relating to the collection, storage, handling and transfer of personal data and potentially intellectual property continue to evolve with increasingly strict enforcement regimes. More privacy and security laws and regulations are being adopted, and more are being enforced, with potential for significant financial penalties. In the E.U., stringent data protection and privacy rules which substantially impact the use of patient data across the healthcare industry became effective in May 2018. The E.U. General Data Protection Regulation (“GDPR”) applies uniformly across the E.U. and includes, among other things, a requirement for prompt notice of data breaches to data subjects and supervisory authorities in certain circumstances and significant fines for non-compliance. The GDPR also requires companies processing personal data of individuals residing in the E.U. to comply with E.U. privacy and data protection rules. In the area of health data, the GDPR is supplemented by national laws and regulations that are less harmonized.
Because data privacy laws and regulations continue to expand, differ from jurisdiction to jurisdiction, and are subject to evolving (and at times inconsistent) governmental interpretation, compliance with these laws and regulations may require significant additional expenditures or changes in products or business that increase competition or reduce revenue. Noncompliance could result in the imposition of fines, penalties, or orders to stop noncompliant activities.
Environmental Health and Safety Laws
We are subject to federal, state, and local laws and regulations related to the protection of the environment, the health and safety of employees and the handling, transportation, storage and disposal of medical specimens, infectious and hazardous waste and radioactive materials. For example, the U.S. Occupational Safety and Health Administration (“OSHA”), has established extensive requirements relating specifically to workplace safety for healthcare employers in the U.S. This includes requirements to develop and implement multi-faceted programs to protect workers from exposure to blood-borne pathogens, including preventing or minimizing any exposure through needle stick injuries. OSHA also regulates the use of hazardous chemicals in the workplace. Likewise, we are subject to EPA and state requirements
16
relating to the management and disposal of hazardous waste, and state requirements relating to the disposal of regulated medical waste. For purposes of transportation, some biological materials and laboratory supplies are classified as hazardous materials and are subject to regulation by one or more of the following agencies: the U.S. Department of Transportation, the U.S. Public Health Service, the United States Postal Service and the International Air Transport Association. We generally use third-party vendors to dispose of regulated medical waste, hazardous waste and radioactive materials that we may use during our research.
Employees and Human Capital
As of December 31, 2023, we had 441 full-time employees, of which 141 worked in sales, sales support, field service, and marketing, 121 worked in engineering and research and development, 117 worked in manufacturing and operations and 62 worked in general and administration. Of our 441 full-time employees, 382 were located in the United States and 59 were located in 13 foreign countries. None of our employees is represented by a labor union or subject to a collective bargaining agreement. We have a strong culture that emphasizes the impact our work has on the detection of neurological and other critical disorders.
Our success depends upon our ability to attract and retain highly qualified employees. Talent management is critical to our ability to execute our long-term growth strategy, and we seek to cultivate a high performing pool of talent by providing career growth, on-the-job learning opportunities and competitive total rewards.
Workforce Compensation and Pay Equity
We provide competitive compensation and benefits programs to help recruit and retain our high performing employees. We utilize third party benchmark compensation data to assist in the evaluation of market wages. Our compensation is designed to attract, retain, and motivate employees to achieve results while balancing short- and long-term company performance. All of our employees are eligible for an annual bonus and/or commission plan, a matching 401(k) Plan (in the case of US employees), healthcare and insurance benefits, paid time off, family leave, employee assistance programs, and behavioral health services. Additionally, all our employees are eligible for annual equity-based grants with vesting conditions designed to award our employees’ performance and encourage retention.
Company Culture
We are committed to an inclusive culture which is grounded on our values of integrity, respect, and equality. In support of our inclusive culture, we sponsor an internal group of culture champions who are focused on diversity, equity, and inclusion. These champions are comprised of employees and executives whose mission is to provide respectful workplace training and awareness to strengthen employee understanding and knowledge of key topics focused on diversity and inclusion. As of December 31, 2023, approximately 43% of our employees were women and approximately 40% of our employees were non-white.
We expect all personnel working at Quanterix, employees, interns, and contractors, to observe the highest levels of business ethics, integrity, and mutual respect. Our employee handbook and Corporate Code of Conduct and Ethics set forth policies that reflect our values and provide guidance for registering complaints in the event of any violation of our policies. An “open door” policy is maintained at all levels of the organization, and any form of retaliation against an employee is strictly prohibited.
Employee Engagement and Wellness
The success of our business is dependent on the physical and mental well-being of our employees. Accordingly, we are committed to creating a safe and healthy workplace for all personnel. We provide our employees with a wide range of policies and practices to ensure an environment of physical and psychological safety and well-being.
17
Corporate Information
We were incorporated under the laws of the State of Delaware in April 2007 under the name “Digital Genomics, Inc.” In August 2007, we changed our name to “Quanterix Corporation.” Our principal executive offices are located at 900 Middlesex Turnpike, Billerica, Massachusetts 01821, and our telephone number is (617) 301-9400.
Information Available on the Internet
Our Internet website address is www.quanterix.com. The information contained on, or that can be accessed through, our website is not a part of or incorporated by reference in this Annual Report on Form 10-K. We have included our website address in this Annual Report on Form 10-K solely as an inactive textual reference. We make available free of charge through our website our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to those reports filed or furnished pursuant to Sections 13(a) and 15(d) of the Securities Exchange Act of 1934, as amended (“Exchange Act”). We make these reports available through the “Investors—Financial Information—SEC Filings” section of our website as soon as reasonably practicable after we electronically file such reports with, or furnish such reports to, the SEC. We also make available, free of charge on our website, the reports filed with the SEC by our executive officers, directors and 10% stockholders pursuant to Section 16 under the Exchange Act as soon as reasonably practicable after copies of those filings are provided to us by those persons. Our electronically filed reports and other information that we file with the SEC can be viewed on the SEC’s website at http://www.sec.gov.
18
ITEM 1A. RISK FACTORS
The following risk factors and other information included in this Annual Report on Form 10-K should be carefully considered. The risks and uncertainties described below are not the only ones we face. Additional risks and uncertainties not presently known to us or that we presently deem less significant may also impair our business operations. Please see page ii of this Annual Report on Form 10-K for a discussion of some of the forward-looking statements that are qualified by these risk factors. If any of the following risks occur, our business, financial condition, results of operations and future growth prospects could be materially and adversely affected.
Risk Factor Summary
Our business is subject to numerous risks and uncertainties. The following summary highlights some of the risks to be considered with respect to our business and prospects. This summary is not complete and the risks summarized below are not the only risks we face. Readers should review and carefully consider the risks and uncertainties described in more detail below, which includes a more complete discussion of these risks.
| ● | Our quarterly and annual operating results and cash flows have fluctuated in the past and might continue to fluctuate, which could cause the value of our common stock to fluctuate or decline significantly. |
| ● | We have incurred annual losses since we were formed and expect to incur losses in the future. We cannot be certain that we will achieve or sustain profitability. |
| ● | Failure to remediate material weaknesses in, or inherent limitations associated with, our internal control over financial reporting could result in material misstatements in our financial statements. |
| ● | If our products fail to achieve and sustain sufficient market acceptance, our revenue will be adversely affected. |
| ● | Sales of our assays for neurological indications have become increasingly important to our business, and any significant decrease in sales of such assays could have a material adverse effect on our business. |
| ● | We may not be successful in penetrating the diagnostics market. |
| ● | Because a significant portion of our revenue comes from a few large customers, any significant decrease in sales to these customers, due to industry consolidation or otherwise, could harm our operating results. |
| ● | Our long-term results depend upon our ability to improve existing products, develop or acquire new technology, and develop, introduce and market new products successfully. |
| ● | Defects or other quality issues in our products could lead to unforeseen costs, product recalls, adverse regulatory actions, negative publicity, and litigation, including product liability claims, any of which could cause customers to decide not to purchase our products, harm our reputation, and negatively affect our sales, operating results and financial condition. |
| ● | We generate a substantial portion of our revenue internationally and we expect this will continue in the future; as a result, our business is subject to various risks relating to our international activities, which could adversely affect our business, operating results and financial condition. |
| ● | We rely on a single contract manufacturer to manufacture and supply our Simoa HD-X instrument and rely on a different single contract manufacturer to manufacture and supply our Simoa SR-X. If either of these manufacturers should fail to perform, or not perform satisfactorily, our ability to supply these instruments would be negatively and adversely affected. |
| ● | We rely on a limited number of suppliers or, in some cases, one supplier, for some of our materials and components used in our consumable products and our SP-X instrument, and we may not be able to find replacements or immediately transition to alternative suppliers if any of these suppliers fail to perform, which could have a material adverse effect on our business, financial condition, results of operations and reputation. |
| ● | We face significant competition in the life sciences research and diagnostic markets. |
| ● | If the FDA determines that our products are subject to regulation as medical devices, if the FDA modifies its regulations to require that our LDTs are subject to regulation as devices, or if we seek to market our products for clinical diagnostic or health screening use, we will be required to obtain regulatory clearance(s) or approval(s) and may be required to cease or limit sales of our then-marketed |
19
| products, which could materially and adversely affect our business, financial condition and results of operations. Any such regulatory process would be expensive, time-consuming and uncertain both in timing and in outcome. |
| ● | If we do not comply with governmental regulations applicable to our CLIA-certified laboratory, we may not be able to continue our operations or continue offering our LDTs. |
| ● | Cybersecurity breaches, loss of data and other disruptions could compromise sensitive information related to our business or prevent us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation. |
| ● | If we are unable to protect our intellectual property, our ability to maintain any technological or competitive advantage over our competitors and potential competitors may be reduced and our business may be harmed. |
| ● | If we or any of our partners are sued for infringing intellectual property rights of third parties, the resulting litigation would be costly and time-consuming, and an unfavorable outcome in that litigation could have a material adverse effect on our business. |
| ● | Our stock price may fluctuate significantly. |
Risks Related to Our Financial Condition
Our quarterly and annual operating results and cash flows have fluctuated in the past and might continue to fluctuate, which could cause the value of our common stock to fluctuate or decline significantly.
Numerous factors, many of which are outside of our control, may cause or contribute to significant fluctuations in our quarterly and annual operating results. These fluctuations may make financial planning and forecasting difficult. In addition, one or more of such factors may cause our revenue or operating expenses in one period to be disproportionately higher or lower relative to the others, and comparing our operating results on a period-to-period basis might not be meaningful. Investors should not rely on our past results as indicative of our future performance. Moreover, our stock price might be based on expectations of future performance that are unrealistic or that we might not meet and, if our revenue or operating results fall below the expectations of investors or securities analysts, the price of our common stock could decline significantly.
We have incurred annual losses since we were formed and expect to incur losses in the future. We cannot be certain that we will achieve or sustain profitability.
We incurred net losses of $32.3 million, $96.7 million and $57.7 million for the years ended December 31, 2023, 2022, and 2021, respectively. As of December 31, 2023, we had an accumulated deficit of $434.5 million. We cannot predict if or when we will achieve profitability or if or when we will be able to sustain profitability once achieved. We expect that our losses will continue at least through the next 24 months as we execute our strategy for our entry into translational pharma and clinical diagnostic markets. We may incur significant losses in the future for a number of reasons, many of which are beyond our control, including the other risks described in this Annual Report on Form 10-K, the market acceptance of our products, competitive products, future product development and our market penetration and margins.
Failure to remediate material weaknesses in, or inherent limitations associated with, internal control over financial reporting could result in material misstatements in our financial statements.
In our Annual Report on Form 10-K for the year ended December 31, 2022, we identified four material weaknesses in our internal control over financial reporting, relating to the operating effectiveness of our internal controls associated with: (i) the accounting for inventory, including excess and obsolescence reserves (the “Inventory MW”), (ii) the accounting for salaries and commissions expense (the “Compensation MW”), (iii) the financial statement close process, including financial reporting, share-based compensation and non-recurring transactions such as impairment of assets and accounting for leases (the “Financial Statement Close Process MW”), and (iv) the accounting for property and equipment, net (the “Property and Equipment MW”).
20
During 2023, we took a number of actions designed to improve our internal control over financial reporting to remediate these material weaknesses. Based on these efforts, and after demonstrating the operating effectiveness of the related internal controls for a sufficient period of time, our management has concluded that the Financial Statement Close Process MW and Compensation MW were remediated as of December 31, 2023. However, management also concluded that control deficiencies did exist as of December 31, 2023, and that these control deficiencies constituted material weaknesses in our internal control over financial reporting. Specifically, management concluded that a portion of the Inventory MW related to the valuation of our inventory, including excess and obsolescence reserves (the “Inventory Valuation MW”) and the Property and Equipment MW continued to exist as of December 31, 2023. The primary cause of the Inventory Valuation MW is our reliance on manual processes to verify the completeness and accuracy of information used in our inventory valuation outputs, and the adequacy and documentation of reviews over these outputs. For the Property and Equipment MW, while the related internal controls were implemented and effective as of December 31, 2023, they were not in all cases in place for a sufficient period of time to demonstrate operating effectiveness as of December 31, 2023. For a discussion of these material weaknesses and our efforts to remediate them, please see “Item 9A. Controls and Procedures”.
Our efforts to remediate the outstanding material weaknesses, and to maintain effective internal control over financial reporting, are ongoing; however, there are inherent limitations in all control systems and no evaluation of controls can provide absolute assurance that all deficiencies have been detected. We cannot assure you that additional material weaknesses in our internal control over financial reporting will not arise or be identified in the future. If after having remediated the remaining material weaknesses we are unable to maintain the effectiveness of our internal control over financial reporting or our disclosure controls and procedures, we could lose investor confidence in the accuracy and completeness of our financial reports, the market price of our common stock could decline, and we could be subject to regulatory scrutiny, civil or criminal penalties or litigation. Continued or future failure to maintain effective internal control over financial reporting could also result in financial statements that do not accurately reflect our financial condition or results of operations, may result in material misstatements in our financial statements and may also restrict our future access to the capital markets.
During the year ended December 31, 2023, we incurred significant expense and dedicated significant internal resources to address the material weaknesses described above, and we expect that the continued execution of the plan to remediate the remaining material weaknesses will be costly and will distract management from other activities. There can be no assurance that we will conclude in the future that we have effectively remediated the remaining material weaknesses or that we will not identify any significant deficiencies or other material weaknesses that will impair our ability to report our financial condition and results of operations accurately or on a timely basis.
Our ability to use net operating losses to offset future income may be subject to certain limitations.
As of December 31, 2023, we had federal net operating loss (“NOLs”) carryforwards to offset future taxable income of approximately $313.4 million, which begin to expire in 2026. A lack of future taxable income would adversely affect our ability to utilize these NOLs. In addition, under Section 382 of the Internal Revenue Code of 1986, as amended (the “Code”), a corporation that undergoes an “ownership change” is subject to limitations on its ability to utilize its NOLs to offset future taxable income. We have already experienced ownership changes as defined under Section 382 of the Code. Depending on the timing of any future utilization of our NOLs, the amount that can be utilized each year may be limited as a result of such previous ownership changes. In addition, future changes in our stock ownership, including changes that may be outside of our control, could result in additional ownership changes under Section 382 of the Code. Our NOLs may also be impaired under similar provisions of state law. We have recorded a full valuation allowance related to our NOLs and other deferred tax assets due to the uncertainty of the ultimate realization of the future benefits of those assets.
Risks Related to Our Business
If our products fail to achieve and sustain sufficient market acceptance, our revenue will be adversely affected.
Our success depends on our ability to develop and market products that are recognized and accepted by our customers and potential customers as reliable, enabling and cost-effective. Continued market acceptance of our Simoa
21
technology platform and products and other platforms and products we may develop in the future will depend on many factors, including our ability to convince potential customers that our technology is an attractive alternative to other available technologies. If we are unable to continue to motivate customers to use Simoa technology or other technologies we may develop, adoption of our technology may be slowed and our ability to retain and grow our customer base and increase our revenue would be adversely affected.
Sales of our assays for neurological indications have become increasingly important to our business, and any significant decrease in sales of such assays could have a material adverse effect on our business.
Neurology has been one of our primary focus areas for commercialization of our Simoa technology and the services that we provide to our customers. Sales from neurological-related biomarkers have become an increasingly important part of our business. There can be no assurance that we will continue to derive meaningful revenues from the sale of our neurological products, from services related to neurodegenerative conditions or from sales of instruments driven by customers desiring access to our technology for work relating to neurological conditions. The adoption by our customers of competitive technologies for detecting biomarkers of neurodegenerative conditions could negatively impact our revenues and have a material adverse effect on our business.
We may not be successful in penetrating the diagnostics market.
We believe our Simoa technology has the capability to enable the development of a new category of less-invasive diagnostic tests that could replace current invasive, expensive, and inconvenient diagnostic methods. Accordingly, we have begun to expand into the diagnostics market. Transitioning from research use only to also serving the diagnostics market entails significant risks, including:
| ● | significant investments in product development, scaling manufacturing processes, marketing and sales activities, regulatory compliance, reimbursement and billing activities and infrastructure to support the foregoing; |
| ● | navigating complex regulatory frameworks, including but not limited to FDA regulations and equivalent agencies internationally; |
| ● | competition from products that may offer superior performance, pricing, or convenience, and prevent us from penetrating target markets effectively; and |
| ● | challenges associated with obtaining adequate reimbursement from government healthcare programs and private insurers. |
Further, our progress in penetrating the diagnostics market may be slower than we intend and may require a substantially larger investment than we expect. If we are unable to manage these risks effectively, our efforts to penetrate the diagnostics market may be unsuccessful, and our business, operating results and financial condition could suffer.
The sales cycle for our Simoa instruments can be lengthy and variable, which makes it difficult for us to forecast revenue and other operating results.
The sales process for our Simoa instruments generally involves numerous interactions with multiple individuals within an organization, and often includes in-depth analysis by potential customers of our technology and products and a lengthy review process. Our customers’ evaluation processes often involve a number of factors, many of which are beyond our control. As a result of these factors, the capital investment required to purchase our systems, and the budget cycles of our customers, the time from initial contact with a customer to our receipt of a purchase order can vary significantly. In 2023, these factors resulted in softness in sales of our instruments. Given the length and uncertainty of our sales cycle, we have in the past experienced, and expect in the future to experience, fluctuations in our sales on a period-to-period basis. In addition, any failure to meet customer expectations could result in customers choosing to retain their existing systems, using existing assays not requiring capital equipment, or purchasing systems other than ours.
22
Because a significant portion of our revenue comes from a few large customers, any significant decrease in sales to these customers, due to industry consolidation or otherwise, could harm our operating results.
One customer accounted for greater than 10% of our total revenue for the year ended December 31, 2023, and several other customers accounted for a significant portion of our total revenue. The loss of a significant amount of business from one or more of our major customers would have a material adverse effect on our business. There can be no assurance that there will not be a loss or reduction in business from one or more of our major customers. In addition, we cannot assure that net sales from customers that have accounted for significant net sales in the past, either individually or as a group, will reach or exceed historical levels in any future period.
Our long-term results depend upon our ability to improve existing products and introduce and market new products successfully.
We generally sell our products in industries that are characterized by rapid technological changes, frequent new product introductions and changing industry standards. Accordingly, our business is dependent on the continued improvement of our existing Simoa products and our development of new products utilizing Simoa or other technology we develop or acquire. As we introduce new products or refine, improve or upgrade versions of existing products, we cannot predict the level of market acceptance or the amount of market share these products will achieve, if any. We cannot guarantee that we will not experience material delays in the introduction of new products in the future. In addition, introduction of new products could result in a decrease in revenues from existing products. Consistent with our strategy of offering new products and product refinements, we have invested substantial capital on research and development, and we expect to continue to use a substantial amount of capital for product research and development. Our research and development initiatives can be costly and time-consuming, and they may fail to achieve the intended benefits. If we do not develop new products and product enhancements based on technological innovation on a timely basis, our products may become obsolete over time and our revenues, cash flow, profitability and competitive position will suffer.
Defects or other quality issues in our products could lead to unforeseen costs, product recalls, adverse regulatory actions, negative publicity, and litigation, including product liability claims, any of which could cause customers to decide not to purchase our products, harm our reputation, and negatively affect our sales, operating results and financial condition.
Our Simoa products are complex and may contain undetected errors or defects, especially when first introduced or as new versions or new products are released. We have in the past devoted, and will continue to devote, funding and resources to technology development, quality assurance and manufacturing initiatives designed to ensure or improve quality, such as the assay redevelopment program initiated in 2022. However, there can be no assurance that we will be successful in our efforts to manufacture products at a level of quality necessary for our customers or to avoid our products containing undiscovered defects or quality issues. Defects, errors or quality issues in our products may discourage customers from purchasing our products and could harm our reputation. We may also be subject to warranty claims and litigation involving claims for damages or incur additional costs, in each case due to errors or defects in our products. In addition, if we do not meet industry or quality standards, if applicable, our products may be subject to recall, and products subject to the FDA’s medical device regulations could be required to be recalled under such regulations. A material liability claim, recall or other occurrence that harms our reputation or decreases market acceptance of our products could harm our business and operating results.
Use of our products or services by us or a customer for diagnostic purposes could result in a product liability claim alleging that one of our products contained a design or manufacturing defect that resulted in the failure to adequately perform, leading to death or injury. A product liability claim could result in substantial damages and be costly and time-consuming to defend, either of which could materially harm our business or financial condition. We cannot guarantee that our product liability insurance would adequately protect our assets from the financial impact of defending a product liability claim. Any product liability claim brought against us, with or without merit, could increase our product liability insurance rates or prevent us from securing insurance coverage in the future.
23
Our reliance on distributors for sales of our products outside of the United States could impact our revenue.
We have established distribution agreements for our Simoa instruments and related consumable products with distributors in certain foreign countries, including Australia, Brazil, China, the Czech Republic, India, Hong Kong, Israel, Japan, New Zealand, Qatar, Saudi Arabia, Singapore, South Africa, South Korea, Taiwan and the UAE. We intend to continue to grow our business internationally, and to do so we must attract additional distributors and retain existing distributors to maximize the commercial opportunity for our products. There is no guarantee that we will be successful in attracting or retaining desirable sales and distribution partners or that we will be able to enter into such arrangements on favorable terms. Distributors may not commit the necessary resources to market and sell our products to the level of our expectations or may choose to favor marketing the products of our competitors. If current or future distributors do not perform adequately, or if we are unable to enter into effective arrangements with distributors in particular geographic areas, we may not realize long-term international revenue growth. In addition, if our distributors fail to comply with applicable laws and ethical standards, including anti-bribery laws, this could damage our reputation and could have a significant adverse effect on our business and our revenues.
We generate a substantial portion of our revenue internationally and we expect this will continue in the future; as a result, our business is subject to various risks relating to our international activities, which could adversely affect our business, operating results and financial condition.
For the years ended December 31, 2023, 2022, and 2021, approximately 37%, 38% and 36%, respectively, of our total revenue was generated from customers located outside of North America. We believe that a substantial percentage of our future revenue will continue to come from international sources as we expand our overseas operations and develop opportunities in additional areas. Engaging in international business involves a number of difficulties and risks, including:
| ● | difficulties and costs of staffing and managing foreign operations; |
| ● | required compliance with existing and changing U.S. or foreign regulatory requirements and laws; |
| ● | a shortage of high-quality salespeople and distributors; |
| ● | pricing pressure that we may experience internationally; |
| ● | difficulties in enforcing our intellectual property rights and in defending against third-party threats and intellectual property enforcement actions against us or any of our distributors, suppliers or collaborators; |
| ● | reduced or varied protection for intellectual property rights in some countries; |
| ● | required compliance with anti-bribery laws, such as the U.S. Foreign Corrupt Practices Act, data privacy requirements, such as the GDPR, labor laws and anti-competition regulations; |
| ● | export or import restrictions and supply chain disruptions; |
| ● | laws and business practices favoring local companies; |
| ● | longer payment cycles and difficulties in enforcing agreements and collecting receivables through certain foreign legal systems; |
| ● | the imposition of restrictions on the activities of foreign agents, representatives and distributors; |
| ● | foreign currency exchange rate fluctuations; |
| ● | the imposition of U.S. or international sanctions against a country, company, person or entity with whom we do business that would restrict or prohibit continued business with the sanctioned country, company, person or entity; |
| ● | the impact of political and economic instability and conflict, which could lead to uncertainty and instability in global financial markets; |
| ● | scrutiny of foreign tax authorities which could result in significant fines, penalties and additional taxes being imposed on us; |
| ● | the imposition of new trade restrictions; and |
| ● | potentially adverse tax consequences, tariffs, customs charges, bureaucratic requirements and other trade barriers. |
24
If we are unable to manage these risks effectively, our business, operating results and financial condition will suffer.
We rely on a single contract manufacturer to manufacture and supply our Simoa HD-X instrument and rely on a different single contract manufacturer to manufacture and supply our Simoa SR-X instrument. If either of these manufacturers should fail to perform, or not perform satisfactorily, our ability to supply these instruments would be negatively and adversely affected.
We currently rely on a single contract manufacturer, STRATEC, an analytical and diagnostic systems manufacturer located in Germany, to manufacture and supply all of our Simoa HD-X instruments. In addition, we currently rely on a single contract manufacturer, Paramit, a contract manufacturer located in California, to manufacture and supply all of our SR-X instruments. Since our contract with STRATEC does not commit them to supply quantities beyond the amounts included in our forecasts and our contract with Paramit does not commit them to carry inventory or make available any particular quantities, we may not be able to obtain adequate supplies in a timely manner or on commercially reasonable terms. If either of these manufacturers were not able to supply instruments, our business would be harmed.
In the event it becomes necessary to utilize a different contract manufacturer for the HD-X instrument or the SR-X instrument, we would experience additional costs, delays and difficulties in doing so as a result of needing to identify and enter into an agreement with a new supplier as well as needing to prepare such new supplier to meet the logistical requirements associated with manufacturing our instruments, and our business would suffer. We may also experience additional costs and delays in the event we need access to or rights under any intellectual property of STRATEC.
In addition, certain of the components used in our instruments are sourced by these manufacturers from limited or sole suppliers. If they were to lose such suppliers, there can be no assurance that they would be able to identify or enter into agreements with alternative suppliers on a timely basis on acceptable terms, if at all. An interruption in our ability to sell and deliver instruments to customers could occur if our manufacturers encounter delays or difficulties in securing these components, or if the quality of the components supplied do not meet specifications, or if they cannot then obtain an acceptable substitute. If any of these events occur, our business and operating results could be harmed.
We rely on a limited number of suppliers or, in some cases, one supplier, for some of our materials and components used in our consumable products and services and our SP-X instrument, and we may not be able to find replacements or immediately transition to alternative suppliers if any of these suppliers fail to perform, which could have a material adverse effect on our business, financial condition, results of operations and reputation.
We rely on limited or sole suppliers for certain reagents and other materials and components that are used in our consumable products and services and in our SP-X instrument. While we have long-term contracts with some critical suppliers, we do not have contracts with all suppliers and instead rely on periodically forecasting our needs for such materials and entering into standard purchase orders with our suppliers. In addition, our use of many of the materials used in our consumable products is limited to research use only. As we expand into diagnostic applications for our products, we will need to secure diagnostic rights to such materials. If we were to lose suppliers or were unable to secure required rights for materials from suppliers, there can be no assurance that we will be able to identify or enter into agreements with alternative suppliers on a timely basis and on acceptable terms, if at all. An interruption in our operations could occur if we encounter delays or difficulties in securing these materials or any required rights to these materials, if the quality of the materials supplied do not meet our requirements, or if we cannot then obtain an acceptable substitute. The time and effort required to qualify a new supplier and ensure that the new materials provide the same or better quality results could result in significant additional costs. Any such interruption could significantly affect our business, financial condition, results of operations and reputation.
25
We could be adversely affected by violations of the U.S. Foreign Corrupt Practices Act and other worldwide anti-bribery laws by us or our agents.
We are subject to the U.S. Foreign Corrupt Practices Act (the “FCPA”), which prohibits companies and individuals from corruptly making payments, directly or indirectly through third parties, to non-U.S. government officials for the purpose of obtaining or retaining business or securing any other improper advantage. We are also subject to the FCPA’s accounting provisions, which require us to keep accurate books and records and to maintain a system of internal accounting controls sufficient to assure management’s control, authority and responsibility over our assets. Our reliance on independent distributors to sell our products internationally demands a high degree of vigilance in maintaining our policy against participation in corrupt activity, because there are circumstances under which we could be held responsible for their actions. Other U.S. companies in the medical device and pharmaceutical fields have faced criminal penalties under the FCPA for allowing their distributors and other third parties to deviate from appropriate practices in doing business with these individuals. We are also subject to similar anti-bribery laws in the jurisdictions in which we operate, including the United Kingdom’s Bribery Act of 2010, which also prohibits commercial bribery and makes it a crime for companies to fail to prevent bribery. These laws are complex and far-reaching in nature, and any violations of these laws, or allegations of such violations, could disrupt our operations, involve significant management distraction, involve significant costs and expenses, including legal fees, and could result in a material adverse effect on our business, prospects, financial condition, or results of operations. We could also incur severe penalties, including criminal and civil penalties, disgorgement and other remedial measures.
The life sciences research and diagnostic markets are highly competitive. If we fail to effectively compete, our business, financial condition and operating results will suffer.
We face significant competition in the life sciences research and diagnostic markets. We currently compete with both established and early-stage companies that design, manufacture and market systems and consumable supplies. Many of our current competitors have competitive advantages over us, including:
| ● | greater name and brand recognition; |
| ● | substantially greater financial and human resources; |
| ● | broader product lines; |
| ● | larger sales forces and more established distributor networks; |
| ● | more substantial intellectual property portfolios; |
| ● | larger and more established customer bases and relationships; and |
| ● | better established, larger scale and lower cost manufacturing capabilities. |
We cannot guarantee that our products will compete favorably or that we will be successful in the face of increasing competition from new products and technologies introduced by our existing competitors or new companies entering our markets. In addition, we cannot guarantee that our competitors do not have or will not develop products or technologies that currently or in the future will enable them to produce competitive products with greater capabilities or at lower costs than ours. Any failure to compete effectively could materially and adversely affect our business, financial condition and operating results.
Integrating any business, product or technology we acquire can be expensive and time-consuming and can disrupt and adversely affect our ongoing business, including product sales, and distract our management.
We may acquire other businesses, products or technologies as well as pursue strategic alliances, joint ventures, technology licenses or investments in complementary businesses. Our ability to successfully integrate any business, product or technology we acquire depends on a number of factors, including, but not limited to, our ability to:
| ● | minimize the disruption and distraction of our management and other employees in connection with the integration of any acquired business, product or technology; |
| ● | avoid acquisition of unanticipated liabilities related to acquired companies; |
| ● | maintain and increase sales of our existing products; |
26
| ● | establish or manage the transition of the manufacture and supply of any acquired product; |
| ● | identify and add the necessary sales, marketing, manufacturing, regulatory and other related personnel, capabilities and infrastructure that are required to successfully integrate any acquired business, product or technology; |
| ● | manage the transition and migration of acquired personnel and all commercial, financial, legal, regulatory and other pertinent information relating to any acquired business, product or technology; |
| ● | comply with legal, regulatory and contractual requirements applicable to any acquired business, product or technology; and |
| ● | maintain and extend intellectual property protection for any acquired product or technology. |
If we are unable to perform the above functions or otherwise effectively integrate any acquired businesses, products or technologies, our business, financial condition and operating results will suffer.
Also, the anticipated benefit of any acquisition may not materialize. Future acquisitions or dispositions could result in potentially dilutive issuances of our equity securities, the incurrence of debt, contingent liabilities or amortization expenses or write-offs of goodwill, any of which could harm our financial condition. We cannot predict the number, timing or size of future joint ventures or acquisitions, or the effect that any such transactions might have on our operating results.
Risks Related to Government Regulation and Diagnostic Product Reimbursement
If the FDA determines that our products are subject to regulation as medical devices, if the FDA modifies its regulations to require that our LDTs are subject to regulation as devices, or if we seek to market our products for clinical diagnostic or health screening use, we will be required to obtain regulatory clearance(s) or approval(s). Any such regulatory process would be expensive, time-consuming and uncertain both in timing and in outcome.
We focused initially on the life sciences research market. This includes offering products for use by laboratories associated with academic and governmental research institutions, as well as pharmaceutical, biotechnology and contract research companies. Accordingly, the majority of our products are labeled as “Research Use Only”. While we focused initially on the life sciences research market and RUO products only, our strategy includes expanding our product line to encompass products that are intended to be used for the diagnosis of disease, including LDTs and IVD devices, either alone or in collaboration with third parties. IVD products are subject to regulation by the FDA, or comparable international agencies, as medical devices including requirements for regulatory clearance or approval of such products before they can be marketed.
The process of obtaining regulatory clearances to market a medical device can be costly and time consuming, and we or our collaborators may not be able to obtain these clearances or approvals on a timely basis, if at all. In general, the FDA permits commercial distribution of a new medical device only after the device has received clearance under Section 510(k) of the Federal Food, Drug and Cosmetic Act (“FDCA”), or is the subject of an approved PMA, unless the device is specifically exempt from those requirements. The FDA will clear marketing of a lower risk medical device through the 510(k) process if the manufacturer demonstrates that the new product is substantially equivalent to a legally marketed predicate device, which can include pre-amendment, 510(k)-exempt, 510(k) cleared products, or PMA-approved products that have subsequently been down-classified. If the FDA determines that the device is not “substantially equivalent” to a predicate device, or if the device is novel, it is automatically classified into Class III, and the device sponsor must then fulfill the much more rigorous premarketing requirements of the PMA approval process, or seek classification of the device through the de novo classification process. The PMA process is more costly, lengthy and uncertain than the 510(k) clearance process. A PMA application must be supported by extensive data, including, but not limited to, technical, preclinical, clinical trial, manufacturing and labeling data, to demonstrate to the FDA’s satisfaction the safety and efficacy of the device for its intended use.
If any of our products are subject to medical device regulation, we would be subject to a substantial number of additional requirements for medical devices, including establishment registration, device listing, QSRs —which cover the design, testing, production, control, quality assurance, labeling, packaging, servicing, sterilization (if required), and storage and shipping of medical devices (among other activities) —product labeling, advertising, recordkeeping,
27
post-market surveillance, post-approval studies, adverse event reporting, and correction and removal (recall) regulations. One or more of the products we may develop using our technology may also require clinical trials in order to generate the data required for a PMA, de novo classification request or 510(k) premarket notification. Complying with these requirements may be time-consuming and expensive. We may be required to expend significant resources to ensure ongoing compliance with the FDA regulations. Failure to comply with these requirements may subject us to a range of enforcement actions, such as warning letters, injunctions, civil monetary penalties, criminal prosecution, recall and/or seizure of products, and revocation of marketing authorization, as well as significant adverse publicity. If we fail to obtain, or experience significant delays in obtaining, regulatory approvals for IVD products, such products may not be able to be launched or successfully commercialized in a timely manner, or at all.
LDTs are a subset of IVD tests that are offered as services by CLIA-certified high complexity clinical laboratories and designed, manufactured and used within a single laboratory. In July 2022, we launched an LDT to quantitatively measure p-Tau 181 in plasma as an aid in diagnostic evaluation of Alzheimer’s disease, and in January 2023, we launched an LDT to quantitatively measure NfL in serum as an aid in the evaluation of individuals for possible neurodegenerative conditions or other causes of neuronal or central nervous system damage. The FDA maintains that LDTs are medical devices and has, for the most part, exercised enforcement discretion for most LDTs, meaning that the FDA has not required LDTs to obtain premarket approval or clearance or comply with post-market medical device requirements. A significant change in the way that the FDA regulates LDTs could affect our business. Most recently, in September 2023, FDA announced a proposed rule regarding LDTs that would make explicit that in vitro diagnostic products are devices under the Federal Food, Drug, and Cosmetic Act, including when the manufacturer is a laboratory. The proposed rule also describes a policy under which FDA would provide greater oversight of LDTs by phasing out its general enforcement discretion approach, and phase in medical device regulation, for LDTs over a period of four years. FDA requested comment on several aspects of its proposal and approach, including whether certain types of LDTs should remain under enforcement discretion. Currently FDA’s target date for final action on this rule is April 2024, but we cannot predict the ultimate timing or form of FDA guidance or regulation, legislative action or their potential impact. Any new regulatory approach for LDTs by the FDA, including as described in the September 2023 proposed rule, would likely lead to an increased regulatory burden, including additional costs and delays in introducing new tests, and potentially a requirement for our current LDTs to receive premarket clearance or approval from FDA to continue offering them after the enforcement discretion phaseout is complete. Any new regulatory approach could also result in our tests being removed from the market if we are not able to secure regulatory clearance or approval from FDA. FDA’s rule could also have impacts on our business more broadly, given that many of our customers would be subject to additional regulation and delays, which could potentially affect the development of new diagnostics that incorporate our instruments or consumables. This also may increase costs and regulatory burdens on laboratories that develop LDTs, thereby reducing the financial incentive for laboratories to develop new LDTs or invest in instruments, which could reduce demand for our instruments and our other products.
Foreign jurisdictions have laws and regulations similar to those described above, which may adversely affect our ability to market our products as planned in such countries. The number and scope of these requirements are increasing. As in the United States, the cost and time required to comply with regulatory requirements may be substantial, and there is no guarantee that we will obtain the necessary authorization(s) required to make our products commercially viable. In addition, the imposition of foreign requirements may also have a material adverse effect on the commercial viability of our operations.
Our products may in the future be subject to product recalls that could harm our reputation, business and financial results.
The FDA and similar foreign governmental authorities have the authority to require the recall of commercialized products, including RUO products, in the event of material deficiencies or defects in design or manufacture. In the case of the FDA, the authority to require a recall of a medical device must be based on an FDA finding that there is a reasonable probability that the device would cause serious injury or death. Manufacturers may, under their own initiative, recall a product if any material deficiency in a device is found. A government-mandated or voluntary recall by us or one of our distributors could occur as a result of component failures, manufacturing errors, design or labeling defects or other deficiencies and issues. Recalls of any of our products would divert managerial and financial resources and have an adverse effect on our financial condition and results of operations.
28
U.S. legislative, FDA or global regulatory reforms may make it more difficult and costly for us to obtain any required regulatory approval of our product candidates and to manufacture, market and distribute our products after approval is obtained.
From time to time, legislation is drafted and introduced in Congress that could significantly change the statutory provisions governing the regulatory approval, manufacture and marketing of regulated products or the reimbursement thereof. For example, in December 2022, Congress enacted the Food and Drug Omnibus Reform Act of 2022 (“FDORA”). FDORA reauthorized the FDA to collect device user fees and contained substantive amendments to the device provisions of the FDCA, including imposing new cybersecurity and clinical trial requirements for devices. Congress has also considered, but not yet passed, legislation to impose a new FDA regulatory framework for all diagnostics, including IVD devices and LDTs. Any new regulations or revisions or reinterpretations of existing regulations may impose additional costs or lengthen review times of future products. In addition, FDA regulations and guidance are often revised or reinterpreted by the agency in ways that may significantly affect our business and our products. For example, in September 2023, the FDA issued a proposed rule to change the FDA’s regulatory approach to LDTs. Under the proposed rule, FDA would phase out its current enforcement discretion approach for LDTs, and phase in medical device regulation, over a period of four years. It is impossible to predict whether legislative changes will be enacted or FDA regulations, guidance or interpretations changed, and what the impact of such changes, if any, may be. Any change in the laws or regulations that govern the clearance and approval processes relating to our current and future products could make it more difficult and costly to obtain clearance or approval for new products, or to produce, market and distribute existing products. Significant delays in receiving clearance or approval, or the failure to receive clearance or approval for our new products would have an adverse effect on our ability to expand our business.
In addition, in the E.U. new regulations recently entered into force that result in greater regulation of medical devices and IVDs. The IVD Regulation is significantly different from the IVD Directive that it replaces in that it ensures that the new requirements apply uniformly and on the same schedule across the member states, includes a risk-based classification system and increases the requirements for conformity assessment. The CE registration for Uman’s NfL ELISA assay kit for cerebral spinal fluid was approved in March 2014 under the IVD Directive. Under the IVD Directive the assay is classified as a general IVD product, and required self-certification with no involvement of a notified body/authority. The IVD Regulation introduces a new classification system for IVDs and assessment by a notified body is required for class B, C and D products. Uman’s NfL ELISA assay kit for CSF is classified as a class B product and must fully comply with (and have a CE mark issued under) the IVD Regulation by May 2027 (subject to proposed extension of the transitional periods in the IVD Regulation). The new requirements include an ISO 13485 certification of the quality system (which Uman received in July 2018) and increased technical evidence and follow-up of performance of the specific product (e.g. clinical evidence and post-market activities). The work to evaluate and to meet the new technical requirements is on-going.
Our failure to continue to comply with applicable foreign regulatory requirements, including those administered by authorities of the European Economic Area (“EEA”) countries, could result in enforcement actions against us, including refusal, suspension or withdrawal of our CE Certificates of Conformity by our notified body, which could impair our ability to market products in the EEA in the future.
If we do not comply with governmental regulations applicable to our CLIA-certified laboratory, we may not be able to continue our Accelerator laboratory operations or continue offering our LDTs.
CLIA is a federal law that regulates clinical laboratories that perform examination of human specimens for the purpose of providing information for the diagnosis, prevention or treatment of any disease or impairment of, or the assessment of health of, human beings. The operation of our CLIA-certified laboratory is subject to regulation by numerous federal, state and local governmental authorities in the United States. This laboratory holds a CLIA certificate of compliance for high-complexity testing and is licensed by California, Maryland, Massachusetts, Pennsylvania and Rhode Island, and we may obtain other state licenses if required in the future. Failure to comply with federal or state regulations or changes in those regulatory requirements could result in a substantial curtailment or even prohibition of the operations of our laboratory and could have an adverse effect on our business. To maintain CLIA certification, laboratories are subject to survey and inspection every two years. Moreover, CLIA inspectors may make unannounced
29
inspections of these laboratories. If we were to lose our CLIA certification or any required state licenses, whether as a result of a revocation, suspension or limitation, it could have a material adverse effect on our business.
We expect to rely on third parties in conducting any required future studies of diagnostic products that may be required by the FDA or other regulatory authorities, and those third parties may not perform satisfactorily.
We do not have the ability to independently conduct clinical trials or other studies that may be required to obtain FDA and other regulatory clearance or approval for future diagnostic products. Accordingly, we expect that we would rely on third parties, such as clinical investigators, contract research organizations, consultants, and collaborators to conduct such studies if needed. For example, we are currently working with the Alzheimer’s Drug Discovery Foundation and the Global Alzheimer’s Platform Foundation on prospective clinical trials for our assays. Our reliance on these third parties for clinical and other development activities would reduce our control over these activities. If these third parties do not successfully carry out their contractual duties or regulatory obligations or meet expected deadlines, if the third parties need to be replaced or if the quality or accuracy of the data they obtain is compromised, we may not be able to obtain regulatory clearance or approval.
If diagnostic procedures that are enabled by our technology are subject to unfavorable pricing regulations or third-party coverage and reimbursement policies, our business could be harmed.
The ability of us, our customers or our collaborators to commercialize diagnostic tests based on our technology, including our recently launched LDTs, will depend in part on the extent to which coverage and reimbursement for these tests will be available from government health care programs, private health insurers and other third-party payors. In the United States, the principal decisions about reimbursement for new technologies are often made by CMS. Private payors often follow CMS’s reimbursement policies to a substantial degree. It is difficult to predict what CMS will decide with respect to reimbursement. A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and third-party payors have attempted to control costs by limiting coverage and the amount of payments for particular products and procedures. We cannot be sure that coverage will be available for any diagnostic tests based on our technology, and, if coverage is available, the level of reimbursement. Payor coverage and reimbursement decisions may impact the demand for those tests. If coverage is not available or the reimbursement amount is inadequate, any tests for which marketing authorization is received may not be able to be successfully commercialized.
Risks Related to Our Operations
We depend on our information technology systems, and any failure of these systems could harm our business.
We depend on information technology and telecommunications systems to operate our business. Our enterprise software systems affect a broad range of business processes and functional areas, including, for example, systems handling human resources, accounting, manufacturing, inventory control, financial controls and reporting, sales administration, and other infrastructure operations. We maintain preventative and detective security controls and seek to enhance such controls by, for example, augmenting the monitoring and alerting functions, network design, and automatic countermeasure operations of our technical systems. We also periodically assess the adequacy of our hardware and systems and are planning to upgrade hardware and systems where appropriate. These information technology and telecommunications systems support a variety of functions, including manufacturing operations, quality control, customer service support, finance, and other general administrative activities.
Information technology and telecommunications systems are vulnerable to damage from a variety of sources, including telecommunications, systems or network failures, malicious human acts, and natural disasters. Moreover, despite network security and back-up measures, some of our servers are potentially vulnerable to physical or electronic break-ins, computer viruses, and similar disruptive problems. Despite the precautionary measures we have taken to prevent unanticipated problems that could affect our information technology and telecommunications systems, those measures may be inadequate and failures or significant downtime of our information technology or telecommunications systems or those used by our third-party suppliers could prevent us from operating our business and managing the administrative aspects of our business. Loss of data or a material delay in our access to our data due to a security breach
30
or other interruption could also prevent us from operating our business. Any disruption or loss of information technology or telecommunications systems on which critical aspects of our operations depend could have an adverse effect on our business.
Cybersecurity breaches, loss of data and other disruptions could compromise sensitive information related to our business or prevent us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation.
In the ordinary course of our business, we collect and store sensitive data, and intellectual property and proprietary business information owned or controlled by ourselves or our customers. This data encompasses a wide variety of business-critical information including research and development information, operational information, commercial information, and business and financial information. We face four primary risks relative to protecting this critical information: loss of access; inappropriate disclosure; inappropriate modification; and inadequate monitoring of our controls over the first three risks.
The secure processing, storage, maintenance, and transmission of this critical information is vital to our operations and business strategy, and we devote significant resources to protecting such information. Although we take measures to protect sensitive information from unauthorized access or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers or viruses, breaches, interruptions due to employee error, malfeasance, faulty password management, lapses in compliance with privacy and security mandates, or other disruptions. The risk of a security breach or disruption, particularly through cyber-attack or cyber intrusion, including by computer hackers, foreign governments, and cyber terrorists, has generally increased as the number, intensity and sophistication of attempted attacks and intrusions from around the world have increased. Our IT networks and related systems are essential to the operation of our business and our ability to perform day-to-day operations. Although we make efforts to maintain the security and integrity of these types of IT networks and related systems, and we have implemented various measures to manage the risk of a security breach or disruption, no security measure is infallible and there can be no assurance that our security efforts and measures will be effective or that attempted security breaches or disruptions will not be successful or damaging. Our information technology systems may have vulnerabilities, and we may not have the resources or technical sophistication to anticipate or prevent rapidly evolving types of cyberattacks, such as ransomware attacks. Although we have experienced cybersecurity incidents from time to time that have not had a material adverse effect on our business, financial condition, or results of operations, there can be no assurance that a cyber-attack, security breach, or other cybersecurity incident will not have a material adverse effect on us in the future. A significant cyber incident, including system failure, security breach, disruption by malware or other damage, could interrupt or delay our operations, result in a violation of applicable cybersecurity and privacy and other laws, damage our reputation, cause a loss of customers, or expose sensitive customer data, or give rise to monetary fines and other penalties, which could be significant.
Third parties may attempt to fraudulently induce employees or other persons into disclosing usernames, passwords, or other sensitive information, which may in turn be used to access our information systems, commit identity theft or carry out other unauthorized or illegal activities. Any such breach could compromise our networks and the information stored there could be accessed, publicly disclosed, lost, or stolen. We engage third-party vendors and service providers to store and otherwise process some of our data, including sensitive and personal information. Our vendors and service providers may also be the targets of the risks described above, including cyberattacks, malicious software, phishing schemes, and fraud. Our ability to monitor our vendors and service providers’ data security is limited, and third parties may be able to circumvent any security measures, resulting in the unauthorized access to, misuse, disclosure, loss or destruction of our data, including sensitive and personal information, and disruption of our or third-party service providers’ systems. We and our third-party service providers may face difficulties in identifying, or promptly responding to, potential security breaches and other instances of unauthorized access to, or disclosure or other loss of, information. Any hacking or other attack on our or our third-party service providers’ or vendors’ systems, and any unauthorized access to, or disclosure or other loss of, information suffered by us or our third-party service providers or vendors, or the perception that any of these have occurred, could result in legal claims or proceedings, loss of intellectual property, liability under laws that protect the privacy of personal information, negative publicity, disruption of our operations and damage to our reputation, which could divert our management’s attention from the operation of our business and materially and adversely affect our business, revenues and competitive position.
31
Any security breach or interruption, as well as any action by us or our employees or contractors that might be inconsistent with the rapidly evolving data privacy and security laws and regulations applicable within the United States and elsewhere where we conduct business, could result in enforcement actions by state or federal governments or foreign governments, liability or sanctions under data privacy laws that protect personally identifiable information, regulatory penalties, other legal proceedings such as but not limited to private litigation, the incurrence of significant remediation costs, disruptions to our development programs, business operations and collaborations, diversion of management efforts and damage to our reputation. Because of the rapidly moving nature of technology and the increasing sophistication of cybersecurity threats, our measures to prevent, respond to and minimize such risks may be unsuccessful.
In addition, our insurance may be insufficient to cover our losses resulting from cyber-attacks, breaches, or other interruptions, and any incidents may result in loss of, or increased costs of, such insurance. The successful assertion of one or more large claims against us that exceed available insurance coverage, the occurrence of changes in our insurance policies, including premium increases or the imposition of large deductible or co-insurance requirements, or denials of coverage, could have a material adverse effect on our business, including our financial condition, results of operations and reputation.
We are currently subject to, and may in the future become subject to additional, U.S. federal and state and international laws and regulations imposing obligations on how we collect, store and process personal information. Our actual or perceived failure to comply with such obligations could harm our business. Ensuring compliance with such laws could also impair our efforts to maintain and expand our future customer base, and thereby decrease our revenue.
In the ordinary course of our business we collect, store, transfer, use or process sensitive data, including personally identifiable information of employees, and intellectual property and proprietary business information owned or controlled by ourselves and other parties. The secure processing, storage, maintenance, and transmission of this critical information are vital to our operations and business strategy. We are, and may increasingly become, subject to various laws and regulations, as well as contractual obligations, relating to data privacy and security in the jurisdictions in which we operate. The regulatory environment related to data privacy and security is increasingly rigorous, with new and constantly changing requirements applicable to our business, and enforcement practices are likely to remain uncertain for the foreseeable future. These laws and regulations may be interpreted and applied differently over time and from jurisdiction to jurisdiction, and it is possible that they will be interpreted and applied in ways that may have a material adverse effect on our business, financial condition, results of operations and prospects.
In the United States, various federal and state regulators, including governmental agencies like the Federal Trade Commission, have adopted, or are considering adopting, laws and regulations concerning personal information and data security. Certain state laws may be more stringent or broader in scope, or offer greater individual rights, with respect to personal information than federal, international, or other state laws, and such laws may differ from each other, all of which may complicate compliance efforts. For example, the California Consumer Privacy Act (the “CCPA”), which increases privacy rights for California residents and imposes obligations on companies that process their personal information, came into effect on January 1, 2020. Among other things, the CCPA requires covered companies to provide disclosures to California consumers regarding the processing of their personal data, as well as data protection and privacy rights, including the ability to opt-out of certain sales or sharing of personal information. The CCPA provides for civil penalties for violations, as well as a private right of action for certain data breaches that result in the loss of personal information. This private right of action may increase the likelihood of, and risks associated with, data breach litigation. In November 2020, California also passed the California Privacy Rights Act (the “CPRA”), which became effective on January 1, 2023 and significantly expands the CCPA, including by introducing additional obligations such as data minimization and storage limitations and granting additional rights to consumers. More recently, other states, including Connecticut, Colorado, Utah and Virginia have passed comprehensive state data privacy laws, and states like Washington and Nevada have enacted consumer health privacy laws. Most of these laws are enforced by state attorneys general, but there is the potential for private actions by plaintiffs in some circumstances under certain laws. In addition, laws in all 50 U.S. states require businesses to provide notice to consumers whose personal information has been disclosed as a result of a data breach. State laws are changing rapidly and there is discussion in the U.S. Congress of a new comprehensive federal data privacy law to which we would become subject if it is enacted. These and future laws and regulations may increase our compliance costs and potential liability.
32
Furthermore, regulations promulgated pursuant to HIPAA establish privacy and security standards that limit the use and disclosure of individually identifiable health information (known as “protected health information”) and require the implementation of administrative, physical, and technological safeguards to protect the privacy of protected health information and ensure the confidentiality, integrity and availability of electronic protected health information. Determining whether protected health information has been handled in compliance with applicable privacy standards and our contractual obligations can require complex factual and statistical analyses and may be subject to changing interpretation. Although we take measures to protect sensitive data from unauthorized access, use or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers or viruses or breached due to employee error, malfeasance or other malicious or inadvertent disruptions. Any such breach or interruption could compromise our networks and the information stored there could be accessed by unauthorized parties, manipulated, publicly disclosed, lost, or stolen. Any such access, breach or other loss of information could result in legal claims or proceedings, and liability under federal or state laws that protect the privacy of personal information, such as the HIPAA, HITECH, and regulatory penalties. Notice of breaches must be made to affected individuals, the Secretary of the Department of Health and Human Services, and for extensive breaches, notice may need to be made to the media. Such a notice could harm our reputation and our ability to compete.
Outside of the United States, many countries have privacy and data security laws and regulations concerning the collection and use of personal data, including but not limited to the GDPR and China’s Personal Information Protection Law (“PIPL”). The GDPR, which governs the collection and use of personal data in the E.U. and is wide-ranging in scope, imposes several requirements relating to the consent of the individuals to whom the personal data relates, the information provided to the individuals, the security and confidentiality of the personal data, data breach notification and the use of third-party processors in connection with the processing of the personal data. The GDPR also imposes strict rules on the transfer of personal data out of the E.U. to the United States, enhances enforcement authority and imposes large penalties for noncompliance, including the potential for fines of up to €20 million or 4% of the annual global revenues of the infringer, whichever is greater. While we have taken steps to comply with the GDPR, including reviewing our security procedures and entering into data processing agreements with relevant contractors, we cannot guarantee that our compliance efforts will be fully successful.
Risks Related to Intellectual Property
If we are unable to protect our intellectual property, our ability to maintain any technological or competitive advantage over our competitors and potential competitors may be reduced, and our business may be harmed.
We rely on patent protection as well as trademark, copyright, trade secret and other intellectual property rights protection and contractual restrictions to protect our proprietary technologies, all of which provide limited protection and may not adequately protect our rights or permit us to gain or keep any competitive advantage. If we fail to protect our intellectual property, third parties may be able to compete more effectively against us, we may lose our technological or competitive advantage, or we may incur substantial litigation costs in our attempts to recover or restrict use of our intellectual property.
Our currently pending or future patent applications may not result in granted patents, and we cannot predict how long it will take for such patents to be granted. It is possible that, for any of our patents that have granted or that may grant in the future, others will design around our patented technologies. Further, other parties may challenge any patents granted to us and courts or regulatory agencies could hold our patents to be invalid or unenforceable. We may not be successful in defending challenges made against our patents and patent applications. Any successful third-party challenge to our patents could result in the unenforceability or invalidity of such patents, or to such patents being interpreted narrowly or otherwise in a manner adverse to our interests. Our ability to establish or maintain a technological or competitive advantage over our competitors may be diminished because of these uncertainties. For these and other reasons, our intellectual property may not provide us with any competitive advantage. To the extent our intellectual property offers inadequate protection, or is found to be invalid or unenforceable, we would be exposed to a greater risk of direct competition. If our intellectual property does not provide adequate coverage over our products and protection against our competitors’ products, our competitive position could be adversely affected, as could our business.
33
In addition to pursuing patents on our technology, we also rely upon trademarks, trade secrets, copyrights and unfair competition laws, as well as license agreements and other contractual provisions, to protect our intellectual property and other proprietary rights. Despite these measures, any of our intellectual property rights could be challenged, invalidated, circumvented or misappropriated. In addition, we take steps to protect our intellectual property and proprietary technology by entering into confidentiality agreements and intellectual property assignment agreements with our employees, consultants, corporate partners and, when needed, our advisors. Such agreements may not be enforceable or may not provide meaningful protection for our trade secrets or other proprietary information in the event of unauthorized use or disclosure or other breaches of the agreements, and we may not be able to prevent such unauthorized disclosure. Moreover, if a party having an agreement with us has an overlapping or conflicting obligation to a third party, our rights in and to certain intellectual property could be undermined. Monitoring unauthorized disclosure is difficult, and we do not know whether the steps we have taken to prevent such disclosure are, or will be, adequate. If we were to enforce a claim that a third party had illegally obtained and was using our trade secrets, it would be expensive and time-consuming, the outcome would be unpredictable, and any remedy may be inadequate. In addition, courts outside of the United States may be less willing to protect trade secrets.
Some of our owned and in-licensed intellectual property has been discovered through government-funded programs and thus is subject to federal regulations such as “march-in” rights, certain reporting requirements, and a preference for U.S. industry. Compliance with such regulations may limit our exclusive rights, subject us to expenditure of resources with respect to reporting requirements, and limit our ability to contract with non-U.S. manufacturers.
Some of the intellectual property rights we own and have in-licensed have been generated through the use of U.S. government funding and are therefore subject to certain federal regulations. For example, some of the issued U.S. patents we own and all of the intellectual property rights licensed to us under our license agreement with Tufts have been generated using U.S. government funds. As a result, the U.S. government has certain rights to intellectual property embodied in our current or future products pursuant to the Bayh-Dole Act of 1980. These U.S. government rights in certain inventions developed under a government-funded program include a non-exclusive, non-transferable, irrevocable worldwide license to use inventions for any governmental purpose. In addition, the U.S. government has the right to require us to grant exclusive, partially exclusive, or non-exclusive licenses to any of these inventions to a third party if the government determines that: (i) adequate steps have not been taken to commercialize the invention; (ii) government action is necessary to meet public health or safety needs; or (iii) government action is necessary to meet requirements for public use under federal regulations (also referred to as “march-in rights”). The U.S. government also has the right to take title to these inventions if we fail, or the applicable licensor fails, to disclose the invention to the government, elect title, and file an application to register the intellectual property within specified time limits. In addition, the U.S. government may acquire title to these inventions in any country in which a patent application is not filed within specified time limits. Intellectual property generated under a government funded program is also subject to certain reporting requirements, compliance with which may require us, or the applicable licensor, to expend substantial resources. In addition, the U.S. government requires that any products embodying the subject invention or produced through the use of the subject invention be manufactured substantially in the United States. The manufacturing preference requirement can be waived if the owner of the intellectual property can show that reasonable but unsuccessful efforts have been made to grant licenses on similar terms to potential licensees that would be likely to manufacture substantially in the United States or that under the circumstances domestic manufacture is not commercially feasible. This preference for U.S. manufacturing may limit our ability to license the applicable patent rights on an exclusive basis under certain circumstances.
Our Simoa bead-based technology is licensed to us by Tufts University. Any loss of our rights to this technology or other technologies we license could prevent us from selling our products.
Our Simoa bead-based technology is licensed exclusively to us from Tufts University (“Tufts”). We do not own the patents that underlie this license. Our rights to use this technology and employ the inventions claimed in the licensed patents are subject to the continuation of and compliance with the terms of the license. Our principal obligations under our license agreement with Tufts are as follows:
| ● | making royalty payments; |
34
| ● | making milestone payments; |
| ● | paying annual maintenance fees for the underlying patents; |
| ● | using commercially reasonable efforts to develop and sell a product using the licensed technology and developing a market for such product; |
| ● | paying and/or reimbursing fees related to prosecution, maintenance and enforcement of patent rights; and |
| ● | providing certain reports. |
If we breach any of these obligations, Tufts may have the right to terminate the license, which could result in our being unable to develop, manufacture and sell products using our Simoa bead-based technology or a competitor gaining access to the Simoa technology. Termination of our license agreement with Tufts would have a material adverse effect on our business.
In addition, we are a party to a number of other agreements that include licenses to intellectual property, including non-exclusive licenses. We expect that we may need to enter into additional license agreements in the future. Our business could suffer materially and adversely, for example, if any current or future licenses terminate, if the licensors fail to abide by the terms of the license, if the licensed patents or other rights are found to be invalid or unenforceable, or if we are unable to enter into necessary licenses on acceptable terms.
If we or any of our partners are sued for infringing intellectual property rights of third parties, the resulting litigation would be costly and time-consuming, and an unfavorable outcome in that litigation could have a material adverse effect on our business.
Our success also depends on our ability to develop, manufacture, market and sell our products and perform our services without infringing upon the proprietary rights of third parties. As part of a business strategy to impede our successful commercialization and entry into new markets, competitors have claimed, and may claim in the future, that our products and/or services infringe their intellectual property rights and have suggested, and may suggest in the future, that we enter into license agreements. We believe any such claims made to date are without merit. However, even if such claims are without merit, we could incur substantial costs and divert the attention of our management and technical personnel in defending ourselves against claims of infringement made by third parties or settling such claims. Any adverse ruling by a court or administrative body, or perception of an adverse ruling, may have a material adverse impact on our ability to conduct our business and our finances. Moreover, third parties making claims against us may be able to obtain injunctive relief against us, which could block our ability to offer one or more products or services and could result in a substantial award of damages against us. In addition, since we sometimes indemnify customers, collaborators or licensees, we may have additional liability in connection with any infringement or alleged infringement of third party intellectual property.
Because patent applications can take many years to issue, there may be pending applications, some of which are unknown to us, that may result in issued patents upon which our products or proprietary technologies may infringe. Moreover, we may fail to identify issued patents of relevance or incorrectly conclude that an issued patent is invalid or not infringed by our technology or any of our products. There is a substantial amount of litigation involving patent and other intellectual property rights in our industry. If a third party claims that we or any of our licensors, customers or collaboration partners infringe upon a third party’s intellectual property rights, we may have to:
| ● | seek to obtain licenses that may not be available on commercially reasonable terms, if at all; |
| ● | abandon any infringing product or redesign our products or processes to avoid infringement; |
| ● | pay substantial damages, including, in exceptional cases, treble damages and attorneys’ fees; |
| ● | pay substantial royalties or fees or grant cross-licenses to our technology; or |
| ● | defend litigation or administrative proceedings that may be costly whether we win or lose, and which could result in a substantial diversion of our financial and management resources. |
35
We may be involved in lawsuits to protect or enforce our patents or the patents of our licensors, which could be expensive, time-consuming and unsuccessful.
Competitors may infringe our patents or the patents that we license. In the event of infringement or unauthorized use, we may file one or more infringement lawsuits. Patent litigation can be very costly and time-consuming, and the outcome is uncertain. In addition, if we or any of our partners were to initiate legal proceedings against a third party to enforce a patent covering one of our products or services, the defendant in such litigation could counterclaim that our patent is invalid and/or unenforceable. In patent litigation, defendant counterclaims alleging invalidity and/or unenforceability are commonplace. The outcome following legal assertions of invalidity and unenforceability is unpredictable. If a defendant were to prevail on a legal assertion of invalidity and/or unenforceability, we would lose at least part, and perhaps all, of the challenged patent. Such a loss of patent protection could have a material adverse impact on our business.
We may not be able to protect our intellectual property rights throughout the world, which could have a material adverse effect on our business.
Filing, prosecuting and defending patents on current and future products in all countries throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside of the United States can be less extensive than those in the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent that federal and state laws do in the United States. Consequently, regardless of whether we are able to prevent third parties from practicing our inventions in the United States, we may not be able to prevent third parties from practicing our inventions in all countries outside of the United States, or from selling or importing products made by using our inventions in and into the United States or other jurisdictions. Competitors may use our technologies in jurisdictions where we have not pursued and obtained patent protection to develop their own products, and further, may export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as it is in the United States. These products may compete with our products and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing. Even if we pursue and obtain issued patents in particular jurisdictions, our patent claims or other intellectual property rights may not be effective or sufficient to prevent third parties from competing. Patent protection must ultimately be sought on a country-by-country basis, which is an expensive and time consuming process with uncertain outcomes. Accordingly, we may choose not to seek patent protection in certain countries, and we will not have the benefit of patent protection in such countries.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, such as China and certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biotechnology, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business, put our patents at risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing, and provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license and may adversely impact our business.
In addition, we and our partners also face the risk that our products are imported or reimported into markets with relatively higher prices from markets with relatively lower prices, which would result in a decrease of sales and any payments we receive from the affected market. Recent developments in U.S. patent law have made it more difficult to stop these and related practices based on theories of patent infringement.
36
We use third-party software that may be difficult to replace or may cause errors or failures of our products that could lead to lost customers or harm to our reputation.
We use software licensed from third parties in our products. In the future, this software may not be available to us on commercially reasonable terms, or at all. Any loss of the right to use any of this software could result in delays in the production of our products until equivalent technology is either developed by us, or, if available, is identified, obtained and integrated, which could harm our business. In addition, any errors or defects in third-party software or other third-party software failures could result in errors, defects or cause our products to fail, which could harm our business and be costly to correct. Many of these providers attempt to impose limitations on their liability for such errors, defects or failures, and if enforceable, we may have additional liability to our customers or third-party providers that could harm our reputation and increase our operating costs.
Risks Related to Our Common Stock and Being a Public Company
Our stock price has fluctuated significantly and may continue to fluctuate significantly.
The market price of shares of our common stock has been and could continue to be subject to wide fluctuations in response to many factors listed in this section, and others beyond our control, including:
| ● | actual or anticipated fluctuations in our financial condition and operating results; |
| ● | announcements by us, our partners or our competitors of new products, significant contracts, restructuring plans, strategic partnerships, joint ventures, collaborations, acquisitions, commercial relationships or capital commitments; |
| ● | competition from existing products or new products that may emerge; |
| ● | failure to meet or exceed financial estimates and projections of the investment community or that we may provide to the public; |
| ● | issuance of new or updated research or reports by securities analysts or recommendations with respect to our stock; |
| ● | positive or adverse regulatory announcements; |
| ● | disputes or other developments related to proprietary rights, including patents, litigation matters and our ability to obtain patent protection for our technologies; |
| ● | commencement of, or our involvement in, litigation; |
| ● | fluctuations in the valuation of companies perceived by investors to be comparable to us; |
| ● | conditions in our markets; |
| ● | manufacturing disputes or delays, product defects or material product quality control issues; |
| ● | any future sales of our common stock or other securities; |
| ● | any change to the composition of our board of directors or key personnel; |
| ● | general economic conditions and slow or negative growth of our markets; |
| ● | a material cybersecurity incident; |
| ● | share price and volume fluctuations attributable to inconsistent trading volume levels of our shares; |
| ● | announcement or expectation of additional debt or equity financing efforts; and |
| ● | other factors described in this Risk Factors section of this Annual Report on Form 10-K. |
These and other market and industry factors may cause the market price and demand for our common stock to fluctuate substantially, regardless of our actual operating performance, which may limit or prevent investors from readily selling their shares of common stock and may otherwise negatively affect the liquidity of our common stock. In addition, the stock market in general, and life science companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. In the past, when the market price of a stock has been volatile, holders of that stock have on occasion instituted securities class action litigation against the company that issued the stock. If any of our stockholders were to bring a lawsuit against us, the defense and disposition of the lawsuit could be costly and divert the time and attention of our management and harm our operating results.
37
We have never paid dividends on our capital stock, and we do not anticipate paying any dividends in the foreseeable future. Consequently, any gains from an investment in our common stock will likely depend on whether the price of our common stock increases.
We have not paid dividends on any of our classes of capital stock to date and we currently intend to retain our future earnings, if any, to fund the development and growth of our business. As a result, capital appreciation, if any, of our common stock will be the sole source of gain for the shareholders in the foreseeable future. Consequently, in the foreseeable future, shareholders will likely only experience a gain from an investment in our common stock if the price of our common stock increases.
Anti-takeover provisions contained in our restated certificate of incorporation and restated by-laws, as well as provisions of Delaware law, could impair a takeover attempt.
Our restated certificate of incorporation, restated by-laws and Delaware law contain provisions which could have the effect of rendering more difficult, delaying or preventing an acquisition deemed undesirable by our board of directors. Our corporate governance documents include provisions:
| ● | authorizing our board of directors to issue up to 5,000,000 shares of preferred stock without stockholder approval upon the terms and conditions and with the rights, privileges and preferences as our board of directors may determine; |
| ● | specifying that special meetings of our stockholders can be called only by our board of directors and that our stockholders may not act by written consent; |
| ● | establishing an advance notice procedure for stockholder proposals to be brought before an annual meeting of our stockholders, including proposed nominations of persons for election to our board of directors; |
| ● | providing that directors may be removed only for cause; |
| ● | providing that our board of directors may create new directorships and that vacancies on our board of directors may be filled only by a majority of directors then in office, even though less than a quorum; |
| ● | establishing that our board of directors is divided into three classes with each class serving staggered three-year terms; |
| ● | providing that our board of directors may amend our restated by-laws without stockholder approval; and |
| ● | requiring a super-majority of votes to amend certain of the above-mentioned provisions. |
These provisions, alone or together, could delay or prevent hostile takeovers and changes in control or changes in our management.
As a Delaware corporation, we are also subject to provisions of Delaware law, including Section 203 of the Delaware General Corporation Law, which prevents some stockholders holding more than 15% of our outstanding common stock from engaging in certain business combinations without approval of the holders of substantially all of our outstanding common stock.
Any provision of our restated certificate of incorporation, restated by-laws or Delaware law that has the effect of delaying or deterring a change in control could limit the opportunity for our stockholders to receive a premium for their shares of our common stock, and could also affect the price that some investors are willing to pay for our common stock
ITEM 1B. UNRESOLVED STAFF COMMENTS
Not applicable.
38
ITEM 1C. CYBERSECURITY
We maintain a cybersecurity risk management program designed to identify, assess, manage, mitigate and respond to cybersecurity threats. Our cybersecurity program is overseen by our Chief Information Officer (“CIO”), who has more than 20 years of experience in information technology. This program incorporates policies, processes, and activities over domains such as access control; facility and data protection; IT systems and data transmission security; threat intelligence and incident response; third-party risk management; disaster recovery; and vulnerability management. We have implemented a risk-based approach to identify and assess the cybersecurity threats that could affect our business and information systems. Thus, we base our program on multiple security frameworks including the National Institute of Standards and Technology (“NIST”), HIPAA, and privacy laws such as the E.U.’s GDPR. We also require that third-party service providers with access to personal or proprietary information implement and maintain cybersecurity practices consistent with applicable legal standards.
Cybersecurity is complex and subject to constantly evolving threats. Accordingly, we engage a range of external experts, including cybersecurity consultants and auditors in evaluating and testing our risk management systems. These partnerships enable us to leverage specialized knowledge and insights. Our collaboration with these third-party experts includes regular audits, threat assessments, and consultation on security enhancements.
Our CIO is tasked with ensuring that the highest levels of management and our Board of Directors are informed about the cybersecurity posture and potential risks facing the Company. Our CIO regularly briefs our CEO about cybersecurity risk management. The CIO’s staff regularly informs the CIO about the latest developments in cybersecurity, including potential threats and risk management techniques. In the event of a cybersecurity incident, the CIO is informed promptly following its detection, and our response is governed by a detailed incident response plan that includes prompt actions to mitigate the impact of the incident and longer-term strategies for remediation and prevention of future incidents. The CIO has also chartered an Information Security Steering Committee made up of cross-functional executive leaders that meets quarterly on topics such as the current cybersecurity landscape and emerging threats; status of ongoing cybersecurity initiatives and strategies; incident reports and learnings from any cybersecurity events; and compliance with regulatory requirements and industry standards.
Cybersecurity risk management is integrated into our broader risk management framework. The Audit Committee of our Board of Directors, which has responsibility for oversight of risk management, also has responsibility for oversight of our program, policies and procedures related to information security and data protection. On a regular basis, the CIO reports to the Audit Committee of our Board of Directors on cybersecurity risks as well as mitigation strategies and the status of initiatives to strengthen our information security systems. The CIO also provides periodic updates to our full Board of Directors, as appropriate.
For a discussion of our risks related to cybersecurity, see the section titled, “Item 1A. Risk Factors – Risks Related to our Operations – Cybersecurity breaches, loss of data and other disruptions could compromise sensitive information related to our business or prevent us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation.”
39
ITEM 2. PROPERTIES
Our corporate headquarters are currently located in Billerica, Massachusetts, which consists of an approximately 91,600 square foot facility we lease for office, laboratory, and manufacturing purposes. We also lease office and laboratory space domestically in Bedford, Massachusetts, and internationally in the Netherlands, Sweden, and China. In the first quarter of 2022, we executed a lease for 85,800 square feet of office and laboratory space in Bedford, Massachusetts. The initial term of the Bedford lease is eight years and nine months beginning on May 1, 2022. We do not currently occupy the Bedford facilities.
We believe our facilities are adequate and suitable for our current operations and that should it be needed, additional or alternative space is available to accommodate our operations.
ITEM 3. LEGAL PROCEEDINGS
In the ordinary course of business, we are from time to time involved in lawsuits, claims, investigations, proceedings, and threats of litigation consisting of intellectual property, contractual, employment, and other matters. While the outcome of any such actions or proceedings cannot be predicted with certainty, as of the date of this Annual Report on Form 10-K, we were not party to any legal proceedings, the outcome of which would be expected to have a material adverse effect on our financial condition or results of operations. Regardless of any outcome, litigation can have a material adverse effect on us due to defense and settlement costs, diversion of management resources, and other factors.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
40
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS, AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our common stock is traded on The Nasdaq Global Market under the symbol “QTRX.”
Holders of Record
As of February 26, 2024, there were approximately 19 stockholders of record of our common stock.
Dividends
Dividends are declared at the discretion of our Board of Directors and depend on actual cash flow from operations, our financial condition, capital requirements, and any other factors our Board of Directors may consider relevant. Since our inception, we have not declared or paid any dividends.
Securities Authorized for Issuance under Equity Compensation Plans
Refer to the section titled “Part III, Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters” of this Annual Report on Form 10-K for information required by Item 201(d) of Regulation S-K.
Unregistered Sales of Securities
There were no unregistered sales of equity securities during the year ended December 31, 2023.
Issuer Purchases of Equity Securities
Not applicable.
41
Stock Performance Graph
The following graph compares the cumulative total shareholder returns over the past five years for our common stock, the NASDAQ Composite Index, and the NASDAQ Biotechnology Index, assuming $100 invested on December 31, 2018, and reinvestment of dividends, if paid:
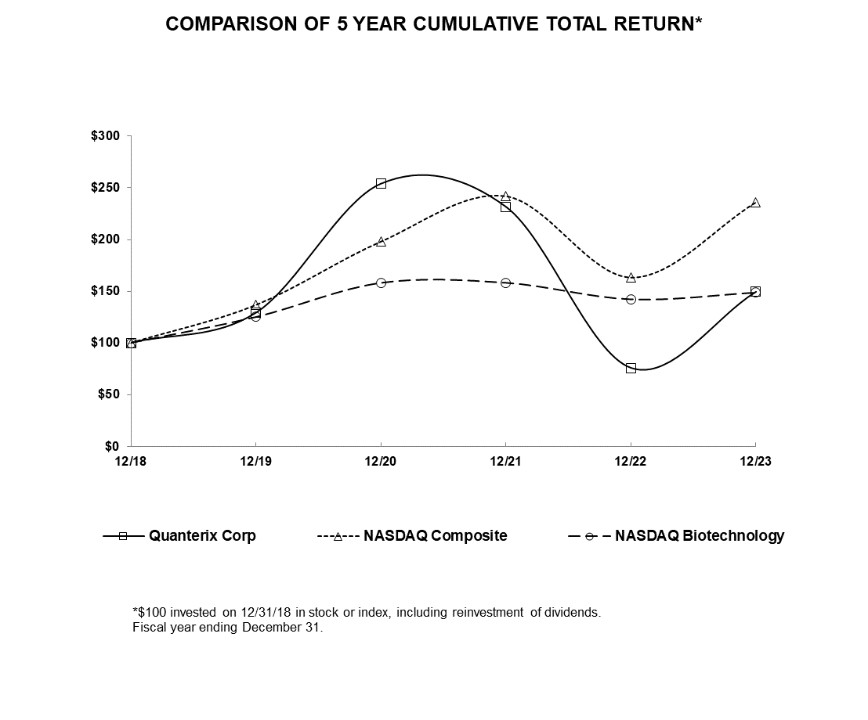
ITEM 6. RESERVED
Not applicable.
42
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis provides information management believes to be relevant to understanding the financial condition and results of operations of Quanterix Corporation for the years ended December 31, 2023 and 2022. For a full understanding of our financial condition and results of operations, this discussion and analysis should be read in conjunction with our Consolidated Financial Statements and accompanying notes included in the section titled “Part II. Item 8. Financial Statements and Supplementary Data” of this Annual Report on Form 10-K. In addition to historical information, the following discussion and analysis contains forward-looking statements that involve risks, uncertainties, and assumptions. Our actual results, performance, or experience may differ materially from those discussed below due to various important factors, risks, and uncertainties, including, but not limited to, those set forth under the sections titled “Part I, Item 1A. Risk Factors” and “Note Regarding Forward-Looking Statements” included in this Annual Report on Form 10-K. Unless the context otherwise requires, the terms “Quanterix,” the “Company,” “we,” “it,” “us,“ and “our” in this Annual Report on Form 10-K refer to Quanterix Corporation and its consolidated subsidiaries.
For additional information on our financial condition as of December 31, 2022 and results of operations for the year ended December 31, 2022 as compared to the year ended December 31, 2021, refer to the section titled “Part II, Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations” in our Annual Report on Form 10-K for the year ended December 31, 2022.
Overview
We are a life sciences company that has developed next-generation, ultra-sensitive digital immunoassay platforms that advance life sciences research and diagnostics. Our platforms are based on our proprietary digital “Simoa” detection technology and enable customers to reliably detect protein biomarkers at ultra-low concentrations in blood, serum, and other fluids that, in many cases, are undetectable using conventional, analog immunoassay technologies. The ability of our Simoa platforms to detect proteins in the femtomolar range is enabling the development of novel therapies and diagnostics and has the potential to facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis, and, ultimately, prevention. Our Simoa platforms have achieved significant scientific validation and commercial adoption, and our Simoa technology has been cited in more than 2,700 scientific publications in areas of high unmet medical need and research interest such as neurology, oncology, cardiology, infectious disease, and inflammation.
Our instruments are designed to be used either with assays fully developed by us, including all antibodies and supplies required to run the assays, or with “homebrew” assay kits where we supply some of the components required for testing, and the customer supplies the remaining required elements. Accordingly, our installed instruments generate a recurring revenue stream. As the installed base of the Simoa instruments increases, we expect total consumables revenue to increase.
We commercially launched our HD-X instrument in the second half of 2019. The HD-X is an upgraded version of the Simoa HD-1 (our first Simoa instrument, launched in January 2014), collectively “HD Instruments”, that is designed to deliver significant productivity and operational efficiency improvements, as well as greater user flexibility. The HD-X uses our bead-based technology and assays run on the HD-X are fully automated. By the end of 2023, approximately 82% of the HD Instrument installed base were HD-X instruments.
Further, we launched our SR-X instrument in 2017 as a compact desktop instrument with a lower price point, more flexible assay preparation, and a wider range of applications. The SR-X utilizes the same Simoa bead-based technology and assay kits as the HD-X.
With our acquisition of Aushon BioSystems, Inc. in 2018, we acquired their CLIA certified laboratory and their proprietary sensitive planar array detection technology. The Clinical Laboratory Improvement Amendments of 1988 (“CLIA”) are federal regulatory standards that apply to all clinical laboratory testing performed on humans in the United States (with the exception of research testing that does not report patient specific results). Leveraging our proprietary
43
sophisticated Simoa image analysis and data analysis algorithms, we further refined the planar array technology to develop the SP-X instrument to provide sensitivity similar to that found in our Simoa bead-based platform. We commercially launched the SP-X instrument in 2019.
Our wholly owned subsidiary UmanDiagnostics AB (“Uman”), a company located in Umeå, Sweden, supplies neurofilament light (“NfL”), antibodies, and enzyme-linked immunoassay (“ELISA”) kits, which are used by researchers and biopharmaceutical and diagnostics companies world-wide in the detection of NfL to advance the development of therapeutics and diagnostics for neurodegenerative conditions.
We also provide contract research services for customers and Laboratory Developed Test (“LDT”) services through our CLIA-certified Accelerator Laboratory (the “Accelerator Laboratory”). The Accelerator Laboratory provides customers with access to Simoa technology, and supports multiple projects and services, including sample testing, homebrew assay development, custom assay development, and blood-based biomarker testing. To date, we have completed over 2,200 projects for more than 480 customers from all over the world using our Simoa platforms.
We have an extensive base of customers including pharmaceutical, biotechnology, contract research organizations, academic and governmental research institutions. We sell our instruments, consumables, and services through a direct field sales and support organizations in North America and Europe, and through our own sales force and distributors in additional countries, including Australia, Brazil, China, Czech Republic, India, Hong Kong, Israel, Japan, New Zealand, Qatar, Saudi Arabia, Singapore, South Africa, South Korea, Taiwan, and the United Arab Emirates. In addition, Uman sells NfL antibodies and NfL ELISA kits directly, and in conjunction with us and another distributor, worldwide.
Our total revenues were $122.4 million and $105.5 million for the years ended December 31, 2023 and 2022, respectively. Since our inception, we have incurred annual net losses, including net losses of $32.3 million and $96.7 million for the years ended December 31, 2023 and 2022, respectively.
We expect to incur significant expenses and operating losses at least through the next 24 months, and we expect our expenses to increase substantially as we:
| ● | expand our sales and marketing efforts to further commercialize our products; |
| ● | expand our research and development efforts to improve our existing products and develop and launch new products, particularly if any of our products become subject to additional or more burdensome regulation by the U.S. Food and Drug Administration (the “FDA”); |
| ● | invest in our diagnostics business in support of the launch of Lucent Diagnostics, additional LDTs, and other diagnostics initiatives including entry into translational pharma and clinical diagnostic markets; |
| ● | seek Premarket Approval (“PMA”) or 510(k) clearance from the FDA for our existing products or new products if or when we decide to market products for use in the prevention, diagnosis, or treatment of a disease or other condition; |
| ● | hire additional personnel to support our growth and research and development; |
| ● | strategically acquire and integrate companies or technologies that may be complementary to our business; |
| ● | enter into collaboration arrangements, or in-license other products and technologies; and |
| ● | add operational, financial, and management information systems. |
Recent Business Developments
In October 2023, we entered into a license agreement with Janssen Sciences Ireland UC, a Johnson & Johnson Company, (“Janssen”). The agreement grants us worldwide, non-exclusive rights to Janssen’s p-Tau 217 antibodies and assay designs for use in clinical research and diagnostic products, including in the production of Simoa p-Tau 217 research use only assay (“RUO”) kits for global distribution. Under this license, we are required to pay royalties on net sales of the licensed products and service activities.
44
In July 2023, we launched Lucent Diagnostics, our brand for clinical testing services for healthcare providers in the field of neurology. Testing services were initially focused on Alzheimer’s disease at the same time newly approved therapies were becoming available. The first product offering was LucentAD, a blood test to aid the evaluation of patients experiencing cognitive symptoms consistent with the early signs of Alzheimer’s disease. In October 2023, we launched an improved blood test for Alzheimer’s disease, LucentAD p-Tau 217. These tests have not been cleared or approved by the FDA. We do not expect material revenues from these tests until late 2024 or later. For further discussion of FDA requirements refer to the section titled “Item 1. Business - Government Regulations”.
In August 2022, we announced a restructuring and strategic realignment plan (the “Restructuring Plan”). The Restructuring Plan included the elimination of 119 positions across the company in 2022 and other cost-saving measures that were substantially completed in 2022. The Restructuring Plan also included an assay redevelopment program with the objective of improving our ability to manufacture and deliver high-quality assays at scale. This six-quarter operational program was substantially completed in the fourth quarter of 2023, and we have now launched five new Simoa Advantage PLUS assays. The Simoa Advantage PLUS assays have been developed using improved protocols, by leveraging manufacturing efficiencies and reagent improvements to provide more consistent results and improved lot-to-lot consistency, and through enabling production of larger lot sizes with extended shelf lives. These assays began shipping to customers in the first quarter of 2024. We expect to continue to apply these improved protocols and manufacturing efficiencies to other existing assays and assays that we may develop in the future.
Components of Results of Operations
Revenues
Product Revenue
Our product revenues are generated from sales of (1) instruments and (2) consumables and related revenues.
Instrument revenues consist of sales of our instruments (HD-X, SR-X, and SP-X). We currently sell our products for RUO applications directly to customers or through distributors. Direct sales of instruments to customers include an initial year of implied service-type warranties. Sales of instruments to distributors include a license to import and resell the instruments. Instrument sales may also be bundled with assays and other consumables, training, installation, and/or an extended service warranty.
Consumable and other revenues consist of sales of assays fully developed by us, including all antibodies and supplies required to run the assays, or with “homebrew” assay kits where we supply some of the components required for testing, and the customer supplies the remaining required elements. Consumable and other revenues also consist of replacement parts, reagents, and antibodies.
Service and Other Revenue
Service revenues consist of fixed fee contract research services through our Accelerator Laboratory, initial implied service-type warranties, extended service warranty contracts, repair services, and other services such as training.
Collaboration and License Revenue
Collaboration and license revenues consist of licensing our technology, intellectual property, and know-how associated with our instruments to third parties and for related services. License arrangements consist of sales or usage-based fees and/or future royalties.
Grant Revenue
Grant revenues consist of funding received to perform specific research and development services under grant arrangements.
45
Cost of Goods Sold and Services
Cost of Product Revenue
Cost of product revenue consists of manufacturing and assembly costs for instruments, related reagents, other consumables, contract manufacturer costs, personnel costs, royalties, overhead, and other direct costs related to product sales. Raw material part costs include inbound freight, and shipping and handling costs associated with purchased goods. Cost of product revenue also includes royalty fees due to third parties from revenue generated by collaboration or license deals.
Cost of Service and Other Revenue
Cost of services and other revenue consists of direct costs associated with operating our Accelerator Laboratory on behalf of customers, including raw materials, personnel costs, royalties, allocated overhead and other related costs. Additional costs include costs related to warranty services and other costs of servicing equipment at customer sites.
Research and Development Expense
Research and development expense consists of personnel costs, research supplies, third-party development costs for new products, materials for prototypes, quality assurance, and allocated overhead costs that include facility and other related costs. We have made substantial investments in research and development since our inception and plan to continue to make substantial investments in the future. Our research and development efforts have focused primarily on supporting development and commercialization of new and existing products and improved product quality. We believe that our continued investment in research and development is essential to our long-term competitive position and expect these expenses to increase in future periods.
Selling, General and Administrative Expense
Selling, general and administrative expense consists of personnel costs for our sales and marketing, finance, legal, human resources, and general management teams, shipping and handling for product sales, other general and administrative costs, as well as professional services costs, such as marketing, advertising, legal and accounting services, and allocated overhead costs that include facility and other related costs. We expect to increase the size of our selling, general and administrative functions to support the growth in our business and newly launched Lucent Diagnostics. However, selling, general and administrative expenses in total are not expected to increase at the same rate in future periods as total revenue or research and development expenses.
The classification of shipping and handling costs for product sales varies from company to company, with some companies recording these as selling, general and administrative expenses and others recording such expenses within costs of goods sold for products. To the extent our classification of these shipping and handling costs differs from the classification used by other companies, our gross margins may not be comparable with those reported by such other companies.
Other Lease Costs
Other lease costs consist of amortization of operating lease right-of-use assets and other facility operating expenses from leased facilities we are not using as a result of the Restructuring Plan.
Impairment and Restructuring
Impairment and restructuring expense primarily consists of charges recorded as a result of the Restructuring Plan.
As a result of the Restructuring Plan, we performed an impairment assessment of our goodwill, long-lived assets, including operating lease right-of-use assets, property and equipment, and intangibles. All of our goodwill, and a
46
portion of our operating lease right-of-use assets (including related property and equipment) were determined to be impaired as their carrying values exceeded their fair values, and corresponding impairment charges were recorded in the year ended December 31, 2022. During the year ended December 31, 2023, we continued to reassess the remaining operating lease right-of-use assets and related property and equipment and recorded an additional impairment charge.
Additional impairment expenses consist of assessments of our intangible and long-lived assets annually, or whenever events or circumstances indicate that the carrying amount of the asset(s) may not be recoverable.
Interest Income
Interest income consists of interest earned on cash, cash equivalents, and marketable securities, and the accretion of discounts from the purchase of marketable securities.
Other Income (Expense), Net
Other income (expense), net primarily consists of unrealized and realized gains and losses on foreign currency, and other non-recurring items that are not a part of our core business operations.
Income Tax Expense
Income tax expense consists primarily of income taxes related to federal, state, and foreign jurisdictions in which we conduct business.
Comparison of Results of Operations for Years Ended December 31, 2023 and 2022:
The following table sets forth select Consolidated Statements of Operations data, and such data as a percentage of total revenues (in thousands, except percentages):
Year Ended December 31, | Increase (Decrease) | ||||||||||||||||
2023 | % of revenue | 2022 | % of revenue | Amount | % | ||||||||||||
Revenues: | |||||||||||||||||
Product revenue | $ | 79,460 |
| 65 | % |
| $ | 69,808 |
| 66 | % |
| $ | 9,652 |
| 14 | % |
Service and other revenue |
| 40,299 |
| 33 | % |
|
| 34,495 |
| 33 | % |
|
| 5,804 |
| 17 | % |
Collaboration and license revenue |
| 1,380 |
| 1 | % |
|
| 649 |
| 1 | % |
|
| 731 |
| 113 | % |
Grant revenue | 1,229 | 1 | % | 570 | 1 | % | 659 | 116 | % | ||||||||
Total revenues |
| 122,368 |
| 100 | % |
|
| 105,522 |
| 100 | % |
|
| 16,846 |
| 16 | % |
Costs of goods sold and services: |
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||
Cost of product revenue |
| 32,636 |
| 27 | % |
|
| 40,809 |
| 39 | % |
|
| (8,173) |
| (20) | % |
Cost of service and other revenue |
| 19,086 |
| 16 | % |
|
| 17,907 |
| 17 | % |
|
| 1,179 |
| 7 | % |
Total costs of goods sold and services |
| 51,722 |
| 42 | % |
|
| 58,716 |
| 56 | % |
|
| (6,994) |
| (12) | % |
Gross profit |
| 70,646 |
| 58 | % |
|
| 46,806 |
| 44 | % |
|
| 23,840 |
| 51 | % |
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
| ||||
Research and development |
| 24,857 |
| 20 | % |
|
| 25,890 |
| 25 | % |
|
| (1,033) |
| (4) | % |
Selling, general and administrative |
| 90,241 |
| 74 | % |
|
| 91,995 |
| 87 | % |
|
| (1,754) |
| (2) | % |
Other lease costs | 3,712 | 3 | % | 1,278 | 1 | % | 2,434 |
| 191 | % | |||||||
Impairment and restructuring | 1,537 | 1 | % | 29,347 | 28 | % | (27,810) |
| (95) | % | |||||||
Total operating expenses |
| 120,347 |
| 98 | % |
|
| 148,510 |
| 141 | % |
|
| (28,163) |
| (19) | % |
Loss from operations |
| (49,701) |
| (41) | % |
|
| (101,704) |
| (96) | % |
|
| 52,003 |
| 51 | % |
Interest income |
| 15,839 |
| 13 | % |
|
| 5,131 |
| 5 | % |
| 10,708 |
| 209 | % | |
Other income (expense), net |
| 2,247 |
| 2 | % |
|
| (62) |
| — | % |
|
| 2,309 |
| 3,702 | % |
Loss before income taxes |
| (31,615) |
| (26) | % |
|
| (96,635) |
| (92) | % |
|
| 65,020 |
| 67 | % |
Income tax expense |
| (719) |
| (1) | % |
|
| (65) |
| — | % |
|
| (654) |
| (1,007) | % |
Net loss | $ | (32,334) |
| (26) | % |
| $ | (96,700) |
| (92) | % |
| $ | 64,366 |
| 67 | % |
47
Revenues
Total revenues increased $16.8 million, or 16%, to $122.4 million for the year ended December 31, 2023, compared to $105.5 million for the year ended December 31, 2022.
Product revenue of $79.5 million for the year ended December 31, 2023 consisted of instrument sales of $15.7 million and sales of consumables and other products of $63.8 million. This represented an increase of $9.7 million, or 14%, compared to product revenue of $69.8 million for the year ended December 31, 2022. The increase in product revenue was primarily due to a $19.0 million increase in sales of consumables and increased average selling prices. This increase was partially offset by a $9.3 million decrease in instrument sales due to reduced demand in what we believe is a constrained capital funding environment. We expect softness in instrument sales to continue in 2024.
Service revenue was $40.3 million for the year ended December 31, 2023, compared to $34.5 million for the year ended December 31, 2022, an increase of $5.8 million, or 17%. This increase was primarily due to a $9.0 million increase in Accelerator Laboratory revenue driven by higher volumes of sample testing and assay development services, and was partially offset by a $4.9 million decrease in revenue recognized from a collaboration agreement with Eli Lilly and Company (the “Lilly Collaboration Agreement”) due to non-recurring upfront payments received in 2022. The Lilly Collaboration Agreement establishes a framework for future projects focused on the development of Simoa immunoassays.
Collaboration and license revenue was $1.4 million for the year ended December 31, 2023, compared to $0.6 million for the year ended December 31, 2022, an increase of $0.7 million, or 113%. The increase was primarily due to $0.5 million of one-time revenue in 2023 related to the expiration of a previously paid for option to expand the scope of a license agreement with Abbott Laboratories entered into in 2020, pursuant to which we granted Abbott a non-exclusive, worldwide, royalty-bearing license, without the right to sublicense, under the Company’s bead-based single molecule detection patents in the field of in vitro diagnostics.
Grant revenue was $1.2 million for the year ended December 31, 2023, compared to $0.6 million for the year ended December 31, 2022, an increase of $0.7 million, or 116%, driven by receipt of a portion of a grant from the National Institutes of Health. Refer to Note 3 – Revenue and Related Matters within the Notes to the Consolidated Financial Statements, for more information regarding this grant.
Cost of Goods Sold and Services
Total cost of goods sold and services decreased $7.0 million, or 12%, to $51.7 million for the year ended December 31, 2023 compared to $58.7 million for the year ended December 31, 2022.
Cost of product revenue decreased $8.2 million, or 20%, to $32.6 million for the year ended December 31, 2023, compared to $40.8 million for the year ended December 31, 2022. The decrease was primarily due to improvement in inventory management and manufacturing processes as well as lower instrument sales, partially offset by higher costs related to increased consumables sales.
Cost of service and other revenue increased $1.2 million, or 7%, to $19.1 million for the year ended December 31, 2023, compared to $17.9 million for the year ended December 31, 2022. This increase was primarily due to an increase in department costs including compensation and benefits costs related to increased headcount, and was partially offset by lower costs related to the Lilly Collaboration Agreement.
Research and Development
Research and development expense decreased $1.0 million, or 4%, to $24.9 million for the year ended December 31, 2023, compared to $25.9 million for the year ended December 31, 2022. This decrease was primarily due to a decrease in compensation and benefit costs related to the reduction in headcount from the Restructuring Plan, which was partially offset by an increase in costs related to the assay redevelopment program under the Restructuring Plan including consulting fees, lab supplies, equipment, and product development activities.
48
Selling, General and Administrative
Selling, general and administrative expense decreased $1.8 million, or 2% to $90.2 million for the year ended December 31, 2023, compared to $92.0 million for the year ended December 31, 2022. The decrease was primarily due to a decrease in compensation and benefit costs related to the reduction in headcount from the Restructuring Plan and a full twelve months of facilities costs from the leased office and laboratory facilities we are not using being recorded in other lease costs instead of selling, general, and administrative expenses on the Consolidated Statements of Operations. These decreases were partially offset by (1) an increase in professional services and consulting fees related to our efforts to remediate the material weaknesses in our internal control over financial reporting described in our Annual Report on Form 10-K for the year ended December 31, 2022, (2) an increase in software and information technology expenses, and (3) an increase in shipping and handling costs for consumables and other products due to higher volume. Included within selling, general and administrative expense are $8.1 million and $7.2 million of shipping and handling costs for product sales for the years ended December 31, 2023 and 2022, respectively.
Other Lease Costs
Other lease costs increased $2.4 million, or 190%, to $3.7 million for the year ended December 31, 2023, compared to $1.3 million for the year ended December 31, 2022. As part of the Restructuring Plan, we are not using two leased office and laboratory facilities and are evaluating alternatives, including sub-leasing the facilities. Other lease costs include the amortization of the related operating lease right-of-use assets and other leased facility operating expenses from periods after the initiation of the Restructuring Plan and the determination that the facilities would not be used. Lease costs in 2022 represent four and a half months of cost in 2022 after the Restructuring Plan was implemented, as compared to twelve months of costs in 2023. Expenses incurred prior to the Restructuring Plan were recorded in selling, general, and administrative on the Consolidated Statements of Operations.
Impairment and Restructuring
Impairment and restructuring costs were $1.5 million for the year ended December 31, 2023, compared to $29.3 million for the year ended December 31, 2022. This decrease reflects the implementation of the Restructuring Plan in August 2022, which did not repeat in 2023.
Costs incurred during the year ended December 31, 2023 primarily relate to long-lived asset impairment charges associated with two leased facilities we are not using. Costs incurred during the year ended December 31, 2022 include (1) $8.2 million of goodwill impairment charges, (2) $16.3 million of long-lived asset impairment charges associated with the leased facilities that we are not using, (3) $1.1 million of software costs related to projects that were rationalized as part of the Restructuring Plan, and (4) $3.8 million of restructuring expenses primarily for severance and one-time termination benefits in connection with the elimination of 119 positions across the Company.
Interest Income
Interest income increased by $10.7 million, or 209% to $15.8 million for the year ended December 31, 2023, compared to $5.1 million for the year ended December 31, 2022. This increase was primarily due to higher interest rates earned on cash, cash equivalents, and marketable securities, and the accretion of discounts from the purchase of marketable securities.
Other Income (Expense), Net
Other income (expense) was $2.2 million of income for the year ended December 31, 2023, compared to $0.1 million of expense for the year ended December 31, 2022. The increase was primarily due to recognizing a $2.4 million receivable under the Employee Retention Credit established by the Coronavirus Aid, Relief, and Economic Security Act in 2021.
49
Income Tax Expense
Income tax expense was $0.7 million for the year ended December 31, 2023, as compared to $0.1 million for the year ended December 31, 2022. The change was primarily due to the increase in the tax expense recorded on the operating results of our foreign subsidiaries.
Liquidity and Capital Resources
Our principal sources of liquidity are cash, cash equivalents, marketable securities, and funds generated from sales of our products and services. As of December 31, 2023, we had cash and cash equivalents of $174.4 million and $146.9 million of available for sale marketable securities. Historically we have financed our operations through equity offerings and borrowings from credit facilities.
We believe our cash, cash equivalents, and marketable securities, along with funds generated from sales of our products and services, will be sufficient to meet our anticipated operating cash requirements for at least 12 months from the date of this Annual Report on Form 10-K.
Our liquidity requirements have consisted, and we expect that they will continue to consist, of sales and marketing expenses, research and development expenses, working capital, and general corporate expenses. Our future capital requirements will depend on many factors, including, but not limited to, our pace of growth, expansion and introduction of new products and services, including Lucent Diagnostics, continuing market acceptance of our products and services, regulatory guidelines or approval of our products or services.
We regularly assess potential acquisitions and have a strategy to pursue acquisitions of complementary businesses, services, and technologies. To the extent our existing cash, cash equivalents, and marketable securities are insufficient to fund future activities or requirements to continue operating our business, we may need to raise additional capital.
If needed, we cannot guarantee that we will be able to obtain additional funds on acceptable terms, or at all. If we raise additional funds by issuing equity or equity-linked securities, our stockholders may experience dilution. Future debt financing, if available, may involve covenants restricting our operations or our ability to incur additional debt. Any debt or equity financing that we raise may contain terms that are not favorable to us or our stockholders. If we raise additional funds through collaboration and licensing arrangements with third parties, it may be necessary to relinquish some rights to our technologies or our products, or grant licenses on terms that are not favorable to us. If we do not have or are not able to obtain sufficient funds, if needed, we may have to delay development or commercialization of our products and services. We also may have to reduce marketing, customer support or other resources devoted to our products, or cease operations.
If the conditions for raising capital are favorable, we may seek to finance future cash needs through public or private equity, debt offerings, or other financings.
50
Cash Flows
The following table summarizes our cash flows (in thousands):
Year Ended December 31, | |||||
2023 |
| 2022 | |||
Net cash used in operating activities | $ | (18,902) | $ | (48,272) | |
Net cash used in investing activities |
| (148,401) |
| (11,206) | |
Net cash provided by financing activities |
| 2,691 |
| 2,311 | |
Net decrease in cash, cash equivalents, and restricted cash | $ | (164,612) | $ | (57,167) | |
Net Cash Used in Operating Activities
We derive cash flows from operations primarily from the sale of our products and services. Our cash flows from operating activities are also significantly influenced by our use of cash for operating expenses to develop new products and services, invest in process and product improvements, and increase our sales and marketing efforts. We have historically experienced negative cash flows from operating activities as we have developed our technology, expanded our business, and built our infrastructure. We expect negative cash flows from operating activities will continue in the future.
Net cash used in operating activities was $18.9 million and $48.3 million for the years ended December 31, 2023 and 2022, respectively. The $29.4 million reduction in net cash used in operating activities was primarily driven by an overall reduction in our net loss, adjusted for non-cash items, consisting of revenue growth, a full year of reduced expenses resulting from the Restructuring Plan, continued improvements in our inventory management and manufacturing processes leading to improved gross margin, and increased interest income from investing in marketable securities and rising interest rates. The reduction was offset by changes in working capital items, primarily an increase in accounts receivable from strong revenue growth through the fourth quarter of 2023 and an increase in inventory as a result of completing the assay development program and manufacturing new assays to begin shipping to customers in the first quarter of 2024.
Net Cash Used in Investing Activities
Our primary investing activities consist of purchases of marketable securities to increase the interest income we would otherwise earn in cash accounts. Additionally, we use funds towards capital expenditures for the purchase of equipment to support our expanding infrastructure and work force. We expect to continue to incur additional capital expenditures related to these efforts in future periods. Cash used towards capital expenditures can be partially offset by proceeds from grants with third parties to purchase assets (refer to the section titled “Grant Revenue” in Note 3 − Revenue and Related Matters in the Notes to Consolidated Financial Statements for more information).
Net cash used in investing activities was $148.4 million during the year ended December 31, 2023, which consisted of the purchase of $175.6 million of marketable securities, proceeds from the maturities of marketable securities of $31.0 million, and $3.8 million of purchases of property and equipment.
Net cash used in investing activities was $11.2 million during the year ended December 31, 2022, which consisted of $11.7 million of purchases of property and equipment which were partially offset by $0.5 million in grant proceeds under the grant received from the National Institutes of Health under its Rapid Acceleration of Diagnostics program. Refer to Note 3 – Revenue and Related Matters, within the Notes to the Consolidated Financial Statements, for more information regarding this grant.
Net Cash Provided by Financing Activities
Financing activities provided $2.7 million and $2.3 million of cash during the years ended December 31, 2023 and 2022, respectively, from sales of our common stock under our employee stock purchase plan and from the exercise of options under our equity incentive plan.
51
Future Cash Obligations
In addition to the future cash obligations described below, we have other payables and liabilities that may be legally enforceable but are not considered contractual commitments. Refer to Note 15 − Commitments and Contingencies in the Notes to Consolidated Financial Statements for a summary of our purchase commitments and other obligations as of December 31, 2023.
Operating Leases
We lease office, laboratory, and manufacturing space for our employees and operations, as well as office equipment, under non-cancellable operating lease agreements (refer to Note 14 − Leases in the Notes to Consolidated Financial Statements). The remaining duration of non-cancellable operating leases ranges from four months to seven years. Remaining lease payments within one year, within two to three years, within four to five years, and greater than five years from December 31, 2023 are $7.1 million, $14.7 million, $15.5 million, and $15.7 million, respectively.
STRATEC Purchase Commitment
During the year ended December 31, 2022, the Company and STRATEC Consumables GmbH (“STRATEC”) entered into an amendment to the supply agreement with STRATEC (as amended, the “STRATEC Supply Agreement”), related to the supply of discs used in Simoa bead-based instruments. As part of the STRATEC Supply Agreement, the Company agreed to purchase a total of 515,000 discs to be shipped at various points starting in 2022 and continuing through 2024 at an agreed purchase price per disc.
The total purchase commitment under the STRATEC Supply Agreement is $3.7 million, of which $2.1 million has been paid, and $1.0 million is due within one year from December 31, 2023.
Critical Accounting Policies and Estimates
Our Consolidated Financial Statements and the related notes included elsewhere in this Annual Report on Form 10-K are prepared in accordance with accounting principles generally accepted in the United States (“U.S. GAAP”). The preparation of these Consolidated Financial Statements requires us to make estimates and assumptions that affect the reported amounts of assets, liabilities, revenue, expenses, and related disclosures. We base our estimates on historical experience, worldwide economic conditions, both general and specific to the life sciences industry, and on various other assumptions we believe to be reasonable under the circumstances. We evaluate our estimates and assumptions on an ongoing basis, and changes in accounting estimates may occur from period to period. Accordingly, actual results could differ significantly from the estimates. To the extent that there are material differences between these estimates and actual results, our future financial statement presentation, financial condition, results of operations, and cash flows will be affected.
Our significant accounting policies are described in Note 2 – Significant Accounting Policies in the Notes to Consolidated Financial Statements. We believe that the assumptions and estimates in the following critical accounting policies involve a greater degree of judgment and complexity and accordingly are the most critical to understanding and evaluating the potential impact to our Consolidated Financial Statements.
Revenue from Contracts with Customers
We generate revenue from the sale of products, services, and licenses, as further described in the section titled “Components of Results of Operations” above.
For contracts with customers, we recognize revenue when a customer obtains control of promised products or services, for an amount that reflects the consideration expected to be received in exchange for those products or services. We follow the five-step framework prescribed by Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) Topic 606 - Revenue from Contracts with Customers (“ASC 606”) to determine revenue recognition: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii)
52
determine the transaction price, including variable consideration, if any; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the entity satisfies a performance obligation. Revenues are presented net of any sales, value added, or similar taxes collected from customers and remitted to the government.
We determine the transaction price based on the amount of consideration we expect to be entitled to, which is generally equal to our contract amounts. In some cases, our contracts contain variable consideration which primarily relates to (1) sales- and usage-based royalties related to the license of intellectual property in collaboration and license contracts and (2) contracts with minimum purchase commitments. For sales and usage-based royalties, ASC 606 provides an exception to estimating variable consideration. Under this exception, we recognize revenues from sales- or usage-based royalty revenue at the later of when the sales or usage occurs or the satisfaction (or partial satisfaction) of the performance obligation to which the royalty has been allocated. All other variable amounts are constrained to the minimum guaranteed contract amount so that a reversal of cumulative revenue does not occur in future periods. Once there is no longer uncertainty over a variable amount, any incremental fees we are entitled to are allocated to the related performance obligations.
Our contracts may include either a single promise (referred to as a performance obligation) to transfer a product or service, or a combination of multiple performance obligations to transfer products or services. We evaluate the existence of multiple performance obligations within our contracts by using judgment to determine if (1) the customer can benefit from each contractual promise on its own or together with readily available resources and (2) the transfer of each contractual promise is separately identifiable from other promises in a contract. When both criteria are met, each promise is accounted for as a separate performance obligation.
Direct instrument sales include installation and an initial year of implied service-type warranties. We have determined that the instrument and installation are a combined performance obligation as the customer cannot benefit from the instrument without the installation and no other vendors can provide installation of our specialized instruments. The implied service-type warranty is considered a separate performance obligation since a customer could benefit from it independently with readily available resources and is capable of being sold on its own. Sales of instruments to distributors include a license to import and resell the instruments. We have determined these distributor licenses are part of a combined performance obligation with the instrument as the distributor only benefits from the combination of the instrument and ability to resell it.
Instrument sales may also be bundled with assays and other consumables, training, and/or an extended service warranty, each of which is considered a separate performance obligation.
Contracts that include rights to additional products or services that are exercisable at a customer’s discretion are generally considered options. We assess if these options provide a material right to the customer and if so, the material right is considered a performance obligation. The identification of material rights requires judgment to determine if the value of the option to purchase additional products and services in relation to options that may be provided to, and prices paid by, customers in the normal course of business. Material rights are recognized when they are exercised by a customer or upon expiration of the right.
For contracts that contain multiple performance obligations, the transaction price is allocated among the performance obligations on a relative basis according to their standalone selling prices (“SSP”). Determining the SSP for performance obligations requires judgment. We determine SSP based on factors including prices charged to customers in observable transactions, internal pricing objectives and list prices, and pricing of similar products, and we use a range of amounts to estimate SSP. We have more than one range of standalone selling price for certain products and services based on the geographic location of the customer and sales channel.
The majority of our products and services are recognized at the point in time we transfer control to the customer.
53
For product revenues, direct instrument sales to customers are recognized upon completion of the instrument’s installation. For instrument sales to distributors, revenue is recognized based on the agreed upon shipping terms (either upon shipment or delivery) as that is when title passes to the customer.
Services revenues generated from contract research services in our Accelerator Laboratory are recognized upon completion and delivery of the research results. In cases where we maintain a contractual right to payment for service performed (including a reasonable profit margin), revenue is recognized over time as the services are provided, using an output method that is based on the number of completed results. Service revenues generated from warranties and service contracts are recognized ratably over the service period as the customer simultaneously receives and benefits from the services.
Collaboration and license revenues are recognized at the point in time the license is delivered as the customer has the right to use the intellectual property when it is received. Royalty revenues that are sales- or usage- based are recognized at the later of when the sales or usage occurs or the satisfaction (or partial satisfaction) of the performance obligation to which the royalty has been allocated.
Inventory Reserves
Inventory is stated at the lower of cost or net realizable value on a first-in, first-out (“FIFO”) basis and includes the cost of materials, labor, and manufacturing overhead. We analyze our inventory levels on each reporting date for slow-moving, excess, and obsolete inventory, and inventory expected to expire prior to being used. Our analysis requires judgment and is based on factors including, but not limited to, our recent historical activity, anticipated or forecasted demand for our products (developed through our planning and sales and marketing inputs), and market conditions. If we identify any of these adverse conditions exist, the carrying value of the inventory is reduced to its estimated net realizable value by providing estimated reserves for excess or obsolete inventory. We adjust the reserves for excess or obsolete inventory and record additional inventory write downs based on unfavorable changes in estimated customer demand or actual market conditions that may differ from management projections.
Impairment of Other Long-Lived Assets
Our long-lived assets consist of operating lease right-of-use assets, property and equipment, and intangible assets. We review the carrying amount of our long-lived assets for impairment whenever events or circumstances indicate that the estimated useful lives may warrant revision, or that the carrying amount of the assets may not be fully recoverable. To assess whether a long-lived asset or asset group has been impaired, the estimated undiscounted and discounted future cash flows for the estimated remaining useful life or estimated lease term of the asset is compared to its carrying value. Significant judgment is required to estimate future cash flows, including, but not limited to, the expected use of the asset, historical client retention rates, technology roadmaps, consumer awareness, trademark and trade name history, contractual provisions that could limit or extend an asset's useful life, market data, discount rates, and potential sublease opportunities, including rent and rent escalation rates, time to sublease, and free rent periods. To the extent that the future cash flows are less than the carrying value, a long-lived asset or asset group is impaired and written down to its estimated fair value.
Non-GAAP Financial Measures
To supplement our financial statements presented on a U.S. GAAP basis, we present non-GAAP gross profit, non-GAAP gross margin, non-GAAP total operating expenses, and non-GAAP loss from operations. These non-GAAP measures are calculated by including shipping and handling costs for product sales within cost of product revenue instead of within selling, general and administrative expenses. We use these non-GAAP measures to evaluate our operating performance in a manner that allows for meaningful period-to-period comparison and analysis of trends in our business and our competitors. We believe that presentation of these non-GAAP measures provides useful information to investors in assessing our operating performance within our industry and to allow comparability to the presentation of other companies in our industry where shipping and handling costs are included in cost of goods sold for products. The non-GAAP financial information presented here should be considered in conjunction with, and not as a substitute for, the financial information presented in accordance with U.S. GAAP.
54
Set forth below is a reconciliation of non-GAAP gross profit, non-GAAP gross margin, non-GAAP total operating expenses, and non-GAAP loss from operations to their most directly comparable GAAP financial measures (in thousands).
Year Ended December 31, | |||||
2023 | 2022 | ||||
GAAP gross profit | $ | 70,646 | $ | 46,806 | |
Shipping and handling costs | (8,146) | (7,211) | |||
Non-GAAP gross profit | $ | 62,500 | $ | 39,595 | |
GAAP revenue | $ | 122,368 | $ | 105,522 | |
GAAP gross margin (gross profit as % of revenue) | 57.7% | 44.4% | |||
Non-GAAP gross margin (non-GAAP gross profit as % of revenue) | 51.1% | 37.5% | |||
GAAP total operating expenses | $ | 120,347 | $ | 148,510 | |
Shipping and handling costs | (8,146) | (7,211) | |||
Non-GAAP total operating expenses | $ | 112,201 | $ | 141,299 | |
GAAP loss from operations | $ | (49,701) | $ | (101,704) | |
Non-GAAP loss from operations | $ | (49,701) | $ | (101,704) | |
Recent Accounting Pronouncements
Refer to Note 2 − Significant Accounting Policies in the Notes to Consolidated Financial Statements for a full description of recent accounting pronouncements, including the expected dates of adoption and effects on our Consolidated Financial Statements.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
We are exposed to a variety of market risks, including fluctuations in foreign currency exchange rates and interest rates affecting the return on our cash, cash equivalents, and marketable securities.
Foreign Currency Exchange Risk
As we expand internationally, our results of operations and cash flows will become increasingly subject to foreign exchange rate fluctuations. For the years ended December 31, 2023 and 2022, approximately 37% and 38%, respectively, of our total revenue was generated from customers located outside of the United States. Our expenses are generally denominated in the currencies in which our operations are located, which is primarily in the United States, with a portion of expenses incurred in Canada, Europe, Japan, and China. Our results of operations and cash flows are, therefore, subject to fluctuations due to changes in foreign exchange rates in Canadian dollars, Euros, British pounds, Swedish krona, Japanese yen, Chinese yuan, and other foreign currencies. Fluctuations in exchange rates could harm our business in the future. As of December 31, 2023, the effect of a hypothetical 10% adverse change in exchange rates on foreign denominated cash and payables would not have been material and a similar adverse change on foreign denominated receivables would decrease potential cash inflows by $0.8 million.
To date, we have not entered into any foreign currency hedging contracts although we may do so in the future.
Interest Rate Risk
We had cash and cash equivalents of $174.4 million and marketable securities of $146.9 million as of December 31, 2023. All cash, cash equivalents, and marketable securities are held at large commercial banks. Marketable securities consisted entirely of highly rated debt securities including commercial paper, U.S. Treasuries, corporate notes and bonds, U.S. Government agency bonds, certificates of deposit, and similar types of debt securities. Due to the short-term nature and investment grade quality of these investments, we do not believe we have material exposure to changes in interest rates. Additionally, if needed, we have the ability to hold our marketable securities until
55
maturity (without giving effect to any future acquisitions or mergers) and we do not hold or issue financial instruments for trading purposes. Therefore, we do not expect our operating results or cash flows to be affected materially by a sudden change in market interest rates.
Declines in interest rates, however, would reduce future investment income. If overall interest rates had decreased by a hypothetical 10% during the year ended December 31, 2023, our interest income would have decreased income by approximately $1.3 million.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
The financial statements required to be filed pursuant to Item 8 are included in this Annual Report on Form 10-K beginning on page F-1.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
Not applicable.
ITEM 9A. CONTROLS AND PROCEDURES
Disclosure Controls and Procedures
We have established disclosure controls and procedures (as such term is defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) that are designed to provide reasonable assurance that information required to be disclosed in the reports we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC and to ensure that such information is accumulated and communicated to management, including our Chief Executive Officer (principal executive officer) and Chief Financial Officer (principal financial officer), to allow timely decisions regarding required disclosures. Under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, we conducted an evaluation of the effectiveness of our disclosure controls and procedures as of December 31, 2023. Based on this evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were not effective at a reasonable assurance level as of December 31, 2023, due to the material weaknesses described below in Management’s Report on Internal Control over Financial Reporting.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as this term is defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act. All internal control systems, no matter how well designed, have inherent limitations. Therefore, even those systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and presentation.
In Part II, Item 9A of our Annual Report on Form 10-K for the fiscal year ended December 31, 2022, we identified four material weaknesses in our internal control over financial reporting, relating to the operating effectiveness of our internal controls associated with: (i) the accounting for inventory, including excess and obsolescence reserves (the “Inventory MW”), (ii) the accounting for salaries and commissions expense (the “Compensation MW”), (iii) the financial statement close process, including financial reporting, share-based compensation and non-recurring transactions such as impairment of assets and accounting for leases (the “Financial Statement Close Process MW”), and (iv) the accounting for property and equipment, net (the “Property and Equipment MW”).
A material weakness is a deficiency, or combination of deficiencies, in internal control over financial reporting such that there is a reasonable possibility that a material misstatement of the Company's annual or interim financial statements will not be prevented or detected on a timely basis.
56
Remediation of Previously-Identified Material Weaknesses
During 2023, we took a number of actions and incurred expenses of approximately $4.2 million, including the efforts outlined below, designed to improve our internal control over financial reporting to remediate the material weaknesses identified above. These efforts included:
| ● | hired additional personnel with public company experience who have the appropriate level of expertise in the respective areas of accounting, SEC financial reporting, and associated internal controls commensurate with the type, volume, and complexity of our accounting operations and reporting requirements, including a Corporate Controller and an Assistant Controller; |
| ● | continued to supplement our team with advisory consultants to provide additional depth and breadth in our period-end closes, technical accounting, financial reporting capabilities, and internal controls compliance; |
| ● | implemented recommendations from a third-party service provider we engaged to perform an assessment of our internal control design and operation and provide us recommendations to enhance the effectiveness of such controls; |
| ● | engaged a third-party consultant to assess our current enterprise resource planning system and identify opportunities to enhance our use of the system through automating certain controls and processes, for which development of system enhancements were made and continue to be underway; and |
| ● | engaged an accounting advisory consultant who conducted additional trainings on a regular basis related to internal control over financial reporting with our team members including, but not limited to, finance and accounting personnel. |
Based on the efforts above and after demonstrating the operating effectiveness of the related internal controls for a sufficient period of time, management has concluded that the Financial Statement Close Process MW and Compensation MW were remediated as of December 31, 2023.
Evaluation of Controls and Procedures
Under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting as of December 31, 2023, based on the framework set forth in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 Framework). Based on our evaluation under the framework set forth in Internal Control-Integrated Framework, as well as the actions outlined above, while our management concluded that the Financial Statement Close Process MW and Compensation MW were remediated as of December 31, 2023, management concluded that our internal control over financing reporting was not effective at the reasonable assurance level as of December 31, 2023 due to the material weaknesses described below.
Our management concluded that control deficiencies existed as of December 31, 2023, and that these control deficiencies constituted material weaknesses in our internal control over financial reporting. Specifically, management concluded that a portion of the Inventory MW related to the valuation of our inventory, including excess and obsolescence reserves (the “Inventory Valuation MW”) and the Property and Equipment MW continued to exist as of December 31, 2023. The primary cause of the Inventory Valuation MW is our reliance on manual processes to verify the completeness and accuracy of information used in our inventory valuation outputs, and the adequacy and documentation of reviews over these outputs. For the Property and Equipment MW, while the related internal controls were implemented and effective as of December 31, 2023, they were not in all cases in place for a sufficient period of time to demonstrate operating effectiveness as of December 31, 2023.
We have not identified any material misstatements as a result of the material weaknesses discussed above. If not remediated, or if we identify further material weaknesses in our internal control, our failure to establish and maintain effective disclosure controls and procedures and internal control over financial reporting could result in material misstatements in our consolidated financial statements or a failure to meet our reporting and financial obligations.
57
The Company’s independent registered public accounting firm, Ernst & Young LLP, has also issued an audit report on the Company’s internal control over financial reporting, which is included elsewhere in this Annual Report on Form 10-K.
Continuing Remediation Efforts
Management, with oversight from the Audit Committee of our Board of Directors, is taking steps to remediate the control deficiencies that resulted in the Inventory Valuation MW and Property and Equipment MW described above by implementing changes to our internal control over financial reporting. Our plans for remediation include, but are not limited to, the efforts summarized below, which have been or are in the process of being implemented:
| ● | we have engaged accounting advisory consultants to implement new software systems to automate manual key inventory valuation processes and outputs; |
| ● | we will strengthen, document, and implement additional compensating controls; |
| ● | we will continue to focus on effectively operating controls that did not have a sufficient period of time to demonstrate operating effectiveness as of December 31, 2023; and |
| ● | we will continue to evaluate, enhance, and add personnel in the finance organization with a focus on the requisite experience in the areas of accounting, SEC financial reporting, and associated internal controls. |
We expect to continue our efforts to remediate the Inventory Valuation MW and Property and Equipment MW as described above through fiscal year 2024. We believe that the implementation of the above steps will allow us to address the deficient controls within our internal control environment, which will facilitate the remediation of the Inventory Valuation MW and Property and Equipment MW. As we continue to evaluate and work to improve our internal control over financial reporting, we will take additional measures to address control deficiencies and we may modify certain of the remediation measures described above. Following our design and implementation of our remediation efforts, we will need to demonstrate their operating effectiveness. We will not be able to consider the Inventory Valuation MW and Property and Equipment MW remediated until the applicable remedial controls operate for a sufficient period of time and our management has concluded, through testing, that our controls are operating effectively.
Changes in Internal Control over Financial Reporting
Other than related to the changes outlined above to remediate the material weaknesses described above, there have been no changes in our internal control over financial reporting during the fiscal quarter ended December 31, 2023 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
58
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and the Board of Directors of Quanterix Corporation
Opinion on Internal Control over Financial Reporting
We have audited Quanterix Corporation’s internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), (the COSO criteria). In our opinion, because of the effect of the material weaknesses described below on the achievement of the objectives of the control criteria, Quanterix Corporation (the Company) has not maintained effective internal control over financial reporting as of December 31, 2023, based on the COSO criteria.
A material weakness is a deficiency, or combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements will not be prevented or detected on a timely basis. The following material weaknesses have been identified and included in management’s assessment. Management has identified material weaknesses in internal controls related to (i) the accounting for inventory valuation, including excess and obsolescence reserves, and (ii) the accounting for property and equipment, net.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of the Company as of December 31, 2023 and December 31, 2022, the related consolidated statements of operations, comprehensive loss, stockholders' equity and cash flows for each of the three years in the period ended December 31, 2023, and the related notes. These material weaknesses were considered in determining the nature, timing and extent of audit tests applied in our audit of the 2023 consolidated financial statements, and this report does not affect our report dated February 29, 2024, which expressed an unqualified opinion thereon.
Basis for Opinion
The Company's management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management's Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company's internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company's internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company's internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management
59
and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company's assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
Boston, Massachusetts
February 29, 2024
60
ITEM 9B. OTHER INFORMATION
Securities Trading Plans of Directors and Executive Officers
During the three months ended December 31, 2023,
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
Not applicable.
61
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The information required by this Item will be set forth in our Proxy Statement for the 2024 Annual Meeting of Stockholders to be filed with the SEC within 120 days of December 31, 2023 and is incorporated into this Annual Report on Form 10-K by reference.
ITEM 11. EXECUTIVE COMPENSATION
The information required by this Item will be set forth in our Proxy Statement for the 2024 Annual Meeting of Stockholders to be filed with the SEC within 120 days of December 31, 2023 and is incorporated into this Annual Report on Form 10-K by reference.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The information required by this Item will be set forth in our Proxy Statement for the 2024 Annual Meeting of Stockholders to be filed with the SEC within 120 days of December 31, 2023 and is incorporated into this Annual Report on Form 10-K by reference.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
The information required by this Item will be set forth in our Proxy Statement for the 2024 Annual Meeting of Stockholders to be filed with the SEC within 120 days of December 31, 2023 and is incorporated into this Annual Report on Form 10-K by reference.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
Our independent registered public accounting firm is Ernst & Young LLP, New York, NY, (PCAOB ID 42).
The other information required by this Item will be set forth in our Proxy Statement for the 2024 Annual Meeting of Stockholders to be filed with the SEC within 120 days of December 31, 2023 and is incorporated into this Annual Report on Form 10-K by reference.
62
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
The Consolidated Financial Statements are included beginning on page F-1 attached hereto and are filed as part of this Annual Report on Form 10-K.
Financial statement schedules have been omitted since they either are not required, not applicable, or the information is otherwise included.
The following is a list of exhibits filed as part of this Annual Report on Form 10-K:
Exhibit Number |
| Exhibit Description |
| Filed |
| Incorporated by |
| Filing Date |
| SEC File/ |
|---|---|---|---|---|---|---|---|---|---|---|
3.1 | 8-K | 12/15/2017 | 001-38319 | |||||||
3.2 | 10-Q | 8/8/2023 | 001-38319 | |||||||
4.1 | 10-K | 3/13/2020 | 001-38319 | |||||||
4.2 | S-1 | 11/9/2017 | 333-221475 | |||||||
10.1.1+ | S-1 | 11/9/2017 | 333-221475 | |||||||
10.1.2+ | Form of Incentive Stock Option Agreement under the 2007 Stock Option and Grant Plan, as amended | S-1 | 11/9/2017 | 333-221475 | ||||||
10.1.3+ | Form of Non-qualified Stock Option Agreement under the 2007 Stock Option and Grant Plan, as amended | S-1 | 11/9/2017 | 333-221475 | ||||||
10.1.4+ | Form of Restricted Stock Agreement under the 2007 Stock Option and Grant Plan, as amended | S-1 | 11/9/2017 | 333-221475 |
63
Exhibit Number |
| Exhibit Description |
| Filed |
| Incorporated by |
| Filing Date |
| SEC File/ |
|---|---|---|---|---|---|---|---|---|---|---|
10.2.1+ | 2017 Employee, Director and Consultant Equity Incentive Plan | S-1/A | 11/27/2017 | 333-221475 | ||||||
10.2.2+ | S-1/A | 11/27/2017 | 333-221475 | |||||||
10.2.3+ | S-1/A | 11/27/2017 | 333-221475 | |||||||
10.2.4+ | X | |||||||||
10.3+ | Employment Agreement, dated August 3, 2023, between the Registrant and Vandana Sriram | 8-K | 8/9/2023 | 001-38319 | ||||||
10.4+ | 8-K | 3/27/2023 | 001-38319 | |||||||
10.5+ | 8-K | 4/29/2022 | 001-38319 | |||||||
10.6+ | Employment Agreement, dated March 28, 2022, between the Registrant and Dan Pikora | 8-K | 9/8/2022 | 001-38319 |
64
Exhibit Number |
| Exhibit Description |
| Filed |
| Incorporated by |
| Filing Date |
| SEC File/ |
|---|---|---|---|---|---|---|---|---|---|---|
10.7.1* | S-1 | 11/9/2017 | 333-221475 | |||||||
10.7.2* | S-1 | 11/9/2017 | 333-221475 | |||||||
10.7.3@ | 10-Q | 11/6/2020 | 001-38319 | |||||||
10.8.1* | S-1 | 11/9/2017 | 333-221475 | |||||||
10.8.2 | S-1 | 11/9/2017 | 333-221475 | |||||||
10.9.1* | S-1 | 11/9/2017 | 333-221475 |
65
Exhibit Number |
| Exhibit Description |
| Filed |
| Incorporated by |
| Filing Date |
| SEC File/ |
|---|---|---|---|---|---|---|---|---|---|---|
10.9.2* | S-1 | 11/9/2017 | 333-221475 | |||||||
10.10* | S-1 | 11/9/2017 | 333-221475 | |||||||
10.11+ | S-1/A | 11/27/2017 | 333-221475 | |||||||
10.12 | Lease Agreement by and between SSI 900 Middlesex MA LP and the Registrant, dated October 2, 2018 | 8-K | 10/5/2018 | 001-38319 | ||||||
10.13 | Lease Agreement by and between the Registrant and XChange Owner LLC, dated January 28, 2022 | 8-K | 1/31/2022 | 001-38319 | ||||||
10.14+ | Amended and Restated Non-Employee Director Compensation Policy | X | ||||||||
21.1 | 10-K | 3/5/2021 | 001-38319 | |||||||
23.1 | X | |||||||||
31.1 | X | |||||||||
31.2 | X |
66
Exhibit Number |
| Exhibit Description |
| Filed |
| Incorporated by |
| Filing Date |
| SEC File/ |
|---|---|---|---|---|---|---|---|---|---|---|
32.1 | X | |||||||||
97.1 | Policy relating to recovery of erroneously awarded compensation | X | ||||||||
101.INS | Inline XBRL Instance Document | X | ||||||||
101.SCH | Inline XBRL Taxonomy Extension Schema Document | X | ||||||||
101.CAL | Inline XBRL Taxonomy Extension Calculation Linkbase Document | X | ||||||||
101.DEF | Inline XBRL Taxonomy Extension Definition | X | ||||||||
101.LAB | Inline XBRL Taxonomy Extension Label Linkbase Document | X | ||||||||
101.PRE | Inline XBRL Taxonomy Presentation Linkbase Document | X | ||||||||
104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
+ | Management contract or compensatory plan or arrangement. |
* | Confidential treatment has been granted for portions of this Exhibit. Redacted portions have been filed separately with the SEC. |
@ | Portions of this document (indicated by “[***]”) have been omitted because such information is not material and is the type of information that the Registrant treats as private or confidential. |
ITEM 16. FORM 10-K SUMMARY
Not applicable.
67
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
QUANTERIX CORPORATION
Date: February 29, 2024 | By: | /s/ MASOUD TOLOUE, PH.D. |
Masoud Toloue, Ph. D. | ||
President and Chief Executive Officer |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
Signature |
| Title |
| Date |
/s/ MASOUD TOLOUE, PH.D. | President and Chief Executive Officer and Director (Principal Executive Officer) | February 29, 2024 | ||
Masoud Toloue, Ph.D. | ||||
/s/ VANDANA SRIRAM | Chief Financial Officer | February 29, 2024 | ||
Vandana Sriram | (Principal Financial Officer and Principal Accounting Officer) | |||
/s/ MARTIN D. MADAUS, PH.D. | Chairman | February 29, 2024 | ||
Martin D. Madaus, Ph.D. | ||||
/s/ BRIAN J. BLASER | Director | February 29, 2024 | ||
Brian J. Blaser | ||||
/s/ WILLIAM DONNELLY | Director | February 29, 2024 | ||
William Donnelly | ||||
/s/ KAREN A. FLYNN | Director | February 29, 2024 | ||
Karen A. Flynn | ||||
/s/ SARAH E. HLAVINKA | Director | February 29, 2024 | ||
Sarah E. Hlavinka | ||||
/s/ PAUL M. MEISTER | Director | February 29, 2024 | ||
Paul M. Meister | ||||
/s/ LAURIE J. OLSON | Director | February 29, 2024 | ||
Laurie J. Olson | ||||
/s/ DAVID R. WALT, PH.D. | Director | February 29, 2024 | ||
David R. Walt, Ph.D. |
68
QUANTERIX CORPORATION
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and the Board of Directors of Quanterix Corporation
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Quanterix Corporation (the Company) as of December 31, 2023 and 2022, the related consolidated statements of operations, comprehensive loss, stockholders' equity and cash flows for each of the three years in the period ended December 31, 2023, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2023, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company's internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), and our report dated February 29, 2024 expressed an adverse opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of the critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the account or disclosure to which it relates.
Excess Inventory | |
Description of the Matter | As of December 31, 2023, the Company had recognized inventory of $22.4 million. As discussed in Note 2 to the consolidated financial statements, the Company analyzes its inventory levels at each reporting date for slow moving, excess or obsolete inventory and inventory expected to expire prior to being used. In the event that the Company identifies adverse conditions, the carrying value of the inventory is reduced to its estimated net realizable value. Auditing management’s valuation of inventory involved judgement in evaluating management’s analysis and significant assumptions related to projections of future demand which is dependent on market factors. |
How We Addressed the Matter in Our Audit | To audit the Company’s valuation of inventory, we performed audit procedures that included, among others, performing inquiries of management and testing the completeness and accuracy of the underlying data used supporting the Company’s estimate. To evaluate the Company’s estimate of future demand, we independently assessed the sensitivity and impact of reasonably possible changes in forecasted demand and the impact on the Company’s calculation of excess inventory. We evaluated inventory levels compared to forecasted demand, historical sales and specific product considerations. We performed inquiries with appropriate non-financial personnel, including supply chain employees, regarding obsolete or discontinued inventory items and other factors as well as obtained supporting evidence to corroborate management's assertions regarding qualitative judgments about discontinued, slow moving and obsolete inventories. We also evaluated management's ability to accurately forecast demand by comparing actual demand to management's prior estimates. |
/s/
We have served as the Company’s auditor since 2008.
February 29, 2024
F-2
QUANTERIX CORPORATION
CONSOLIDATED BALANCE SHEETS
(amounts in thousands, except per share data)
| |||||
December 31, 2023 |
| December 31, 2022 | |||
ASSETS | |||||
Current assets: |
|
| |||
Cash and cash equivalents | $ | | $ | | |
Marketable securities | | — | |||
Accounts receivable, net of allowance for expected credit losses |
| |
| | |
Inventory |
| |
| | |
Prepaid expenses and other current assets |
| |
| | |
Total current assets | |
| | ||
Restricted cash |
| |
| | |
Property and equipment, net |
| |
| | |
Intangible assets, net |
| |
| | |
Operating lease right-of-use assets | | | |||
Other non-current assets |
| |
| | |
Total assets | $ | | $ | | |
LIABILITIES AND STOCKHOLDERS' EQUITY |
|
|
|
| |
Current liabilities: |
|
|
|
| |
Accounts payable | $ | | $ | | |
Accrued compensation and benefits |
| |
| | |
Accrued expenses and other current liabilities |
| |
| | |
Deferred revenue |
| |
| | |
Operating lease liabilities | | | |||
Total current liabilities |
| |
| | |
Deferred revenue, net of current portion |
| |
| | |
Operating lease liabilities, net of current portion | | | |||
Other non-current liabilities |
| |
| | |
Total liabilities |
| |
| | |
Commitments and contingencies (Note 15) | |||||
Stockholders’ equity: |
|
|
|
| |
Common stock, $ |
|
| |||
Authorized: |
| |
| | |
Additional paid-in capital |
| |
| | |
Accumulated other comprehensive loss | ( | ( | |||
Accumulated deficit |
| ( |
| ( | |
Total stockholders’ equity |
| |
| | |
Total liabilities and stockholders’ equity | $ | | $ | | |
The accompanying notes are an integral part of these Consolidated Financial Statements.
F-3
QUANTERIX CORPORATION
CONSOLIDATED STATEMENTS OF OPERATIONS
(amounts in thousands, except per share data)
Year Ended December 31, | ||||||||
2023 |
| 2022 |
| 2021 | ||||
Revenues: | ||||||||
Product revenue | $ | | $ | | $ | | ||
Service and other revenue |
| |
| |
| | ||
Collaboration and license revenue |
| |
| |
| | ||
Grant revenue | | | | |||||
Total revenues |
| |
| |
| | ||
Costs of goods sold and services: |
|
|
|
|
|
| ||
Cost of product revenue |
| |
| |
| | ||
Cost of service and other revenue |
| |
| |
| | ||
Total costs of goods sold and services |
| |
| |
| | ||
Gross profit | | | | |||||
Operating expenses: |
|
|
|
|
|
| ||
Research and development |
| |
| |
| | ||
Selling, general and administrative |
| |
| |
| | ||
Other lease costs | | | — | |||||
Impairment and restructuring | | | — | |||||
Total operating expenses |
| |
| |
| | ||
Loss from operations |
| ( |
| ( |
| ( | ||
Interest income (expense), net |
| |
| |
| ( | ||
Other income (expense), net |
| |
| ( |
| | ||
Loss before income taxes | ( | ( | ( | |||||
Income tax (expense) benefit | ( | ( | | |||||
Net loss | $ | ( | $ | ( | $ | ( | ||
Net loss per common share, basic and diluted | $ | ( | $ | ( | $ | ( | ||
Weighted-average common shares outstanding, basic and diluted |
| |
| |
| | ||
The accompanying notes are an integral part of these Consolidated Financial Statements.
F-4
QUANTERIX CORPORATION
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
(amounts in thousands)
Year Ended December 31, | ||||||||
2023 |
| 2022 | 2021 | |||||
Net loss | $ | ( | $ | ( | $ | ( | ||
Other comprehensive loss, net of tax: | ||||||||
Unrealized gains on marketable securities | | — | — | |||||
Foreign currency translation | | ( | ( | |||||
Total other comprehensive income (loss) | | ( | ( | |||||
Comprehensive loss | $ | ( | $ | ( | $ | ( | ||
The accompanying notes are an integral part of these Consolidated Financial Statements.
F-5
QUANTERIX CORPORATION
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(amounts in thousands)
Year ended December 31, 2023 | |||||||||||||||||
Common Stock |
|
| |||||||||||||||
Shares |
| Amount |
| Additional paid-in capital |
| Accumulated other comprehensive loss | Accumulated | Total shareholders' equity | |||||||||
Balance at December 31, 2022 | | $ | | $ | | $ | ( | $ | ( | $ | | ||||||
Issuance of common stock under stock plans, including tax effects | | | | — | — | | |||||||||||
Stock-based compensation expense | — | — | | — | — | | |||||||||||
Unrealized gains on marketable securities, net of tax | — | — | — | | — | | |||||||||||
Foreign currency translation | — | — | — | | — | | |||||||||||
Net loss | — |
| — |
| — |
| — | ( | ( | ||||||||
Balance at December 31, 2023 | | $ | | $ | | $ | ( | $ | ( | $ | | ||||||
Year ended December 31, 2022 | |||||||||||||||||
Common Stock |
|
| |||||||||||||||
Shares |
| Amount |
| Additional paid-in capital |
| Accumulated other comprehensive loss | Accumulated | Total shareholders' equity | |||||||||
Balance at December 31, 2021 | | $ | | $ | | $ | | $ | ( | $ | | ||||||
Issuance of common stock under stock plans, including tax effects | | — | | — | — | | |||||||||||
Stock-based compensation expense | — | — | | — | — | | |||||||||||
Foreign currency translation | — | — | — | ( | — | ( | |||||||||||
Net loss | — |
| — |
| — |
| — | ( | ( | ||||||||
Balance at December 31, 2022 | | $ | | $ | | $ | ( | $ | ( | $ | | ||||||
Year ended December 31, 2021 | |||||||||||||||||
Common Stock |
|
| |||||||||||||||
Shares |
| Amount |
| Additional paid-in capital |
| Accumulated other comprehensive loss | Accumulated | Total shareholders' equity | |||||||||
Balance at December 31, 2020 | | $ | | $ | | $ | | $ | ( | $ | | ||||||
Issuance of common stock under stock plans, including tax effects | | | | — | — | | |||||||||||
Sale of common stock in underwritten public offering, net | | | | — | — | | |||||||||||
Stock-based compensation expense | — | — | | — | — | | |||||||||||
Foreign currency translation | — | — | — | ( | — | ( | |||||||||||
Net loss | — |
| — |
| — |
| — | ( | ( | ||||||||
Balance at December 31, 2021 | | $ | | $ | | $ | | $ | ( | $ | | ||||||
The accompanying notes are an integral part of these Consolidated Financial Statements.
F-6
QUANTERIX CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
(amounts in thousands)
Year Ended December 31, | ||||||||
2023 |
| 2022 |
| 2021 | ||||
Cash flows from operating activities: |
|
|
|
|
|
| ||
Net loss | $ | ( | $ | ( | $ | ( | ||
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
| |||
Depreciation and amortization expense |
| |
| |
| | ||
Credit losses (gains) on accounts receivable | | ( | | |||||
Accretion of marketable securities | ( | — | — | |||||
Operating lease right-of-use asset amortization | | | | |||||
Stock-based compensation expense |
| |
| |
| | ||
Impairment | | | — | |||||
Other operating activity | | ( | | |||||
Changes in assets and liabilities: |
|
|
| |||||
Accounts receivable |
| ( |
| |
| ( | ||
Inventory |
| ( |
| |
| ( | ||
Prepaid expenses and other current assets |
| ( |
| ( |
| ( | ||
Other non-current assets |
| ( |
| ( |
| ( | ||
Accounts payable |
| |
| ( |
| | ||
Accrued compensation and benefits, accrued expenses, and other current liabilities |
| |
| ( |
| | ||
Deferred revenue |
| |
| |
| | ||
Operating lease liabilities | ( | ( | ( | |||||
Other non-current liabilities | ( | | ( | |||||
Net cash used in operating activities | ( | ( | ( | |||||
Cash flows from investing activities: |
|
|
|
| ||||
Purchases of marketable debt securities | ( | — | — | |||||
Proceeds from marketable debt securities | | — | — | |||||
Purchases of property and equipment |
| ( |
| ( |
| ( | ||
Proceeds from RADx grant on assets purchased | — | | | |||||
Net cash used in investing activities | ( | ( | ( | |||||
Cash flows from financing activities: |
|
|
| |||||
Proceeds from common stock issued under stock plans |
| |
| |
| | ||
Payments for employee taxes withheld on stock-based compensation awards | ( | — | — | |||||
Sale of common stock in underwritten public offering, net | — | — | | |||||
Payments on notes payable |
| — |
| — |
| ( | ||
Net cash provided by financing activities | | | | |||||
Net increase (decrease) in cash, cash equivalents, and restricted cash |
| ( |
| ( |
| | ||
Effect of exchange rate changes on cash, cash equivalents, and restricted cash | | ( | ( | |||||
Cash, cash equivalents, and restricted cash at beginning of period |
| |
| |
| | ||
Cash, cash equivalents, and restricted cash at end of period | $ | | $ | | $ | | ||
Supplemental disclosure of cash flow information: |
|
|
|
|
|
| ||
Cash paid for taxes | $ | | $ | | $ | — | ||
Cash paid for interest | $ | — | $ | — | $ | | ||
Purchases of property and equipment in accounts payable and accruals | $ | | $ | | $ | | ||
Operating lease right-of-use assets obtained in exchange for lease liabilities | $ | — | $ | | $ | — | ||
Non-cash consideration received under product sales agreement (Note 3, 6) | $ | | $ | — | $ | — | ||
The accompanying notes are an integral part of these Consolidated Financial Statements.
F-7
QUANTERIX CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 1. Organization and Nature of Business
Quanterix Corporation (“Quanterix” or the “Company”) is a life sciences company that has developed next-generation, ultra-sensitive digital immunoassay platforms that advance life sciences research and diagnostics. The Company’s platforms are based on its proprietary digital “Simoa” detection technology and enable customers to reliably detect protein biomarkers in ultra- low concentrations in blood, serum, and other fluids that, in many cases, are undetectable using conventional, analog immunoassay technologies. The ability of the Company’s Simoa platforms to detect proteins in the femtomolar range is enabling the development of novel therapies and diagnostics and has the potential to facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis, and, ultimately, prevention.
The Company also provides contract research services for customers and Laboratory Developed Test (“LDT”) services through its CLIA-certified Accelerator Laboratory (the “Accelerator Laboratory”). The Accelerator Laboratory provides customers with access to Simoa technology and supports multiple projects and services, including sample testing, homebrew assay development, custom assay development, and blood-based biomarker testing. To date, the Company has completed over
Liquidity
The Company has recognized annual losses from operations since inception and has an accumulated deficit of $
Note 2. Significant Accounting Policies
Basis of Presentation
The accompanying Consolidated Financial Statements and notes herein have been prepared in accordance with generally accepted accounting principles in the United States of America (“U.S. GAAP”) and pursuant to the rules and regulations of the U.S. Securities and Exchange Commission (“SEC”) regarding annual financial reporting on Form 10-K.
The Company’s fiscal year is the twelve-month period from January 1 through December 31, and all references to “2023,” “2022,” and “2021,” refer to the fiscal year unless otherwise noted. Certain amounts in the prior years’ Consolidated Financial Statements have been reclassified to conform to the current year’s presentation.
Use of Estimates
The preparation of the Consolidated Financial Statements and Notes to Consolidated Financial Statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities at the dates of the Consolidated Financial Statements, and the reported amounts of revenues and expenses during each fiscal year. Such estimates include, but are not limited to, revenue recognition, valuation of inventory, leases, valuation and impairment of long-lived assets, recoverability of deferred tax assets, and stock-based compensation expense. The Company bases its estimates on historical experience, known trends, worldwide economic conditions, both general and specific to the life sciences industry, and other relevant factors it believes to be reasonable under the circumstances. On an ongoing basis, management evaluates its estimates and changes in estimates are recorded in the period in which they become known. Actual results could differ from those estimates.
F-8
Principles of Consolidation
The Consolidated Financial Statements include the accounts of Quanterix and its wholly-owned subsidiaries. All intercompany transactions have been eliminated in consolidation.
In accordance with Accounting Standards Codification (“ASC”) 810 – Consolidation, the Company assesses the terms of its non-marketable equity investments in entities to determine if any meet the definition of a variable interest entity (“VIE”) and require consolidation into its Consolidated Financial Statements. Refer to Note 19 − Variable Interest Entities for further discussion.
Foreign Currency
The functional currency of the Company’s subsidiaries is their respective local currencies. These subsidiary financial statements are translated into U.S. dollars using the period-end exchange rates for assets and liabilities, average exchange rates during the corresponding period for revenue and expenses, and historical rates for equity. The effects of foreign currency translation adjustments are recorded in accumulated other comprehensive income (loss), a component of stockholders’ equity on the Consolidated Balance Sheets.
Foreign currency transaction gains (losses) are included in other income (expense), net on the Consolidated Statements of Operations. Foreign exchange losses were not material during the year ended December 31, 2023, and were $
Segment Information
Operating segments are defined as components of an enterprise about which separate discrete information is available for evaluation by the chief operating decision-maker (“CODM”), in deciding how to allocate resources and assess performance. The Company’s CODM is the chief executive officer, who reviews the Company's operations and manages its business as a single operating segment as of December 31, 2023.
Revenue from Contracts with Customers
The Company generates the majority of its revenues from contracts with customers and accounts for them pursuant to the Financial Accounting Standards Board (“FASB”) Accounting Standards Codification Topic 606 - Revenue from Contracts with Customers (“ASC 606”). Refer to the section titled “Grant Revenue” below for discussion the Company’s accounting policy for revenue generated from grant awards.
For contracts with customers, the Company recognizes revenue when a customer obtains control of promised products or services, for an amount that reflects the consideration expected to be received in exchange for those products or services. The Company follows the five step revenue model prescribed by ASC 606 to determine revenue recognition: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price, including variable consideration, if any; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the entity satisfies a performance obligation. Revenues are presented net of any sales, value added, or similar taxes collected from customers and remitted to the government.
The Company accounts for a contract when it has approval and commitment from both parties, the fees, payment terms and rights of the parties regarding the products or services to be transferred are identified, the contract has commercial substance, and it is probable that substantially all of the consideration for the products and services expected to be transferred is collectible. The Company applies judgment in determining the customer’s ability and intention to pay for services expected to be transferred, which is based on factors including the customer’s payment history, management’s ability to mitigate exposure to credit risk (for example, requiring payment in advance of the transfer of products or services, or the ability to stop transferring promised products or services in the event a customer fails to pay consideration when due), and experience selling to similarly situated customers.
F-9
The Company’s contracts may include either a single promise (referred to as a performance obligation) to transfer a product or service, or a combination of multiple performance obligations to transfer products or services. The Company evaluates the existence of multiple performance obligations within its contracts by using judgment to determine if (1) the customer can benefit from each contractual promise on its own or together with readily available resources and (2) the transfer of each contractual promise is separately identifiable from other promises in a contract. When both criteria are met, each promise is accounted for as a separate performance obligation. Additionally, the Company has elected the practical expedient under ASC 606 to account for shipping and handling as an activity to fulfill a promise to transfer a product, and therefore does not evaluate whether shipping and handling activities are promised services to its customers.
Contracts that include rights to additional products or services that are exercisable at a customer’s discretion are generally considered options. The Company assesses if these options provide a material right to the customer and if so, the material right is considered a performance obligation. The identification of material rights requires judgment to determine the value of the option to purchase additional products and services in relation to options that may be provided to, and prices paid by, customers in the normal course of business. Material rights are recognized when exercised by a customer or upon expiration of the right.
The Company determines the transaction price of its contracts based on the amount of consideration it expects to be entitled to, which is generally equal to the contract amount. In some cases, contracts contain variable consideration which primarily relates to (1) sales- and usage-based royalties related to the license of intellectual property in collaboration and license contracts and (2) contracts with minimum purchase commitments. For sales and usage-based royalties, ASC 606 provides an exception to estimating variable consideration. Under this exception, the Company recognizes revenues from sales- or usage-based royalty revenue at the later of when the sales or usage occurs or the satisfaction (or partial satisfaction) of the performance obligation to which the royalty has been allocated. All other variable amounts are constrained to the minimum guaranteed contract amount so that a reversal of cumulative revenue does not occur in future periods. Once there is no longer uncertainty over a variable amount, any incremental fees the Company is entitled to are allocated to the related performance obligation(s).
For contracts that contain multiple performance obligations, the Company allocates the transaction price among the performance obligations on a relative basis according to their standalone selling prices (“SSP”). Determining the SSP for performance obligations requires judgment and is based on factors including prices charged to customers in observable transactions, internal pricing objectives and list prices, and pricing of similar products. The Company uses a range of amounts to estimate SSP and has more than one range for certain products and services based on the geographic location of customers and sales channel.
Product Revenue
The Company’s product revenues are composed of instruments, assay kits, replacement parts, and other consumables such as reagents and antibodies. Products are sold directly to customers and are also sold through distributors in EMEA and Asia Pacific regions. Direct instrument sales include installation and an initial year of implied service-type warranties. The Company has determined that the instrument and installation are a combined performance obligation as the customer cannot benefit from the instrument without the installation and no other vendors can provide the installation of the Company’s specialized instruments. The implied service-type warranty is considered a separate performance obligation since a customer could benefit from it independently with readily available resources and is capable of being sold on its own. Sales of instruments to distributors include a license to import and resell the instruments. The Company has determined these distributor licenses are part of a combined performance obligation with the instrument as the distributor only benefits from the combination of the instrument and ability to resell it.
Instrument sales may also be bundled with assays and other consumables, training, and/or an extended service warranty, each of which is considered a separate performance obligation.
Product revenues for direct instrument sales to customers are recognized upon completion of the instrument’s installation. For instrument sales to distributors, revenue is recognized based on the agreed upon shipping terms (either upon shipment or delivery) as that is when title passes to the customer.
F-10
Service Revenue
Service revenues are composed of contract research services, initial year of implied service-type warranties, extended services warranty contracts, repair services, and other services such as training. Contract research services are provided through the Company’s Accelerator Laboratory and generally consist of fixed fee contracts.
Revenues from contract research services are recognized at the point in time when the Company completes and delivers its research results on each individually completed study, or over time if the contractual provisions allow for the collection of transaction consideration for costs incurred plus a reasonable margin through the period of performance of the services. For contract research services recognized over time, the Company uses the output method based on the number of completed results. Revenues from other services are recognized at the point in time when the training or other services are delivered as the customer simultaneously receives and benefits from the services. Revenues from service-type warranties are recognized ratably over the contract service period.
Collaboration and License Revenue
Collaboration and license revenues are composed of revenue associated with licensing to third parties the Company’s technology, intellectual property, and know-how associated with our instruments and for related services. License arrangements consist of sales or usage-based fees and/or future royalties. Revenues are recognized at the point in time the license is delivered as the underlying license is considered functional intellectual property. Royalty revenues that are sales- or usage- based are recognized at the later of when the sales or usage occurs and the satisfaction (or partial satisfaction) of the performance obligation to which the royalty has been allocated.
Contract Assets and Liabilities
Accounts Receivable and Allowance for Credit Losses
The Company is exposed to credit losses primarily through accounts receivable from sales of its products and services. Accounts receivable includes amounts billed and currently due from customers. Since the only condition for payment of the Company’s invoices is the passage of time, the Company records a receivable on the date the invoice is issued. Also included in accounts receivable are unbilled amounts resulting from revenue exceeding the amount billed to the customer, where the right to payment is unconditional. If the right to payment for services performed was conditional on something other than the passage of time, the unbilled amount would be recorded as a separate contract asset.
The Company’s expected loss allowance methodology is developed using historical collection experience, current and future economic and market conditions, and a review of the current status of customers’ accounts receivable. Specific allowance amounts are established to record the appropriate provision for customers that have a higher probability of default. The Company monitors changes to the receivables balance on a timely basis, and balances are written off as they are determined to be uncollectable after collection efforts have been exhausted.
Generally, the Company’s contracts are non-cancellable. For contracts that are cancellable by the customer, the Company does not record a receivable when it issues an invoice. The Company records accounts receivable on these contracts only up to the amount of revenue recognized but not yet collected.
The Company’s payment terms vary by the type and location of the customer and the products or services offered. Payment from customers is generally required
The Company refers to contract liabilities as deferred revenue on the Consolidated Balance Sheets. For deferred revenue, the Company applies the practical expedient under ASC 606 that allows for the exclusion of (1) contracts with original expected length of one year or less and (2) contracts for which revenue is recognized at the amount to which the Company has the right to invoice for services performed from the amount of remaining performance obligations disclosed.
F-11
Costs to Obtain Contracts
The Company capitalizes commissions paid to its sales representatives and related fringe benefit costs that are incremental to obtaining customer contracts. Capitalized commissions are recorded in prepaid expenses and other current assets and other non-current assets on the Consolidated Balance Sheets. These commissions are amortized over the life of the contract and are recorded in cost of goods sold and selling, general and administrative expense on the Consolidated Statements of Operations. The Company has elected the practical expedient allowing commissions with an amortization period of one year or less to be expensed as incurred.
Commissions associated with instrument sales are generally earned when installation is complete and title to the instrument has transferred. Commissions associated with consumables sales are earned when title to the product transfers. Commissions associated with warranty and extended service contracts are earned upon booking.
Warranties
The Company provides an initial year of warranty and maintenance service related to its instruments sold directly to customers and sells extended warranty contracts for additional periods. The Company defers revenue associated with these warranty services and recognizes them ratably over the service period.
Grant Revenue
Accounting for grants does not fall under ASC 606, as the grantor will not benefit directly from the Company’s expansion or product development, and no products or services are transferred to the grantor. As there is no authoritative guidance under U.S. GAAP on accounting for grants to for profit business entities from government entities, the Company accounts for grants by analogy to International Accounting Standards Topic 20, Accounting for Government Grants and Disclosure of Government Assistance (“IAS 20”) and ASC Topic 958, Not for-Profit Entities (“ASC 958”). The decision to account for a grant under IAS 20 or ASC 958 is based on whether the grantor is a government entity.
The Company records grants related to assets as a deduction in calculating the carrying value of the asset, and to record grants related to income separately on the Consolidated Statements of Operations on a gross basis as grant revenue. The related expenses are recorded on a gross basis within operating expenses. These methods are elections an entity can make under both IAS 20 and ASC 958.
The Company recognizes grant revenue as the Company performs services under the arrangement when the funding is committed and as each grant’s activities are performed. The timing of revenue recognition and receipt of funding varies by grant and can be independent from performance of the related activities, such as an upfront payment of the award value, or subsequent to the Company’s requests for reimbursement for already performed activities (subject to the approval of the granting organization).
Cost of Goods Sold and Services
Cost of Product Revenue
Cost of product revenue consists of manufacturing and assembly costs for instruments, related reagents, other consumables, contract manufacturer costs, personnel costs, royalties, overhead, and other direct costs related to product sales. Raw material part costs include inbound freight, shipping and handling costs associated with purchased goods, contract manufacturer costs, personnel costs, royalties, overhead and other direct costs related to product sales. Additionally included in cost of product revenue are royalty fees due to third parties from revenue generated by collaboration or license deals.
Cost of Service and Other Revenue
Cost of services and other revenue consists of direct costs associated with operating the Company’s Accelerator Laboratory on behalf of its customers, including raw materials, personnel costs, royalties, allocated overhead costs that include facility and other related costs, and other direct costs. Additional costs include costs related to warranties, and other costs of servicing equipment at customer sites.
F-12
Research and Development Expenses
Research and development expense consists primarily of personnel costs, research supplies, third-party development costs for new products, materials for prototypes, quality assurance, and allocated overhead costs that include facility and other related costs. The Company accounts for nonrefundable advance payments for products and services that will be used in future research and development activities as expense when the service has been performed or when the products have been received.
For arrangements in which the Company receives funding from third parties for research and development activities (excluding the government sponsored arrangements), the Company assesses whether the arrangement is within the scope of ASC 730 – Research and Development. When the Company is entitled to receive reimbursements, does not have an obligation to repay, does not transfer products or services, and is the primary beneficiary of the activities, the Company records the reimbursable amounts as a reduction to research and development expense. Amounts reimbursed in excess of the cost incurred by the Company are recorded within other income (expense), net on the Consolidated Statements of Operations.
Reimbursable amounts recorded as a reduction to research and development expenses were not material during the years ended December 31, 2023, 2022, and 2021. Amounts reimbursed in excess of costs incurred by the Company related to activities funded by third parties were $
Selling, General and Administrative Expenses
Selling, general and administrative expense consists primarily of personnel costs for our sales and marketing, finance, legal, human resources, and general management teams, shipping and handling for product sales, other general and administrative costs, as well as professional services costs, such as marketing, advertising, legal and accounting services, and allocated overhead costs that include facility and other related costs. The classification of shipping and handling costs for product sales as selling, general and administrative expenses varies from company to company with some companies recording these as selling, general and administrative expenses and others recording such expenses within costs of goods sold for products. To the extent the classification of its shipping and handling costs differs from the reporting approach used by other companies, the Company’s gross margins may not be comparable with those reported by such other companies.
Net Loss Per Share
Basic net loss per common share attributable to common stockholders is calculated by dividing the loss attributable to common stockholders by the weighted-average number of common shares outstanding. Diluted net loss per common share attributable to common stockholders is calculated under the treasury stock method by dividing the loss attributable to common stockholders by the diluted weighted-average number of common shares outstanding. Diluted weighted-average shares outstanding reflect the dilutive effect, if any, of potential common shares issued, such as unvested common stock, unvested restricted stock units (“RSUs”), common stock options, and shares estimated to be purchased under the Company’s employee stock purchase plan (“ESPP”). During periods when the Company is in a net loss position, these potential common shares are excluded from the diluted net loss per common share attributable to common stockholders because their effect would be anti-dilutive. Accordingly, basic and diluted net loss per common share attributable to common stockholders were the same for all periods presented.
Cash and Cash Equivalents
Cash and cash equivalents consist of cash deposits and short-term, highly liquid marketable securities that are readily convertible into cash, with original maturities of three months or less. Cash and cash equivalents consist of the following (in thousands):
As of December 31, | |||||
2023 |
| 2022 | |||
Cash | $ | | $ | | |
Money market funds | | | |||
Marketable securities | | — | |||
Total cash and cash equivalents | $ | | $ | | |
F-13
Restricted Cash
The following table summarizes the period ending cash and cash equivalents as presented on the Consolidated Balance Sheets and the total cash, cash equivalents, and restricted cash as presented on the Consolidated Statements of Cash Flows (in thousands):
As of December 31, | |||||
2023 |
| 2022 | |||
Cash and cash equivalents | $ | | $ | | |
Restricted cash | | | |||
Cash, cash equivalents, and restricted cash | $ | | $ | | |
Restricted cash consists of collateral for a letter of credit issued as security for two of the Company’s leased facilities and to secure the Company’s corporate credit card program. The short-term or long-term classification is determined in accordance with the expiration of the underlying letter of credit and security.
Marketable Securities
The Company’s current portfolio of marketable securities is entirely debt securities and may at any time include commercial paper, U.S. Treasuries, corporate notes and bonds, U.S. Government agency bonds, certificates of deposit, and similar types of debt securities. Marketable debt securities with original maturities of three months or less at the time of purchase are recorded in cash equivalents on the Consolidated Balance Sheets as they are considered highly liquid and readily convertible into cash. All other marketable securities, including those with maturities beyond one year, are recorded as current assets on the Consolidated Balance Sheets based on their highly liquid nature and because such securities are available for use in current operations.
The Company classifies its marketable securities as either held to maturity, available-for-sale, or trading at the time of purchase and re-evaluates such classification at each balance sheet date. All of the Company’s marketable securities are currently classified as available-for-sale as it may use them in current operations. Available-for-sale securities are recorded at fair value (refer to Note 6 − Fair Value of Financial Instruments).
Unrealized gains and losses (other than impairment or credit related losses) are recorded in accumulated other comprehensive income (loss), a component of stockholders’ equity on the Consolidated Balance Sheets. Realized gains and losses are determined using the specific identification method and are recorded in other income (expense), net on the Consolidated Statements of Operations.
Quarterly, or more frequently if circumstances warrant, the Company monitors its marketable securities for impairment. In the event a security’s fair value is less than its amortized cost basis, the Company evaluates whether an impairment exists and if the impairment is a result of credit loss or other factors. For a security in an unrealized loss position, if the Company intends to sell the security in an unrealized loss position, or it is more likely than not that the Company will be required to sell the security before recovery of its amortized cost basis, an impairment loss equal to the difference between the security’s fair value and amortized cost basis is recorded in other income (expense), net. Additionally, the Company determines if a credit loss exists by considering information about the collectability of the security, current market conditions, and the issuer’s financial condition. If a decline in fair value is a result of a credit loss, an allowance for credit losses is recorded in other income (expense), net on the Consolidated Statement of Operations, limited to the portion attributed to the credit loss.
The Company has also elected the practical expedient to separately present accrued interest receivable from its marketable securities balance. Such accrued interest is recorded in prepaid expenses and other current assets on the Consolidated Balance Sheets and is not included in the assessment and measurement of impairment of its marketable securities.
Inventory
Inventory consists of instruments, assays, and the materials required to manufacture instruments and assays.
F-14
Inventory is stated at the lower of cost or net realizable value on a first-in, first-out (“FIFO”) basis and includes the cost of materials, labor, and manufacturing overhead. The Company analyzes its inventory levels on each reporting date for slow-moving, excess, and obsolete inventory, and inventory expected to expire prior to being used. These analyses require judgment and are based on factors including, but not limited to, recent historical activity, anticipated or forecasted demand for the Company’s products (developed through its planning and sales and marketing inputs, and market conditions). If the Company identifies adverse conditions exist, the carrying value of the inventory is reduced to its estimated net realizable value by providing estimated reserves for excess or obsolete inventory.
The Company adjusts the reserves for excess or obsolete inventory and records additional inventory write downs based on unfavorable changes in estimated customer demand or actual market conditions that may differ from its projections.
Property and Equipment
Property and equipment, including leasehold improvements, are stated at cost, net of accumulated depreciation. These assets are depreciated over their estimated useful lives using the straight-line method. Expenditures for maintenance and repairs are charged to expense as incurred, whereas significant expenditures that extend the useful lives of existing assets are capitalized as additions to property and equipment.
Depreciation is calculated based upon the following estimated useful lives:
Estimated Useful Life | |
Laboratory and manufacturing equipment | |
Office furniture and equipment | |
Computers and software | |
Leasehold improvements | Shorter of asset's life or remaining lease term |
Leases
The Company enters into operating leases for office, laboratory, and manufacturing spaces, as well as office equipment, and determines whether an arrangement is a lease at inception of the arrangement. The Company accounts for a lease when it has the right to control the leased asset for a period of time, while obtaining substantially all of the assets’ economic benefits. Leases are recorded on the Consolidated Balance Sheets as operating lease right-of-use (“ROU”) assets and current or non-current operating lease liabilities, as applicable. All of the Company’s leases are classified as operating leases. Additionally, the Company does not separate lease and non-lease components.
Operating lease ROU assets and lease liabilities are recognized at the lease commencement date based on the estimated present value of the future minimum lease payments over the lease term and any initial direct costs incurred. Initial direct costs are incremental costs of a lease that would not have been incurred had the lease not been executed. The discount rate used to determine the present value of the lease payments is the Company’s incremental borrowing rate on a collateralized basis for a similar term and amount, as generally an implicit rate in the lease is not readily determinable. To estimate its incremental borrowing rate, a credit rating applicable to the Company is estimated using a synthetic credit rating analysis since the Company does not currently have an agency-based credit rating.
The Company’s lease agreements can contain lease and non-lease components. The Company accounts for the lease and fixed payments for non-lease components as a single lease component under ASC 842– Leases, which increases the amount of the ROU assets and lease liabilities. Most of the Company’s lease agreements also contain variable payments, primarily maintenance, utility, and other-related costs, which are expensed as incurred and not included in the measurement of the ROU assets and lease liabilities.
Some of the Company’s leases contain options to extend or terminate the lease. When determining the lease term, these options are included in the measurement and recognition of the Company’s ROU assets and lease liabilities when it is reasonably certain that the Company will exercise the option(s). The Company considers various economic factors when making this determination, including, but not limited to, the significance of leasehold improvements incurred in the leased space, the difficulty in replacing the asset, underlying contractual obligations, and specific characteristics unique to a particular lease. Subsequent to
F-15
entering into a lease arrangement, the Company reassesses the certainty of exercising options to extend or terminate a lease. When it becomes reasonably certain that the Company will exercise an option that was not included in the lease term, the Company accounts for the change in circumstances as a lease modification, which results in the remeasurement of the ROU asset and lease liability as of the modification date.
Leases with a term of 12 months or less upon commencement are not recorded on the Consolidated Balance Sheets and are recorded to expense on a straight-line basis over the lease term.
Impairment of Goodwill
The Company assesses goodwill for impairment at the reporting unit level annually and whenever events occur or circumstances change that would more likely than not reduce the fair value of a reporting unit below its carrying amount. Such events or circumstances could include the occurrence of operating losses, a significant decline in earnings, or significant changes in or restructuring of the business. The impairment test is first performed at the reporting unit level using a qualitative assessment to determine whether it is more likely than not that the fair value of the reporting unit is less than the carrying value. If the reporting unit does not pass the qualitative assessment, the reporting unit’s carrying value is compared to its fair value, using estimates including forecasts of discounted future cash flows and peer market multiples. An impairment charge is recorded equal to the excess of the reporting unit’s carrying value over its fair value.
Impairment of Long-Lived Assets
The Company’s long-lived assets consist of operating lease ROU assets, property and equipment, and intangible assets. The Company reviews the carrying amount of its long-lived assets for impairment whenever events or circumstances indicate that the estimated useful lives may warrant revision, or that the carrying amount of the assets may not be fully recoverable. To assess whether a long-lived asset or group of assets has been impaired, the estimated undiscounted and discounted future cash flows for the estimated remaining useful life or estimated lease term of the asset (or the primary asset in the asset group) are compared to their carrying values. Significant judgment is required to estimate future cash flows, including, but not limited to, the expected use of the asset (group), historical customer retention rates, technology roadmaps, customer awareness, trademark and trade name history, contractual provisions that could limit or extend an asset's useful life, market data, discount rates, potential sublease opportunities including rent and rent escalation rates, time to sublease, and free rent periods. To the extent that the future cash flows are less than the carrying value, the asset(s) are impaired and written down to its estimated fair value.
Software Development Costs
The Company develops and modifies software related to the operation of some of its instruments and internal use software supporting the Company’s operations. Certain costs incurred during the application development stage including external direct costs of services used in the development or internal personnel costs for employees directly associated with the development, are capitalized. The Company begins depreciating these costs over the life of the related asset upon completion of a working model or when it is ready for its intended use. Capitalized software development costs related to software owned or developed by the Company are recorded in property and equipment on the Consolidated Balance Sheets. Capitalized software development costs related to software hosted by a vendor (i.e. cloud computing) are recorded in prepaid expenses and other current assets, and other non-current assets on the Consolidated Balance Sheets.
Costs incurred during the preliminary project stage and post-configuration stages are expensed as incurred.
Fair Value of Financial Instruments
The carrying amount reflected in the Consolidated Balance Sheets for cash, restricted cash, accounts receivable, prepaid expenses and other current assets, accounts payable, accrued compensation and benefits, and accrued expenses and other current liabilities approximate their fair values due to their short-term nature.
Additionally, the Company has certain financial assets that are required to be measured at fair value on a recurring basis including cash equivalents and marketable securities. Pursuant to the accounting standards for fair value measurements, the fair values of these financial assets are classified as Level 1, 2, or 3 within the fair value hierarchy as follows:
F-16
| ● | Level 1: Observable inputs based on unadjusted quoted prices in active markets for identical assets. |
| ● | Level 2: Inputs, other than Level 1 inputs, that are observable either directly or indirectly, such as quoted prices for similar assets, quoted prices in markets that are not active, other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets. |
| ● | Level 3: Unobservable inputs for which there is little or no market data and such inputs are significant to the fair value of the assets. These inputs reflect the Company’s assumptions about the inputs that market participants would use in pricing the asset. |
To the extent that the valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment.
Income Taxes
The Company recognizes deferred tax assets and liabilities for the expected future tax consequences of events that have been recognized in the Consolidated Financial Statements or tax returns. Deferred tax assets and liabilities are determined based on differences between the carrying amount and the tax basis of the assets and liabilities using the enacted tax rates in effect in the years in which the differences are expected to reverse. A valuation allowance against deferred tax assets is recorded if, based on the weight of the available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized.
The Company accounts for uncertain tax positions using a more likely than not threshold for recognizing uncertain tax positions, in accordance with ASC 740 – Income Taxes. The evaluation of uncertain tax positions is based on factors that include, but are not limited to, changes in tax law, the measurement of tax positions taken or expected to be taken in tax returns, the effective settlement of matters subject to audit, new audit activity, and changes in facts or circumstances related to a tax position. The Company evaluates uncertain tax positions on an ongoing basis and adjusts any liability recorded to reflect subsequent changes in the relevant facts surrounding the uncertain positions. Amounts recorded for uncertain tax positions, including interest and penalties, are recorded in income tax (expense) benefit on the Consolidated Statement of Operations.
Credit, Product, and Supplier Concentrations and Off-Balance-Sheet Risk
Financial instruments that potentially expose the Company to concentrations of credit risk primarily consist of cash, cash equivalents, marketable securities, and accounts receivable. The Company limits its risk exposure by having its cash, cash equivalents, and marketable securities held at large commercial banks.
Customers outside the United States represented
For the year ended December 31, 2023,
The Company is also subject to supply chain risks related to outsourced manufacturing of its instruments. Although there are a limited number of manufacturers for its instruments, the Company believes that other suppliers could provide similar manufacturing on comparable terms. A change in suppliers, however, could cause a delay in manufacturing and a possible loss of sales, which would adversely affect operating results. In addition to outsourced manufacturing of its instruments, the Company also purchases antibodies through a number of different suppliers. Although a disruption in service from any one of its antibody suppliers is possible, the Company believes that it would be able to find an adequate supply from alternative suppliers.
Stock-Based Compensation
The Company measures and recognizes stock-based compensation expense by calculating the estimated fair value of stock options, RSUs, or purchase rights issued under the Company’s ESPP on the grant date. The Company generally issues new common
F-17
shares upon exercise of options and vesting of RSUs. Awards granted by the Company are routine in nature including new hire, annual, and promotion grants.
The fair value of stock options and purchase rights under the ESPP is estimated using the Black-Scholes option-pricing model. The Black-Scholes model requires the Company to make assumptions about the expected or contractual term of the option or purchase right, the expected volatility, risk-free interest rates, and expected dividend yield. The Company estimates the expected term of options granted to employees utilizing historical exercise data. The expected term is applied to the stock option grant group as a whole, as the Company does not expect substantially different exercise or post-vesting termination behavior among its employee population. During 2023, the expected volatility was based on the Company’s historical volatility. Prior to 2023, the expected volatility was estimated based on both the Company’s volatility and the average volatility for comparable publicly traded companies over a period equal to the expected term of the related stock-based awards. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant, commensurate with the expected term. The expected dividend yield is
The fair value of RSUs is determined using the closing market price of the Company’s common stock on the grant date.
The Company recognizes stock-based compensation expense on a straight-line basis over the award’s requisite service period, which is the vesting period for stock options and RSUs, and the offering period for purchase rights under the ESPP. The Company recognizes forfeitures as they occur.
Advertising
The Company expenses the cost of advertising as incurred and records them in selling, general and administrative expense in the Consolidated Statements of Operations. Advertising expense was $
Recent Accounting Pronouncements
Recent Accounting Standards To Be Adopted
In June 2022, the FASB issued ASC Update No. 2022-03, Fair Value Measurement (Topic 820): Fair Value Measurement of Equity Securities Subject to Contractual Sale Restrictions. This update clarifies the guidance in Topic 820 related to measuring the fair value of an equity security subject to contractual restrictions that prohibit the sale of an equity security, as well as introduces new disclosure requirements for these types of equity securities. The new standard will be effective for the Company on January 1, 2024. The adoption of this standard is not expected to have a material impact on the Company’s financial position or results of operations.
In November 2023, the FASB issued ASC Update No. 2023-07, Segment Reporting (Topic 280): Improvement to Reportable Segment Disclosures. This update enhances reportable segment disclosure requirements by requiring public entities to provide disclosures of significant segment expenses and other segment items, as well as disclosures about a reportable segment’s profit or loss and assets that are currently required annually in interim periods. The new standard will be effective for the Company’s annual financial statements for the period beginning on January 1, 2024. The adoption of this standard is not expected to have a material impact on the Company’s disclosures.
In December 2023, the FASB issued ASC Update No. 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures. This update enhances income tax disclosure requirements by requiring public entities to provide additional information in its tax rate reconciliation and additional disclosures about income taxes paid. The update is effective for annual periods beginning after December 15, 2024. Early adoption is permitted for annual financial statements that have not yet been issued or made available for issuance and the amendments in this update should be applied prospectively, but entities have the option to apply it retrospectively. The Company is currently evaluating the impact that the standard will have on its Consolidated Financial Statements.
F-18
Note 3. Revenue and Related Matters
Revenue from Contracts with Customers
The Company’s customers primarily consist of entities engaged in the life sciences research that pursue the discovery and development of new drugs for a variety of neurologic, oncologic, cardiovascular, infectious disease, and other protein biomarkers associated with diseases. The Company’s customer base includes pharmaceutical, biotechnology, contract research organizations, academic, and government institutions.
Disaggregated Revenue
When disaggregating revenue, the Company considers all of the economic factors that may affect its revenues. The following tables disaggregate the Company’s revenue from contracts with customers by geography, based on the location products and services are consumed, and revenue type (in thousands):
Year Ended December 31, 2023 | ||||||||||||
(in thousands) | North America |
| EMEA |
| Asia Pacific |
| Total | |||||
Product revenue: | ||||||||||||
Instruments | $ | | $ | | $ | | $ | | ||||
Consumable and other products | | | | | ||||||||
Total | $ | |
| $ | |
| $ | |
| $ | | |
Service revenue: | ||||||||||||
Service-type warranties | $ | | $ | | $ | | $ | | ||||
Research services | | | |
| | |||||||
Other services | | | ( | | ||||||||
Total | $ | | $ | | $ | | $ | | ||||
Collaboration and license revenue: | | — | — | | ||||||||
Total | $ | | $ | — | $ | — | $ | | ||||
Year Ended December 31, 2022 | ||||||||||||
(in thousands) | North America |
| EMEA |
| Asia Pacific |
| Total | |||||
Product revenue: | ||||||||||||
Instruments | $ | | $ | | $ | | $ | | ||||
Consumable and other products | | | | | ||||||||
Total | $ | |
| $ | |
| $ | |
| $ | | |
Service and other revenues: | ||||||||||||
Service-type warranties | $ | | $ | | $ | |
| $ | | |||
Research services | | | |
| | |||||||
Other services | | | |
| | |||||||
Total | $ | | $ | | $ | | $ | | ||||
Collaboration and license revenue: | ||||||||||||
Total | $ | | $ | | $ | | $ | | ||||
F-19
Year Ended December 31, 2021 | ||||||||||||
(in thousands) | North America |
| EMEA |
| Asia Pacific |
| Total | |||||
Product revenue: | ||||||||||||
Instruments | $ | | $ | | $ | |
| $ | | |||
Consumable and other products | | | |
| | |||||||
Total | $ | |
| $ | |
| $ | |
| $ | | |
Service and other revenue: | ||||||||||||
Service-type warranties | $ | | $ | | $ | |
| $ | | |||
Research services | | | |
| | |||||||
Other services | | | |
| | |||||||
Total | $ | | $ | | $ | | $ | | ||||
Collaboration and license revenue: | ||||||||||||
Total | $ | | $ | | $ | — | $ | | ||||
UltraDx Product Sales Agreement
On May 26, 2022, the Company and UltraDx Limited (“UltraDx”), a company formed by ARCH Venture Partners (“ARCH”), entered into an agreement (the “UltraDx Agreement”). Under the UltraDx Agreement, the Company agreed to supply UltraDx with HD-X instruments (both fully assembled and disassembled), assays and assay components, and granted a co-exclusive license to manufacture, seek Chinese regulatory approval of (including performance of any necessary research and development activities), and commercialize, HD-X instruments assembled in China and related assays in the Chinese in vitro diagnostic (“IVD”) market. Refer to Note 16 − Related Party Transactions for a discussion of the related party relationships between Quanterix and these entities.
The Company determined that the instruments, components, and licenses formed a single, combined performance obligation. The consideration due to the Company included (1) cash proceeds of $
In the second quarter of 2023, UltraDx completed the qualified preferred share financing and issued to the Company million ordinary shares. Refer to Note 6 − Fair Value of Financial Instruments for the Company’s disclosures related to determining the fair value of the shares received. Also refer to Note 19 − Variable Interest Entities for additional information on the Company’s investment in UltraDx as a result of the share issuance.
During the year ended December 31, 2023, the Company recognized $
Eli Lilly and Company Service Revenue Agreements
On February 25, 2022, the Company entered into a Master Collaboration Agreement with Eli Lilly and Company (“Lilly”) establishing a framework for future projects focused on the development of Simoa immunoassays (the “Lilly Collaboration Agreement”). The Company also entered into a statement of work under the Lilly Collaboration Agreement to perform assay research and development services within the field of Alzheimer’s disease. In connection with the Lilly Collaboration Agreement, the Company received a non-refundable up-front payment of $
F-20
Concurrent with the execution of the Lilly Collaboration Agreement, the Company entered into a Technology License Agreement (the “Lilly License”) under which Lilly granted the Company a non-exclusive license to Lilly’s proprietary p-Tau 217 antibody technology for use in research use only products and services and future IVD applications within the field of Alzheimer’s disease. In consideration of the Lilly License, the Company paid an upfront fee, is required to make milestone payments based on the achievement of predetermined regulatory and commercial events, and will pay royalties on net sales of licensed products.
The Company recognized revenue from the Lilly Collaboration Agreement of $
Contract Assets
There were
Deferred Revenue
During the years ended December 31, 2023 and 2022, the Company recognized $
Remaining Performance Obligations
As of December 31, 2023, the aggregate amount of transaction prices allocated to performance obligations that were not yet satisfied, or were partially satisfied, was $
Costs to Obtain a Contract
Changes in costs to obtain a contract were as follows (in thousands):
| 2023 |
| 2022 |
| 2021 | ||||
Balance at December 31 of prior year |
| $ | | $ | | $ | | ||
Capitalization of costs to obtain a contract |
| |
| |
| | |||
Recognition of costs to obtain a contract |
| ( |
| ( |
| ( | |||
Balance at December 31 |
| $ | | $ | | $ | | ||
The Company evaluates potential impairment of these amounts at each balance sheet date, and
Grant Revenue
All of the Company's grant revenue is generated within North America. Grant revenue for the years ended December 31, 2023, 2022, and 2021 was $
NIH Grant
On September 21, 2022, the Company and the National Institutes of Health (the “NIH”), an agency of the U.S. Department of Health and Human Services, entered into a contract (the “NIH Grant”) with a total award value of $
F-21
During the year ended December 31, 2023, grant revenue recognized and research and development expenses incurred were $
ADDF Grant
On March 24, 2022, the Company and the Alzheimer’s Drug Discovery Foundation (the “ADDF”) entered into a contract (the “ADDF Grant”) with a total funding value of $
During the year ended December 31, 2023, grant revenue recognized and research and development expenses incurred was $
RADx Grant
On September 29, 2020, the Company entered into a contract with the NIH under its Rapid Acceleration of Diagnostics (“RADx”) program (the “RADx Grant”), with a total award value of $
During the years ended December 31, 2023 and 2022, the Company recognized
Note 4. Allowance for Credit Losses
The change in the allowance for credit losses on accounts receivable is summarized as follows (in thousands):
2023 | 2022 | 2021 | ||||||
Balance at December 31 of prior year | $ | | $ | | $ | | ||
Provision for expected credit losses | | | | |||||
Write-offs and recoveries collected | ( | ( | ( | |||||
Balance at December 31 | $ | | $ | | $ | | ||
F-22
Note 5. Marketable Securities
The amortized cost, gross unrealized gains, gross unrealized losses, and fair value of the Company’s marketable securities by major security type were as follows (in thousands):
As of December 31, 2023 | Amortized cost |
| Unrealized Gains |
| Unrealized Losses |
| Fair Value | |||||
Commercial paper | $ | | $ | | $ | ( | $ | | ||||
U.S. Treasuries | | | — | | ||||||||
U.S. Government agency bonds | | | ( | | ||||||||
Corporate bonds | | | ( | | ||||||||
Total marketable securities | $ | | $ | | $ | ( | $ | | ||||
Marketable securities are reported in the following Consolidated Balance Sheets captions: | ||||||||||||
Cash and cash equivalents | $ | | ||||||||||
Marketable securities | | |||||||||||
Total marketable securities | $ | | ||||||||||
The Company did
The following table shows the fair value and gross unrealized losses of the Company’s available-for-sale securities with unrealized losses that are not deemed to be other-than-temporary, aggregated by major security type and length of time that the individual securities have been in a continuous unrealized loss position (in thousands):
Less Than 12 Months | ||||||
As of December 31, 2023 | Fair Value |
| Unrealized Losses | |||
Commercial paper | $ | | $ | ( | ||
U.S. Government agency bonds | | ( | ||||
Corporate bonds | | ( | ||||
Total | $ | | $ | ( | ||
The Company did not have any individual securities in a continuous loss position for greater than 12 months, and there were no individual securities that were in a significant unrealized loss position as of December 31, 2023. For marketable securities in an unrealized loss position, the Company does not intend to sell them before recovery of their amortized cost bases, it is not more likely than not that the Company will be required to sell them before recovery of their amortized cost bases, and the unrealized losses are not credit related. Accordingly, the Company has not recorded any impairment losses or a credit loss allowance.
The Company did not sell any marketable securities or record any realized gains or losses for the year ended December 31, 2023. At December 31, 2023, the Company had $
The following table summarizes the contractual maturities of the Company’s marketable securities (in thousands):
As of December 31, 2023 | Amortized cost | Fair Value | ||||
Due within one year | $ | | $ | | ||
Due in one to two years | | | ||||
Total | $ | | $ | | ||
F-23
Note 6. Fair Value of Financial Instruments
Recurring Fair Value Measurements
The following tables present the Company’s fair value hierarchy for its financial assets that are measured at fair value on a recurring basis (in thousands):
As of December 31, 2023 | Total |
| Level 1 |
| Level 2 |
| Level 3 | ||||
Financial assets: |
|
|
|
|
|
| |||||
Cash equivalents: (1) | |||||||||||
Money market funds | $ | |
| $ | | $ | — |
| $ | — | |
Commercial paper | | — | | — | |||||||
U.S. Treasuries | | — | | — | |||||||
Total cash equivalents | | | | — | |||||||
Marketable securities: (2) | |||||||||||
Commercial paper | | — | | — | |||||||
U.S. Treasuries | — | — | — | — | |||||||
U.S. Government agency bonds | | — | | — | |||||||
Corporate bonds | | — | | — | |||||||
Total marketable securities | | — | | — | |||||||
Total financial assets | $ | | $ | | $ | | $ | — | |||
As of December 31, 2022 | Total |
| Level 1 |
| Level 2 |
| Level 3 | ||||
Financial assets: |
|
|
|
|
|
| |||||
Money market funds (1) | $ | |
| $ | | $ | — |
| $ | — | |
Total financial assets | $ | | $ | | $ | — | $ | — | |||
(1) Included in cash and cash equivalents on the Consolidated Balance Sheets.
(2) Marketable securities are initially valued at their purchase price and subsequently fair valued at the end of each reporting period utilizing third party pricing services or other observable data. The pricing services utilize industry standard valuation methods, including both income and market-based approaches and observable market inputs to determine the fair value. These observable market inputs include reportable trades, benchmark yields, credit spreads, broker/dealer quotes, bids, offers, current spot rates, and other industry and economic events.
Nonrecurring Fair Value Measurements
The Company has several non-marketable equity investments in entities that are evaluated under the VIE guidance (refer to Note 19 - Variable Interest Entities for further discussion). Pursuant to ASC 321 – Investments – Equity Securities, the Company uses the measurement alternative for equity investments without readily determinable fair values and recognizes its non-marketable equity investments at cost, less any impairment, and adjusted for any observable price changes in orderly transactions.
On June 26, 2023, the Company received ordinary shares in UltraDx (refer to Note 3 − Revenue and Related Matters) which were valued at $
During the year ended December 31, 2023, the Company recorded $
F-24
Other Fair Value Disclosures
During the years ended December 31, 2023 and 2022, the Company did not transfer financial assets between levels of the fair value hierarchy. Additionally, there have been no changes to the valuation techniques for Level 2 or Level 3 financial assets.
Note 7. Inventory
Inventory, net of inventory reserves, consisted of the following (in thousands):
As of December 31, | ||||||
| 2023 |
| 2022 | |||
Raw materials | $ | | $ | | ||
Work in process |
| |
| | ||
Finished goods |
| |
| | ||
Total inventory | $ | | $ | | ||
Note 8. Property and Equipment
Property and equipment consisted of the following (in thousands):
As of December 31, | ||||||
| 2023 |
| 2022 | |||
Laboratory and manufacturing equipment | $ | | $ | | ||
Office furniture and equipment |
| |
| | ||
Computers and software |
| |
| | ||
Leasehold improvements |
| |
| | ||
Total cost | $ | | $ | | ||
Less: accumulated depreciation |
| ( |
| ( | ||
Property and equipment, net | $ | | $ | | ||
The Company incurred depreciation expense of $
During the year ended December 31, 2023, the Company had
For the years ended December 31, 2023, and 2022, the Company recorded impairments related to leasehold improvements associated with two leased facilities not being used. Refer to Note 14 − Leases for further discussion. There were
As of December 31, 2023, the Company capitalized $
F-25
Note 9. Accrued Expenses and Other Current Liabilities
Accrued expenses and other current liabilities consisted of the following (in thousands):
As of December 31, | |||||
2023 |
| 2022 | |||
Accrued professional services | $ | | $ | | |
Accrued royalties | | | |||
Accrued tax liabilities | | | |||
Other accrued expenses |
| |
| | |
Total accrued expenses and other current liabilities | $ | | $ | | |
Note 10. Stock-Based Compensation
Stock-Based Compensation Plans
In December 2017, the Company adopted the 2017 Employee, Director and Consultant Equity Incentive Plan (the “2017 Plan”), under which it may grant incentive stock options, non-qualified stock options, RSUs, and other stock-based awards. As of December 31, 2017, the 2017 Plan allowed for the issuance of (1) up to
In December 2017, the Company adopted the 2017 Employee Stock Purchase Plan (the “2017 ESPP”). As of December 31, 2019, the 2017 ESPP allowed for the issuance of up to
The 2017 ESPP provides for six-month offering periods commencing and ending on March 1 through August 31, and September 1 through February 28. During the years ended December 31, 2023, 2022, and 2021, employees purchased
Stock Options
Under the 2017 Plan, stock options may not be granted with exercise prices of less than fair market value on the date of the grant. Options generally vest ratably over a
F-26
Stock option activity for the year ended December 31, 2023 is presented below (in thousands, except per share and contractual life amounts):
Weighted-average | Weighted-average remaining contractual | Aggregate | |||||||||
| Number of options |
| exercise price per share |
| life (in years) |
| intrinsic value | ||||
Outstanding at December 31, 2022 |
| |
| $ | | $ | | ||||
Granted | | | |||||||||
Exercised | ( | | |||||||||
Forfeited/expired | ( | | |||||||||
Outstanding at December 31, 2023 | | $ | | $ | | ||||||
Exercisable at December 31, 2023 | | $ | | $ | | ||||||
Vested and expected to vest at December 31, 2023 | | $ | | $ | | ||||||
The total intrinsic value of stock options exercised was $
Restricted Stock Units
RSUs represent the right to receive shares of common stock upon meeting specified vesting requirements. Shares are delivered to the grantee upon vesting, less shares for the payment of withholding taxes. RSU activity for the year ended December 31, 2023 is presented below (in thousands, except per share amounts):
Weighted-average grant | |||||
| Number of shares |
| date fair value per share | ||
Unvested as of December 31, 2022 |
| |
| $ | |
Granted | | | |||
Vested | ( | | |||
Forfeited | ( | | |||
Unvested as of December 31, 2023 | | $ | | ||
Expected to convert at December 31, 2023 | | $ | | ||
The weighted average grant-date fair value per share of awards granted was $
Stock-Based Compensation Expense
Stock-based compensation expense was recorded in the following categories on the Consolidated Statements of Operations (in thousands):
Year Ended December 31, | ||||||||
2023 |
| 2022 |
| 2021 | ||||
Cost of product revenue | $ | | $ | | $ | | ||
Cost of service and other revenue |
| |
| |
| | ||
Research and development |
| |
| |
| | ||
Selling, general and administrative |
| |
| |
| | ||
Total stock-based compensation | $ | | $ | | $ | | ||
As of December 31, 2023, there was $
F-27
The fair value of the Company’s stock options granted and purchase rights to the ESPP were estimated using the Black-Scholes valuation model with the following assumptions:
Year Ended December 31, | ||||||||
2023 | 2022 | 2021 | ||||||
Stock Options: | ||||||||
Risk-free interest rate | ||||||||
Expected dividend yield | ||||||||
Expected term (in years) | ||||||||
Expected volatility | ||||||||
Weighted-average grant date fair value per share | $ | | $ | | $ | | ||
Employee Stock Purchase Plan: | ||||||||
Risk-free interest rate | ||||||||
Expected dividend yield | ||||||||
Expected term (in years) | ||||||||
Expected volatility | ||||||||
Weighted-average grant date fair value per share | $ | | $ | | $ | | ||
Note 11. Net Loss Per Share
The following table presents the computation of basic and diluted net loss per share (in thousands, except per share data):
Year Ended December 31, | ||||||||
| 2023 |
| 2022 |
| 2021 | |||
Numerator: | ||||||||
Net loss | $ | ( | $ | ( | $ | ( | ||
Denominator: | ||||||||
Weighted average common shares outstanding, basic and diluted | | | | |||||
Net loss per share, basic and diluted | $ | ( | $ | ( | $ | ( | ||
As the Company was in a net loss position, the following common share equivalents (calculated on a weighted average basis) were excluded from the calculation of diluted net loss per share (in thousands):
Year Ended December 31, | ||||||||
| 2023 |
| 2022 |
| 2021 | |||
Stock options |
| |
| |
| | ||
Common stock and RSUs |
| |
| |
| | ||
Estimated ESPP purchases | | | | |||||
Total dilutive shares |
| |
| |
| | ||
Note 12. Income Taxes
The following table presents the components of loss before income taxes (in thousands):
Year Ended December 31, | ||||||||
2023 | 2022 |
| 2021 | |||||
United States | $ | ( | $ | ( | $ | ( | ||
Foreign |
| |
| ( |
| ( | ||
Total loss before income taxes | $ | ( | $ | ( | $ | ( | ||
F-28
The following table summarizes income tax (expense) benefit (in thousands):
Year Ended December 31, | ||||||||
2023 |
| 2022 |
| 2021 | ||||
Current: | ||||||||
United States | ||||||||
Federal | $ | — |
| $ | — |
| $ | — |
State | ( | ( | ( | |||||
Foreign | ( | ( | ( | |||||
Total current income tax provision | ( | ( | ( | |||||
Deferred | ||||||||
United States | ||||||||
Federal | — | | | |||||
State | — | | ( | |||||
Foreign | | | | |||||
Total deferred income tax benefit | | | | |||||
Total income tax (expense) benefit | $ | ( | $ | ( | $ | | ||
A reconciliation of the federal statutory income tax rate to the effective tax rate is as follows:
Year Ended December 31, | ||||||
2023 |
| 2022 |
| 2021 |
| |
Federal statutory income tax rate | | % | | % | | % |
Non-deductible executive compensation | ( | % | ( | % | ( | % |
State taxes, net of federal benefit | | % | | % | | % |
Tax credits | | % | | % | | % |
Stock-based compensation | ( | % | ( | % | | % |
Permanent items | ( | % | ( | % | ( | % |
Deferred tax rate change | — | % | — | % | | % |
Change in valuation allowance | ( | % | ( | % | ( | % |
Impairment of goodwill | — | % | ( | % | — | % |
Other | | % | — | % | ( | % |
Effective income tax rate | ( | % | ( | % | | % |
The effective income tax rate differs from the U.S. federal statutory rate of
F-29
Deferred tax assets and liabilities reflect the net tax effects of temporary differences between the carrying amount of assets and liabilities for financial reporting and the amounts used for income tax purposes. Significant components of the Company’s deferred tax assets and liabilities were as follows (in thousands):
As of December 31, | |||||
2023 |
| 2022 | |||
Deferred tax assets: |
|
|
| ||
Net operating loss carryforwards | $ | | $ | | |
Tax credits |
| |
| | |
Deferred revenue | | | |||
Amortization | | | |||
Stock-based compensation |
| |
| | |
Inventory | | | |||
Capitalized R&D Costs | | | |||
Lease liability | | | |||
Other deferred tax assets | | | |||
Total deferred tax assets |
| |
| | |
Less: valuation allowances | ( | ( | |||
Net deferred tax assets | | | |||
Deferred tax liabilities: | |||||
Right-of-Use Assets | ( | ( | |||
Depreciation | ( | ( | |||
Amortization acquired intangibles | ( | ( | |||
Goodwill | — | — | |||
Other deferred tax liabilities | — | ( | |||
Net deferred tax (liability) asset | $ | ( | $ | ( | |
The Company records deferred tax liabilities in other non-current liabilities on the Consolidated Balance Sheets.
The Company’s change in its valuation allowance account related to deferred tax assets was as follows (in thousands):
2023 |
| 2022 | |||
Balance at December 31 of prior year | $ | | $ | | |
Change in valuation allowance | | | |||
Balance at December 31 | $ | | $ | | |
The valuation allowance increased during the year ended December 31, 2023 primarily as a result of the U.S. operating losses incurred.
In determining the need for a valuation allowance, the Company considers the cumulative book income and loss positions of each of its entities as well as its worldwide cumulative loss position. The Company has assessed, on a jurisdictional basis, the available means of recovering deferred tax assets, including the ability to carryback net operating losses (“NOLs”), the existence of reversing taxable temporary differences, the availability of tax planning strategies, and forecasted future taxable income. At December 31, 2023, the Company maintained a full valuation allowance against its worldwide net deferred tax assets, as it concluded that it was more likely than not that the deferred assets will not be utilized.
As of December 31, 2023, the Company had U.S. federal net operating losses (“NOLs”) of approximately $
As of December 31, 2023, the Company had $
F-30
Under Sections 382 and 383 of the U.S. Internal Revenue Code, if a corporation undergoes an ownership change, the corporation’s ability to use its pre-ownership change NOLs and other pre-ownership change tax attributes, such as research tax credits, to offset its post-change income and taxes may be limited. In general, an ownership change occurs if there is a cumulative change in an entity’s ownership by 5% stockholders that exceeds 50 percentage points over a rolling three-year period. Similar rules may apply under U.S. state tax laws. Under the Tax Cuts and Jobs Act of 2017 (“TCJA”), the use of federal NOLs arising in taxable years beginning after December 31, 2017 is limited to 80% of current year taxable income and NOLs arising in taxable years ending after December 31, 2017 may not be carried back (though any such NOLs may be carried forward indefinitely).
The Company may have experienced an ownership change in the past and may experience ownership changes in the future as a result of future transactions in its share capital, some of which may be outside of the control of the Company. As a result, if the Company earns net taxable income, its ability to use its pre-ownership change NOLs, or other pre-ownership change tax attributes, to offset U.S. federal and state taxable income and taxes may be subject to significant limitations.
For the years ended December 31, 2023, 2022, and 2021, the Company had
The Company is subject to taxation in the United States as well as the Netherlands, Sweden, and China. At December 31, 2023, the Company is generally no longer subject to examination by taxing authorities in the United States for years prior to 2020. However, NOLs and tax credits in the United States may be subject to adjustments by taxing authorities in future years in which they are utilized. The Company’s foreign subsidiaries remain open to examination by taxing authorities from 2018 onward.
As of December 31, 2023, the Company’s foreign subsidiaries had immaterial undistributed earnings and the tax payable on the earnings that are indefinitely reinvested would be immaterial.
Note 13. Goodwill and Intangible Assets
During the third quarter of 2022, the Company identified certain indicators of impairment, including the significant decline in the Company’s stock price, actions taken under the Restructuring Plan (Refer to Note 17 – Restructuring), and a reduction of forecasted sales and profitability. As a result, the Company performed a goodwill impairment test and determined the carrying amount of the Company’s sole reporting unit exceeded its estimated fair value. The Company concluded that its entire goodwill balance was impaired and recognized an $
Acquired intangible assets consisted of the following (in thousands, except useful life and weighted average life amounts):
As of December 31, 2023 | |||||||||||||||
Estimated | Cumulative | ||||||||||||||
Useful | Gross Carrying | Accumulated | Translation | Net Carrying | Weighted Average | ||||||||||
Life (in years) |
| Value |
| Amortization |
| Adjustment |
| Value |
| Life Remaining (in years) | |||||
Know-how | $ | | $ | ( | $ | ( | $ | | |||||||
Developed technology | | ( | — | | |||||||||||
Customer relationships |
| |
| ( | ( |
| | ||||||||
Non-compete agreements | | ( | ( | | |||||||||||
Trade names |
| |
| ( | — |
| — | — | |||||||
Total | $ | | $ | ( | $ | ( | $ | | |||||||
F-31
As of December 31, 2022 | |||||||||||||||
Estimated | Cumulative | ||||||||||||||
Useful | Gross Carrying | Accumulated | Translation | Net Carrying | Weighted Average | ||||||||||
Life (in years) |
| Value |
| Amortization |
| Adjustment |
| Value |
| Life Remaining (in years) | |||||
Know-how | $ | | $ | ( | $ | ( | $ | | |||||||
Developed technology | | ( | — | | |||||||||||
Customer relationships |
| |
| ( | ( |
| | ||||||||
Non-compete agreements | | ( | ( | | |||||||||||
Trade names |
| |
| ( | — |
| — | — | |||||||
Total | $ | | $ | ( | $ | ( | $ | | |||||||
The Company recorded amortization expense of $
Future estimated amortization expense is as follows (amounts in thousands):
As of December 31, 2023 | ||
2024 | $ | |
2025 | | |
2026 | | |
2027 | | |
2028 | | |
Thereafter | — | |
Total amortization expense | $ | |
Note 14. Leases
As part of the Restructuring Plan in the third quarter of 2022 (refer to Note 17 − Restructuring), the Company decided not to use two leased facilities in its operations and intends to sublease the vacant space to recover a portion of the total lease costs. The Company’s decision to not use the leased facilities triggered an impairment assessment and the related ROU assets and leasehold improvements became their own asset group. The impairment analysis evaluated the present value of net cash flows under the original lease and the estimated cash flows under estimated subleases to identify any impairment amount. The impairment assessment considered all industry and economic factors such as rental rates, interest rates, and recent real estate activities to estimate the net cash flows analysis and impairment amount. During 2023, the Company continued to reassess the remaining ROU assets and leasehold improvements associated with the leased facilities that remained vacant.
The above assessments resulted in the Company recording an impairment charge of $
There were
F-32
The components of the lease costs and supplemental cash flow information relating to the Company's leases were as follows (in thousands):
Year Ended December 31, | |||||||||
2023 | 2022 | 2021 | |||||||
Operating lease cost | $ | | $ | | $ | | |||
Short-term and variable lease cost | | | | ||||||
Total lease cost | $ | | $ | | $ | | |||
Year Ended December 31, | ||||||
2023 | 2022 | |||||
Cash paid for amounts included in the measurement of operating lease liabilities | $ | | $ | | ||
Operating ROU assets obtained in exchange for lease obligations | $ | — | $ | | ||
Weighted average remaining lease term - operating leases (years) | ||||||
Weighted average discount rate - operating leases | ||||||
The undiscounted future lease payments for non-cancelable operating leases were as follows (in thousands):
Maturity of lease liabilities | As of December 31, 2023 | ||
2024 | $ | | |
2025 | | ||
2026 | | ||
2027 | | ||
2028 | | ||
Thereafter | | ||
Total lease payments | $ | | |
Less: imputed interest | ( | ||
Total operating lease liabilities | $ | | |
Operating lease balances presented on the Consolidated Balance Sheets were as follows (in thousands):
As of December 31, 2023 | |||
Operating lease ROU assets | $ | | |
Operating lease liabilities | $ | | |
Operating lease liabilities, net of current portion | | ||
Total operating lease liabilities | $ | | |
Note 15. Commitments and Contingencies
Purchase Commitments
STRATEC
During 2022, the Company and STRATEC Consumables GmbH (“STRATEC”) entered into an amendment to the supply agreement with STRATEC (as amended, the “STRATEC Supply Agreement”), related to the supply of discs used in Simoa bead-based instruments. As part of the STRATEC Supply Agreement, the Company agreed to purchase a total of
The total purchase commitment under the STRATEC Supply Agreement is $
In 2023 and 2022, STRATEC shipped
F-33
Other Purchase Commitments
The Company’s other non-cancellable purchase commitments primarily consist of purchases of raw materials for manufacturing operations under annual and multi-year agreements, some of which have minimum quantity requirements. The Company’s total purchase commitments under these agreements as of December 31, 2023 was $
License Agreements
Harvard University
In August 2022, the Company and Harvard University (“Harvard”) entered into an exclusive license agreement (the “Harvard License Agreement”) for certain intellectual property owned by Harvard. Pursuant to the Harvard License Agreement, the Company paid an upfront fee of $
Refer to Note 16 − Related Party Transactions for a discussion of a related party relationship with Harvard.
Tufts University
In June 2007, the Company and Tufts University (“Tufts”) entered into a license agreement (the “Tufts License Agreement”) for certain intellectual property owned by Tufts. The Tufts License Agreement, which was subsequently amended, is exclusive and sub-licensable, and will continue in effect on a country by country basis as long as there is a valid claim of a licensed patent in a country. The Company is contractually obligated to pay license and maintenance fees that are creditable against royalties, in addition to low single-digit royalties on direct sales and services, and a royalty on sublicense income. The Company incurred royalty expenses related to the Tufts License Agreement of $
Refer to Note 16 − Related Party Transactions for a discussion of a related party relationship with Tufts.
Legal Contingencies
The Company is subject to claims in the ordinary course of business; however, the Company is not currently a party to any pending or threatened litigation, the outcome of which would be expected to have a material adverse effect on its financial condition or results of operations. The Company accrues for contingent liabilities when losses are probable and estimable. If an estimate of a probable loss is a range and no amount within the range is more likely than any other amount in the range, the Company accrues the minimum amount of the range.
Note 16. Related Party Transactions
In June 2007, the Company entered into the Tufts License Agreement for certain intellectual property owned by Tufts (refer to Note 15 − Commitments and Contingencies). A member of the Company’s Board of Directors was previously affiliated with Tufts. This Board member continues to receive compensation from Tufts on a formulaic basis based on royalties and license payments the Company makes to Tufts. At December 31, 2023 and 2022, open payable balances to Tufts were not material.
In August 2022, the Company entered into the Harvard License Agreement for certain intellectual property owned by Harvard (refer to Note 15 − Commitments and Contingencies). Harvard is obligated to pay a portion of the payments received from the Company under the Harvard License Agreement to a member of the Company’s Board of Directors. A member of the Company’s Board of Directors is also affiliated with Harvard and Mass General Brigham. Revenue recorded from sales of products and services to Harvard and its affiliates and to Mass General Brigham and its affiliates totaled $
F-34
product revenue and operating expenses with Harvard and its affiliates and Mass General Brigham and its affiliates was $
As discussed in Note 3 − Revenue and Related Matters, on May 26, 2022, the Company and UltraDx, a company formed by ARCH, entered into the UltraDx Agreement to supply certain instruments and to grant certain licenses. At contract inception, the Company determined that UltraDx was a related party because a member of the Company’s Board of Directors was affiliated with ARCH and UltraDx. As of June 7, 2023, this individual was no longer a member of the Company’s Board of Directors. Cost of product revenue with UltraDx was $
Note 17. Restructuring
Following a strategic review and assessment of the Company’s operations and cost structure, on August 8, 2022, the Company announced a plan of restructuring and strategic re-alignment plan (the “Restructuring Plan”). As part of the Restructuring Plan, the Company began an assay redevelopment program with the ultimate objective of improving its ability to manufacture and deliver high-quality assays at scale. The Restructuring Plan aligned the Company’s investments to best serve the needs of its customers, focused the Company’s innovation efforts on key platforms, and provided a foundation for the Company’s entry into translational pharma and clinical markets. In accordance with the Restructuring Plan, the Company implemented a workforce reduction, which was substantially completed by the end of the third quarter of 2022. The Restructuring Plan included the elimination of
During the year ended December 31, 2022, the Company incurred approximately $
As a result of the Restructuring Plan, the Company performed an impairment assessment of its goodwill, long-lived assets, including ROU assets and related leasehold improvements, and intangibles. The assessments resulted in the Company recording an impairment charge of $
The following table presents the restructuring reserve and provision activity for the year ended December 31, 2023 (in thousands):
Severance and Employee Benefit Costs | ||
Balance at December 31, 2022 | $ | |
Accrual adjustments | ( | |
Cash payments | ( | |
Foreign currency translation | | |
Balance at December 31, 2023 | $ | |
The Company did not have any restructuring activities related to the Restructuring Plan during the year ended December 31, 2023.
F-35
Note 18. Employee Benefit Plans
The Company sponsors a 401(k) savings plan for employees and may make discretionary contributions. During the years ended December 31, 2023, 2022, and 2021, the Company made contributions of $
Note 19. Variable Interest Entities
The Company enters into relationships with, or has investments in, other entities that may be VIEs. The Company assesses the criteria in ASC 810 – Consolidation to determine if any of these entities meet the definition of a VIE and require consolidation into its financial statements. The Company’s analysis determines whether it has a controlling financial interest and also identifies the primary beneficiary of a VIE as the enterprise that has both (1) the power to direct activities of a VIE that most significantly impact the entity’s economic performance and (2) the obligation to absorb losses of, or the right to receive benefits from, the entity that could potentially be significant to that entity.
As discussed in Note 3 − Revenue and Related Matters, during the second quarter of 2023 the Company received million ordinary shares of UltraDx under the UltraDx Agreement. Primarily due to having less than a
Based on the Company’s assessments, it does not have any controlling financial interests in any VIEs, and therefore did not consolidate any VIEs into its Consolidated Financial Statements during the years ended December 31, 2023 and 2022. As of December 31, 2023 and 2022, the carrying value of the Company’s investments in VIEs was $
F-36


