UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 20-F
(Mark One)
| REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR (g) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
OR
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
(For the fiscal year ended December 31 , 2023
OR
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
OR
| SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
Commission file number: 001-40627
(Exact name of Registrant as specified in its charter)
(Jurisdiction of incorporation or organization)
+41 21 694 10 60
(Address of principal executive offices)
SOPHiA GENETICS, Inc.
(617 ) 982-1210
(Name, telephone, e-mail and/or facsimile number and address of company contact person)
Securities registered or to be registered, pursuant to Section 12(b) of the Act.
Title of each class | Trading symbol(s) | Name of each exchange on which registered | ||||||||||||
The | ||||||||||||||
Securities registered or to be registered pursuant to Section 12(g) of the Act: None
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act: None
Indicate the number of outstanding shares of each of the issuer’s classes of capital stock or common stock as of the close of business covered by the annual report. Ordinary shares, nominal value CHF 0.05 per share: 65,230,559
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. o Yes x No
If this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934. o Yes x No
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. x Yes o No
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). x Yes o No
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer | o | x | ||||||||||||
| Non-accelerated Filer | o | Emerging growth company | ||||||||||||
If an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards† provided pursuant to Section 13(a) of the Exchange Act. o
† The term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification after April 5, 2012.
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. o
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. o
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). o
Indicate by check mark which basis of accounting the registrant has used to prepare the financial statements included in this filing:
U.S. GAAP o | Other o | |||||||
If “Other” has been checked in response to the previous question indicate by check mark which financial statement item the registrant has elected to follow. Item 17 o Item 18 o
If this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). o Yes x No
TABLE OF CONTENTS
F-1 | ||||||||
ABOUT THIS ANNUAL REPORT
Unless otherwise indicated or the context otherwise requires, all references in this Annual Report to “SOPHiA GENETICS,” “SOPH,” the “Company,” “we,” “our,” “ours,” “us” or similar terms refer to SOPHiA GENETICS SA and its consolidated subsidiaries.
Trademarks
We own various trademark registrations and applications, and unregistered trademarks, including for “SOPHiA GENETICS,” “SOPHiA DDMTM,” “Alamut,” “SOPHiA Trial Match,” “SOPHiA Insights,” “SOPHiA CDx,” “SOPHiA Awareness” and our corporate logo. All other trade names, trademarks and service marks of other companies appearing in this Annual Report are the property of their respective owners. Solely for convenience, the trademarks and trade names in this Annual Report may be referred to without the ® and ™ symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. We do not intend to use or display other companies’ trademarks and trade names to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
Presentation of Financial Information
Our consolidated financial statements are presented in U.S. dollars and have been prepared in accordance with IFRS Accounting Standards as issued by the International Accounting Standards Board (“IASB”). None of the consolidated financial statements were prepared in accordance with generally accepted accounting principles in the United States (“U.S. GAAP”). The terms “dollar,” “USD” and “$” refer to U.S. dollars and the terms “Swiss franc” and “CHF” refer to the legal currency of Switzerland, unless otherwise indicated. We have made rounding adjustments to some of the figures included in this Annual Report. Accordingly, any numerical discrepancies in any table between totals and sums of the amounts listed are due to rounding.
Our fiscal year ends on December 31. References in this Annual Report to a fiscal year relate to our fiscal year ended on December 31 of that calendar year.
Market and Industry Data
This Annual Report contains industry, market and competitive position data that are based on general and industry publications, surveys and studies conducted by third parties, some of which may not be publicly available, and our own internal estimates and research. Third-party publications, surveys and studies generally state that they have obtained information from sources believed to be reliable, but do not guarantee the accuracy and completeness of such information. While we are not aware of any misstatements regarding the industry, market and competitive position data presented herein, these data involve a number of assumptions and limitations and contain projections and estimates of the future performance of the industries in which we operate that are subject to a high degree of uncertainty.
1
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report contains statements that constitute forward-looking statements. All statements other than statements of historical facts contained in this Annual Report, including statements regarding our future results of operations and financial position, business strategy, technology, collaborations and partnerships, as well as plans and objectives of management for future operations are forward-looking statements. Many of the forward-looking statements contained in this Annual Report can be identified by the use of forward-looking words such as “anticipate,” “believe,” “could,” “expect,” “should,” “plan,” “intend,” “estimate,” “will” and “potential,” among others.
Forward-looking statements appear in a number of places in this Annual Report and include, but are not limited to, statements regarding our intent, belief or current expectations. Forward-looking statements are based on our management’s beliefs and assumptions and on information currently available to our management. Such statements are subject to risks and uncertainties, and actual results may differ materially from those expressed or implied in the forward-looking statements due to various factors, including, but not limited to, those identified in the section titled “Item 3. Key Information—D. Risk Factors” in this Annual Report. These forward-looking statements include, among others:
•our expectations regarding our revenue, gross margin, expenses, other operating results, and cash usage;
•our plans regarding further development of our SOPHiA DDM Platform and its expansion into additional features, applications and data modalities;
•future investments in our business, our anticipated capital expenditures and our estimates regarding our capital requirements, future revenues, expenses, reimbursement rates and needs for additional financing;
•our expectations regarding the market size for our platform, applications, products, and services and the market acceptance they will be able to achieve;
•our expectations regarding changes in the healthcare systems in different jurisdictions, in particular with respect to the manner in which electronic health records are collected, distributed and accessed by various stakeholders;
•the timing or outcome of any domestic and international regulatory submissions;
•impact from future regulatory, judicial, and legislative changes or developments in the United States and foreign countries;
•our ability to acquire new customers and successfully engage and retain customers;
•the costs and success of our marketing efforts, and our ability to promote our brand;
•our ability to increase demand for our applications, products, and services, obtain favorable coverage and reimbursement determinations from third-party payors and expand geographically;
•our expectations of the reliability, accuracy and performance of our applications, products, and services, as well as expectations of the benefits to patients, medical personnel and providers of our applications, products and services;
•our expectations regarding our ability, and that of our manufacturers, to manufacture our products;
•our efforts to successfully develop and commercialize our applications, products, and services;
•our competitive position and the development of and projections relating to our competitors or our industry;
2
•our ability to identify and successfully enter into strategic collaborations in the future, and our assumptions regarding any potential revenue that we may generate thereunder;
•our ability to obtain, maintain, protect and enforce intellectual property protection for our technology, applications, products, and services, and the scope of such protection;
•our ability to operate our business without infringing, misappropriating or otherwise violating the intellectual property or proprietary rights of third parties;
•our ability to attract and retain qualified key management and technical personnel; and
•our expectations regarding the time during which we will be an emerging growth company under the Jumpstart our Business Startups Act of 2012 (“JOBS Act”) and a foreign private issuer.
These forward-looking statements speak only as of the date of this Annual Report and are subject to a number of risks, uncertainties and assumptions described in the sections in this Annual Report titled “Item 3. Key Information—D. Risk Factors” and “Item 5. Operating and Financial Review and Prospects” and elsewhere in this Annual Report. Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control, you should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. Moreover, we operate in an evolving environment. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this Annual Report, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements.
3
PART I
Item 3. Key Information
D. Risk Factors
Our business faces significant risks and uncertainties. You should carefully consider all of the information set forth in this Annual Report and in other documents we file with or furnish to the SEC, including the following risk factors, before deciding to invest in or to maintain an investment in our securities. If any of the events or developments described below were to occur, our business, results of operations, financial condition and prospects could suffer materially and the trading price of our ordinary shares could decline. The risks and uncertainties described below are not the only ones we face. Additional risks and uncertainties not presently known to us or that we currently believe to be immaterial may also adversely affect our business.
•We may not be successful in expanding features, applications and data modalities of our SOPHiA DDM Platform and related solutions, applications, products, and services.
•We may experience challenges with the acquisition, development, enhancement and deployment of technology necessary for our data analytics platform technologies.
•If we are unable to expand our sales and marketing capabilities, including through additional strategic relationships, in a cost-effective manner, we may not be able to grow our revenue.
•The coverage and reimbursement status of newly developed applications and products, such as data analytics platforms and related solutions, applications, products, and services, particularly in a new category of diagnostics and therapeutics, is uncertain. An inability to obtain or maintain adequate coverage and reimbursement could limit the commercial potential of our SOPHiA DDM Platform and related solutions, applications, products, and services.
•If we cannot maintain our current relationships and enter into new relationships with hospitals, reference and specialty laboratories and biopharmaceutical companies, our revenue prospects could be reduced.
•We are highly dependent on our senior management team and other key personnel, and our business could be harmed if we are unable to retain and attract such personnel.
•Our industry is subject to rapid change, which could make our SOPHiA DDM Platform and related solutions, applications, products, and services obsolete. If we are unable to continue to innovate and improve our SOPHiA DDM Platform and related solutions, applications, products, and services, we could fail to attract new customers and expand our market share and we could lose existing customers and market share.
•We face competition from many sources and we may be unable to compete successfully.
•Cybersecurity or data privacy breaches, other unauthorized or improper access, or (distributed) denial service lack of access (e.g., ransomware, persistent DoS/DDoS) could result in additional costs, loss of revenue, significant liabilities, harm to our brand and decreased use of our SOPHiA DDM Platform and related solutions, applications, products, or services.
•If we are not able to obtain, maintain, defend and enforce patent and other intellectual property protection or if the scope of such patent and other intellectual property protection is not sufficiently broad, our competitors could develop and commercialize applications, products, services and technology similar or identical to ours.
•We license patent rights from third-party owners. If such owners do not properly or successfully obtain, maintain, defend and enforce the patents underlying such licenses, or if they retain or license to others any competing rights, our competitive position and business prospects may be adversely affected. If we fail to comply with our obligations in the agreements under which we license intellectual property rights from third parties or otherwise experience disruptions to our
4
relationships with any of our licensors, we could lose license rights that are important to our business.
•We have incurred net losses since our inception and expect to continue to incur losses for the foreseeable future. We may never achieve or sustain profitability.
Risks Related to the Development of Our SOPHiA DDM Platform and Related Solutions, Applications, Products, and Services
We may not be successful in expanding features, applications and data modalities of our SOPHiA DDM Platform and related solutions, applications, products, and services.
Our SOPHiA DDM Platform offers a broad range of genomics applications across oncology, rare diseases, infectious diseases, cardiology, neurology, metabolism and other disease areas. A major part of our long-term strategy is bringing new high-impact content to our customers through updates to our platform, which may include expanding our platform with additional features, applications and data modalities and related solutions, applications, products, and services. However, enhancing our platform and developing new related solutions, applications, products, and services poses various risks and challenges, including the inability to develop features, applications, data modalities and services that achieve the desired results, analytical accuracy and utility; the need to alter our platform, solutions, applications, products, or services; the time and resources required for platform, solution, application, service, and product development; uncertainty regarding and potential limitation in the scope of new features, solutions, applications, data modalities, products, or services; and the potential need to obtain U.S. Food and Drug Administration (the “FDA”), European Medicines Agency (the “EMA”), United Kingdom (the “UK”) Medicines and Healthcare products Regulatory Agency (the “MHRA”) and other regulatory clearances, authorizations or approvals. Even if we successfully develop a feature, application, data modality, solution, service or product and receive any necessary regulatory clearance, authorization or approval, such feature, application, data modality, solution, service or product may never achieve significant market acceptance among various stakeholders and be commercially successful. The expenses or losses associated with unsuccessful platform innovation or expansion could adversely affect our business revenue growth and results of operations.
This risk may be heightened as we generally sell our platform, solutions, applications, products, and services in industries that are characterized by rapid technological changes, frequent new application and product introductions and changing industry standards. Operating in and servicing such industries require us to develop platform enhancements based on technological innovation on a timely basis. In addition, whereas we purposefully built our SOPHiA DDM Platform in a decentralized manner and strategically positioned it as a “universal operating-system” for multimodal data analytics in order to provide for a broad range of application, product, and service expansion opportunities, certain jurisdictions, such as the Netherlands and the United Kingdom, have implemented or is expected to implement centralized services architectures for electronic healthcare records (“EHR”) where all patient data passes through a single, often government-run, entity rather than being shared directly between the healthcare providers. The widespread implementation of such centralized systems may limit the adoption and development of our platform.
Strong platform, application, product, and service performance, security and reliability are necessary to maintain and grow our business.
We need to maintain and continuously improve the performance, security and reliability of our SOPHiA DDM Platform and related solutions, applications, products, and services. Our platform, solutions, applications, and other products may contain errors or defects, and while we have made efforts to test them and are not aware of any widespread material errors, defects or other performance-related issues, there can be no assurance that our platform, solutions, applications, products, and services do not or will not have performance problems. As we continue to launch more platform features, applications, data modalities, solutions, products and services, these risks may increase. Poor performance, security and reliability could adversely impact our customers and lead to customer dissatisfaction, adversely affect our reputation and revenues and increase our application, service, and product care, and distribution costs and working capital requirements.
In particular, our SOPHiA DDM Platform and related solutions, applications, products and services use artificial intelligence, machine learning, data analytics and similar tools that collect, aggregate and analyze data
5
(collectively, “Data Tools”). The use of such Data Tools poses risks such as incomplete or inaccurate data, human error, or lack of appropriate controls and processes, the technologies underlying how we store and leverage data and their use cases are subject to a variety of laws. Uncertainty around new and emerging Data Tools may require additional investment in the development of proprietary datasets, machine learning, large language models and systems to test for accuracy, bias and other variables and computing capabilities.
We may experience challenges with the acquisition, development, enhancement and deployment of technology necessary for our data analytics platform technologies.
Our business requires sophisticated computer systems and software in order to accurately and efficiently capture, service and process increasing volumes of health data, in particular a growing number of genomic profiles generated by our customers through various NGS test kits, sequencers and sample materials from different manufacturers. Some of the technologies are changing rapidly and we must continue to adapt to these changes in a timely and effective manner at an acceptable cost. There can be no assurance that we will be able to develop, acquire, enhance, deploy or integrate new technologies, including technologies needed to integrate new genomics test kits into our data analytics platform, that these new technologies will be effective and efficient, will meet our needs or achieve our expected goals or that we will be able to do so as quickly or cost-effectively as our competitors. Significant technological change could render our data analytics platform and technologies obsolete and incompatible with new or improved genomics test kits. In addition, we may face challenges in expanding into markets without suitable cloud infrastructure compatible with our SOPHiA DDM Platform. Our continued success will depend on our ability to adapt to changing technologies, manage and process ever-increasing amounts of data and information and improve the performance features of our data analytics platform technologies in response to an ever-changing patient population. We may experience difficulties that could delay or prevent the successful design, development, testing and introduction of new versions of our data analytics platform technologies, limiting our ability to identify new solutions, applications, products, and services. Any of these challenges could have a material adverse effect on our operating results and financial condition.
Any failure to offer high-quality support for our applications, products, and services may adversely affect our relationships with customers and collaborators and negatively impact our reputation and our business, financial condition and results of operations.
In implementing and using our SOPHiA DDM Platform and related solutions, applications, products, and services, our customers and collaborators depend on our support to resolve issues in a timely manner. We may be unable to respond quickly enough to accommodate short-term increases in demand for customer support. Increased customer demand for support could increase costs and adversely affect our financial condition and results of operations. In addition, we need highly trained technical support personnel. Hiring technical support personnel is very competitive in our industry due to the limited number of people available with the necessary scientific and technical backgrounds and ability to understand our technology at a technical level. Our sales are highly dependent on our reputation and on positive recommendations from our customers, users, care collaborators, providers, laboratories, hospitals and clinics. If we do not maintain high-quality customer support, or if the market perceives that we do not maintain high-quality customer support, our reputation and our business, financial condition and results of operations could be adversely affected.
Delays in the commencement and successful completion of multimodal clinical studies, and negative or ambiguous data generated from such studies, could increase costs and delay or prevent regulatory approval of our SOPHiA DDM Platform and related solutions, applications, and products.
To further improve our SOPHiA DDM Platform and its capabilities, for example by developing new predictive algorithmic models that we can deploy on our platform, we are sponsoring and intend to continue to sponsor observational multimodal clinical studies in various disease areas. These clinical studies are subject to numerous risks. Events that may prevent successful or timely commencement and completion of multimodal clinical studies include:
•delays in receiving the required regulatory clearance from the appropriate regulatory authorities to commence the studies, delays in reaching an agreement on acceptable terms with prospective clinical research organizations (“CROs”) and participating sites and difficulties in obtaining required Institutional Review Board (“IRB”) or ethics committee approval at each participating site;
6
•challenges in recruiting and enrolling suitable patients that meet the study criteria to participate in the studies, which challenges may be heightened for any clinical study that seeks to enroll patients with characteristics that are found in a small population;
•our CROs or participating sites failing to comply with regulatory requirements or meet their contractual obligations to us, which risk may be heightened by our reliance on third parties;
•lower than anticipated patient retention rates and difficulties in maintaining contact with patients, resulting in incomplete data;
•changes in regulatory requirements and guidance;
•lack of adequate funding to continue the study; or
•delays and disruptions as a result of unforeseen external events.
Furthermore, there can be no assurance that any multimodal clinical study will produce the data necessary to support further development of our platform in a particular disease area or to support any potential regulatory submission. Even if a study is completed, the data generated may be negative, ambiguous or otherwise insufficient. To obtain sufficient data, we may be required to conduct additional studies beyond those that we current plan for, which would increase our costs and delay potential regulatory submissions and commercialization activities.
If we do not have the support of key opinion leaders or clinical data using our applications and products is not published in peer-reviewed journals, it may be difficult to drive adoption of our applications and products.
We have established relationships with leading thought leaders. If these key opinion leaders determine that our SOPHiA DDM Platform and related solutions, applications, products, and services are not accurate or that alternative technologies, applications, products and services are more accurate or more cost-effective, or if we fail to establish new relationships with key opinion leaders in different markets, geographies and among various stakeholders, we may see lower demand for our SOPHiA DDM Platform and related solutions, applications, products, and services, which would limit our revenue growth and our ability to achieve profitability.
The publication of clinical data using our applications and products in peer-reviewed journals is also crucial to our success. We are unable to control when, if ever, results of current or future trials and projects are published, which may delay or limit adoption of our SOPHiA DDM Platform and related solutions, applications, products, and services. Such peer-reviewed publications may be limited by many factors, including delays in the completion of, poor design of, or lack of compelling data from, clinical studies, as well as delays in the review, acceptance and publication process. If our SOPHiA DDM Platform and related solutions, applications, products, and services do not receive sufficient favorable exposure in peer-reviewed publications, the rate of adoption of our SOPHiA DDM Platform and related solutions, applications, products, and services among medical personnel and positive reimbursement coverage determinations for them could be adversely affected.
Risks Related to Commercialization
If we are unable to expand our sales and marketing capabilities, including through additional strategic relationships, in a cost-effective manner, we may not be able to grow our revenue.
Our future sales will depend in large part on our ability to develop, train, retain and expand our sales force, to increase the scope of our marketing efforts, including into markets and geographies where our presence is currently limited, and to maintain our current strategic relationships and enter into new strategic relationships.
We are continuously expanding our sales and marketing infrastructure. Identifying, recruiting and training qualified sales and marketing personnel requires significant time, expense and attention. Competition for such personnel is intense. It often takes several months or more before a sales representative is fully trained and productive, depending on the target market or geography. Our sales force may subject us to higher fixed costs than those incurred by our competitors that utilize independent third parties, which could place us at a competitive disadvantage.
7
In addition, we currently have multiple strategic relationships with third-party providers of solutions, applications, products, and services that can be bundled with our SOPHiA DDM Platform and offer our SOPHiA DDM Platform and related solutions, applications, products, and services through various global and local distributors. There is no guarantee that we will be successful in maintaining our existing arrangements or entering into new arrangements on favorable terms. Most of our distribution relationships are non-exclusive and permit such distributors to distribute competing applications and products. As such, our distributors may not commit the necessary resources to market our applications and products to the level of our expectations or may choose to favor marketing the applications and products of our competitors. If current or future distributors do not perform adequately or we are unable to enter into effective arrangements with distributors in particular geographies, we may not achieve revenue growth and realize our expansion strategy.
Sales and marketing activities in the healthcare space are subject to various rules and regulations. In addition, our marketing messaging can be complex and nuanced, and there may be errors or misunderstandings in our sales force’s communication of such messaging. As we continue to grow our sales and marketing efforts, we face an increased need to continuously monitor and improve our policies, processes and procedures to maintain compliance with a growing number and variety of laws and regulations. To the extent that there is any violation, whether actual, perceived or alleged, of our policies or applicable laws and regulations, we could incur additional training and compliance costs, receive inquiries from third parties or be held liable or otherwise responsible for such acts of noncompliance. Any of the foregoing could adversely affect our business, reputation and results of operations.
The commercial success of our SOPHiA DDM Platform and current and future applications, products, and services depend on attaining significant market acceptance.
Our commercial success depends, in part, on market acceptance of our SOPHiA DDM Platform and our applications, products, and services. We cannot predict how quickly, if at all, our SOPHiA DDM Platform and related solutions, applications, products, and services will attain significant market acceptance or, if accepted, how frequently they will be used. These constituents must believe that our SOPHiA DDM Platform and related solutions, applications, products, and services offer benefits over other available alternatives. The degree of market acceptance of our SOPHiA DDM Platform and related solutions, applications, products, and services depends on a number of factors, including:
•whether there is adequate utilization of our SOPHiA DDM Platform and related solutions, applications, products, and services based on their potential and perceived advantages over those of our competitors;
•the safety, accuracy and ease of use of our SOPHiA DDM Platform and related solutions, applications, products, and services relative to those currently on the market;
•our ability to develop, commercialize and obtain regulatory clearance or approval for in vitro diagnostic (“IVD”) applications and products for diagnostic use and our compliance with the FDA’s “Distribution of In Vitro Diagnostic Products Labeled for Research Use Only or Investigational Use Only” (the “RUO Guidance”) and other laws and regulations governing Research Use Only (“RUO”) and IVD applications and products in the United States, the European Union (the “EU”) and other geographies;
•the clinical flexibility, operational versatility and technology agnostic nature of our SOPHiA DDM Platform and related solutions, applications, products, and services;
•the prices at which we and our distributors offer our SOPHiA DDM Platform and related solutions, applications, products and services;
•the effectiveness of our sales and marketing efforts;
•our ability to provide incremental data that show the clinical benefits and cost-effectiveness, and operational benefits, of our SOPHiA DDM Platform and related solutions, applications, products and services;
8
•our ability to build and maintain robust data sets with respect to patient populations, both in geographic regions that we have historically served and in geographic regions that we may seek to enter or further penetrate in the future;
•the coverage and reimbursement acceptance of our applications, products, and services;
•pricing pressure, including from group purchasing organizations (“GPOs”), seeking to obtain discounts on our SOPHiA DDM Platform and related solutions, applications, products, and services based on the collective bargaining power of the GPO members;
•negative publicity regarding our or our competitors’ platforms, applications, products, and services; and
•the accuracy of our SOPHiA DDM Platform and related solutions, applications, products, and services relative to those of our competitors.
Additionally, even if our SOPHiA DDM Platform and related solutions, applications, products, and services achieve widespread market acceptance, they may not maintain that market acceptance over time if more cost-effective or more favorably received platforms, applications, products, services or technologies are introduced. Failure to achieve or maintain market acceptance and/or market share would limit our ability to generate revenue.
There can be no assurance that we will be able to further penetrate our existing markets, that our existing markets will be able to sustain our current and future solutions, applications, products, and services offerings and that we will be able to expand into new markets. Any failure to increase penetration in our existing markets or expand into new ones would adversely affect our revenues and results of operations.
The market opportunities for our SOPHiA DDM Platform and related solutions, applications, products, and services may be smaller than we estimate.
Our estimates of the addressable market for our SOPHiA DDM Platform and related solutions, applications, products, and services are derived from a variety of sources, including scientific literature, surveys of clinicians, medical personnel and healthcare professionals and other forms of market research. These estimates may be inaccurate or based on imprecise data. Further, these estimates are based on various assumptions, including the outcomes of clinical studies, and whether the clinical studies will achieve objectives needed to meet clinical and payor expectations, the number of people who have a particular disease or condition, our expansion into other features, applications and data modality opportunities and disease areas, maintenance and expansion of our clinical and multimodal data sets for patient populations in specific geographic regions, the prices at which we and our distributors provide or sell our SOPHiA DDM Platform and related solutions, applications, products, and services in the market, the regulatory framework governing the development, sale and use of our SOPHiA DDM Platform and related solutions, applications, products, and services, including the laws and regulations governing RUO and IVD applications and products, the degree of coverage and reimbursement, the cost-containment efforts by payors, customers and collaborators as well as obtaining necessary clearance or regulatory approvals. While we believe our assumptions and estimates are reasonable, these assumptions and estimates may prove to be incorrect and the conditions supporting our assumptions or estimates may change at any time. If the addressable market for our SOPHiA DDM Platform and related solutions, applications, products, and services is smaller than our estimates, or if the prices at which we can sell our SOPHiA DDM Platform and related solutions, applications, products, and services are lower than our estimates, our business, financial condition, and results of operations could be negatively impacted.
The coverage and reimbursement status of newly developed applications and products, such as data analytics platforms and related solutions, applications, products, and services, particularly in a new category of diagnostics and therapeutics, is uncertain. An inability to obtain or maintain adequate coverage and reimbursement could limit the commercial potential of our SOPHiA DDM Platform and related solutions, applications, products, and services.
The availability and extent of reimbursement by governmental and private payors is essential for most patients to be able to afford our current and future platforms, solutions, applications, products, and services, if approved for IVD use. In addition, because our SOPHiA DDM Platform and related solutions, applications, products, and services represent new approaches to the research, diagnosis, detection and treatment of diseases, we cannot
9
accurately estimate how they would be priced, whether reimbursement could be obtained or any potential revenue generated. Sales of our SOPHiA DDM Platform and related solutions, applications, products, and services, if approved for IVD use, may depend substantially on the extent to which they are covered by health maintenance, managed care and similar healthcare management organizations, or reimbursed by government health administration authorities, private health coverage insurers and other third-party payors. If reimbursement is not available, or is available only to limited levels, we may not be able to successfully commercialize our SOPHiA DDM Platform and related solutions, applications, products, and services. Even if coverage is provided, the available reimbursement amount may not be high enough to allow us to establish or maintain pricing sufficient to realize an adequate return on our research and development and sales and marketing costs.
Coverage and reimbursement are ever changing, and we are not in control of how our competitors’ coverage and pricing strategies are established. Some of our competitors have widespread brand recognition and substantially greater financial and technical resources and development, production and marketing capabilities than we do. Others may develop lower-priced, less complex tests that payors and healthcare professionals could view as functionally equivalent to our applications and products, which could force us to lower the list price of our tests and impact our operating margins and our ability to achieve and maintain profitability. Payors may compare our applications and products to our competitors and utilize them as precedents, which may impact our coverage and reimbursement. In addition, technological innovations that result in the creation of enhanced diagnostic tools that are more effective than ours may enable other clinical laboratories, hospitals, medical personnel or medical providers to provide specialized diagnostic tests similar to ours in a more patient-friendly, efficient or cost-effective manner than is currently possible.
Cost-containment efforts of our customers and third-party payors could have a material adverse effect on our sales and profitability.
Increasing efforts by governmental and third-party payors to cap or reduce healthcare costs may cause such organizations to limit both coverage and level of reimbursement for newly cleared, authorized or approved devices and medicines and, as a result, they may not cover or provide adequate payment for our platform and related solutions, applications, products and services. Such efforts include legislation and regulations designed to control pharmaceutical and biological pricing and hospitals’ participation in group purchasing organizations (“GPOs”) and Integrated Delivery Networks (the “IDNs”) that negotiate pricing arrangements with medical device companies and distributors.
We expect that a significant portion of our revenue will be derived from sales to customers for research and development applications, including for CROs. The demand for our SOPHiA DDM Platform and related solutions, applications, products, and services will depend in part upon the research and development budgets of these customers, which are impacted by factors beyond our control. In addition, academic, governmental and other research institutions that fund research and development activities may be subject to stringent budgetary constraints that could results in spending reductions, reduced allocations or budget cutbacks, which could jeopardize the ability of these customers to purchase our applications and products.
Risks Related to Our Business Strategy
Our results of operations will be materially harmed if we are unable to accurately forecast customer demand for, and utilization of, our SOPHiA DDM Platform and related solutions, applications, products and services and manage our inventory.
To ensure adequate inventory supply, we must forecast inventory needs and assemble products related to our SOPHiA DDM Platform and services based on our estimates of future demand. Our ability to accurately forecast demand could be negatively affected by various factors, including our failure to accurately manage our expansion strategy, application and product introductions by competitors, change in customer demand, changes in customer acceptance, changes in general market conditions or regulatory matters and weakening of economic conditions or consumer confidence in future economic conditions. Inventory levels in excess of customer demand may result in inventory write-downs or write-offs, which would adversely affect our gross margin and impair the strength of our brand. Conversely, if we underestimate customer demand for our SOPHiA DDM Platform and related solutions, applications, products, and services, our supply chain, manufacturing collaborators and/or internal manufacturing team may not be able to deliver components to meet our requirements, which could damage our reputation, sales growth, and customer relationships. In addition, if we experience a significant increase in demand, additional supplies of raw materials or additional manufacturing
10
capacity may not be available when required on terms that are acceptable to us, if at all, or suppliers may not be able to allocate sufficient capacity in order to meet our increased requirements, which could adversely affect our business, reputation, and results of operations.
We have in the past and may in the future acquire other businesses, which could require significant management attention, disrupt our business, dilute shareholder value and adversely affect our results of operations.
As part of our business strategy, we have in the past and may in the future acquire complementary companies, platforms, applications, products, or technologies that we believe fit within our business model and can address the needs of our current and potential customers. There can be no assurance that we can acquire or successfully integrate such companies, platforms, applications, products, or technologies into our business, in particular that we can successfully integrate any acquired technology into our SOPHiA DDM Platform. We may not be able to find suitable acquisition candidates, and we may not be able to complete such acquisitions on favorable terms, if at all. In addition, the pursuit of potential acquisitions may divert the attention of management and cause us to incur additional expenses in identifying, investigating and pursuing suitable acquisitions, whether or not they are consummated. If we do complete acquisitions, we may not ultimately strengthen our competitive position or achieve our strategic goals and any acquisitions we complete could be viewed negatively by our customers, investors and industry analysts. We may not be aware of all of the risks associated with the acquired business. In addition, an acquisition may result in unforeseen operating difficulties and expenditures, such as:
•difficulties integrating businesses, services, personnel, operations and financial and other controls and systems and retaining key employees;
•assumption of unknown liabilities, known contingent liabilities, that become realized or known liabilities that prove greater than anticipated;
•difficulties retaining the customers or employees of any acquired business;
•incurrence of debt, contingent liabilities or future write-offs of intangible assets or goodwill;
•entry into a new market or business line in which we have no prior experience and in which we may not successfully compete;
•integration of an acquired company, which may disrupt ongoing operations and require management resources that would otherwise be used in developing our existing business; and
•divergent interests from those of our collaborators.
Foreign acquisitions involve unique risks in addition to those mentioned above, including those related to integration of operations across different cultures and languages, currency risks, and the particular economic, political and regulatory risks associated with specific countries.
Any such acquisitions may reduce cash available for operations and other uses and could result in amortization expense related to identifiable assets acquired. We may have to pay cash, incur debt or issue equity securities to pay for any such acquisition, each of which could adversely affect our financial condition and the value of our ordinary shares. The sale or issuance of equity securities to finance any such acquisitions would result in dilution to our shareholders. The incurrence of indebtedness to finance any such acquisition would result in fixed obligations and could also include restrictive covenants that impede our ability to manage our operations. In addition, our results of operations may be adversely affected by the dilutive effect of an acquisition, performance earn-outs or contingent bonuses associated with an acquisition.
11
Risks Related to Our Relationships with Third Parties
If we cannot maintain our current relationships and enter into new relationships with hospitals, reference and specialty laboratories and biopharmaceutical companies, our revenue prospects could be reduced.
We collaborate with various constituents in the healthcare ecosystem, such as hospitals, reference and specialty laboratories, and biopharmaceutical companies to analyze patient samples for multiple applications; strategic partners with whom we combine our SOPHiA DDM Platform with their offerings and with whom we jointly develop application, product, and service offerings; and manufacturers, suppliers and distributors of our products and offerings. See “Item 10. Additional Information—Material Contracts.” There can be no assurance that these collaborations will be successful or provide benefits to us as we expect. The revenue attributable to such relationships may fluctuate from period to period, which could adversely affect our financial condition and results of operations. In addition, the termination of these relationships could result in a temporary or permanent loss of revenue and could negatively impact our reputation.
Our future success depends in part on our ability to maintain these relationships and to establish new relationships, including with additional partners and collaborators and with governmental and third-party payors and patients. Many factors have the potential to impact such relationships, including our customers’ and collaborators’ satisfaction with our SOPHiA DDM Platform and related solutions, applications, products, and services, their perceptions of our SOPHiA DDM Platform, which may be impacted by decisions of our other collaborators, and our ability to respond to the evolving needs of our customers. Furthermore, our customers may decide to decrease or discontinue their use of our SOPHiA DDM Platform and related solutions, applications, products, and services due to changes in clinical routine, research and development plans, financial constraints or utilization of internal testing resources or tests. In addition, our collaborators may decide to discontinue providing services or manufacturing products, for instance testing kits, complementary to or compatible with our SOPHiA DDM Platform and related solutions, applications, products, and services, in particular, applications and products offered as part of “bundle” solutions together with our SOPHiA DDM Platform. Furthermore, our collaborators with whom we entered into both manufacturing and distribution agreements may be dis-incentivized from adequately performing their obligations under the applicable distribution agreement if we substantially decrease the quantities of products purchased from them under the manufacturing agreement or terminate the manufacturing agreement. In addition to reducing our revenue, the loss of one or more of these relationships may reduce our exposure to clinical routine and research that facilitate the collection and incorporation of new data, including new genomic profiles, into our SOPHiA DDM Platform.
We engage in conversations with potential collaborators regarding commercial opportunities on an ongoing basis. There can be no assurance that any of these conversations will result in a commercial agreement, or if an agreement is reached, that the resulting relationship will be successful or that clinical or research studies conducted as part of the engagement will produce successful outcomes.
Our operating results depend on the performance of third-party distributors.
A portion of our sales is made through independent global and regional distributors that are not under our control. We rely on distributors to grow and develop our customer base and anticipate customer needs, and any lack of such actions by our distributors may adversely affect our results of operations. If the business relationship with such distributor is terminated, whether through industry consolidation or otherwise, and we are unable to find a suitable replacement, our operations and operating results could be materially adversely affected. These independent distributors also generally represent applications and products offered by several companies and are not subject to any minimum sales requirements or obligation to market our applications and products to their customers. In turn, distributors could reduce their sales efforts for our applications and products or choose to terminate their representation of us. They may also fail to perform their obligations under the agreements with us, including their obligations to ensure that end users of our SOPHiA DDM Platform are aware that informed consent is required from patients prior to obtaining access to our SOPHiA DDM Platform.
12
We rely on third-party service providers to host and deliver our SOPHiA DDM Platform and related services, and any interruptions or delays in these services could harm our business.
We currently serve our customers from third-party data center hosting facilities located in the United States, Canada, Brazil, Europe, Turkey and Australia. Our operations depend, in part, on our third-party facility providers’ ability to protect these facilities against damage or interruption from natural disasters, power or telecommunications failures, criminal acts, and similar events. In the event that our data center arrangements are terminated, or if there are any lapses of service or damage to a center, we could experience lengthy interruptions in providing our SOPHiA DDM Platform and related solutions, applications, products, and services as well as delays and additional expenses in making new arrangements.
In addition, we currently use Microsoft Corporation (“Microsoft”) and Microsoft Azure Services for a substantial portion of our computing, storage, data processing, networking and other services. Our platform can also be deployed onto other platforms, including Amazon Web Services (“AWS”) or Google Cloud Platform (“Google Cloud”). Any significant disruption of, or interference with, our use of Microsoft Azure Services, AWS, Google Cloud or other similar cloud platforms, could affect the use of, and our customers’ satisfaction with, our applications, products, and services and could harm our business, results of operations and reputation.
Cloud providers have broad discretion to change and interpret the terms of service and other policies with respect to us, and those actions may be unfavorable to our business operations. Cloud providers may also take actions beyond our control that could seriously harm our business, including discontinuing or limiting our access to one or more services, increasing pricing terms, terminating or seeking to terminate our contractual relationship altogether or altering how we are able to process data in a way that is unfavorable or costly to us. If our arrangements with cloud providers were terminated, we could experience interruptions on our platform and in our ability to make our content available to users, as well as delays and additional expenses in arranging for alternative cloud infrastructure services. Any transition to new cloud providers would be difficult to implement and would cause us to incur significant delays and expense.
We rely on third-party manufacturers for the supply, manufacture and production of our products. Our reliance on these third parties may impair the advancement and commercialization of our products.
We rely, and expect that we will continue to rely, on third parties for the manufacturing and supply of our products offered with our SOPHiA DDM Platform, and such reliance on third-party manufacturers may expose us to different risks than if we were to manufacture products ourselves. If our agreements with these third parties expire or are terminated, there can be no assurance that we would be able to negotiate new agreements with them or other third parties on equally favorable terms as the current agreements, or at all.
Reliance on third-party providers exposes us to different risks than if we were to manufacture and supply products ourselves. For example, third-party manufacturers may fail to deliver the required commercial quantities of materials on a timely basis and at commercially reasonable prices, and we may be unable to find one or more replacement manufacturers capable of production at a substantially equivalent cost in substantially equivalent volumes and quality on a timely basis. Further, as we do not have control over a manufacturer’s compliance with applicable manufacturing standards and other laws and regulations, we may receive required materials for, or components of, our products that are defective, contaminated or otherwise not conforming to applicable specifications, warranties and statutory and regulatory requirements. Any failure to achieve and maintain compliance with laws, regulations and standards could lead to suspension of the manufacturing and sales of our products, revocation of obtained regulatory clearance, product recalls, marketing or promotional restrictions, litigation, customer loss and reputational harm. Further, our manufacturing collaborators may be unable to successfully increase the manufacturing capacity for our products in a timely or cost-effective manner, or at all, as needed for our development efforts or, if our additional products are developed and approved, our commercialization efforts.
Establishing additional or replacement manufacturers could take a substantial amount of time and be expensive, which may result in interruptions in our operations and product delivery, negatively affect the quality and performance of our products or require that modifications be made to our products’ designs. Even if we are able to find replacement manufacturers, we will be required to verify that the new manufacturer maintains facilities, procedures and operations that comply with our quality expectations and applicable regulatory requirements. If we are unable to find an adequate replacement or another acceptable solution in time, our research and development and commercial activities could be harmed.
13
We rely on third parties to conduct multimodal clinical studies. If they do not properly and successfully perform their obligations to us, we may not be able to gather data necessary to support further development of our SOPHiA DDM Platform in a particular disease area or to support potential regulatory submissions and commercial activities.
We rely, and we expect that we will continue to rely, on third parties to assist in managing, monitoring and otherwise carrying out multimodal clinical studies of the performance of our SOPHiA DDM Platform and related solutions, applications, and products. As a result of our reliance on these third parties, we have less direct control over the conduct, timing and completion of these studies than we would otherwise have if we relied entirely upon our own staff. If these third parties do not successfully carry out their duties under their agreements, or if the quality or accuracy of the data they obtain is compromised, or if they fail to comply with study protocols or meet expected deadlines, the multimodal clinical studies of our SOPHiA DDM Platform and related solutions, applications, and products, may fail to generate data necessary to support further development of our platform in a particular disease area or to support potential regulatory submissions and commercial activities and could subject us to liability claims. If third parties fail to comply with applicable regulatory requirements, the data generated in the multimodal clinical studies may be unreliable and these studies may be extended, delayed, suspended or terminated and we could be subject to liability claims.
We compete with many other companies for the resources of these third parties. These third parties may have contractual relationships with other entities, some of which may be our competitors, which may draw time and resources away from our studies. If any of our relationships with these third parties terminate, we may not be able to enter into alternative arrangements or to do so on commercially reasonable terms. As a result, delays may occur in our studies, which can materially impact our ability to meet our desired development, regulatory and commercialization timelines. There can be no assurance that we will not encounter challenges or delays in the future or that these delays or challenges will not have a material adverse impact on our business, results of operations, financial condition and prospects.
Performance issues, service interruptions or price increases by our shipping carriers and warehousing providers could adversely affect our business, reputation and ability to provide our products on a timely basis.
Expedited, reliable shipping and delivery services and secure warehousing are essential to our operations. We rely on providers of transport services for reliable and secure point-to-point transport of our research and diagnostic products and for tracking of these shipments, and from time to time require warehousing for our products. Should a carrier encounter delivery performance issues such as loss, damage or destruction of any systems, it would be costly to replace such systems in a timely manner and such occurrences may damage our reputation, reduce demand for our SOPHiA DDM Platform and related solutions, applications, products, and services and increase costs and expenses to our business. In addition, any significant increase in shipping or warehousing rates could adversely affect our operating margins and results of operations. Similarly, strikes, severe weather, natural disasters, civil unrest and disturbances or other service interruptions affecting delivery or warehousing services we use would adversely affect our ability to process orders for our products on a timely basis and adversely affect specimen integrity and our ability to process samples in a timely manner.
Risks Related to Our Business and Industry
We are highly dependent on our senior management team and other key personnel, and our business could be harmed if we are unable to retain and attract such personnel.
We are highly dependent on our senior management, including our Chief Executive Officer Dr. Jurgi Camblong. Our success will depend on our ability to retain senior management and to attract and retain qualified personnel in the future, including sales and marketing professionals, scientists, clinical specialists and other highly skilled personnel. The inability to attract and retain members of our senior management, sales and marketing professionals, scientists, IT and data experts or clinical and regulatory specialists could result in delays in application and product development and commercialization and harm our business.
To induce valuable employees to remain at our company, in addition to salary and cash incentives, we have granted and intend to continue to grant share-based compensation awards that vest over time. The value to employees of such awards is significantly affected by movements in our share price due to market volatility, and such awards may at any time be insufficient to retain key employees and senior management, as well as to
14
counteract more lucrative offers from other companies. Despite our efforts to retain valuable employees, members of our management, scientific and development teams may terminate their employment with us on short notice. We do not maintain “key person” insurance policies.
Our industry is subject to rapid change, which could make our SOPHiA DDM Platform and related solutions, applications, products, and services obsolete. If we are unable to continue to innovate and improve our SOPHiA DDM Platform and related solutions, applications, products, and services, we could fail to attract new customers and expand our market share and we could lose existing customers and market share.
Our industry is characterized by rapid changes, including technological and scientific breakthroughs, frequent new application, product, or service introductions and enhancements and evolving industry standards, all of which could make our SOPHiA DDM Platform and related solutions, applications, products and services and others we are developing obsolete. Our future success will depend on our ability to keep pace with the evolving needs of our customers on a timely and cost-effective basis and to pursue new market opportunities that develop as a result of scientific and technological advances.
In recent years, there have been numerous advancements in genomics and our understanding of cancer, rare diseases, cardiology, neurology, metabolism and infectious diseases. There have also been advancements in methods used to analyze very large amounts of molecular information. New technologies, including new AI/ML-powered technologies, and evolving business models in the field of precision medicine continue to develop rapidly. We must continuously enhance our offerings and develop new and improved features, applications and data modalities of our SOPHiA DDM Platform and related solutions, applications, products, and services to keep pace with scientific and industry developments. If we do not leverage or scale our database of genomic profiles or update our data analytics platform and improve our services and research and diagnostic applications and products to reflect new scientific knowledge, including in the fields of oncology and hereditary disorders, our SOPHiA DDM Platform and related solutions, applications, products, and services could become obsolete and sales of our SOPHiA DDM Platform and related solutions, applications, products, and services could decline or fail to grow as expected. A failure to make continuous improvements to our SOPHiA DDM Platform and related solutions, applications, products, and services to keep ahead of those of our competitors could result in the loss of customers or market share.
We face competition from many sources and we may be unable to compete successfully.
There are a number of healthcare technology companies providing bioinformatics analysis solutions, applications, services, and products in North and South America, Europe and Asia. See “Item 4. Information on the Company—Business Overview—Competition.” These competitors provide AI-driven precision medicine platforms, applications, services, and research and diagnostic products to hospitals, researchers, medical personnel, laboratories and other medical facilities. Many of these organizations, particularly in the United States, are more established, possess regulatory clearances and approval, have broader or deeper relations with healthcare professionals, customers and third-party payors, have greater ability to price their platforms, solutions, applications, products, and services competitively and have significantly greater financial and personnel resources and market share than we do. As a consequence, they may be able to spend more on application and product development, marketing, sales and other application and product initiatives than we can. Our continued success depends on our ability to:
•further penetrate the disease diagnostic solutions market and increase utilization of our SOPHiA DDM Platform and related solutions, applications, products, and services;
•maintain and widen our technology lead over competitors by continuing to innovate and deliver new application and product enhancements on a continuous basis;
•cost-effectively develop and improve our SOPHiA DDM Platform and related solutions, applications, products, and services;
•add new clinically relevant features, applications and data modalities to our SOPHiA DDM Platform and related solutions, applications, products, and services, such as anatomical pathology and proteomics, and generate suitable evidence supporting the research and clinical utility of our multimodal analytical approach ahead of our competitors; and
15
•maintain existing and establish additional research and development, manufacturing, distribution and commercialization collaborations and partnerships.
Our competitors also compete with us in recruiting and retaining qualified scientific, management and commercial personnel, as well as in acquiring technologies complementary to, or necessary for, development of our SOPHiA DDM Platform and related solutions, applications, products, and services. Because of the complex and technical nature of data-driven healthcare analysis and the dynamic market in which we compete, any failure to attract and retain a sufficient number of qualified employees could materially harm our ability to develop and commercialize our SOPHiA DDM Platform and related solutions, applications, products, and services, which would have a material adverse effect on our business, financial condition and results of operations.
As we attain greater commercial success, our competitors are likely to develop technology, platforms, applications, products, and services that offer features and functionality similar to ours. Improvements in existing competitive technology, platforms, applications, products, and services or the introduction of new competitive technology, platforms, applications, products, and services may make it more difficult for us to compete for sales, particularly if competitors demonstrate better accuracy, reliability, convenience or effectiveness or price their platforms, applications, products, and services less expensively.
Our competitors may develop data analytics platforms, applications, and products or adopt and implement standards or technologies not compatible with our SOPHiA DDM Platform and our other applications, services, and products. This may inhibit our efforts to develop our platform, applications, services, and products in a technology-agnostic manner, which could narrow the addressable market for our SOPHiA DDM Platform and our other applications, services, and products, adversely impact their sales and market acceptance, and limit our revenue growth and potential profitability.
In addition, we operate in an ecosystem where we and our customers have multiple offerings and our own customers may become our competitors or may view us as potential competitors. This could dis-incentivize our customers or potential customers from adopting our offerings and sharing data with us, which would adversely impact sales and market acceptance and limit our revenue growth and potential profitability.
Cybersecurity or data privacy breaches, other unauthorized or improper access, or (distributed) denial service lack of access (e.g., ransomware, persistent DoS/DDoS) could result in additional costs, service outages, loss of revenue, significant liabilities, harm to our brand and decreased/denied use of our SOPHiA DDM Platform and related solutions, applications, products, or services.
Any failure to prevent or mitigate cybersecurity incidents or improper access to, use, disclosure or other misappropriation of our data or customers’ personal data or the inability to rightfully access/control any such data (e.g., ransomware, persistent DoS/DDoS, system compromise, IP leak) could result in significant liability under state (e.g., state breach notification and privacy laws such as the California Consumer Privacy Act (“CCPA”)), federal (e.g., the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act (“HITECH”), collectively “HIPAA”) and international laws (e.g., the General Data Protection Regulation (“GDPR”) and the UK General Data Protection Regulation (“UK GDPR”)). Such an incident may also cause a material loss of revenue from the potential adverse impact to our reputation and brand, affect our ability to retain or attract new users and customers of our applications, products, and services and potentially disrupt our business.
As we become more dependent on information technologies, to conduct our operations, cybersecurity incidents, including deliberate cybersecurity attacks and attempts to gain unauthorized access to computer systems and cloud/hybrid networks, may increase in frequency and sophistication. Next generation sequencing (NGS) is becoming a new standard in public health sector. Its growing visibility inevitably attracts malicious activity threats, as it combines mission-critical infrastructure and high-value data. In addition to the nature of the data we hold and process, the geopolitical environment induced by the war in Ukraine and increasing pressure in Asia Pacific may make cybersecurity attacks more common and targeted.
These cybersecurity threats pose a risk to the security of our systems and networks, the confidentiality and the availability and integrity of our data, and these risks apply both to us (including via our corporate systems and any employees that may be working remotely) and to third parties on whose systems we rely for the conduct of our business. The techniques used to obtain unauthorized access, disable or degrade service or sabotage
16
systems develop and change frequently and often are not recognized until launched against a target, we and our collaborators may be unable to anticipate these techniques or to implement adequate preventative measures. We may in the future experience security incidents. In particular, we may be subject to cybersecurity incidents as we continue to adapt and upgrade our platform architecture. If we do not allocate and effectively manage the resources necessary to build and sustain the proper technology and cybersecurity infrastructure, we could suffer significant business disruption, data loss or damage to intellectual property or other proprietary information. While no security incidents in the past have had a material adverse effect on our business, financial condition and results of operations, we cannot predict the impact of any such future events. Further, although we are obligated under certain laws and regulations to ensure that our platform, systems and servers and those of our service providers remain compliant with the relevant legal requirements with respect to data privacy and cybersecurity, we do not have any control over the operations of the facilities or technology of such providers, including any third-party vendors that collect, process and store personal data on our behalf. Our platform, systems and servers and those of our service providers may be vulnerable to malware or physical or electronic break-ins that our or their security measures may not detect, including via supply chain attacks. Individuals able to circumvent such security measures may misappropriate our confidential or proprietary information, disrupt our operations, damage our computers or otherwise impair our reputation and business. We may need to expend significant resources and make significant capital investments to protect against cybersecurity breaches or to mitigate (compensating controls) the impact of any such breaches. In addition, to the extent that our platform, systems and servers and those of our service providers experience security breaches that result in the unauthorized or improper use of confidential data, employee data or personal data, we may not be indemnified for any losses resulting from such breaches. Currently, we carry cybersecurity coverage to mitigate certain potential losses, but this insurance is limited in amount and by the relevant policy’s terms and conditions and there can be no assurance that such potential losses will not exceed our policy limits.
There can be no assurance that we or our third-party providers will be successful in preventing cyberattacks or successfully mitigating their effects. If we are unable to prevent or mitigate the impact of such security breaches, our ability to attract and retain new customers, patients and other collaborators could be harmed as they may be reluctant to entrust their data to us, and we could be exposed to litigation and governmental investigations, proceedings and regulatory actions by federal, state and local regulatory entities in the United States and by other national regulatory entities, and we could breach our contractual obligations, all of which could result in significant legal and financial exposure and reputational damages and lead to a potential disruption to our business or other adverse consequences.
If we experience significant disruptions in our information technology systems, our business may be adversely affected.
We depend on our information technology systems for the efficient functioning of our business, including the performance, distribution and maintenance of our SOPHiA DDM Platform and related solutions, applications, products, and services, as well as for accounting, data storage, compliance, purchasing and inventory management, and our continued growth is dependent on our ability to adapt and upgrade our platform architecture without suffering significant business disruption, data loss or the loss of or damage to intellectual property or other proprietary information. Our information technology systems may fail and are vulnerable to breakdown, cybersecurity breach, interruption or damage from computer viruses, ransomware, DoS/DDoS attacks or other malware, attacks by computer hackers, including sophisticated nation-state and nation-state-supported actors, employee error or malfeasance, theft or misuse, failures during the process of upgrading or replacing software, databases or components thereof, power outages, damage or interruption from fires or other natural disasters, hardware failures, telecommunication failures and user errors, among other malfunctions. We could be subject to an unintentional event that involves a third party gaining unauthorized access to our systems, which could disrupt our operations, corrupt our data or result in release of our confidential information. Technological interruptions would disrupt our operations, including our ability to timely ship and track diagnostic test orders and results, project inventory requirements, manage our supply chain and otherwise adequately service our customers or disrupt our customers’ ability to use our applications, products, and services. In the event we experience significant disruptions, we may be unable to repair our systems in an efficient and timely manner. Accordingly, such events may disrupt or reduce the efficiency of our entire operation and have a material adverse effect on our business, financial condition and results of operations.
Currently, we carry business interruption coverage to mitigate certain potential losses, but this insurance is limited in amount and by the relevant policy’s terms and conditions and there can be no assurance that such potential losses will not exceed our policy limits. The successful assertion of one or more large claims against us that exceed or are not covered by our insurance coverage, or changes in our insurance policies, including
17
premium increases or the imposition of large deductible or co-insurance requirements, could have a material adverse effect on our business, financial condition and results of operations. Further, such insurance may not cover all potential claims to which we are exposed. We are increasingly dependent on complex information technology to manage our infrastructure. Our information systems require an ongoing commitment of significant resources to maintain, protect and enhance our existing systems. Failure to maintain or protect our information systems and data integrity effectively could have a material adverse effect on our business, financial condition and results of operations.
Our business could be adversely affected by health epidemics, pandemics and unplanned events.
Our business could be adversely affected by health epidemics, pandemics and natural disasters. To the extent any pandemic, epidemic or outbreak of an infectious disease adversely affects our business and financial results, it may also have the effect of heightening many of the other risks described in this “Item 3D. Risk Factors” section.
In addition, any unplanned event, such as a flood, fire, explosion, earthquake, extreme weather condition, power shortage, telecommunication failure or other natural or man-made accidents or incidents that result in us being unable to fully use our facilities, which may render it difficult or impossible for us to operate our platform for some period of time or result in damages to our inventory that require sensitive storage conditions. Certain of these natural events and disasters may become more frequent and severe as a result of the effects of climate change. We do not presently have an emergency back-up generator in place at our Rolle, Switzerland laboratory facility which would otherwise mitigate, to some extent, the effects of a prolonged power outage. Furthermore, our facility and the equipment we use to perform our research and development work could be unavailable or costly and time-consuming to repair or replace, which may increase backlog. It would be difficult, time-consuming and expensive to rebuild our facility, to locate new facilities or license or transfer our proprietary technologies to a third party.
We carry business continuity insurance and insurance for damage to our property in amounts and pursuant to terms that we believe are reasonable, but this insurance may not cover all of the risks associated with damage, may not provide coverage in amounts sufficient to cover our potential losses and may not continue to be available to us on acceptable terms, if at all.
If product liability lawsuits are brought against us, we may incur substantial liabilities and may be required to limit or halt the marketing and sale of our SOPHiA DDM Platform and related solutions, applications, products, and services.
We face an inherent risk of product liability as a result of the marketing and sale of our SOPHiA DDM Platform and related solutions, applications, products, and services and the testing of our SOPHiA DDM Platform in clinical studies. Such claims may be made against us even if the apparent injury is due to the actions of others or the preexisting health of the patient. This risk may be heightened by our reliance on third parties for the manufacturing, marketing and research and development of our SOPHiA DDM Platform and related solutions, applications, products, and services. If we cannot successfully defend ourselves against product liability claims, we may incur substantial liabilities or be required to limit or halt the marketing and sale of our products. Even successful defense would require significant financial and management resources, harm our reputation in our industry and reduce sales.
We believe that we have adequate product liability insurance, but it may not prove to be adequate to cover all liabilities that we may incur. Insurance coverage is increasingly expensive. We may not be able to maintain or obtain insurance at a reasonable cost or in an amount adequate to satisfy any liability that may arise. Our insurance policy contains various exclusions, and we may be subject to a product liability claim for which we have no coverage. The potential inability to obtain sufficient product liability insurance at an acceptable cost to protect against product liability claims could prevent or inhibit the marketing and sale of our applications, products, and services. In addition, any product liability claims brought against us, with or without merit, could increase our product liability insurance rates or prevent us from securing continuing coverage.
If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial condition and results of operations in a timely manner or prevent fraud.
18
Effective internal control over financial reporting necessary for us to provide reliable financial reports and, together with adequate disclosure controls and procedures, are designed to prevent and/or detect error and/or fraud in a timely manner . Any failure to implement required new or improved controls, or difficulties encountered in their implementation could cause us to fail to meet our reporting obligations. As required by Section 404 of the Sarbanes-Oxley Act, our management has conducted an assessment on the effectiveness of our internal control over financial reporting. However, as an emerging growth company, our independent registered public accounting firm has not provided an attestation report on our internal control over financial reporting. There can be no assurance that any future testing conducted by us or, if required, by our independent registered public accounting firm will not reveal deficiencies in our internal control over financial reporting. If we do not maintain an effective system of internal control over financial reporting, we may be unable to accurately report our financial condition and results of operations in a timely manner, may incur additional financial and management resources to remediate any deficiencies, or may be unable to detect and prevent fraud, all of which would adversely affect our business.
Litigation and other legal proceedings may adversely affect our business.
From time to time, we may become involved in legal proceedings relating to patent and other intellectual property matters, product liability claims, employee claims, tort or contract claims, regulatory investigations, securities class action and other legal proceedings or investigations, which could have an adverse impact on our reputation, business and financial condition and divert the attention of our management from the operation of our business. Litigation is inherently unpredictable and can result in excessive or unanticipated judgments and/or injunctive relief that affect how we operate our business. We could incur judgments or enter into settlements of claims for monetary damages or for agreements to change the way we operate our business, or both. There may be an increase in the scope of these matters or there may be additional lawsuits, claims, proceedings or investigations in the future, which could have a material adverse effect on our business, financial condition and results of operations. Adverse publicity about regulatory or legal action against us could damage our reputation and brand image, undermine our customers’ confidence and reduce long-term demand for our solutions, applications, products, and services, even if the regulatory or legal action is unfounded or not material to our operations.
Our business is subject to economic, political, regulatory and other risks associated with international operations.
Our results could be adversely affected by a variety of risks associated with our international operations, including economic weakness, such as inflation and rising interest rates, or political instability in economies and markets; global trends towards pharmaceutical pricing; differing regulatory requirements for bioinformatics analysis services and research and diagnostic products approvals; differing reimbursement, pricing and insurance regimes; potentially reduced protection for, and complexities and difficulties in obtaining, maintaining, protecting and enforcing, intellectual property rights; difficulties in compliance with U.S. and non-U.S. laws and regulations, including data security and data protection laws, which may result in increased compliance costs to us, and anti-corruption and anti-bribery laws; changes in regulations and customs, tariffs and trade barriers; changes in currency exchange rates and currency controls; changes in a specific country’s or region’s political or economic environment; trade protection measures, economic sanctions and embargoes on certain countries and persons, import or export licensing requirements or other restrictive actions by governments, including with respect to our applications, products, and services, in particular IT solutions, services and technologies on which our operations rely; changes in tax laws; compliance with tax, employment, immigration and labor laws for employees living or traveling abroad; workforce uncertainty in countries where labor unrest is more common than in Switzerland and the United States; difficulties associated with staffing and managing international operations, including differing labor relations; production shortages resulting from any events affecting raw material supply or manufacturing capabilities abroad; business interruptions resulting from geopolitical actions, including war and terrorism (including the ongoing conflict between Russia and Ukraine and in the Middle East), or natural disasters including earthquakes, typhoons, floods and fires; and the impact of public health epidemics on employees and the global economy. Certain of our suppliers and service providers are located in geopolitically sensitive regions, including Ukraine and other eastern European countries, and are negatively impacted in their ability to conduct their operations and perform services for us as a result of the ongoing conflict between Russia and Ukraine. As a result, we may experience supply chain disruptions and interruptions in our operations. Any of these factors could require us to modify our business plans and strategy and significantly harm our future international expansion and operations and, consequently, our revenue and results of operations.
19
Risks Related to Governmental Regulation
Currently our applications and products in the United States are labeled as RUO. We intend to seek regulatory clearance or approval to offer our applications and products as IVD applications and products for diagnostic use. We cannot guarantee when, if at all, we will apply for regulatory clearance or approval or that we will be successful in obtaining such clearances or approvals.
While we have several CE-IVD marked applications and products in the European Union (“EU”), our currently available applications and products in the United States are labeled as RUO applications and products and are not intended for diagnostic use. Although we have focused initially on the RUO applications and products only, our strategy is to expand our application and product line to encompass applications and products that are intended to be used as IVDs. Such IVD products will be subject to regulation by the FDA as medical devices, including requirements for regulatory clearance or approval of such applications and products before they can be marketed. Accordingly, we will be required to obtain FDA 510(k) clearance or premarket approval (“PMA”) in order to sell our applications and products in a manner consistent with FDA laws and regulations. Such regulatory approval processes or clearances are expensive, time-consuming and uncertain; our efforts may never result in any premarket approval or 510(k) approval or clearance for our applications and products; and failure by us to obtain or comply with such approvals and clearances could have an adverse effect on our business, financial condition or operating results.
Regulatory authorities have substantial discretion in the clearance and approval process. They may refuse to accept any application or may decide that our data are insufficient for clearance or approval and require additional studies. Therefore, even if we believe the data collected from studies of our platform are promising, such data may not be sufficient to support clearance or approval by any regulatory authority. If we are required to conduct additional studies or other testing of any of our platform beyond those we contemplate, we may incur significant additional costs and regulatory approval may be delayed or prevented. Furthermore, approval by one regulatory authority does not ensure approval by regulatory authorities in other jurisdictions, and we may be required to undertake additional studies to access particular markets.
If we successfully obtain such clearance or approvals, we will be subject to a substantial number of additional requirements for medical devices, including establishment registration, device listing, and compliance with the Quality Systems Regulation (“QSR”) which cover the design, testing, production, control, quality assurance, labeling, packaging, servicing, sterilization (if required), and storage and shipping of medical devices (among other activities), advertising, record keeping, post-market surveillance, post-approval studies, adverse event reporting, and correction and removal (recall) regulations. We may be required to expend significant resources to ensure ongoing compliance with the FDA regulations and/or take satisfactory corrective action in response to enforcement action, which may have a material adverse effect on the ability to design, develop and commercialize applications and products using our technology as planned. Failure to comply with these requirements may subject us to a range of enforcement actions, such as warning letters, injunctions, civil monetary penalties, criminal prosecution, recall and/or seizure of products, and revocation of marketing authorization, as well as significant adverse publicity. If we fail to obtain, or experience significant delays in obtaining, regulatory approvals for IVD or other applications and products, such applications and products may not be able to be launched or successfully commercialized in a timely manner, or at all.
Laboratory developed tests (“LDTs”) are a subset of IVD tests that are designed, manufactured and used within a single laboratory. The FDA maintains that LDTs are medical devices and has historically exercised enforcement discretion for most LDTs. The FDA recently proposed a rule that would subject LDTs to a new and phased-in regulatory framework. This rule, if finalized, or if there are any other significant changes in the way that the FDA regulates any LDTs that our customers develop using our RUO applications and products could affect our business. If the FDA requires laboratories to undergo premarket review, as proposed, and comply with other applicable FDA requirements in the future, the cost and time required to commercialize an LDT will increase substantially, and may reduce the financial incentive for laboratories to develop LDTs, which could reduce demand for our RUO applications and products.
We develop applications and products for clinical laboratories, which may be qualified as LDTs, as well as market RUO applications and products. Our customers may decide to validate our applications and products to use as an LDT, which will be covered under Clinical Laboratory Improvement Amendments
20
of 1988 (“CLIA”) and CMS, although future developments may cause us to be subject to additional FDA requirements.
The laws and regulations governing the marketing of diagnostic applications and products are evolving, extremely complex and in many instances, there are no significant regulatory or judicial interpretations of these laws and regulations. Pursuant to its authority under the Federal Food, Drug, and Cosmetic Act (the “FDCA”), the FDA has jurisdiction over medical devices, including in vitro diagnostics and, therefore, potentially our applications and products.
Pursuant to the FDCA and its implementing regulations, the FDA regulates the research, testing, manufacturing, safety, labeling, storage, record keeping, premarket clearance or approval, marketing and promotion, and sales and distribution of medical devices in the United States to ensure that medical products distributed domestically are safe and effective for their intended uses. Although the FDA has asserted that it has authority to regulate the development and use of LDTs, such as our and many other laboratories’ tests, as medical devices, it has generally exercised enforcement discretion and is currently not regulating most tests developed and performed within a single high-complexity CLIA-certified laboratory. The FDA has proposed a new regulatory framework to regulate LDTs as a subset of in vitro diagnostics.
We currently do not offer any diagnostic applications and products in the United States. We believe that our research applications and products, as utilized in clinical laboratories by our customers, are and would be considered LDTs and that as a result, the FDA does not require that they obtain regulatory clearances or approvals for the LDTs or their components pursuant to the FDA’s current policies and guidance. Although we believe that our products and test components delivered to our customers, when validated as LDTs, are either exempt from FDA medical device regulations or are subject to an enforcement discretion policy, it is possible that the FDA would not agree with these determinations or that the FDA will change its regulations and policies such that our applications and products become regulated as medical devices.
On September 29, 2023, the FDA announced its proposal to amend its regulations to make explicit that In Vitro Diagnostic Products (“IVDs”) are devices under the Federal Food, Drug, and Cosmetic Act including when the manufacturer of the IVD is a laboratory. In conjunction with the proposed amendment, the FDA has proposed a policy under which it intends to phase out its general enforcement discretion approach for LDTs so that IVDs manufactured by a laboratory would generally fall under the same enforcement approach as other IVDs. Some of our customers in the United States may decide to discontinue the use of RUO products and test components in their LDTs.
In addition, changes in the current regulatory framework for diagnostic applications, products, and services can impose additional regulatory burdens on us. For example, the FDA’s Center for Devices and Radiological Health is currently considering a total product lifecycle-based regulatory framework for AI/ML technologies. On January 12, 2021, the FDA released its Artificial Intelligence/Machine Learning-Based Software as a Medical Device Action Plan. As the regulatory framework evolves, we may incur substantial costs to ensure compliance with new or amended laws and regulations. Failure to comply with any of these laws and regulations could result in enforcement actions against us, damage to our reputation and loss of goodwill, any of which could have a material adverse effect on our business. In addition, legislation could require us to seek FDA-premarket review of other applications, products, and services, including reports that we do not currently believe require premarket authorization but could be subject to additional regulation including premarket review. For any such review, we may be required to conduct extensive analytical validation and user comprehension studies to demonstrate the accuracy of our test results and that they are appropriate for sale directly to consumers. This process will likely be costly, time-consuming and uncertain.
Further, on September 28, 2022, the FDA has issued the guidance on Clinical Decision Support software. It is unclear how any such guidance will impact our SOPHiA DDM Platform.
Our operations, therefore, are or may become subject to extensive regulation by the FDA in the United States, the IVDR in the EU, the MHRA in the UK, and various health authorities, in particular SwissMedic in Switzerland, and the ANSM in France, and in other jurisdictions in which we conduct business. Government regulations specific to medical devices are wide-ranging and govern, among other things:
•test design, development, manufacture, and release;
•laboratory and clinical testing, labeling, packaging, storage and distribution;
21
•product safety and efficacy;
•premarketing clearance, approval or certification;
•service operations;
•record keeping;
•application and product marketing, promotion and advertising, sales and distribution;
•post-marketing surveillance, including reporting of deaths or serious injuries, recalls, correction and removals;
•post-market approval studies; and
•product import and export.
As discussed above, although we believe that our current line of products and their components, as utilized in clinical laboratories by our customers, are LDTs, subject to state licensing requirements and federal regulation by CMS under CLIA, it is possible that the FDA or comparable regulatory authorities would not agree with our determinations. If our products become subject to 510(k) or other similar FDA regulations, we would need to comply with the applicable regulations or face significant civil and criminal penalties. Exposure to these additional regulatory requirements would also affect our business, financial condition and results of operations.
Failure to comply with federal, state, and foreign laboratory licensing requirements if we begin to provide diagnostic applications and products in the United States could result in significant penalties and materially adversely affect our operations.
CLIA is a federal law that regulates clinical laboratories that perform testing on specimens derived from humans for the purpose of providing information for the diagnosis, prevention, or treatment of disease, or impairment of, or the assessment of the health of, human beings. CLIA regulations require, among other things, clinical laboratories to obtain a certificate and mandate specific standards in the areas of personnel qualifications, administration, participation in proficiency testing, test management, and quality assurance. In addition to federal certification requirements of laboratories under CLIA, CLIA provides that states may adopt laboratory regulations and licensure requirements that are more stringent than those under federal law. A number of states have implemented their own licensure and more stringent laboratory regulatory requirements. Such laws, among other things, establish standards for the day-to-day operation of a clinical laboratory, including the training and skills required of personnel and quality control. Failure to comply with CLIA and applicable state clinical laboratory licensure requirements may result in a range of enforcement actions, including license suspension, limitation, or revocation, directed plan of action, onsite monitoring, civil monetary penalties, and criminal sanctions as well as significant adverse publicity.
Based on our current scope of operations, we do not currently operate a CLIA-certified laboratory and our customers are responsible for their own CLIA certification. However, if we begin to provide diagnostic applications and products in the United States, we will become subject to such requirements.
We may fail to obtain required clearances or approvals in additional jurisdictions for any of our applications, products, or services and, even if we do, we may never be able to commercialize them in additional jurisdictions, which would limit our ability to realize their full market potential.
In order to eventually market any of our current or future applications, products, and services in any particular jurisdiction, we must establish and comply with numerous and varying regulatory requirements on a jurisdiction-by-jurisdiction basis regarding quality, safety, performance and efficacy. In addition, regulatory clearance, authorization or approval in one country does not guarantee regulatory clearance, authorization or approval in any other country. For example, the performance characteristics of our applications, products, and services may need to be validated separately in specific ethnic and genetic populations. Approval processes vary among countries and can involve additional product testing and validation and additional administrative review periods.
Seeking regulatory clearance, authorization or approval could result in difficulties and costs. Regulatory requirements and ethical approval obligations can vary widely from country to country and could delay or
22
prevent the introduction of our applications, products, and services in those countries. We have no experience in obtaining regulatory clearance, authorization or approval in international markets. If we fail to comply with regulatory requirements in international markets or to obtain and maintain required regulatory clearances, authorizations or approvals in international markets, or if those approvals are delayed, our target market will be reduced and our ability to realize the full market potential of our applications, products, and services will be unrealized.
Our products or services may be subject to product or service recalls in the future. A recall of products or services, either voluntarily or at the direction of a regulatory authority, or the discovery of serious safety issues with our products or services, could have a significant adverse impact on us.
Regulatory authorities can require the recall of commercialized products or services that are subject to its regulation. Manufacturers may, under their own initiative, recall a product or service if any deficiency is found. A government-mandated or voluntary recall by us or one of our distributors could occur as a result of an unacceptable health risk, component failures, failures in laboratory processes, malfunctions, manufacturing errors, design or labeling defects, or other deficiencies and issues. Recalls of any of our commercialized products or services would divert managerial and financial resources, subject us to liability claims and require us to bear other costs and adversely affect our business, results of operations, financial condition and reputation. If we initiate a recall, including a correction or removal, for one of our commercialized products or services, issue a safety alert, or undertake a field action or recall to reduce a health risk, this could lead to increased scrutiny by the FDA, other governmental and regulatory enforcement bodies, and our customers regarding the quality and safety of our products and services, and to negative publicity, including FDA alerts, press releases, or administrative or judicial actions. Furthermore, the submission of these reports could be used against us by competitors and cause customers to delay purchase decisions or cancel orders, which would harm our reputation.
We are subject to stringent privacy and, information security laws and regulations and changes in such laws and regulations could adversely affect our business and increase our costs.
We are subject to numerous state, federal and foreign laws and regulations that govern the collection, transmission, storage, dissemination, use, privacy, confidentiality, security, availability, integrity and processing of individually identifiable information. “Item 4. Information on the Company—B. Business Overview—Government Regulation—Data Privacy and Security.” These laws and regulations are complex and change frequently, at times due to changes in political climate, and existing laws and regulations are subject to different and conflicting interpretations, which adds to the complexity of processing personal data from these jurisdictions. Failure to comply with any of these laws and regulations could result in enforcement actions against us, including fines, imprisonment of company officials, public censure, claims for damages by affected individuals, damage to our reputation and loss of goodwill, any of which could have a material adverse effect on our business.
This risk is heightened as there is uncertainty regarding the applicability of data privacy regulations to certain types of data that we process, such as health-related and genetic data. While we deploy specific measures and processes designed to de-identify data, we may fail in the comprehensive application of the process resulting in a breach of requirements and our contractual obligation. Some of our product and services rely on anonymized data and while the legal definition is clear, the effective application of the anonymization techniques are subject to multiple interpretation and can prove to be highly difficult and costly.
The transfer of data has been subject to increasing scrutiny. Although there are legal mechanisms to allow for the transfer of personal data from the EEA, Switzerland and the UK to the United States, uncertainty remains about such mechanisms. There have been and there may in the future be legal challenges surrounding the mechanisms that allow companies to transfer personal data from the EU and EEA to the United States, which could limit our ability to transfer personal data across borders, particularly if governments are unable or unwilling to reach new or maintain existing agreements that support cross-border data transfers. Inability to transfer personal data from the EU, EEA, Switzerland or the UK to the United States may restrict our research and development activities in these territories and limit our ability to offer applications, products, and services we may develop.
Because the interpretation and application of health-related and data protection laws, regulations, standards and other obligations are uncertain, and often contradictory and in flux, it is possible that the scope and requirements of these laws may be interpreted and applied in a manner that is inconsistent with our practices and our efforts to comply with the evolving data protection rules may be unsuccessful. If so, this could result in
23
government-imposed fines or orders requiring that we change our practices, which could adversely affect our business. In addition, these privacy regulations may differ from country to country, and may vary based on whether testing is performed in the United States or in the local country, and our operations or business practices may not comply with these regulations in each country.
In addition to the possibility of fines, sanctions, lawsuits, regulatory investigations, public censure, other claims and penalties, and significant costs for remediation and damage to our reputation, we could be materially and adversely affected if legislation or regulations are expanded to require changes in our data processing practices and policies or if governing jurisdictions interpret or implement their legislation or regulations in ways that negatively impact our business. Complying with these various laws could cause us to incur substantial costs or require us to change our business practices and compliance procedures in a manner adverse to our business. Any inability to adequately address data privacy or security-related concerns, even if unfounded, or to comply with applicable laws, regulations, standards and other obligations relating to data privacy and security, could result in additional cost and liability to us, harm our reputation and brand, damage our relationships with customers and have a material and adverse impact on our business.
Any failure to comply with our privacy policies or contractual or statutory notification obligations could result in significant liability or reputational harm.
We make public statements about our use and disclosure of personal information through our privacy policy, information provided on our internet platform and press statements. Although we endeavor to comply with our public statements and documentation, we may be alleged to have failed to do so. The publication of our privacy policy and other statements that provide promises and assurances about data privacy and security can subject us to potential government or legal action if they are found to be deceptive, unlawful, unfair or misrepresentative of our actual practices. Any failure, real or perceived, by us to comply with our posted privacy policies or with any legal or regulatory requirements, standards, certifications or orders or other privacy or consumer protection-related laws and regulations applicable to us could cause our customers to reduce their use of our applications, products, and services and could materially and adversely affect our business, financial condition and results of operations. In many jurisdictions, enforcement actions and consequences for noncompliance can be significant and are rising. In addition, from time to time, concerns may be expressed about whether our applications, products, services, or processes compromise the privacy of customers and others. Concerns about our practices with regard to the collection, use and reuse, retention, security, disclosure, transfer and other processing of personal information or other privacy-related or security-related matters, even if unfounded, could damage our reputation and materially and adversely affect our business, financial condition and results of operations.
Many statutory requirements, both in the United States and abroad, include obligations for companies to notify individuals of security breaches involving certain personal information, which could result from breaches experienced by us or our third-party service providers. For example, the GDPR requires from companies experiencing a personal data breach to report the breach within 72 hours to the supervisory authority and where necessary to the individuals affected by the breach. Also, laws in all 50 U.S. states and the District of Columbia require businesses to provide notice to consumers whose personal information has been disclosed as a result of a data breach. These laws are not consistent, and compliance in the event of a widespread data breach is difficult and may be costly. Moreover, jurisdictions have been frequently amending existing laws, requiring attention to changing regulatory requirements. We also may be contractually required to notify customers or other counterparties of a security breach. Although we may have contractual protections with our third-party service providers, contractors and consultants, any actual or perceived security breach could harm our reputation and brand, expose us to potential liability or require us to expend significant resources on data security and in responding to any such actual or perceived breach. Any contractual protections we may have from our third-party service providers, contractors or consultants may not be sufficient to adequately protect us from any such liabilities and losses, and we may be unable to enforce any such contractual protections.
Our operations may subject us to various healthcare laws and regulations and could face substantial penalties if we are unable to fully comply with such laws.
Our operations may subject us to health care regulation and enforcement by both the federal government and the states and foreign jurisdictions in which we conduct our business, which influence, among other things, how we structure our sales offerings, including discount practices, customer support, education and training
24
programs, and physician consulting and other service arrangements. Applicable U.S. federal and state healthcare laws and regulations include the following:
•the federal Anti-Kickback Statute (the “AKS”), which prohibits, among other things, persons or entities from soliciting, receiving, offering or providing remuneration, directly or indirectly, overtly or covertly, in cash or in kind, in return for, or to induce, either the referral of an individual for, or the purchase, lease, order or recommendation of, any good, facility, item or services for which payment may be made under a federal healthcare program such as the Medicare and Medicaid programs;
•the federal physician self-referral prohibition, commonly known as the Stark Law, which prohibits physicians from referring Medicare or Medicaid patients to providers of “designated health services” with whom the physician or a member of the physician’s immediate family has an ownership interest or compensation arrangements, unless a statutory or regulatory exception applies;
•the federal Eliminating Kickbacks in Recovery Act of 2018 (the “EKRA”) prohibits payments for referrals to recovery homes, clinical treatment facilities, and laboratories. EKRA’s reach extends beyond federal health care programs to include private insurance (i.e., it is an “all payor” statute);
•HIPAA, which established additional federal civil and criminal liability for, among other things, knowingly and willfully executing a scheme to defraud any healthcare benefit program or making false statements in connection with the delivery of or payment for healthcare benefits, items or services;
•HIPAA, as amended by HITECH and its implementing regulations, which imposes certain requirements relating to the privacy, security and transmission of individually identifiable health information;
•federal false claims and civil monetary penalties laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, false or fraudulent claims for payment to the federal government;
•the federal Physician Payments Sunshine Act requirements, which require certain manufacturers of drugs, devices, biologics and medical supplies to report to CMS information related to payments and other transfers of value made to or at the request of physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors) and teaching hospitals, and certain ownership and investment interests held by physicians and their immediate family members; and
•state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws, which may apply to items or services reimbursed by any third-party payor, including commercial insurers.
Ensuring that our business and operations comply with applicable healthcare laws and regulations is costly. Any action brought against us for violation of these laws or regulations, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. If our operations are found to be in violation of any of these laws and regulations, we may be subject to any applicable penalty associated with the violation, including, among others, significant administrative, civil and criminal penalties, damages, fines, disgorgement, imprisonment, integrity oversight and reporting obligations, and exclusion from participation in government-funded healthcare programs such as Medicare and Medicaid. Additionally, we could be required to refund payments received by us, and we could be required to curtail or cease our operations. Any of the foregoing consequences could seriously harm our business, financial condition and results of operations.
25
Our employees, collaborators, distributors, agents, contractors and collaborators may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
We cannot ensure that our compliance controls, policies and procedures will in every instance protect us from acts committed by our employees, collaborators, distributors, agents, contractors or collaborators that would violate the laws or regulations of the jurisdictions in which we operate, including, without limitation, healthcare, employment, anti-corruption, environmental, competition, and patient privacy and other privacy laws and regulations. Misconduct by these parties could include intentional failures to comply with FDA, EMA, MHRA or other applicable regulations, including, without limitation, regulations governing the marketing, sale, labeling and use of RUO and IVD applications and products, provide accurate information to the FDA, the EMA, the MHRA and comparable regulatory authorities, comply with healthcare fraud and abuse laws and regulations in the United States and abroad, report financial information or data accurately or disclose unauthorized activities to us. In addition, because business is heavily regulated and therefore involves significant interaction with public officials, including officials of non-U.S. governments, government purchasers and healthcare providers who are employed by governments, we face heightened risk with respect to compliance with the Foreign Corrupt Practices Act (the “FCPA”) and similar anti-bribery or anti-corruption laws, regulations or rules of other countries in which we operate, including the UK Bribery Act 2010 and the French Law n° 2016-1691 (Sapin II). These laws prohibit offering, promising, giving, or authorizing others to give anything of value, either directly or indirectly, to a government official in order to influence official action, or otherwise obtain or retain business.
There is no certainty that all of our employees, collaborators, distributors, agents, contractors and collaborators, or those of our affiliates, will comply with all applicable laws and regulations, particularly given the high level of complexity of these laws. We have provisions in our Code of Business Conduct and Ethics (the “Code of Ethics”), an anti-corruption policy, certain provisions in some of our agreements with third parties, including our collaborators and distributors, and certain controls and procedures in place that are designed to mitigate the risk of noncompliance with anti-corruption and anti-bribery laws. However, it is not always possible to identify and deter misconduct by employees and agents, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from government investigations or other actions stemming from a failure to comply with these laws or regulations. Violations of these laws and regulations could result in, among other things, significant administrative, civil and criminal fines and sanctions against us, our officers, or our employees, the closing down of our facilities, exclusion from participation in federal healthcare programs, implementation of compliance programs, integrity oversight and reporting obligations and prohibitions on the conduct of our business. Any such violations could include prohibitions on our ability to offer our SOPHiA DDM Platform and related solutions, applications, products, and services in one or more countries and could materially damage our reputation, our brand, our international expansion efforts, our ability to attract and retain employees, and our business, prospects, operating results and financial condition.
We face risks related to handling of hazardous materials and other regulations governing environmental safety.
Our activities currently require and may in the future continue to require the use of hazardous chemicals and biohazardous waste, including chemical, biological agents and compounds, blood and bone marrow samples, and other human tissue. We cannot eliminate the risk of accidental contamination or injury to employees or third parties from the use, storage, handling or disposal of these materials. In the event of contamination or injury, we could be held liable for any resulting damages, and any liability could exceed our resources or any applicable insurance coverage we may have. Additionally, we are subject on an ongoing basis to laws and regulations governing the use, storage, handling and disposal of these materials and specified waste services that both public officials and private individuals may seek to enforce. We could discover that we, an acquired business or our suppliers are not in material compliance with these regulations. The cost of compliance with these laws and regulations may become significant and could negatively affect our business, financial condition and results of operations. We do not carry specific biological waste or hazardous waste insurance coverage, workers’ compensation or property and casualty and general liability insurance policies that include coverage for damages and fines arising from biological or hazardous waste exposure or contamination.
26
If a clinical trial subject’s or a clinical research study participant’s informed consent is challenged or proven invalid, unlawful, or otherwise inadequate for our purposes, our application and product development efforts may be hindered and we could become involved in legal challenges.
We seek to ensure that all data and biological samples that we receive from our collaborators and customers have been collected from subjects or participants who have provided appropriate informed consent for purposes that extend to our development activities. We also strive to make sure such data and samples are provided to us in a subject de-identified manner. Our collaborators currently conduct clinical trials and clinical research studies in a number of different countries. The collection of data and samples in many different countries results in complex legal questions regarding the adequacy of informed consent and the status of genomic material under a large number of different legal systems. Therefore, we rely on our collaborators and customers to comply with the informed consent requirements and with applicable local law and international regulation. The subject’s or participant’s informed consent obtained in any particular country could be challenged in the future, and those could prove invalid, unlawful or otherwise inadequate for our purposes. Any findings against us, or our collaborators and customers, could deny us access to or force us to stop using some of our data and clinical samples, which would hinder our application and product development efforts, potentially involve us in costly and prolonged litigation, result in reputational harm and adversely affect our business, financial condition and results of operations.
If we or our suppliers fail to comply with ongoing FDA or comparable regulatory authority requirements, or if we experience unanticipated problems with our research and diagnostic, they could be subject to restrictions or withdrawal from the market.
Any medical device that we manufacture, including those for which we obtain regulatory clearance, approval, or certification and the manufacturing processes, reporting requirements, post-approval clinical data and promotional activities for such diagnostic test, will be subject to continued regulatory review, oversight and periodic inspections by the FDA and comparable regulatory authorities. In particular, we and our suppliers may be required to comply with the FDA’s QSR for medical devices, the International Standards Organization (“ISO”) 13485 standards for the manufacture of our diagnostic products and other regulations that cover the methods and documentation of the design, testing, production, control, quality assurance, labeling, packaging, storage and shipping of any diagnostic test for which we obtain clearance or approval. Regulatory authorities such as the FDA enforce the QSR and other regulations through periodic inspections. The failure by us or one of our suppliers to comply with applicable statutes and regulations administered by the FDA and comparable regulatory authorities, or the failure to timely and adequately respond to any adverse inspection observations or product safety issues, could result in, among other things, one or more of the following enforcement actions:
•untitled letters, warning letters, fines, injunctions, consent decrees and civil penalties;
•unanticipated expenditures to address or defend such actions;
•customer notifications for repair, replacement or refunds;
•recall, detention or seizure of our diagnostics products;
•operating restrictions or partial suspension or total shutdown of production;
•refusing or delaying our requests for 510(k) clearance or PMA of new diagnostics products or modified versions of such products currently manufactured, as well as CE marking certification (in the EU);
•operating restrictions;
•withdrawing 510(k) clearances on PMA approvals that have already been granted;
•suspending or withdrawing our CE mark certifications; and
•criminal prosecution.
In addition, we are required to conduct surveillance to monitor the safety or effectiveness of our research and diagnostic products, and we must comply with medical device reporting requirements, including the reporting of adverse events and malfunctions related to our research and diagnostic products. Later discovery of previously
27
unknown problems with our diagnostic products, including unanticipated adverse events or adverse events of unanticipated severity or frequency, manufacturing problems, or failure to comply with regulatory requirements such as QSR, may result in changes to labeling restrictions on such products or manufacturing processes, withdrawal of the research and diagnostic products from the market, voluntary or mandatory recalls, a requirement to repair, replace or refund the cost of any medical device we manufacture or distribute, fines, suspension of regulatory approvals, product seizures, injunctions or the imposition of civil or criminal penalties, which would adversely affect our business, operating results and prospects.
Risks Related to Intellectual Property
If we are not able to obtain, maintain, defend and enforce patent and other intellectual property protection or if the scope of such patent and other intellectual property protection is not sufficiently broad, our competitors could develop and commercialize applications, products, services, and technology similar or identical to ours.
Our success depends in part on our ability to obtain, maintain, defend, and enforce patents and other forms of intellectual property rights, including in-licenses of intellectual property rights of others, as well as our ability to preserve our trade secrets and to prevent third parties from infringing, misappropriating or otherwise violating our intellectual property and proprietary rights. Our ability to protect our applications, products, or services from unauthorized use by third parties depends on the extent to which valid and enforceable patents cover them or they are effectively protected as trade secrets. Although we have filed a number of patents, our patent portfolio is in an earlier stage of prosecution, and we own a limited number of issued patents related to our applications, products, and technology. For information regarding our patent portfolio, see “Item 4. Information on the Company—B. Business Overview—Intellectual Property.”
The patent position of biotechnology and information technology companies generally is highly uncertain, involves complex legal and factual questions, and has been the subject of much litigation in recent years. As a result, the issuance, scope, validity, enforceability, and commercial value of our patent rights are highly uncertain. There can be no assurance that our patent rights will not be invalidated or held to be unenforceable, will adequately protect our technology, applications, products, or services or provide any competitive advantage, or that any of our pending or future patent applications will issue as valid and enforceable patents. Our ability to obtain and maintain patent protection for our methods and related solutions, applications, products, or services is uncertain due to a number of factors, including that:
•we or our licensors may not have been the first to invent the technology covered by our pending patent applications or issued patents;
•we or our licensors may not be the first to file all patent applications, as patent applications in the United States and most other countries are confidential for a period of time after filing;
•our methods and related solutions, applications, and products may not be patentable;
•our disclosures in patent applications may not be sufficient to meet the statutory requirements for patentability;
•any or all of our pending patent applications may not result in issued patents;
•others may independently develop identical, similar or alternative technologies;
•others may design around our patent claims to produce competitive technologies, methods, applications, or products that fall outside of the scope of our patents;
•we may fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection;
•we may not seek or obtain patent protection in countries that may eventually provide us a significant business opportunity;
28
•any patents issued to us may not provide a basis for commercially viable methods, applications, or products, may not provide any competitive advantages or may be successfully challenged by third parties;
•a third party may challenge our patents in court and, upon such a challenge, a court may not hold that our patents are valid, enforceable and non-infringing;
•a third party may challenge our patents in various patent offices and, if challenged, we may be compelled to limit the scope of our pending, allowed or granted claims or lose some or all of the pending, allowed or granted claims altogether;
•the patents of others could harm our business; and
•our competitors could conduct research and development activities in countries where we will not have enforceable patent rights and then use the information learned from such activities to develop competitive methods, applications, or products for sale in our major commercial markets.
While we will endeavor to protect our technology with intellectual property rights such as patents, as appropriate, the process of obtaining patents is time-consuming, expensive and sometimes unpredictable, and we may not be able to file, prosecute, maintain, enforce, or license all necessary or desirable patent applications at a reasonable cost or in a timely manner. Although we enter into non-disclosure and confidentiality agreements with parties who have access to confidential or patentable aspects of our research and development output, such as our employees, corporate collaborators, outside scientific collaborators, contract research organizations or manufacturers, consultants, advisors and other third parties, any of these parties may breach the agreements and disclose such output before a patent application is filed, thereby jeopardizing our ability to seek patent protection. Furthermore, we cannot guarantee that any patents will be issued from any of our pending or future patent applications. The standards applied by the U.S. Patent and Trademark Office (“the USPTO”) and foreign patent offices in granting patents are not always applied uniformly or predictably. For example, there is no uniform worldwide policy regarding patentable subject matter or the scope of claims allowable in biotechnology or information technology patents. Moreover, the coverage claimed in a patent application can be significantly reduced before the patent is issued, and its scope can be reinterpreted after issuance. As such, we do not know the degree of future protection that we will have on our proprietary applications, products, services, and technology. Thus, even if our patent applications issue as patents, they may not issue in a form that will provide us with meaningful protection, prevent competitors from competing with us or otherwise provide us with any competitive advantage.
Even if we have or obtain patents, we may still be barred from making, using, and selling such methods, applications, products, or services because of the patent rights of others. Others may have filed, and in the future may file, patent applications covering compositions, software applications, products, or methods that are similar or identical to ours, which could materially affect our ability to successfully develop our technology or to successfully commercialize any approved assays alone or with collaborators. Patent applications in the United States and elsewhere are generally published approximately 18 months after the earliest filing for which priority is claimed, with such earliest filing date being commonly referred to as the priority date. Therefore, patent applications could have been filed by others without our knowledge. Additionally, pending claims in patent applications which have been published can, subject to certain limitations, be later amended in a manner that could cover our platform technologies or related solutions, applications, products, and services. These patent applications may have priority over patent applications filed by us.
The issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and our owned and licensed patents may be challenged in the courts or patent offices in the United States and abroad. We may be subject to third party pre-issuance submissions of prior art to the USPTO, or become involved in opposition, derivation, revocation, reexamination, post-grant and inter partes review, or interference proceedings challenging our patent rights. An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate, our patent rights, allow third parties to commercialize our applications, products, services and technology and compete directly with us, without payment to us, or result in our inability to manufacture or commercialize applications, products, or provide services without infringing third-party patent rights. Moreover, we, or our licensors, may have to participate in interference proceedings declared by the USPTO to determine priority of invention or in post-grant challenge proceedings, such as oppositions in a
29
foreign patent office, that challenge priority of invention or other features of patentability. Such challenges may result in loss of patent rights, loss of exclusivity or freedom to operate or in patent claims being narrowed, invalidated or held unenforceable, in whole or in part, which could limit our ability to stop others from using or commercializing similar or identical applications, products, services, and technology, or limit the duration of the patent protection of our applications, products, services, and technology. Such proceedings also may result in substantial cost and require significant time from our employees and management, even if the eventual outcome is favorable to us. In addition, if the breadth or strength of protection provided by the patents and patent applications we own or in-license is threatened, it could dissuade companies from collaborating with us to license, develop or commercialize current or future technology.
In addition, third parties may be able to develop technology that is similar to, or better than, ours in a way that is not covered by the claims of our patents or may have blocking patents that could prevent us from marketing our applications and products or practicing our own patented technology. Moreover, patents have a limited lifespan. In the United States, the natural expiration of a patent is generally 20 years after it is filed and the life of a patent, and the protection it affords, is limited. Without patent protection for current or future methods and related solutions, applications, products, and services, we may face competing technology. Given the amount of time required for the development and testing, and regulatory review where necessary, patents protecting such technology might expire before or shortly after such technology is commercialized. At the same time, given the rapid pace of technological advancement and innovation in the information technology field, the time needed to obtain patents for novel information technology solutions often renders the protection, once obtained, ineffective if the protected solution has become obsolete or widely-adopted while the patent protection was pending. As a result, our patent portfolio may not provide us with sufficient rights to exclude others from commercializing technology similar or identical to that we or our collaborators may develop.
Moreover, certain of our patents and patent applications may in the future be, co-owned with third parties. If we are unable to obtain an exclusive license to any such third party co-owners’ interest in such patents or patent applications, such co-owners may be able to use or license their rights to other third parties, including our competitors, and our competitors could market competing applications, products, and technology. In addition, we may need the cooperation of any such co-owners of our patents in order to enforce such patents against third parties, and such cooperation may not be provided to us. Any of the foregoing could have a material adverse effect on our business, financial conditions, results of operations, and prospects.
We may in the future be involved in lawsuits to defend or enforce our patents and proprietary rights. Such disputes could result in substantial costs or loss of productivity, delay or prevent the development and commercialization of our technology, applications, products, and services, prohibit our use of proprietary technology or put our patents and other proprietary rights at risk.
Competitors and other third parties may infringe, misappropriate or otherwise violate our patents and intellectual property rights or the patents and intellectual property rights of our licensors. The enforcement of such claims can be expensive and time-consuming. In an infringement proceeding, a court may decide that a patent owned or in-licensed by us is invalid or unenforceable or may refuse to stop the other party from using the technology at issue on the grounds that our owned and in-licensed patents do not cover the technology in question. An adverse result in any litigation proceeding could put one or more of our owned or in-licensed patents at risk of being invalidated or interpreted narrowly. In addition, our ability to enforce our patents or other intellectual property rights depends on our ability to detect infringement. It may be difficult to detect those who infringe on our intellectual property rights who do not advertise the components or methods that are used in connection with their applications, products, and services. Moreover, it may be difficult or impossible to obtain evidence of infringement in a competitor’s or potential competitor’s application, product, or service.
If we were to initiate legal proceedings against any other third party to enforce a patent covering our technology, the defendant could assert that our patent is invalid or unenforceable. In patent litigation in the United States and Europe, defendants alleging invalidity or unenforceability are common responses to a claim of infringement. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, for example, lack of novelty, obviousness, overbreadth or lack of utility. Third parties might allege unenforceability of our patents because during prosecution of the patent, an individual connected with such prosecution withheld relevant information or made a misleading statement. Third parties may also raise challenges to the validity of our patent claims before administrative bodies in the United States or abroad, even outside the context of litigation. Such mechanisms include re-examination, post-grant review, inter partes review, interference proceedings, derivation proceedings, and equivalent proceedings in foreign jurisdictions (e.g., opposition
30
proceedings). Such proceedings could result in the revocation of, cancellation of, or amendment to, our patents in such a way that they no longer cover our technology, applications, or products. The outcome of proceedings involving assertions of invalidity and unenforceability, including during patent litigation, is unpredictable. With respect to the validity of patents, for example, we cannot be certain that there is no invalidating prior art of which we and the patent examiner were unaware during prosecution, but that an adverse third party may identify and submit in support of such assertions of invalidity. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, we would lose at least part, and perhaps all, of the patent protection on our technology. Such a loss of patent protection could have a material adverse effect on our business. Our patents and other intellectual property rights also will not protect our technology if competitors design around our protected technology without infringing our patents or other intellectual property rights.
Even if resolved in our favor, litigation or other legal proceedings relating to intellectual property claims may cause us to incur significant expenses and could distract our personnel from their normal responsibilities. There can be no assurance that we will have sufficient financial or other resources to file and pursue infringement claims, which typically last for years before they are concluded. We may or may not choose to pursue litigation or other actions against those who have infringed on our patents, or have used them without authorization, due to the associated expense and time commitment of monitoring these activities. In addition, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions, or other interim proceedings or developments, and if securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our ordinary shares. Such litigation or proceedings could substantially increase our operating losses and reduce the resources available for development activities or any future sales, marketing or commercialization activities. We may not have sufficient financial or other resources to conduct such litigation or proceedings adequately. Uncertainties resulting from patent and other intellectual property litigation or other proceedings could have a material adverse effect on our ability to compete in the marketplace, our ability to raise additional funds, and could otherwise have a material adverse effect on our business, financial condition, results of operations, and prospects.
We may in the future be subject to claims against us alleging that we are infringing, misappropriating or otherwise violating the intellectual property rights of third parties, the outcome of which would be uncertain and could have a material adverse effect on our business.
Our commercial success depends in part upon our ability to develop, manufacture, market, and sell our applications, products, and services and use our proprietary technology without infringing, misappropriating or otherwise violating the patents or other intellectual property or proprietary rights of third parties. Litigation relating to infringement, misappropriation or other violations of patents and other intellectual property rights in the biotechnology industry is common, including patent infringement lawsuits, trade secret lawsuits, interferences, oppositions, and inter partes review, post-grant review and re-examination proceedings before the USPTO, and corresponding international patent offices.
In the future, we may be subject to third-party claims and similar adversarial proceedings or litigation regarding our infringement, misappropriation, or any other violation of a third party’s patent or other intellectual property rights. If any such claim or proceeding is brought against us, our collaborators or our third-party service providers, our development, manufacturing, marketing, sales, and other commercialization activities could be similarly adversely affected. Even if we believe third-party intellectual property claims are without merit, there is no assurance that a court would find in our favor on questions of infringement, validity, enforceability or priority. A court of competent jurisdiction could hold that third-party patents asserted against us are valid, enforceable, and infringed, which could materially and adversely affect our ability to develop, manufacture, market, sell and commercialize any of our applications, products, or services. In order to successfully challenge the validity of any such U.S. patent in federal court, we would need to overcome a presumption of validity. As this burden is a high one requiring us to present clear and convincing evidence as to the invalidity of any such U.S. patent claim, there is no assurance that a court of competent jurisdiction would invalidate the claims of any such U.S. patent. If we are found to infringe any third party’s patents or other intellectual property rights, and we are unsuccessful in demonstrating that such patents or other intellectual property are invalid or unenforceable, we could be required to obtain a license from such third party to continue developing, manufacturing, marketing, selling, and commercializing our applications, products, and services. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Even if we were able to obtain a license, it could be non-exclusive, which would give our competitors and other third parties access to the same technologies licensed to
31
us, and it could require us to make substantial licensing, royalty, and other payments. We also could be forced, including by court order, to cease developing, manufacturing, marketing, selling, and commercializing the infringing application, product, or technology. In addition, we could be found liable for significant monetary damages, including treble damages and attorneys’ fees, if we are found to have willfully infringed a patent or other intellectual property right. Claims that we have misappropriated the confidential information or trade secrets of third parties could have a similar material adverse effect on our business, financial condition, results of operations, and prospects.
The various markets in which we plan to operate are subject to frequent and extensive litigation regarding patents and other intellectual property rights. It is possible that one or more organizations will hold patent rights to which we will need a license. If those organizations refuse to grant us a license to such patent rights on reasonable terms, we may be unable to develop, manufacture, market, sell, and commercialize applications, products, or services or perform research and development or other activities covered by these patents. Some claimants may have substantially greater resources than we do and may be able to sustain the costs of complex intellectual property litigation to a greater degree and for longer periods of time than we could. In addition, many companies in intellectual property-dependent industries, including the biotechnology industry, have employed intellectual property litigation as a means to gain an advantage over their competitors. Furthermore, patent holding companies that focus solely on extracting royalties and settlements by enforcing patent rights may target us. As the biotechnology industry expands and more patents are issued, and as we gain greater visibility and market exposure as a public company, the risk increases that our applications, products, services, and technology may be subject to intellectual property-related claims by third parties.
Even if resolved in our favor, litigation or other legal proceedings relating to intellectual property claims may cause us to incur significant expenses and could distract our personnel from their normal responsibilities. In addition, intellectual property litigation, regardless of its outcome, may cause negative publicity, adversely impact prospective customers, cause product shipment delays or prohibit us from manufacturing, marketing, selling or otherwise commercializing our applications, products, services, and technology. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions, or other interim proceedings or developments, and if securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our ordinary shares. Such litigation or proceedings could substantially increase our operating losses and reduce the resources available for development activities or any future sales, marketing or commercialization activities. We may not have sufficient financial or other resources to conduct such litigation or proceedings adequately. Uncertainties resulting from patent and other intellectual property litigation or other proceedings could have a material adverse effect on our ability to compete in the marketplace, our ability to raise additional funds, and could otherwise have a material adverse effect on our business, financial condition, results of operations, and prospects.
We license patent rights from third-party owners. If such owners do not properly or successfully obtain, maintain, defend and enforce the patents underlying such licenses, or if they retain or license to others any competing rights, our competitive position and business prospects may be adversely affected. If we fail to comply with our obligations in the agreements under which we license intellectual property rights from third parties or otherwise experience disruptions to our relationships with any of our licensors, we could lose license rights that are important to our business.
Even though we actively file patent applications, we also rely on intellectual property rights licensed from third parties to protect our technology, including licenses that give us rights to third-party intellectual property that is necessary or useful for our business. For example, we are dependent on licenses from Normandie Valorisation for certain products we commercialize. If one or both of our license agreements with Normandie Valorisation were to terminate for any reason, we may be required to cease the manufacturing, marketing, selling, and commercialization of certain products. For more information regarding these license agreements, please see “Item 10. Additional Information—C. Material Contracts.”
We also may license additional third-party intellectual property in the future. Our success will depend in part on the ability of our licensors to obtain, maintain, protect, and enforce patent protection for our licensed intellectual property, in particular, those patents to which we have secured exclusive rights. These licenses, and other licenses we may enter into in the future, may not provide adequate rights to use such intellectual property and proprietary technologies in all relevant fields of use or in all territories in which we may wish to develop or
32
commercialize technology, applications, products, and services in the future. In some cases, patent prosecution of our licensed technology is controlled by the licensor. Therefore, we cannot be certain that these patents and patent applications will be prepared, filed, prosecuted, and maintained in a manner consistent with the best interests of our business. For example, under our license agreements with Normandie Valorisation, Normandie Valorisation controls the prosecution, maintenance and defense of the patents licensed to us pursuant to the agreements. Our licensors may not successfully prosecute the patent applications licensed to us, by failing to draft or prosecute the patents and patent applications licensed to us in compliance with applicable laws and regulations, which may affect the validity and enforceability of such patents or any patents that may issue from such applications. Even if patents issue or are granted, our licensors may fail to maintain these patents, may determine not to pursue litigation against other companies that are infringing these patents, or may pursue litigation less aggressively than we would. Further, we may not obtain exclusive rights, which would allow for third parties to develop competing applications and products. In addition, our licensors may have relied on third-party consultants or collaborators or on funds from third parties such that our licensors are not the sole and exclusive owners of the patents we in-licensed. If our licensors fail to obtain and maintain a patent or other protection for the proprietary intellectual property we license from such licensor, we could lose our rights to such intellectual property or the exclusivity of such rights, and our competitors could market competing technology using such intellectual property. In that event, we may be required to expend significant time and resources to develop or license replacement technology. If we are unable to do so, we or our collaborators may be unable to develop or commercialize the affected technology, which could adversely affect our competitive business position and harm our business prospects.
Our existing license agreements impose, and we expect that future license agreements will impose, various development, commercialization, royalty, diligence, patent prosecution and enforcement, and other obligations on us. If we breach any of these obligations, we may be required to pay damages and the licensor may have the right to terminate the license, which could result in our being unable to commercialize related solutions, applications, products, or services that are covered by these agreements, which could materially adversely affect the value of any such technology and our business. In spite of our efforts, our licensors might conclude that we have breached our obligations under such license agreements, and might therefore terminate the license agreements. Termination of these agreements or reduction or elimination of our rights under these agreements may result in our having to negotiate new or reinstated agreements with less favorable terms, or cause us to lose our rights under these agreements, including our rights to important intellectual property or technology. In addition, while we cannot currently determine the amount of the royalty obligations we would be required to pay on sales of future applications and products, if any, the amounts may be significant. The amount of our future royalty obligations will depend on the technology and intellectual property we use in the applications, products, and services that we successfully develop and commercialize, if any. Therefore, even if we successfully develop and commercialize applications, products, and services, we may be unable to achieve or maintain profitability.
In addition, disputes may arise under our license agreements, including regarding the payment of the royalties or other payments due to licensors in connection with our exploitation of the rights we license from them. For example, licensors may contest the basis of royalties we retained and claim that we are obligated to make payments under a broader basis. In addition to the costs of any litigation we may face as a result, any legal action against us could increase our payment obligations under the respective agreement and require us to pay interest and potentially damages to such licensors.
Disputes may arise regarding intellectual property subject to a license agreement, including those relating to:
•the scope of rights, if any, granted under the license agreement and other interpretation-related issues;
•the amounts of royalties due under the license agreement;
•whether, and the extent to which, our technology and processes infringe, misappropriate or otherwise violate intellectual property of the licensor that is not subject to the license agreement;
•our diligence obligations under the license agreement and what activities satisfy those diligence obligations;
•the sublicensing of patent and other rights under the license agreements;
33
•the inventorship and ownership of inventions and know-how resulting from the creation or use of intellectual property by our licensors and by us and our collaborators; and
•the priority of invention of patented technology.
The agreements under which we currently license intellectual property or technology from third parties are complex, and certain provisions in such agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could narrow what we believe to be the scope of our rights to the relevant intellectual property or technology or increase what we believe to be our financial or other obligations under the relevant agreement. Such disputes may be costly to resolve and may divert management’s attention away from day-to-day activities. If disputes over intellectual property that we have licensed from third parties prevent or impair our ability to maintain our licensing arrangements on acceptable terms, we or our collaborators may be unable to successfully develop and commercialize the affected technology, which could have a material adverse effect on our business, financial condition, results of operations, and prospects.
We may not be successful in obtaining necessary rights to any applications, products, or services we may develop through acquisitions and in-licenses.
We currently have rights to intellectual property, through licenses from third parties, to identify and develop certain applications, products, services and technology. Many pharmaceutical companies, biotechnology companies and academic institutions are competing with us and filing patent applications potentially relevant to our business. In order to avoid infringing these third-party patents, we may find it necessary or prudent to obtain licenses from such third-party intellectual property holders.
However, we may be unable to secure such licenses or otherwise acquire or in-license any compositions, methods of use, processes or other intellectual property rights from third parties that we identify as necessary for our business. The licensing or acquisition of third-party intellectual property rights is a competitive area, and several more established companies may pursue strategies to license or acquire third-party intellectual property rights that we may consider attractive or necessary. These established companies may have a competitive advantage over us due to their size, capital resources, and greater clinical development and commercialization capabilities. In addition, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. We also may be unable to license or acquire third-party intellectual property rights on terms that would allow us to make an appropriate return on our investment or at all. If we are unable to successfully obtain rights to required third-party intellectual property rights or maintain the existing intellectual property rights we have, it could have a material adverse effect on our business, financial condition, results of operations, and prospects.
We will not seek to protect our intellectual property rights in all jurisdictions throughout the world, and we may not be able to adequately enforce our intellectual property rights even in the jurisdictions where we seek protection.
Filing, prosecuting, and defending patents in all countries and jurisdictions throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States could be less extensive than those in the United States, assuming that rights are obtained in the United States. In-licensing patents covering our technology in all countries throughout the world may similarly be prohibitively expensive, if such opportunities are available at all. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States, even in jurisdictions where we do pursue patent protection. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, even in jurisdictions where we do pursue patent protection, or from selling or importing our technology in and into the United States or other jurisdictions.
We generally apply for patents in those countries where we intend to make, have made, use or offer for sale our applications, products, or services, and where we assess the risk of infringement to justify the cost of seeking patent protection. However, we may not seek protection in all countries where we will commercialize our applications, products, and services and we may not accurately predict all the countries where patent protection would ultimately be desirable. If we fail to timely file a patent application in any such country or major market, we may be precluded from doing so at a later date. Competitors may use our technology in jurisdictions where
34
we do not pursue and obtain patent protection to develop their own assays and products and may export otherwise infringing assays and products to territories where we have patent protection, but where our ability to enforce our patent rights is not as strong as in the United States. These applications, products, and services may compete with technologies that we or our collaborators may develop, and our patents or other intellectual property rights may not be effective or sufficient to prevent such competition.
The laws of some other countries do not protect intellectual property rights to the same extent as the laws of the United States. Patent protection must ultimately be sought on a country-by-country basis, which is an expensive and time-consuming process with uncertain outcomes. Accordingly, we may choose not to seek patent protection in certain countries, and we will not have the benefit of patent protection in such countries. In addition, the legal systems of some countries, particularly developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biopharmaceuticals or biotechnologies. As a result, many companies have encountered significant difficulties in protecting and defending intellectual property rights in certain jurisdictions outside the United States. Such issues may make it difficult for us to stop the infringement of our patents, if obtained, or the misappropriation or other violation of our other intellectual property rights. For example, many other countries, including countries in the EU, have compulsory licensing laws under which a patent owner must grant licenses to third parties. In addition, many countries limit the enforceability of patents against third parties, including government agencies or government contractors. In these countries, patents may provide limited or no benefit. In those countries, we and our licensors may have limited remedies if patents are infringed or if we or our licensors are compelled to grant a license to a third party, which could materially diminish the value of those patents and could limit our potential revenue opportunities. Accordingly, our and our licensors’ efforts to enforce intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we own or license.
Furthermore, proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business, subject our patents to the risk of being invalidated or interpreted narrowly, subject our patent applications to the risk of not issuing or provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate, and the damages or other remedies awarded to us, if any, may not be commercially meaningful, while the damages and other remedies we may be ordered to pay such third parties may be significant.
If we are unable to execute invention assignment agreements with our employees and consultants or protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In addition to seeking patent protection for certain aspects of our technology, we also consider trade secrets, including confidential and unpatented know-how, important to the maintenance of our competitive position. We protect trade secrets and confidential and unpatented know-how, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to such knowledge, such as our employees, corporate collaborators, outside scientific collaborators, contract research organizations or manufacturers, consultants, advisors and other third parties. We also enter into confidentiality and invention or patent assignment agreements with our employees and consultants that obligate them to maintain confidentiality and assign their inventions to us.
We cannot guarantee that we have entered into such agreements with each party that may have or have had access to our trade secrets or proprietary technology and processes or that the assignment agreements that have been entered into are self-executing. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, or claim ownership in intellectual property that we believe is owned by us. Monitoring unauthorized uses and disclosures of our intellectual property is difficult, and we do not know whether the steps we have taken to protect our intellectual property will be effective. In addition, we may not be able to obtain adequate remedies for such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts in the United States and certain foreign jurisdictions are less willing or unwilling to protect trade secrets.
Moreover, our competitors or other third parties may independently develop knowledge, methods and know-how equivalent to our trade secrets or seek to reverse-engineer our technology for which we do not have patent protection. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor or other third parties, we would have no right to prevent them from using that technology or information to
35
compete with us. If any of our trade secrets were to be disclosed to or independently developed by a competitor, our competitive position would be harmed.
We are also subject both in the United States and outside the United States to various regulatory schemes regarding requests for the information we provide to regulatory authorities, which may include, in whole or in part, trade secrets or confidential commercial information. While we are likely to be notified in advance of any disclosure of such information and would likely object to such disclosure, there can be no assurance that our challenge to the request would be successful. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operations, and prospects.
We have in the past and may in the future be subject to claims that our employees, consultants or advisors have wrongfully used or disclosed trade secrets or other confidential information of their current or former employers or claims asserting ownership of what we regard as our own intellectual property.
Many of our employees, consultants and advisors are currently or were previously employed at universities, research institutes or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees, consultants and advisors do not use the proprietary information or know-how of others in their work for us, we have in the past and may in the future be subject to claims that we or these individuals have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such individual’s current or former employer. Litigation may be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management.
In addition, while it is our policy to require our employees and contractors who may be involved in the conception or development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who, in fact, conceives or develops intellectual property that we regard as our own. The assignment of intellectual property rights may not be self-executing, or the assignment agreements may be breached or invalid, and we may be forced to bring claims against third parties, or defend claims that they may bring against us, to determine the ownership of what we regard as our intellectual property. Such claims could have a material adverse effect on our business, financial condition, results of operations, and prospects.
If our trademarks and trade names are not adequately protected, we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
Our registered or unregistered trademarks or trade names may be challenged, infringed, circumvented, declared generic, cancelled or determined to be infringing on other marks. As a means to enforce our trademark rights and prevent infringement, we may be required to file trademark claims against third parties or initiate trademark opposition proceedings. This can be expensive, particularly for a company of our size, and time-consuming. In addition, in an infringement proceeding, a court may decide that a trademark of ours is not valid or is unenforceable, or may refuse to stop the other party from using the trademark at issue. We may not be able to protect our rights to these and other trademarks and trade names or may be forced to stop using these names, which we may need to build name recognition with potential collaborators or customers in our markets of interest.
We have certain trademark applications pending in the United States and abroad, but there can be no assurance that these applications will be allowed and not opposed. Any denial of our trademark applications or adverse ruling in any opposition proceedings could prevent us from differentiating our applications, products and/or services and maintaining consistency across our brand. Even if these applications proceed to registration, third parties may challenge our use or registration of these trademarks in the future. In the event that our trademarks are successfully challenged, we could be forced to rebrand our applications and products, which could result in loss of brand recognition and could require us to devote resources to advertising and marketing new brands. Other biotechnology companies may be using trademarks that are similar to ours, thereby impeding our ability to build brand identity and possibly leading to market confusion. In addition, they may infringe our trademarks and we may not have adequate resources to enforce our trademarks. If we attempt to enforce our trademarks and assert trademark infringement claims, a court may determine that the party against whom we have asserted trademark infringement has superior rights to the marks in question. In this
36
case, we could ultimately be forced to cease use of such trademarks. Furthermore, there could be potential trade name or trademark infringement claims brought by owners of other trademarks or trademarks that incorporate variations of our registered or unregistered trademarks or trade names. Failure to maintain our trademark registrations, or to obtain new trademark registrations in the future, could limit our ability to protect our trademarks and impede our marketing efforts in the countries in which we operate. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be adversely affected.
Our use of “open-source” software could subject our proprietary software to general release, adversely affect our ability to sell our applications and products or provide our services, and subject us to possible litigation.
A portion of the applications, products, or technologies licensed, developed or distributed by us incorporate so-called “open-source” software, and we may incorporate open-source software into other applications, products, or technologies in the future. Such open-source software is generally licensed by its authors or other third parties under open-source licenses. Some open-source licenses may contain requirements that we disclose source code for modifications we make to the open-source software and that we license such modifications to third parties at no cost. In some circumstances, distribution of our software in connection with open-source software could require that we disclose and license some or all of our proprietary code in that software as well as distribute our applications and products that use particular open-source software at no cost to the user.
We monitor our use of open-source software in an effort to avoid uses in a manner that would require us to disclose or grant licenses under our proprietary source code; however, there can be no assurance that such efforts will be successful. Open-source license terms are often ambiguous and such use could inadvertently occur. There is little legal precedent governing the interpretation of many of the terms of certain of these licenses, and the potential impact of these terms on our business may result in unanticipated obligations regarding our applications, products, and technologies.
Companies that incorporate open-source software into their applications and products have, in the past, faced claims seeking enforcement of open-source license provisions and claims asserting ownership of open-source software incorporated into their applications and products. If an author or other third party that distributes such open-source software were to allege that we had not complied with the conditions of an open-source license, we could incur significant legal costs from defending ourselves against such allegations. In the event such claims were successful, we could be subject to significant damages or be enjoined from the distribution of our applications and products. In addition, if we combine our proprietary software with open-source software in certain ways, under some open-source licenses we could be required to release the source code of our proprietary software, which could substantially help our competitors develop applications, products, and services that are similar to or better than ours and otherwise have a material adverse effect on our business, financial condition, results of operations, and prospects.
Changes in U.S. patent law could diminish the value of patents in general, thereby impairing our ability to protect our applications and products.
Changes in either the patent laws or interpretation of the patent laws in the United States could increase the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of issued patents. Changes in either the patent laws or the interpretation of the patent laws in the United States or other jurisdictions could increase the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of issued patents. Depending on future actions by the U.S. Congress, the U.S. courts, the USPTO and the relevant law-making bodies in other countries, the laws and regulations governing patents could change in unpredictable ways that could have a material adverse effect on our existing patent portfolio and our ability to protect and enforce our intellectual property in the future.
37
Risks Related to Our Financial Position and Capital Requirements
We have incurred net losses since our inception and expect to continue to incur losses for the foreseeable future. We may never achieve or sustain profitability.
We have incurred losses since our inception and expect to continue to incur losses for the foreseeable future. For the years ended December 31, 2023 and 2022, we reported net losses of $79.0 million and $87.4 million, respectively. As of December 31, 2023, we had an accumulated deficit of $377.8 million.
We expect to continue to incur net losses for the foreseeable future as we continue to devote substantial resources to (i) research and development, in particular to further expand the features, applications and data modalities of our SOPHiA DDM Platform in order to accommodate multimodal data analytics capabilities across a wide range of disease areas; (ii) expanding selling and marketing efforts for our SOPHiA DDM Platform, in particular to drive new customer adoption with clinical customers and biopharmaceutical companies; (iii) establishing and maintaining relationships with our collaborators and customers across the healthcare system; and (iv) obtaining regulatory clearance or approval to offer our applications and products as IVD applications and products for diagnostic use. We may encounter unforeseen expenses, difficulties, complications, delays and unknown factors that may adversely affect our business. The size of our future net losses will depend, in part, on the rate of growth of our expenses and of our revenue. In addition, we have incurred and expect to continue to incur increased general and administrative expenses associated with operating as a public company. Our net losses may fluctuate significantly from quarter to quarter and from year to year.
Because of the numerous risks and uncertainties associated with our research and development and commercialization efforts, although we have stated our desire to become cash flow positive in the next two or more years, we may not be able to achieve this goal in the timeframe indicated, and we may never become profitable. Even if we achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Our failure to achieve or sustain profitability would depress our market value and could impair our ability to execute our business plan, raise capital, develop additional applications, products, and services and continue our operations. A decline in the value of our company could cause our shareholders to lose all or part of their investment.
We may need to raise additional capital to fund our existing operations; further develop our SOPHiA DDM Platform, applications, and products; commercialize our applications, products, and services; and expand our operations.
Since our inception, we have used substantial amounts of cash. The research and development process as well as selling and marketing efforts are capital intensive and we expect that we will continue to expend substantial resources for the foreseeable future to develop, commercialize and market additional features, applications and data modalities of our SOPHiA DDM Platform and related solutions, applications, products, and services. In addition, we may also raise capital to expand our business and pursue strategic investments, to take advantage of financing opportunities or for other reasons. We do not have any committed external source of funds and additional funds may not be available when we need them or on terms that are acceptable to us. Our ability to raise additional funds will depend on financial, economic and market conditions and other factors, over which we may have no or limited control. Further, as a Swiss company, we have less flexibility to raise capital, particularly in a quick and efficient manner, as compared to U.S. companies. See “—Risks Related to Our Securities—Our shareholders enjoy certain rights that may limit our flexibility to raise capital, issue dividends and otherwise manage ongoing capital needs.” If adequate funds are not available to us on a timely basis or on terms acceptable to us, we may be required to delay, limit, reduce or terminate our research and development, commercialization and growth efforts.
We may seek additional capital through a variety of means, including through public and private equity offerings and debt financings, credit and loan facilities and collaborations. If we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms of such equity or convertible debt securities may include liquidation or other preferences that are senior to or otherwise adversely affect your rights as a shareholder. If we raise additional capital through the sale of debt securities or through entering into credit or loan facilities, we may be restricted in our ability to take certain actions, such as incurring additional debt, making capital expenditures, acquiring or licensing intellectual property rights, declaring dividends or encumbering our assets to secure future indebtedness. Such restrictions could adversely impact
38
our ability to conduct our operations and execute our business plan. If we raise additional capital through collaborations with third parties, we may be required to relinquish valuable rights to our intellectual property, technology, applications, and products or we may be required to grant licenses for our intellectual property, technology, applications, and products on unfavorable terms.
Our operating results may fluctuate significantly or may fall below the expectations of investors or securities analysts, each of which may cause the price of our ordinary shares to fluctuate or decline.
Our quarterly and annual operating results may fluctuate significantly. This fluctuation may be as a result of a variety of factors, many of which are outside our control and, as a result, may not fully reflect the underlying performance of our business. The cumulative effects of these factors could result in large fluctuations and unpredictability in our quarterly and annual financial results. Because a substantial portion of our expenses are relatively fixed in the short-term and require time to adjust, our results of operations and liquidity would suffer if revenue falls below our expectations in a particular period. If our operating results fall below the expectations of investors or securities analysts, the price of our ordinary shares could decline substantially. Furthermore, any fluctuations in our operating results may, in turn, cause the price of our ordinary shares to fluctuate substantially. We believe that quarterly comparisons of our financial results are not necessarily meaningful and should not be relied upon as an indication of our future performance.
We may not be able to sufficiently reduce our costs to achieve sustainable gross margins.
Operating our business is costly, and we expect our expenses to continue to increase in the future as we broaden our customer base and expand our platform, applications, services, and product offerings. In particular, a significant portion of our business, including our SOPHiA DDM Platform, is provided through a cloud-native software platform and computational and storage-related costs and fees constitute a significant portion of our cost of revenue. While we seek to negotiate favorable economic arrangements with respect to computational and storage-related costs and fees, in the near term, we expect that our gross profit margin will be adversely impacted by such fees and costs as we have purchased, and may be required to continue to purchase, increased capacity at less favorable rates in order to address increased demand for our SOPHiA DDM Platform and related solutions, applications, products and services. In addition, we collaborate with manufacturers in the assembly and development of our research and diagnostic products, in particular DNA enrichment kits. While we are undertaking a number of initiatives designed to reduce our costs, including provisions in our manufacturing and supply agreements that limit our counterparty’s ability to increase prices for the manufactured products if certain conditions are met by us, and expect that our gross margin will increase as we broaden our customer base and increase customer engagement, there can be no assurance that we will be able to achieve planned cost reductions. There may also be unforeseen occurrences that increase our costs, such as increased prices of the components of our products, increased costs of hosting and consumer support services, changes to labor costs or less favorable terms with third-party suppliers, service providers or manufacturing collaborators. In addition, if our platform, applications, services and product mix becomes more customer-specific and diversified, our costs may increase. If we are unable to reduce our costs, or if cost reductions are less significant or less timely than those we project, we will not be able to achieve sustainable gross margins, which would adversely affect our ability to invest in and grow our business.
We customize a substantial portion of our research and diagnostic products to address the needs of individual customers and collaborators. If we cannot sell our customized products in the event an order is cancelled, we may be unable to cover our costs and may be left with substantial unsaleable inventory, which could have a material adverse effect on our financial condition and results of operations.
We assemble a substantial portion of our products to address the needs of individual customers. Some of the agreements with our customers require us to cover the initial manufacturing and assembly costs of such products. We will be paid only upon delivery of such products to our customers once the products have been ordered. If our customers fail to purchase these customized products from us in sufficient quantities, do not purchase such products from us at all or otherwise fail to perform their obligations under the agreements with us, we may bear the full cost of manufacturing and assembling of such products, fail to cover our costs and have substantial unsaleable inventory, each of which could have a material adverse effect on our financial condition and results of operations.
39
Our ability to use tax loss carryforwards in Switzerland, the United States and other jurisdictions may be limited.
We are entitled to carry forward losses incurred in Switzerland, the United States and other jurisdictions in which we conduct business, which could be used to offset future taxable income. Due to our limited income, there is a significant risk that our tax loss carryforwards will expire in part or in their entirety and cannot be used to offset future taxable income for corporate income tax purposes. Furthermore, any tax loss carryforwards that we report on our tax returns are subject to review and confirmation by the competent tax authorities in their tax assessment of the tax year for which the tax loss carryforwards are used to offset taxable income. Consequently, we are exposed to the risk that the competent tax authorities may not accept the reported tax loss carryforwards in part or in their entirety.
We are subject to risks related to taxation in multiple jurisdictions.
We are subject to income taxes in Switzerland and foreign jurisdictions. Significant judgments based on interpretations of existing tax laws or regulations may be required in determining our provision for income taxes. Our effective income tax rate could be adversely affected by various factors, including, but not limited to, changes in the combination of earnings in tax jurisdictions with different statutory tax rates, changes in the valuation of deferred tax assets and liabilities, changes in existing tax policies, laws, regulations or rates, changes in the level of non-deductible expenses (including share-based compensation), changes in the location of our operations, changes in our future levels of research and development spending, mergers and acquisitions or the result of examinations by various tax authorities. Although we believe our tax estimates are reasonable, if taxing authorities disagree with the positions taken on our tax returns, we could have additional tax liability, including interest and penalties.
Exchange rate fluctuations have in the past materially affected and may in the future materially affect our results of operations and financial condition.
We operate internationally and a meaningful portion of our revenue, expenses, assets and liabilities are denominated in currencies other than the U.S. dollar, our presentation currency, and the Swiss franc, SOPHiA GENETICS SA’s functional currency. In preparing our consolidated financial statements, those revenues, expenses, assets and liabilities are translated into U.S. dollars at applicable exchange rates. Increases or decreases in exchange rates between the U.S. dollar and other currencies affect the U.S. dollar value of those items, as reflected in the consolidated financial statements. We expect that a significant part of our revenues and expenses will continue to be denominated in currencies other than the U.S. dollar, including the euro and Swiss franc, and to a lesser extent, British pound, Australian dollar, Brazilian real, Turkish lira and Canadian dollar. Therefore, unfavorable developments in the value of the U.S. dollar relative to other relevant currencies could adversely affect our results of operations, financial condition and liquidity. For example, fluctuations in the exchange rate between the U.S. dollar and the Swiss franc has impacted our revenues and results of operations for the year ended December 31, 2023 and 2022.
The exchange rates of the U.S. dollar and other currencies are affected by many factors, including forces of supply and demand in the foreign exchange markets and global economic events. These rates are also affected by the international balance of payments and other economic and financial conditions, government intervention, speculation and other factors. We currently do not engage in hedging transactions to protect against uncertainty in future exchange rates between particular foreign currencies and the U.S. dollar and even if we engage in hedging operations in the future, there can be no assurance as to the success of any hedging operations that we may implement. Foreign currency fluctuations may adversely affect our results of operations, financial condition and liquidity.
We are subject to risks related to the accounting treatment of our pension and other post-employment benefit plans.
We provide retirement benefits to our employees as required by Swiss law by means of a pension fund that is maintained by a life insurance company. The life insurance company operates a pension plan for all of our employees as a defined benefit plan under International Accounting Standards (“IAS”) 19, Employee Benefits (“IAS 19”). As of December 31, 2023, we reported an employee benefit obligation, before deduction of plan assets, of $23.0 million in accordance with IAS 19. The obligation represents our projected obligations towards current and future pensioners discounted at an annual rate of 1.50%. Under Swiss statutory rules and
40
pursuant to our contract with the group life assurance provider, all risks including investment risk are fully covered. That said, no underfunding exists under Swiss law. The variance between Swiss statutory rules and IFRS Accounting Standards is apparent in many Swiss companies, and the IFRS Accounting Standards obligation of our pension plan does not necessarily reflect a true payment obligation under Swiss law because Swiss law allows us to maintain flexibility to adjust benefit levels under the plans and we could use this flexibility to mitigate any liability. For more information, see Note 22—“Post-employment benefits” to the audited consolidated financial statements included elsewhere in this Annual Report. However, should the Swiss statutory rules at any time require a determination that our pension plan is significantly underfunded, we could be obliged to make additional contributions to the pension plan in addition to our obligation to make regular contributions as defined in the pension plan regulation. If such risk materializes, this could have a material adverse effect on our financial position or results of operations.
We have elected to reserve capital under our directors and officers insurance policy to reduce the associated premiums. Our use of such designated capital for other purposes could increase our premiums.
We currently hold $15.0 million in a separate bank account to be used exclusively to settle potential liabilities arising from claims against our directors and officers. Our use of such a designated account reduces our directors and officers insurance premiums below those that we would pay absent such a designated account. Therefore, our current insurance premiums may not reflect those that we will incur in the future. Although we expect to continue to hold cash in a separate bank account for such purposes, we are under no obligation to do so and may withdraw the funds at any time, in which case we expect our insurance premiums to increase significantly. In addition, our practice diverts capital that can be used for other purposes, and there can be no assurance that the benefits of our practice (in the form of lower insurance premiums) outweigh the costs of such practice (in the form of benefits foregone by not deploying the reserved capital for other purposes). Moreover, our insurance provider may require additional capital to be reserved in the future in order to maintain our insurance premiums at current levels or reduce the rate of increases in our insurance premiums, and we may be unable to meet such requirements or we may find it disadvantageous to do so, which would increase our insurance premiums.
Risks Related to Our Securities
The market price of our ordinary shares has historically been, and in the future may continue to be, volatile and may fluctuate due to factors beyond our control.
In the past, we have experienced significant fluctuations in the price of our ordinary shares. The market price of our ordinary shares could be subject to wide fluctuations in response to many risk factors listed in this “Risk Factors” section, some of which are beyond our control, including:
•actual or anticipated fluctuations in our financial condition and operating results;
•effectiveness, accuracy and efficiency of our SOPHiA DDM Platform and related solutions, applications, products, and services;
•public concern relating to the commercial value or safety of any of our SOPHiA DDM Platform and related solutions, applications, products, and services;
•the timing and results of multimodal clinical studies of our SOPHiA DDM Platform;
•our inability to adequately protect our proprietary and intellectual property rights, including patents, trademarks and trade secrets;
•our inability to raise additional capital and the terms on which we raise it;
•our ability to enter into strategic collaboration or licensing agreements and the commencement, termination and terms of such agreements;
•regulatory developments, including actions with respect to our and our competitors’ platforms, applications, products, and services;
41
•publication of research reports by securities analysts about us or our competitors or our industry;
•our failure or the failure of our competitors to meet analysts’ projections or guidance that we or our competitors may give to the market;
•additions and departures of key personnel;
•the passage of legislation or other regulatory developments affecting us or our industry;
•fluctuations in the valuation of companies perceived by investors to be comparable to us;
•sales of our ordinary shares by us, our insiders or our other shareholders;
•changes in market conditions for our industry, including changes in the structure of healthcare payment systems; and
•changes in general market and economic conditions.
In addition, the stock market has historically experienced significant volatility, particularly with respect to healthcare technology company stocks. As we operate in a single industry, we are particularly vulnerable to these factors to the extent that they affect our industry. In the past, securities class action litigation has often been initiated against companies following periods of volatility in their stock price. This risk is particularly relevant for healthcare technology companies, which have experienced significant stock price volatility in recent years. Securities litigation could result in substantial costs and divert our management’s attention and resources and could also require us to make substantial payments to satisfy judgments or to settle litigation.
Future sales, or the possibility of future sales, of a substantial number of our ordinary shares could adversely affect the price of our ordinary shares.
Future sales of a substantial number of our ordinary shares, or the perception that such sales will occur, could cause a decline in the market price of our ordinary shares. Ordinary shares that were issued prior to our initial public offering can be sold pursuant to Rule 144 under the Securities Act, subject to current public information and volume and manner of sale limitations applicable to affiliates. In addition, we have registered under the Securities Act all ordinary shares that we may issue under our share-based compensation plans, such that they can be freely sold in the public market upon issuance, subject to volume limitations applicable to affiliates. We intend to adopt a sell-to-cover approach for withholding and other taxes upon RSU settlement for executive officers and certain directors, which may result in substantial sales of ordinary shares into the market in relatively short periods of time, particularly around the date of our annual general meeting. If our shareholders sell substantial numbers of ordinary shares in the public market or if the market perceives that such sales may occur, the market price of our ordinary shares and our ability to raise capital through the issuance and sale of equity or equity-linked securities in the future could be adversely affected.
Under Swiss law, shareholders benefit from pre-emptive rights to subscribe on a pro rata basis for issuances of equity or other securities that are convertible into equity, unless such pre-emptive rights are excluded in accordance with Swiss law and our articles of association. However, due to the laws and regulations in certain jurisdictions, shareholders in certain jurisdictions may not be able to exercise such rights, unless the company registers or otherwise qualifies the rights offering, including by complying with Annual Report requirements under the laws of that jurisdiction. There can be no assurance that we will take any action to register or otherwise qualify an offering of subscription rights or shares under the laws of any jurisdiction where the offering of such rights is restricted, other than the United States. If shareholders in such jurisdictions are unable to exercise their subscription rights, their ownership interest will be diluted.
We have never paid dividends and do not expect to pay any dividends in the foreseeable future.
Since inception, we have not paid any dividends. Even if future operations lead to significant levels of distributable profits, we currently intend to reinvest any earnings in our business and do not anticipate declaring or paying any dividends until we have an established revenue stream to support dividend payments. In addition, any proposal for the payment of future dividends will be at the discretion of our board of directors after taking into account various factors including our business prospects, liquidity requirements, financial performance and
42
new application and product development. Furthermore, payment of future dividends is subject to certain limitations pursuant to our current and future debt instruments, Swiss law and our articles of association. See “Item 10. Additional Information—Memorandum and Articles of Association.” Accordingly, investors cannot rely on dividend income from our ordinary shares, and any returns on an investment in our ordinary shares will likely depend entirely upon any future appreciation in the price of our ordinary shares.
If securities or industry analysts publish inaccurate or unfavorable research about our business or case to publish research about our business, the price of our ordinary shares and our trading volume could decline.
The trading market for our ordinary shares depends, in part, on the research and reports that securities or industry analysts publish about us or our business. If one or more of the analysts who cover us downgrade our ordinary shares or publish inaccurate or unfavorable research about us or our business, the price of our ordinary shares will likely decline. In addition, if our operating results fail to meet the forecast of analysts, the price of our ordinary shares will likely decline. If one or more of these analysts cease coverage of our company or fail to publish reports on us regularly, demand for our ordinary shares could decrease, which might cause the price of our ordinary shares and trading volume to decline.
The rights of our shareholders may be different from the rights of shareholders in companies governed by the laws of U.S. jurisdictions.
We are a Swiss corporation. Our corporate affairs are governed by our articles of association and by the laws governing companies, including listed companies, incorporated in Switzerland. The rights of our shareholders and the responsibilities of members of our board of directors may be different from the rights and obligations of shareholders and directors of companies governed by the laws of U.S. jurisdictions.
In the performance of its duties, our board of directors is required by Swiss law to consider the interests of our company, our shareholders, our employees and other stakeholders, in all cases with due observation of the principles of reasonableness and fairness. It is possible that some of these parties will have interests that are different from, or in addition to, shareholders’ interests. Swiss law limits the ability of our shareholders to challenge resolutions made or other actions taken by our board of directors in court. Our shareholders generally are not permitted to file a suit to reverse a decision or an action taken by our board of directors, but are instead only permitted to seek damages for breaches of fiduciary duty. As a matter of Swiss law, shareholder claims against a member of our board of directors for breach of fiduciary duty would have to be brought to the competent courts in Switzerland, or where the relevant member of our board of directors is domiciled. In addition, under Swiss law, any claims by our shareholders against us must be brought exclusively to the competent courts in Switzerland. For a further summary of applicable Swiss company law contained in this Annual Report, see “Item 10. Additional Information—Memorandum and Articles of Association.” However, there can be no assurance that Swiss law will not change in the future, which could adversely affect the rights of our shareholders, or that Swiss law will protect our shareholders in a similar fashion as under U.S. corporate law principles.
Our shareholders enjoy certain rights that may limit our flexibility to raise capital, issue dividends and otherwise manage ongoing capital needs.
Swiss law reserves for approval by shareholders certain corporate actions over which a board of directors would have authority in some other jurisdictions. For example, the payment of dividends and cancellation of treasury shares must be approved by shareholders. Swiss law also requires that our shareholders themselves resolve to, or authorize our board of directors to, increase our share capital. While our shareholders may authorize share capital that can be issued by our board of directors without additional shareholder approval, Swiss law limits this authorization to 50% of the issued share capital at the time of the authorization. The authorization, furthermore, has a limited duration of up to five years and must be renewed by the shareholders from time to time thereafter in order to be available for raising capital. Additionally, subject to specified exceptions, including exceptions explicitly described in our articles of association, Swiss law grants pre-emptive subscription rights to existing shareholders to subscribe for new issuances of shares. Swiss law also does not provide as much flexibility in the various rights and regulations that can attach to different categories of shares as do the laws of some other jurisdictions. These Swiss law requirements relating to our capital management may limit our flexibility, and situations may arise where greater flexibility would have provided benefits to our shareholders. See “Item 10. Additional Information—Memorandum and Articles of Association.”
43
Our ordinary shares are not listed in Switzerland, our home jurisdiction. As a result, our shareholders will not benefit from certain provisions of Swiss law that are designed to protect shareholders in a public takeover offer or a change-of-control transaction.
Because our ordinary shares will be listed exclusively on Nasdaq and not in Switzerland, our shareholders will not benefit from the protection afforded by certain provisions of Swiss law that are designed to protect shareholders in the event of a public takeover offer or a change-of-control transaction. For example, Article 120 of the Swiss Financial Market Infrastructure Act and its implementing provisions require investors to disclose their interest in our company if they reach, exceed or fall below certain ownership thresholds. Similarly, the Swiss takeover regime imposes a duty on any person or group of persons who acquires more than one-third of a company’s voting rights to make a mandatory offer for all of the company’s outstanding listed equity securities. In addition, the Swiss takeover regime imposes certain restrictions and obligations on bidders in a voluntary public takeover offer that are designed to protect shareholders. However, these protections are applicable only to issuers that list their equity securities in Switzerland, and because our ordinary shares will be listed exclusively on Nasdaq, they will not be applicable to us. Furthermore, since Swiss law restricts our ability to implement rights plans or U.S.-style “poison pills,” our ability to resist an unsolicited takeover attempt or to protect minority shareholders in the event of a change-of-control transaction may be limited. Therefore, our shareholders may not be protected in the same degree in a public takeover offer or a change-of-control transaction as are shareholders in a Swiss company listed in Switzerland.
U.S. shareholders may not be able to obtain judgments or enforce civil liabilities against us or our executive officers or members of our board of directors.
We are organized under the laws of Switzerland and our registered office and domicile is located in Rolle, Canton of Vaud, Switzerland. Moreover, a number of our directors and executive officers are not residents of the United States, and all or a substantial portion of the assets of such persons are located outside the United States. As a result, it may not be possible for investors to effect service of process within the United States upon us or upon such persons or to enforce against them judgments obtained in U.S. courts, including judgments in actions predicated upon the civil liability provisions of the federal securities laws of the United States. We have been advised by our Swiss counsel that there is doubt as to the enforceability in Switzerland of original actions, or in actions for enforcement of judgments of U.S. courts, of civil liabilities to the extent solely predicated upon the U.S. federal and state securities laws. Original actions against persons in Switzerland based solely upon the federal or state securities laws are governed, among other things, by the principles set forth in the Swiss Federal Act on Private International Law (the “PILA”). This statute provides that the application of provisions of non-Swiss law by the courts in Switzerland shall be precluded if the result is incompatible with Swiss public policy (ordre public). Also, certain mandatory provisions of Swiss law may be applicable regardless of any other law that would otherwise apply.
Switzerland and the United States do not have a treaty providing for reciprocal recognition and enforcement of judgments in civil and commercial matters. The recognition and enforcement of a judgment of the courts of the United States in Switzerland is governed by the principles set forth in the PILA. This statute provides in principle that a judgment rendered by a non-Swiss court may be enforced in Switzerland only if:
•the non-Swiss court had jurisdiction pursuant to the PILA;
•the judgment of such non-Swiss court has become final and non-appealable;
•the judgment does not contravene Swiss public policy;
•the court procedures and the service of documents leading to the judgment were in accordance with the due process of law; and
•no proceeding involving the same parties and the same subject matter was first brought in Switzerland, or adjudicated in Switzerland, or was earlier adjudicated in a third state, and this decision is recognizable in Switzerland.
44
Anti-takeover provisions in our articles of association could make an acquisition of us, which may be beneficial to our shareholders, more difficult.
Our articles of association contain provisions that may have the effect of discouraging, delaying or preventing a change in control of us that shareholders may consider favorable, including transactions in which our shareholders may receive a premium for their shares. Our articles of association include provisions that:
•in certain cases, allow our board of directors to place up to 33,199,082 ordinary shares and rights to acquire an additional 33,199,082 ordinary shares (in aggregate, 51% of our share capital) with affiliates or third parties, without existing shareholders having statutory pre-emptive rights in relation to this share placement;
•allow our board of directors not to record any acquirer of ordinary shares, or several acquirers acting in concert, in our share register as a shareholder with voting rights with respect to more than 15% of our share capital as set forth in the commercial register;
•limit the exercise of voting rights by shareholders, acting alone or in concert with others, to a maximum of 15% of the share capital recorded in the commercial register;
•limit the size of our board of directors to eight members; and
•require two-thirds of the votes represented at a general meeting of the shareholders for amending or repealing most of the above-mentioned authorizations to place shares as well as the above-mentioned voting and recording restrictions, for amending the provision setting a maximum board size or providing for indemnification of our directors and members of our executive committee and for removing the chairman or any member of the board of directors before the end of his or her term of office.
These and other provisions, alone or together, could delay or prevent takeovers and changes in control. See “Item 10. Additional Information—Memorandum and Articles of Association.” These provisions could also limit the price that investors might be willing to pay in the future for our ordinary shares, thereby depressing the market price of our ordinary shares.
There can be no assurance that we were not a passive foreign investment company for any taxable year, which could subject U.S investors in our ordinary shares to significant adverse U.S. federal income tax consequences.
Under the Internal Revenue Code of 1986, as amended (the “Code”), we will be a passive foreign investment company (“PFIC”), for any taxable year in which, after the application of certain look-through rules with respect to subsidiaries, either (i) 75% or more of our gross income consists of “passive income” or (ii) 50% or more of the average quarterly value of our assets consists of assets that produce, or are held for the production of, “passive income” (including cash). Passive income generally includes dividends, interest, certain non-active rents and royalties, and capital gains.
Cash is generally characterized as a passive asset for these purposes. Goodwill is generally characterized as a non-passive or passive asset based on the nature of the income produced in the activity to which the goodwill is attributable. The extent to which our goodwill should be characterized as a non-passive asset is not entirely clear. We hold a substantial amount of cash, and while this continues to be the case our PFIC status for any taxable year will depend largely on the value of our goodwill and the characterization of our goodwill as passive or non-passive. The value of our goodwill for any taxable year may be determined in large part by reference to the average of our market capitalization for that year. Although we believe that we were a PFIC for our 2022 taxable year, based on the nature of our operations and assets, as well as certain estimates as to our income and the relative values of our assets during our 2023 taxable year, we believe that we were not a PFIC for our 2023 taxable year (subject to the discussion in the next paragraph with respect to U.S. Holders who held our ordinary shares during our 2022 taxable year). There can be no assurance that the IRS will agree with our conclusion, however.
If we are a PFIC for any taxable year during which a U.S. investor holds ordinary shares, we generally will continue to be treated as a PFIC with respect to that U.S. investor for all succeeding years during which the U.S. investor holds ordinary shares, even if we cease to meet the threshold requirements for PFIC status.
45
Accordingly, since we believe that we were a PFIC for our 2022 taxable year, we believe that we will remain a PFIC with respect to a U.S. Holder that held our ordinary shares during our 2022 taxable year unless certain mitigating steps were taken. For further discussion, see “Item 10. Additional Information—. Taxation—Material U.S. Federal Income Tax Consequences for U.S. Holders.”
If we are deemed to be a PFIC with respect to a particular U.S. investor, such U.S. investor may be subject to adverse U.S. federal income tax consequences, including (i) the treatment of all or a portion of any gain on disposition as ordinary income; (ii) the application of a deferred interest charge on such gain and the receipt of certain dividends; and (iii) compliance with certain reporting requirements. A “mark-to-market” or “qualified electing fund” (“QEF”) election may be available that will alter the consequences of PFIC status if our ordinary shares are regularly traded on a qualified exchange. For further discussion, see “Item 10. Additional Information—E. Taxation—Material U.S. Federal Income Tax Consequences for U.S. Holders.”
Risks Related to Our Status Under U.S. Securities Laws
We are a foreign private issuer, and, as a result, we are not subject to certain rules and obligations that are applicable to a U.S. domestic public company and are not subject to certain Nasdaq corporate governance listing standards that are applicable to a Nasdaq-listed U.S. domestic public company.
We report under the Exchange Act as a non-U.S. company with foreign private issuer status. Because we qualify as a foreign private issuer under the Exchange Act and although we intend to furnish quarterly financial information to the SEC, we are exempt from certain provisions of the Exchange Act that are applicable to U.S. domestic public companies, including (i) the sections of the Exchange Act regulating the solicitation of proxies, consents or authorizations in respect of a security registered under the Exchange Act; (ii) the sections of the Exchange Act requiring insiders to file public reports of their stock ownership and trading activities, and liability for insiders who profit from trades made in a short period of time; and (iii) the rules under the Exchange Act requiring the filing with the SEC of quarterly reports on Form 10-Q containing unaudited financial and other specified information, or current reports on Form 8-K upon the occurrence of specified significant events. In addition, foreign private issuers are not required to file their annual report on Form 20-F until four months after the end of each financial year, while U.S. domestic issuers are required to file their annual report on Form 10-K in less time. Foreign private issuers are also exempt from the Regulation Fair Disclosure, aimed at preventing issuers from making selective disclosures of material information.
Furthermore, because we are a foreign private issuer, we comply with our home country governance requirements and certain exemptions thereunder, rather than complying with certain of the Nasdaq corporate governance listing standards that are applicable to U.S. companies listed on Nasdaq. For example, we are exempt from Nasdaq listing standards that require a listed U.S. company to have (i) a majority of the board of directors consist of independent directors, (ii) regularly scheduled executive sessions with only independent directors and (iii) a compensation committee and a nomination and corporate governance committee consisting entirely of independent directors. Furthermore, Nasdaq listing standards generally require Nasdaq-listed U.S. companies to, among other things, seek shareholder approval for the implementation of certain equity compensation plans and issuances of securities, which we are not required to follow as a foreign private issuer. Accordingly, our shareholders may not have the same protections afforded to shareholders of companies that are not foreign private issuers. For an overview of the material differences between our governance principles and Nasdaq corporate governance listing standards, see “Item 16G—Corporate Governance.”
We may lose our foreign private issuer status, which would then require us to comply with the Exchange Act’s domestic reporting regime and cause us to incur significant legal, accounting and other expenses.
We qualify as a foreign private issuer, and therefore we are not required to comply with all of the periodic disclosure and current reporting requirements of the Exchange Act applicable to U.S. domestic issuers. We may no longer be a foreign private issuer as of June 30, 2024, which would require us to comply with all of the periodic disclosure and current reporting requirements of the Exchange Act applicable to U.S. domestic issuers, as of January 1, 2025. In order to maintain our current status as a foreign private issuer, either (a) a majority of our ordinary shares must be either directly or indirectly owned of record by non-residents of the United States or (b)(i) a majority of our executive officers or directors may not be United States citizens or residents, (ii) more than 50% of our assets cannot be located in the United States and (iii) our business must be administered principally outside the United States. If we lose this status, we will be required to comply with the Exchange
46
Act reporting and other requirements applicable to U.S. domestic issuers, which are more detailed and extensive than the requirements for foreign private issuers. We will also be required to make changes in our corporate governance practices in accordance with various SEC and stock exchange rules. The regulatory and compliance costs to us under U.S. securities laws, if we are required to comply with the reporting requirements applicable to a U.S. domestic issuer, may be significantly higher than the cost we would incur as a foreign private issuer. As a result, we expect that a loss of foreign private issuer status would increase our legal and financial compliance costs and would make some activities highly time-consuming and costly. We also expect that if we were required to comply with the rules and regulations applicable to U.S. domestic issuers, it would be more difficult and expensive for us to obtain director and officer liability insurance, and we may be required to accept reduced coverage or incur substantially higher costs to obtain coverage. These rules and regulations could also make it more difficult for us to retain and attract qualified members of our board of directors.
We are an emerging growth company, and the reduced reporting requirements applicable to emerging growth companies may make our ordinary shares less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act. For as long as we continue to be an emerging growth company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies, including, but not limited to, (i) not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, (ii) reduced disclosure obligations regarding executive compensation in this Annual Report and our periodic reports and proxy statements and (iii) exemptions from the requirements of holding a non-binding advisory vote on executive compensation.
We could be an emerging growth company for up to five years from our initial public offering, although circumstances could cause us to lose that status earlier, including if the market value of our ordinary shares held by non-affiliates equals or exceeds $700.0 million as of any June 30 (the end of our second fiscal quarter) before that time or if we have total annual gross revenues of $1.235 billion or more during any fiscal year before that time, in which cases we would no longer be an emerging growth company as of the following December 31 (our fiscal year end); or, if we issue more than $1.0 billion in non-convertible debt during any three-year period before that time, we would cease to be an emerging growth company immediately. Even after we no longer qualify as an emerging growth company, if we no longer qualify as a foreign private issuer, we may still qualify as a “smaller reporting company,” which, under certain circumstances, would allow us to take advantage of many of the same exemptions from disclosure requirements, including not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act and reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements. We cannot predict if investors will find our ordinary shares less attractive because we may rely on these exemptions. If some investors find our ordinary shares less attractive as a result, there may be a less active trading market for our ordinary shares and the price of our ordinary shares may be more volatile. When these exemptions cease to apply, we expect to incur additional expenses and devote increased management effort towards ensuring compliance with them, and we cannot predict or estimate the amount or timing of such additional costs.
47
Item 4. Information on the Company
A. History and Development of the Company
SOPHiA GENETICS SA was incorporated as a Swiss stock corporation (société anonyme) under the laws of Switzerland on March 18, 2011. Our principal executive office is located at La Pièce 12, CH-1180 Rolle, Switzerland and our telephone number is +41 21 694 10 60. Our agent for service of process in the United States is SOPHiA GENETICS, Inc., 185 Dartmouth Street, Floor 5, Boston, MA 02116, and its telephone number is (617) 982-1210.
Our website is www.sophiagenetics.com. The reference to our website is an inactive textual reference only and information contained therein or connected thereto are not incorporated into this Annual Report. We file reports and other information with the SEC, including annual reports on Form 20-F and reports on Form 6-K. The SEC maintains an Internet site at www.sec.gov that contains reports, proxy and information statements and other information we have filed electronically with the SEC.
In July 2021, we completed our initial public offering on the Nasdaq Global Select Market (“Nasdaq”) and our ordinary shares are listed under the ticker symbol “SOPH”.
B. Business Overview
Our Mission
SOPHiA GENETICS was founded to generate clinically actionable insights from data to improve patient outcomes. Our mission is to provide equal access to knowledge and capabilities by democratizing data-driven medicine.
We observed that across the healthcare ecosystem, a vast amount of digital healthcare data was being generated, fueled by technologies such as NGS, and which held promise to accelerate the understanding of biology and disease. However, this data has been generated primarily using non-standardized methods and by clinicians and researchers across many healthcare institutions. As a result, the data remained siloed and complex and was not fully leveraged for the benefit of patients.
We founded SOPHiA GENETICS to address this issue. We are unlocking data silos, leveraging AI to generate actionable insights from data and helping healthcare professionals work together as a community and deploy their collective expertise for the benefit of patients around the world.
We refer to data-driven medicine as the practice of drawing insights from complex data sets to improve diagnosis, treatment and drug development. Using data-driven medicine, healthcare professionals supplement their own experience and intuition with data insights and shared knowledge from their peers to inform the best course of action for their patients or research. Our goal is to empower clinicians and researchers around the world to practice data-driven medicine and improve clinical and scientific outcomes.
Overview
We are a cloud-native software technology company in the healthcare space dedicated to establishing the practice of data-driven medicine as the standard of care and for life sciences research. We purposefully built a cloud-native software platform capable of analyzing data and generating insights from complex multimodal data sets and different diagnostic modalities. Our platform standardizes, computes and analyzes digital health data and is used across decentralized locations to break down data silos. This enables healthcare institutions to share knowledge and experiences and to build a collective intelligence. We envision a future in which all clinical diagnostic test data is channeled through a decentralized analytics platform that will provide insights powered by large real-world data sets and AI. We believe that a decentralized platform is the most powerful and effective solution to create the largest network, leverage data and bring the benefits of data-driven medicine to customers and patients globally. In doing so, we can both support and benefit from growth across the healthcare ecosystem.
In 2014, we launched the first application of our platform to analyze NGS data for cancer diagnosis. We offer a broad range of applications used by healthcare providers, clinical and life sciences research laboratories and biopharmaceutical companies for precision medicine across oncology, rare diseases, infectious diseases,
48
cardiology, neurology, metabolism and other disease areas. In 2019, we launched our solution for radiomics data that enables longitudinal monitoring of cancer patients and tumor progression throughout their disease journey. In 2022, we unveiled SOPHiA CarePath, a new multimodal module on our SOPHiA DDM Platform powered by our artificial intelligence and machine learning algorithms, which also incorporates our original radiomics offering. The module will allow healthcare practitioners to visualize data across multiple modalities (including genomic, radiomic, clinical, and biological) for individual patients in a longitudinal manner and derive additional insights through cohort design and comparison. SOPHiA CarePath has already been deployed as part of our Deep-Lung IV multimodal clinical study on non-small cell lung cancer and is now live at 30 sites across the world.
Today, we believe that our SOPHiA DDM Platform, commercialized under the name “SOPHiA DDM,” is one of the most widely used decentralized analytics platform globally for clinical genomics. As of December 31, 2023, we served more than 770 hospital, laboratory and biopharma customers globally through our SOPHiA DDM Platform and related solutions, applications, products and services, and our SOPHiA DDM Platform has supported the analysis of more than 1,500,000 genomic profiles and has been utilized in clinical trials and research projects discussed in more than 617 peer-reviewed publications. As of December 31, 2023, we had approximately 450 core genomics SOPHiA DDM Platform customers (defined as the number of customers who generated revenue through usage of our bundle access, dry lab, and integrated access models during the specified time period, which, in this case, is the twelve months ended December 31, 2023). We commercialize our SOPHiA DDM Platform and related solutions, applications, products and services as RUO and CE-IVD products.
In the United States, our products are labeled and sold for research use only. Because such applications and products are not intended for use in clinical practice and diagnostics and cannot make clinical or diagnostic claims, the FDA regulations require that RUO applications and products be labeled “For Research Use Only. Not for use in diagnostic procedures.” In the EU, we have self-certified our applications and products without the intervention of a notified body in order to affix the CE marking.
Data-driven medicine has become possible through technological breakthroughs, like NGS, that have driven creation of digital healthcare data and an accelerated understanding of biology and disease. While genomics has played a large role in these advances, emerging technologies such as radiomics, digital pathology and proteomics are creating new data sets that add phenotypic context to genomic information. Additionally, the adoption of EHRs has enabled the matching of clinical outcome data to these data sets. The digital format of these data sets makes them ideal candidates for data exploration, analysis and interpretation by advanced algorithmic computing solutions. We believe that analytics approaches have traditionally primarily focused on analyzing data from a single modality and not on combining structured data from multiple modalities. Although some institutions and laboratories have created service-based business models designed to capture multimodal data, these approaches are typically centralized at a single institution, which we believe limits their ability to scale globally.
With our SOPHiA DDM Platform, we have the potential to serve and collaborate with all types of institutions in the global life sciences ecosystem, including healthcare providers, clinical and life sciences research laboratories and biopharmaceutical companies. Our platform is built on a decentralized model in which we push data analytics solutions to our customers’ sites, rather than a centralized model that requires samples to be sent to a central location. Our customers therefore generally perform testing on their own samples, retain custody of both their sample and data, and use our SOPHiA DDM Platform to analyze the pseudonymized data and share insights with other sites in our network. Through this process, we create and grow a global collective intelligence. Our platform is designed to improve as we analyze more data over time, leveraging AI and then sharing the benefits of this growing collective intelligence with our customers.
We believe that our global platform empowers better patient care through data-driven medicine by offering the following benefits for customers:
•high accuracy genomic analysis to support clinical diagnosis and life sciences research;
•rapid turnaround time for data analysis and insights;
•ability to lower cost of data analysis through higher efficiency;
49
•capacity to develop their own in-house precision medicine expertise and operations, retain custody of their samples and data and use their preferred instrument setup; and
•option to rapidly launch new precision medicine applications on our SOPHiA DDM Platform.
We believe that our strategic positioning as a universal healthcare analytics platform for multimodal data analytics offers us a broad range of application, product, and service expansion opportunities and significant long-term growth in our total addressable market opportunity. We estimate the total addressable market opportunities in 2023 for our current commercial clinical applications and for our current biopharma applications were approximately $25 billion and $15 billion, respectively.
We offer a range of platform access models to meet our customers’ needs. Our primary pricing strategy for our clinical customers is a pay-per-use model, in which customers can access our platform free of charge but pay for each analysis performed using our platform. To commercialize our applications and products, we employ our direct sales force, use local distributors and form collaborations with other global product and service providers in the healthcare ecosystem to assemble solutions to address customer needs. For example, we combine our solution and applications with other products used in the genomic testing process to provide customers integrated products in the testing workflow. We offer our SOPHiA DDM Platform and related solutions, applications, products, and services across 68 countries through our direct sales force and our distributor partners. As of December 31, 2023, our direct sales team consisted of more than 84 field-based commercial representatives with a direct presence in 64 countries. To supplement our direct sales force, we also offer our SOPHiA DDM Platform and related solutions, applications, products, and services in 39 countries through our distributor partners.
The Importance of Data-Driven Medicine
Over the last decade, there has been an explosion in the amount of healthcare data. This growth has been fueled by technologies that enable high throughput analysis and data generation at large scale, as well as the collection and digitization of real-world health data in EHRs. The ability to draw insights from this data has led to an acceleration in the understanding of biology and disease and paved the way for data-driven medicine.
Data-driven medicine aims to produce better clinical and scientific outcomes by drawing insights from complex data sets to improve diagnosis, treatment and drug development. Using data-driven medicine, healthcare professionals are able to supplement their own experience and intuition with shared knowledge and data insights from their peers and have the potential to select the best course of action for their patients or research.
Genomics is propelling data-driven medicine. The development of large-scale genomics data is advancing data-driven medicine. With broad access to NGS technologies, the life sciences field is beginning to successfully document the relationship between the genome and various diseases and is deploying this information to improve clinical and scientific outcomes. This has given rise to the field of precision medicine, which is having an increasing impact on a range of life sciences areas. In oncology, for example, advancements in genomics and the understanding of cancer have fueled the growth of a large precision oncology ecosystem, in which genomic information is critical to informing diagnostic, treatment and drug development decisions. In other areas such as rare diseases, cardiology, neurology, metabolism and infectious diseases, the adoption of data-driven medicine is just beginning and represents a significant opportunity for growth. For instance, in cardiology, clinical genomics is becoming more common for screening, diagnostic and therapy selection for certain inherited conditions, while in neurology, clinical genomics is helping direct treatment decisions for therapeutic intervention.
Multimodal data provides novel and deep insights to assess health and disease states. While the growing understanding of genomics has dramatically advanced the life sciences field and data-driven medicine, it is only one piece of the biological equation. Phenotypic information is also needed to put genomic information into context and provide a more complete picture of biology and disease. Driven by this need, innovation is accelerating across new health technologies, such as radiomics, digital pathology and proteomics, providing this phenotypic context. We believe that combining data from different instrument modalities, or a multimodal approach, will transform clinical and scientific outcomes by generating clinically actionable insights from combined relevant healthcare data sets. If leveraged properly, these data sets have the potential to provide a stronger “signal,” or window into biology and disease, than any single modality alone. In oncology, for example, oncologists can characterize the genetic determinants of a tumor at the time of diagnosis and complement this
50
with phenotypic information through radiomics analysis of CT, MRI, SPECT and PET imaging, digital pathology analysis of histology slides and proteomics analysis of the tumor stroma and blood samples. Then, throughout a patient’s disease journey, the oncologist can collect longitudinal insights through imaging, liquid biopsies and proteomics assessment of repeat blood sampling. This information can be aggregated and linked to clinical outcome data to find associations between disease evolution and response to therapy. In addition, deep-learning algorithms applied to multimodal data sets now make it possible to predict the evolution of a disease or the response to a specific treatment with high accuracy, in order to inform the best treatment decisions for the patient. These unique insights are driving the opportunity and demand for analytics platforms that can draw clinically actionable insights from this information.
AI/ML produce novel insights from large and complex data sets. The output of these new health technologies is generated in a digital format, making data highly amenable to advanced algorithmic computing solutions for exploration, analysis and interpretation. AI approaches have enabled the ability to standardize, classify, analyze and interpret massive volumes of data, and separate the signal from the noise. Large volumes of digital information across modalities can then be mined using AI approaches to generate novel insights, enabling truly data-driven medicine.
Challenges to the Adoption of Data-Driven Medicine Today
While we believe that data-driven medicine has the potential to transform healthcare, currently, there are significant challenges that limit its democratization and adoption at scale. These challenges include:
•Lack of data harmonization and standardization across the healthcare ecosystem. Data is often produced with different approaches and methodologies, which can result in dramatic variability in data quality. In the clinical genomics field, for example, every experimental step, from using different technologies for nucleic acid extraction and DNA or RNA amplification to using different models of NGS instruments, could lead to inconsistent data across sites and experiments, or “noise” in the data. As a result, obtaining a comparable set of clinical genomics data can be challenging, particularly in decentralized settings in which inter-laboratory variability can be considerable.
•Data silos and lack of knowledge sharing. Most healthcare data is produced by different healthcare institutions and by centralized laboratories that use different instrument modalities. As a result, data is created and remains in silos. In hospitals, for example, clinicians may struggle to collect and piece together data sets from clinical genomics to pathology to medical imaging for patients that have been produced in different, non-standardized ways. Pharmaceutical companies face similar challenges when reconciling their clinical trial data with disparate real-world data sources, resulting in highly variable quality of insights.
•Barriers to collaboration. Healthcare professionals and researchers may be limited in their ability to share their patients’ healthcare data for various reasons, such as privacy or concerns over losing control of their data. In addition, they have difficulties collaborating with peers from different sites or different fields. As a result, collaborations among healthcare professionals and researchers across different sites and fields is suboptimal.
•Healthcare infrastructure is designed to facilitate healthcare delivery at a local or regional level, rather than on a global scale. Healthcare infrastructure is generally designed around centralized institutions, such as hospitals and laboratories, that generate data within their own facilities. This centralized design is not built to scale or to provide equal access to data-driven medicine globally.
•Existing software analytics approaches are limited in their ability to generate insights from multimodal data. Traditional approaches to software analytics solutions have primarily focused on analyzing data from a single modality and not on combining structured data from multiple modalities. Existing analytical software solutions thus have limited utility to generate insights from multimodal information.
51
Our SOPHiA DDM Platform
We believe that a decentralized platform is necessary to create the largest network that will bring the benefits of big data to customers and to both support, and benefit from, growth across the healthcare ecosystem. We purposefully built a cloud-native software platform capable of being used in decentralized locations and of analyzing data from multiple modalities and that can be scaled globally. With our SOPHiA DDM Platform, we have the potential to serve and collaborate with a variety of types of institutions in the healthcare ecosystem, including healthcare providers, centralized laboratories and biopharmaceutical companies.
Our SOPHiA DDM Platform is a global, cloud-native software platform that we began building in 2011. It is powered by our SOPHiA AI that standardizes, computes and analyzes digital health data, generating insights from complex multimodal data sets that have the potential to improve diagnosis, therapy selection and drug development. Our customers generally perform testing on their own samples, retain custody of both their sample and data, and use our SOPHiA DDM Platform to analyze the pseudonymized data and share insights with each other. Through this process, we create and grow a collective intelligence. We offer multiple platform access models that enable customers to choose how they want to use our platform and customer network. These range from models in which customers produce their own data independently through their own testing operations to those in which customers produce the data through testing operations provided by our network of customer institutions. In all cases, customers access their data and our analytics through our SOPHiA DDM Platform. Our platform is designed to continually improve as we analyze more data over time, leveraging AI and then sharing the benefits of this growing collective intelligence with our customers.
We believe that our SOPHiA DDM Platform addresses key challenges to the adoption and democratization of data-driven medicine by:
•Enabling data harmonization and standardization across the healthcare ecosystem. The accuracy of our pattern-recognition AI/ML-based algorithms enables our platform to separate the signal from the noise and standardize data at high-quality levels.
•Breaking down data silos. We empower our customers to practice data-driven medicine through a decentralized model and support clinicians, laboratories and researchers across the healthcare ecosystem to improve clinical and scientific outcomes.
•Empowering clinicians and researchers to collaborate with peers from different sites or different fields. Our customers use our platform to share insights with each other across sites in our network. Our platform is designed to improve as we analyze more data over time, leveraging AI and then sharing the benefits of this growing collective intelligence with our customers.
•Offering a highly scalable platform. We designed our cloud-native software platform to be capable of scaling globally and to use AI to leverage the data that this scale provides.
•Generating insights from complex multimodal data sets. We believe our platform is uniquely positioned to combine high-quality data at the patient level to generate multimodal insights, leveraging the power of advanced AI/ML models.
52
The following figure shows how our SOPHiA DDM Platform functions within the healthcare ecosystem.
Our SOPHiA DDM Platform within the Healthcare Ecosystem

We launched the first commercial application of our platform in 2014 to analyze NGS data for cancer diagnosis. We offer a broad range of applications focused on precision medicine across oncology, rare diseases, infectious diseases, cardiology, neurology, metabolism and other disease areas. In 2019, we launched our solution for radiomics data that enables longitudinal monitoring of cancer patients and tumor progression throughout their disease journey. In 2022, we unveiled SOPHiA CarePath, a new module that will further enhance our solution for longitudinal monitoring of cancer patients and tumor progression by integrating data across multiple modalities including genomic, radiomic, clinical, and biological). CarePath serves as the successor to our radiomic solution, which we plan to integrate into the module.
Our Network and Data
Today, we believe that our SOPHiA DDM Platform, commercialized under the name “SOPHiA DDM,” is one of the most widely used decentralized analytics platform globally for clinical genomics. As of December 31, 2023, we served more than 770 hospitals and laboratory customers globally through our SOPHiA DDM Platform who are part of our clinical genomics network. The establishment and creation of this network of customers has enabled us to capture and compute more than 1.5 million raw clinical genomics profiles in oncology and other genetic-related disorders as of December 31, 2023, growing by more than 25,000 new profiles on a monthly basis.
Our SOPHiA DDM Platform Architecture
We believe that our platform architecture allows our platform to be highly flexible and scalable in terms of analyzing larger volumes of data, supporting additional data modalities, expanding to new geographies and deploying new applications and functionalities. This flexibility and scalability comes from our platform’s underlying architecture that is developed based on a deep understanding of our users’ needs and a thorough domain model, allowing us to build re-useable User Interface (“UI”) components and services to interact with the data. Our SOPHiA DDM Platform includes multiple tailored analytics engines, each tuned to specific domains and use cases. Each domain is responsible for its own data with a common shared data model that allows powerful Extract-Transform-Load (“ETL”) pipelines to process specific data sets, integrating data into a series of regional data warehouses that enable comprehensive and performant multimodal queries to be run across the entire global data set. We have eight regional data centers in Switzerland, France, the Netherlands, the United States, Canada, Brazil, Australia, and Turkey.
53
As of December 31, 2023, this platform architecture was deployed in 68 countries through our cloud-based solution. We have developed significant operational experience by running such a large-scale cloud-based platform, which we believe enables us to deploy rapidly in new geographies. We have demonstrated that we can deploy in a new geography in approximately four weeks if appropriate cloud infrastructure exists such as Microsoft Azure, Amazon Web Services or Google Cloud Platform, or in approximately twelve weeks if such cloud infrastructure does not exist.
We regularly release platform updates. Through these updates, we offer our customers either new content, in the form of new applications, or improvements to existing applications, such as new functionalities. We believe the frequency of our updates is a competitive advantage in a rapidly evolving precision medicine ecosystem and allows our customers to benefit from new biological discoveries, such as genomic associations, that are reflected on the platform.
Cybersecurity
As part of our business, we collect, transmit, receive, process, use and store pseudonymized data provided by our customers. Our customers are required to obtain their patients’ consent to our use of the data. We use security techniques designed to safeguard data received from our customers using a combination of data architecture, pseudonymization, anonymization, minimization and segregation, and process and store this data only in accordance with our agreements with customers and applicable data protection laws and regulations. This data is aggregated and analyzed by our proprietary algorithms and models in our SOPHiA DDM Platform to generate insights. These insights, which show aggregated and general trends without identifying specific patients and without providing personally identifiable information, form the growing collective intelligence that we provide to our customers.
Cybersecurity and data protection are core tenets of our company. We have processed over 1.5 million genomic profiles and continue to process more than 50 terabytes of data each month for our customers around the world, subject to applicable data protection laws and regulations, including HIPAA and the GDPR. We accomplish this through our global compliance framework that integrates specialized and dedicated personnel, procedures and controls and ISO/IEC 27001:2013 security infrastructure to protect data against damage, loss and unauthorized access, use, modification, disclosure or other misuse.
Applications of Our Platform
We currently have commercial applications targeting both clinical and biopharma markets. We serve our clinical market customers through two offerings of our SOPHiA DDM Platform. Our first offering is our SOPHiA DDM platform for clinical genomics, spanning a broad range of applications that we market for analyzing genomic data across oncology, rare diseases, infectious diseases, cardiology, neurology, metabolism and other disease areas. Our SOPHiA DDM platform empowers customers to build their own precision medicine operations, including testing, and then use our platform to generate insights from their data. Our second offering is our Alamut suite of genomics mutation interpretation software, which is connected to our SOPHiA DDM platform and gives our customers advanced analytics capabilities for a deeper and more informed genomic data interpretation.
For revenues generated from applications for which the disease end market is known, approximately 70% of our revenue from clinical customers in the year ended December 31, 2023 was attributable to oncology applications, including hereditary cancer, while approximately 30% was attributable to other disease areas such as rare diseases, cardiology, neurology and metabolism, with applications ranging from targeted gene panels to whole-exome solutions. In the future, we intend to pursue additional IVD status and FDA approval for specific solutions. We also intend to support external collaborators in deploying their own IVD or FDA-approved solutions on our SOPHiA DDM Platform.
We serve our biopharma customers by leveraging the capabilities and Data of our SOPHiA DDM Platform to help customers solve bottlenecks across the biopharma value chain, including throughout the Discovery, Development and Deployment stages. We currently have four branded applications for biopharma customers: SOPHiA Insights for generating insights pre- and post-approval of a drug based on our own proprietary SOPHiA DDM Platform data sets or on the biopharma customers’ own data sets; SOPHiA Trial Match for clinical trial
54
recruitment of biomarker-defined patient populations; SOPHiA CDx for companion diagnostics development and deployment in our decentralized network of customer institutions; and SOPHiA Awareness for providing real-world insights into NGS testing to inform market-shaping and commercialization strategies. We launched our initial applications for the biopharma market in 2019.
The following figure shows our applications that we currently commercialize across both clinical and biopharma markets.
Our SOPHiA DDM Platform’s Applications Currently in Market

Clinical Applications
In the clinical market, we currently serve three main customer segments: academic and non-academic hospitals (including comprehensive cancer centers and children’s hospitals), reference laboratories and specialty laboratories. We currently serve our clinical market customers through two offerings of our SOPHiA DDM Platform: our SOPHiA DDM platform for clinical genomics and our Alamut suite of genomics mutation interpretation software. We have also unveiled SOPHiA CarePath, a multimodal module on our SOPHiA DDM Platform, encompassing the capabilities of our radiomic solution, which we plan to commercialize across our clinical customer base in the future.
Oncology Applications
Our oncology applications support both germline and somatic oncology testing across both solid and liquid tumors. Our commercial oncology applications support diagnosis, therapy selection and disease monitoring. Our SOPHiA DDM Platform also supports the deployment of novel oncology testing applications. In genomics, this includes liquid biopsy-based early cancer screening as well as treatment response monitoring and minimal residual disease monitoring. In multimodal, we are able to complement and enhance our genomics capabilities with other data modalities, such as radiomic and clinical data, to include diagnosis, prediction of disease evolution and response to specific therapies, as well as longitudinal follow-up of the tumor for treatment response monitoring. The following figure shows our current applications in oncology.
55
Our SOPHiA DDM Platforms’ Oncology Applications
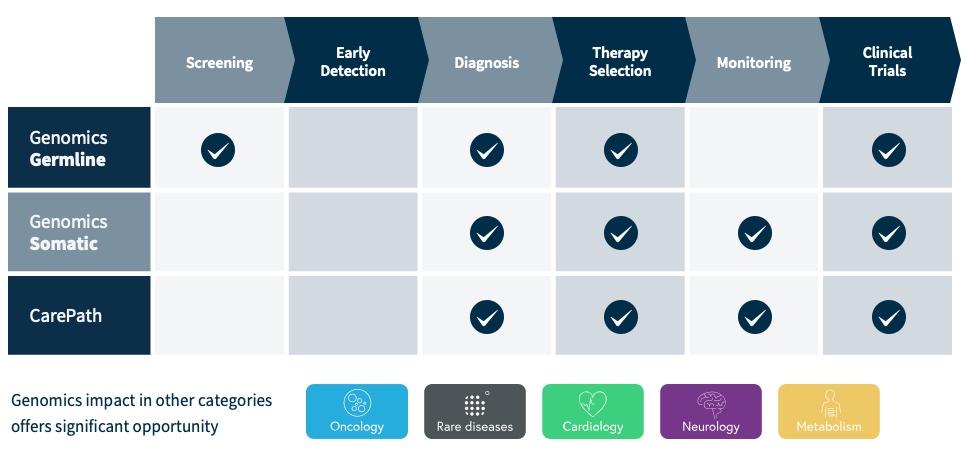
Non-Oncology Applications
Our non-oncology applications currently focus on disease areas such as rare diseases, cardiology, neurology and metabolism, with applications ranging from targeted gene panels to whole-exome solutions. While clinical genomics applications are still emerging in these disease areas, we expect significant opportunity as the life sciences field continues to establish the genetic determinants of high-profile diseases such as hereditary cardiovascular conditions, multiple sclerosis, Alzheimer’s disease, autism and metabolic syndrome. We also see significant promise of multimodality in these other disease areas, for example, in cardiology by generating novel multimodal insights stemming from the joint analysis of genomics data, radiomic analysis of ultrasound images and analysis of electrocardiograms.
Our SOPHiA DDM Platform in Genomics
We believe that our technical capabilities are cutting-edge in the genomics space. Our platform can process data from any type of biological sample, including fresh frozen tissue, formalin-fixed paraffin-embedded samples as well as liquid circulating tumor DNA samples. It can also process data from any nucleic acid source across DNA and RNA. We can identify with confidence any type of genomic alteration, including single nucleotide variants (“SNVs”), insertions-deletions (“indels”), copy-number variations (“CNVs”) and gene fusions, as well as more complex mutational signatures such as microsatellite instability (“MSI”), tumor mutational burden (“TMB”), homologous recombination deficiency or minimal residual disease. Our smart algorithms allow us to reach high accuracy on the detection and identification of challenging genomic alterations, such as mutations in CEBPA or FLT3-ITD, MET exon14 skipping mutations, or rare gene fusions. The following figure shows our SOPHiA DDM Platform’s capabilities in genomics.
56
Our SOPHiA DDM Platform’s Capabilities in Genomics
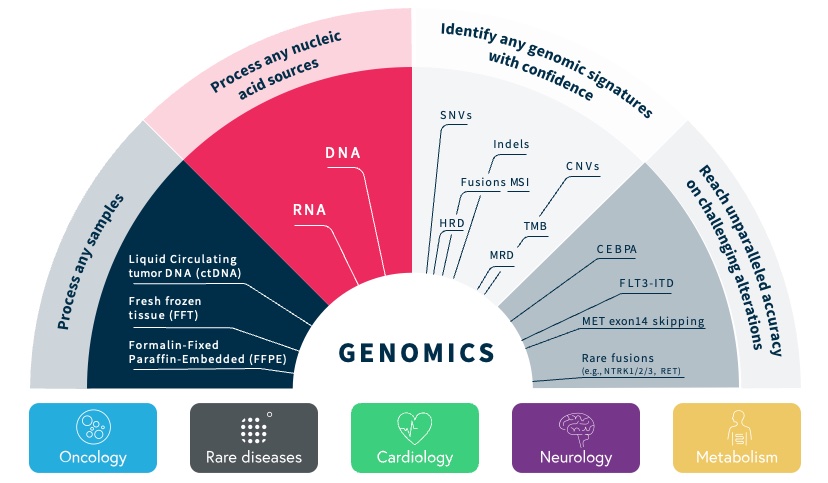
Our SOPHiA DDM Platform clinical genomics workflow involves CLIA-CAP and equivalent laboratories in academic hospitals, comprehensive cancer centers, children’s hospitals and reference and specialty laboratories collecting patient samples and conducting the genetic sequencing on their premises. In doing so, they can use different NGS solutions on different NGS sequencing instruments.
For somatic oncology applications, for example, the laboratory technician logs into our SOPHiA DDM Platform and loads the raw, pseudonymized, NGS data of multiple patients from the sequencing run, indicating the oncology indication to be investigated. The genomics data is securely transferred to our platform that operates globally in eight different regional data centers, keeping the data closest to the customer and complying with all local data handling requirements. The data is automatically recognized by our AI-based smart algorithms that check data quality. All types of genomic variants and signatures are then detected and identified with high accuracy, including SNVs, indels, CNVs, fusions, MSI and TMB. This molecular information is then annotated and pre-classified using AI/ML techniques.
The principal investigator, usually a pathologist or geneticist (for germline applications), accesses the results and completes the interpretation. The principal investigator may flag or store the genomic variants that he or she has recognized as being associated with a certain disease on our platform. Because of the decentralized nature of our network, other users in different sites can see the aggregated flagging of a specific variant from the community to further assist their own interpretation. The more interpretations being conducted in our platform, the more novel knowledge is generated and made available to our community. Our platform is particularly easy to use as it does not require an additional technician and provides a user-friendly interface to upload data and navigate data analytics. We believe that this ease-of-use, coupled with the scale of our decentralized platform, will empower our users to continue to rapidly uncover new variants.
This workflow is technology-agnostic in terms of sequencer type and sample preparation technology and supports a broad range of different commercial NGS solutions. The following figure shows our SOPHiA DDM Platform’s genomics workflow.
57
Our SOPHiA DDM Platform’s End-to-End Genomics Workflow
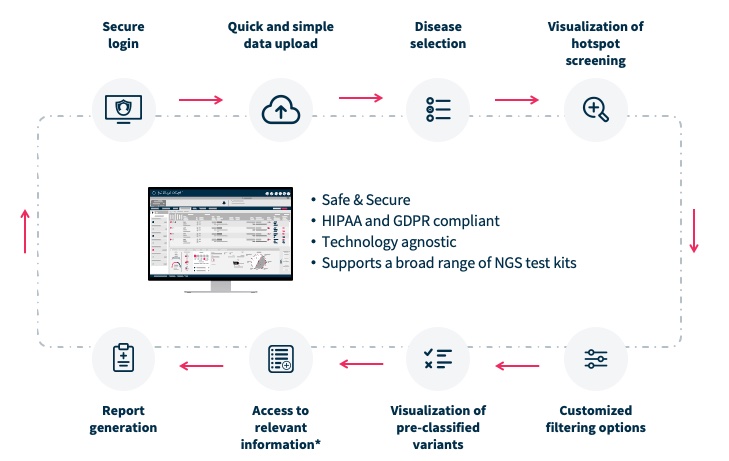
Our SOPHiA DDM Platform in Multimodal Data Analytics
Through our SOPHiA CarePath module on our SOPHiA DDM Platform, we can support the multimodal analysis of any source of digital health data, developing machine learning predictive models of aggregated multimodal data stacks for the same patient. We can support the analysis of clinical, biological, genomics, radiomics data today and intend to support additional data modalities such as digital pathology, proteomics, spatial genomics and metabolomics in the future.
We offer a range of predictive modeling applications ranging from disease screening, disease early detection, disease diagnosis and subtype discrimination, prediction of disease evolution, prediction of response to therapy, therapy selection and monitoring. We develop these multimodal predictive models in close collaboration with leading academic institutions together with cross-functional teams consisting of treating physicians (such as oncologists), radiologists and pathologists. Illustrative examples of advanced clinical research projects in which we have proof-of-concept data include prediction of response to anti-PD-1 immunotherapy for patients with metastatic non-small cell lung cancer, and prediction of pathological complete response after neoadjuvant therapy for patients with triple-negative breast cancer. For example, we sponsored a retrospective 57-patient analysis of the use of nivolumab for the treatment of relapsed or refractory non-small cell lung cancer to identify predictive markers of immune-oncology response based on multiple sources of data through machine learning analysis. We found that machine learning could help predict a patient’s response using baseline data and can help identify markers that are predictive of the patient’s response. We are sponsoring multimodal clinical studies to refine and assess the clinical significance of some of these multimodal signatures, which we believe will enable us to further improve our SOPHiA DDM Platform and develop new predictive algorithmic models that we can then deploy on our platform to serve a wide range of stakeholders, including oncologists and other treating physicians.
58
The following figure shows our SOPHiA DDM Platform’s capabilities in multimodal data analytics.
Our SOPHiA DDM Platform’s Capabilities in Multimodal Data Analytics
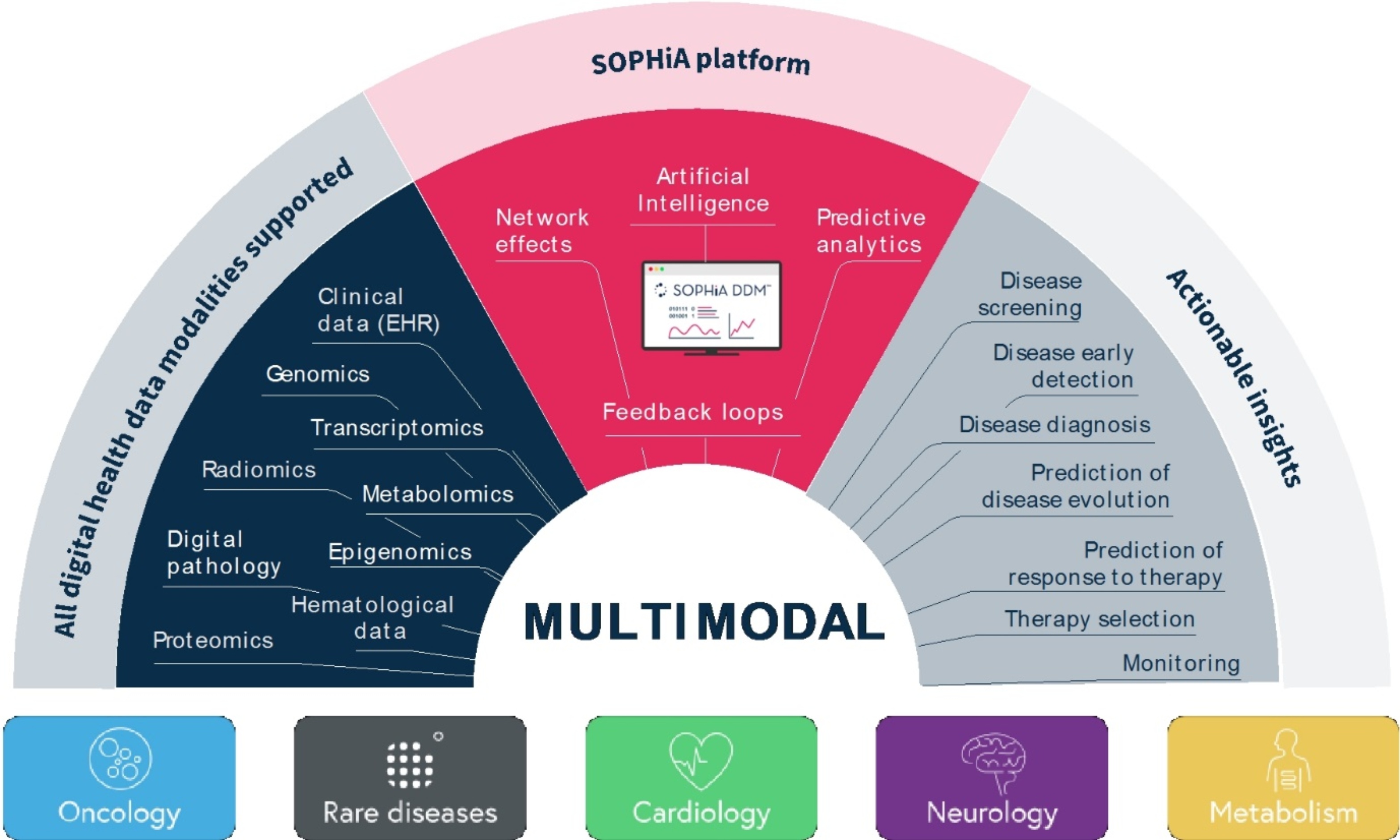
Among our multimodal capabilities, besides our initial core competency in genomics, we have built out technical capabilities that we believe are cutting-edge in the radiomics space. These capabilities are integrated into and offered through our SOPHiA CarePath module. We can process and analyze data from any type of three-dimensional medical imaging technology, including CT, PET, MRI and SPECT scanners. We have developed AI/ML-powered segmentation algorithms that detect tumors in the scans and that segment and reconstruct tumors in three dimensions on our SOPHiA DDM Platform. Our current segmentation applications cover a wide range of major tumor types, including lung, breast, liver, kidney and brain cancer, supporting the analysis of primary and secondary (i.e., metastatic disease of another organ origin) tumors. In addition, we are developing new applications in areas such as colorectal, prostate, ovarian or neuroendocrine cancers. Radiomics features extraction is conducted on segmented tumors, generating hundreds of data points across volumetric, morphological, first order (i.e., heterogeneity), second order (i.e., texture), and deep-learning generated features. Our features extraction process is compliant with the Image Biomarker Standardization Initiative (“IBSI”) recommendations, such that results from our radiomics analyses are standardized and can be readily compared with similar analyses globally. Through our segmentation and radiomics features extraction steps, we turn existing medical images into hundreds of novel data points. We offer radiomics applications ranging from disease detection, discrimination of disease histological subtypes, prediction of tumor evolution and prediction of disease progression. In the future, we intend to develop additional radiomics applications for existing and new tumor types, as well as for disease areas outside of oncology, such as cardiology, neurology and metabolism.
Additionally, we may expand applications to other imaging modalities such as two-dimensional modalities (e.g., ultrasound, traditional x-rays) as well as testing modalities that can be processed through imaging-based approaches (e.g., electrocardiograms). The following figure shows our SOPHiA DDM Platform’s capabilities in radiomics.
59
Our SOPHiA DDM Platform’s Capabilities in Radiomics
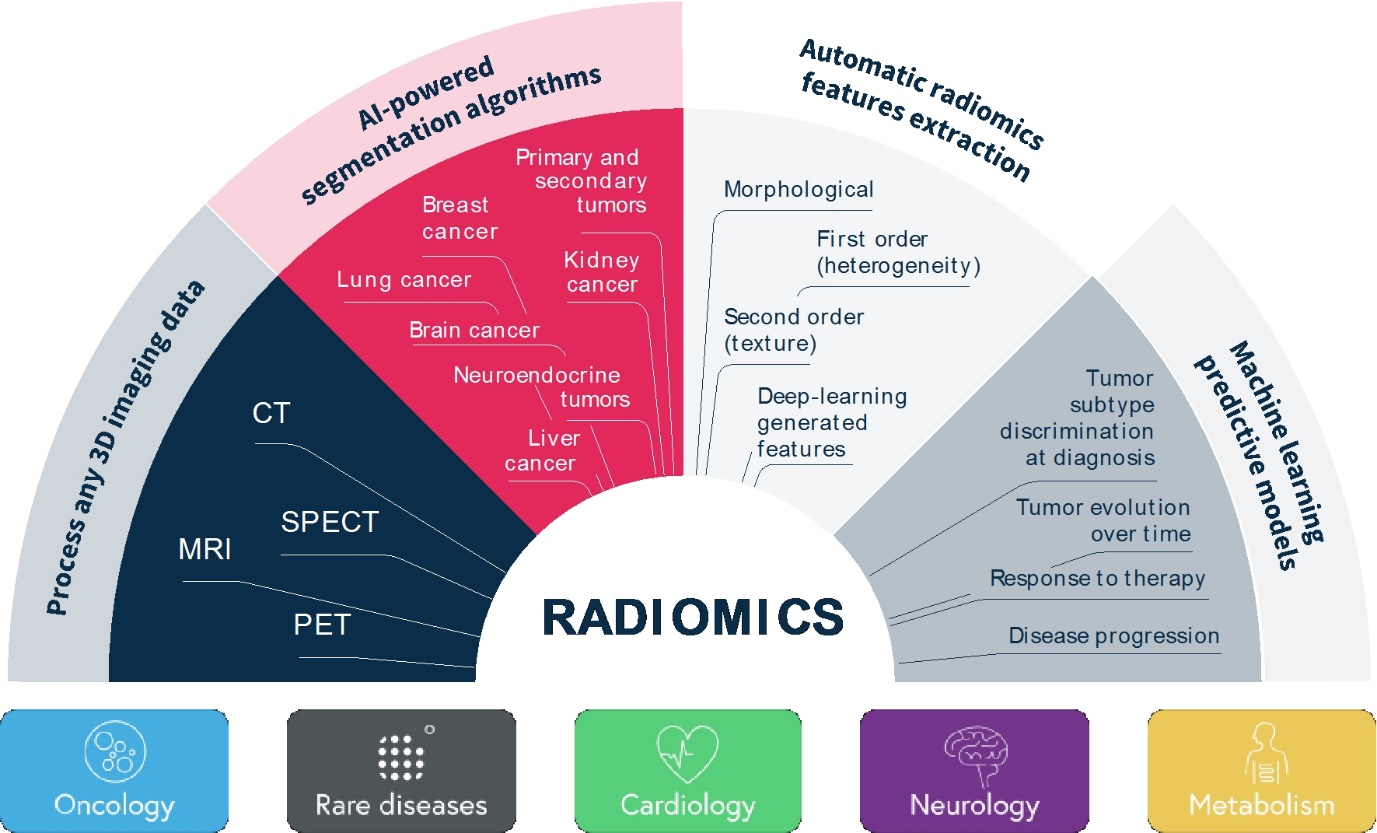
While the data modality is different for radiomics compared to genomics, the same overall workflow and principles apply. The user, typically a radiologist, identifies the relevant medical images for a specific patient in the local picture archiving and communication system. The user then uploads the images into our SOPHiA DDM Platform. For a metastatic lung cancer case, for example, deep learning and other machine learning proprietary algorithms automatically detect the imaging modality, recognize the organ, segment the tumor and extract more than 200 radiomics features from the tumor image. These radiomics features can then be aggregated with genomics, clinical and biological data from the same patient. The following figure shows our SOPHiA DDM Platform workflow for radiomics.
Our SOPHiA DDM Platform’s End-to-End Radiomics Workflow
Biopharma Applications
In the biopharma market, we currently serve three types of customers: pharmaceutical companies, biotechnology companies, and CROs. Leveraging both our SOPHiA DDM Platform data and our customers’ own proprietary data through our AI/ML-powered multimodal analytics capabilities, we help customers solve bottlenecks across the biopharma value chain. We categorize our applications based on a framework to address specific needs across the Discovery, Development, and Deployment stages of the drug and therapeutic life cycle both pre- and post-commercialization supported by the underlying data generated on our Platform. Across our application portfolio, we offer four branded applications: SOPHiA Insights, SOPHiA Trial Match, SOPHiA Awareness and SOPHiA CDx, to address the needs of our biopharmaceutical and biotechnology customers, including diagnostic assay development, clinical trial matching and application, drug and therapeutic design optimization, and diagnostic assay deployment. The following figure shows our capabilities across the biopharma value chain.
60
SOPHiA GENETICS’ Offerings across Biopharma Value Chain
We began commercializing biopharma applications in 2019. Our biopharma applications are competitively positioned as insights programs, which utilize data already uploaded to the SOPHiA DDM platform or proprietary data provided directly by a biopharma customer. We believe that our customers value our biopharma applications for their ability to identify unique patient populations in clinical research and asset commercialization efforts. We signed our first biopharma customer in 2019. We served six biopharma customers as of December 31, 2023
Discovery Applications.
While Moore’s law has generally played out accordingly in the computation and software space with greater efficiency for lower cost being achieved over time, Eroom’s law has been used to explain an observed trend in the drug discovery space as the inflation-adjusted costs to bring a drug to market continue to rise over time. Despite the improvements in technology over time, drug discovery has remained very complex with many critical decisions that need to be made along the way at which point could ultimately lead to an unviable path forward.
To address the growing costs of drug discovery, biopharma and biotechnology companies are increasingly turning to technology solutions, particularly artificial intelligence and machine learning, to apply the benefits of Moore’s law to reverse the observed costs according to Eroom’s law. With our SOPHiA DDM Platform, we can help our biopharma and biotechnology customers make more informed decisions in the drug discovery process by leveraging the data we generate on our SOPHiA DDM platform across our global network and applying our proprietary artificial intelligence and machine learning algorithms to the data across our network and the proprietary data of our customers. Within the Discovery segment, our current primary focus is on the later sub-stages post target identification and prior to Development, such as assay development and lead optimization.
Development Applications
The drug and therapeutic development process can be very long and capital intensive. There are both significant financial and opportunity costs that are incurred but never recouped should a drug or therapeutic fail in development stage. To increase the likelihood of success, biopharmaceutical and biotechnology companies are increasingly turning to algorithm based approaches powered by machine learning and artificial intelligence to better predict drug properties and aid in drug and therapeutic design and clinical trial testing and applications.
We are supporting our biopharma and biotechnology customers in the drug and therapeutic development process through our decentralized, technology agnostic SOPHiA DDM Platform powered by our machine learning and artificial intelligence algorithms. Our SOPHiA DDM Platform’s unique ability to harmonize data derived from diverse genomic instruments and deploy as a robust, standardized solution enables a new model for clinical trial testing through a decentralized approach, allowing our biopharma and biotechnology customers to optimize patient selection and clinical trial design. Leveraging our SOPHiA DDM’s capabilities and multimodal data, we can also support our customers in developing companion diagnostics (“CDx”) to improve testing across the target patient populations and help them in optimizing their drug and therapeutic designs. Through
61
our SOPHiA CarePath module, we will also enable our customers to track patients longitudinally across multiple data modalities to support their clinical trials.
Deployment Applications
As biopharmaceutical companies begin deploying their drugs and therapies, they can face challenges associated with the identification of eligible patients. To increase adoption, biopharma companies have partnered with clinical institutions to expand access to diagnostic testing that could help identify new patients eligible for and who can benefit from the new drugs and therapies.
We can help our biopharma customers with their deployment-related challenges in multiple ways. In the absence of an applicable diagnostic assay, the versatility of our SOPHiA DDM Platform allows us to support our customers by helping them develop new targeted assays, including companion diagnostics, to diagnose and identify eligible patients. We can then help our customers expand access to diagnostic testing by deploying a SOPHiA GENETICS developed diagnostic assay or a third-party developed diagnostic assay across our broad global network of hospital and laboratory customers.
SOPHiA Insights
Faced with a complex and fragmented precision medicine environment, we believe biopharmaceutical companies need access to high quality real-world data sets and advanced data analytics capabilities to generate insights from these data sets to inform their decision-making. However, currently, these data sets are often fragmented, siloed and of variable quality, while data analytics capabilities are typically more focused on single-modality applications.
We offer solutions to support biopharma customers by generating insights pre- and post-approval of a drug throughout the entire pharma value chain, including the research, development and commercialization stages. We can generate these insights both based on our SOPHiA DDM Platform real-world data sets and by leveraging our AI/ML-powered multimodal analytics capabilities on the biopharma customer’s own data sets, including data from their clinical trials. For example, a biopharmaceutical company may ask us to generate insights from our platform regarding the real-life molecular epidemiology of rare genomic variants in a specific cancer type, including NGS sequencing install base and testing practices across geographies. A biopharmaceutical company may also ask us to support it in the AI/ML-powered multimodal analysis of its own data sets, which could include genomics, clinical, biological and medical imaging data from its clinical trials, for example to identify new biomarkers associated with patient subgroups that may have a higher likelihood of response to an investigational therapy. As we generate increasingly more multimodal patient-level data stacks in our SOPHiA DDM Platform, in the future, we may support biopharmaceutical companies on novel use cases, including real-world virtual control arms for clinical trials.
SOPHiA Trial Match
Challenges of clinical trial patient enrollment is a major bottleneck for clinical trial sponsors, which leads to delays, increased costs and clinical trial failures. This challenge is magnified in the case of biomarker-targeted investigational therapies associated with rare genomic variants due to the difficulty of finding and recruiting patients with the desired genomic traits.
With SOPHiA Trial Match, sponsors can place “molecular alerts” in our SOPHiA DDM Platform for specific genomic variants or signature that may indicate eligibility for a clinical trial. When a genomic profile matching the recruitment criteria is detected in our platform, the participating local healthcare Institutions or research centers are notified in real-time and given the opportunity to connect with the clinical trial sponsor. We provide the real-time trial matching services for our participating customers.
SOPHiA CDx
We are witnessing a steady growth in the number of regulatory approvals for therapies linked to companion diagnostic assays in oncology and other disease areas. Today, these CDx assays are typically used in a centralized model in which healthcare institutions lose access to their samples and data and which can suffer from poor turnaround times due to logistical issues. We believe that in the future CDx assays will become increasingly decentralized which will drive further testing uptake at scale and enable faster turnaround times.
62
We offer strategic and operational support for biopharma CDx programs. Biopharma customers can leverage our capabilities to develop genomic variant detection and identification solutions with high accuracy and precision, as well as our ability to decentralize such CDx solutions at scale through our global footprint. We believe that, in the future, CDx programs may become multimodal in nature, which we would be in a position to support through our multimodal analytics capabilities.
SOPHiA Awareness
As biopharmaceutical companies commercially launch new biomarker-targeted therapies (e.g., linked to a specific companion diagnostic assay), they face significant challenges in driving broad adoption and testing rates of specific biomarkers of interest. For example, the genomics testing landscape is currently fragmented with important regional and local variations. In that context, we believe it is imperative for biopharmaceutical companies to adequately manage parallel and interdependent adoption curves across the biomarker testing and therapy prescription dimensions. While biopharmaceutical companies tend to have insights into prescription patterns by health practitioners, they typically lack insights into real-life genomics testing practices across geographies.
We support biopharma customers with real-world insights on NGS testing trends to support their market-shaping and commercial strategies. For example, when novel therapies enter the market, a biopharmaceutical company may ask us to provide regular aggregated statistical reports on NGS testing results in specific geographies to optimize resource allocation for its go-to-market strategy. A biopharmaceutical company may also collaborate with us to increase the NGS testing rate of specific biomarkers in our network, thus supporting the identification of relevant genomic variants for targeted therapies, for example by sponsoring increased testing volumes and NGS panel upgrades. As more data modalities are computed in our SOPHiA DDM Platform, we envision additional market opportunities for SOPHiA Awareness.
Alamut Suite of Genomics Analysis Software
Our offering in the clinical genomics space also includes the Alamut suite of genomics mutations interpretation software. This add-on software is connected to our SOPHiA DDM platform through an API and provides our customers with advanced analytics capabilities for a deeper and more informed genomic data interpretation. It simplifies and accelerates variant interpretation workflows by providing an exploration and visualization application powered by an extensive collection of top-ranked external databases and proprietary prediction tools, which has the potential to be particularly impactful in deepening genomic investigations in rare diseases.
In the future, we plan to offer other add-on software solutions that are integrated with our SOPHiA DDM Platform, including software solutions from external collaborators, and that provide additional analytics capabilities. Such offerings have the potential to further increase the value of our SOPHiA DDM Platform through indirect network effects, attracting new types of customers and solutions to our network.
Clinical Publications
Our technology and its broad applications have been utilized in clinical trials and research projects discussed in more than 617 peer-reviewed publications as of December 31, 2023. These publications support scientists in new discoveries and applications across oncology, immunology, cardiology, neurology, rare diseases and other disease categories.
Access Models
We currently have three models through which customers access our SOPHiA DDM Platform. In the dry lab access model and the bundle access model, we empower customers to produce their own data. In the integrated access model, we help customers produce data through our existing network of institutions. In all cases, clinical customers access their data through our SOPHiA DDM Platform. Our biopharma customers can access our SOPHiA DDM Platform through the same three models, but they may also have access to data generated through our SOPHiA DDM Platform in the form of custom reports and analytics.
The dry lab access model involves customers using the testing instruments and consumables of their choice and our SOPHiA DDM Platform and algorithms for variant detection and identification. In this model, we provide clinical genomics analytics capabilities without influencing the tools that customers use to generate the data. For
63
example, in genomics, a laboratory might order an NGS kit directly from the manufacturer, conduct the sequencing using its installed sequencer and then use our smart algorithms in our SOPHiA DDM Platform for data analytics.
The bundle access model enables us to support our customers end-to-end across the data generation, analytics and reporting steps. In this model, we bundle third-party instruments and consumable products with our analytics solution to provide customers the ability to perform end-to-end workflows. By bundling our algorithmic capabilities with specific high-performance instruments and consumables from third parties, we can further increase the accuracy of our genomics solutions.
The integrated access model provides customers with the ability to access high-quality data on our platform even when they cannot generate data themselves. Customers that are not able or do not wish to locally conduct the sequencing steps, for example, due to a lack of appropriate resources, can have their samples processed and sequenced within our SOPHiA clinical network. We route their samples to selected SOPHiA DDM Platform collaborators who conduct the sequencing process for the customer and upload the resulting data into our SOPHiA DDM Platform. The customer is then able to access the data through our SOPHiA DDM Platform. Through this model, the selected SOPHiA DDM Platform collaborators can increase their sequencing volumes, while our SOPHiA DDM Platform is further enriched by the data produced.
The same conceptual access models apply to our multimodal solution SOPHiA CarePath. For multimodal, including our integrated radiomics solution, we are currently offering a dry lab access model, in which we provide the algorithmic analytics solutions while leaving the data generation at the discretion of our customers. We have developed, and intend to continue to develop, smart algorithms for specific radiomics applications, such as deep learning-enabled algorithms that can automatically recognize a lung CT scan image, detect an advanced lung cancer tumor, segment the tumor and extract radiomics features for further analysis. In the future, we may also offer a bundle access model, in which we offer solutions linked to specific imaging contrasting agents or imaging procedure modalities to optimize the performance of the final signal analysis.
We intend to apply the same conceptual access models to additional data modalities that we may support in our SOPHiA DDM Platform in the future, such as digital pathology, proteomics, spatial genomics and metabolomics.
Benefits to Customers
Our platform has the potential to offer the following benefits for customers, empowering them to adopt data-driven medicine to improve clinical and scientific outcomes:
•Accuracy. Our platform design and data analytics capabilities provide high accuracy analytics for our customers, who have access to high quality, standardized data through our SOPHiA DDM Platform.
•Turnaround time. We empower our customers to generate data themselves locally, which avoids delays associated with shipment, logistics and processing of samples through an external collaborator. We therefore significantly reduce the turnaround time, which is a critical factor in driving toward timely diagnosis and treatment of disease.
•Cost-control through increased efficiency. Customers can compute, detect and annotate any type of genomic alterations through our SOPHiA DDM Platform without the need for specific orthogonal assays, thus reducing additional testing costs.
•Maintenance and development of in-house expertise. By empowering our customers to retain ownership and access to their biological samples and data, we enable them to build in-house expertise while benefiting from world-class analytics accuracy through our SOPHiA DDM Platform’s network effects.
•Accelerated launch of new precision medicine applications. The universal nature of our SOPHiA DDM Platform facilitates adding new applications to the same workflow once an institution adopts our platform. Our customers can avoid having to set up parallel and sometimes redundant workflows for different assays and technologies.
64
Markets
We estimate that our clinical and biopharma applications targeted a $40 billion global total addressable market opportunity in 2023, approximately $9 billion of which was in the United States based on our addressable clinical market. These estimates are primarily based on epidemiological data, including incidence and prevalence estimates of addressable populations for each application, as well as a range of price assumptions for our applications and products taking into account differences in panel sizes. Further, these estimates do not depend on obtaining regulatory clearances or approvals to market our applications and products as in vitro diagnostics (“IVD”) and / or software as a medical device (“SaMD”) in the United States. Over time, we believe that our platform and insights enable market opportunity expansion through new application and product development. The following figure shows our estimated total addressable market in 2023.
Our Total Addressable Market

Clinical Market Opportunity
We estimate our total addressable clinical market opportunity for our current offerings was $25 billion in 2023, with the largest market opportunity being in oncology.
Oncology. While the majority of commercial business today in the clinical market comprises diagnosis and advanced therapy selection, our capabilities enable us to serve the oncology testing market across the full patient journey. We can also support healthcare practitioners across tumor and sample types at any stage of the patient journey as long as genomic information or 3D medical imaging is applicable. Our clinical oncology market opportunity consists of five market segments: screening, early detection, diagnosis, therapy selection and monitoring.
Rare diseases. In rare diseases, we believe the adoption of data-driven medicine is just beginning and represents a significant opportunity for growth. In the rare disease market, there has been growing preference among clinical institutions for larger genetic testing panels, including whole exome and whole genome panels, to test for a more comprehensive array of rare diseases in contrast to the smaller, more targeted panels that are common in the market today. We believe we are well adapted to address this market opportunity with the exome solutions offered through our SOPHiA DDM Platform. In addition to our SOPHiA DDM Platform, our Alamut suite of genomics mutation interpretation software could be particularly impactful in deepening genomic investigations in rare diseases.
Other disease areas and conditions. We believe that the aggregate market opportunity linked to other disease areas beyond oncology and rare diseases could ultimately be larger than our current opportunity in
65
oncology and rare diseases given their higher prevalence compared to cancer. Our SOPHiA DDM Platform has applications in areas such as cardiology, neurology and metabolism, through applications ranging from targeted gene panels to whole-exome solutions. While genomics applications are still emerging in these disease areas, we expect significant opportunity in the coming years as novel findings establish the genetic determinants of high-profile diseases such as inherited cardiovascular conditions, multiple sclerosis, Alzheimer’s disease, autism and metabolic syndrome. We provide applications in each of these disease areas today, and plan to further penetrate the testing landscape in these disease areas through genomics and other data modalities in the future.
Other data modalities beyond genomics. We designed our platform architecture to be able to scale with new digital healthcare data modalities beyond genomics. We have already taken the next step in that direction by developing and deploying our proprietary radiomics analytics capabilities onto our SOPHiA DDM Platform. In radiomics, we offer analytics solutions for three-dimensional medical imaging technologies, including CT, MRI, SPECT and PET scanners, regardless of manufacturers. Additionally, we may expand applications to other imaging modalities such as two-dimensional modalities (e.g., ultrasound, traditional x-rays) as well as testing modalities that can be processed through imaging-based approaches (e.g., electrocardiograms). Beyond radiomics, we intend to support additional data modalities in the future, for example digital pathology, proteomics, spatial genomics and metabolomics. We believe that supporting additional data modalities in our SOPHiA DDM Platform, both as stand-alone modalities and in a multimodal approach, has the potential to open significant new market opportunities and increase our total addressable market in the future.
Biopharma Opportunity
Oncology and rare diseases. We estimate our total addressable biopharma opportunity for our current offerings was $15 billion in 2023 based on three market segments: Discovery, Development, and Deployment. We believe also that our strategic positioning as a healthcare data analytics platform will enable other business opportunities to become available in the future, in, for example, global public health solutions.
Other Market Opportunities Accessible with our Business Model
We believe that our strategic positioning as a healthcare data analytics platform has the potential to enable other business opportunities to become available in the future.
Universal analytics platform for digital health data. Leveraging our global network and customer base, we may enter into collaboration agreements with third-party providers of solutions and services that can be deployed through our SOPHiA DDM Platform and to our customer network, thereby generating new indirect network effects. We believe that we could provide a single, unified analytical workflow through our SOPHiA DDM Platform for instruments generating many kinds of digital health data, such as digital pathology, proteomics, single-cell sequencing and other similar applications.
Global public health solutions. Our SOPHiA DDM Platform could provide a fast and reliable ecosystem to gather data on a global scale and inform public health agencies on significant health-related events, such as pandemics. The COVID-19 pandemic has demonstrated the need for solutions able to harmonize and analyze vast data sets on a global scale, for example, to track the evolution of new variants of the SARS-CoV-2 virus over time and across geographies. This may apply to other infectious diseases, and to human host factors such as detecting specific susceptibility characteristics through genomics and other phenotypic information across populations.
Value-based medicine. As data-driven medicine and multimodality diagnostic approaches are further adopted in the future, we may collaborate with healthcare stakeholders such as payors, providers and integrated healthcare systems to increase the overall affordability of healthcare. We may develop outcomes-based business models in which we enter into risk-sharing agreements with these stakeholders to support the optimal care management of specific patient populations with the goal of achieving better health and economic outcomes.
Our Platform’s Advantages
We believe our SOPHiA DDM Platform has several advantages over alternative genomics analytics platforms as well as other business models aimed at providing data-driven medicine.
66
Unique Value Proposition as a Genomics Analytics Platform
Our SOPHiA DDM Platform enables highly sensitive and specific testing and rapid turnaround time, enabling customers to compute, detect and annotate genomic alterations with high confidence. Our platform and its many applications also allow customers to rapidly build and scale precision medicine operations with different applications. We believe that a crucial characteristic for customers and a key differentiator of our platform is accuracy, leading to quality of insight. The accuracy of our pattern-recognition, AI/ML-based algorithms enable our platform to separate the signal from the noise and standardize data at high-quality levels. Our smart algorithms have high accuracy across applications, from oncology to rare diseases and cardiology, and reduce testing costs by obviating the need for orthogonal assays. The accuracy of our algorithms is a result of the scale and diversity of data within our database.
The following table shows how our SOPHiA DDM Platform performs on genomic variant detection from NGS data across a range of selected genomics applications versus the analytical performance of widely used orthogonal assays such as Sanger Sequencing, MLPA, array CGH and digital PCR.
Our SOPHiA DDM Platform’s Analytical Performance in Selected Current Genomics Applications
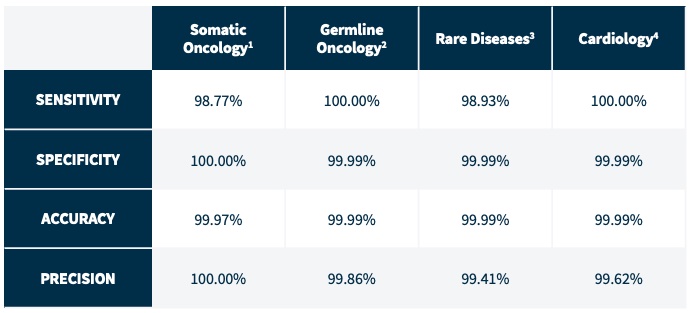
1.Results of the CE-IVD study based on our Solid Tumor Solution (STS) that included data from 6 different sequencing centers and a total of 155 clinical and commercial FFPE samples in which 192 confirmed variants were used as the standard.
2.Results of the CE-IVD study based on our Hereditary Cancer Solution (HCS) that included data from 7 different sequencing centers and a total of 159 clinical and commercial samples in which 1252 confirmed variants were used as the standard.
3.Results based on the clinical exome analysis of the Ashkenazim trio (mother, father and son’s DNA) from the Genome In a Bottle consortium that included data from 2 different sequencing centers and a total of 9 samples (including replicates) in which an average of 6241.2 confirmed variants per sample were used as the standard.
4.Results based on two similar studies that included data from 2 different sequencing centers and a total of 113 clinical and commercial samples in which 833 confirmed variants were used as the standard.
Sensitivity measures how often a test correctly generates a positive result for samples in which a certain genomic variant is present (“true positive” rate). Specificity measures how often a test correctly generates a negative result for samples in which a certain genomic variant is not present (“true negative” rate). Accuracy measures the proportion of tested samples that are correctly classified (“true positives” plus “true negatives”). Precision measures the ability for repeated analyses on the same samples to give similar results.
Broad and Growing Multimodal Application Offering
The breadth of our applications and multimodal capabilities enables our customers to deploy and scale their data-driven medicine operations rapidly and to incorporate additional clinically relevant data sets over time. We believe our platform is uniquely positioned to combine high-quality data at the patient level to generate multimodal insights, leveraging the power of advanced AI/ML models. We have developed proprietary capabilities in AI/ML-enabled exploration of multimodal signatures. Through these, we can unlock the synergistic power of next-generation healthcare data to advance predictive capabilities. We believe that over time, multimodal data will provide a superior means to diagnose and treat disease relative to the current approach focusing on just a single modality.
67
Software-based Platform Facilitates Rapid Global Scaling and Data Collection
We designed our cloud-native software platform to be capable of scaling globally and to use AI to leverage the data that this scale provides. As of December 31, 2023, we served more than 770 hospital, laboratory and biopharma customers globally through our SOPHiA DDM Platform and related solutions, applications, products, and services. We believe that this global footprint is unique and enables us to capture a wide variety of real-world clinical data around the world. The following figure shows our customer base by region as of December 31, 2023.
Our Customer Base by Region
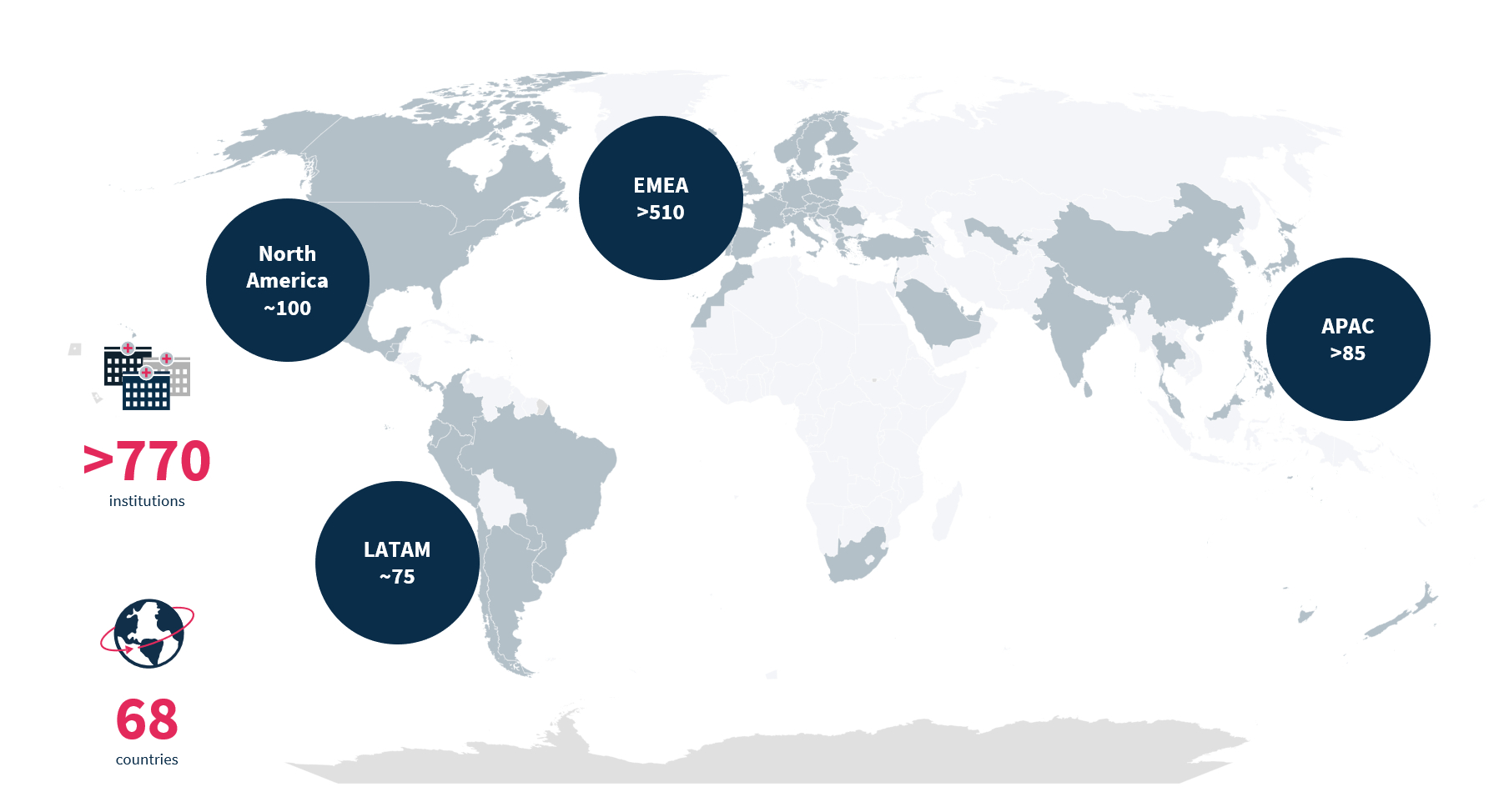
We have been expanding our customer base as well as the volume of data that we analyze. From December 31, 2016 to December 31, 2023, our number of active customers grew from 182 to more than 770. During the same period, the aggregate number of genomic profiles analyzed using our SOPHiA DDM Platform grew from approximately 80,000 profiles to approximately 1,500,000 profiles, recently growing by more than 25,000 new profiles on a monthly basis.
We regularly release platform updates, currently at a pace of once every two to three weeks. Through these updates, we offer our customers either new content, in the form of new applications or improvements to existing applications, such as new functionalities. We believe this update frequency is a competitive advantage in a rapidly evolving precision medicine ecosystem and allows our customers to benefit from new biological discoveries, such as genomic associations, that are reflected on the platform.
Ability to Work with All Stakeholders in the Healthcare Ecosystem
We are empowering our customers through a decentralized model and are able to support clinicians, laboratories and researchers across the healthcare ecosystem. This enables us to benefit from growth across the industry and provide the benefits of our network to different stakeholders. We are also able to collaborate with other product providers in the ecosystem to bundle our solutions to provide differentiated end-to-end solutions. For example, we collaborate with testing kit companies, testing hardware providers, software analytics companies, and diagnostic companies operating with a centralized model. We collaborate with companies including Twist, IDT and Agilent to create an integrated solution using our analytics platform and their library preparation products, including DNA enrichment kits, and with hardware providers such as Hamilton and PerkinElmer. We believe that we can support and collaborate with any industry player for their data analytics needs and are therefore not dependent on any specific business model or industry segment.
68
We believe that this unique ecosystem positioning strategy, coupled with our industry-leading analytics capabilities and our global footprint, position us as a global leading healthcare data analytics company. The following figure shows our unique position in the healthcare ecosystem.
SOPHiA GENETICS’ Unique Position as an “Operating System”

Real-time Visibility into the Healthcare Ecosystem Provides Product and Application Expansion Opportunities
Our strategic positioning as a universal healthcare data analytics platform gives us real-time visibility into data and events in the healthcare ecosystem, including diagnosis, clinical data, customer behavior, performance of third-party technology solutions and other data important to stakeholders. We believe that we are well positioned to provide value to stakeholders across the healthcare ecosystem and to benefit from product and application expansion opportunities.
High Visibility and Predictability into Our Business
Once onboarded onto our SOPHiA DDM Platform, our customers tend to steadily increase their use of our SOPHiA DDM Platform, which offers a level of predictability that helps us project and manage our growth. In addition, customers rarely leave our SOPHiA DDM Platform given that we are generally integrated into their processes. These observed trends hold particularly well for our dry lab and bundle access model customers. We have a revenue churn rate, which we define as the annualized revenues we estimate to have lost from customers who access our platform through our dry lab access and bundle access models and have not generated revenue over the past 12 months in that period based on their average quarterly revenue contributions from point of onboarding as a percentage of total recurring platform revenue, of 4% across our customer base over the year ending December 31, 2023. Furthermore, our customers generally increase their use and adopt new applications of our SOPHiA DDM Platform as our relationship with them grows.
Our Growth Strategy
Our mission is to empower clinicians and researchers around the world to practice data-driven medicine and improve clinical and scientific outcomes. Our growth strategy is to:
•Drive innovation and advancement of our SOPHiA DDM Platform to increase its capabilities and broaden its applications. We plan to continue to invest in scientific innovation to bring new, high-impact content to our customers through regular updates to our platform. This may include new features, new applications, new data modalities and new services. Furthermore, we intend to augment our offering across a multimodality framework, generating novel insights enabled by our expanding data assets, including genomics data, radiomics analysis of medical imaging, clinical data and future additional data modalities such as digital pathology, proteomics, spatial genomics and metabolomics.
•Drive new customer adoption with clinical customers worldwide. We intend to continue to raise awareness of the benefits of data-driven medicine and drive adoption of our platform around the world through our direct sales force, our distributors and our collaborator network. We plan to further penetrate the U.S. market, which we see as our largest opportunity, by significantly investing in our direct sales force to further scale the size of our network, both in terms of the number and types of customers. In addition, we also plan to focus on commercializing our solutions by forming additional collaborations with reference and specialty
69
laboratories. Outside the United States, we believe there is significant growth opportunity across EMEA and Latin America markets, as well as untapped potential in APAC, including in China, India, Korea and Japan. In selected geographies outside the United States, we intend to utilize a hybrid commercial model including direct sales force or direct collaborations and distributors.
•Increase utilization within our clinical customer base. We employ a “land and expand” commercial model that is focused on winning new customers and then driving utilization of our solution by those customers. Once we secure a customer, we use our direct sales force to build further engagement and help that customer profitably increase its testing operations. For example, we may initially support a customer in setting up its NGS testing operations for hereditary cancer screening, including operational support through our set-up programs. Once the customer is fully onboarded onto our SOPHiA DDM Platform, it is then comparatively easier to deploy additional germline testing solutions as well as somatic oncology testing solutions, creating synergies across the offerings and a unified workflow. We also target incremental users within each institution, for example, additional clinicians within a provider across expanded departments such as radiology or pathology.
•Leverage our platform and database to drive adoption by biopharmaceutical companies. We have a distinct sales force focused on biopharma opportunities across the discovery, clinical development and commercialization value chain. We continue to promote our current applications, products, and services, which we believe will strengthen existing collaborations with biopharmaceutical companies as well as lead to new relationships. For example, we may collaborate with a biopharmaceutical company to generate insights on the real-world molecular epidemiology of specific genomic variants relevant for an investigational targeted therapy, including insights on testing trends across our network of customers. This may lead to additional collaborations on a multimodal program to investigate new biomarkers of response to the investigational therapy, a tailored companion diagnostic program, clinical trial recruitment efforts, and market-shaping activities on biomarker testing to support the asset go-to-market strategy. Additionally, we plan to develop new offerings for biopharma as we expand the number and type of new applications and data modalities on our platform. Our biopharma strategy is also highly synergistic to our virtuous cycle.
•Establish and grow industry collaborations across the healthcare ecosystem. We intend to establish new industry collaborations with other companies providing applications, products, and services to our customers. We intend to collaborate with a diverse array of industry participants, including instruments, reagent and software companies in genomics and in other fields such as digital pathology and proteomics. We intend to collaborate with service providers such as centralized laboratory players and interpretation services providers to expand the breadth of our capabilities. We believe that each new collaboration we develop helps facilitate further adoption of our platform, the evolution of the solution we provide to customers and the growth of our network and application and product capabilities. A larger network enables us to continue to collaborate with customers to develop new solutions and to commercialize these solutions, benefiting all users across the healthcare ecosystem.
Commercial
We sell our SOPHiA DDM Platform and related solutions, applications, products, and services to healthcare providers, centralized laboratories and biopharmaceutical companies through our own sales force as well as through distributors and industry collaborators. As of December 31, 2023, our direct sales team consisted of approximately 84 field-based commercial representatives, including sales and business development managers, key account managers and biopharma alliance managers who are engaged in sales efforts and promotional activities towards our customers. We also employ subject matter experts, clinical genomic experts and biopharma operations specialists who provide customer-facing technical and scientific support. As of December 31, 2023, we had a sales presence in 68 countries, including a direct sales presence in 64 countries and 39 countries in which we offer our SOPHiA DDM Platform and related solutions, applications, products, and services through distributors. The following figure shows our global commercial footprint as of December 31, 2023.
70
SOPHiA GENETICS’ Global Footprint – Countries and Sales Force
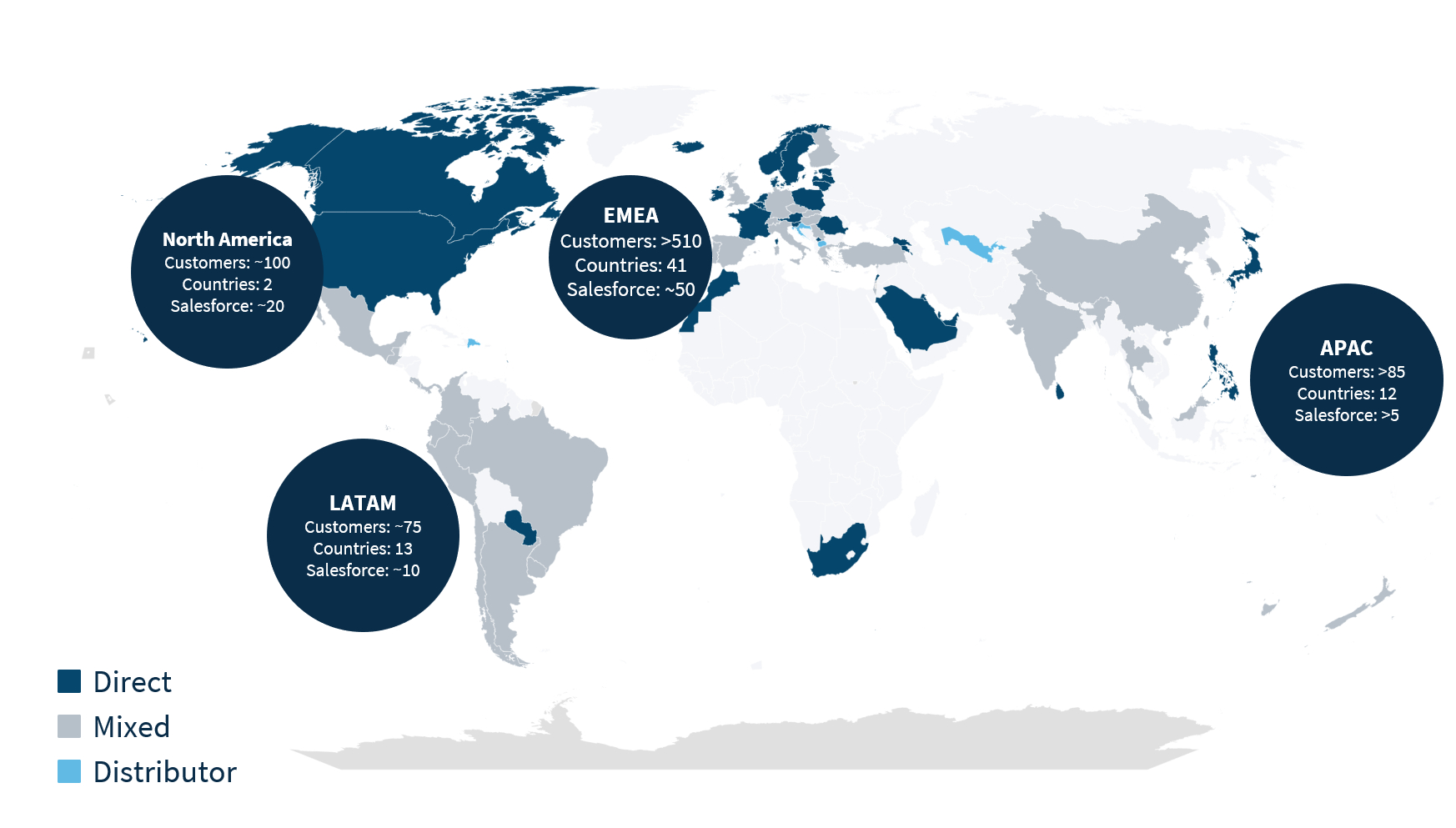
Our initial focus has been on winning clinical customers in order to drive data capture and building our reputation for accuracy and quality in the clinical community. We estimate that there are more than 10,000 laboratories globally that are using NGS instrumentation. We believe that there is significant opportunity to expand our customer base as well as grow utilization of our SOPHiA DDM Platform by our existing customers. Our sales strategy is focused on both attracting new customers to our platform and driving their utilization and adoption of our applications. Once we win a new customer, our direct sales team provides set-up programs to accelerate the adoption of our SOPHiA DDM Platform and facilitates our customers to adopt our platform into their routines.
We started commercializing our biopharma services in 2019. Our initial focus was to establish pilot programs with large pharma and biotech companies to develop customer trust and raise awareness about our offerings. Our biopharma business development and operations team is now focused on developing and scaling joint collaborations and on continuing to refine our application and product offering across large pharma companies, biotech companies and CROs.
Suppliers and Manufacturers
Our platform is a cloud-native software platform. To deploy our platform, we rely on cloud-based service providers. We also collaborate with consumables and hardware suppliers for the bundle access model and with platform customers for the integrated access model.
Platform Suppliers. Our platform production environment currently runs on Microsoft Azure. As our platform architecture is vendor-agnostic, we could readily deploy our solutions onto any cloud infrastructure, as well as on-premise if necessary. We have ongoing research and development projects on all major cloud solution providers, including Microsoft Azure, Amazon Web Services and Google Cloud Platform. This allows us a strong degree of flexibility and helps manage vendor risks.
Consumables and Hardware Suppliers. In the bundle access model, we work with Integrated DNA Technologies (“IDT”), Twist Biosciences (“Twist”), Qiagen, Beckman Coulter, Thermo Fisher Scientific (“Thermo Fisher”) and others for consumables and with Hamilton, PerkinElmer and others for hardware equipment.
71
Platform Customers. In the integrated access model, we route a customer’s samples to selected SOPHiA DDM Platform collaborators who conduct the sequencing process for the customer and upload the resulting data into our SOPHiA DDM Platform. As of December 31, 2023, we collaborated with eleven laboratories across eight countries to provide this service.
We continually assess our dependence on our suppliers and manufacturers and evaluate alternative solutions. We have built our business such that we do not rely on any single supplier or manufacturer, such that we are able to switch suppliers and manufacturers as necessary. We believe that this mitigates risks to our business and provides us the opportunity to drive down costs.
Competition
We operate in a market characterized by rapidly advancing technologies and a strong emphasis on intellectual property. Our main competitors are institutions that collect multimodal data that have developed in-house analytics solutions, such as Tempus Labs, F. Hoffmann-La Roche and Caris Life Sciences, but we believe these competitors also represent our potential customers. In addition, other companies such as Siemens, Koninklijke Philips and Konika Minolta are also positioning themselves in the market with data analytics platform capabilities to build a multimodal world. We also face competition from companies that have developed software analytics platforms for genomics data, such as Agilent, Fabric Genomics, Illumina, Qiagen Digital Insights, Velsera Inc., Congenica Ltd and Thermo Fisher. We believe that our proprietary technology and the agility and the scalability of our platform distinguishes us from other players. We believe that our position as a “universal operating system” enables us to empower and sell to many different players in the ecosystem, including competitors. See “Item 3. Key Information—D. Risk Factors—Risks Related to Our Business and Industry—We face competition from many sources and we may be unable to compete successfully.”
Intellectual Property
Intellectual property is of vital importance in the biotechnology field. Our success depends in part on our ability to obtain and maintain intellectual property and proprietary protection for our technology, defend and enforce our intellectual property rights, preserve the confidentiality of our trade secrets, and operate without infringing, misappropriating or otherwise violating valid and enforceable intellectual property and proprietary rights of others.
We are actively involved in research and development and therefore seek to protect the investments made into the development of our technology by relying on a combination of patents, trademarks, copyrights, trade secrets, including know-how, and license agreements. We also seek to protect our proprietary technology, in part, by requiring our employees, consultants, contractors and other third parties to execute confidentiality agreements and invention assignment agreements and by implementing technological measures and other methods.
Our ability to stop third parties from making, using, selling, offering to sell or importing our platform, applications, services, and products depends on the extent to which we have rights under valid and enforceable patents, trade secrets or other intellectual property and proprietary rights that cover these activities. We pursue intellectual property protection to the extent we believe it would advance our business objectives. Notwithstanding these efforts, there can be no assurance that we will adequately protect our intellectual property or provide any competitive advantage. For more information regarding risks relating to intellectual property, see “Item 3. Key Information—D. Risk Factors—Risks Related to Intellectual Property.”
Patents
Our intellectual property strategy is focused on protecting our ongoing research and development through patents and other intellectual property rights.
As of December 31, 2023, we solely owned 6 issued U.S. patents, 21 pending U.S. patent applications, 41 issued patents and approximately 40 pending patent applications in foreign jurisdictions, including Europe, Canada, Australia, Brazil, China, and India, wherein 4 are pending Patent Cooperation Treaty applications relating to laboratory methods and/or software to provide molecular diagnosis in germline diseases. These include filings for 22 families of utility patents and 2 families of design patents relating to graphical user interfaces. Such issued patents and any patents derived from such applications or applications that claim
72
priority from such applications, if granted, would be expected to expire between (2034 and 2044), excluding any additional term for patent term adjustments.
As of December 31, 2023, our most material patents and patent applications consisted of (i) one issued patent in Israel, one issued patent in Australia, three pending U.S. patent applications and five pending foreign patent applications in Brazil (allowed), Canada, China, Europe, and India relating to our algorithm for next generation sequencing data which is used in our SOPHiA DDM platform, (ii) one issued European Patent and one pending, but allowed, U.S. patent application relating to a method for processing certain genomic data which is used in our SOPHiA DDM platform, (iii) one issued European Patent and one allowed U.S. patent application for a method to improve the accuracy of the estimated length of homopolymer and heteropolymer regions, which is used in our SOPHiA DDM platform, (iv) one pending U.S. patent application, one pending European patent application, and five pending foreign patent applications in Australia, Brazil, Canada, Japan, and South Korea relating to a unique molecular identifier and related analytics workflow which is incorporated in our software applications and products and into our SOPHiA DDM platform, (v) one European pending patent application and one U.S. pending patent application for a method to detect microsatellite instability that is used in our SOPHiA DDM platform, (vi) one pending European patent application, three U.S. pending patent application, and seven pending foreign patent applications in Hong Kong, Australia, Brazil, Canada, China, Japan, and South Korea relating to a method to detect homologous recombination deficiency (“HRD”), which is used in our SOPHiA DDM platform and SOPHiA DDM HRD solution, and (vii) one pending European patent application and one pending U.S. patent application relating to a Limit of Detection aware variant calling method which is not currently used in our software applications and products. Any patents derived from such applications or applications that claim priority from such applications, if granted, would be expected to expire between 2036 and 2042, excluding any additional term for patent term adjustments.
The term of individual patents depends upon the legal term of the patents in the countries in which they are obtained. In most countries in which we file or intend to file, including the United States, the patent term is 20 years from the earliest date of filing a non-provisional patent application. In the United States, a patent’s term may be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the USPTO in examining and granting a patent, or may be shortened if a patent is terminally disclaimed over an earlier filed patent. We cannot be sure that patents will be granted with respect to any current pending patent application or with respect to any patent applications filed by us in the future, and we cannot be sure that any current or future patents will be commercially useful in protecting our platform, applications, products, services, technologies and processes. In addition, any patents that we may hold, whether owned or licensed, may be challenged, circumvented or invalidated by third parties.
Trademarks
The success of our business strategy depends on our continued ability to use our existing intellectual property in order to increase brand awareness and develop our branded services.
As of December 31, 2023, we owned 11 registered U.S. trademarks, 4 pending U.S. trademarks, approximately 131 registered foreign trademarks and 7 pending foreign trademark applications. Our trademark portfolio is designed to protect the brands of our current and future applications and products and includes U.S. trademark registrations for our company name, “SOPHIA GENETICS”, and application and product names, such as “SOPHIA DDM” and “ALAMUT”. We have granted licenses to certain of our trademarks to our domestic and international collaborators.
Trade Secrets
We also rely on trade secrets, including know-how, unpatented technology and other proprietary information, to strengthen our competitive position. We have determined that certain technologies that are not amenable to, or that we do not presently consider appropriate for, patent protection, such as our analysis techniques and analysis generated using our proprietary algorithms in the context of our SOPHiA DDM platform, are better kept as trade secrets in order to protect and maintain our competitive position and aspects of our business and prevent competitors from reverse-engineering or copying our technologies.
We seek to protect trade secrets and confidential and unpatented know-how, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to such knowledge, such as our employees, corporate collaborators, outside scientific collaborators, contract research organizations or manufacturers, consultants, advisors and other third parties. We also seek to enter into confidentiality and
73
invention or patent assignment agreements with our employees and consultants that obligate them to maintain confidentiality and assign their inventions to us. We cannot guarantee that we have entered into such agreements with each party that may have or have had access to our trade secrets or proprietary technology and processes or that the assignment agreements that have been entered into are self-executing. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, or claim ownership in intellectual property that we believe is owned by us. In addition, we take other appropriate precautions, such as physical and technological security measures, to guard against misappropriation of our proprietary information by third parties.
Government Regulation
Laboratory Developed Tests
CLIA and State Laboratory Licensing
CLIA is a U.S. federal law that regulates clinical laboratories that perform testing on specimens derived from humans for the purpose of providing information for the diagnosis, prevention, or treatment of disease, or impairment of, or the assessment of the health of, human beings. CLIA regulations require, among other things, clinical laboratories to obtain a certificate and mandate specific standards in the areas of personnel qualifications, administration, participation in proficiency testing, test management, and quality assurance. CLIA certification is also required for us to be eligible to bill state and federal healthcare programs, if such reimbursement is otherwise available, as well as many private third-party payors, for our applications and products.
In addition to federal certification requirements of laboratories under CLIA, CLIA provides that states may adopt laboratory regulations and licensure requirements that are more stringent than those under federal law. A number of states have implemented their own more stringent laboratory regulatory requirements. Such laws, among other things, establish standards for the day-to-day operation of a clinical laboratory, including the training and skills required of personnel and quality control. For example, New York laws and regulations establish standards for day-to-day operation of a clinical laboratory, including training and skill levels.
We do not currently operate a CLIA-certified laboratory. Our customers are responsible for their own CLIA certification.
Federal Oversight of Laboratory Developed Tests
The laws and regulations governing the marketing of clinical laboratory testing and diagnostic products are evolving and extremely complex and, in many instances, there are no significant regulatory or judicial interpretations of these laws and regulations. Clinical laboratory tests are regulated under CLIA, as administered by CMS, as well as by applicable state laws. In addition, the FDCA defines a medical device to include any instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article, including a component part, or accessory, intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals. Among other things, pursuant to the FDCA and its implementing regulations, the FDA regulates the research, testing, manufacturing, safety, labeling, storage, recordkeeping, pre-market clearance or approval, marketing and promotion, and sales and distribution of medical devices in the United States to ensure that medical products distributed domestically are safe and effective for their intended uses. In addition, the FDA regulates the export of medical devices manufactured in the United States to international markets.
Although the FDA has statutory authority to assure that medical devices are safe and effective for their intended uses, the FDA has generally exercised its enforcement discretion and not enforced applicable regulations with respect to in vitro diagnostics that are designed, manufactured and used within a single laboratory for use only in that laboratory. These tests are referred to as LDTs.
Legislative and administrative proposals proposing to amend the FDA’s oversight of LDTs have been introduced in recent years and we expect that new legislative and administrative proposals will continue to be introduced from time to time. For example on September 29, 2023, the FDA announced its proposal to amend its regulations to make explicit that all IVDs are devices under the Federal Food, Drug, and Cosmetic Act including when the manufacturer of the IVD is a laboratory. In conjunction with the proposed amendment, the FDA has
74
proposed a policy under which it intends to phase out its general enforcement discretion approach for LDTs so that IVDs manufactured by a laboratory would generally fall under the same enforcement approach as other IVDs. It is also possible that legislation could be enacted into law or regulations or guidance could be issued by the FDA which may result in new or increased regulatory requirements. It is also possible that legislation could be enacted into law or regulations or guidance could be issued by the FDA which may result in new or increased regulatory requirements.
AI/ML-Based Medical Software
The FDA recognizes that the traditional paradigm of medical device regulation was not designed for adaptive AI/ML technologies. The FDA has cleared or approved several AI/ML-based software as medical devices (“SaMD”). Typically, these have only included algorithms that are “locked” prior to marketing, where algorithm changes likely require FDA premarket review for changes beyond the original market authorization. However, not all AI/ML-based SaMD are locked; some algorithms can adapt over time. Following distribution, these types of continuously learning and adaptive AI/ML algorithms may provide a different output in comparison to the output initially cleared for a given set of inputs.
The FDA’s Center for Devices and Radiological Health is currently considering a total product lifecycle-based regulatory framework for AI/ML technologies. On January 12, 2021, the FDA released its Artificial Intelligence/Machine Learning-Based Software as a Medical Device Action Plan, which outlines five actions that the FDA intends to take, including:
•further developing the proposed regulatory framework, including through issuance of draft guidance on a predetermined change control plan (for software’s learning over time);
•supporting the development of good machine learning practices to evaluate and improve machine learning algorithms;
•fostering a patient-centered approach, including device transparency to users;
•developing methods to evaluate and improve machine learning algorithms; and
•advancing real-world performance monitoring pilots.
U.S. Medical Device Regulatory Framework
Pursuant to its authority under the FDCA, the FDA has jurisdiction over medical devices, which are defined to include, among other things, IVDs and SaMD. The FDA regulates the research, design, development, preclinical and clinical testing, manufacturing, safety, effectiveness, packaging, labeling, storage, recordkeeping, pre-market clearance or approval, adverse event reporting, marketing, promotion, sales, distribution and import and export of medical devices. Specifically, if the FDA begins to actively regulate LDTs, then, unless an exemption applies, each new or significantly modified medical device we seek to commercially distribute in the United States could require either a premarket notification to the FDA requesting permission for commercial distribution under Section 510(k) of the FDCA (“510(k) clearance”) or approval from the FDA of a PMA application. Both the 510(k) clearance and PMA processes can be resource-intensive, expensive, and lengthy, and require payment of significant user fees.
Device Classification
Under the FDCA, medical devices are classified into one of three classes (Class I, Class II or Class III) depending on the degree of risk associated with each medical device and the extent of control needed to provide reasonable assurances with respect to safety and effectiveness.
Class I includes devices with the lowest risk to the patient and are those for which safety and effectiveness can be reasonably assured by adherence to General Controls for Medical Devices, which require compliance with the applicable portions of the FDA’s Quality System Regulation, facility registration and product listing, reporting of adverse events and malfunctions, and appropriate, truthful and non-misleading labeling and promotional materials. While some Class I devices also require premarket clearance by the FDA through the 510(k) premarket notification process described below, most Class I products are exempt from the premarket notification requirements.
75
Class II devices are those that are subject to the General Controls, as well as Special Controls as deemed necessary by the FDA to ensure the safety and effectiveness of the device. These Special Controls can include performance standards, patient registries, FDA guidance documents and post-market surveillance. Most Class II devices are subject to premarket review and clearance by the FDA. Premarket review and clearance by the FDA for Class II devices is accomplished through the 510(k) premarket notification process.
Class III devices include devices deemed by the FDA to pose the greatest risk, such as life-supporting, life-sustaining devices or implantable devices, in addition to those deemed novel and not substantially equivalent following the 510(k) process. The safety and effectiveness of Class III devices cannot be reasonably assured solely by the General Controls and Special Controls described above. Therefore, these devices are subject to the PMA process, which is generally more costly and time-consuming than the 510(k) process. Through the PMA process, the applicant must submit data and information demonstrating reasonable assurance of the safety and effectiveness of the device for its intended use to the FDA’s satisfaction. Accordingly, a PMA typically includes, but is not limited to, extensive technical information regarding device design and development, preclinical and clinical trial data, manufacturing information and labeling and financial disclosure information for the clinical investigators in device studies. The PMA application must provide valid scientific evidence that demonstrates to the FDA’s satisfaction a reasonable assurance of the safety and effectiveness of the device for its intended use.
The 510(k) Clearance Process
Under the 510(k) clearance process, the manufacturer must submit to the FDA a premarket notification, demonstrating that the device is “substantially equivalent” to a legally marketed predicate device. A predicate device is a legally marketed device that is not subject to a PMA, i.e., a device that was legally marketed prior to May 28, 1976 (pre-amendments device) and for which a PMA is not required, a device that has been reclassified from Class III to Class II or I, or a device that was previously found substantially equivalent through the 510(k) process. To be “substantially equivalent,” the proposed device must have the same intended use as the predicate device, and either have the same technological characteristics as the predicate device or have different technological characteristics and not raise different questions of safety or effectiveness than the predicate device. Clinical data is sometimes required to support substantial equivalence.
After a 510(k) premarket notification is submitted, the FDA determines whether to accept it for substantive review. If it lacks necessary information for substantive review, the FDA will refuse to accept the 510(k) premarket notification. If it is accepted for filing, the FDA begins a substantive review. By statute, the FDA is required to complete its review of a 510(k) notification within 90 days of receiving the 510(k) notification. As a practical matter, clearance often takes longer, and clearance is never assured. Although many 510(k) premarket notifications are cleared without clinical data, the FDA may require further information, including data from samples collected in a clinical setting, to make a determination regarding substantial equivalence which may significantly prolong the review process. If the FDA agrees that the device is substantially equivalent, it will grant clearance to commercially market the device.
If the FDA determines that the device is not “substantially equivalent” to a predicate device, or if the device is automatically classified into Class III, the device sponsor must then fulfill the much more rigorous premarketing requirements of the PMA approval process or seek reclassification of the device through the De Novo classification process.
After a device receives 510(k) clearance, any modification that could significantly affect its safety or effectiveness, or that would constitute a new or major change in its intended use, will require a new 510(k) clearance or, depending on the modification, could require a PMA application. The FDA requires each manufacturer to determine whether the proposed change requires a new submission in the first instance, but the FDA can review any such decision and disagree with a manufacturer’s determination. Many minor modifications are accomplished by an internal letter-to-file in which the manufacture documents its reasoning for why a change does not require premarket submission to the FDA. The letter-to-file is in lieu of submitting a new 510(k) to obtain clearance for such change. The FDA can always review these letters-to-file in an inspection. If the FDA disagrees with a manufacturer’s determination regarding whether a new premarket submission is required for the modification of an existing 510(k)-cleared device, the FDA can require the manufacturer to cease marketing and/or recall the modified device until 510(k) clearance or approval of a PMA application is obtained. In addition, in these circumstances, the FDA can impose significant regulatory fines or penalties for failure to submit the requisite application(s).
76
The De Novo Process
The De Novo classification process is an alternate pathway to classify medical devices that are automatically classified into Class III but which are low to moderate risk. A manufacturer can submit a petition for direct De Novo review if the manufacturer is unable to identify an appropriate predicate device and the new device or new use of the device presents a moderate or low risk. De Novo classification may also be available after receipt of a “not substantially equivalent” letter following submission of a 510(k) to the FDA.
The PMA Approval Process
Following receipt of a PMA application, the FDA conducts an administrative review to determine whether the application is sufficiently complete to permit a substantive review. If it is not, the agency will refuse to file the PMA. If it is, the FDA will accept the application for filing and begin the review. The FDA has 180 days to review a filed PMA application, although the review of an application more often occurs over a significantly longer period of time. During this review period, the FDA may request additional information or clarification of information already provided, and the FDA may issue a major deficiency letter to the applicant, requesting the applicant’s response to deficiencies communicated by the FDA.
Before approving or denying a PMA, an FDA advisory committee may review the PMA at a public meeting and provide the FDA with the committee’s recommendation on whether the FDA should approve the submission, approve it with specific conditions, or not approve it. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
Prior to approval of a PMA, the FDA may conduct inspections of the clinical trial data and clinical trial sites, as well as inspections of the manufacturing facility and processes. Overall, the FDA review of a PMA application generally takes between one and three years, but may take significantly longer. The FDA can delay, limit or deny approval of a PMA application for many reasons, including:
•The device may not be shown safe or effective to the FDA’s satisfaction;
•The data from pre-clinical studies and/or clinical trials may be found unreliable or insufficient to support approval;
•The manufacturing process or facilities may not meet applicable requirements; and
•Changes in FDA clearance or approval policies or adoption of new regulations may require additional data.
If the FDA evaluation of a PMA is favorable, the FDA will issue either an approval letter or an approvable letter, the latter of which usually contains a number of conditions that must be met in order to secure final approval of the PMA. When and if those conditions have been fulfilled to the satisfaction of the FDA, the agency will issue a PMA approval letter authorizing commercial marketing of the device, subject to the conditions of approval and the limitations established in the approval letter. If the FDA’s evaluation of a PMA application or manufacturing facilities is not favorable, the FDA will deny approval of the PMA or issue a not-approvable letter. The FDA also may determine that additional tests or clinical trials are necessary, in which case the PMA approval may be delayed for several months or years while the trials are conducted and data is submitted in an amendment to the PMA, or the PMA is withdrawn and resubmitted when the data are available. The PMA process can be expensive, uncertain and lengthy, and a number of devices for which FDA approval has been sought by other companies have never been approved by the FDA for marketing.
New PMA applications or PMA supplements are required for modification to the manufacturing process, equipment or facility, quality control procedures, sterilization, packaging, expiration date, labeling, device specifications, ingredients, materials or design of a device that has been approved through the PMA process. PMA supplements often require submission of the same type of information as an initial PMA application, except that the supplement is limited to information needed to support any changes from the device covered by the approved PMA application and may or may not require as extensive technical or clinical data or the convening of an advisory panel, depending on the nature of the proposed change.
In approving a PMA application, as a condition of approval, the FDA may also require some form of post-approval study or post-market surveillance, whereby the applicant conducts a follow-up study or follows certain
77
patient groups for a number of years and makes periodic reports to the FDA on the clinical status of those patients when necessary to protect the public health or to provide additional or longer-term safety and effectiveness data for the device. The FDA may also approve a PMA application with other post-approval conditions intended to ensure the safety and effectiveness of the device, such as, among other things, restrictions on labeling, promotion, sale, distribution and use. New PMA applications or PMA supplements may also be required for modifications to any approved diagnostic tests, including modifications to manufacturing processes, device labeling and device design, based on the findings of post-approval studies.
Clinical Trials
In the United States, absent certain limited exceptions, human clinical trials intended to support medical device clearance or approval require an investigational device exemption (“IDE”) application. Some types of studies deemed to present “non-significant risk” are deemed to have an approved IDE–without affirmative submission of an IDE application to the FDA–once certain requirements are addressed and IRB approval is obtained. If the device presents a “significant risk” to human health, as defined by the FDA, the sponsor must submit an IDE application to the FDA and obtain IDE approval prior to commencing the human clinical trials. The IDE application must be supported by appropriate data, such as animal and laboratory testing results, showing that it is safe to test the device in humans and that the testing protocol is scientifically sound. Generally, clinical trials for a significant risk device may begin once the IDE application is approved by the FDA and the study protocol and informed consent are approved by appropriate IRBs at the clinical trial sites. Submission of an IDE will not necessarily result in the ability to commence clinical trials, and although the FDA’s approval of an IDE allows clinical testing to go forward for a specified number of subjects, it does not bind the FDA to accept the results of the trial as sufficient to prove the product’s safety and efficacy, even if the trial meets its intended success criteria.
Such clinical trials must be conducted in accordance with the FDA’s IDE regulations that govern investigational device labeling, prohibit promotion and specify an array of recordkeeping, reporting and monitoring responsibilities of study sponsors and study investigators. Clinical trials must further comply with good clinical practice regulations for IRB approval and for informed consent and other human subject protections. Required records and reports are subject to inspection by the FDA for any clinical trials subject to FDA oversight. The results of clinical testing may be unfavorable, or, even if the intended safety and efficacy success criteria are achieved, may not be considered sufficient for the FDA to grant marketing approval or clearance of a product. The commencement or completion of any clinical trial may be delayed or halted, or be inadequate to support approval of a PMA application or clearance of a 510(k) premarket notification, for numerous reasons.
The Breakthrough Devices Program is a voluntary program intended to expedite the development, assessment and review of certain medical devices that provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating human diseases or conditions for which no approved or cleared treatment exists or that offer significant advantages over existing approved or cleared alternatives. All submissions for devices designated as Breakthrough Devices will receive priority review, meaning that the review of the submission is placed at the top of the appropriate review queue and receives additional review resources, as needed. Although Breakthrough Device designation or access to any other expedited program may expedite the development or approval process, it does not change the standards for approval. Access to an expedited program may also be withdrawn by the FDA if it believes that the designation is no longer supported by data from our clinical development program. Additionally, qualification for any expedited review procedure does not ensure that we will ultimately obtain regulatory clearance or approval for such product.
Research Use Only
In the United States, SOPHiA applications and products labeled and sold for research use only, and not for the diagnosis or treatment of disease, are sold to a variety of parties, including biopharmaceutical companies, academic institutions and molecular laboratories. Because such applications and products are not intended for use in clinical practice in diagnostics, and the applications and products cannot include clinical or diagnostic claims, they are exempt from many regulatory requirements otherwise applicable to medical devices. In particular, while the FDA regulations require that RUO applications and products be labeled “For Research Use Only. Not for use in diagnostic procedures,” the regulations do not otherwise subject such applications and products to the FDA’s pre- and post-market controls for medical devices.
78
A significant change in the laws governing RUO products or how they are enforced may require a change to our business model in order to maintain compliance. For instance, in November 2013 the FDA issued the RUO Guidance, which highlights the FDA’s interpretation that distribution of RUO products with any labeling, advertising or promotion that suggests that clinical laboratories can validate the test through their own procedures and subsequently offer it for clinical diagnostic use as a laboratory, developed test is in conflict with RUO status. The RUO Guidance further articulates the FDA’s position that any assistance offered in performing clinical validation or verification, or similar specialized technical support, to clinical laboratories conflicts with RUO status. If we engage in any activities that the FDA deems to be in conflict with the RUO status held by the applications and products that we sell, we may be subject to immediate, severe and broad FDA enforcement action that would adversely affect our ability to continue operations. Accordingly, if the FDA finds that we are distributing our RUO applications and products in a manner that is inconsistent with its regulations or guidance, we may be forced to stop distribution of our RUO tests until we are in compliance, which would reduce our revenues, increase our costs and adversely affect our business, prospects, results of operations and financial condition. In addition, the FDA’s proposed implementation for a new framework for the regulation of LDTs may negatively impact the LDT market and thereby reduce demand for RUO applications and products.
If the FDA requires marketing authorization of our RUO applications and products in the future, there can be no assurance that the FDA will ultimately grant any clearance or approval requested by us in a timely manner, or at all.
Post-Market Regulation
After a device is cleared or approved for marketing, numerous and pervasive regulatory requirements continue to apply. These include:
•establishment registration and device listing with the FDA;
•QSR requirements, which require manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation and other quality assurance procedures during all aspects of the design and manufacturing process;
•labeling regulations and FDA prohibitions against the promotion of investigational products or the promotion of “off-label” uses of cleared or approved products;
•requirements related to promotional activities;
•clearance or approval of product modifications to 510(k)-cleared devices that could significantly affect safety or effectiveness or that would constitute a major change in intended use of one of our cleared devices, or approval of certain modifications to PMA-approved devices;
•medical device reporting regulations, which require that a manufacturer report to the FDA if a device it markets may have caused or contributed to a death or serious injury, or has malfunctioned and the device or a similar device that it markets would be likely to cause or contribute to a death or serious injury if the malfunction were to recur;
•correction, removal and recall reporting regulations, which require that manufacturers report to the FDA field corrections and product recalls or removals if undertaken to reduce a risk to health posed by the device or to remedy a violation of the FDCA that may present a risk to health;
•the FDA’s recall authority, whereby the agency can order device manufacturers to recall from the market a product that is in violation of governing laws and regulations; and
•post-market surveillance activities and regulations, which apply when deemed by the FDA to be necessary to protect the public health or to provide additional safety and effectiveness data for the device.
Device manufacturing processes are required to comply with the applicable portions of the QSR, which cover the methods and the facilities and controls for the design, manufacture, testing, production, processes, controls, quality assurance, labeling, packaging, distribution, installation and servicing of finished devices intended for human use. The QSR also requires, among other things, maintenance of a device master file, device history file
79
and complaint files. In January 2024, the FDA published a final rule to amend the QSR to align the regulations more closely with the international consensus standard for devices by converging with the quality management system (QMS) requirements used by other regulatory authorities from other countries. Manufacturers have until February 2, 2026, to modify their quality systems to meet QMS Regulations (QMSR).
Manufacturers are subject to periodic scheduled or unscheduled inspections by the FDA. A failure to maintain compliance with the regulatory requirements could result in the shut-down of, or restrictions on, manufacturing operations and the recall or seizure of products. The discovery of previously unknown problems with products, including unanticipated adverse events or adverse events of increasing severity or frequency, whether resulting from the use of the device within the scope of its clearance or off-label by a physician in the practice of medicine, could result in restrictions on the device, including the removal of the product from the market or voluntary or mandatory device recalls.
The FDA has broad regulatory compliance and enforcement powers. If the FDA determines that a manufacturer has failed to comply with applicable regulatory requirements, it can take a variety of compliance or enforcement actions, including the following:
•issuance of warning letters, untitled letters, fines, injunctions, consent decrees and civil penalties;
•requesting or requiring recalls, withdrawals or administrative detention, or seizure of our products;
•imposing operating restrictions or partial suspension or total shutdown of production;
•refusing or delaying requests for 510(k) marketing clearance or PMA approvals of new products or modified products;
•withdrawing 510(k) clearances or PMA approvals that have already been granted;
•refusal to grant export approvals for our products; or
•criminal prosecution.
Authorization to Market In Vitro Medical Devices in the European Economic Area
In the EEA, in vitro medical devices are currently required to conform with the essential requirements of the EU In Vitro Diagnostic Directive (IVDD Directive No 98/79/EC, as amended, the “IVDD”). The scope of 98/79/EC applies to IVD medical devices and accessories, which can include not just reagents and kits but also instruments and software. To demonstrate compliance, ISO 13485 is recognized as the harmonized standard for regulatory quality system compliance. Companies are required to meet the essential requirements of the IVDD.
EU IVD Regulatory Classification
The risk presented by a device determines the classification and therefore the level of control and regulatory review required. Annex II of the IVDD identifies specific device types that are categorized as either high risk (List A) or moderate risk (List B). General IVDs may self-certify without the intervention of a Notified Body in order to affix the CE Marking. Self-test IVDs, because of the greater risk associated with being used by untrained lay users, have special requirements, while all other devices not classified as either List A, List B or self-test are regarded as general IVDs. SOPHiA currently has self-certified applications and products in the EU market through SwissMedic.
On April 5, 2017, the EU adopted the new In Vitro Device Regulation (EU) 2017/746 (the “IVDR”), which repeals and replaces Directive No 98/79/EC effective May 2022. Unlike directives, which must be implemented into the national laws of the EU member states, a regulation is directly applicable, i.e., without the need for adoption of EU member state laws implementing them, in all EEA member states. The IVDR, among other things, is intended to establish a uniform, transparent, predictable and sustainable regulatory framework across the EU for in vitro diagnostic medical devices and ensure a high level of safety and health while supporting innovation.
80
The IVDR will not become fully applicable until five years following its entry into force. Once applicable, the IVDR will, among other things:
•strengthen the rules on placing devices on the market and reinforce surveillance once they are available;
•establish explicit provisions on manufacturers’ responsibilities for the follow-up of the quality, performance and safety of devices placed on the market;
•improve the traceability of medical devices throughout the supply chain to the end-user or patient through a unique identification number; and
•set up a central database to provide patients, healthcare professionals and the public with comprehensive information on products available in the EU.
On October 14, 2021, the EU announced a proposal for an updated phased transitional period for in vitro diagnostic medical devices with a certificate issued by a notified body in accordance with the Directive. The proposal was approved by the EU Parliament and Council on December 15, 2021. The new transitional periods are May 2025 for class D devices, May 2026 for class C devices and May 2027 for class B and A sterile devices. However, the European Parliament is currently considering a proposal announced by the European Commission on January 23, 2024 to extend the transitional periods. The proposed transitional periods are December 31, 2027 for class D devices, December 31, 2028 for class C devices and December 31, 2029 for class B and A sterile devices. Moreover, the application of certain requirements for devices manufactured and used in the same health institution (so-called ‘in-house devices') is delayed by two years until May 2024. If, however, the health institutions prove the unavailability of an equivalent device on the market, the transitional periods will end in May 2028. Any products currently on the market with a CE-IVD label before May 2022 may remain on the market until the new deadline or until the product undergoes a significant change, at which point, it must comply with all the requirements of the IVDR.
Brexit and the Regulatory Framework in the UK
On June 23, 2016, the electorate in the UK voted in favor of leaving the EU, commonly referred to as Brexit. On December 24, 2020, the UK and the EU entered into a Trade and Cooperation Agreement. The agreement sets out certain procedures for approval and recognition of medical products in each jurisdiction. Since the regulatory framework for medical products in the UK covering quality, safety and efficacy of medical products, clinical trials, marketing authorization, commercial sales and distribution of pharmaceutical medical is derived from EU directives and regulations, Brexit could materially impact the future regulatory regime which applies to medical products in the UK, as the UK legislation now has the potential to diverge from EU legislation. It remains to be seen how Brexit will impact regulatory requirements for medical products in the UK in the long-term. The Medicines and Healthcare products Regulatory Agency published detailed guidance for industry and organizations to follow from January 1, 2021, which will be updated as the UK’s regulatory position on medicinal products evolves over time.
A new UKCA mark will replace the CE mark in Great Britain (CE marks or UKNI marks will be required in Northern Ireland). However, all medical devices and IVD's must be registered with the Medicine and Healthcare products Regulatory Agency to be placed on the Great Britain market, CE marked devices can be placed on the Great Britain market until, at least, 30 June 2030, depending on the device type and classification.
EU-Swiss Institutional Framework Agreement
In May 2022, Switzerland adopted the new Ordinance on In vitro Diagnostic Medical Devices (“IvDO”), which replaced the former agreement. Under the new regulation, in order to market products in Switzerland, companies that manufacture diagnostic and medical devices are required to register the products with the Swiss Agency for Therapeutic Products (“Swissmedic”) and label the products accordingly. Additionally, products manufactured in the Swiss facilities of companies are subject to additional registration and representation requirements in order to be marketed within the European Economic Area (“EEA”), including but not limited to the appointment of an Authorized Representative.
81
Other Jurisdictions
Outside the United States, the EU, the UK and Switzerland, regulatory pathways for the marketing of medical devices vary greatly from country to country. In many countries, local regulatory agencies conduct an independent review of medical devices prior to granting marketing approval and may require specific disclosure or localization to access the local market.
For instance, in Brazil all medical devices imported into or distributed within Brazil must first be registered with the Agência Nacional de Vigilância Sanitária (“ANVISA”) or the National Health Surveillance Agency. ANVISA is an autonomous regulatory agency responsible for the regulation and oversight of medical devices and other medical products in Brazil, including the registration of medical devices and the maintenance of a registered products database. The medical device company must be located in Brazil or arrange for a licensed third-party company to be the Brazilian registration certificate title holder. Resolution RDC 36/2015 is the central regulation applicable to registration of in vitro diagnostic devices and Resolution RDC 751/2022, which became effective on March 1, 2023, covers software as medical devices in Brazil, describing the protocol and documents required, including localization into Brazilian Portuguese. Chapter II set forth of RDC 36/2015 the classification scheme, assigning devices to one of four risk classes, based upon various rules enumerated therein. This classification structure is aligned with the EU’s one. If a device fits into more than one risk classification, its final risk class is the one associated with the highest risk level. Class I and Class II registrations do not expire. Class III and IV registrations are valid for ten years. Registration renewals must be initiated no earlier than one year and no later than six months prior to expiration. Manufacturers are also subject to audits to ensure compliance with the Brazilian Good Manufacturing Practices (“BGMP”) prior to receiving authorization to sell from ANVISA. BGMP audits may be fulfilled through other audits by recognized entities through the Medical Device Single Audit Program. RUO products are labeled accordingly and are not subject to these registration requirements. While we are currently able to market our SOPHiA DDM Platform and related solutions, products and services in Brazil, including through our Brazilian subsidiary, any changes to the regulatory framework for RUO products could result in additional costs to us, including expenses related to additional audits, translations and registration fees, or delays in accessing the Brazilian market, including due to the time required to obtain necessary ANVISA approvals.
In Turkey, medical devices are regulated by the Medicines and Medical Devices Agency within Ministry of Health and pursuant to the Medical Device Regulation, the Regulation on Active Implantable Medical Devices and the Regulation on In Vitro Diagnostic Medical Devices. These regulations generally resemble analogous EU directives and regulations. To be sold in Turkey, medical devices must bear a CE mark and must subsequently be registered in the Turkish Ministry’s online database (Turkish Drug and Medical Device National Databank, or TITUBB) in order to be marketed in Turkey. Manufacturers without local presence in Turkey must appoint a Local Authorized Representative. A product will generally be considered a medical device if it is marketed as a medical device in the EU. In recent years, software and mobile application medical devices have been increasing and the Medicines and Medical Devices Agency has considered certain software and mobile applications as medical devices, taking into consideration their intended use. In March 2021, the Product Safety and Technical Regulations Law No. 7223 (the “Product Safety Law”) became effective. The Product Safety Law reconciled some outstanding differences between Turkish and EU product safety standards, providing in part for manufacturer and importer liability in the event that a noncompliant or unsafe product causes harm or damage and mandating recall of such products. Because the current regulatory framework in Turkey closely parallels the EU’s framework, we do not currently experience material difficulties in marketing our SOPHiA DDM Platform and related solutions, applications, products and services in Turkey that are unique to that jurisdiction.
Federal and State Health Care Laws
Federal Physician Self-Referral Prohibition
We are subject to the federal physician self-referral prohibition, commonly known as the Stark Law. Under this law, physicians who have an ownership interest or a compensation relationship with a clinical laboratory may not, unless an exception applies, refer Medicare or Medicaid patients for testing to the laboratory, regardless of the intent of the parties. Similarly, laboratories may not bill Medicare or Medicaid for services furnished pursuant to a prohibited self-referral. Several Stark Law exceptions are relevant to many common financial relationships involving clinical laboratories and referring physicians, including: (1) fair market value compensation for the provision of items or services; (2) payments by physicians to a laboratory for clinical laboratory services; (3) space and equipment rental arrangements that satisfy certain requirements and (4)
82
personal services arrangements that satisfy certain requirements. Penalties for violating the Stark Law include significant denial of payment, the return of funds received for all prohibited referrals, fines, civil monetary penalties, and exclusion from the federal health care programs. In addition, knowing violations of the Stark Law may also serve as the basis for liability under the federal False Claims Act (the “FCA”), which can result in additional civil and criminal penalties. Many states have their own self-referral laws as well, which in some cases apply to all patient referrals, not just government reimbursement programs.
Federal Anti-Kickback Law
The federal Anti-Kickback Statute (AKS) prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving any remuneration (i.e., anything of value), directly or indirectly, overtly or covertly, in cash or in kind, in order to induce or in return either for the referral of an individual, or for purchasing, leasing, ordering, or arranging for the purchase, lease, or order of any healthcare item or service that is reimbursable under any federal health care program, including Medicare or Medicaid. Liability under the AKS may be established without proving actual knowledge of the statute or specific intent to violate it. Federal and state law enforcement authorities scrutinize arrangements between health care providers and potential referral sources to ensure that the arrangements are not designed as a mechanism to induce patient care referrals or induce the purchase or prescribing of particular applications, products, or services. Generally, courts have taken a broad interpretation of the scope of the AKS, holding that the statute may be violated if merely one purpose of a payment arrangement is to induce referrals or purchases. Although there is a number of statutory exceptions and regulatory safe harbors protecting certain common business arrangements and activities from prosecution or regulatory sanctions, the exceptions and safe harbors are drawn narrowly and practices that involve remuneration intended to induce prescribing, purchasing or recommending may be subject to scrutiny if they do not fit squarely within an exception or safe harbor. In addition, the government may assert that a claim that includes items or services resulting from a violation of the AKS constitutes a false or fraudulent claim under the federal civil False Claims Act (FCA), which is discussed in greater detail below. Although the AKS applies only to items and services reimbursable under any federal health care program, a number of states has passed statutes substantially similar to the AKS that apply to all payors. Violations of the AKS are punishable by imprisonment, criminal fines, damages, civil monetary penalties, and exclusion from participation in federal healthcare programs.
False Claims Act
The FCA prohibits, among other things, a person from knowingly presenting, or causing to be presented, a false or fraudulent claim for payment or approval and from making, using, or causing to be made or used, a false record or statement material to a false or fraudulent claim in order to secure payment or retain an overpayment by the federal government. In addition to actions initiated by the government itself, the statute authorizes actions to be brought on behalf of the federal government by a private party having knowledge of the alleged fraud. Because the complaint is initially filed under seal, the action may be pending for some time before the defendant is even aware of the action. If the government intervenes and is ultimately successful in obtaining redress in the matter or if the plaintiff succeeds in obtaining redress without the government’s involvement, then the plaintiff will receive a percentage of the recovery. Finally, the Social Security Act includes its own provisions that prohibit the filing of false claims or submitting false statements in order to obtain payment. Several states have enacted comparable false claims laws which may be broader in scope and apply regardless of payor.
Other Health Care Laws
In addition to the requirements discussed above, several other health care fraud and abuse laws could have an effect on our business
The Social Security Act includes civil monetary penalty provisions that impose penalties against any person or entity that, among other things, is determined to have presented or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent. In addition, a person who offers or provides to a Medicare or Medicaid beneficiary any remuneration, including waivers of co-payments and deductible amounts (or any part thereof), that the person knows or should know is likely to influence the beneficiary’s selection of a particular provider, practitioner or supplier of Medicare or Medicaid payable items or services may be liable under the civil monetary penalties statute. Moreover, in certain cases, providers who routinely waive copayments and deductibles for Medicare
83
and Medicaid beneficiaries, for example, in connection with patient assistance programs, can also be held liable under the AKS and FCA. One of the statutory exceptions to the prohibition is non-routine, unadvertised waivers of copayments or deductible amounts based on individualized determinations of financial need or exhaustion of reasonable collection efforts. The Office of Inspector General of the HHS emphasizes, however, that this exception should only be used occasionally to address special financial needs of a particular patient.
The Health Insurance Portability and Accountability Act of 1996 and its implementing regulations (collectively, HIPAA) imposes criminal and civil liability for, among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private third-party payors, and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement or representation in connection with the delivery of or payment for healthcare benefits, items or services. Like the AKS, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
The Eliminating Kickbacks in Recovery Act (EKRA) is an all-payor anti-kickback law that makes it a criminal offense to pay any remuneration to induce referrals to, or in exchange for, patients using the services of recovery homes, substance use clinical treatment facilities, or laboratories. Although the enactment of EKRA focused on patient brokering and similar arrangements to induce the patronage of substance use recovery and treatment, EKRA’s statutory prohibition is broadly written. The full scope and application of EKRA is uncertain.
The Physician Payments Sunshine Act, enacted as part of the ACA, also imposed annual requirements on manufacturers of certain devices, drugs and biologics to report annually to CMS information related to payments and other transfers of value made to physicians, other health care professionals, and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members.
Also, many states have laws similar to those listed above that may be broader in scope and may apply regardless of payor.
If our operations are found to be in violation of any of the fraud and abuse laws described above or any other laws that apply to us, we may be subject to penalties, including potentially significant criminal, civil and administrative penalties, damages, fines, disgorgement, imprisonment, exclusion from participation in government healthcare programs, contractual damages, reputational harm, integrity oversight and reporting obligations, limitations to the sale of certain applications, products, or services, diminished profits and future earnings, and the curtailment or restructuring of our operations. Efforts to ensure that our internal operations and business arrangements with third parties comply with applicable laws and regulations involve substantial costs. Any action brought against us for violation of these or other laws or regulations, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business.
Coverage and Reimbursement
Sales of our SOPHiA DDM Platform and related solutions, applications, products and services, if approved for IVD use in the United States, may depend substantially on the extent to which health maintenance organizations, managed care organizations, pharmacy benefit managers, federal and state government health administration authorities, private health coverage insurers and other third-party payors provide coverage for and establish adequate reimbursement levels for such solutions, applications, products, and services.
In the United States, many significant decisions about coverage and reimbursement for new diagnostics and medicines are made by CMS, which decides whether and to what extent a new diagnostic or medicine will be covered and reimbursed under Medicare, although it frequently delegates this authority to local Medicare Administrative Contractors (MACs), and by states under their Medicaid programs. Medicare is a federally funded program managed by CMS through MACs and carriers that administer coverage and reimbursement for certain healthcare items and services furnished to the elderly, disabled individuals, and individuals with certain medical conditions. Medicaid is an insurance program for certain categories of patients, including pregnant women, whose income and assets fall below state defined levels, that is both federally and state funded and managed by each state. In the United States, private health insurers and other third-party payors often provide reimbursement for products and services based on the level at which the government provides reimbursement through the Medicare or Medicaid programs for such products and services. It is difficult to predict what CMS,
84
state Medicaid programs, and other third-party payors will decide with respect to coverage and reimbursement for novel platforms, applications, products, and services such as ours.
Outside the United States, the reimbursement process and timelines vary significantly. Certain countries, including a number of member states of the EU, set prices and make reimbursement decisions for diagnostics and pharmaceutical products, or medicinal products, as they are commonly referred to in the EU, with limited participation from the marketing authorization holders or medical device manufacturers, or may take decisions that are unfavorable to the marketing authorization holders or medical device manufacturers where they have participated in the process.
Health Reform
In the United States and some foreign jurisdictions, there has been significant interest in implementing cost-containment programs to limit the growth of government-paid healthcare costs, including price controls and restrictions on reimbursement. Because private payers often follow Medicare and Medicaid coverage policy and payment limitations in setting their own reimbursement rates, any reduction in reimbursement that results from federal legislation or regulation may result in a similar reduction in payments from private payers. We expect to experience pricing pressures in connection with the sale of any products that we develop due to the trend toward managed healthcare, the increasing influence of health maintenance organizations, and additional legislative and regulatory measures.
Such legislative changes in the United States include the Affordable Care Act (ACA), which intended to broaden access to health insurance, reduced or constrained the growth of healthcare spending, enhanced remedies against healthcare fraud and abuse, added new transparency requirements for healthcare and health insurance industries, and imposed additional health policy reforms. We expect that additional federal, state, and foreign healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal, state, and foreign governments will pay for healthcare products and services, which could result in limited coverage and reimbursement and reduced demand for our products, once approved, or additional pricing pressures.
Data Privacy and Security
Health Insurance Portability And Accountability Act and Other U.S. Laws and Regulations
Under HIPAA, as amended by HITECH, HHS has issued security, privacy and breach notification regulations pertaining to PHI used or disclosed by certain entities, including certain health care providers such as us.
Three standards have been promulgated under HIPAA’s and HITECH’s regulations: the Standards for Privacy of Individually Identifiable Health Information, which restrict the use and disclosure of certain individually identifiable health information, the Standards for Electronic Transactions, which establish standards for common healthcare transactions, such as claims information, plan eligibility, payment information and the use of electronic signatures, and the Security Standards for the Protection of Electronic Protected Health Information, which require covered entities and business associates to implement and maintain certain security measures to safeguard certain electronic health information, including the adoption of administrative, physical and technical safeguards to protect such information.
The HIPAA privacy regulations cover the use and disclosure of PHI by covered entities as well as business associates, which are defined to include subcontractors that create, receive, maintain or transmit PHI on behalf of a covered entity or business associate, as well as their covered subcontractors. They also set forth certain rights that an individual has with respect to his or her PHI maintained by a covered entity, including the right to access or amend certain records containing PHI, or to request restrictions on the use or disclosure of PHI. The HIPAA security regulations establish requirements for safeguarding the confidentiality, integrity and availability of PHI that is electronically transmitted or electronically stored. HITECH, among other things, established certain health information security breach notification requirements. A covered entity must notify any individual whose PHI is breached according to the specifications set forth in the breach notification rule. The HIPAA privacy and security regulations establish a uniform federal “floor” for PHI and do not preempt state laws that are more stringent or provide individuals with greater rights with respect to the privacy or security of, and access to, their records containing PHI or insofar as such state laws apply to personal information that is broader in scope than PHI. In addition, individuals (or their personal representatives, as applicable) generally have the
85
right to access test reports directly from laboratories and to direct that copies of those reports be transmitted to persons or entities designated by the individual.
HIPAA authorizes U.S. state attorneys general to file suit on behalf of their residents for violations. Courts are able to award damages, costs and attorneys’ fees related to violations of HIPAA in such cases. While HIPAA does not create a private right of action allowing individuals to file suit against us in civil court for violations of HIPAA, its standards have been used as the basis for duty-of-care cases in state civil suits such as those for negligence or recklessness in the misuse or breach of PHI. In addition, violations of HIPAA could result in significant penalties imposed by the HHS’s Office for Civil Rights. HIPAA also mandates that the Secretary of HHS conduct periodic compliance audits of HIPAA-covered entities, such as us, and their business associates for compliance with the HIPAA privacy and security standards. It also tasks HHS with establishing a methodology whereby harmed individuals who were the victims of breaches of unsecured PHI may receive a percentage of the civil monetary penalty paid by the violator. Our company may receive, as part of the normal course of its business, PHI that is covered by HIPAA. Considering this, we have certain obligations under HIPAA regarding the use and disclosure of any PHI that may be provided to us. Therefore, noncompliance with privacy and security requirements imposed by HIPAA and HITECH could subject us to significant administrative, civil and criminal penalties.
In addition, many states in which we operate have laws that protect the privacy and security of sensitive and personal information. Certain state laws, such as those of California and other states that have adopted versions of the Consumer Data Privacy Act, are more stringent or broader in scope, or offer greater rights to individuals, with respect to sensitive and personal information than federal, international or other state laws, and such laws may differ from each other, which may complicate compliance efforts. In addition, new legislation or constitutional amendments proposed or enacted in various states impose, or have the potential to impose, additional obligations on companies that collect, store, use, retain, disclose, transfer and otherwise process confidential, sensitive and personal information, and will continue to shape the data privacy environment nationally. State laws are changing rapidly and there is discussion in the U.S. Congress of a new federal data protection and privacy law to which we would become subject if it is enacted. All of these evolving compliance and operational requirements impose significant costs that are likely to increase over time, and may require us to modify our data processing practices and policies, divert resources from other initiatives and projects, and could restrict the way applications, products, and services involving data are offered, all of which may have a material and adverse impact on our business, financial condition and results of operations.
Numerous other federal and state laws, including consumer protection laws and regulations, govern the collection, dissemination, use, access to, confidentiality and security of patient health information. We intend to continue to comprehensively protect all personal information and to comply with all applicable laws regarding the protection of such information through our policies and procedures as well as through administrative, physical and technical safeguards.
General Data Protection Regulation and Other Foreign Laws and Regulations
As we are operating worldwide, including in the EU and the EEA member states, the UK, and Switzerland, we have to ensure the compliance of our processing activities with different data protection laws and regulations. Non-compliance with these data protection laws and regulations may not only result in high penalties, it can also cause a loss of reputation and trust.
In the EU and the EEA, processing operations of personal data, including health and genetic personal data, are governed by the GDPR. The GDPR strengthens the powers of the relevant authorities and adds a broad array of requirements for handling personal data, including, for example, requirements to establish a legal basis for processing, higher standards for obtaining consent from individuals to process their personal data, more robust disclosures to individuals and a strengthened individual data rights regime, requirements to implement safeguards to protect the security and confidentiality of personal data that requires the adoption of administrative, physical and technical safeguards, shortened timelines for data breach notifications to appropriate data protection authorities or data subjects, limitations on retention and secondary use of information, increased requirements pertaining to health data and additional obligations when we contract third-party processors in connection with the processing of the personal data. EU and EEA member states are tasked under the GDPR to enact, and have enacted, certain implementing legislation that adds to and/or further interprets the GDPR requirements and potentially extends our obligations and potential liability for failing to meet such obligations. The GDPR, together with national legislation, regulations and guidelines of the EU and
86
the EEA member states governing the processing of personal data, impose strict obligations and restrictions on the ability to collect, use, retain, protect, disclose, transfer and otherwise process personal data. In particular, the GDPR includes obligations and restrictions concerning the consent and rights of individuals to whom the personal data relates, the transfer of personal data out of the EEA, security breach notifications and the security and confidentiality of personal data, including the following:
•Lawfulness, fairness and transparency: Personal data must be processed lawfully, fairly and in a transparent manner.
•Purpose limitation: Personal data must be obtained for specified, explicit and legitimate purposes and not further processed in a manner that is incompatible with those purposes.
•Data minimization: Personal data processed must be adequate, relevant and limited to what is necessary.
•Accuracy: Personal data must be accurate and, where necessary, kept up to date.
•Storage limitation: Personal data must not be kept longer than is necessary.
•Integrity and confidentiality: Appropriate technical and organizational measures must be put in place to guard against unauthorized or unlawful processing, loss, damage or destruction.
The GDPR authorizes fines for certain violations of up to 4% of global annual revenue or €20 million, whichever is greater, and other administrative penalties. The UK has transposed the GDPR into domestic law, with its version of the GDPR that took effect on January 1, 2021, which could expose us to two parallel regimes, each of which potentially authorizes similar fines for certain violations.
In addition, processing of personal data may be governed by the New Federal Act on Data Protection (FADP). The FADP entered into force in September 2023 and provides for data protection principles that are substantially similar to those applied under the GDPR. The purpose of the FADP is to protect the personality rights, including privacy rights, and the fundamental rights of data subjects. The FADP is broad in its material scope and applies to personal data processing activities carried out by federal authorities, private organizations and individual private persons (excluding processing activities for exclusively personal use). The territorial scope of the FADP goes beyond those processing operations carried out in Switzerland, also covering operations that have an effect in Switzerland, even if they originate in another country. Sensitive personal data, including health data, genetic data and biometric data, which unequivocally identify a natural person, are subject to stricter protective measures in various respects. For example, (i) if consent is required, it must be given expressly in the case of processing of sensitive personal data, (ii) controllers must keep record of their data processing activities, (iii) sensitive data must not be disclosed to third parties without justification, (iv) the controller of a data file is obliged to inform the data subject of the collection personal data and (v) disclosing personal data in breach of a confidentiality obligation may be criminally prosecuted. Processing activities by companies must not harm the privacy or personality of the data subject. If the FADP is violated, the FDPIC may request that the processing is fully or partially adjusted, suspended or terminated. Additionally, the new FADP authorize criminal fines for certain violations of up to CHF 250,000. Such fines are mainly imposed upon the individual responsible for the violation and most likely levied against C-level executives and those responsible for the organization’s data protection program. The FADP also authorizes fines of up to CHF 50,000 on the responsible data controller or processor. Fines under the FADP may be imposed in addition to fines under other data protection regimes. As part of our processing activities, we implemented a global compliance plan with applicable laws and regulations, which includes in particular:
•the appointment of a Data Protection Officer;
•the creation of the Data Protection Committee and the Information Security Committee, of which the Data Protection Officer and the Vice President of Information Security are the respective manager;
•the implementation of contractual documentation with our collaborators, aligned with the GDPR requirements;
87
•the preparation of procedures and guidelines, such as a global data protection policy, a data breach responses plan and standard operating procedures for data subject requests; and
•the realization of global data mapping and record by the Data Protection Officer, the Vice President of Information Security and the Compliance Manager.
In particular, the purpose of the Data Protection Committee is to ensure the data and information we process are protected against data protection risks (in compliance with various data privacy regulations and principles of good governance) as well as to assess the effectiveness of our systems, controls and procedures.
The Data Protection Officer is in charge, in particular, of establishing and maintaining processes for receiving, documenting, tracking, investigating and taking actions on all complaints concerning data protection and considering the risks associated with processing operations, taking into account the nature, scope, context and purposes of processing.
For more information regarding risks relating to data privacy and security laws and regulations, see “Item 3. Key Information—D. Risk Factors—Risks Related to Governmental Regulation—We are subject to stringent privacy and, information security laws and regulations and changes in such laws and regulations could adversely affect our business.”
Information Security
We have implemented protections consistent with the ISO/IEC 27001:2013 standard with respect to technical and physical security in an effort to ensure a level of security appropriate to the risk of our processing activities, in particular with respect to protecting the personal data and customer data we process against damage, loss and unauthorized access, use, modification, disclosure, destruction or other misuse. For this purpose, we have what we believe are adequate data breach response plans, disaster recovery plans and security arrangements in place. However, there can be no assurance that our efforts will be successful in protecting against adverse events or successfully mitigating their effects. ISO/IEC 27001:2022 was released on October 25, 2022, replacing ISO/IEC 27001:2013. The new standard allows for a three-year transition period, and we plan to implement the appropriate changes to our security to remain consistent with the updated standard within that timeframe. These changes include: reorganization of controls into four categories / themes from 14 control domains, reduction of total control count by 21, merging of 24 controls from the previous 2013 standard, addition of 11 new controls and introduction of five types of “attributes of control” to improve categorization. For more information regarding risks relating to information security, see “Item 3. Key Information—D. Risk Factors—Risks Related to Our Business and Industry—Cybersecurity or data privacy breaches, other unauthorized or improper access, or (distributed) denial service lack of access (e.g., ransomware, persistent DoS/DDoS) could result in additional costs, loss of revenue, significant liabilities, harm to our brand and decreased use of our SOPHiA DDM Platform and related solutions, applications, products, or services.”
Data Use Rights
As part of our activities, we process thousands of genetic profiles for our customers around the world. As a result, and in accordance with applicable data protection laws and regulations, we may produce aggregate anonymized statistical data from the results of all analyses performed using our proprietary algorithms (“Insights”), which are our sole and exclusive property.
Hence, a distinction is made between customer data (i.e., the data uploaded by our customers on our SOPHiA DDM Platform) on the one hand and other data generated and developed by us (i.e., the results of the performance of our proprietary algorithms, such as Insights) on the other hand. In this respect, Insights are generated using our proprietary algorithms in the context of our SOPHiA DDM Platform and constitute our know-how and trade secrets.
As part of the performance of our services related to our commercial and research and development activities and in accordance with our contractual documentations accepted by our customers and collaborators, we may use our customers’ and collaborators’ data in particular: (i) for the performance of our contractual obligations; (ii) to pseudonymize and anonymize data (and consequently reuse anonymized data); (iii) for statistical, scientific or research purposes; (iv) for providing biomarker identification; (v) for researching, developing, maintaining, or promoting our technology, products or services and (vi) as permitted by applicable laws and regulations.
88
In addition, in accordance with the FADP and the GDPR, we can reuse customer data (including personal data) for further processing activities for statistical purposes. European data protection authorities have previously noted that processing for statistical purposes and for research purposes (including marketing research) are contexts where legitimate purpose can arise. In addition, the processing of personal data for purposes other than those for which the personal data was initially collected should be allowed where the processing is compatible with the purposes for which the personal data was initially collected.
Specific derogations apply for processing operations for statistical purposes, in accordance with the GDPR, as follows:
•personal data can be stored for longer periods insofar as the personal data will be processed solely for statistical purposes subject to implementation of the appropriate technical and organizational measures; information obligations in processing for statistical purposes do not apply if they would involve a disproportionate effort; consideration of this takes into account the number of data subjects and the age of the data, and appropriate safeguards must be adopted; and
•restrictions of the right of a data subject to exercise its “right to erasure” apply if it is likely to significantly impair processing for statistical purposes.
Meanwhile, processing for statistical purposes is subject to certain requirements to:
•set up appropriate safeguards to protect the rights and freedoms of the data subject; and
•implement adequate technical and security measures entrenching the principle of data minimization and using pseudonymized data as the default.
Insights consist of aggregated data providing general trends without identifying individual data subjects and do not contain personally identifiable information. The GDPR does not apply to data that does not relate to or identify an individual, such as aggregated data sets. Consequently, such data sets do not constitute personal data or identifiable information under the GDPR. We believe we have taken reasonable measures to ensure appropriate safeguards and adequate technical and security measures for the processing activities required to generate Insights.
Environmental, Health and Safety Regulations
We are subject to various federal, state, local and foreign environmental, health and safety laws and regulations and permitting and licensing requirements. Such laws include those governing laboratory practices, the generation, storage, use, manufacture, handling, transportation, treatment, remediation, release and disposal of, and exposure to, hazardous materials and wastes, and worker health and safety. Our operations involve the generation, use, storage and disposal of hazardous materials, and the risk of injury, contamination or non-compliance with environmental, health and safety laws and regulations or permitting or licensing requirements cannot be eliminated. Compliance with environmental laws and regulations has not had a material effect on our capital expenditures, financial position, or competitive position.
International Regulations
Many countries in which we may offer any of our diagnostic tests in the future have anti-kickback regulations prohibiting providers from offering, paying, soliciting or receiving remuneration, directly or indirectly, in order to induce business that is reimbursable under any national health care program. In situations involving physicians employed by state-funded institutions or national health care agencies, violation of the local anti-kickback law may also constitute a violation of the FCPA.
The FCPA prohibits individuals and companies, and their employees, agents, and intermediaries from offering, providing, giving or authorizing the provision of, directly or indirectly through a third party, including any potential distributors we may rely on in certain markets, anything of value to a foreign government official with corrupt intent to influence an award or continuation of business or to gain an unfair advantage, whether or not such conduct violates local laws. We can also be held liable for the corrupt or illegal activities of our agents and intermediaries, even if we do not explicitly authorize or have actual knowledge of such activities. In addition, the
89
FCPA requires public companies to maintain accurate books or records and to maintain a system of internal accounting controls.
Violations of the FCPA’s anti-bribery provisions for corporations and other business entities are subject to a fine of up to $2 million, and officers, directors, stockholders, employees, and agents are subject to a fine of up to $100,000 and imprisonment for up to five years. Other countries, including the UK and other member states of the OECD Convention on Combating Bribery of Foreign Public Officials in International Business Transactions, have similar anti-corruption regulations, such as the United Kingdom Bribery Act 2010.
When marketing our diagnostic tests outside of the United States, we may be subject to foreign regulatory requirements governing human clinical testing, prohibitions on the import of tissue necessary for us to perform our diagnostic tests or restrictions on the export of tissue imposed by countries outside of the United States or the import of tissue into the United States, and marketing approval. These requirements vary by jurisdiction, differ from those in the United States and may in some cases require us to perform additional pre-clinical or clinical testing. In many countries outside of the United States, coverage, pricing and reimbursement approvals are also required.
C. Organizational Structure
We have the following wholly owned subsidiaries:
| Name of Subsidiary | Jurisdiction of incorporation | ||||
| SOPHiA GENETICS S.A.S. | France | ||||
| SOPHiA GENETICS LTD | UK | ||||
| SOPHiA GENETICS, Inc. | Delaware (USA) | ||||
| SOPHiA GENETICS Intermediação de Negócios LTDA | Brazil | ||||
| SOPHiA GENETICS PTY LTD | Australia | ||||
| SOPHiA GENETICS S.R.L. | Italy | ||||
D. Property, Plants and Equipment
We do not own any real property. We believe that our facilities meet our present needs and we are continuously reviewing our space requirements. The table below sets forth the sizes and uses of our facilities as of December 31, 2023:
| Location | Primary Function | Approximate Size | ||||||
| A-ONE Park Building B2 Z.A La Pièce 12, 1180 Rolle Switzerland | Office, Laboratory & Warehouse | 65,860 sq ft | ||||||
| Technopole Izarbel 158 Allée Fauste d'Elhuyar 64210 Bidart France | Office | 13,509 sq ft | ||||||
| Bâtiment GIENAH 11 avenue de Canteranne 33600 Pessac France | Office | 3,450 sq ft | ||||||
| 185 Dartmouth Street Boston, Massachusetts 02116 USA | Office | 14,070 sq ft | ||||||
We continuously review our anticipated requirements for facilities and, on the basis of that review, may from time to time acquire or lease additional facilities and/or dispose of existing facilities. We are not aware of any environmental issues or other constraints that would materially impact the intended use of our facilities.
Item 4A. Unresolved Staff Comments
None.
Item 5. Operating and Financial Review and Prospects
90
A. Operating Results
For a comparison of our results of operations and KPIs for the years ended December 31, 2022 and 2021, see “Item 5. Operating and Financial Review and Prospects—A. Operating Results—Results of Operations” in our Annual Report on Form 20-F filed with the SEC on March 7, 2023.
Overview
We are a cloud-native software company in the healthcare space dedicated to establishing the practice of data-driven medicine as the standard of care and for life sciences research. We purposefully built a cloud-native software platform capable of analyzing data and generating insights from complex multimodal data sets and different diagnostic modalities. Our platform standardizes, computes and analyzes digital health data and is used across decentralized locations to break down data silos. This enables healthcare institutions to share knowledge and experiences and to build a collective intelligence. We envision a future in which all clinical diagnostic test data is channeled through a decentralized analytics platform that will provide insights powered by large real-world data sets and AI. We believe that a decentralized platform is the most powerful and effective solution to create the largest network, leverage data and bring the benefits of data-driven medicine to customers and patients globally. In doing so, we can both support and benefit from growth across the healthcare ecosystem.
In 2014, we launched the first application of our platform to analyze NGS data for cancer diagnosis. We have a broad range of applications used by healthcare providers, clinical and life sciences research laboratories and biopharmaceutical companies for precision medicine across oncology, rare diseases, infectious diseases, cardiology, neurology, metabolism and other disease areas. In 2019, we launched our solution for radiomics data that enables longitudinal monitoring of cancer patients and tumor progression throughout their disease journey. In 2022, we unveiled SOPHiA CarePath, our multimodal solution that integrates the capabilities of our genomics and radiomics solutions with additional modalities to further enable clinical decision-making. Today, we believe that our SOPHiA DDM Platform, commercialized under the name “SOPHiA DDM,” is one of the most widely used decentralized analytics platforms globally for clinical genomics. As of December 31, 2023, we served more than 770 hospital, laboratory and biopharma customers globally through our SOPHiA DDM Platform and related solutions, applications, products and services, and our SOPHiA DDM Platform has supported the analysis of more than 1.5 million genomic profiles and has been utilized in clinical trials and research projects discussed in more than 617 peer-reviewed publications. As of December 31, 2023, we had 450 recurring SOPHiA DDM Platform customers (defined as the number of customers who generated revenue during the specified time period, which, in this case, is the twelve months ended December 31, 2023). We commercialize our SOPHiA DDM Platform and related solutions, applications, products, and services as RUO and CE-IVD applications and products. In the United States, our applications and products are labeled and sold for research use only. Because such products are not intended for use in clinical practice in diagnostics and the applications and products cannot include clinical or diagnostic claims, the FDA regulations require that RUO applications and products be labeled “For Research Use Only. Not for use in diagnostic procedures.” In the EU, we have self-certified our applications and products without the intervention of a notified body in order to affix the CE marking.
Our clinical customers primarily include academic and non-academic hospitals and reference and specialty laboratories. Our biopharma customers primarily include pharmaceutical companies, biotechnology companies, and CROs. Our customers are able to access our SOPHiA DDM Platforms through three primary access models: dry lab access, bundle access and integrated access. As of December 31, 2023, we operated a global direct sales team of more than 84 field-based commercial representatives across 64 countries in all four of our major regions of operations (North America, Latin America, EMEA and Asia-Pacific (“APAC”)) and further supplemented our direct sales team with distributors in 39 additional countries. For the years ended December 31, 2023 and 2022, we generated $62.4 million and $47.6 million in revenue, respectively, representing 31% year-over-year growth.
We have funded our operations primarily through equity financings that have generated $498.3 million in gross proceeds as of December 31, 2023 and, to a lesser extent, through revenue generated from the sale of access to our SOPHiA DDM Platform and related licenses, solutions, applications, products, and services. As of December 31, 2023, we had cash and cash equivalents of $123.3 million and no term deposits. Since our inception, we have incurred net losses, which have been significant in recent periods. For the years ended December 31, 2023 and 2022, our net losses were $79.0 million and $87.4 million, respectively. As of
91
December 31, 2023, we had an accumulated deficit of $377.8 million. We expect to continue to incur net losses for the foreseeable future as we continue to devote substantial resources to (i) research and development, in particular to further expand the features, applications and data modalities of our SOPHiA DDM Platform in order to accommodate multimodal data analytics capabilities across a wide range of disease areas, (ii) expanding our selling and marketing efforts for our SOPHiA DDM Platform and related solutions, applications, products, and services, in particular to drive new customer adoption with clinical customers and biopharmaceutical companies, (iii) establishing and maintaining relationships with our collaborators and customers across the healthcare system, and (iv) obtaining regulatory clearance or approval to offer our applications and products as IVD applications and products for diagnostic use. Our ability to achieve profitability depends on the successful commercialization and further development of our SOPHiA DDM Platform and related solutions, applications, products, and services.
Factors Affecting Our Performance
We believe that our financial performance has primarily been driven by, and in the foreseeable future will continue to be primarily driven by, the factors discussed below. While these factors present significant opportunities for our business, they also pose challenges that we must successfully address in order to sustain our growth and improve the results of our operations. Our ability to successfully address these challenges is subject to various risks and uncertainties described elsewhere in this prospectus, particularly in the section titled “Item 3. Key Information—D. Risk Factors.”
Customer Acquisition and Analysis Volume
We principally derive revenue from the use of our SOPHiA DDM Platform by our customers as well as the sales of related licenses, solutions, applications, products, and services. Our analysis volume is dependent on both the acquisition of new customers as well as usage volume from our existing customers. We employ a “land and expand” commercial model focused on winning new customers and then driving subsequent recurring utilization of our solutions by those acquired customers. Once we secure a customer, we use our direct sales force to build further engagement and help that customer increase its testing operations. For example, we may initially support a customer in setting up its NGS testing operations for hereditary cancer screening, including operational support through our set-up programs. Once the customer is fully onboarded on our SOPHiA DDM Platform, it is then comparatively easier to deploy additional germline testing solutions as well as somatic oncology testing solutions, creating synergies across the offerings and a unified workflow. We also target incremental users within each customer, for example, additional clinicians within a provider across expanded departments such as radiology or pathology.
We expect our analysis volume to increase and new customer acquisitions to accelerate as we further expand the features, applications and data modalities of our SOPHiA DDM Platform, expand our presence into new geographies and further penetrate existing geographies, particularly geographies that represent largely under-penetrated opportunities such as North America. We intend to significantly invest in the development of our SOPHiA DDM Platform to accommodate multimodal data analytics capabilities across a wide range of disease areas, including under-penetrated disease areas such as cardiology and neurology, which we believe will allow us to attract new customers and increase usage of our SOPHiA DDM Platform within our existing customer base. While we believe that our existing sales force can support our near-term growth plans, to continue expanding our presence into new geographies and further penetrate existing geographies, we will continue to opportunistically invest in our direct sales force to further scale the size of our network in under-penetrated geographies such as North America, form additional collaborations with reference and specialty laboratories, and collaborate with collaborators and distributors in selected geographies outside of North America.
Revenue Mix
We derive revenue from the use of our SOPHiA DDM Platform by our customers as well as the sales of related licenses, solutions, applications, products, and services. Our clinical customers can access our platform using three different models: dry lab access, bundle access and integrated access. In the dry lab access model, our customers use the testing instruments and consumables of their choice and our SOPHiA DDM Platform and algorithms for variant detection and identification. In the bundle access model, we bundle DNA enrichment kits with our analytics solution to provide customers the ability to perform end-to-end workflows. In the integrated access model, our customers have their samples processed and sequenced through select SOPHiA DDM Platform collaborators within our clinical network and access their data through our SOPHiA DDM Platform. Our
92
biopharma customers can access our SOPHiA DDM Platform through the same three models, but they may also have access to data generated through our SOPHiA DDM Platform in the form of custom reports and analytics.
We have experienced fluctuations in how our clinical customers access our SOPHiA DDM Platform across the three access models. Specifically, certain customers may transition from one access model to another over time. For example, we have observed a trend with certain customers being onboarded onto our platform through the dry lab access model, but, over time, as our relationships with them grow, these customers transition to the bundle access model as customers trust us to curate a set of instruments and consumable products to help increase the accuracy of the analysis they generate. This trend is one illustration of our “land and expand” commercial model, as bundle access is typically a higher revenue-generating model compared to dry lab access based on the incremental value from the sale of consumables and instruments as well as higher platform usage on average for bundle access customers. Certain types of customers are also more likely to access our SOPHiA DDM Platform using one access model compared to other customers. For example, customers who are unable or do not wish to conduct sequencing locally are inclined to use the integrated access model. These customers have historically represented a small percentage of our customer base relative to customers that use the bundle access and dry lab access models. We expect that the revenue contribution from each of the three access models will vary depending on our customer base and the rate of new customer acquisition.
We also derive revenue from the sale of licenses for our Alamut suite of genomics mutations interpretation software. While we view Alamut as a complementary add-on to our SOPHiA DDM platform, there are a number of Alamut users who currently are not customers of our SOPHiA DDM platform. We expect that revenue contribution from Alamut will continue to vary based on the number of stand-alone Alamut users as well as our ability to cross-sell our SOPHiA DDM platform to Alamut users and vice versa.
Seasonality
We typically experience lower usage of our SOPHiA DDM Platform in the first and third quarters compared to the second and fourth quarters, which tend to be seasonally stronger. We typically see relatively lower usage in the first quarter as customers across our global network return from their holidays and new customers are still being onboarded onto our platform. Meanwhile, we believe the relatively lower usage in the third quarter is due to the seasonal slowdown at our customers’ European facilities attributable to vacations and European holiday schedules. As we expand in the North American market, we expect that we will be subject to lower seasonal variations in our usage per customer. We typically experience the highest usage of our SOPHiA DDM Platform in the fourth quarter as we bring new customers and new applications and products with existing customers into routine usage over the course of the year.
Biopharma Expansion
To date, the majority of our revenue is generated through our clinical customers, including academic and non-academic hospitals, and reference and specialty laboratories. However, we see potential for our biopharma business to comprise a more significant portion of our revenues. We began commercializing our biopharma application, product, and service offerings in 2019. While we have the ability to offer a robust package of pre- and post-market solutions to our biopharma customers across the Discovery, Development, and Deployment stages through a broad set of solutions, including SOPHiA Trial Match, SOPHiA Insights, SOPHiA CDx and SOPHiA Awareness, our biopharma business is still nascent with the initial focus on establishing pilot programs with large pharmaceutical and biotech companies to build customer trust and raise awareness about our offerings. We intend to leverage our platform and database to drive adoption by biopharmaceutical companies through our sales force focused on biopharma opportunities across the value chain. In addition, we plan to develop new offerings for biopharma as we expand the number and type of new applications and data modalities on our platform.
SOPHiA CarePath
In 2022, we unveiled SOPHiA CarePath, a new multimodal module on our SOPHiA DDM Platform powered by our artificial intelligence and machine learning algorithms. The module will allow healthcare practitioners to visualize data across multiple modalities (including genomic, radiomic, clinical, and biological) for individual patients in a longitudinal manner and derive additional insights through cohort design and comparison. SOPHiA
93
CarePath has already been deployed as part of our Deep-Lung IV multimodal clinical study on non-small cell lung cancer and is now live at 30 sites across the world. We believe that the module will be adopted by our existing customers and help drive additional adoption of our SOPHiA DDM Platform by clinical institutions and biopharmaceutical companies.
Strategic Acquisitions and Collaborations
We vigilantly monitor the market for potential investments to expand or add key technologies to our offerings that we believe will improve our platform’s ability to address our customers’ needs and catalyze the commercialization of new applications, products, and services. Our investment strategy could take the form of a business acquisition, asset acquisition or strategic licensing of patented technology, all of which may affect our future financial results. For example, our acquisition of Interactive Biosoftware (“IBS”) in 2018 expanded the functionality of our SOPHiA DDM Platform. The Alamut suite of genomics mutation interpretation software is connected to our SOPHiA DDM platform and gives our customers advanced analytics capabilities for a deeper and more informed genomic data interpretation. We view Alamut as a complement to our SOPHiA DDM platform and expect to be able to accelerate our growth by cross-selling our SOPHiA DDM platform to Alamut users and vice versa.
To complement our investment strategy, we have also collaborated, and intend to form additional collaborations, with other product providers in the ecosystem to bundle our solutions to provide differentiated end-to-end solutions. We currently collaborate with testing kit companies, testing hardware providers, software analytics companies and diagnostic companies operating with a centralized model. For example, we formed collaborations with companies including Twist, IDT and Agilent to create an integrated solution using our analytics platform and their library preparation products, including DNA enrichment kits. We continue to regularly evaluate our role in the genomics and radiomics value chain in order to provide both our existing and new customers with a comprehensive product offering, enhance our overall market and competitive position and expand into adjacent untapped markets and new geographies.
Research and Development
A significant aspect of our business is our continued investment in research and development, including new features, new applications, new data modalities and new services. We plan to continue investing in scientific innovation to bring innovative, high-impact content to our customers through regular updates of our platform.
Exchange Rates
We operate internationally and a majority of our revenue, expenses, assets, liabilities and cash flows are denominated in currencies other than our presentation currency, the U.S. dollar and the functional currency of SOPHiA GENETICS SA, the Swiss franc. Our revenues are generated primarily in the U.S. dollar, the euro and Swiss franc and, to a lesser extent, British pound, Australian dollar, Brazilian real, Turkish lira and Canadian dollar depending on our customers’ geographic location. Our expenses are incurred primarily in the U.S. dollar, the euro and Swiss franc and, to a lesser extent, British pound, Australian dollar and Brazilian real. We expect that a part of our revenues and expenses will continue to be denominated in currencies other than the U.S. dollar. Therefore, part of the fluctuations in our operating results in any period may result from changes in exchange rates. We currently do not use any financial instruments to manage our exchange rate risks, which we have been partially mitigating by matching costs in the same foreign currency.
Key Operating Performance Indicators
We regularly monitor a number of key operating performance indicators and metrics to evaluate our business, measure our performance, identify key operating trends and formulate financial projections and strategic plans. We believe that the following metrics are representative of our current business, but the metrics we use to measure our performance could change as our business continues to evolve. Our key operating performance indicators primarily focus on metrics related to our SOPHiA DDM Platform metrics, as platform revenue comprises the majority of our revenues.
As used in this section, the term “customer” refers to any customer who accesses our SOPHiA DDM Platform through the dry lab and bundle access models. We exclude from this definition any customers accessing our SOPHiA DDM Platform using the integrated business model because they tend to use our platform in an ad hoc
94
manner compared to our dry lab and bundle access customers who typically do so in a recurring fashion, generate an immaterial portion of our revenue and analysis volume and constitute a small part of our customer base. We also exclude from this definition customers who only use Alamut through our SOPHiA DDM Platform.
Platform Analysis Volume
Quarterly Platform Analysis Volume from Q1 2017 to Q4 2023*
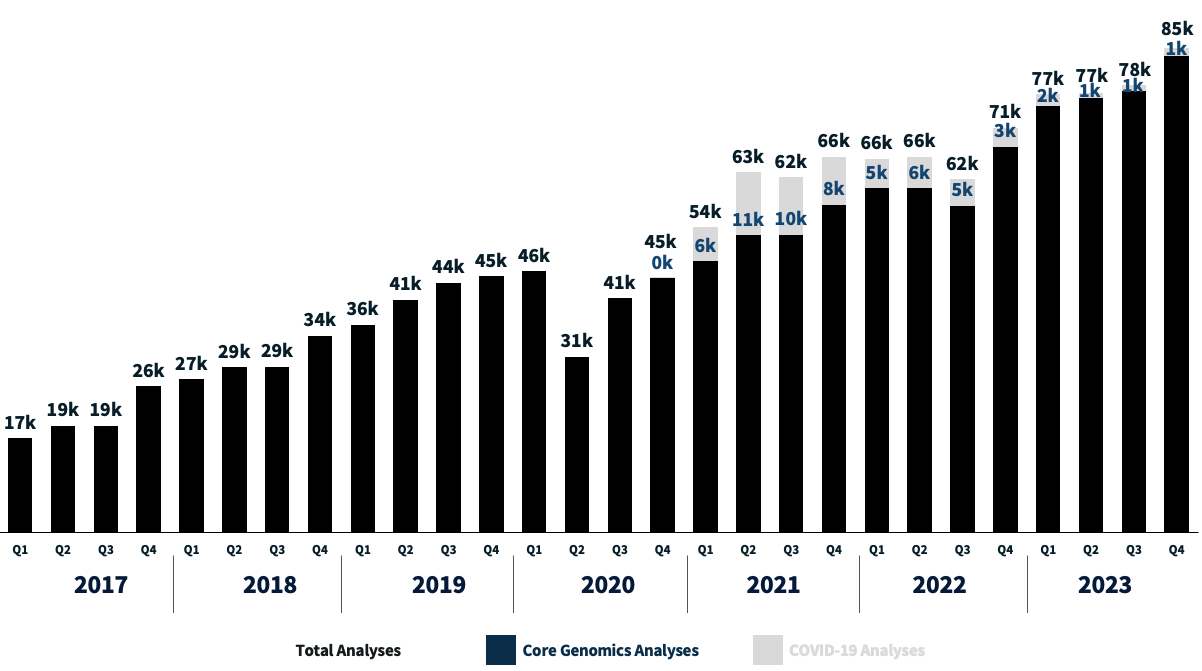
| Year ended December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| SOPHiA DDM Platform analysis volume* | 317,062 | 264,291 | ||||||||||||
*The figures in the bar chart and table above have been adjusted to exclude analyses conducted during the period but for which chargebacks were issued or other adjustments were made to customers after the period. We do not believe that such adjustments are material to the periods presented.
Platform analysis volume represents a key business metric that reflects our overall business performance, as we generate revenue on a pay-per-analysis basis. Platform analysis volume measures the number of analyses that generated revenue to us and were conducted by our customers. Analysis volume is a direct function of the number of active customers and usage rates across our customer base during a specified time period. While our platform analysis volume is a major driver of our revenue growth, other factors, including application and product pricing, access model used and customer size mix, also affect our revenue. Because of that, our revenue may increase in periods in which our analysis volume decreases and vice versa.
Analysis volume increased to 317,062 in the year ended December 31, 2023 from 264,291 in the year ended December 31, 2022. We observed an increase in chargeable analysis volume of 20% for the year ended December 31, 2023, as compared to the year ended December 31, 2022. This increase is primarily attributable to increased usage from our existing customers as well as new customers onboarded onto our platform, with outsized growth across our newer Oncology applications in Homologous Recombination Deficiency (“HRD”), Comprehensive Genomic Profiling (“CGP”), and Liquid Biopsy, albeit from a smaller base relative to some of our more established applications. The increase in analyses was partially offset by the continued reduction of COVID-19 related analyses. We increase prices annually or as necessary throughout the year. While platform analysis volume is a primary driver of our overall revenue, there are other important factors that also contribute
95
to our revenue performance, including access model mix, Alamut license sales, biopharma service revenue and workflow equipment and services revenue. These factors also contributed to year-over-year growth in our overall revenue in 2023.
Total Core Genomics Customers
The following table shows the change in the number of existing Core Genomics Customers, as of December 31, 2023 and 2022, new Core Genomics Customers that went into routine usage during the year ended December 31, 2023 and 2022, and the total number of Core Genomics Customers as of December 31, 2023 and 2022:
| As of December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| Existing Core Genomics Customers | 425 | 415 | ||||||||||||
| New Core Genomics Customers | 25 | 19 | ||||||||||||
| Total Core Genomics Customers | 450 | 434 | ||||||||||||
We track the number of our Core Genomics Customers, defined as the number of customers who generated revenue through usage of our bundle access, dry lab, and integrated access models during the specified time period, as a key measure of our ability to generate recurring revenue from our install base. We further define our Core Genomics Customers as “Existing” or “New” Core Genomics Customers based on the year in which they first accessed our SOPHiA DDM Platform and generated revenue for us.
The numbers exclude customers without any usage of our SOPHiA DDM Platform over the past twelve months and customers who have executed agreements with us that have not generated any revenue to us, including customers that are in the process of being onboarded onto our SOPHiA DDM Platform.
Total Core Genomics Customers increased to 450 as of December 31, 2023 from 434 as of December 31, 2022. The increase is primarily attributable to our continued customer acquisition momentum over the course of the intervening period net of churn.
Net Dollar Retention (NDR)
| As of December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| Net dollar retention (NDR) | 130 | % | 102 | % | ||||||||||
We track net dollar retention for our dry lab and bundle access customers as a measure of our ability to grow the revenue generated from our core genomics customers through our “land and expand” strategy net of customer churn. To calculate net dollar retention, we first specify a measurement period consisting of the trailing two-year period from our fiscal year end. Next, we define a measurement cohort consisting of platform customers who use our dry lab access and bundle access models from whom we have generated revenues during the first month of the measurement period, which we believe is generally representative of our overall dry lab access and bundle access customer base. We then calculate our net dollar retention as the ratio between the revenue generated from this cohort in the second year of the measurement period and the revenue generated in the first year. Any customer in the cohort that did not use our platform in the second year are included in the calculation as having contributed zero revenue in the second year.
Net dollar retention increased to 130% as of December 31, 2023 from 102% as of December 31, 2022. The year-over-year increase in revenue growth momentum is attributable to the continued volume growth across our existing customer base, with regards to usage of existing applications as well as new applications, a slight benefit from favorable foreign exchange movements for revenue generated in key transactional currencies other than the U.S. dollar, particularly the euro and the Swiss franc, and a stable annualized churn rate of 4% in line with expectations, partially offset by a decrease in COVID-19-related revenues as our customers reduced their COVID-19-related operations and business.
96
Components of Results of Operations
Revenue
We generate revenue from goods and services rendered to our clinical customers and from our biopharma customers. Our clinical customers include academic and non-academic hospitals (including comprehensive cancer centers and children’s hospitals), and reference and specialty laboratories. Our biopharma customers include companies along the full biopharma value chain. We group our solutions that we offer our customers into two primary reporting segments: our SOPHiA DDM Platform and workflow equipment and services.
SOPHiA DDM Platform revenue comprises the bulk of our revenue and includes goods and services related to the use of our SOPHiA DDM platform, including our clinical genomics solutions, which span across a broad range of unique applications for analyzing genomic data; our Alamut suite of genomics mutation interpretation software, which gives our clinical customers advanced analytics capabilities for a deeper and more informed genomic data interpretation; and biopharma applications designed to help customers solve bottlenecks across the biopharma value chain, including discovery, clinical development and commercialization; and the sale of third-party instruments and consumables to our bundle access customers.
For clinical customers, our primary pricing strategy for our SOPHiA DDM platform is a pay-per-use model, in which customers access our platform free of charge but pay for each use of our platform. Pricing varies based on our customer mix, as customers require differing levels of customization. For Alamut, our primary pricing strategy is a licensing model, in which customers access our platform for a contracted price. For biopharma customers, we are continuing to refine our pricing strategy since we launched our initial applications for the biopharma market in 2019. We recognize revenue when our customer obtains control of promised goods or services, in an amount that reflects the consideration that we expect to receive in exchange for those goods or services. For revenue generated from our SOPHiA DDM platform customers, we recognize revenue from analyses as the analyses are conducted and revenue from bundled instruments and consumables at the point of delivery. For revenue generated from Alamut licenses, we recognize revenue over the course of the license period. Payments from our customers are typically due up to 180 days from the invoice date. We have a diverse range of customers and no single end customer accounted for more than 5% of our revenue for the years ended December 31, 2023 or 2022.
Workflow equipment and services revenue includes all revenue from the sale of materials and services that do not form part of a contract for the provision of platform services rendered primarily to clinical customers. These include the provision of set-up programs and training and the sale of equipment that are not linked to the use of the platform, such as automation equipment. Set-up programs and training are typically combined with a customer’s first order prior to the customer being onboarded onto our SOPHiA DDM Platform. Revenue from services is generally recognized when the services are performed. Revenue from materials are recognized when control of the goods is transferred to the customer, generally at the time of delivery.
We have demonstrated continued revenue growth during 2023 and 2022 as a result of the continued development of our platform and technology and further penetration of the market. Revenue performance is reflective of the strong foundation that has been built, focused around clinical and biopharma customers. This category of revenue also includes the revenue from the sale of DNA sequencing automation equipment accounted for under IFRS 16, Leases (“IFRS 16”), leasing and the fees charged for the maintenance of this equipment.
Cost of Revenue
Cost of revenue comprises costs directly incurred in earning revenue, including computational and storage-related costs and fees paid to hosting providers, manufacturing costs, materials and consumables, the cost of equipment leased out under finance leases, personnel-related expenses and amortization of capitalized development costs. Capitalized software development costs are amortized using the straight-line method over an estimated life of five years.
While we currently expect increased investments to accelerate growth, we also expect to realize increased efficiencies and economies of scale and undertake cost containment measures to reduce the cost of using cloud infrastructure. Over time, we expect our gross profit margin to increase as we broaden our customer base, increase customer engagement, expand our cloud infrastructure and negotiate additional arrangements
97
with service providers, including with respect to computational and storage-related costs and fees paid to hosting providers. However, in the near term, we expect that our gross profit margin will be adversely impacted by increased computational and storage-related costs and fees as we have purchased, and may be required to continue to purchase, increased capacity at less favorable rates in order to address increased demand for our SOPHiA DDM Platform and related solutions, applications, products, and services. Our cost of revenue as a percentage of revenue may fluctuate from period to period depending on the interplay of the various components of cost of revenue.
Operating Expenses
Operating expenses consist of research and development, selling and marketing, general and administrative, and other operating income (expense), net.
Research and Development Costs
Research and development costs consist of personnel and related expenses for technology, application, and product development, depreciation and amortization, laboratory supplies, consulting services, computational and storage-related costs and fees paid to hosting providers related to research and development and allocated overhead costs. These costs are stated net of government grants for research and development and innovation received as tax credits and net of capitalized costs.
In the short and long term, we expect our research and development costs to increase in absolute dollars, but not necessarily as a percentage of revenue, while we continue to develop, refine and optimize our platform, technology, applications, products, and services as we seek to expand the features, applications and data modalities of our SOPHiA DDM Platform, broaden our customer base and increase customer engagement to drive revenue growth. We expect research and development costs to continue to comprise the largest component of our overall operating expenses. Our research and development costs as a percentage of revenue may fluctuate from period to period due to the timing and extent of such expenses.
Selling and Marketing Costs
Selling and marketing costs consist of personnel and related expenses for the employees of our sales and marketing organization, costs of communications materials that are produced to generate greater awareness and utilization of our platform among our customers, costs of third-party market research, costs related to transportation and distribution of our products and allocated overhead costs, and commissions to sales employees.
In the short term, we expect our selling and marketing costs to increase in absolute dollars and as a percentage of revenue as we seek to broaden our customer base and increase customer engagement to drive revenue growth and as we hire additional sales personnel and related account management and sales support personnel to properly service our growing customer base. However, in the long term, we expect our selling and marketing costs to gradually and modestly decrease as a percentage of revenue. Our selling and marketing costs as a percentage of revenue may fluctuate from period to period due to the timing and extent of such expenses.
General and Administrative Costs
General and administrative costs consist of personnel and related expenses for our executive, accounting and finance, legal, quality, support and human resources functions, depreciation and amortization, professional services fees incurred by these functions, general corporate costs and allocated overhead costs, which include occupancy costs and information technology costs.
In the short term, we expect that our general and administrative costs may vary in absolute dollars and as a percentage of revenue in line with our business needs. However, in the long term, we expect our general and administrative costs to gradually and modestly decrease as a percentage of revenue.
Other Operating Income (Expense), Net
Other operating income (expense), net consists of benefits from the COVID-19 loans and grants with a below-market interest rate (see “Liquidity and Capital Resources—Sources of Capital Resources”), gains and losses
98
related to the disposal of tangible assets, write-offs of intangible assets and other operating income and expenses. We cannot predict the amount of other operating income (expense), net for future periods.
Interest Income (Expense), net
Interest income (expense), net consists of interest income earned on cash and cash equivalents, term deposits and short-term investments, and lease receivables and interest expense incurred on the lease liabilities.
We currently do not use any financial instruments to manage our interest risk exposure.
Foreign Exchange and Other Losses
Foreign exchange and other losses consist of foreign exchange realized and unrealized gains and losses arising principally from intercompany receivable balances in the parent company denominated in U.S. Dollar, whose functional currency is the Swiss franc.
Taxation
We are subject to corporate taxation in Switzerland and other jurisdictions in which we operate, in particular, the United States, France, the UK, Italy, Brazil and Australia, where our wholly owned subsidiaries are incorporated.
Pursuant to a written agreement with the Swiss government, we were exempted from corporate taxes (including capital tax) in Switzerland until December 31, 2022. Effective as of January 1, 2023, we are subject to ordinary cantonal and Swiss federal corporate taxes, including capital tax.
We are entitled under Swiss laws to carryforward any losses incurred for a period of seven years, which could be used to offset future taxable income. As of December 31, 2023, we had total tax loss carryforwards totaling $349.0 million with $344.9 million of which no deferred tax asset has been recorded as they are expected to expire prior to being utilized to offset future incomes. We currently have tax loss carryforwards in Switzerland, the U.S., the U.K. and Brazil, and, of our loss carryforwards, $342.0 million are in Switzerland and can be carried forward through future periods that will expire at various dates between January 1, 2024 and December 31, 2030. There is no certainty that we will make sufficient profits to be able to utilize these tax loss carryforwards in full during the allotted time periods.
Results of Operations
The following table summarizes our results of operations for the years ended December 31, 2023 and 2022:
| Year ended December 31, | Change | |||||||||||||||||||||||||
| (Amounts in USD thousands, except %) | 2023 | 2022 | $ | % | ||||||||||||||||||||||
| Revenue | $ | 62,371 | $ | 47,560 | $ | 14,811 | 31 | % | ||||||||||||||||||
| Cost of revenue | (19,458) | (16,306) | (3,152) | 19 | % | |||||||||||||||||||||
| Gross profit | 42,913 | 31,254 | 11,659 | 37 | % | |||||||||||||||||||||
| Research and development costs | (36,969) | (35,371) | (1,598) | 5 | % | |||||||||||||||||||||
| Selling and marketing costs | (28,423) | (28,267) | (156) | 1 | % | |||||||||||||||||||||
| General and administrative costs | (53,301) | (55,816) | 2,515 | (5) | % | |||||||||||||||||||||
| Other operating income, net | 954 | 377 | 577 | 153 | % | |||||||||||||||||||||
| Operating loss | (74,826) | (87,823) | 12,997 | (15) | % | |||||||||||||||||||||
| Interest income (expense), net | 3,959 | 685 | 3,274 | 478 | % | |||||||||||||||||||||
| Foreign exchange and other losses | (7,628) | (447) | (7,181) | 1606 | % | |||||||||||||||||||||
| Loss before income taxes | (78,495) | (87,585) | 9,090 | (10) | % | |||||||||||||||||||||
| Income tax (expense) benefit | (486) | 136 | (622) | (457) | % | |||||||||||||||||||||
| Loss for the period | $ | (78,981) | $ | (87,449) | $ | 8,468 | (10) | % | ||||||||||||||||||
99
Revenue
| Year ended December 31, | Change | |||||||||||||||||||||||||
| (Amounts in USD thousands, except %) | 2023 | 2022 | $ | % | ||||||||||||||||||||||
| SOPHiA DDM Platform | $ | 60,904 | $ | 45,679 | $ | 15,225 | 33 | % | ||||||||||||||||||
| Workflow equipment and services | 1,467 | 1,881 | (414) | (22) | % | |||||||||||||||||||||
| Total revenue | $ | 62,371 | $ | 47,560 | $ | 14,811 | 31 | % | ||||||||||||||||||
Revenue was $62.4 million for the year ended December 31, 2023, compared to $47.6 million for the year ended December 31, 2022. This increase was primarily attributable to an increase in SOPHiA DDM Platform revenue and partially by a $0.5 million benefit in foreign exchange impact over the course of the year related to the appreciation in the exchange rates between key transactional currencies, particularly the euro and the Swiss franc, and our reporting currency, the U.S. dollar. SOPHiA DDM Platform revenue was $60.9 million for the year ended December 31, 2023 compared to $45.7 million for the year ended December 31, 2022. This increase was primarily attributable to an increase in analysis volume, particularly across our Solid Tumors application portfolio driven by HRD, and an increase in biopharma revenue of $3.8 million. Workflow equipment and services revenue was $1.5 million for the year ended December 31, 2023, compared to $1.9 million for the year ended December 31, 2022. This decrease was primarily attributable to a decrease in workflow automation-related equipment revenue.
Cost of Revenue
| Year ended December 31, | Change | |||||||||||||||||||||||||
| (Amounts in USD thousands, except %) | 2023 | 2022 | $ | % | ||||||||||||||||||||||
| Cost of revenue | $ | (19,458) | $ | (16,306) | $ | (3,152) | 19 | % | ||||||||||||||||||
| Gross profit | $ | 42,913 | $ | 31,254 | $ | 11,659 | 37 | % | ||||||||||||||||||
| Gross margin | 69 | % | 66 | % | ||||||||||||||||||||||
Cost of revenue was $19.5 million for the year ended December 31, 2023, compared to $16.3 million for the year ended December 31, 2022. This increase was primarily attributable to an increase in materials and services-related costs of $1.5 million, an increase in computational and storage-related costs of $0.4 million, and an increase in amortization of capitalized software development expenses of $1.1 million. The increase in gross profit margin to 69% for the year ended December 31, 2023 as compared to 66% for the year ended December 31, 2022 was due to benefits from economies of scale achieved with regards to our computational and storage-related costs and our materials-related costs.
Operating Expenses
| Year ended December 31, | Change | |||||||||||||||||||||||||
| (Amounts in USD thousands, except %) | 2023 | 2022 | $ | % | ||||||||||||||||||||||
| Research and development costs | (36,969) | (35,371) | $ | (1,598) | 5% | |||||||||||||||||||||
| Selling and marketing costs | (28,423) | (28,267) | (156) | 1% | ||||||||||||||||||||||
| General and administrative costs | (53,301) | (55,816) | 2,515 | (5)% | ||||||||||||||||||||||
| Other operating income, net | 954 | 377 | 577 | 153% | ||||||||||||||||||||||
| Total operating expenses | $ | (117,739) | $ | (119,077) | $ | 1,338 | (1)% | |||||||||||||||||||
Research and Development Costs
Research and development costs were $37.0 million for the year ended December 31, 2023, compared to $35.4 million for the year ended December 31, 2022. This increase was primarily attributable to an increase in
100
employee-related expenses, including bonuses and share-based compensation, of $2.7 million for R&D initiatives related to the development of new applications and products, as we retain and hire key positions, and an increase of $1.0 million in professional fees related to the development of new applications and products, partially offset by an increase in the capitalization of research and development costs of $2.0 million.
Selling and Marketing Costs
Selling and marketing costs were $28.4 million for the year ended December 31, 2023, compared to $28.3 for the year ended December 31, 2022. The slight decrease was primarily attributed to a decrease in marketing and conference-related expenses of $0.6 million as we launched more focused campaigns, partially offset by a slight increase of $0.1 million in travel expenses, as our commercial team continues to ramp up travel to customer sites, and an increase in shipping costs of $0.3 million in line with our continued revenue growth.
General and Administrative Costs
General and administrative costs were $53.3 million for the year ended December 31, 2023, compared to $55.8 million for the year ended December 31, 2022. This decrease was primarily attributable to a reduction of $1.9 million of public company-related expenses and $1.9 million of professional fees as we reduced our reliance on external resources, partially offset by an increase of $1.2 million in employee-related expenses, including bonuses and share-based compensation, as we retain and hire key positions.
Other Operating Income , net
Other operating income, net was $1.0 million for the year ended December 31, 2023, compared to $0.4 million for the year ended December 31, 2022.
Interest Income, net
| Year ended December 31, | Change | |||||||||||||||||||||||||
| (Amounts in USD thousands, except %) | 2023 | 2022 | $ | % | ||||||||||||||||||||||
| Interest income (expense), net | $ | 3,959 | $ | 685 | $ | 3,274 | 478 | % | ||||||||||||||||||
Interest income (expense), net was $4.0 million for the year ended December 31, 2023, compared to $0.7 million for the year ended December 31, 2022. The increase was primarily driven by an increase in the interest earned on cash held in short term deposits and money market funds of $3.2 million due to higher interest rates.
Foreign exchange and other losses
| Year ended December 31, | Change | |||||||||||||||||||||||||
| (Amounts in USD thousands, except %) | 2023 | 2022 | $ | % | ||||||||||||||||||||||
| Foreign exchange and other losses | $ | (7,628) | $ | (447) | $ | (7,181) | 1606 | % | ||||||||||||||||||
Foreign exchange and other losses were $7.6 million for the year ended December 31, 2023, compared to foreign exchange and other losses of $0.4 million for the year ended December 31, 2022. This increase in foreign exchange and other losses is primarily driven by an increase unrealized net foreign exchange losses of $8.1 million, primarily related to the outstanding intercompany receivable balances held by the Swiss parent entity that have not been settled with other subsidiaries, partially offset by an increase of $0.9 million in realized net foreign exchange gains. Unrealized gains and losses do not constitute a cash impact until the related transactions are settled.
101
Income Tax (Expense) Benefit
| Year ended December 31, | Change | |||||||||||||||||||||||||
| (Amounts in USD thousands, except %) | 2023 | 2022 | $ | % | ||||||||||||||||||||||
| Income tax (expense) benefit | $ | (486) | $ | 136 | $ | (622) | (457) | % | ||||||||||||||||||
Income tax expense was $0.5 million for the year ended December 31, 2023, compared to a tax benefit of $0.1 million for the year ended December 31, 2022. This tax expense is primarily attributed to current and deferred tax expenses recorded in France, the U.S., and Italy, and, to a lesser extent, to the provision for uncertain tax positions.
Off-Balance Sheet Arrangements and Commitments
We entered into an agreement with Microsoft as of November 1, 2022. As part of the agreement, we have commitments of approximately $69.4 million in computational and hosting-related costs through October 31, 2027.
We did not have, during the periods presented, and we do not currently have, any off-balance sheet arrangements or commitments that may have a material current or future effect on financial condition, changes in financial condition, results of operations, liquidity, capital expenditures, capital resources, or significant components of revenues or expenses.
B. Liquidity and Capital Resources
For a discussion of our liquidity and capital sources and cash flows for the year ended December 31, 2022 and comparison to the year ended December 31, 2021, see “Item 5. Operating and Financial Review and Prospects—B. Liquidity and Capital Resources” in our Annual Report on Form 20-F filed with the SEC on March 7, 2023.
Sources of Capital Resources
Our principal sources of liquidity were cash and cash equivalents totaling $123.3 million as of December 31, 2023 which were held for a variety of growth initiatives and investments in our SOPHiA DDM Platform and related solutions, applications, products and services as well as working capital purposes. Our cash and cash equivalents are comprised of bank and short-term deposits with maturities up to three months. Separately, we did not hold any term deposits with maturities between three and twelve months as of December 31, 2023.
On June 21, 2022 we entered into a credit agreement (the “Credit Facility”) with Credit Suisse SA for up to CHF 5.0 million. Borrowings under the credit facility will bear interest at a rate to be established between us and Credit Suisse at the time of each draw down. Borrowings under the Credit Facility have no restrictions related to its use. As of December 31, 2023, we had no borrowings outstanding under the Credit Facility.
In August 2023, we established an at-the-market offering program pursuant to which we may sell, from time to time, ordinary shares having an aggregate offering price of $50 million. For the year ended December 31, 2023, we did not sell any ordinary shares under this program.
We have funded our operations primarily through equity financing and, to a lesser extent, through revenue generated from the sale of access to our SOPHiA DDM Platform and related licenses and services. Invoices for our products and services are a substantial source of revenue for our business, which are included on our consolidated balance sheet as trade receivables prior to collection. Accordingly, collections from our customers have a material impact on our cash flows from operating activities. As we expect our revenue to grow, we also expect our accounts receivable and inventory balances to increase, which could result in greater working capital requirements.
Uses of Capital Resources
Since our inception, we have incurred net losses, which have been significant in recent periods. For the years ended December 31, 2023 and 2022, our net losses were $79.0 million and $87.4 million, respectively. As of
102
December 31, 2023, we had an accumulated deficit of $377.8 million. Our primary use of capital sources has been to fund our operations and grow our business.
Operating Capital Requirements
We expect to continue to incur net losses for the foreseeable future as we continue to devote substantial resources to research and development, in particular, to further expand the applications and modalities of our SOPHiA DDM Platform in order to accommodate multimodal data analytics capabilities across a wide range of disease areas; selling and marketing efforts for our SOPHiA DDM Platform to establish and maintain relationships with our collaborators and customers; and obtaining regulatory clearances or approvals for our SOPHiA DDM Platform and our applications, products, and services. We believe that our existing cash and cash equivalents will be sufficient to meet our working capital and capital expenditure needs for at least the next 12 months. Our future funding requirements will depend on many factors, including:
•our ability to achieve revenue growth;
•our ability to secure any required regulatory clearance or approval for additional features, applications and data modalities of our SOPHiA DDM Platform and related solutions, applications, products, and services;
•the ability of our customers and collaborators to secure any required regulatory clearance or approval for their product candidates, other products, and services the development of which they rely on our SOPHiA DDM Platform and related solutions, applications, products and services;
•our rate of progress in, and cost of the sales and marketing activities associated with, establishing adoption of our SOPHiA DDM Platform and related solutions, applications, products, and services;
•the rate of progress in establishing payor coverage and reimbursement arrangements with domestic and international commercial third-party payors and government payors by us with respect to our application and products, if approved for IVD use, and by our customers and collaborators, with respect to their product candidates, other products, and services;
•the cost of expanding our research and development; manufacturing and laboratory operations; and applications, products, and services offerings;
•the cost of building out our facilities, including our corporate headquarters in Switzerland and our locations around the world;
•our ability to maintain and expand our collaborations with biopharmaceutical companies, both advanced and early stage, and reference and specialist laboratories;
•our rate of progress in, and cost of research and development activities associated with, early research and development efforts;
•the effect of competing technological and market developments;
•market acceptance of our platform, solutions, applications, products, and services;
•costs related to international expansion; and
•the potential cost of, and delays in, application and product development as a result of regulatory oversight.
Unless and until we can generate sufficient revenue to finance our cash requirements, which may never happen, we may seek additional capital through a variety of means, including through public and private equity offerings and debt financings, credit and loan facilities and collaborations. Additional funds may not be available when we need them or on terms that are acceptable to us. See “Item 3. Key Information—D. Risk Factors—Risks Related to Our Financial Position and Capital Requirements.”
103
Cash Flows
The following table summarizes our cash flows for the years ended December 31, 2023 and 2022:
| Year ended December 31, | ||||||||||||||
| (in USD thousands) | 2023 | 2022 | ||||||||||||
| Net cash from (used in): | ||||||||||||||
| Operating activities | $ | (48,575) | $ | (70,093) | ||||||||||
| Investing activities | 8,320 | 41,973 | ||||||||||||
| Financing activities | (2,817) | (1,568) | ||||||||||||
| Net (decrease) increase in cash and cash equivalents | $ | (43,072) | $ | (29,688) | ||||||||||
| Effect of exchange rate differences on cash and cash equivalents | $ | 5,018 | $ | (1,969) | ||||||||||
Operating Activities
For the year ended December 31, 2023, net cash used in operating activities was $48.6 million, primarily attributable to our net loss of $79.0 million, which was reflective of our continued research and development of and commercialization activities for our SOPHiA DDM Platform, partially offset by an increase in working capital and interest received.
For the year ended December 31, 2022, net cash used in operating activities was $70.1 million, primarily attributable to our net loss of $87.4 million, which was reflective of our continued research and development of and commercialization activities for our SOPHiA DDM platform and an increase in general and administrative expenses as we transitioned to a public company, offset by an increase in share-based compensation.
Investing Activities
For the year ended December 31, 2023, net cash provided by investing activities was $8.3 million, primarily attributable to the proceeds associated with the maturity of investments in term deposits partially offset by our capital expenditures to support research and development and revenue-generation activities.
For the year ended December 31, 2022, net cash provided by investing activities was $42.0 million, primarily attributable to the proceeds associated with the maturity of investments in term deposits partially offset by our capital expenditures to support research and development and revenue-generation activities and investments in new term deposits.
Financing Activities
For the year ended December 31, 2023, net cash used in financing activities was $2.8 million, primarily attributable to cash payments related to our leases, partially offset by proceeds from the exercise of stock options.
For the year ended December 31, 2022, net cash used in financing activities was $1.6 million, primarily attributable to cash payments related to our leases, partially offset by proceeds from the exercise of stock options.
C. Research and Development, Patents and Licenses
See “Item 4. Information on the Company—B. Business Overview” and “Item 5. Operating and Financial Review and Prospects—A. Operating Results—Results of Operations.”
D. Trend Information
See “Item 5. Operating and Financial Review and Prospects—A. Operating Results.”
104
E. Critical Accounting Estimates
The preparation of financial statements in conformity with IFRS Accounting Standards requires the use of accounting estimates. It also requires management to exercise judgment in applying our accounting policies. Disclosed below are the areas which require a high degree of judgment, significant assumptions and/or estimates.
Revenue
We recognize revenue when control of promised goods or services is transferred to customers in an amount that reflects the consideration that is expected to be received for those goods or services. Significant judgment is required to determine the stand-alone selling price (“SSP”) for each performance obligation in our SOPHiA DDM Platform, the amount allocated to each performance obligation and whether it depicts the amount that we expect to receive in exchange for the related application use, product, and/or service. As the selling prices of our analyses are highly variable, we estimate SSP of our analyses using the residual approach when the analyses are sold with other products and services and observable SSPs exist for the other products and services. While the majority of sales agreements contain standard terms and conditions, we do enter into biopharma contracts that contain multiple products or services or non-standard terms and conditions.
We enter into arrangements with multiple performance obligations where it could be difficult to determine whether there is more than one performance obligation under a sales agreement; in such cases, how and when revenue should be recognized is subject to certain estimates or assumptions. Should these judgments and estimates not be correct, revenue recognized for any reporting period could be adversely affected.
SOPHiA DDM Platform
The majority of SOPHiA DDM Platform revenue is derived from each use of our SOPHiA DDM Platform by customers to generate analyses on their patient data. Analysis revenue is recognized as analysis results are made available to the customer on our SOPHiA DDM Platform. Contract assets are recognized on the balance sheet as accrued contract revenue for any analyses performed by customers that have not been invoiced at the reporting period date. Any payments received in advance of customers generating analyses are recorded as deferred contract revenue until the analyses are performed.
Customers use our SOPHiA DDM Platform to perform analyses under three different models: dry lab access; bundle access; and integrated access.
For dry lab arrangements, customers use the testing instruments and consumables of their choice and our SOPHiA DDM Platform and algorithms for variant detection and identification. In these arrangements, we have identified one performance obligation, which is the delivery of the analysis result to the customer.
For bundle arrangements, customers purchase a DNA enrichment kit along with each analysis. Customers use the DNA enrichment kit in the process of performing their own sequencing of each sample. Customers then upload their patient data to our SOPHiA DDM Platform for analysis. In these arrangements, we have identified two performance obligations: the delivery of the DNA enrichment kits and the performance of the analyses. Revenue is recognized for the DNA enrichment kits when control of products has transferred to the customer, which is generally at the time of delivery, as this is when title and risk of loss have been transferred. Revenue for the performance of the analyses is recognized on delivery of the analysis results to the customer. Refer to “—Arrangements with multiple performance obligations” below for how revenue is allocated between the performance obligations.
Deferred contract revenue balances relating to analyses not performed within 12 months from the date of the delivery date are recognized as revenue. This policy is not based on contractual conditions but on the Company’s experience of customer behavior and expiration of the kits associated with the analyses.
For integrated arrangements, customers have their samples processed and sequenced through selected SOPHiA DDM Platform partners within the clinical network and access their data through our SOPHiA DDM Platform. We have identified one performance obligation, which is delivery of the analysis results to the customer.
105
Through our SOPHiA DDM Platform, we also sell access to our Alamut software application. Some arrangements with customers allow customers to use Alamut as a hosted software service over the contract period without the customer taking possession of the software. Other customers take possession of the software, but the utility of that software is limited by access to our proprietary SOPHiA database, which is provided to the customer on a fixed term basis. Under both models, revenue is recognized on a straight-line basis over the duration of the agreement.
We also derive revenue from our SOPHiA DDM Platform by providing services to biopharma customers who engage us to (i) develop and perform customized genomic analyses and/or (ii) access the database for use in clinical trials and other research projects.
The Company does enter into biopharma contracts that contain multiple products or services or non-standard terms and conditions. The biopharma contracts are generally unique in nature and each contract is assessed upon execution. Contracts may contain multiple performance obligations or performance obligations that are recognized over time, at a point in time upon achievement of milestones, or through a combination of both methods depending on the Company’s ability to satisfy the requirements to recognize revenue over time and reasonably estimate the amount of revenue to recognize. See “Arrangements with multiple performance obligations” below for further discussion on treatment of biopharma contracts.
Generally, the primary performance obligation in these arrangements is the delivery of analysis results in the form of a final report, resulting in revenue being recognized, in most cases, upon the issuance of the final report or successful recruitment of clinical trial participants.
Workflow Materials and Services
Revenue from workflow materials and services includes all revenue from the sale of materials and services that do not form part of a contract for the provision of platform services. These include the provision of set-up programs and training and the sale of kits and tests that are not linked to use of the platform. Set-up programs and training are typically combined with a customer’s first order prior to the customer beginning to use our SOPHiA DDM Platform.
Revenue from services is generally recognized when the services are performed. Revenue from materials is recognized when control of the goods is transferred to the customer, generally at the time of delivery. This category of revenue also includes the revenue from the sale of DNA sequencing automation equipment accounted for under IFRS 16, Leases (“IFRS 16”) and the fees charged for the maintenance of this equipment.
Arrangements with Multiple Performance Obligations
For multi-element arrangements the following steps are performed to determine the amount of revenue to be recognized and when it should be recognized: (1) identify the contract or contracts; (2) determine whether the promised goods or services are performance obligations, including whether they are distinct in the context of the contract; (3) measure the transaction price, including the constraint on variable consideration; (4) allocate the transaction price to the performance obligations based on estimated selling prices; and (5) recognize revenue when (or as) each performance obligation is satisfied.
We have determined that the stand-alone selling prices for services and DNA enrichment kits are directly observable. For set-up programs and training services sold along with dry lab arrangements or bundle arrangements, the stand-alone selling price of these services is determined on a time and materials basis. For DNA enrichment kits sold as part of a bundle, the stand-alone selling price is based on an expected cost-plus-margin approach.
We have determined that the stand-alone selling price for the analyses, in both a dry lab arrangement and bundle arrangement, is highly variable and therefore a representative stand-alone selling price is not discernible from past transactions. As a result, the residual approach is used to determine the stand-alone selling price of the analyses in dry lab arrangements that include services and in bundle arrangements that include DNA enrichment kits and, in some cases, services.
We also have a small number of bundle contracts with a fixed term, generally four years, that also include providing the customer with DNA sequencing automation equipment, which we have determined is an IFRS 16 leasing component. In these arrangements, we provide DNA sequencing automation equipment to the customer
106
over the fixed term and at completion of the contract term the customer takes possession of the equipment. We have determined that we are a dealer lessor and provision of this equipment to the customer is classified as a finance lease. As a result, upon delivery of leased equipment at the inception of the agreement, a selling profit is recognized based on the fair value of the underlying equipment less the cost of the equipment. Over the term of the agreement, the minimum lease payment is deducted from the proceeds of the bundle sales in order to reduce the net investment in the corresponding lease receivable over the contract term and interest income is recognized as the discount on the lease receivable unwinds. The remaining proceeds from the contract are accounted for under IFRS 15, Revenue from Contracts with Customers (“IFRS 15”), using the policies described above.
The Company assesses biopharma contracts upon execution of each contract given their project based nature. The Company establishes each performance obligation within the contract and determines the appropriate value to be ascribed to be each performance obligation. When relevant to the biopharma contract, the Company utilizes previously established SSPs for its dry lab and bundle solutions or other services. When the performance obligation(s) is only specific to the biopharma contract, the Company utilizes all available information to reasonably estimate the correct value allocated to each performance obligation
Capitalized Internal Software Development Costs
All work performed by our research and development personnel is tracked and allocated to codes based on the nature of the work done. The hours spent are costed on the basis of the related salaries, benefits and share-based compensation. The cost of work attributable to the development of new data analytics solutions and services or to the improvement or enhancement of existing solutions and services is capitalized, once it is evident that the project is technically and financially feasible and that it will bring economic benefits to us. Capitalized software development costs are amortized using the straight-line method over an estimated life of five years.
Costs incurred for research, for development projects that do not meet the capitalization criteria, for maintenance and for minor modifications are expensed when incurred and presented as research and development costs. Other, administrative costs are expensed and presented as general and administrative costs.
Share-Based Compensation
For the years ended December 31, 2023 and 2022, we granted share options under one plan - the SOPHiA GENETICS 2021 Equity Incentive Plan (the “2021 Equity Incentive Plan” or the “2021 EIP”). Under this plan, directors may offer options to directors, employees and advisors. The exercise price of the share options is set at the time they are granted. Options, once vested, can be exchanged for an equal number of ordinary shares.
Measuring the Cost of Share Options
The fair value of the options outstanding under all plans are measured at each reporting date using an adjusted form of the Black-Scholes option pricing model, taking into account the terms and conditions upon which the options were granted.
For options up to September 2020, the fair value at grant date is independently determined using an adjusted form of the Black-Scholes option pricing model that takes into account the strike price, the fair value of the share at grant date, the expected life of the award, the expected price volatility of the underlying share, the risk-free interest rate for the term of the award and the expected dividend yield. For options granted from September 2020 to the IPO in July 2021, the fair value at grant date is based on a probability-weighted expected returns method that takes account of both the value derived by using an adjusted form of the Black-Scholes option pricing model, as described above, and a discounted estimate of the price that might be achieved in a future transaction. For options granted after the IPO in July 2021, the fair value at grant date is determined using a Black-Scholes option pricing model.
107
The key inputs used in the valuation model for the stock options are outlined below.
| EIP 2021 | ||||||||||||||||||||||||||
| Year Ended December 31, | ||||||||||||||||||||||||||
| 2023 | 2022 | |||||||||||||||||||||||||
| Weighted average strike price (in USD) | $4.44 | $6.03 | ||||||||||||||||||||||||
| Share price at grant date (in USD) | $2.53 | - | $4.72 | $2.06 | - | $8.36 | ||||||||||||||||||||
| Expected life of share options (years) | 5.50 | - | 7.00 | 5.50 | - | 7.00 | ||||||||||||||||||||
| Expected volatility (%) | 69.50 | % | - | 74.96% | 62.65 | % | - | 69.43% | ||||||||||||||||||
| Risk free interest rate (%) | 3.45 | % | - | 4.67% | 2.42 | % | - | 4.00% | ||||||||||||||||||
| Dividend yield (%) | —% | —% | ||||||||||||||||||||||||
Subsequent to the IPO, we used the most recent public market close price of our stock on the date of grant as the strike price.
Recognizing the Cost of Share Options
At each reporting date, we take a charge for the vested options granted and for partially earned but non-vested portions of options granted. This results in a front-loaded charge to the statement of income/loss. In addition, at each reporting date we reappraise our estimate of the likelihood and date of a future transaction that would cause all outstanding options to vest and, if necessary, accelerates the recognition of the unrecognized cost in the statement of income/loss. We account for these plans as equity-settled. The charge to the statement of income/loss therefore results in a corresponding credit being booked to “Other reserves” within equity.
Goodwill Impairment Testing
We operate as one segment or as a single cash-generating unit (“CGU”). As a single CGU, goodwill is tested by considering its recoverability in terms of the entire business. We assess the recoverable value of goodwill by comparing our equity value, either from observable market prices or based on discounted cash flow forecasts, to the net assets as reported in our consolidated financial statements. The value as of October 1, 2023 was based on our market capitalization which is a factor of our outstanding shares multiplied by the price of the Company’s stock on October 1, 2023.
The value as of October 1, 2022 was based on our discounted cash flow projections, which in turn were based on historical results and ratios updated to reflect our expectations of future growth and profitability and discounted using a weighted average cost of capital derived from an analysis of comparable selected public companies.
Capitalized internally developed software costs
Capitalized costs are based on the employment costs of individuals working on software development and based on timesheets. Special attention is paid to distinguishing between costs incurred on developing new software or software upgrades, which may be eligible for capitalization, and costs incurred in maintenance and in the correction of problems, which is not eligible.
Judgment is required in identifying whether individual projects meet all of the criteria required to permit capitalization, in particular, whether the software will generate probable future economic benefits.
Defined Benefit Pension Liabilities
The liability or asset recognized on the balance sheet in respect of defined benefit pension plans is the present value of the defined benefit obligation at the end of the reporting period less the fair value of plan assets. The defined benefit obligation is calculated annually by independent actuaries using the projected unit credit method.
The present value of the defined benefit obligation is determined by discounting the estimated future cash outflows using interest rates of high-quality corporate bonds that are denominated in the currency in which the
108
benefits will be paid, and that have terms approximating to the terms of the related obligation. In countries where there is no deep market in such bonds, the market rates on government bonds are used.
The net interest cost is calculated by applying the discount rate to the net balance of the defined benefit obligation and the fair value of plan assets. This cost is included in employee benefit expense in the statement of income/loss.
Remeasurement gains and losses arising from experience adjustments and changes in actuarial assumptions are recognized in the period in which they occur, directly in other comprehensive income. They are included in retained earnings in the statement of changes in equity and on the balance sheet.
Changes in the present value of the defined benefit obligation resulting from plan amendments or curtailments are recognized immediately in income as past service costs.
For defined contribution plans, we pay contributions to publicly or privately administered pension insurance plans. Employee contributions to these plans is voluntary and these contributions are matched by the employer. We have no further payment obligations once the contributions have been paid. The contributions are recognized as employee benefit expense when they are due. Prepaid contributions are recognized as an asset to the extent that a cash refund or a reduction in the future payments is available. Contributions are charged to the statement of income/loss as incurred.
Expected Credit Losses
We have adopted the simplified method indicated in IFRS 9, Financial Instruments (“IFRS 9”), to build our allowance for expected credit losses (“ECL”). We use a matrix based on a calculation of collectability rates according to historical accounts receivable. Allowance is made for lifetime expected credit losses as invoices are issued. The amount of allowance initially recognized is based on historical experience, tempered by expected changes in future cash collections, due to, for example, expected improved customer liquidity or more active credit management.
Accounts receivable-trade balances are non-interest bearing and payment terms are generally under agreements with payment terms of up to 180 days. Our customers primarily consist of government-owned or government-funded hospitals, laboratories with low credit risk, and biopharmaceutical companies. We had minimal instances of actual credit losses and believe that this will continue to be the case.
Income Taxes
Uncertain Tax Positions
The Company files tax returns as prescribed by the tax laws of the jurisdictions in which it operates and is therefore subject to tax examination by various taxing authorities. In the normal course of business, the Company is subject to examination by local tax authorities in Switzerland, France, Italy, Brazil, the UK and the U.S. With the exception of a tax assessment rendered by the French tax authority during an audit of its 2018 and 2019 tax returns, the Company is not aware of any additional issues that could result in any other significant payments, accruals or material deviation from its tax positions. There are no other tax examinations in progress.
The Company records tax liabilities or benefits for all years subject to examination based upon management’s evaluation of the facts, circumstances and information available at the reporting date. There is inherent uncertainty in quantifying income tax positions, especially considering the complex tax laws and regulations in each of the jurisdictions in which the Company operates.
109
Item 6. Directors, Senior Management and Employees
A. Directors and Senior Management
The following table presents information about our executive officers and directors. Ages are as of February 15, 2024.
| Name | Position(s) | Age | ||||||||||||
| Executive Officers and Directors | ||||||||||||||
| Jurgi Camblong | Chief Executive Officer and Director | 45 | ||||||||||||
| Ross Muken | Chief Financial Officer and Chief Operating Officer | 44 | ||||||||||||
| Daan van Well | Chief Legal and Compliance Officer | 49 | ||||||||||||
| Manuela da Silva Valente | Chief People Officer | 54 | ||||||||||||
| Zhenyu Xu | Chief Scientific Officer | 41 | ||||||||||||
| Philippe Menu | Chief Medical Officer and Chief Product Officer | 42 | ||||||||||||
| Non-Executive Directors | ||||||||||||||
| Troy Cox | Chairman of the Board of Directors | 59 | ||||||||||||
| Tomer Berkovitz | Director | 44 | ||||||||||||
| Kathy Hibbs | Director | 60 | ||||||||||||
| Didier Hirsch | Director | 72 | ||||||||||||
| Vincent Ossipow | Director | 55 | ||||||||||||
| Lila Tretikov | Director | 46 | ||||||||||||
| Jean-Michel Cosséry | Director | 64 | ||||||||||||
Executive Officers
Jurgi Camblong, Ph.D., M.B.A., has served as our Chief Executive Officer and a member of our board of directors since March 2011 when he co-founded our company with Dr. Pierre Hutter and Professor Lars Steinmetz. From 2010 to 2011, Dr. Camblong served as the Chief Executive Officer of Gene Predictis SA. Prior to that, Dr. Camblong was a post-doctoral associate researcher at Oxford University and at the University of Geneva. Dr. Camblong was a member of the Advisory Council on Digital Transformation to the Swiss Government and is a Board member of the Swiss Biotech Association. Dr. Camblong holds a Ph.D. in life sciences from the University of Geneva and an Executive M.B.A. in management of technology from EPFL/HEC Lausanne.
Ross Muken, B.Sc., has served as our Chief Financial Officer since February 2021 and our Chief Operating Officer since March 2023. From 2019 to 2020, Mr. Muken served as the Chief Financial Officer of Click Therapeutics, Inc. From 2012 to 2019, Mr. Muken served as the Senior Managing Director and Partner of Equity Research at Evercore/ISI Group. Prior to that, Mr. Muken served in various roles at Deutsche Bank, including as Managing Director of Equity Research, and at Thomas Weisel Partners. Mr. Muken holds a B.Sc. in business administration from Boston University.
Daan van Well, LL.M., M.B.A., has served as our Chief Legal Officer since June 2019 and Chief Compliance Officer since March 2023. Mr. Van Well has more than 20 years of legal, governance and compliance experience. From 2018 to 2019, Mr. Van Well served as the founder and managing partner of consulting firm SpringWorks Sàrl. From 2010 to 2017, Mr. Van Well served in various legal positions with PwC Switzerland, including as the Head of Legal from 2011 to 2017. Prior to that, Mr. Van Well served as corporate secretary and senior legal counsel of Koninklijke Ahold N.V. (currently Koninklijke Ahold Delhaize N.V.) (Royal Ahold Delhaize) and practiced law at Loyens & Loeff N.V. in Rotterdam, The Netherlands. Mr. Van Well holds an LL.M. in Dutch civil law from Utrecht University and an Executive M.B.A. in management and corporate finance from HEC Lausanne.
110
Manuela da Silva Valente, B.A., has served as our Chief People Officer since January 2019. Ms. Da Silva Valente has more than 20 years of human resources experience. From 2011 to 2018, Ms. Da Silva Valente served in various human resources leadership roles at IQVIA (formerly Quintiles and IMS Health Inc.), including as Global Senior Director of Human Resources from 2016 to 2018. Prior to that, Ms. Da Silva Valente held various human resources roles at Outcome Sciences, Inc. prior to its acquisition by IQVIA. Ms. Da Silva Valente holds a B.A. in business administration from the Business Management School of Zurich and a Management & Human Resources Diploma from CEFCO Lausanne.
Zhenyu Xu, Ph.D., has served as our Chief Scientific Officer since January 2021 and was previously our Chief Technology Officer since May 2014. Dr. Xu was the leader of the technology team that developed our SOPHiA DDM Platform. Prior to that, Dr. Xu was a post-doctoral fellow at the European Molecular Biology Laboratory. Dr. Xu holds a Ph.D. in molecular and computational biology from the European Molecular Biology Laboratory.
Philippe Menu, M.D., Ph.D., M.B.A., has served as our Chief Medical Officer since February 2020 and our Chief Product Officer since March 2023. From 2011 to 2020, Dr. Menu was a management consultant with McKinsey & Company, focusing on the biopharmaceutical sector and in particular innovative therapies and diagnostics in oncology and rare diseases. Prior to that, Dr. Menu was a post-doctoral fellow at the University of Lausanne. Dr. Menu holds an M.D./Ph.D. in life sciences from the University of Lausanne and an M.B.A. from the Open University Business School.
Non-Executive Directors
Troy Cox, M.B.A., has served as the Chairman of our board of directors since June 2019. From 2017 to 2019, Mr. Cox served as the chief executive officer of Foundation Medicine Inc. From 2010 to 2017, Mr. Cox served as the senior vice president of U.S. commercial of Genentech, Inc. Prior to that, Mr. Cox held various executive and senior positions at UCB S.A., Sanofi-Aventis U.S. LLC and Schering-Plough Corporation. In addition to our board of directors, Mr. Cox serves on the board of directors of Zymeworks Inc., LetsGetChecked, Standard BioTools (formerly SomaLogic, Inc.), BioSplice Therapeutics, Inc. and previously served on the board of directors of Foundation Medicine Inc. Mr. Cox holds an M.B.A. from the University of Missouri.
Tomer Berkovitz, Ph.D., has served as a member of our board of directors since March 2021. Since 2018, Dr. Berkovitz has served as General Partner of aMoon Fund, where he co-leads its Growth fund. From 2014 to 2018, Dr. Berkovitz served as the Chief Operating Officer and Chief Financial Officer of Alcobra Ltd. Prior to that, Mr. Berkovitz served as an Executive Director in J.P. Morgan’s investment banking division in New York. In addition to our board of directors, Dr. Berkovitz serves on the board of directors of several other healthcare companies in the aMoon portfolio. Dr. Berkovitz holds a Ph.D. in finance from Columbia Business School.
Kathy Hibbs, J.D., has served as a member of our board of directors since March 2021. Since 2022, Ms. Hibbs has served as the Chief Administrative Officer of 23andMe, Inc. and prior to that role as the Chief Legal and Regulatory Officer from 2014 to 2022. Before joining 23andMe, Inc., Ms. Hibbs held various leadership roles in legal, business development and compliance functions at Genomic Health, Inc., Monogram Biosciences Inc. and Varian Medical Systems, Inc. Ms. Hibbs previously served on the board of directors of Decipher Biosciences, Inc. Ms. Hibbs holds a J.D. from the University of California, Hastings College of Law.
Didier Hirsch, M.Sc., M.S., has served as a member of our board of directors since June 2020. From 2010 to 2018, Mr. Hirsch served as the senior vice president and chief financial officer of Agilent Technologies, Inc. Prior to that, Mr. Hirsch held various leadership roles in finance at Agilent Technologies, Inc. and Hewlett-Packard Company. In addition to our board of directors, Mr. Hirsch serves on the board of directors of Knowles Corporation and Azenta. Mr. Hirsch previously served on the board of directors of Logitech International S.A. and International Rectifier Corporation. Mr. Hirsch holds an M.Sc. in computer science from Toulouse University and an M.S. in industrial administration from Purdue University.
Vincent Ossipow, Ph.D., has served as a member of our board of directors since June 2014. Dr. Ossipow has been a partner at Omega Funds. Dr. Ossipow has also served as the Chief Scientific Officer of Omega Alpha SPAC. Prior to that, Dr. Ossipow held various investment management positions at Sectoral Asset Management and Pictet Bank. In addition to our board of directors, Dr. Ossipow serves on the board of directors of Immunic, Inc., BioInvent International AB, FoRx SA, SwissThera SA, and eTheRNA immunotherapies NV, and previously served on the board of directors of Andrew Alliance S.A., Lifespan, Inc., Raindance Technologies, CNx SA and Kuros Biosciences AG. Dr. Ossipow holds a Ph.D. in molecular biology from the University of Geneva.
111
Lila Tretikov has served as a member of our board of directors since June 2023. Since August 2020, Ms. Tretikov has served as the Deputy Chief Technology Officer of Microsoft and, from 2018 to August 2020, served as the Corporate Vice President, AI, Perception and Mixed Reality of Microsoft. Prior to that, Ms. Tretikov was the chief executive officer of Terrawatt and the chief executive officer of Wikipedia. In addition to our board of directors, Ms. Tretikov currently as a member of the board of directors of Volvo Cars, Xylem and Onfido LTD. Ms. Tretikov holds a [B.S.] in computer science, art and artificial intelligence from the University of California, Berkeley and has completed executive graduate programs at Stanford University and the University of Oxford.
Jean-Michel Cosséry, Ph.D., Pharm.D., M.B.A., has served as a member of our board of directors since June 2022. From 2012 to 2018, Dr. Cosséry served in various senior leadership positions at Eli Lilly and Company, including as Vice President, North America Oncology, as well as Vice President and Managing Director of Lilly UK and Northern Europe. Prior to that, he served as Vice President and Chief Marketing Officer of GE Healthcare as well as in senior positions at Novartis International AG and Serono (now Merck (Schweiz) AG). Dr. Cosséry serves on the board of directors of Malin Corporation plc and Eracal Therapeutics Ltd., and serves as the non-executive chairman of Scancell Holdings PLC. Previously he served on the board of directors of ABPI (UK) LIMITED, Immunocore Holdings Limited, Kymab Ltd, Exact Therapeutics AS, and Diurnal PLC, and as chairman of the board of directors of the American Pharmaceutical Group in the UK. Dr. Cosséry holds an M.B.A. from the Rotterdam School of Management, a Ph.D. with honors in nuclear chemistry and neurobiology from Paris Sud University, and a Pharm.D. with honors in pharmacology from Paris Sud University.
Relationships
There are no family relationships among any of our directors or executive officers.
B. Compensation
Compensation of Directors and Executive Officers
For the year ended December 31, 2023, the aggregate compensation paid or accrued to the members of our board of directors and our executive officers for services in all capacities, including retirement and similar benefits, was $4.5 million, the total fair value of stock options and non-vested share awards granted to the members of our board of directors and our executive officers was $13.9 million, and the amount set aside or accrued by us to provide pension, retirement or similar benefits to the members of our board of directors and our executive officers was $0.2 million.
Equity Incentive Plans
On June 29, 2021, our shareholders approved the 2021 Equity Incentive Plan that our board of directors had previously adopted. The purpose of the 2021 Equity Incentive Plan is to motivate and reward performance of our employees, directors, consultants and advisors and further the best interests of the Company and our shareholders.
Plan Administration. The 2021 Equity Incentive Plan is administered by the compensation committee of our board of directors, subject to the board of directors’ discretion to administer or appoint another committee to administer it.
Awards. Equity incentive awards under the 2021 Equity Incentive Plan may be granted in the form of options (including incentive stock options and non-qualified stock options), share appreciation rights, restricted shares, restricted share units, performance awards or other share-based awards. Options and share appreciation rights will have an exercise price determined by the compensation committee and, in the case of options granted to a participant subject to U.S. taxation, will not be less than fair market value of the underlying ordinary shares on the date of grant (or, if such options consist of incentive stock options and the participant owns (or is deemed to own) at least 10% of the total combined voting power of all classes of our capital stock (a “ten percent shareholder”), an exercise price not be less than 110% of the fair market value of the underlying ordinary shares on the date of grant). In addition, under the 2021 Equity Incentive Plan, options and share appreciation rights may not have a term that exceeds ten years (or, in the case of an incentive stock option granted to a ten percent shareholder, a term that exceeds five years).
112
Eligible Participants. The compensation committee is able to offer equity awards at its discretion under the 2021 Equity Incentive Plan to (1) any employees of us or any of our subsidiaries, (2) any non-employee directors serving on our board of directors and (3) any consultants or other advisors to us or any of our subsidiaries; provided that only employees of our company or certain of our subsidiaries may be granted incentive stock options. To the extent required by applicable law and our articles of association in effect from time to time, all awards and rights, payments and benefits granted or made under the 2021 Equity Incentive Plan to our directors and executive officers are subject to the approval of the relevant total amount of compensation by our shareholders.
Share Reserve. The maximum number of ordinary shares initially reserved for issuance pursuant to awards under the 2021 Equity Incentive Plan is 7,800,740 ordinary shares, which will be increased on the first day of each fiscal year of the Company, beginning with the 2022 fiscal year, in an amount equal to the least of (i) a number of ordinary shares equal to five percent (5%) of the total number of shares of all classes of shares of the Company outstanding on the last day of the immediately preceding fiscal year, (ii) such number of shares determined by our board of directors, and (iii) the aggregate number of shares available to our board of directors under our articles of association or otherwise that may be granted as, or be subject to, equity incentive awards on such date. To ensure that our board of directors can reserve a sufficient number of ordinary shares for purposes of the 2021 Equity Incentive Plan, we plan to request its shareholders approve annual increases to the Company’s conditional share capital for employee participation. Notwithstanding the foregoing, no more than 7,800,740 ordinary shares may be issued in respect of incentive stock options. In addition, ordinary shares reserved for issuance under the 2021 Equity Incentive Plan are subject to adjustment in the event of certain corporate transactions or events if necessary to prevent dilution or enlargement of the benefits made available under the 2021 Equity Incentive Plan.
Vesting. The vesting conditions for grants under the equity incentive awards under the 2021 Equity Incentive Plan are set forth in the applicable award documentation.
Termination of Service and Change in Control. In the event of a participant’s termination of employment or service, the compensation committee may determine in the applicable award agreement the extent to which an equity incentive award may be exercised, settled, vested, paid or forfeited. Unless otherwise provided in the applicable award agreement, in the event of a change in control by way of a merger, a sale of the Company’s securities, a sale of all or substantially all of the Company’s assets or similar transaction, each award that is outstanding as of immediately prior to such change in control will, (i) to the extent not then vested, accelerate and become fully vested (with any performance award assumed to have achieved the applicable performance criteria at the greater of target and maximum level of performance), and (ii) be cancelled and converted into the right to receive a payment in cash with a value equal to the value of such award based on the per share value of consideration received or to be received by other shareholders of the Company in such change in control, with the value of the any such award that is an option or a share appreciation right reduced by the applicable exercise price. In the event of a change in control, the compensation committee may also, in lieu of the acceleration and cash out of outstanding awards described above, take any one or more of the following actions with respect to outstanding awards that the compensation committee determines to be appropriate: (i) cancel any such award in exchange for a payment in securities or other property other than cash or any combination thereof with a value equal to the value of such award based on the per share value of consideration received or to be received by other shareholders in the event (or without payment of consideration if the compensation committee determines that no amount would have been realized upon the exercise of the award or other realization of the participant’s rights); (ii) require the exercise of any outstanding option; (iii) provide for the assumption, substitution, replacement or continuation of any award by the successor or surviving corporation, along with appropriate adjustments with respect to the number and type of securities (or other consideration) of the successor or surviving corporation, subject to any replacement awards, the terms and conditions of the replacement awards (including performance targets) and the grant, exercise or purchase price per share for the replacement awards; (iv) make any other adjustments in the number and type of securities (or other consideration) subject to awards that may be granted in the future; (v) provide that any such award shall be accelerated and become exercisable, payable and/or fully vested with respect to all ordinary shares covered thereby or (vi) provide that any award shall not vest, be exercised or become payable as a result of such event.
Termination and Amendment. Unless terminated earlier, the 2021 Equity Incentive Plan will continue for a term of ten years. Our board of directors has the authority to amend or terminate the 2021 Equity Incentive Plan subject to shareholder approval with respect to certain amendments. However, no such action may materially
113
adversely affect the rights of any participant under any outstanding award without the consent of the affected participant.
During the year ended December 31, 2023, we have granted to the members of our board of directors and to our executive officers, in the aggregate, the right to acquire 2,164,370 ordinary shares under the 2021 Equity Incentive Plan. Options granted to executive officers during 2023 vest either 25 % on the first anniversary of the grant date and the remaining 75 % vesting ratably on a monthly basis over the remaining three years , 25 % on the first anniversary of the grant date and the remaining 75 % vesting ratably on a quarterly basis over the remaining three years , on the second anniversary of the grant date. Options grants to non-executive board members vest annually over four years on each anniversary of the grant date. The weighted-average exercise price for the options granted during 2023 was $3.00. During the year ended December 31, 2023, we granted to the members of our board of directors and to our executive officers, in the aggregate, 1,621,573 RSUs under the 2021 Equity Incentive Plan. The restricted share units granted to an executive officer vest ratably over a four-year period, subject to the executive officer’s continued employment with us, and any unvested RSUs will be forfeited should the executive officer terminate his or her employment with us. The restricted share units granted to a non-executive member of the board of directors shall vest between the Company’s 2023 Annual General Meeting and the Company’s 2024 Annual General Meeting, subject to continued service on our board of directors.
Employment Agreements
We have entered into employment agreements with certain of our executive officers. Each of these agreements provides for an initial salary and annual bonus opportunity, as well as participation in certain pension and welfare benefit plans. These agreements may require advance notice of termination and in some cases provide for paid garden leave. Some of our executive officers have agreed to covenants not to compete against us or solicit our employees or customers during employment and for a period of up to one year following termination. We may be required to pay some of our executive officers compensation for their covenant not to compete with us following termination. If we experience a “change in control”, then our executive officers’ then-unvested awards will become fully vested at such time.
Additional Information
We are required under Swiss law to provide additional disclosure regarding the compensation of our executive officers and directors. We incorporate by reference the information contained in Exhibit 99.4 of the Report on Form 6-K filed with the SEC on March 5, 2024 (other than the Report of the Statutory Auditor and Section 4 Equity and Equity-Linked Instruments Held by Members of the Board of Directors and the Executive Committee).
C. Board Practices
Board Composition and Election of Directors
Our board of directors is composed of eight members. Each director is elected for a one-year term. The current members of our board of directors will serve until our annual general meeting of shareholders in 2024.
Board Practices
We are a foreign private issuer under the rules of the SEC. As a result, in accordance with Nasdaq listing standards, we rely on home country governance requirements and certain exemptions thereunder rather than on Nasdaq corporate governance requirements. For an overview of our corporate governance principles, see “Item 10. Additional Information—B. Memorandum and Articles of Association” and “Item 16G—Corporate Governance.”
Director Independence
Our board of directors has affirmatively determined that each of Troy Cox, Tomer Berkovitz, Kathy Hibbs, Didier Hirsch, Vincent Ossipow, Lila Tretikov, and Jean-Michel Cosséry is an independent director within the meaning of applicable Nasdaq standards.
114
Diversity
Our board of directors values diversity among its members. Our nomination and corporate governance committee, within the purview of its mandate, has the responsibility to take diversity into consideration as part of the overall director selection and nomination processes and to make the identification of diverse candidates a search criterion. The matrix below sets forth a summary of the diversity of our board of directors as of February 1, 2024:
Country of Principal Executive Offices: Switzerland
Foreign Private Issuer: Yes
Disclosure Prohibited under Home Country Law: No
Total Number of Directors: 8
| Female | Male | Non-Binary | Did Not Disclose | ||||||||||||||||||||
| Part I: Gender Identity | 2 | 6 | — | — | |||||||||||||||||||
| Part II: Demographic Background | |||||||||||||||||||||||
| Underrepresented individual in home country jurisdiction | 3 | ||||||||||||||||||||||
| LGBTQ+ | 2 | ||||||||||||||||||||||
| Did not disclose | 3 | ||||||||||||||||||||||
The matrix below sets forth a summary of the diversity of our board of directors as of February 1, 2023:
Country of Principal Executive Offices: Switzerland
Foreign Private Issuer: Yes
Disclosure Prohibited under Home Country Law: No
Total Number of Directors: 8
| Female | Male | Non-Binary | Did Not Disclose | ||||||||||||||||||||
| Part I: Gender Identity | 1 | 7 | — | — | |||||||||||||||||||
| Part II: Demographic Background | |||||||||||||||||||||||
| Underrepresented individual in home country jurisdiction | 2 | ||||||||||||||||||||||
| LGBTQ+ | 1 | ||||||||||||||||||||||
| Did not disclose | — | ||||||||||||||||||||||
Board Meetings
In 2023, our board of directors held 8 meetings.
Committees of the Board of Directors
Our board of directors has three committees: an audit committee, a compensation committee and a nomination and corporate governance committee.
115
Audit Committee
The audit committee, which consists of Didier Hirsch (chair), Tomer Berkovitz, and Lila Tretikov, assists our board of directors in overseeing our accounting and financial reporting processes and the audits of our consolidated financial statements. In addition, the audit committee is directly responsible for the compensation, retention and oversight of the work of our independent registered public accounting firm that our shareholders elect as our external auditors. The audit committee consists exclusively of members of our board of directors who are financially literate, and each of Didier Hirsch and Tomer Berkovitz is considered an “audit committee financial expert” as defined by the SEC. Our audit committee complies with Rule 10A-3(b)(1) of the Exchange Act. Our board of directors has determined that each of Didier Hirsch, Lila Tretikov and Tomer Berkovitz satisfy the “independence” requirements under Nasdaq listing standards and Rule 10A-3 under the Exchange Act.
The audit committee is governed by a charter that complies with the Nasdaq listing standards that apply to us. The audit committee has the responsibility to, among other things:
•select, appoint, compensate, retain, terminate and oversee the work of any accounting firm engaged for the purpose of preparing or issuing an audit report or performing other audit, review or attest services;
•pre-approve the audit services and non-audit services (including the fees and terms thereof) to be provided by the independent auditor pursuant to pre-approval policies and procedures;
•review and approve the planned scope and timing of our independent registered public accounting firm’s annual audit plan(s);
•discuss significant findings from the audit and any problems or difficulties encountered, including any restrictions on the scope of our independent registered public accounting firm’s activities or on access to requested information, and any significant disagreements with management;
•evaluate the independent auditor’s qualifications, performance and independence, and present its conclusions with respect to the independent auditor to the board of directors on at least an annual basis;
•supervise the ethics committee as provided in the Code of Ethics, consider related party transactions and supervise compliance with any other policies over which the audit committee has oversight authority;
•review and discuss with management and the independent auditor the annual audited consolidated and stand-alone financial statements and make its recommendation to the board of directors for their presentation to the general meeting of shareholders for approval;
•review and discuss with management and the independent auditor the unaudited quarterly financial statements
•review with management and the independent auditor (i) any analyses or other written communications prepared by management and/or the independent auditor setting forth significant financial reporting issues and judgments made in connection with the preparation of the financial statements, (ii) our critical accounting policies and practices, (iii) the effect of regulatory and accounting initiatives, as well as off-balance-sheet transactions and structures, on our financial statements and (iv) any major issues regarding accounting principles and financial statement presentations;
•in conjunction with the chief executive officer and chief financial officer, review disclosure controls and procedures and internal control over financial reporting;
•review and discuss with the independent auditor any audit problems or difficulties and management’s response thereto;
•discuss with the chief financial officer and the chief executive officer the results of its review of the management or internal control letter issued by the independent auditor;
116
•resolve disagreements between management and the auditor regarding our financial reporting;
•review our risk assessment and risk management policies and practices;
•establish procedures for the receipt, retention and treatment of complaints received regarding accounting, internal accounting controls or auditing matters, as well as the receipt of summary whistleblower reports and the confidential, anonymous submission by employees of concerns regarding questionable accounting or auditing matters;
•review our compliance with laws and regulations; and
•review any major litigation or investigations against us that may have a material impact on our financial statements.
The audit committee meets as often as it determines is appropriate to carry out its responsibilities, but in any event meets at least quarterly.
Compensation Committee
The compensation committee, which consists of Kathy Hibbs (chair), Jean-Michel Cosséry, and Vincent Ossipow, supports our board of directors in establishing and reviewing the compensation and benefits strategy and guidelines as well as in preparing the proposals to the annual general meeting of shareholders regarding the compensation of the members of the board of directors and the executive officers. The compensation committee may submit proposals to the board of directors on other compensation-related matters. Swiss law requires that we have a compensation committee, so in accordance with Nasdaq listing standards, we follow home country requirements with respect to the compensation committee. As a result, our practice varies from Nasdaq listing standards, which set forth certain requirements as to the responsibilities, composition and independence of compensation committees for domestic issuers. Swiss law requires that our board of directors submit the aggregate amount of compensation of all members of our board of directors and of all executive officers to a binding shareholder vote every year. The members of the compensation committee will be elected by our annual general meeting of shareholders. The board of directors appoints the chair of the compensation committee and fills any vacancies until the following annual general meeting of shareholders.
The compensation committee has the responsibility to, among other things:
•regularly review and make recommendations to the board of directors regarding our compensation and benefits strategy and guidelines;
•review and make recommendations to the board of directors regarding the compensation of the members of the board of directors, of the executive committee and of our extended management team;
•prepare the proposals to the shareholders’ meeting regarding the compensation of the members of the board of directors and of the executive committee;
•review and approve the recommendation of our chief executive officer regarding the fixed and variable compensation, including incentive plan participation and benefits, of the members of the management team other than members of the executive committee;
•review and make recommendations to the board of directors regarding our compensation and benefits plans (cash and/or equity-based plans) and, where appropriate or required, make recommendations to adopt, amend and terminate such plans;
•to the extent not delegated by the compensation committee to a different body or a third party, administer our compensation and benefits plans; and
•review and assess risks arising from our employee compensation policies and practices and whether any such risks are reasonably likely to have a material adverse effect on us.
117
Nomination and Corporate Governance Committee
The nomination and corporate governance committee, which consists of Troy Cox (chair), Kathy Hibbs and Didier Hirsch, is responsible for director and board committee nominations, succession planning, performance evaluation and reviewing and amending, if required, our corporate governance framework and guidelines. The members of the nomination and corporate governance committee and its chair are appointed by our board of directors.
The nomination and corporate governance committee has the responsibility to, among other things:
•determine selection criteria for the succession of the members of the board of directors and board committees, our chief executive officer and our chief financial officer, and establish such succession planning (including for the event of the incapacitation, retirement or removal of such individuals) by making recommendations to the board of directors;
•oversee searches, identify qualified individuals and recommend individuals for membership on the board of directors and for the position of chief executive officer;
•recommend individuals for appointment to the audit committee annually and as vacancies or newly created positions occur;
•at least annually, prepare the board of directors’ assessment of the performance of the board of directors and board committees and of our chief executive officer;
•review the recommendations of the other board committees based on their self-evaluations and discuss its own evaluation with the board of directors;
•monitor and assess developments and trends in corporate governance to the extent that these do not have an impact on the activities and tasks of the audit committee or the compensation committee;
•review proposals to be made to the board of directors for the amendment of our articles of association, our organizational regulations, and any other charter, rules or regulations;
•approve in advance any acceptance by a member of our management of a position as member of the board of directors in companies not belonging to our group;
•periodically review and assess the adequacy of the charter of the nomination and corporate governance committee and recommend any proposed changes to the board of directors for approval;
•if it deems necessary, develop and recommend to the board of directors corporate governance guidelines for us;
•periodically review and reassess the adequacy of the Code of Ethics and recommend any proposed changes to the board of directors;
•oversee compliance with the Code of Ethics and report on such compliance to the board of directors;
•supervise the ethics committee as provided in the Code of Ethics; and
•review and consider any requests for waivers of the Code of Ethics for members of our board of the directors, our management and other senior financial officers, and make a recommendation to our board of directors with respect to such request for a waiver.
D. Employees
We employ great minds in biotechnology and machine learning who continuously advance our algorithms, applications, products, and services to benefit clinical researchers around the world. Approximately 30% of our employees hold doctoral degrees in diverse fields that range from cell biology to computer science. Our
118
employees bring widely varied expertise and competencies to our company. Our multidisciplinary team includes bioinformaticians, medical and genetic experts, scientists, software engineers, web developers, graphic designers, commercial experts and lab specialists, as well as staff in our administrative and corporate teams.
We pride ourselves on the excellence and integrity of our employees. We work towards the best quality and target the highest performance. Our corporate DNA, rooted in quality, precision and robustness, is the key to our success. We strive to foster an entrepreneurial, innovative and unique culture that ignites employees’ passion and inspires them to challenge the status quo. We create work environments that preserve and value individuality and diversity of viewpoints and approaches such that our employees trust each other and collaborate to achieve our collective goals.
The following strategies help ensure that we attract and retain high quality employees:
•Attracting Talent. Our dedicated and experienced global talent acquisition team identifies and attracts the most qualified candidates. Our locations were strategically selected to attract highly educated talent from renowned universities and engineering schools, and we regularly attend events and use social media to increase awareness of our brand to prospective candidates. As part of our hiring process, we conduct scientific and technical assessment to ensure that candidates have the appropriate skills and expertise.
•Retaining and Developing Talent. As part of our effort to continuously motivate and engage our employees and provide professional development for our employees, we provide corporate talent reviews and follow-up individual development plans for our employees and have created career ladders with grading systems for all departments with detailed job descriptions on what is required at each level. We also perform employee engagement surveys that inform our dedicated task forces as they continuously strive to increase employee satisfaction and morale.
•Training. To help our employees integrate into our company, advance their knowledge and skills and remain at the forefront of innovation, we created Learning@SOPHiA, which consists of (i) an onboarding program with a new hire learning path, a manager’s guide to onboarding and a buddy system for new hires, (ii) ongoing learning paths with department specific training modules, technical and non-technical training, cross-functional information sessions, mentoring and soft skill training, and (iii) leadership and development programs for managers. In addition, for our salespersons, our sales success department provides commercial training, including consultative sales, negotiations skills and cold call training.
As of December 31, 2023, we had 449 employees across 24 countries, of whom 351 were located in EMEA, 77 were located in North America, 13 were located in Latin America and 8 were located in Asia Pacific. Of which, we had 19 temporary employees located in EMEA and 3 were located in North America. Over the course of the year ended December 31, 2023, we employed, on average, 459 employees. Approximately 45% of our employees are engaged in research and development.
In certain countries in which we operate, we are subject to, and comply with, local labor law requirements, which may automatically make our employees subject to industry-wide collective bargaining agreements. For instance, our employees in France are covered by the Syntec Collective Bargaining Agreement. In addition, pursuant to French regulations, we have established at our French subsidiary a Comité Social et Économique or Social and Economic Committee. We are not subject to any other collective bargaining agreements. We believe that our relationship with our employees is good.
E. Share Ownership
See “Item 7. Major Shareholders and Related Party Transactions—A. Major shareholders.”
F. Disclosure of a Registrant’s Action to Recover Erroneously Awarded Compensation
None.
Item 7. Major Shareholders and Related Party Transactions
119
A. Major Shareholders
The following table presents information relating to the beneficial ownership of our ordinary shares as of February 15, 2024 by:
•each person, or group of affiliated persons, known by us to own beneficially 5% or more of our outstanding ordinary shares;
•each of our executive officers and directors and persons nominated to serve in such positions; and
•all executive officers and directors and persons nominated to serve in such positions as a group.
The number of ordinary shares beneficially owned by each entity, person, executive officer or director is determined in accordance with the rules of the SEC, and the information is not necessarily indicative of beneficial ownership for any other purpose. Under such rules, beneficial ownership includes any ordinary shares over which the individual has sole or shared voting power or investment power as well as any ordinary shares that the individual has the right to acquire within 60 days from February 15, 2024 through the exercise of any option or other right. Except as otherwise indicated, and subject to applicable community property laws, we believe that the persons named in the table have sole voting and investment power with respect to all ordinary shares held by that person based on information provided to us by such person.
The percentage of outstanding ordinary shares beneficially owned is computed on the basis of 65,301,358 ordinary shares outstanding as of February 15, 2024. Ordinary shares that a person has the right to acquire within 60 days are deemed outstanding for purposes of computing the percentage ownership of the person holding such rights, but are not deemed outstanding for purposes of computing the percentage ownership of any other person, except with respect to the percentage ownership of all executive officers and directors as a group. Unless otherwise indicated below, the business address for each beneficial owner is SOPHiA GENETICS SA, La Pièce 12, CH-1180 Rolle, Switzerland.
120
| Number of Ordinary Shares | Percentage of Ordinary Shares | |||||||||||||
| Principal Shareholders | Beneficially Owned | Beneficially Owned | ||||||||||||
| 5% or Greater Shareholders | ||||||||||||||
Alychlo NV(1) | 6,993,800 | 10.71% | ||||||||||||
Generation IM Sustainable Solutions Fund III, L.P.(2) | 6,789,560 | 10.40% | ||||||||||||
Balderton Capital VI, S.L.P.(3) | 3,361,880 | 5.15% | ||||||||||||
| Executive Officers and Directors | ||||||||||||||
Jurgi Camblong(4) | 2,460,081 | 3.77% | ||||||||||||
Zhenyu Xu(4) | 414,249 | * | ||||||||||||
| Vincent Ossipow | 395,502 | * | ||||||||||||
| Troy Cox | 185,942 | * | ||||||||||||
| Daan van Well | 91,827 | * | ||||||||||||
| Didier Hirsch | 74,522 | * | ||||||||||||
| Jean-Michel Cosséry | 63,411 | * | ||||||||||||
| Ross Muken | 61,537 | * | ||||||||||||
| Philippe Menu | 41,749 | * | ||||||||||||
| Manuela da Silva Valente | 39,900 | * | ||||||||||||
| Kathy Hibbs | 37,414 | * | ||||||||||||
| Tomer Berkovitz | — | * | ||||||||||||
| Lila Tretikov | — | * | ||||||||||||
| All executive officers and directors as a group (13 persons) | 3,866,134 | 5.92% | ||||||||||||
*Less than 1% of our total outstanding ordinary shares.
(1)This information is based solely on a Schedule 13G filed by Alychlo NV and Marc Coucke with the SEC on February 14, 2022. Marc Coucke is the principal shareholder, chairman and managing director of Alychlo NV. The principal business address of each of the foregoing persons or entities is Lembergsesteenweg 19, 9820 Merelbeke, Belgium.
(2)This information is based solely on a Schedule 13G filed by Generation Investment Management LLP, Generation IM Sustainable Solutions III, GP Ltd and Generation IM Sustainable Solutions Fund III, L.P. with the SEC on February 13, 2024. The principal business address of each of the foregoing entities is 20 Air Street, 7th floor, London, United Kingdom W1B 5AN.
(3)This information is based solely on a Schedule 13G filed by Balderton Capital VI, S.L.P. with the SEC on February 14, 2023. Balderton Capital General Partner VI, S.a.r.l. is the managing general partner of Balderton Capital VI, S.L.P. and may be deemed to have voting, investment and dispositive power with respect to these securities. Adrian Rainey, Donatien-Xavier Martin and Marie Calinet are the managers of Balderton Capital General Partner VI, S.a.r.l. and may each be deemed to share voting, investment, and dispositive power with respect to these securities.
(4)The shares owned by the parties have been pledged pursuant to lending arrangements.
As of February 15, 2024, we had approximately 169 shareholders of record of our ordinary shares. We estimate that as of February 15, 2024, approximately 61.55% of our outstanding ordinary shares are held by 18 U.S. record holders. The actual number of shareholders is greater than this number of record holders and includes shareholders who are beneficial owners but whose shares are held in street name by brokers and other nominees. This number of holders of record also does not include shareholders whose shares may be held in trust or by other entities such as Cede & Co. as nominee for the Depository Trust Company.
We have experienced significant changes in the percentage ownership held by major shareholders as a result of our initial public offering. Prior to our initial public offering, our principal shareholders were Alychlo NV,
121
Generation Investment Management LLP and Balderton Capital VI, S.L.P., which held ordinary shares representing 14.2%, 13.8% and 6.8% of our outstanding ordinary shares prior to our initial public offering.
B. Related Party Transactions
The following is a description of certain related party transactions we have entered into since January 1, 2023 with any of our executive officers and directors or their affiliates and holders of more than 10% of any class of our voting securities in the aggregate, which we refer to as related parties, other than compensation arrangements, which are described under “Item 6. Directors, Senior Management and Employees—B. Compensation.”
Indemnification Agreements
We have entered into indemnification agreements with our executive officers and directors. The indemnification agreements and our articles of association require us to indemnify our executive officers and directors to the fullest extent permitted by law.
Related Person Transaction Policy
We have adopted a related person transaction policy. Our related person transaction policy states that any related person transaction must be approved or ratified by our audit committee or board of directors. In determining whether to approve or ratify a transaction with a related person, our audit committee or board of directors will consider all relevant facts and circumstances, including, without limitation, the commercial reasonableness of the terms of the transaction, the benefit and perceived benefit, or lack thereof, to us, the opportunity costs of an alternative transaction, the materiality and character of the related person’s direct or indirect interest and the actual or apparent conflict of interest of the related person. Our audit committee or board of directors will not approve or ratify a related person transaction unless it has determined that, upon consideration of all relevant information, such transaction is in, or not inconsistent with, our best interests and the best interests of our shareholders.
Item 8. Financial Information
A. Consolidated Statements and Other Financial Information
Financial Statements
See “Item 18. Financial Statements,” which contains our financial statements prepared in accordance with IFRS Accounting Standards.
Legal Proceedings
From time to time, we may be subject to various legal proceedings and claims that arise in the ordinary course of our business activities. The results of litigation and claims cannot be predicted with certainty. As of the date of this Annual Report, we do not believe that we are party to any claim or litigation the outcome of which would, individually or in the aggregate, be reasonably expected to have a material adverse effect on our business.
Dividends and Dividend Policy
We have never declared or paid cash dividends on our share capital. We intend to retain all available funds and any future earnings, if any, to fund the development and expansion of our business, and we do not anticipate paying any cash dividends in the foreseeable future. Any future determination related to dividend policy will be made at the discretion of our board of directors and will depend upon, among other factors, our results of operations, financial condition, capital requirements, contractual restrictions and business prospects and other factors our board of directors may deem relevant.
Under Swiss law, any dividend must be approved by our shareholders. In addition, our auditors must confirm that the dividend proposal of our board of directors to the shareholders conforms to Swiss statutory law and our articles of association. A Swiss corporation may pay dividends only if it has sufficient distributable profits from the previous business year (bénéfice de l’exercice) or brought forward from previous business years (report des
122
bénéfices) or if it has distributable reserves (réserves à libre disposition), each as evidenced by its audited stand-alone statutory balance sheet prepared pursuant to Swiss law and after allocations to reserves required by Swiss law and its articles of association have been deducted. Distributable reserves are generally booked either as free reserves (réserves libres) or as reserves from capital contributions (apports en capital). Distributions out of share capital, which is the aggregate par value of a corporation’s issued shares, may be made only by way of a share capital reduction. See “Item 10. Additional Information—B. Memorandum and Articles of Association.”
B. Significant Changes
There have been no significant changes in our business since the date of the annual financial statements, other than those described under “Item 4. Information on the Company—B. Business Overview.”
Item 9. The Offer and Listing.
A. Offer and Listing Details
Our ordinary shares are listed on Nasdaq under the symbol “SOPH.” For a description of our ordinary shares, see “Item 10. Additional Information—B. Memorandum and Articles of Association.”
C. Markets
See “—A. Offer and Listing Details.”
Item 10. Additional Information
B. Memorandum and Articles of Association
See Exhibit 2.1 to this Annual Report for a description of our ordinary shares and articles of association.
C. Material Contracts
The following descriptions of our material agreements are not complete and are qualified in their entirety by reference to the full text of such agreements, which are filed as exhibits to this Annual Report and incorporated herein by reference.
License Agreements
Normandie Valorisation—Exclusive License Agreements
In March of 2018, we entered into an exclusive license of patents and results (the “2018 Normandie Agreement”) with the University of Rouen Normandy, the Henri Becquerel Centre, INSERM Transfert SA (collectively, the “Co-Owners”) and Normandy University, acting through Normandie Valorisation, pursuant to which we obtained an exclusive, royalty-bearing, non-sublicensable license under certain patents related to methods for diagnosing hematological malignancies and an associated diagnostic kit to develop, manufacture and sell products for the diagnosis of acute myeloblastic leukemia, acute lymphoblastic leukemia and chronic myeloid leukemia in the countries covered by the licensed patents.
In May 2019, we entered into an additional Exclusive License of Patents and Results (the “2019 Normandie Agreement”) with the same parties, pursuant to which we obtained an identical license under the same patents for products for the diagnosis of carcinomas other than acute myeloblastic leukemia, acute lymphoblastic leukemia and chronic myeloid leukemia.
Under each agreement, we are obligated to act with a standard of care to develop, manufacture and sell the licensed products. Our failure to meet the standard of care requirement could subject us to a reduction of applicable territory or respective field of use by way of an amendment to the applicable agreement if (i) we have not marketed nor implemented necessary steps for the marketing of the applicable licensed products within a reasonable time following the end of the agreed-upon development plan for the commercialization of such products or (ii) our delay in executing the development plan exceeds a certain specified time. Pursuant to both
123
agreements, Normandie Valorisation is responsible for the prosecution, maintenance and defense of the licensed patents.
Pursuant to the 2018 Normandie Agreement, we paid Normandie Valorisation a low five-digit Euro upfront fee and are obligated to pay to Normandie Valorisation a low-tens Euro fee per each analysis utilizing Myeloid Plus Solution (MYS+). Pursuant to the 2019 Normandie Agreement, we paid Normandie Valorisation a low five-digit Euro upfront fee and are obligated to pay Normandie Valorisation a mid-single-digit Euro fee per each analysis utilizing Solid Tumor Plus Solution (STS+).
The term of each agreement continues until the expiration of the last-to-expire licensed patent in September 2033, unless automatically terminated upon our cessation of business, bankruptcy or insolvency or upon the invalidation of the applicable licensed patents in France. In addition, Normandie Valorisation may terminate either agreement if we challenge the validity or enforceability of the licensed patents. Either party may terminate either agreement in the event of non-performance by the other party of one or more of its obligations under such agreement which is not cured within three months of receipt of written notice of such non-performance. Either party may also terminate the 2019 Normandie Agreement at any time, with one year’s prior written notice.
SATT Aquitaine—Software Sublicense Agreement
In November 2017, we entered into a software sublicense agreement with Aquitaine Science Transfert (“SATT Aquitaine”), pursuant to which SATT Aquitaine granted us a non-exclusive, worldwide sublicense under certain intellectual property rights exclusively licensed to SATT Aquitaine to reproduce, adapt, use and distribute, and create derivative works based on, certain modeling software applications in the field of oncology. The right to use such software applications granted to us includes the right to provide software-related services to our customers.
In exchange for the rights granted to us, we paid SATT Aquitaine a low six-digit Euro upfront payment. Intellectual property rights in any derivative software applications developed by us shall be owned by us in accordance with French intellectual property law.
The agreement is effective for 15 years unless automatically terminated upon our cessation of business, dissolution, bankruptcy or liquidation. In addition, either party may terminate the agreement in the event of non-performance by the other party of one or more of its obligations under the agreement which are not cured within three months of receipt of written notice of such non-performance.
Collaboration Agreements
We have built an agnostic platform that enables others to get the most out of their data. As such, our model is valuable to all types of players within the health care ecosystem. For hardware and consumable players our platform and smart algorithms bring large benefits to their customer base as it allows for a more rapid set-up of their technology, better results in terms of clinical outcomes on the same hardware, bringing value to a larger number of samples and as such higher volumes.
We collaborate not only to provide valuable technologies to hospitals and laboratories around the world directly or through collaborators, but also to increase our channels and to offer a variety of application and product choices to our large customer base.
GE Healthcare—Master Alliance Agreement
In November 2021, we entered into a Master Alliance Agreement with GE Healthcare. Under this agreement, we will be working together with GE Healthcare on a variety of opportunities in the healthcare market and will collaborate on different initiatives and projects in the fields of digital oncology and radiogenomic analysis. The first projects we and GE Healthcare are working on together relate to the creation of infrastructure to integrate data between GE Healthcare’s Edison platform and our SOPHiA DDM Platform, as well as a commercial collaboration focused on co-marketing and pilot site recruitment in the digital oncology and radiogenomic analysis space. We also intend to work together to combine our and GE’s existing capabilities to jointly develop and provide multimodal analytics.
124
The term of the Master Alliance Agreement is five years, with automatic one-year renewals thereafter, unless earlier terminated for cause or, after three years after the execution of the Agreement, if no statements of work are in effect.
Agilent—Co-Marketing Agreement
In December 2020, we entered into an agreement for the co-marketing of products and services (the “Co-Marketing Agreement”), with Agilent, pursuant to which we agreed to collaborate with Agilent for the development and commercialization of sequencing diagnostic products.
Pursuant to the Co-Marketing Agreement, we agreed to collaborate with Agilent to develop a diagnostic product for worldwide commercialization that will be specifically compatible with Agilent’s Magnis SureSelect assays. The collaboration under the Co-Marketing Agreement is non-exclusive, and neither party is restricted from entering into similar collaborations with other third parties so long as it does not breach its obligations to perform under the Co-Marketing Agreement.
Each party retains all rights to its background intellectual property provided in connection with the development or commercialization of any diagnostic product developed under the Co-Marketing Agreement, including the commercialization rights thereto, subject to certain non-exclusive licenses granted by each party to the other under its trademarks and confidential information. Any intellectual property arising from the performance of the Co-Marketing Agreement that is substantially developed solely by a party will be owned by such party. Any intellectual property that is substantially developed jointly under the Co-Marketing Agreement shall be jointly owned by the parties, and both parties are prohibited from using such jointly-owned intellectual property for any purposes other than certain specified activities relating to gene panels. The parties may enter into additional agreements regarding the use and rights relating to such jointly-developed intellectual property.
The Co-Marketing Agreement is effective for two years, with potential successive one-year renewal options exercisable by the parties, and may be terminated by either party upon (i) a material breach by the other party (subject to a certain specified cure period) or (ii) 180 days’ prior written notice, provided that the parties must follow an express wind-down procedure upon any expiration or termination.
IDT—Manufacturing and Supply Agreement
In December 2015, we entered into a manufacturing and supply agreement with IDT for the manufacture and supply of various commercial and research products, in particular DNA enrichment kits. The agreement was amended and restated in October 2018 and amended in March 2019.
Under the agreement, IDT committed to manufacture and supply to us or directly to our customers, as applicable, various products, in particular DNA enrichment kits. IDT shall manufacture the products covered by this agreement in accordance with product specifications provided by us, (such products, “commercial products”). IDT is required to certify that the delivered products were manufactured in accordance with said specifications. In addition, we can purchase products already manufactured by IDT pursuant to its own specifications, (such products, “research products”) and resell such products to our customers and collaborators.
Purchase prices for both commercial and research products are set upon completion of the corresponding master specification document. We are obligated to provide IDT on a quarterly basis with written good faith non-binding purchasing forecasts of the aggregate quantities of commercial products to be purchased by us in the following twelve months and IDT is obligated to ensure sufficient inventory and manufacturing capacity to deliver the forecasted quantities. We are not required to purchase the commercial products from IDT in the previously forecasted quantities. However, if we fail to purchase a specified percentage of the forecasted quantities of commercial products, IDT will have the right to increase the prices of such products by amounts in excess of the additional manufacturing costs associated with our reduced purchase. At the same time, we have the right to place purchase orders for products beyond the previously forecasted quantities and IDT is obligated to use its commercially reasonable efforts to fill such orders at regular prices. We are not required to provide IDT with forecasts for research products that we intend to purchase in any given period. The purchase prices for such products are not adjustable based on the quantities actually ordered by us.
All inventions made directly as a result of the manufacture of commercial and research products that are improvements to such products will be owned by us. IDT will obtain a fully-paid up, royalty-free, worldwide
125
license under such improvements to make, have made, use and sell the commercial and research products exclusively for and to us.
The agreement has an initial term of five years. It will automatically renew for additional one-year periods unless a party notifies the other of its decision not to renew the agreement. We also have the right to terminate the agreement if we do not agree with changes made by IDT in accordance with the provisions of the agreement, including in connection with changes to master specification documents for any research or commercial product. In addition, each party has the right to terminate the agreement effective immediately if the other party fails to meet essential terms of the agreement and such failure is not cured.
Qiagen—OEM Supply Agreement
In January 2018, we entered into an OEM supply agreement with Qiagen for the supply of certain amplification technologies required to complement our NGS technologies. The agreement was amended in June 2019.
Under the agreement, we receive the exclusive right to offer and resell the products manufactured by Qiagen as part of “bundle” solutions. We cannot offer such products as stand-alone or single items, unless it is required for product replacement or quality control reasons or in other special circumstances discussed by the parties in good faith. Qiagen is obligated to use its commercially reasonable efforts to manufacture and deliver products in quantities ordered by us. We are required to provide Qiagen in writing with a monthly rolling forecast covering the next six months. In addition, both parties shall meet at least once every quarter to discuss any adjustments to the forecast. Furthermore, we are obligated to purchase minimum quantities of products every year. If we fail to do so in any given calendar year, we will be obligated to make a one-time payment at the end of the year in an amount equal to the difference between the value of the products forecasted to be purchased in that year and the products actually purchased.
The agreement had an initial term of three years. It automatically extended for one 2-year period which will be followed by 1-year periods unless a party notifies the other of its decision not to renew the agreement. The parties may terminate the agreement effective immediately for cause or upon notice in certain situations.
NEB—Supply Agreement
In January 2019, we entered into a supply agreement with NEB for the supply of various reagents. These products may only be used in the research field and in compliance with applicable intended use statements, limited use statements or limited label licenses.
We are required to provide NEB with twelve-months' rolling written forecasts of our estimated product supply requirements. The forecasts for the certain months of each twelve-month rolling period are binding, and the forecasts for the remaining months are non-binding and serve only as a good faith estimate to facilitate NEB’s production scheduling.
New England Biolabs shall not unreasonably reject any purchase order and shall use commercially reasonable efforts to fill purchase orders for any quantity of product that, alone or cumulatively with other purchase orders submitted to it for product delivery during the relevant calendar quarter, exceeds the amount specified in the binding portion of the then-current rolling forecast.
Prior to each shipment of products, NEB shall perform quality control procedures reasonably necessary to ensure that the products to be shipped conform fully to the specifications as agreed upon by us with NEB from time to time.
The agreement had an initial term of three years. It has been automatically extended for a two-year period and thereafter it will automatically extend for successive two-year periods, unless either party notifies the other party in writing at least six months prior to the expiration of the then-current term that such party does not wish to continue the agreement.
Twist—Supply Agreement
In November 2019, we entered into a supply agreement with Twist for the manufacture and supply of various RUO products, in particular target enrichment and library construction reagent sets.
126
Under the agreement, we may offer and resell products manufactured by Twist as part of “bundle” solutions. We may not offer such product as stand-alone products, unless we obtain Twist’s written consent. Twist is obligated to use its commercially best efforts to manufacture and deliver products in quantities ordered by us. We are required to provide Twist with a non-binding good faith six-months’ rolling written forecast, setting out forecasted quantities of the products to be purchased by us in monthly increments.
We can provide Twist with specifications of the products to be supplied by Twist or order ready-to-use products already manufactured by Twist. Once Twist accepts a purchase order from us, we are not allowed to cancel such an order, but Twist may cancel the purchase order if it cannot fulfill such order despite using commercially best efforts.
The agreement has an initial term of three years. It will automatically extend for one-year periods unless a party notifies the other of its decision not to renew the agreement. In addition, either party may terminate it without cause.
D. Exchange Controls
There are no Swiss governmental laws, decrees or regulations that restrict, in a manner material to us, the export or import of capital, including any foreign exchange controls, or that generally affect the remittance of dividends or other payments to non-residents or non-citizens of Switzerland who hold our ordinary shares.
E. Taxation
The following discussion is based on the tax laws, regulations and regulatory practices of Switzerland and the United States as in effect on the date hereof, which are subject to change (or subject to changes in interpretation), possibly with retroactive effect.
Current and prospective shareholders are advised to consult their own tax advisors in light of their particular circumstances as to the Swiss or U.S. tax laws, regulations and regulatory practices that could be relevant for them, the acquiring, owning and selling or otherwise disposing of our ordinary shares and receiving dividends and similar cash or in-kind distributions on our ordinary shares (including dividends on liquidation proceeds and share dividends) or distributions on our ordinary shares based upon a capital reduction or reserves paid out of capital contributions and the consequences thereof under the tax laws, regulations and regulatory practices of Switzerland or the United States.
Swiss Tax Considerations
Swiss Federal Withholding Tax
Dividends and other cash or in-kind distributions (including scrip or stock dividends), if any, on ordinary shares made or paid by us out of reserves from capital contributions (réserves d’apports de capital) and distributions, if any, on ordinary shares made or paid by us based upon a reduction of nominal value of ordinary shares (réduction de la valeur nominale) and the purchase price for ordinary shares bought back, if any, by us for a capital reduction booked against reserves from capital contributions and nominal value of ordinary shares, are exempt from Swiss federal withholding tax. The proceeds from the offering of the ordinary shares (net of certain deductions) will qualify as reserves from capital contributions and as nominal value of the ordinary shares.
Dividends and other cash or in-kind distributions (including scrip or stock dividends), if any, on ordinary shares made or paid by us out of profit or reserves other than reserves from capital contributions and the purchase price for ordinary shares bought back, if any, by us for a capital reduction booked against reserves other than reserves from capital contributions, are subject to Swiss federal withholding tax at a rate of 35%. Any Swiss federal withholding tax must be withheld by us on the gross amount of the dividend or distribution or purchase price, as applicable, and be remitted to the Swiss Federal Tax Administration.
Capital gains realized on the sale of ordinary shares in the secondary market are not subject to Swiss federal withholding tax.
127
Refund of Withholding Tax on Taxable Distributions
The Swiss Federal Tax Administration or the relevant cantonal tax authority, as applicable, will refund or credit Swiss federal withholding tax on dividends or other cash or in-kind distributions (including scrip or stock dividends), if any, on ordinary shares made or paid by us, or the purchase price paid by us for ordinary shares bought back, if any, for a capital reduction, out of or against profit or reserves other than tax-exempt reserves from capital contributions in full to holders who are individuals resident in Switzerland and to holders, who hold the ordinary shares on which the dividends or other distributions have been paid, as part of a trade or business in Switzerland, and, who, in each case, inter alia, are the beneficial owners of the ordinary shares and duly report the dividend or distributions or the purchase price paid by us for ordinary shares bought back for a capital reduction in the income tax return or the financial statements, respectively, for the relevant tax period.
A holder who is a resident of the United States for purposes of the double taxation agreement between the United States and Switzerland (the “Treaty”) without taxable presence in Switzerland to which the ordinary shares are attributable or who is a qualified U.S. pension fund and who, in each case, is the beneficial owner of the ordinary shares and the dividend or distribution and who meets the conditions of the Treaty may apply for a full refund of the Swiss federal withholding tax in the case of qualified U.S. pension funds or in excess of the amount of the 15% treaty rate in all other cases. The claim for refund must be filed on Swiss Tax Form 82 (82C for corporations, 82I for individuals, 82E for other entities and 82R for regulated investment companies), which forms together with an instruction form may be obtained from any Swiss consulate general in the United States, the Swiss Federal Tax Administration at the address below or be downloaded from the Swiss Federal Tax Administration’s website. Four copies of the form must be duly completed and signed before a notary public of the United States, and three of them must be sent to the Swiss Federal Tax Administration (currently at Eigerstrasse 65, CH-3003, Bern, Switzerland). The form must be accompanied by suitable evidence of deduction of the Swiss federal withholding tax, such as certificates of deduction, bank vouchers or credit slips. The form must be filed no later than December 31 of the third year following the calendar year in which the dividend subject to the tax became payable.
Any other holder who is not resident in Switzerland and who does not hold the ordinary shares as part of a trade or business in Switzerland, may be entitled to a full or partial refund of the Swiss federal withholding tax deducted if the country in which the recipient resides for tax purposes has entered into a bilateral treaty for the avoidance of double taxation with Switzerland, the recipient is the beneficial owner of the ordinary shares and the dividend or distribution or the purchase price and the other conditions of the treaty are met. Refund forms are available on the Swiss Federal Tax Administration’s website.
Swiss Federal Issue Stamp Tax
We will be liable to Swiss federal issue stamp tax on the issuance (droit d’émission sur capital propre) of the ordinary shares of 1% of the proceeds from the offering, net of certain deductions.
Swiss Federal Securities Turnover Tax
The delivery of ordinary shares to the initial purchasers of the ordinary shares against payment of the offer price will not be subject to Swiss securities turnover tax (droit de négociation).
Any subsequent transactions in ordinary shares in the secondary markets are subject to Swiss federal securities turnover tax at a rate of 0.15% of the purchase price of the ordinary shares if a Swiss or Liechtenstein domestic bank or securities dealer (as defined in the Swiss Federal Stamp Tax Act) is a party or an intermediary to the transaction, and none of the exemptions provided for in the Swiss Federal Stamp Tax Act applies. Generally, half of the tax is charged to the one party to the transaction and the other half to the other party, subject to applicable statutory exemptions in respect of the one or the other party to the transaction and their respective halves of the tax. Secondary market dealings in ordinary shares where no domestic bank or securities dealer is a party or an intermediary to the transaction are not subject to Swiss federal securities turnover tax.
128
Swiss Federal, Cantonal and Communal Income Taxes
Ordinary Shares Held By Holders Resident Outside Of Switzerland and With No Trade or Business in Switzerland
Holders of ordinary shares who are not residents of Switzerland for tax purposes, and who during the taxable year have not held ordinary shares through a permanent establishment within Switzerland for tax purposes, are not subject to any Swiss federal, cantonal or communal income tax in respect of the receipt of dividends, or other distributions, if any, on ordinary shares, or gain realized on the sale or other disposition of ordinary shares.
For a discussion of the Swiss federal withholding tax treatment of dividends and distributions or capital gains on ordinary shares, see above “—Swiss Federal Withholding Tax.” For a discussion of the automatic exchange of information in tax matters, see below “—International Automatic Exchange of Information in Tax Matters” and for a discussion of the Swiss facilitation of the implementation of the FATCA, see below “—Swiss Facilitation of the Implementation of FATCA.”
Ordinary Shares Held By Swiss Resident Individuals as Private Investments
Dividends and other cash or in-kind distributions (including scrip or stock dividends), if any, on ordinary shares, to the extent made or paid by us out of reserves from capital contributions and distributions, if any, to the extent made or paid by us on ordinary shares based upon a capital reduction, and the purchase price of ordinary shares bought back for a capital reduction charged to reserves from capital contributions are exempt from Swiss federal, cantonal and communal income tax for holders of ordinary shares who are individuals resident in Switzerland for tax purposes and who hold the ordinary shares as private investments.
Conversely, any dividends and other cash or in-kind distributions (including scrip or stock dividends), if any, on ordinary shares to the extent made or paid by us out of profit and reserves other than reserves from capital contributions, and the purchase price for ordinary shares bought back, if any, by us for a capital reduction to the extent booked against reserves other than reserves from capital contributions, will be subject to Swiss federal, cantonal and communal taxable income for such holders.
A capital gain realized by a holder on the sale of ordinary shares (other than a sale to us in a share buy-back for capital reduction) held as private investments classifies as tax-exempt private capital gain and, vice versa, a capital loss as non-tax deductible private capital loss for purposes of Swiss federal, cantonal and communal income tax.
See below “—Ordinary shares held as assets of a Swiss business” for a summary of the taxation treatment of Swiss resident individuals who, for income tax purposes, are classified as “professional securities dealers.”
Ordinary Shares Held As Assets of a Swiss Business
For a corporate or an individual who holds the ordinary shares as part of a trade or business carried on in Switzerland, any dividends and any other distributions, if any, made or paid by us on ordinary shares, and any capital gain or loss realized on the sale of ordinary shares, are includible in, or deductible from, respectively, the taxable income in the relevant taxation period for purposes of Swiss federal, cantonal and communal individual or corporate income tax. This taxation treatment also applies to Swiss resident private individuals who, for income tax purposes, are classified as “professional securities dealers.”
Corporate taxpayers will be eligible for dividend relief (réduction pour participations) in respect of dividends and distributions, if any, on ordinary shares if either the market value of the ordinary shares held by them equals or exceeds CHF 1.0 million or the ordinary shares represent 10% or more of our share capital.
International Automatic Exchange of Information in Tax Matters
Switzerland has concluded a multilateral agreement with the EU on the international automatic exchange of information (“AEOI”) in tax matters, which applies to all EU member states. In addition, Switzerland signed the multilateral competent authority agreement on the automatic exchange of financial account information (the “MCAA”) and a number of bilateral AEOI agreements with other countries, most of them on the basis of the MCAA. Based on these agreements and the implementing laws of Switzerland, Switzerland collects and exchanges data in respect of financial assets held in, and income derived thereon and credited to, accounts or
129
deposits (including ordinary shares held in such accounts or deposits) with a paying agent in Switzerland for the benefit of individuals resident in an EU member state or in another treaty state. An up-to-date list of the AEOI agreements to which Switzerland is a party that are in effect, or signed but not yet in effect, can be found on the website of the State Secretariat for International Financial Matters SIF.
Swiss Facilitation of the Implementation of FATCA
The United States and Switzerland entered into an intergovernmental agreement (the “U.S.-Switzerland IGA”) to facilitate the implementation of FATCA. Under the U.S.-Switzerland IGA, financial institutions acting out of Switzerland generally are directed to become participating foreign financial institutions. The U.S.-Switzerland IGA ensures that accounts held by U.S. persons with Swiss financial institutions (including accounts in which ordinary shares are held) are disclosed to the U.S. tax authorities either with the consent of the account holder or by means of group requests within the scope of administrative assistance, on the basis of the Treaty. The Treaty, as amended in 2019, includes a mechanism for the exchange of information in tax matters upon request between Switzerland and the United States, which is in line with international standards, and allows the United States to make group requests under FATCA concerning non-consenting U.S. accounts and non-consenting non-participating foreign financial institutions for periods from June 30, 2014. Furthermore, the Swiss Federal Council approved a mandate for negotiations with the United States on October 8, 2014, with regard to a change from the current direct-notification-based regime to a regime where the relevant information is sent to the Swiss Federal Tax Administration, which in turn provides the information to the U.S. tax authorities. It is not yet known when negotiations will continue and if and when any new regime would come into force.
Material U.S. Federal Income Tax Consequences for U.S. Holders
The following is a description of the material U.S. federal income tax consequences to U.S. Holders, as defined below, of owning and disposing of our ordinary shares. It does not describe all tax consequences that may be relevant to a particular person’s decision to acquire ordinary shares.
This discussion applies only to a U.S. Holder that holds ordinary shares as capital assets for U.S. federal income tax purposes within the meaning of Section 1221 of the Code (generally, property held for investment). In addition, it does not describe any tax consequences other than U.S. federal income tax consequences, including state and local tax consequences and estate or gift tax consequences, and does not describe all of the U.S. federal income tax consequences that may be relevant in light of the U.S. Holder’s particular circumstances, including alternative minimum tax consequences, the special tax accounting rules under Section 451(b) of the Code, the potential application of the Medicare contribution tax on net investment income, and tax consequences applicable to U.S. Holders subject to special rules, such as:
•certain banks, insurance companies and other financial institutions;
•brokers, dealers or traders in securities who use a mark-to-market method of tax accounting;
•persons holding ordinary shares as part of a straddle, wash sale, conversion transaction or other integrated transaction or persons entering into a constructive sale with respect to the ordinary shares;
•persons whose functional currency for U.S. federal income tax purposes is not the U.S. dollar;
•entities or arrangements classified as partnerships or S corporations for U.S. federal income tax purposes and investors in such entities;
•tax-exempt entities, including an “individual retirement account” or “Roth IRA” or governmental entities;
•real estate investment trusts or regulated investment companies;
•former U.S. citizens or long-term residents of the United States;
•persons that own or are deemed to own 10% or more of the voting power or value of our shares; or
130
•persons holding ordinary shares in connection with a trade or business conducted outside of the United States or in connection with a permanent establishment or other fixed place of business outside of the United States.
If an entity or arrangement that is classified as a partnership for U.S. federal income tax purposes holds ordinary shares, the U.S. federal income tax treatment of a partner will generally depend on the status of the partner and the activities of the partnership. Partnerships holding ordinary shares and partners in such partnerships should consult their tax advisors as to the particular U.S. federal income tax consequences of owning and disposing of the ordinary shares in their particular circumstances.
This discussion is based on the Code, administrative pronouncements, judicial decisions, final, temporary and proposed Treasury regulations, and the Treaty, all as of the date hereof, any of which is subject to change or differing interpretations, possibly with retroactive effect.
A “U.S. Holder” is a holder who, for U.S. federal income tax purposes, is a beneficial owner of ordinary shares, who is eligible for the benefits of the Treaty and who is:
•an individual who is a citizen or resident of the United States;
•a corporation, or other entity taxable as a corporation, created or organized in or under the laws of the United States, any state therein or the District of Columbia; or
•an estate or trust, the income of which is subject to U.S. federal income taxation regardless of its source.
U.S. Holders should consult their tax advisors concerning the U.S. federal, state, local and non-U.S. tax consequences of owning and disposing of ordinary shares in their particular circumstances.
Treasury regulations that apply to taxable years beginning on or after December 28, 2021 (the “Foreign Tax Credit Regulations”), may in some circumstances prohibit a U.S. person from claiming a foreign tax credit with respect to certain non-U.S. taxes that are not creditable under applicable income tax treaties. Accordingly, U.S. investors that are not eligible for Treaty benefits should consult their tax advisors regarding the creditability or deductibility of any Swiss taxes imposed on dividends on, or dispositions of, the ordinary shares. The discussions below regarding the creditability or deductibility of Swiss taxes, if any, do not apply to investors in this special situation. However, the Internal Revenue Service released a notice that provides relief from certain
of the provisions of the Treasury regulations described above for taxable years ending before the date that a
notice or other guidance withdrawing or modifying the temporary relief is issued (or any later date specified in
such notice or other guidance).
Passive Foreign Investment Company Rules
Under the Code, we will be a PFIC for any taxable year in which, after the application of certain “look-through” rules with respect to subsidiaries, either (i) 75% or more of our gross income consists of “passive income,” or (ii) 50% or more of the average quarterly value of our assets consists of assets that produce, or are held for the production of, “passive income.” For purposes of the above calculations, we will be treated as if we hold our proportionate share of the assets of, and receive directly our proportionate share of the income of, any other corporation in which we directly or indirectly own at least 25%, by value, of the shares of such corporation. Passive income generally includes dividends, interest, rents, certain non-active royalties and capital gains.
Cash is generally characterized as a passive asset for these purposes. Goodwill, is generally characterized as a non-passive or passive asset based on the nature of the income produced in the activity to which the goodwill is attributable. The extent to which our goodwill should be characterized as a non-passive asset is not entirely clear. We hold a substantial amount of cash, and while this continues to be the case our PFIC status for any taxable year will depend largely on the value of our goodwill and the characterization of our goodwill as passive or non-passive. The value of our goodwill for any taxable year may be determined in large part by reference to the average of our market capitalization for that year. Although we believe that we were a PFIC for our 2022 taxable year, based the nature of our operations and assets, as well as certain as to our income and the relative values of our assets during our 2023 taxable year, we believe that we were not a PFIC for our 2023 taxable year
131
(subject to the discussion in the next paragraph with respect to U.S. Holders who held our ordinary shares during our 2022 taxable year). There can be no assurance that the IRS will agree with our conclusion. In addition, we have not obtained any valuation of our assets (including goodwill). U.S. Holders should consult their tax advisors regarding the value and characterization of our assets for purposes of the PFIC rules, which are subject to some uncertainties. In addition, our PFIC status is a factual annual determination that can be made only after the end of the relevant taxable year and will depend on the composition of our income and assets and the value of our assets from time to time. Accordingly, our PFIC status for 2024 and any future taxable year is uncertain.
If we are a PFIC for any year during which a U.S. Holder holds ordinary shares, we would generally continue to be treated as a PFIC with respect to such holder for all succeeding years during which such holder holds ordinary shares, even if we ceased to meet the threshold requirements for PFIC status. Accordingly, since we believe that we were a PFIC for our 2022 taxable year, we believe that we will remain a PFIC with respect to a U.S. Holder that held our ordinary shares during our 2022 taxable year. U.S. Holders should consult their tax advisors regarding the advisability of making a “deemed sale” election that will allow them to eliminate the continuing PFIC status under certain circumstances, but may require them to recognize gain taxed under the general PFIC rules described in this section. Additionally, in the event that the U.S. Holder made a QEF or mark to market election (as described below) for the year that we were a PFIC, special rules may apply that mitigate the rules described in this paragraph.
If we were a PFIC for any taxable year and any of our subsidiaries or other companies in which we owned or were treated as owning equity interests were also a PFIC (any such entity, a “Lower-tier PFIC”), a U.S. Holder would be deemed to own a proportionate amount (by value) of the shares of each Lower-tier PFIC and would be subject to U.S. federal income tax according to the rules described in the subsequent paragraph on (i) certain distributions by a Lower-tier PFIC and (ii) dispositions of shares of Lower-tier PFICs, in each case as if such holder held such shares directly, even though such holder will not have received the proceeds of those distributions or dispositions.
If we are a PFIC for any taxable year during which a U.S. Holder holds any of our ordinary shares, such holder will generally be subject to adverse tax consequences. Unless a U.S. Holder makes a timely “mark to market” election or “qualified electing fund” ("QEF") election, each as discussed below, gain recognized upon a disposition (including, under certain circumstances, a pledge) of ordinary shares will be allocated ratably over a U.S. Holder’s holding period for the ordinary shares. The amounts allocated to the taxable year of disposition and to years before we became a PFIC will be taxed as ordinary income. The amount allocated to each other taxable year will be subject to tax at the highest rate in effect for that taxable year for individuals or corporations, as appropriate, and an interest charge will be imposed on the tax on such amount. Further, to the extent that any distributions received on a U.S. Holder’s ordinary shares during a taxable year exceed 125% of the average of the annual distributions on those shares during the preceding three years or such holder’s holding period, whichever is shorter, those distributions will be subject to taxation in the same manner as gain, described immediately above.
Alternatively, if we are a PFIC and if the ordinary shares are “regularly traded” on a “qualified exchange,” a U.S. Holder will be eligible to make a mark-to-market election that will result in tax treatment different from the general tax treatment for PFICs described above. The ordinary shares will be treated as “regularly traded” if more than a de minimis amount of the ordinary shares are traded on a qualified exchange on at least 15 days during each calendar quarter (the “15-Day Test”). The Nasdaq, on which the ordinary shares are listed, is a qualified exchange for this purpose. Once made, the election cannot be revoked without the consent of the IRS unless the shares cease to be marketable.
If a U.S. Holder makes the mark-to-market election, such holder will generally recognize as ordinary income any excess of the fair market value of such holder’s ordinary shares at the end of each taxable year over their adjusted tax basis, and will recognize an ordinary loss in respect of any excess of the adjusted tax basis of the ordinary shares over their fair market value at the end of the taxable year (but only to the extent of the net amount of income previously included as a result of the mark-to-market election). If a U.S. Holder makes the election, such holder’s tax basis in their ordinary shares will be adjusted to reflect these income or loss amounts. Any gain recognized on the sale or other disposition of ordinary shares in a year when we are a PFIC will be treated as ordinary income and any loss will be treated as an ordinary loss (but only to the extent of the net amount of income previously included as a result of the mark-to-market election). This election will not apply to any of our non-U.S. subsidiaries. Accordingly, a U.S. Holder may continue to be subject to tax under the PFIC
132
excess distribution regime with respect to any Lower-tier PFICs notwithstanding a mark-to-market election for the ordinary shares.
In addition, if we are a PFIC for any taxable year in which we pay a dividend or for the prior taxable year, the preferential dividend rates discussed below with respect to dividends paid to certain non-corporate U.S. Holders will not apply.
If a company that is a PFIC provides certain information to U.S. Holders, a U.S. Holder can then avoid certain adverse tax consequences described above by making a QEF election to be taxed currently on its proportionate share of the PFIC’s ordinary income and net capital gains.
The QEF election is made on a shareholder-by-shareholder basis and, once made, can be revoked only with the consent of the IRS. A U.S. Holder generally makes a QEF election by attaching a completed IRS Form 8621, including the information provided in a PFIC Annual Information Statement, to a timely filed U.S. federal income tax return for the taxable year to which the election relates. U.S. Holders should consult their tax advisor regarding the availability and tax consequences of a retroactive QEF election under their particular circumstances. In order to comply with the requirements of a QEF election, a U.S. Holder must receive a PFIC Annual Information Statement from us.
If a U.S. Holder owns ordinary shares during any year in which we are a PFIC, such holder must generally file annual reports containing such information as the U.S. Treasury may require on IRS Form 8621 (or any successor form) with respect to us, generally with such holder’s federal income tax return for that year.
The rules dealing with PFICs and with the mark-to-market and QEF elections are complex and are affected by various factors in addition to those described above. Accordingly, U.S. Holders should consult their tax advisors concerning the application of the PFIC rules to our ordinary share under their particular circumstances. As discussed above, special rules may apply if a U.S. Holder made a QEF election with respect to our 2022 taxable year. U.S. Holders who made a QEF election for our 2022 taxable year should consult their tax advisors.
Information Returns
If a U.S. Holder owns ordinary shares during any year in which we are a PFIC or in which we hold a direct or indirect equity interest in a Lower-tier PFIC, the U.S. Holder generally must file an annual report on IRS Form 8621 with respect to each such PFIC containing such information as the U.S. Treasury may require, generally with the U.S. Holder’s U.S. federal income tax return for the relevant year. A U.S. Holder’s failure to file the annual report will cause the statute of limitations for such U.S. Holder’s U.S. federal income tax return to remain open with respect to the items required to be included in such report until three years after the U.S. Holder files the annual report and, unless such failure is due to reasonable cause and not willful neglect, the statute of limitations for the U.S. Holder’s entire U.S. federal income tax return will remain open during such period.
PROSPECTIVE U.S. HOLDERS SHOULD CONSULT THEIR TAX ADVISORS REGARDING THE CONSEQUENCES OF OUR POTENTIAL PFIC STATUS ON AN INVESTMENT IN ORDINARY SHARES.
Taxation of Distributions
The following is subject to the discussion regarding the PFIC rules described above.
As discussed above under “Dividend Policy,” we do not currently expect to make distributions on our ordinary shares. In the event that we do make distributions of cash or other property, distributions paid on ordinary shares, other than certain pro rata distributions of ordinary shares, will generally be treated as dividends to the extent paid out of our current or accumulated earnings and profits (as determined under U.S. federal income tax principles). Because we do not maintain calculations of our earnings and profits under U.S. federal income tax principles, we expect that distributions generally will be reported to U.S. Holders as dividends. For so long as our ordinary shares are listed on the Nasdaq or we are eligible for benefits under the Treaty, dividends paid to certain non-corporate U.S. Holders will be eligible for taxation as “qualified dividend income” and therefore, subject to applicable holding period requirements, will be taxable at rates not in excess of the long-term capital gain rate applicable to such U.S. Holder.
The amount of a dividend will include any amounts withheld by us in respect of Swiss income taxes. The amount of the dividend will be treated as foreign-source dividend income to U.S. Holders and will not be eligible
133
for the dividends-received deduction generally available to U.S. corporations under the Code. Dividends will be included in a U.S. Holder’s income on the date of the U.S. Holder’s receipt of the dividend. The amount of any dividend income paid in Swiss francs will be the U.S. dollar amount calculated by reference to the exchange rate in effect on the date of actual or constructive receipt, regardless of whether the payment is in fact converted into U.S. dollars at that time. If the dividend is converted into U.S. dollars on the date of receipt, a U.S. Holder should not be required to recognize foreign currency gain or loss in respect of the dividend income. A U.S. Holder may have foreign currency gain or loss if the dividend is converted into U.S. dollars after the date of receipt.
Subject to applicable limitations, some of which vary depending upon the U.S. Holder’s particular circumstances, Swiss income taxes withheld from dividends on ordinary shares (at a rate not exceeding the rate provided by the Treaty) may be creditable against the U.S. Holder’s U.S. federal income tax liability. The rules governing foreign tax credits are complex and U.S. Holders should consult their tax advisors regarding the creditability of foreign taxes in their particular circumstances. In lieu of claiming a foreign tax credit, U.S. Holders may, at their election, deduct foreign taxes, including any Swiss income tax, in computing their taxable income, subject to generally applicable limitations under U.S. law. An election to deduct foreign taxes instead of claiming foreign tax credits applies to all foreign taxes paid or accrued in the taxable year.
Sale or Other Disposition of Ordinary Shares
The following is subject to the discussion regarding the PFIC rules described above.
Gain or loss realized by a U.S. Holder on the sale or other disposition of ordinary shares will be capital gain or loss, and will be long-term capital gain or loss if the U.S. Holder’s holding period for such ordinary shares was more than one year as of the date of the sale or other disposition. The amount of the gain or loss will equal the difference between the U.S. Holder’s tax basis in the ordinary shares disposed of and the amount realized on the disposition, in each case as determined in U.S. dollars. Long-term capital gain recognized by a non-corporate U.S. Holder is subject to U.S. federal income tax at rates lower than the rates applicable to ordinary income and short-term capital gains, while short-term capital gains are subject to U.S. federal income tax at the rates applicable to ordinary income. This gain or loss will generally be U.S.-source gain or loss for foreign tax credit purposes. The deductibility of capital losses is subject to various limitations.
Information Reporting and Backup Withholding
Payments of dividends and sales proceeds that are made within the United States or through certain U.S.-related financial intermediaries generally are subject to information reporting, and may be subject to backup withholding, unless (i) the U.S. Holder is a corporation or other exempt recipient or (ii) in the case of backup withholding, the U.S. Holder provides a correct taxpayer identification number and certifies that it is not subject to backup withholding.
The amount of any backup withholding from a payment to a U.S. Holder will be allowed as a credit against the U.S. Holder’s U.S. federal income tax liability and may entitle it to a refund, provided that the required information is timely furnished to the IRS.
Information with Respect to Foreign Financial Assets
Certain U.S. Holders who are individuals (and, under proposed regulations, certain entities) may be required to report information relating to an interest in our ordinary shares, subject to certain exceptions (including an exception for ordinary shares held in accounts maintained by certain U.S. financial institutions). Such U.S. Holders who fail to timely furnish the required information may be subject to a penalty. Additionally, if a U.S. Holder does not file the required information, the statute of limitations with respect to tax returns of the U.S. Holder to which the information relates may not close until three years after such information is filed. U.S. Holders should consult their tax advisors regarding the effect, if any, of this legislation on their ownership and disposition of the ordinary shares.
H. Documents on Display
We are subject to the informational requirements of the Exchange Act. Accordingly, we file reports and other information with the SEC, including annual reports on Form 20-F and reports on Form 6-K. The SEC maintains an Internet site at www.sec.gov that contains reports, proxy and information statements and other information
134
we have filed electronically with the SEC. As a foreign private issuer, we are exempt under the Exchange Act from, among other things, the rules prescribing the furnishing and content of proxy statements, and our executive officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. In addition, we are not required under the Exchange Act to file periodic reports and financial statements with the SEC as frequently or as promptly as U.S. companies whose securities are registered under the Exchange Act.
Additionally, pursuant to Swiss law, any shareholder of record has the right to receive a free copy of this Annual Report and to inspect this Annual Report at any time at our registered office in Rolle, Canton of Vaud, Switzerland.
We also make available on our website, free of charge, our Annual Report and the text of our reports on Form 6-K, including any amendments to these reports, as well as certain other SEC filings, as soon as reasonably practicable after they are electronically filed with or furnished to the SEC. Our website address is www.sophiagenetics.com. The reference to our website is an inactive textual reference only, and information contained therein or connected thereto is not incorporated into this Annual Report.
J. Annual Report to Security Holders
If we are required to provide an annual report to security holders in response to the requirements of Form 6-K, we will submit the annual report to security holders in electronic format in accordance with the EDGAR Filer Manual.
Item 11. Quantitative and Qualitative Disclosures About Market Risk
Interest Rate Risk
We had cash and cash equivalents totaling $123.3 million and $161.3 million as of December 31, 2023 and 2022, respectively, which are comprised of bank and short-term deposits with maturities up to three months. We also had no term deposits and term deposits of $17.3 million as of December 31, 2023 and 2022, respectively. Our cash and cash equivalents are subject to market risk due to changes in interest rates. Fixed rate securities may have their market value adversely affected due to a rise in interest rates. Due in part to these factors, our future investment income may fall short of expectation due to changes in interest rates or we may suffer losses in principal if we are forced to sell securities that decline in market value due to changes in interest rates.
As we currently do not have any outstanding debt, we are not subject to interest rate risk related to debt obligations.
We do not believe that a hypothetical 100 basis points change in interest rates would have a material effect on our business, financial condition or results of operations. We do not enter into investments for trading or speculative purposes. We do not use any financial instruments to manage our interest rate risk exposure.
Foreign Exchange Risk
We operate internationally and a portion of our revenue, expenses, assets, liabilities and cash flows are denominated in currencies other than our presentation currency. As a result, we are exposed to fluctuations in foreign exchange rates.
The sensitivity of our income to possible changes in foreign exchange rates is measured at the local entity level as it depends on the functional currency of each entity. For the years ended December 31, 2023 and 2022, we
135
were exposed principally to movements in four cross-currency pairs. The sensitivity of our loss before tax to such changes was as follows:
| December 31, | ||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||||||||||||||||||||
| Increase / (decrease) in USD/CHF exchange rate by 10% | 3,034 | / | (3,034) | 6,614 | / | (6,614) | 19,499 | / | (19,499) | |||||||||||||||||||||||||||||
| Increase / (decrease) in EUR/CHF exchange rate by 10% | 508 | / | (508) | (94) | / | 94 | 648 | / | (648) | |||||||||||||||||||||||||||||
| Increase / (decrease) in GBP/CHF exchange rate by 10% | (23) | / | 23 | (83) | / | 83 | (18) | / | 18 | |||||||||||||||||||||||||||||
| Increase / (decrease) in USD/EUR exchange rate by 10% | (513) | / | 513 | 503 | / | (503) | 726 | / | (726) | |||||||||||||||||||||||||||||
We do not believe that foreign exchange risk associated with other cross-currency pairs is material to our business, financial condition or results of operations.
Credit Risk
We are exposed to credit risk from our operating activities, primarily trade receivables. Credit risk is the risk that a counterparty will be unable to meet its obligations under a financial instrument or customer contract. Allowance is made for lifetime expected credit losses as invoices are issued. The amount of allowance initially recognized is based on historical experience, tempered by expected changes in future cash collections, due to, for example, expected improved customer liquidity or more active credit management.
We do not believe that credit risk had a material effect on our business, financial condition or results of operations. The largest outstanding balance represented 24% of trade and other receivables in 2023, which is attributable to one of our biopharma customers. This is due to a large upfront payment related to significant portions of related contracts completed in December 2023.The customer has a strong payment history and is in good standing with us. Our cash and cash equivalents are deposited with reputable financial institutions. If customers representing a significant percentage of our trade receivables are unable to meet their payment obligations to us, we may suffer harm to our business, financial condition or results of operations.
Inflation Risk
We believe our business is able to pass along increases in the costs of providing our applications, products, and services caused by inflation by increasing the prices of our applications, products, and services. For multi-year contracts, our general terms and conditions allow us to increase prices, at minimum on an annual basis. However, we do not believe that inflation had a material effect on our business, financial condition or results of operations. If our costs were to become subject to significant inflationary pressures, we may not be able to fully offset such higher costs through price increases. Our inability or failure to do so could harm our business, financial condition or results of operations.
Item 12. Description of Securities Other than Equity Securities
Not applicable.
136
PART II
Item 13. Defaults, Dividend Arrearages and Delinquencies.
None.
Item 14. Material Modifications to the Rights of Security Holders and Use of Proceeds
On July 22, 2021, the SEC declared effective our Registration Statement on Form F-1 (No. 333-257646) filed in connection with our initial public offering. J.P. Morgan Securities LLC, Morgan Stanley & Co. LLC, Cowen and Company, LLC and Credit Suisse Securities (USA) LLC acted as underwriters in the initial public offering. In the initial public offering, we issued and sold a total of 13,519,493 ordinary shares, including 519,493 ordinary shares pursuant to the underwriters’ exercise of their option to purchase additional shares. The public offering price was $18.00 per share. Our net proceeds from the initial public offering was $220.2 million, after deducting underwriting discounts and commissions of $17.0 million and offering expenses of $6.2 million. There has been no material changes in the planned use of proceeds as described in the our prospectus dated July 22, 2021, filed with the SEC pursuant to Rule 424(b)(4) under the Securities Act.
On July 27, 2021, in a private placement exempt from registration under Section 4(a)(2) of the Securities Act and the rules and regulations promulgated thereunder, we issued and sold ordinary shares to GE Healthcare. No underwriter or placement agent acted on our behalf in the private placement. In the private placement, we issued and sold 1,111,111 ordinary shares. The offering price was $18.00 per share. Our net proceeds from the private placement was $19.6 million, after deducting offering expenses of $0.4 million. We intend to use the net proceeds from the private placement for working capital and general corporate purposes.
Item 15. Controls and Procedures
A. Disclosure Controls and Procedures
As required by Rule 13a-15 under the Exchange Act, management, including our Chief Executive Officer and our Chief Financial Officer, has evaluated the effectiveness of our disclosure controls and procedures as of the end of the period covered by this report. Disclosure controls and procedures refer to controls and other procedures designed to ensure that information required to be disclosed in the reports we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by us in reports that we file or submit under the Exchange Act is accumulated and communicated to management, including our principal executive and principal financial officers, or persons performing similar functions, as appropriate to allow timely decisions regarding our required disclosures.
The Company’s management, with the participation of the Company’s Chief Executive Officer and Chief Financial Officer, has evaluated the effectiveness of the Company’s disclosure controls and procedures as of the end of the period covered by this report. Based on such evaluations, our Chief Executive Officer and Chief Financial Officer have concluded that, as of the end of such period, our disclosure controls and procedures were effective.
B. Management’s Annual Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Rule 13a-15(f) under the Exchange Act. Our internal control over financial reporting is a process designed by or under the supervision of the Chief Executive Officer and Chief Financial Officer, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of our financial statements for external reporting purposes in accordance with the IFRS Accounting Standards as issued by the International Accounting Standards Board (“IASB”).
As of December 31, 2023, our management conducted an assessment of the effectiveness of the Company’s internal control over financial reporting based on the criteria established in the Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission
137
(“COSO”). Based on this assessment, our management has determined that the Company’s internal control over financial reporting as of December 31, 2023 is effective.
C. Attestation Report of the Registered Public Accounting Firm
This Annual Report does not include an attestation report of the company’s independent registered public accounting firm because we are an emerging growth company under the JOBS Act.
D. Changes in Internal Control Over Financial Reporting
There were no changes to internal control over financial reporting during the year ended December 31, 2023 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 16A. Audit Committee Financial Expert
Our board of directors has determined that each of Didier Hirsch and Tomer Berkovitz are considered an “audit committee financial expert” as defined by the SEC. Our board of directors has determined that each of Didier Hirsch, Tomer Berkovitz, and Lila Tretikov satisfy the “independence” requirements under Nasdaq listing standards and Rule 10A-3 under the Exchange Act.
Item 16B. Code of Ethics
We have adopted the Code of Ethics, which is applicable to all of our employees, executive officers and directors. The Code of Ethics is available on our website www.sophiagenetics.com. Our board of directors is responsible for overseeing the Code of Ethics and will be required to approve any waivers of the Code of Ethics for our executive officers and directors. We expect that any amendments to the Code of Ethics, or any waivers of its requirements, will be disclosed on our website. For the year ended December 31, 2023, we did not grant any waivers of the Code of Ethics.
Item 16C. Principal Accountant Fees and Services
Fees
| For the year ended December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| Audit fees | $ | 905,525 | $ | 997,379 | ||||||||||
| Audit-related fees | 5,916 | 4,752 | ||||||||||||
| Tax fees | — | 5,426 | ||||||||||||
| All other fees | 9,875 | — | ||||||||||||
| Total fees | $ | 921,316 | $ | 1,007,557 | ||||||||||
For the year ended December 31, 2023 and 2022, PricewaterhouseCoopers SA was the Company’s auditor for the IFRS Accounting Standards and statutory accounts.
Audit fees include the standard audit work performed each fiscal year necessary to allow the auditor to issue an opinion on our financial statements and to issue an opinion on the local statutory financial statements. Audit fees also include services that can be provided only by the external auditor such as reviews of quarterly financial results and review of our securities offering documents.
Audit-related fees consist of fees billed for assurance and related services reasonably related to the performance of the audit or review of our financial statements or for services traditionally performed by an external auditor.
Tax services represent tax compliance, assistance with historical tax matters, and other tax-related services.
Other services include for statutory non-financial requirements and other reporting guidance databases.
138
Pre-Approval Policies and Procedures
In accordance with the requirements of the Sarbanes-Oxley Act and rules issued by the SEC, we review and pre-approve of any services performed by PricewaterhouseCoopers SA. The procedures require that all proposed future engagements of PricewaterhouseCoopers SA for audit and permitted non-audit work are submitted to the audit committee of our board of directors for approval prior to the beginning of any such service. In accordance with this policy, all services performed by and fees paid to PricewaterhouseCoopers SA in this Item 16C. were approved by the audit committee.
Item 16D. Exemptions from the Listing Standards for Audit Committees
None.
Item 16E. Purchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
Item 16F. Change in Registrant’s Certifying Accountant
None.
Item 16G. Corporate Governance
As a “foreign private issuer,” as defined by the SEC, we are permitted to follow home country corporate governance practices instead of certain corporate governance standards required by Nasdaq for U.S. companies. Accordingly, we follow Swiss corporate governance rules in lieu of certain of Nasdaq’s corporate governance requirements. The significant differences between our Swiss corporate governance rules and Nasdaq’s corporate governance requirements are set forth below:
•Exemption from the requirement that a majority of the board of directors be comprised of independent directors and that there be regularly scheduled meetings with only the independent directors present. Swiss law does not have such a requirement.
•Exemption from the requirements that the compensation committee and the nomination and corporate governance committee be comprised of independent directors. Swiss law does not have such requirements.
•Exemption from quorum requirements applicable to meetings of shareholders. Swiss law does not have such quorum requirements.
•Exemption from the requirement that independent directors meet at regularly scheduled executive sessions. Swiss law does not have such a requirement.
•Exemption from the requirement to disclose within four business days of any determination to grant a waiver of the Code of Ethics to directors and executive officers. Although we will require approval by our board of directors for any such waiver, we may choose not to disclose the waiver in the manner set forth in the Nasdaq listing standards.
•Exemption from the requirement to obtain shareholder approval for certain issuances of securities, including shareholder approval of share option plans. Our articles of association provide that our board of directors is authorized, in certain instances, to issue a certain number of ordinary shares without re-approval by our shareholders.
Furthermore, Nasdaq Rule 5615(a)(3) provides that a foreign private issuer may rely on home country corporate governance practices in lieu of certain of the rules in the Nasdaq Rule 5600 Series and Rule 5250(d), provided that it nevertheless complies with Nasdaq’s Notification of Noncompliance requirement (Rule 5625) and the Voting Rights requirement (Rule 5640) and that it has an audit committee that satisfies Rule 5605(c)(3), consisting of committee members that meet the independence requirements of Rule 5605(c)(2)(A)(ii). We intend to use these exemptions for as long as we continue to qualify as a foreign private issuer.
139
Item 16H. Mine Safety Disclosure
Not applicable.
Item 16I. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable.
Item 16J. Insider Trading Policies
Not applicable.
Item 16K. Cybersecurity
At SOPHiA GENETICS, cybersecurity risk management is an integral part of our enterprise risk management program. Our cybersecurity risk management program is derived from the ISO27001 family of standards and underpins our Information Security Management System (“ISMS”) framework. This determines how we design our polices, guidelines, controls, and best practices to manage the organization’s information security risks while permitting us to leverage NIST, STRIDE, and SANS frameworks to inform our handling of cybersecurity threats and incidents. Additionally, we are certified in accordance to and maintain our cybersecurity framework consistent with ISO 27001 standards.
The scope of applicability includes our globally accessible cloud-native solution offering along with all physical associated geographical locations: Switzerland, the US, and France. This is designed to achieve comprehensive global risk coverage across operations, application development, wet-lab analysis, and R&D. Many of these capabilities are delivered in-house, however third-party services are engaged in a modest capacity to support specific activities, such as security operations center capabilities and annual penetration testing. Where engaged, the organization has vendor management procedures in place that require cross-functional participation from the areas of Quality, Procurement, Regulatory, Legal, and Cybersecurity. These procedures are also interlinked directly with cybersecurity’s ISMS.
Our framework includes tools and processes to identify and assess the severity of cybersecurity threats and incidents including, but not limited to, data loss, theft, and system penetration. Additionally our framework allows us to identify if the threat or incident is associated with a third-party provider, dynamically implementing cybersecurity countermeasures and mitigation strategies and informing management and our board of directors of material cybersecurity threats and incidents
The cybersecurity team is further responsible for assessing our cybersecurity risk management program and communicating these risks to senior leadership and the Board. Although the assessment is solely performed within the cybersecurity team, external engagements influence such determination e.g.:
–Annual penetration testing and Tabletop exercises
–Quarterly phishing exercises
–Monthly SOC read-out calls
–External threat feeds
–Cyber insurance evaluation
–External Audit activities (ISO27001, ISO15485)
–Board and Executive team feedback
The cybersecurity team is also responsible for providing all employee training, awareness, and education. It performs this by carrying out:
–Quarterly phishing exercises
–Digital awareness campaigns through intranet posts, email updates, digital presentations
–Reach-out engagements to cross-functional teams aligned to their delivery schedules.
140
Our board of directors has overall oversight responsibility for our risk management and delegates cybersecurity risk management oversight to the audit committee of the board of directors. The audit committee is responsible for ensuring that management has processes in place designed to identify and evaluate cybersecurity risks to which the company is exposed and implement processes and programs to manage cybersecurity risks and mitigate cybersecurity incidents. The audit committee also reports material cybersecurity risks to our full board of directors. Management is responsible for identifying, considering and assessing material cybersecurity risks on an ongoing basis, establishing processes to ensure that such potential cybersecurity risk exposures are monitored, putting in place appropriate mitigation measures and maintaining cybersecurity programs. Our cybersecurity programs are under the direction of our Chief Legal and Compliance Officer (“CLCO”) who receives reports from our cybersecurity team and monitors the prevention, detection, mitigation, and remediation of cybersecurity incidents. Our dedicated personnel hold a multitude of certifications, including but not limited too Certified information systems security professional (“CISSP”), Certified Information Systems Auditor (“CISA”), Certified Information Security Manager (“CISM”), Certified ethical hacker (“CEH”), ITIL Foundations certification, and Masters of Science in Information Assurance, and experienced information systems security professionals and information security managers with many years of experience. Management, including the CLCO and our cybersecurity team, update the audit committee on the company’s cybersecurity programs, material cybersecurity risks and mitigation strategies and provide cybersecurity reports annually that cover, among other topics, such as, key performance metrics, developments in cybersecurity and updates to the company’s cybersecurity programs and mitigation strategies.
In 2023, we did not identify any cybersecurity incidents that have materially affected or are reasonably likely to materially affect our business strategy, results of operations, or financial condition. However, despite our efforts, we cannot eliminate all risks from cybersecurity threats, or provide assurances that we have not experienced an undetected cybersecurity incident. For more information about these risks, please see “Risk Factors – Cybersecurity or data privacy breaches, other unauthorized or improper access, or (distributed) denial service lack of access (e.g., ransomware, persistent DoS/DDoS) could result in additional costs, service outages, loss of revenue, significant liabilities, harm to our brand and decreased/denied use of our SOPHiA DDM Platform and related solutions, applications, products, or services.” in this annual report on Form 20-F.
141
PART III
Item 17. Financial Statements
See “Item 18—Financial Statements.”
Item 18. Financial Statements
The financial statements are filed as part of this Annual Report, beginning on page F-1.
Item 19. Exhibits
The following documents are filed as part of Annual Report on Form 20-F:
| Incorporation by Reference | ||||||||||||||||||||||||||||||||
| Exhibit No. | Description | Form | File No. | Exhibit No. | Filing Date | |||||||||||||||||||||||||||
| 1.1 | 6-K | 001-40627 | 99.1 | June 27, 2023 | ||||||||||||||||||||||||||||
| 2.1 | ||||||||||||||||||||||||||||||||
| 4.1# | F-1 | 333-257646 | 10.1 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.2# | F-1 | 333-257646 | 10.2 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.3# | F-1 | 333-257646 | 10.3 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.4# | F-1 | 333-257646 | 10.4 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.5# | F-1 | 333-257646 | 10.5 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.6# | F-1 | 333-257646 | 10.6 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.7# | F-1 | 333-257646 | 10.7 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.8# | F-1 | 333-257646 | 10.8 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.9# | F-1 | 333-257646 | 10.9 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.10#† | 20-F | 001-40627 | 4.10 | March 15, 2022 | ||||||||||||||||||||||||||||
| 4.11#† | 20-F | 001-40627 | 4.11 | March 15, 2022 | ||||||||||||||||||||||||||||
| 4.12§ | F-1 | 333-257646 | 10.10 | July 2, 2021 | ||||||||||||||||||||||||||||
| 4.13§ | F-1 | 333-257646 | 10.13 | July 2, 2021 | ||||||||||||||||||||||||||||
8.1 | ||||||||||||||||||||||||||||||||
142
| 12.1 | ||||||||||||||||||||||||||||||||
| 12.2 | ||||||||||||||||||||||||||||||||
| 13.1 | ||||||||||||||||||||||||||||||||
| 13.2 | ||||||||||||||||||||||||||||||||
| 15.1 | ||||||||||||||||||||||||||||||||
| 97.1 | ||||||||||||||||||||||||||||||||
| 101.INS | Inline XBRL Instance Document | |||||||||||||||||||||||||||||||
| 101.SCH | Inline XBRL Taxonomy Extension Schema Document | |||||||||||||||||||||||||||||||
| 101.CAL | Inline XBRL Taxonomy Extension Calculation Linkbase Document | |||||||||||||||||||||||||||||||
| 101.DEF | Inline XBRL Taxonomy Extension Definition Linkbase Document | |||||||||||||||||||||||||||||||
| 101.LAB | Inline XBRL Taxonomy Extension Label Linkbase Document | |||||||||||||||||||||||||||||||
| 101.PRE | Inline XBRL Taxonomy Extension Presentation Linkbase Document | |||||||||||||||||||||||||||||||
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |||||||||||||||||||||||||||||||
# Portions of this exhibit have been omitted because they are both (i) not material and (ii) customarily and actually treated by the registrant as private or confidential.
† Certain schedules to this exhibit have been omitted pursuant to Item 601(b)(2) of Regulation S-K. A copy of any omitted schedule will be furnished supplementally to the SEC upon request; provided, however, that the parties may request confidential treatment pursuant to Rule 24b-2 of the Exchange Act for any document so furnished.
§ Indicates a management contract or compensatory plan.
143
Signatures
The registrant hereby certifies that it meets all of the requirements for filing on Form 20-F and that it has duly caused and authorized the undersigned to sign this Annual Report on its behalf.
| SOPHiA GENETICS SA | ||||||||
| By: | /s/ Jurgi Camblong | |||||||
| Name: | Jurgi Camblong | |||||||
Title: | Chief Executive Officer | |||||||
| Date: | March 5, 2024 | |||||||
144
INDEX TO FINANCIAL STATEMENTS
| Contents | Page | ||||
Report of Independent Registered Public Accounting Firm (PCAOB ID: | F-2 | ||||
F-3 | |||||
F-4 | |||||
F-5 | |||||
F-6 | |||||
F-7 | |||||
F-8 | |||||
F-1
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Shareholders of SOPHiA GENETICS SA
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of SOPHiA GENETICS SA and its subsidiaries (the “Company”) as of December 31, 2023 and 2022, and the related consolidated statements of loss, comprehensive loss, changes in equity and cash flows for each of the three years in the period ended December 31, 2023, including the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2023 in conformity with IFRS Accounting Standards as issued by the International Accounting Standards Board.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits of these consolidated financial statements in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ PricewaterhouseCoopers SA
March 5, 2024
We have served as the Company’s auditor since 2020.
F-2
SOPHiA GENETICS SA, Rolle
Consolidated Statements of Loss
(Amounts in USD thousands, except per share data)
| Year ended December 31, | ||||||||||||||||||||||||||
| Notes | 2023 | 2022 | 2021 | |||||||||||||||||||||||
| Revenue | 4 | $ | $ | $ | ||||||||||||||||||||||
| Cost of revenue | 5 | ( | ( | ( | ||||||||||||||||||||||
| Gross profit | ||||||||||||||||||||||||||
| Research and development costs | 6 | ( | ( | ( | ||||||||||||||||||||||
| Selling and marketing costs | 6 | ( | ( | ( | ||||||||||||||||||||||
| General and administrative costs | 6 | ( | ( | ( | ||||||||||||||||||||||
| Other operating income, net | 7 | |||||||||||||||||||||||||
| Operating loss | ( | ( | ( | |||||||||||||||||||||||
| Interest income (expense), net | 8 | ( | ||||||||||||||||||||||||
| Foreign exchange and other losses | 8 | ( | ( | ( | ||||||||||||||||||||||
| Loss before income taxes | ( | ( | ( | |||||||||||||||||||||||
| Income tax (expense) benefit | 9 | ( | ( | |||||||||||||||||||||||
| Loss for the year | ( | ( | ( | |||||||||||||||||||||||
| Attributable to the owners of the parent | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||
| Basic and diluted loss per share | 10 | $ | ( | $ | ( | $ | ( | |||||||||||||||||||
The Notes form an integral part of these consolidated financial statements
F-3
SOPHiA GENETICS SA, Rolle
Consolidated Statements of Comprehensive Loss
(Amounts in USD thousands)
| Year ended December 31, | ||||||||||||||||||||||||||
| Notes | 2023 | 2022 | 2021 | |||||||||||||||||||||||
| Loss for the year | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||
| Other comprehensive (loss) income: | ||||||||||||||||||||||||||
| Items that may be reclassified to statement of loss (net of tax) | ||||||||||||||||||||||||||
| Currency translation differences | ( | ( | ||||||||||||||||||||||||
| Total items that may be reclassified to statement of loss | $ | $ | ( | $ | ( | |||||||||||||||||||||
| Items that will not be reclassified to statement of loss (net of tax) | ||||||||||||||||||||||||||
| Remeasurement of defined benefit plans | 22 | ( | ||||||||||||||||||||||||
| Total items that will not be reclassified to statement of loss | $ | ( | $ | $ | ||||||||||||||||||||||
| Other comprehensive (loss) income for the year | $ | $ | ( | $ | ( | |||||||||||||||||||||
| Total comprehensive loss for the year | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||
| Attributable to owners of the parent | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||
The Notes form an integral part of these consolidated financial statements
F-4
SOPHiA GENETICS SA, Rolle
Consolidated Balance Sheets
(Amounts in USD thousands)
| As of December 31, | ||||||||||||||||||||
| Notes | 2023 | 2022 | ||||||||||||||||||
| Assets | ||||||||||||||||||||
| Current assets | ||||||||||||||||||||
| Cash and cash equivalents | 11 | $ | $ | |||||||||||||||||
| Term deposits | 12 | |||||||||||||||||||
| Accounts receivable | 13 | |||||||||||||||||||
| Inventory | 14 | |||||||||||||||||||
| Prepaids and other current assets | 15 | |||||||||||||||||||
| Total current assets | ||||||||||||||||||||
| Non-current assets | ||||||||||||||||||||
| Property and equipment | 16 | |||||||||||||||||||
| Intangible assets | 17 | |||||||||||||||||||
| Right-of-use assets | 18 | |||||||||||||||||||
| Deferred tax assets | 9 | |||||||||||||||||||
| Other non-current assets | 19 | |||||||||||||||||||
| Total non-current assets | ||||||||||||||||||||
| Total assets | $ | $ | ||||||||||||||||||
| Liabilities and equity | ||||||||||||||||||||
| Current liabilities | ||||||||||||||||||||
| Accounts payable | 20 | $ | $ | |||||||||||||||||
| Accrued expenses | 21 | |||||||||||||||||||
| Deferred contract revenue | 4 | |||||||||||||||||||
| Current portion of lease liabilities | 18 | |||||||||||||||||||
| Total current liabilities | ||||||||||||||||||||
| Non-current liabilities | ||||||||||||||||||||
| Lease liabilities, net of current portion | 18 | |||||||||||||||||||
| Defined benefit pension liabilities | 22 | |||||||||||||||||||
| Other non-current liabilities | ||||||||||||||||||||
| Total non-current liabilities | ||||||||||||||||||||
| Total liabilities | ||||||||||||||||||||
| Equity | ||||||||||||||||||||
| Share capital | ||||||||||||||||||||
| Share premium | ||||||||||||||||||||
| Treasury shares | ( | ( | ||||||||||||||||||
| Other reserves | ||||||||||||||||||||
| Accumulated deficit | ( | ( | ||||||||||||||||||
| Total equity | ||||||||||||||||||||
| Total liabilities and equity | $ | $ | ||||||||||||||||||
The Notes form an integral part of these consolidated financial statements
F-5
SOPHiA GENETICS SA, Rolle
Consolidated Statements of Changes in Equity
(Amounts in USD thousands, except share data)
| Notes | Shares | Share capital | Treasury Shares | Treasury Share capital | Share premium | Other reserves | Accumulated deficit | Total | ||||||||||||||||||||||||||||||||||||||||||||||||
| January 1, 2021 | $ | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||||||||||||||
| Loss for the period | — | — | — | — | — | — | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive loss | — | — | — | — | — | ( | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive loss | — | — | — | — | — | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | 23 | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||
| Transactions with owners | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share options exercised | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sale of ordinary shares in initial public offering, net of transaction costs | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sale of ordinary shares in private placement, net of transaction costs | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sale of ordinary shares in greenshoe offering, net of transaction costs | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2021 | $ | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||||||||||||||
| Loss for the period | — | — | — | — | — | — | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive loss | — | — | — | — | — | ( | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive loss | — | — | — | — | — | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | 23 | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||
| Transactions with owners | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share options exercised and vesting of Restricted Stock Units | 23 | — | — | ( | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares to be held as treasury shares | ( | ( | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2022 | $ | ( | $ | ( | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||||||||||||
| Loss for the period | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive loss | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive loss | — | — | — | — | — | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | 23 | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transactions with owners | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share options exercised and vesting of Restricted Stock Units | 23 | — | — | ( | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares to be held as treasury shares | ( | ( | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2023 | $ | ( | $ | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||
The Notes form an integral part of these consolidated financial statements
F-6
SOPHiA GENETICS SA, Rolle
Consolidated Statements of Cash Flows
(Amounts in USD thousands)
| Year ended December 31, | ||||||||||||||||||||||||||
| Notes | 2023 | 2022 | 2021 | |||||||||||||||||||||||
| Operating activities | ||||||||||||||||||||||||||
| Loss before income tax | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||
| Adjustments for non-monetary items | ||||||||||||||||||||||||||
| Depreciation | 16,18 | |||||||||||||||||||||||||
| Amortization | 17 | |||||||||||||||||||||||||
| Finance expense (income), net | ( | |||||||||||||||||||||||||
| Gain on TriplePoint success fee | ( | |||||||||||||||||||||||||
| Expected credit loss allowance | 13 | ( | ( | |||||||||||||||||||||||
| Share-based compensation | 23 | |||||||||||||||||||||||||
| Intangible assets write-off | 17 | |||||||||||||||||||||||||
| Movements in provisions, pensions, and government grants | 14,22 | ( | ||||||||||||||||||||||||
| Research tax credit | 6 | ( | ( | ( | ||||||||||||||||||||||
| Loss on disposal of property and equipment | 16 | |||||||||||||||||||||||||
| Gain on disposal of lease liability | ( | |||||||||||||||||||||||||
| Working capital changes | ||||||||||||||||||||||||||
| (Increase) decrease in accounts receivable | ( | |||||||||||||||||||||||||
| (Increase) decrease in prepaids and other assets | ( | ( | ||||||||||||||||||||||||
| (Increase) decrease in inventory | ( | ( | ( | |||||||||||||||||||||||
| Increase (decrease) in accounts payables, accrued expenses, deferred contract revenue, and other liabilities | ( | |||||||||||||||||||||||||
| Cash used in operating activities | ||||||||||||||||||||||||||
| Income tax received (paid) | ( | ( | ||||||||||||||||||||||||
| Interest paid | ( | ( | ( | |||||||||||||||||||||||
| Interest received | ||||||||||||||||||||||||||
| Net cash flows used in operating activities | ( | ( | ( | |||||||||||||||||||||||
| Investing activities | ||||||||||||||||||||||||||
| Purchase of property and equipment | 16 | ( | ( | ( | ||||||||||||||||||||||
| Acquisition of intangible assets | 17 | ( | ( | ( | ||||||||||||||||||||||
| Capitalized development costs | 17 | ( | ( | ( | ||||||||||||||||||||||
| Proceeds upon maturity of term deposits and short-term investments | 12 | |||||||||||||||||||||||||
| Purchase of term deposits and short-term investments | 12 | ( | ( | |||||||||||||||||||||||
| Net cash flow provided from (used in) investing activities | ( | |||||||||||||||||||||||||
| Financing activities | ||||||||||||||||||||||||||
| Proceeds from exercise of share options | 23 | |||||||||||||||||||||||||
| Proceeds from initial public offering, net of transaction costs | ||||||||||||||||||||||||||
| Proceeds from greenshoe, net of transaction costs | ||||||||||||||||||||||||||
| Proceeds from private placement, net of transaction costs | ||||||||||||||||||||||||||
| Payment of TriplePoint success fee | ( | |||||||||||||||||||||||||
| Repayments of borrowings | 24 | ( | ||||||||||||||||||||||||
| Payments of principal portion of lease liabilities | 18 | ( | ( | ( | ||||||||||||||||||||||
| Net cash flow (used in) provided from financing activities | ( | ( | ||||||||||||||||||||||||
| Increase (decrease) in cash and cash equivalents | ( | ( | ||||||||||||||||||||||||
| Effect of exchange differences on cash balances | ( | ( | ||||||||||||||||||||||||
| Cash and cash equivalents at beginning of the year | ||||||||||||||||||||||||||
| Cash and cash equivalents at end of the year | $ | $ | $ | |||||||||||||||||||||||
The Notes form an integral part of these consolidated financial statements
F-7
SOPHiA GENETICS SA, Rolle
Notes to the Consolidated Financial Statements
1. Company information and operations
General information
SOPHiA GENETICS SA and its consolidated subsidiaries (NASDAQ: SOPH) (“the Company”) is a cloud-native software company in the healthcare space, incorporated on March 18, 2011, and headquartered in Rolle, Switzerland. The Company is dedicated to establishing the practice of data-driven medicine as the standard of care in healthcare and for life sciences research. The Company has built a cloud-native software platform capable of analyzing data and generating insights from complex multimodal datasets and different diagnostic modalities. This platform, commercialized as “SOPHiA DDMTM,” standardizes, computes, and analyzes digital health data and is used in decentralized locations to break down data silos.
On June 26, 2023, during the Company’s Annual General Meeting, the move of the statutory seat from Saint-Sulpice, Canton Vaud, Switzerland to Rolle, Canton Vaud, Switzerland was approved.
As of December 31, 2023, the Company had the following wholly-owned subsidiaries:
| Name | Country of domicile | ||||
Interactive Biosoftware S.A.S., a wholly owned subsidiary located in France and acquired in 2018, was merged into SOPHiA GENETICS S.A.S. in 2020.
On April 9, 2021, SOPHiA GENETICS PTY LTD, a wholly owned subsidiary located in Australia, was incorporated.
On May 27, 2021, SOPHiA GENETICS S.R.L., a wholly owned subsidiary located in Italy, was incorporated.
On December 12, 2022, the Company changed the name of SOPHiA GENETICS Intermediação de Negócios EIRELI to SOPHiA GENETICS Intermediação de Negócios LTDA.
The Company’s Board of Directors approved the issue of the consolidated financial statements on March 5, 2024.
Share split
On June 30, 2021, the Company effected a one-to-twenty share split of its outstanding shares. Accordingly, all share and per share amounts for all periods presented in these consolidated financial statements and notes thereto have been adjusted retroactively, where applicable, to reflect this share split.
Initial public offering
In July 2021, the Company completed its initial public offering (“IPO”) in the United States on the Nasdaq Global Market (“Nasdaq”) under the trading ticker symbol “SOPH”. Trading on the Nasdaq commenced at market open on July 23, 2021. The Company completed the IPO of 13,000,000 ordinary shares, at an IPO price of $18.00 per share, par value $0.05 (CHF 0.05 ). The aggregate net proceeds received from the IPO, net of underwriting discounts and commissions and offering expenses, was $211.7 million. Immediately prior to the completion of the IPO, all then outstanding shares of preferred shares were converted into 24,561,200 shares of ordinary shares on a one -to-one basis.
F-8
Concurrent with the IPO, the Company closed a private placement, in which it sold 1,111,111 ordinary shares to an affiliate of GE Healthcare at a price of $18.00 per share, par value $0.05 (CHF 0.05 ). The aggregate net proceeds received from the private placement, net of offering expenses, was $19.6 million.
On August 25, 2021, the underwriters of the IPO elected to exercise in part their option to purchase an additional 519,493 ordinary shares (“greenshoe”) at the IPO price of $18.00 per share, par value $0.05 (CHF 0.05 ). The aggregate net proceeds received from the greenshoe, net of underwriting discounts and commissions and offering expenses, was $8.5 million.
Issued share capital
As of December 31, 2023, the Company had issued 76,898,164 shares of which 65,230,559 are outstanding and 11,667,605 are held by the Company as treasury shares. As of December 31, 2022, the Company had issued outstanding shares of 64,231,220 . All shares were considered paid as of December 31, 2023.
Treasury shares
During the first quarter of 2022, the Company issued 2,540,560 registered shares to SOPHiA GENETICS LTD pursuant to a share delivery and repurchase agreement, which were immediately exercised, and repurchased the shares to hold as treasury shares for the purposes of administering the Company's equity incentive programs. During the second quarter of 2023, the Company issued 10,500,000 registered shares to SOPHiA GENETICS LTD pursuant to a share delivery and repurchase agreement, which were immediately exercised, and repurchased the shares to hold as treasury shares. The Company held 11,667,605 and 2,166,944 treasury shares as of December 31, 2023 and 2022, respectively.
Treasury shares are recognized at acquisition cost and recorded as treasury shares at the time of the transaction. Upon exercise of share options or vesting of restricted stock units, the treasury shares are subsequently transferred. Any consideration received is included in shareholders’ equity.
2. Material accounting policies
Basis of preparation
Compliance with International Financial Reporting Standards
The consolidated financial statements of the Company have been prepared in accordance with IFRS Accounting Standards and interpretations issued by the IFRS Interpretations Committee (“IFRS IC”) applicable to companies reporting under IFRS. The consolidated financial statements comply with IFRS Accounting Standards as issued by the International Accounting Standards Board (“IASB”).
Basis of consolidation
A subsidiary is an entity over which the Company has control. The Company controls an entity when it has the power to direct its activities and has rights to its variable returns. Subsidiaries are fully consolidated from the date on which control is transferred to the Company and deconsolidated from the date that control ceases.
During the consolidation process intercompany transactions, balances, and unrealized gains on transactions between companies are eliminated. Unrealized losses are also eliminated unless there is evidence of an impairment of the transferred asset. In order to ensure consistency with the accounting policies of the Company, the accounting policies of subsidiaries have been changed where necessary.
Foreign currency translation
Items included in the consolidated financial statements of each of the Company’s entities are measured using the currency of the primary economic environment in which the entity operates (“functional currency”). In individual entities, transactions in foreign currencies are translated as of transaction date. Monetary assets and liabilities in foreign currencies are translated at month end rates. The Company’s reporting currency of the Company’s consolidated financial statements is the U.S. dollar (“USD”). Assets and liabilities denominated in foreign currencies are translated at the month-end spot exchange rates, income statement accounts are
F-9
translated at average rates of exchange for the period presented, and equity is translated at historical exchange rates.
On consolidation, assets and liabilities of foreign operations reported in their local functional currencies are translated into USD. Differences arising from the retranslation of opening net assets of foreign operations, together with differences arising from the translation of the net results for the year of foreign operations, are recognized in other comprehensive income under currency retranslations. Gains or losses resulting from foreign currency transactions are included in net income.
The Company selected the U.S. dollar as its presentation currency for purposes of its consolidated financial statements instead of the Company’s functional currency, the Swiss franc, because of the global nature of its business, its expectation that an increasing portion of revenues and expenses will be denominated in USD, and its plans to continue to access U.S. capital markets.
Use of estimates
The preparation of consolidated financial statements in conformity with IFRS Accounting Standards requires the use of accounting estimates. It also requires management to exercise judgment in applying the Company’s accounting policies. The Company’s significant estimates and judgments included in the preparation of the consolidated financial statements are related to revenue recognition, capitalized internal software development costs, share-based compensation, expected credit loss, goodwill, defined benefit pension liabilities, uncertain tax positions, and derivatives.
Disclosed in the corresponding sections within the footnotes are the areas which require a high degree of judgment, significant assumptions, and/or estimates.
Going concern basis
Historical cost convention
The consolidated financial statements have been prepared on a historical cost basis except for certain assets and liabilities, which are carried at fair value.
Accounting policies
The material accounting policies adopted in the preparation of the consolidated financial statements have been consistently applied, unless otherwise stated.
Provisions and contingencies
Provisions comprise liabilities of uncertain timing or amount. The provisions and liabilities are recognized when the Company has a present legal or constructive obligation as a result of past events, it is probable that an outflow of resources will be required to settle the obligation, and the amount can be reliably estimated. Provisions are not recognized for future operating losses. Provisions are measured at the present value of management’s best estimate of the expenditure required to settle the present obligation at the end of the reporting period, unless the impact of discounting is immaterial. The discount rate used to determine the present value is a pre-tax rate that reflects current market assessments of the time value of money and the risks specific to the liability. The increase in the provision due to the passage of time is recognized as interest expense.
Contingent liabilities are possible obligations that arise from past events and whose existence will be confirmed only by the occurrence or non-occurrence of one or more uncertain future events not fully within the control of the Company.
The likelihood of occurrence of provisions and contingent liabilities requires use of judgment. Judgment is also required to determine if an outflow of economic resources is probable, or possible but not probable. Where it is probable, a liability is recognized, and further judgment is used to determine the level of the provision. Where it is possible but not probable, further judgment is used to determine if the likelihood is remote, in which case no
F-10
disclosures are provided; if the likelihood is not remote then judgment is used to determine the contingent liability disclosed.
Financial assets classification
Upon recognition, financial assets are classified on the basis of how the financial assets are measured: at amortized cost or fair value through income.
The classification of financial assets at initial recognition depends on the financial asset’s contractual cash flow characteristics and the Company’s business model for managing them. Except for accounts receivable that do not contain a significant financing component, the Company initially measures a financial asset at its fair value plus, in the case of a financial asset not at fair value through income, transaction costs. Accounts receivable that do not contain a significant financing component are measured at the transaction price.
The Company’s business model for managing financial assets is defined by whether cash flows will result from collecting contractual cash flows, selling the financial assets, or both. Financial assets held in order to collect contractual cash flows are measured at amortized cost. Financial assets held both to collect contractual cash flows and for sale are measured at fair value through other comprehensive income/loss.
Purchases or sales of financial assets that require delivery of assets within a time frame established by regulation or convention in the marketplace (regular way trades) are recognized on the trade date, i.e., the date that the Company commits to purchase or sell the asset.
Financial assets measured at amortized cost
Financial assets initially measured at amortized cost are subsequently measured using the effective interest rate (“EIR”) method and are subject to impairment. Gains and losses are recognized in income when the asset is derecognized, modified, or impaired. The Company’s financial assets at amortized cost include cash, term deposits and accounts receivable.
Financial assets—derecognition
A financial asset (or, where applicable, a part of a financial asset or part of a group of similar financial assets) is primarily derecognized (i.e., removed from the Company’s consolidated balance sheet) when:
•the rights to receive cash flows from the asset have expired or;
•the Company has transferred its rights to receive cash flows from the asset or has assumed an obligation to pay the received cash flows in full without material delay to a third party under a ‘pass-through’ arrangement; and either;
•the Company has transferred substantially all the risks and rewards of the asset, or;
•the Company has neither transferred nor retained substantially all the risks and rewards of the asset but has transferred control of the asset.
When the Company has transferred its rights to receive cash flows from an asset or has entered into a pass-through arrangement, it evaluates if, and to what extent, it has retained the risks and rewards of ownership.
When the Company has neither transferred nor retained substantially all of the risks and rewards of the asset, nor transferred control of the asset, the Company continues to recognize the transferred asset to the extent of its continuing involvement. In that case, the Company also recognizes an associated liability. The transferred asset and the associated liability are measured on a basis that reflects the rights and obligations that the Company has retained.
Continuing involvement that takes the form of a guarantee over the transferred asset is measured at the lower of the original carrying amount of the asset and the maximum amount of consideration that the Company could be required to repay.
F-11
Financial assets—impairment
For cash, cash equivalents, and term deposits, the Company invests in assets where it has never incurred and does not expect to incur credit losses.
For accounts receivable the Company recognizes a loss allowance based on lifetime estimated credit losses (“ECL”) at each reporting date. When estimating the ECL the Company takes into consideration: readily available relevant and supportable information (this includes quantitative and qualitative data), the Company’s historical experience and forward-looking information specific to the receivables and the economic environment.
See Note 13 – “Accounts receivable” for further information about the Company’s accounting for trade receivables.
Financial liabilities classification
Financial liabilities are classified upon initial recognition as financial liabilities measured at fair value through income or at amortized cost. The Company’s financial liabilities include accounts payable and debt (including borrowings and lease liabilities), which are measured at amortized cost, and derivatives, which are measured at fair value through income.
Interest-bearing borrowings are initially recognized at fair value less directly attributable costs and subsequently measured at amortized cost using the EIR method. Gains and losses are recognized in income when the liabilities are derecognized as well as through the EIR amortization process.
Amortized cost is calculated by taking into account any discount or premium on acquisition and fees or costs that are an integral part of the EIR. The EIR amortization is included as finance costs in the statement of income/loss.
Financial liabilities—derecognition
A financial liability is derecognized when the obligation under the liability is discharged or canceled or expires. When an existing financial liability is replaced by another from the same lender on substantially different terms, or the terms of an existing liability are substantially modified, such an exchange or modification is treated as the derecognition of the original liability and the recognition of a new liability. The difference in the respective carrying amounts is recognized in the statements of loss.
New standards, amendments to standards and interpretations
New standards, amendments to standards, and interpretations issued recently effective
There are no new IFRS Accounting Standards, amendments or interpretations that are mandatory as of January 1, 2023 that are relevant to the Company.
New standards, amendments to standards, and interpretations issued not yet effective
In January 2020, IASB issued amendments to paragraphs 69 to 76 of International Accounting Standard (“IAS”) 1, Presentation of Financial Statements (“IAS 1”), to specify the requirements for classifying liabilities as current or non-current, effective for annual reporting periods beginning on or after January 1, 2024. The Company determined the amendment has no impact.
3. Segment reporting
The Company operates in a single operating segment. The Company’s financial information is reviewed, and its performance assessed as a single segment by the senior management team led by the Chief Executive Officer (“CEO”), the Company’s Chief Operating Decision Maker (“CODM”).
F-12
For the years ended December 31, 2023 and 2022, respectively, the Company had a physical presence in three countries outside of its headquarters in Switzerland: France, the United States, and Brazil. An analysis of the location of non-current assets other than financial instruments and deferred tax assets by country is as follows (in USD thousands):
| As of December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| Switzerland | $ | $ | ||||||||||||
| France | ||||||||||||||
| United States | ||||||||||||||
| Brazil | ||||||||||||||
| Total non-current assets other than financial instruments and deferred tax assets | $ | $ | ||||||||||||
4. Revenue
Material accounting estimates and judgments
The Company recognizes revenue when control of promised goods or services is transferred to customers in an amount that reflects the consideration that is expected to be received for those goods or services. Significant judgment is required to determine the stand-alone selling price (“SSP”) for each performance obligation in the SOPHiA DDM Platform, the amount allocated to each performance obligation and whether it depicts the amount that the Company expects to receive in exchange for the related product and/or service.
The Company enters into arrangements with multiple performance obligations where it could be difficult to determine the performance obligations under a sales agreement; in such cases, how and when revenue should be recognized is subject to certain estimates or assumptions. Should these judgments and estimates not be correct, revenue recognized for any reporting period could be adversely affected.
Accounting policies
Revenue represents amounts received and receivable from third parties for goods supplied and services rendered to customers. Revenues are reported net of rebates and discounts and net of sales and value added taxes in an amount that reflects the consideration that is expected to be received for goods or services. The majority of the sales revenue is recognized: (i) when customers generate analyses on their patient data through the SOPHiA DDM Platform, (ii) when consumables, namely DNA enrichment kits, are delivered to customers at which point control transfers, (iii) when services, namely set-up programs, are performed and (iv) over the duration of the software licensing arrangements for the Alamut software offerings.
Products and services are sold both directly to customers and through distributors, generally under agreements with payment terms of up to 180 days. Therefore, contracts do not contain a significant financing component.
For all contracts with customers the following steps are performed to determine the amount of revenue to be recognized and when it should be recognized: (1) identify the contract or contracts; (2) determine whether the promised goods or services are performance obligations, including whether they are distinct in the context of the contract; (3) measure the transaction price, including the constraint on variable consideration; (4) allocate the transaction price to the performance obligations based on estimated selling prices; and (5) recognize revenue when (or as) each performance obligation is satisfied.
SOPHiA DDM Platform
The majority of the SOPHiA DDM Platform revenue is derived from each use of the SOPHiA DDM Platform by customers to generate analyses on their patient data. Analysis revenue is recognized as analysis results are made available to the customer on the SOPHiA DDM Platform. The Company recognizes accrued contract revenue in accounts receivable for any analyses performed by customers that have not been invoiced at the reporting date and where the right to consideration is unconditional. Any payments received in advance of customers generating analyses are recorded as deferred contract revenue until the analyses are performed.
F-13
Customers use the SOPHiA DDM Platform to perform analyses under three different models: dry lab access; bundle access; and integrated solutions.
For dry lab contracts, customers use the testing instruments and consumables of their choice and the SOPHiA DDM Platform and algorithms for variant detection and identification. In these arrangements, the Company has identified one performance obligation, which is the delivery of the analysis result to the customer.
For bundle arrangements, customers purchase a DNA enrichment kit along with each analysis. Customers use the DNA enrichment kit in the process of performing their own sequencing of each sample. Customers then upload their patient data to the SOPHiA DDM Platform for analysis. In these arrangements, the Company has identified two performance obligations: the delivery of the DNA enrichment kits and the performance of the analyses. Revenue is recognized for the DNA enrichment kits when control of products has transferred to the customer, which is generally at the time of delivery, as this is when title and risk of loss have been transferred. Revenue for the performance of the analyses is recognized on delivery of the analysis results to the customer. Refer to Arrangements with multiple performance obligations below for how revenue is allocated between the performance obligations.
Deferred contract revenue balances relating to analyses not performed within 12 months from the date of the delivery date are recognized as revenue. This policy is not based on contractual conditions but on the Company’s experience of customer behavior and expiration of the kits associated with the analyses.
For integrated arrangements, customers have their samples processed and sequenced through selected SOPHiA DDM Platform partners within the clinical network and access their data through the SOPHiA DDM Platform. The Company has identified one performance obligation, which is delivery of the analysis results to the customer through the SOPHiA DDM Platform.
The Company also sells access to its Alamut software application (“Alamut”) through the SOPHiA DDM Platform. Some arrangements with customers allow customers to use Alamut as a hosted software service over the contract period without the customer taking possession of the software. Other customers take possession of the software, but the utility of that software is limited by access to the Company’s proprietary SOPHiA database, which is provided to the customer on a fixed term basis. Under both models, revenue is recognized on a straight-line basis over the duration of the agreement.
The Company also derives revenue from the SOPHiA DDM Platform by providing services to biopharma customers who engage the Company to (i) develop and perform customized genomic analyses and/or (ii) access the database for use in clinical trials and other research projects.
The Company does enter into biopharma contracts that contain multiple products or services or non-standard terms and conditions. The biopharma contracts are generally unique in nature and each contract is assessed upon execution. Contracts may contain multiple performance obligations or performance obligations that are recognized overtime, at a point-in-time, or a combination depending on the Company’s ability to satisfy the requirements to recognize revenue over time and reasonably estimate the amount of revenue to recognize. See “Arrangements with multiple performance obligations” below for further discussion on treatment of biopharma contracts.
Generally, the primary performance obligation in these arrangements is the delivery of analysis results in the form of a final report, resulting in revenue being recognized, in most cases, upon the issuance of the final report or successful recruitment of clinical trial participants.
Workflow materials and services
Revenue from workflow materials and services includes all revenue from the sale of materials and services that do not form part of a contract for the provision of platform services. These include the provision of set-up programs and training and the sale of kits and tests that are not linked to use of the platform. Set-up programs and training are typically combined with a customer’s first order prior to the customer beginning to use the SOPHiA DDM Platform.
Revenue from services is generally recognized when the services are performed. Revenue from materials is recognized when control of the goods is transferred to the customer, generally at the time of delivery. This category of revenue also includes the revenue from the sale of DNA sequencing automation equipment
F-14
accounted for under IFRS 16, Leases (“IFRS 16”), leasing and the fees charged for the maintenance of this equipment.
Arrangements with multiple performance obligations
The Company sells different combinations of analyses, consumables, and services to its customers under its various SOPHiA DDM Platform models.
The Company has determined that the stand-alone selling prices for services and DNA enrichment kits are directly observable. For set-up programs and training sold along with dry lab arrangements or bundle arrangements, the stand-alone selling price of these services is determined on a time and materials basis. For DNA enrichment kits sold as part of a bundle, the SSP is based on an expected cost-plus-margin approach of the kit portion of the bundle.
The Company has determined that the SSP for the analyses, in both a dry lab arrangement and bundle arrangement, is highly variable and therefore a representative SSP is not discernible from past transactions. As a result, the residual approach is used to determine the stand-alone selling price of the analyses in dry lab arrangements that include services and in bundle arrangements that include DNA enrichment kits and, in some cases, services.
The Company also has a small number of bundle contracts with a fixed term that also include providing the customer with DNA sequencing automation equipment, which the Company has determined is an IFRS 16 leasing component. In these arrangements the Company provides DNA sequencing automation equipment to the customer over the fixed term and at completion of the contract term the customer takes possession of the equipment. The Company has determined that it is a dealer lessor and provision of this equipment to the customer is classified as a finance lease. As a result, upon delivery of the leased equipment at the inception of the arrangement, a selling profit is recognized based on the fair value of the underlying equipment less the cost of the equipment. Over the term of the agreement, the minimum lease payment is deducted from the proceeds of the bundle sales in order to reduce the net investment in the corresponding lease receivable over the contract term and interest income is recognized as the discount on the lease receivable unwinds. The remaining proceeds from the contract are accounted for under IFRS 15, Revenue from Contracts with Customers (“IFRS 15”), using the policies described above.
The Company assess biopharma contracts upon execution of each contract given their unique nature. The Company establishes each performance obligation within the contract and determines the appropriate value to be ascribed to be each performance obligation. When relevant the Company utilizes previous established SSPs of its dry lab and bundle solutions or other service. When the performance obligation is specific to only the contract the Company utilizes all available information to reasonable estimate the correct value allocated to the performance obligation.
Contract Balances
Deferred contract costs
Deferred contract costs comprise deferred fulfillment costs related to biopharma, prepayments on contracts, and prepaid maintenance costs relating to DNA sequencing automation equipment.
Costs are incurred to fulfill obligations under certain contracts once obtained, but before transferring goods or services to the customer. Fulfillment costs are recognized as an asset, provided these costs are not addressed by other accounting standards, if the following criteria are met: (i) the costs relate directly to a contract or an anticipated contract that the Company can specifically identify, (ii) the costs generate or enhance resources of the Company that will be used in satisfying (or continuing to satisfy) performance obligations in the future and (iii) the costs are expected to be recovered.
The asset recognized from deferring the costs to fulfill a contract is recorded in the consolidated balance sheet as deferred contract costs within other current assets and amortized on a systematic basis consistent with the pattern of the transfer of the goods or services to which the asset relates, which depends on the nature of the performance obligation(s) in the contract. The amortization of these assets is recorded in cost of revenue.
F-15
The timing of revenue recognition and billings can result in accrued contract revenue, which are presented within accounts receivable in the consolidated balance sheet and deferred contract revenue which is presented on the face of the consolidated balance sheet.
Deferred contract revenue
Deferred contract revenue relates to prepayments received from customers before revenue is recognized and is primarily related to SOPHiA DDM Platform analyses invoiced in advance of the customers performing the analyses, deferred Alamut software revenue and progress payments received as part of biopharma contracts.
Deferred contract revenue brought forward as of January 1, 2023 and 2022 amounts to $3.4 million and $4.0 million, respectively. During the twelve months ended December 31, 2023 and 2022, the Company satisfied the performance obligations associated with that deferred contract revenue to the extent that revenue was recognized of $3.4 million and $4.0 million, respectively.
The majority of the platform revenue is derived from contracts with an original expected length of one year or less. However, there are certain biopharma and Alamut contracts in which performance obligations extend over multiple years. The Company has elected to apply the practical expedient not to disclose the value of remaining performance obligations associated with these types of contracts.
F-16
Disaggregated Revenue
When disaggregating revenue, the Company considered all of the economic factors that may affect its revenues. The Company assess its revenues by four geographic regions Europe, the Middle East, and Africa (“EMEA”); North America (“NORAM”); Latin America (“LATAM”); and Asia-Pacific (“APAC”). Additionally, the Company assess revenues generated in its domiciled country and any country with significant revenue. The following tables disaggregate the Company's revenue from contracts with customers by geographic market (in USD thousands):
| Year ended December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Switzerland | $ | $ | $ | |||||||||||||||||
| France | ||||||||||||||||||||
| Italy | ||||||||||||||||||||
| Spain | ||||||||||||||||||||
| Rest of EMEA | ||||||||||||||||||||
| EMEA | $ | $ | $ | |||||||||||||||||
| United States | $ | $ | $ | |||||||||||||||||
| Rest of NORAM | ||||||||||||||||||||
| NORAM | $ | $ | $ | |||||||||||||||||
| LATAM | $ | $ | $ | |||||||||||||||||
| APAC | $ | $ | $ | |||||||||||||||||
| Total revenue | $ | $ | $ | |||||||||||||||||
Revenue streams
The Company’s revenue from contracts with customers has been allocated to the revenue streams indicated in the table below (in USD thousands):
| Year ended December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| SOPHiA DDM Platform | $ | $ | $ | |||||||||||||||||
| Workflow equipment and services | ||||||||||||||||||||
| Total revenue | $ | $ | $ | |||||||||||||||||
Workflow equipment and services includes revenues from payments from leased equipment recognized under IFRS 16, Leases, of less than $0.1 million, $0.1 million, and $0.2 million for the years ended December 31, 2023, 2022, and 2021, respectively.
5. Cost of revenue
Accounting policies
Cost of revenue comprises costs directly incurred in earning revenue, including computer costs and data storage fees paid to hosting providers, manufacturing costs, materials and consumables, the cost of equipment leased out under finance leases, personnel-related expenses and amortization of capitalized development costs.
F-17
6. Operating expense
Accounting policies
Research and development
Research and development costs consist of personnel and related expenses for technology, application, and product development, depreciation and amortization, laboratory supplies, consulting services, computer costs and data storage fees paid to hosting providers related to research and development and allocated overhead costs. These costs are stated net of government grants for research and development and innovation received as tax credits and net of capitalized costs.
Government grants for research and development and innovation received as tax credits
The Company receives government grants in France for research and development and innovation by way of tax credits. Total government grants for research and development and innovation recognized in the statement of loss amounts to $1.1 million, $1.3 million, $1.6 million for the years ended December 31, 2023, 2022, and 2021, respectively.
Selling and marketing costs
Selling and marketing costs consist of personnel and related expenses for the employees of the sales and marketing organization, costs of communications materials that are produced to generate greater awareness and utilization of the platform among customers, costs of third-party market research, costs related to transportation and distribution of our products, and allocated overhead costs. The Company also records increases to, reversals of, and write-offs of the allowance for expected credit losses to selling and marketing costs.
The Company pays sales commission to its employees for obtaining contracts. These costs are expensed as part of employee compensation in selling and marketing costs. They are not capitalized as contract costs as the commissions either represent bonuses payable for revenue earned in the period or have a service condition attached.
General and administrative costs
General and administrative costs consist of personnel and related expenses for our executive, accounting and finance, legal, quality, support and human resources functions, depreciation and amortization, professional services fees incurred by these functions, general corporate costs and allocated overhead costs, which include occupancy costs and information technology costs.
F-18
Operating expense by nature
The table presents operating expenses by nature (in USD thousands):
| For the year ended December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Changes in inventories of finished goods and work in progress | $ | $ | $ | |||||||||||||||||
| Raw materials and consumables used | ( | ( | ( | |||||||||||||||||
| Employee benefit expenses | ( | ( | ( | |||||||||||||||||
| Social charges | ( | ( | ( | |||||||||||||||||
| Research tax credit | ||||||||||||||||||||
| Share-based compensation | ( | ( | ( | |||||||||||||||||
| Depreciation | ( | ( | ( | |||||||||||||||||
| Amortization | ( | ( | ( | |||||||||||||||||
| Professional fees | ( | ( | ( | |||||||||||||||||
| Laboratory and office expenses | ( | ( | ( | |||||||||||||||||
| Travel | ( | ( | ( | |||||||||||||||||
| Marketing | ( | ( | ( | |||||||||||||||||
| Licenses | ( | ( | ( | |||||||||||||||||
| Less: capitalized software development costs ("Note 17 - Intangible assets”) | ||||||||||||||||||||
| Other expense | ( | ( | ( | |||||||||||||||||
| Total | $ | ( | $ | ( | $ | ( | ||||||||||||||
Depreciation and amortization have been charged in the following expense categories (in USD thousands):
| For the year ended December 31, | ||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||||||||||||||||||||
| Depreciation | Amortization | Depreciation | Amortization | Depreciation | Amortization | |||||||||||||||||||||||||||||||||
| Cost of revenue | $ | $ | ( | $ | $ | ( | $ | $ | ( | |||||||||||||||||||||||||||||
| Research and development costs | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| Selling and marketing costs | ( | ( | ( | |||||||||||||||||||||||||||||||||||
| General and administrative costs | ( | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||
| Total | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||||||
The table presents employee costs by function, which consists of “Employee benefit expenses”, “Social charges” and “Share-based compensation” from the operating expense table (in USD thousands):
| For the year ended December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Research and development costs | ||||||||||||||||||||
| Selling and marketing costs | ||||||||||||||||||||
| General and administrative costs | ||||||||||||||||||||
| Total | $ | $ | $ | |||||||||||||||||
F-19
7. Other operating income, net
Accounting policies
The Company records income and expenses that are not regularly occurring or normal business income and expense to other operating income (expense). Other operating income (expense) consists of government grants, gains on disposal of tangible assets, intangible write-offs, and other operating income (expense).
8. Interest income (expense), net and foreign exchange and other losses
| December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Interest income | $ | $ | $ | |||||||||||||||||
| Total interest income | $ | $ | $ | |||||||||||||||||
| Interest on loans | ( | |||||||||||||||||||
| Interest on lease liabilities | ( | ( | ( | |||||||||||||||||
| Other interest | ( | ( | ( | |||||||||||||||||
| Total interest expense | $ | ( | $ | ( | $ | ( | ||||||||||||||
| Total Interest income (expense), net | $ | $ | $ | ( | ||||||||||||||||
| December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Derivative fair value (losses) | ( | |||||||||||||||||||
| Foreign exchange gains (losses), net | ( | ( | ||||||||||||||||||
| Total foreign exchange and other losses | $ | ( | $ | ( | $ | ( | ||||||||||||||
Accounting policies
Interest income consists of interest income earned on cash and cash equivalents, short-term investments, and lease receivables.
Interest expense on lease liabilities and loans, which includes, interest on commercial borrowings.
The foreign exchange gains and losses arise principally on intercompany receivable balances in the parent company, whose functional currency is the Swiss Franc.
9. Income tax
Material accounting estimates and judgments
Uncertain tax positions
The Company files tax returns as prescribed by the tax laws of the jurisdictions in which it operates and therefore could be subject to tax examination by various taxing authorities. In the normal course of business, the Company is subject to examination by local tax authorities in Switzerland, France, Italy, Brazil, the UK, the US, and Australia. In 2022 a tax assessment examination was rendered by the French tax authority during an audit of the Company’s 2018 and 2019 tax returns. In 2023, a tax assessment was rendered by the French tax authority during the review of the 2022 tax return as discussed below. The Company is not aware of any additional issues that could result in any other significant payments, accruals or material deviation from its tax positions. There were no other tax examinations in progress as of December 31, 2023.
The Company records tax liabilities or benefits for all years subject to examination based upon management’s evaluation of the facts, circumstances and information available at the reporting date. There is inherent
F-20
uncertainty in quantifying income tax positions, especially considering the complex tax laws and regulations in each of the jurisdictions in which the Company operates.
Accounting policies
The Company is subject to taxes in different countries. Taxes and related fiscal assets and liabilities recognized in the Company’s consolidated financial statements reflect management’s best estimate of the outcome based on the facts known at the balance sheet date in each individual country. These facts may include but are not limited to change in tax laws and interpretation thereof in the various jurisdictions where the Company operates. They may have an impact on the income tax as well as the resulting income tax assets and liabilities. Any differences between tax estimates and final tax assessments are charged to the statement of income/loss in the period in which they are incurred. Taxes include current and deferred taxes on income as well as actual or potential withholding taxes on current and expected transfers of income from subsidiaries and tax adjustments relating to prior years. Income tax is recognized in the statement of income/loss, except to the extent that it relates to an item directly taken to other comprehensive income/loss or equity, in which case it is recognized against other comprehensive income/loss or equity, respectively.
Current income tax liabilities refer to the portion of the tax on the current year taxable profit (as determined according to the rules of the taxation authorities) and includes uncertain tax liabilities. The Company determines the taxable profit (tax loss), tax bases, unused tax losses, unused tax credits and tax rates consistently with the tax treatment used or planned to be used in its income tax filings if the Company concludes it is probable that the taxation authority will accept an uncertain tax treatment.
Otherwise, the Company reflects the effect of uncertainty using either the most likely outcome or the expected value outcome, depending on which method the entity expects to better predict the resolution of the uncertainty.
Deferred taxes are based on the temporary differences that arise when taxation authorities recognize and measure assets and liabilities with rules that differ from the accounting policies of the Company’s consolidated financial statements. They also arise on temporary differences stemming from tax losses carried forward. Deferred taxes are measured at the rates of tax expected to prevail when the temporary differences reverse, subject to such rates being substantively enacted at the balance sheet date. Any changes of the tax rates are recognized in the statement of income/loss unless related to items directly recognized against other comprehensive income. Deferred tax liabilities are recognized on all taxable temporary differences excluding non-deductible goodwill. Deferred tax assets are recognized for unused tax losses, unused tax credits and deductible temporary differences to the extent that it is probable that future taxable profits will be available against which they can be used. Future taxable profits are determined based on the reversal of relevant taxable temporary differences. If the amount of taxable temporary differences is insufficient to recognize a deferred tax asset in full, then future taxable profits, adjusted for reversals of existing temporary differences, are considered, on the basis of the business plans for individual subsidiaries in the Company. Deferred tax assets are reviewed at each reporting date and are reduced to the extent that it is no longer probable that the related tax benefit will be realized; such reductions are reversed when the probability of future taxable profits improves.
The tax impact of a transaction or item can be uncertain until a conclusion is reached with the relevant tax authority or through a legal process. The Company uses in-house tax experts when assessing uncertain tax positions and seeks the advice of external professional advisors where appropriate. The assessment of the uncertain tax position is done by first making a determination of whether it is more likely than not that a tax position would be sustained upon an examination, and then by calculating the amount of the benefit, of that tax position that meets the more likely than not threshold, that should be recognized in the financial statements.
As of December 31, 2023, and 2022, the Company recorded a provision of less than $0.1 million and a release of $0.3 million for unrecognized tax liabilities including interest and penalties. The Company records interest and penalties related to income tax amounts as a component of income tax expense.
France tax audit
The French tax authority issued a tax assessment in December 2022 that reduced the balance of the Company’s tax losses carryforward in France by $1.8 million ($0.5 million tax effected amount) stemming from a review of the Company’s transfer pricing policy. The tax assessment is subject to appeal. However, the Company has elected to take a conservative approach and adjusted the balance of its deferred tax assets to
F-21
reflect the reduction in the balance of tax losses carryforward. The tax assessment in France has resulted in no other material tax liability or payment. In 2023, the French tax authority performed a review of the Company’s 2022 French tax return. No tax assessments resulted from the review of the 2022 French tax return.
Presentation of tax (expense) benefits
The following table presents the current and deferred tax (expense) benefits (in USD thousands):
| For the year ended December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Current income tax expense | ||||||||||||||||||||
| Current year | $ | ( | $ | ( | $ | |||||||||||||||
| Uncertain tax positions | ( | ( | ||||||||||||||||||
| Total current income tax expense | $ | ( | $ | $ | ( | |||||||||||||||
| Deferred income tax (expense) benefit | ||||||||||||||||||||
| Origination and reversal of temporary differences | $ | ( | $ | $ | ( | |||||||||||||||
| Total deferred income tax (expense) benefit | $ | ( | $ | $ | ( | |||||||||||||||
| Total income tax (expense) benefit | $ | ( | $ | $ | ( | |||||||||||||||
The following table presents the reconciliation of the expected tax expense to the tax expense report in the statement of loss (in USD thousands):
| For the year ended December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Loss before tax | $ | ( | $ | ( | $ | ( | ||||||||||||||
| Tax at Swiss statutory rate | ||||||||||||||||||||
| Effect of tax rates in foreign jurisdictions | ( | ( | ( | |||||||||||||||||
| Tax effect of: | ||||||||||||||||||||
| Unrecognized deferred tax assets | ( | ( | ( | |||||||||||||||||
| Income not subject to tax (expense not deductible for tax purposes) | ( | ( | ( | |||||||||||||||||
| Uncertain tax positions | ( | ( | ||||||||||||||||||
| Recognition of deferred tax assets from previously unrecognized tax assets | ||||||||||||||||||||
| 2018-2019 French tax assessment | ( | |||||||||||||||||||
| Other | ( | |||||||||||||||||||
| Income tax (expense)/benefit | $ | ( | $ | $ | ( | |||||||||||||||
F-22
Movement in the deferred tax balances
During the year ended December 31, 2023, the Company recognized deferred tax assets for its foreign subsidiaries due to intercompany transfer pricing arrangements that will assure realization of their respective deferred tax assets in each country. The following table presents the changes in the Company’s deferred tax assets and deferred tax liabilities (in USD thousands):
| Depreciation & amortization | Bad debt reserves | Accrued pension | ROU asset | Lease liability | Other | Net operating loss carryforward | Total | |||||||||||||||||||||||||||||||||||||||||||
| January 1, 2023 | $ | ( | $ | $ | $ | ( | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||
| Recognized in profit or loss | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Recognized in OCI | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Currency translation differences | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2023 | $ | ( | $ | $ | $ | ( | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||
| Deferred tax assets | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Deferred tax liabilities | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||
| Depreciation & amortization | Bad debt reserves | Accrued pension | ROU asset | Lease liability | Other | Net operating loss carryforward | Total | |||||||||||||||||||||||||||||||||||||||||||
| January 1, 2022 | $ | ( | $ | $ | $ | ( | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||
| Recognized in profit or loss | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||
| Recognized in OCI | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Currency translation differences | ( | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
| December 31, 2022 | $ | ( | $ | $ | $ | ( | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||
| Deferred tax assets | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Deferred tax liabilities | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||
Unrecognized deferred tax assets
As of December 31, 2023 and 2022, the Company recognized deferred tax assets to the extent that it was probable that they would be realized. The following table consists of the deferred tax assets that have not been recognized because it is not probable that there will be future taxable profits to use these benefits (in USD thousands):
| December 31, | ||||||||||||||||||||||||||
| 2023 | 2022 | |||||||||||||||||||||||||
| Gross amount | Tax effect | Gross amount | Tax effect | |||||||||||||||||||||||
| Deductible temporary differences | $ | $ | $ | $ | ||||||||||||||||||||||
| Net operating loss carryforwards | ||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | ||||||||||||||||||||||
F-23
Net operating loss carryforwards
As of December 31, 2023 and 2022, the Company had various net operating loss (“NOL”) carryforwards in Switzerland, France, the UK, the US, and Brazil that are available to reduce future taxable income and income taxes, the majority of which will expire at various dates through 2030. As of December 31, 2023 and 2022, the Company had the following expiring amounts of unrecognized NOL carryforwards (in USD thousands):
| December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| One year | $ | $ | ||||||||||||
| Two years | ||||||||||||||
| Three years | ||||||||||||||
| Four years | ||||||||||||||
| Thereafter and unlimited | ||||||||||||||
| Net operating loss carryforwards | $ | $ | ||||||||||||
Future realization of the tax benefits of existing temporary differences and NOL carryforwards ultimately depends on the existence of sufficient taxable income within the carryforward period. As of December 31, 2023, the Company performed an evaluation to determine the likelihood of realization of these tax benefits. In assessing the realization of the deferred tax assets, the Company considered whether it is more likely than not that some portion or all of the deferred tax assets will not be realized. The Company considered all available evidence, both positive and negative, which included the results of operations for the current and preceding years. The Company determined that it was not possible to reasonably quantify future taxable income and determined that it is not probable that all of the deferred tax assets will be realized in Switzerland and Brazil but has recognized deferred tax assets in France, the UK and the US.
Unrecognized deferred tax liability on retained earnings of subsidiaries
The Company reviews its plan to indefinitely reinvest on a periodic basis for each one of its foreign subsidiaries. In making its decision to indefinitely reinvest, the Company evaluates its plans of reinvestment, its ability to control repatriation and to mobilize funds without triggering basis differences, and the profitability of its Swiss operations and associated cash requirements and the need, if any, to repatriate funds. If the assessment of the Company with respect to any earnings of its foreign subsidiaries’ changes, deferred Swiss income taxes, foreign income taxes, and foreign withholding taxes may have to be accrued.
The Company does not provide for foreign income and withholding taxes, Swiss income taxes or tax benefits on the excess of the financial reporting basis over the tax basis of its investments in foreign subsidiaries to the extent that such amounts are indefinitely reinvested to support operations and continued growth plans outside of Switzerland, or if the Company has determined that no tax liability would arise in case of distribution.
As of December 31, 2023, the Company plans to indefinitely reinvest any undistributed foreign earnings for all its foreign subsidiaries except France. During the year 2023, the Company received a dividend payment from its French subsidiary in the amount of $2.4 million, paid on December 12, 2023. During the year 2022, the Company received a dividend payment from its French subsidiary in the amount of $3.4 million.
The Company has determined that the repatriation of foreign earnings from France does not trigger a tax liability, based on the application of Swiss Participation Exemption rules and exemptions provided by the Double Tax Treaty signed between France and Switzerland, based on which dividends are exempt from withholding tax. The total amount of temporary differences associated with the other investments in subsidiaries is not material.
F-24
10. Loss per share
The Company’s shares comprised of ordinary shares. Each share has a nominal value of $0.05 (CHF 0.05 ). The basic loss per share is calculated by dividing the net loss attributable to shareholders by the weighted average number of shares in issue during the period. The table presents the loss for the year ended December 31, 2023, 2022, and 2021, respectively (in USD thousands, except shares and loss per share):
| Year ended December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Net loss attributed to shareholders | $ | ( | $ | ( | $ | ( | ||||||||||||||
| Weighted average number of shares in issue | ||||||||||||||||||||
| Basic and diluted loss per share | $ | ( | $ | ( | $ | ( | ||||||||||||||
11. Cash and cash equivalents
Accounting policies
Cash and cash equivalents include cash on hand, deposits held at call with external financial institutions and other short- term highly liquid investments with original maturities of three months or less. They are both readily convertible to known amounts of cash and so near to their maturity that they present insignificant risk of changes in value because of changes in interest rates. Amounts held in money market funds held as cash equivalents and are classified as level 1 fair value financial instrument.
The following table presents the allocation between the Company’s cash and cash equivalents (in USD thousands):
| December 31, | |||||||||||
| 2023 | 2022 | ||||||||||
| Bank balances | $ | $ | |||||||||
| Total cash | $ | $ | |||||||||
| Money market funds | $ | $ | |||||||||
| Term deposits less than 3 months | $ | $ | |||||||||
| Total cash equivalents | $ | $ | |||||||||
| Cash and cash equivalents | $ | $ | |||||||||
Designated cash
In July 2021, the Company designated $30.0 million to a separate bank account to be used exclusively to settle potential liabilities arising from claims against Directors and Officers covered under the Company’s Directors and Officers Insurances Policy (“D&O Policy”). Setting up the designated account has significantly reduced the premiums associated with the D&O Policy. In June 2023, the Company obtained a new D&O Policy that allowed it to reduce the designated cash amount set aside in the separate bank account from $30.0 million to $15 million. The new D&O policy and reduction of designated cash are effective as of July 2023. The Company expects to continue to designate this cash balance for this sole use under the D&O Policy.
F-25
12. Term deposits
The following table presents the allocation between the Company’s term deposits (in USD thousands):
| December 31, | |||||||||||
| 2023 | 2022 | ||||||||||
| Term deposits, over 3 months, up to 12 months | $ | $ | |||||||||
| Total term deposits | $ | $ | |||||||||
13. Accounts receivable
Material accounting estimates and judgments
The Company has adopted the simplified method indicated in IFRS 9, Financial Instruments (“IFRS 9”), to build its allowance for expected credit losses (“ECL”). The Company uses a matrix based on a calculation of collectability rates according to historical accounts receivable. Allowance is made for lifetime expected credit losses as invoices are issued. The amount of allowance initially recognized is based on historical experience, tempered by expected changes in future cash collections, due to, for example, expected improved customer liquidity or more active credit management.
Accounting policies
Accounts receivable balances are non-interest bearing and payment terms are generally under agreements with payment terms of up to 180 days. The Company’s customers primarily consist of government-owned or government-funded hospitals, laboratories with a low credit risk, and biopharmaceutical companies. The Company has had minimal instances of actual credit losses and considers that this will continue to be the case.
The following table presents the accounts receivable and lease receivable less the expected credit loss (in USD thousands):
| As of December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| Accounts receivable | $ | $ | ||||||||||||
| Accrued contract revenue | ||||||||||||||
| Lease receivable | ||||||||||||||
| Allowance for expected credit losses | ( | ( | ||||||||||||
| Net accounts receivable | $ | $ | ||||||||||||
The movement in the allowance for expected credit losses in accounts receivable is presented below (in USD thousands):
| 2023 | 2022 | |||||||||||||
| As of January 1 | $ | $ | ||||||||||||
| Increase | ||||||||||||||
| Reversals | ( | ( | ||||||||||||
| Write-off | ( | ( | ||||||||||||
| Currency translation differences | ( | |||||||||||||
| As of December 31 | $ | $ | ||||||||||||
As of December 31, 2023 and 2022, the Company’s largest customer balance represented 24 % and 15 % of accounts receivable. All customer balances that individually exceeded 1% of accounts receivable in aggregate amounted to $6.7 million and $5.4 million as of December 31, 2023 and 2022, respectively.
F-26
Accounts receivable includes amounts receivable that relate to leases. The Company is the lessor under finance leases related to the leasing out of DNA sequencing automation equipment. The Company recorded no 0.1 million and $0.2 million.
14. Inventory
Accounting policies
Raw materials and finished goods are stated at the lower of cost calculated using the first-in, first-out (“FIFO”) method and net realizable value. Work in progress is stated at the lower of its weighted average cost and net realizable value. Cost comprises direct materials, direct labor and an appropriate proportion of variable and fixed overhead expenditure, the latter being allocated on the basis of normal operating capacity.
Inventory consists of the following (in USD thousands):
| December 31, | |||||||||||
| 2023 | 2022 | ||||||||||
| Raw materials | $ | $ | |||||||||
| Work in progress | |||||||||||
| Finished goods | |||||||||||
| Provision | ( | ( | |||||||||
| Total | $ | $ | |||||||||
Inventory provision movement for the years ended December 31, 2023 and 2022, respectively are as follows (in USD thousands):
| 2023 | 2022 | ||||||||||
| As of January 1 | $ | ( | $ | ( | |||||||
| Increase in provision | ( | ( | |||||||||
| Currency Translation Adjustment | ( | ( | |||||||||
| As of December 31 | $ | ( | $ | ( | |||||||
15. Prepaids and other current assets
The following table presents the other current assets (in USD thousands):
| As of December 31, | |||||||||||
| 2023 | 2022 | ||||||||||
| Prepayments | $ | $ | |||||||||
| VAT receivable | |||||||||||
| Government grants receivable | |||||||||||
| Other | |||||||||||
| Total | $ | $ | |||||||||
F-27
16. Property and equipment
Accounting policies
Property and equipment include leasehold improvements, computer hardware, machinery and furniture and fixtures.
Property and equipment are shown on the balance sheet at their historical cost. The cost of an asset, less any residual value, is depreciated using the straight-line method over the useful life of the asset. For this purpose, assets with similar useful lives have been grouped as follows:
•Leasehold improvements—Shorter of the useful life of the asset or the remaining term of the lease
•Computer hardware— to five years
•Machinery and equipment—Five years
•Furniture and fixtures—Five years
Useful lives, components, and residual amounts are reviewed annually. Such a review takes into consideration the nature of the assets, their intended use, including but not limited to the closure of facilities, and the evolution of the technology and competitive pressures that may lead to technical obsolescence. Depreciation of property and equipment is allocated to the appropriate headings of expenses by function in the statement of loss.
Reviews of the carrying amount of the Company’s property and equipment are performed when there is an indication of impairment. If any such indication exists, then the asset’s recoverable amount is estimated. The recoverable amount of an asset is the greater of its value in use and its fair value less costs of disposal. In assessing the value in use, the estimated future cash flows are discounted to their present value, based on the time value of money and the risks specific to the country where the assets are located.
For the year ended December 31, 2023 and 2022, the Company recorded $0.2 million and $0.1 million in accrued expense related to amounts to be paid within the next 12 months, respectively.
Property and equipment, net movement for the years ended December 31, 2023 and 2022, respectively are as follows (in USD thousands):
| Leasehold improvements | Machinery and equipment | Computer hardware | Furniture and fixtures | Total | ||||||||||||||||||||||||||||
| January 1, 2023 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||||||||
| Disposals | ( | ( | ( | ( | ||||||||||||||||||||||||||||
| Currency Translation Adjustment | ||||||||||||||||||||||||||||||||
| December 31, 2023 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Accumulated depreciation | ||||||||||||||||||||||||||||||||
| January 1, 2023 | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||
| Additions | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
| Disposals | ||||||||||||||||||||||||||||||||
| Currency Translation Adjustment | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
| December 31, 2023 | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||
| Net book value at December 31, 2023 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
F-28
| Leasehold improvements | Machinery and equipment | Computer hardware | Furniture and fixtures | Total | ||||||||||||||||||||||||||||
| January 1, 2022 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||||||||
| Disposals | ( | ( | ( | |||||||||||||||||||||||||||||
| Currency Translation Adjustment | ( | ( | ( | |||||||||||||||||||||||||||||
| December 31, 2022 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Accumulated depreciation | ||||||||||||||||||||||||||||||||
| January 1, 2022 | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||
| Additions | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
| Disposals | ||||||||||||||||||||||||||||||||
| Currency Translation Adjustment | ( | ( | ||||||||||||||||||||||||||||||
| December 31, 2022 | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||
| Net book value at December 31, 2022 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
17. Intangible Assets
Material accounting estimates and judgments
Goodwill
The Company operates as one segment or as a single cash-generating unit (“CGU”). As a single CGU, goodwill is tested by considering its recoverability in terms of the entire business. Management assesses the recoverable value of goodwill by comparing the Company’s equity value, either from observable market prices or based on discounted cash flow forecasts, to the net assets as reported in the Company’s consolidated financial statements. The value as of October 1, 2023 was based on the Company’s market capitalization, which is a factor of the Company’s outstanding shares multiplied by the price of the Company’s stock on October 1, 2023.
The value as of October 1, 2022 was based on the Company’s discounted cash flow projections, which in turn were based on historical results and ratios updated to reflect management’s expectations of future growth and profitability and discounted using a weighted average cost of capital derived from an analysis of comparable selected public companies.
Capitalized internally developed software costs
Capitalized costs are based on the employment costs of individuals working on software development and based on timesheets. Special attention is paid to distinguishing between costs incurred on developing new software or software upgrades, which may be eligible for capitalization, and costs incurred in maintenance and in the correction of problems, which is not eligible.
Judgment is required in identifying whether individual projects meet all of the criteria required to permit capitalization, in particular, whether the software will generate probable future economic benefits.
Accounting policies
Goodwill
Goodwill is initially measured as the difference between the aggregate of the value of the consideration transferred and the fair value of net assets acquired. Goodwill is not amortized but it is tested for impairment annually, or more frequently if events or changes in circumstances indicate that it might be impaired and is carried at cost less accumulated impairment losses. Gains and losses on the disposal of an entity include the carrying amount of goodwill relating to the entity sold.
F-29
Impairment testing
Intangible assets are allocated to CGUs for the purpose of impairment testing. The allocation is made to those CGUs or groups of CGUs that are expected to benefit from the business combination in which the goodwill arose. The CGUs or groups of CGUs are identified at the lowest level at which goodwill is monitored for internal management purposes, being the operating segments. As the Company operates as a single operating segment or CGU, the Company has only a single cash generating unit for impairment testing.
Management assesses the recoverable value of goodwill by comparing the value of the Company equity value, either inferred from the public prices of share issues based on the fair value less cost of disposal (“FVLCOD”) method or based on discounted cash flow forecasts, with the net assets as reported in its consolidated financial statements based on the value in use (“ViU”) method. The discounted cash flow approach involves key assumptions that leave considerable scope for judgment.The Company typically compares the two methods and utilizes the greater recoverable amount for the purposes of its impairment testing. Impairment testing is performed on an annual basis as of October 1. The value as of October 1, 2023 was based on our market capitalization, which is a factor of the Company’s outstanding shares multiplied by the price of the Company’s stock on October 1, 2023. The Company used the discounted cash flow method for the fiscal year ended as of December 31, 2022.
Purchased software
The costs of accessing software services are not capitalized if the Company does not have any contractual right to take possession of the software at any time during the term of the agreement and it is not feasible for the Company either to run the software on its own hardware or to contract with a third party unrelated to the vendor. Such costs represent software as a service costs and are expensed as incurred.
The Company does capitalize software implementation costs, such as fees paid to outside consultants to set up a software arrangement.
For cloud computing costs, the Company capitalized costs for certain configuration and customization costs paid by a customer in a cloud computing or hosting arrangement. The guidance aligns the accounting treatment of these costs incurred in a hosting arrangement treated as a service contract with the requirements for capitalization and amortization costs to develop or obtain an intangible asset.
Purchased software and associated capitalized costs are amortized using the straight-line method over an estimated life of five years .
Capitalized internally developed software costs
Costs incurred in the internal development of software are capitalized as intangible assets when the criteria required by IAS 38 as set out below is satisfied.
Software development costs consist entirely of capitalized internally generated costs that are directly attributable to the design, testing and enhancement of identifiable and unique software applications and products controlled by the Company and incorporated principally within the Company’s SOPHiA DDM Platform. They are recognized as intangible assets where the following criteria are met:
•it is technically feasible to complete the software so that it will be available for use;
•management intends to complete the software and use or sell it;
•there is an ability to use or sell the software;
•it can be demonstrated how the software will generate probable future economic benefits;
•adequate technical, financial and other resources to complete the development and to use or sell the software are available, and;
•the expenditure attributable to the software during its development can be reliably measured.
F-30
Directly attributable costs that are capitalized as part of the software comprise principally employee costs. Capitalized development costs are recorded as intangible assets and amortized from the point at which the asset is ready for use on a straight-line basis over its expected useful life. Capitalized software development costs are amortized using the straight-line method over an estimated life of five years .
The Company considers that it is only since the beginning of 2020 that development costs have fulfilled the criteria for recognition as intangible assets set out in IAS 38.
Intangible assets, net movement for the years ended December 31, 2023 and 2022, respectively are as follows (in USD thousands):
| Goodwill | Purchased software | Capitalized internally developed software costs | Total intangible assets | |||||||||||||||||||||||
| January 1, 2023 | $ | $ | $ | $ | ||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||
| Disposals | ||||||||||||||||||||||||||
| Currency Translation Adjustment | ||||||||||||||||||||||||||
| December 31, 2023 | $ | $ | $ | $ | ||||||||||||||||||||||
| Accumulated amortization | ||||||||||||||||||||||||||
| January 1, 2023 | $ | $ | ( | $ | ( | $ | ( | |||||||||||||||||||
| Additions | ( | ( | ( | |||||||||||||||||||||||
| Disposals | ||||||||||||||||||||||||||
| Currency Translation Adjustment | ( | ( | ( | |||||||||||||||||||||||
| December 31, 2023 | $ | $ | ( | $ | ( | $ | ( | |||||||||||||||||||
| Net book value at December 31, 2023 | $ | $ | $ | $ | ||||||||||||||||||||||
| Goodwill | Purchased software | Capitalized internally developed software costs | Total intangible assets | |||||||||||||||||||||||
| January 1, 2022 | $ | $ | $ | $ | ||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||
| Disposals | ( | ( | ||||||||||||||||||||||||
| Currency Translation Adjustment | ( | ( | ( | |||||||||||||||||||||||
| December 31, 2022 | $ | $ | $ | $ | ||||||||||||||||||||||
| Accumulated amortization | ||||||||||||||||||||||||||
| January 1, 2022 | $ | $ | ( | $ | ( | $ | ( | |||||||||||||||||||
| Additions | ( | ( | ( | |||||||||||||||||||||||
| Disposals | ||||||||||||||||||||||||||
| Currency Translation Adjustment | ( | ( | ( | |||||||||||||||||||||||
| December 31, 2022 | $ | $ | ( | $ | ( | $ | ( | |||||||||||||||||||
| Net book value at December 31, 2022 | $ | $ | $ | $ | ||||||||||||||||||||||
F-31
Goodwill arose from the Company’s acquisition of Interactive Biosoftware (“IBS”) in June 2018. Through this acquisition the Company added Alamut (a genomic mutation interpretation software) to its existing SOPHiA DDM Platform.
Goodwill is tested for impairment on an annual basis as of October 1 and at the occurrence of a potential indication of impairment. A triggering assessment is performed each quarter to ensure no occurrence of impairment triggering events. As of December 31, 2023 and 2022, respectively, no
As of October 1, 2023 the Company utilized the equity method (“FVLCOD”) to perform its annual assessment. The estimated equity value of the Company was $165.8 million, which exceeds the reported net assets of the Company of $158.4 million at that date by $7.3 million.
As of October 1, 2022 the Company utilized a discounted cash flow (“ViU”) method to perform its annual assessment. The Company assessed both the value of goodwill and intangibles using the discounted cash flow method. The Company used the discounted cash flow method in its annual assessment in 2022 given the significant drop in its share price from January 1, 2022, which resulted in a decline in the market capitalization of the Company.
The Company computed the value of the CGU using a discounted cash flow analysis. The discounted cash flow analysis used a forecast of seven years in order to project to a point at which the Company’s financial profile is expected to be more mature, which will allow for a more accurate valuation of the recoverable amount of the CGU. The basis of the projection for the discounted cash flow analysis was an internal plan reviewed and approved by management. The Company based its forecast on an expected compound annual growth rate (“CAGR”) of revenue and applied a weighted average cost of capital (“WACC”) and a terminal free cash flow growth rate to the discounted cash flow projections to calculate its CGU’s value. The Company performed a sensitivity analysis over the WACC, the terminal free cash flow growth rate, and the revenue CAGR.
The Company used a WACC of 12 % that was consistent with the range used in publicly available analyst valuations. The Company used a terminal free cash flow growth rate of 3 % based on an internal assessment of historical sustainable market growth rates and historical GDP growth figures that was consistent with the range of rates used in publicly available analyst valuations. The Company performed a sensitivity analysis on the WACC and the terminal free cash flow growth rate to determine the impact on the valuation. The Company determined that the level of WACC and cash flow growth rate at which an impairment of the CGU would occur are 20 % for the WACC and a negative cash flow growth rate, respectively.
The Company projected revenue over the seven-year period at a CAGR of 37 %, which is consistent with internal forecasts reviewed and approved by Management. The Company performed a sensitivity analysis to determine the CAGR at which an impairment would occur. The Company determined that at a CAGR of 25 % over a seven-year period an impairment of the CGU would occur..
On the basis of the analyses performed, the Company concludes that the recoverable amount exceeds the carrying amount of the goodwill and no
18. Leases
Accounting policies
Lessee
The Company assesses at inception of the contract whether a contract is or contains a lease. This assessment involves determining whether the Company obtains substantially all the economic benefits from the use of that asset, and whether the Company has the right to direct the use of the asset. When these conditions are met, the Company recognizes a right-of-use (“ROU”) asset and a lease liability at the lease commencement date, except for short-term leases of 12 months or less, which are expensed in the statement of income/loss on a straight-line basis over the lease term.
F-32
At inception, the ROU asset comprises the initial lease liability, initial direct costs, and any obligations to refurbish the asset, less any incentives granted by the lessors.
The ROU asset is depreciated over the shorter of the duration of the lease contract (including contractually agreed optional extension periods whose exercise is deemed to be reasonably certain) and the useful life of the underlying asset.
The ROU asset is subject to testing for impairment if there is an indicator for impairment, as for owned assets.
The lease liability is initially measured at the present value of the lease payments that are not paid at the commencement date, discounted using the interest rate implicit in the lease or, if that is not readily determinable, the incremental borrowing rate (“IBR”) at the lease commencement date. The IBR is the rate of interest that the Company would have to pay to borrow over a similar term, and with a similar security, the funds necessary to obtain an asset of a similar value to the ROU asset in a similar economic environment. Lease payments can include fixed payments; variable payments that depend on an index or rate known at the commencement date; and extension option payments or purchase options that the Company is reasonably certain to exercise.
The lease liability is subsequently measured at amortized cost using the effective interest rate method and remeasured (with a corresponding adjustment to the related ROU asset) when there is a change in future lease payments due to renegotiation, changes in an index or rate or a reassessment of options.
Some of the Company’s leases include options to extend the lease, and these options are included in the lease term to the extent they are reasonably certain to be exercised.
Lessor
The Company leases out laboratory equipment to certain customers. These leases are classified as finance leases as the Company transfers substantially all the risks and rewards incidental to ownership of the asset to the customer.
At the commencement of the lease term, the Company records revenue and the associated costs of sales, being the sale proceeds at fair value of the asset (computed at cost plus a margin) and the cost of the asset, derecognizes the leased asset from inventory, and recognizes a finance lease receivable on the balance sheet equal to the net investment in the lease. As of December 31, 2023, the Company did not have any leases of laboratory equipment.
Company leases
During the year ended December 31, 2023, the Company entered into one significant lease as described below.
Rolle office
On March 3, 2021, the Company entered into a 120-month lease for office space in Rolle, Switzerland primarily to support the expansion of the research and development department. The lease in total is for approximately 65,860 square feet, including an additional 21,258 square feet based on a lease amendment as described below, with the Company gaining access to areas on prescribed dates. The Company gained access to 11,840 square feet on July 1, 2021. The Company gained access to 7,535 square feet on January 1, 2022 and the remaining 21,258 square feet on February 1, 2023. The expected lease commitments resulting from this contract are less than $0.1 million in 2021, $0.5 million in 2022, $1.0 million in 2023 onwards, and $1.14 million from 2024 onward. The expected lease commitments are linked to changes in the Swiss Consumer Price Index as published by Swiss Federal Statistical Office.
On January 25, 2022, the Company entered into an amendment to the lease for office space in Rolle, Switzerland. The amendment provides the Company with an additional floor of approximately 21,258 square feet with lease commencement initiating on April 1, 2022. Upon commencement of the lease, the Company recorded a right-of-use asset of $4.5 million and a lease liability of $4.5 million.
The Company makes fixed payments and additional variable payments depending on the usage of the asset during the contract period. Upon commencement of the lease, the Company recorded a ROU asset of
F-33
$7.7 million and a lease liability of $8.5 million. The difference between the ROU and lease liability of $0.8 million is driven by lease incentives and expected restoration costs.
Boston office
On August 9, 2021, the Company entered into a 40-month new lease for office space in Boston, Massachusetts to support the expansion of the Company’s growth in the United States. The lease in total for the expansion of the Boston office is approximately 9,192 square feet. The expected lease commitments resulting from this contract are $0.5 million a year starting in 2022 through the end of the lease in 2024. The Company makes fixed payments and additional variable payments depending on the usage of the asset during the contract period. Upon commencement of the lease, the Company recorded a right-of-use asset of $1.2 million and a lease liability of $1.4 million. The difference between the ROU and lease liability of $0.2 million is driven by lease incentives.
Bidart office
On June 1, 2023, the Company entered into a 108-month lease for office space in Bidart, France primarily to support the expansion of the research and development department. The lease in total is for approximately
Leases
Generally, lease terms for office buildings are between and ten years . Any leases with terms less than 12 months and/or with low value are expensed in accordance with the IFRS 16 practical expedients for short-term leases and low-value leases. These expenses amounted to less than $0.1 million and $0.2 million for the years ended December 31, 2023 and 2022, respectively. The Company had cash outflows related to leases less than 12 months and/or with low value of less than $0.1 million and $0.2 million for the years ended December 31, 2023 and 2022, respectively.
The Company has lease liabilities amounting to $17.8 million and $14.7 million for the years ended December 31, 2023 and 2022, respectively, that are linked to consumer price indices in Switzerland and France.
The future cash flow in relation to short-term leases and leases of low value assets is disclosed in Note 27 – “Commitments and contingencies.”
The future cash flow in relation to leases accounted for under IFRS 16 is disclosed in Note 28 – “Financial instruments.”
The Company has several leases with extension and termination options. Management determines, on the basis of the business needs, whether they expect to exercise these options.
The lease liability is initially measured at the present value of the lease payments that are not paid at the commencement date, discounted using the interest rate implicit in the lease or, if that is not readily determinable, the IBR at the lease commencement date. The IBR is the rate of interest that the Company would have had to pay to borrow over a similar term, and with a similar security, the funds necessary to obtain an asset of a similar value to the ROU asset in a similar economic environment. On the basis of this policy, the IBRs used by the Company to discount lease payments outstanding at December 31, 2023 and 2022, respectively, in the countries in which it has recognized right-of-use assets and lease liabilities have been in the range of 2.61 % to 4.82 % and 2.61 % to 3.47 %, respectively.
F-34
The following table presents the movements in the ROUs (in USD thousands):
| 2023 | 2022 | |||||||||||||
| As of January 1 | $ | $ | ||||||||||||
| Additions | ||||||||||||||
| Depreciation charge | ( | ( | ||||||||||||
| Currency translation effects | ||||||||||||||
| As of December 31 | $ | $ | ||||||||||||
The following table presents the movements in the lease liabilities (in USD thousands):
| 2023 | 2022 | |||||||||||||
| As of January 1 | $ | $ | ||||||||||||
| Additions | ||||||||||||||
| Cash outflows (principle and interest) | ( | ( | ||||||||||||
| Non-cash interest | ||||||||||||||
| Currency translation effects | ||||||||||||||
| As of December 31 | $ | $ | ||||||||||||
19. Other non-current assets
Other non-current assets consist of the following (in USD thousands):
| December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| Research tax credit receivable | $ | $ | ||||||||||||
| Guarantee deposits | ||||||||||||||
| Total | $ | $ | ||||||||||||
20. Accounts payable
Accounts payable consist of the following (in USD thousands):
| As of December 31, | |||||||||||
| 2023 | 2022 | ||||||||||
| Trade payables | |||||||||||
| Employee related payables | |||||||||||
| VAT, sales, and other taxes | |||||||||||
| Total | $ | $ | |||||||||
F-35
21. Accrued expenses
Accrued expenses consist of the following (in USD thousands):
| As of December 31, | |||||||||||
| 2023 | 2022 | ||||||||||
| Accrued Compensation | $ | $ | |||||||||
| Accrued Professional Fees | |||||||||||
| Accrued Inventory Purchases | |||||||||||
| Accrued IT Support | |||||||||||
| Accrued Legal Fees | |||||||||||
| Accrued Other | |||||||||||
| Total | $ | $ | |||||||||
22. Post-employment benefits
Material accounting estimates and judgments
The liability or asset recognized on the balance sheet in respect of defined benefit pension plans is the present value of the defined benefit obligation at the end of the reporting period less the fair value of plan assets. The defined benefit obligation is calculated annually by independent actuaries using the projected unit credit method.
The present value of the defined benefit obligation is determined by discounting the estimated future cash outflows using interest rates of high-quality corporate bonds that are denominated in the currency in which the benefits will be paid, and that have terms approximating to the terms of the related obligation. In countries where there is no deep market in such bonds, the market rates on government bonds are used.
The net interest cost is calculated by applying the discount rate to the net balance of the defined benefit obligation and the fair value of plan assets. This cost is included in employee benefit expense in the statement of income/loss.
Remeasurement gains and losses arising from experience adjustments and changes in actuarial assumptions are recognized in the period in which they occur, directly in other comprehensive income. The remeasurement gains and losses are included in retained earnings in the statement of changes in equity and on the balance sheet.
Changes in the present value of the defined benefit obligation resulting from plan amendments or curtailments are recognized immediately in income as past service costs.
For defined contribution plans, the Company pays contributions to publicly or privately administered pension insurance plans. Employee contributions to these plans is voluntary and these contributions are matched by the employer. The Company has no further payment obligations once the contributions have been paid. The contributions are recognized as employee benefit expense when they are due. Prepaid contributions are recognized as an asset to the extent that a cash refund or a reduction in the future payments is available. Contributions are charged to the statement of income/loss as incurred.
Accounting policies
The Company operates defined benefit and defined contribution pension plans. Funded schemes are generally funded through payments to insurance companies or trustee-administered funds, determined by periodic actuarial calculations. A defined contribution plan is a pension plan under which the Company pays fixed contributions into a separate entity (a fund) and has no legal or constructive obligations to pay further contributions if the fund does not hold sufficient assets to pay all employees the benefits relating to employee service in the current and prior periods. A defined benefit plan is a pension plan that is not a defined contribution
F-36
plan. Typically, defined benefit plans define an amount of pension benefit that an employee will receive on retirement, usually dependent on one or more factors such as age, years of service and compensation.
The actual return on plan assets, excluding interest income measured at the discount rate, is recognized in other comprehensive income/loss within defined benefit plan remeasurements.
The Company has a funded defined benefit plan in Switzerland, an unfunded defined benefit plan in France and a defined contribution plans in the US. The Company has no occupational pension plans in the UK and Brazil.
Swiss pension plan
The Company contracted with the Swiss Life Collective BVG Foundation based in Zurich for the provision of occupational benefits. All benefits in accordance with the regulations are reinsured in their entirety with Swiss Life SA within the framework of the corresponding contract. This pension solution fully reinsures the risks of disability, death and longevity with Swiss Life. Swiss Life invests the vested pension capital and provides a 100% capital and interest guarantee. The pension plan is entitled to an annual bonus from Swiss Life comprising the effective savings, risk and cost results.
Although the amount of ultimate pension benefit is not defined, certain legal obligations of the plan create constructive obligations on the employer to pay further contributions to fund an eventual deficit; this results in the plan nevertheless being accounted for as a defined benefit plan.
French pension plan
In France, the bulk of pensions are paid by national pension schemes, which are unfunded. In addition, French employers are obliged by law to pay a retirement indemnity. Its amount depends on the last salary of the employee and on the period of activity with its employer. Rights to this benefit are acquired during the service life with the same employer on the condition that the employee will be with its employer at retirement date; it means that the rights are only vested on retirement date. This indemnity is in substance a defined benefit plan.
The following table provides additional details on the defined benefit plans’ funded status (in USD thousands):
| December 31, | |||||||||||
| 2023 | 2022 | ||||||||||
| Present value of defined benefit obligation | $ | ( | $ | ( | |||||||
| Fair value of plan assets | |||||||||||
| Net pension liability | $ | ( | $ | ( | |||||||
F-37
The following table presents the movement in the defined benefit obligation (in USD thousands):
| 2023 | 2022 | ||||||||||||||||||||||||||||||||||
| Funded | Unfunded | Total | Funded | Unfunded | Total | ||||||||||||||||||||||||||||||
| January 1 | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||
| Service Cost | ( | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||
| of which current service cost | ( | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||
| of which past service cost including effects from curtailment | |||||||||||||||||||||||||||||||||||
| Interest expense | ( | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||
| Actuarial gains (losses) | |||||||||||||||||||||||||||||||||||
| Actual plan participants’ contributions | ( | ( | ( | ( | |||||||||||||||||||||||||||||||
| Transfers (in) out due to (joiners) leavers | ( | ( | |||||||||||||||||||||||||||||||||
| Currency translation differences | ( | ( | ( | ||||||||||||||||||||||||||||||||
| December 31 | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||
The service cost and interest expense are charged to the statement of income/loss as pension cost. Actuarial gains (losses) are credited or charged to other comprehensive income (loss) as defined benefit plan remeasurements.
As of December 31, 2023, the Swiss and French plans had 233 and 97 active members, respectively. As of December 31, 2022, the Swiss and French plans had 248 and 102 active members, respectively.
As a result of the reduction in conversion factors, the Company incurred a past service cost gain including curtailment of $0.4 million for the year ended December 31, 2023.
The following table presents the movement in the defined benefit plans’ assets (in USD thousands):
| 2023 | 2022 | ||||||||||
| As of January 1 | $ | $ | |||||||||
| Interest income | |||||||||||
| Return on plan assets, excl. interest income | ( | ( | |||||||||
| Administrative expenses | ( | ( | |||||||||
| Employer contributions | |||||||||||
| Employee contributions | |||||||||||
| Transfers in (out) due to joiners (leavers) | ( | ||||||||||
| Currency translation differences | ( | ||||||||||
| As of December 31 | $ | $ | |||||||||
The following table presents the defined benefit plan assets, which include the following (in USD thousands):
| December 31, | |||||||||||
| 2023 | 2022 | ||||||||||
| Cash | $ | $ | |||||||||
| Insurance policies | |||||||||||
| Total | $ | $ | |||||||||
F-38
The Swiss Life Collective BVG Foundation, to which the Swiss pension plan is affiliated, manages its funds in the interests of all members, with due attention to the priorities of liquidity, security, and return. The Company’s pension plan benefits from the economies of scale and diversification of risk available through this affiliation. The Company has no influence over the investment policy.
The follow table presents the pension costs recognized in statement of loss (in USD thousands):
| December 31, | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2021 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Funded | Unfunded | Total | Funded | Unfunded | Total | Funded | Unfunded | Total | |||||||||||||||||||||||||||||||||||||||||||||
| Service cost | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||||||||||||||
| Interest cost | ( | ( | ( | ( | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
| Total recognized | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||||||||||||||
The follow table presents the pension remeasurement recognized in statement of other comprehensive loss (in USD thousands):
| December 31, | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2021 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Funded | Unfunded | Total | Funded | Unfunded | Total | Funded | Unfunded | Total | |||||||||||||||||||||||||||||||||||||||||||||
| Changes in demographic assumptions | $ | $ | $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||
| Changes in financial assumptions | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Experience adjustments | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Total actuarial gains (losses) | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Return on plan assets | ( | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Currency translation differences | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Total recognized | $ | ( | $ | $ | ( | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||
The positive impact of changes in demographic assumptions in 2023 was due principally to an increase in the weighted turnover from 15.80 % to 19.50 %.
The positive impact of changes in demographic assumptions in 2022 was due principally to an increase in the expected employee salaries from 1.25 % to 3.00 %. This implies that more members are expected to have a higher pensionable amount before pensionable age.
The negative impact of changes in financial assumptions in 2023 was due to a decrease in the discount rate from 2.25 % to 1.50 %.
The positive impact of changes in financial assumptions in 2022 was due to an increase in the discount rate from 0.30 % to 2.25 %.
The positive experience adjustments in 2023 was due largely to the surplus between the additional defined benefit obligation attributable to new joiners and the assets that they transferred into the plan.
F-39
Key actuarial assumptions by plan
Discount rate
In estimating the defined benefit obligation, the discount rates used were, for the Swiss plan, 1.50 % and 2.25 % and, for the French plan, 3.55 % and 3.90 % for the years ended December 31, 2023 and 2022, respectively.
Expected rate of salary increase
The expected rate of annual salary increase was assumed to be, for the Swiss plan 2.75 % and 3.00 % and for the French plan 3.00 % and 3.00 % for the years ended December 31, 2023 and 2022, respectively.
Pension plan modified duration
The weighted average modified duration of the Swiss plan is 11.7 and 13.2 years and of the French plan 16.1 and 16.0 years for the years ended December 31, 2023 and 2022, respectively.
Interest rates
For the Swiss plan, the interest on old age accounts is based, for the LPP account, on the LPP interest rate, which was 1.50 % and 2.25 % and, for the extra mandatory part, is equivalent to the discount rate, which was 1.50 % and 2.25 % for the years ended December 31, 2023 and 2022, respectively.
Inflation
For the Swiss plan, the expected annual rate of inflation is based on the inflation forecast of the Swiss National Bank and was assumed to be 1.25 % and 1.50 % for the years ended December 31, 2023 and 2022, respectively.
Mortality tables
Assumptions regarding future mortality experience are set based on actuarial advice provided in accordance with published statistics and experience and are based on the mortality generational tables BGV 2020 (Swiss) and TH/TF 00-02 (French). For the Swiss plan, the average life expectancy in years after retirement of a pensioner retiring at age 65 (male) and 65 (female) on the balance sheet date is, respectively, 22.82 and 22.70 and 24.59 and 22.48 , for the years ended December 31, 2023 and 2022, respectively.
Sensitivity analysis
The following tables demonstrate the sensitivity of the defined benefit obligations to changes in the discount rate, expected rates of salary increase, interest credited on savings accounts, inflation and life expectancy at retirement age.
F-40
The table below presents the sensitivity analysis for the funded plans (in USD thousands):
| 2023 | 2022 | ||||||||||
| Discount rates | |||||||||||
Increase of | ( | ( | |||||||||
Decrease of | |||||||||||
| Expected rates of salary increases | |||||||||||
Increase of | |||||||||||
Decrease of | ( | ( | |||||||||
| Interest rate | |||||||||||
Increase of | |||||||||||
Decrease of | ( | ( | |||||||||
| Inflation | |||||||||||
Increase of | |||||||||||
Decrease of | ( | ( | |||||||||
| Life expectancy | |||||||||||
Increase of | |||||||||||
Decrease of | ( | ( | |||||||||
The table below presents the sensitivity analysis for the unfunded plans (in USD thousands):
| 2023 | 2022 | ||||||||||
| Discount rates | |||||||||||
Increase of | ( | ( | |||||||||
Decrease of | |||||||||||
| Expected rates of salary increases | |||||||||||
Increase of | |||||||||||
Decrease of | ( | ( | |||||||||
The above sensitivity analyses are based on a change in an assumption while holding all other assumptions constant. In practice, this is unlikely to occur, and changes in some of the assumptions may be correlated. When calculating the sensitivity of the defined benefit obligation to significant actuarial assumptions the same method (present value of the defined benefit obligation calculated with the projected unit credit method at the end of the reporting period) has been applied as when calculating the pension liability recognized on the balance sheet.
The methods and types of assumptions used in preparing the sensitivity analysis did not change compared to the prior period.
Future employer contributions
Expected employer contributions to the Swiss defined benefit pension plan for the year ending December 31, 2024 amount to $1.7 million.
Defined contribution plans
US pension plan
The Company has a multiple employer 401(k) defined contribution plan in the USA. The expense recognized in respect of the defined contribution plan in the USA was $0.4 million and $0.4 million and for the years ended December 31, 2023 and 2022, respectively.
F-41
23. Share-based compensation
Material accounting estimates and judgments
Share-based Compensation
For the years ended December 31, 2023 and 2022, we granted share options under one plan - the SOPHiA GENETICS 2021 Equity Incentive Plan (the “2021 Equity Incentive Plan” or the “2021 EIP”). Under this plan, directors may offer options to directors, employees and advisors. The exercise price of the share options is set at the time they are granted. Options, once vested, can be exchanged for an equal number of ordinary shares.
Measuring the cost of share options
The fair value of the options under all plans are measured at each grant date using the Black-Scholes option pricing model, taking into account the terms and conditions upon which the options were granted.
For options up to September 2020, the fair value at grant date is independently determined using an adjusted form of the Black-Scholes option pricing model that takes into account the strike price, the fair value of the share at grant date, the expected life of the award, the expected price volatility of the underlying share, the risk-free interest rate for the term of the award and the expected dividend yield. For options granted on and subsequent to September 2020 until July 22, 2021, the fair value at grant date is based on a probability-weighted expected returns method that takes account of both the value derived by using an adjusted form of the Black-Scholes option pricing model, as described above, and a discounted estimate of the price that might be achieved in a future transaction. For options granted on and subsequent to July 22, 2021, the fair value at grant date is determined by using the Black-Scholes option pricing model.
The Company has used an independent valuation firm to assist in calculating the fair value of the award grants per participant.
The key inputs used in the valuation model, for the stock options granted in the years ended December 31, 2023 and 2022, respectively, are outlined below. Stock options were only granted under the 2021 Employee Incentive Plan (“2021 EIP”). No
Prior to the Company’s IPO, the price of the ordinary shares at grant date, which represents a critical input into this model, has been determined on one of the following two bases:
•By reference to a contemporaneous transaction involving another class of share, using an adjusted form of the Black-Scholes option pricing model as described above, and considering the timing, amount, liquidation preferences and dividend rights of issues of other classes of shares.
•On the basis of discounted cash flow forecasts, where there was no contemporaneous or closely contemporaneous transaction in another class of share and the time interval was too large to permit an assumption that there had been no significant change in the Company’s equity value.
Subsequent to the IPO, the price of the ordinary shares at grant date, which represents a critical input into this model, has been determined on the most recent close price of the Company’s stock price on the date of grant.
Accounting policies
The Company has three share option plans for directors, employees, and advisors which are accounted for as equity-settled share-based compensation plans.
The fair value of options granted under these plans is recognized as an employee benefits expense, with a corresponding increase in equity. The total amount to be expensed is determined by reference to the fair value of the options granted:
•including any market performance conditions (e.g., the entity’s share price);
F-42
•excluding the impact of any service and non-market performance vesting conditions (e.g., profitability, sales growth; targets and remaining an employee of the entity over a specified time period), and;
•including the impact of any non-vesting conditions (e.g., the requirement for employees to save or hold shares for a specific period of time).
The total expense is recognized over the vesting period, which is the period over which all of the specified vesting conditions are to be satisfied. At the end of each period, the entity revises its estimates of the number of options that are expected to vest based on the non-market vesting and service conditions. It recognizes the impact of the revision to original estimates, if any, in income, with a corresponding adjustment to equity.
Estimating fair value for share-based payment transactions requires determination of the most appropriate valuation model, which depends on the terms and conditions of the grant. This estimate also requires determination of the most appropriate inputs to the valuation model including the share price, or the fair value of a share, the expected life of the share option, the volatility of the share price, the risk-free interest rate, the dividend yield, and making certain assumptions about the inputs. The assumptions used for estimating fair value for share-based payment transactions are disclosed below.
The volatility used in the estimation of fair value is calculated utilizing a mix of the Company’s own share price volatility and the volatility of the share prices of a set list of publicly traded peer companies based on a defined proportion. Share price volatility is calculated for each tranche of share options on a historical basis over a period of time equal to the average life of the share options granted in each tranche. In the event that a company used in the volatility calculation has not been publicly traded for the requisite amount of time, the entirety of its trading history was used.
If the shares are not listed, estimating their fair value also requires determination of the most appropriate valuation model, such as:
•By reference to a contemporaneous transaction involving another class of share, using an adjusted form of an option pricing model above, and considering the timing, amount, liquidation preferences and dividend rights of issues of other classes of shares;
•On the basis of discounted cash flow forecasts, where there was no contemporaneous or closely contemporaneous transaction in another class of share and the time interval was too large to permit an assumption that there had been no significant change in the Company’s equity value;
•Share based compensation expense is measured at the fair value of the options at the grant date and recognized over the vesting period. Share based compensation expense is presented in the statement of income/loss and allocated to the various expense categories based on the functions of the employees to whom the options are granted (e.g., research and development, selling and marketing, general & administrative).
The calculation of the cost of the Company’s share option grants and of the fair value of the ordinary shares at the grant date requires the selection of an appropriate valuation model and is based on key assumptions that leave considerable scope for judgment.
Recognizing the cost of share options
At each reporting date, the Company takes a charge for the vested options granted and for partially earned but non-vested portions of options granted. This results in a front-loaded charge to the statement of loss. Prior to the IPO, at each reporting date, the Company reappraised its estimate of the likelihood and date of a future transaction that would cause all options which would vest six months from the transaction date to vest and, if necessary, accelerated the recognition of the unrecognized cost in the statements of loss. The Company accounts for these plans as equity-settled transactions. The charge to the statements of loss therefore results in a corresponding credit being booked to “Other reserves” within equity.
F-43
The plans
The Company has three share option plans: the 2013 ISOP (launched in September 2013), the 2019 ISOP (launched March 2019), and the 2021 EIP (launched June 2021). Under these plans, directors may offer options to directors, employees and advisors. The exercise price of the share options is set at the time they are granted. Options, once vested, can be exchanged for an equal number of ordinary shares. Under the 2021 EIP, the Company can grant restricted stock units (“RSUs”) which represent the right to receive ordinary shares upon meeting specific vesting requirements. RSUs are able to be granted to directors, executives, and employees.
The options have a life of ten years. Options under the 2013 ISOP vest 50 % on the second anniversary of the grant date and a further 50 % on the third anniversary of the grant date. Options under the 2019 ISOP vest 25 % on each anniversary of the grant date over four years . The options under the 2021 EIP vest either 25 % on the first anniversary of the grant date and the remaining 75 % vesting ratably on a monthly basis over the remaining three years , 25 % on the first anniversary of the grant date and the remaining 75 % vesting ratably on a quarterly basis over the remaining three years , on the second anniversary of the grant date, or annually over four years on each anniversary of the grant date. Refer to Restricted Stock Units below for the vesting schedules of the RSUs under the 2021 EIP.
On April 22, 2021, the Board amended the 2019 ISOP to the effect that, in the event of a successful IPO or public listing of the Company’s shares, only those unvested options that otherwise would vest within six months following the effective date of the IPO or such public listing should become fully vested immediately as of such date (accelerated vesting). The remaining unvested options (i.e., unvested options that would only vest after the six-month period following the effective date of the IPO or public listing) would not be subject to accelerated vesting and, subject to certain conditions, would vest on the basis of the original vesting schedule. Additionally, the Board instituted a black-out period, irrespective of a successful IPO or public listing of the Company, in which no options could be exercised from May 1, 2021 to January 19, 2022, and to accelerate the vesting of options that would otherwise vest during that period.
2013 ISOP
Activity for the year ended December 31, 2023, under the 2013 ISOP was as follows:
| Number of options | Weighted average exercise price | Weighted average remaining life in years | ||||||||||||||||||
| Outstanding as of January 1, 2023 | $ | |||||||||||||||||||
| Exercised | ( | — | ||||||||||||||||||
| Forfeited | ( | — | ||||||||||||||||||
| Outstanding as of December 31, 2023 | $ | |||||||||||||||||||
| Exercisable as of December 31, 2023 | $ | |||||||||||||||||||
Activity for the year ended December 31, 2022, under the 2013 ISOP was as follows:
| Number of options | Weighted average exercise price | Weighted average remaining life in years | ||||||||||||||||||
| Outstanding as of January 1, 2022 | $ | |||||||||||||||||||
| Exercised | ( | — | ||||||||||||||||||
| Forfeited | ( | — | ||||||||||||||||||
| Outstanding as of December 31, 2022 | $ | |||||||||||||||||||
| Exercisable as of December 31, 2022 | $ | |||||||||||||||||||
Options outstanding as of December 31, 2023, under the 2013 ISOP expire between 2024 and 2029.
F-44
The weighted average share price at the date of exercise were $4.82 and $7.41 for the years ended December 31, 2023 and 2022, respectively.
2019 ISOP
Activity for the year ended December 31, 2023, under the 2019 ISOP was as follows:
| Number of options | Weighted average exercise price | Weighted average remaining life in years | ||||||||||||||||||
| Outstanding as of January 1, 2023 | ||||||||||||||||||||
| Exercised | ( | — | ||||||||||||||||||
| Forfeited | ( | — | ||||||||||||||||||
| Outstanding as of December 31, 2023 | $ | |||||||||||||||||||
| Exercisable as of December 31, 2023 | $ | |||||||||||||||||||
Activity for the year ended December 31, 2022, under the 2019 ISOP was as follows:
| Number of options | Weighted average exercise price | Weighted average remaining life in years | ||||||||||||||||||
| Outstanding as of January 1, 2022 | $ | |||||||||||||||||||
| Exercised | ( | — | ||||||||||||||||||
| Forfeited | ( | — | ||||||||||||||||||
| Outstanding as of December 31, 2022 | $ | |||||||||||||||||||
| Exercisable as of December 31, 2022 | $ | |||||||||||||||||||
The valuation inputs for the 2019 ISOP grants were as follows:
| Twelve months ended December 31, | ||||||||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||||||||
| Share price at grant date (in USD) | N/A | N/A | $ | |||||||||||||||||||||||
| Expected life of share options (years) | N/A | N/A | - | |||||||||||||||||||||||
| Expected volatility | N/A | N/A | % | - | ||||||||||||||||||||||
| Risk free interest rate | N/A | N/A | ( | % | - | ( | ||||||||||||||||||||
| Dividend yield (%) | N/A | N/A | ||||||||||||||||||||||||
Options outstanding as of December 31, 2023, under the 2019 ISOP expire between 2024 and 2031.
The weighted average share price at the date of exercise were $4.65 and $6.19 for the years ended December 31, 2023 and 2022, respectively.
F-45
2021 EIP
Activity for the year ended December 31, 2023, under the 2021 EIP was as follows:
| Number of options | Weighted average exercise price | Weighted average remaining life in years | ||||||||||||||||||
| Outstanding as of January 1, 2023 | $ | |||||||||||||||||||
| Granted | — | |||||||||||||||||||
| Exercised | ( | — | ||||||||||||||||||
| Forfeited | ( | $ | — | |||||||||||||||||
| Outstanding as of December 31, 2023 | $ | |||||||||||||||||||
| Exercisable as of December 31, 2023 | $ | |||||||||||||||||||
Activity for the year ended December 31, 2022, under the 2021 EIP was as follows:
| Number of options | Weighted average exercise price | Weighted average remaining life in years | ||||||||||||||||||
| Outstanding as of January 1, 2022 | $ | |||||||||||||||||||
| Granted | — | |||||||||||||||||||
| Forfeited | ( | — | ||||||||||||||||||
| Outstanding as of December 31, 2022 | $ | |||||||||||||||||||
| Exercisable as of December 31, 2022 | $ | |||||||||||||||||||
The valuation inputs for the 2021 EIP grants were as follows:
| Year Ended December 31, | ||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||||||||||||||||||||
| Share price at grant date (in USD) | $ | - | $ | $ | - | $ | $ | - | $ | |||||||||||||||||||||||||||||
| Expected life of share options (years) | | - | | - | - | |||||||||||||||||||||||||||||||||
| Expected volatility (%) | % | - | % | - | % | - | ||||||||||||||||||||||||||||||||
| Risk free interest rate (%) | % | - | % | - | % | - | ||||||||||||||||||||||||||||||||
| Dividend yield (%) | ||||||||||||||||||||||||||||||||||||||
Options outstanding as of December 31, 2023, under the 2021 EIP expire between 2025 and 2033.
The weighted average share price at the date of exercise was $3.79 for the year ended December 31, 2023 and no options were exercised for the year ended December 31, 2022.
Share options outstanding at the year ended December 31, 2023
The weighted average fair value of options granted during the years ended December 31, 2023 and 2022, respectively (in USD):
| 2023 | 2022 | |||||||||||||
| 2021 EIP | $ | $ | ||||||||||||
F-46
Restricted Stock Units
As part of the 2021 EIP, the Company initiated granting of RSUs, which represent the right to receive shares of ordinary shares upon meeting specified vesting requirements. In the year ended December 31, 2023, the Company granted 2,658,150 RSUs under the 2021 plan. Under the terms of the 2021 plan, 2,260,649 of the RSUs granted are subject to a four-year vesting schedule with 25 % vesting on the first anniversary of the grant date and the remaining 75 % ratably on a quarterly basis over the remaining three years , 107,647 are subject to a year vesting period on the second anniversary from the date of grant, and the remaining 289,854 of the RSUs granted to non-executive members of the Company’s board of directors are subject to a vesting period set to be completed upon the Company’s 2024 Annual General Meeting. The activity for the year ended December 31, 2023 was as follows:
| Number of RSUs | Weighted-average grant date fair value per share | |||||||||||||
| Unvested as of January 1, 2023 | $ | |||||||||||||
| Granted | $ | |||||||||||||
| Vested | ( | $ | ||||||||||||
| Forfeited | ( | $ | ||||||||||||
| Unvested as of December 31, 2023 | $ | |||||||||||||
In the year ended December 31, 2022, the Company issued 1,776,832 RSUs under the 2021 plan. Under the terms of the 2021 plan, 1,396,366 of the RSUs issued are subject to a four-year vesting schedule with 25 % vesting on the first anniversary of the grant date and the remaining 75 % ratably on a monthly basis over the remaining three years , and the remaining 380,466 of the RSUs issued to non-executive members of the Company’s board of directors are subject to a vesting period set to be completed upon the Company’s 2023 Annual General Meeting. The activity for the year ended December 31, 2022 was as follows:
| Number of RSUs | Weighted-average grant date fair value per share | |||||||||||||
| Unvested as of January 1, 2022 | $ | |||||||||||||
| Granted | ||||||||||||||
| Vested | ( | |||||||||||||
| Forfeited | ( | |||||||||||||
| Unvested as of December 31, 2022 | $ | |||||||||||||
Share-based compensation expense
Movements in the share-based compensation reserve were as follows (in USD thousands):
| Total | |||||
| January 1, 2022 | $ | ||||
| Movement in the period | |||||
| December 31, 2022 | |||||
| Movement in the period | |||||
| December 31, 2023 | $ | ||||
Share-based compensation expense by financial statement caption for all stock awards consists of the following (in USD thousands):
F-47
| Year ended December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Research and development | $ | $ | ||||||||||||||||||
| Sales and marketing | ||||||||||||||||||||
| General and administrative | ||||||||||||||||||||
| Total | $ | $ | $ | |||||||||||||||||
24. Borrowings
Revolving credit facility
On June 21, 2022 the Company entered into a credit agreement (“the Credit Facility”) with Credit Suisse SA for up to CHF 5.0 million. Borrowings under the credit facility will bear interest at a rate to be established between the Company and Credit Suisse at the time of each draw down. Borrowings under the Credit Facility have no restrictions related to its use. As of December 31, 2023, the Company had no borrowings outstanding under the Credit Facility.
During the period since January 1, 2020, the Company has not been subject to any externally imposed capital requirements.
25. Share capital issuance
On June 30, 2021, the Company performed a one-to-twenty share split and converted all preferred shares to ordinary shares. Refer to Note 1 – “Company information and operations - Share split.”
On July 22, 2021 as part of the Company IPO, the Company converted all preferred shares to ordinary shares. Refer to Note 1- “Company information and operations - Initial public offering.”
At the next ordinary Annual General Meeting, the Board of Directors will not propose any dividend in respect of the year ended December 31, 2023.
26. Related parties
Related parties comprise the Company’s executive officers and directors, including their affiliates, and any person that directly, or indirectly through one or more intermediaries, controls, is controlled by, or is under common control, with the Company.
Key management personnel comprised of six Executive Officers and Directors and seven Non-Executive Directors for the year ended December 31, 2023. Key management personnel comprised of six Executive Officers and Directors and seven Non-Executive Directors for the year ended December 31, 2022. Key management personnel comprised of six Executive Officers and Directors and six Non-Executive Directors for the year ended December 31, 2021.
F-48
Compensation for key management and non-executive directors recognized during the year comprised (in USD thousands):
| December 31, | ||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||
| Salaries and other short-term employee benefits | $ | $ | $ | |||||||||||||||||
| Pension costs | ||||||||||||||||||||
| Share-based compensation expense | ||||||||||||||||||||
| Total | $ | $ | $ | |||||||||||||||||
27. Commitments and contingencies
Commitments
The Company has no commitments for future lease payments under short-term leases not recognized on the balance sheet as of December 31, 2023. As of December 31, 2022 the company had commitments for future lease payments under short-term leases not recognized on the balance sheet of $0.2 million.
The Company entered into an agreement with Microsoft Corporation as of November 1, 2022. As part of the agreement, the Company has commitments of approximately $69.4 million in computational and hosting-related costs through October 31, 2027.
Contingencies
As of December 31, 2023 and 2022 the Company had no
F-49
28. Financial instruments and risks
A financial instrument is any contract that gives rise to a financial asset of one entity and a financial liability or equity instrument of another entity.
The Company holds the following financial instruments (in USD thousands):
| December 31, | ||||||||||||||
| 2023 | 2022 | |||||||||||||
| Financial assets at amortized cost | ||||||||||||||
| Cash and cash equivalents | $ | $ | ||||||||||||
| Term deposits | ||||||||||||||
| Accounts receivable | ||||||||||||||
| Other financial non-current assets | ||||||||||||||
| Total financial assets at amortized cost | $ | $ | ||||||||||||
| Financial assets at fair value through statement of loss | ||||||||||||||
| Total financial assets | $ | $ | ||||||||||||
| Financial liabilities at amortized cost | ||||||||||||||
| Accounts payable | ||||||||||||||
| Accrued expenses | ||||||||||||||
| Lease liabilities | ||||||||||||||
| Total financial liabilities at amortized cost | ||||||||||||||
| Financial liabilities at fair value through statement of loss | ||||||||||||||
| Total financial liabilities | $ | $ | ||||||||||||
The Company’s exposure to various risks associated with the financial instruments is discussed below in “Financial risk management.” The maximum exposure to credit risk at the end of the reporting period is the carrying amount of each class of financial assets mentioned above. See Note 13 - “Accounts receivable” for expected credit loss provisions on accounts receivable.
Fair value measurement
As of December 31, 2023 and 2022, the carrying amount was a reasonable approximation of fair value for the following financial assets and liabilities:
Financial assets
•Cash and cash equivalents
•Term deposits
•Accounts receivable
•Other non-current assets—lease deposits and lease receivable
Financial liabilities
•Accounts payable
•Accrued liabilities
•Lease liabilities
F-50
Fair value measurement methodology
The Company measures financial instruments at fair value at each balance sheet date. Fair value is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. The fair value measurement is based on the presumption that the transaction to sell the asset or transfer the liability takes place either in the principal market for the asset or liability or, in the absence of a principal market, in the most advantageous market for the asset or liability.
The fair value of an asset or a liability is measured using the assumptions that market participants would use when pricing the asset or liability, assuming that market participants act in their economic best interest.
A fair value measurement of a non-financial asset takes into account a market participant’s ability to generate economic benefits by using the asset or by selling it to another market participant.
The Company uses valuation techniques to measure fair value maximizing the use of relevant observable inputs and minimizing the use of unobservable inputs.
All assets and liabilities for which fair value is measured or disclosed in the consolidated financial statements are categorized within the fair value hierarchy, described as follows, based on the lowest level input that is significant to the fair value measurement as a whole:
Level 1—Quoted (unadjusted) market prices in active markets for identical assets or liabilities.
Level 2—Valuation techniques for which the lowest level input that is significant to the fair value measurement is directly or indirectly observable.
Level 3—Valuation techniques for which the lowest level input that is significant to the fair value measurement is unobservable.
For assets and liabilities that are recognized in the consolidated financial statements at fair value on a recurring basis, the Company determines whether transfers have occurred between levels in the hierarchy by re-assessing categorization (based on the lowest level input that is significant to the fair value measurement as a whole) at the end of each reporting period.
Management determines the policies and procedures for both recurring fair value measurement and for non-recurring measurement with the involvement of experts and external consultants when needed.
The Company holds money market funds which are classified as cash equivalents which are measured as a level 1 valuation. Refer to Note 11 - “Cash and cash equivalents”.
In 2023 and 2022 there were no significant changes in the business or economic circumstances that affect the fair value of the Company’s financial assets and financial liabilities. There were also no transfers between categories.
Financial risk management
Financial risks
Senior management regularly reviews the Company’s cash forecast and related risks. They also perform the risk assessment, define any necessary measures and ensure the monitoring of the internal control system.
The Company’s principal financial liabilities include accounts payable and lease liabilities. The Company’s principal financial assets include cash and cash equivalents, term deposits and short-term investments and accounts receivable.
In the course of its business, the Company is exposed to a number of financial risks including credit and counterparty risk, funding and liquidity risk and market risk (i.e. foreign currency risk and interest rate risk). This note presents the Company’s objectives, policies, and processes for managing these risks.
F-51
Credit and counterparty risk management
Credit risk is the risk that a counterparty will not meet its obligations under a financial instrument or customer contract, leading to a financial loss. The Company is exposed to credit risk from its operating activities, primarily accounts receivable.
Concentration risk arises when a number of counterparties are engaged in similar business activities, or activities in the same geographical region, or have economic features that would cause their ability to meet contractual obligations to be similarly affected by changes in economic, political or other conditions.
The Company’s policy with regard to assessing and providing for expected credit losses on accounts receivable is set out in Note 13 - “Accounts receivable.”
Credit risk from balances with banks and financial institutions is managed by the Company’s treasury department in accordance with the Company’s policy.
Financial transactions are predominantly entered into with investment grade financial institutions and in principle the Company requires a minimum long-term rating of A3/A- for its cash investments and term deposits. The Company may deviate from this requirement from time to time for operational reasons. The highest exposure to a single financial counterparty within cash and cash equivalents and term deposits and short-term investments amounted to $43.7 million and $57.3 million as of December 31, 2023 and 2022, respectively.
Other non-current financial assets include cash deposits for leases.
Funding and liquidity risk management
Funding and liquidity risk is the risk that a company may encounter difficulties in meeting its obligations associated with financial liabilities that are settled by delivering cash or other financial assets. Such risk may result from inadequate market depth or disruption or refinancing problems.
The Company views equity funding as its primary source of liquidity only partly complemented with revenue generated from the sale of the platform, applications, products, and services and some borrowings. The Company has no outstanding borrowing facilities. Short term liquidity is managed based on projected cash flows. As of December 31, 2023 and 2022, the Company’s liquidity consisted of $123.3 million and $161.3 million in cash and cash equivalents, respectively. On the basis of the current operating performance and liquidity position, management believes that the available cash balances will be sufficient for operating activities, working capital, interest, capital expenditures and scheduled debt repayments for the next 12 months.
F-52
The table below summarizes the maturity profile of the Company’s financial liabilities based on contractual undiscounted cash flows (in USD thousands):
| Net carrying amount | Within 1 year | Between 1 and 5 years | After 5 years | Total | ||||||||||||||||||||||||||||
| December 31, 2023 | ||||||||||||||||||||||||||||||||
| Lease liabilities | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Accounts payable | ||||||||||||||||||||||||||||||||
| Accrued expenses | ||||||||||||||||||||||||||||||||
| Total contractual liabilities | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| December 31, 2022 | ||||||||||||||||||||||||||||||||
| Lease liabilities | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Accounts payable | ||||||||||||||||||||||||||||||||
| Accrued expenses | ||||||||||||||||||||||||||||||||
| Total contractual liabilities | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
Market risk
Market risk includes currency risk and interest rate risk.
Currency risk
Foreign currency risk is the risk that the fair value or future cash flows of an exposure will fluctuate because of changes in foreign exchange rates.
The significant exchange rates that have been applied to these consolidated financial statements are listed below:
| December 31, | For the twelve months ended December 31, | ||||||||||||||||||||||||||||
| 2023 | 2022 | 2023 | 2022 | 2021 | |||||||||||||||||||||||||
| Currency | Spot rate | Spot rate | Average rate | Average rate | Average rate | ||||||||||||||||||||||||
| USD/CHF | |||||||||||||||||||||||||||||
| USD/EUR | |||||||||||||||||||||||||||||
| USD/GBP | |||||||||||||||||||||||||||||
| USD/BRL | |||||||||||||||||||||||||||||
The sensitivity of the Company’s income to possible changes in foreign exchange rates is measured at the local entity level as it depends on the functional currency of each entity. As of December 31, 2023, 2022, and 2021
F-53
the Company was exposed principally to movements in four cross currency pairs. The sensitivity of the Company’s loss before tax to such changes was as follows (in USD thousands):
| December 31, | ||||||||||||||||||||||||||||||||||||||
| 2023 | 2022 | 2021 | ||||||||||||||||||||||||||||||||||||
Increase / (decrease) in USD/CHF exchange rate by | / | ( | / | ( | / | ( | ||||||||||||||||||||||||||||||||
Increase / (decrease) in EUR/CHF exchange rate by | / | ( | ( | / | / | ( | ||||||||||||||||||||||||||||||||
Increase / (decrease) in GBP/CHF exchange rate by | ( | / | ( | / | ( | / | ||||||||||||||||||||||||||||||||
Increase / (decrease) in USD/EUR exchange rate by | ( | / | / | ( | / | ( | ||||||||||||||||||||||||||||||||
The Company’s exposure to foreign currency changes for all other currencies is not material. The significant increase/decrease between USD/CHF resulted from the Company’s IPO, which occurred in USD. The Company does not use derivative financial instruments to hedge exposures and under no circumstances may enter into derivative instruments for speculative purposes.
The sensitivity of the Company’s reported equity or net assets to possible changes in foreign exchange rates is measured at the consolidated level as it depends on the presentation currency selected for the consolidated financial statements. Such effects are reported not in income but in the currency translation account within other reserves. As of December 31, 2023 and 2022 the sensitivity of the Company’s equity to such changes, measured against the USD, was as follows (in USD thousands):
| December 31, | ||||||||||||||||||||||||||
| 2023 | 2022 | |||||||||||||||||||||||||
Increase / (decrease) in USD/CHF exchange rate by | ( | / | / | ( | ||||||||||||||||||||||
Increase / (decrease) in USD/EUR exchange rate by | / | ( | ( | / | ||||||||||||||||||||||
Increase / (decrease) in USD/GBP exchange rate by | / | ( | / | ( | ||||||||||||||||||||||
Interest rate risk
The Company’s cash and cash equivalents and term deposits are subject to market risk associated with interest rate fluctuations. Fixed rate securities may have their market value adversely affected due to a rise in interest rates. The Company concluded that fluctuations in the interest rate did not have a material impact on our cash equivalents and term deposit balances.
29. Capital management
The Company considers equity as equivalent to the IFRS Accounting Standards equity on the balance sheet (including share capital, share premium and all other equity reserves attributable to the owners of the Company).
The primary objective of the Company’s capital management is to maximize shareholder value. The Board regularly reviews its shareholders’ return strategy. For the foreseeable future, the Board will maintain a capital structure that supports the Company’s strategic objectives through managing funding and liquidity risks and optimizing shareholder return.
As of December 31, 2023 and 2022, the Company’s cash and cash equivalents amounted to $123.3 million and $161.3 million, respectively.
The Board of Directors believes that the Company has sufficient financial resources to meet all of its obligations for at least the next twelve months. Moreover, the Company is not exposed to liquidity risk through requests for early repayment of loans.
F-54
30. Events after the reporting date
The Company has evaluated, for potential recognition and disclosure, events that occurred prior to the date at which the consolidated financial statements were available to be authorized for issuance. There were no material subsequent events.
F-55